Biodiversity Conservation of National Parks and Nature-Protected Areas in West Africa: The Case of Kainji National Park, Nigeria
Abstract
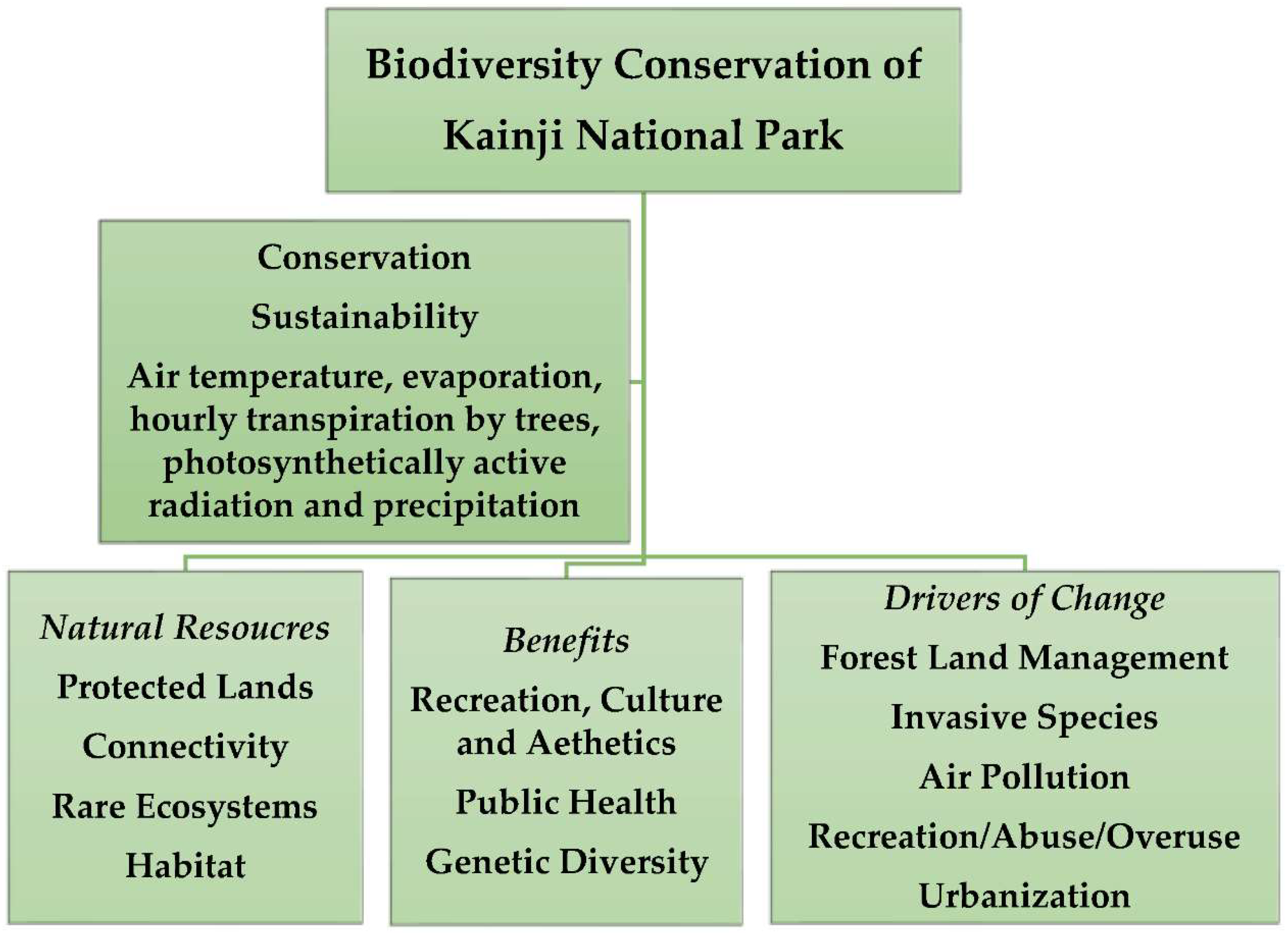
:1. Introduction
2. Literature Review: Biodiversity, Protected Areas, and National Parks
