Diversity and Distribution of Sabellida (Annelida) under Protection Regimes
Abstract
1. Introduction
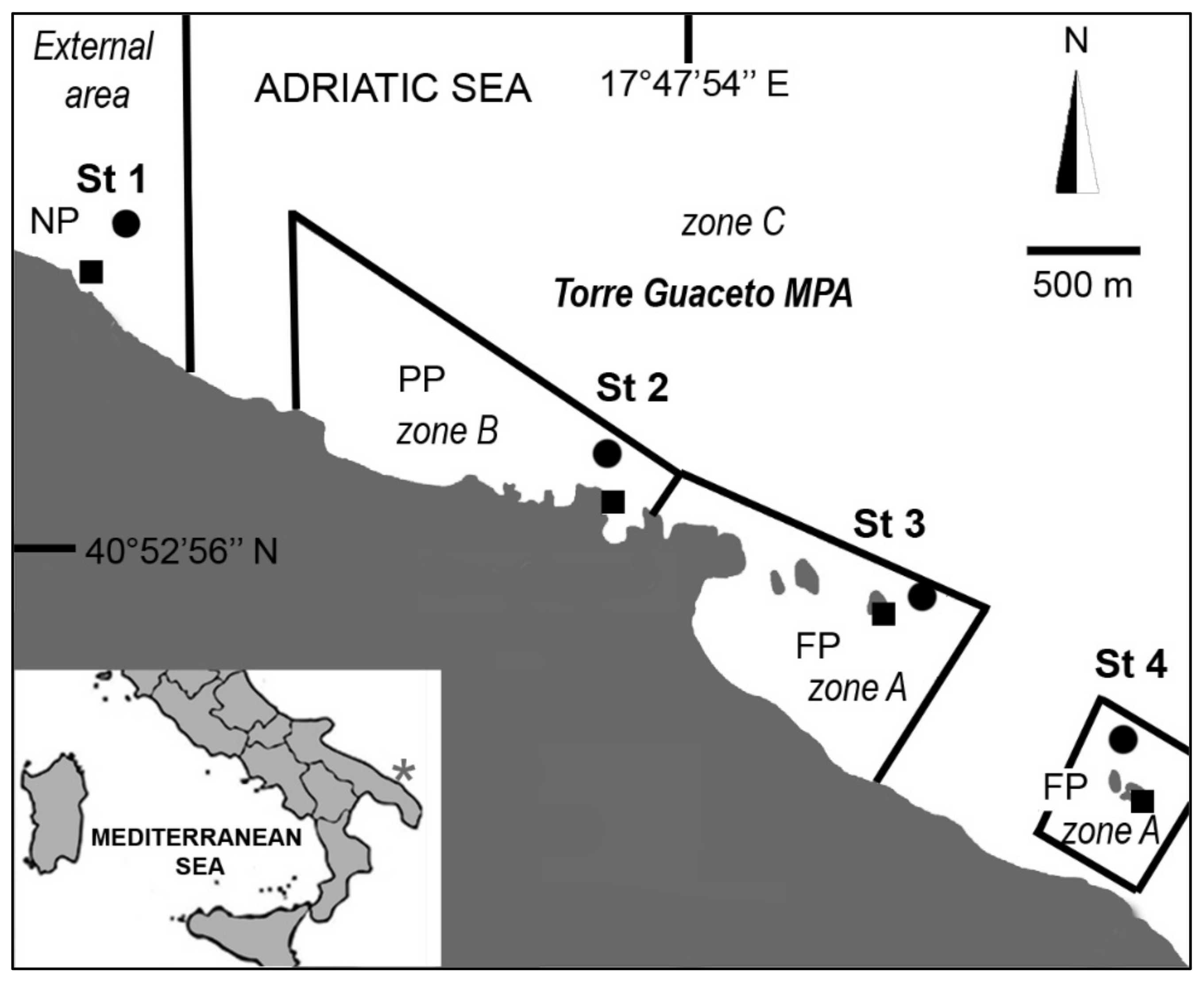
2. Materials and Methods
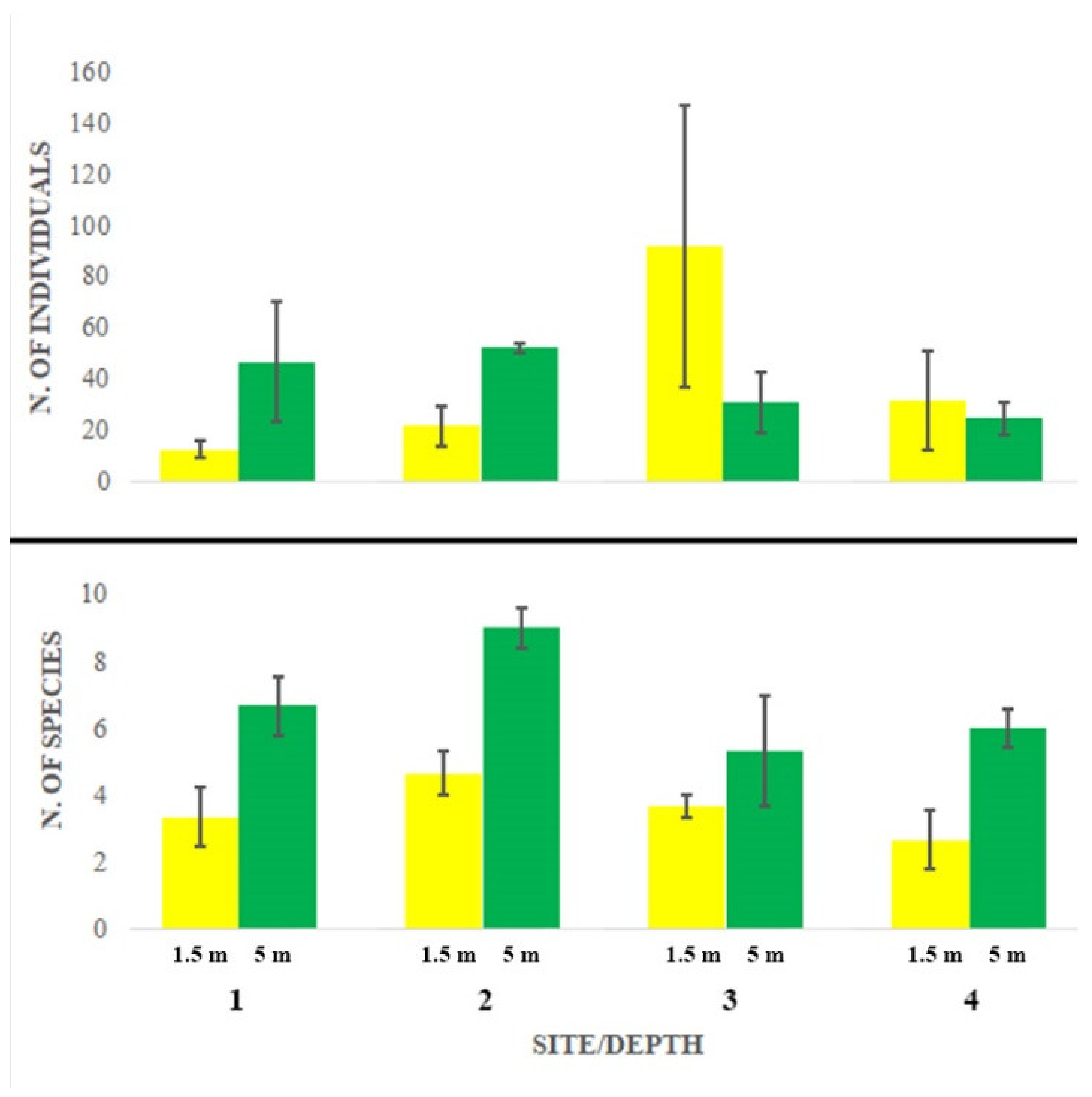
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Station 1 | Station 2 | Station 3 | Station 4 | Total | |||||
|---|---|---|---|---|---|---|---|---|---|
| 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | ||
| Rhodophyta | |||||||||
| Amphiroa sp. | 38.245 | - | 22.831 | - | 38.41 | - | 9.209 | - | 108.695 |
| Peysonnelia sp. | - | 2.388 | - | 4.707 | 0.535 | 3.752 | - | 0.484 | 11.866 |
| Pseudolithophyllum sp. | 0.241 | - | 1.989 | 1.475 | 0.638 | - | - | 0.337 | 4.68 |
| Laurencia obtusa (Hudson) J.V.Lamouroux | - | - | - | - | - | - | 2.485 | - | 2.485 |
| Lithophyllum sp. | - | - | 2.409 | - | - | - | - | - | 2.409 |
| Chlorophyta | |||||||||
| Halimeda tuna (J.Ellis & Solander) J.V.Lamouroux, 1816 | 2.259 | 5.307 | 10.775 | 4.469 | 1.261 | 2.71 | 3.901 | 6.24 | 36.922 |
| Udotea petiolata (Turra) Børgesen 1925 | - | 0.471 | - | 0.994 | 0.055 | 1.47 | - | 1.209 | 4.199 |
| Ochrophyta | |||||||||
| Dictyota dichotoma (Hudson) J.V.Lamouroux | 0.069 | 0.469 | 0.656 | 0.382 | 0.081 | 0.986 | 0.029 | - | 2.672 |
| Porifera | |||||||||
| Sarcotragus sp. | - | 1.365 | 0.115 | 1.932 | 0.03 | 10.238 | - | 2.492 | 16.172 |
| Chondrilla nucula Schmidt, 1862 | - | - | 0.318 | 0.341 | - | - | 1.978 | 2.637 | |
| Petrosia sp. | - | - | - | 1.078 | - | - | - | - | 1.078 |
| Station 1 | Station 2 | Station 3 | Station 4 | Total | |||||
|---|---|---|---|---|---|---|---|---|---|
| 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | ||
| Annelida | |||||||||
| Syllidae | 95 | 254 | 113 | 222 | 265 | 144 | 89 | 90 | 1272 |
| Cirratulidae | 31 | 4 | 17 | 4 | 191 | 10 | 22 | 10 | 289 |
| Nereididae | 84 | 58 | 13 | 15 | 57 | 13 | 10 | 11 | 261 |
| Eunicidae | 20 | 35 | 20 | 42 | 53 | 35 | 6 | 19 | 230 |
| Lumbrineridae | 25 | 9 | 20 | 26 | 43 | 10 | 8 | 9 | 150 |
| Ophelidae | 8 | 14 | 24 | 24 | 20 | 29 | 6 | 19 | 144 |
| Sipunculidae | 4 | 7 | - | 35 | 61 | 15 | - | 9 | 131 |
| Terebellidae | 9 | 16 | 17 | 29 | 17 | 14 | 13 | 16 | 131 |
| Sabellariidae | 8 | 18 | 7 | 19 | 12 | 4 | 2 | 10 | 80 |
| Capitellidae | 3 | 5 | 2 | 8 | 8 | 15 | 8 | 7 | 56 |
| Spionidae | - | 10 | 2 | 17 | 6 | 8 | 5 | 1 | 49 |
| Hesionidae | - | 2 | - | 7 | 2 | 20 | 2 | 9 | 42 |
| Mollusca | |||||||||
| Gastropoda (Prosobranchia) | 23 | 82 | 75 | 80 | 62 | 49 | 63 | 67 | 501 |
