Electron Probe Microanalysis and Microscopy of Polishing-Exposed Solid-Phase Mineral Inclusions in Fuxian Kimberlite Diamonds
Abstract
1. Introduction
2. Geology of the Fuxian Kimberlite
3. Sample Preparation
4. Analytical Methods
5. Features of Diamond Hosts and Their Mineral Inclusions
6. Results and Discussion
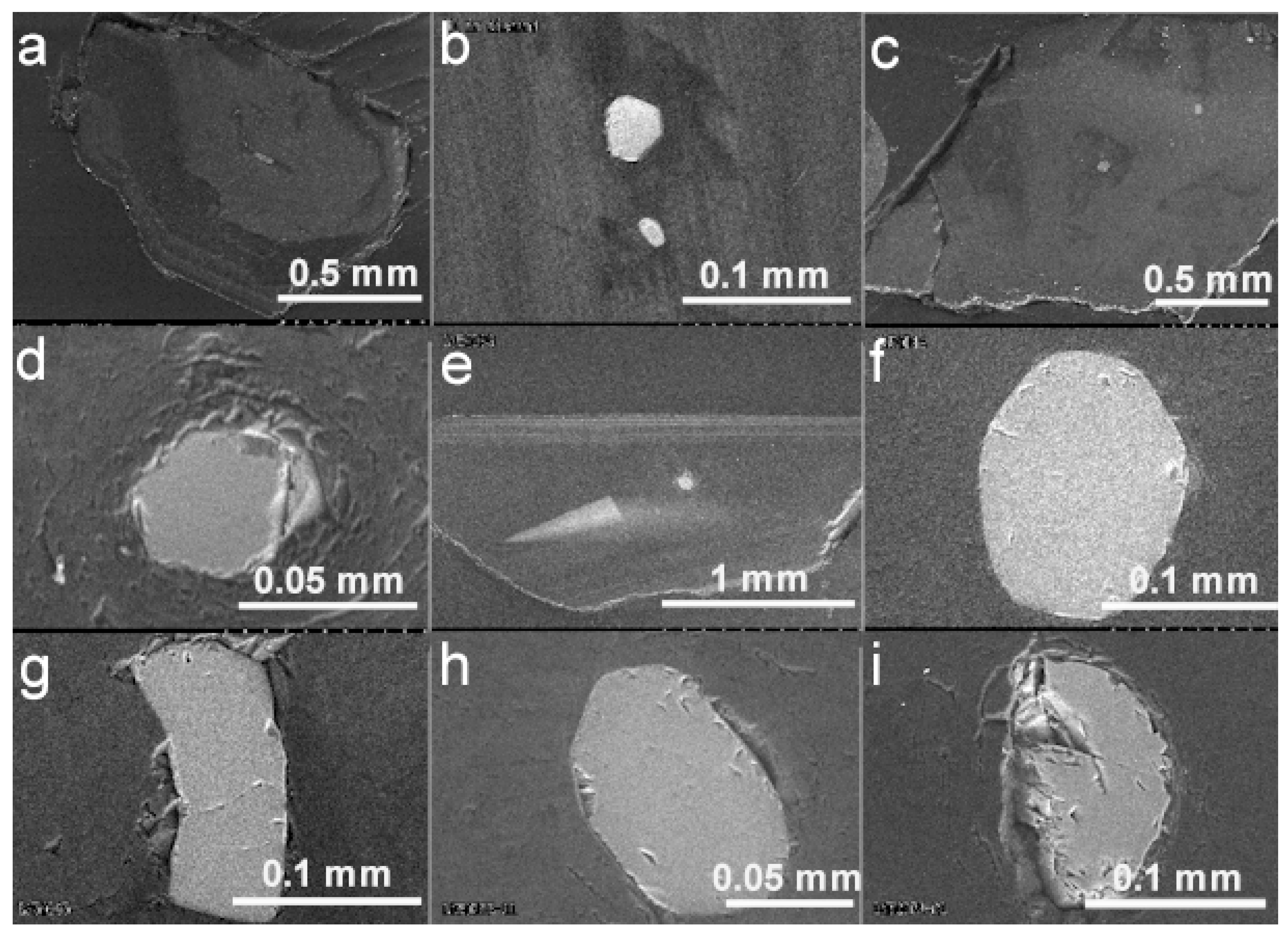
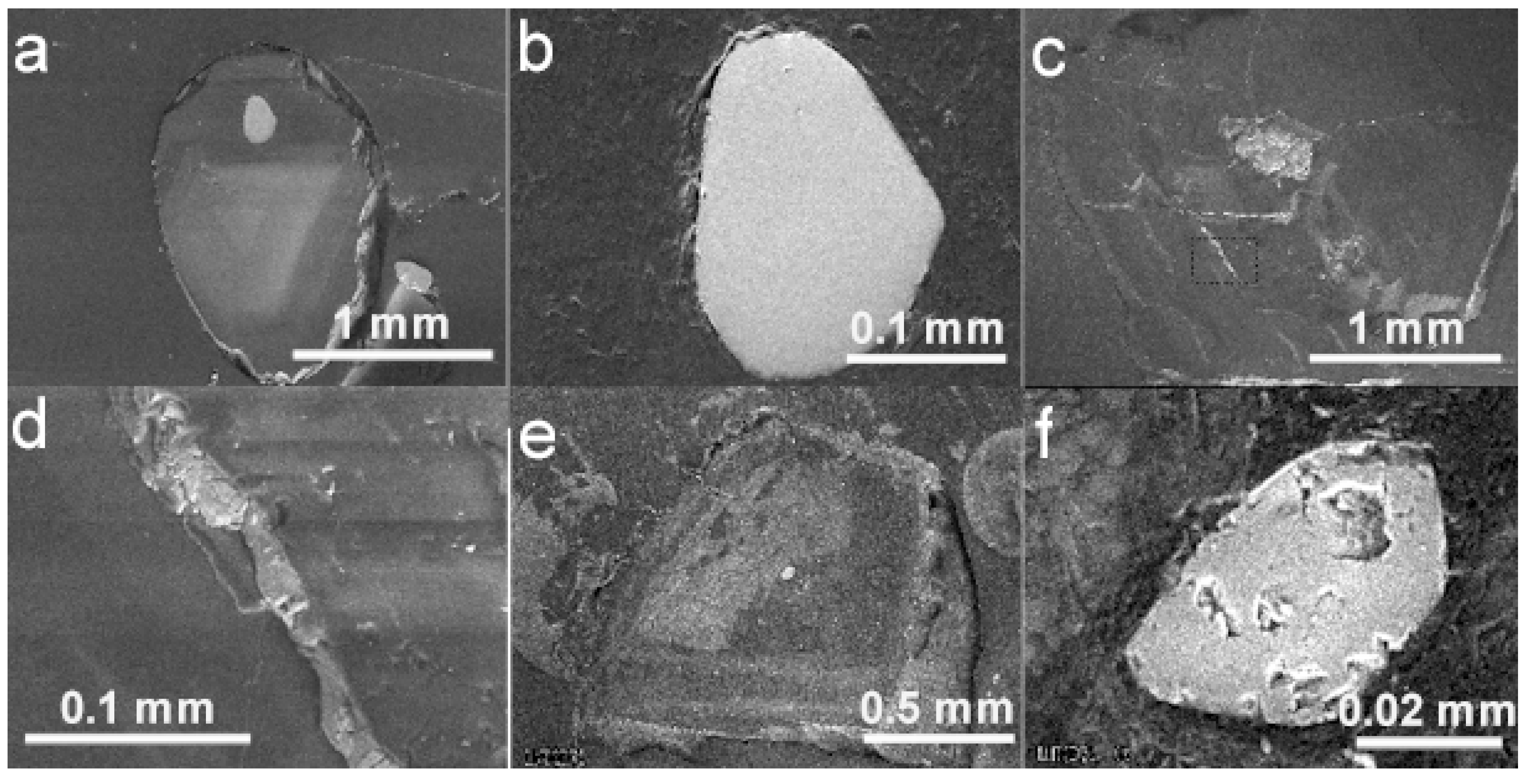
6.1. BSE Images and Cathodoluminescence of Diamond Hosts
6.2. EDS and X-ray Mapping of Mineral Inclusions
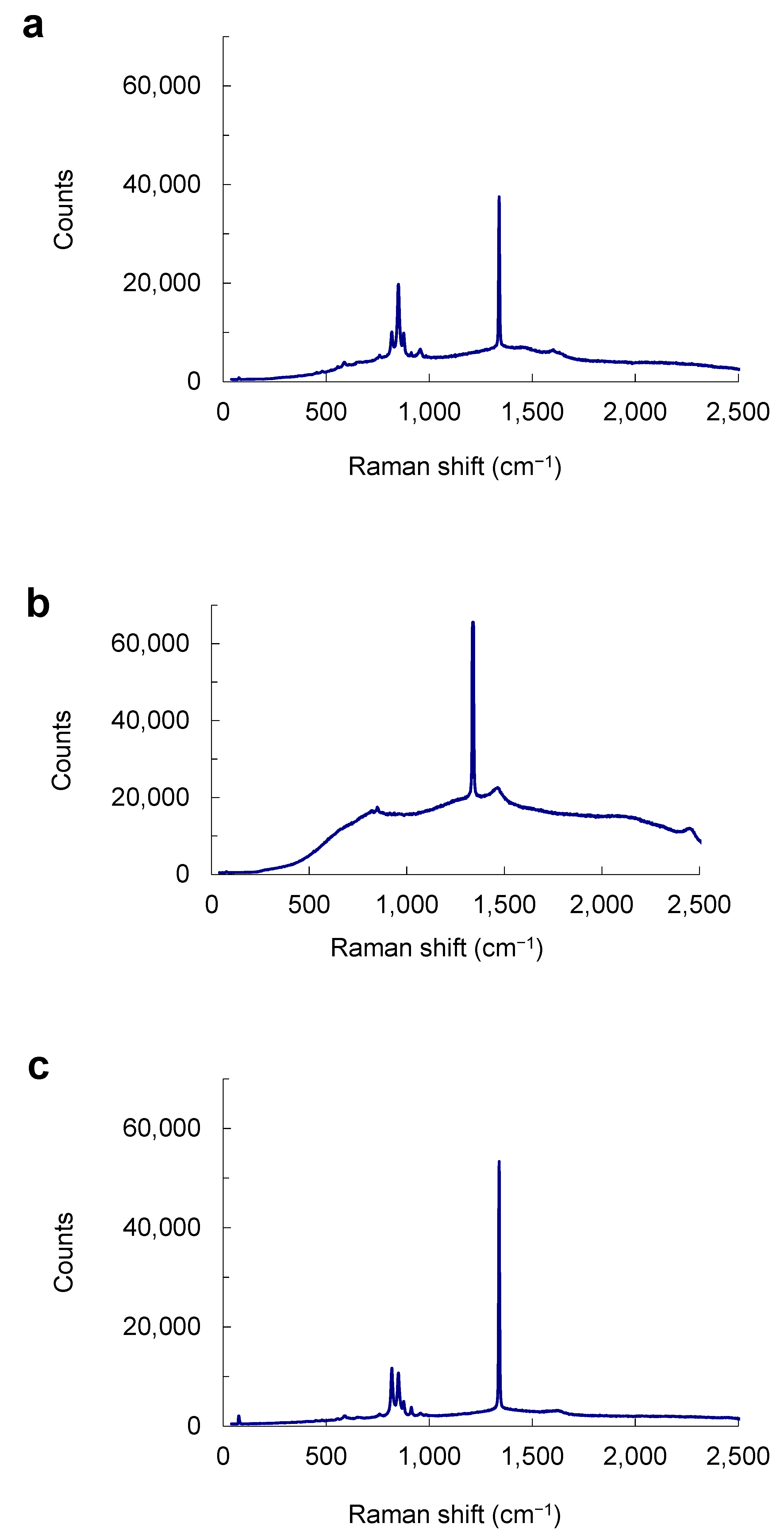
6.3. Micro-Raman Spectra of Mineral Inclusions
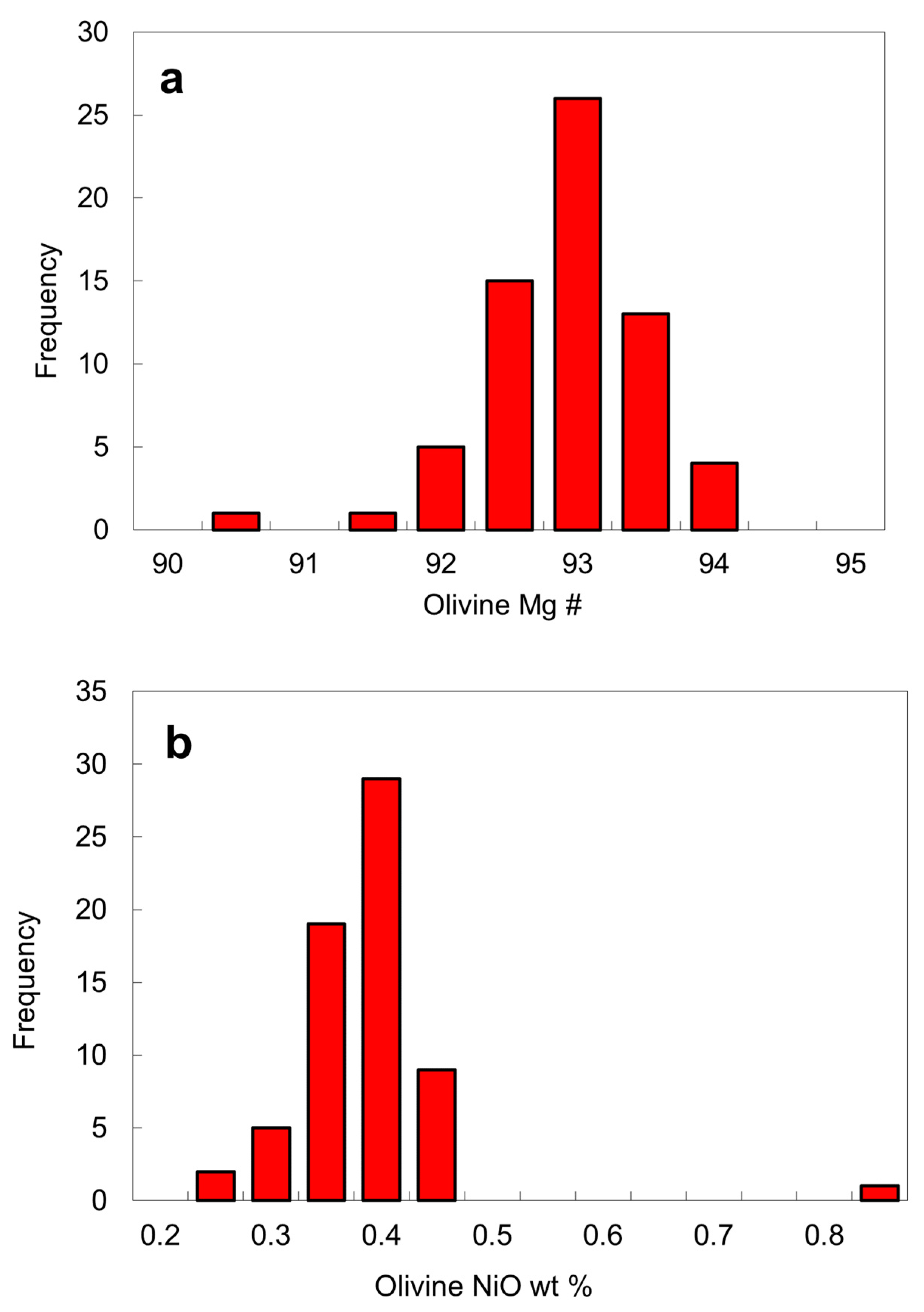
6.4. Chemical Compositions of Peridotitic Mineral Inclusions
6.5. Chemical Compositions of Rare Mineral Inclusions
| LN50D67 Domain 1 | LN50D67 Domain 2 | Miao et al. [10] | Smyth & Kawamoto [61] | Burnley and Navrotsky [60] | |||||
|---|---|---|---|---|---|---|---|---|---|
| Mineral | Wadsleyite II | Phase D | Phase E | Phase F | |||||
| Average | 3 Analyses | 3 Analyses | 4 Analyses | 1 | 2 | ||||
| SiO2 | 47.27 | 41.29 | 65.81 | SiO2 | 40.04 | 40.01 | 50.74 | 39.65 | 60.71 |
| TiO2 | 0.01 | 0.01 | 0.06 | TiO2 | na | na | 0.00 | 0.00 | 0.00 |
| Al2O3 | 10.01 | 10.42 | 0.91 | Al2O3 | 0.41 | 0.34 | 0.00 | 0.00 | 0.00 |
| Cr2O3 | 0.08 | 0.10 | 0.11 | Cr2O3 | 0.19 | 0.22 | 0.00 | 0.00 | 0.00 |
| FeO | 8.19 | 11.90 | 0.22 | FeO | 8.80 | 10.60 | 0.00 | 0.00 | 0.00 |
| NiO | 0.13 | 0.21 | na | NiO | na | na | 0.00 | 0.00 | 0.00 |
| MnO | 0.01 | 0.02 | 0.02 | MnO | na | na | 0.00 | 0.00 | 0.00 |
| MgO | 20.37 | 24.39 | 29.36 | MgO | 47.57 | 44.37 | 34.04 | 48.94 | 27.15 |
| CaO | 0.87 | 0.38 | 0.11 | CaO | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Σ | 86.95 | 88.72 | 96.60 | H2O | 2.99 | 4.46 | 15.21 | 11.41 | 12.14 |
| H2O | 12.90 | 12.68 | 4.84 | Σ | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| Σ | 99.85 | 101.40 | 101.44 | ||||||
| Si | 4.395 | 3.906 | 4.08 | Si | 0.975 | 0.980 | 1.000 | 1.250 | 1.800 |
| Al | 1.097 | 1.162 | 0.07 | Al | 0.012 | 0.010 | 0.000 | 0.000 | 0.000 |
| Ti | 0.001 | 0.000 | 0.00 | Ti | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Cr | 0.006 | 0.008 | 0.01 | Cr | 0.004 | 0.004 | 0.000 | 0.000 | 0.000 |
| Fe | 0.637 | 0.942 | 0.01 | Fe | 0.179 | 0.217 | 0.000 | 0.000 | 0.000 |
| Ni | 0.008 | 0.013 | 0.00 | Ni | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Mn | 0.000 | 0.001 | 0.00 | Mn | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Mg | 2.822 | 3.439 | 2.71 | Mg | 1.726 | 1.620 | 1.000 | 2.300 | 1.200 |
| Ca | 0.087 | 0.039 | 0.01 | Ca | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Σcation | 9.053 | 9.509 | 6.88 | H | 0.243 | 0.364 | 2.000 | 2.400 | 2.400 |
| ΣO | 14.000 | 14.000 | 11.00 | Σcation | 3.139 | 3.195 | 4.000 | 5.950 | 5.400 |
| Mg # | 81.6 | 78.5 | 99.58 | ΣO | 4.000 | 4.000 | 4.000 | 6.000 | 6.000 |
| LN50D09 (Goethite or Limonite?) | Miao et al. [10] | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Analysis | a1 | a2 | b1 | b2 | b3 | b4 | Average | Goethite (?) | Ideal Goethite |
| Al2O3 | 0.11 | 0.37 | 0.26 | 0.10 | 0.35 | 0.18 | 0.25 | 0.54 | 0.00 |
| SiO2 | 3.77 | 3.53 | 3.66 | 1.39 | 3.64 | 3.63 | 3.27 | na | 0.00 |
| Cr2O3 | 0.02 | 0.03 | 0.12 | 0.00 | 0.04 | 0.09 | 0.05 | na | 0.00 |
| MnO | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.09 | 0.00 |
| Fe2O3 | 80.68 | 81.75 | 80.54 | 78.86 | 82.04 | 79.92 | 80.63 | 85.30 | 87.98 |
| CoO | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | na | 0.00 |
| NiO | 0.10 | 0.02 | 0.08 | 0.11 | 0.04 | 0.09 | 0.07 | na | 0.00 |
| CuO | 0.00 | 0.00 | 0.06 | 0.06 | 0.08 | 0.33 | 0.10 | na | 0.00 |
| BaO | 0.14 | 0.08 | 0.14 | 0.02 | 0.07 | 0.10 | 0.09 | na | 0.00 |
| H2O | 15.19 | 14.21 | 15.14 | 19.47 | 13.74 | 15.66 | 15.53 | 14.07 | 12.02 |
| Σ | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| Oxygen normalized to 3 | |||||||||
| Al | 0.004 | 0.013 | 0.009 | 0.004 | 0.012 | 0.007 | 0.009 | 0.020 | 0.000 |
| Si | 0.114 | 0.106 | 0.111 | 0.045 | 0.109 | 0.111 | 0.100 | 0.000 | 0.000 |
| Cr | 0.000 | 0.001 | 0.003 | 0.000 | 0.001 | 0.002 | 0.001 | 0.000 | 0.000 |
| Mn | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.002 | 0.000 |
| Fe3+ | 1.841 | 1.844 | 1.837 | 1.933 | 1.840 | 1.836 | 1.853 | 1.979 | 2.000 |
| Co | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Ni | 0.002 | 0.001 | 0.002 | 0.003 | 0.001 | 0.002 | 0.002 | 0.000 | 0.000 |
| Cu | 0.000 | 0.000 | 0.001 | 0.001 | 0.002 | 0.008 | 0.002 | 0.000 | 0.000 |
| Ba | 0.002 | 0.001 | 0.002 | 0.000 | 0.001 | 0.001 | 0.001 | 0.000 | 0.000 |
| Σcation | 1.963 | 1.965 | 1.965 | 1.986 | 1.965 | 1.967 | 1.968 | 2.001 | 2.000 |
| Σcharge | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 |
| H2O molecule | 1.265 | 1.183 | 1.261 | 1.621 | 1.144 | 1.304 | 1.293 | 1.172 | 1.000 |
6.6. Origin of Sulfide Inclusions in Diamond
6.7. Source Rocks, Metasomatism, and Diamond Formation
Funding
Acknowledgments
Conflicts of Interest
References
- Meyer, H.O.A. Inclusions in diamonds. In Mantle Xenoliths; Nixon, P.H., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 1987; pp. 501–522. [Google Scholar]
- Moore, R.O.; Gurney, J.J. Inclusions in diamond from the Monastery kimberlite, South Africa. In Kimberlites and Related Rocks Volume 2: Their Mantle/Crustal Setting, Diamonds and Diamond Exploration, Proceedings of the Fourth International Kimberlite Conference, Perth, Australia, 11–15 August 1986; Ross, J., Jaques, A.L., Ferguson, J., Green, D.H., O’Reilly, S.Y., Danchin, R.Y., Janse, A.J.A., Eds.; Geological Society of Australia Special Publication 14 (2); Geological Society of Australia: Hornsby, Australia, 1989; pp. 1029–1041. [Google Scholar]