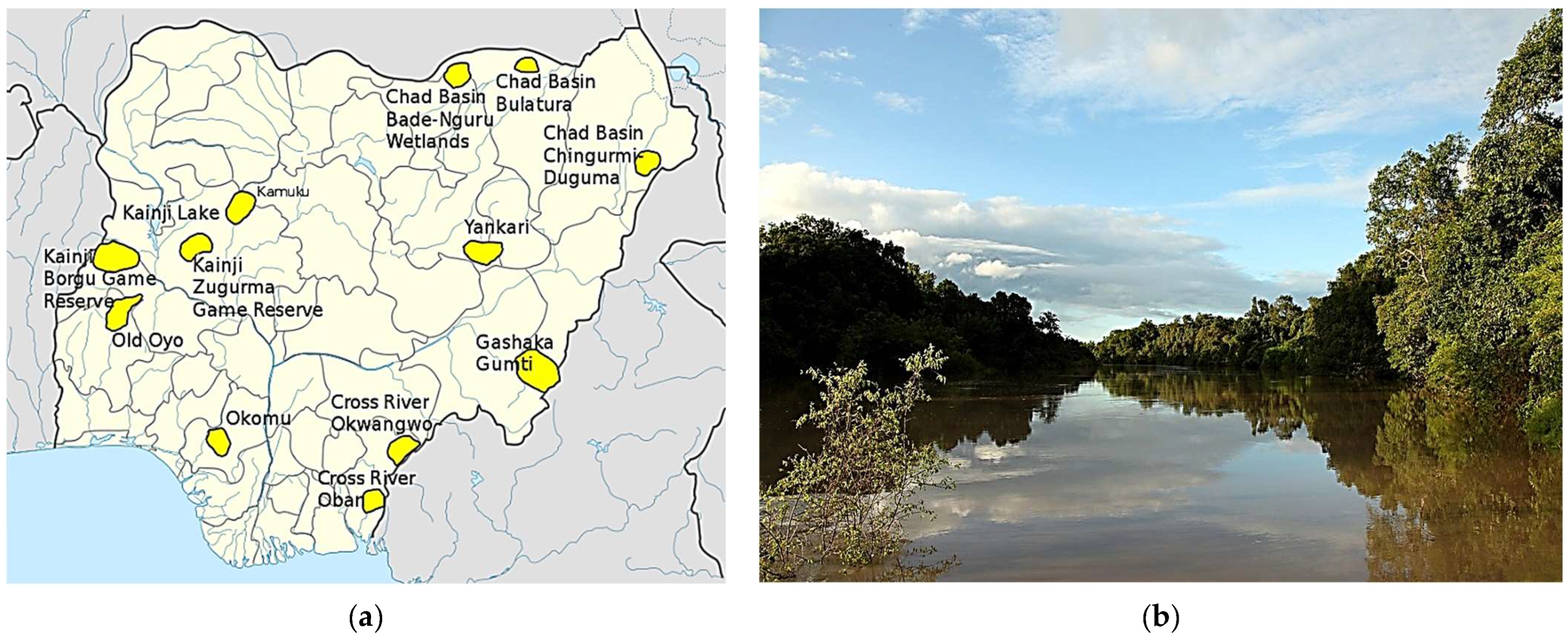
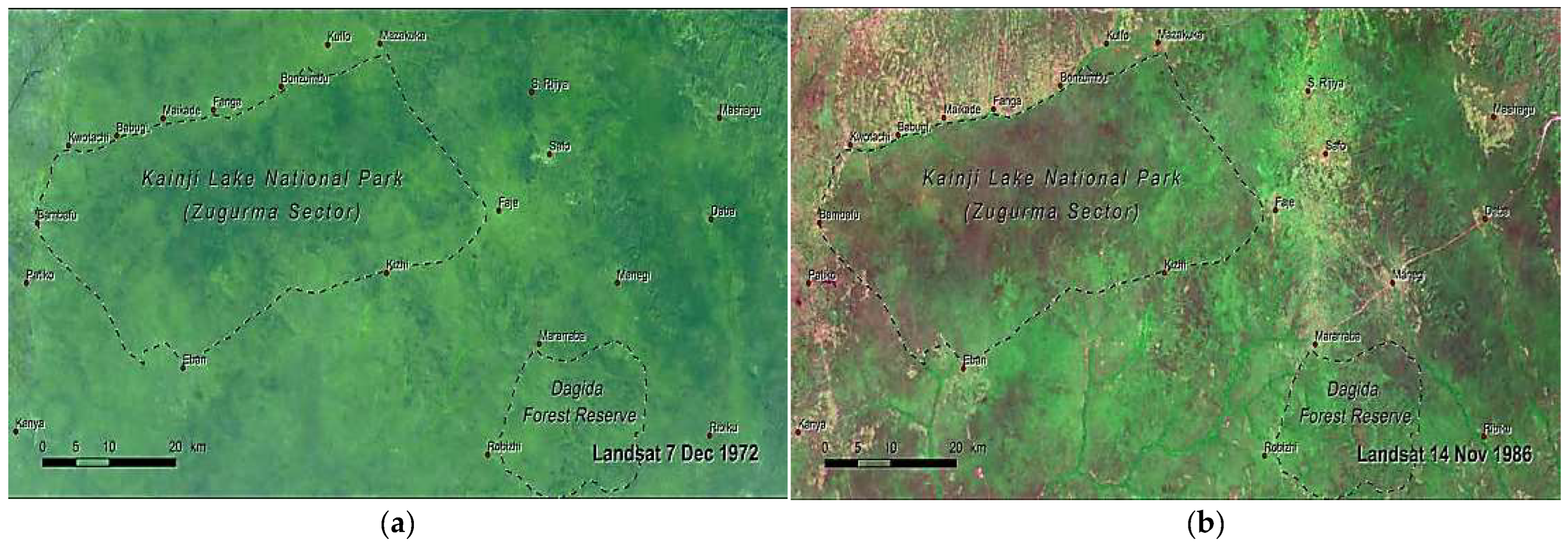
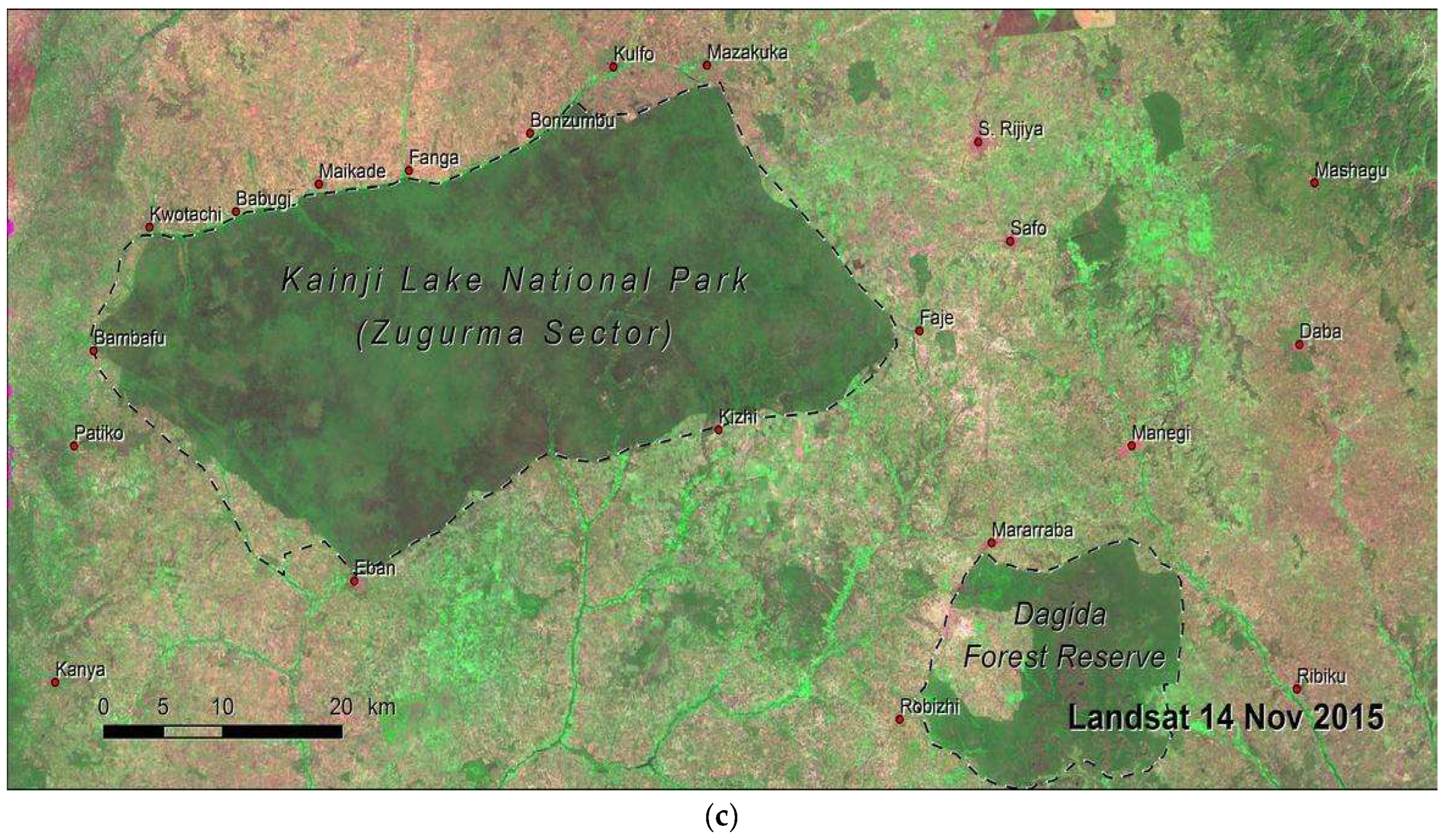
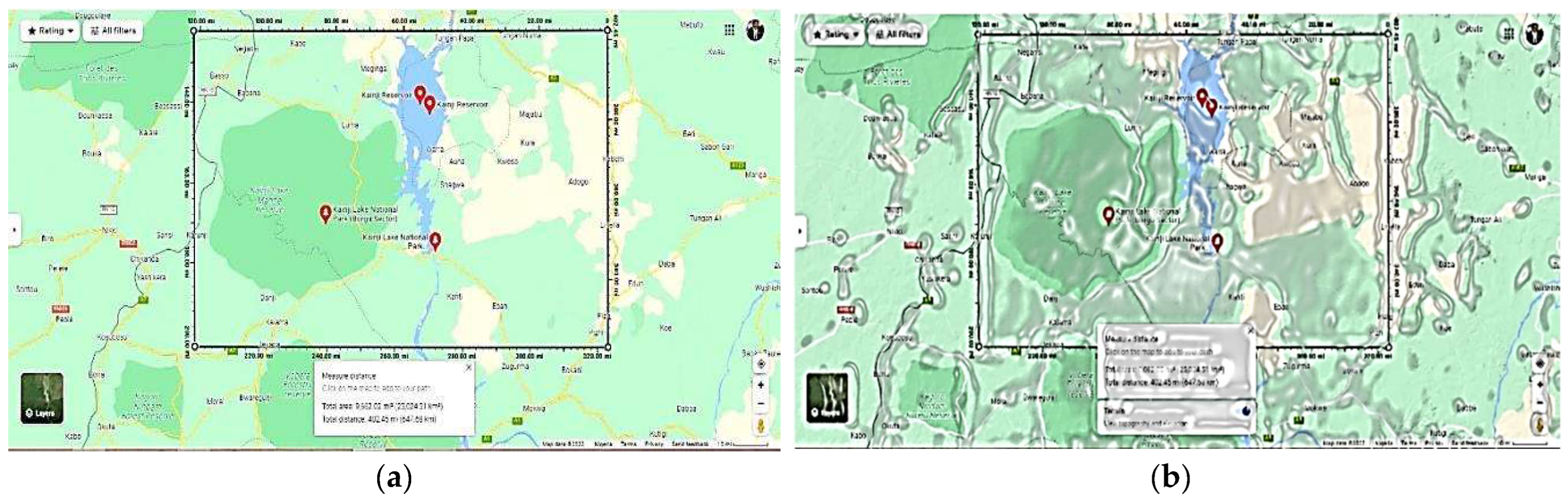
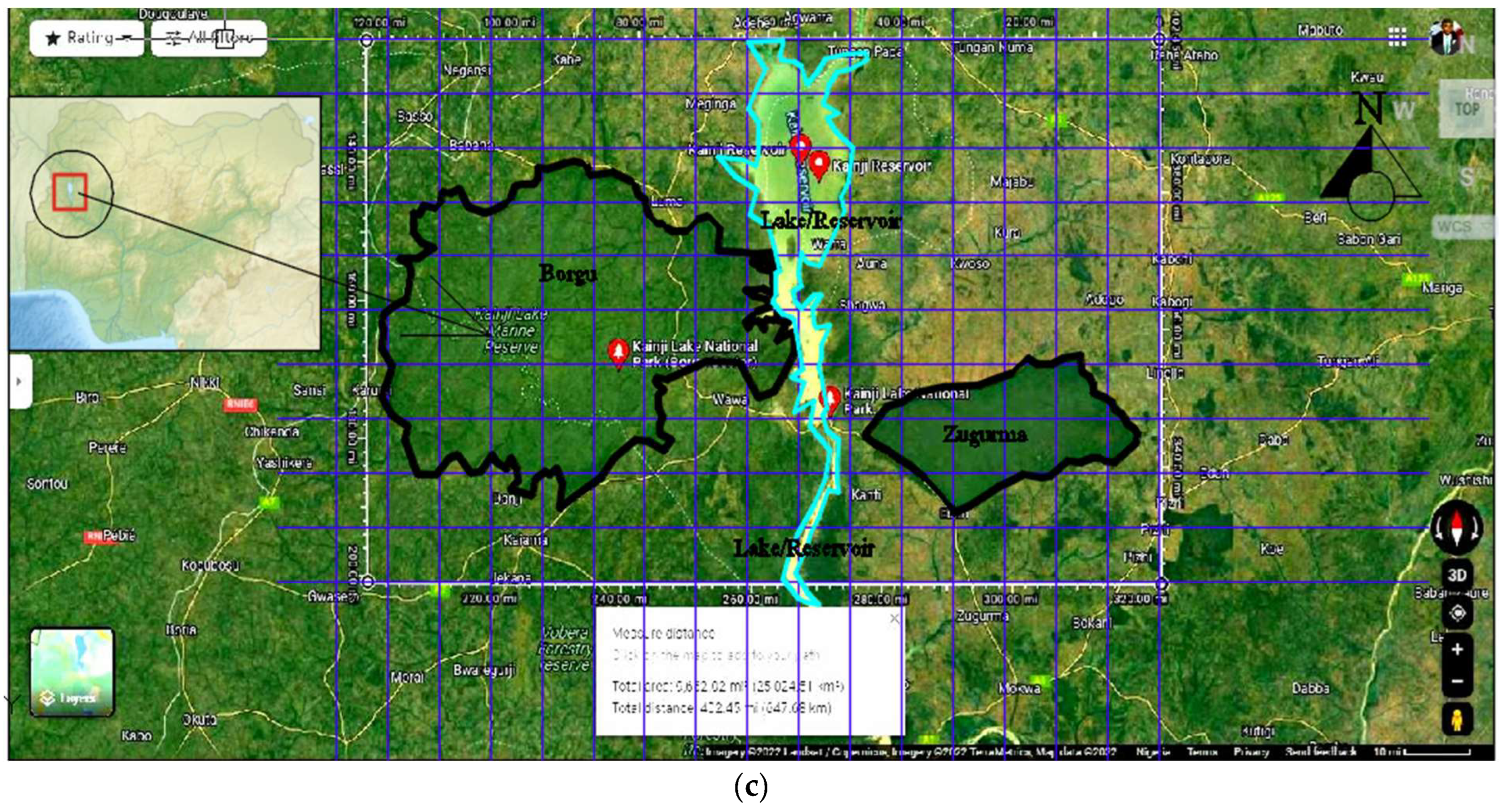
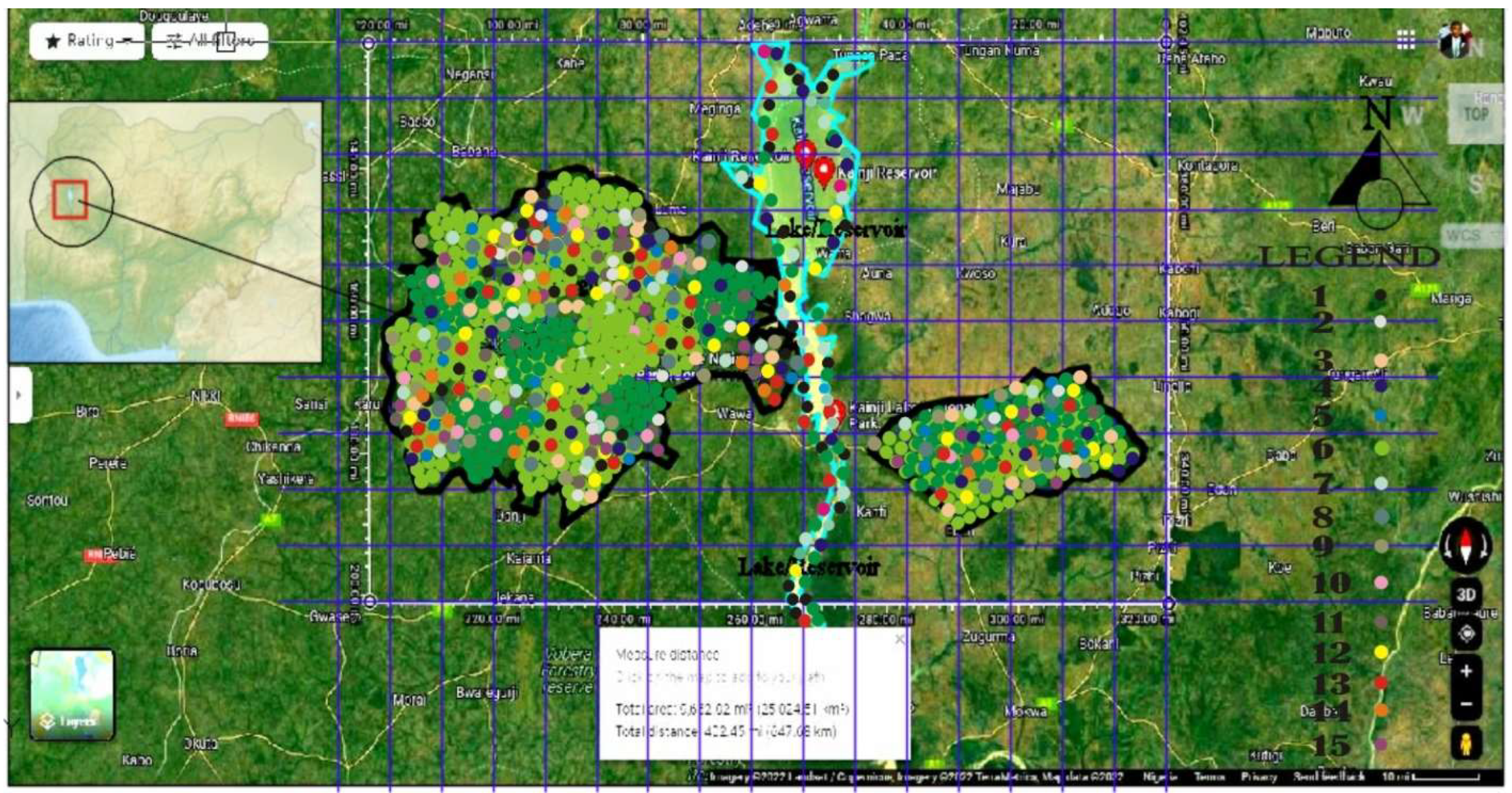
The Study Area: Kainji National Park, Nigeria (West Africa)
3. Methods: The Study Area Protocol
Data Collection
4. Results
- Crown Health (dieback): Excellent = 0% dieback, Good = 1–10% dieback, Fair = 10–25% dieback, Poor = 25–50% dieback, Critical = 50–75% dieback, Dying = 75–99% dieback, Dead = 100% dieback.