| Bivalvia | 237 | 62 | 49 | 40 | 42 | 42 | 15 | 11 | 498 |
| Polyplacophora | 41 | 15 | 71 | 5 | 70 | - | 27 | 1 | 230 |
| Gastropoda (Vermicularidae) | 36 | - | 40 | - | 13 | - | - | - | 89 |
| Arthropoda | |||||||||
| Amphipoda | 369 | 127 | 364 | 180 | 592 | 192 | 281 | 108 | 2213 |
| Tanaidacea | 30 | 40 | 174 | 29 | 178 | 54 | 121 | 19 | 645 |
| Isopoda | 72 | 17 | 79 | 5 | 77 | 26 | 23 | 3 | 302 |
| Decapoda | 29 | 9 | 10 | 13 | 20 | 19 | 3 | 11 | 114 |
| Insecta (Diptera, Chironomidae) | 6 | - | 25 | - | 62 | - | - | - | 93 |
| Cirripedia | 22 | 33 | 15 | - | 1 | 3 | - | - | 74 |
| Nematoda | 36 | 48 | 77 | 91 | 377 | 47 | 63 | 48 | 787 |
| Nemertea | - | - | 29 | - | 15 | - | 4 | - | 48 |
References
- Capa, M.; Kupriyanova, E.; Nogueira, J.M.D.M.; Bick, A.; Tovar-Hernández, M.A. Fanworms: Yesterday, Today and Tomorrow. Diversity 2021, 13, 130. [Google Scholar] [CrossRef]
- Musco, L. Ecology and diversity of Mediterranean hard bottom Syllidae (Annelida): A community-level approach. Mar. Ecol. Prog. Ser. 2012, 461, 107–119. [Google Scholar] [CrossRef]
- Giangrande, A.; Pierri, C.; Arduini, D.; Borghese, J.; Licciano, M.; Trani, R.; Corriero, G.; Basile, G.; Cecere, E.; Petrocelli, A. An innovative IMTA System: Polychaetes, sponges and macroalgae co-cultured in a southern Italian in-shore mariculture plant (Ionian Sea). J. Mar. Sci. Eng. 2020, 8, 733. [Google Scholar] [CrossRef]
- Ingrosso, G.; Abbiati, M.; Badalamenti, F.; Bavestrello, G.; Belmonte, G.; Cannas, R.; Benedetti-Cecchi, L.; Bertolino, M.; Bevilacqua, S.; Bianchi, C.N.; et al. Mediterranean Bioconstructions Along the Italian Coast. Adv. Mar. Biol. 2018, 79, 61–136. [Google Scholar]
- Tovar-Hernández, M.A.; Yáñez-Rivera, B.; Giangrande, A.; Gambi, M.C. Notes on the species of Perkinsiana (Polychaeta: Sabellidae) from Antarctica with the description of P. brigittae sp. nov. Zootaxa 2012, 3485, 56–68. [Google Scholar] [CrossRef]
- Capa, M.; Nishi, E.; Tanaka, K.; Fujikura, K. First record of a Bispira species (Sabellidae: Polychaeta) from a hydrothermal vent. Mar. Biodivers. Rec. 2013, 6, e68. [Google Scholar] [CrossRef]
- Gambi, M.C.; Musco, L.; Giangrande, A.; Badalamenti, F.; Micheli, F.; Kroeker, K.J. Distribution and functional traits of polychaetes in a CO2 vent system: Winners and losers among closely related species. Mar. Ecol. Prog. Ser. 2016, 550, 121–134. [Google Scholar] [CrossRef]
