Abstract
Biodiversity in ecosystems is threatened by plastic waste, a significant pollution problem. The research aims to use plastic, Pleorotus ostreatus mycelium, and sugarcane bagasse to develop an eco-friendly biomaterial. The preparation consisted of three phases: (1) Treatment of the underlying layer, (2) inoculation of the fungus (three different concentrations: 15%, M1; 20%, M2; and 25%, M3), and (3) dehydration of the biomaterial. The physical characterization results (density, water absorption, and texture) showed that the average density varies between 127.86 and 131.19 kg/m3; also, the M1 sample had a high percentage of water absorption (23.55%), while M2 obtained lower percentages (11.79%). In the bending test, samples M2 and M3 showed higher structural resistance with stress values of 0.682 and 0.68 MPa, respectively. Based on the results, the produced biomaterials may serve as an alternative to expanded polystyrene (EPS). Due to their moldable characteristics, they can be used in non-structural construction, packaging, and various architectural applications. The research provided promising results; however, additional parameters must be evaluated to be able to commercialize them.
1. Introduction
In recent years, the world population has exceeded 7.7 billion inhabitants, and it will probably rise to 8.6 billion by 2030 and 9.8 billion by 2050 [1]. This exponential world demographic growth, accompanied by excessive industrialization, low diffusion, and the application of alternative technologies, causes an environmental crisis. Likewise, the consumption of polymers or plastics has increased rapidly in the last five decades; these petrochemicals have been partially replaced in specific circumstances and many natural materials are used such as wood, cotton, paper, wool, and leather, among others. The United Nations Environment Program (UNEP) tells us in its research that the magnitude of the generation of greenhouse gases (GHGs) linked to the production, use, and elimination of plastics from fossil fuels will increase by around 2.1 gigatons of carbon dioxide (Gt CO2) by the year 2040 [2]. Similarly, other sources calculate that these emissions from plastics were 1.7 Gt CO2 in 2015 and will increase to about 6.5 Gt CO2 by the year 2050, equivalent to 15% of the global carbon budget.
At the same time, Lozano et al. (2020) indicated that worldwide 33% of the total organic waste obtained is not managed correctly; for example, in Peru, only 1% of this waste is used, causing a disproportionate emission of GHG, highlighting methane and CO2, contributing significantly to climate change [3]. On the other hand, regarding industrialization in Peru, Muñoz et al. (2019) indicates the outstanding sector is the agro-industrial sector, as the sugar industry originates several organic wastes, which present a high content of lignin and cellulose, the latter being one of the compounds found in large quantities throughout the world. However, they are not used or valued [4,5,6,7]. Mendez-Matias et al. (2018) stressed that approximately 75% of each ton of sugarcane is organic solid waste, having a greater volume of bagasse generated in industrial and artisanal processes [8]. The extensive use of expanded polystyrene (EPS) leads to the accumulation of plastic waste due to its non-degradability property in its natural state, which has generated severe environmental impacts (white pollution). One of the challenges in our society is the correct disposal of the waste produced by anthropogenic activities; as the production of synthetic polymers grows, the amount of plastic waste will also increase [9,10,11]. Unfortunately, only a tiny fraction is recycled. It is an unavoidable dilemma due to the collection, cleaning, and separation processes; the materials obtained are not competitive with virgin materials. That is why there is an urgent need for more significant efforts to carry out monitoring in all periods, since these data on plastic waste, especially EPS, are of great importance to restore and improve the perspective of the global, regional, and local model, thereby optimizing prevention and collection strategies [12,13].
Given this increase in consumption and considering that toxic resources have been used in the manufacturing, use, and disposal process of EPS, some countries have regulated the manufacturing process. In contrast, others, such as the United States, have prohibited or restricted its use as food packaging [14,15,16]. These measures have undoubtedly made it easier to reduce the amount of production; however, they do not solve the problem since, according to the data previously presented, except in Asia, the consumption of products destined for food packaging represents little more than 30% of total consumption, leaving pending the integral management of the remaining 70% [17,18]. In 1986, the United States Environmental Protection Agency (EPA) reported that the polystyrene manufacturing process was the fifth most significant source of hazardous waste, linking it to adverse health effects in humans, and in 2014 it was classified as a potential carcinogen [19,20]. Faced with this problem, the present research project was justified by proposing a new alternative in green chemistry to reduce the use of additives and thus generate a sustainable biomaterial [21,22,23].
In this research, the use of mycelium was analyzed, referring to the associations that fungi generate in the roots for better absorption of water and nutrients as a consequence of the growth of filaments (hyphae) [24,25,26]. Likewise, the use of waste as raw material or substrate, together with the biodegradability of the biomaterial at the end of its useful life and the extensive scale of materials that it could replace, guarantees better waste management in the future if it is applied efficiently [27,28]. The contribution of this research is made up of the physical and mechanical characterization with which a biomaterial composed of Pleurotus ostreatus mycelium and sugarcane bagasse was obtained as a substitute for polystyrene foams, presenting itself as an alternative for the use of lignocellulosic waste from the agro-industrial sector, in such a way that it can serve future researchers generating harmony between ecosystems and the fundamental needs of human beings, contributing to the circular economy [29].
Due to this, the objective of the research is to produce a composite biomaterial based on Pleurotus ostreatus mycelium and sugarcane bagasse as an alternative to polystyrene foams. For this purpose, the physical and mechanical parameters (density, water absorption, and texture) of the biomaterial were evaluated using physical–mechanical tests of the compound biomaterials of Pleurotus ostreatus mycelium and bagasse.
2. Materials and Methods
2.1. Culture of the Pleurotus ostreatus
The fungal Pleurotus ostreatus culture came from the Fungi Peru company (Cusco, Peru). This company is a pioneer in the cultivation and commercial business of gourmet and medicinal mushrooms in Cusco. Fungal culture was provided in the form of mycelium attached to wheat grains.
2.2. Preparation of Sugarcane Bagasse and Pleurotus Ostreatus Inoculum
The bagasse was sieved with N°10 mesh to achieve homogeneous 2.0 mm fibers. Afterward, it was hydrated with distilled water in a 1:9 ratio (50 g of substrate + 450 mL) and homogenized for 10 min. After 30 min of hydrating the substrate, excess water was removed, and relative humidity was measured [30,31]. Three amounts of bagasse (300, 320, and 340 g) were used, which were distributed in six bags for each concentration (18 bags in total). Then, they were sterilized at 121 °C for 15 min at 1 atm of pressure.
On the other hand, the mycelium of fungus attached to wheat grains (inoculum) was distributed in 18 first-use Ziploc bags. The quantity of mycelium employed was 60, 80, and 100 g [32].
2.3. Preparation of Biomaterial
The substrate was added to previously weighed mycelium and was manually shaken for 1 min for uniform growth. The mixed product was placed in sterile 1 L beakers and incubated at 26 °C for 10 days. During this period, the mixtures were hydrated with 5–10 mL of sterile distilled water and homogenized every 3 days, obtaining the biomaterial [30]. The biomaterial (M1, M2, and M3) was prepared with three inoculum concentrations, where blocks M1 consisted of 340 g of bagasse + 60 g of inoculum (15%), M2 had 80 g of bagasse + 320 g of inoculum (20%), and finally, M3 contained 300 g of bagasse + 100 g of inoculum (25%). To make the biomaterial-based blocks, 20 × 15 × 3 cm wooden molds containing a combination of bagasse and the Pleurotus ostreatus inoculum were employed. It was evenly distributed and then incubated at 26 °C for 14 days under aseptic conditions. The mixtures were hydrated with 3–5 mL of sterile distilled water. After 14 days, the wooden molds were removed, and the biomaterial-based blocks were obtained, which were left to incubate for 5 days at 26 °C. Subsequently, this biomaterial was placed in an oven at 60 °C for 24 h. Finally, the blocks were allowed to cool to store them in first-use bags for later analysis of density, texture, and moisture absorption.
2.4. Physical Parameters of Biomaterial
To calculate the density of the biomaterials, a Vernier scale (150 mm) was used and the measurements of length, width and thickness of the 6 replicas (blocks) were taken, as well as an electric balance for the measurement of the mass; the following formula was used:
where D is density of biomaterial, M is the mass, and V is the volume.
D (kg/m3) = M/V
For the measurement of texture, the TA.HD plus Texture Analyzer from Stable Micro Systems (UK) was used, where the 7 × 6 cm fragments of the biomaterial were placed on the test bed in the double-column frame. It exerted a force perpendicular to the blocks, using a P/75 at a speed of 1 mm/s. The data obtained were processed and graphically analyzed through its Exponent Connect software (www.stablemicrosystems.com). For the moisture determination, six replicates (blocks) for each inoculum concentration were cut into pieces of approximately 7 × 6 cm, and their initial mean weight (P1) was calculated. The samples were immersed in distilled water at room temperature for a period of 45 min. They were removed from the trays, excess water was removed with absorbent paper, and they were weighed again to obtain the final weight (P2). The following equation was used to calculate the weight percent:
∆M = (P2 − P1/P1) × 100
3. Results
The humidity tests (Table 1 and Table 2) carried out on the sugarcane bagasse found that it is within the optimal percentages for mycelial growth, having a range between 85 and 87%.

