Uptake and Transfer of Polyamide Microplastics in a Freshwater Mesocosm Study
Abstract
:1. Introduction
2. Materials and Methods
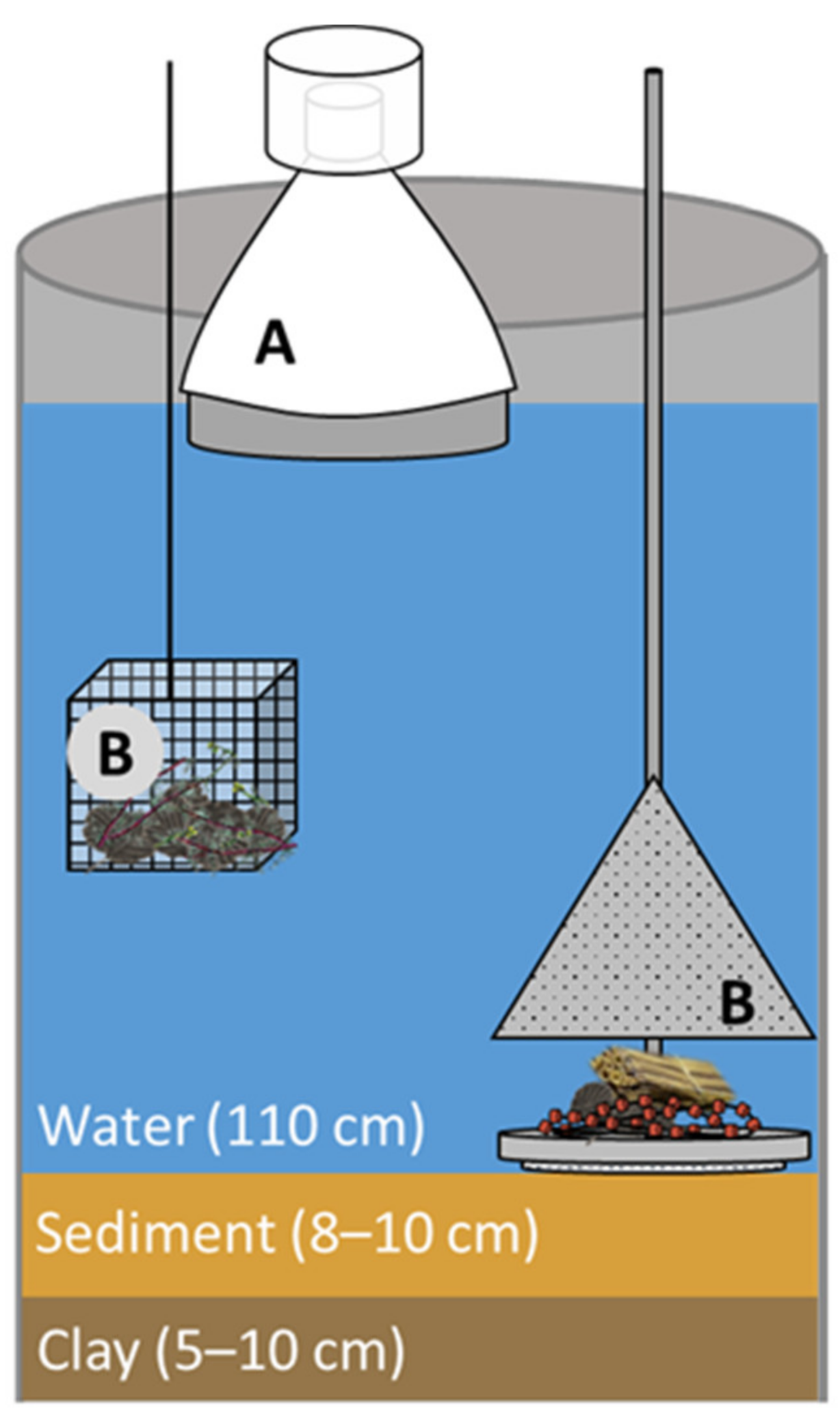
2.1. Preparation of the Test System
2.2. Microplastic Application
2.3. Macroinvertebrate Sampling
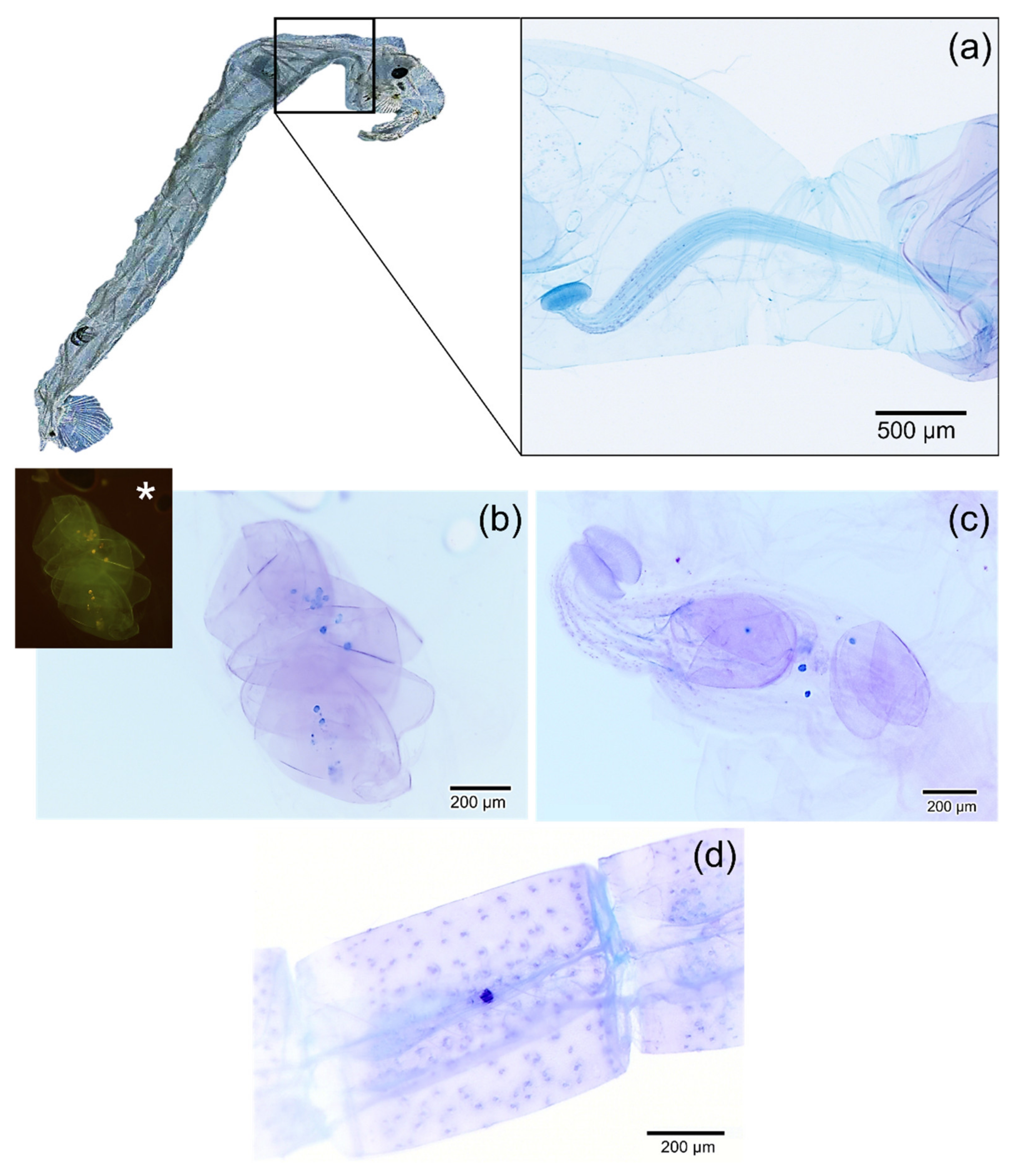
2.4. Detection of Microplastics in Chaoborus sp.
3. Results
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haegerbaeumer, A.; Mueller, M.-T.; Fueser, H.; Traunspurger, W. Impacts of Micro- and Nano-Sized Plastic Particles on Benthic Invertebrates: A Literature Review and Gap Analysis. Front. Environ. Sci. 2019, 7, 17. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Zhang, Y.; Kang, S.; Yang, L.; Shi, H.; Tripathee, L.; Gao, T. Research progresses of microplastic pollution in freshwater systems. Sci. Total Environ. 2021, 795, 148888. [Google Scholar] [CrossRef] [PubMed]
- Dris, R.; Imhof, H.K.; Löder, M.G.; Gasperi, J.; Laforsch, C.; Tassin, B. Microplastic Contamination in Freshwater Systems: Methodological Challenges, Occurrence and Sources. In Microplastic Contamination in Aquatic Environments: An Emerging Matter of Environmental Urgency; Zeng, E.Y., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 51–93. ISBN 9780128137475. [Google Scholar]
- Sarijan, S.; Azman, S.; Said, M.I.M.; Jamal, M.H. Microplastics in freshwater ecosystems: A recent review of occurrence, analysis, potential impacts, and research needs. Environ. Sci. Pollut. Res. Int. 2021, 28, 1341–1356. [Google Scholar] [CrossRef] [PubMed]
- Ivleva, N.P.; Wiesheu, A.C.; Niessner, R. Microplastic in Aquatic Ecosystems. Angew. Chem. Int. Ed. Engl. 2017, 56, 1720–1739. [Google Scholar] [CrossRef]
- Scherer, C.; Weber, A.; Lambert, S.; Wagner, M. Interactions of Microplastics with Freshwater Biota. In Freshwater Microplastics: Emerging Environmental Contaminants? Wagner, M., Lambert, S., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 153–180. ISBN 978-3-319-61615-5. [Google Scholar]
- Lebreton, L.C.M.; van der Zwet, J.; Damsteeg, J.-W.; Slat, B.; Andrady, A.; Reisser, J. River plastic emissions to the world’s oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef]
- Sá, L.C.; de Oliveira, M.; Ribeiro, F.; Rocha, T.L.; Futter, M.N. Studies of the effects of microplastics on aquatic organisms: What do we know and where should we focus our efforts in the future? Sci. Total Environ. 2018, 645, 1029–1039. [Google Scholar] [CrossRef]
