Scorpion Species with Smaller Body Sizes and Narrower Chelae Have the Highest Venom Potency
Abstract
:1. Introduction
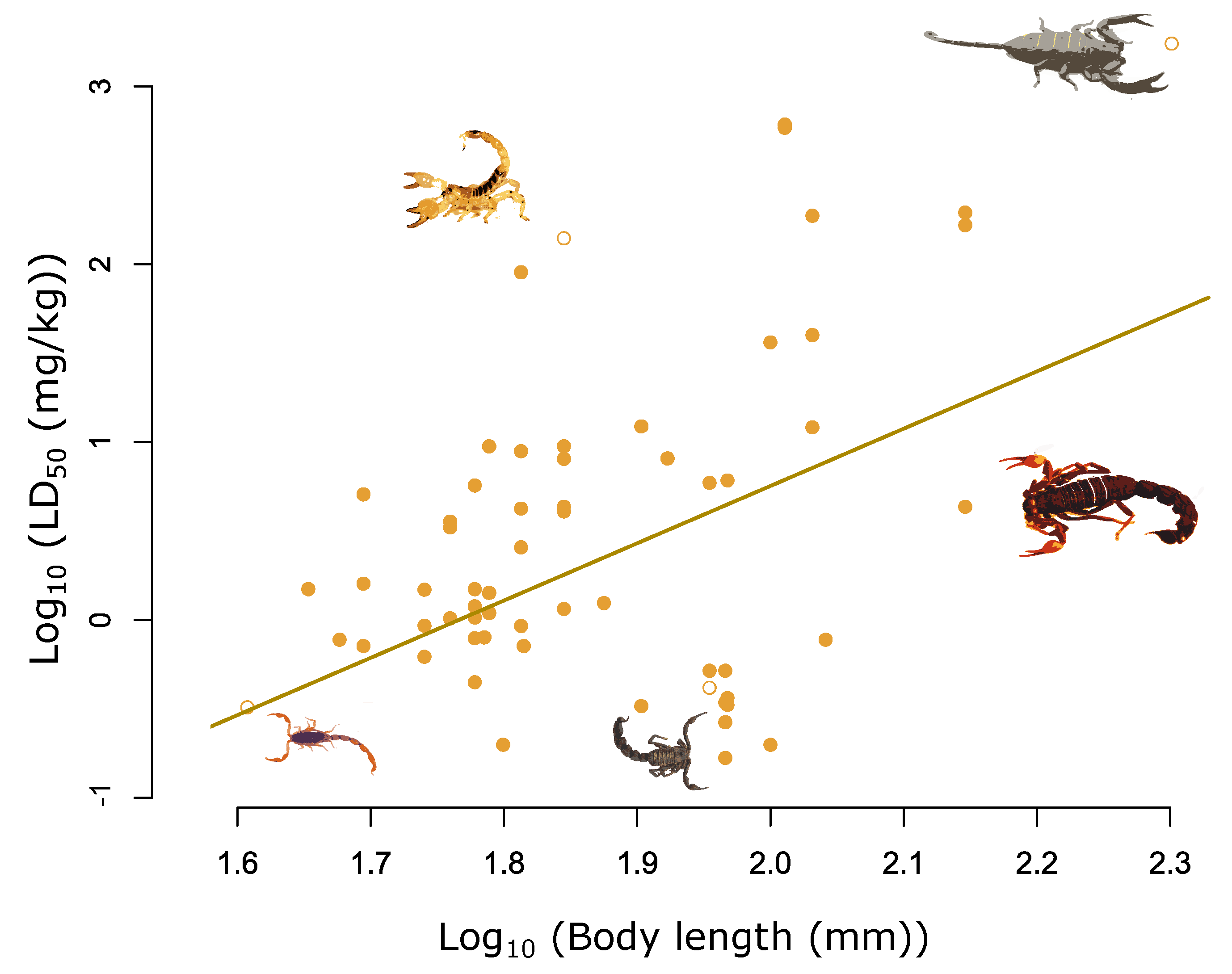
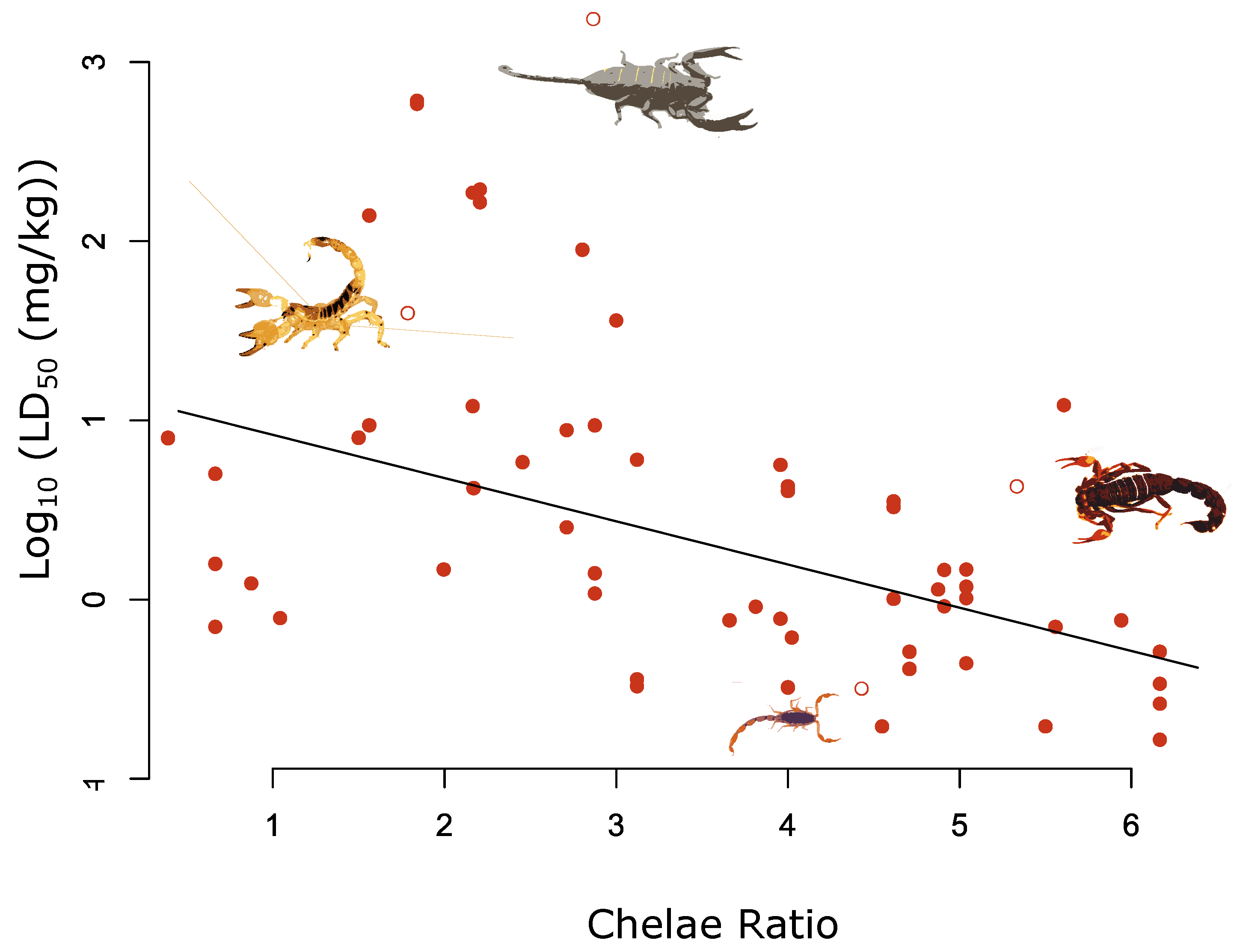
2. Results
3. Discussion
4. Conclusions
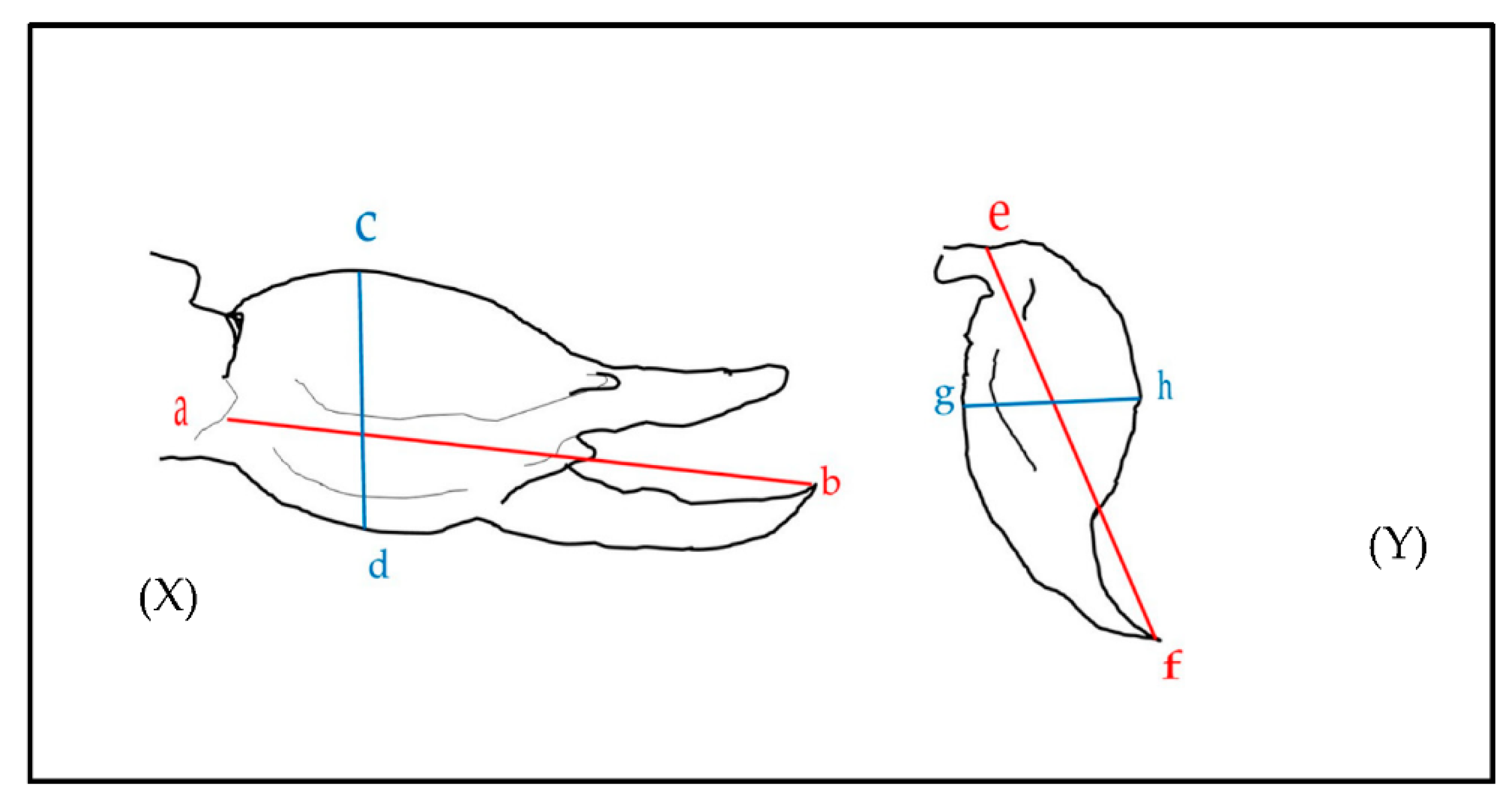
5. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chippaux, J.-P. Emerging options for the management of scorpion stings. Drug Des. Dev. Ther. 2012, 6, 165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Hidan, M.A.; Touloun, O.; Boumezzough, A. An epidemiological study on scorpion envenomation in the Zagora oases (Morocco). J. Coast. Life Med. 2015, 3, 704–707. [Google Scholar] [CrossRef]
- Adawi, S.H.A.A.; Adawi, S.H.A.A.; Adawi, D.H.A.A. The Scorpion sting: Epidemiology, clinical symptoms, treatment, surveillance and reporting obstacles in the Salfit district (West Bank) (2014–2015). Int. J. Trop. Dis. Health 2016, 14, 1–10. [Google Scholar] [CrossRef]