Table 1.
Polypropylene bag with 340 g of substrate.

Table 2.
Polypropylene bag with 310 g of substrate.
3.1. Physical and Chemical Parameters of the Biomaterial
3.2. Biomaterials Obtained
Figure 1, Figure 2 and Figure 3 show the biomaterials obtained from the different dosages and the necessary conditions for their development, such as darkness, temperature (26 °C), and substrate humidity (85–87%) during the 29 days of growth. The colonization of the mycelium gave a whitish appearance with some yellowish and brown tones. This is due to the increase in temperature in the final stage for the inhibition of the fungus. By standardizing the particle size of the substrate, its efficiency for the development and growth of the mycelium increased. This result was obtained as a consequence of increased oxygen exchange and increased moisture retention. In addition, it presented a soft and cottony texture typical of the mycelium formed on the surface, also called “fungal skin,” which developed irregularly; significant differences were observed since different treatments were elaborated, obtaining, as a result, a total of eighteen inoculated samples and only one presented contamination by mold on day 14 of sowing inside the substrate.

Figure 1.
Biomaterials obtained from sample “M1” with 340 g bagasse and 60 g of mycelium.

Figure 2.
Biomaterials obtained from the sample “M2” with 320 g bagasse and 80 g of mycelium.

Figure 3.
Biomaterials were obtained from the “M3” sample with 300 g bagasse and 100 g of mycelium.
3.3. Control Treatments
Figure 4 shows the control treatments of the substrates without inoculation; they were given the same controlled conditions: darkness, temperature (26 °C), and substrate humidity (85–87%) during the 29 days. However, it was observed that unlike samples M1, M2, and M3, they did not achieve consistency or obtain the shape of the mold to which they were transferred.

