Identification and Evaluation of Diterpenoids from Glandular Trichome Secretions of Air/Sun-Cured Tobacco Germplasm Resources
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Field Trial Design
2.3. Sampling and Treatment
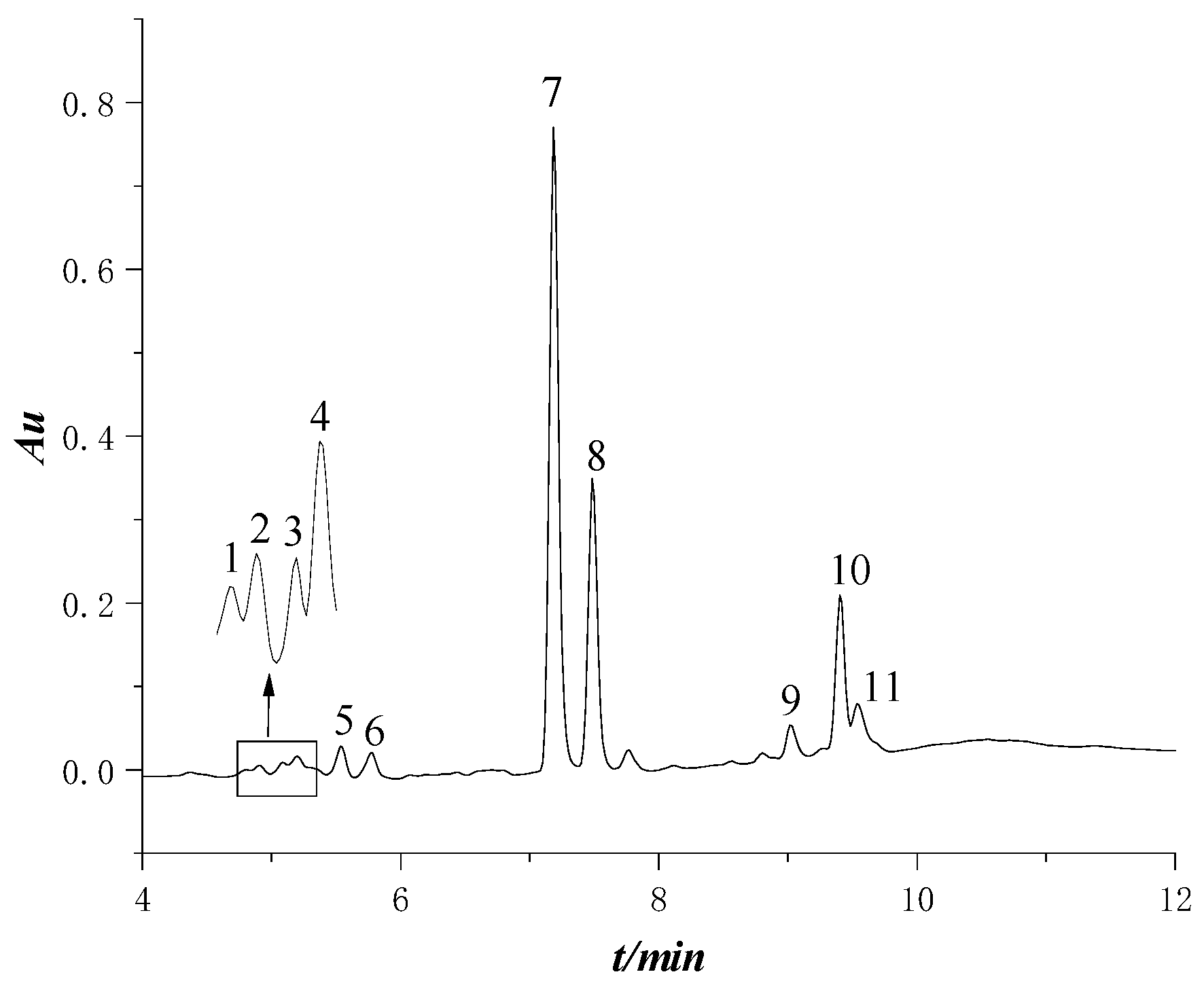
2.4. UPLC Analysis
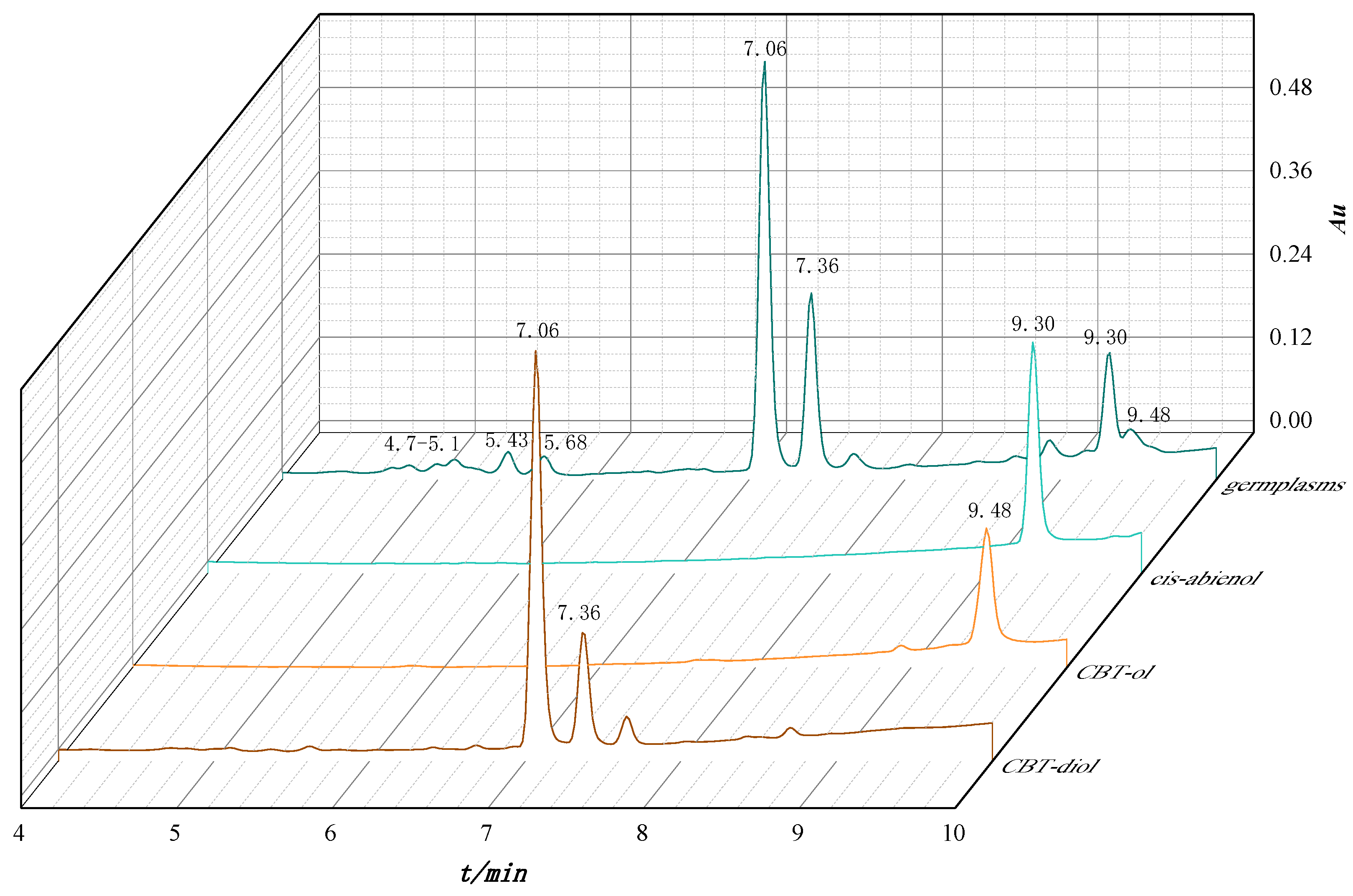
2.5. UPLC-Q-TOF-MS Analysis
2.6. Content Calculation
2.7. Data Analysis
3. Results
3.1. Subsection Diterpenoid Components of Glandular Trichome Secretion
3.2. Correlation Analysis
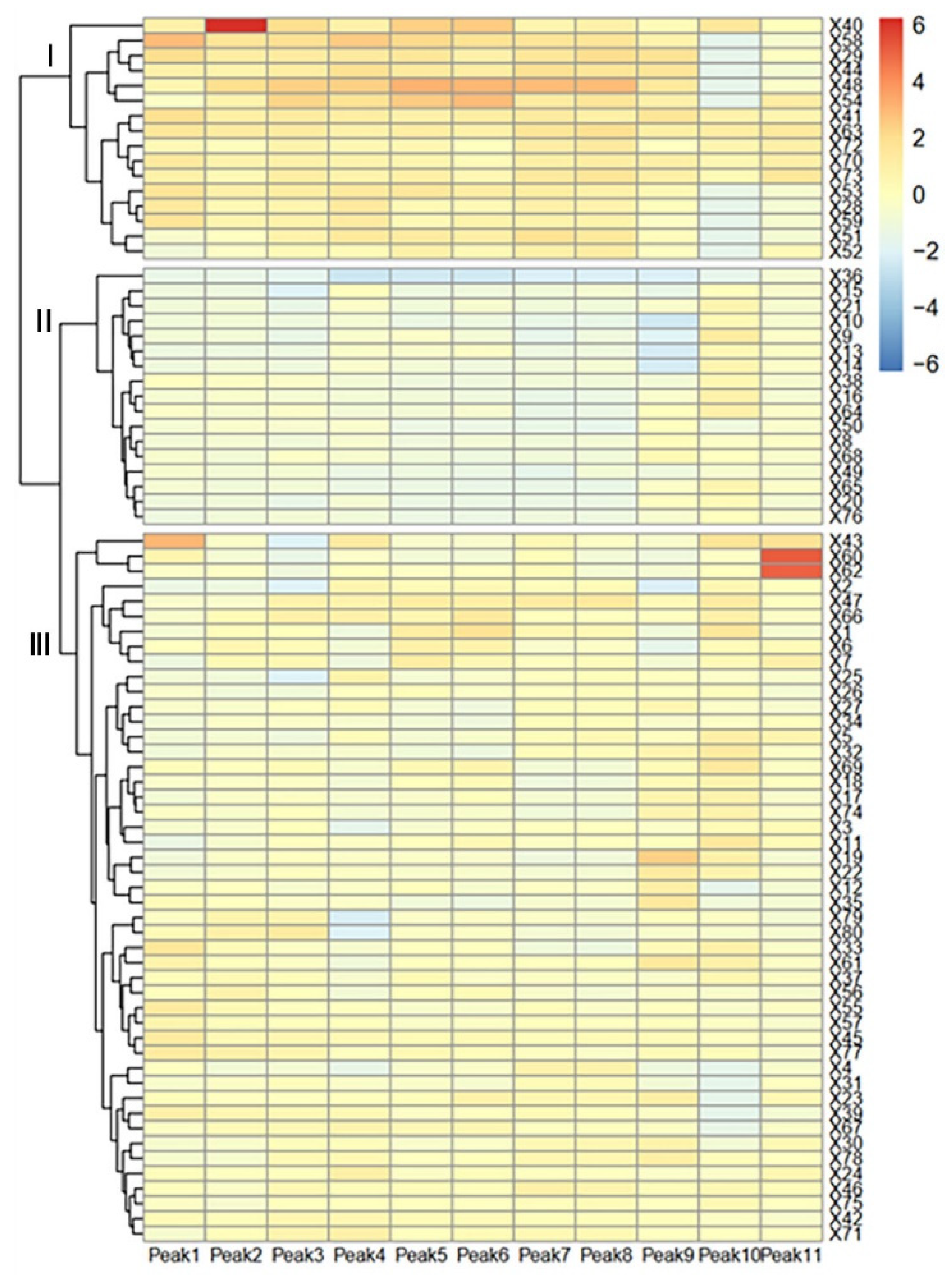
3.3. Cluster Analysis
3.4. Principal Component Analysis
4. Discussion
4.1. Diterpenoid Components in the Glandular Trichome Secretions of Tobacco
4.2. Correlation Analysis of Diterpenoid Components
4.3. Comprehensive Evaluation of Selected Air/Sun-Cured Tobacco Germplasms
4.4. Obtaining Diterpenoid Components
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Severson, R.F.; Arrendale, R.F.; Chortyk, O.T.; Green, C.R.; Thome, F.A.; Stewart, J.L.; Johnson, A.W. Isolation and characterization of the sucrose esters of the cuticular waxes of green tobacco leaf. J. Agric. Food Chem. 1985, 33, 870–875. [Google Scholar] [CrossRef]
- Xu, K.; Du, X.Y.; Ren, X.; Li, X.X.; Li, H.; Fu, X.J.; Wei, X.Y. Structural Modifications and Biological Activities of Natural alpha- and beta-Cembrenediol: A Comprehensive Review. Pharmaceuticals 2022, 15, 601. [Google Scholar] [CrossRef] [PubMed]
- El Sayed, K.A.; Laphookhieo, S.; Baraka, H.N.; Yousaf, M.; Hebert, A.; Bagaley, D.; Rainey, F.A.; Muralidharan, A.; Thomas, S.; Shah, G.V. Biocatalytic and semisynthetic optimization of the anti-invasive tobacco (1S,2E,4R,6R,7E,11E)-2,7,11-cembratriene-4,6-diol. Bioorg. Med. Chem. 2008, 16, 2886–2893. [Google Scholar] [CrossRef] [PubMed]
- Aqil, F.; Zahin, M.; El Sayed, K.A.; Ahmad, I.; Orabi, K.Y.; Arif, J.M. Antimicrobial, antioxidant, and antimutagenic activities of selected marine natural products and tobacco cembranoids. Drug Chem. Toxicol. 2011, 34, 167–179. [Google Scholar] [CrossRef]
- He, L.; Liu, H.; Cheng, C.; Xu, M.; He, L.; Li, L.; Yao, J.; Zhang, W.; Zhai, Z.; Luo, Q.; et al. RNA sequencing reveals transcriptomic changes in tobacco (Nicotiana tabacum) following NtCPS2 knockdown. BMC Genom. 2021, 22, 467. [Google Scholar] [CrossRef]
- Menetrez, M.L.; Spurr, H.W.; Danehower, D.A.; Lawson, D.R. Influence of tobacco leaf surface chemicals on germination of peronospora-tabacina adam sporangia. J. Chem. Ecol. 1990, 16, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Baraka, H.N.; Khanfar, M.A.; Williams, J.C.; El-Giar, E.M.; El Sayed, K.A. Bioactive natural, biocatalytic, and semisynthetic tobacco cembranoids. Planta Med. 2011, 77, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Hailat, M.M.; Ebrahim, H.Y.; Mohyeldin, M.M.; Goda, A.A.; Siddique, A.B.; El Sayed, K.A. The tobacco cembranoid (1S,2E,4S,7E,11E)-2,7,11-cembratriene-4,6-diol as a novel angiogenesis inhibitory lead for the control of breast malignancies. Bioorg. Med. Chem. 2017, 25, 3911–3921. [Google Scholar] [CrossRef]
