Non-Dairy Fermented Beverages as Potential Carriers to Ensure Probiotics, Prebiotics, and Bioactive Compounds Arrival to the Gut and Their Health Benefits
Abstract
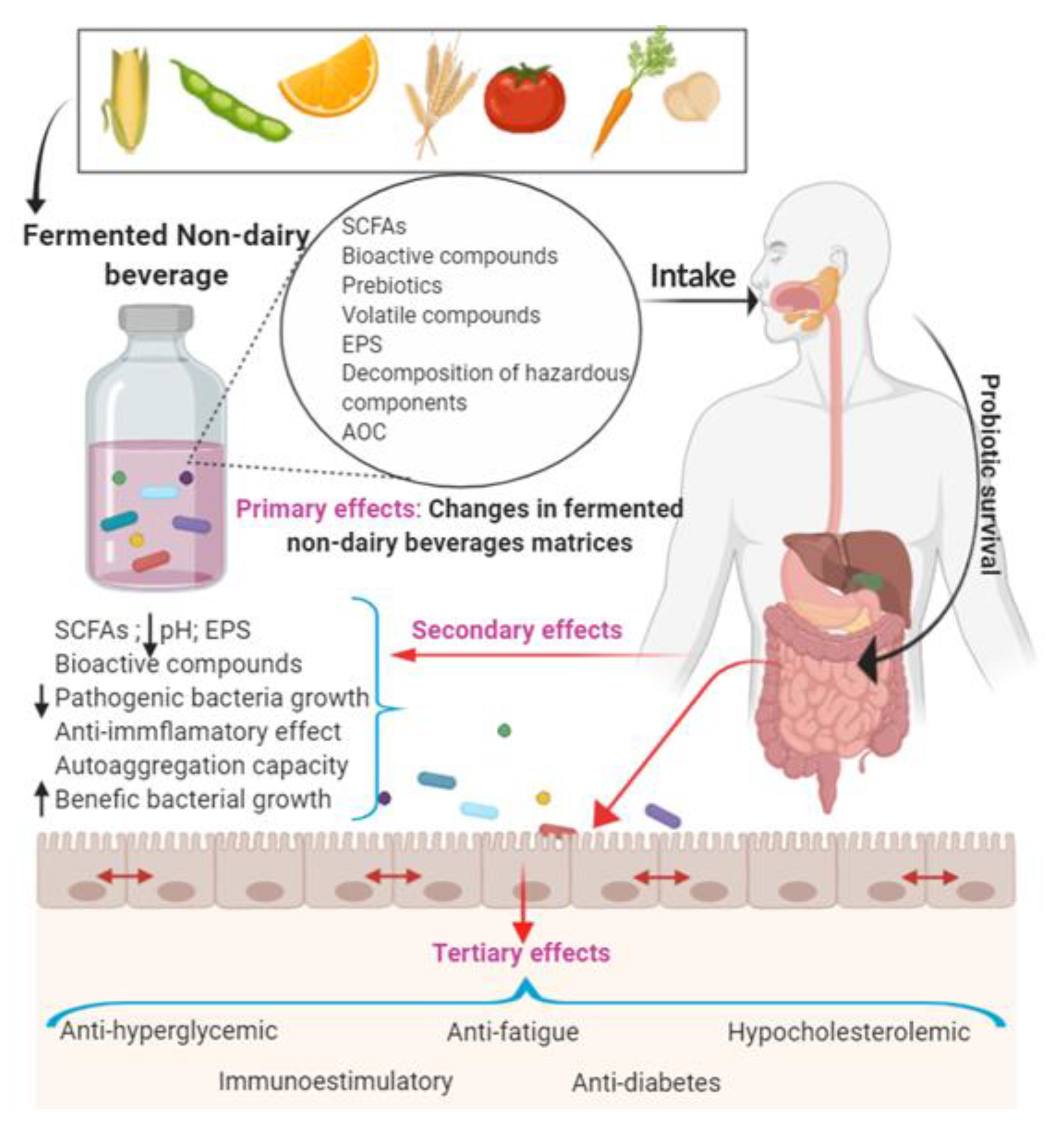
1. Introduction
2. Non-Dairy Fermented Beverages as Vehicles for Bioactive Compound, Probiotic, and Prebiotic Delivery to the Gut and their Health Benefits
2.1. Fermented Legume Beverages
2.2. Fermented Cereal Beverages
2.3. Fermented Pseudocereal Beverages
2.4. Fermented Fruit and Vegetable Beverages
3. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Ajmone Marsan, P.; Cocconcelli, P.S.; Masoero, F.; Miggiano, G.; Morelli, L.; Moro, D.; Rossi, F.; Sckokai, P.; Trevisi, E. Food for healthy living and active ageing. Stud. Health Technol. Inform. 2014, 203, 32–43. [Google Scholar]
- Guerrero, C.; Wilson, L. Chapter 5—Enzymatic Production of Lactulose. In Lactose-Derived Prebiotics; Illanes, A., Conejeros, R., Eds.; Academic Press: London, UK, 2016; pp. 191–227. [Google Scholar]
- Villaño, D.; Gironés-Vilapana, A.; García-Viguera, C.; Moreno, D.A. Chapter 10—Development of Functional Foods. In Innovation Strategies in the Food Industry; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 191–210. [Google Scholar]
- Gouw, V.P.; Jung, J.; Zhao, Y. Functional properties, bioactive compounds, and in vitro gastrointestinal digestion study of dried fruit pomace powders as functional food ingredients. LWT Food Sci. Technol. 2017, 80, 136–144. [Google Scholar] [CrossRef]
- Rathore, S.; Salmeron, I.; Pandiella, S.S. Production of potentially probiotic beverages using single and mixed cereal substrates fermented with lactic acid bacteria cultures. Food Microbiol. 2012, 30, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Vieira da Silva, B.; Barreira, J.C.M.; Oliveira, M.B.P.P. Natural phytochemicals and probiotics as bioactive ingredients for functional foods: Extraction, biochemistry and protected-delivery technologies. Trends Food Sci. Technol. 2016, 50, 144–158. [Google Scholar] [CrossRef]
- Yasmin, A.; Butt, M.S.; van Baak, M.; Shahid, M.Z. Supplementation of prebiotics to a whey-based beverage reduces the risk of hypercholesterolaemia in rats. Int. Dairy J. 2015, 48, 80–84. [Google Scholar] [CrossRef]
- Marques, T.M.; Cryan, J.F.; Shanahan, F.; Fitzgerald, G.F.; Ross, R.P.; Dinan, T.G.; Stanton, C. Gut microbiota modulation and implications for host health: Dietary strategies to influence the gut–brain axis. Innov. Food Sci. Emerg. Technol. 2014, 22, 239–247. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligne, B.; Ganzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Plé, C.; Breton, J.; Daniel, C.; Foligné, B. Maintaining gut ecosystems for health: Are transitory food bugs stowaways or part of the crew? Int. J. Food Microbiol. 2015, 213, 139–143. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations and World Health Organization Expert Consultation Report (FAO/WHO). Guidelines for the Evaluation of Probiotics in Food; Joint FAO/WHO Working Group Meeting: London, ON, Canada, 2002. [Google Scholar]
- Salminen, S.; von Wright, A.; Morelli, L.; Marteau, P.; Brassart, D.; de Vos, W.M.; Fonden, R.; Saxelin, M.; Collins, K.; Mogensen, G.; et al. Demonstration of safety of probiotics—A review. Int. J. Food Microbiol. 1998, 44, 93–106. [Google Scholar] [CrossRef]
- Ricci, A.; Allende, A.; Bolton, D.; Chemaly, M.; Davies, R.; Girones, R.; Koutsoumanis, K.; Lindqvist, R.; Nørrung, B.; Robertson, L.; et al. Update of the list of QPS-recommended biological agents intentionally added to food or feed as notified to EFSA 6: Suitability of taxonomic units notified to EFSA until March 2017. EFSA J. 2017, 15, 4884. [Google Scholar]
- Tripathi, M.K.; Giri, S.K. Probiotic functional foods: Survival of probiotics during processing and storage. J. Funct. Foods 2014, 9, 225–241. [Google Scholar] [CrossRef]
- Ranadheera, C.S.; Evans, C.A.; Adams, M.C.; Baines, S.K. In vitro analysis of gastrointestinal tolerance and intestinal cell adhesion of probiotics in goat’s milk ice cream and yogurt. Food Res. Int. 2012, 49, 619–625. [Google Scholar] [CrossRef]
- Shori, A.B. Influence of food matrix on the viability of probiotic bacteria: A review based on dairy and non-dairy beverages. Food Biosci. 2016, 13, 1–8. [Google Scholar] [CrossRef]
