Management of Low Rectal Cancer Complicating Ulcerative Colitis: Proposal of a Treatment Algorithm
Abstract
Simple Summary
Abstract
1. Introduction
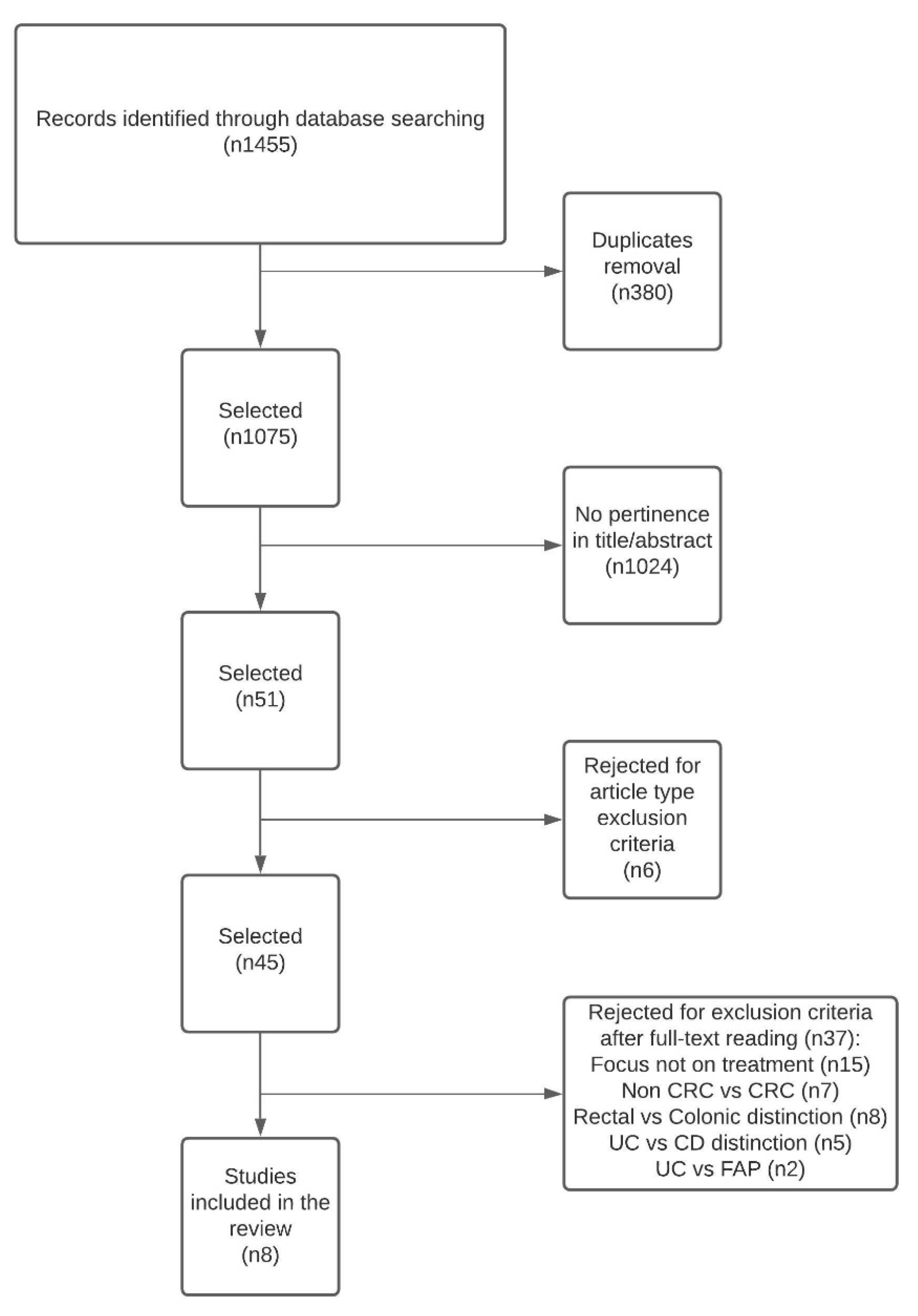
2. Methods
3. Results of Literature Search
4. Treatment of Rectal Adenocarcinoma Complicating Ulcerative Colitis
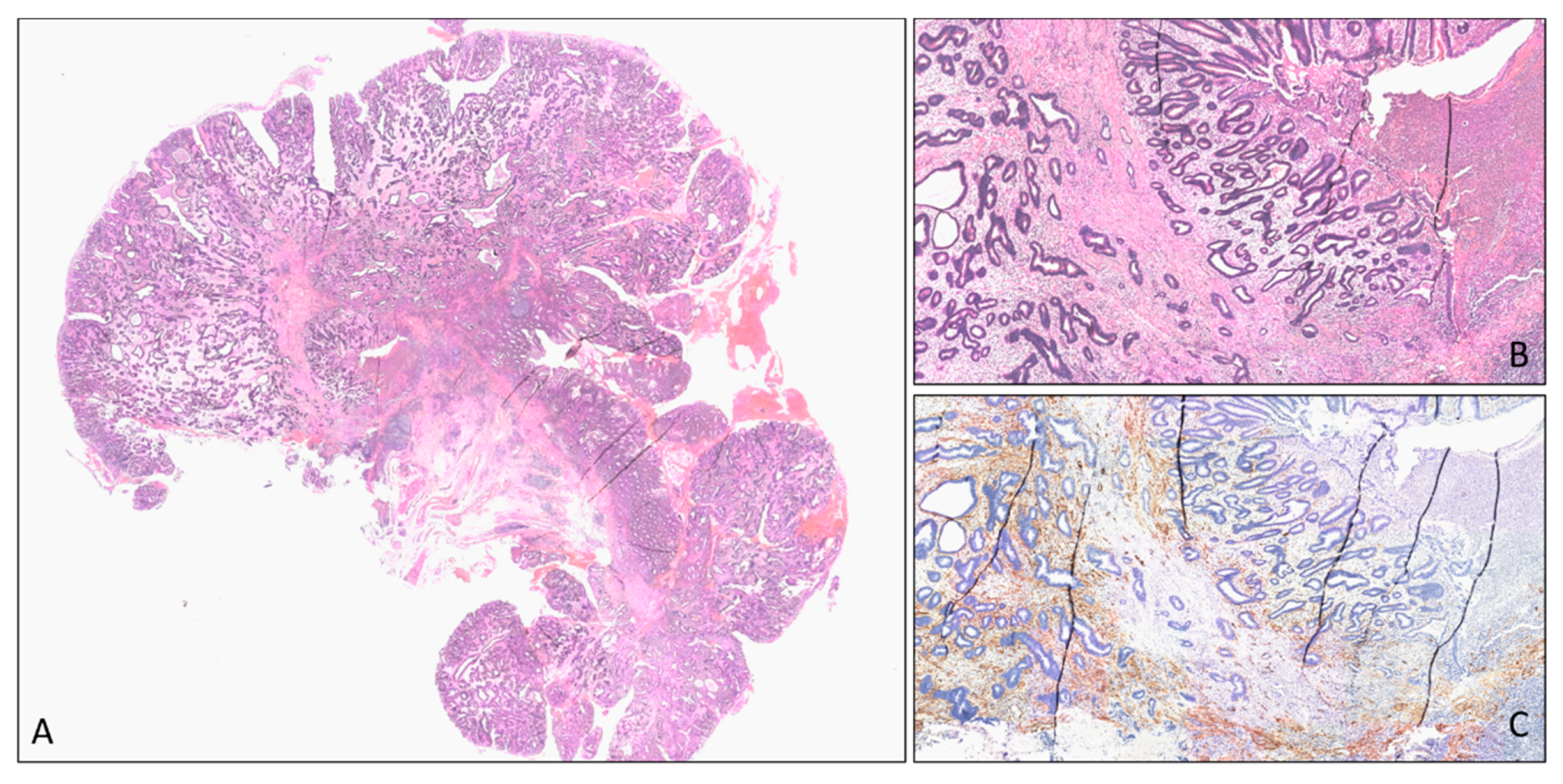
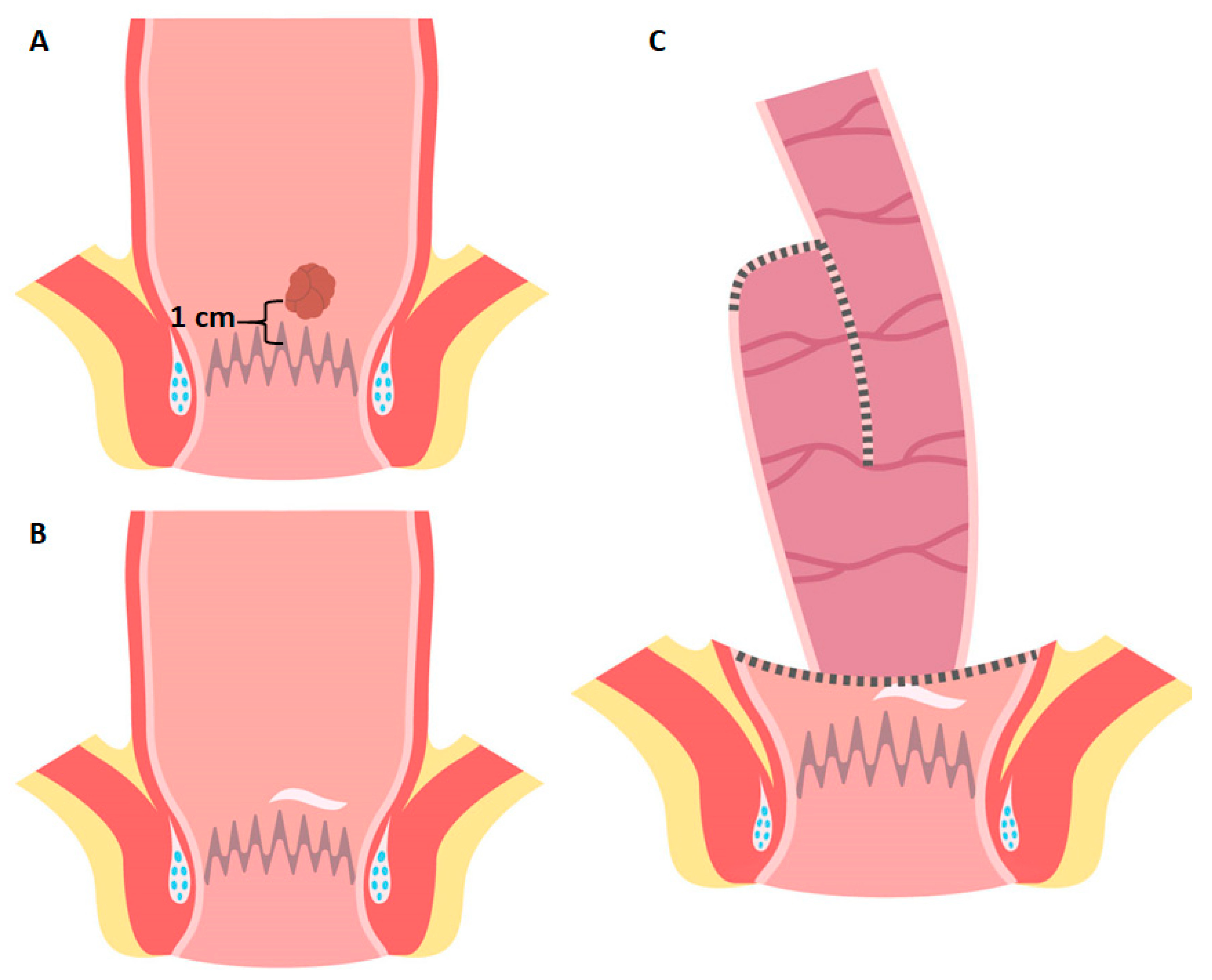
4.1. Low Rectal Cancer
4.2. Ultra-Low Rectal Cancer
4.3. Early Stage Ultra-Low Rectal Cancer
5. Proposal of a Treatment Algorithm
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jess, T.; Simonsen, J.; Jørgensen, K.T.; Pedersen, B.O.V.; Nielsen, N.M.; Frisch, M. Decreasing Risk of Colorectal Cancer in Patients with Inflammatory Bowel Disease Over 30 Years. Gastroenterol. Clin. Biol. 2012, 143, 375–381. [Google Scholar] [CrossRef]
- Selinger, C.P.; Andrews, J.M.; Titman, A.; Norton, I.; Jones, D.B.; McDonald, C.; Barr, G.; Selby, W.; Leong, R.W. Long-term Follow-up Reveals Low Incidence of Colorectal Cancer, but Frequent Need for Resection, Among Australian Patients with Inflammatory Bowel Disease. Clin. Gastroenterol. Hepatol. 2014, 12, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Eaden, J.A.; Abrams, K.R.; Mayberry, J.F. The risk of colorectal cancer in ulcerative colitis: A meta-analysis. Gut 2001, 48, 526–535. [Google Scholar] [CrossRef] [PubMed]
- OCEBM Levels of Evidence Working Group. 2011. Available online: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence (accessed on 9 January 2021).
- Shimizu, S.; Myojo, S.; Nagashima, M.; Okuyama, Y.; Sugeta, N.; Sakamoto, S. A patient with rectal cancer associated with ulcerative colitis in whom endoscopic ultrasonography was useful for diagnosis. J. Gastroenterol. 1999, 34, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Remzi, F.H.; Preen, M. Rectal cancer and ulcerative colitis: Does it change the therapeutic approach? Colorectal Dis. 2003, 5, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Gorfine, S.R.; Harris, M.T.; Bub, D.S.; Bauer, J.J. Restorative Proctocolectomy for Ulcerative Colitis Complicated by Colorectal Cancer. Dis. Colon Rectum 2004, 47, 1377–1385. [Google Scholar] [CrossRef]
- Zmora, O.; Spector, D.; Dotan, I.; Klausner, J.M.; Rabau, M.; Tulchinsky, H. Is stapled ileal pouch anal anastomosis a safe option in ulcerative colitis patients with dysplasia or cancer? Int. J. Colorectal Dis. 2009, 24, 1181–1186. [Google Scholar] [CrossRef]
- Merchea, A.; Wolff, B.G.; Dozois, E.J.; Abdelsattar, Z.M.; Harmsen, W.S.; Larson, D.W. Clinical Features and Oncologic Outcomes in Patients with Rectal Cancer and Ulcerative Colitis. Dis. Colon Rectum 2012, 55, 881–885. [Google Scholar] [CrossRef]
- Inoue, Y.; Araki, T.; Okugawa, Y.; Kawamoto, A.; Hiro, J.; Toiyama, Y.; Tanaka, K.; Uchida, K.; Mohri, Y.; Kusunoki, M. Chemoradiotherapy followed by restorative proctocolectomy with partial intersphincteric resection for advanced rectal cancer associated with ulcerative colitis: Report of a case. Surg. Today 2013, 44, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, K.; Nanashima, A.; Ikeda, T.; Minami, S. A case of concomitant colitic cancer and intrahepatic cholangiocarcinoma during follow-up for ulcerative colitis. Clin. J. Gastroenterol. 2017. [Google Scholar] [CrossRef]