Figure 4.
Biomaterials obtained from control treatments.
To determine the physical and mechanical results of the biomaterial, destructive tests had to be carried out; these were carried out in test tubes or samples of the biomaterial because they were subjected to different analyses and forces until some change in their composition occurred, these being the properties that gave us a perspective on in which circumstances the biomaterial can be used and if these characteristics are similar to other biomaterials already produced [33,34]. The three samples (M1, M2, and M3) were able to pass the proposed physical tests; on the contrary, the controls, due to the low firmness and consistency of the bagasse, were not able to withstand the proposed tests.
Table 3 shows the average values obtained for this test, having as results for sample M1 an average density of 130.99 kg/m3, for M2 of 131.19 kg/m3, and for sample M3 of 127.86 kg/m3, the latter presenting a lower density of the samples analyzed.

Table 3.
Average values of the data recorded for the biomaterial density test.
Table 4 shows the average values obtained in this test, having as results for sample M1 an average water absorption percentage of 23.55%, for sample M2 of 11.79%, and sample M3 of 15.35%; in this test M2 was the one that obtained the lowest water absorption.

Table 4.
Average values of the data recorded for the biomaterial water absorption test.
Table 5 shows the average values obtained from the hardness, elasticity, and resilience tests of the three samples.

Table 5.
Average values of the data recorded for the hardness, elasticity, and resilience tests of the biomaterial.
In Figure 5, it can be seen that the average hardness of sample M2 is greater than that of samples M1 and M3; that is to say, it has greater strength and resistance to wear.
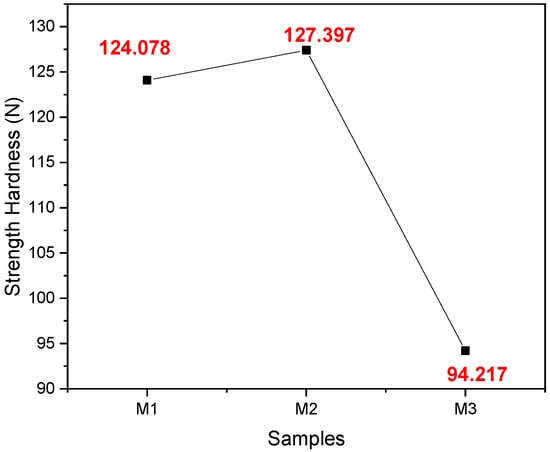
Figure 5.
Average values were obtained from the hardness test of samples M1, M2, and M3 with their respective replicates.
In Figure 6, it can be seen that the average elasticity of sample M2 is greater than sample M1, while M2 and M3 had equal values, that is, they had the same elasticity capacity.
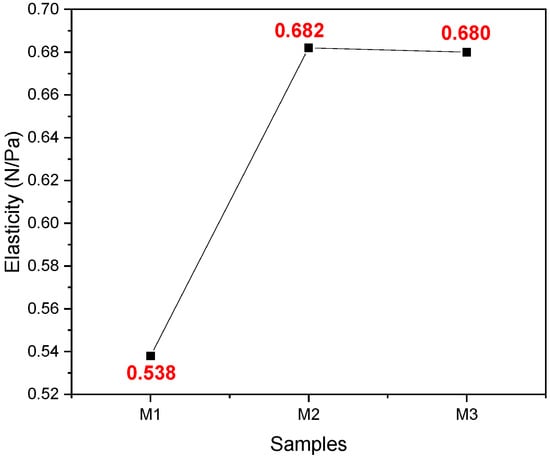
Figure 6.
Average values were obtained from the elasticity test of the samples M1, M2, and M3 with their respective replicates.
In Figure 7, it can be seen that the average resilience of sample M3 is higher than samples M1 and M2, that is, it had a greater ability to recover its shape when subjected to force.
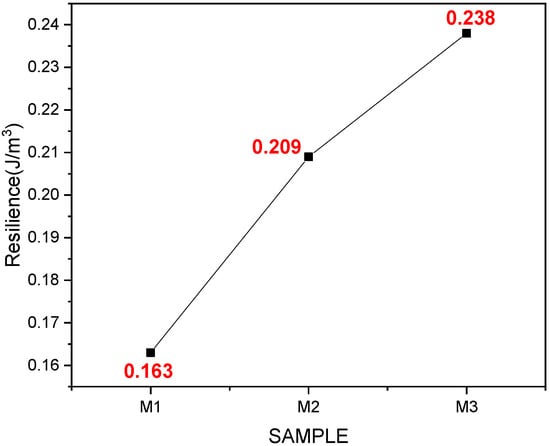
Figure 7.
Average values were obtained from the resilience test of samples M1, M2, and M3 with their respective replicates.
A significance test was performed on the data obtained for each type of sample. This analysis was applied to decide if the difference between the measured quantity and the known quantity is significant, attributing a random error; contrasts are widely applied in the field of experimental research [35,36]. Specifically, the Grubbs contrast was used for anomalous data; it is used as long as the sample size is between three and seven since, in the results, some replicates of the same sample showed values that differ from the rest. This test mentions that if the G value exceeds the critical value, that is, G > 1.887, the hypothesis that the value is not anomalous is rejected. However, after this exhaustive analysis, only two samples rejected the hypothesis because two values were anomalous, so they were withdrawn from our databases.
In Table 6, it is observed that the hardness results of the M1, M2, and M3 samples are lower than the critical value G > 1.887 according to the Grubbs test; the values are abnormal or atypical.

