New Evaluation of Isoflavone Exposure in the French Population
Abstract
1. Introduction
2. Materials and Methods
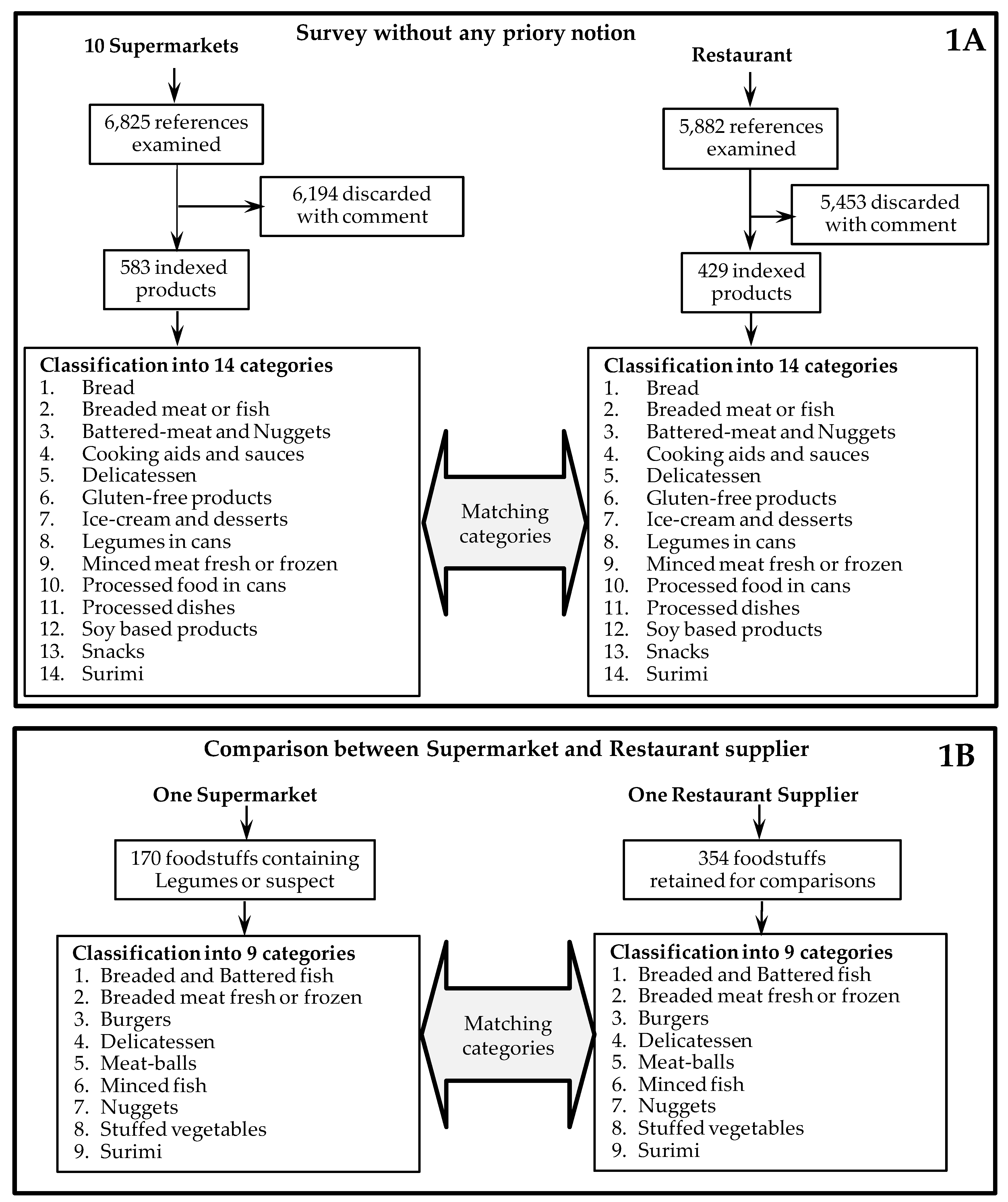
2.1. Supermarket Study
2.2. Restaurant Supplier Study
2.3. Isoflavone Determination in Selected Foodstuffs
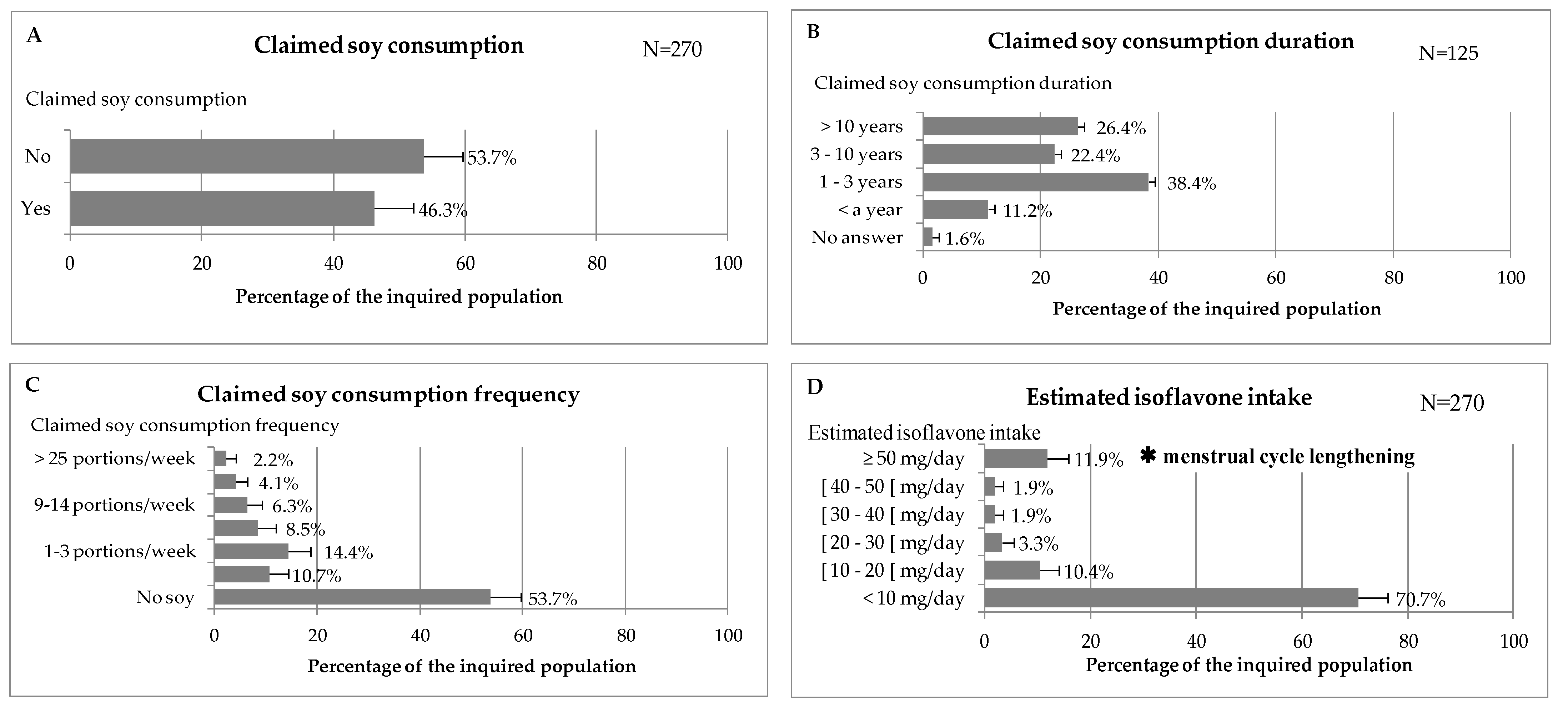
2.4. Survey on French Premenopausal Women
2.5. Statistical Analysis
3. Results
3.1. Identification of Foodstuffs from Supermarkets Containing Legumes
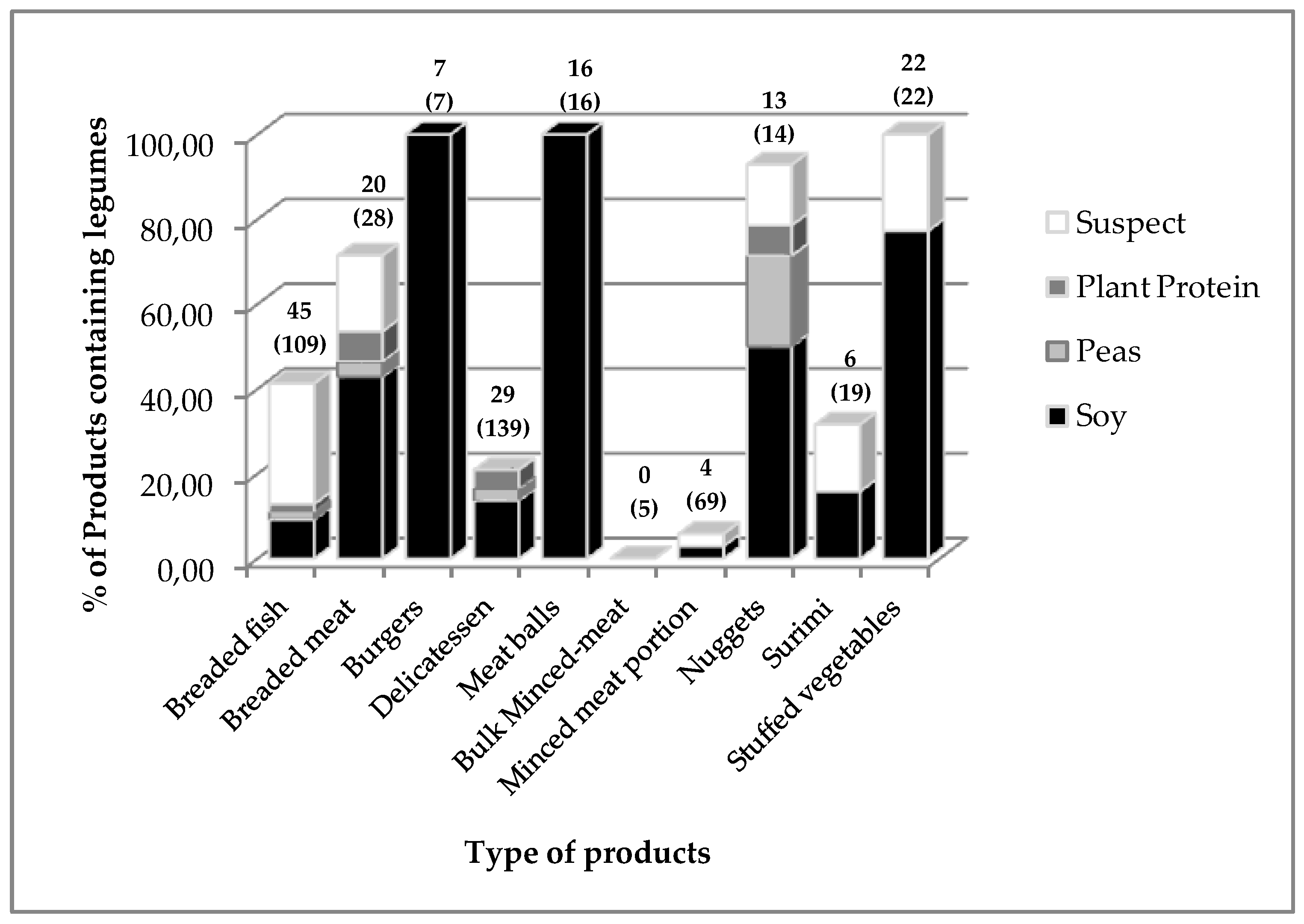
3.2. Respective Proportion of Each Selected Legume in the Main Foodstuff Categories
3.3. Study by Price
3.4. Theoretical Exposure to Isoflavones from Canned Vegetable Preparations
3.5. Assays Performed on Some Foodstuffs from the Supermarket Offerings
3.6. Analysis of the Restaurant-Supplier Website
3.7. Comparison of the Two Distribution Circuits
3.8. Consumption Survey and Isoflavone Exposure
4. Discussion
4.1. Presence of Pulses in Processed Food
4.1.1. Under-Estimation of Pulses and Isoflavone Exposure
4.1.2. The Differences between the Distribution Circuits
4.1.3. The Price Analysis
4.2. Analysis of the Premenopausal Women Survey
4.3. Consequences of Plant Protein Incorporation in Processed Food
4.3.1. Technological and Nutritional Arguments
4.3.2. Allergenic Effects
4.3.3. Soy Phytoestrogen Content and Health Effects
4.4. Limitation of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bradbury, R.B.; White, D.E. Estrogens and related compounds in plants. Vit. Horm. 1954, 12, 207–233. [Google Scholar]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment, Nitric oxide synthase-mediated phytoalexin accumulation in soybean cotyledons in response to the Diaporthe phaseolorum f. sp. meridionalis elicitor. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef] [PubMed]
- Modolo, L.V.; Cunha, F.Q.; Braga, M.R.; Salgado, I. Nitric Oxide Synthase-Mediated Phytoalexin Accumulation in Soybean Cotyledons in Response to the Diaporthe phaseolorum f. sp. meridionalis Elicitor. Plant Physiol. 2002, 130, 1288–1297. [Google Scholar] [CrossRef] [PubMed]