- Hotta, S.; Shimada, Y.; Nakano, M.; Yamada, S.; Abe, K.; Oyanagi, H.; Yagi, R.; Tajima, Y.; Nakano, M.; Kameyama, H.; et al. Feasibility of restorative proctocolectomy in patients with ulcerative colitis-associated lower rectal cancer: A retrospective study. Asian J. Surg. 2019, 42, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Pellino, G.; Keller, D.S.; Sampietro, G.M.; Carvello, M.; Celentano, V.; Coco, C.; Colombo, F.; Geccherle, A.; Luglio, G.; Rottoli, M.; et al. Inflammatory bowel disease position statement of the Italian Society of Colorectal Surgery (SICCR): Ulcerative colitis. Tech. Coloproctol. 2020, 24, 397–419. [Google Scholar] [CrossRef] [PubMed]
- Pellino, G.; Keller, D.S.; Sampietro, G.M.; Annese, V.; Carvello, M.; Celentano, V.; Coco, C.; Colombo, F.; Cracco, N.; Di Candido, F.; et al. Inflammatory bowel disease (IBD) position statement of the Italian Society of Colorectal Surgery (SICCR): General principles of IBD management. Tech. Coloproctol. 2020, 24, 105–126. [Google Scholar] [CrossRef]
- Ullman, T.; Croog, V.; Harpaz, N.; Sachar, D.; Itzkowitz, S. Progression of flat low-grade dysplasia to advanced neoplasia in patients with ulcerative colitis. Gastroenterology 2003, 125, 1311–1319. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.A.; Pemberton, J.H.; Carpenter, H.A.; Levin, K.E.; Schroeder, K.W.; Welling, D.R.; Spencer, M.P.; Zinsmeister, A.R. Dysplasia in Chronic Ulcerative Colitis: Implications for Colono-scopic Surveillance. Dis. Colon Rectum. 1992, 35, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, C.N.; Shanahan, F.; Weinstein, W.M. Articles Are we telling patients the truth about surveillance in ulcerative colitis? Colonoscopy. Lancet 1994, 99, 71–74. [Google Scholar]
- Connell, W.R.; Lennard-Jones, J.E.; Williams, C.B.; Talbot, I.C.; Price, A.B.; Wilkinson, K.H. Factors affecting the outcome of endoscopic surveillance for cancer in ulcerative colitis. Gastroenterology 1994, 107, 934–944. [Google Scholar] [CrossRef]
- Rutter, M.D.; Saunders, B.P.; Wilkinson, K.H.; Rumbles, S.; Schofield, G.; Kamm, M.A.; Williams, C.B.; Price, A.B.; Talbot, I.C.; Forbes, A. Thirty-Year Analysis of a Colonoscopic Surveillance Program for Neoplasia in Ulcerative Colitis. Gastroenterology 2006, 130, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Frontali, A.; Cohen, L.; Bridoux, V.; Myrelid, P.; Sica, G.; Poggioli, G.; Espin, E.; Beyer-Berjot, L.; Laharie, D.; Spinelli, A.; et al. Segmental Colectomy for Ulcerative Colitis: Is There a Place in Selected Patients Without Active Colitis? An International Multicentric Retrospective Study in 72 Patients. J. Crohns Coliti 2020, 14, 1687–1692. [Google Scholar] [CrossRef]
- Khan, N.; Cole, E.; Shah, Y.; Paulson, E.C. Segmental resection is a safe oncologic alternative to total proctocolectomy in elderly patients with ulcerative colitis and malignancy. Colorectal Dis. 2017. [Google Scholar] [CrossRef]
