Abstract
Complex interactions in soil carbon and nitrogen (C-N) synchronisation in tropical perennial orchards are highly responsive to fertiliser chemistry. However, the intensity and stage-specific dynamics of these interactions are not well quantified. Six nitrogen regimes, namely, urea (URT), ammonium (AMT), nitrate (NT), slow-release fertiliser (SRT), bio-organic fertiliser (BFT), and an unfertilised control, were assessed at the vegetative, flowering, fruit-set, and maturity stages of durian cultivated on highly weathered tropical soils. A two-way ANOVA indicated high to very high treatment × phenology interactions for almost all soil properties (p < 0.001), indicating that nutrient responses were highly stage-dependent. The highest soil organic carbon (SOC) and cation exchange capacity (CEC) values were consistently obtained with the BFT, which was often associated with significant differences compared with synthetic treatments. In contrast, the SRT showed the most consistent nutrient release behaviour, especially in flowering. On the other hand, soil pH did not differ significantly among the treatments during the vegetative and maturity stages. A significant decrease in pH was observed for the URT and NT treatments during the flowering stage, indicating temporary acidification at this stage and steep increases in nitrate nitrogen (NO3—N), indicating strong nitrification and attenuated carbon (C) stabilisation. Leaf nutrient responses were increased in phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg) by 23% in response to the SRT and BFT. The NT and URT tended to enhance leaf nitrogen (N) primarily, and PCA (59–69% variance explained) clearly displayed clustering of the fertiliser effects, with the maximum difference at flowering, the peak period of nutrient demand in the crop. In general, fertiliser chemistry and phenophase jointly controlled the C-N partitioning, soil chemical paths, and nutrient yield correlations. The BFT and SRT showed the greatest significant gains in soil fertility and nutrient retention, making them the best high-performance alternatives in sustainable durian production in tropical systems.
1. Introduction
Durian (Durio zibethinus) is an important fruit crop in Southeast Asia, of great economic importance [1], but with excessive nutrient requirements [2]. The durability and sustainability of the durian fruit, together with the perennial nature of the crop, have caused an endemic burden on the soils of the orchards, and caused a productivity and sustainability crisis [3,4]. This is particularly significant in tropical conditions, which naturally have low soil organic matter (SOM), high mineralisation, and high rainfall, which in combination contribute to soil health degradation, destabilise the nitrogen (N) and present a risk to the long-term soil fertility [5]. To escape this hurdle, the mechanistic conceptualisation of soil nutrient interactions, i.e., the interaction that exists in the background of soil organic carbon (C) and N, would have to be adopted by the empirical fertiliser methods [6].
Carbon–nitrogen (C–N) coupling is indeed a central nexus in biogeochemistry and strongly controls soil fertility [7]. The C pools aid the N cycle: they provide energy for the minute microbial mineralisation of organic N, cation exchange capacity (CEC) to store ammonium, and they help reverse the acidifying influence of nitrification [8]. On the other hand, the evolution of the C space can be radically altered as the number and duration of N inputs increase or decrease, thereby favouring or deterring the decomposition process of organic matter and changing the chemical composition of the soil [9,10]. This mutualism in significantly developed perennial systems, such as durian orchards, not only provides immediate access to nutrient resources but also ensures future soil health.
C-N coupling, which lies at the heart of a durian plantation, has not been duly computed, even though fertilisers are still at the mercy of the utilisation of empirical urea and mineral N [11]. Traditional fertilisers are readily available to N but tend to be less sensitive to crop-specific physiological needs as well as having long-term (changing), less predictable impacts on the most labile soil properties of soil organic carbon (SOC), soil CEC, soil pH, and availability of base cations [12,13]. Another alternative is new fertiliser technologies that create a temporarily dynamic environment, to which current fertiliser regimes are unable to react [14,15]. As demonstrated by the formulations applied (SRT), the number of plants required is reduced, and losses and nutrient utilisation increase as the quantity of N released per plant decreases [16]. Meanwhile, bio-organic fertilisers (BFT) have the potential to supplement nutrient levels, favour the growth of microorganisms, and fix SOCs, thereby increasing the soil’s ability to store nutrients [17]. However, the degree to which they can mediate coupled C-N dynamics of the soils of the tropical orchard is distinct, especially at the most critical stage, at the time of the growth of the durian, which is also an open urgent question which has never been answered in the context of optimising the management of durian soils. The quantitative responses of SOC, CEC, pH, the inorganic N pools, and multi-nutrient availability to six different fertiliser plans to the durian phenological cycle are thus being determined. This study will (i) explain the mechanical interplay between nitrogen sources and soil carbon stability, (ii) explain the changes in the primary soil chemical signatures as a result of phenology, and (iii) clarify what fertiliser regimes produce the best productivity coupled with the simultaneous preservation of soil chemical stability. Provision of solutions to these knowledge gaps will provide a scientific basis for policy on fertilisers, making tropical perennial horticulture more profitable and sustainable despite increasing environmental pressures.
2. Materials and Methods
2.1. Study Site
The field study was conducted at the Durian Science and Technology Courtyard, situated within Hainan Wan He Bao Biotechnology Co., Ltd., Sanya, Hainan, ChinaHainan Province (18°21′25″ N 109°10′19″ E). This area features a tropical monsoon climate, with an average temperature of around 24.8 °C. The district experiences a tropical wet and dry or savanna climate (Classification: Aw), receiving approximately 1588 mm of rainfall distributed across 161.37 rainy days per year (44.21% of the year). The area records two rainy seasons: the primary wet season (May to October) driven by the summer monsoon, and the shorter secondary wet season (March to April) caused by pre-monsoon rainfall. According to the World Reference Base for Soil Resources, the soils in the area are classified as Ferrosol, with granite serving as the primary parent material [18].
2.2. Experimental Design
The experiment was a field study conducted during the 2024 crop season, with a randomised block design (RBD). Treatment plots had three durian trees, and this was repeated three times, giving a total of nine trees per treatment plot. All the trees were 5 years old, and spacing was 8 m × 8 m, providing sufficient buffer space to avoid cross-treatment effects between plots and to maintain uniform growth conditions across the different treatments. Fertiliser applications were organised into three operational intervals aligned with durian phenology: pre-flowering (preparation and vegetative build-up), flowering → fruit-set (peak reproductive demand), and maturity (fruit enlargement and ripening). Each tree received a total of 1.60 kg N, 0.60 kg P2O5, and 2.95 kg K2O per growth cycle. Nutrients were applied in a circular band at the canopy drip line to target the active feeder roots. The interval doses per tree were as follows: pre-flowering: 0.56 kg N, 0.30 kg P2O5, 0.44 kg K2O; flowering → fruit-set: 0.72 kg N, 0.30 kg P2O5, 1.18 kg K2O; maturity: 0.32 kg N, 0.00 kg P2O5, 1.33 kg K2O. Nitrogen was supplied using five chemically distinct fertiliser sources obtained from different fertiliser manufacturers in China: urea (amide-N, 46% N; Sinofert Holding Co., Ltd.), calcium nitrate (nitrate-N, 15.5% N with Ca2+; Dalian Saiyang Chemical Group Co., Ltd.), ammonium sulphate (ammonium-N, 21% N and 24% S; Shandong Hualu-Hengsheng Chemical Co., Ltd.), a polymer-coated slow-release nitrogen fertiliser (controlled-release urea, ~40% N; Kingenta Ecological Engineering Group Co., Ltd.), and a bio-organic nitrogen fertiliser containing organic N compounds (amino acids and microbial-derived N; Hainan Wanhebao Biotechnology Co., Ltd.). None of the nitrogen fertilisers contained phosphorus or potassium. Phosphorus (reported as P2O5) and potassium (reported as K2O) were supplied separately using mineral P and K fertilisers and were applied across all nitrogen treatments, ensuring that P and K availability was balanced among nitrogen regimes. The split proportions were chosen to reflect durian’s physiological demands (greater N and P2O5 support during the reproductive transition and increased K2O demand during fruit fill). Applications were implemented as single events at each interval; where field operations required, the second interval might be applied as two closely timed sub-applications (flowering and fruit-set) without changing total interval amounts. All other management (irrigation, manual weed control, tillage) followed standard orchard practices [19].
2.3. Soil Sampling and Physicochemical Characterisation
Before the experiment commenced in March 2024, soil samples were collected using an auger from 20 locations across the experimental field, including the centre, at a depth of 0–20 cm. The samples were homogenised to form composite soil samples for baseline soil characterisation. The termination of a treatment was determined by completion of the corresponding stage, following which soil and leaf samples were obtained to undergo analysis. One portion was air-dried to assess physicochemical properties. At the same time, the other part was stored at −20 °C to study the enzymatic characteristics of soil enzymes, including urease, dehydrogenase, alkaline phosphatase, and β-glucosidase. Bulk density was determined using a core sampler [20]. Soil pH was assessed with a pH meter using a glass electrode and a soil/water (w/v) ratio of 1:5 [21], while electrical conductivity (EC) was measured with a multimeter electrode (SX 731, Sanxin) at 25 °C [22]. Soil organic matter was determined using the potassium dichromate oxidation method described by Walkley and Black [23]. Soil ammonium N (NH4+-N) was extracted with 2 M KCl at a soil-to-solution ratio of 1:5 and subsequently quantified by indophenol blue colourimetry at 625 nm [24]. Soil nitrate (NO3−-N) was extracted with 2 M KCl at a soil-to-solution ratio of 1:5 and then quantified using dual-wavelength ultraviolet spectrophotometry at 220 and 275 nm [24]. Soil available phosphorus (AP) was analysed using the phosphomolybdate blue colorimetric method at a wavelength of 700 nm [25]. Soil available potassium (AK) was extracted using 1 M NH4OAc and analysed using flame photometry [26]. Cation exchange capacity was determined using an atomic absorption spectrometer using the ammonium acetate (NH4) method at pH 7.0 [22]. Method blanks and duplicate samples were included in each analytical batch to assess contamination and ensure analytical precision, and calibration standards were run to verify measurement accuracy. The initial soil physicochemical properties of the experimental sites are presented in Table 1.

Table 1.
The initial soil physicochemical properties at the experimental site.
2.4. Enzyme Assays
We analysed four soil extracellular enzyme activities (EEAS): β-glucosidase (β-G), dehydrogenase (DHA), urease, and alkaline phosphatase (ALP). Enzyme tests were conducted using 96-well microplates [27]. Urease activity was assayed by incubating 1 g of fresh soil with 0.5 mL of 0.2 M urea in 0.1 M phosphate buffer (pH 6.7) at 37 °C for two hours in 96-well microplates, followed by extraction with 2 M KCl and quantification of NH4+ using the indophenol blue method at 625 nm [28]. β-glucosidase activity was measured by incubating 0.5 g of soil with 0.25 mL of 25 mM p-nitrophenyl-β-D-glucopyranoside (pNPG) in acetate buffer (pH 5.5) at 37 °C for 1 h, with p-nitrophenol (pNP) release detected at 400 nm. Alkaline phosphatase activity was assessed using 1 g of soil incubated with 0.5 mL of 0.1 M p-nitrophenyl-phosphate (pNPP) in Tris buffer (pH 11) at 37 °C for one hour, and the liberated pNP was measured at 410 nm according to Gerritse and van Dijk [29]. Dehydrogenase activity (DHA) was determined by incubating 5 g of soil with 1 mL of 3% 2,3,5-triphenyltetrazolium chloride (TTC) in 0.1 M Tris-HCl buffer (pH 7.6) at 30 °C for 24 h in the dark, followed by extraction of triphenyl formazan (TPF) with methanol and measurement at 485 nm. All enzyme activities were expressed on a dry soil weight basis and quantified using a microplate reader (BioTek Instruments, Winooski, VT, USA) [30].