- Elyamani, R.; Soulaymani, A.; Serheir, Z. Epidemiology of scorpion envenoming in the prefecture of Figuig, Morocco. Int. J. Med. Toxicol. Forensic Med. 2019, 9, 17–24. [Google Scholar]
- Santos, M.S.; Silva, C.G.; Neto, B.S.; Grangeiro Junior, C.R.; Lopes, V.H.; Teixeira Junior, A.G.; Bezerra, D.A.; Luna, J.V.; Cordeiro, J.B.; Junior, J.G.; et al. Clinical and Epidemiological Aspects of Scorpionism in the World: A Systematic Review. Wilderness Environ. Med. 2016, 27, 504–518. [Google Scholar] [CrossRef] [Green Version]
- Chippaux, J.P.; Goyffon, M. Epidemiology of scorpionism: A global appraisal. Acta Trop. 2008, 107, 71–79. [Google Scholar] [CrossRef]
- Mohammed-Geba, K.; Obuid-Allah, A.H.; El-Shimy, N.A.; Mahbob, M.A.E.-M.; Ali, R.S.; Said, S.M. DNA Barcoding for Scorpion Species from New Valley Governorate in Egypt Reveals Different Degrees of Cryptic Speciation and Species Misnaming. Conservation 2021, 1, 228–240. [Google Scholar] [CrossRef]
- Khatony, A.; Abdi, A.; Fatahpour, T.; Towhidi, F. The epidemiology of scorpion stings in tropical areas of Kermanshah province, Iran, during 2008 and 2009. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 45. [Google Scholar] [CrossRef] [Green Version]
- Murray, K.A.; Martin, G.; Iwamura, T. Focus on snake ecology to fight snakebite. Lancet 2020, 395, e14. [Google Scholar] [CrossRef]
- Chippaux, J.-P.; Williams, V.; White, J. Snake venom variability: Methods of study, results and interpretation. Toxicon 1991, 29, 1279–1303. [Google Scholar] [CrossRef]
- Vaucel, J.-A.; Larréché, S.; Paradis, C.; Labadie, M.; Courtois, A.; Grenet, G.; Langrand, J.; Tournoud, C.; Nisse, P.; Gallart, J.-C. Relationship between scorpion stings events and environmental conditions in mainland France. J. Med. Entomol. 2021, 58, 2146–2153. [Google Scholar] [CrossRef]
- Indiana Jones Wiki. Available online: https://indianajones.fandom.com/wiki/Scorpion (accessed on 18 January 2022).