- Caini, S.; Bagnoli, S.; Palli, D.; Saieva, C.; Ceroti, M.; Bendinelli, B.; Assedi, M.; Masala, G. Total and cancer mortality in a cohort of ulcerative colitis and Crohn’s disease patients: The Florence inflammatory bowel disease study, 1978–2010. Dig. Liver Dis. 2016, 48, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Glynne-Jones, R.; Wyrwicz, L.; Tiret, E.; Brown, G.; Rödel, C.; Cervantes, A.; Arnold, D. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv22–iv40. [Google Scholar] [CrossRef] [PubMed]
- Sica, G.S.; Biancone, L. Surgery for inflammatory bowel disease in the era of laparoscopy. World J. Gastroenterol. 2013, 19, 2445–2448. [Google Scholar] [CrossRef]
- Radice, E.; Nelson, H.; Devine, R.M.; Dozois, R.R.; Nivatvongs, S.; Pemberton, J.H.; Wolff, B.G.; Fozard, B.J.; Ilstrup, D. Ileal Pouch-Anal Anastomosis in Patients with Colorectal Cancer Long-Term Functional and Oncologic Outcomes. Dis Colon Rectum 1994, 41, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Monson, J.R.T.; Weiser, M.R.; Buie, W.D.; Chang, G.J.; Rafferty, J.; Guillem, J.; Boushey, R.; Feingold, D.; Fleshner, P.; Genua, J.; et al. Practice Parameters for the Management of Rectal Cancer (Revised). Dis. Colon Rectum 2013, 56, 535–550. [Google Scholar] [CrossRef]
- Song, D.Y.; Lawrie, W.T.; Abrams, R.A.; Kafonek, D.R.; Bayless, T.M.; Welsh, J.S.; DeWeese, T.L. Acute and late Radiotherapy toxicity in patients with inflammatory bowel disease Patient characteristics. Int. J. Radiat. Oncol. 2001, 51, 455–459. [Google Scholar] [CrossRef]
- Green, S.; Stock, R.G.; Greenstein, A.J. Rectal cancer and inflammatory bowel disease: Natural history and implications for radiation therapy. Int. J. Radiat. Oncol. 1999, 44, 835–840. [Google Scholar] [CrossRef]
- Spinelli, A.; Carvello, M.; Kotze, P.G.; Maroli, A.; Montroni, I.; Montorsi, M.; Buchs, N.C.; Ris, F. Ileal pouch–anal anastomosis with fluorescence angiography: A case—Matched study. Colorectal Dis. 2019, 21, 827–832. [Google Scholar] [CrossRef]
- Øresland, T.; Bemelman, W.A.; Sampietro, G.M.; Spinelli, A.; Windsor, A.; Ferrante, M.; Marteau, P.; Zmora, O.; Kotze, P.G.; Espin-Basany, E.; et al. European evidence based consensus on surgery for ulcerative colitis. J. Crohn’s Colitis 2015, 9, 4–25. [Google Scholar] [CrossRef] [PubMed]
- Schinzari, F.; Armuzzi, A.; De Pascalis, B.; Mores, N.; Tesauro, M.; Melina, D.; Cardillo, C. Tumor Necrosis Factor-α Antagonism Improves Endothelial Dysfunction in Patients with Crohn’s Disease. Clin. Pharmacol. Ther. 2008, 83, 70–76. [Google Scholar] [CrossRef]
- Formica, V.; Luccchetti, J.; Cunningham, D.; Smyth, E.C.; Ferroni, P.; Nardecchia, A.; Tesauro, M.; Cereda, V.; Guadagni, F.; Roselli, M. Systemic inflammation, as measured by the neutrophil/lymphocyte ratio, may have differential prognostic impact before and during treatment with fluorouracil, irinotecan and bevacizumab in metastatic colorectal cancer patients. Med. Oncol. 2014, 31, 166. [Google Scholar] [CrossRef] [PubMed]