- Crown Health (condition): Excellent = 100% condition, Good = 90–99% condition, Fair = 75–90% condition, Poor = 50–75% condition, Critical = 25–50% condition, Dying = 1–25% condition, Dead = 0% condition.
Tree Characteristics of Kainji National Park
5. Discussion
5.1. Trees’ Benefits for Biodiversity Conservation
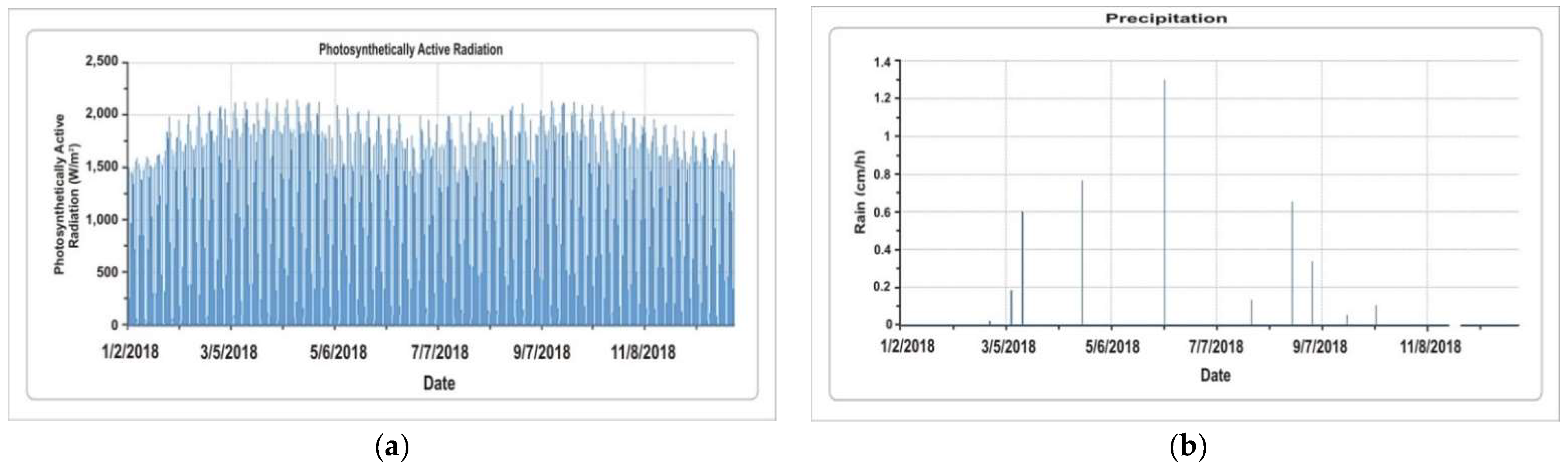
5.1.1. Photosynthetically Active Radiation and Precipitation
5.1.2. Temperature and Transpiration
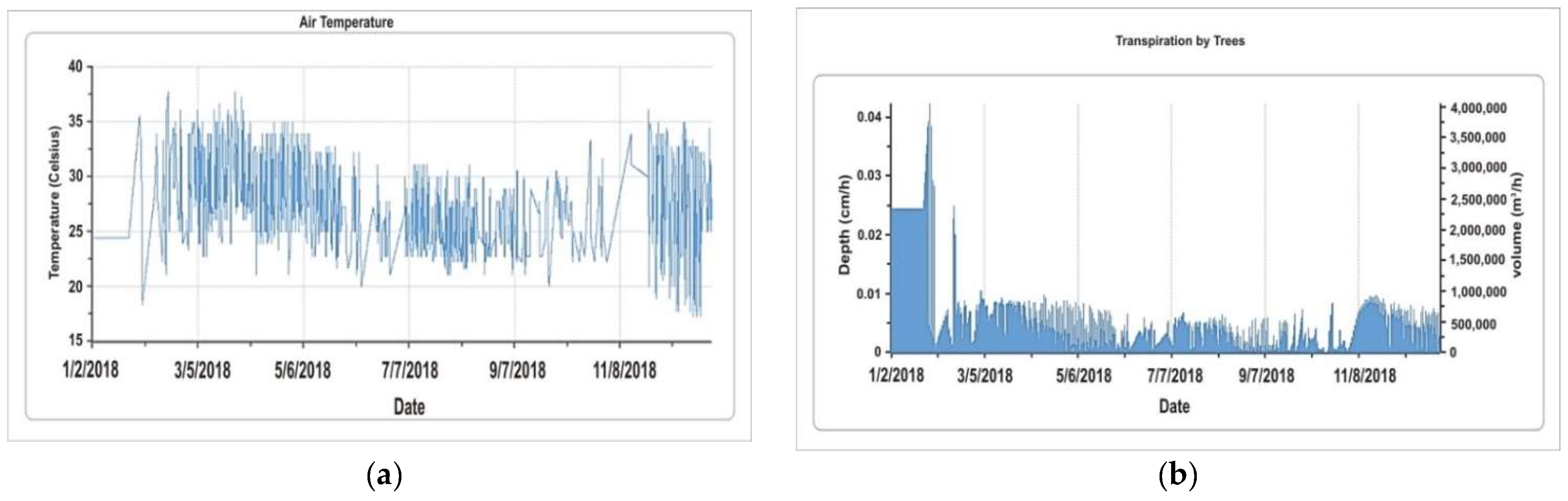
5.1.3. Evaporation and Water Intercepted by Trees

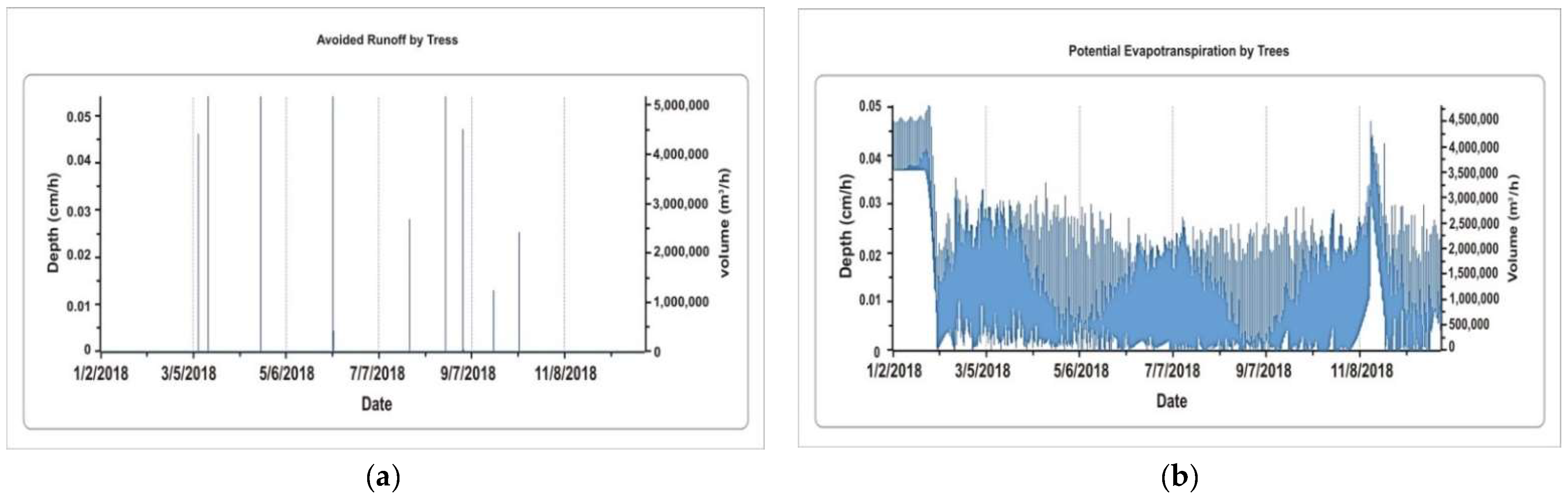
5.1.4. Avoided Runoff by Trees and Potential Evaporation by Trees
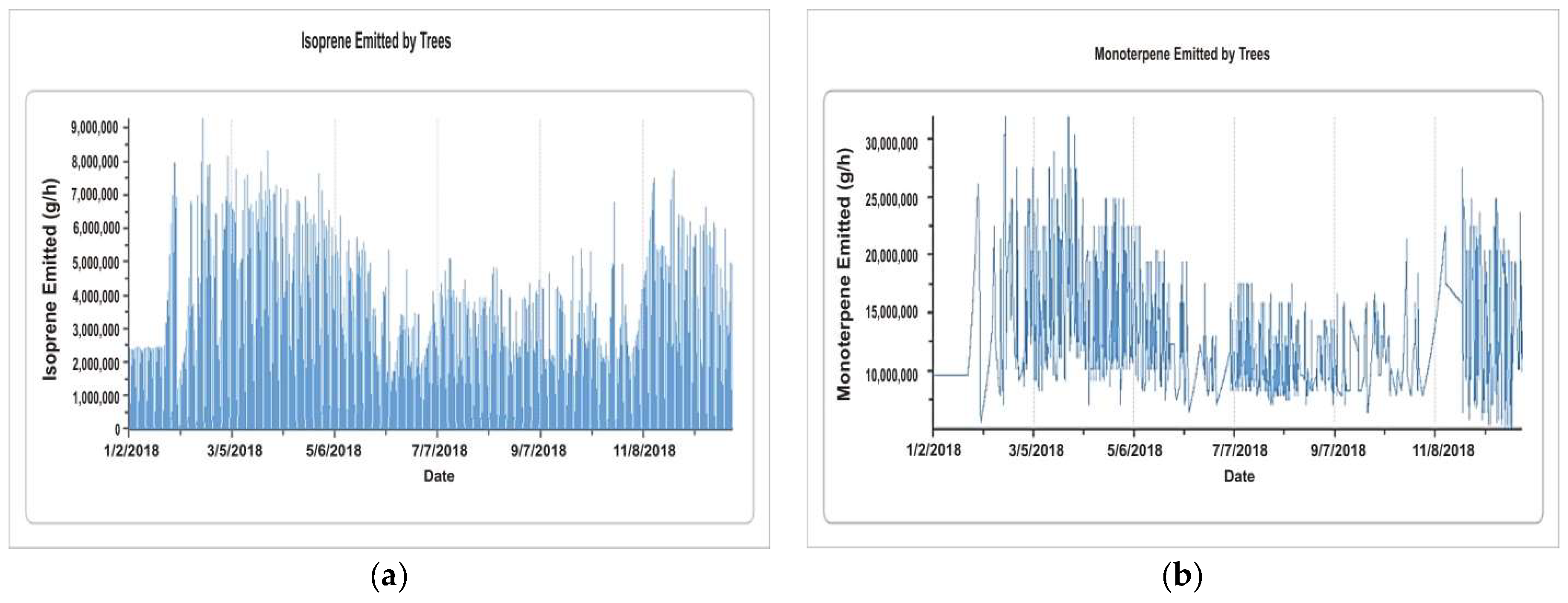
5.1.5. Isoprene and Monoterpene by Trees
5.2. Air Pollution Removal by Urban Trees
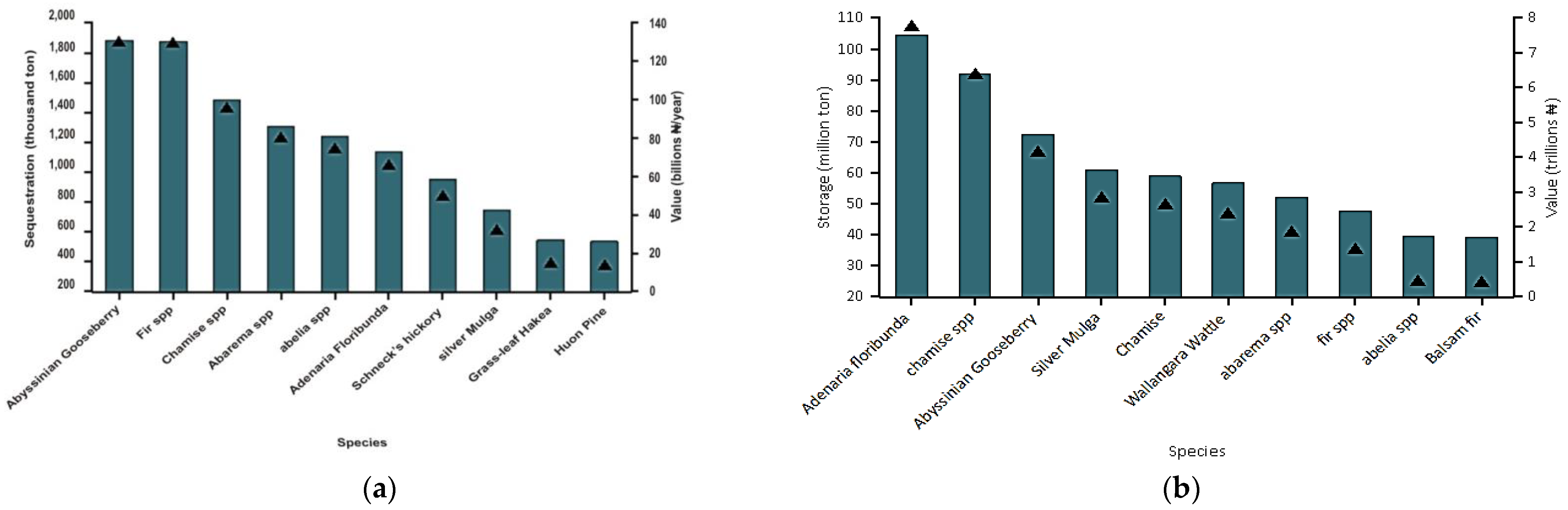
5.3. Carbon Storage and Sequestration
5.4. Oxygen Production
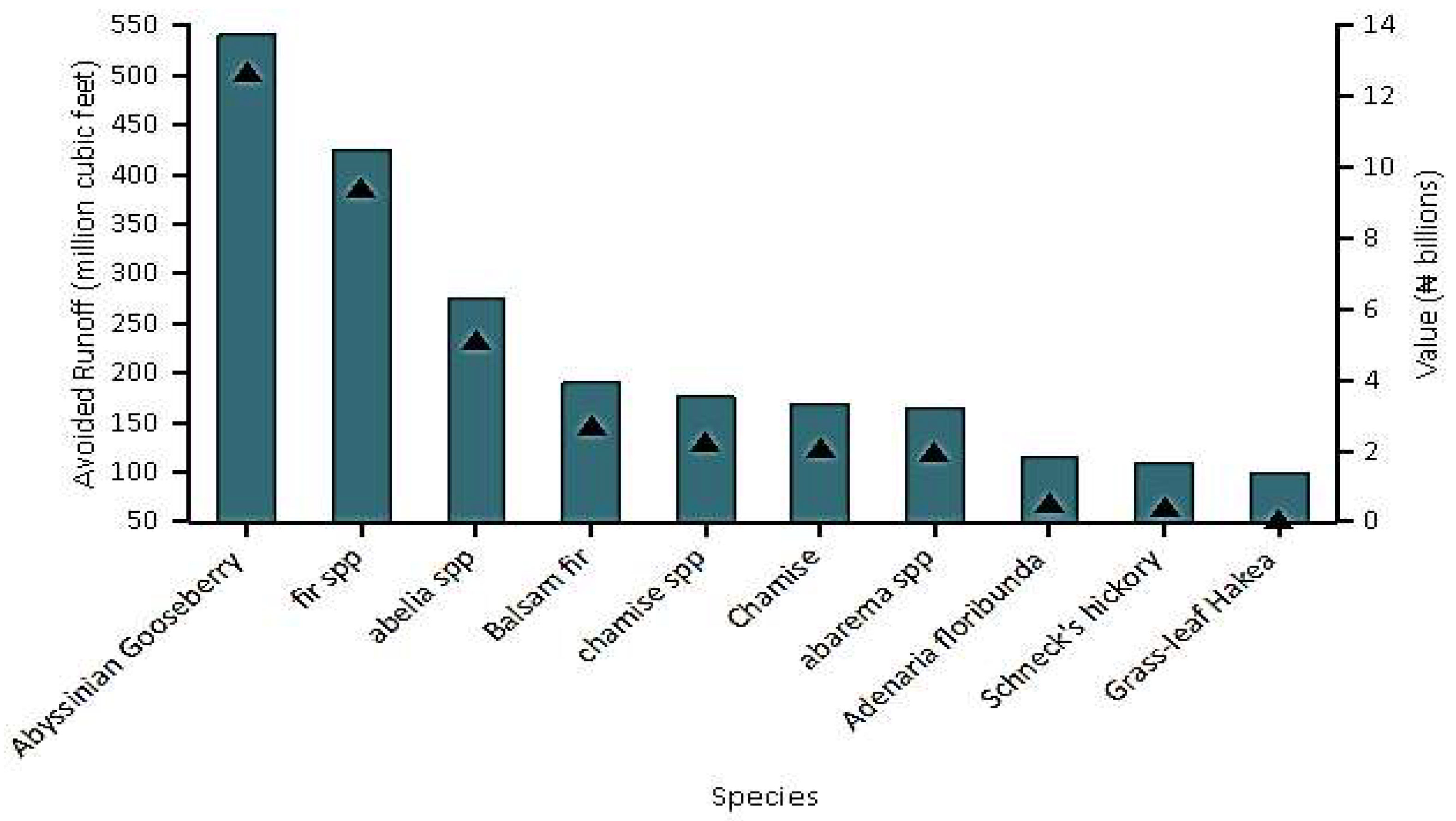
5.5. Avoided Runoff
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Conservation International. 2007. Available online: https://www.yumpu.com/en/document/view/37491711/annual-report-2008-library-conservation-international (accessed on 1 December 2021).
- United Nations Environment Programme, World Conservation Monitoring Centre. Kainji Lake National Park. 2007. Available online: https://web.archive.org/web/20070930012150/unep-wcmc.org/sites/pa/0302p.htm (accessed on 1 December 2021).