- Aljewicz, M.; Cichosz, G. The effect of probiotic Lactobacillus rhamnosus HN001 on the in vitro availability of minerals from cheeses and cheese-like products. LWT Food Sci. Technol. 2015, 60, 841–847. [Google Scholar] [CrossRef]
- Mani-Lopez, E.; Palou, E.; Lopez-Malo, A. Probiotic viability and storage stability of yogurts and fermented milks prepared with several mixtures of lactic acid bacteria. J. Dairy Sci. 2014, 97, 2578–2590. [Google Scholar] [CrossRef]
- O’Brien, K.V.; Aryana, K.J.; Prinyawiwatkul, W.; Ordonez, K.M.C.; Boeneke, C.A. Short communication: The effects of frozen storage on the survival of probiotic microorganisms found in traditionally and commercially manufactured kefir. J. Dairy Sci. 2016, 99, 7043–7048. [Google Scholar] [CrossRef]
- Rutella, G.S.; Tagliazucchi, D.; Solieri, L. Survival and bioactivities of selected probiotic lactobacilli in yogurt fermentation and cold storage: New insights for developing a bi-functional dairy food. Food Microbiol. 2016, 60, 54–61. [Google Scholar] [CrossRef]
- Granato, D.; Branco, G.F.; Nazzaro, F.; Cruz, A.G.; Faria, J.A.F. Functional foods and nondairy probiotic food development: Trends, concepts, and products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 292–302. [Google Scholar] [CrossRef]
- Vijaya Kumar, B.; Vijayendra, S.V.; Reddy, O.V. Trends in dairy and non-dairy probiotic products—A review. J. Food Sci. Technol. 2015, 52, 6112–6124. [Google Scholar] [CrossRef]
- Marsh, A.J.; Hill, C.; Ross, R.P.; Cotter, P.D. Fermented beverages with health-promoting potential: Past and future perspectives. Trends Food Sci. Technol. 2014, 38, 113–124. [Google Scholar] [CrossRef]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introduccing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Sirisomboon, P.; Pornchaloempong, P.; Romphophak, T. Physical properties of green soybean: Criteria for sorting. J. Food Eng. 2007, 79, 18–22. [Google Scholar] [CrossRef]
- Katz, Y.; Gutierrez-Castrellon, P.; Gonzalez, M.G.; Rivas, R.; Lee, B.W.; Alarcon, P. A comprehensive review of sensitization and allergy to soy-based products. Clin. Rev. Allergy Immunol. 2014, 46, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Osorio-Diaz, P.; Agama-Acevedo, E.; Mendoza-Vinalay, M.; Tovar, J.; Bello-Perez, L.A. Pasta added with chickpea flour: Chemical composition, in vitro starch digestibility and predicted glycemic index. CyTA J. Food 2008, 6, 6–12. [Google Scholar]
- Rossi, E.A.; Vendramini, R.C.; Carlos, I.Z.; Pei, Y.C.; De Valdez, G.F. Development of a novel fermented soymilk product with potential probiotic properties. Eur. Food Res. Technol. 1999, 209, 305–507. [Google Scholar] [CrossRef]
- Bedani, R.; Rossi, E.A.; Isay Saad, S.M. Impact of inulin and okara on Lactobacillus acidophilus La-5 and Bifidobacterium animalis Bb-12 viability in a fermented soy product and probiotic survival under in vitro simulated gastrointestinal conditions. Food Microbiol. 2013, 34, 382–389. [Google Scholar] [CrossRef]
- Mridula, D.; Sharma, M. Development of non-dairy probiotic drink utilizing sprouted cereals, legume and soymilk. LWT Food Sci. Technol. 2015, 62, 482–487. [Google Scholar] [CrossRef]
- Santos, C.C.A.A.; Libeck, B.S.; Schwan, R.F. Co-culture fermentation of peanut-soy milk for the development of a novel functional beverage. Int. J. Food Microbiol. 2014, 186, 32–41. [Google Scholar] [CrossRef]
- Cabello-Olmo, M.; Oneca, M.; Torre, P.; Sainz, N.; Moreno-Aliaga, M.J.; Guruceaga, E.; Diaz, J.V.; Encio, I.J.; Barajas, M.; Arana, M. A fermented food product containing lactic acid bacteria protects ZDF rats from the development of type 2 diabetes. Nutrients 2019, 11, 2530. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Nagino, T.; Kaga, C.; Kano, M.; Masuoka, N.; Anbe, M.; Moriyama, K.; Maruyama, K.; Nakamura, S.; Shida, K.; Miyazaki, K. Effects of fermented soymilk with Lactobacillus casei Shirota on skin condition and the gut microbiota: A randomised clinical pilot trial. Benef. Microbes 2018, 9, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chelikani, V.; Serventi, L. Evaluation of chickpea as alternative to soy in plant-based beverages, fresh and fermented. LWT Food Sci. Technol. 2018, 97, 570–572. [Google Scholar] [CrossRef]
