Abstract
To define breeding strategies, the understanding of trait inheritance is critical. The objective of this study was to determine the inheritance of popcorn under different water regimes. To this end, Hayman’s diallel methodology was used, with 8 parents and 28 hybrids. The experiment was carried out under well-watered conditions (WW) and water stress (WS). For popping expansion (PE) under both water regimes, the effects of complete dominance and greater importance of the components associated with the dominance effects were observed. In contrast, the number of dominant genes was zero and the determination coefficient in the narrow sense was >50%; additive effects were also present. For the number of grains per row (GR), ear length (EL), and grain yield (GY) under WS and WW conditions, the dominance effects were the most relevant, and the mean degree of dominance with overdominance effects and greatest relevance of the components associated with this effect were also observed. The same breeding methods can be applied under the studied WS and WW conditions. Exploiting heterosis for GY and related components is a promising way to adapt popcorn to WS. To be able to capitalize on additive and dominance effects, a reciprocal recurrent selection is recommended.
1. Introduction
Of all environmental stresses, drought causes the greatest losses in global agricultural production [1,2,3,4]. In response to the severe effects of this abiotic stress on plant growth and development, breeding is an effective tool to reduce the vulnerability of crops [5]. Breeders have developed studies to understand the morphological, physiological, and agronomic responses of water-stressed plants, as well as to select higher-yield genotypes of the major crops under water stress [1,2,6,7,8] with a view to mitigating the effects of climate change, mainly perceptible in the poor temporal and spatial distribution of rainfall.
Abiotic stresses are deleterious environmental influences on the plant life cycle and affect crop growth and structure development, preventing the expression of the full genetic potential [9]. In this sense, water stress causes physiological and biochemical disturbances in plants, which can result in reduced leaf expansion and metabolic cell activity, stomatal closure, photosynthesis inhibition, and changes in carbon partitioning, among others, thus reducing yields [9,10].
Understanding the genetic basis of agronomically and economically important traits is one of the initial phases of breeding programs for higher yields in water-stressed environments. In this sense, diallel crosses have been widely used for parent selection and the study of the genetic mode of action (additive or non-additive effects) under full irrigation and water stress conditions [2,11]. However, other basic information about the mechanism of gene action and trait inheritance is scarce, e.g., the number of genes involved and the mean degree of dominance and heritability. For specialty maize types, such as popcorn, studies addressing drought adaptation [8] or analyses of the gene action in the control of the relevant crop traits are even rarer.
A robust option for the study of inheritance is the methodology of Hayman [12]. This method is not based on previously established statistical models, but on knowledge about the environmental and genetic nature of statistics, such as means, variances, and covariances, listed in a diallel table. This is an interesting approach, in particular because it exempts the need to establish the F2 generation, instead requiring only the parents and F1. The application of the methodology requires some assumptions, as pointed out by Hayman [12], namely: (a) Diploid segregation, (b) parental homozygosis, (c) no maternal effects, (d) no multiple allelism, (e) independent distribution of parents, and (f) no epistasis.
Studies on the genetic effects on trait inheritance of popcorn under full irrigation [13,14,15,16,17] and under biotic stress [18,19], and among these, those using Hayman’s diallel analysis [15,20], showed that the gene action of popping expansion is additive while that of grain yield and related components is mostly non-additive. For popcorn in water-stressed environments, this information is not available. Thus, the following questions arise: What is the mode of gene action of traits of drought-stressed popcorn? Is the mode of gene action of the traits the same under full irrigation and drought? Can the same breeding methods be used for both cultivation conditions?
The objective of this study was to determine the inheritance patterns of economically important popcorn traits under different water availability conditions. An additional objective was to estimate the number of genes involved in drought stress tolerance as well as the mean degree of dominance, trait heritability, and other genetic parameters and components by Hayman’s methodology to establish a guideline for choosing the best-suited strategies for breeding programs for improved genotypes.
2. Materials and Methods
Twenty-eight simple popcorn hybrids and the eight parents were evaluated under contrasting water regimes, in a full diallel mating scheme without reciprocals. The parents were selected from a group of 20 lines of the UENF (Universidade Estadual do Norte Fluminense Darcy Ribeiro) genebank, 3 of which were high-yielding (P3, P6, and P7), 3 were low-yielding (L61, L63, and L65), and 2 were intermediate (L71 and L76) under drought stress [8]. This set of parents were selected in this way to meet the assumptions of allelic symmetry for grain yield under drought stress, as defined by the methodology of Hayman [12].
The use of a limited number of parents (p < 10) and the fixed genetic model is suggested by Hayman [12] to improve the effectiveness of its diallel methodology and to prevent estimates of genetic parameters from being compromised by assumption failures. For this reason, the analysis was performed with 8 parents. It was also decided not to use reciprocal hybrids because normally, there are no reciprocal effects for grain yield traits under well-irrigated [17,21] and water-stress conditions [11,22].
The experiments were initiated in a low rainfall period, i.e., from May to August (autumn/winter), at the "Antonio Sarlo" State College of Agriculture, Campos dos Goytacazes—RJ, in 2018. The phenotypic traits of plants grown at two levels of water availability were evaluated: The first level consisted of adequate irrigation—well-watered (WW) conditions, maintaining soil moisture at field capacity (−0.01 MPa) and the second level consisted of irrigation suspension between the phenological stages pre-flowering (10 days before flowering) and grain filling—water stress (WS). We tried to compare whether the genetic effects present under well-irrigated conditions were the same under water stress. Therefore, water stress was applied during the period considered critical for the crop (flowering and grain filling) [23,24].