- Seo, S.; Gomi, K.; Kaku, H.; Abe, H.; Seto, H.; Nakatsu, S.; Neya, M.; Kobayashi, M.; Nakaho, K.; Ichinose, Y.; et al. Identification of natural diterpenes that inhibit bacterial wilt disease in tobacco, tomato and Arabidopsis. Plant Cell Physiol. 2012, 53, 1432–1444. [Google Scholar] [CrossRef] [PubMed]
- Mischko, W.; Hirte, M.; Roehrer, S.; Engelhardt, H.; Mehlmer, N.; Minceva, M.; Brueck, T. Modular biomanufacturing for a sustainable production of terpenoid-based insect deterrents. Green Chem. 2018, 20, 2637–2650. [Google Scholar] [CrossRef]
- Ju, F.; Zhang, H.; Yan, N.; Du, Y.; Zhang, Z.; Fu, Q.; LIU, l.; Liu, J.; Liu, Y. Genetic analysis of cembratrien-diols content in tobacco. Chin. Tob. Sci. 2021, 42, 1–6. [Google Scholar] [CrossRef]
- Ma, X.; Wang, Z.; Yan, X.; Cui, H.; Zhang, H. Comparative studies on secretory glandular trichome morphology and leaf surface chemistry between tobacco varieties. Tob. Sci. Technol. 2021, 54, 10–16. [Google Scholar]
- Huang, M.Y.; Zhang, H.Y.; Wang, Z.J.; Niu, D.X.; Li, Y.H.; Cui, H. Comparative studies of leaf surface chemical biosynthesis in different tobacco cultivars. Acta Physiol. Plant. 2018, 40, 67. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, G.; Zhao, H.; Han, Y.; Liu, X.; Zhang, C.; Li, H. Screening and evaluation of salt and alkali tolerance of rice at tillering stage in cold regions. J. South. Agric. 2021, 52, 28–36. [Google Scholar] [CrossRef]
- Wu, J.; Wang, Y.; Li, K.; Zhou, X.; Li, Z. Evaluation of noodles made under different proofing conditions based on principal component analysis. Food Sci. 2016, 37, 119–123. [Google Scholar]
- Wang, J.; Xu, K.; Zhang, J.; Ren, G.; Yang, X.; Zhang, Z.; Zhang, Y.; Xiao, Y.; Du, Y. Systematic activity-oriented separation and structure-activity relationship of tobacco cembranoids. Ind. Crops Prod. 2021, 173, 114136. [Google Scholar] [CrossRef]
- Wang, E.M.; Wagner, G.J. Elucidation of the functions of genes central to diterpene metabolism in tobacco trichomes using posttranscriptional gene silencing. Planta 2003, 216, 686–691. [Google Scholar] [CrossRef]
- Zhang, Y.; Bian, S.; Liu, X.; Fang, N.; Wang, C.; Liu, Y.; Du, Y.; Timko, M.P.; Zhang, Z.; Zhang, H. Synthesis of cembratriene-ol and cembratriene-diol in yeast via the MVA pathway. Microb. Cell Factories 2021, 20, 29. [Google Scholar] [CrossRef]
- Xu, S.; Han, W.; Cao, K.; Li, B.; Zheng, C.; Xie, K.; Li, W.; He, L. Knockdown of NtCPS2 promotes plant growth and reduces drought tolerance in Nicotiana tabacum. Front. Plant Sci. 2022, 13, 968738. [Google Scholar] [CrossRef]
- Kong, W.; Lv, N.; Luo, D.; Ling, J.; Gao, Q.; Liu, C.; Li, Y.; Liu, X.; Xiang, H.; Mi, Q.; et al. Chromone Derivatives from the Stems of Sichuan Local Sun Cured Tobacco and Their Anti-TMV Activity. Chem. Nat. Compd. 2021, 57, 260–264. [Google Scholar] [CrossRef]
