The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
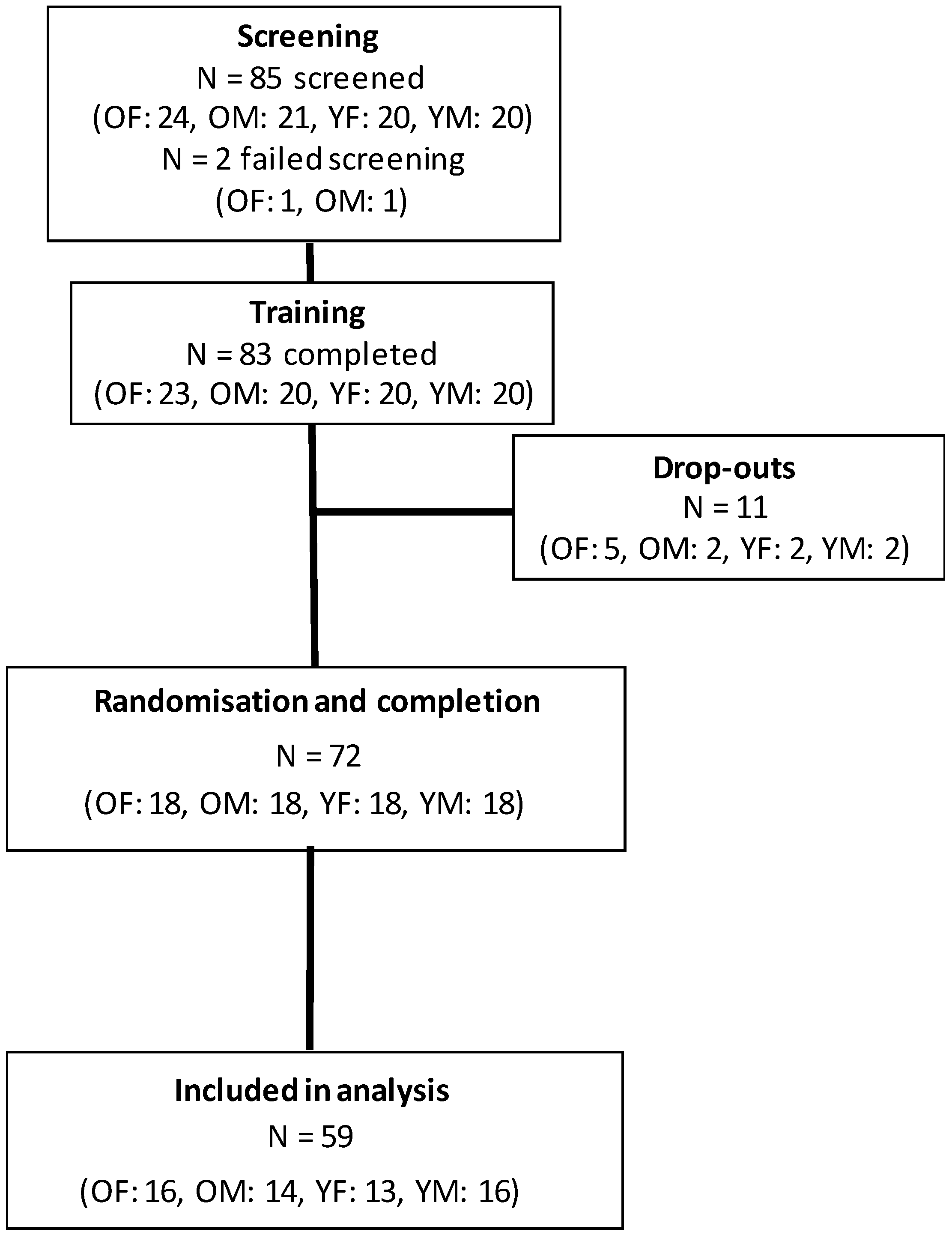
2.2. Participants
2.3. Treatment
- 220 mL water mixed with 2.5 g coffee flavouring (placebo)
- 220 mL regular coffee (without milk and sugar) containing 100 mg caffeine
- 220 mL decaffeinated coffee (without milk and sugar) containing ~5 mg caffeine
2.4. Salivary Caffeine Levels
2.5. Cognitive and Mood Measures
2.6. Caffeine Research Visual Analogue Scales
2.7. Computerised Location Learning—Learning Phase
2.8. Computerised Location Learning—Delayed Trial
2.9. Driving Ability
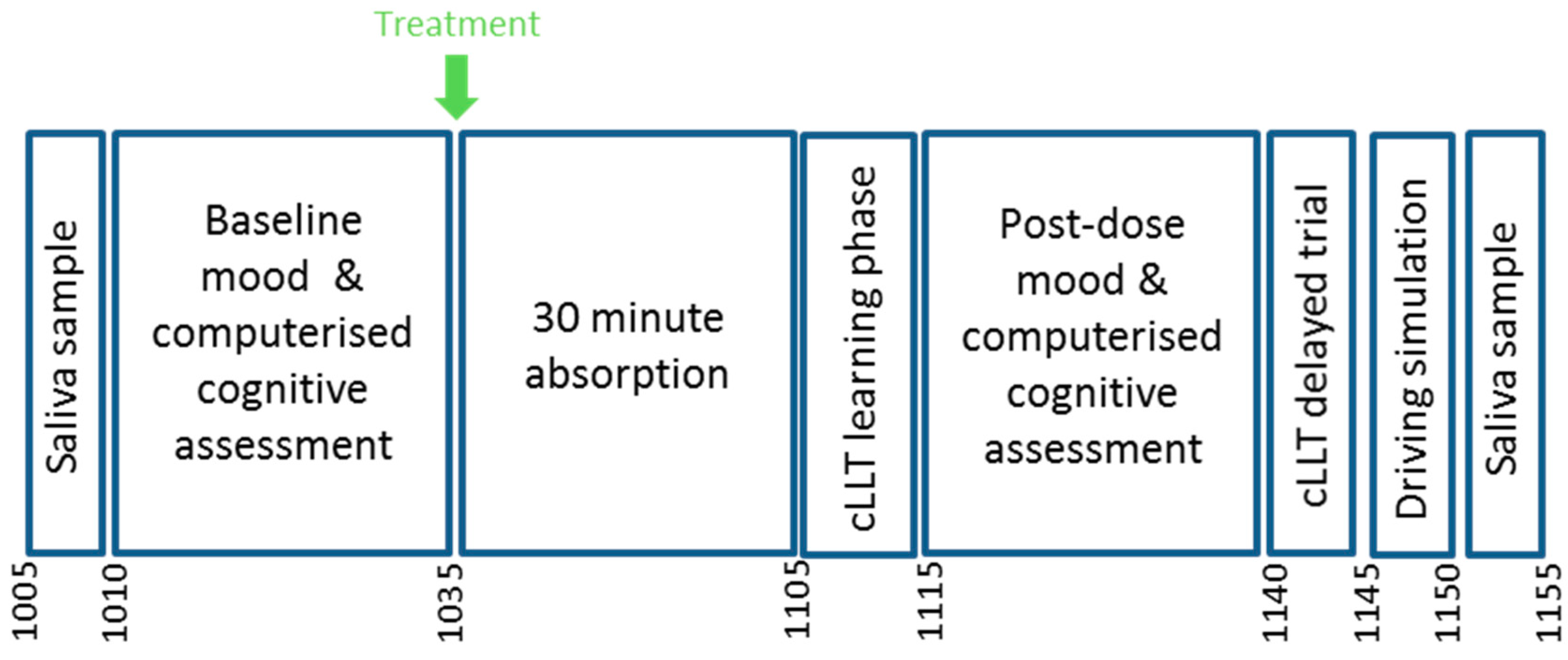
2.10. Procedure
2.11. Statistics
3. Results
3.1. Treatment-Related Effects
3.1.1. Salivary Caffeine
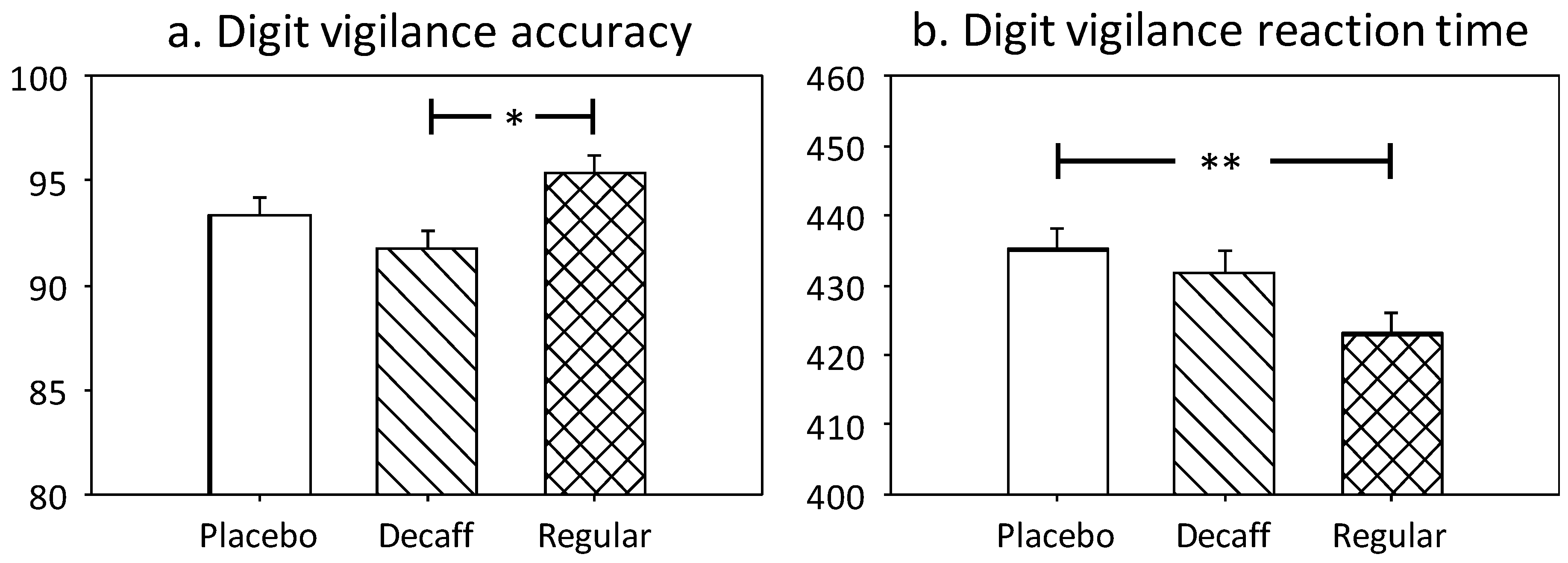
3.1.2. Digit Vigilance
3.1.3. Rapid Visual Information Processing
3.1.4. Computerised Location Learning Delayed Trial
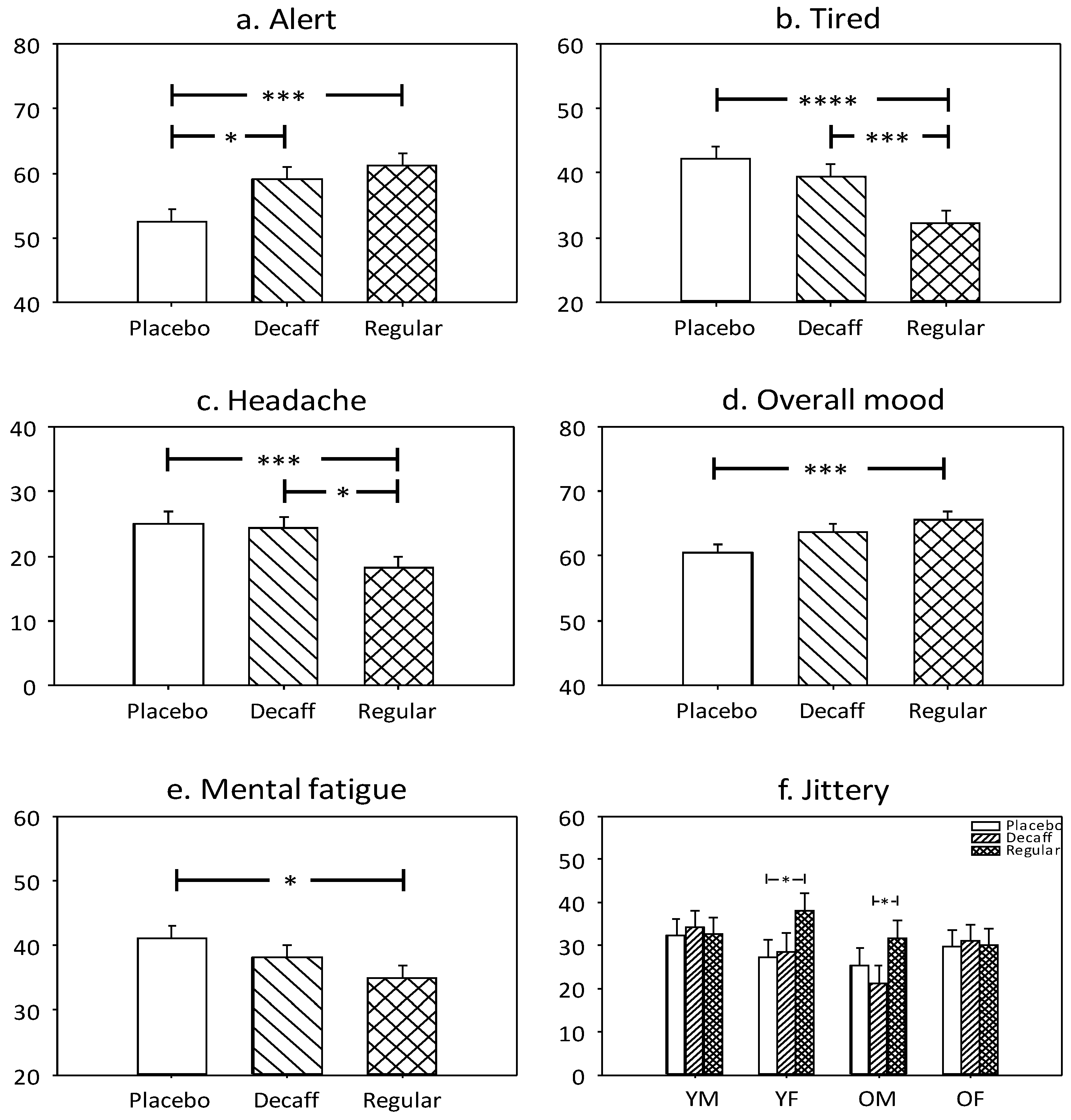
3.1.5. Alert
3.1.6. Tired
3.1.7. Headache
3.1.8. Overall Mood
3.1.9. Mental Fatigue
3.1.10. Jittery
3.2. Treatment Guess
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rodriguez-Artalejo, F.; Lopez-Garcia, E. Coffee consumption and cardiovascular disease: A condensed review of epidemiological evidence and mechanisms. J. Agric. Food Chem. 2018, 66, 5257–5263. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.B.; Zhang, D.F.; Jiang, W.J. Coffee and caffeine intake and incidence of type 2 diabetes mellitus: A meta-analysis of prospective studies. Eur. J. Nutr. 2014, 53, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.F.; Jacobs, D.R., Jr.; Carlsen, M.H.; Blomhoff, R. Consumption of coffee is associated with reduced risk of death attributed to inflammatory and cardiovascular diseases in the Iowa women’s health study. Am. J. Clin. Nutr. 2006, 83, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.A.; Dunbar, C.C.; Schnoll, R.; Kokolis, R.; Kokolis, S.; Kassotis, J. Caffeinated beverage intake and the risk of heart disease mortality in the elderly: A prospective analysis. Am. J. Clin. Nutr. 2007, 85, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Freedman, N.D.; Park, Y.; Abnet, C.C.; Hollenbeck, A.R.; Sinha, R. Association of coffee drinking with total and cause-specific mortality. N. Engl. J. Med. 2012, 366, 1891–1904. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.J.; Murphy, N.; Cross, A.J.; Dossus, L.; Dartois, L.; Fagherazzi, G.; Kaaks, R.; Kuhn, T.; Boeing, H.; Aleksandrova, K.; et al. Coffee drinking and mortality in 10 european countries: A multinational cohort study. Ann. Intern. Med. 2017, 167, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Freedman, N.D.; Haiman, C.A.; Le Marchand, L.; Wilkens, L.R.; Setiawan, V.W. Association of coffee consumption with total and cause-specific mortality among nonwhite