- PlasticsEurope. Plastics–the Facts 2018. An Analysis of European Plastics Production, Demand and Waste Data; Plastics Europe AISBL: Bruessels, Belgium, 2018. [Google Scholar]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Goodhead, R.; Moger, J.; Galloway, T.S. Microplastic ingestion by zooplankton. Environ. Sci. Technol. 2013, 47, 6646–6655. [Google Scholar] [CrossRef]
- Scherer, C.; Brennholt, N.; Reifferscheid, G.; Wagner, M. Feeding type and development drive the ingestion of microplastics by freshwater invertebrates. Sci. Rep. 2017, 7, 17006. [Google Scholar] [CrossRef] [Green Version]
- Browne, M.A.; Galloway, T.; Thompson, R. Microplastic—An emerging contaminant of potential concern? Integr. Environ. Assess. Manag. 2007, 3, 559–561. [Google Scholar] [CrossRef]
- Thompson, R.C.; Moore, C.J.; vom Saal, F.S.; Swan, S.H. Plastics, the environment and human health: Current consensus and future trends. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2153–2166. [Google Scholar] [CrossRef]
- Silva, C.J.M.; Silva, A.L.P.; Gravato, C.; Pestana, J.L.T. Ingestion of small-sized and irregularly shaped polyethylene microplastics affect Chironomus riparius life-history traits. Sci. Total Environ. 2019, 672, 862–868. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Environmentally relevant concentrations of polyethylene microplastics negatively impact the survival, growth and emergence of sediment-dwelling invertebrates. Environ. Pollut. 2018, 236, 425–431. [Google Scholar] [CrossRef]
- Cuthbert, R.N.; Al-Jaibachi, R.; Dalu, T.; Dick, J.T.A.; Callaghan, A. The influence of microplastics on trophic interaction strengths and oviposition preferences of dipterans. Sci. Total Environ. 2019, 651, 2420–2423. [Google Scholar] [CrossRef] [Green Version]
- Gouin, T. Toward an Improved Understanding of the Ingestion and Trophic Transfer of Microplastic Particles: Critical Review and Implications for Future Research. Environ. Toxicol. Chem. 2020, 39, 1119–1137. [Google Scholar] [CrossRef]
- Provencher, J.F.; Ammendolia, J.; Rochman, C.M.; Mallory, M.L. Assessing plastic debris in aquatic food webs: What we know and don’t know about uptake and trophic transfer. Environ. Rev. 2019, 27, 304–317. [Google Scholar] [CrossRef]
- Al-Jaibachi, R.; Cuthbert, R.N.; Callaghan, A. Examining effects of ontogenic microplastic transference on Culex mosquito mortality and adult weight. Sci. Total Environ. 2019, 651, 871–876. [Google Scholar] [CrossRef] [Green Version]
- Setyorini, L.; Michler-Kozma, D.; Sures, B.; Gabel, F. Transfer and effects of PET microfibers in Chironomus riparius. Sci. Total Environ. 2021, 757, 143735. [Google Scholar] [CrossRef]