- el-Adawy, T.A. Effect of soaking process on nutritional quality and protein solubility of some legume seeds. Plant Foods Hum. Nutr. 2002, 57, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Dia, V.P.; Gomez, T.; Vernaza, G.; Berhow, M.; Chang, Y.K.; de Mejia, E.G. Bowman-Birk and Kunitz protease inhibitors among antinutrients and bioactives modified by germination and hydrolysis in Brazilian soybean cultivar BRS 133. J. Agric. Food Chem. 2012, 60, 7886–7894. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, T.; Ono, T.; Wada, T.; Tsukamoto, C.; Kono, Y. Changes in soybean phytate content as a result of field growing conditions and influence on tofu texture. Biosci. Biotech. Biochem. 2006, 70, 874–880. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hubert, J.; Berger, M.; Daydé, J. Use of a simplified HPLC-UV analysis for soyasaponin B determination: Study of saponin and isoflavone variability in soybean cultivars and soy-based health food products. J. Agric. Food Chem. 2005, 53, 3923–3930. [Google Scholar] [CrossRef] [PubMed]
- Shutt, D.A. The effects of plant estrogens on animal reproduction. Endeavour 1976, 35, 110–113. [Google Scholar] [CrossRef]
- Latonnelle, K.; Le Menn, F.; Kaushik, S.J.; Bennetau-Pelissero, C. Effects of dietary phytoestrogens in vivo and in vitro in rainbow trout and Siberian sturgeon: Interests and limits of the in vitro studies of interspecies differences. Gen. Comp. Endocrinol. 2002, 126, 39–51. [Google Scholar] [CrossRef]
- National Toxicology Program. Multigenerational reproductive study of genistein (Cas No. 446-72-0) in Sprague-Dawley rats (feed study). Nat. Toxicol. Prog. Tech. Rep. Ser. 2008, 539, 1–266. [Google Scholar]
- National Toxicology Program. Toxicology and carcinogenesis studies of genistein (Cas No. 446-72-0) in Sprague-Dawley rats (feed study). Nat. Toxicol. Prog. Tech. Rep. Ser. 2008, 545, 1–240. [Google Scholar]