- Huang, Y.; Qin, S.; Zhou, X.; Deng, C.; Zhao, D.; Yang, J.; Guo, Y.; Gao, Y.; Li, G. Kimberlites and Diamonds in the North China Craton; Geological Publishing House: Beijing, China, 1992; 206p. (In Chinese) [Google Scholar]
- Leung, I.S. The discovery of calcite inclusion in natural diamond and its implications on the genesis of diamond, kimberlite and carbonatite. In Geological Society of America 97th Annual Meeting Abstract and Programs, Proceedings of the Geological Society of America 97th Annual Meeting, Reno, NV, USA, 5–8 November 1984; Geological Society of America: Boulder, CO, USA, 1984; Volume 16, p. 574. [Google Scholar]
- Leung, I.S. Silicon carbide cluster entrapped in a diamond from Fuxian, China. Am. Mineral. 1990, 75, 1110–1119. [Google Scholar]
- Leung, I.S. Metasomatized olivine, garnet and diopside entrapped in diamonds from Fuxian. Eos 1994, 74, 192. [Google Scholar]
- Zhang, A.; Meyer, H.O.A. Inclusions in diamonds from Chinese kimberlites. In Extended Abstracts of the 28th IGC Workshop on Diamonds; The Congress: Washington, DC, USA, 1989; pp. 1–3. [Google Scholar]
- Deng, C.; Huang, Y.; Zhao, D. Mineral inclusions in the Chinese diamonds. In Extended Abstracts of the 15th IMA General Meeting; International Mineralogical Association: Aubière, France, 1990; Volume 1, pp. 487–488. [Google Scholar]
- Dong, Z. Mineral inclusions in diamonds. Mineral. Petrol. 1991, 11, 64–71, (In Chinese with English Abstract). [Google Scholar]
- Miao, Q.; Liu, G.; Lu, Q.; Wang, X.; Zheng, C. New discovery of complicated inclusions in diamonds and their origin. Geol. Sci. Technol. Inf. 1991, 10, 117–123. (In Chinese) [Google Scholar]
- Wang, A.; Wang, W.; Zhang, A. Microstructural variations of a pyrope inclusion in diamond, as revealed by a micro-Raman spectroscopic study. Can. Mineral. 1991, 29, 517–524. [Google Scholar]
- Wang, A.; Dhamelincourt, P.; Meyer, H.O.A.; Guo, L.; Zhang, A. A carbon-rich multiphase inclusion in a Chinese diamond and its geochemical implications. Contrib. Mineral. Petrol. 1994, 117, 15–24. [Google Scholar] [CrossRef]
- Chen, F.; Wang, M.; Wang, S.; Ding, Z.; Guo, J.; Wang, Z.; Lei, P.; Zhang, Y. First discovery of High-Cu and high-Cl inclusions in diamond. Kexue Tongbao (Chin. Sci. Bull.) 1992, 37, 1782–1784. (In Chinese) [Google Scholar]
- Miao, Q. The characteristic of diamonds from Toudaogou placer deposit, Southern Liaoning, China. Collect. Mineral. Petrol. 1992, 8, 25–32, (In Chinese with English Abstract). [Google Scholar]
- Zhao, D.; Deng, C.; Yang, J. Diamonds from East China: Morphology and inclusions. Eos 1993, 74, 168. [Google Scholar]
- Harris, J.W.; Duncan, D.J.; Zhang, F.; Miao, Q.; Zhu, Y. The physical characteristics and syngenetic inclusion geochemistry of diamonds from Pipe 50, Liaoning Province, People’s Republic of China. In Diamonds: Characterization, Genesis and Exploration, Proceedings of the Fifth International Kimberlite Conference, Araxa, Brazil, 18 June–4 July 1991; Meyer, H.O.A., Leonardos, O.H., Eds.; CPRM-Special Publication; Companhia de Pesquisa de Recursos Minerais: Rio de Janeiro, Brazil, 1994; Volume 2, pp. 106–115. [Google Scholar]
- Meyer, H.O.A.; Zhang, A.; Milledge, H.J.; Mendelssohn, M.J. Diamonds and inclusions in diamonds from Chinese kimberlites. In Diamonds: Characterization, Genesis and Exploration, Proceedings of the Fifth International Kimberlite Conference, Araxa, Brazil, 18 June–4 July 1991; Meyer, H.O.A., Leonardos, O.H., Eds.; CPRM-Special Publication; Companhia de Pesquisa de Recursos Minerais: Rio de Janeiro, Brazil, 1994; Volume 2, pp. 98–105. [Google Scholar]
- Wang, Z.; Guo, G. Characteristics of inclusions from diamonds in No. 50 pipe at Wafangdian, South Liaoning. Liaoning Geol. 1994, 263–274, (In Chinese with English Abstract). [Google Scholar]
- Gorshkov, A.I.; Bao, Y.; Bershov, L.V.; Ryabchikov, I.D.; Sivtsov, A.V.; Lapina, M.I. Inclusions in diamond from the Liaoning deposit (China) and their genetic meaning. Geochem. Int. 1997, 35, 51–57. [Google Scholar]
- Gorshkov, A.I.; Bao, Y.; Bershov, L.V.; Ryabchikov, I.D.; Sivtsov, A.V.; Lapina, M.I. Inclusions of native metals and other minerals in diamond from kimberlite pipe 50, Liaoning, China. Geochem. Int. 1997, 35, 695–703. [Google Scholar]
- Zhao, D. Electron Probe Microanalysis and Microscopy: Focused Electron Beam Techniques and Applications in Characterization of Mineral Inclusions in Diamond. Microsc. Microanal. 2015, 21 (Suppl. 3), 1319–1320. [Google Scholar] [CrossRef][Green Version]
- Bulanova, G.P. The formation of diamond. J. Geochem. Explor. 1995, 53, 1–23. [Google Scholar] [CrossRef]
- Kopylova, M.G.; Gurney, J.J.; Daniels, L.R.M. Mineral inclusions in diamonds from the River Ranch kimberlite, Zimbabwe. Contrib. Mineral. Petrol. 1997, 129, 366–384. [Google Scholar] [CrossRef]
- Stachel, T.; Harris, J.W. Syngenetic inclusions in diamond from the Birim field (Ghana)—A deep peridotitic profile with a history of depletion and re-enrichment. Contrib. Mineral. Petrol. 1997, 127, 336–352. [Google Scholar] [CrossRef]
- Zhao, D. Kimberlite, Diamond and Mantle Xenolith from Northwest Territories, Canada and North China. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 1998. [Google Scholar]
- Zhao, D.; Zhang, Y.; Essene, E.J. Electron Probe Microanalysis and Microscopy: Principles and Applications in Characterization of Mineral Inclusions in Chromite from Diamond Deposit. Ore Geol. Rev. 2015, 65, 733–748. [Google Scholar] [CrossRef]
- Harris, J.W. Diamond geology. In The Properties of Natural and Synthetic Diamond; Field, J.E., Ed.; Academic Press: London, UK, 1992; pp. 345–393. [Google Scholar]
- Harris, J.W. Black material on mineral inclusions and in internal fracture planes in diamond. Contrib. Mineral. Petrol. 1972, 129, 22–33. [Google Scholar] [CrossRef]