- Weis, J.S.; Palmquist, K.H. Reality Check: Experimental Studies on Microplastics Lack Realism. Appl. Sci. 2021, 11, 8529. [Google Scholar] [CrossRef]
- Ockenden, A.; Tremblay, L.A.; Dikareva, N.; Simon, K.S. Towards more ecologically relevant investigations of the impacts of microplastic pollution in freshwater ecosystems. Sci. Total Environ. 2021, 792, 148507. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Protection Products and Their Residues. Guidance on tiered risk assessment for plant protection products for aquatic organisms in edge-of-field surface waters. EFSA J. 2013, 11, 3290. [Google Scholar] [CrossRef]
- Boyle, T.P.; Fairchild, J.F. The role of mesocosm studies in ecological risk analysis. Ecol. Appl. 1997, 7, 1099–1102. [Google Scholar] [CrossRef]
- Caquet, T.; Lagadic, L.; Jonot, O.; Baturo, W.; Kilanda, M.; Simon, P.; Le Bras, S.; Echaubard, M.; Ramade, F. Outdoor experimental ponds (mesocosms) designed for long-term ecotoxicological studies in aquatic environment. Ecotoxicol. Environ. Saf. 1996, 34, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Caquet, T.; Lagadic, L.; Sheffield, S.R. Mesocosms in ecotoxicology (1): Outdoor aquatic systems. Rev. Environ. Contam. Toxicol. 2000, 165, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Hasselerharm, P.E.; Gort, G.; Peeters, E.T.H.M.; Koelmans, A.A. Nano- and microplastics affect the composition of freshwater benthic communities in the long term. Sci. Adv. 2020, 6, eaay4054. [Google Scholar] [CrossRef] [Green Version]
- Stanković, J.; Milošević, D.; Jovanović, B.; Savić-Zdravković, D.; Petrović, A.; Raković, M.; Stanković, N.; Stojković Piperac, M. In Situ Effects of a Microplastic Mixture on the Community Structure of Benthic Macroinvertebrates in a Freshwater Pond. Environ. Toxicol. Chem. 2021. [Google Scholar] [CrossRef]
- Berendonk, T.U.; Bonsall, M.B. The phantom midge and a comparison of metapopulation structures. Ecology 2002, 83, 116–128. [Google Scholar] [CrossRef]
- Yan, N.D.; Keller, W.; MacIsaac, H.J.; McEachern, L.J. Regulation of Zooplankton Community Structure of an Acidified Lake by Chaoborus. Ecol. Appl. 1991, 1, 52–65. [Google Scholar] [CrossRef]
- Parma, S. The life cycle of Chaoborus crystallinus, Diptera, Chaoboridae in a Dutch pond. Int. Ver. Theor. Angew. Limnol. 1969, 17, 888–889. [Google Scholar]
- Pickford, D.B.; Finnegan, M.C.; Baxter, L.R.; Böhmer, W.; Hanson, M.L.; Stegger, P.; Hommen, U.; Hoekstra, P.F.; Hamer, M. Response of the mayfly (Cloeon dipterum) to chronic exposure to thiamethoxam in outdoor mesocosms. Environ. Toxicol. Chem. 2018, 37, 1040–1050. [Google Scholar] [CrossRef] [Green Version]