- Cassidy, A.; Bingham, S.; Setchell, K.D.R. Biological effects of a diet of soy protein rich in isoflavones on the menstrual cycle of premenopausal women. Am. J. Clin. Nutr. 1994, 60, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Bennetau-Pelissero, C. Positive or negative effects of isoflavones: Toward the end of a controversy. Food Chem. 2017, 225, 293–301. [Google Scholar] [CrossRef] [PubMed]
- OECD. OECD guideline for the testing of chemicals. In Test No. 440: Uterotrophic Bioassay in Rodents. A Short-Term Screening Test for Estrogenic Properties; OCDE Publication: Paris, France, 2007; 21p. [Google Scholar] [CrossRef]
- Fernandez-Lopez, A.; Lamothe, V.; Delample, M.; Denayrolles, M.; Bennetau-Pelissero, C. Removing isoflavones from modern soyfood: Why and how? Food Chem. 2016, 210, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Zhu, T.; Parray, A.; Siddique, H.R.; Yang, W.; Saleem, M.; Bosland, M.C. Differential effects of genistein on prostate cancer cells depend on mutational status of the androgen receptor. PLoS ONE 2013, 8, e78479. [Google Scholar] [CrossRef]
- Sathyapalan, T.; Manuchehri, A.M.; Thatcher, N.J.; Rigby, A.S.; Chapman, T.; Kilpatrick, E.S.; Atkin, S.L. The effect of soy phytoestrogen supplementation on thyroid status and cardiovascular risk markers in patients with subclinical hypothyroidism: A randomized, double-blind, crossover study. J. Clin. Endocrinol. Metab. 2011, 96, 1442–1449. [Google Scholar] [CrossRef] [PubMed]
- Phrakonkham, P.; Brouland, J.P.; Saad Hel, S.; Bergès, R.; Pimpie, C.; Pocard, M.; Canivenc-Lavier, M.C.; Perrot-Applanat, M. Dietary exposure in utero and during lactation to a mixture of genistein and an anti-androgen fungicide in a rat mammary carcinogenesis model. Reprod. Toxicol. 2015, 54, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Patisaul, H.B. Endocrine disruption by dietary phytoestrogens: Impact on dimorphic sexual systems and behaviours. Proc. Nutr. Soc. 2016, 8, 1–15. [Google Scholar]
- Bennetau-Pelissero, C. Risks and benefits of phytoestrogens: Where are we now? Curr. Opin. Clin. Nutr. Metab. Care. 2016, 19, 477–483. [Google Scholar] [CrossRef]