- Wolmark, N.; Fisher, B. An analysis of survival and treatment failure following abdominoperineal and sphincter-saving resection in Dukes’ B and C rectal carcinoma. Ann. Surg. 1986, 4, 480–489. [Google Scholar] [CrossRef]
- Williams, N.S.; Dixon, M.F.; Johnston, D. Reappraisal of the 5 centimetre rule of distal excision for carcinoma of the rectum: A study of distal intramural spread and of patients’ survival. Br. J. Surg. 1983, 70, 150–154. [Google Scholar] [CrossRef]
- Guillem, J.G.; Chessin, D.B.; Shia, J.; Suriawinata, A.; Riedel, E.; Moore, H.G.; Minsky, B.D.; Wong, W.D. A Prospective Pathologic Analysis Using Whole-Mount Sections of Rectal Cancer Following Preoperative Combined Modality Therapy. Ann. Surg. 2007, 245, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Kuvshinoff, B.; Maghfoor, I.; Miedema, B.; Bryer, M.; Westgate, S.; Wilkes, J.; Ota, D. Distal Margin Requirements After Preoperative Chemoradiotherapy for Distal Rectal Carcinomas: Are <1 cm Distal Margins Sufficient? Ann. Surg. Oncol. 2001, 8, 163–169. [Google Scholar] [PubMed]
- Leo, E.; Belli, F.; Miceli, R.; Mariani, L.; Gallino, G.; Battaglia, L.; Vannelli, A.; Andreola, S. Distal clearance margin of 1 cm or less: A safe distance in lower rectum cancer surgery. Int. J. Colorectal Dis. 2008, 24, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Franzè, E.; Monteleone, I.; Cupi, M.L.; Mancia, P.; Caprioli, F.; Marafini, I.; Colantoni, A.; Ortenzi, A.; Laudisi, F.; Sica, G.; et al. Interleukin-34 sustains inflammatory pathways in the gut. Clin. Sci. 2015, 129, 271–280. [Google Scholar] [CrossRef]
- Marafini, I.; Monteleone, I.; Di Fusco, D.; Cupi, M.L.; Paoluzi, O.A.; Colantoni, A.; Ortenzi, A.; Izzo, R.; Vita, S.; De Luca, E.; et al. TNF-α Producing Innate Lymphoid Cells (ILCs) Are Increased in Active Celiac Disease and Contribute to Promote Intestinal Atrophy in Mice. PLoS ONE 2015, 10, e0126291. [Google Scholar] [CrossRef]
- Cupi, M.L.; Sarra, M.; Marafini, I.; Monteleone, I.; Franzè, E.; Ortenzi, A.; Colantoni, A.; Sica, G.; Sileri, P.; Rosado, M.M.; et al. Plasma Cells in the Mucosa of Patients with Inflammatory Bowel Disease Produce Granzyme B and Possess Cytotoxic Activities. J. Immunol. 2014, 192, 6083–6091. [Google Scholar] [CrossRef]
- Sileri, P.; Sica, G.; Gentileschi, P.; Venza, M.; Manzelli, A.; Palmieri, G.; Spagnoli, L.; Testa, G.; Benedetti, E.; Gaspari, A. Ischemic preconditioning protects intestine from prolonged ischemia. Transplant. Proc. 2004, 36, 283–285. [Google Scholar] [CrossRef]
- Franzè, E.; Dinallo, V.; Rizzo, A.; Giovangiulio, M.D.; Bevivino, G.; Stolfi, C.C.F.; Colantoni, A.; Ortenzi, A.; Grazia, A.D.; Sica, G.; et al. Interleukin-34 sustains pro-tumorigenic signals in colon cancer tissue. Oncotarget 2017, 9, 3432–3445. [Google Scholar] [CrossRef]