- Imhof, H.K.; Rusek, J.; Thiel, M.; Wolinska, J.; Laforsch, C. Do microplastic particles affect Daphnia magna at the morphological, life history and molecular level? PLoS ONE 2017, 12, e0187590. [Google Scholar] [CrossRef] [Green Version]
- Elizalde-Velázquez, A.; Carcano, A.M.; Crago, J.; Green, M.J.; Shah, S.A.; Cañas-Carrell, J.E. Translocation, trophic transfer, accumulation and depuration of polystyrene microplastics in Daphnia magna and Pimephales promelas. Environ. Pollut. 2020, 259, 113937. [Google Scholar] [CrossRef]
- Sturm, M.T.; Horn, H.; Schuhen, K. The potential of fluorescent dyes-comparative study of Nile red and three derivatives for the detection of microplastics. Anal. Bioanal. Chem. 2021, 413, 1059–1071. [Google Scholar] [CrossRef]
- Shim, W.J.; Song, Y.K.; Hong, S.H.; Jang, M. Identification and quantification of microplastics using Nile Red staining. Mar. Pollut. Bull. 2016, 113, 469–476. [Google Scholar] [CrossRef]
- Jin, Y.; Li, H.; Mahar, R.B.; Wang, Z.; Nie, Y. Combined alkaline and ultrasonic pretreatment of sludge before aerobic digestion. J. Environ. Sci. 2009, 21, 279–284. [Google Scholar] [CrossRef]
- Erdbeer, L.; Kiel, E. Methodische Entwicklung zur Sichtbarmachung von Mikroplastik in Chironomiden. In Proceedings of the Annual Conference of the German Limnological Society (DGL) and the German and Austrian Section of the International Limnological Society, Kamp-Lintfort, Germany, 10–14 September 2018. [Google Scholar]
- Tamminga, M. Nile Red Staining as a Subsidiary Method for Microplastic Quantifica-tion: A Comparison of Three Solvents and Factors Influencing Application Reliability. SDRP J. Earth Sci. Environ. Stud. 2017, 2, 165–172. [Google Scholar] [CrossRef] [Green Version]
- Rehse, S.; Kloas, W.; Zarfl, C. Short-term exposure with high concentrations of pristine microplastic particles leads to immobilisation of Daphnia magna. Chemosphere 2016, 153, 91–99. [Google Scholar] [CrossRef]
- Chae, Y.; Kim, D.; Kim, S.W.; An, Y.-J. Trophic transfer and individual impact of nano-sized polystyrene in a four-species freshwater food chain. Sci. Rep. 2018, 8, 284. [Google Scholar] [CrossRef]
- Aljaibachi, R.; Laird, W.B.; Stevens, F.; Callaghan, A. Impacts of polystyrene microplastics on Daphnia magna: A laboratory and a mesocosm study. Sci. Total Environ. 2020, 705, 135800. [Google Scholar] [CrossRef]
- Setälä, O.; Norkko, J.; Lehtiniemi, M. Feeding type affects microplastic ingestion in a coastal invertebrate community. Mar. Pollut. Bull. 2016, 102, 95–101. [Google Scholar] [CrossRef]