- Müller, G.; Modler, H.; Wium, C.; Veale, D. Scorpion sting in southern Africa: Diagnosis and management. Contin. Med. Educ. 2012, 30, 356–361. [Google Scholar]
- Lourenço, W.R. Scorpion incidents, misidentification cases and possible implications for the final interpretation of results. J. Venom. Anim. Toxins Incl. Trop. Dis. 2016, 22, 21. [Google Scholar] [CrossRef]
- Lourenço, W.R. The coevolution between telson morphology and venom glands in scorpions (Arachnida). J. Venom. Anim. Toxins Incl. Trop. Dis. 2020, 26, e20200128. [Google Scholar]
- Van Der Meijden, A.; Lobo Coelho, P.; Sousa, P.; Herrel, A. Choose your weapon: Defensive behavior is associated with morphology and performance in scorpions. PLoS ONE 2013, 8, e78955. [Google Scholar] [CrossRef] [Green Version]
- Coelho, P.; Kaliontzopoulou, A.; Rasko, M.; van der Meijden, A. A ‘striking’relationship: Scorpion defensive behaviour and its relation to morphology and performance. Funct. Ecol. 2017, 31, 1390–1404. [Google Scholar] [CrossRef] [Green Version]
- Bergman, N.J. Scorpion sting in Zimbabwe. S. Afr. Med. J. 1997, 87, 163–167. [Google Scholar]
- Kovařík, F. A review of the genus Heterometrus Ehrenberg, 1828, with descriptions of seven new species (Scorpiones, Scorpionidae). Euscorpius 2004, 2004, 1–60. [Google Scholar] [CrossRef]
- Hoang, A.N.; Vo, H.D.; Vo, N.P.; Kudryashova, K.S.; Nekrasova, O.V.; Feofanov, A.V.; Kirpichnikov, M.P.; Andreeva, T.V.; Serebryakova, M.V.; Tsetlin, V.I. Vietnamese Heterometrus laoticus scorpion venom: Evidence for analgesic and anti-inflammatory activity and isolation of new polypeptide toxin acting on Kv1. 3 potassium channel. Toxicon 2014, 77, 40–48. [Google Scholar] [CrossRef]
- van der Valk, T.; van der Meijden, A. Toxicity of scorpion venom in chick embryo and mealworm assay depending on the use of the soluble fraction versus the whole venom. Toxicon 2014, 88, 38–43. [Google Scholar] [CrossRef]
- Touloun, O.; Slimani, T.; Boumezzough, A. Epidemiological survey of scorpion envenomation in southwestern Morocco. J. Venom. Anim. Toxins 2001, 7, 199–218. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Omran, M.A.A.; Abdel-Nabi, I.M.; Ueda, H.; McVean, A. Intraspecific variation in the Egyptian scorpion Scorpio maurus palmatus venom collected from different biotopes. Toxicon 2009, 53, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Evans, E.R.; Northfield, T.D.; Daly, N.L.; Wilson, D.T. Venom costs and optimization in scorpions. Front. Ecol. Evol. 2019, 7, 196. [Google Scholar] [CrossRef] [Green Version]
- El-Abd Ahmed, A.; Hassan, M.H.; Rashwan, N.I.; Sayed, M.M.; Meki, A.-R.M. Myocardial injury induced by scorpion sting envenoming and evidence of oxidative stress in Egyptian children. Toxicon 2018, 153, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Healy, K.; Carbone, C.; Jackson, A.L. Snake venom potency and yield are associated with prey-evolution, predator metabolism and habitat structure. Ecol. Lett. 2019, 22, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Newlands, G. Scorpion defensive behaviour. Afr. Wildl. 1969, 23, 147–153. [Google Scholar]
- De Sousa, L.; Parrilla-Alvarez, P.; Quiroga, M. An epidemiological review of scorpion stings in Venezuela: The Northeastern region. J. Venom. Anim. Toxins 2000, 6, 128–166. [Google Scholar] [CrossRef]
- Dent, M.A.; Possani, L.D.; Ramírez, G.A.; Fletcher, P.L., Jr. Purification and characterization of two mammalian toxins from the venom of the Mexican scorpion Centruroides noxius Hoffmann. Toxicon 1980, 18, 343–350. [Google Scholar] [CrossRef]
- El-Asmar, M.; Ismail, M.; Osman, O. Immunological studies of scorpion (Buthus minax, L. Koch) venom. Toxicon 1973, 11, 9–14. [Google Scholar] [CrossRef]
- Habermehl, G.G. Arthropoda. In Venomous Animals and Their Toxins; Springer: Berlin/Heidelberg, Germany, 1981; pp. 21–91. [Google Scholar]
- Ismall, M.; Osman, O.; Gumaa, K.; Karrar, M. Some pharmacological studies with scorpion (Pandinus exitialis) venom. Toxicon 1974, 12, 75–78. [Google Scholar] [CrossRef]
- Latifi, M.; Tabatabai, M. Immunological studies on Iranian scorpion venom and antiserum. Toxicon 1979, 17, 617–620. [Google Scholar] [CrossRef]
- Lourenço, W.R.; Cloudsley-Thompson, J. Stridulatory apparatus and the evolutionary significance of sound production in Rhopalurus species (Scorpiones: Buthidae). J. Arid Environ. 1995, 4, 423–429. [Google Scholar] [CrossRef]
- Marinkelle, C.; Stahnke, H. Toxicological and clinical studies on Centruroides margaritatus (Gervais), a common scorpion in western Colombia. J. Med. Entomol. 1965, 2, 197–199. [Google Scholar] [CrossRef]