- Chavan, M.; Gat, Y.; Harmalkar, M.; Waghmare, R. Development of non-dairy fermented probiotic drink based on germinated and ungerminated cereals and legume. LWT Food Sci. Technol. 2018, 91, 339–344. [Google Scholar] [CrossRef]
- Schwan, R.F.; Ramos, C.L. Chapter 10: Functional Beverages from Cereals. In Functional and Medicinal Beverages; Grumezescu, A., Holban, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 351–379. [Google Scholar]
- Angelov, A.; Yaneva-Marinova, T.; Gotcheva, V. Oats as a matrix of choice for developing fermented functional beverages. J. Food Sci. Technol. 2018, 55, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Kedia, G.; Vazquez, J.A.; Charalampopoulos, D.; Pandiella, S.S. In vitro fermentation of oat bran obtained by debranning with a mixed culture of human fecal bacteria. Curr. Microbiol. 2009, 58, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Luana, N.; Rossana, C.; Curiel, J.A.; Kaisa, P.; Marco, G.; Rizzello, C.G. Manufacture and characterization of a yogurt-like beverage made with oat flakes fermented by selected lactic acid bacteria. Int. J. Food Microbiol. 2014, 185, 17–26. [Google Scholar] [CrossRef]
- Bernat, N.; Chafer, M.; Gonzalez-Martinez, C.; Rodriguez-Garcia, J.; Chiralt, A. Optimisation of oat milk formulation to obtain fermented derivatives by using probiotic Lactobacillus reuteri microorganisms. Food Sci. Technol. Int. 2015, 21, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Cox, S.; Abu-Ghannam, N. Process optimization for the development of a functional beverage based on lactic acid fermentation of oats. Biochem. Eng. J. 2010, 52, 199–204. [Google Scholar] [CrossRef]
- Gupta, M.; Bajaj, B. Development of fermented oat flour beverage as a potential probiotic vehicle. Food Biosci. 2017, 2017, 104–107. [Google Scholar] [CrossRef]
- Kedia, G.; Vazquez, J.A.; Pandiella, S.S. Fermentability of whole oat flour, PeriTec flour and bran by Lactobacillus plantarum. J. Food Eng. 2008, 89, 246–249. [Google Scholar] [CrossRef][Green Version]
- Wang, C.; Liang, S.; Wang, H.; Guo, M. Physiochemical properties and probiotic survivability of symbiotic oat-based beverage. Food Sci. Biotechnol. 2018, 27, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Funck, G.; Marques, J.; Cruxen, C.; Sehn, C.; Haubert, L.; Dannenberg, G.; Klajn, V.M.; Silva, W.; Fiorentini, A. Probiotic potential of Lactobacillus curvatus P99 and viability in fermented oat dairy beverage. J. Food Process. Preserv. 2019, 43, e14286. [Google Scholar] [CrossRef]
- Johansson, M.L.; Nobaek, S.; Berggren, A.; Nyman, M.; Bjorck, I.; Ahrne, S.; Jeppsson, B.; Molin, G. Survival of Lactobacillus plantarum DSM 9843 (299v), and effect on the short-chain fatty acid content of faeces after ingestion of a rose-hip drink with fermented oats. Int. J. Food Microbiol. 1998, 42, 29–38. [Google Scholar] [CrossRef]
- Ghosh, K.; Ray, M.; Adak, A.; Halder, S.K.; Das, A.; Jana, A.; Parua Mondal, S.; Vagvolgyi, C.; Das Mohapatra, P.K.; Pati, B.R.; et al. Role of probiotic Lactobacillus fermentum KKL1 in the preparation of a rice based fermented beverage. Bioresour. Technol. 2015, 188, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Giri, S.S.; Sen, S.S.; Saha, S.; Sukumaran, V.; Park, S.C. Use of a potential probiotic, Lactobacillus plantarum L7, for the preparation of a rice-based fermented beverage. Front. Microbiol. 2018, 9, 473. [Google Scholar] [CrossRef] [PubMed]
- Ranum, P.; Pena-Rosas, J.P.; Garcia-Casal, M.N. Global maize production, utilization, and consumption. Ann. N. Y. Acad. Sci. 2014, 1312, 105–112. [Google Scholar] [CrossRef]
- Menezes, A.G.T.; Ramos, C.L.; Dias, D.R.; Schwan, R.F. Combination of probiotic yeast and lactic acid bacteria as starter culture to produce maize-based beverages. Food Res. Int. 2018, 111, 187–197. [Google Scholar] [CrossRef]
- Wacoo, A.P.; Mukisa, I.M.; Meeme, R.; Byakika, S.; Wendiro, D.; Sybesma, W.; Kort, R. Probiotic enrichment and reduction of aflatoxins in a traditional African maize-based fermented food. Nutrients 2019, 11, 265. [Google Scholar] [CrossRef]
- Freire, A.L.; Ramos, C.L.; Schwan, R.F. Effect of symbiotic interaction between a fructooligosaccharide and probiotic on the kinetic fermentation and chemical profile of maize blended rice beverages. Food Res. Int. 2017, 100, 698–707. [Google Scholar] [CrossRef]