To control the irrigation level, a drip system (flow rate 2.3 mm h−1) with one katif dripper per plant was used under both water regimes. The soil water potential was monitored by Decagon MPS-6 tensiometers installed between plants, at a depth of 0.20 m. The well-watered environment was irrigated whenever necessary to maintain field capacity. The weekly applied irrigation levels are listed in Table 1. In the WS treatment, irrigation was suspended 10 days before the date of male flowering (Table 1). The soil water potential was monitored throughout the experiment (Figure S1).

Table 1.
Precipitation and irrigation levels (mm) applied to popcorn under well-watered (WW) and water-stressed (WS) conditions in the weeks after sowing.
The WS environment was watered with 217.5 mm and the WW with 335.1 mm (Table 1). Rainfall during the experiment was measured by an automatic weather station of the National Institute of Meteorology (INMET), installed at the experimental site (Table 1). The mean temperature was 22 °C, relative air humidity was 77%, and the photosynthetic photon flow was 1208 μmol m2 s−1 (Figure S2).
In the water-stressed environment, the soil reached the permanent wilting point (−1.5 MPa) twice (Figure S1)—the first time during flowering (68 days after sowing) and the second during grain filling (90 days after sowing).
The experiment was arranged in a randomized complete block design with three replications. Each plot consisted of one 4.80-m row, with plants spaced 0.20 m and rows 0.80 m apart, with a total of 23 plants per row. Three seeds per planting hole were sown in the conventional planting system. Fertilization at sowing consisted of 30 kg N ha−1 (as urea), 60 kg P2O5·ha−1 (triple superphosphate), and 60 kg K2O ha−1 (potassium chloride). In the stage of two well-defined leaves, the stand was thinned to 5 plants m−1. About 30 days after sowing, 100 kg N ha −1 (as urea) was applied.
The following traits were assessed: Mean grain number per row (GR), by counting; ear diameter (ED), measured with a caliper (cm); ear length (EL), measured with a ruler (cm); 100-grain weight (W100), by weighing and averaging (g) two 100-grain samples per plot; grain yield (GY), weighed after threshing the ears of each plot and corrected to 13% moisture (kg.ha−1); popping expansion (PE), by measuring the volume of a 30 g grain sample irradiated in a special microwaveable paper bag at 1000 W for 2 min 10 s in a beaker and expressed as the ratio of popcorn volume by grain weight (in mL.g−1); and expanded popcorn volume per hectare (PV), by multiplying GY and PE (m3 ha−1).
The statistical design used for the experiment was based on a combined analysis between water conditions, i.e., , where is the ith genotype in the jth water condition in the kth block; is the general constant; _i is the fixed effect of the ith genotype; is the effect of the ith block within environment j; is the fixed effect of the jth water condition, with NID(0, σ2); is the fixed effect of the interaction between the ith genotype with the jth water condition with NID (0, σ2); and is the random experimental error associated with the Y_ijk observation, with NID (0, σ2). As it is not possible to perform a combined analysis by the Hayman diallel [12], it was decided to present only the individual analysis for each water condition.
The data were subjected to individual variance analysis for each water regime (WS and WW), based on the statistical model: , where o is the observed value of the ith genotype in the jth block; μ is the general constant; is the fixed effect attributed to the ith genotype; is the effect of block j; and is the random error associated with observation , normally independently distributed (NID) (0, σ2).
Hayman’s diallel analysis [12] is based on previously established statistical models, associated with knowledge of the environmental and genetic nature of statistics, such as means, variances, and covariance. This method imposes the following assumptions: Homozygous parents, diploid segregation, absence of maternal effect, absence of multiple allelism, genes independently distributed among parents, and absence of epistasis. The parameters are obtained from a diallel table involving p homozygous parents. These are supposed to differ by one locus (T/t), while there is still a proportion of u with favorable alleles and another with unfavorable alleles [24]. It is also considered that the genes in question have an independent distribution among the parents, and the genotypic values related to the TT, Tt, and tt genotypes are, respectively, dt, ht, and −dt. It is assumed that, normally, between hybrids and their reciprocal, there are no differences. Thus, the diallel table uses symmetry, in which the hybrid combination is represented by (Yij + Yji)/2. Thus, considering only the environmental (residual) components, we have: E (Y2ii) = ε; E (Y2ij) = ½ ε, where ε and ε ’are the residual variances associated, respectively, with parents and hybrids.
Thereafter, the Hayman diallel analysis [12] was performed. To validate the assumptions of Hayman (1954), the sufficiency of the model was evaluated. It was tested based on linear regression analysis of (covariance between parent means and the mean of crosses between the parents) as a function of (variance between the mean of the cross between parents). Thus, two tests were performed: The first tested whether the regression coefficient did not differ from 1 and the hypothesis was evaluated by the t-test, associated with p − 2 degrees of freedom; the second was based on the t-statistic, a test that weighted (covariance between parent means and ith means and variance between means of the ith line, respectively) by a 45° rotation of the axes represented by and . Based on the new values of and , hypothesis H0 was tested: b’: 0 (b’ is the regression coefficient after rotation) by the t or F test, associated with 1 and p − 2 degrees of freedom.
Then, the genetic and environmental components were estimated: is the environmental variance component, represented by the mean square error (experimental error); is the variance component associated with additive effects; a are the variance components associated with dominance deviations; is the quadratic component determined by the difference between hybrid and parent means; is the component associated with covariance between additive and non-additive effects; and is the component expressing the difference between additive and dominant gene effects. In addition, the following genetic parameters were estimated: is the mean degree of dominance; is the distance between alleles (symmetry); is the dominant/recessive ratio; is the number of dominant genes; is the determination coefficient in the narrow sense; is the determination coefficient in the broad sense; correlations between the mean values of parents () and the sum of covariance between parent means and means of the line () and the variance between means of the line (); expected coordinate values ; and and the predicted value for the parent with the maximum number of dominant () and recessive () alleles. The terms’ components and parents were used as explained by Hayman [12] and Cruz et al. [25]. For the genetic statistical analysis, the genotype effect was considered fixed. The analyses were performed with software GENES [26].