- Fina, D.; Franze, E.; Rovedatti, L.; Corazza, G.R.; Biancone, L.; Sileri, P.P.; Sica, G.; Macdonald, T.T.; Pallone, F.; Di Sabatino, A.; et al. Interleukin-25 production is differently regulated by TNF-α and TGF-β1 in the human gut. Mucosal Immunol. 2010, 4, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.T.; Heneghan, H.M.; Winter, D.C. Systematic review of outcomes after intersphincteric resection for low rectal cancer. Br. J. Surg. 2012, 99, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Saiki, Y.; Takano, S.; Iwamoto, K.; Tanaka, M.; Fukunaga, M.; Noguchi, T.; Nakamura, Y.; Hisano, S.; Fukami, K.; et al. Long-term results of intersphincteric resection for low rectal cancer in Japan. Surg. Today 2019, 49, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Fazio, V.W.; Kiran, R.P.; Remzi, F.H.; Coffey, J.C.; Heneghan, H.M.; Kirat, H.T.; Manilich, E.; Shen, B.; Martin, S.T. Ileal Pouch Anal Anastomosis. Ann. Surg. 2013, 257, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Biancone, L.; Onali, S.; Calabrese, E.; Petruzziello, C.; Zorzi, F.; Condino, G.; Sica, G.S.; Pallone, F. Non-invasive techniques for assessing postoperative recurrence in Crohn’s disease. Dig. Liver Dis. 2008, 40, 265–270. [Google Scholar] [CrossRef]
- Onali, S.; Calabrese, E.; Petruzziello, C.; Zorzi, F.; Sica, G.; Lolli, E.; Ascolani, M.; Condino, G.; Pallone, F.; Biancone, L. Endoscopic vs ultrasonographic findings related to Crohn’s Disease recurrence: A prospective longitudinal study at 3 years. J. Crohns Coliti 2010, 4, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Rouanet, P.; Rivoire, M.; Gourgou, S.; Lelong, B.; Rullier, E.; Jafari, M.; Mineur, L.; Pocard, M.; Faucheron, J.L.; Dravet, F.; et al. Sphincter—Saving surgery for ultra—Low rectal carcinoma initially indicated for abdominoperineal resection: Is it safe on a longterm followup? J. Surg. Oncol. 2021, 123, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Park, S.Y.; Kim, H.J.; Cho, S.H.; Kwak, S.G.; Choi, G.-S. Long-term Oncologic Outcomes After Neoadjuvant Chemoradiation Followed by Intersphincteric Resection with Coloanal Anastomosis for Locally Advanced Low Rectal Cancer. Dis. Colon Rectum 2019, 62, 408–416. [Google Scholar] [CrossRef]
- Luna-Pérez, P.; Rodríguez-Ramírez, S.; Hernández-Pacheco, F.; De La Barrera, M.G.; Fernández, R.; Labastida, S. Anal sphincter preservation in locally advanced low rectal adenocarcinoma after preoperative chemoradiation therapy and coloanal anastomosis. J. Surg. Oncol. 2002, 82, 3–9. [Google Scholar] [CrossRef]
- Lorenzi, B.; Brading, A.F.; Martellucci, J.; Cetta, F.; Mortensen, N.J.M. Short-Term Effects of Neoadjuvant Chemoradiotherapy on Internal Anal Sphincter Function. Dis. Colon Rectum 2012, 55, 465–472. [Google Scholar] [CrossRef]
- da Silva, G.M.; Berho, M.; Wexner, S.D.; Efron, J.; Weiss, E.; Nogueras, J.; Verneva, A.; Connor, J.; Gervaz, P. Histologic Analysis of the Irradiated Anal Sphincter. Dis. Colon Rectum 2003, 46, 1492–1497. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Saito, N.; Sugito, M.; Kobayashi, A.; Nishizawa, Y.; Tsunoda, Y. Analysis of Clinical Factors Associated with Anal Function after Intersphincteric Resection for Very Low Rectal Cancer. Dis. Colon Rectum 2009, 52, 64–70. [Google Scholar] [CrossRef]