populations. Ann. Intern. Med. 2017, 167, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, M.J. Does caffeine intake enhance absolute levels of cognitive performance? Psychopharmacology 1993, 110, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Araujo, L.F.; Giatti, L.; Reis, R.C.; Goulart, A.C.; Schmidt, M.I.; Duncan, B.B.; Ikram, M.A.; Barreto, S.M. Inconsistency of association between coffee consumption and cognitive function in adults and elderly in a cross-sectional study (elsa-brasil). Nutrients 2015, 7, 9590–9601. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Kivipelto, M. Caffeine as a protective factor in dementia and Alzheimer’s disease. J. Alzheimers Dis. 2010, 20, S167–S174. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Ngandu, T.; Tuomilehto, J.; Soininen, H.; Kivipelto, M. Midlife coffee and tea drinking and the risk of late-life dementia: A population-based caide study. J. Alzheimers Dis. 2009, 16, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Maia, L.; de Mendonca, A. Does caffeine intake protect from alzheimer’s disease? Eur. J. Neurol. 2002, 9, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.; Laurin, D.; Verreault, R.; Hebert, R.; Helliwell, B.; Hill, G.B.; McDowell, I. Risk factors for alzheimer’s disease: A prospective analysis from the canadian study of health and aging. Am. J. Epidemiol. 2002, 156, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.P.; Wu, Y.F.; Cheng, H.Y.; Xia, T.; Ding, H.; Wang, H.; Wang, Z.M.; Xu, Y. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutrition 2016, 32, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; Battig, K.; Holmen, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar] [PubMed]
- Haskell, C.F.; Kennedy, D.O.; Wesnes, K.A.; Scholey, A.B. Cognitive and mood improvements of caffeine in habitual consumers and habitual non-consumers of caffeine. Psychopharmacology 2005, 179, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Smit, H.J.; Rogers, P.J. Effects of low doses of caffeine on cognitive performance, mood and thirst in low and higher caffeine consumers. Psychopharmacology 2000, 152, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Childs, E.; de Wit, H. Subjective, behavioral, and physiological effects of acute caffeine in light, nondependent caffeine users. Psychopharmacology 2006, 185, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Renouf, M.; Marmet, C.; Giuffrida, F.; Lepage, M.; Barron, D.; Beaumont, M.; Williamson, G.; Dionisi, F. Dose-response plasma appearance of coffee chlorogenic and phenolic acids in adults. Mol. Nutr. Food Res. 2014, 58, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Dodd, F.L.; Kennedy, D.O.; Riby, L.M.; Haskell-Ramsay, C.F. A double-blind, placebo-controlled study evaluating the effects of caffeine and l-theanine both alone and in combination on cerebral blood flow, cognition and mood. Psychopharmacology 2015, 232, 2563–2576. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Kennedy, D.O.; Milne, A.L.; Wesnes, K.A.; Scholey, A.B. The effects of l-theanine, caffeine and their combination on cognition and mood. Biol. Psychol. 2008, 77, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Giles, G.E.; Mahoney, C.R.; Brunye, T.T.; Taylor, H.A.; Kanarek, R.B. Caffeine and theanine exert opposite effects on attention under emotional arousal. Can. J. Physiol. Pharmacol. 2017, 95, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.E.; Blumenthal, T.D.; Flaten, M.A. Effects of caffeine and caffeine-associated stimuli on the human startle eyeblink reflex. Pharmacol. Biochem. Behav. 1998, 59, 39–44. [Google Scholar] [CrossRef]
- Ferruzzi, M.G. The influence of beverage composition on delivery of phenolic compounds from coffee and tea. Physiol. Behav. 2010, 100, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Cropley, V.; Croft, R.; Silber, B.; Neale, C.; Scholey, A.; Stough, C.; Schmitt, J. Does coffee enriched with chlorogenic acids improve mood and cognition after acute administration in healthy elderly? A pilot study. Psychopharmacology 2012, 219, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Camfield, D.A.; Silber, B.Y.; Scholey, A.B.; Nolidin, K.; Goh, A.; Stough, C. A randomised placebo-controlled trial to differentiate the acute cognitive and mood effects of chlorogenic acid from decaffeinated coffee. PLoS ONE 2013, 8, e82897. [Google Scholar] [CrossRef] [PubMed]
- Duff, S.J.; Hampson, E. A sex difference on a novel spatial working memory task in humans. Brain Cognit. 2001, 47, 470–493. [Google Scholar] [CrossRef] [PubMed]
- Herlitz, A.; Rehnman, J. Sex differences in episodic memory. Curr. Dir. Psychol. Sci. 2008, 17, 52–56. [Google Scholar] [CrossRef]
- van Gelder, B.M.; Buijsse, B.; Tijhuis, M.; Kalmijn, S.; Giampaoli, S.; Nissinen, A.; Kromhout, D. Coffee consumption is inversely associated with cognitive decline in elderly european men: The fine study. Eur. J. Clin. Nutr. 2007, 61, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Arab, L.; Biggs, M.L.; O’Meara, E.S.; Longstreth, W.T.; Crane, P.K.; Fitzpatrick, A.L. Gender differences in tea, coffee, and cognitive decline in the elderly: The cardiovascular health study. J. Alzheimers Dis. 2011, 27, 553–566. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, K.; Carriere, I.; de Mendonca, A.; Portet, F.; Dartigues, J.F.; Rouaud, O.; Barberger-Gateau, P.; Ancelin, M.L. The neuroprotective effects of caffeine: A prospective population study (the three city study). Neurology 2007, 69, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Henry, C.J.; Lightowler, H.J.; Marchini, J. Intra-individual variation in resting metabolic rate during the menstrual cycle. Br. J. Nutr. 2003, 89, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.D.; Steege, J.F.; Rupp, S.L.; Kuhn, C.M. Menstrual cycle effects on caffeine elimination in the human female. Eur. J. Clin. Pharmacol. 1992, 43, 543–546. [Google Scholar] [CrossRef] [PubMed]