- Reguera, M.; Haros, C.M. Structure and composition kernels. In Pseudocereals Chemistry and Technology; Haros, C.M., Schoenlechner, R., Eds.; Wiley-Blackwell: West Sussex, UK, 2017; pp. 28–48. [Google Scholar]
- Bhargava, A.; Shukla, S.; Ohri, D. Chenopodium quinoa—An Indian perspective. Ind. Crop. Prod. 2006, 23, 73–87. [Google Scholar] [CrossRef]
- Ranilla, L.G.; Apostolidis, E.; Genovese, M.I.; Lajolo, F.M.; Shetty, K. Evaluation of indigenous grains from the Peruvian Andean region for antidiabetes and antihypertension potential using in vitro methods. J. Med. Food 2009, 12, 704–713. [Google Scholar] [CrossRef]
- Ludena-Urquizo, F.E.; García-Torres, S.M.; Tolonen, T.; Jaakkola, M.; Pena-Niebuhr, M.G.; von Wright, A.; Repo-Carrasco-Valencia, R.; Korhonen, H.; Plumed-Ferrer, C. Development of a fermented quinoa-based beverage. Food Sci. Nutr. 2017, 5, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Vera-Pingitore, E.; Jimenez, M.E.; Dallagnol, A.; Belfiore, C.; Fontana, C.; Fontana, P.; von Wright, A.; Vignolo, G.; Plumed-Ferrer, C. Screening and characterization of potential probiotic and starter bacteria for plant fermentations. LWT Food Sci. Technol. 2016, 71, 288–294. [Google Scholar] [CrossRef]
- Bianchi, F.; Rossi, E.A.; Sakamoto, I.K.; Tallarico Adorno, M.A.; Van de Wiele, T.; Sivieri, K. Beneficial effects of fermented vegetal beverages on human gastrointestinal microbial ecosystem in a simulator. Food Res. Int. 2014, 64, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Molly, K.; Vandewoestyne, M.; Desmet, I.; Verstraete, W. Validation of the simulator of the human intestinal microbial ecosystem (SHIME) reactor using microorganism associated activities. Microb. Ecol. Health Dis. 1994, 7, 191–200. [Google Scholar]
- Ichikawa, H.; Sakata, T. Stimulation of epithelial cell proliferation of isolated distal colon of rats by continuous colonic infusion of ammonia or short-chain fatty acids is nonadditive. J. Nutr. 1998, 128, 843–847. [Google Scholar] [CrossRef]
- Matejčeková, Z.; Liptáková, D.; Valík, L. Functional probiotic products based on fermented buckwheat with Lactobacillus rhamnosus. LWT Food Sci. Technol. 2017, 81, 35–41. [Google Scholar] [CrossRef]
- Kocková, M.; Valík, L. Development of new cereal-, pseudocereal-, and cereal-leguminous-based probiotic foods. Czech J. Food Sci. 2014, 32, 391–397. [Google Scholar] [CrossRef]
- Kocková, M.; Dilongová, M.; Hybenová, E.; Valík, L. Evaluation of cereals and pseudocereals suitability for the development of new probiotic foods. J. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Bustos, A.Y.; Gerez, C.L.; Mohtar Mohtar, L.G.; Paz Zanini, V.I.; Nazareno, M.A.; Taranto, M.P.; Iturriaga, L.B. Lactic acid fermentation improved textural behaviour, phenolic compounds and antioxidant activity of chia (Salvia hispanica L.) dough. Food Technol. Biotechnol. 2017, 55, 381–389. [Google Scholar] [CrossRef]
- Kumar, H.; Salminen, S.; Verhagen, H.; Rowland, I.; Heimbach, J.; Banares, S.; Lalonde, M. Novel probiotics and prebiotics: Road to the market. Curr. Opin. Biotechnol. 2015, 32, 99–103. [Google Scholar] [CrossRef]
- Valero-Cases, E.; Frutos, M.J. Effect of inulin on the viability of L. plantarum during storage and in vitro digestion and on composition parameters of vegetable fermented juices. Plant Foods Hum. Nutr. 2017, 72, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Valero-Cases, E.; Frutos, M.J. Development of prebiotic nectars and juices as potential substrates for Lactobacillus acidophilus: Special reference to physicochemical characterization and consumer acceptability during storage. LWT Food Sci. Technol. 2017, 81, 136–143. [Google Scholar] [CrossRef]
- Mantzourani, I.; Terpou, A.; Bekatorou, A.; Mallouchos, A.; Alexopoulos, A.; Kimbaris, A.; Bezirtzoglou, E.; Koutinas, A.A.; Plessas, S. Functional pomegranate beverage production by fermentation with a novel synbiotic L. paracasei biocatalyst. Food Chem. 2020, 308, 125658. [Google Scholar] [CrossRef]
- Mantzourani, I.; Terpou, A.; Alexopoulos, A.; Bezirtzoglou, E.; Bekatorou, A.; Plessas, S. Production of a potentially synbiotic fermented Cornelian cherry (Cornus mas L.) beverage using Lactobacillus paracasei K5 immobilized on wheat bran. Biocatal. Agric. Biotechnol. 2019, 17, 347–351. [Google Scholar] [CrossRef]