- Gorshkov, A.I.; Titkov, S.V.; Sivtsov, A.V.; Bershov, L.V.; Marfunin, A.S. The first finds of native Cr, Ni, and Fe in carbonado from the diamond deposits of Yakutia. Geochem. Int. 1996, 33, 59–63. [Google Scholar]
- Foster, N.F.; Wozniakiewicz, P.J.; Price, M.C.; Kearsley, A.T.; Burchell, M.J. Identification by Raman spectroscopy of Mg–Fe content of olivine samples after impact at 6 km s−1 onto aluminium foil and aerogel: In the laboratory and in Wild-2 cometary samples. Geochim. Cosmochim. Acta 2013, 121, 1–14. [Google Scholar] [CrossRef]
- Kleppe, A.K.; Jephcoat, A.P.; Smyth, J.R. High-pressure Raman spectroscopic studies of hydrous wadsleyite II. Am. Mineral. 2006, 91, 1102–1109. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Logvinova, A.M.; Zedgenizov, D.A.; Seryotkin, Y.V.; Yefimova, E.S.; Floss, C.; Taylor, L.A. Mineral inclusions in microdiamonds and macrodiamonds from kimberlites of Yakutia: A comparative study. Lithos 2004, 77, 225–242. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Logvinova, A.M.; Zedgenizov, D.A.; Pokhilenko, N.P.; Kuzmin, D.V.; Sobolev, A.V. Olivine inclusions in Siberian diamonds: High precision approach to minor elements. Eur. J. Mineral. 2008, 20, 305–315. [Google Scholar] [CrossRef]
- Griffin, W.L.; Cousens, D.R.; Ryan, C.G.; Sie, S.H.; Suter, G.F. Ni in chrome pyrope garnets: A new geothermometer. Contrib. Mineral. Petrol. 1989, 103, 199–202. [Google Scholar] [CrossRef]
- Ryan, C.G.; Griffin, W.L.; Pearson, N.J. Garnet geotherms—Pressure-temperature data from Cr-pyrope garnet xenocrysts in volcanic rocks. J. Geophys. Res. 1996, 101, 5611–5625. [Google Scholar] [CrossRef]
- Ryabchikov, I.D.; Green, D.H.; Wall, V.; Brey, G.P. The oxidation state of carbon in the reduced velocity zone. Geochem. Int. 1981, 18, 148–158. [Google Scholar]
- Smith, J.V.; Steele, I.M. Lunar mineralogy; a heavenly detective story: Part II. Am. Mineral. 1976, 61, 1059–1116. [Google Scholar]
- Li, J.-P.; O’Neill, H.S.C.; Seifert, F. Subsolidus phase relations in the system MgO-SiO2-Cr-O in equilibrium with metallic Cr, and their significance for the petrochemistry of chromium. J. Petrol. 1995, 36, 107–132. [Google Scholar] [CrossRef]
- Sutton, S.R.; Bajt, S.; Rivers, M.L.; Smith, J.V.; Blanchard, D.; Black, D. X-ray microprobe determination of chromium oxidation state in olivine from lunar basalt and kimberlitic diamonds. In Abstracts of Papers Submitted to the Lunar and Planetary Science Conference; American Geophysical Union: Washington, DC, USA, 1993; Volume 24, pp. 1383–1384. [Google Scholar]
- Köhler, T.P.; Brey, G.P. Calcium exchange between olivine and clinopyroxene calibrated as a geothermobarometer for natural peridotites from 2–60 kb with applications. Geochim. Cosmochim. Acta 1990, 54, 2375–2388. [Google Scholar] [CrossRef]
- Lee, D.-C.; Halliday, A.N.; Davies, G.R.; Essene, E.J.; Fitton, J.G.; Temdjim, R. Melt enrichment of shallow depleted mantle: A detailed petrological, trace element and isotopic study of mantle derived xenoliths and megacrysts from the Cameroon line. J. Petrol. 1996, 37, 415–441. [Google Scholar] [CrossRef]
- Woodland, A.B.; O’Neill, H.S.C. Synthesis and stability of Fe32+Fe23+Si3O12 garnet and phase relations with Fe3Al2Si3O12—Fe32+Fe23+Si3O12 solutions. Am. Mineral. 1993, 78, 1002–1015. [Google Scholar]
- Brey, G.; Doroshev, A.; Kogarko, L. The join pyrope-knorringite: Experimental constraints for a new geothermobarometer for coexisting garnet and spinel. In Extended Abstracts of the Fifth International Kimberlite Conference; CPRM-Special Publication, 2/91; Meyer, H.O.A., Leonardos, O.H., Eds.; Companhia de Pesquisa de Recursos Minerais: Araxa, Brazil, 1991; pp. 26–28. [Google Scholar]
- Gurney, J.J.; Harris, J.W.; Rickard, R.S. Minerals associated with diamonds from the Roberts Victor Mine. In Kimberlites II: Their Mantle and Crust-Mantle Relationships, Proceedings of the Third International Kimberlite Conference, Clermont-Ferrand, France, September 1982; Kornprobst, J., Ed.; Developments in Petrology, 11B; Elsevier: Amsterdam, The Netherlands, 1984; pp. 25–32. [Google Scholar]
- Wilding, M.C.; Harte, B.; Harris, J.W. Inclusion chemistry, carbon isotopes and nitrogen distribution in Bultfontein diamonds: The evolution of natural diamond growth conditions. In Extended Abstracts of the Fifth International Kimberlite Conference; CPRM-Special Publication, 2/91; Meyer, H.O.A., Leonardos, O.H., Eds.; Companhia de Pesquisa de Recursos Minerais: Araxa, Brazil, 1991; p. 459. [Google Scholar]
- Sobolev, N.V.; Lavrent’ev, Y.G.; Pokhilenko, N.P.; Usova, L.V. Chrome-rich garnets from the kimberlites of Yakutia and their paragenesis. Contrib. Mineral. Petrol. 1973, 40, 39–52. [Google Scholar] [CrossRef]
- Gurney, J.J.; Zweistra, P. The interpretation of the major element compositions of mantle minerals in diamond exploration. J. Geochem. Explor. 1995, 53, 293–309. [Google Scholar] [CrossRef]
- Ringwood, A.E. Composition and Petrology of the Earth’s Upper Mantle; McGraw-Hill: London, UK, 1975. [Google Scholar]
- Moore, R.O.; Gurney, J.J. Pyroxene solid solution in garnets included in diamonds. Nature 1985, 318, 553–555. [Google Scholar] [CrossRef]
- Irifune, T. An experimental investigation of the pyroxene garnet transformation in a pyrolite composition and its bearing on the constitution of the mantle. Phys. Earth Planet. Inter. 1987, 45, 324–336. [Google Scholar] [CrossRef]
- Wang, L.; Essene, E.J.; Zhang, Y. Mineral inclusions in pyrope crystals from Garnet Ridge, Arizona, USA: Implications for processes in the upper mantle. Contrib. Mineral. Petrol. 1999, 135, 164–178. [Google Scholar] [CrossRef]