3. Results
3.1. Analysis of Variance, Effects of Drought Stress, and Fitting of the Additive Dominance Model
The evaluated traits differed statistically (p < 0.01) between the two water regimes (Table 2). The coefficients of variation were low (<21%). The percentage losses, in comparison between the environments, were highest for the traits GY and PV (41.25% and 46.15%, respectively) and lowest for W100 and PE (8.88% and 9.08%, respectively) (Table 2).

Table 2.
Summary of the analysis of variance, overall means, coefficient of experimental variation (CV) (%), and proportional loss (%) in a comparison of the two treatments, under water-stress (WS) and well-watered (WW) conditions.
The prerequisites for the use of the model proposed by Hayman [12] were evaluated by sufficiency tests of the additive dominance model. Thus, with the exception of PV under WS, W100 in WW, and ED in both environments, the non-rejection of the hypothesis Ho: β = 1 for regression and of the hypothesis Ho: β′ = β −1 = 0, after axis rotation, shows the validity of the model assumptions (Table 3). This indicates the validity of the additive dominance model in the genetic studies of almost all evaluated traits, whereas the traits that did not meet the assumptions were excluded from further analysis.

Table 3.
Sufficiency tests of the additive dominance model based on linear regression analysis of as a function of of the traits evaluated in popcorn genotypes under water-stress (WS) and well-watered (WW) conditions.
3.2. Additive and Dominance Effects
Based on the estimates of the mean degree of dominance, dominance effects were predominant for PE, under both water regimes and for W100 under WS. For the other traits, under both water regimes, the mean degree of dominance was >1.2, indicating over-dominance effects (Table 4). These results are in line with the estimates of components associated with dominance and additivity effects (Table 5), in which the components associated with dominance effects (, ) were mostly more important than the components associated with additive effects () in both environments, for all evaluated traits. This was also confirmed by the negative values of , for all traits, under both water regimes. In these cases, dominance is the main type of gene action affecting the studied traits.

Table 4.
Estimates of genetic and non-genetic parameters, according to Hayman’s diallel analysis (1954) of the traits evaluated in popcorn genotypes under water-stress (WS) and well-watered (WW) conditions.

Table 5.
Estimates of genetic and non-genetic components, according to Hayman’s diallel analysis (1954), of the traits evaluated in popcorn genotypes under water-stress (WS) and well-watered (WW) conditions.
The allelic symmetry of the traits was <0.25 (Table 4), which indicates that the alleles are symmetrically distributed among the parents. In the relationship between dominant/recessive genes (Table 4), the dominant homozygous forms of trait PE were predominant (−1.74 under WS and 2.47 in WW), contradicting the values of allele distribution that apparently suggested symmetry. The values of the other traits were >1, indicating a tendency of predominantly dominant genes in the parents.
The estimates of the number of genes highlighted that, in general, under WS, the number of genes/gene blocks involved in trait inheritance was lower than in the WW environment (Table 4). In both environments, no dominant genes or genes/gene blocks were observed for PE. Under WS, trait GY had around three genes; for the other traits, around two genes/gene blocks were estimated. In WW, the number of genes involved was around five for GR and close to four for the other traits (Table 4).
The determination coefficient in the narrow sense for PE in the WW environment was moderately higher (0.56) than that under WS (0.50) (Table 4). For the other traits, the values of this coefficient were higher under WS (Table 4). On the other hand, for the determination coefficient in the broad sense, only GR effectively expressed a higher value (0.87 for WS compared to 0.83 for WW), while the others had the same magnitude. The determination coefficient in the narrow sense ranged from 0.32 for GY to 0.57 for W100 under WS and from 0.19 for PV to 0.56 for PE in WW. The determination coefficient in the broad sense ranged from 0.84 for PE to 0.93 for GY under WS and 0.81 for EL to 0.94 for GY in WW (Table 4).
The results of the genetic parameters and components of PE (Table 4 and Table 5) were controversial with regard to the expression of additive and dominance effects. The mean degree of dominance (Table 4) and estimates of genetic components (Table 5) expressed the superiority of dominance, while the number of dominant genes with a high determination coefficient in the narrow sense (Table 4) indicated additivity.
3.3. Genetic Merit of Parents and Theoretical Limits of Selection
By the graphs of linear regression analyses of (covariance between parent means and means of the ith row) as a function of (variance between means within the ith row, respectively) (Figure 1), the proportion of additive and dominant D/R alleles based on the parent distribution along the regression line for each trait can be analyzed. In this way, the parents can be sorted in descending order of dominance. This distribution is based on the principle that the genotypes derived from parents with the highest number of dominant genes will have minimal variance (, ) and minimum parent–progeny covariance (, ). Thus, the closer the genotype location is to the origin of the line in the graph of linear regression, the higher the concentration of dominant alleles. This information about parent allele concentration can also be observed by ranking (Table 6), where the higher the value, the higher the concentration of recessive alleles.
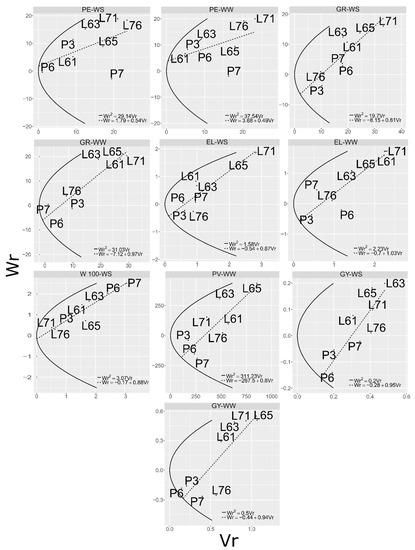
Figure 1.