- Kapos, V.; Werner, A.; Kurz, T.; Gardner, J.F.; Guariguata, M.; Koh, L.P.; Mansourian, S. Impacts of forest and land management on biodiversity and carbon. IUFRO World Ser. 2012, 31, 53–80. Available online: https://www.fs.usda.gov/treesearch/pubs/47890 (accessed on 1 December 2021).
- Eagles, P.F.J.; McCool, S.F. Tourism in National Parks and Protected Areas: Planning and Management; Cabi: Wallingford, UK, 2002. [Google Scholar]
- Brown, J.; Brent, M. Extending the reach of national parks and protected areas: Local stewardship initiatives. In National Parks and Protected Areas; Springer: Berlin/Heidelberg, Germany, 1997; pp. 103–116. [Google Scholar] [CrossRef]
- Clerici, N.; Salazar, C.; Pardo-Díaz, C.; Jiggins, C.D.; Richardson, J.E.; Linares, M. Peace in Colombia is a critical moment for Neotropical connectivity and conservation: Save the northern Andes–Amazon Biodiversity Bridge. Conserv. Lett. 2019, 12, e12594. [Google Scholar] [CrossRef]
- Mittermeier, R.A. Conservation International and biodiversity conservation. Nature 2000, 405, 254. [Google Scholar] [CrossRef]