- James, J.E.; Rogers, P.J. Effects of caffeine on performance and mood: Withdrawal reversal is the most plausible explanation. Psychopharmacology 2005, 182, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.P.; Christopher, G.; Sutherland, D. Acute effects of caffeine on attention: A comparison of non-consumers and withdrawn consumers. J. Psychopharmacol. 2013, 27, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Wightman, E.L.; Reay, J.L.; Lietz, G.; Okello, E.J.; Wilde, A.; Haskell, C.F. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: A double-blind, placebo-controlled, crossover investigation. Am. J. Clin. Nutr. 2010, 91, 1590–1597. [Google Scholar] [CrossRef] [PubMed]
- Stonehouse, W.; Conlon, C.A.; Podd, J.; Hill, S.R.; Minihane, A.M.; Haskell, C.; Kennedy, D. Dha supplementation improved both memory and reaction time in healthy young adults: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 1134–1143. [Google Scholar] [CrossRef] [PubMed]
- Rogers, P.J.; Martin, J.; Smith, C.; Heatherley, S.V.; Smit, H.J. Absence of reinforcing, mood and psychomotor performance effects of caffeine in habitual non-consumers of caffeine. Psychopharmacology 2003, 167, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Haskell, C.F. Cerebral blood flow and behavioural effects of caffeine in habitual and non-habitual consumers of caffeine: A near infrared spectroscopy study. Biol. Psychol. 2011, 86, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kessels, R.P.; Nys, G.M.; Brands, A.M.; van den Berg, E.; Van Zandvoort, M.J. The modified location learning test: Norms for the assessment of spatial memory function in neuropsychological patients. Arch. Clin. Neuropsychol. 2006, 21, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, K.; Shibamoto, T. Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem. 2008, 106, 217–221. [Google Scholar] [CrossRef]
- Griffeth, V.E.; Perthen, J.E.; Buxton, R.B. Prospects for quantitative fmri: Investigating the effects of caffeine on baseline oxygen metabolism and the response to a visual stimulus in humans. Neuroimage 2011, 57, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Geraets, L.; Moonen, H.J.; Wouters, E.F.; Bast, A.; Hageman, G.J. Caffeine metabolites are inhibitors of the nuclear enzyme poly(adp-ribose)polymerase-1 at physiological concentrations. Biochem. Pharmacol. 