- Pimentel, T.C.; Madrona, G.S.; Prudencio, S.H. Probiotic clarified apple juice with oligofructose or sucralose as sugar substitutes: Sensory profile and acceptability. LWT Food Sci. Technol. 2015, 62, 838–846. [Google Scholar] [CrossRef]
- da Costa, G.; Silva, J.; Mingotti, J.; Barão, C.; Klososki, S.; Pimentel, T. Effect of ascorbic acid or oligofructose supplementation on L. paracasei viability, physicochemical characteristics and acceptance of probiotic orange juice. LWT Food Sci. Technol. 2016, 75, 195–201. [Google Scholar] [CrossRef]
- Miranda, R.F.; da Silva, J.P.; Machado, A.R.F.; da Silva, E.C.; de Souza, R.C.; Marcolino, V.A.; Klososki, S.J.; Pimentel, T.C.; Barão, C.E. Impact of the addition of Lactobacillus casei and oligofructose on the quality parameters of orange juice and hibiscus tea mixed beverage. Food Process. Preserv. 2019, 43, e14249. [Google Scholar] [CrossRef]
- Bernal-Castro, C.A.; Díaz-Moreno, C.; Gutiérrez-Cortés, C. Inclusion of prebiotics on the viability of a commercial Lactobacillus casei subsp. rhamnosus culture in a tropical fruit beverage. J. Food Sci. Technol. 2019, 56, 987–994. [Google Scholar] [CrossRef]
- Esmaeilinezhad, Z.; Babajafari, S.; Sohrabi, Z.; Eskandari, M.H.; Amooee, S.; Barati-Boldaji, R. Effect of synbiotic pomegranate juice on glycemic, sex hormone profile and anthropometric indices in PCOS: A randomized, triple blind, controlled trial. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 201–208. [Google Scholar] [CrossRef]
- Li, Z.; Teng, J.; Lyu, Y.; Hu, X.; Zhao, Y.; Wang, M. Enhanced antioxidant activity for apple juice fermented with Lactobacillus plantarum ATCC14917. Molecules 2018, 24, 51. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhou, J.; Fan, L.; Qin, Z.; Chen, Q.; Zhao, L. Antioxidant properties of a vegetable-fruit beverage fermented with two Lactobacillus plantarum strains. Food Sci. Biotechnol. 2018, 27, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Valero-Cases, E.; Nuncio-Jauregui, N.; Frutos, M.J. Influence of fermentation with different lactic acid bacteria and in vitro digestion on the biotransformation of phenolic compounds in fermented pomegranate juices. J. Agric. Food Chem. 2017, 65, 6488–6496. [Google Scholar] [CrossRef]
- Zhao, D.; Shah, N.P. Lactic acid bacterial fermentation modified phenolic composition in tea extracts and enhanced their antioxidant activity and cellular uptake of phenolic compounds following in vitro digestion. J. Funct. Foods 2016, 20, 182–194. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, X.L.; Sun, Y.; Hu, B.; Sun, Y.; Jabbar, S.; Zeng, X.X. Fermentation in vitro of EGCG, GCG and EGCG300″Me isolated from Oolong tea by human intestinal microbiota. Food Res. Int. 2013, 54, 1589–1595. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Miao, Y.; Cao, J.; Wu, Z.; Weng, P. The modulatory effect of (-)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3’’Me) on intestinal microbiota of high fat diet-induced obesity mice model. Food Res. Int. 2017, 92, 9–16. [Google Scholar] [CrossRef]
- Yang, Y.; Qiao, L.; Zhang, X.; Wu, Z.; Weng, P. Effect of methylated tea catechins from Chinese oolong tea on the proliferation and differentiation of 3T3-L1 preadipocyte. Fitoterapia 2015, 104, 45–49. [Google Scholar] [CrossRef]
- Cueva, C.; Moreno-Arribas, M.V.; Martin-Alvarez, P.J.; Bills, G.; Vicente, M.F.; Basilio, A.; Rivas, C.L.; Requena, T.; Rodriguez, J.M.; Bartolome, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Fessard, A.; Kapoor, A.; Patche, J.; Assemat, S.; Hoarau, M.; Bourdon, E.; Bahorun, T.; Remize, F. Lactic fermentation as an efficient tool to enhance the antioxidant activity of tropical fruit juices and teas. Microorganisms 2017, 5, 23. [Google Scholar] [CrossRef]
- Filannino, P.; Cavoski, I.; Thlien, N.; Vincentini, O.; De Angelis, M.; Silano, M.; Gobbetti, M.; Di Cagno, R. Lactic acid fermentation of cactus cladodes (Opuntia ficus-indica L.) generates flavonoid derivatives with antioxidant and anti-Inflammatory properties. PLoS ONE 2016, 11. [Google Scholar]
- Di Cagno, R.; Filannino, P.; Vincentini, O.; Lanera, A.; Cavoski, I.; Gobbetti, M. Exploitation of Leuconostoc mesenteroides strains to improve shelf life, rheological, sensory and functional features of prickly pear (Opuntia ficus-indica L.) fruit puree. Food Microbiol. 2016, 59, 176–189. [Google Scholar] [CrossRef] [PubMed]