- Mallon, C.A.; Poly, F.; Le Roux, X.; Marring, I.; van Elsas, J.D.; Salles, J.F. Resource pulses can alleviate the biodiversity–invasion relationship in soil microbial communities. Ecology 2015, 96, 915–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Union for Conservation of Nature; International Union for Conservation of Nature, Natural Resources; Species Survival Commission; IUCN Species Survival Commission. IUCN Red List Categories and Criteria; IUCN: Gland, Switzerland, 2001. [Google Scholar]
- Imarhiagbe, O.; Egboduku, W.O.; Nwankwo, B.J. A review of the biodiversity conservation status of Nigeria. J. Wildl. Biodivers. 2020, 4, 73–83. [Google Scholar] [CrossRef]
- Akindele, E.O.; Ekwemuka, M.C.; Apeverga, P.; Amusa, T.O.; Olajuyigbe, S.; Coker, O.M.; Fatsuma, O.; Abiodun, A. Assessing awareness on biodiversity conservation among Nigerians: The Aichi Biodiversity Target 1. Biodivers. Conserv. 2021, 30, 1947–1970. [Google Scholar] [CrossRef]
- Radić, B.; Gavrilovic, S. Natural Habitat Loss: Causes and Implications of Structural and Functional Changes. In Life on Land; Springer: Berlin/Heidelberg, Germany, 2021; pp. 699–712. [Google Scholar]
- Mukherjee, S. Globalisation and the political economy of protected areas. In Globalisation, Environment and Social Justice; Routledge: Delhi, India, 2018; pp. 109–129. Available online: https://www.taylorfrancis.com/chapters/edit/10.4324/9780429455766-5/globalisation-political-economy-protected-areas-sudeshna-Mukherjee (accessed on 1 December 2021).