Table 6.
Values were obtained for the hardness test of samples M1, M2, and M3, with their 6 replicates.
In Table 7, it can be seen from the results obtained in the Grubbs contrast of the M1 resilience test, when compared with the critical value G > 1.887, that only one value exceeds it, that is, the hypothesis that the value is not anomalous is only rejected for the first value of M1. The results obtained in the Grubbs contrast of the M2 elasticity test, when compared with the critical value G > 1.887, do not exceed it, that is, the hypothesis that the value is not anomalous is accepted. Furthermore, it shows that the results obtained in the Grubbs contrast of the M3 elasticity test, when compared with the critical value G > 1.887, do not exceed it, that is, the hypothesis that the value is not anomalous is accepted.

Table 7.
Values obtained for the elasticity test of sample M1, M2, and M3, with its 6 replicates.
In Table 8, it can be seen from the results obtained in the Grubbs contrast of the M1 resilience test, when compared with the critical value G > 1.887, that only one value exceeds it, that is, the hypothesis that the value is not anomalous is rejected only for the first value of M1. Likewise, the results obtained in the Grubbs contrast of the M2 resilience test, when compared with the critical value G > 1.887, do not exceed it, that is, the hypothesis that the value is not anomalous is accepted. The results obtained in the Grubbs contrast of the M3 resilience test, when compared with the critical value G > 1.887, do not exceed it, that is, the hypothesis that the value is not anomalous is accepted.

Table 8.
Values were obtained for the resilience test of samples M1, M2, and M3 with their 6 replicates.
After removing the abnormal values from our database, the coefficient of variation was measured for each sample and in each test. Being an example of a relative error, which is why it is expressed as a percentage, it is calculated for each of the distributions, and the values obtained are compared with each other [36]. In Table 9, the coefficient of variation for each sample in the hardness test can be observed, with M1 and M2 having a high variability with values of 46.10 and 37.36%, respectively; their data are heterogeneous, and the means are not representative, while M3 had a moderate variability with 27.06%, so its data are homogeneous and the average is representative.

Table 9.
Coefficient of variation in the hardness test for each sample.
In Table 10 the coefficient of variation for each sample in the elasticity test can be observed, with M1 being the one that presented the least dispersion or very low variability with 3.25% while, on the other hand, M2 and M3 had a low variability of exactly 7.33 and 7.25%, respectively. In the three samples the data are homogeneous, their means being representative.

Table 10.
Coefficient of variation in the elasticity test for each sample.
Table 11 shows the coefficient of variation in the resilience test for each sample; M1 is the one that had the least dispersion or very low variability with 5.88% while, on the other hand, M2 and M3 presented a low variability with 9.60 and 10.80%, respectively. In the three samples the data are homogeneous, their means being representative.

Table 11.
Coefficient of variation in the resilience test for each sample.
Table 12 shows the comparison between our results obtained in the physical characterization (density, moisture absorption, and elasticity) of the biomaterial carried out in this project and the results of other authors.

Table 12.
Comparison with the values of other biomaterials.
Table 13 shows the comparison between our results obtained in the physical characterization (density, moisture absorption, and elasticity) of the biomaterial in this project and the standard data for polystyrene foam.