- Caquet, T.; Lagadic, L.; Monod, G.; Lacaze, J.C.; Couté, A. Variability of physicochemical and biological parameters between replicated outdoor freshwater lentic mesocosms. Ecotoxicology 2001, 10, 51–66. [Google Scholar] [CrossRef]
- Maes, T.; Jessop, R.; Wellner, N.; Haupt, K.; Mayes, A.G. A rapid-screening approach to detect and quantify microplastics based on fluorescent tagging with Nile Red. Sci. Rep. 2017, 7, 44501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baruah, A.; Sharma, A.; Sharma, S.; Nagraik, R. An insight into different microplastic detection methods. Int. J. Environ. Sci. Technol. 2021, 1–10. [Google Scholar] [CrossRef]
- Wagner, M.; Lambert, S. (Eds.) Freshwater Microplastics: Emerging Environmental Contaminants? Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-61615-5. [Google Scholar]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the marine environment: A review of the methods used for identification and quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, D.; Kowalski, N.; Waniek, J.J. Effects of biofouling on the sinking behavior of microplastics. Environ. Res. Lett. 2017, 12, 124003. [Google Scholar] [CrossRef] [Green Version]
- Lagarde, F.; Olivier, O.; Zanella, M.; Daniel, P.; Hiard, S.; Caruso, A. Microplastic interactions with freshwater microalgae: Hetero-aggregation and changes in plastic density appear strongly dependent on polymer type. Environ. Pollut. 2016, 215, 331–339. [Google Scholar] [CrossRef]
- Leiser, R.; Wu, G.-M.; Neu, T.R.; Wendt-Potthoff, K. Biofouling, metal sorption and aggregation are related to sinking of microplastics in a stratified reservoir. Water Res. 2020, 176, 115748. [Google Scholar] [CrossRef]
- Hübner, M.K.; Michler-Kozma, D.N.; Gabel, F. Microplastic concentrations at the water surface are reduced by decreasing flow velocities caused by a reservoir. Fundam. Appl. Limnol. 2020, 194, 49–56. [Google Scholar] [CrossRef]
- Lindeque, P.K.; Cole, M.; Coppock, R.L.; Lewis, C.N.; Miller, R.Z.; Watts, A.J.R.; Wilson-McNeal, A.; Wright, S.L.; Galloway, T.S. Are we underestimating microplastic abundance in the marine environment? A comparison of microplastic capture with nets of different mesh-size. Environ. Pollut. 2020, 265, 114721. [Google Scholar] [CrossRef]
- Rios Mendoza, L.M.; Balcer, M. Microplastics in freshwater environments: A review of quantification assessment. TrAC Trends Anal. Chem. 2019, 113, 402–408. [Google Scholar] [CrossRef]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef] [Green Version]