- Day, J.; Dudley, N.; Hockings, M.; Holmes, G.; Laffoley, D.D.A.; Stolton, S.; Wells, S.M. Guidelines for Applying the IUCN Protected Area Management Categories to Marine Protected Areas; IUCN: Gland, Switzerland, 2012. [Google Scholar]
- Ortiz, M.J. Aichi biodiversity targets direct and indirect drivers of biodiversity loss. Environ. Law Rev. 2011, 13, 100–106. [Google Scholar] [CrossRef]
- Newbold, T.; Adams, G.L.; Robles, G.A.; Boakes, E.H.; Ferreira, G.B.; Chapman, A.S.A.; Etard, A.; Gibb, R.; Millard, J.; Outhwaiteet, C.L.; et al. Climate and land-use change homogenise terrestrial biodiversity, with consequences for ecosystem functioning and human well-being. Emerg. Top. Life Sci. 2019, 3, 207–219. [Google Scholar] [CrossRef]
- Essl, F.; Guillaume, L.; Lenzner, B.; Pagad, S.; Seebens, H.; Smith, K.; Wilson, J.R.U.; Genovesi, P. The Convention on Biological Diversity (CBD)’s Post-2020 target on invasive alien species—What should it include and how should it be monitored? NeoBiota 2020, 62, 99. Available online: https://neobiotapensoft.net/article/53972/download/pdf (accessed on 1 December 2021). [CrossRef]
- Morgera, E. Dawn of a New Day: The Evolving Relationship between the Convention on Biological Diversity and International Human Rights Law. Wake For. Rev. 2018, 53, 691. Available online: https://heinonline.org/hol-cgi-bin/get_pdf.cgi?handle=hein.journals/wflr53§ion=30 (accessed on 1 December 2021). [CrossRef] [Green Version]
- Prip, C. The Convention on Biological Diversity as a legal framework for safeguarding ecosystem services. Ecosyst. Serv. 2018, 29, 199–204. [Google Scholar] [CrossRef] [Green Version]
- Opoku, A. Biodiversity and the built environment: Implications for the Sustainable Development Goals (SDGs). Resour. Conserv. Recycl. 2019, 141, 1–7. [Google Scholar] [CrossRef]
- Osunsina, I.O.O.; Osunsina, J.; Oduntan, O.O.; Oladiran, Y.; Moyaki, O.T. Awareness and perception of climate change and variability among indigenous people living around Nigerian national parks. Afr. J. Agric. Technol. Environ. 2019, 8, 47–61. Available online: https://www.researchgate.net/profile/Israel-Osunsina/publication/.pdf (accessed on 1 December 2021).
- Elijah, E.; Ojeh, V.N.; Philip, A.H.; Joshua, M.M.; Bonchak, G. Land Transformation of Tropical Forest Conservation of Gashaka-Gumti National Park, Nigeria from 1987–2014. Asian J. Environ. Ecol. 2019, 9, 1–17. Available online: https://www.researchgate.net/publication/332975202_Land_Transformation_of_Tropical_Forest_Conservation_of_Gashaka-Gumti_National_Park_Nigeria_from_1987-2014 (accessed on 1 December 2021).
- Brankov, J.; Glavonjić, T.J.; Pešić, A.M.; Petrović, M.D.; Tretiakova, T.N. Residents’ perceptions of tourism impact on the community in national parks in Serbia. Eur. Ctry. 2019, 11, 124–142. Available online: https://www.researchgate.net/publication/332456114_Residents'_Perceptions_of_Tourism_Impact_on_Community_in_National_Parks_in_Serbia (accessed on 1 December 2021). [CrossRef] [Green Version]
- Convention on Biological Diversity Secretariat of the Convention on Biological Diversity. 2007. Montreal. Available online: https://www.cbd.int/doc/reports/cbd-report-2006-en.pdf (accessed on 1 December 2021).
- Dwyer, J.F.; Nowak, D.J. A national assessment of the urban forest: An overview. Soc. Am. For. 2000, 2000, 157–162. Available online: https://www.researchgate.net/publication/279562620_Sustaining_urban_forests (accessed on 1 December 2021).
- Rana, K.; Munesh, K.; Amit, K. Assessment of annual shoot biomass and carbon storage potential of Grewia optiva: An approach to combat climate change in Garhwal Himalaya. Water Air Soil Pollut. 2020, 231, 1–13. [Google Scholar] [CrossRef]
- Nunes, L.J.R.; Meireles, C.I.R.; Pinto Gomes, C.J.; Almeida Ribeiro, N. Forest contribution to climate change mitigation: Management oriented to carbon capture and storage. Climate 2020, 8, 21. [Google Scholar] [CrossRef] [Green Version]
- Tschora, H.; Cherubini, F. Co-benefits and trade-offs of agroforestry for climate change mitigation and other sustainability goals in West Africa. Glob. Ecol. Conserv. 2020, 22, e00919. [Google Scholar] [CrossRef]
- Goodwin, M.J.; North, M.P.; Zald, H.S.J.; Hurteau, M.D. Changing climate reallocates the carbon debt of frequent-fire forests. Glob. Chang. Biol. 2020, 26, 6180–6189. [Google Scholar] [CrossRef]
- Jovanelly, T.J.; Rodríguez-Montero, L.; Sánchez-Gutiérrez, R.; Mena-Rivera, L.; Thomas, D. Evaluating watershed health in Costa Rican national parks and protected areas. Sustain. Water Resour. Manag. 2020, 6, 1–14. [Google Scholar] [CrossRef]
- Abd-Elaty, I.; Alban, K.; El Shahawy, A. Environmental rethinking of wastewater drains to manage environmental pollution and alleviate water scarcity. Nat. Hazards 2021, 110, 2353–2380. [Google Scholar] [CrossRef]
- Sikdar, P.K. Environmental Management: Issues and Concerns. In Environmental Management: Issues and Concerns in Developing Countries; Springer: Berlin/Heidelberg, Germany, 2021; pp. 1–21. [Google Scholar] [CrossRef]
- Thakholi, L. Conservation labour geographies: Subsuming regional labour into private conservation spaces in South Africa. Geoforum 2021, 123, 1–11. [Google Scholar] [CrossRef]
- Cotillon, S.E. West Africa Land Use and Land Cover Time Series; No. 2017-3004; US Geological Survey: Reston, VA, USA, 2017. [Google Scholar] [CrossRef]
- Akodéwou, A.; Oszwald, J.; Saïdi, S.; Gazull, L.; Akpavi, S.; Akpagana, K.; Gond, V. Land use and land cover dynamics analysis of the Togodo Protected Area and its surroundings in Southeastern Togo, West Africa. Sustainability 2020, 12, 5439. [Google Scholar] [CrossRef]