Table 13.
Comparison with standard values for polystyrene foam.
4. Discussion
This type of research, where microorganisms are immersed, is complex since there are factors involved in mycelial development, so constant and vigorous monitoring must be carried out [41]. Even though the substrate was sterilized and the samples, including the controls, were kept under controlled conditions (darkness, temperature, humidity, and time), one of the samples presented contamination by external agents since the mycelium is susceptible and vulnerable in its development phase [42]. To clarify the difference in performance in the samples, there are probable factors, such as, when injecting each sample, it can be deduced that there was not an adequate distribution of the mycelium seeds. Adding to the above, Elsacker et al. (2020) emphasize that in a large percentage of laboratories, there is a tendency to work with microorganisms such as bacteria and fungi, among others, which is why the probability of contamination of the substrate or mycelium, as well as the equipment, continues to exist during the sowing phase. Instruments are in an aseptic condition [43]. The slightest change in any of the parameters can trigger a substantial variation in the development and homogeneity of the colonization [44] and the manipulation of the sowing within the substrate, evidencing how complex the process is. Work with living organisms depends on controlled conditions to develop biomaterials based on mycelial growth [45]. After all these considerations, it was possible to elaborate on the biomaterial composed of Pleurotus ostreatus mycelium and sugarcane bagasse in question; the procedure is written in the Materials and Methods. This biomaterial does not have its own standards. Therefore, it was compared with the data obtained from biomaterials reported in the literature and with standard values of expanded polystyrene.
Results obtained from the physical–mechanical tests of the biomaterial composed of Pleurotus ostreatus mycelium and sugarcane bagasse were compared with previous investigations of other biomaterials and polystyrene foam, finding significant differences. The low density is considered necessary in competitiveness with synthetic foams; in our results, an average density of 127.86–131.19 kg/m3 was obtained, which means that their density is low; that is, they are light. These values are relatively close compared to the compounds made from Pleurotus ostreatus and Trametes versicolor mycelium with cereal stubble and nut shells by Rodríguez (2018), which obtained a density of 149.37 kg/m3 [37]. Likewise, Ocegueda et al. (2021), in their bioplastics made with Pleurotus ostreatus mycelium in lignocellulosic residues of oak (Quercus castanea Née) and corn cob (Zea mays), the average density values varied from 96–198 kg/m3 [38]. Apples et al. (2019), in their mycelial compounds with Trametes multicolor and Pleurotus ostreatus in substrates such as straw, sawdust, and cotton, obtained average values of 100–390 kg/m3 [39]. On the other hand, Jones et al. (2020) mention that biomaterials containing forest residue substrates, such as sawdust, have a density ranging between 60 and 300 kg/m3. In contrast, those that contain “filler” agro-industrial waste substrates, such as straw fibers, among others, have low densities of around 130 kg/m3; this could be verified since, in our research, we used agro-industrial waste, and our values are within the range indicated by the authors [40]. Supporting this statement, we have the work of Rey et al. (2018), who made biomaterials from Pleurotus ostreatus and residues such as sugarcane bagasse and rice grass, having an average density of 132.7 kg/m3. The value was more similar to our result, and the authors highlight that the development of this type of biomaterial based on fungi for different applications is no longer an insubstantial issue and that it represents a high degree of originality [30]. Likewise, Susel et al. (2021), in their bioplastics made with Ganoderma lucidum and walnut shell residues and sawdust, obtained a density of 169 kg/m3, indicating that they can be used in the non-structural construction field and recommending further analysis since these types of compounds are new and innovative [41]. However, compared with the standard values of EPS, they present intervals that oscillate from 10–50 kg/m3, a much lower range than the one obtained in this investigation. Despite this, the authors affirm that these biomaterials can be used as an alternative packaging material to traditional polystyrene foams for packaging different items or food, where their combination of low density and low cost gives a competitive advantage and, even though production is still limited, the production process is improving rapidly.
According to Pelletier et al. (2019), compounds based on mycelial growth can show variation even if the same type of fiber is used since it is directly proportional to the expansion of the mycelium’s tissue within the substrate, with more excellent moisture absorption in those that contain intertwined fibers, facilitating the propagation of water in the biomaterial [46]. In our results, the percentage of water absorption was 23.55% for sample M1, 11.79% for sample M2, and 15.35% for sample M3, presenting M2 as having the best behavior. That is, it has a lower absorption. This percentage obtained is relatively low compared to the results by Rodríguez (2018), who found an absorption of 15%; they mention that this water absorption capacity of mycelium-based materials generates tremendous interest since they can be superabsorbent [37]. Similarly, Apples et al. (2019) obtained a result of around 11.63%; this result is similar to ours, showing the homogeneity in the degree of colonization, as well as that the thickness of the fungal skin and the type of substrate influence the rigidity and the water resistance of biomaterials [39].