- Wang, D.; Sun, J.; Peng, Y.; Zhang, X.; Li, L.; Song, Y.; Yang, T. Metabolism differences of cembratrien-diols in different tobacco cultivars. Acta Tabacaria Sin. 2014, 20, 63–69. [Google Scholar]
- Chiocchio, I.; Mandrone, M.; Tacchini, M.; Guerrini, A.; Poli, F. Phytochemical profile and in vitro bioactivities of plant-based by-products in view of a potential reuse and valorization. Plants 2023, 12, 795. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Bai, X.; Zhou, Z.; Liu, Y.; Zhong, C.; Feng, J. Synthesis and antifungal activity of beta-amino alcohol derivatives of sclareol. Chin. J. Pestic. Sci. 2023, 25, 586–594. [Google Scholar] [CrossRef]
- Sui, J.; Wang, C.; Liu, X.; Fang, N.; Liu, Y.; Wang, W.; Yan, N.; Zhang, H.-B.; Du, Y.; Liu, X.; et al. Formation of alpha- and beta-Cembratriene-Diols in Tobacco (Nicotiana tabacum L.) Is Regulated by Jasmonate-Signaling Components via Manipulating Multiple Cembranoid Synthetic Genes. Molecules 2018, 23, 2511. [Google Scholar] [CrossRef] [PubMed]
- Atuegwu, N.C.; Mortensen, E.M.; Krishnan-Sarin, S.; Laubenbacher, R.C.; Litt, M.D. Prospective predictors of electronic nicotine delivery system initiation in tobacco naive young adults: A machine learning approach. Prev. Med. Rep. 2023, 32, 102148. [Google Scholar] [CrossRef]
- Oreizi, E.; Rahiminejad, M.R.; Asghari, G. Influence of environment on glandular trichomes and composition of essential oil of Perovskia abrotanoides Karel. Jundishapur J. Nat. Pharm. Prod. 2014, 9, e16432. [Google Scholar] [CrossRef]


| No. | Name | Type | Source | No. | Name | Type | Source |
|---|---|---|---|---|---|---|---|
| X1 | Havana-1 | cigar | introduced | X41 | Tangpeng | sun-cured | local |
| X2 | Havana No. 1 | cigar | introduced | X42 | Jiangyouyan | sun-cured | local |
| X3 | Beinhart 1000-1 | cigar | introduced | X43 | Wushanxiaolanyan | sun-cured | local |
| X4 | Criollo Salteno 11 | cigar | introduced | X44 | Tiebanqing | sun-cured | local |
| X5 | S-2 | cigar | introduced | X45 | Meitanshaiyan | sun-cured | local |
| X6 | Havana IIc | cigar | introduced | X46 | Hefengheiyan | sun-cured | local |
| X7 | Zrenjanin | cigar | introduced | X47 | Liufengmaoba | sun-cured | local |
| X8 | Yinnixuejiabaopi | cigar | introduced | X48 | Jianpingpiaoba | sun-cured | local |
| X9 | 112–117 | cigar | introduced | X49 | Dayeziqingyan | sun-cured | local |
| X10 | Bad Geudertheimer Landsorte | cigar | introduced | X50 | Lichuanmaoyan | sun-cured | local |
| X11 | Begej | cigar | introduced | X51 | Zhushandaliuzi | sun-cured | local |
| X12 | Connecticut Broad Leaf | cigar | introduced | X52 | Fengjiedamaoyan | sun-cured | local |
| X13 | Connecticut Shade | cigar | introduced | X53 | Shai9118 | sun-cured | breeding |
| X14 | E 18 | cigar | introduced | X54 | Shifangpipaliu | sun-cured | local |
| X15 | Geudetthelmex | cigar | introduced | X55 | Shiyan1 | sun-cured | local |