2.5. Statistical Analysis
A two-way ANOVA was conducted to test the main effects of stage and N treatment on these variables, as well as their interaction. The model was used to test how phenophase and N source (urea, ammonium, nitrate, slow-release fertiliser, bio-fertiliser, and control) impacted each outcome variable. Any significant main effects or interactions were followed by Tukey’s post hoc significant difference (HSD) to reveal treatment differences. Kruskal–Wallis tests were used for nonparametric comparisons when normality or homogeneity of variances had been violated. Furthermore, Pearson’s correlation was conducted to assess the relationship between microbial biomass and enzyme activities and principal component analysis (PCA) was performed to visualise the overall variation in the ecoenzymatic stoichiometry across treatments. Statistical analyses were performed using R statistical software (version 4.5.2). All statistical tests were performed with a significance level of p < 0.05. Post hoc tests were used to perform mean separations, and the results are presented in the Supplementary Materials.
3. Results
3.1. Influence of Fertiliser Regime and Phenological Stage on Soil Properties
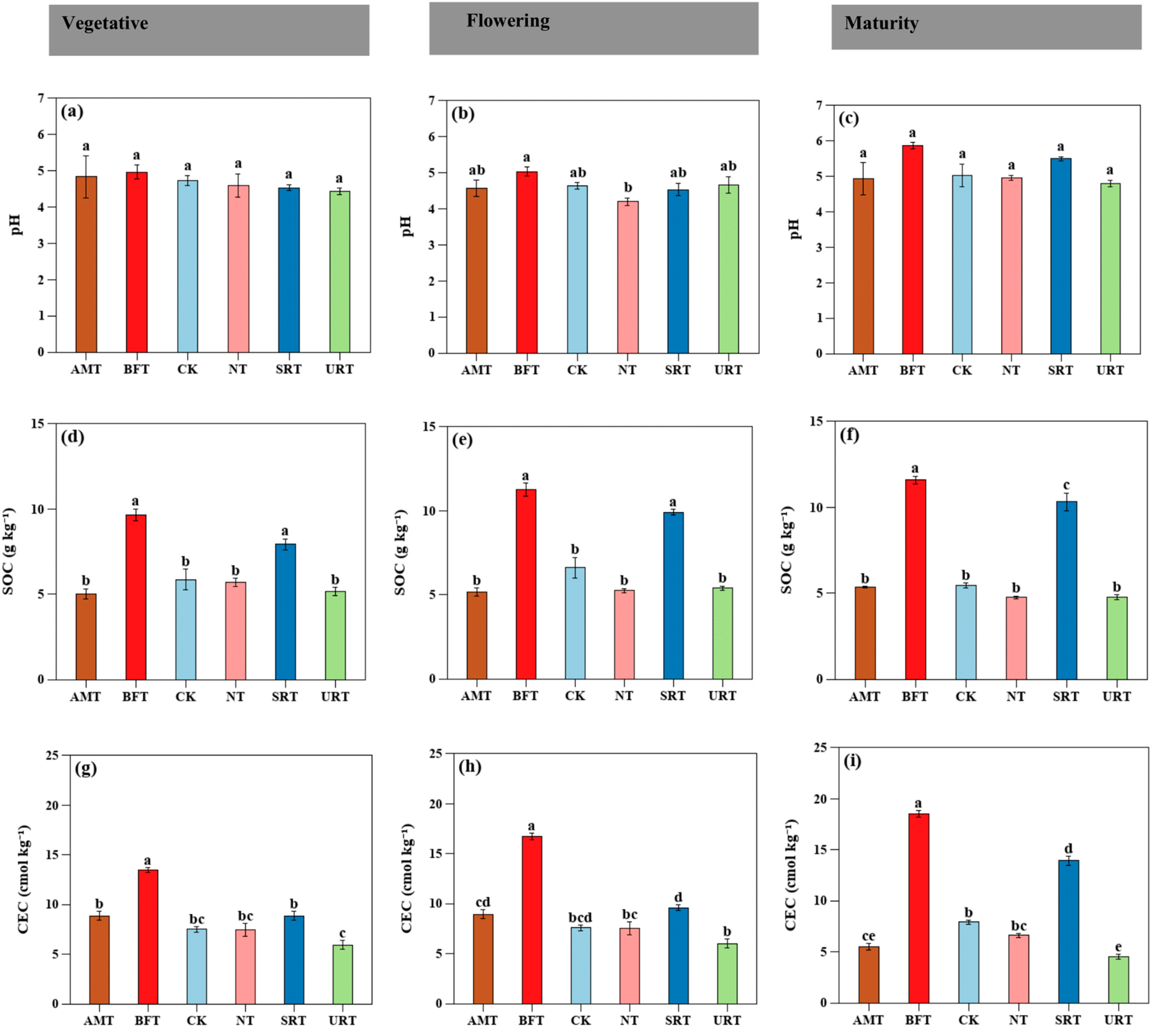
A one-way ANOVA revealed that nitrogen fertiliser type significantly influenced most soil physicochemical properties within each phenological stage. Soil pH showed no treatment effects during the vegetative stage but exhibited marginal differences at flowering and maturity. In contrast, SOC was strongly affected by fertiliser type across all stages (p < 0.001), with the BFT consistently producing the highest SOC levels. The CEC responded strongly to fertiliser type during vegetative, flowering, and maturity stages (p < 0.001), with the BFT maintaining the highest exchange capacity (Figure 1a–g). A two-way ANOVA showed that soil physicochemical properties were strongly influenced by nitrogen fertiliser type and durian growth stage, and in many cases by their interaction. Soil pH was significantly affected by both treatment and stage (p < 0.05), whereas EC exhibited significant main and interaction effects (p < 0.05). Soil organic carbon (SOC) showed intense treatment, stage, and interaction effects (all p < 0.001).
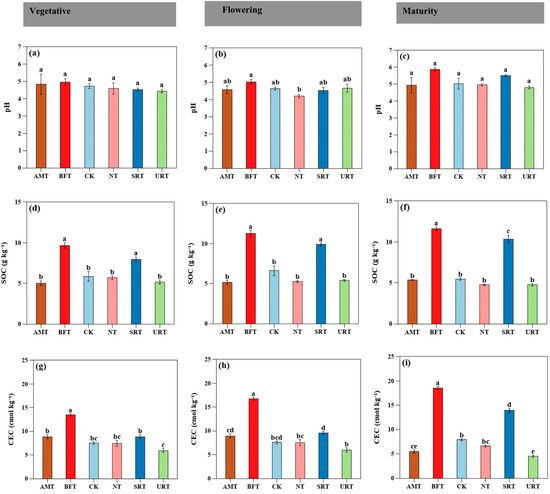
Figure 1.
The influence of various treatments of nitrogen fertiliser (AMT, BFT, CK, NT, SRT, and URT) on the soil pH (a–c), soil organic carbon (SOC; d–f), and cation exchange capacity (CEC; g–i) at three different stages of durian growth: vegetative, flowering, and maturity. Different lowercase letters above bars indicate significant differences among treatments at p < 0.05 (one-way ANOVA followed by Tukey’s HSD test). Values are means ± standard error (SE).
3.2. Stage-Wise Nutrient and Soil Chemical Responses to Nitrogen Regimes
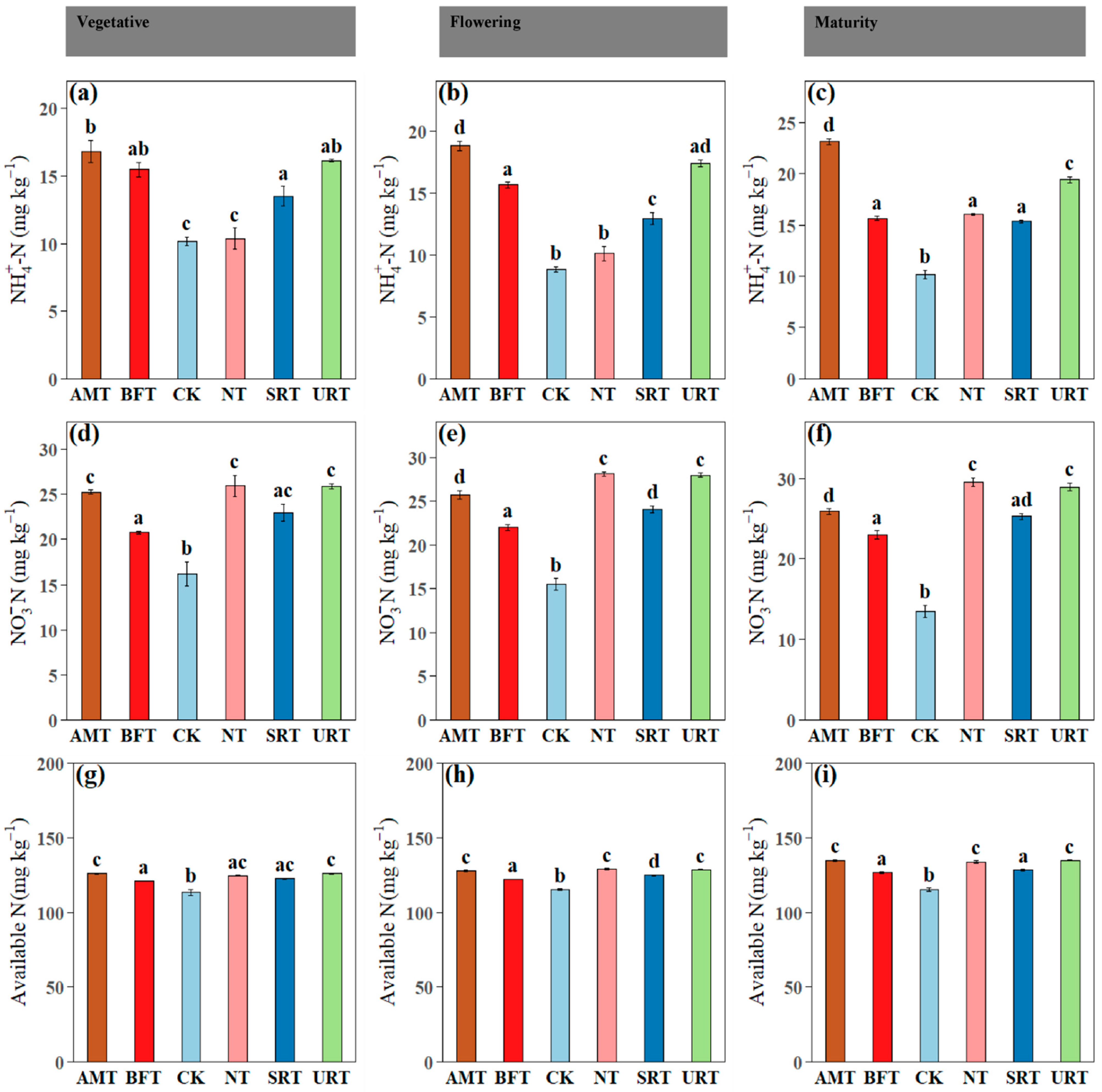
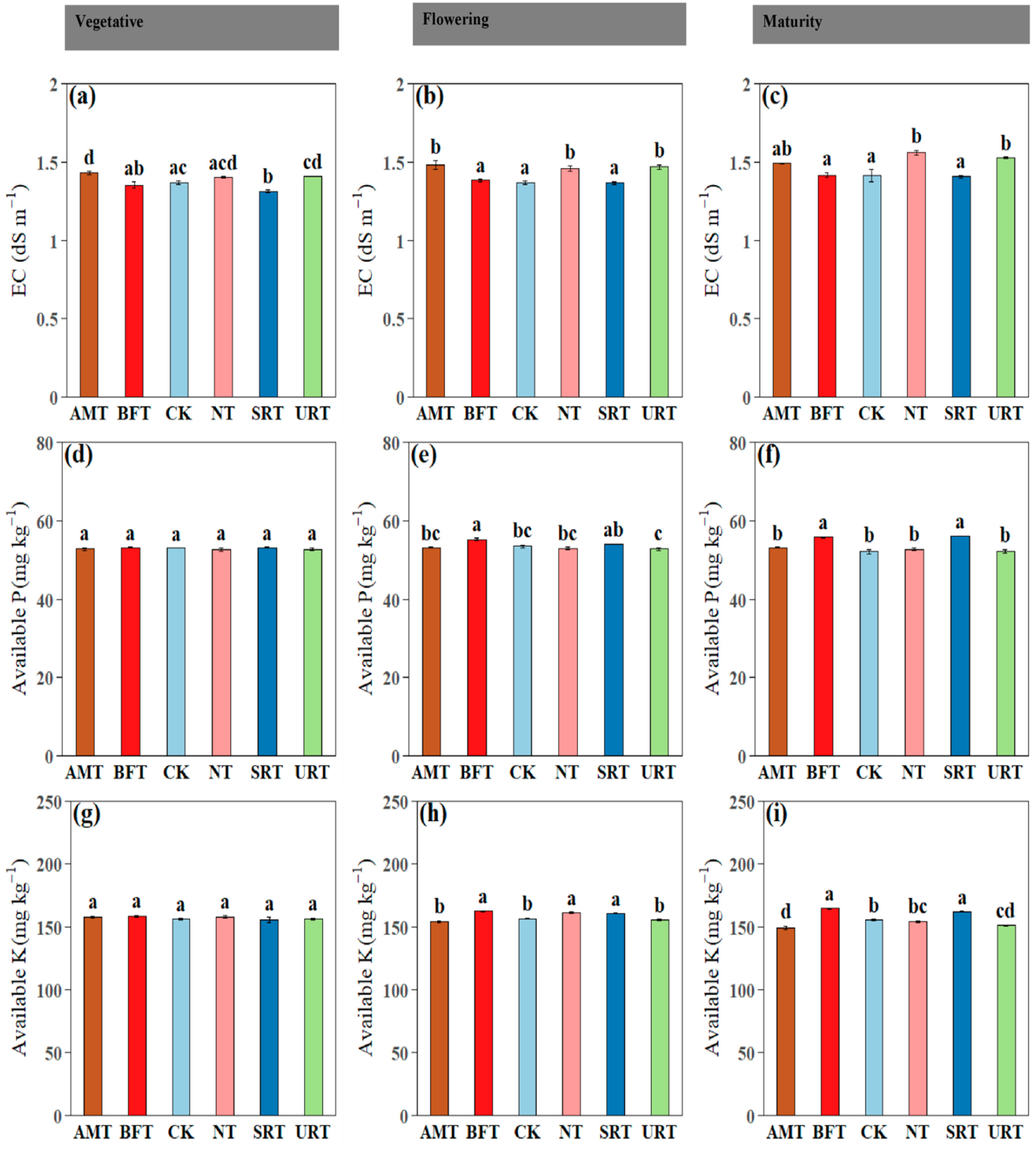