On the other hand, Ocegueda Vega (2021) obtained a high degree of water absorption, between 33.66 and 40.2%, in their bioplastics. The author indicates that instead of seeing their high absorption capacity as a disadvantage, it can be leveraged in different areas [38]. Checking these data, they were verified with the hypothesis proposed by Jones et al. (2020), which indicates that the absorption percentage of biomaterials made with forest residues varies from 30–43%, while that of those that include only agro-industrial residues is much lower with a range of 10–26%. This could also be verified with our data [40]. Likewise, Rodríguez, Sarache, and Orrego (2014) mention that the percentage of water absorption of these compounds can directly alter the physical tests since both the substrates and the mycelium absorb a significant amount of water [47]. Adding to the above, one of the drawbacks of the materials made from mycelium is its moisture absorption since it is higher compared to the standard values of expanded polystyrene (0.03–9% by weight) and can be a possible difficulty if it is applied within the construction sector (roofs, walls, among others) because it would cause leaks. The limitation in the volume of water absorption of these biomaterials increases their resistance to degradation when exposed to environmental conditions such as humidity, sunlight, and temperature variation, preserving their mechanical properties and dimensional stability [48].
In the texture test, results of hardness, elasticity, and resilience were obtained. Samples M1, M2, and M3 obtained values of 0.538, 0.682, and 0.68 MPa, respectively, for elasticity. It was observed that the maximum stress in the fibers was recorded for sample M3, showing a difference between samples M1 and M3, the latter results being more significant. In research conducted by Appels et al. (2019), they subjected their samples to hot, cold, and unpressed processes, and the heat-pressed samples had a higher elastic modulus than the cold-pressed and non-pressed ones, obtaining a modulus of 0.24 MPa [39]. Likewise, Susel et al. (2021) obtained a value of 0.392 MPa, concluding that the material has a high compressive strength and stressing that these compounds depend on the type of waste used as a substrate for the mycelium [41]. Likewise, Jones et al. (2020) obtained average values of 1.43 M/Pa, indicating that the material can be used in applications where the support of high flexural loads is not demanded and recommending that an additional improvement of mycelium materials to promote colonization in the central part of the substrate is needed [40]. Adding to the above, the authors in the literature mention that this characteristic can be affected by the applied techniques, such as pressing, since heat pressing can substantially increase tensile strength. Compared with expanded polystyrene, materials have registered values from 1.24–3.45 MPa. Therefore, density and flexural strength have a directly proportional relationship; conversely, biomaterials are consistent and firm but brittle. The resilience depends on the deviator stress for the lowest density; for higher density, this effect is very slight, increasing as the density of the material increases [49]. Samples M1, M2, and M3 achieved values of 0.163, 0.209, and 0.238 J/m3, respectively, about the same as those mentioned above; as emphasized, density influences mechanical properties. According to the literature, the mycelium’s composition, morphology, and physical–mechanical performance are influenced by substrate content, incubation conditions, and manufacturing processes [50].
On the other hand, this type of biomaterial is still a pioneering field, and a standardized process to produce optimized material properties has yet to be identified. How to customize the types of substrates for certain fungal species still needs to be defined to maximize mycelium yield and optimize mechanical performance. It is of utmost importance to synthesize information from the scientific literature, patents, and experience to identify barriers and possibilities for effective implementation of mycelium-based compounds in industrial manufacturing, in the architectural interior design of apartments, or the packaging sector.
5. Conclusions
Fabrication of a substitute biomaterial for polystyrene foam through mycelial growth in sugarcane bagasse was possible. A significant contribution to the biological research of mycelium was made, showing a viable and alternative option for solid waste management. In the results obtained, the average density of the three samples varies between 127.86 and 131.19 kg/m3. In addition, the M1 sample had a high percentage of water absorption at 23.55%, while M2 showed a better behavior with 11.79%. In the bending test, the samples M2 and M3 presented more significant stresses with values of 0.682 and M3 0.68 MPa, respectively, being structurally more resistant to bending and, simultaneously, lighter. Results also show that M2 was the sample that obtained the most efficient and constant values in all tests compared to the others. When comparing physical–mechanical characteristics with EPS, it was found that fabricated biomaterial has noticeable and significant differences. However, similar values to those of the mycelium-based compounds made by other authors were observed. Research suggests fabricated material has advantages such as low environmental impact and low production cost, making it a promising ecological alternative. The drawbacks are their percentage of water absorption and the processing time, which can take a minimum of a month.