2006, 72, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Zulli, A.; Smith, R.M.; Kubatka, P.; Novak, J.; Uehara, Y.; Loftus, H.; Qaradakhi, T.; Pohanka, M.; Kobyliak, N.; Zagatina, A.; et al. Caffeine and cardiovascular diseases: Critical review of current research. Eur. J. Nutr. 2016, 55, 1331–1343. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, J.A.; Benitez, J. Clinically significant pharmacokinetic interactions between dietary caffeine and medications. Clin. Pharmacokinet. 2000, 39, 127–153. [Google Scholar] [CrossRef] [PubMed]
- Gunes, A.; Dahl, M.L. Variation in cyp1a2 activity and its clinical implications: Influence of environmental factors and genetic polymorphisms. Pharmacogenomics 2008, 9, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Gambaro, S.E.; Moretti, R.; Tiribelli, C.; Gazzin, S. Brain cytochrome p450 enzymes: A possible therapeutic targets for neurological diseases. Ther. Targets Neurol. Dis. 2015, 2, e598. [Google Scholar]
- Sansone, R.; Ottaviani, J.I.; Rodriguez-Mateos, A.; Heinen, Y.; Noske, D.; Spencer, J.P.; Crozier, A.; Merx, M.W.; Kelm, M.; Schroeter, H.; et al. Methylxanthines enhance the effects of cocoa flavanols on cardiovascular function: Randomized, double-masked controlled studies. Am. J. Clin. Nutr. 2017, 105, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Dulloo, A.G.; Duret, C.; Rohrer, D.; Girardier, L.; Mensi, N.; Fathi, M.; Chantre, P.; Vandermander, J. Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans. Am. J. Clin. Nutr. 1999, 70, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, K.; Nakayama, K.; Nakamura, M.; Sookwong, P.; Tsuduki, T.; Niino, H.; Kimura, F.; Miyazawa, T. Effects of co-administration of tea epigallocatechin-3-gallate (EGCG) and caffeine on absorption and metabolism of EGCG in humans. Biosci. Biotechnol. Biochem. 2009, 73, 2014–2017. [Google Scholar] [CrossRef] [PubMed]
- Reis, C.E.G.; Paiva, C.; Amato, A.A.; Lofrano-Porto, A.; Wassell, S.; Bluck, L.J.C.; Dorea, J.G.; da Costa, T.H.M. Decaffeinated coffee improves insulin sensitivity in healthy men. Br. J. Nutr. 2018, 119, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, T.; Comi, R.; Sluss, P.; Keisari, R.; Manwar, S.; Kim, J.; Larson, R.; Baron, J.A. Metabolic and hormonal effects of caffeine: Randomized, double-blind, placebo-controlled crossover trial. Metabolism 2007, 56, 1694–1698. [Google Scholar] [CrossRef] [PubMed]
- Battram, D.S.; Arthur, R.; Weekes, A.; Graham, T.E. The glucose intolerance induced by caffeinated coffee ingestion is less pronounced than that due to alkaloid caffeine in men. J. Nutr. 2006, 136, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Adisakwattana, S.; Moonsan, P.; Yibchok-Anun, S. Insulin-releasing properties of a series of cinnamic acid derivatives in vitro and in vivo. J. Agric. Food Chem. 2008, 56, 7838–7844. [Google Scholar] [CrossRef] [PubMed]