Across phenophases, soil inorganic N fractions differed significantly among treatments (Figure 2). Ammonium-N (NH4+–N) was consistently highest under the AMT at the vegetative, flowering, and maturity stages, with the URT also maintaining elevated NH4+–N relative to the CK, while the CK and NT generally recorded the lowest values (Figure 2a–c). In contrast, nitrate-N (NO3−–N) showed a clear treatment shift, with the NT and URT producing the highest NO3−–N concentrations at flowering and maturity, and the AMT, NT, and URT showing higher NO3-N concentrations than the CK at the vegetative stage (Figure 2d–f). This pattern indicates stronger nitrification under the synthetic N regimes, particularly the NT and URT, likely reflecting greater NH4+ availability and enhanced microbial nitrification compared with the control. Plant available N followed similar trends, with the AMT and URT maintaining significantly higher levels than the CK at the vegetative, flowering, and maturity stages, while the NT and SRT also exceeded the control at flowering and maturity (Figure 2g–i). Across phenophases, soil electrical conductivity (EC) and available P and K exhibited treatment- and stage-dependent responses (Figure 3). Soil EC differed significantly among treatments at all stages, with the AMT and URT generally showing higher EC than the CK at the vegetative and flowering stages, while NT recorded the highest EC at maturity (Figure 3a–c). Available phosphorus showed no significant differences among treatments during the vegetative stage (p > 0.05), confirming uniform P availability early in crop growth (Figure 3d); however, at flowering and maturity, treatment effects became evident, with the BFT and SRT often maintaining higher available P than the CK, while the URT showed comparatively lower values at later stages (Figure 3e,f). Available potassium did not differ significantly among treatments at the vegetative stage (p > 0.05; Figure 3g), but clear separation emerged thereafter, with the BFT, NT, and SRT sustaining higher K availability than the AMT and CK at flowering, and the BFT and SRT maintaining the highest K levels at maturity (Figure 3h,i).
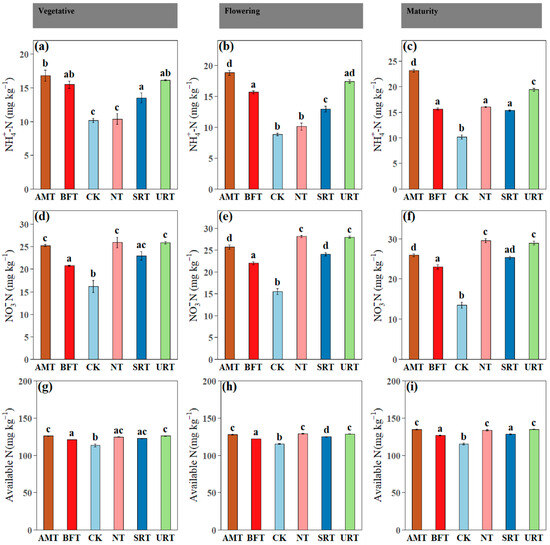
Figure 2.
The influence of various N treatments (AMT, BFT, CK, NT, SRT, and URT) on soil ammonium nitrogen (NH4+N; a–c), nitrate nitrogen (NO3-N; d–f), and available nitrogen (g–i) at three different stages of durian growth: vegetative, flowering, and maturity. Different lowercase letters above bars indicate significant differences among treatments at p < 0.05 (one-way ANOVA followed by Tukey’s HSD test). Values are means ± standard error (SE).
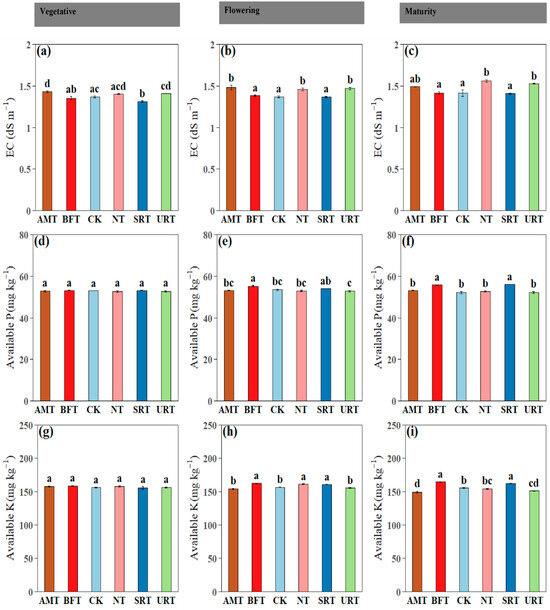
Figure 3.
The influence of various nitrogen fertiliser treatments (AMT, BFT, CK, NT, SRT, and URT) on soil electrical conductivity (EC) (a–c), available P (d–f), and available K (g–i) at three different stages of durian growth: vegetative, flowering, and maturity. Different lowercase letters above bars indicate significant differences among treatments at p < 0.05 (one-way ANOVA followed by Tukey’s HSD test). Values are means ± standard error (SE).