Straight line and parabola established by the relationships between covariance between parent means and means of the crosses with the parent (), with the means of the crosses with the parent ( ), of the traits evaluated in in popcorn genotypes under water-stress (WS) and well-watered (WW) conditions. PE: popping expansion; GY: grain yield; EL: ear length; GR: mean number of grains per row; W100: 100-grain weight; and PV: expanded popcorn volume per hectare.

Table 6.
Sum of covariance values between parent means and means of crosses with the parent ( ), with variance between means of crosses with the parent ( ) and classification of the sum and parent means and their classification of popcorn genotypes under water-stress (WS) and well-watered (WW) conditions.
For trait PE, parent L71 had the highest concentration of recessive alleles under WW conditions and the second highest under WS (Table 6), and was located near the origin of the line in the linear regression graph (Figure 1). Consequently, this parent had the highest means in both environments (Table 6) and the highest mean and highest dominant allele concentrations for W100 under WS.
For the traits GY and related components (Table 6), the parents with the highest concentration of dominant genes, located at the lower end of the graph (Figure 1) and consequently those with the lowest , were P6 for GY under WS and WW and for GR in WW; P3 for EL in both environments and GR under WS; and P7 for PV, in WW (Table 6). With regard to the overall mean, the best parents were P3 for trait GY under WS and WW; GR and EL under WS; and P7 for PV, EL, and GR in WW (Table 6).
The correlation between and () indicates which type of intralocus allelic interaction is most important for the trait, i.e., dominant when the correlation is negative, or recessive when the correlation is positive (Table 7). For PE, the correlation analyses between and () clearly showed a value close to zero, with a slight tendency to a higher relevance of recessive alleles for the trait in both environments, i.e., with = 0.01 under WS and = 0.06 under WW conditions (Table 7). For the other traits, under both water regimes, the correlation values were highly negative, indicating that the dominant alleles are the most important (Table 7).

Table 7.
Estimates of the expected values of the sum of the coordinates and , and genotypic values ( ) of the parent with the highest concentration of recessive ( ) and dominant ( ) alleles of traits evaluated in popcorn genotypes grown under water-stress (WS) and well-watered (WW) conditions.
Considering the theoretical limits of selection, for trait PE in both environments, lines with higher allele concentrations can be established in the segregating generations of the studied parents, since the values of the parents (Table 6) were not higher than the estimates of Table 7. For the other traits, under both WS and WW, superior segregating generations can also be obtained for all traits, since the values of the parents (Table 6) were not below the estimates of Table 7.
4. Discussion
4.1. Analysis of Variance, Effects of Drought Stress, and Fitting of the Additive Dominance Model
The presence of significant differences between the two studied environments for all evaluated traits indicates that superior parents and hybrids can be identified and selected.
Compared with the fully irrigated environment, water stress reduced GY by 41.25%. Reductions in GY between 25% and 92% were reported for field corn [23]. The very high GY loss was attributed to the combined reduction in GR, EL, and W100. Popping expansion was less strongly reduced in the comparison between environments (9.08%). In the study of Kamphorst et al. [8], in which S7 lines were screened, losses of >29% for the same trait were detected, showing that water stress can severely affect grain expansion. Stress-induced changes in grain physicochemical properties may be causing these losses.
Globally, maize, the main of C4 carbon fixation cereal species produced in the world, is more sensitive compared to wheat, the main of C3 carbon fixation cereal species [3]. Overall, corn has a 40% reduction in yield due to drought while wheat has an average loss of 20% [3]. According to the same authors, even though C4 plants have higher photosynthetic potential, this greater loss is partly attributed to the fact that maize originates from wetter regions, which probably contributes to greater yield loss under water-stress conditions.
In general, in Hayman’s [12] diallel analysis, most of the constraints imposed by the model are fully satisfied for the trait under selection. When a small number of parents (p < 10) are involved, the fixed genotype effect is used; thus, estimates of genetic parameters are barely affected by failures in the assumptions [27]. However, when several traits are simultaneously analyzed, it is rather unlikely that the assumptions of the additive dominance model are met for all variables. In this case, the additive dominance model was not valid for PV under WS, for W100 in WW, and for ED in either environment. According to Cruz et al. [27], in this case, there may be a targeted distribution of the alleles that regulate the trait, causing the non-fitting to the additive dominance model.
4.2. Additive and Dominance Effects
The evaluated genetic parameters and components were similarly expressed, regardless of the water regime, for all analyzed traits. Similar responses between well-irrigated and water-stress conditions have been reported, where the reduction due to water stress is up to 50% for cereals [1,28,29] and grasses [30,31].
The relationships between the gene actions present in each trait for each water condition are extremely important in determining the magnitude of heterosis in hybrids. Hybrid vigor depends mainly on the type of gene action, such as additive, dominant, additive × additive, and dominant × dominant effects, associated with the biological viability and genetic divergence of parents [32,33]. These types of gene actions will be translated into hybrids as additive effects when the value of F1 refers to the parental mean; partial dominance, when the F1 value is between the parental mean and the superior parent mean; total dominance, when the value of F1 is equal to the mean of the superior parent; and over-dominance, when the deviation of F1 is greater than the mean of the superior mean [33].