- Afssa-Afssaps. Sécurité et Bénéfices des Phyto-Estrogènes Apportés par L’alimentation—Recommandations, Afssa edition France; Afssa Repport; ANSES: Maison Alfort, France, 2005; 440p.
- Vergne, S.; Bennetau-Pelissero, C.; Lamothe, V.; Chantre, P.; Potier, M.; Asselineau, J.; Perez, P.; Durand, M.; Moore, N.; Sauvant, P. Higher bioavailability of isoflavones after a single ingestion of a soya-based supplement than a soya-based food in young healthy males. Br. J. Nutr. 2008, 99, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Le Houérou, C.; Bennetau-Pelissero, C.; Lamothe, V.; Le Menn, F.; Babin, P.; Bennetau, B. Synthesis of novel hapten-protein conjugates for production of highly specific antibodies to formononetin, daidzein and genistein. Tetrahedron 2000, 56, 295–301. [Google Scholar] [CrossRef]
- Bennetau-Pelissero, C.; Le Houérou, C.; Lamothe, V.; Le Menn, F.; Babin, P.; Bennetau, B. Synthesis of haptens and conjugates for ELISAs of phytoestrogens. Development of the immunological tests. J. Agric. Food. Chem. 2000, 48, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Shinkaruk, S.; Durand, M.; Lamothe, V.; Carpaye, A.; Martinet, A.; Chantre, P.; Vergne, S.; Nogues, X.; Moore, N.; Bennetau-Pelissero, C. Bioavailability of glycitein relatively to other soy isoflavones in healthy young Caucasian men. Food Chem. 2012, 35, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Ardilly, P. Les Techniques de Sondage; Technip: London, UK, 2006. [Google Scholar]
- Conso glob. Les chiffres du Kebab en France. Available online: https://www.planetoscope.com/restauration/1736-consommation-de-kebabs-en-france.html (accessed on 25 July 2017).
- ANSES. Etude de L’alimentation Totale Infantile; 376 Anses Edition France; Anses Repport; ANSES: Maison Alfort, France, 2016; Volume 2, p. 284.
- Bhagwat, S.; Haytowitz, D.B.; Holden, J.M. USDA Database for the Isoflavone Content of Selected Foods. 2008. Available online: https://www.ars.usda.gov/ARSUserFiles/80400525/Data/isoflav/ Isoflav_R2-1.pdf (accessed on 19th July 2019).