- Veereman-Wauters, G.; Vlayen, J.J.; Robays, J.; Fairon, N.N.; Stordeur, S.; Rolfo, C.C.; Bielen, D.J.L.E.D.; Bols, A.; Demetter, P.; D’Hoore, A.; et al. Systematic review and meta-analysis of local resection or transanal endoscopic microsurgery versus radical resection in stage i rectal cancer: A real standard? Crit. Rev. Oncol. 2017, 114, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Bach, S.P.; Gilbert, A.; Brock, K.; Korsgen, S.; Geh, I.; Hill, J.; Gill, T.; Hainsworth, P.; Tutton, M.G.; Khan, J.; et al. Radical surgery versus organ preservation via short-course radiotherapy followed by transanal endoscopic microsurgery for early-stage rectal cancer (TREC): A randomised, open-label feasibility study. Lancet Gastroenterol. Hepatol. 2021, 6, 92–105. [Google Scholar] [CrossRef]
- Clancy, C.; Burke, J.P.; Albert, M.R.; O’Connell, P.R.; Winter, D.C. Transanal Endoscopic Microsurgery Versus Standard Transanal Excision for the Removal of Rectal Neoplasms. Dis. Colon Rectum 2015, 58, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Arezzo, A.; Passera, R.; Saito, Y.; Sakamoto, T.; Kobayashi, N.; Sakamoto, N.; Yoshida, N.; Naito, Y.; Fujishiro, M.; Niimi, K.; et al. Systematic review and meta-analysis of endoscopic submucosal dissection versus transanal endoscopic microsurgery for large noninvasive rectal lesions. Surg. Endosc. 2014, 28, 427–438. [Google Scholar] [CrossRef]
- Sica, G.S. Comparative study of laparoscopicvsopen gastrectomy in gastric cancer management. World J. Gastroenterol. 2011, 17, 4602–4606. [Google Scholar] [CrossRef]
- Christoforidis, D.; Cho, H.-M.; Dixon, M.R.; Mellgren, A.F.; Madoff, R.D.; Finne, C.O. Transanal Endoscopic Microsurgery Versus Conventional Transanal Excision for Patients with Early Rectal Cancer. Ann. Surg. 2009, 249, 776–782. [Google Scholar] [CrossRef]
- Nascimbeni, R.; Burgart, L.J.; Nivatvongs, S.; Larson, D.R. Risk of Lymph Node Metastasis in T1 Carcinoma of the Colon and Rectum. Dis. Colon Rectum 2002, 45, 200–206. [Google Scholar] [CrossRef]
- Kikuchi, R.; Takano, M.; Takagi, K.; Fujimoto, N.; Nozaki, R.; Fujiyoushi, T.; Uchida, Y. Management of Early Invasive Colorectal Cancer Risk of Recurrence and Clinical Guidelines. Dis. Colon Rectum 1995, 38, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Nash, G.M.; Weiser, M.R.; Guillem, J.G.; Temple, L.K.; Shia, J.; Gonen, M.; Wong, W.D.; Paty, P.B. Long-Term Survival After Transanal Excision of T1 Rectal Cancer. Dis. Colon Rectum 2009, 52, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Bosch, S.L.; Teerenstra, S.; De Wilt, J.H.W.; Cunningham, C.; Nagtegaal, I.D. Predicting lymph node metastasis in pT1 colorectal cancer: A systematic review of risk factors providing rationale for therapy decisions. Endoscopy 2013, 45, 827–841. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, H.; Eishi, Y.; Ueno, H.; Nemoto, T.; Fujimori, T.; Iwashita, A.; Ajioka, Y.; Ochiai, A.; Ishiguro, S.; Shimoda, T.; et al. A three-tier classification system based on the depth of submucosal invasion and budding/sprouting can improve the treatment strategy for T1 colorectal cancer: A retrospective multicenter study. Mod. Pathol. 2015, 28, 872–879. [Google Scholar] [CrossRef]