The mean degree of dominance revealed full dominance effects on PE under both water regimes. This indicates that the hybrid mean is close to the mean of the best parent of a cross. On the other hand, Silva et al. [15] and Schwantes et al. [20], studying the inheritance of this trait under well-watered conditions, diagnosed partial dominance effects. These partial dominance values are in better agreement with most diallel studies with popcorn, almost all of which reported an additive action for PE [13,16,17,19,20]. However, in an evaluation of the same trait by SSR mapping, Babu et al. [34] found QTLs with both dominant and partially dominant gene action. Similarly, in diallel studies on PE, Oliveira et al. [35] and Santos et al. [36] detected dominant gene action.
In contrast to the reports of Oliveira et al. [35] and Santos et al. [36], in this study, the number of dominant genes for PE was close to zero (−0.04 under WS and−0.08 under WW), demonstrating that there are no dominant alleles in the genes that control PE. Moreover, the determination coefficient in the narrow sense was >50%; therefore, the additive effect is predominant over the non-additive effects, regardless of the water regime [15,20]. For such cases, Silva et al. [15] suggested the occurrence of additive inheritance, so that intrapopulation methods would be more effective to improve the trait. This was also recommended by Larish and Brewbaker [13] and Pereira and Amaral Júnior [14], who defined the inheritance of popping expansion as additive.
Based on the results found for PE, both additivity and dominance effects are present in the trait. In a study on the inheritance of popping expansion in crosses between popcorn and flint-type corn, Coan at al. [37] suggested two possible types of inheritance for the trait: A large gene with additive action, together with polygenes with additive and dominance action (mixed inheritance), and another with only polygenes with additive and dominance action. In both types, the two forms of gene action are present (mixed inheritance). Thus, additivity still seems to be the main mode of gene action for PE, although dominance in an equal or lower proportion can be observed. These results are also in line with the findings of Babu et al. [34]. Therefore, the occurrence of mixed inheritance in PE expression is suggested.
On the other hand, it is noteworthy that PE is usually negatively correlated with GY (base trait underlying parent selection) and according to Cabral et al. [38], the correlation may be moderately negative or even null [39]. Thus, the allelic symmetry may not have been fully met, even though the results did not show it. To draw more assertive conclusions with regard to PE inheritance, a diallel mating system with the selection of parents based on this trait would be ideal. None of the inheritance studies that established inferences about PE were based on the selection of discrepant parents for PE [15,20].
Overdominance effects are the most important for GY, EL, and GR in both environments and PV in WW, which is why the exploitation of heterosis is a promising genetic solution to mitigate environmental stress caused by drought. Heterosis has been one of the most important factors of efficient selection, which has resulted in the exponential increase in corn grain yield in the past decades [40]. This gene effect has been described to improve adaptation of field corn to stress conditions [40,41], including drought [1,11].
Non-additive effects are most important for grain yield and the related components (EL and GR). Genes expressing dominance, with low determination coefficients in the narrow sense and higher relevance of components associated with dominance effects, confirm this statement. Thus, the dominance gene action, the main factor determining high heterosis, can maximize genetic gain under WS [11]. Therefore, heterosis is a factor that should be exploited to improve the adaptation of popcorn to drought-induced environmental stress with regard to grain yield and yield components.
Among the grain yield components, W100 was the only trait with no overdominance effect under WS. In a study on the adaptation of field corn to WS, Araus et al. [1] found that heterosis due to overdominance for W100 is only relevant under full irrigation. The authors explained that the parents under WW are affected by the lack of sufficient photoassimilates for grain filling caused by their lower stay green, and that the hybrids are more effective for grain filling in this environment. Thus, it is believed that despite the greater adaptive power of the hybrid to WS, the stress is limiting for its development. In this sense, in this study, it can be argued that grain filling of the hybrids was restricted under WS compared to their parents, since the hybrids had, on average, 30% more GR. In these cases, under WS, Holá et al. [42] suggested that further hybrid development becomes disadvantageous. The authors explained that the main cause is the increased loss of photosynthetic efficiency due to drought. This may be directly due to the loss of photosynthetic capacity or the loss of potential to translocate assimilates to grain [43].
The loss of vigor of the hybrids due to the lack of over-dominance of the W100, an important component for GY, in the WS condition may be detrimental to the final yield. Araus et al. [1] and Cairns et al. [43] reported that a reduction in the grain number and weight are the main factors among the production components that affect GY. Thus, the loss of W100 over-dominance can be pointed as a cause of GY reduction. Thus, the selection of parents with higher combinatorial capacity for W100 can provide a better water status for the hybrid.
A reduction in the number of genes or gene blocks involved in trait expression under WS compared to WW was observed. This reduction may be related to the lower yield of genotypes under WS, where they fall short of their maximum genetic potential [4]. Water stress reduces plant development and productivity, which may be largely determined by the negative relationships between yield potential and the ability to maintain yield levels under water scarcity [44].
Hybrid exploitation is the best option to improve the adaptation of popcorn to water stress, particularly for GY and related components. Nevertheless, the mixed inheritance of PE must be taken into account. An effective strategy is the development of lines from improved genotypes, with a higher frequency of favorable alleles for PE that ensure a higher value per se of the line. In this respect, the reciprocal recurrent selection method appears to be a more appropriate strategy because it capitalizes on additive and dominance effects. This method favors the concentration of additive alleles with gains within each breeding population and allows gains among dominant populations by exploiting hybrids [33]. Thus, it is possible to ensure an improved adaptation of popcorn to water stress, providing higher yields as well as better popcorn quality due to the increased popping expansion.