- Bhat, B.; Bajaj, B.K. Hypocholesterolemic potential and bioactivity spectrum of an exopolysaccharide from a probiotic isolate Lactobacillus paracasei M7. Bioact. Carbohydr. Diet. Fibre 2019, 19, 100191. [Google Scholar] [CrossRef]
- Han, J.; Xu, X.; Gao, C.; Liu, Z.; Wu, Z. Levan-producing Leuconostoc citreum strain BD1707 and its growth in tomato juice supplemented with sucrose. Appl. Environ. Microbiol. 2016, 82, 1383–1390. [Google Scholar] [CrossRef]
- Panda, S.K.; Behera, S.K.; Qaku, X.W.; Sekar, S.; Ndinteh, D.T.; Nanjundaswamy, H.M.; Ray, R.C.; Kayitesi, E. Quality enhancement of prickly pears (Opuntia sp.) juice through probiotic fermentation using Lactobacillus fermentum—ATCC 9338. LWT Food Sci. Technol. 2017, 75, 453–459. [Google Scholar] [CrossRef]
- Koh, W.Y.; Uthumporn, U.; Rosma, A.; Irfan, A.R.; Park, Y.H. Optimization of a fermented pumpkin-based beverage to improve Lactobacillus mali survival and α-glucosidase inhibitory activity: A response surface methodology approach. Food Sci. Hum. Wellness 2018, 7, 57–70. [Google Scholar] [CrossRef]
- Gamboa-Gómez, C.I.; Simental-Mendía, L.E.; González-Laredo, R.F.; Alcantar-Orozco, E.J.; Monserrat-Juarez, V.H.; Ramírez-Espana, J.C.; Gallegos-Infante, J.A.; Moreno-Jiménez, M.R.; Rocha-Guzmán, N.E. In vitro and in vivo assessment of anti-hyperglycemic and antioxidant effects of Oak leaves (Quercus convallata and Quercus arizonica) infusions and fermented beverages. Food Res. Int. 2017, 102, 690–699. [Google Scholar] [CrossRef]
- Yan, Y.; Zhang, F.; Chai, Z.; Liu, M.; Battino, M.; Meng, X. Mixed fermentation of blueberry pomace with L. rhamnosus GG and L. plantarum-1: Enhance the active ingredient, antioxidant activity and health-promoting benefits. Food Chem. Toxicol. 2019, 131, 110541. [Google Scholar] [CrossRef]
- Harima-Mizusawa, N.; Kano, M.; Nozaki, D.; Nonaka, C.; Miyazaki, K.; Enomoto, T. Citrus juice fermented with Lactobacillus plantarum YIT 0132 alleviates symptoms of perennial allergic rhinitis in a double-blind, placebo-controlled trial. Benef. Microbes 2016, 7, 649–658. [Google Scholar] [CrossRef]
- Valero-Cases, E.; Roy, N.C.; Frutos, M.J.; Anderson, R.C. Influence of the fruit juice carriers on the ability of Lactobacillus plantarum DSM20205 to improve in vitro intestinal barrier integrity and its probiotic properties. J. Agric. Food Chem. 2017, 65, 5632–5638. [Google Scholar] [CrossRef]
- Martins de Sá, L.; Castro, P.; Lino, F.; Bernardes, M.; Viegas, J.; Dinis, T.; Santana, M.; Romao, W.; Vaz, G.B.; Lião, L.; et al. Antioxidant potential and vasodilatory activity of fermented beverages of jabuticaba berry (Myrciaria jaboticaba). J. Funct. Foods 2014, 8, 169–179. [Google Scholar] [CrossRef]
- Cheng, Y.; Wu, T.; Chu, X.; Tang, S.; Cao, W.; Liang, F.; Fang, Y.; Pan, S.; Xu, X. Fermented blueberry pomace with antioxidant properties improves fecal microbiota community structure and short chain fatty acids production in an in vitro mode. LWT Food Sci. Technol. 2020, 125, 109260. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, M.; Shi, Y.; Lu, T.; Xu, W.; Sun, Y.; Yang, L.; Gan, Z.; Xie, L. Effects of a fermented beverage of Changbai Mountain fruit and vegetables on the composition of gut microbiota in mice. Plant Foods Hum. Nutr. 2019, 74, 468–473. [Google Scholar] [CrossRef] [PubMed]

| Legume Fermented Beverage | Probiotic Bacteria | Results | Reference |
|---|---|---|---|
| Soy milk with inulin and okara flour | L. acidophilus La-5, B. animalis Bb-12 | Probiotic viability above the minimum recommended after 28 days of storage, high probiotic viability after in vitro gastrointestinal digestion | Bedani et al. [29] |
| Soy milk with green gram | L. acidophilus NCDC14 | High probiotic viability after fermentation, high sensory acceptability score | Mridula and Sharma [30] |
| Peanut-soy milk | P. acidilactici UFLA BFFCX 27.1, L. lactis CCT 0360, L. rhamnosus LR 32, L. acidophilus LACA 4 | High probiotic viability after fermentation, high acid lactic contents | Santos et al. [31] |
| Soya flour, alfalfa meal, barley sprouts | Different LAB, mostly Lactobacillus genus | Enrichment of the gut microbiota population | Cabello-Olmo et al. [32] |
| Soy milk | L. casei Shirota | Beneficial modulation of the gut microbiota | Nagino et al. [34] |
| Chickpeas | S. thermophilus, L. bulgaricus, L. acidophilus | High probiotic viability after fermentation | Wang et al. [35] |