- Tsai, H.M.; Meyer, H.O.; Mozeau, J.; Milledge, H.J. Mineral inclusions in diamond: Premier, Jagersfontein and Finch kimberlites, South Africa, and Williamson Mine, Tanzania. In Kimberlites, Diatremes and Diamonds: Their Geology and Petrology and Geochemistry; Boyd, F.R., Meyer, H.O.A., Eds.; American Geophysical Union: Washington, DC, USA, 1979; pp. 16–26. [Google Scholar]
- Gurney, J.J.; Harris, J.W.; Rickard, R.S. Silicate and oxide inclusions in diamonds from the Finsch kimberlite pipe. In Kimberlites, Diatremes and Diamonds: Their Geology and Petrology and Geochemistry; Boyd, F.R., Meyer, H.O.A., Eds.; American Geophysical Union: Washington, DC, USA, 1979; pp. 1–15. [Google Scholar]
- Wang, A.; Pasteris, J.D.; Meyer, H.O.A.; Deleduboi, M.L. Magnesite-bearing inclusion assemblage in natural diamond. Earth Planet. Sci. Lett. 1996, 141, 293–306. [Google Scholar] [CrossRef]
- Meyer, H.O.A.; McCallum, M.E. Mineral inclusions in diamonds from the Sloan kimberlites, Colorado. J. Geol. 1986, 94, 600–612. [Google Scholar] [CrossRef]
- Phillips, D.; Harris, J.W. Geothermobarometry of diamond inclusions from the De Beers Pool Mines, Kimberley, South Africa. In Extended Abstracts of the Sixth International Kimberlite Conference; Sobolev, N.V., Ed.; United Institute of Geology, Geophysics and Mineralogy, Siberian Branch of Russian Academy of Sciences: Novosibirsk, Russia, 1995; pp. 441–443. [Google Scholar]
- Stachel, T.; Harris, J.W.; Brey, G.P. Rare and unusual mineral inclusions in diamonds from Mwadui, Tanzania. Contrib. Mineral. Petrol. 1998, 132, 34–47. [Google Scholar] [CrossRef]
- Logvinova, A.M.; Wirth, R.; Zedgenizov, D.A.; Taylor, L.A. Carbonate–Silicate–Sulfide Polyphase Inclusion in Diamond from the Komsomolskaya Kimberlite Pipe, Yakutia. Geochem. Int. 2018, 56, 283–291. [Google Scholar] [CrossRef]
- Bulanova, G.P.; Griffin, W.L.; Ryan, C.G.; Shestakova, O.Y.; Barnes, S.J. Trace elements in sulfide inclusions from Yakutian diamonds. Contrib. Mineral. Petrol. 1996, 124, 111–125. [Google Scholar] [CrossRef]
- Burnley, P.C.; Navrotsky, A. Synthesis of high-pressure hydrous magnesium silicates: Observations and analysis. Am. Mineral. 1996, 81, 317–326. [Google Scholar] [CrossRef]
- Smyth, J.R.; Kawamoto, T. Wadsleyite II: A new high pressure hydrous phase in the peridotite-H2O system. Earth Planet. Sci. Lett. 1997, 146, E9–E16. [Google Scholar] [CrossRef]
- Shieh, S.R.; Mao, H.K.; Konzett, J.; Hemley, R.J. In-situ high pressure X-ray diffraction of phase E to 15 GPa. Am. Mineral. 2000, 85, 765–769. [Google Scholar] [CrossRef]
- Sharp, W.E. Pyrrhotite: A common inclusion in South Africa diamonds. Nature 1966, 211, 402–403. [Google Scholar] [CrossRef]
- Gurney, J.J. Diamonds. In Kimberlites and Related Rocks Volume 2: Their Mantle/Crustal Setting, Diamonds and Diamond Exploration, Proceedings of the Fourth International Kimberlite Conference, Perth, Australia, 11–15 August 1986; Ross, J., Jaques, A.L., Ferguson, J., Green, D.H., O’Reilly, S.Y., Danchin, R.Y., Janse, A.J.A., Eds.; Geological Society of Australia Special Publication 14 (2); Geological Society of Australia: Hornsby, Australia, 1989; pp. 935–965. [Google Scholar]
- Yefimova, E.S.; Sobolev, N.V.; Pospelova, L.N. Sulfide inclusions in diamond and specific features of their paragenesis. Zap. Vsesoyuznogo Mineral. Obs. 1983, 112, 300–310. (In Russian) [Google Scholar]
- Zhimulev, E.I.; Chepurov, A.I.; Sinyakova, E.F.; Sonin, V.M.; Chepurov, A.A.; Pokhilenko, N.P. Diamond crystallization in the Fe-Co-S-C and Fe-Ni-S-C systems and the role of sulfide-metal melts in the genesis of diamond. Geochem. Int. 2012, 50, 205–216. [Google Scholar] [CrossRef]
- Palyanov, Y.N.; Borzdov, Y.M.; Bataleva, Y.V.; Kupriyanov, I.N. Diamond formation during sulfidation of metal–carbon melts. Diam. Relat. Mater. 2021, 120, 108660. [Google Scholar] [CrossRef]
- Fleet, M.E.; MacRae, N.D.; Herzberg, C.T. Partition of nickel between olivine and sulfide: A test for immiscible liquids. Contrib. Mineral. Petrol. 1977, 65, 191–197. [Google Scholar] [CrossRef]
- Fleet, M.E.; MacRae, N.D. Partition of Ni between olivine and sulfide: The effect of temperature, fO2 and fS2. Contrib. to Mineral. Petrol. 1987, 95, 75–81. [Google Scholar] [CrossRef]
- Fleet, M.E.; MacRae, N.D. Partition of nickel between olivine and sulfide: Equilibria with sulfide-oxide liquids. Contrib. Mineral. Petrol. 1988, 100, 462–469. [Google Scholar] [CrossRef]
- Deines, P.; Harris, J.W. Sulfide inclusion chemistry and carbon isotopes of African diamonds. Geochim. Cosmochim. Acta 1995, 59, 3173–3188. [Google Scholar] [CrossRef]
- Harris, J.W.; Gurney, J.J. Inclusions in diamond. In The Properties of Diamond; Field, J.E., Ed.; Academic Press: London, UK, 1979; pp. 555–591. [Google Scholar]
- Kennedy, C.S.; Kennedy, G.C. The equilibrium boundary between graphite and diamond. J. Geophys. Res. 1976, 81, 2467–2470. [Google Scholar] [CrossRef]
- Craig, J.R.; Kullerud, G. Phase relations in the Cu-Fe-Ni-S system and their application to magmatic ore deposits. Econ. Geol. Monogr. 1969, 4, 344–358. [Google Scholar]
- Sugaki, A.; Kitakaze, A. High form of pentlandite and its thermal stability. Am. Mineral. 1998, 83, 133–140. [Google Scholar] [CrossRef]
- Andersen, T.; Griffin, W.L.; O’Reilly, S.Y. Primary sulphide melt inclusions in mantle-derived megacrysts and pyroxenites. Lithos 1987, 20, 279–294. [Google Scholar] [CrossRef]
- Meyer, H.O.A.; Svisero, D.P. Mineral inclusions in Brazilian diamonds. Phys. Chem. Earth 1975, 9, 785–795. [Google Scholar] [CrossRef]
- Otter, M.L.; Gurney, J.J. Inclusions in diamonds from the Sloan diatremes, Colorado-Wyoming State Line kimberlite district, North America. In Kimberlites and Related Rocks Volume 2: Their Mantle/Crustal Setting, Diamonds and Diamond Exploration, Proceedings of the Fourth International Kimberlite Conference, Perth, Australia, 11–15 August 1986; Ross, J., Jaques, A.L., Ferguson, J., Green, D.H., O’Reilly, S.Y., Danchin, R.Y., Janse, A.J.A., Eds.; Geological Society of Australia Special Publication 14 (2); Geological Society of Australia: Hornsby, Australia, 1989; pp. 1042–1053. [Google Scholar]