- Stabili, L.; Licciano, M.; Giangrande, A.; Fanelli, G.; Cavallo, R.A. Sabella spallanzanii filter-feeding on bacterial community: Ecological implications and applications. Mar. Environ. Res. 2006, 61, 74–92. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Schirosi, R.; Licciano, M.; Giangrande, A. The mucus of Sabella spallanzanii (Annelida, Polychaeta): Its involvement in chemical defence and fertilization success. J. Exp. Mar. Biol. Ecol. 2009, 374, 144–149. [Google Scholar] [CrossRef]
- Licciano, M.; Stabili, L.; Giangrande, A. Clearance rates of Sabella spallanzanii and Branchiomma luctuosum (Annelida: Polychaeta) on a pure culture of Vibrio alginolyticus. Water Res. 2005, 39, 4375–4384. [Google Scholar] [CrossRef]
- Licciano, M.; Stabili, L.; Giangrande, A.; Cavallo, R.A. Bacterial accumulation by Branchiomma luctuosum (Annelida: Polychaeta): A tool for biomonitoring marine systems and restoring polluted waters. Mar. Environ. Res. 2007, 63, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Tovar-Hernández, M.A.; Méndez, N.; Villalobos-Guerrero, T.F. Fouling polychaete worms from the Southern Gulf of California: Sabellidae and Serpulidae. Syst. Biodivers. 2009, 7, 319–336. [Google Scholar] [CrossRef]
- Clapin, G.; Evans, D.R. The Status of the Introduced Marine Fan Worm, Sabella spallanzanii, in Western Australia: A Preliminary Investigation; Technical report N. 2; CSIRO Division of Fisheries, Centre for Research into Marine Species: Clayton, Australia, 1995. [Google Scholar]
- Patti, F.; Gambi, M. Phylogeography of the invasive polychaete Sabella spallanzanii (Sabellidae) based on the nucleotide sequence of internal transcribed spacer 2 (ITS2) of nuclear rDNA. Mar. Ecol. Prog. Ser. 2001, 215, 169–177. [Google Scholar] [CrossRef]
- Read, G.; Inglis, G.; Stratford, P.; Ahyong, S. Arrival of the alien fanworm Sabella spallanzanii (Gmelin, 1791) (Polychaeta: Sabellidae) in two New Zealand harbours. Aquat. Invasions 2011, 6, 273–279. [Google Scholar] [CrossRef]
- Cochrane, S.J. Snowflakes and feather-dusters—Some challenges for soft-bottom fanworm systematics. Hydrobiologia 2003, 496, 49–62. [Google Scholar] [CrossRef]
- Fraschetti, S.; Giangrande, A.; Terlizzi, A.; Della Tommasa, L.; Miglietta, M.P.; Boero, F. Spatio-temporal variation of hydroids and polychaetes associated to Cystoseira amentacea (Fucales, Phaeophyceae): A regional scale approach. Mar. Biol. 2002, 140, 949–957. [Google Scholar]