| Germinated and ungerminated cereals and legumes: barley, ragi, moth bean, soybean | L. acidophilus | High probiotic viability after fermentation, good sensory acceptability | Chavan et al. [36] |
| Cereal Fermented Beverage | Probiotic Bacteria | Results | Reference |
|---|---|---|---|
| Oat | Human fecal cultures | Increase in SCFAs 1, increase of healthy intestinal bacteria, reduction of harmful bacteria | Kedia et al. [39] |
| Oat | Different Lactobacillus strains | High levels of antioxidant capacity, high polyphenols content, increase of β-glucan content during fermentation, decrease in the hydrolysis index of starch, high probiotic viability | Luana et al. [40], Bernat et al. [41], Gupta et al. [42], Gupta and Bajaj [43], Kedia et al. [44], Wang et al. [45] |
| Oat | L. curvatus P99 | High probiotic viability after in vitro digestion, antimicrobial activity, blocking of the adhesion of pathogenic bacteria to epithelial cells, autoaggregation capacity | Funck et al. [46] |
| Oat | L. plantarum DSM9843 | In vivo study: increase of lactic, acetic and propionic acid, high probiotic viability after digestion; decrease in flatulence; and softer stool consistency | Johansson et al. [47] |
| Rice | L. fermentum KKL1 | Strong antioxidant capacity, glucoamylase and α-amylase production, phytase activity, high hydrosoluble vitamins, antibiotic susceptibility | Ghosh et al. [48] |
| Rice | L. plantarum L7 | High antioxidant capacity; increase in lactic, succinic, and acetic acid during fermentation; high probiotic viability after in vitro digestion; antibiotic susceptibility; antimicrobial activities; increase in minerals and phytase activity | Giri et al. [49] |
| Maize | L. paracasei LBC-81 with S. cerevisiae CCMA 0731 and L. paracasei LBC-81 S. cerevisiae CCMA 0732 | Probiotic viability above the minimum recommended, high production of lactic acid during fermentation, 70 volatile compounds identified | Menezes et al. [51] |
| Kwete | L. rhamnosus yoba 2012 and S. thermophilus C106 | Decrease in aflatoxins content = detoxification of beverage | Wacoo et al. [52] |
| Cereals mixing | Mixed or single LAB | Malt was the best substrate for microbial growth used as single or mixed beverages | Rathore et al. [5] |
| Rice and maize | L. acidophilus LACA 4 and L. pantarum CCMA 0743 supplemented with FOS | High probiotic viability, increase in lactic and acetic acids during fermentation, good sensorial acceptance | Freire et al. [53] |
| Pseudocereal Fermented Beverage | Probiotic Bacteria | Results | Reference |
|---|---|---|---|
| Two quinoa varieties (Rosada de Huancayo, Pasankalla) | L. plantarum Q823, L. casei Q11, L. lactis ARH74 | Probiotic viability above the minimum recommended after 28 days of storage | Ludena-Urquizo et al. [57] |
| Quinoa | L. plantarum Q823 | High probiotic viability after the gastrointestinal digestion | Vera-Pingitore et al. [58] |
| Aqueous extracts of soybean and quinoa grains with FOS | L. casei Lc-01 | Positive modulation of the gut microbiota, decrease in toxic elements (ammonia) | Bianchi et al. [59] |
| Mashed buckwheat previously fermented with LAB | L. rhamnosus GG | High probiotic viability after 14 days of cold storage, good sensorial acceptance | Matejčeková et al. [62] |
| Buckwheat, dark buckwheat | L. rhamnosus GG | Probiotic viability above the minimum recommended after 21 days of cold storage | Kocková and Valík [63] |
| Amaranth flour, amaranth grain, buckwheat flour, whole buckwheat flour | L. rhamnosus GG | High probiotic viability after 21 days of storage, except for the beverage with whole buckwheat flour | Kocková et al. [64] |
| Fruits and Vegetables Fermented Beverage | Probiotic Bacteria | Results | Reference |
|---|---|---|---|
| Synbiotic beverages: Carrot–orange juices and nectars + inulin | Different LAB 1 strains | Prebiotic ingredients + vegetable beverage matrices = good compatibility Prebiotic supplementation = viability of probiotic strains above the minimum recommended. High viability of probiotic strains after gastrointestinal in vitro digestion | Valero-Cases and Frutos [67,68], Mantzourani et al. [69], Mantzourani et al. [70], Pimentel et al. [71], Miranda et al. [73], Bernal-Castro et al. [74] |
| Orange + hibiscus tea + oligofructose | |||