4.3. Genetic Merit of Parents and Theoretical Limits of Selection
Recessive genes determine the expression of popping expansion. A higher positive correlation between and () was clearly shown in the studies of Silva et al. [15] and Schwantes et al. [20], pointing out that a vast majority of recessive genes determine PE expression. The recessive genes were considered the most relevant and parent L71 united a high concentration of recessive alleles (high ) and a high mean. Therefore, this parent had the highest merit for the trait in both environments. The theoretical limits of selection for recessive genes () exceeded those found for the parent, indicating the possibility of selecting parents with higher PE values from the segregating populations.
The low correlation between and () for PE was due to some peculiarities of the parents in the study environments; the concentrations of recessive alleles of the lines with the best means for the trait were sometimes high and sometimes low. The performance pattern of the parents was similar in the environment under WS WW conditions. The number of genes expressing dominance for trait PE was close to zero in both environments, reinforcing the predominance of an additive gene action for this trait. Nevertheless, the influence of dominance is not discarded, albeit very low, resulting in mixed inheritance for PE.
Dominant genes are those that determine the expression of trait GY and its components, regardless of the water regime. These results were confirmed by Silva et al. [15] under well-watered conditions. The lines P3, P6, and P7 stand out as parents with the highest concentrations of dominant alleles with low estimates and high means. Thus, these were the parents with high genetic merit for the traits in both studied environments. The theoretical selection limits for dominant genes () were lower than those found for the studied parents, indicating that higher-yielding genotypes can be obtained from the segregating populations derived from the diallel crosses.
The high performance of parent L71 for PE and W100 is interesting for plant breeding under both water regimes since this genotype can improve popcorn quality by increasing PE and yield component W100 under WS. In a first instance, crosses between parents with a good performance for grain yield and its components (P3, P6, and P7 with L71), may be promising. In genetic diversity studies, estimated by microsatellite markers (SSR-EST), the largest genetic distances were found between P6 and L71 [8,45], indicating a better chance for allelic complementation. Hybrids resulting from the combination of these parents can have high performance for both popping expansion and grain yield. Therefore, the development of lines from diallel-derived segregating populations may provide a generation of better parents for the studied popcorn traits. This might be a successful strategy for the popcorn breeding program since the selection limits can be reached under both water regimes.
5. Conclusions
Based on the results, for the applied stress levels, the same breeding methods applied to popcorn under well-watered conditions can be applied to develop superior genotypes under water stress.
Overdominance was the predominant gene effect for grain yield and its components, regardless of the water regime. In this case, genetic gains can be achieved by exploiting heterosis.
For popping expansion, the dominance and additivity effects were predominant (mixed inheritance). Thus, the methods of interpopulation recurrent selection may be a promising option to obtain selection gains.
The theoretical limits of selection were not exceeded and genotypes with higher yields could be obtained from the segregated populations of the diallel.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4395/9/12/782/s1, Figure S1: Soil water potential (−MPa) of the experiment with popcorn genotypes under water-stress (WS) and well-watered (WW) conditions. Figure S2: Mean weekly values, in days after sowing, of temperature (°C), relative air humidity (%), and photosynthetic photon flow (mol m2·s−1) throughout the period of experimental cultivation of popcorn genotypes.
Author Contributions
Conceptualization, V.J.d.L., A.T.d.A.J. and S.H.K.; methodology, V.J.d.L., S.H.K., R.B.B., J.T.L., T.d.O.S., K.F.M.S., M.M.C., U.A.d.O. and G.M.B.G.; software, V.J.d.L., R.B.B. and P.H.A.D.S.; validation, V.J.d.L.; formal analysis, V.J.d.L. and S.H.K.; investigation, V.J.d.L., A.T.d.A.J., S.H.K., R.B.B., J.T.L., T.d.O.S., K.F.M.S., M.M.C., U.A.d.O., P.H.A.D.S., G.M.B.G. and L.J.M.G.; resources, A.T.d.A.J.; data curation, A.T.d.A.J.; writing—original draft preparation, V.J.d.L., A.T.d.A.J., S.H.K., J.T.L., P.H.A.D.S., S.K. and L.J.M.G.; writing—review and editing, V.J.d.L., A.T.d.A.J., S.H.K., P.H.A.D.S. and S.K.; supervision, A.T.d.A.J., S.H.K. and L.J.M.G.; project administration, A.T.d.A.J.; funding acquisition, V.J.d.L., A.T.d.A.J. and S.H.K.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Araus, J.L.; Sánchez, C.; Cabrera-Bosquet, L. Is heterosis in maize mediated through better water use? New Phytol. 2010, 187, 392–406. [Google Scholar] [CrossRef]