- Ward, H.A.; Kuhnle, G.G.; Mulligan, A.A.; Lentjes, M.A.; Luben, R.N.; Khaw, K.T. Breast, colorectal, and prostate cancer risk in the European Prospective Investigation into Cancer and Nutrition-Norfolk in relation to phytoestrogen intake derived from an improved database. Am J. Clin. Nutr. 2010, 91, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Clarke, D.B.; Lloyd, A.S.; Lawrence, J.M.; Brown, J.E.; Storey, L.; Raats, M.; Rainsbury, R.M.; Culliford, D.J.; Bailey-Horne, V.A.; Parry, B.M. Development of a food compositional database for the estimation of dietary intake of phytoestrogens in a group of postmenopausal women previously treated for breast cancer and validation with urinary excretion. Brit. J. Nutr. 2013, 109, 2261–2268. [Google Scholar] [CrossRef] [PubMed]
- Horn-Ross, P.L.; John, E.M.; Lee, M.; Stewart, S.L.; Koo, J.; Sakoda, L.C.; Shiau, A.C.; Goldstein, J.; Davis, P.; Perez-Stable, E.J. Phytoestrogen consumption and breast cancer risk in a multiethnic population: The Bay Area Breast Cancer Study. Am. J. Epidemiol. 2001, 154, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Bai, W.; Wang, C.; Ren, C. Intakes of total and individual flavonoids by US adults. Int. J. Food Sci. Nutr. 2014, 65, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, D.; Debeuf, C.; Joubrel, G.; Kocken, M.; Planchenault, N. Les aliments au soja: Consommation en France, qualités nutritionnelles et données scientifiques récentes sur la santé. OCL 2016, 23, D405–D420. [Google Scholar]
- Degen, G.H.; Blaszkewicz, M.; Shi, L.; Buyken, A.E.; Remer, T. Urinary isoflavone phytoestrogens in German children and adolescents—A longitudinal examination in the DONALD cohort. Mol. Nutr. Food Res. 2011, 55, 359–367. [Google Scholar] [CrossRef]
- Berman, T.; Goldsmith, R.; Göen, T.; Spungen, J.; Novack, L.; Levine, H.; Amitai, Y.; Shohat, T.; Grotto, I. Urinary concentrations of environmental contaminants and phytoestrogens in adults in Israel. Environ. Int. 2013, 59, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Mumford, S.L.; Kim, S.; Chen, Z. Urinary phytoestrogens are associated with subtle indicators of semen quality among male partners of couples desiring pregnancy. J. Nutr. 2015, 145, 2535–2541. [Google Scholar] [CrossRef] [PubMed]
- Wada, K.; Tsuji, M.; Tamura, T.; Konishi, K.; Kawachi, T.; Hori, A.; Tanabashi, S.; Matsushita, S.; Tokimitsu, N.; Nagata, C. Soy isoflavone intake and stomach cancer risk in Japan: From the Takayama study. Int. J. Cancer. 2015, 137, 885–892. [Google Scholar] [CrossRef]
- Baglia, M.L.; Gu, K.; Zhang, X.; Zheng, Y.; Peng, P.; Cai, H.; Bao, P.P.; Zheng, W.; Lu, W.; Shu, X.O. Soy isoflavone intake and bone mineral density in breast cancer survivors. Cancer Causes Control. 2015, 26, 571–580. [Google Scholar] [CrossRef]
- Mathey, J.; Lamothe, V.; Coxam, V.; Potier, M.; Sauvant, P.; Bennetau-Pelissero, C. Concentrations of isoflavones in plasma and urine of post-menopausal women chronically ingesting high quantities of soy isoflavones. J. Pharm. Biomed. Anal. 2006, 41, 957–965. [Google Scholar] [CrossRef]
- Kumar, N.B.; Cantor, A.; Allen, K.; Riccardi, D.; Cox, C.E. The specific role of isoflavones on estrogen metabolism in premenopausal women. Cancer 2002, 94, 1166–1174. [Google Scholar] [CrossRef] [PubMed]
- Bennetau-Pelissero, C.; Sauvant, P.; Peltre, G.; Auriol, P.; Rocca, A.; Rancé, F. Phyto-oestrogènes du soja: Problèmes posés chez le nourrisson allergique au lait de vache et consommant des formules à base de soja. Cah. Nutr. Diét. 2004, 39, 1. [Google Scholar] [CrossRef]
- Singh, P.; Kumar, R.; Sabapathy, S.N.; Bawa, A.S. Functional and Edible Uses of Soy Protein Products. Comp. Rev. Food Sci. Food Saf. 2008, 7, 15–28. [Google Scholar] [CrossRef]
- Tömösközi, S.; Lásztity, R.; Haraszi, R.; Baticz, O. Isolation and study of the functional properties of pea proteins. Nahrung 2001, 45, 399–401. [Google Scholar] [CrossRef]
- Aluko, R.E.; Mofolasayo, O.A.; Watts, B.M. Emulsifying and foaming properties of commercial yellow pea (Pisum sativum L.) seed flours. J. Agric. Food Chem. 2009, 57, 9793–9800. [Google Scholar] [CrossRef]
- Edelman, M.; Colt, M. Nutrient Value of Leaf vs. Seed. Front. Chem. 2016, 4, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Tosh, S.M.; Yada, S. Dietary fibres in pulse seeds and fractions: Characterization, functional attributes, and applications. Food Res. Int. 2010, 43, 450–460. [Google Scholar] [CrossRef]