- Torronen, R.; Kolehmainen, M.; Sarkkinen, E.; Mykkanen, H.; Niskanen, L. Postprandial glucose, insulin, and free fatty acid responses to sucrose consumed with blackcurrants and lingonberries in healthy women. Am. J. Clin. Nutr. 2012, 96, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Torronen, R.; Kolehmainen, M.; Sarkkinen, E.; Poutanen, K.; Mykkanen, H.; Niskanen, L. Berries reduce postprandial insulin responses to wheat and rye breads in healthy women. J. Nutr. 2013, 143, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Torronen, R.; Sarkkinen, E.; Niskanen, T.; Tapola, N.; Kilpi, K.; Niskanen, L. Postprandial glucose, insulin and glucagon-like peptide 1 responses to sucrose ingested with berries in healthy subjects. Br. J. Nutr. 2012, 107, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Sapwarobol, S.; Adisakwattana, S.; Changpeng, S.; Ratanawachirin, W.; Tanruttanawong, K.; Boonyarit, W. Postprandial blood glucose response to grape seed extract in healthy participants: A pilot study. Pharmacogn. Mag. 2012, 8, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.T.; Head, K.; Morris, P.G.; Macdonald, I.A. The effect of flavanol-rich cocoa on the fMRI response to a cognitive task in healthy young people. J. Cardiovasc. Pharmacol. 2006, 47, S215–S220. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Pal, D.; Moutsiana, C.; Field, D.T.; Williams, C.M.; Spencer, J.P.; Butler, L.T. The effect of flavanol-rich cocoa on cerebral perfusion in healthy older adults during conscious resting state: A placebo controlled, crossover, acute trial. Psychopharmacology 2015, 232, 3227–3234. [Google Scholar] [CrossRef] [PubMed]
- Liguori, A.; Hughes, J.R.; Grass, J.A. Absorption and subjective effects of caffeine from coffee, cola and capsules. Pharmacol. Biochem. Behav. 1997, 58, 721–726. [Google Scholar] [CrossRef]
- Stalmach, A.; Williamson, G.; Crozier, A. Impact of dose on the bioavailability of coffee chlorogenic acids in humans. Food Funct. 2014, 5, 1727–1737. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, J.; Sawers, S.J. Comparative pharmacokinetics of caffeine in young and elderly men. J. Pharmacokinet. Biopharm. 1983, 11, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Haskell-Ramsay, C.F.; Jackson, P.A.; Forster, J.S.; Robertson, B.C.; Kennedy, D.O. Acute effects of three doses of caffeine on attention, motor speed and mood over an 8-hour period. Appetite 2018, in press. [Google Scholar] [CrossRef]
- van Boxtel, M.; Schmitt, J. Coffee, Tea, Chocolate, and the Brain, 1st ed.; Nehlig, A., Ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Office for National Statistics (ONS). 2011 Census: Key Statistics for England and Wales. March 2011. Available online: http://www.ons.gov.uk/ons/dcp171778_290685.pdf (accessed on 28 September 2018).