- Nishikawa, A.; Caricati, C.P.; Lima, M.; Dos Santos, M.; Kipnis, T.; Eickstedt, V.; Knysak, I.; Da Silva, M.; Higashi, H.G.; Da Silva, W.D. Antigenic cross-reactivity among the venoms from several species of Brazilian scorpions. Toxicon 1994, 32, 989–998. [Google Scholar] [CrossRef]
- Ozkan, O.; Yagmur, E.; Ark, M. A newly described scorpion species, Leiurus abdullahbayrami (Scorpion: Buthidae), and the lethal potency and in vivo effects of its venom. J. Venom. Anim. Toxins Incl. Trop. Dis. 2011, 17, 414–421. [Google Scholar]
- Tu, A.T. Handbook of Natural Toxins: Insect poisons, allergens, and other invertebrate venoms. In Handbook of Natural Toxins: Insect Poisons, Allergens, and Other Invertebrate Venoms; Marcel Dekker Inc.: New York, NY, USA, 1984. [Google Scholar]
- Vazirianzadeh, B.; Fatoallahi, I.; Sharififard, M.; Moghadam, A.T. Determination of scorpion venom LD50 of Apistobuthus susanae species in Khuzestan province;(Southwest of Iran). Toxicon 2019, 159, S24. [Google Scholar] [CrossRef]
- Watt, D.D.; Simard, J.M. Neurotoxic proteins in scorpion venom. J. Toxicol. Toxin Rev. 1984, 3, 181–221. [Google Scholar] [CrossRef]
- Whittemore, F., Jr.; Keegan, H.; Borowitz, J. Studies of scorpion antivenins: 1. Paraspecificity. Bull. World Health Organ. 1961, 25, 185. [Google Scholar]
- Yağmur, E.A.; Özkan, Ö.; Karaer, K.Z. Determination of the median lethal dose and electrophoretic pattern of Hottentotta saulcyi (Scorpiones, Buthidae) scorpion venom. J. Arthropod-Borne Dis. 2015, 9, 238. [Google Scholar]
- Zlotkin, E.; Fraenkel, G.; Miranda, F.; Lissitzky, S. The effect of scorpion venom on blowfly larvae—A new method for the evaluation of scorpion venoms potency. Toxicon 1971, 9, 1–2. [Google Scholar] [CrossRef]
- Zlotkin, E.; Martinez, G.; Rochat, H.; Miranda, F. A protein from scorpion venom toxic to crustaceans. Anim. Plant Microb. Toxins 1976, 1, 73–80. [Google Scholar]
- Aboshaala, F.; Badry, A.; Sadine, S.E. Ecological considerations on Buthiscus bicalcaratus Birula, 1905 with a new locality in northern Libya (Scorpiones, Buthidae). Arch. L’Institut Pasteur D’Algérie 2020, 15, 97–101. [Google Scholar]
- Da Rosa, C.M.; Abegg, A.D.; Borges, L.M.; Bitencourt, G.S.; Di Mare, R.A. New record and occurrence map of Tityus serrulatus Lutz & Mello, 1922 (Scorpiones, Buthidae) in the state of Rio Grande do Sul, southern Brazil. Check List 2015, 11, 1556. [Google Scholar]
- De Armas, L.F.; Martín-Frías, E. El género" Centruroides" Marx, 1890 (Scorpiones: Buthidae) en el estado de Veracruz, México. Boletín SEA 2008, 43, 7–22. [Google Scholar]
- Fet, V.; Polis, G.A.; Sissom, W.D. Life in sandy deserts: The scorpion model. J. Arid Environ. 1998, 39, 609–622. [Google Scholar] [CrossRef]
- Fox, G.A.; Cooper, A.M.; Hayes, W.K. The dilemma of choosing a reference character for measuring sexual size dimorphism, sexual body component dimorphism, and character scaling: Cryptic dimorphism and allometry in the scorpion Hadrurus arizonensis. PLoS ONE 2015, 10, e0120392. [Google Scholar] [CrossRef] [Green Version]
- King, H. Notes on Sudan scorpions. Sudan Notes Rec. 1925, 8, 79–84. [Google Scholar]
- Kovařík, F. Taxonomic position of species of the genus Buthacus Birula, 1908 described by Ehrenberg and Lourenço, and description of a new species (Scorpiones: Buthidae). Euscorpius 2005, 2005, 1–13. [Google Scholar] [CrossRef]
- Kovařík, F. A revision of the genus Hottentotta Birula, 1908, with descriptions of four new species (Scorpiones, Buthidae). Euscorpius 2007, 2007, 1–107. [Google Scholar] [CrossRef]
- Kovařík, F.; Lowe, G.; Mazuch, T.; Awale, A.I.; Štundlová, J.; Šťáhlavský, F. Scorpions of the Horn of Africa (Arachnida: Scorpiones). Part XII. Pandinurus hangarale sp. n. (Scorpionidae) from Somaliland and a review of type locality and true distribution of Pandinurus smithi (Pocock, 1897). Euscorpius 2017, 253, 1–18. [Google Scholar] [CrossRef]
- Lourenço, W.; Huber, D.; Cloudsley-Thompson, J. Notes on the ecology, distribution and postembryonic development of Tityus cambridgei Pocock, 1897 (Scorpiones, Buthidae) from French Guyana and Oriental Amazonia. Entomol. Mitt. Aus Dem Zool. Mus. Hambg. 2000, 13, 197–203. [Google Scholar]
- Lourenço, W.R. Révision du Genre Rhopalurus Thorell, 1876 (Scorpiones, Buthidae). Available online: https://www.european-arachnology.org/esa/wp-content/uploads/2015/08/129-137_Lourenco.pdf (accessed on 3 February 2022).