- Giangrande, A.; Delos, A.L.; Fraschetti, S.; Musco, L.; Licciano, M.; Terlizzi, A. Polychaete assemblages along a rocky shore on the South Adriatic coast (Mediterranean Sea): Patterns of spatial distribution. Mar. Biol. 2003, 143, 1109–1116. [Google Scholar] [CrossRef]
- Giangrande, A.; Delos, A.L.; Musco, L.; Licciano, M.; Pierri, C. Polychaete assemblages of rocky shore along the South Adriatic coast (Mediterranean Sea). Cah. Biol. Mar. 2004, 45, 85–95. [Google Scholar]
- Musco, L.; Terlizzi, A.; Licciano, M.; Giangrande, A. Taxonomic structure and the effectiveness of surrogates in environmental monitoring: A lesson from polychaetes. Mar. Ecol. Prog. Ser. 2009, 383, 199–210. [Google Scholar] [CrossRef]
- Dorgham, M.M.; Hamdy, R.; El-Rashidy, H.H.; Atta, M.M.; Musco, L. Distribution patterns of shallow water polychaetes (Annelida) along the coast of Alexandria, Egypt (eastern Mediterranean). Mediterr. Mar. Sci. 2014, 15, 635–649. [Google Scholar] [CrossRef][Green Version]
- Fresi, E.; Colognola, R.; Gambi, M.C.; Giangrande, A.; Scardi, M. Ricerche sui popolamenti bentonici di substrato duro del Porto di Ischia. Infralitorale fotofilo: Policheti. Cah. Biol. Mar. 1983, 24, 1–19. [Google Scholar]
- Fresi, E.; Colognola, R.; Gambi, M.C.; Giangrande, A.; Scardi, M. Ricerche sui popolamenti bentonici di substrato duro del Porto di Ischia. Infralitorale fotofilo: Policheti (II). Cah. Biol. Mar. 1984, 25, 33–47. [Google Scholar]
- Giangrande, A. Polychaete zonation and its relation to algal distribution down a vertical cliff in the western Mediterranean (Italy): A structural analysis. J. Exp. Mar. Biol. Ecol. 1988, 120, 263–276. [Google Scholar] [CrossRef]
- Mikac, B.; Licciano, M.; Jaklin, A.; Iveša, L.; Giangrande, A.; Musco, L. Diversity and distribution patterns of hard bottom polychaete assemblages in the North Adriatic Sea (Mediterranean). Diversity 2020, 12, 408. [Google Scholar] [CrossRef]
- Witman, J.D.; Dayton, P.K. Rocky Subtidal Communities. In Marine Community Ecology; Bertness, M.D., Gaines, S.D., Hay, M.E., Eds.; Sinauer Associates: Sunderland, MA, USA, 2001; pp. 339–366. [Google Scholar]
- Giangrande, A.; Licciano, M.; Musco, L. Polychaetes as environmental indicators revisited. Mar. Pollut. Bull. 2005, 50, 1153–1162. [Google Scholar] [CrossRef]
- Guidetti, P. Marine reserves reestablish lost predatory interactions and cause community changes in rocky reefs. Ecol. Appl. 2006, 16, 963–976. [Google Scholar] [CrossRef]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E: Plymouth, UK, 2008. [Google Scholar]