- Efsa. Regulation (eu) no 1169/2011 of the European parliament and of the council. Off. J. Eur. Union 2011, L 304/18, 18–63. [Google Scholar]
- Turnbull, J.L.; Adams, H.N.; Gorard, D.A. Review article: The diagnosis and management of food allergy and food intolerances. Alim. Pharmacol. Ther. 2015, 41, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Holzhauser, T.; Wackermann, O.; Ballmer-Weber, B.K.; Bindslev-Jensen, C.; Scibilia, J.; Perono-Garoffo, L.; Utsumi, S.; Poulsen, L.K.; Vieths, S. Soybean (Glycine max) allergy in Europe: Gly m 5 (beta-conglycinin) and Gly m 6 (glycinin) are potential diagnostic markers for severe allergic reactions to soy. J. Allergy Clin. Immunol. 2009, 123, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Foucard, T.; Malmheden Yman, I. A study on severe food reactions in Sweden--is soy protein an underestimated cause of food anaphylaxis? Allergy 1999, 54, 261–265. [Google Scholar] [CrossRef]
- Yagami, A.; Inaba, Y.; Kuno, Y.; Suzuki, K.; Tanaka, A.; Sjolander, S.; Saito, H.; Matsunaga, K. Two cases of pollen-food allergy syndrome to soy milk diagnosed by skin prick test, specific serum immunoglobulin E and microarray analysis. J. Dermatol. 2009, 36, 50–55. [Google Scholar] [CrossRef]
- Ciccarelli, A.; Calabrò, C.; Imperatore, C.; Scala, G. Prick by prick induced anaphylaxis in a patient with peanuts and lupine allergy: Awareness of risks and role of component resolved diagnosis. Case Rep. Med. 2014, 2014, 892394. [Google Scholar] [CrossRef] [PubMed]
- Senna, G.E.; Crivellaro, M.; Bonadonna, P.; Dama, A.; Mezzelani, P.; Passalacqua, G. Pizza, an unsuspected source of soybean allergen exposure. Allergy 1998, 53, 1106–1107. [Google Scholar] [CrossRef]
- Gomez-Andre, S.A.; Deschildre, A.; Bienvenu, F.; Just, J. Soybean, an emerging allergy. Rev. Fra. Allergol. 2012, 52, 448–453. [Google Scholar] [CrossRef]
- Franck, P.; Moneret-Vautrin, D.A.; Dousset, B.; Kanny, G.; Nabet, P.; Guénard-Bilbaut, L.; Parisot, L. The allergenicity of soybean-based products is modified by food technologies. Int. Arch. Allergy Immunol. 2002, 128, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Seppen, J. A diet containing the soy phytoestrogen genistein causes infertility in female rats partially deficient in UDP glucuronyltransferase. Biochim. Toxicol. App. Pharmacol. 2012, 264, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Takatsuka, N.; Inaba, S.; Kawakami, N.; Shimizu, H. Effect of soymilk consumption on serum estrogen concentrations in premenopausal Japanese women. J. Natl. Cancer Inst. 1998, 90, 1830–1835. [Google Scholar] [CrossRef]
- Watanabe, S.; Terashima, K.; Sato, Y.; Arai, S.; Eboshida, A. Effects of isoflavone supplement on healthy women. Biofactors 2000, 12, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Ryder, J.J.; Kurzer, M.S.; Lampe, J.W.; Messina, M.J.; Phipps, W.R.; Cassidy, A. Effects of soy protein and isoflavones on circulating hormone concentrations in pre- and post-menopausal women: A systematic review and meta-analysis. Hum. Reprod. Update 2009, 15, 423–440. [Google Scholar] [CrossRef]
- Chavarro, J.E.; Toth, T.L.; Sadio, S.M.; Hauser, R. Soy food and isoflavone intake in relation to semen quality parameters among men from an infertility clinic. Hum. Reprod. 2008, 23, 2584–2590. [Google Scholar] [CrossRef]
- Toshima, H.; Suzuki, Y.; Imai, K.; Yoshinaga, J.; Shiraishi, H.; Mizumoto, Y.; Hatakeyama, S.; Onohara, C.; Tokuoka, S. Endocrine disrupting chemicals in urine of Japanese male partners of subfertile couples: A pilot study on exposure and semen quality. Int. J. Hyg. Environ. Health. 2012, 215, 502–506. [Google Scholar] [CrossRef]
- Xia, Y.; Chen, M.; Zhu, P.; Lu, C.; Fu, G.; Zhou, X.; Chen, D.; Wang, H.; Hang, B.; Wang, S.; et al. Urinary phytoestrogen levels related to idiopathic male infertility in Chinese men. Environ. Int. 2013, 59, 161–167. [Google Scholar] [CrossRef]
- Yuan, G.; Liu, Y.; Liu, G.; Wei, L.; Wen, Y.; Huang, S.; Guo, Y.; Zou, F.; Cheng, J. Associations between semen phytoestrogens concentrations and semen quality in Chinese men. Environ. Int. 2019, 129, 136–144. [Google Scholar] [CrossRef]