| X16 | Hanica | cigar | introduced | X56 | Kuiliu | sun-cured | local |
| X17 | Havana 211 | cigar | introduced | X57 | Bamaoliu | sun-cured | local |
| X18 | Havana 510 | cigar | introduced | X58 | Mianzhu | sun-cured | local |
| X19 | Manila | cigar | introduced | X59 | Bashan1 | sun-cured | local |
| X20 | Havana | cigar | introduced | X60 | Chongzhoushaiyan2 | sun-cured | local |
| X21 | Tuerqixueji | cigar | introduced | X61 | Quanyan | sun-cured | local |
| X22 | Conn Shade | cigar | introduced | X62 | Zhouyan | sun-cured | local |
| X23 | Dexue 1 | cigar | introduced | X63 | Xuejia5 | sun-cured | introduced |
| X24 | Dexue2 | cigar | introduced | X64 | New Havana | cigar | introduced |
| X25 | Dexue 3 | cigar | introduced | X65 | Comstock Spanish | cigar | introduced |
| X26 | Habana92 | cigar | introduced | X66 | Mont Calme Brun | cigar | introduced |
| X27 | Cubra-Brazil | cigar | introduced | X67 | Trapesond 288 | cigar | introduced |
| X28 | Kangzhoukuoye | cigar | introduced | X68 | CA0705 | cigar | breeding |
| X29 | OLOR | cigar | introduced | X69 | CA0709 | cigar | breeding |
| X30 | Duominijiachangxin | cigar | introduced | X70 | Nilajiagua Changxin | cigar | introduced |
| X31 | Duominijiaduanxin | cigar | introduced | X71 | Besuki | cigar | introduced |
| X32 | MFPP | cigar | introduced | X72 | Ha20 | cigar | introduced |
| X33 | MFZS | cigar | introduced | X73 | Criollo | cigar | introduced |
| X34 | CP2011 | cigar | introduced | X74 | Ha12 | cigar | introduced |
| X35 | MSCA | cigar | introduced | X75 | Ha19 | cigar | introduced |
| X36 | CS0708 | cigar | breeding | X76 | Jilindabaihua | sun-cured | local |
| X37 | Shangzhiyiduohua | sun-cured | local | X77 | Mulengdaqingjin | sun-cured | local |
| X38 | Shandongdaye | sun-cured | local | X78 | Juanyeshaiyan | oriental | local |
| X39 | Xinbinxiaotuanye | sun-cured | local | X79 | Huangmaoyan | sun-cured | local |
| X40 | Liaoduo | sun-cured | local | X80 | Baimaoyan | sun-cured | local |
| Sum of Squares | DOF | Mean Square | F | P | R² | Adjust R² | |
|---|---|---|---|---|---|---|---|
| environment | 6141.664 | 3 | 11.557 | 57.074 | 0.000 ** | 0.999 | 0.998 |
| germplasm resources | 5,897,001.563 | 79 | 74,645.589 | 2081.029 | 0.000 ** | ||
| proper noun error | 8501.085 | 237 | 35.87 | NaN |
| Peak1 | Peak2 | Peak3 | Peak4 | Peak5 | Peak6 | |
| 1 | 94.77 ± 66.30 a | 88.65 ± 63.61 a | 88.85 ± 43.62 a | 182.16 ± 69.50 a | 161.48 ± 65.13 a | 164.45 ± 61.59 b |
| 2 | 75.99 ± 46.16 a | 82.11 ± 53.56 a | 78.30 ± 52.31 a | 159.97 ± 91.35 a | 199.98 ± 105.38 a | 230.60 ± 108.35 a |
| 3 | 83.55 ± 39.96 a | 78.86 ± 41.64 a | 81.51 ± 45.03 a | 166.59 ± 64.95 a | 183.22 ± 86.29 a | 206.17 ± 83.10 ab |
| 4 | 79.96 ± 48.70 a | 74.37 ± 56.96 a | 81.93 ± 79.13 a | 157.86 ± 103.30 a | 192.12 ± 151.52 a | 229.74 ± 148.62 b |
| Peak7 | Peak8 | Peak9 | Peak10 | Peak11 | ||