- Lourenco, W.R. Compléments à la faune de scorpions (Arachnida) de l’Afrique du Nord, avec des considérations sur le genre Buthus Leach, 1815. Rev. Suisse Zool. 2003, 110, 875–912. [Google Scholar] [CrossRef]
- Lourenço, W.R. Nouvelles considérations taxonomiques sur les espèces du genre Androctonus Ehrenberg, 1828 et description de deux nouvelles espèces (Scorpiones, Buthidae). Rev. Suisse Zool. 2005, 112, 145–171. [Google Scholar] [CrossRef]
- Lourenço, W.R. What do we know about some of the most conspicuous scorpion species of the genus Tityus? A historical approach. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Pipelzadeh, M.H.; Jalali, A.; Taraz, M.; Pourabbas, R.; Zaremirakabadi, A. An epidemiological and a clinical study on scorpionism by the Iranian scorpion Hemiscorpius lepturus. Toxicon 2007, 50, 984–992. [Google Scholar] [CrossRef]
- Prendini, L. The systematics of southern African Parabuthus Pocock (Scorpiones, Buthidae): Revisions to the taxonomy and key to the species. J. Arachnol. 2004, 32, 109–187. [Google Scholar] [CrossRef]
- Quijano-Ravell, A.F.; Ponce-Saavedra, J. A new species of scorpion of the genus Centruroides (Scorpiones: Buthidae) from the state of Michoacán, Mexico. Rev. Mex. Biodivers. 2016, 87, 49–61. [Google Scholar] [CrossRef] [Green Version]
- Salama, W.M.; Sharshar, K.M. Surveillance study on scorpion species in Egypt and comparison of their crude venom protein profiles. J. Basic Appl. Zool. 2013, 66, 76–86. [Google Scholar] [CrossRef] [Green Version]
- Seiter, M.; Turiel, C. First record of Androctonus australis (Linnaeus, 1758) from Jordan (Scorpiones: Buthidae). Rev. Ibérica Aracnol. 2013, 23, 95–98. [Google Scholar]
- Teruel, R.; Ponce-Saavedra, J.; Quijano-Ravell, A.F. Redescription of Centruroides noxius and description of a closely related new species from western Mexico (Scorpiones: Buthidae). Rev. Mex. Biodivers. 2015, 86, 896–911. [Google Scholar] [CrossRef] [Green Version]
- Karataş, A.; Kürtüllü, M. Duplication of pedipalp segments in the scorpion Androctonus crassicauda (Olivier, 1807). Euscorpius 2006, 2006, 1–4. [Google Scholar] [CrossRef]
- Kovařík, F. Review of Tunisian species of the genus Buthus with descriptions of two new species and a discussion of Ehrenberg’s types (Scorpiones: Buthidae). Euscorpius 2006, 2006, 1–16. [Google Scholar] [CrossRef]
- Kovařík, F.; Yağmur, E.A.; Fet, V.; Navidpour, S. On two subspecies of Mesobuthus eupeus (CL Koch, 1839) in Turkey (Scorpiones: Buthidae). Euscorpius 2011, 2011, 1–15. [Google Scholar]
- Lourenço, W.R. Further considerations on the genus Buthacus Birula, 1908. Boletín Soc. Entomol. Aragonesa 2006, 1, 59–70. [Google Scholar]
- Lourenço, W.R.; Cloudsley-Thompson, J.L. A new species of Buthus Leach, 1815 from Egypt (Scorpiones, Buthidae). Entomol. Mitt. Aus Dem Zool. Mus. Hambg. 2012, 16, 11–18. [Google Scholar]
- Lourenço, W.R.; Rossi, A. Sicily (Scorpiones, Buthidae). Biogeographical Implications. Available online: http://sea-entomologia.org/PDF/RIA22/009014RIA22ButhusSicilia.pdf (accessed on 3 February 2022).