- Johnson-Kozlow, M.; Kritz-Silverstein, D.; Barrett-Connor, E.; Morton, D. Coffee consumption and cognitive function among older adults. Am. J. Epidemiol. 2002, 156, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Weisskopf, M.G.; O’Reilly, E.J.; McCullough, M.L.; Calle, E.E.; Rodriguez, C.; Thun, M.J. Coffee consumption, gender, and parkinson’s disease mortality in the cancer prevention study II cohort: The modifying effects of estrogen. Am. J. Epidemiol. 2004, 160, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, R.L.; Barrett-Connor, E. Caffeine intake and endogenous sex steroid levels in postmenopausal women. The rancho bernardo study. Am. J. Epidemiol. 1996, 144, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Hartley, T.R.; Lovallo, W.R.; Whitsett, T.L. Cardiovascular effects of caffeine in men and women. Am. J. Cardiol. 2004, 93, 1022–1026. [Google Scholar] [CrossRef] [PubMed]
- Kiyohara, C.; Kono, S.; Honjo, S.; Todoroki, I.; Sakurai, Y.; Nishiwaki, M.; Hamada, H.; Nishikawa, H.; Koga, H.; Ogawa, S.; et al. Inverse association between coffee drinking and serum uric acid concentrations in middle-aged japanese males. Br. J. Nutr. 1999, 82, 125–130. [Google Scholar] [PubMed]
- Perna, L.; Mons, U.; Schottker, B.; Brenner, H. Association of cognitive function and serum uric acid: Are cardiovascular diseases a mediator among women? Exp. Gerontol. 2016, 81, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, B.B.; Brix, T.H.; Kyvik, K.O.; Brosen, K. The interindividual differences in the 3-demthylation of caffeine alias cyp1a2 is determined by both genetic and environmental factors. Pharmacogenetics 2002, 12, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Alsene, K.; Deckert, J.; Sand, P.; de Wit, H. Association between A2a receptor gene polymorphisms and caffeine-induced anxiety. Neuropsychopharmacology 2003, 28, 1694–1702. [Google Scholar] [CrossRef] [PubMed]
- Childs, E.; Hohoff, C.; Deckert, J.; Xu, K.; Badner, J.; de Wit, H. Association between adora2a and drd2 polymorphisms and caffeine-induced anxiety. Neuropsychopharmacology 2008, 33, 2791–2800. [Google Scholar] [CrossRef] [PubMed]
- Gajewska, A.; Blumenthal, T.D.; Winter, B.; Herrmann, M.J.; Conzelmann, A.; Muhlberger, A.; Warrings, B.; Jacob, C.; Arolt, V.; Reif, A.; et al. Effects of ADORA2A gene variation and caffeine on prepulse inhibition: A multi-level risk model of anxiety. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 40, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Domschke, K.; Gajewska, A.; Winter, B.; Herrmann, M.J.; Warrings, B.; Muhlberger, A.; Wosnitza, K.; Glotzbach, E.; Conzelmann, A.; Dlugos, A.; et al. ADORA2A gene variation, caffeine, and emotional processing: A multi-level interaction on startle reflex. Neuropsychopharmacology 2012, 37, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C.; El-Sohemy, A.; Campos, H. Genetic polymorphism of the adenosine A2A receptor is associated with habitual caffeine consumption. Am. J. Clin. Nutr. 2007, 86, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Rogers, P.J.; Hohoff, C.; Heatherley, S.V.; Mullings, E.L.; Maxfield, P.J.; Evershed, R.P.; Deckert, J.; Nutt, D.J. Association of the anxiogenic and alerting effects of caffeine with ADORA2A and adora1 polymorphisms and habitual level of caffeine consumption. Neuropsychopharmacology 2010, 35, 1973–1983. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. Interindividual differences in caffeine metabolism and factors driving caffeine consumption. Pharmacol. Rev. 2018, 70, 384–411. [Google Scholar] [CrossRef] [PubMed]


| Young | Older | |||||||
|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | |||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Age | 26.3 | 4.4 | 26.2 | 3.6 | 67.7 | 6.3 | 67.1 | 3.4 |
| Years in education | 18 | 3 | 17 | 3 | 16 | 5 | 14 | 4 |
| Body Mass Index (BMI) | 25.7 | 3.8 | 23.8 | 3.6 | 25.9 | 3.4 | 26.1 | 3.9 |
| Caffeine consumption (mg/day) | 327 | 88.2 | 351 | 110.4 | 426 | 74.4 | 394 | 87.8 |
| Coffee consumption (cups/day) | 2.88 | 1.54 | 2.54 | 1.45 | 2.64 | 1.13 | 3.59 | 0.93 |
| Fruit and vegetables (portions/day) | 4.3 | 1.5 | 4.1 | 1.4 | 4.4 | 1.9 | 5.4 | 1.8 |
| Task | Descriptor | Scoring | Domain |
|---|---|---|---|
| Word presentation | A series of words is displayed on the screen, one word at a time. In this case, 15 words were presented with a display time of 1 s and interstimulus interval of 1 s | - | |