- Shike, M.; Doane, A.S.; Russo, L.; Cabal, R.; Reis-Filho, J.S.; Gerald, W.; Cody, H.; Khanin, R.; Bromberg, J.; Norton, L. The effects of soy supplementation on gene expression in breast cancer: A randomized placebo-controlled study. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef]
- Touillaud, M.; Gelot, A.; Mesrine, S.; Bennetau-Pelissero, C.; Clavel-Chapelon, F.; Arveux, P.; Bonnet, F.; Gunter, M.; Boutron-Ruault, M.C.; Fournier, A. Use of dietary supplements containing soy isoflavones and breast cancer risk among women aged >50 y: A prospective study. Am. J. Clin. Nutr. 2019, 109, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Möller, F.J.; Pemp, D.; Soukup, S.T.; Wende, K.; Zhang, X.; Zierau, O.; Muders, M.H.; Bosland, M.C.; Kulling, S.E.; Lehmann, L.; et al. Soy isoflavone exposure through all life stages accelerates 17β-estradiol-induced mammary tumor onset and growth, yet reduces tumor burden, in ACI rats. Arch. Toxicol. 2016, 90, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Boucher, B.A.; Cotterchio, M.; Anderson, L.N.; Kreiger, N.; Kirsh, V.A.; Thompson, L.U. Use of isoflavone supplements is associated with reduced postmenopausal breast cancer risk. Int. J. Cancer 2013, 132, 1439–1450. [Google Scholar] [CrossRef] [PubMed]
- Hydovitz, J.D. Occurrence of goiter in an infant on a soy diet. N. Engl. J. Med. 1960, 262, 351–353. [Google Scholar] [CrossRef]
- Ripp, J.A. Soybean-induced goiter. Am. J. Dis. Child. 1961, 102, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Bell, D.S.; Ovalle, F. Use of soy protein supplement and resultant need for increased dose of levothyroxine. Endocr. Pract. 2001, 7, 193–194. [Google Scholar] [CrossRef]
- Nakamura, Y.; Ohsawa, I.; Goto, Y.; Tsuji, M.; Oguchi, T.; Sato, N.; Kiuchi, Y.; Fukumura, M.; Inagaki, M.; Gotoh, H. Soy isoflavones inducing overt hypothyroidism in a patient with chronic lymphocytic thyroiditis: A case report. J. Med. Case Rep. 2017, 11, 253–258. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sathyapalan, T.; Köhrle, J.; Rijntjes, E.; Rigby, A.S.; Dargham, S.R.; Kilpatrick, E.S.; Atkin, S.L. The Effect of High Dose Isoflavone Supplementation on Serum Reverse T3 in Euthyroid Men With Type 2 Diabetes and Post-menopausal Women. Front. Endocrinol. (Lausanne) 2018, 9, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Auxietre, T.A.; Dumontier, M.F.; Balguy, I.; Frapart, Y.; Canivenc-Lavier, M.C.; Berges, R.; Boudalia, S.; Auger, J.; Corvol, M.T.; Savouret, J.F. Sub-NOAEL amounts of vinclozolin and xenoestrogens target rat chondrogenesis in vivo. Biochim. Biochim. 2014, 99, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Thongprakaisang, S.; Thiantanawat, A.; Rangkadilok, N.; Suriyo, T.; Satayavivad, J. Glyphosate induces human breast cancer cells growth via estrogen receptors. Food Chem. Toxicol. 2013, 59, 129–136. [Google Scholar] [CrossRef]
- Bennetau-Pelissero, C. Plant Proteins from Legumes. In Bioactive Molecules in Food; Mérillon, J.M., Ramawat, K., Eds.; Reference Series in Phytochemistry; Springer: Cham, Switzerland, 2019; pp. 223–265. [Google Scholar]
- Mantovani, D.; Cardozo Filho, L.; Santos, L.C.; de Souza, V.L.; Watanabe, C.S. Chromatographic quantification of isoflavone content from soy derivates using HPLC technique. J. Chromatogr. Sci. 2009, 47, 766–769. [Google Scholar] [CrossRef] [PubMed]
- Pieretti, M.M.; Chung, D.; Pacenza, R.; Slotkin, T.; Sicherer, S.H. Audit of manufactured products: Use of allergen advisory labels and identification of labeling ambiguities. J. Allergy Clin. Immunol. 2009, 124, 337–341. [Google Scholar] [CrossRef] [PubMed]
| Food Categories | Number of Foodstuffs | Total of Indexed Foodstuff | Products Claiming Containing Pulse | |||||
|---|---|---|---|---|---|---|---|---|
| Soy | Pea | Lupine | Broad Bean | Suspects | Traces | |||
| 1. Bread | 11 | - | - | 37 | 3 | 1 | 52 | 48 |
| 2. Breaded meat or fish | 12 | 14 | - | - | 3 | - | 29 | 26 |
| 3. Battered-meat and Nuggets | 5 | 10 | - | - | 6 | - | 23 | 15 |
| 4. Cooking aids and sauces | 22 | - | - | 1 | 9 | - | 32 | 23 |
| 5. Delicatessen | 11 | 10 | - | - | 2 | - | 26 | 21 |
| 6. Gluten-free products | 13 | 6 | 3 | - | 3 | - | 27 | 22 |
| 7. Ice-cream and desserts | 7 | 3 | - | - | 6 | - | 16 | 10 |
| 8. Legumes in cans | 0 | 33 | 4 | - | - | - | 54 | 37 |
| 9. Minced meat fresh or frozen | 52 | 7 | - | - | 5 | - | 65 | 59 |