- Bucci, K.; Tulio, M.; Rochman, C.M. What is known and unknown about the effects of plastic pollution: A meta-analysis and systematic review. Ecol. Appl. 2020, 30, e02044. [Google Scholar] [CrossRef]
- Meisch, C. Crustacea: Ostracoda; Schwoerbel, J., Zwick, P., Eds.; Spektrum Akad. Verl.: Heidelberg, Germany, 2000; pp. 1–522. ISBN 9783827410016. [Google Scholar]
- Gersch, M. Experimentelle Untersuchungen über den Verdauungstraktus der Larve von Chaoborus (Corethra). Z. Vgl. Physiol. 1952, 34, 346–369. [Google Scholar] [CrossRef]
- Kruckenfellner, L.; Ebke, K.P. Institut für Gewässerschutz Mesocosm GmbH, Homberg (Ohm), Germany. 2022; manuscript in preparation. [Google Scholar]
- Bosker, T.; Olthof, G.; Vijver, M.G.; Baas, J.; Barmentlo, S.H. Significant decline of Daphnia magna population biomass due to microplastic exposure. Environ. Pollut. 2019, 250, 669–675. [Google Scholar] [CrossRef] [Green Version]
- Cáceres, C.E.; Knight, C.J.; Hall, S.R. Predator-spreaders: Predation can enhance parasite success in a planktonic host-parasite system. Ecology 2009, 90, 2850–2858. [Google Scholar] [CrossRef] [Green Version]
- Von Ende, C.N. Fish Predation, Interspecific Predation, and the Distribution of Two Chaoborus Species. Ecology 1979, 60, 119–128. [Google Scholar] [CrossRef]

| Larvae | Imagines | ||||
|---|---|---|---|---|---|
| Treatment (mg L−1 PA) | 150 | 15 | 0 | 150 | 0 |
| Sample size per treatment | 200 | 200 | 200 | 100 | 100 |
| Individuals with PA | 92 | 19 | 4 | 1 | 0 |
| N total particles | 245 | 35 | 7 | 2 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michler-Kozma, D.N.; Kruckenfellner, L.; Heitkamp, A.; Ebke, K.P.; Gabel, F. Uptake and Transfer of Polyamide Microplastics in a Freshwater Mesocosm Study. Water 2022, 14, 887. https://doi.org/10.3390/w14060887
Michler-Kozma DN, Kruckenfellner L, Heitkamp A, Ebke KP, Gabel F. Uptake and Transfer of Polyamide Microplastics in a Freshwater Mesocosm Study. Water. 2022; 14(6):887. https://doi.org/10.3390/w14060887
Chicago/Turabian StyleMichler-Kozma, Diana Noemi, Lukas Kruckenfellner, Anna Heitkamp, Klaus Peter Ebke, and Friederike Gabel. 2022. "Uptake and Transfer of Polyamide Microplastics in a Freshwater Mesocosm Study" Water 14, no. 6: 887. https://doi.org/10.3390/w14060887
APA StyleMichler-Kozma, D. N., Kruckenfellner, L., Heitkamp, A., Ebke, K. P., & Gabel, F. (2022). Uptake and Transfer of Polyamide Microplastics in a Freshwater Mesocosm Study. Water, 14(6), 887. https://doi.org/10.3390/w14060887