- Colletti, A.; Savinelli, B.; Di Muzio, G.; Rizzo, L.; Tamburello, L.; Fraschetti, S.; Musco, L.; Danovaro, R. The date mussel Lithophaga lithophaga: Biology, ecology and the multiple impacts of its illegal fishery. Sci. Total Environ. 2020, 744, 140866. [Google Scholar] [CrossRef]
- Lepore, E.; Sciscioli, M.; Mastrodonato, M.; Gherardi, M.; Giangrande, A.; Musco, L. Sperm ultra-structure and spermiogenesis in Syllis krohni (Polychaeta: Syllidae), with some observations on its reproductive biology. Sci. Mar. 2006, 70, 585–592. [Google Scholar]
- Çinar, M.E.; Ergen, Z. Faunistic analysis of Syllidae (Polychaeta: Annelida) from the Aegean Sea. Cah. Biol. Mar. 2002, 43, 171–176. [Google Scholar]
- Serrano, A.; San Martín, G.; López, E. Ecology of Syllidae (Annelida: Polychaeta) from shallow rocky environments in the Cantabrian Sea (South Bay of Biscay). Sci. Mar. 2005, 70, 225–235. [Google Scholar] [CrossRef]
- Çinar, M.E.; Gönlügür-Demirci, G. Polychaete assemblages on shallow-water benthic habitats along the Sinop Peninsula (Black Sea, Turkey). Cah. Biol. Mar. 2005, 4, 253–263. [Google Scholar]
- Giangrande, A.; Putignano, M.; Licciano, M.; Gambi, M.C. The Pandora’s box: Morphological diversity within the genus Amphiglena Claparède, 1864 (Sabellidae, Annelida) in the Mediterranean Sea, with description of nine new species. Zootaxa 2021, 4949, 201–239. [Google Scholar] [CrossRef] [PubMed]
| Taxon | Station 1 | Station 2 | Station 3 | Station 4 | Total | ||||
|---|---|---|---|---|---|---|---|---|---|
| 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | 1.5 m | 5 m | ||
| Sabellidae | |||||||||
| Amphicorina armandi (Claparède, 1864) | - | 2 | - | 5 | - | 3 | 5 | 5 | 20 |
| Amphicorina armandi (Claparède, 1864) | - | - | - | - | - | - | 20 | 1 | 21 |
| Amphiglena spp. | 28 | 54 | 20 | 82 | 224 | 61 | 59 | 50 | 578 |
| Branchiomma bombyx (Dalyell, 1853) | - | - | - | 2 | - | - | - | 1 | 3 |
| Chone collaris Langerhans, 1881 | - | - | - | - | - | 10 | - | 3 | 13 |
| Parasabella saxicola (Grube, 1861) | - | - | - | - | - | 1 | - | - | 1 |
| Parasabella langerhansi (Knight-Jones, 1983) | - | 1 | - | 3 | - | 4 | - | 1 | 9 |
| Parasabella tenuicollaris (Grube, 1861) | 1 | 2 | - | 2 | - | - | - | - | 5 |
| Laonome sp. | - | - | - | - | - | 1 | - | - | 1 |
| Perkinsiana socialis (Langerhans, 1884) | - | - | - | - | - | - | 9 | 2 | 11 |