| Red fruit beverage + FOS, GOS, Inulin | |||
| Pomegranate + Cornelian cherry + delignified wheat bran | |||
| Clarified apple juice + oligofructose | |||
| Pomegranate + inulin | Three species of Lactobacillus | In vivo study: improve testosterone level, insulin, insulin resistance, body mass index, weight and waist circumference in polycystic ovarian syndrome | Esmaeilinezhad et al. [75] |
| Apple juice | L. plantarum ATCC14917 | Improved antioxidant capacity, increasing quercetin phloretin and 5-O-caffeoylquinic acid contents | |
| Mixed beverages from apples, carrots and pears | L. plantarum 115 and Vege Start 60 | Increase of total flavonoids content and the antioxidant activity | Yang et al. [77] |
| Pomegranate juices | L.plantarum CECT220, L.acidophilus CECT903, B.longum subsp. infantis CECT4551, B.bifidum CECT870 | The biotransformation of phenolic compounds during fermentation and gastrointestinal digestion, suggesting a prebiotic effect | Li et al. [76] |
| Tea infusion | L. plantarum ASCC276, L. plantarum ASCC292, L. acidhophilus CSCC2400, L.plantarum WCFSI, L. rhamnosus WQS, L. brevis NPS-QW145 | Increase in the antioxidant capacity, modification of the phenolic composition, increase in cellular uptake after in vitro gastrointestinal digestion | Zhao and Shah [79] |
| Oolong tea | Fecal bacteria | Improved the host health generating SCFAs 2 and modulating the human gut microbiota, anti-obesity therapy | Zhang et al. [80] |
| Prickly pear | Leuconostoc mesenteroides strains | Production of EPS 3, improvement of consistency and antioxidant capacity | Di Cagno et al. [86] |
| Prickly pear | L. fermentum ATCC 9338 | Decomposition of some risky organic compounds present in the fresh juice like: 2-propenenitrile, 2-(acetyloxy); furfuryl alcohol; acetaldehyde; 2,2-diethyl-3-methyloxazolidine, 4h-Pyran-4-one, 3,5-dihydroxy-2-methyl and furan | Panda et al. [89] |
| Pumpkin | L. mali K8 | α-glucosidase inhibitory activity with anti-hyperglycemic effect | Koh et al. [90] |
| Infusion of oak leaves | Kombucha culture | Anti-hyperglycemic effect and antioxidant activity | Gamboa-Gómez et al. [91] |
| Blueberry pomace | L. rhamnosus GG. L. plantarum-1 and L. plantarum-2 | Hypocholesterolemic and anti-fatigue effect | Yan et al. [92] |
| Citrus | L. plantarum YIT0132 | Good effect in relieving perennial allergic rhinitis symptoms | Harima-Mizusawa et al. [93] |
| Tomato, feijoa, blueberry-blackberry, cactus pear, and prickly pear fruits | Different LAB strains | Great in vitro anti-inflammatory capacity and help to maintain the integrity of intestinal barrier | Di Cagno et al. [86], Valero-Cases et al. [94], Filannino et al. [85] |
| Blueberry pomace | L. casei CICC20280 | Increase of SCFA production and an improvement of fecal microbiota | Cheng et al. [96] |
| Changbai Mountain vegetables and fruits | Naturalized species of bacteria | Reduction of Firmicutes/Bacteroidetes ratio, increase of Bacteroidales S24–7 group, Bacteroidaceae, genus Bacteroides and Prevotellaceae in a mouse model study | Wang et al. [97] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valero-Cases, E.; Cerdá-Bernad, D.; Pastor, J.-J.; Frutos, M.-J. Non-Dairy Fermented Beverages as Potential Carriers to Ensure Probiotics, Prebiotics, and Bioactive Compounds Arrival to the Gut and Their Health Benefits. Nutrients 2020, 12, 1666. https://doi.org/10.3390/nu12061666
Valero-Cases E, Cerdá-Bernad D, Pastor J-J, Frutos M-J. Non-Dairy Fermented Beverages as Potential Carriers to Ensure Probiotics, Prebiotics, and Bioactive Compounds Arrival to the Gut and Their Health Benefits. Nutrients. 2020; 12(6):1666. https://doi.org/10.3390/nu12061666
Chicago/Turabian StyleValero-Cases, Estefanía, Débora Cerdá-Bernad, Joaquín-Julián Pastor, and María-José Frutos. 2020. "Non-Dairy Fermented Beverages as Potential Carriers to Ensure Probiotics, Prebiotics, and Bioactive Compounds Arrival to the Gut and Their Health Benefits" Nutrients 12, no. 6: 1666. https://doi.org/10.3390/nu12061666
APA StyleValero-Cases, E., Cerdá-Bernad, D., Pastor, J.-J., & Frutos, M.-J. (2020). Non-Dairy Fermented Beverages as Potential Carriers to Ensure Probiotics, Prebiotics, and Bioactive Compounds Arrival to the Gut and Their Health Benefits. Nutrients, 12(6), 1666. https://doi.org/10.3390/nu12061666