- Sobolev, N.V.; Kaminsky, F.V.; Griffin, W.L.; Yefimova, E.S.; Win, T.T.; Ryan, C.G.; Botkunov, A.I. Mineral inclusions in diamonds from the Sputnik kimberlite pipe, Yakutia. Lithos 1997, 39, 135–157. [Google Scholar] [CrossRef]
- Taylor, L.A.; Liu, Y. Sulfide inclusions in diamonds: Not monosulfide solid solution. Russ. Geol. Geophys. 2009, 50, 1201–1211. [Google Scholar] [CrossRef]
- Li, C.; Barnes, S.-J.; Makovicky, E.; Rose-Hansen, J.; Makovicky, M. Partitioning of nickel, copper, iridium, rhenium, platinum, and palladium between monosulfide solid solution and sulfide liquid: Effects of composition and temperature. Geochim. Cosmochim. Acta 1996, 60, 1231–1238. [Google Scholar] [CrossRef]





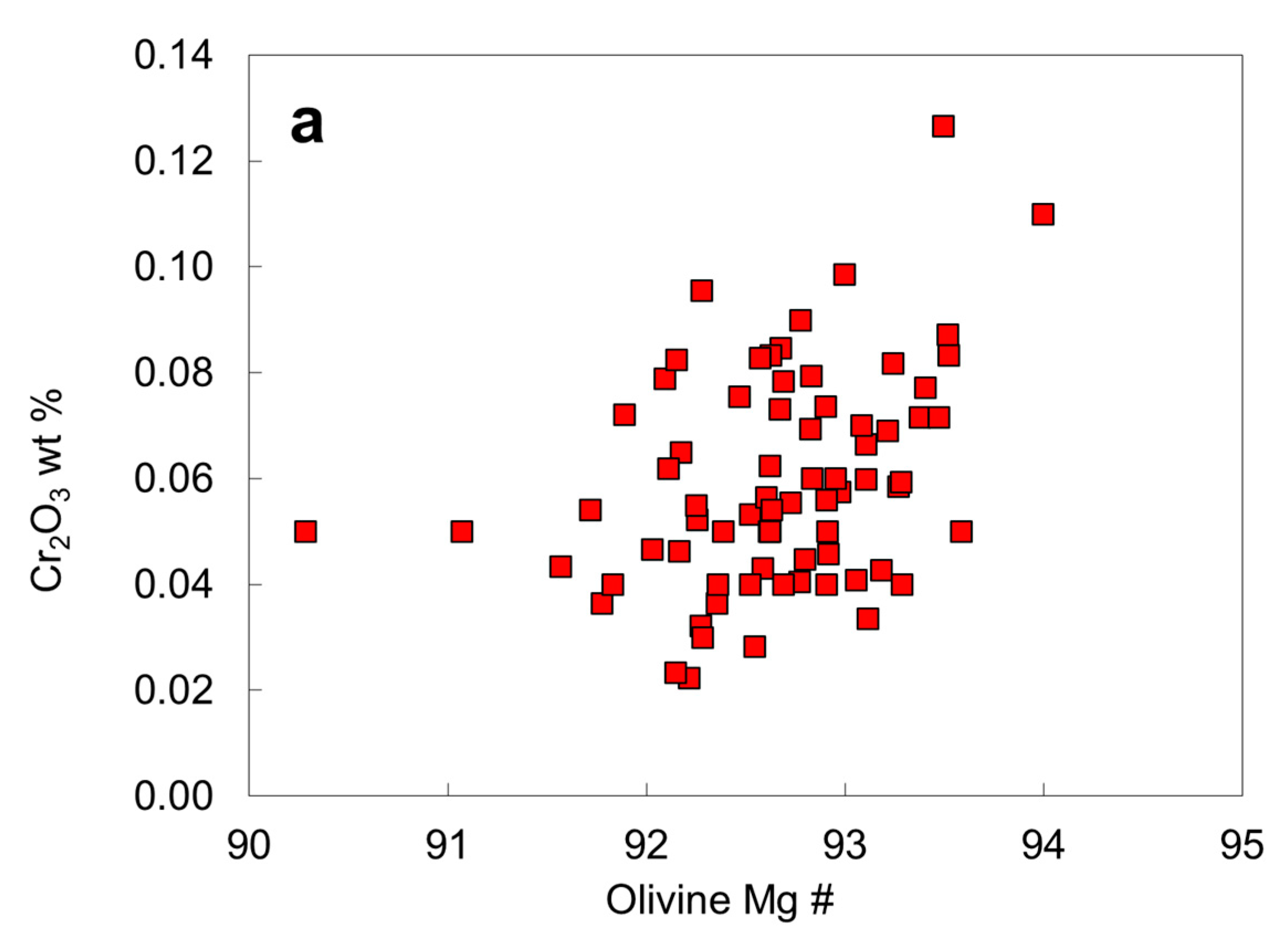
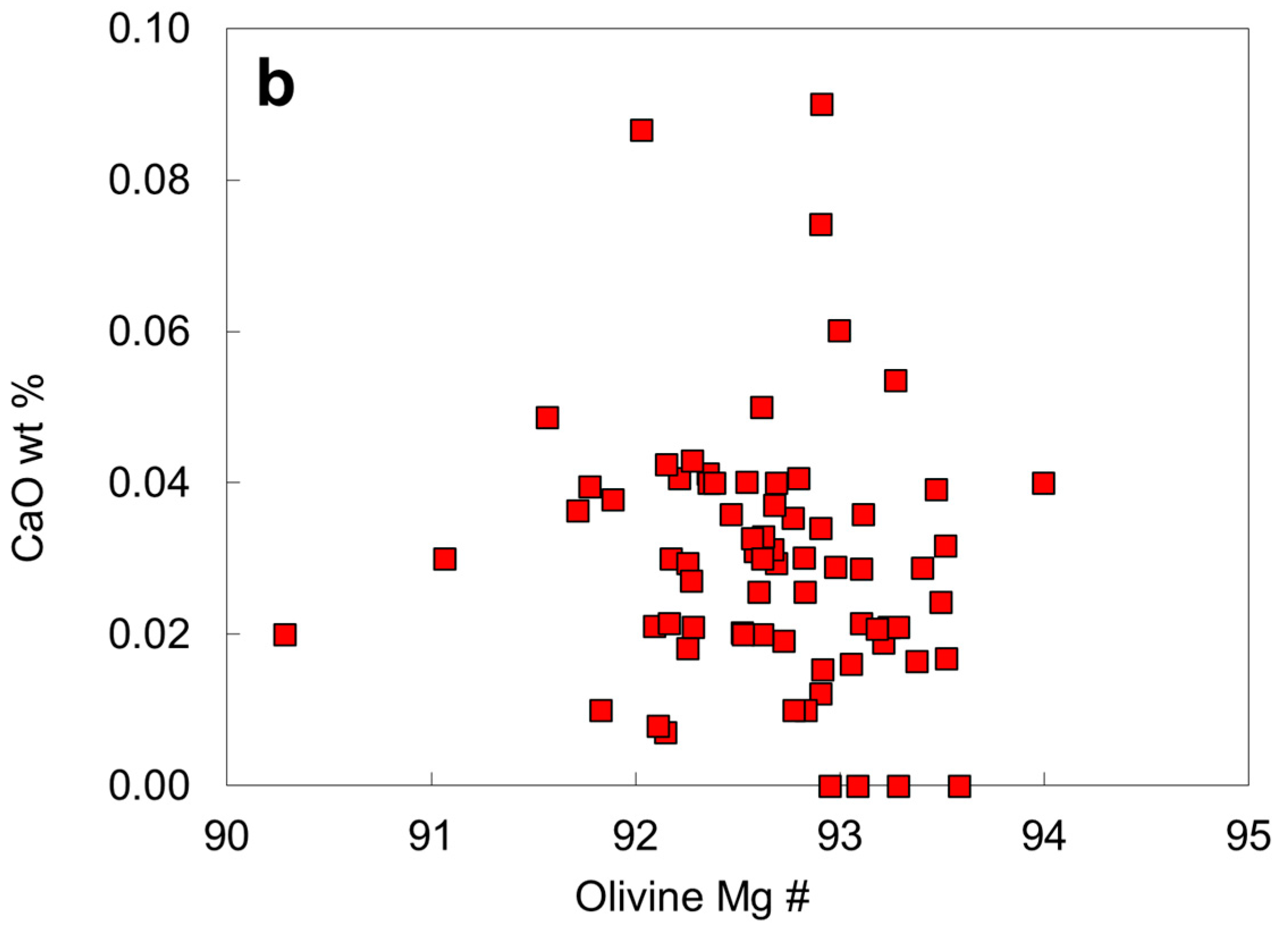
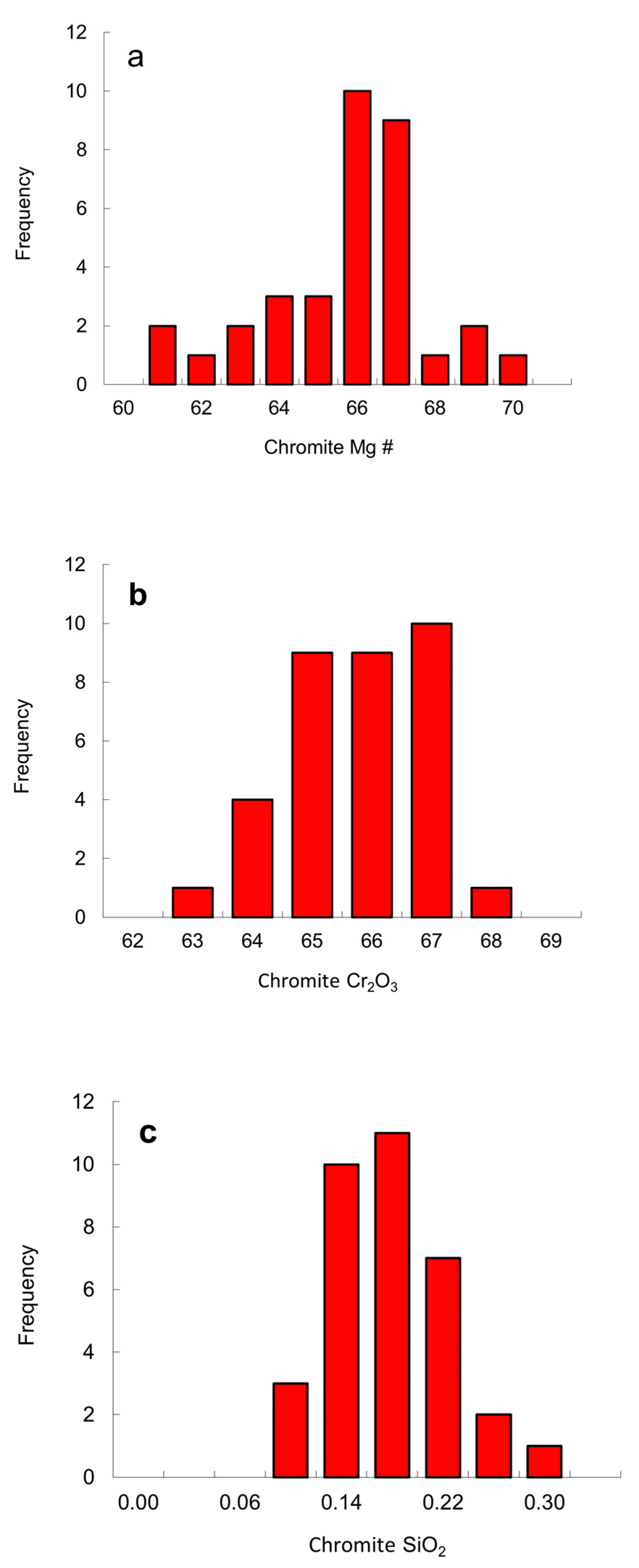


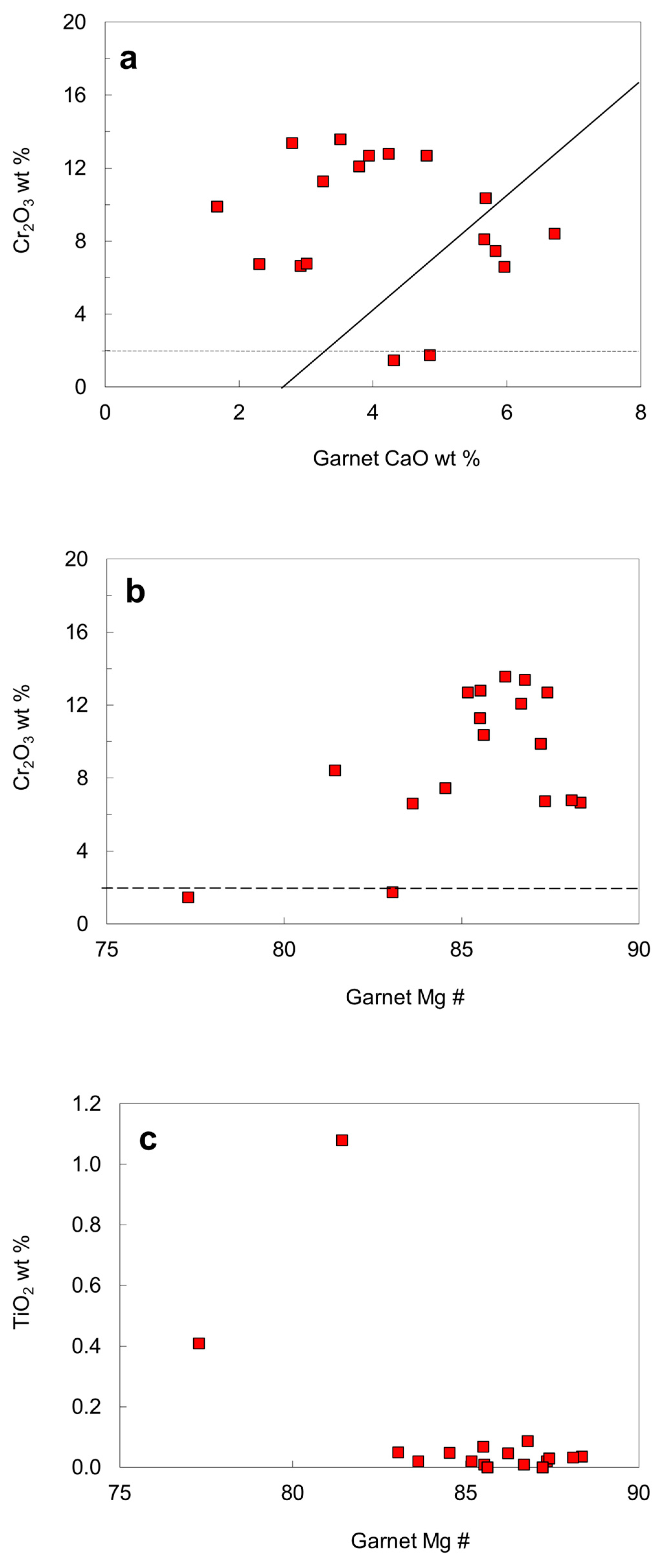
| Protogenetic/Syngenetic | Epigenetic | Uncertain | |
|---|---|---|---|
| Ultramafic | Eclogitic | ||
| forsterite * | Omphacite [3,20] | calcite * | phlogopite [3,20] |
| enstatite * | coesite (quartz) [9] | magnesite * | magnetite [20] |
| Diopside [3,15,20] | rutile [20] | graphite * | apatite [3,20] |
| pyrope * | ilmenite [20] | moissanite [5] | |
| spinel * | graphite * | graphite * | |
| ilmenite [20] | native iron [9,20] | ||
| sulfides * | Fe-dominant phase * | ||
| zircon [20] | Cr-Fe-Ni alloy [20] | ||
| diamond * | |||
| graphite * | |||
| native iron [9,20]? | |||
| native chromium [20]? | |||
| magnesite * | |||
| Ca-carbonate * | |||
| hydrous Mg-silicate * | |||
| Sample | LN50D01 | LN50D02 | LN50D02 | LN50D03 | LN50D03 | LN50D03 | LN50D04 | LN50D04 | LN50D06 | LN50D06 | LN50D06 | LN50D11 | LN50D11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inclusion | 1 (large) | 2 (small) | 1 (small) | 2 (large) | 3 | 1 | 2 | 1 | 2 | 3 | 1 | 2 | ||
| SiO2 | 41.21 | 39.98 | 40.61 | 41.08 | 41.36 | 41.22 | 41.38 | 42.02 | 41.20 | 40.69 | 40.69 | 40.58 | 41.14 | |
| TiO2 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.00 | 0.01 | 0.01 | 0.00 | |
| Al2O3 | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 0.00 | 0.01 | 0.02 | 0.00 | 0.01 | |
| Cr2O3 | 0.05 | 0.04 | 0.04 | 0.08 | 0.08 | 0.13 | 0.07 | 0.09 | 0.07 | 0.06 | 0.04 | 0.04 | 0.02 | |
| TFeO | 7.39 | 7.33 | 7.11 | 6.38 | 6.52 | 6.35 | 6.48 | 6.39 | 7.08 | 7.24 | 7.05 | 7.49 | 7.56 | |
| NiO | 0.37 | 0.35 | 0.34 | 0.34 | 0.35 | 0.39 | 0.34 | 0.26 | 0.37 | 0.36 | 0.34 | 0.39 | 0.38 | |
| MnO | 0.11 | 0.10 | 0.10 | 0.09 | 0.13 | 0.06 | 0.11 | 0.09 | 0.09 | 0.12 | 0.12 | 0.11 | 0.09 | |
| MgO | 51.28 | 51.32 | 51.17 | 51.62 | 51.74 | 51.14 | 51.27 | 51.72 | 51.35 | 51.02 | 50.98 | 50.72 | 50.18 | |
| CaO | 0.02 | 0.03 | 0.04 | 0.02 | 0.03 | 0.02 | 0.02 | 0.03 | 0.03 | 0.03 | 0.04 | 0.04 | 0.04 | |
| Na2O | na | na | na | na | na | 0.02 | 0.00 | 0.00 | na | na | na | na | 0.02 | |
| Σ | 100.45 | 99.17 | 99.42 | 99.62 | 100.22 | 99.37 | 99.69 | 100.61 | 100.20 | 99.53 | 99.29 | 99.37 | 99.44 | |
| Si | 0.994 | 0.975 | 0.988 | 0.995 | 0.997 | 1.003 | 1.004 | 1.009 | 0.996 | 0.990 | 0.992 | 0.990 | 1.005 | |
| Al | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | |
| Ti | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | |
| Cr | 0.001 | 0.001 | 0.001 | 0.002 | 0.001 | 0.002 | 0.001 | 0.002 | 0.001 | 0.001 | 0.001 | 0.001 | 0.000 | |
| Fe | 0.149 | 0.149 | 0.145 | 0.129 | 0.131 | 0.129 | 0.132 | 0.128 | 0.143 | 0.147 | 0.144 | 0.153 | 0.154 | |
| Ni | 0.007 | 0.007 | 0.007 | 0.007 | 0.007 | 0.008 | 0.007 | 0.005 | 0.007 | 0.007 | 0.007 | 0.008 | 0.007 | |
| Mn | 0.002 | 0.002 | 0.002 | 0.002 | 0.003 | 0.001 | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | |
| Mg | 1.845 | 1.865 | 1.856 | 1.865 | 1.859 | 1.854 | 1.854 | 1.852 | 1.850 | 1.851 | 1.853 | 1.845 | 1.828 | |
| Ca | 0.001 | 0.001 | 0.001 | 0.000 | 0.001 | 0.001 | 0.000 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | |
| Na | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | |
| Σcation | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | |
| ΣO | 3.995 | 3.975 | 3.989 | 3.996 | 3.998 | 4.004 | 4.005 | 4.010 | 3.997 | 3.991 | 3.993 | 3.991 | 4.005 | |
| Mg # | 92.5 | 92.6 | 92.8 | 93.5 | 93.4 | 93.5 | 93.4 | 93.5 | 92.8 | 92.6 | 92.8 | 92.4 | 92.2 | |
| LN50D12 | LN50D12 | LN50D14 | LN50D14 | LN50D20 | LN50D21 | LN50D30 | LN50D35 | LN50D35 | LN50D39 | LN50D44 | LN50D45 | LN50D50 | LN50D50 | |
| 1 (large) | 2 (small) | 1 | 2 | 1 (large) | 2 (small) | 1 (large) | 2 | |||||||
| 41.52 | 40.92 | 40.94 | 40.48 | 40.81 | 40.83 | 39.71 | 41.13 | 41.18 | 41.34 | 41.54 | 41.11 | 40.76 | 40.95 | |
| 0.02 | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.01 | 0.01 | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | |
| 0.05 | 0.12 | 0.04 | 0.34 | 0.02 | 0.02 | 0.02 | 0.01 | 0.01 | 0.02 | 0.00 | 0.01 | 0.01 | 0.02 | |
| 0.08 | 0.07 | 0.08 | 0.05 | 0.02 | 0.08 | 0.04 | 0.04 | 0.07 | 0.08 | 0.10 | 0.06 | 0.05 | 0.07 | |
| 7.78 | 7.56 | 7.70 | 7.79 | 7.73 | 7.05 | 8.07 | 6.87 | 6.77 | 7.33 | 7.59 | 6.97 | 7.61 | 7.92 | |
| 0.36 | 0.43 | 0.42 | 0.44 | 0.40 | 0.35 | 0.42 | 0.35 | 0.30 | 0.38 | 0.39 | 0.45 | 0.41 | 0.40 | |
| 0.11 | 0.13 | 0.06 | 0.12 | 0.11 | 0.09 | 0.13 | 0.09 | 0.10 | 0.15 | 0.10 | 0.12 | 0.12 | 0.05 | |
| 50.78 | 49.89 | 50.68 | 50.41 | 50.90 | 51.20 | 49.15 | 51.62 | 52.10 | 50.45 | 50.86 | 51.73 | 50.80 | 50.35 | |
| 0.02 | 0.03 | 0.04 | 0.09 | 0.01 | 0.03 | 0.05 | 0.02 | 0.02 | 0.04 | 0.04 | 0.03 | 0.03 | 0.04 | |
| 0.02 | 0.01 | 0.01 | 0.01 | na | 0.01 | 0.02 | 0.01 | 0.01 | na | na | 0.01 | 0.01 | 0.02 | |