| Pseudopotamilla saxicava (Quatrefages, 1866) | 1 | - | 1 | 11 | 7 | 7 | - | - | 27 |
| Fabriciidae | |||||||||
| Fabricia sabella (Ehrenberg, 1836) | - | - | - | - | - | 1 | - | - | 1 |
| Novafabricia posidoniae Licciano & Giangrande, 2006 | - | 1 | - | - | 7 | - | 1 | - | 9 |
| Serpulidae | |||||||||
| Hydroides elegans (Haswell, 1883) | - | - | - | - | - | 1 | - | - | 1 |
| Hydroides nigra Zibrowius, 1971 | 2 | 2 | 2 | 3 | - | - | - | - | 9 |
| Hydroides pseudouncinata Zibrowius, 1968 | 3 | 12 | 6 | 9 | 1 | - | - | 2 | 33 |
| Hydroides ind. | - | 38 | 33 | 30 | 35 | 2 | - | 5 | 143 |
| Janua heterostropha (Montagu, 1803) | - | 18 | - | - | - | - | - | - | 18 |
| Serpula concharum Langerhans, 1880 | 1 | 4 | - | - | - | 1 | - | - | 6 |
| Spirobranchus polytrema (Philippi, 1844) | 1 | 3 | - | - | - | - | - | 2 | 6 |
| Vermiliopsis striaticeps (Grube, 1862) | - | 3 | 2 | 4 | 2 | - | - | 1 | 12 |
| Spirorbidae ind. | - | - | - | 5 | - | - | - | - | 5 |
| df | MS | Pseudo-F | P(perm) | U.P. | P(MC) | |
|---|---|---|---|---|---|---|
| (a) | ||||||
| st | 3 | 1438.1 | 0.90797 | 0.4929 | 9958 | 0.4519 |
| de | 1 | 4.1667 | 1.4059 × 10−3 | 0.9151 | 425 | 0.9734 |
| st × de | 3 | 2963.6 | 1.8711 | 0.1407 | 9953 | 0.1714 |
| Res | 16 | 1583.9 | ||||
| (b) | ||||||
| st | 3 | 7.8889 | 3.3216 | 0.0431 | 9917 | 0.0478 |
| de | 1 | 60.167 | 32.818 | 0.0313 | 113 | 0.0091 |
| st × de | 3 | 1.8333 | 0.77193 | 0.5344 | 9877 | 0.522 |
| Res | 16 | 2.375 | ||||
| df | MS | Pseudo-F | P(perm) | U.P. | P(MC) | |
|---|---|---|---|---|---|---|
| (a) | ||||||
| st | 3 | 2508.6 | 1.6939 | 0.055 | 9910 | 0.074 |
| de | 1 | 2624.6 | 1.1371 | 0.3891 | 424 | 0.3772 |
| st × de | 3 | 2308.1 | 1.5585 | 0.1099 | 9925 | 0.124 |
| Res | 16 | 1480.9 | ||||
| (b) | ||||||
| st | 3 | 3756.4 | 3.3305 | 0.0001 | 9935 | 0.0011 |
| de | 1 | 4554.7 | 2.4863 | 0.1547 | 425 | 0.1065 |
| st × de | 3 | 1831.9 | 1.6242 | 0.09 | 9947 | 0.1136 |
| Res | 16 | 1127.9 | ||||
| Stations 1 and 2. Average Dissimilarity = 46.45 | ||||||
| Stat 1 | Stat 2 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Vermiliopsis striaticeps | 0.17 | 0.83 | 6.51 | 1.41 | 14.02 | 14.02 |
| Spirobranchus polytrema | 0.50 | 1.00 | 5.30 | 0.91 | 11.41 | 25.43 |
| Pseudopotamilla saxicava | 0.17 | 0.67 | 5.03 | 1.11 | 10.82 | 36.26 |
| Hydroides nigra | 0.50 | 0.67 | 4.78 | 0.91 | 10.29 | 46.55 |
| Hydroides ind. | 0.50 | 0.00 | 4.18 | 0.94 | 8.99 | 55.53 |
| Parasabella langerhansi | 0.17 | 0.50 | 3.84 | 0.97 | 8.26 | 63.80 |