- Levic, K.; Bulut, O.; Hesselfeldt, P.; Bülow, S. The outcome of rectal cancer after early salvage TME following TEM compared with primary TME: A case-matched study. Tech. Coloproctol. 2012, 17, 397–403. [Google Scholar] [CrossRef]
- Morino, M.; Allaix, M.E.; Arolfo, S.; Arezzo, A. Previous transanal endoscopic microsurgery for rectal cancer represents a risk factor for an increased abdominoperineal resection rate. Surg. Endosc. 2013, 27, 3315–3321. [Google Scholar] [CrossRef]
- Slors, F.J.; Van Zuijlen, P.P.; Van Dijk, G.J. Sexual and bladder dysfunction after total mesorectal excision for benign diseases. Scand. J. Gastroenterol. Suppl. 2000, 232, 48–51. [Google Scholar]
- EuroSurg Collaborative. EuroSurg: A new European student-driven research network in surgery. Colorectal Dis. 2016, 18, 214–215. [Google Scholar] [CrossRef]
- Lindsey, I.; George, B.D.; Kettlewell, M.G.W.; Mortensen, N.J.M.C. Impotence after mesorectal and close rectal dissection for inflammatory bowel disease. Dis. Colon Rectum 2001, 44, 831–835. [Google Scholar] [CrossRef]
- Rink, A.D.; Radinski, I.; Vestweber, K.-H. Does Mesorectal Preservation Protect the Ileoanal Anastomosis after Restorative Proctocolectomy? J. Gastrointest. Surg. 2008, 13, 120–128. [Google Scholar] [CrossRef]
- Sica, G.S.; Djapardy, V.; Westaby, S.; Maynard, N.D. Diagnosis and Management of Aortoesophageal Fistula Caused by a Foreign Body. Ann. Thorac. Surg. 2004, 2217–2218. [Google Scholar] [CrossRef]
- Overstraeten, A.D.B.V.; Wolthuis, A.M.; D’Hoore, A. Transanal completion proctectomy after total colectomy and ileal pouch-anal anastomosis for ulcerative colitis: A modified single stapled technique. Color. Dis. 2016, 18, O141–O144. [Google Scholar] [CrossRef] [PubMed]
- Lovegrove, R.E.; Constantinides, V.A.; Heriot, A.G.; Athanasiou, T.; Darzi, A.; Remzi, F.H.; Nicholls, R.J.; Fazio, V.W.; Tekkis, P.P. A Comparison of Hand-Sewn Versus Stapled Ileal Pouch Anal Anastomosis (IPAA) Following Proctocolectomy. Ann. Surg. 2006, 244, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Um, J.W.; M’Koma, A.E. Pouch-related dysplasia and adenocarcinoma following restorative proctocolectomy for ulcerative colitis. Tech. Coloproctol. 2011, 15, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Bemelman, W.A.; Warusavitarne, J.; Sampietro, G.M.; Serclova, Z.; Zmora, O.; Luglio, G.; Overstraeten, A.D.B.V.; Burke, J.P.; Buskens, C.J.; Francesco, C.; et al. ECCO-ESCP Consensus on Surgery for Crohn’s Disease. J. Crohn’s Colitis 2017, 12, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Moslein, G. Surgical considerations in FAP-related pouch surgery: Could we do better? Fam. Cancer 2016, 15, 457–466. [Google Scholar] [CrossRef]



| Publication Year | First Author | Article Type | Patients (n) | Stage (n) | Distance from Dentate Line (n) | Radiotherapy (n) | Surgical Procedure (n) | DSA/MHSA (n) |
|---|---|---|---|---|---|---|---|---|
| 1999 | Shimizu [5] | Case Report | 1 | Not reported | Upper rectum | Not reported | Subtotal colectomy | Not reported |