- Booncham, U.; Sitthicharoenchai, D.; Pradatsundarasar, A.-O.; Prasarnpun, S.; Thirakhupt, K. Sexual dimorphism in the Asian giant forest scorpion, Heterometrus laoticus Couzijn, 1981. NU Int. J. Sci. 2007, 4, 42–52. [Google Scholar]
- Bucaretchi, F.; Fernandes, L.C.; Fernandes, C.B.; Branco, M.M.; Prado, C.C.; Vieira, R.J.; De Capitani, E.M.; Hyslop, S. Clinical consequences of Tityus bahiensis and Tityus serrulatus scorpion stings in the region of Campinas, southeastern Brazil. Toxicon 2014, 89, 17–25. [Google Scholar] [CrossRef]
- de Armas, L.F.; Teruel, R.; Kovařík, F. On Centruroides margaritatus (Gervais, 1841) and closely related species (Scorpiones: Buthidae). Euscorpius 2011, 2011, 1–16. [Google Scholar] [CrossRef]
- De Souza, C.A.R.; Candido, D.M.; Lucas, S.M.; Brescovit, A.D. On the Tityus stigmurus complex (Scorpiones, Buthidae). Zootaxa 2009, 1987, 1–38. [Google Scholar] [CrossRef]
- Esposito, L.A.; Yamaguti, H.Y.; Souza, C.A.; Pinto-Da-Rocha, R.; Prendini, L. Systematic revision of the neotropical club-tailed scorpions, Physoctonus, Rhopalurus, and Troglorhopalurus, revalidation of Heteroctenus, and descriptions of two new genera and three new species (Buthidae: Rhopalurusinae). Bull. Am. Mus. Nat. Hist. 2017, 2017, 1–136. [Google Scholar] [CrossRef]
- González-Santillán, E.; Galán-Sánchez, M.A.; Valdez-Velázquez, L.L. A new species of Centruroides (Scorpiones, Buthidae) from Colima, Mexico. Comptes Rendus Biol. 2019, 342, 331–344. [Google Scholar] [CrossRef]
- Mozaffari, E.; Sedaghat, M.M.; Dehkordi, A.S.; Akbarzadeh, K. Bidiversity and species composition of scorpions (Arachnida, Scorpiones) in Ilam County, Iran. J. Appl. Sci. Res. 2013, 9, 5412–5418. [Google Scholar]
- Navidpour, S.; Lowe, G. Revised diagnosis and redescription of Apistobuthus susanae (Scorpiones, Buthidae). J. Arachnol. 2009, 37, 45–59. [Google Scholar] [CrossRef]
- Prendini, L. Further additions to the scorpion fauna of Trinidad and Tobago. J. Arachnol. 2001, 29, 173–188. [Google Scholar] [CrossRef]
- Teruel, R.; de Armas, L.F. Redescripción de Rhopalurus junceus (Herbst 1800) (Scorpiones: Buthidae). Boletín Soc. Entomol. Aragonesa 2012, 50, 153–174. [Google Scholar]
- Teruel, R.; Kovařík, F. The true identity of the enigmatic scorpion Centruroides subgranosus (Kraepelin, 1898), with some taxonomic comments on Centruroides vittatus (Say, 1821) and Centruroides suffusus Pocock, 1902 (Scorpiones: Buthidae). Euscorpius 2010, 2010, 1–9. [Google Scholar] [CrossRef]
- Teruel, R.; Kovařík, F. Redescription of Androctonus bicolor Ehrenberg, 1828, and Description of Androctonus turieli sp. n. from Tunisia (Scorpiones: Buthidae). Euscorpius 2014, 186, 1–15. [Google Scholar] [CrossRef]
- Yağmur, E.A.; Koç, H.; Kunt, K.B. Description of a new species of Leiurus Ehrenberg, 1828 (Scorpiones: Buthidae) from southeastern Turkey. Euscorpius 2009, 2009, 1–20. [Google Scholar] [CrossRef]
- van der Meijden, A.; Kleinteich, T.; Coelho, P. Packing a pinch: Functional implications of chela shapes in scorpions using finite element analysis. J. Anat. 2012, 220, 423–434. [Google Scholar] [CrossRef]
- Williams, S. Scorpion bionomics. Annu. Rev. Entomol. 1987, 32, 275–295. [Google Scholar] [CrossRef]
- Edmunds, M.C.; Sibly, R.M. Optimal sting use in the feeding behavior of the scorpion Hadrurus spadix. J. Arachnol. 2010, 38, 123–125. [Google Scholar] [CrossRef] [Green Version]
- Simone, Y.; Garcia, L.F.; Lacava, M.; van der Meijden, A.; Viera, C. Predatory versatility in females of the scorpion Bothriurus bonariensis (Scorpiones: Bothriuridae): Overcoming prey with different defensive mechanisms. J. Insect Behav. 2018, 31, 402–415. [Google Scholar] [CrossRef]
- Li, M.; Fry, B.; Kini, R.M. Eggs-only diet: Its implications for the toxin profile changes and ecology of the marbled sea snake (Aipysurus eydouxii). J. Mol. Evol. 2005, 60, 81–89. [Google Scholar] [CrossRef]
- Niermann, C.N.; Tate, T.G.; Suto, A.L.; Barajas, R.; White, H.A.; Guswiler, O.D.; Secor, S.M.; Rowe, A.H.; Rowe, M.P. Defensive Venoms: Is Pain Sufficient for Predator Deterrence? Toxins 2020, 12, 260. [Google Scholar] [CrossRef] [Green Version]
- Pekár, S.; Líznarová, E.; Bočánek, O.; Zdráhal, Z. Venom of prey-specialized spiders is more toxic to their preferred prey: A result of prey-specific toxins. J. Anim. Ecol. 2018, 87, 1639–1652. [Google Scholar] [CrossRef]