| Parasabella tenuicollaris | 0.33 | 0.33 | 3.75 | 0.84 | 8.07 | 71.87 |
| Stations 1 and 3. Average Dissimilarity = 61.47 | ||||||
| Stat 1 | Stat 3 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Hydroides pseudouncinata | 0.83 | 0.17 | 8.49 | 1.28 | 13.82 | 13.82 |
| Spirobranchus polytrema | 0.50 | 0.67 | 6.12 | 0.91 | 9.95 | 23.77 |
| Hydroides ind. | 0.50 | 0.00 | 5.14 | 0.95 | 8.36 | 32.13 |
| Hydroides nigra | 0.50 | 0.00 | 4.68 | 0.94 | 7.61 | 39.74 |
| Serpula concharum | 0.33 | 0.17 | 4.47 | 0.69 | 7.27 | 47.01 |
| Pseudopotamilla saxicava | 0.17 | 0.33 | 4.41 | 0.72 | 7.18 | 54.18 |
| Novafabricia posidoniae | 0.17 | 0.33 | 4.40 | 0.74 | 7.16 | 61.34 |
| Parasabella tenuicollaris | 0.33 | 0.00 | 3.65 | 0.68 | 5.94 | 67.28 |
| Amphicorina armandi | 0.17 | 0.33 | 3.56 | 0.77 | 5.79 | 73.07 |
| Stations 2 and 3. Average Dissimilarity = 55.57 | ||||||
| Stat 2 | Stat 3 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Hydroides pseudouncinata | 1.00 | 0.17 | 7.93 | 1.75 | 14.28 | 14.28 |
| Vermiliopsis striaticeps | 0.83 | 0.17 | 6.48 | 1.38 | 11.67 | 25.94 |
| Hydroides nigra | 0.67 | 0.00 | 6.23 | 1.25 | 11.21 | 37.15 |
| Pseudopotamilla saxicava | 0.67 | 0.33 | 4.97 | 1.05 | 8.95 | 46.10 |
| Parasabella langerhansi | 0.50 | 0.33 | 4.18 | 0.98 | 7.53 | 53.63 |
| Amphicorina armandi | 0.33 | 0.33 | 3.56 | 0.87 | 6.41 | 60.04 |
| Spirobranchus polytrema | 1.00 | 0.67 | 3.28 | 0.64 | 5.90 | 65.93 |
| Novafabricia posidoniae | 0.00 | 0.33 | 3.23 | 0.67 | 5.82 | 71.75 |
| Stations 1 and 4. Average Dissimilarity = 62.07 | ||||||
| Stat 1 | Stat 4 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Hydroides pseudouncinata | 0.83 | 0.33 | 8.02 | 0.98 | 12.92 | 12.92 |
| Perkinsiana socialis | 0.00 | 0.67 | 7.21 | 1.23 | 11.62 | 24.54 |
| Hydroides ind. | 0.50 | 0.50 | 5.54 | 0.95 | 8.92 | 33.46 |
| Spirobranchus politrema | 0.50 | 0.17 | 5.39 | 0.93 | 8.69 | 42.14 |
| Amphicorina armandi | 0.17 | 0.50 | 5.39 | 0.91 | 8.68 | 50.83 |
| Hydroides nigra | 0.50 | 0.00 | 4.79 | 0.93 | 7.72 | 58.55 |
| Parasabella tenuicollaris | 0.33 | 0.00 | 3.76 | 0.67 | 6.06 | 64.61 |
| Amphicorina eimeri | 0.00 | 0.33 | 3.39 | 0.66 | 5.47 | 70.07 |
| Stations 2 and 4. Average Dissimilarity = 60.34 | ||||||
| Stat 2 | Stat 4 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Hydroides pseudouncinata | 1.00 | 0.33 | 7.00 | 1.19 | 11.61 | 11.61 |
| Vermiliopsis striaticeps | 0.83 | 0.17 | 6.89 | 1.36 | 11.42 | 23.02 |