It is advisable to investigate the use of agro-industrial residues, such as wood sawdust, mixtures of different substrates, as well as other species of fungus. Additionally, a prior study of the agro-industrial substrates (sugarcane) used should be considered since they may contain pesticide residues as well as a lower amount of ash, which may adversely affect the mycelial development of the fungus and, consequently, the biomaterial’s properties.
Author Contributions
Conceptualization, M.M.A.-C. and A.R.-L.; methodology, M.A.Q.-A. and R.S.; software, M.M.A.-C. and A.R.-L.; validation, M.M.A.-C. and A.R.-L.; formal analysis, M.M.A.-C. and A.R.-L.; investigation, L.C.-C., M.M.A.-C. and A.R.-L.; resources, M.M.A.-C. and A.R.-L.; data curation, M.M.A.-C. and A.R.-L.; writing—original draft preparation, W.R.-V. and S.M.B.; writing—review and editing, W.R.-V. and S.R.-F.; visualization, M.A.Q.-A.; supervision, R.S. and S.R.-F.; project administration, W.R.-V.; funding acquisition, M.M.A.-C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- United Nations. World Population Prospects 2022: Summary of Results; United Nations: San Francisco, CA, USA, 2022; ISBN 9789210014380.
- United Nations Environment Programme. From Pollution to Solution: A Global Assessment of Marine Litter and Plastic Pollution. Available online: https://malaysia.un.org/en/171922-pollution-solution-global-assessment-marine-litter-and-plastic-pollution (accessed on 17 May 2023).
- Lozano Zamora, L.V.; Rojas Fraile, E.A. Elaboración de Paneles Termoaislantes Para Cubierta a Partir de Fibras de Bagazo de la Caña de Azúcar; Universidad La Gran Colombia: Armenia, Colombia, 2019. [Google Scholar]
- Díaz Muñoz, K.; Casanova Guajardo, M.; León Torres, C.A.; Gil Ramírez, L.A.; Bardales Vásquez, C.B.; Cabos Sánchez, J. Producción de Pleurotus Ostreatus (Pleurotaceae) ICFC 153/99 Cultivado Sobre Diferentes Residuos Lignocelulósicos. Arnaldoa 2019, 26, 1177–1184. [Google Scholar] [CrossRef]
- Gopinath, A.; Bahurudeen, A.; Appari, S.; Nanthagopalan, P. A Circular Framework for the Valorisation of Sugar Industry Wastes: Review on the Industrial Symbiosis between Sugar, Construction and Energy Industries. J. Clean. Prod. 2018, 203, 89–108. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Kesari, K.K.; Shurpali, N. Multidisciplinary Approaches to Handling Wastes in Sugar Industries. Water Air Soil Pollut. 2016, 227, 11. [Google Scholar] [CrossRef]
- Fonseca Ballesteros, P.G.; Monroy Munévar, J.F. Propuesta de un Medio Filtrante Biodegradable Mediante Fibras de Celulosa a Partir del Bagazo de Caña Azúcar; Fundación Universidad de América: Bogotá, Colombia, 2022. [Google Scholar]
- Méndez-Matías, A.; Robles, C.; Ruiz-Vega, J.; Castañeda-Hidalgo, E. Compostaje de Residuos Agroindustriales Inoculados Con Hongos Lignocelulósicos y Modificación de La Relación C/N. Rev. Mex. Cienc. Agric. 2018, 9, 271–280. [Google Scholar] [CrossRef]
- Hu, X.Q.; Chen, F.; Li, N.; Bai, W.; Bai, R.K. Photo-and Chemo-Degradable Polystyrene Containing & ITo&IT-Nitrobenzyl Ester and Trithiocarbonate Moieties. Acta Polym. Sin. 2017, 12, 1939–1946. [Google Scholar]
- Poeta, G.; Fanelli, G.; Pietrelli, L.; Acosta, A.T.; Battisti, C. Plastisphere in Action: Evidence for an Interaction between Expanded Polysty-Rene and Dunal Plants. Environ. Sci. Pollut. Res. 2017, 24, 11856–11859. [Google Scholar] [CrossRef]
- Barrera Castro, G.P.; Olaya Florez, J.J.; Ocampo Carmona, L.M. Production and Characterization of the Mechanical and Thermal Properties of Expanded Polystyrene with Recycled Material. Ing. Univ. 2017, 21, 177–194. [Google Scholar] [CrossRef]
- van Calcar, C.J.; van Emmerik, T.H.M. Abundance of Plastic Debris across European and Asian Rivers. Environ. Res. Lett. 2019, 14, 124051. [Google Scholar] [CrossRef]
- Turner, A. Foamed Polystyrene in the Marine Environment: Sources, Additives, Transport, Behavior, and Impacts. Environ. Sci. Technol. 2020, 54, 10411–10420. [Google Scholar] [CrossRef]
- Ward, C.P.; Armstrong, C.J.; Walsh, A.N.; Jackson, J.H.; Reddy, C.M. Sunlight Converts Polystyrene to Carbon Dioxide and Dissolved Organic Carbon. Environ. Sci. Technol. Lett. 2019, 6, 669–674. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Marine Litter: Socio-Economic Study; UN Environment: Nairobi, Kenya, 2017; ISBN 9789280737011. [Google Scholar]
- Yeo, B.G.; Mizukawa, K.; Takada, H.; Tait, H.; Frechou, J. Polycyclic Aromatic Hydrocarbons (PAHs) in New Unexposed and Beached Expanded Polystyrene Foams. Environ. Monit. Contam. Res. 2022, 2, 14–21. [Google Scholar] [CrossRef]
- Rahimi, A.; García, J.M. Chemical Recycling of Waste Plastics for New Materials Production. Nat. Rev. Chem. 2017, 1, 0046. [Google Scholar] [CrossRef]
- Tian, Z.; Kim, S.-K.; Hyun, J.-H. Environmental Distribution of Styrene Oligomers (SOs) Coupled with Their Source Characteristics: Tracing the Origin of SOs in the Environment. J. Hazard. Mater. 2020, 398, 122968. [Google Scholar] [CrossRef] [PubMed]
- Farrelly, T.A.; Shaw, I.C. Polystyrene as Hazardous Household Waste. In Household Hazardous Waste Management; InTech: London, UK, 2017; ISBN 9789535129097. [Google Scholar]
- Maafa, I.M. Pyrolysis of Polystyrene Waste: A Review. Polymers 2021, 13, 225. [Google Scholar] [CrossRef] [PubMed]
- Thornberry, M. Expanding the Knowledge Base of Expanded and Extruded Polystyrene: A Report. 2020. Available online: https://repak.ie/images/uploads/reports/Expanding_the_Knowledge_Base_of_Expanded_and_Extruded_Polystyrene.pdf (accessed on 20 February 2020).
- Espinoza, L.; Israel, B. Diseño de Contenedores Biodegradables Para el Transporte de Alimentos; Universidad del Azuay: Cuenca, Ecuador, 2019; Available online: http://dspace.uazuay.edu.ec/handle/datos/9182 (accessed on 20 February 2020).