3.3. Fertiliser-Driven Variation in Leaf Nutrient Dynamics
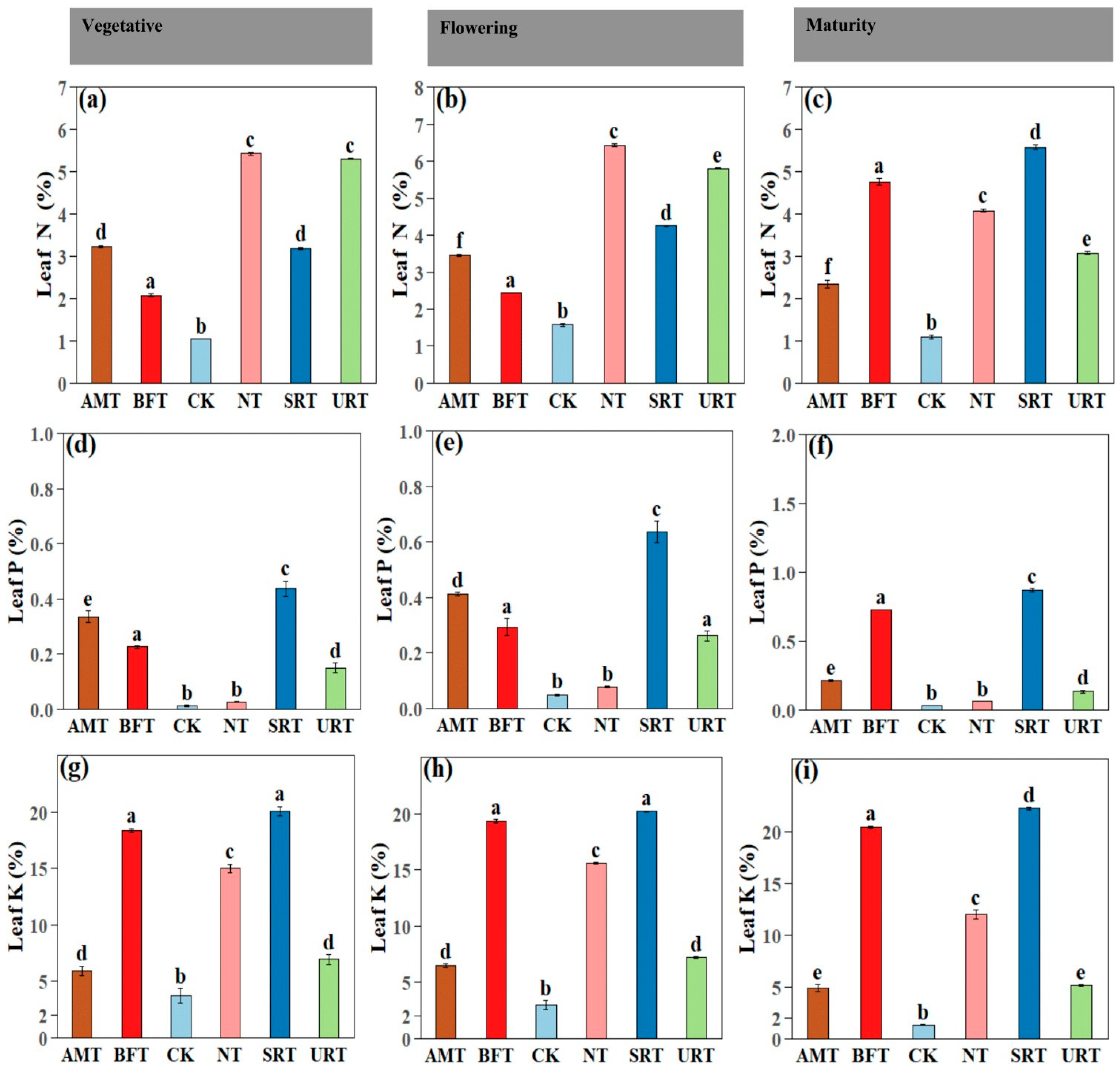
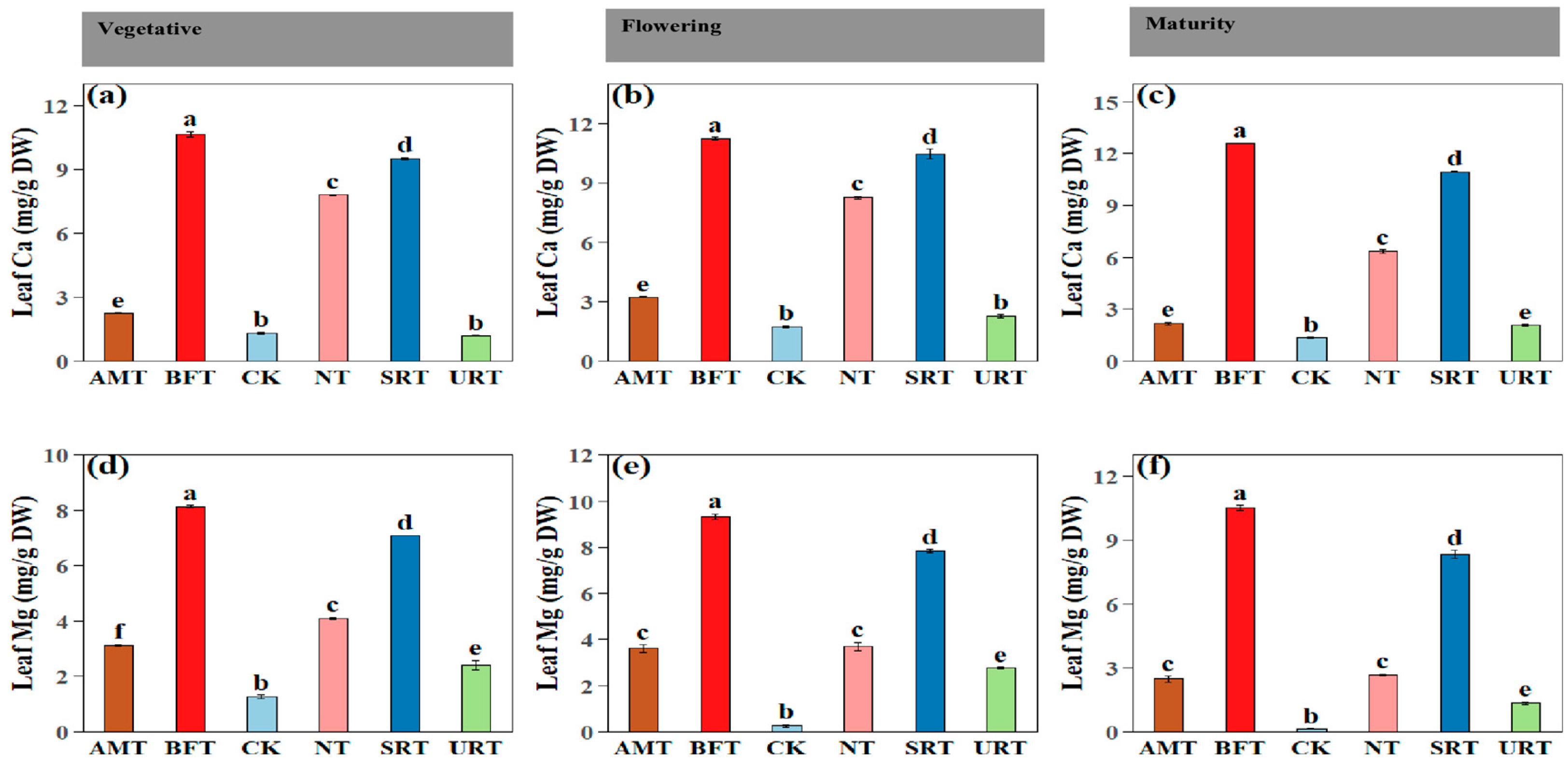
Fertiliser treatments had a highly significant effect on leaf nutrient concentrations across all durian growth stages (ANOVA, p < 0.001). At maturity, treatment effects remained pronounced, with SRT producing the highest leaf P and K concentrations and among the highest leaf N values (Figure 4). During the vegetative stage, the NT and URT significantly increased leaf N, whereas the SRT and BFT resulted in the highest leaf P and K concentrations (Figure 4). The CK consistently recorded the lowest leaf nutrient concentrations across all stages. Similar patterns were observed at flowering, with the NT maintaining the highest leaf N levels and the SRT and BFT again promoting greater accumulation of P and K (Figure 4). Across all phenophases, nitrogen-based treatments (NT and URT) primarily enhanced leaf N accumulation, with comparatively smaller effects on P and K, whereas the SRT and BFT consistently improved leaf P and K concentrations. The AMT generally produced intermediate nutrient levels. Similar patterns were observed at flowering, SRT and BFT promoting greater accumulation of leaf Ca and leaf Mg (Figure 5) Overall, slow-release and bio-fertiliser treatments resulted in more balanced and sustained multi-nutrient uptake, while the untreated control remained nutrient-deficient throughout crop development.
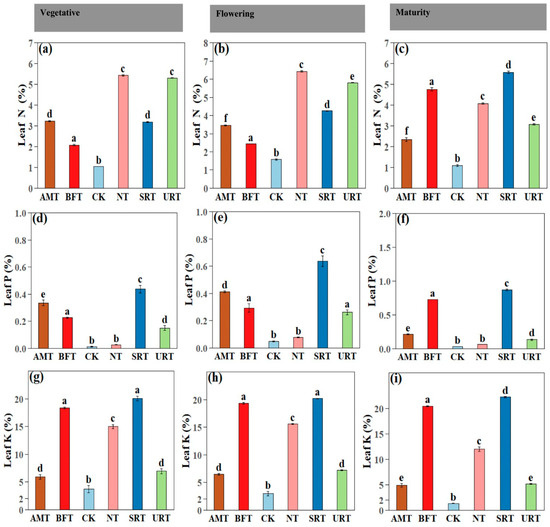
Figure 4.
The effects of different treatments of nitrogen fertiliser (AMT, BFT, CK, NT, SRT, and URT) on leaf nitrogen (a–c), phosphorus (d–f), and potassium (g–i) concentrations at three different stages of durian growth: vegetative, flowering, and maturity. Different lowercase letters above bars indicate significant differences among treatments at p < 0.05 (one-way ANOVA followed by Tukey’s HSD test). Values are means ± standard error (SE).
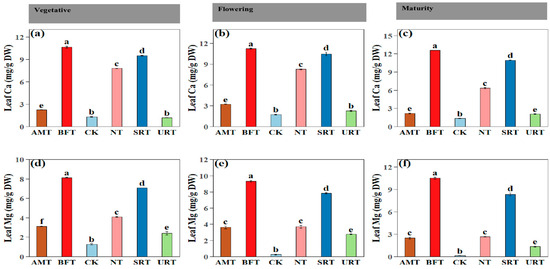
Figure 5.
The effects of different treatments of nitrogen fertiliser (AMT, BFT, CK, NT, SRT, and URT) on Ca (a–c) and Mg (d–f) concentrations at three different stages of durian growth: vegetative, flowering, and maturity. Different lowercase letters above bars indicate significant differences among treatments at p < 0.05 (one-way ANOVA followed by Tukey’s HSD test). Values are means ± standard error (SE).
3.4. Multivariate Analysis of Soil Chemical and C-N Dynamics Across Growth Stages
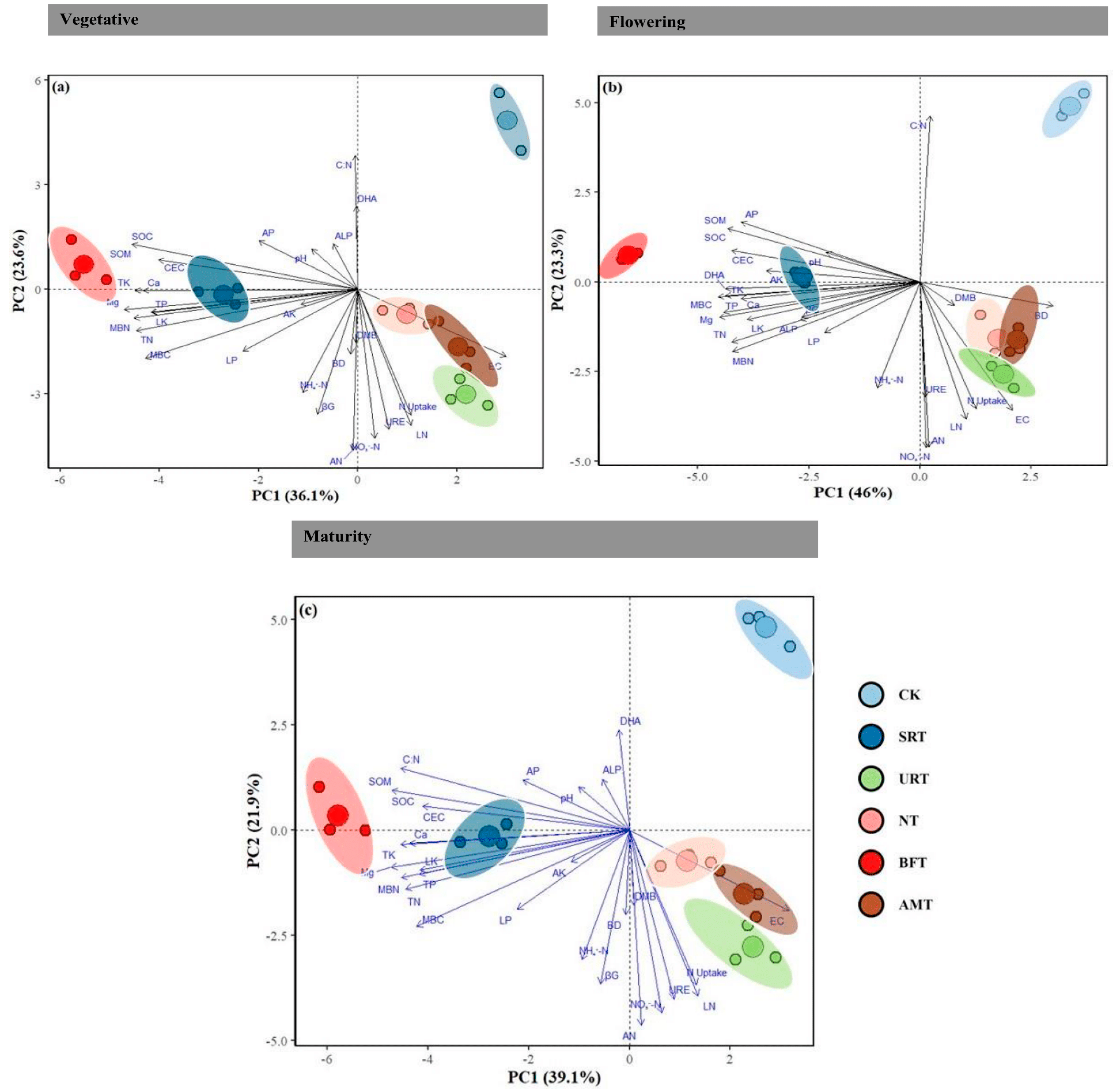
Principal component analysis (PCA) demonstrated clear differentiation of fertiliser treatments across all durian growth stages, with the first two components explaining 59.7%, 69.3%, and 61.0% of total variation at the vegetative, flowering, and maturity stages, respectively (Figure 6a–c). Treatments were moderately separated during the vegetative stage, with the NT and URT aligning with nitrogen-related variables, the BFT and AMT clustering with organic matter-related attributes, and the CK remaining distinct along PC2. Separation became most pronounced at the flowering stage, where the BFT was strongly associated with carbon- and nutrient-related soil properties, the SRT corresponded to SOC and AP, and the NT/URT aligned with mineral N forms and leaf N. At maturity, clustering patterns resembled those in the vegetative stage, with the BFT, SRT, and AMT associating with soil nutrient parameters, and the NT/URT again linked to N-related variables, while the CK remained isolated along PC2.