| 10. Processed foods in can | 13 | 12 | 1 | - | - | - | 26 | 26 |
| 11. Processed dishes | 23 | 18 | - | 1 | 15 | - | 57 | 42 |
| 12. Soy based products | 138 | - | - | - | - | - | 138 | 138 |
| 13. Snacks | 12 | 9 | - | 1 | 10 | - | 32 | 22 |
| 14. Surimi | 4 | - | - | - | 2 | - | 6 | 4 |
| Total | 323 | 122 | 8 | 40 | 64 | 1 | 583 | 493 |
| Product Category | Supermarkets | Restaurants | Significance Products with Legumes vs. no Legumes | Significance Products with Soy vs. no Soy | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Products per Category | Products with Legumes or Suspect | % with Legumes or Suspect | Products with Soy | % with Soy | Products per Category | Products with Legumes or Suspect | % with Legumes or Suspect | Products with Soy | % with Soy | |||
| 1. Breaded and Battered fish | 14 | 1 | 7% | 0 | 0% | 109 | 45 | 41% | 10 | 9% | na | na |
| 2. Breaded meat fresh or frozen | 18 | 15 | 83% | 4 | 22% | 28 | 20 | 71% | 12 | 43% | ns | na |
| 3. Burgers fresh or frozen | 7 | 4 | 57% | 2 | 29% | 7 | 7 | 100% | 7 | 100% | na | na |
| 4. Delicatessen | 49 | 9 | 18% | 0 | 0% | 139 | 29 | 21% | 19 | 14% | ns | na |
| 6. Meatballs fresh or frozen | 10 | 10 | 100% | 5 | 50% | 16 | 16 | 100% | 16 | 100% | na | na |
| 7. Nuggets fresh or frozen | 24 | 19 | 19% | 2 | 8% | 14 | 13 | 93% | 7 | 50% | na | na |
| 8. Stuffed vegetables fresh or frozen | 5 | 4 | 80% | 3 | 60% | 22 | 22 | 100% | 17 | 77% | na | na |
| 9. Surimi * | 43 | 7 | 16% | 4 | 9% | 19 | 6 | 32% | 3 | 16% | p < 0.01 | na |
| Total number of products (%) | 170 | 69 | 41% | 20 | 11.76% | 354 | 158 | 45% | 91 | 25.71% | ns | p < 0.01 |
| Price-Quintiles for All Product Categories* | Pea | Soy | Plant Protein + Suspect | Total Legumes | No Legumes | Total | Significance Legume vs. no Legumes |
|---|---|---|---|---|---|---|---|
| Price 1 | 7 (15%) | 11 (23%) | 4 (9%) | 22 (48%) | 24 (52%) | 46 | na |
| Price 2 | 14 (27%) | 10 (19%) | 3 (6%) | 27 (51%) | 26 (49%) | 53 | |
| Price 3 | 6 (12%) | 1 (2%) | 4 (8%) | 11 (23%) | 37 (77%) | 48 | |
| Price 4 | 6 (13%) | 2 (4.3%) | 6 (13%) | 14 (32%) | 30 (68%) | 44 | |
| Price 5 | 1 (4%) | 3 (12%) | 4 (16%) | 8 (32%) | 17 (68%) | 25 | |
| Price 1 + 2 | 21 (14%) | 21 (14%) | 7 (5%) | 49 (33%) | 50 (34%) | 99 | p < 0.01 |
| Price 3 + 4 + 5 | 13 (9%) | 6 (4%) | 14 (9%) | 33 (22%) | 84 (56%) | 117 | |
| Significance soy vs. no legumes | |||||||
| Number by category | 34 | 27 | 21 | 82 | 134 | 216 | p < 0.01 |
| Price 1 + 2 | 21 (62%) | 21 (78%) | 7 (33%) | 49 (60%) | 50 (37%) | 99 | |
| Price 3 + 4 + 5 | 13 (38%) | 6 (22%) | 14 (67%) | 33 (40%) | 84 (63%) | 117 |
| Classes of Isoflavone Exposure | Number of Subjects | Confidence Intervals |
|---|---|---|
| < 0.5 mg /day | 84 | 25.6% < 31.1 < 36.6% |
| (0.5–1) mg/day | 48 | 13.2% < 17.8 < 22.3% |
| (1–2) mg/day | 37 | 9.6% < 13.7 < 17.8% |
| (2–3) mg/day | 31 | 7.7% < 11.5 < 15.3% |
| (3–5) mg/day | 42 | 11.2% < 15.6 < 19.9% |
| (5–8) mg/day | 21 | 4.6% < 7.8 < 11.0% |
| ≥ 8 mg/day | 7 | 0.7% < 2.6 < 4.5% |
| TOTAL | 270 |
| Isoflavone Consumption | <45 mg/day | ≥45 mg/day | TOTAL |
|---|---|---|---|
| Apart from you who consume soy at yours? | |||
| Nobody | 43 (43%) | 18 (47%) | 61 |
| My husband | 25 (25%) | 9 (24%) | 34 |
| My child below 3 years-old | 1 (1%) | 1 (2.6%) | 2 |
| My child over 3 years-old | 4 (4%) | 2 (5.3%) | 6 |
| My adolescent | 9 (9%) | 4 (10.5%) | 13 |
| Others | 18 (18%) | 4 (10.5%) | 22 |
| Total | 100 (100%) | 38 (100%) | 138 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, A.; Beaubernard, L.; Lamothe, V.; Bennetau-Pelissero, C. New Evaluation of Isoflavone Exposure in the French Population. Nutrients 2019, 11, 2308. https://doi.org/10.3390/nu11102308
Lee A, Beaubernard L, Lamothe V, Bennetau-Pelissero C. New Evaluation of Isoflavone Exposure in the French Population. Nutrients. 2019; 11(10):2308. https://doi.org/10.3390/nu11102308
Chicago/Turabian StyleLee, Alexandre, Laetitia Beaubernard, Valérie Lamothe, and Catherine Bennetau-Pelissero. 2019. "New Evaluation of Isoflavone Exposure in the French Population" Nutrients 11, no. 10: 2308. https://doi.org/10.3390/nu11102308
APA StyleLee, A., Beaubernard, L., Lamothe, V., & Bennetau-Pelissero, C. (2019). New Evaluation of Isoflavone Exposure in the French Population. Nutrients, 11(10), 2308. https://doi.org/10.3390/nu11102308