| Immediate word recall | Participants are instructed to write down the words that were presented. In this case, 60 s were given to complete the task | Number correct and number of errors | Episodic memory |
| Picture presentation | A series of photographic images are displayed on the screen, one at a time. In this case, 15 images were presented with a display time of 2 s and an interstimulus interval of 1 s | - | |
| Simple reaction time | An upwards pointing arrow is displayed on the screen at irregular intervals. Participants must respond as quickly as they can as soon as they see the arrow appear. In this case, 50 stimuli were presented | Reaction time (ms) | Attention |
| Digit vigilance | A fixed number appears on the right of the screen and a series of changing numbers appear on the left of the screen at the rate of 150 per minute. Participants are required to make a response when the number on the left matches the number on the right. In this case the task lasted for 3 min | Accuracy (%), reaction time for the correct responses (ms) and false alarms (number) | Attention |
| Numeric working memory | Five single target numbers are displayed on the screen, one at a time. Participants are required to memorise these numbers as they appear. Once the target series has been presented, numbers are displayed one at a time and participants are required to indicate if each number was presented in the previous list or not. In this case, three trials were completed | Accuracy (%) and reaction time for the correct responses (ms) | Working memory |
| Verbal fluency | Participants are presented with a letter on screen and asked to write down as many words as they can, beginning with that letter. In this case, the letters presented were A, T, C, F, M, and S and 60 s were given to complete the task | Number correct permitted words, with names and perseverations discounted from the total score | Language |
| Delayed word recall | Participants are instructed to write down the words that were presented to them at the beginning of the assessment. In this case, 60 s were given to complete the task | Number correct and number of errors | Episodic memory |
| Rapid visual information processing | A continuous series of single digits are presented in the centre of the screen at the rate of 100 per minute. Participants are required to make a response when three consecutive odd or three consecutive even digits are displayed. In this case, the task lasted for 5 min, with eight correct target strings presented in each minute. | Accuracy (%), reaction time for the correct responses (ms) and false alarms (number) | Attention |
| Delayed word recognition | All target words that were shown during Word presentation plus an equal number of decoys are displayed on the screen one at a time. Participants indicate if they remember seeing the word earlier or not. | Accuracy (%) and reaction time for the correct responses (ms) | Episodic memory |
| Delayed picture recognition | All target pictures shown during Picture presentation plus an equal number of decoys are displayed on the screen one at a time. Participants indicate if they remember seeing the picture earlier or not. | Accuracy (%) and reaction time for the correct responses (ms) | Episodic memory |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haskell-Ramsay, C.F.; Jackson, P.A.; Forster, J.S.; Dodd, F.L.; Bowerbank, S.L.; Kennedy, D.O. The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults. Nutrients 2018, 10, 1386. https://doi.org/10.3390/nu10101386
Haskell-Ramsay CF, Jackson PA, Forster JS, Dodd FL, Bowerbank SL, Kennedy DO. The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults. Nutrients. 2018; 10(10):1386. https://doi.org/10.3390/nu10101386
Chicago/Turabian StyleHaskell-Ramsay, Crystal F., Philippa A. Jackson, Joanne S. Forster, Fiona L. Dodd, Samantha L. Bowerbank, and David O. Kennedy. 2018. "The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults" Nutrients 10, no. 10: 1386. https://doi.org/10.3390/nu10101386
APA StyleHaskell-Ramsay, C. F., Jackson, P. A., Forster, J. S., Dodd, F. L., Bowerbank, S. L., & Kennedy, D. O. (2018). The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults. Nutrients, 10(10), 1386. https://doi.org/10.3390/nu10101386