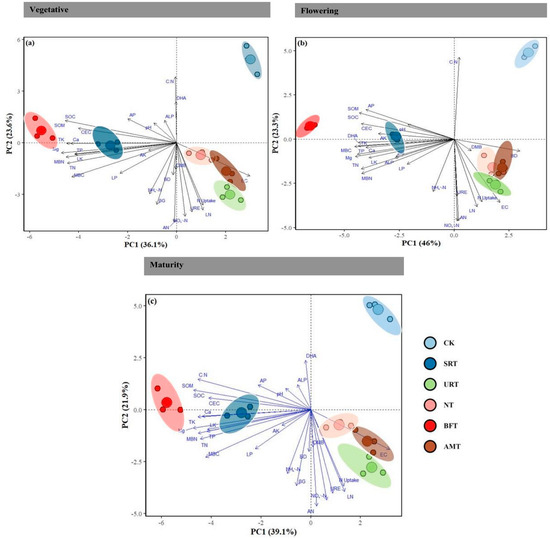
Figure 6.
Principal component analysis (PCA) of soil physicochemical and biological properties under different nitrogen fertiliser treatments across durian growth stages: (a) vegetative; (b) flowering; (c) maturity. Vectors represent the contribution of soil variables to the ordination. Colored ellipses indicate treatment groups (CK, control; SRT, slow-release nitrogen; URT, urea; NT, nitrate; BFT, bio-fertiliser; AMT, ammonium). Percentages on axes denote the variation explained by each principal component. Dashed vertical and horizontal lines represent the zero values (origin) of PC1 and PC2, respectively. Abbreviations: SOC, soil organic carbon; SOM, soil organic matter; TN, total nitrogen; CEC, cation exchange capacity; AP, available phosphorus; AK, available potassium; TK, total potassium; Ca, calcium; Mg, magnesium; MBC, microbial biomass carbon; MBN, microbial biomass nitrogen; ALP, alkaline phosphatase; BG, β-glucosidase; URE, urease; DHA, dehydrogenase activity; NO3−-N, nitrate nitrogen; NH4+-N, ammonium nitrogen; EC, electrical conductivity; pH, soil pH; LK, leaf potassium; LN, leaf nitrogen; LP, leaf phosphorus; BD, bulk density; AN, available nitrogen; DMB, dry matter biomass; N uptake, nitrogen uptake; C:N, carbon-to-nitrogen ratio.
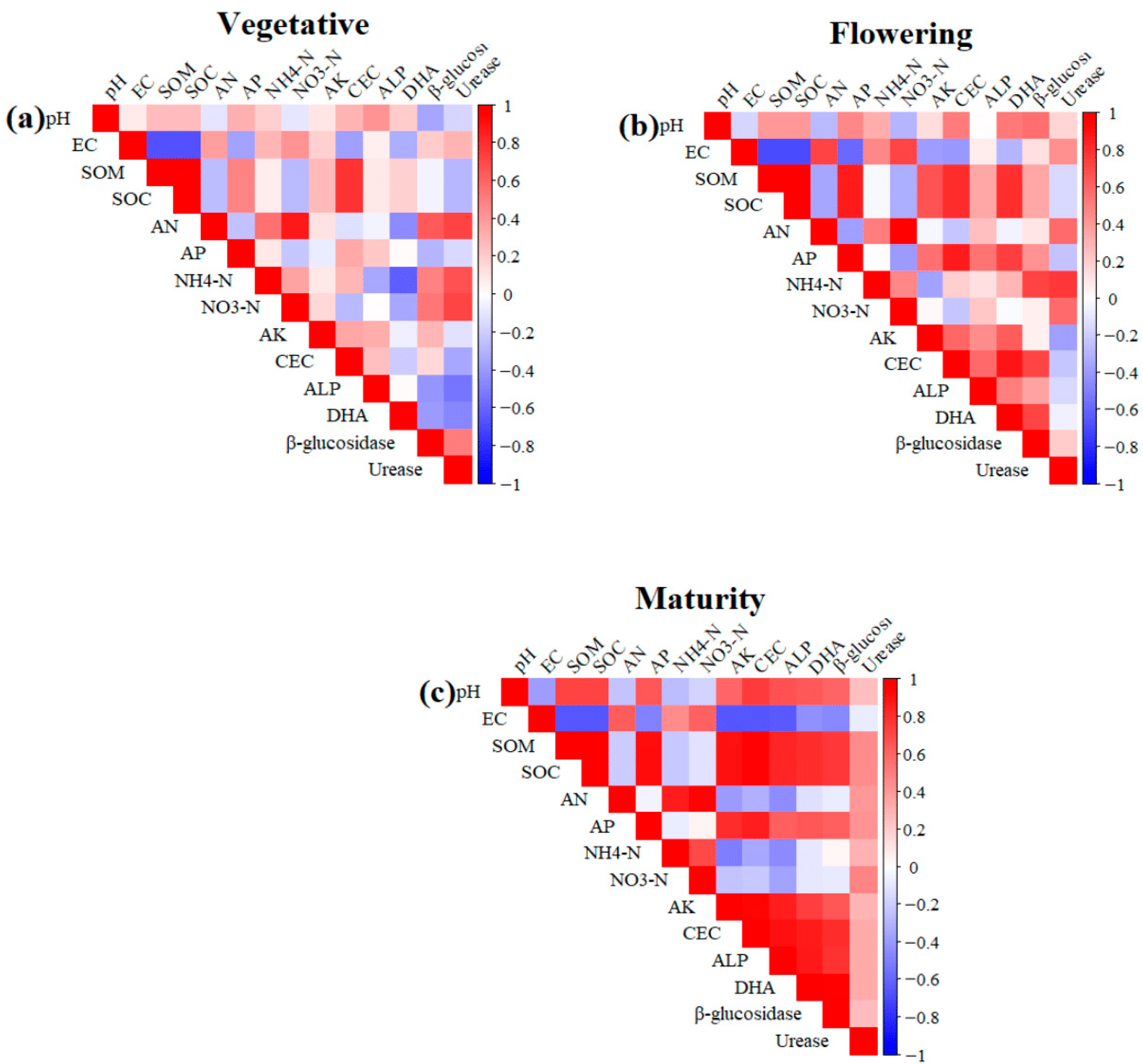
Overall, the PCA biplots revealed that fertiliser treatments formed distinct clusters throughout the growth cycle, with the most significant divergence occurring at flowering, indicating that soil–plant interactions influenced by fertilisation were strongly stage-dependent. The heatmaps revealed clear stage-specific correlation patterns among soil physicochemical properties and yield-related characteristics (Figure 7). During the vegetative stage, strong positive correlations were observed among key soil fertility variables, including SOM, SOC, TP, and CEC, indicating that favourable nutrient conditions were already established early in growth (Figure 7a). Soil pH and EC consistently formed distinct clusters and showed negative correlations with most nutrient variables. By the flowering stage, the correlation structure became more integrated, reflecting tighter linkages among soil fertility attributes as plants entered a more nutrient-demanding period (Figure 7b). At maturity, yield traits such as fruit number, fruit size, single-fruit weight, canopy volume, and leaf nutrient concentrations were strongly and positively correlated with soil fertility indicators, demonstrating that enhanced soil conditions translated directly into higher productivity (Figure 7c). Across all stages, pH and EC remained negatively associated with soil fertility parameters and yield traits, suggesting their potential to limit crop performance. Overall, the findings highlight dynamic soil–plant interactions across the durian growth cycle, with strong nutrient-based networks established early, strengthened during flowering, and ultimately underpinning the positive yield relationships observed at maturity.
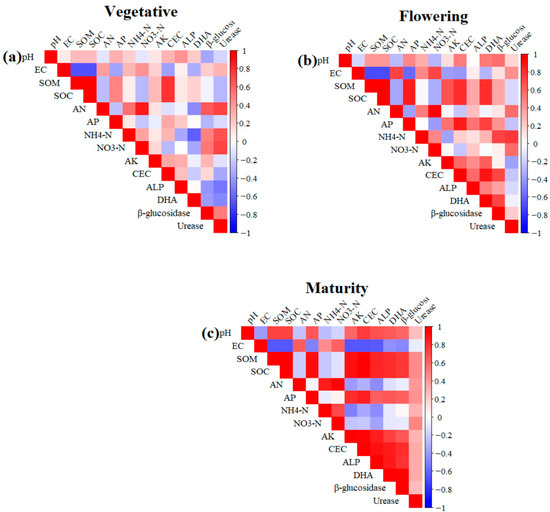
Figure 7.
Correlation heatmap of soil physicochemical, biochemical and plant nutrient parameters at various stages of crop growth: (a) vegetative; (b) flowering; (c) maturity. Pearson’s correlation coefficients are shown in the heatmaps, with red showing positive correlations and blue showing negative correlations.