| 100.74 | 99.14 | 99.97 | 99.73 | 100.01 | 99.67 | 97.62 | 100.15 | 100.55 | 99.78 | 100.63 | 100.48 | 99.82 | 99.82 | |
| 1.002 | 1.004 | 0.995 | 0.986 | 0.990 | 0.991 | 0.990 | 0.993 | 0.989 | 1.006 | 1.003 | 0.989 | 0.991 | 0.997 | |
| 0.001 | 0.003 | 0.001 | 0.010 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | |
| 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | |
| 0.002 | 0.001 | 0.002 | 0.001 | 0.000 | 0.002 | 0.001 | 0.001 | 0.001 | 0.001 | 0.002 | 0.001 | 0.001 | 0.001 | |
| 0.157 | 0.155 | 0.156 | 0.159 | 0.157 | 0.143 | 0.168 | 0.139 | 0.136 | 0.149 | 0.153 | 0.140 | 0.155 | 0.161 | |
| 0.007 | 0.008 | 0.008 | 0.009 | 0.008 | 0.007 | 0.008 | 0.007 | 0.006 | 0.007 | 0.008 | 0.009 | 0.008 | 0.008 | |
| 0.002 | 0.003 | 0.001 | 0.002 | 0.002 | 0.002 | 0.003 | 0.002 | 0.002 | 0.003 | 0.002 | 0.002 | 0.002 | 0.001 | |
| 1.827 | 1.825 | 1.835 | 1.831 | 1.842 | 1.853 | 1.827 | 1.858 | 1.865 | 1.831 | 1.831 | 1.856 | 1.841 | 1.828 | |
| 0.001 | 0.001 | 0.001 | 0.002 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | |
| 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.001 | |
| 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | |
| 4.003 | 4.006 | 3.996 | 3.991 | 3.991 | 3.993 | 3.990 | 3.993 | 3.990 | 4.007 | 4.004 | 3.990 | 3.992 | 3.998 | |
| 92.1 | 92.2 | 92.1 | 92.0 | 92.1 | 92.8 | 91.6 | 93.1 | 93.2 | 92.5 | 92.3 | 93.0 | 92.3 | 91.9 | |
| LN50D69 | LN50D69 | LN50D69 | LN50D70 | LN50D72 | LN50D74 | LN50D79 | LN50D91 | LN50D91 | LN50D91 | LN50D91 | LN50D94 | LN50D95 | LN50D96 | LN50D99 |
| 1 (large) | 2 (small) | 1 (large) | 2 (small) | 3 | 4 | |||||||||
| 40.54 | 41.21 | 41.06 | 41.39 | 41.83 | 40.75 | 40.87 | 41.70 | 40.82 | 40.76 | 40.98 | 41.29 | 41.27 | 40.19 | 41.01 |
| 0.01 | 0.00 | 0.00 | 0.01 | 0.00 | 0.02 | 0.01 | 0.01 | 0.01 | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 |
| 0.02 | 0.03 | 0.02 | 0.01 | 0.05 | 0.04 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.00 |
| 0.10 | 0.07 | 0.06 | 0.06 | 0.08 | 0.06 | 0.07 | 0.03 | 0.06 | 0.05 | 0.03 | 0.03 | 0.04 | 0.03 | 0.04 |
| 6.82 | 6.65 | 6.67 | 6.53 | 7.29 | 6.75 | 6.94 | 7.68 | 7.61 | 7.75 | 7.65 | 6.85 | 6.72 | 7.29 | 7.08 |
| 0.34 | 0.36 | 0.35 | 0.31 | 0.36 | 0.29 | 0.35 | 0.36 | 0.36 | 0.33 | 0.34 | 0.36 | 0.32 | 0.33 | 0.40 |
| 0.12 | 0.07 | 0.06 | 0.06 | 0.14 | 0.12 | 0.10 | 0.11 | 0.09 | 0.06 | 0.08 | 0.11 | 0.10 | 0.07 | 0.04 |
| 50.78 | 50.33 | 51.82 | 50.85 | 50.94 | 51.12 | 50.92 | 51.39 | 50.84 | 51.13 | 51.27 | 51.94 | 51.55 | 50.73 | 52.02 |
| 0.06 | 0.03 | 0.05 | 0.02 | 0.03 | 0.02 | 0.03 | 0.03 | 0.02 | 0.02 | 0.02 | 0.04 | 0.02 | 0.04 | 0.01 |
| 0.00 | 0.02 | 0.03 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.00 | 0.00 | 0.01 | 0.01 |
| 98.78 | 98.76 | 100.12 | 99.24 | 100.74 | 99.17 | 99.30 | 101.32 | 99.81 | 100.12 | 100.38 | 100.64 | 100.05 | 98.72 | 100.61 |
| 0.993 | 1.011 | 0.990 | 1.009 | 1.008 | 0.993 | 0.996 | 0.999 | 0.992 | 0.987 | 0.990 | 0.992 | 0.997 | 0.985 | 0.985 |
| 0.000 | 0.001 | 0.001 | 0.000 | 0.002 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 |
| 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| 0.002 | 0.001 | 0.001 | 0.001 | 0.002 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 |
| 0.140 | 0.136 | 0.134 | 0.133 | 0.147 | 0.138 | 0.141 | 0.154 | 0.155 | 0.157 | 0.154 | 0.138 | 0.136 | 0.149 | 0.142 |
| 0.007 | 0.007 | 0.007 | 0.006 | 0.007 | 0.006 | 0.007 | 0.007 | 0.007 | 0.006 | 0.007 | 0.007 | 0.006 | 0.007 | 0.008 |
| 0.002 | 0.001 | 0.001 | 0.001 | 0.003 | 0.002 | 0.002 | 0.002 | 0.002 | 0.001 | 0.002 | 0.002 | 0.002 | 0.002 | 0.001 |
| 1.854 | 1.841 | 1.863 | 1.848 | 1.830 | 1.858 | 1.850 | 1.836 | 1.842 | 1.846 | 1.846 | 1.859 | 1.857 | 1.854 | 1.863 |
| 0.002 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.000 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.000 |
| 0.000 | 0.001 | 0.002 | 0.000 | 0.001 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| 3.994 | 4.011 | 3.990 | 4.010 | 4.009 | 3.995 | 3.997 | 3.999 | 3.993 | 3.988 | 3.990 | 3.992 | 3.998 | 3.986 | 3.985 |
| 93.0 | 93.1 | 93.3 | 93.3 | 92.6 | 93.1 | 92.9 | 92.3 | 92.3 | 92.2 | 92.3 | 93.1 | 93.2 | 92.5 | 92.9 |
| Sample | LN50D03 | LN50D04 | LN50D07 | LN50D12 | LN50D20 | LN50D58 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grain | Sp2 | Sp 3 | Sp 4 | Sp 5 | Sp 1 | Sp 2 | Sp 3 | Sp 4 | Sp | Sp 1 | Sp 2 | Sp 1 | Sp 2 | |
| SiO2 | 0.20 | 0.18 | 0.16 | 0.19 | 0.19 | 0.12 | 0.12 | 0.11 | 0.15 | 0.17 | 0.14 | 0.14 | 0.16 | 0.21 |
| TiO2 | 0.20 | 0.22 | 0.19 | 0.18 | 0.17 | 0.41 | 0.32 | 0.42 | 0.41 | 0.45 | 0.05 | 0.06 | 0.03 | 0.04 |
| Al2O3 | 6.28 | 5.83 | 6.08 | 5.77 | 5.88 | 4.83 | 4.82 | 4.82 | 4.73 | 4.20 | 4.92 | 4.96 | 6.72 | 4.76 |
| Cr2O3 | 64.80 | 66.76 | 66.48 | 65.93 | 65.34 | 66.10 | 66.31 | 66.28 | 66.74 | 66.72 | 64.26 | 65.00 | 64.31 | 66.90 |
| V2O3 | 0.18 | 0.14 | 0.16 | 0.17 | 0.14 | 0.16 | 0.17 | 0.18 | 0.22 | 0.30 | 0.25 | 0.26 | 0.14 | 0.19 |
| TFeO | 13.38 | 13.41 | 13.25 | 13.32 | 13.24 | 15.41 | 15.39 | 15.16 | 15.03 | 15.79 | 16.42 | 16.62 | 13.76 | 14.22 |
| NiO | 0.00 | 0.09 | 0.15 | 0.10 | 0.13 | 0.04 | 0.13 | 0.09 | 0.14 | 0.16 | 0.08 | 0.09 | 0.15 | 0.00 |
| MnO | 0.00 | 0.23 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.24 | 0.32 | 0.00 | 0.00 |
| MgO | 14.22 | 13.74 | 14.23 | 13.28 | 14.43 | 12.74 | 12.72 | 13.46 | 13.43 | 12.56 | 11.96 | 12.08 | 13.70 | 13.37 |
| ZnO | 0.10 | 0.04 | 0.07 | 0.07 | 0.01 | 0.05 | 0.03 | 0.09 | 0.06 | 0.08 | 0.09 | 0.08 | 0.06 | 0.03 |
| Σ | 99.36 | 100.64 | 100.77 | 99.01 | 99.55 | 99.86 | 100.00 | 100.60 | 100.90 | 100.43 | 98.41 | 99.61 | 99.03 | 99.71 |
| Fe2O3 | 2.10 | 1.26 | 1.71 | 0.78 | 2.41 | 1.84 | 1.88 | 2.57 | 2.27 | 1.89 | 2.78 | 2.84 | 2.02 | 1.80 |