- Richards, D.P.; Barlow, A.; Wüster, W. Venom lethality and diet: Differential responses of natural prey and model organisms to the venom of the saw-scaled vipers (Echis). Toxicon 2012, 59, 110–116. [Google Scholar] [CrossRef]
- Van der Meijden, A.; Koch, B.; Van der Valk, T.; Vargas-Muñoz, L.J.; Estrada-Gómez, S. Target-specificity in scorpions; comparing lethality of scorpion venoms across arthropods and vertebrates. Toxins 2017, 9, 312. [Google Scholar] [CrossRef] [Green Version]
- Lyons, K.; Dugon, M.M.; Healy, K. Diet breadth mediates the prey specificity of venom potency in snakes. Toxins 2020, 12, 74. [Google Scholar] [CrossRef] [Green Version]
- Nejati, J.; Saghafipour, A.; Rafinejad, J.; Mozaffari, E.; Keyhani, A.; Abolhasani, A.; Kareshk, A.T. Scorpion composition and scorpionism in a high-risk area, the southwest of Iran. Electron. Physician 2018, 10, 7138. [Google Scholar] [CrossRef] [Green Version]
- El Hidan, M.A.; Touloun, O.; Bouazza, A.; Laaradia, M.A.; Boumezzough, A. Androctonus genus species in arid regions: Ecological niche models, geographical distributions, and envenomation risk. Vet. World 2018, 11, 286. [Google Scholar] [CrossRef]
- Ward, M.J.; Ellsworth, S.A.; Nystrom, G.S. A global accounting of medically significant scorpions: Epidemiology, major toxins, and comparative resources in harmless counterparts. Toxicon 2018, 151, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Van Der Meijden, A.; Herrel, A.; Summers, A. Comparison of chela size and pincer force in scorpions; getting a first grip. J. Zool. 2010, 280, 319–325. [Google Scholar] [CrossRef]
- van der Meijden, A.; Kleinteich, T. A biomechanical view on stinger diversity in scorpions. J. Anat. 2017, 230, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Hadfield, J.D. MCMC methods for multi-response generalized linear mixed models: The MCMCglmm R package. J. Stat. Softw. 2010, 33, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Hinchliff, C.E.; Smith, S.A.; Allman, J.F.; Burleigh, J.G.; Chaudhary, R.; Coghill, L.M.; Crandall, K.A.; Deng, J.; Drew, B.T.; Gazis, R. Synthesis of phylogeny and taxonomy into a comprehensive tree of life. Proc. Natl. Acad. Sci. USA 2015, 112, 12764–12769. [Google Scholar] [CrossRef] [Green Version]
- Hadfield, J.; Nakagawa, S. General quantitative genetic methods for comparative biology: Phylogenies, taxonomies and multi-trait models for continuous and categorical characters. J. Evol. Biol. 2010, 23, 494–508. [Google Scholar] [CrossRef]
- Brooks, S.P.; Gelman, A. General methods for monitoring convergence of iterative simulations. J. Comput. Graph. Stat. 1998, 7, 434–455. [Google Scholar]
- Brown, J.H.; Gillooly, J.F.; Allen, A.P.; Savage, V.M.; West, G.B. Toward a metabolic theory of ecology. Ecology 2004, 85, 1771–1789. [Google Scholar] [CrossRef]
| β | Lower CI | Upper CI | pMCMC | |
|---|---|---|---|---|
| Fixed Terms | ||||
| Intercept | −4.82 | −8.40 | −1.68 | 0.004 |
| Log10 body length (mm) | 3.24 | 1.57 | 4.99 | <0.001 |
| LD50 methodSC | ||||
| IV | −0.04 | −0.35 | 0.29 | 0.81 |
| IP | 0.17 | −0.36 | 0.73 | 0.88 |
| IM | 0.10 | −1.05 | 1.35 | 0.52 |
| Chela ratio | −0.26 | −0.40 | −0.11 | 0.005 |
| Telson ratio | 0.04 | −0.15 | 0.20 | 0.63 |
| Random Terms | ||||
| Phylogeny (h2) | 0.60 | 0.25 | 0.87 | |
| Species | 0.01 | 0.00 | 0.46 | |
| Residuals | 0.26 | 0.10 | 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forde, A.; Jacobsen, A.; Dugon, M.M.; Healy, K. Scorpion Species with Smaller Body Sizes and Narrower Chelae Have the Highest Venom Potency. Toxins 2022, 14, 219. https://doi.org/10.3390/toxins14030219
Forde A, Jacobsen A, Dugon MM, Healy K. Scorpion Species with Smaller Body Sizes and Narrower Chelae Have the Highest Venom Potency. Toxins. 2022; 14(3):219. https://doi.org/10.3390/toxins14030219
Chicago/Turabian StyleForde, Alannah, Adam Jacobsen, Michel M. Dugon, and Kevin Healy. 2022. "Scorpion Species with Smaller Body Sizes and Narrower Chelae Have the Highest Venom Potency" Toxins 14, no. 3: 219. https://doi.org/10.3390/toxins14030219
APA StyleForde, A., Jacobsen, A., Dugon, M. M., & Healy, K. (2022). Scorpion Species with Smaller Body Sizes and Narrower Chelae Have the Highest Venom Potency. Toxins, 14(3), 219. https://doi.org/10.3390/toxins14030219