| 2002 | Remzi [6] | Retrospective case series | 26 | Stage 0, 7; Stage I, 9; Stage II, 3; Stage III, 7; | Not reported | 1 adjuvant CRT → pouch loss | IPAA | 10 vs. 16 |
| 2004 | Gorfine [7] | Retrospective case series | 14 | Stage I, 9; Stage 2, 2; Stage 3, 3; | Not reported | 1 neoadjuvant CRT → pouch survival; 1 adjuvant CRT → pouch loss | IPAA | 0 vs. 14 |
| 2009 | Zmora [8] | Retrospective case series | 7 | Stage I, 3; Stage II 3, Stage III, 1; | Upper rectum, 3; Mid rectum, 3; Low rectum, 1 | 1 neoadjuvant CRT → pouch loss; 1 adjuvant CRT → pouch loss | IPAA | 7 vs. 0 |
| 2012 | Merchea [9] | Retrospective case series | 41 | Stage I, 18; Stage II, 10; Stage III, 10; Stage IV, 3 | Upper rectum, 8; Mid rectum, 19; Low rectum, 13; Unknown, 1; | 4 neoadjuvant (0 in IPAA group); 14 adjuvant (1 in IPAA group) → 1 pouch loss | IPAA, 11; PPC, 27; APR, 2; Subtotal colectomy, 1 | 6 vs. 5 |
| 2013 | Inoue [10] | Case Report | 1 | Stage III | 2 cm | Neoadjuvant CRT | IPAA with ISR | HSA |
| 2017 | Tsuchiya [11] | Case report | 1 | Stage I | Upper rectum | Not used | PPC | Not reported |
| 2018 | Hotta [12] | Retrospective case series | 11 | Stage 0, 5; Stage I, 2; Stage III, 2; Unknown, 2 | Lower rectum | Not used | IPAA, 8; IPAA with ISR, 1; PPC, 2 | 0 vs. 9 |
| Paper | Patients (n) | Follow-up | Oncologic Outcomes |
|---|---|---|---|
| Remzi 2002 [6] | 26 | Mean 6.1 years; 2 lost to fu | 95.8% overall survival |
| Gorfine 2004 [7] | 14 | Not specified | 83.5% estimated overall survival at 5 years |
| Merchea 2012 [9] | 41 | Median 4.4 years | 62% overall survival |
| Inoue 2013 [10] | 1 | 1 year | Disease free survival |
| Tsuchiya 2017 [11] | 1 | 2 years | Disease free survival |
| Hotta 2018 [12] | 11 | 5 years | 100% disease free survival |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sensi, B.; Bagaglini, G.; Bellato, V.; Cerbo, D.; Guida, A.M.; Khan, J.; Panis, Y.; Savino, L.; Siragusa, L.; Sica, G.S. Management of Low Rectal Cancer Complicating Ulcerative Colitis: Proposal of a Treatment Algorithm. Cancers 2021, 13, 2350. https://doi.org/10.3390/cancers13102350
Sensi B, Bagaglini G, Bellato V, Cerbo D, Guida AM, Khan J, Panis Y, Savino L, Siragusa L, Sica GS. Management of Low Rectal Cancer Complicating Ulcerative Colitis: Proposal of a Treatment Algorithm. Cancers. 2021; 13(10):2350. https://doi.org/10.3390/cancers13102350
Chicago/Turabian StyleSensi, Bruno, Giulia Bagaglini, Vittoria Bellato, Daniele Cerbo, Andrea Martina Guida, Jim Khan, Yves Panis, Luca Savino, Leandro Siragusa, and Giuseppe S. Sica. 2021. "Management of Low Rectal Cancer Complicating Ulcerative Colitis: Proposal of a Treatment Algorithm" Cancers 13, no. 10: 2350. https://doi.org/10.3390/cancers13102350
APA StyleSensi, B., Bagaglini, G., Bellato, V., Cerbo, D., Guida, A. M., Khan, J., Panis, Y., Savino, L., Siragusa, L., & Sica, G. S. (2021). Management of Low Rectal Cancer Complicating Ulcerative Colitis: Proposal of a Treatment Algorithm. Cancers, 13(10), 2350. https://doi.org/10.3390/cancers13102350