| FeO | 11.49 | 12.28 | 11.71 | 12.62 | 11.08 | 13.76 | 13.70 | 12.84 | 12.99 | 14.09 | 13.92 | 14.07 | 11.94 | 12.60 |
| Σ | 99.57 | 100.77 | 100.94 | 99.09 | 99.79 | 100.05 | 100.19 | 100.86 | 101.13 | 100.62 | 98.69 | 99.90 | 99.24 | 99.90 |
| Si | 0.006 | 0.006 | 0.005 | 0.006 | 0.006 | 0.004 | 0.004 | 0.004 | 0.005 | 0.006 | 0.005 | 0.005 | 0.005 | 0.007 |
| Alvi | 0.242 | 0.224 | 0.232 | 0.226 | 0.227 | 0.189 | 0.188 | 0.186 | 0.182 | 0.164 | 0.196 | 0.195 | 0.261 | 0.185 |
| Fe3+ | 0.052 | 0.031 | 0.042 | 0.019 | 0.059 | 0.046 | 0.047 | 0.063 | 0.056 | 0.047 | 0.071 | 0.071 | 0.050 | 0.045 |
| Ti | 0.005 | 0.005 | 0.005 | 0.005 | 0.004 | 0.010 | 0.008 | 0.010 | 0.010 | 0.011 | 0.001 | 0.002 | 0.001 | 0.001 |
| Cr | 1.678 | 1.719 | 1.702 | 1.729 | 1.690 | 1.733 | 1.737 | 1.717 | 1.726 | 1.747 | 1.715 | 1.714 | 1.673 | 1.749 |
| V | 0.005 | 0.004 | 0.004 | 0.004 | 0.004 | 0.004 | 0.005 | 0.005 | 0.006 | 0.008 | 0.007 | 0.007 | 0.004 | 0.005 |
| Fe2+ | 0.315 | 0.335 | 0.317 | 0.350 | 0.303 | 0.382 | 0.380 | 0.352 | 0.355 | 0.390 | 0.393 | 0.392 | 0.329 | 0.348 |
| Ni | 0.000 | 0.002 | 0.004 | 0.003 | 0.003 | 0.001 | 0.003 | 0.002 | 0.004 | 0.004 | 0.002 | 0.002 | 0.004 | 0.000 |
| Mn | 0.000 | 0.006 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.007 | 0.009 | 0.000 | 0.000 |
| Mg | 0.694 | 0.667 | 0.687 | 0.657 | 0.704 | 0.630 | 0.628 | 0.658 | 0.655 | 0.620 | 0.602 | 0.601 | 0.672 | 0.659 |
| Zn | 0.002 | 0.001 | 0.002 | 0.002 | 0.000 | 0.001 | 0.001 | 0.002 | 0.001 | 0.002 | 0.002 | 0.002 | 0.001 | 0.001 |
| Σcation | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| ΣO | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 |
| Mg # | 68.8 | 66.6 | 68.4 | 65.2 | 69.9 | 62.3 | 62.3 | 65.1 | 64.8 | 61.4 | 60.5 | 60.5 | 67.2 | 65.4 |
| Cr # | 87.4 | 88.5 | 88.0 | 88.5 | 88.2 | 90.2 | 90.2 | 90.2 | 90.4 | 91.4 | 89.8 | 89.8 | 86.5 | 90.4 |
| Sample | LN50D10 | LN50D10 | LN50D13 | LN50D19 | LN50D68 | LN50D71 | 40A | 43A | 52A | WG1 | WG5 | WG6 | WG7 | WG8 | L1 | L2 | L3 | L4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 41.83 | 42.38 | 41.22 | 40.78 | 41.05 | 40.52 | 42.83 | 42.21 | 41.24 | 41.49 | 41.70 | 41.00 | 41.34 | 40.22 | 41.50 | 40.60 | 41.20 | 42.00 |
| TiO2 | 0.04 | 0.03 | 0.07 | 0.05 | 0.05 | 0.09 | 0.02 | 0.05 | 0.41 | 0.02 | 0.01 | 0.02 | 1.08 | 0.00 | 0.01 | 0.03 | 0.00 | 0.00 |
| Al2O3 | 19.19 | 19.13 | 15.29 | 14.64 | 18.06 | 15.34 | 18.56 | 21.94 | 22.27 | 18.84 | 13.50 | 13.50 | 15.26 | 15.23 | 14.40 | 13.60 | 16.40 | 17.30 |
| Cr2O3 | 6.66 | 6.78 | 11.29 | 13.59 | 7.46 | 13.38 | 6.74 | 1.76 | 1.48 | 6.61 | 12.80 | 12.70 | 8.42 | 10.37 | 12.10 | 12.70 | 9.90 | 8.10 |
| TFeO | 5.43 | 5.46 | 6.66 | 6.15 | 6.69 | 5.88 | 6.08 | 7.66 | 10.36 | 6.99 | 6.52 | 6.52 | 7.75 | 6.23 | 5.95 | 5.65 | 6.19 | 6.99 |
| NiO | 0.03 | 0.03 | 0.05 | 0.00 | 0.05 | 0.03 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | na | na | 0.00 | 0.02 | 0.01 | 0.07 |
| MnO | 0.22 | 0.17 | 0.22 | 0.15 | 0.24 | 0.21 | 0.36 | 0.32 | 0.38 | 0.39 | 0.35 | 0.36 | 0.15 | 0.21 | 0.30 | 0.32 | 0.26 | 0.38 |
| MgO | 23.11 | 22.65 | 22.04 | 21.59 | 20.52 | 21.64 | 23.52 | 21.04 | 19.77 | 20.01 | 21.60 | 21.00 | 19.05 | 20.80 | 21.70 | 22.00 | 23.70 | 20.30 |
| CaO | 2.91 | 3.01 | 3.25 | 3.51 | 5.83 | 2.79 | 2.30 | 4.85 | 4.31 | 5.96 | 4.23 | 4.80 | 6.71 | 5.68 | 3.79 | 3.94 | 1.67 | 5.66 |
| Na2O | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.03 | 0.01 | 0.04 | 0.07 | 0.02 | 0.00 | 0.00 | 0.07 | na | 0.00 | 0.02 | 0.01 | 0.04 |
| Σ | 99.42 | 99.64 | 100.10 | 100.46 | 99.97 | 99.92 | 100.42 | 99.87 | 100.29 | 100.33 | 100.71 | 99.90 | 99.83 | 98.74 | 99.75 | 98.88 | 99.34 | 100.84 |
| Fe2O3 | 0.65 | 0.00 | 1.19 | 0.55 | 2.18 | 0.00 | 0.15 | 1.52 | 2.39 | 1.28 | 1.03 | 1.58 | 0.42 | 3.01 | 0.18 | 1.97 | 1.81 | 0.94 |
| FeO | 4.85 | 5.46 | 5.59 | 5.66 | 4.74 | 5.88 | 5.95 | 6.29 | 8.21 | 5.84 | 5.59 | 5.10 | 7.37 | 3.53 | 5.79 | 3.87 | 4.56 | 6.15 |
| Σ | 99.49 | 99.64 | 100.21 | 100.51 | 100.18 | 99.92 | 100.43 | 100.02 | 100.53 | 100.46 | 100.81 | 100.06 | 99.87 | 99.04 | 99.77 | 99.08 | 99.52 | 100.93 |
| Si | 2.986 | 3.026 | 2.988 | 2.965 | 2.960 | 2.956 | 3.033 | 2.993 | 2.941 | 2.980 | 3.027 | 3.004 | 3.031 | 2.956 | 3.027 | 2.986 | 2.972 | 3.016 |
| Aliv | 0.014 | 0.000 | 0.012 | 0.035 | 0.040 | 0.044 | 0.000 | 0.007 | 0.059 | 0.020 | 0.000 | 0.000 | 0.000 | 0.044 | 0.000 | 0.014 | 0.028 | 0.000 |
| Alvi | 1.601 | 1.610 | 1.294 | 1.219 | 1.494 | 1.274 | 1.549 | 1.827 | 1.813 | 1.575 | 1.155 | 1.166 | 1.318 | 1.275 | 1.238 | 1.165 | 1.366 | 1.464 |
| Fe3+ | 0.035 | 0.000 | 0.065 | 0.030 | 0.118 | 0.000 | 0.008 | 0.081 | 0.128 | 0.069 | 0.056 | 0.087 | 0.023 | 0.166 | 0.010 | 0.109 | 0.098 | 0.051 |
| Ti | 0.002 | 0.002 | 0.004 | 0.003 | 0.003 | 0.005 | 0.001 | 0.003 | 0.022 | 0.001 | 0.001 | 0.001 | 0.060 | 0.000 | 0.001 | 0.002 | 0.000 | 0.000 |
| Cr | 0.376 | 0.383 | 0.647 | 0.781 | 0.425 | 0.772 | 0.377 | 0.099 | 0.083 | 0.375 | 0.735 | 0.736 | 0.488 | 0.603 | 0.698 | 0.739 | 0.565 | 0.460 |
| Fe2+ | 0.290 | 0.326 | 0.339 | 0.344 | 0.286 | 0.358 | 0.352 | 0.373 | 0.490 | 0.351 | 0.340 | 0.312 | 0.452 | 0.217 | 0.353 | 0.238 | 0.275 | 0.369 |
| Ni | 0.002 | 0.002 | 0.003 | 0.000 | 0.003 | 0.002 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.001 | 0.004 |
| Mn | 0.013 | 0.010 | 0.013 | 0.009 | 0.014 | 0.013 | 0.022 | 0.019 | 0.023 | 0.024 | 0.022 | 0.022 | 0.009 | 0.013 | 0.019 | 0.020 | 0.016 | 0.023 |
| Mg | 2.459 | 2.410 | 2.382 | 2.340 | 2.205 | 2.353 | 2.483 | 2.224 | 2.102 | 2.143 | 2.337 | 2.294 | 2.082 | 2.279 | 2.359 | 2.412 | 2.549 | 2.173 |
| Ca | 0.223 | 0.230 | 0.252 | 0.273 | 0.450 | 0.218 | 0.174 | 0.368 | 0.329 | 0.459 | 0.329 | 0.377 | 0.527 | 0.447 | 0.296 | 0.311 | 0.129 | 0.435 |