| 1 | 1674.78 ± 692.89 b | 1236.21 ± 508.28 b | 406.78 ± 106.11 b | 464.46 ± 291.33 b | 112.36 ± 60.67 b | |
| 2 | 2181.81 ± 1163.86 ab | 1629.43 ± 96.75 ab | 950.40 ± 850.20 a | 868.58 ± 763.87 a | 325.97 ± 423.77 a | |
| 3 | 2181.83 ± 1060.16 ab | 1603.29 ± 79.19 ab | 739.22 ± 405.21 a | 807.03 ± 625.70 a | 246.30 ± 198.51 ab | |
| 4 | 2497.54 ± 1793.07 a | 1816.06 ± 272.88 a | 892.48 ± 627.62 a | 799.67 ± 608.21 a | 264.29 ± 219.05 a |
| Fraction | Min. | Max. | Avg. | Median | SD | CV (%) | Skewness | Kurtosis |
|---|---|---|---|---|---|---|---|---|
| μg/g | ||||||||
| Peak1 | 0.00 | 294.39 | 94.77 | 75.45 | 66.30 | 69.96 | 1.00 | 0.51 |
| Peak2 | 0.00 | 486.96 | 88.65 | 75.82 | 63.61 | 71.75 | 3.27 | 18.63 |
| Peak3 | 0.00 | 194.48 | 88.85 | 89.08 | 43.62 | 49.09 | 0.06 | −0.02 |
| Peak4 | 5.73 | 361.71 | 182.16 | 174.24 | 69.50 | 38.15 | 0.19 | 0.21 |
| Peak5 | 5.76 | 369.58 | 161.48 | 152.24 | 65.13 | 40.33 | 0.74 | 0.94 |
| Peak6 | 11.07 | 353.12 | 164.45 | 158.31 | 61.59 | 37.45 | 0.69 | 1.16 |
| Peak7 | 123.10 | 3740.91 | 1674.78 | 1675.95 | 692.89 | 41.37 | 0.29 | −0.04 |
| Peak8 | 92.78 | 2732.21 | 1236.21 | 1227.20 | 508.28 | 41.12 | 0.46 | −0.04 |
| Peak9 | 146.84 | 664.67 | 406.78 | 406.89 | 106.12 | 26.09 | −0.41 | 0.50 |
| Peak10 | 0.00 | 949.81 | 464.46 | 522.56 | 291.33 | 62.72 | −0.37 | −0.99 |
| Peak11 | 0.00 | 946.32 | 112.36 | 64.39 | 160.67 | 143.00 | 3.61 | 15.86 |
| Peak1 | Peak2 | Peak3 | Peak4 | Peak5 | Peak6 | Peak7 | Peak8 | Peak9 | Peak10 | Peak11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Peak1 | 1 | ||||||||||
| Peak2 | 0.58 ** | 1 | |||||||||
| Peak3 | 0.50 ** | 0.75 ** | 1 | ||||||||
| Peak4 | 0.54 ** | 0.42 ** | 0.49 ** | 1 | |||||||
| Peak5 | 0.49 ** | 0.72 ** | 0.81 ** | 0.68 ** | 1 | ||||||
| Peak6 | 0.45 ** | 0.70 ** | 0.77 ** | 0.66 ** | 0.95 ** | 1 | |||||
| Peak7 | 0.54 ** | 0.51 ** | 0.67 ** | 0.76 ** | 0.80 ** | 0.74 ** | 1 | ||||
| Peak8 | 0.46 ** | 0.51 ** | 0.70 ** | 0.75 ** | 0.82 ** | 0.76 ** | 0.97 ** | 1 | |||
| Peak9 | 0.41 ** | 0.38 ** | 0.56 ** | 0.41 ** | 0.41 ** | 0.38 ** | 0.47 ** | 0.46 ** | 1 | ||
| Peak10 | −0.17 | −0.08 | −0.25 * | −0.27 * | −0.17 | −0.12 | −0.32 ** | −0.29 ** | −0.11 | 1 | |
| Peak11 | 0.17 | 0.03 | −0.06 | 0.15 | 0.1 | 0.1 | 0.25 * | 0.14 | −0.04 | 0.14 | 1 |
| Group | Peak1 | Peak2 | Peak3 | Peak4 | Peak5 | Peak6 |
| Group Ⅰ | 165.05 ± 66.56 a | 161.51 ± 94.78 a | 145.97 ± 26.46 a | 275.69 ± 42.77 a | 252.42 ± 53.32 a | 239.46 ± 56.53 a |
| Group II | 36.93 ± 18.78 c | 36.93 ± 18.78 c | 45.75 ± 21.04 c | 129.59 ± 39.36 c | 88.93 ± 29.16 c | 95.87 ± 29.92 c |
| Group III | 90.59 ± 55.66 b | 82.56 ± 32.00 b | 85.00 ± 32.38 b | 169.34 ± 54.11 b | 156.76 ± 34.89 b | 163.72 ± 38.27 b |
| Peak7 | Peak8 | Peak9 | Peak10 | Peak11 | ||
| Group Ⅰ | 2654.41 ± 389.41 a | 1985.34 ± 282.72 a | 489.59 ± 72.14 a | 278.58 ± 364.41 b | 145.78 ± 134.94 a | |