- Adebayo, M.A.; Menkir, A.; Blay, E.; Gracen, V.; Danquah, E.; Hearne, S. Genetic analysis of drought tolerance in adapted × exotic crosses of maize inbred lines under managed stress conditions. Euphytica 2014, 196, 261–270. [Google Scholar] [CrossRef]
- Daryanto, S.; Wang, L.; Jacinthe, P.-A. Global Synthesis of Drought Effects on Maize and Wheat Production. PLoS ONE 2016, 11, e0156362. [Google Scholar] [CrossRef]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop Production under Drought and Heat Stress: Plant Responses and Management Options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Challinor, A.J.; Koehler, A.-K.; Ramirez-Villegas, J.; Whitfield, S.; Das, B. Current warming will reduce yields unless maize breeding and seed systems adapt immediately. Nat. Clim. Chang. 2016, 6, 954–958. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nicholls, C.I. The adaptation and mitigation potential of traditional agriculture in a changing climate. Clim. Chang. 2017, 140, 33–45. [Google Scholar] [CrossRef]
- Dias, K.O.D.G.; Gezan, S.A.; Guimarães, C.T.; Parentoni, S.N.; Guimarães, P.E.D.O.; Carneiro, N.P.; Portugal, A.F.; Bastos, E.A.; Cardoso, M.J.; Anoni, C.D.O.; et al. Estimating Genotype × Environment Interaction for and Genetic Correlations among Drought Tolerance Traits in Maize via Factor Analytic Multiplicative Mixed Models. Crop Sci. 2018, 58, 72. [Google Scholar] [CrossRef]
- Kamphorst, S.H.; de Lima, V.J.; Júnior, A.T.D.A.; Schmitt, K.F.M.; Leite, J.T.; Carvalho, C.M.; Silva, R.M.R.; Xavier, K.B.; Fereira, F.R.A.; Santos, P.H.A.; et al. Popcorn breeding for water-stress tolerance or for agronomic water-use efficiency? Genet. Mol. Res. 2018, 17, gmr18184. [Google Scholar] [CrossRef]
- Dalal, M.; Sharma, T.R. Biotechnological Applications for Improvement of Drought Tolerance. In Abiotic Stress Management for Resilient Agriculture; Springer: Singapore, 2017; pp. 299–312. [Google Scholar]
- Lopes, M.S.; Araus, J.L.; van Heerden, P.D.R.; Foyer, C.H. Enhancing drought tolerance in C4 crops. J. Exp. Bot. 2011, 62, 3135–3153. [Google Scholar] [CrossRef]
- Adebayo, M.A.; Menkir, A.; Hearne, S.; Kolawole, A.O. Gene action controlling normalized difference vegetation index in crosses of elite maize (Zea mays L.) inbred lines. Cereal Res. Commun. 2017, 45, 675–686. [Google Scholar] [CrossRef]
- Hayman, B.I. The Theory and Analysis of Diallel Crosses. II. Genetics 1958, 43, 63–85. [Google Scholar] [PubMed]
- Burnham Larish, L.L.; Brewbaker, J.L. Diallel analyses of temperate and tropical popcorns. MAYDICA 1999, 44, 279–284. [Google Scholar]
- Pereira, M.G.; Amaral Júnior, A.T. Estimation of Genetic Components in Popcorn Based on the Nested Design. Crop Breed. Appl. Biotechnol. 2001, 1, 3–10. [Google Scholar] [CrossRef]
- Silva, V.; Amaral Júnior, A.; Scapim, C.; Freitas Júnior, S.; Gonçalves, L. Inheritance for economically important traits in popcorn from distinct heterotic groups by Hayman’s diallel. Cereal Res. Commun. 2010, 38, 272–284. [Google Scholar] [CrossRef]
- Jele, P.; Derera, J.; Siwela, M. Assessment of popping ability of new tropical popcorn hybrids. Aust. J. Crop Sci. 2014, 8, 831–839. [Google Scholar]
- Cabral, P.D.S.; Amaral Júnior, A.T.; Pio Viana, A.; Duarte Vieira, H.; de Jesus Freitas, I.L.; Vittorazzi, C.; Vivas, M. Combining ability between tropical and temperate popcorn lines for seed quality and agronomic traits. Aust. J. Crop Sci. 2015, 9, 256. [Google Scholar]
- Schwantes, I.A.; do Amaral Júnior, A.T.; Gerhardt, I.F.S.; Vivas, M.; e Silva, F.H.D.L.; Kamphorst, S.H. Diallel analysis of resistance to Fusarium ear rot in Brazilian popcorn genotypes. Trop. Plant Pathol. 2017, 42, 70–75. [Google Scholar] [CrossRef]
- Mafra, G.S.; Amaral Junior, A.T.D.; Vivas, M.; Santos, J.S.D.; Guimarães, A.G.; Pena, G.F. The combining ability of popcorn S7 lines for Puccinia polysora resistance purposes. Bragantia 2018, 77, 519–526. [Google Scholar] [CrossRef]
- Schwantes, I.A.; Amaral Júnior, A.T.D.; Vivas, M.; Almeida Filho, J.E.D.; Kamphorst, S.H.; Guimarães, A.G.; Khan, S. Inheritance of resistance to Fusarium ear rot in popcorn. Crop Breed. Appl. Biotechnol. 2018, 18, 81–88. [Google Scholar] [CrossRef]
- Schmitt, K.F.M.; Lima, V.J.; do Amaral Júnior, A.T.; Santos, J.S.; Mafra, G.S.; Vivas, M.; Kamphorst, S.H.; Souza, Y.P.; Oliveira, F.T.; Ferreira, F.R.A.; et al. Combining ability of popcorn lines for resistance to the fungus Puccinia polysora (Pucciniaceae). Genet. Mol. Res. 2019, 18, gmr18330. [Google Scholar] [CrossRef]
- Wattoo, F.M.; Saleem, M.; Sajjad, M. Identification of Potential F1 Hybrids in Maize Responsive to Water Deficient Condition. Am. J. Plant Sci. 2014, 5, 1945–1955. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant Drought Stress: Effects, Mechanisms and Management. In Sustainable Agriculture; Springer: Dordrecht, The Netherlands, 2009; pp. 153–188. [Google Scholar]