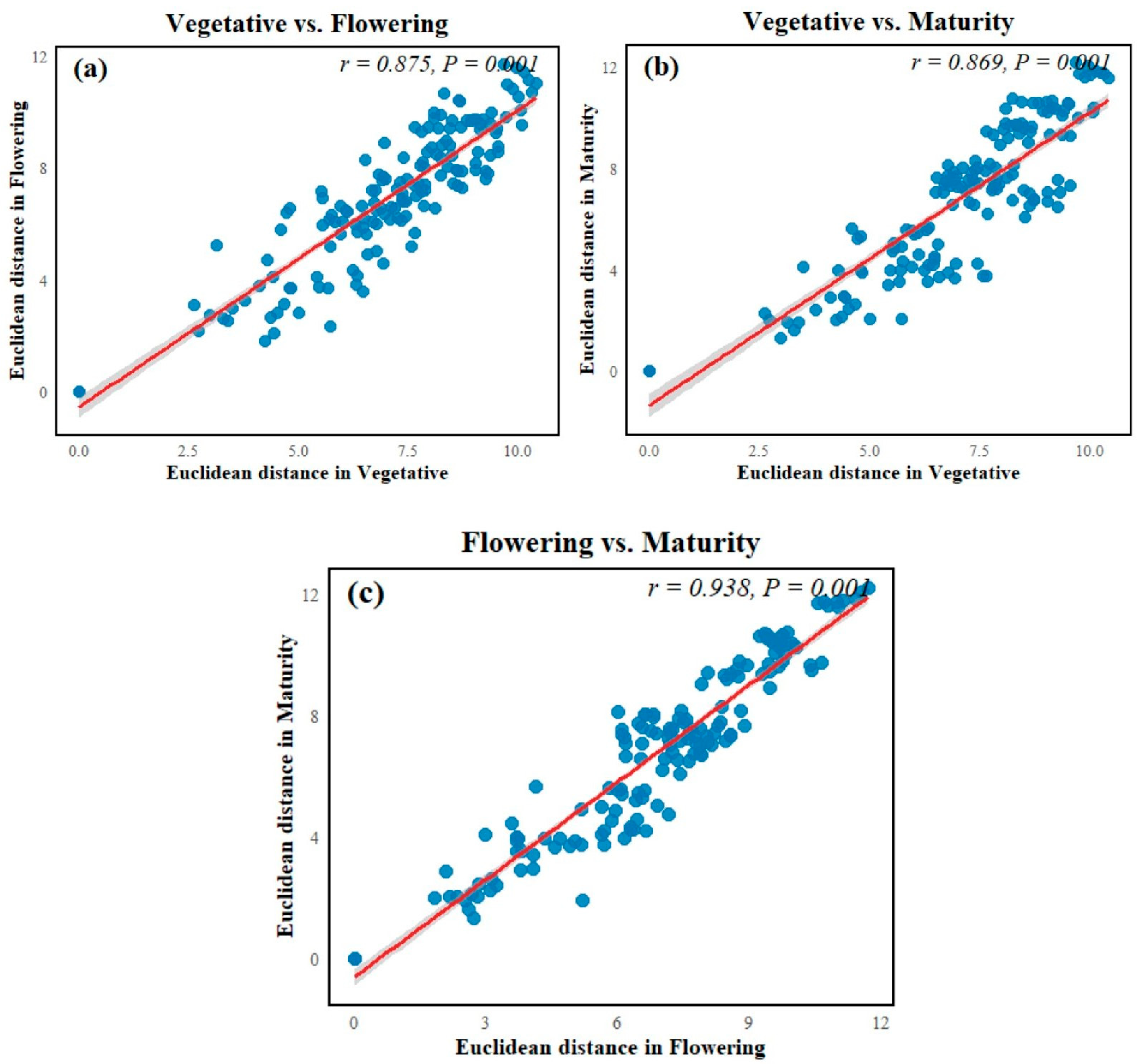
The Mantel test revealed significant correlations between soil physicochemical properties across all crop growth stages. A strong positive relationship was observed between the vegetative and flowering stages (Mantel r = 0.875, p = 0.001), indicating that soil nutrient availability remained broadly comparable between these periods. The vegetative and maturity stages were also highly correlated (Mantel r = 0.869, p < 0.001), suggesting minimal shifts in soil conditions during transition from early to late development. The strongest correlation occurred between the flowering and maturity stages (Mantel r = 0.938, p < 0.001), demonstrating substantial continuity in soil properties during the later stages of crop growth (Figure 8).
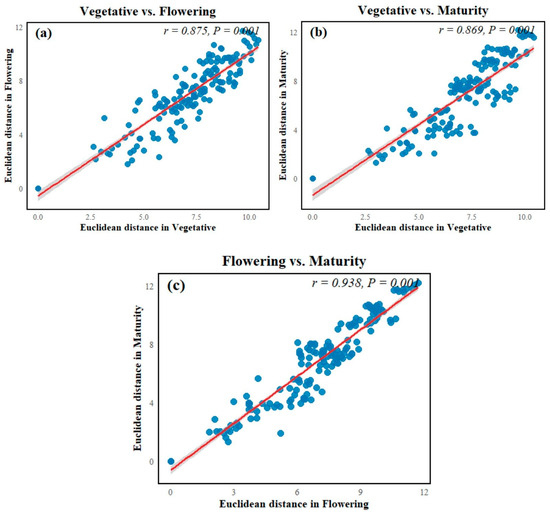
Figure 8.
The Mantel test comparisons of soil physicochemical and biochemical properties across crop growth stages. Scatter plots show pairwise relationships between Euclidean distance matrices of soil properties in (a) vegetative vs. flowering, (b) vegetative vs. maturity, and (c) flowering vs. maturity stages. Mantel correlation coefficients (r) and associated p-values are displayed within each panel.
4. Discussion
4.1. Fertiliser Control of C–N Synchronisation in Tropical Soils
The carbon/nitrogen (C-N) synchronisation in this tropical durian orchard was placed under solid control by the fertiliser chemistry and by the treatments’ evident ability to stabilise soil C levels, in addition to their regulation of phenophase N transformations. Interactive relationships among SOC, CEC, soil pH, and inorganic N pools suggest that C-N relationships were regulated by fertiliser form and crop development stage, which is why fertiliser-mediated pedogenic control models are acceptable in highly weathered tropical soils [31].
Bio-organic fertiliser attained the highest and the most uniform C-N synchronisation, evidenced by the sustained growth in SOC and CEC throughout the different stages (Figure 1d–i). This reaction indicates improved microbial immobilisation and mineral-related organic matter, the prevalent route of stabilisation in tropical soils [32]. The comparatively moderate inorganic N levels in the BFT (Figure 2a–i) suggest that biological N retention is rather more efficient than mineralisation or loss, which is in agreement with previous studies [33] that organic inputs enhance the C utilisation efficiency of the microbial community and increase the C–N coupling. Contrary to previous studies by [34] priming losses observed with organic amendments, SOC depletion was not observed here, suggesting a high level of organo-mineral protection in soils with high sesquioxide levels [35].
Slow-release fertiliser (SRT) enhanced C-N synchronisation by regulating the supply of N kinetically as opposed to C addition [36]. Even though the SOC levels were smaller than under BFT, they still were greater than in fast-soluble mineral N treatments (Figure 1d–f). The accumulation of attenuated NO3−-N in flowering and maturity (Figure 2d) relative to the control (Figure 2e,f) and the comparatively constant soil pH (Figure 1a) are indicative of nitrification-driven acidification suppression, as has been found with controlled-release fertilisers in warm and high-rainfall environments [37].
Fast-soluble N fertilisers (URT and NT), in contrast, disturbed C–N synchronisation, especially at flowering. Sudden rises in NO3−-N (Figure 2d–f) and acidification of the soil (Figure 1a–c) indicate increased rates of nitrification and proton loss, both of which have the potential to decouple the C and N cycles [38]. Inorganic N availability was also high, yet SOC and CEC were low (Figure 1d–i), which suggested the provision of N supply in the short term instead of stabilising C [39]. This is in contrast to the reports of mineral N-induced SOC gains being higher in cooler systems, and is probably influenced by high leaching, low buffering, and high turnover of the microorganisms in the tropical environment [40].
The ammonium-based treatment (AMT) responded in the middle, preserving the high NH4+-N (Figure 2a–c) with no significant increases in SOC or CEC (Figure 1d–i). Long-term acidic nitrification may compensate for possible microbial N immobilisation, which highlights the importance of N form in itself to guarantee C–N coupling [41]. Throughout, C-N synchronisation in tropical soils needed to be effective by ensuring that N release was matched to biological demand and by enabling the stabilisation of C pathways. The BFT realised this by means of microbial C interactions, the SRT by means of regulated N kinetics, and fast-soluble mineral N fertilisers by encouraging N-rich cycling and destabilising soil chemical resilience [42]. The strong separation in treatment at flowering (Figure 6b) marks this stage as a key control point in C–N dynamics in fertiliser-driven tropical perennial systems.
4.2. Phenology-Driven Soil Chemical Trajectories Across Fertiliser Regimes
The chemical conditions of the soil where tropical orchards are developed are dynamically conditional on the phenological stage, but the fertiliser regimes are the determining factors, with the development of a given behaviour towards stabilisation or degradation [43]. The variations in soil pH, SOC, and CEC in response to changes in the seasonal dynamics of the plant, and the changes in microbial processes and geochemistry caused by fertiliser, are all measurable [44]. The underlying mineralogy (kaolinite, sesquioxides, and low-activity clays are dominant in the tropics) provides weak buffering against acidity, and the type of fertiliser is particularly significant [45]. Ammonium fertilisers lower the pH by releasing protons during nitrification, especially during active biological processes such as flowering [45]. These consequences of acidification have been regularly reported in tropical orchard systems and cause loss of base cations, mobilisation of aluminium, and low microbial diversity [46]. Biological amendments, on the other hand, incorporate biological ligands, weak bases, and base cations that raise buffering capacity and also provide resistance against pH degradation [47]. The studies of tropical cocoa, rambutan, and citrus orchard reveal that these organic additions do not cause variability in pH despite the high rainfall and N input [48,49].
The nature of SOC curves across growth stages is a manifestation of a balance between carbon intake in the form of roots and degradation by microbes [50,51]. The tropical fruit trees at reproductive stages tend to allocate C more to roots that produce a strong underground C pulse [52]. Regimes of fertiliser that had the potential to stabilise this carbon, through either encouraging microbial uptake of carbon-enriched compounds or discouraging N-induced spikes of decomposition, were likely to result in a growing SOC with age [53]. Soluble mineral N, on the other hand, is more likely to enhance mineralisation and reduce carbon retention, which is reported in large proportions in tropical agroforestry systems [54]. These contrary trends give credence to the idea that the SOC responses of the tropics are strongly dependent on the nature of the input, the stabilisation of the nutrient by the microorganisms, in addition to the allocation of the nutrients with the phenophase of the plants [55].