| Group II | 784.44 ± 242.58 c | 647.72 ± 198.25 c | 309.15 ± 107.22 c | 533.73 ± 221.84 a | 39.38 ± 25.83 a | |
| Group III | 1663.33 ± 357.37 b | 1194.05 ± 280.95 b | 413.90 ± 87.66 b | 502.68 ± 264.68 a | 127.37 ± 188.61 a |
| Principal Component | Eigenvalue | Variance Contribution (%) | Cumulative Contribution (%) |
|---|---|---|---|
| 1 | 6.07 | 55.16 | 55.16 |
| 2 | 1.20 | 10.88 | 66.04 |
| 3 | 1.06 | 9.66 | 75.70 |
| 4 | 0.81 | 7.33 | 83.03 |
| 5 | 0.69 | 6.27 | 89.29 |
| 6 | 0.51 | 4.63 | 93.93 |
| 7 | 0.28 | 2.54 | 96.47 |
| 8 | 0.19 | 1.74 | 98.21 |
| 9 | 0.13 | 1.21 | 99.42 |
| 10 | 0.04 | 0.40 | 99.82 |
| 11 | 0.02 | 0.18 | 100.00 |
| Component | Principal Components | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| 1 | 0.66 | 0.10 | 0.05 |
| 2 | 0.75 | −0.05 | 0.43 |
| 3 | 0.86 | −0.23 | 0.24 |
| 4 | 0.79 | 0.10 | −0.29 |
| 5 | 0.93 | 0.04 | 0.12 |
| 6 | 0.89 | 0.06 | 0.16 |
| 7 | 0.90 | 0.13 | −0.28 |
| 8 | 0.90 | 0.04 | −0.23 |
| 9 | 0.58 | −0.21 | 0.17 |
| 10 | −0.29 | 0.52 | 0.70 |
| 11 | 0.13 | 0.89 | −0.22 |
| Tobacco Number | Comprehensive Index Value | Subordinate Function Value | Comprehensive Evaluation (D Value) | Order | ||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | U(x1) | U(x2) | U(x3) | |||
| X1 | 0.21 | 0.47 | 1.55 | 0.49 | 0.33 | 0.57 | 0.48 | 19 |
| X2 | −0.54 | 1.08 | −1.24 | 0.35 | 0.44 | 0.11 | 0.33 | 59 |
| X3 | −0.47 | 0.18 | 0.22 | 0.36 | 0.28 | 0.35 | 0.35 | 52 |
| X4 | −0.28 | −0.71 | −1.63 | 0.40 | 0.12 | 0.04 | 0.31 | 62 |
| X5 | −0.40 | 1.13 | −0.37 | 0.38 | 0.45 | 0.25 | 0.37 | 45 |
| X6 | 0.02 | 0.55 | 0.80 | 0.45 | 0.34 | 0.44 | 0.44 | 28 |
| X7 | 0.01 | 0.71 | 0.55 | 0.45 | 0.37 | 0.40 | 0.43 | 29 |
| X8 | −0.92 | −0.36 | −0.20 | 0.28 | 0.18 | 0.28 | 0.27 | 68 |
| X9 | −1.26 | 0.61 | 0.57 | 0.22 | 0.35 | 0.41 | 0.26 | 70 |
| X10 | −1.53 | −0.01 | −0.12 | 0.17 | 0.24 | 0.29 | 0.20 | 79 |
| X11 | −0.27 | 0.72 | 0.85 | 0.40 | 0.37 | 0.45 | 0.40 | 39 |
| X12 | −0.15 | −1.37 | −0.74 | 0.42 | 0.00 | 0.19 | 0.33 | 58 |
| X13 | −1.17 | 0.41 | −0.27 | 0.24 | 0.32 | 0.27 | 0.25 | 71 |
| X14 | −1.20 | 0.42 | −0.22 | 0.23 | 0.32 | 0.28 | 0.25 | 72 |
| X15 | −1.28 | 0.13 | −1.05 | 0.22 | 0.27 | 0.14 | 0.21 | 76 |
| X16 | −1.15 | −0.23 | 0.94 | 0.24 | 0.20 | 0.47 | 0.26 | 69 |
| X17 | −0.48 | −0.21 | 0.98 | 0.36 | 0.21 | 0.48 | 0.35 | 51 |
| X18 | −0.44 | 0.07 | 1.09 | 0.37 | 0.26 | 0.49 | 0.37 | 46 |
| X19 | −0.43 | −0.78 | 1.51 | 0.37 | 0.11 | 0.56 | 0.36 | 49 |
| X20 | −1.34 | −0.48 | 0.08 | 0.20 | 0.16 | 0.33 | 0.21 | 77 |
| X21 | −1.05 | 0.01 | 0.18 | 0.26 | 0.25 | 0.34 | 0.27 | 67 |
| X22 | −0.41 | −0.31 | 0.70 | 0.38 | 0.19 | 0.43 | 0.36 | 50 |
| X23 | 0.54 | −0.34 | −1.17 | 0.55 | 0.18 | 0.12 | 0.44 | 24 |
| X24 | 0.13 | 0.48 | −0.62 | 0.47 | 0.33 | 0.21 | 0.42 | 33 |
| X25 | −0.55 | −0.06 | −1.38 | 0.35 | 0.23 | 0.08 | 0.30 | 64 |
| X26 | −0.32 | −0.21 | −0.35 | 0.39 | 0.21 | 0.25 | 0.35 | 53 |
| X27 | −0.22 | −0.67 | −0.63 | 0.41 | 0.12 | 0.21 | 0.34 | 56 |