- Gutiérrez Moreno, D.R. Gestión Integral de los Residuos Sólidos Domiciliarios Para Mejorar la Calidad Ambiental Urbana en el Distrito de Piura–2017. 2018. Available online: https://repositorio.ucv.edu.pe/handle/20.500.12692/11774 (accessed on 17 May 2023).
- Furci, G. Guía de Campo: Hongos de Chile; Fundación Fungi: Santiago, Chile, 2013. [Google Scholar]
- Freundt Espinosa, P.A. Producción y Comercialización de Hongos Comestibles Para El Mercado Nacional e Internacional; Universidad de Piura: Piura, Perú, 2012. [Google Scholar]
- Pérez, J.C. Pleurotus ostreatus. Available online: https://www.fungipedia.org/hongos/pleurotus-ostreatus.html (accessed on 17 May 2023).
- Gandia, A.; van den Brandhof, J.G.; Appels, F.V.W.; Jones, M.P. Flexible Fungal Materials: Shaping the Future. Trends Biotechnol. 2021, 39, 1321–1331. [Google Scholar] [CrossRef]
- Chavez Franco, A.M.; de los Angeles Tamayo Henao, M. Papel de la Participación Social y Política en Los Procesos Liderados Por Mujeres: Incidencia Real Frente a la Materialización de Derechos, el Bienestar Social y la Calidad de Vida; Corporación Universitaria Minuto de Dios (UNIMINUTO): Bogotá, Colombia, 2012. [Google Scholar]
- Zea, B.; Gary, J.J. Reciclado de Plástico PET; Universidad Católica San Pablo: Arequipa, Perú, 2019. [Google Scholar]
- Rey, J.; David, F. Elaboración de un Material Biológico a Partir del Hongo Pleurotus Ostreatus Para su Utilización en Aplicaciones de Ingeniería; Universidad de Costa Rica: San Pedro, Costa Rica, 2018. [Google Scholar]
- Peña Contreras, K. Caracterización y Biodegradación de los Residuos Lignocelulósicos Domésticos; Universidad Michoacana de San Nicolás de Hidalgo: Morelia, Mexico, 2016. [Google Scholar]
- AOAC International. Official Methods of Analysis of Aoac Int; AOAC International: Rockville, MD, USA, 2022; ISBN 9780197610145. [Google Scholar]
- ASTM C303-10; Standard Test Method for Dimensions and Density of Preformed Block and Board—Type Thermal Insulation. ASTM: West Conshohocken, PA, USA, 2016.
- ASTM D570-98; Standard Test Method for Water Absorption of Plastics. ASTM: West Conshohocken, PA, USA, 2018.
- Shettar, M.; Doshi, M.; Rawat, A.K. Study on Mechanical Properties and Water Uptake of Polyester-Nanoclay Nanocomposite and Analysis of Wear Property Using RSM. J. Mater. Res. Technol. 2021, 14, 1618–1629. [Google Scholar] [CrossRef]
- Miller, J.N.; Miller, J.C. Estadística y Quimiometría Para Química Analítica; No. 543.062: 519.23 MIL; Estadisicca y Quimiometria para quimica analítica: Madrid, Spain, 2002. [Google Scholar]
- Rodríguez, S. Material biobasado compuesto por el micelio de hongos descomponedores de madera y residuos agroindustriales. RChD Creación Pensam. 2018, 3. [Google Scholar] [CrossRef]
- Ocegueda Vega, D.S. Bioplástico por Biodesarrollo Micelial de Pleurotus ostreatus en Sustratos de Residuos Lignocelulósicos de Encino (Quercus castanea neé) y Olote de Maíz (Zea mayz). 2021. Available online: http://bibliotecavirtual.dgb.umich.mx:8083/xmlui/handle/DGB_UMICH/6379 (accessed on 17 May 2023).
- Appels, F.V.W.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.B.; Dijksterhuis, J.; Krijgsheld, P.; Wösten, H.A.B. Fabrication Factors Influencing Mechanical, Moisture- and Water-Related Properties of Mycelium-Based Composites. Mater. Des. 2019, 161, 64–71. [Google Scholar] [CrossRef]
- Jones, M.; Mautner, A.; Luenco, S.; Bismarck, A.; John, S. Engineered Mycelium Composite Construction Materials from Fungal Biorefineries: A Critical Review. Mater. Des. 2020, 187, 108397. [Google Scholar] [CrossRef]
- Susel, G.D.E.; Domínguez, E.M.H.; Fernández, A.G.E.; Cervantes, J.Á.; Medellín, L.D.R.; Mendoza, B.M. Biomaterial obtenido a partir de micelio de hongo (ganoderma lucidum) y residuos agrícolas: Biomaterial obtained from fungus micellium (ganoderma lucidum) and agricultural wastes. South Fla. J. Dev. 2021, 2, 4663–4681. [Google Scholar] [CrossRef]
- Sydor, M.; Bonenberg, A.; Doczekalska, B.; Cofta, G. Mycelium-based composites in art, architecture, and interior design: A review. Polymers 2022, 14, 145. [Google Scholar] [CrossRef]
- Elsacker, E.; Vandelook, S.; Van Wylick, A.; Ruytinx, J.; De Laet, L.; Peeters, E. A comprehensive framework for the production of mycelium-based lignocellulosic composites. Sci. Total Environ. 2020, 725, 138431. [Google Scholar] [CrossRef] [PubMed]
- Attias, N.; Danai, O.; Tarazi, E.; Pereman, I.; Grobman, Y.J. Implementing bio-design tools to develop mycelium-based products. Des. J. 2019, 22, 1647–1657. [Google Scholar] [CrossRef]
- Gil Jaime, B.S.; Rojas Sanabria, D.F. Entropía Aislante Termoacústico a Partir de Fique, Micelio y Heno. 2021. Available online: https://repository.ugc.edu.co/handle/11396/7091 (accessed on 17 May 2023).
- Pelletier, M.G.; Holt, G.A.; Wanjura, J.D.; Greetham, L.; McIntyre, G.; Bayer, E.; Kaplan-Bie, J. Acoustic Evaluation of Mycological Biopolymer, an All-Natural Closed Cell Foam Alternative. Ind. Crops Prod. 2019, 139, 111533. [Google Scholar] [CrossRef]
- Rodríguez, L.J.; Sarache, W.A.; Orrego, C.E. Compuestos de Poliéster Reforzados con Fibra de Plátano/Banano (Musa paradisiaca) Modificada Químicamente: Comparación con Fibra de Vidrio y Fique (Furcraea andina). Inf. Tecnol. 2014, 25, 27–34. [Google Scholar] [CrossRef]
- Yang, L.; Park, D.; Qin, Z. Material Function of Mycelium-Based Bio-Composite: A Review. Front. Mater. 2021, 8, 737377. [Google Scholar] [CrossRef]
- Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A.; Athanassiou, A. Advanced materials from fungal mycelium: Fabrication and tuning of physical properties. Sci. Rep. 2017, 7, 41292. [Google Scholar] [CrossRef]
- Islam, M.R.; Tudryn, G.; Bucinell, R.; Schadler, L.; Picu, R.C. Morphology and Mechanics of Fungal Mycelium. Sci. Rep. 2017, 7, 13070. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).