The potential for resilience building through carbon enrichment is also evident in the CEC trajectories [56]. Organic matter is also a significant source of the exchange capacity in the very weathered tropical soils, rather than clay minerals [57]. Consequently, the increase in the CEC will be directly proportional to the increase in SOC gains, which would entail greater cation retention, lesser leaching, and relatively high fertility [58,59]. Organic inputs are characterised by high potential in creating an increase in CEC through increasing permanent and pH-dependent charges on humic compounds [60,61]. The converse of this is that soluble mineral N systems are exposed to loss in CEC numerous times because of the diminution in the contributions of organic matter and the discharge of exchangeable bases by leaching and acid release reactions [61]. Long-term tropical studies have always shown that CEC is optimally produced under conditions in which primary emphasis is on the long-term carbon additions rather than the dependence on mineral additions [62,63]. Taken together, these trends point to the idea that fertiliser regimes comprising either organic fertiliser or regulated N-release cause the soils to be set on the stabilising chemical trajectories, and high-rate N-release regimes cause the pathways connected with acidification, nutrient loss, and declining exchange capacity. This kind of mechanistic knowledge is compulsory to perennial tropical orchards, where the interaction of the belowground resources is the immediate factor for long-term productivity.
4.3. Applied Implications and Scalability for Orchard Management
The high fertiliser × phenophase relationships observed in this study provide a clear, scalable recommendation for nutrient management in orchards, with flowering appearing to be the most sensitive period for intervention. This step exhibited the best multivariate statistical separation (Figure 6b) and the strongest association between soil fertility indicators, leaf nutrient status, and yield-related characteristics (Figure 7b,c), which indicated that fertiliser timing ought to be put on peak reproductive demand and not on even distribution of soil fertility across the seasons. The most robust management options included in the evaluated regimes were slow-release fertiliser (SRT) and bio-organic fertiliser (BFT): the SRT resulted in the maintenance of nutrient availability synchrony and restriction of the accumulation of NO3− during flowering (Figure 2 and Figure 4), which is appropriate in large-scale orchards, whereas the BFT continued to increase SOC, CEC, and retention of base cations across all phenophases (Figure 1 and Figure 3), which is reflected in enhanced multi-nutrient uptake and yield stability. To scale effectively, it is necessary to monitor soil pH trends, accumulation of NO3−-N, CEC and base cation position, nutrient utilisation efficiency, and yield stability routinely, which reveal productivity improvements and future risks. Specifically, the further application of fast-soluble N sources (URT and NT) can facilitate a gradual process of acidification, nitrate accumulation, and loss of base cations (Figure 1, Figure 2, and Figure 6), but the application of organic inputs can threaten nutrient accumulation or elevated EC unless adequately controlled (Figure 3). Even though the current findings effectively show the stage-specific benefits of the SRT and BFT, multi-season monitoring and long-term monitoring are needed to ascertain the sustainability of SOC gains, nutrient balance, and yield stability, which will prove the effectiveness of the long-term efficacy of the fertiliser-mediated C–N synchronisation strategy in the growth of tropical durian orchards.
5. Conclusions
This work demonstrates that fertiliser form possesses a stage and overriding influence on C-N coupling, nutrient retention, and soil chemical stability in tropical durian orchards. The most notable gains in each of the major fertility indicators were in the use of bio-organic fertiliser (BFT), which performed better than mineral fertilisers by very large margins in all the indicators of SOC, CEC, and multi-nutrient availability. Such benefits are good signs of enhanced microbial activity, improved carbon retention, and enhanced buffering against acidification.
Slow-release nitrogen (SRT) provided an extra stability-providing mechanism, which provided a better-regulated N release and fewer nitrifier bursts and high C–N synchrony compared to the most nutrient-blind phenophases. On the other hand, fast solubility sources of nitrogen (URT, NT) caused extreme acidification of the soil, high concentration of NO3−-N, and low concentration of C stabilisation, which confirmed their relative weakness in long-term soil health in highly weathered tropical soils.
Phenology further contributed to the differences in treatments by flowering being the most differentiated stage and nutrient availability and productivity of the nutrient maturity-stage improved downstream. The multivariate analyses showed that the BFT and SRT were equal to optimum fruiting, canopy, and leaf nutrient attributes, and the control was invariably equal to the minimum fertility and poorest performances of the plants.
Taken together, the regimes of fertilisers that increase C–N synchronisation, soil chemical resilience, and nutrient loss mitigation are most beneficial to the production of tropical durians. The best performing bio-organic and controlled-release fertilisers appeared to be scientifically sound and best, and can be applied as a long-term sustainable strategy in order to maximize the production of the orchard and well-being of the soil.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agronomy16030366/s1.
Author Contributions
O.O.N. contributed to the literature review, data compilation, figure preparation, table preparation, writing—original draft, and writing—review and editing. X.L. contributed to the literature review, data compilation, figure preparation, table preparation, writing—original draft, and writing—review and editing. Q.Z. and R.G.N. contributed to the literature review, data compilation, figure preparation, and table preparation. L.C., H.Z., X.Z., Q.L., X.D., J.Z. and A.S.E. contributed to the literature review and data compilation. L.M. contributed to conceptualisation, supervision, funding acquisition, writing—original draft, writing—review and editing, and project administration. All authors contributed to the article and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Hainan Provincial Industrial Technology System for High-quality and Rare Fruits: Durian High-efficiency Cultivation Position (Grant No. HNARS-11-G02), and the Hainan Provincial Key Research and Development Program: Sanya Durian Quality Control Research and Demonstration (Grant No. ZDYF2024XDNY251).
Institutional Review Board Statement
Ethical review and approval were not required for this study.
Data Availability Statement
All data and materials used in this study are included in the article.
Conflicts of Interest
Author Xiangdong Zhang is affiliated with Wanbao Agriculture and Animal Husbandry Group Co., Ltd., Sanya 572025, China, which also developed/supplied the product used in this study. The authors declare that this affiliation did not influence the study design, data collection, analysis, or interpretation of the results. No commercial or financial relationships that could be construed as a potential conflict of interest exist.
References
- Aziz, N.A.; Wong, L.M.; Bhat, R.; Cheng, L.H. The Durian: A Review of Nutritional Properties, Dietary Importance, and Health Benefits. Int. J. Agric. Sustain. 2021, 19, 209–221. [Google Scholar]
- Salakpetch, S.; Turner, D.W. Durian (Durio zibethinus) flower and fruit set in response to paclobutrazol and potassium nitrate. Thai J. Agric. Sci. 2000, 33, 1–12. [Google Scholar]
- Li, Y.; Zhang, J.; Liu, Y.; Wang, H.; Ma, C. Monoculture and long-term cultivation of durian (Durio zibethinus) alter soil chemical properties and microbial community structure. Appl. Soil Ecol. 2021, 168, 104117. [Google Scholar] [CrossRef]
- Mohamed, A.W.; Jamaluddin, A.F.; Zainal, Z. The Sustainability Crisis of Durian Orchards: A Soil Health Perspective. J. Clean. Prod. 2023, 414, 137562. [Google Scholar] [CrossRef]
- Lal, R. The soil–energy–food security nexus in the tropical environment. J. Plant Nutr. Soil Sci. 2020, 183, 14–27. [Google Scholar]
- Chivenge, P.; Vanlauwe, B.; Gentile, R.; Six, J.; Merckx, R. Soil Carbon and Nitrogen Cycling and Ecosystem Service Delivery in a Conservation Agriculture Maize-Legume System. Agron. Sustain. Dev. 2022, 42, 95. [Google Scholar] [CrossRef]
- Schimel, J.P.; Bennett, J. Nitrogen mineralization: Challenges of a changing paradigm. Ecology 2004, 85, 591–602. [Google Scholar] [CrossRef]
- Chapin, F.S.; Matson, P.A.; Vitousek, P.M. Principles of Terrestrial Ecosystem Ecology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Brookshire, E.N.J.; Hedin, L.O.; Newbold, J.D.; Sigman, D.M.; Jackson, J.K. The Nitrogen Paradox in Tropical Forest Ecosystems. Nat. Rev. Earth Environ. 2012, 1, 1–8. [Google Scholar]
- Blagodatskaya, E.; Kuzyakov, Y. Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: Critical review. Biol. Fertil. Soils 2008, 45, 115–131. [Google Scholar] [CrossRef]
- Brahmaprempit, S.; Phattaralerphong, J.; Sdoodee, S.; Sasaki, N. Nutrient budgets and soil chemical property changes in durian (Durio zibethinus) orchards of Chanthaburi Province, Thailand. Int. J. Agric. Sustain. 2020, 18, 471–488. [Google Scholar]
- Jian, S.; Li, J.; Wang, G.; Mayes, M.A.; Dzantor, E.K.; Hui, D. Nitrogen fertilization and soil carbon storage: The role of mineral-associated organic matter. Glob. Change Biol. 2020, 26, 3933–3944. [Google Scholar] [CrossRef]
- Geisseler, D.; Miller, K.S.; Leighton, P.M. Long-term effects of nitrogen fertilization on soil chemical properties, nutrient cycling, and soil organic carbon quality in a temperate grassland. Soil Biol. Biochem. 2022, 174, 108823. [Google Scholar]
- Timilsena, Y.N.; Adhikari, R.; Casey, P.; Muster, T.; Gill, H.; Adhikari, B. Controlled release fertilizers: A review on coating materials and mechanism of release. Plants 2015, 4, 574–622. [Google Scholar]
- Lawrenko, J.A.; Mikkelsen, R.L.; Goyal, S. Polymer-coated controlled release fertilizers: Fundamentals, development and agricultural performance. Adv. Agron. 2020, 164, 69–118. [Google Scholar] [CrossRef]
- Li; Wang, M.; Li, Z.; Li, R.; Zhao, B.; Zhang, F. Polymer-coated urea improves nitrogen use efficiency and grain yield of summer maize in the North China Plain. Field Crops Res. 2022, 286, 108629. [Google Scholar] [CrossRef]