| Hydroides nigra | 0.67 | 0.00 | 6.40 | 1.21 | 10.60 | 33.62 |
| Perkinsiana socialis | 0.00 | 0.67 | 6.00 | 1.27 | 9.95 | 43.57 |
| Hydroides ind. | 1.00 | 0.50 | 5.76 | 0.90 | 9.55 | 53.12 |
| Pseudopotamilla saxicava | 0.67 | 0.00 | 5.54 | 1.30 | 9.18 | 62.30 |
| Amphicorina armandi | 0.33 | 0.50 | 4.57 | 0.93 | 7.57 | 69.87 |
| Parasabella langerhansi | 0.50 | 0.17 | 4.13 | 0.95 | 6.84 | 76.72 |
| Stations 3 and 4. Average Dissimilarity = 60.09 | ||||||
| Stat 3 | Stat 4 | |||||
| Species | Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum.% |
| Perkinsiana socialis | 0.00 | 0.67 | 7.55 | 1.25 | 12.57 | 12.57 |
| Spirobranchus polytrema | 0.67 | 0.50 | 6.73 | 0.89 | 11.20 | 23.77 |
| Amphicorina armandi | 0.33 | 0.50 | 5.78 | 0.92 | 9.61 | 33.38 |
| Novafabricia posidoniae | 0.33 | 0.17 | 4.95 | 0.73 | 8.23 | 41.61 |
| Hydroides pseudouncinata | 0.17 | 0.33 | 4.43 | 0.74 | 7.37 | 48.98 |
| Chone collaris | 0.33 | 0.17 | 3.94 | 0.76 | 6.56 | 55.54 |
| Parasabella langerhansi | 0.33 | 0.17 | 3.94 | 0.76 | 6.56 | 62.10 |
| Pseudopotamilla saxicava | 0.33 | 0.00 | 3.66 | 0.65 | 6.09 | 68.19 |
| Amphicorina eimeri | 0.00 | 0.33 | 3.55 | 0.66 | 5.90 | 74.09 |
| Variable | SS(Trace) | Pseudo-F | p | Prop. | Cumul. |
|---|---|---|---|---|---|
| Syllidae | 7039.8 | 45.916 | 0.0014 | 0.17267 | 0.17267 |
| sediment | 3718.1 | 26.016 | 0.0269 | 0.091198 | 0.26387 |
| Sabellariidae | 3925.7 | 30.098 | 0.0097 | 0.09629 | 0.36016 |
| Cirripedia | 2657.7 | 21.554 | 0.041 | 0.065189 | 0.42535 |
| Tanaidacea | 2769.0 | 24.125 | 0.0137 | 0.067917 | 0.49327 |
| Halimeda tuna | 2289.1 | 21.183 | 0.0404 | 0.056146 | 0.54941 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musco, L.; Licciano, M.; Giangrande, A. Diversity and Distribution of Sabellida (Annelida) under Protection Regimes. Water 2021, 13, 1491. https://doi.org/10.3390/w13111491
Musco L, Licciano M, Giangrande A. Diversity and Distribution of Sabellida (Annelida) under Protection Regimes. Water. 2021; 13(11):1491. https://doi.org/10.3390/w13111491
Chicago/Turabian StyleMusco, Luigi, Margherita Licciano, and Adriana Giangrande. 2021. "Diversity and Distribution of Sabellida (Annelida) under Protection Regimes" Water 13, no. 11: 1491. https://doi.org/10.3390/w13111491
APA StyleMusco, L., Licciano, M., & Giangrande, A. (2021). Diversity and Distribution of Sabellida (Annelida) under Protection Regimes. Water, 13(11), 1491. https://doi.org/10.3390/w13111491