- Reynolds, M.P.; Quilligan, E.; Aggarwal, P.K.; Bansal, K.C.; Cavalieri, A.J.; Chapman, S.C.; Chapotin, S.M.; Datta, S.K.; Duveiller, E.; Gill, K.S.; et al. An integrated approach to maintaining cereal productivity under climate change. Glob. Food Sec. 2016, 8, 9–18. [Google Scholar] [CrossRef]
- Cruz, C.D.; Carneiro, P.C.S.; Regazzi, A.J. Modelos Biométricos Aplicados ao Melhoramento Genético; UFV: Viçosa, Brazil, 2014; Volume 2. [Google Scholar]
- Cruz, C.D. GENES-A software package for analysis in experimental statistics and quantitative genetics. Acta Sci. Agron. 2013, 35, 271–276. [Google Scholar] [CrossRef]
- Cruz, C.D.; Regazzi, A.J.; Carneiro, P.C.S. Modelos Biométricos Aplicados ao Melhoramento Genético; UFV: Viçosa, Brazil, 2012. [Google Scholar]
- Cattivelli, L.; Rizza, F.; Badeck, F.-W.; Mazzucotelli, E.; Mastrangelo, A.M.; Francia, E.; Marè, C.; Tondelli, A.; Stanca, A.M. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crops Res. 2008, 105, 1–14. [Google Scholar] [CrossRef]
- Rauf, S.; Al-Khayri, J.M.; Zaharieva, M.; Monneveux, P.; Khalil, F. Breeding Strategies to Enhance Drought Tolerance in Crops. In Advances in Plant Breeding Strategies: Agronomic, Abiotic and Biotic Stress Traits; Springer International Publishing: Cham, Switzerland, 2016; pp. 397–445. [Google Scholar]
- Basnayake, J.; Jackson, P.A.; Inman-Bamber, N.G.; Lakshmanan, P. Sugarcane for water-limited environments. Genetic variation in cane yield and sugar content in response to water stress. J. Exp. Bot. 2012, 63, 6023–6033. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Basnayake, J.; Jackson, P.A.; Chen, X.; Zhao, J.; Zhao, P.; Yang, L.; Bai, Y.; Xia, H.; Zan, F.; et al. Growth and yield of sugarcane genotypes are strongly correlated across irrigated and rainfed environments. Field Crops Res. 2016, 196, 418–425. [Google Scholar] [CrossRef]
- Mulualem, T.; Abate, M. Heterotic Response in Major Cereals and Vegetable Crops. Int. J. Plant Breed. Genet. 2016, 10, 69–78. [Google Scholar] [CrossRef][Green Version]
- Hallauer, A.R.; Carena, M.J.; Miranda Filho, J.B. Quantitative Genetics in Maize Breeding; Springer: Berlin, Germany, 2010. [Google Scholar]
- Babu, R.; Nair, S.K.; Kumar, A.; Rao, H.S.; Verma, P.; Gahalain, A.; Singh, I.S.; Gupta, H.S. Mapping QTLs for popping ability in a popcorn × flint corn cross. Theor. Appl. Genet. 2006, 112, 1392–1399. [Google Scholar] [CrossRef]
- Oliveira, G.H.F.d.; Amaral, C.; Revolti, L.T.M.; Buzinaro, R.; Moro, G.V. Genetic variability in popcorn synthetic population. Acta Sci. Agron. 2018, 41, 39497. [Google Scholar] [CrossRef]
- Santos, J.S.; Souza, Y.P.; Vivas, M.; Amaral Júnior, A.T.; Almeida Filho, J.E.; Mafra, G.S.; Viana, A.P.; Gravina, G.A.; Ferreira, F.R.A. Genetic merit of popcorn lines and hybrids for multiple foliar diseases and agronomic properties. Funct. Plant Breed. J. 2020. no prelo. [Google Scholar]
- Coan, M.M.D.; Pinto, R.J.B.; Kuki, M.C.; do Amaral Júnior, A.T.; Figueiredo, A.S.T.; Scapim, C.A.; Warburton, M. Inheritance Study for Popping Expansion in Popcorn vs. Flint Corn Genotypes. Agron. J. 2019, 111, 2174. [Google Scholar] [CrossRef]
- Cabral, P.D.S.; Amaral Júnior, A.T.D.; Freitas, I.L.D.J.; Ribeiro, R.M.; Silva, T.R.D.C. Cause and effect of quantitative characteristics on grain expansion capacity in popcorn. Rev. Ciênc. Agron. 2016, 47, 108–117. [Google Scholar] [CrossRef]
- Ematné, H.J.; Nunes, J.A.R.; Dias, K.O.G.; Prado, P.E.R.; Souza, J.C. Estimate of genetic gain in popcorn after cycles of phenotypic recurrent selection. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Tollenaar, M.; Lee, E.A. Physiological dissection of grain yield in maize by examining genetic improvement and heterosis. Maydica 2006, 51, 399–408. [Google Scholar]
- Tollenaar, M.A.A.L.E.A. Physiological Basis of Heterosis for Grain Yield in Maize. Crop Sci. 2004, 44, 2086–2094. [Google Scholar] [CrossRef]
- Holá, D.; Benešová, M.; Fischer, L.; Haisel, D.; Hnilička, F.; Hniličková, H.; Jedelský, P.L.; Kočová, M.; Procházková, D.; Rothová, O.; et al. The disadvantages of being a hybrid during drought: A combined analysis of plant morphology, physiology and leaf proteome in maize. PLoS ONE 2017, 12, e0176121. [Google Scholar] [CrossRef] [PubMed]
- Cairns, J.E.; Sanchez, C.; Vargas, M.; Ordoñez, R.; Araus, J.L. Dissecting Maize Productivity: Ideotypes Associated with Grain Yield under Drought Stress and Well-watered Conditions. J. Integr. Plant Biol. 2012, 54, 1007–1020. [Google Scholar] [CrossRef]
- Blum, A. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. F. Crop. Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Vittorazzi, C.; Júnior, A.T.A.; Guimarães, A.G.; Silva, F.H.L.; Pena, G.F.; Daher, R.F.; Gerhardt, I.F.S.; Oliveira, G.H.F.; Santos, P.H.A.D.; Souza, Y.P.; et al. Evaluation of genetic variability to form heterotic groups in popcorn. Genet. Mol. Res. 2018, 17, gmr18083. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).