- Yan, Y.; Wang, X.; Li, Y.; Wang, J.; Wang, L.; Zhang, Y.; Wang, E.; Wang, Z. Bio-organic fertilizers stimulate indigenous soil microbiota and change the microbial community structure to enhance soil health and crop yield. Appl. Soil Ecol. 2024, 195, 105245. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps, 4th ed.; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022. [Google Scholar]
- Shamin-Shazwan, K.; Shahari, R.; Aini, C.; Go, R. Propagation methods, agronomic practices and fruit production of Durio zibethinus L. in Malaysia: A review. Malay. Nat. J. 2021, 73, 85–98. [Google Scholar]
- Walter, K.; Don, A.; Tiemeyer, B.; Freibauer, A. Determining Soil Bulk Density for Carbon Stock Calculations: A Systematic Method Comparison. Soil Sci. Soc. Am. J. 2016, 80, 579–591. [Google Scholar] [CrossRef]
- Pansu, M.; Gautheyrou, J. Handbook of Soil Analysis: Mineralogical, Organic and Inorganic Methods; Springer: Berlin/Heidelberg, Germany, 2006; pp. 599–603. [Google Scholar] [CrossRef]
- Rayment, G.E.; Lyons, D.J. Soil Chemical Methods: Australasia; CSIRO Publishing: Melbourne, Australia, 2011. [Google Scholar]
- Walkley, A.; Black, I. An Examination of the Degtjareff Method for Determining Soil Organic Matter and a Proposed Modification of the Chromic Acid Titration Method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Keeney, D.R.; Nelson, D.W. Nitrogen-Inorganic Forms. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1982; pp. 643–698. [Google Scholar] [CrossRef]
- Olsen, S.R.; Sommers, L.E. Phosphorus. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1982; pp. 403–430. [Google Scholar] [CrossRef]
- Thomas, G.W. Exchangeable Cations. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1982; pp. 159–165. [Google Scholar] [CrossRef]
- German, D.P.; Weintraub, M.N.; Grandy, A.S.; Lauber, C.L.; Rinkes, Z.L.; Allison, S.D. Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol. Biochem. 2011, 43, 1387–1397. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Assay of urease activity in soils. Soil Biol. Biochem. 1972, 4, 479–487. [Google Scholar] [CrossRef]
- Gerritse, R.G.; van Dijk, H. Determination of phosphatase activities of soils and animal wastes. Soil Biol. Biochem. 1978, 10, 545–551. [Google Scholar] [CrossRef]
- Wortmann, S.; Kutta, R.J.; Nuernberger, P. Monitoring the photochemistry of a formazan over 15 orders of magnitude in time. Front. Chem. 2022, 10, 983342. [Google Scholar] [CrossRef]
- Slessarev, E.W.; Schwab, V.F.; Cornejo, N.S.; Perez, C.A.; Torn, M.S. Microbial processing of plant litter is associated with contrasting aggregate formation and stabilization in the mineral soil of a tropical montane forest. Glob. Change Biol. 2022, 28, 1118–1135. [Google Scholar] [CrossRef]
- Jian, S.; Li, J.; Wang, J.; Liu, Y.; Kuzyakov, Y.; Razavi, B.S.; Zhu, B. Microbial necromass dominates mineral-associated organic matter in tropical agricultural soils. Soil Biol. Biochem. 2024, 189, 109280. [Google Scholar] [CrossRef]
- Liu, L.; Muneer, M.A.; Zhong, Y.; He, B.; Li, X. Organic farming significantly improves microbial community structure, network complexity, and functional diversity in the Gannan navel orange orchard. BMC Microbiol. 2025, 25, 561. [Google Scholar] [CrossRef]
- Li, Q.; Hu, W.; Li, L.; Li, Y. Interactions between organic matter and Fe oxides at soil micro-interfaces: Quantification, associations, and influencing factors. Sci. Total Environ. 2023, 855, 158710. [Google Scholar] [CrossRef]
- Peng, X.; Yan, X.; Zhou, H.; Zhang, Y.Z.; Sun, H. Assessing the contributions of sesquioxides and soil organic matter to aggregation in an Ultisol under long-term fertilization. Soil Tillage Res. 2015, 146, 89–98. [Google Scholar] [CrossRef]
- Li, X.; Li, Z. Global Trends and Current Advances in Slow/Controlled-Release Fertilizers: A Bibliometric Analysis from 1990 to 2023. Agriculture 2024, 14, 1502. [Google Scholar] [CrossRef]
- Ayiti, O.E.; Babalola, O.O. Factors Influencing Soil Nitrification Process and the Effect on Environment and Health. Front. Sustain. Food Syst. 2022, 6, 821994. [Google Scholar] [CrossRef]
- Wang, J.; Tu, X.; Zhang, H.; Cui, J.; Ni, K.; Chen, J.; Cheng, Y.; Zhang, J.; Chang, S.X. Effects of ammonium-based nitrogen addition on soil nitrification and nitrogen gas emissions depend on fertilizer-induced changes in pH in a tea plantation soil. Sci. Total Environ. 2020, 747, 141340. (In English) [Google Scholar] [CrossRef]
- Bailey, T.; Robinson, N.; Macdonald, B.; McGowan, J.; Weaver, T.; Antille, D.L.; Farrell, M. Opposing patterns of carbon and nitrogen stability in soil organic matter fractions compared to whole soil. Eur. J. Soil Sci. 2024, 75, e13495. [Google Scholar] [CrossRef]
- Laub, M.; Corbeels, M.; Couëdel, A.; Ndungu, S.M.; Mucheru-Muna, M.W.; Mugendi, D.; Necpalova, M.; Waswa, W.; Van de Broek, M.; Vanlauwe, B.; et al. Managing soil organic carbon in tropical agroecosystems: Evidence from four long-term experiments in Kenya. Soil 2023, 9, 301–323. [Google Scholar] [CrossRef]
- Mooshammer, M.; Wanek, W.; Hammerle, I.; Fuchslueger, L.; Hofhansl, F.; Knoltsch, A.; Schnecker, J.; Takriti, M.; Watzka, M.; Wild, B.; et al. Adjustment of microbial nitrogen use efficiency to carbon:nitrogen imbalances regulates soil nitrogen cycling. Nat. Commun. 2014, 5, 3694. [Google Scholar] [CrossRef]
- Shaviv, A.; Raban, S.; Zaidel, E. Advances in controlled-release fertilizers: Mechanisms, formulations and applications. Adv. Agron. 2020, 164, 1–49. [Google Scholar] [CrossRef]
- Murugan, R.; Kumar, S.; Rani, A.; Kumar, P.; Dang, Y.P.; Bell, M.J. Long-term nutrient management in a tropical plantation alters the chemical and microbial community structure of soils. Geoderma 2022, 405, 115398. [Google Scholar] [CrossRef]
- Xu, Q.; Liang, C.; Chen, J.; Li, Y.; Wang, H.; Wu, J.; Jiang, P. Seasonal and interannual variations of soil physicochemical and microbial properties in a chronosequence of Moso bamboo (Phyllostachys edulis) plantations under different fertilization management. Geoderma 2020, 361, 114034. [Google Scholar] [CrossRef]
- Aini, F.Z.; Hashidoko, Y.; Hattori, Y.; Tamura, K.; Melling, L. Ammonium-based fertilizers enhance nitrification and acidification in a tropical oil palm plantation. Soil Sci. Plant Nutr. 2022, 68, 279–290. [Google Scholar]
- Illias, M.K.; Melling, L.; Wasli, M.E.; Husni, M.H.; Yeng, W.S. Long-term acidification of an oil palm plantation soil in Sarawak, Malaysia. Sci. Total Environ. 2020, 703, 134576. [Google Scholar] [CrossRef]
- Shi, R.; Liu, Z.; Li, Y.; Jiang, T.; Xu, M.; Li, J.; Xu, R. Organic amendments increase the soil pH buffering capacity in acidic soils through managing microbial feedback. Geoderma 2022, 420, 115884. [Google Scholar]
- Snoeck, D.; Abdoellah, S.; Kageyama, H.; Afandi, A.; Hartemink, A.E. Soil fertility and nutrient management in perennial crops: A case study of rambutan and citrus in Southeast Asia. Soil Use Manag. 2020, 36, 124–136. [Google Scholar] [CrossRef]
- Liu, S.; Li, Y.; Wang, J.; Chen, Y.; Deng, O.; Zhang, L. Soil pH buffering capacity and nitrogen availability following compost and biochar applications in a tropical orchard soil. Soil Use Manag. 2022, 38, 1215–1229. [Google Scholar] [CrossRef]
- Sayer, E.J.; Lopez-Sangil, L.; Crawford, J.A.; Brechet, L.M.; Birkett, A.J.; Baxendale, C.; Castro, B.; Rodtassana, C.; Kerdraon, D.; Soong, J.L. Plant carbon allocation and microbial processing of soil organic carbon in temperate and tropical forests. Glob. Change Biol. 2021, 27, 3692–3705. [Google Scholar] [CrossRef]
- Liang, C.; Zhu, X.; Zhang, J.; Yuan, Y.; Schädler, M. Root-derived carbon and microbial decomposition drive soil organic carbon dynamics under prolonged fertilization. Soil Biol. Biochem. 2022, 174, 108819. [Google Scholar]
- Wurzburger, N.; Wright, S.J.; Hedin, L.O. Carbon allocation in tropical trees: Reproductive stages drive belowground investment. Ecology 2021, 102, e03305. [Google Scholar] [CrossRef]
- Ge, T.; Li, B.; Zhu, Z.; Hu, Y.; Yuan, H.; Dorodnikov, M.; Jones, D.L.; Wu, J. Long-term fertilization alters the relative importance of nitrate- and carbon-driven priming effects in soil organic matter decomposition. Glob. Change Biol. 2022, 28, 1129–1142. [Google Scholar] [CrossRef]
- Chen, J.; van Groenigen, K.J.; Yang, Y.; Liang, C.; Huang, Y.; Tian, J.; Zhu, A.; Wu, J.; van Groenigen, J.W. Nitrogen fertilization reduces soil organic carbon sequestration in tropical agroforestry systems: A meta-analysis. Glob. Change Biol. 2022, 28, 5256–5272. [Google Scholar] [CrossRef]
- van Lent, J.; Haddix, M.L.; Cotrufo, M.F.; Paul, E.A.; Nogueira, L.; Cerri, C.E.P. Plant- and microbial-derived carbon inputs drive contrasting soil carbon dynamics in tropical agricultural systems. Nat. Geosci. 2024, 17, 45–53. [Google Scholar] [CrossRef]
- Oldfield, E.E.; Possinger, A.R.; Sokol, N.W.; Michalski, G.M.; Grandy, A.S. Evidence for the existence and ecological relevance of fast-cycling mineral-associated organic matter. Commun. Earth Environ. 2025, 6, 690. [Google Scholar] [CrossRef]
- Segnini, A.; de Brogniez, D.; Milori, D.M.B.P.; de Novais, R.F.; de Souza, A.J.; de Almeida, R.F.; de Almeida, E.; de Almeida, L.F.; de Almeida, L.F.; de Almeida, L.F. Soil organic matter is the dominant contributor to cation exchange capacity in highly weathered tropical soils. Geoderma 2022, 408, 115573. [Google Scholar]
- Wang, H.; Zhang, Y.; Zhou, P.; Li, D.; Huang, D.; Wu, J. The quantitative relationship between soil organic carbon and cation exchange capacity in acidic soils under long-term organic fertilization. Soil Tillage Res. 2024, 235, 105894. [Google Scholar] [CrossRef]
- Liu, L.; Wang, J.; Li, Y.; Zhang, S.; He, P.; Xu, X. Organic amendments enhance soil cation exchange capacity through increasing soil organic carbon in tropical agricultural soils. Agric. Ecosyst. Environ. 2023, 356, 108647. [Google Scholar] [CrossRef]
- Chen, J.; Chen, C.; Li, Y.; Wu, L. Mechanisms of humic substances enhancing cation exchange capacity in tropical acidic soils: Role of functional groups and molecular structure. Geoderma 2024, 431, 116382. [Google Scholar] [CrossRef]
- Yang; Wang, L.; Zhang, W.; Wang, H.; Fu, X.; Shen, J. Long-term mineral nitrogen fertilization decreases soil organic carbon and cation exchange capacity in acidic soils by promoting proton release and base cation leaching. Nat. Geosci. 2023, 16, 789–795. [Google Scholar] [CrossRef]
- Powlson, D.S.; Stirling, C.M.; Thierfelder, C.; White, R.P.; Jat, M.L. Organic inputs and reduced tillage enhance cation exchange capacity and carbon sequestration in tropical soils. Soil Tillage Res. 2023, 234, 105838. [Google Scholar] [CrossRef]
- Diekow, J.; Mielniczuk, J.; Knicker, H.; Bayer, C.; Dick, D.; Kögel-Knabner, I. Soil C and N stocks as affected by cropping systems and nitrogen fertilisation in a southern Brazil Acrisol managed under no-tillage for 17 years. Soil Tillage Res. 2005, 81, 87–95. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.