| Na | 0.001 | 0.001 | 0.001 | 0.000 | 0.002 | 0.005 | 0.001 | 0.005 | 0.010 | 0.003 | 0.000 | 0.000 | 0.010 | 0.000 | 0.000 | 0.003 | 0.001 | 0.006 |
| Σcation | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 | 8.000 |
| ΣO | 12.000 | 12.024 | 12.000 | 12.000 | 12.000 | 12.003 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 | 12.000 |
| Mg # | 88.3 | 88.1 | 85.5 | 86.2 | 84.5 | 86.8 | 87.3 | 83.0 | 77.3 | 83.6 | 85.5 | 85.2 | 81.4 | 85.6 | 86.7 | 87.4 | 87.2 | 83.8 |
| Ca # | 8.3 | 8.7 | 9.6 | 10.5 | 17.0 | 8.5 | 6.6 | 14.2 | 13.5 | 17.6 | 12.3 | 14.1 | 20.2 | 16.4 | 11.2 | 11.4 | 4.8 | 16.7 |
| Cr # | 18.9 | 19.2 | 33.1 | 38.4 | 21.7 | 36.9 | 19.6 | 5.1 | 4.3 | 19.1 | 38.9 | 38.7 | 27.0 | 31.4 | 36.0 | 38.5 | 28.8 | 23.9 |
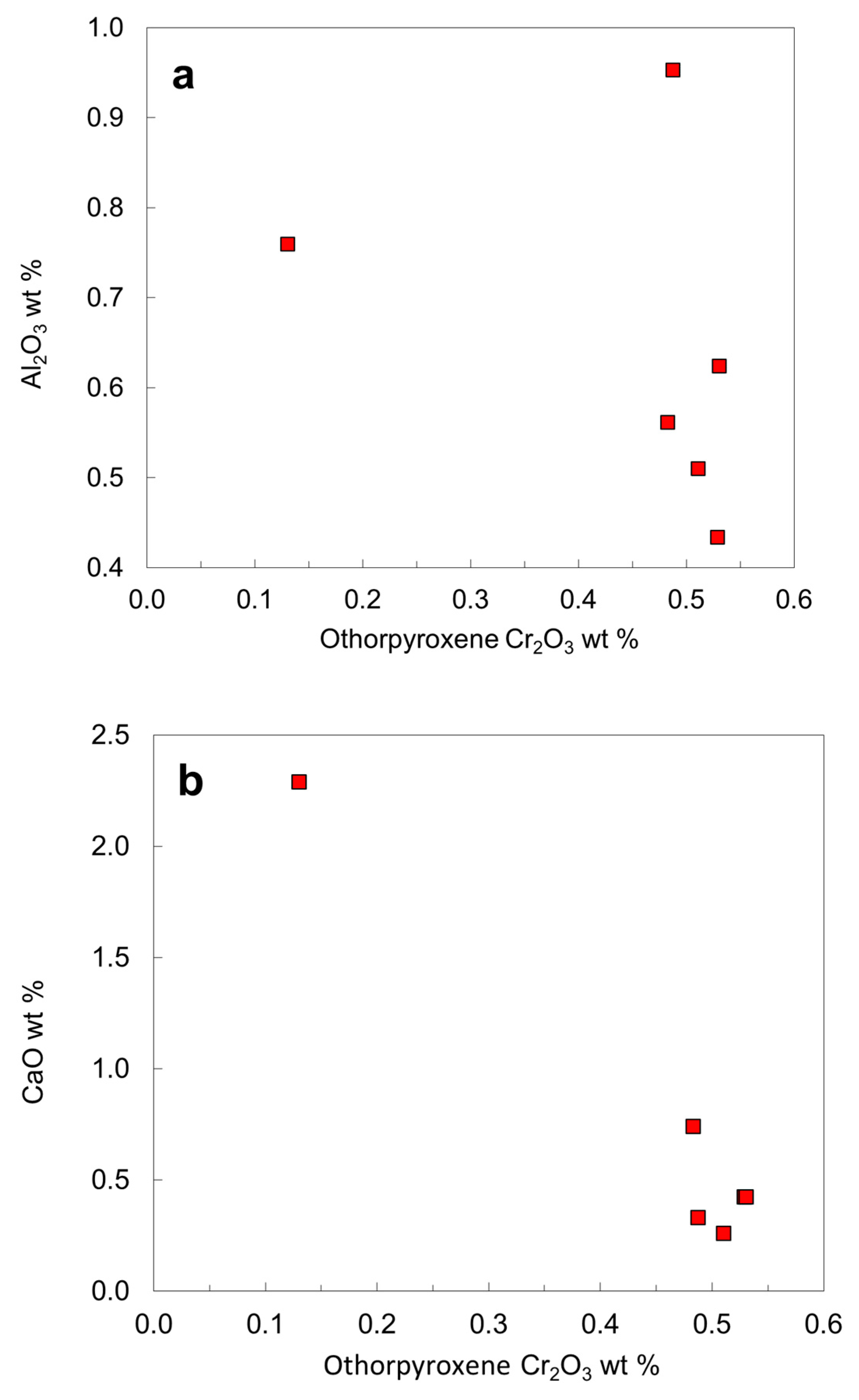
| Orthopyroxene | Clinopyroxene | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sample | LN50D40 | LN50D40 | LN50D52 | LN50D65 | 38A | 52B | Sample | 28B | 43B | 52B |
| Grain | 1 | 2 | Grain | |||||||
| SiO2 | 58.24 | 57.75 | 57.69 | 57.39 | 58.62 | 56.63 | SiO2 | 55.25 | 55.41 | 54.48 |
| TiO2 | 0.00 | 0.02 | 0.01 | 0.02 | 0.00 | 0.11 | TiO2 | 0.07 | 0.01 | 0.19 |
| Al2O3 | 0.43 | 0.62 | 0.95 | 0.56 | 0.51 | 0.76 | Al2O3 | 0.76 | 1.23 | 2.35 |
| Cr2O3 | 0.53 | 0.53 | 0.49 | 0.48 | 0.51 | 0.13 | Cr2O3 | 1.51 | 0.36 | 0.64 |
| TFeO | 4.30 | 4.33 | 4.26 | 4.59 | 4.18 | 7.47 | TFeO | 1.98 | 2.52 | 3.95 |
| NiO | 0.16 | 0.10 | 0.13 | 0.10 | 0.11 | 0.00 | NiO | 0.00 | 0.00 | 0.00 |
| MnO | 0.15 | 0.12 | 0.14 | 0.11 | 0.14 | 0.13 | MnO | 0.09 | 0.07 | 0.14 |
| MgO | 35.22 | 35.08 | 36.41 | 35.49 | 35.62 | 32.22 | MgO | 18.40 | 18.23 | 16.97 |
| CaO | 0.42 | 0.42 | 0.33 | 0.74 | 0.26 | 2.29 | CaO | 20.89 | 21.48 | 18.75 |
| Na2O | 0.11 | 0.12 | 0.03 | 0.14 | 0.00 | 0.31 | Na2O | 0.84 | 0.77 | 1.81 |
| Σ | 99.57 | 99.09 | 100.44 | 99.63 | 99.95 | 100.05 | ∑ | 99.79 | 100.08 | 99.28 |
| Si | 1.002 | 0.998 | 0.981 | 0.986 | 1.005 | 0.984 | Si | 0.999 | 0.998 | 0.989 |
| Aliv | 0.000 | 0.002 | 0.019 | 0.011 | 0.000 | 0.016 | Aliv | 0.001 | 0.002 | 0.011 |
| Alvi | 0.009 | 0.011 | 0.000 | 0.000 | 0.010 | 0.000 | Alvi | 0.016 | 0.024 | 0.039 |
| Ti | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | Ti | 0.001 | 0.000 | 0.003 |
| Cr | 0.007 | 0.007 | 0.007 | 0.007 | 0.007 | 0.002 | Cr | 0.022 | 0.005 | 0.009 |
| Fe | 0.062 | 0.063 | 0.061 | 0.066 | 0.060 | 0.108 | Fe | 0.030 | 0.038 | 0.060 |
| Ni | 0.002 | 0.001 | 0.002 | 0.001 | 0.002 | 0.000 | Ni | 0.000 | 0.000 | 0.000 |
| Mn | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | Mn | 0.001 | 0.001 | 0.002 |
| Mg | 0.904 | 0.904 | 0.923 | 0.909 | 0.910 | 0.834 | Mg | 0.496 | 0.490 | 0.459 |
| Ca | 0.008 | 0.008 | 0.006 | 0.014 | 0.005 | 0.043 | Ca | 0.405 | 0.415 | 0.365 |
| Na | 0.004 | 0.004 | 0.001 | 0.005 | 0.000 | 0.010 | Na | 0.029 | 0.027 | 0.064 |
| Σcation | 2.000 | 2.000 | 2.000 | 2.000 | 2.000 | 2.000 | ∑cation | 2.000 | 2.000 | 2.000 |
| ΣO | 3.009 | 3.006 | 2.993 | 2.993 | 3.013 | 2.988 | ∑O | 3.005 | 3.001 | 2.989 |
| Mg # | 93.6 | 93.5 | 93.8 | 93.2 | 93.8 | 88.5 | Mg# | 94.3 | 92.8 | 88.5 |
| Ca# | 44.9 | 45.9 | 44.3 | |||||||
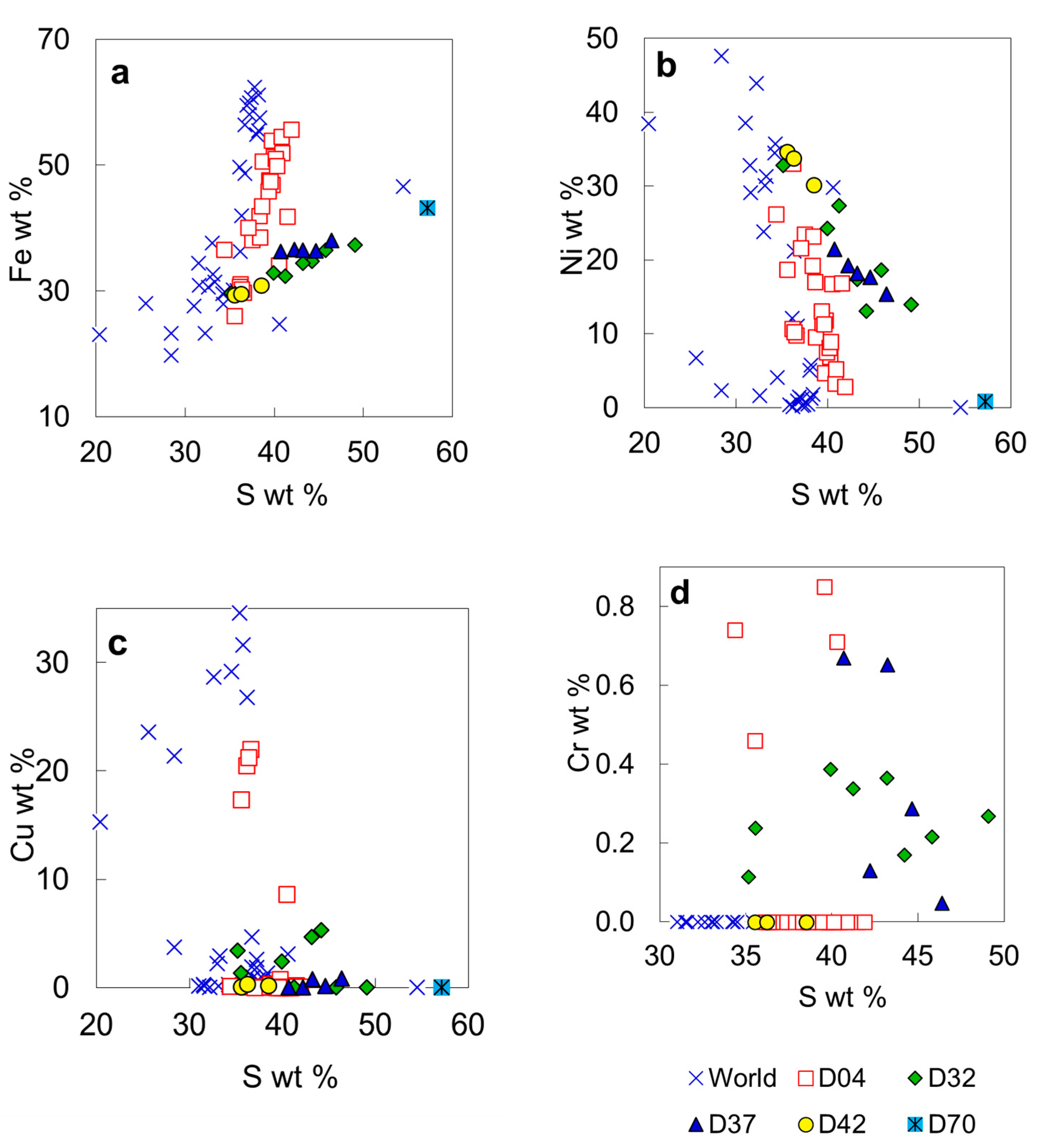
| Sample | LN50D04 | LN50D32 | LN50D37 | LN50D42 | LN50D70 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mineral | grg | grg | trl | Cu po | pnt | R3S4 | R3S4 | po | py | ||
| Fe | 34.13 | 55.66 | 31.10 | 29.81 | 36.55 | 37.35 | 34.43 | 36.55 | 36.49 | 29.54 | 43.21 |
| Ni | 16.73 | 2.84 | 33.08 | 9.78 | 26.19 | 13.91 | 17.33 | 19.22 | 18.13 | 33.75 | 0.80 |
| Cu | 8.64 | 0.06 | 0.06 | 22.00 | 0.15 | 0.00 | 4.65 | 0.00 | 0.76 | 0.33 | 0.00 |
| Zn | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.00 | 0.00 |
| Mn | 0.00 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.01 | 0.01 |
| Co | 0.31 | 0.00 | 0.42 | 0.01 | 0.30 | 0.78 | 0.52 | 0.67 | 0.81 | 0.34 | 0.80 |
| Cr | na | na | na | na | 0.74 | 0.27 | 0.37 | 0.13 | 0.65 | na | na |
| S | 40.47 | 41.90 | 36.18 | 36.54 | 34.36 | 49.08 | 43.18 | 42.22 | 43.22 | 36.23 | 57.17 |
| Σ | 100.29 | 100.46 | 100.83 | 98.15 | 98.29 | 101.39 | 100.47 | 98.81 | 100.07 | 100.18 | 102.00 |
| Sulfur normalized to 4 | |||||||||||
| Fe | 1.936 | 3.051 | 1.974 | 1.873 | 2.442 | 1.747 | 1.831 | 1.988 | 1.938 | 1.872 | 1.736 |
| Ni | 0.903 | 0.148 | 1.998 | 0.585 | 1.665 | 0.619 | 0.877 | 0.995 | 0.916 | 2.035 | 0.030 |
| Cu | 0.431 | 0.003 | 0.003 | 1.215 | 0.009 | 0.000 | 0.217 | 0.000 | 0.035 | 0.018 | 0.000 |
| Zn | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Mn | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 |
| Co | 0.017 | 0.000 | 0.025 | 0.001 | 0.019 | 0.035 | 0.026 | 0.034 | 0.041 | 0.020 | 0.030 |
| Cr | 0.000 | 0.000 | 0.000 | 0.000 | 0.053 | 0.013 | 0.021 | 0.008 | 0.037 | 0.000 | 0.000 |
| S | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 |
| Σcation | 3.287 | 3.202 | 4.000 | 3.674 | 4.189 | 2.415 | 2.972 | 3.025 | 2.969 | 3.947 | 1.797 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, D. Electron Probe Microanalysis and Microscopy of Polishing-Exposed Solid-Phase Mineral Inclusions in Fuxian Kimberlite Diamonds. Minerals 2022, 12, 844. https://doi.org/10.3390/min12070844
Zhao D. Electron Probe Microanalysis and Microscopy of Polishing-Exposed Solid-Phase Mineral Inclusions in Fuxian Kimberlite Diamonds. Minerals. 2022; 12(7):844. https://doi.org/10.3390/min12070844
Chicago/Turabian StyleZhao, Donggao. 2022. "Electron Probe Microanalysis and Microscopy of Polishing-Exposed Solid-Phase Mineral Inclusions in Fuxian Kimberlite Diamonds" Minerals 12, no. 7: 844. https://doi.org/10.3390/min12070844
APA StyleZhao, D. (2022). Electron Probe Microanalysis and Microscopy of Polishing-Exposed Solid-Phase Mineral Inclusions in Fuxian Kimberlite Diamonds. Minerals, 12(7), 844. https://doi.org/10.3390/min12070844