| X28 | 0.95 | −0.99 | −1.17 | 0.62 | 0.07 | 0.12 | 0.48 | 18 |
| X29 | 1.92 | −0.72 | −0.79 | 0.80 | 0.12 | 0.18 | 0.63 | 7 |
| X30 | 0.10 | −0.14 | −1.06 | 0.47 | 0.22 | 0.14 | 0.39 | 43 |
| X31 | −0.13 | −0.72 | −1.39 | 0.43 | 0.12 | 0.08 | 0.34 | 57 |
| X32 | −0.58 | 0.31 | 0.50 | 0.34 | 0.30 | 0.39 | 0.34 | 54 |
| X33 | −0.23 | 0.05 | 1.55 | 0.41 | 0.25 | 0.57 | 0.41 | 36 |
| X34 | −0.50 | −0.06 | −0.65 | 0.36 | 0.23 | 0.20 | 0.32 | 60 |
| X35 | −0.37 | −1.25 | −0.19 | 0.38 | 0.02 | 0.28 | 0.32 | 61 |
| X36 | −2.44 | −1.30 | −1.15 | 0.00 | 0.01 | 0.12 | 0.02 | 80 |
| X37 | −0.10 | 0.07 | 0.77 | 0.43 | 0.26 | 0.44 | 0.41 | 35 |
| X38 | −0.89 | −0.20 | 0.57 | 0.29 | 0.21 | 0.41 | 0.29 | 65 |
| X39 | 0.36 | −1.08 | −0.83 | 0.52 | 0.05 | 0.17 | 0.41 | 37 |
| X40 | 2.24 | 0.48 | 4.14 | 0.86 | 0.33 | 1.00 | 0.80 | 1 |
| X41 | 1.47 | 0.81 | 0.83 | 0.72 | 0.39 | 0.45 | 0.64 | 6 |
| X42 | 0.36 | −0.44 | 0.09 | 0.52 | 0.17 | 0.33 | 0.44 | 23 |
| X43 | −0.02 | 2.94 | −0.32 | 0.45 | 0.77 | 0.26 | 0.47 | 21 |
| X44 | 1.78 | −1.11 | −1.00 | 0.78 | 0.05 | 0.15 | 0.59 | 9 |
| X45 | 0.57 | −0.15 | 0.30 | 0.55 | 0.22 | 0.36 | 0.48 | 17 |
| X46 | 0.31 | 0.38 | −0.22 | 0.51 | 0.31 | 0.28 | 0.45 | 22 |
| X47 | 0.81 | 0.60 | 0.34 | 0.60 | 0.35 | 0.37 | 0.53 | 12 |
| X48 | 2.99 | −0.82 | −0.59 | 1.00 | 0.10 | 0.22 | 0.77 | 2 |
| X49 | −1.26 | −0.79 | −0.38 | 0.22 | 0.10 | 0.25 | 0.21 | 78 |
| X50 | −1.07 | −1.08 | −0.43 | 0.25 | 0.05 | 0.24 | 0.22 | 74 |
| X51 | 1.15 | −0.94 | −1.62 | 0.66 | 0.08 | 0.04 | 0.50 | 15 |
| X52 | 0.57 | −0.34 | −1.60 | 0.55 | 0.18 | 0.05 | 0.44 | 27 |
| X53 | 1.47 | −0.90 | −0.54 | 0.72 | 0.08 | 0.22 | 0.57 | 11 |
| X54 | 2.11 | 0.15 | −0.76 | 0.84 | 0.27 | 0.19 | 0.67 | 4 |
| X55 | 0.04 | −0.27 | −0.11 | 0.46 | 0.20 | 0.29 | 0.40 | 40 |
| X56 | −0.10 | −0.71 | 0.72 | 0.43 | 0.12 | 0.43 | 0.39 | 44 |
| X57 | 0.04 | −0.43 | 0.06 | 0.46 | 0.17 | 0.32 | 0.40 | 41 |
| X58 | 2.40 | −0.83 | −0.52 | 0.89 | 0.10 | 0.23 | 0.69 | 3 |
| X59 | 1.04 | −0.83 | −0.98 | 0.64 | 0.10 | 0.15 | 0.50 | 14 |
| X60 | −0.57 | 4.24 | −1.88 | 0.35 | 1.00 | 0.00 | 0.40 | 42 |
| X61 | −0.01 | 0.07 | 1.18 | 0.45 | 0.26 | 0.51 | 0.43 | 32 |
| X62 | −0.26 | 4.04 | −1.45 | 0.40 | 0.97 | 0.07 | 0.44 | 25 |
| X63 | 1.59 | 1.59 | 0.76 | 0.74 | 0.53 | 0.44 | 0.67 | 5 |
| X64 | −0.92 | −0.10 | 1.09 | 0.28 | 0.23 | 0.49 | 0.30 | 63 |
| X65 | −1.40 | −0.17 | 0.57 | 0.19 | 0.21 | 0.41 | 0.22 | 73 |
| X66 | 0.50 | 0.23 | 1.01 | 0.54 | 0.28 | 0.48 | 0.50 | 16 |
| X67 | 0.36 | −0.90 | −0.75 | 0.52 | 0.08 | 0.19 | 0.41 | 34 |
| X68 | −0.84 | −0.65 | 0.02 | 0.30 | 0.13 | 0.32 | 0.27 | 66 |
| X69 | −0.12 | 0.28 | 1.76 | 0.43 | 0.29 | 0.61 | 0.43 | 30 |
| X70 | 1.08 | 0.98 | 0.35 | 0.65 | 0.42 | 0.37 | 0.58 | 10 |
| X71 | 0.33 | −0.07 | −0.31 | 0.51 | 0.23 | 0.26 | 0.44 | 26 |
| X72 | 0.74 | 1.27 | −0.18 | 0.59 | 0.47 | 0.28 | 0.53 | 13 |
| X73 | 1.26 | 1.38 | −0.11 | 0.68 | 0.49 | 0.29 | 0.61 | 8 |
| X74 | −0.57 | −0.21 | 1.05 | 0.35 | 0.21 | 0.49 | 0.34 | 55 |
| X75 | −0.03 | 0.18 | −0.01 | 0.44 | 0.28 | 0.31 | 0.40 | 38 |
| X76 | −1.26 | −0.47 | −0.14 | 0.22 | 0.16 | 0.29 | 0.22 | 75 |
| X77 | 0.41 | −0.30 | 1.01 | 0.52 | 0.19 | 0.48 | 0.47 | 20 |
| X78 | 0.26 | −0.18 | −0.18 | 0.50 | 0.21 | 0.28 | 0.43 | 31 |
| X79 | −0.33 | −1.02 | 1.25 | 0.39 | 0.06 | 0.52 | 0.36 | 48 |
| X80 | −0.25 | −1.15 | 1.34 | 0.40 | 0.04 | 0.53 | 0.37 | 47 |
| No. | Peak1 | Peak2 | Peak3 | Peak4 | Peak5 | Peak6 | Peak7 | Peak8 | Peak9 | Peak10 | Peak11 | Ranking |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| X40 | 160.20 | 486.96 | 179.86 | 245.14 | 323.25 | 322.56 | 2138.43 | 1712.40 | 434.73 | 909.92 | 126.82 | 1 |
| X48 | 128.41 | 219.42 | 194.48 | 352.08 | 369.58 | 353.12 | 3740.91 | 2732.21 | 557.31 | 3.23 | 55.11 | 2 |
| X58 | 288.94 | 194.94 | 174.88 | 361.71 | 302.47 | 280.04 | 2878.61 | 2092.68 | 495.61 | 4.60 | 22.40 | 3 |
| X54 | 86.06 | 144.31 | 192.30 | 307.94 | 324.69 | 342.00 | 2605.10 | 2100.89 | 499.96 | 3.95 | 309.26 | 4 |
| X63 | 197.90 | 174.59 | 148.02 | 243.57 | 237.69 | 222.59 | 2852.54 | 2247.44 | 522.27 | 812.95 | 339.22 | 5 |
| X41 | 219.75 | 152.43 | 144.94 | 265.80 | 220.59 | 234.82 | 2577.59 | 1801.52 | 590.46 | 703.19 | 224.92 | 6 |
| X29 | 233.20 | 164.64 | 147.73 | 284.44 | 264.25 | 222.40 | 2786.32 | 2255.71 | 598.34 | 0.00 | 104.58 | 7 |
| X73 | 158.33 | 115.06 | 139.30 | 242.54 | 229.95 | 202.64 | 2704.66 | 2067.63 | 521.88 | 609.14 | 363.39 | 8 |
| X44 | 155.15 | 139.13 | 140.84 | 313.10 | 258.68 | 240.32 | 3027.85 | 2091.26 | 588.76 | 14.04 | 6.58 | 9 |
| X70 | 192.57 | 140.21 | 131.23 | 241.74 | 204.42 | 181.16 | 2371.70 | 1864.34 | 522.64 | 644.50 | 274.78 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, C.; Liu, J.; Du, Y.; Han, X.; Zhang, H.; Fu, Q.; Zhang, J.; Liu, Y. Identification and Evaluation of Diterpenoids from Glandular Trichome Secretions of Air/Sun-Cured Tobacco Germplasm Resources. Agronomy 2023, 13, 2433. https://doi.org/10.3390/agronomy13092433
Qi C, Liu J, Du Y, Han X, Zhang H, Fu Q, Zhang J, Liu Y. Identification and Evaluation of Diterpenoids from Glandular Trichome Secretions of Air/Sun-Cured Tobacco Germplasm Resources. Agronomy. 2023; 13(9):2433. https://doi.org/10.3390/agronomy13092433
Chicago/Turabian StyleQi, Chaofan, Jing Liu, Yongmei Du, Xiao Han, Hongbo Zhang, Qiujuan Fu, Jianhui Zhang, and Yanhua Liu. 2023. "Identification and Evaluation of Diterpenoids from Glandular Trichome Secretions of Air/Sun-Cured Tobacco Germplasm Resources" Agronomy 13, no. 9: 2433. https://doi.org/10.3390/agronomy13092433
APA StyleQi, C., Liu, J., Du, Y., Han, X., Zhang, H., Fu, Q., Zhang, J., & Liu, Y. (2023). Identification and Evaluation of Diterpenoids from Glandular Trichome Secretions of Air/Sun-Cured Tobacco Germplasm Resources. Agronomy, 13(9), 2433. https://doi.org/10.3390/agronomy13092433