- Bah, O.A.; Tidiani, K.; Yaffa, S.; Ndiaye, M.L. Land use and land cover dynamics in central river region of The Gambia, West Africa from 1984 to 2017. Am. J. Mod. Energy 2019, 5, 5–18. [Google Scholar] [CrossRef]
- Reid, R.S.; Kruska, R.L.; Muthui, N.; Taye, A.; Wotton, S.; Wilson, C.J.; Mulatu, W. Land-use and land-cover dynamics in response to changes in climatic, biological and socio-political forces: The case of southwestern Ethiopia. Landsc. Ecol. 2020, 15, 339–355. [Google Scholar] [CrossRef]
- Ouedraogo, I.; Tigabu, M.; Savadogo, P.; Compaoré, H.; Odén, P.C.; Ouadba, J.M. Land cover change and its relation with population dynamics in Burkina Faso, West Africa. Land Degrad. Dev. 2010, 21, 453–462. [Google Scholar] [CrossRef]
- Houessou, L.G.; Teka, O.; Imorou, I.T.; Lykke, A.M.; Sinsin, B. Land use and land-cover change at “W” Biosphere Reserve and its surrounding areas in the Benin Republic (West Africa). Environ. Nat. Resour. Res. 2013, 3, 87. [Google Scholar] [CrossRef]
- Kumi, S.; Addo-Fordjour, P.; Fei-Baffoe, B.; Belford, E.J.D.; Ameyaw, Y. Land use land cover dynamics and fragmentation-induced changes in woody plant community structure in a mining landscape, Ghana. Trees For. People 2021, 4, 100070. [Google Scholar] [CrossRef]
- Makwinja, R.; Mengistou, K.S.; Alamirew, T. Impact of land use/land cover dynamics on ecosystem service value—A case from Lake Malombe, Southern Malawi. Environ. Monit. Assess. 2021, 193, 1–23. [Google Scholar] [CrossRef]
- Sinonmatohou, T.D.; Hu, S.; Fan, X.; Ali, M.R. Tropical coastal land-use and land cover changes impact on ecosystem service value during rapid urbanization of Benin, West Africa. Int. J. Environ. Res. Public Health 2021, 18, 7416. [Google Scholar] [CrossRef]
- Gbedzi, D.D.; Ofosu, E.A.; Mortey, E.M.; Obiri-Yeboah, A.; Nyantakyi, E.K.; Siabi, E.K.; Abdallah, F.; Domfeh, M.K.; Amankwah-Minkah, A. Impact of mining on land use land cover change and water quality in the Asutifi North District of Ghana, West Africa. Environ. Chall. 2022, 6, 100441. [Google Scholar] [CrossRef]
- Kindu, M.; Tibebe, D.; Nigussie, D.; Schneider, T.; Döllerer, M.; Teketay, D.; Knoke, T. Understanding Land Use/Land Cover Dynamics in and Surrounding the Ethiopian Church Forests. In State of the Art in Ethiopian Church Forests and Restoration Options; Springer: Berlin/Heidelberg, Germany, 2022; pp. 11–30. [Google Scholar] [CrossRef]
- Achugbu, I.C.; Olufayo, A.A.; Balogun, I.A.; Adefisan, E.A.; Dudhia, J.; Naabil, E. Modeling the spatiotemporal response of dew point temperature, air temperature and rainfall to land use land cover change over West Africa. Modeling Earth Syst. Environ. 2022, 8, 173–198. [Google Scholar] [CrossRef]
- Tang, X.; Adesina, J.A. Integrated Watershed Management Framework and Groundwater Resources in Africa—A Review of West Africa Sub-Region. Water 2022, 14, 288. [Google Scholar] [CrossRef]


| Plot ID | Tree ID | Species Name | DBH | Height | Crown Height | Crown Width | Canopy Cover | Tree Condition | Leaf Area | Leaf Biomass | Leaf Area Index | Basal Area |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (cm) | (m) | (m) | (m) | (m2) | (m2) | (kg) | (m2) | |||||
| 1 | 1 | Abies | 35.0 | 30.0 | 10.0 | 15.0 | 176.7 | Critical | 558.1 | 78.6 | 3.2 | 0.1 |
| 1 | 2 | Abelia | 35.4 | 20.0 | 9.0 | 8.0 | 50.3 | Poor | 334.1 | 17.5 | 6.6 | 0.1 |
| 1 | 3 | Dovyalis abyssinica | 35.0 | 60.0 | 10.0 | 20.0 | 314.2 | Critical | 728.9 | 62.3 | 2.3 | 0.1 |
| 1 | 4 | Abarema | 35.0 | 35.0 | 10.0 | 17.5 | 240.5 | Critical | 171.8 | 14.8 | 0.7 | 0.1 |
| 1 | 5 | Abies balsamea | 50.0 | 30.0 | 2.0 | 15.0 | 176.7 | Critical | 209.1 | 21.8 | 1.2 | 0.2 |
| 1 | 6 | Acacia adunca | 50.0 | 20.0 | 3.0 | 12.5 | 122.7 | Critical | 11.6 | 2.8 | 0.1 | 0.2 |
| 1 | 7 | Lagarostrobos franklinii | 15.0 | 30.0 | 6.0 | 12.5 | 122.7 | Poor | 11.5 | 1.4 | 0.1 | 0.0 |
| 1 | 8 | Adenostoma | 40.0 | 65.0 | 10.0 | 12.5 | 122.7 | Critical | 187.5 | 14.1 | 1.5 | 0.1 |
| 1 | 9 | Adenaria floribunda | 25.0 | 30.0 | 11.0 | 17.5 | 240.5 | Critical | 60.2 | 7.8 | 0.3 | 0.0 |
| 1 | 10 | Salix fragilis | 10.0 | 25.0 | 1.0 | 12.5 | 122.7 | Critical | 5.5 | 0.3 | 0.0 | 0.0 |
| 1 | 11 | Hakea francisiana | 20.0 | 20.0 | 2.0 | 12.5 | 122.7 | Critical | 73.6 | 9.0 | 0.6 | 0.0 |
| 1 | 12 | Acacia argyrophylla | 40.0 | 35.0 | 12.0 | 17.0 | 227.0 | Critical | 43.6 | 10.5 | 0.2 | 0.1 |
| 1 | 13 | Adenaria floribunda | 50.0 | 40.0 | 15.0 | 25.0 | 490.9 | Dying | 36.3 | 4.7 | 0.1 | 0.2 |
| 1 | 14 | Carya x schneckii | 28.0 | 30.0 | 10.0 | 16.5 | 213.8 | Critical | 89.4 | 6.0 | 0.4 | 0.1 |
| 1 | 15 | Adenostoma fasciculatum | 45.0 | 35.0 | 5.0 | 10.0 | 78.5 | Dying | 177.2 | 13.3 | 2.3 | 0.2 |
| Total | 2822.7 | 2698.6 | 265.0 | 1.6 |
| Species Name | Tree Count Value % | Canopy Cover (m2) Value % | Leaf Area (m2) Value % | Leaf Biomass (kg) Value % | Basal Area (m2) Value % | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Abarema | 1 | 6.7 | 240.5 | 8.5 | 171.8 | 6.4 | 14.8 | 5.6 | 0.1 | 6.2 |
| Abelia | 1 | 6.7 | 50.3 | 1.8 | 334.1 | 12.4 | 17.5 | 6.6 | 0.1 | 6.3 |
| Abies | 1 | 6.7 | 176.7 | 6.3 | 558.1 | 20.7 | 78.6 | 29.7 | 0.1 | 6.2 |
| Abies balsamea | 1 | 6.7 | 176.7 | 6.3 | 209.1 | 7.7 | 21.8 | 8.2 | 0.2 | 12.6 |
| Acacia adunca | 1 | 6.7 | 122.7 | 4.3 | 11.6 | 0.4 | 2.8 | 1.1 | 0.2 | 12.6 |
| Acacia argyrophylla | 1 | 6.7 | 227.0 | 8.0 | 43.6 | 1.6 | 10.5 | 4.0 | 0.1 | 8.1 |
| Adenaria floribunda | 2 | 13.3 | 731.4 | 25.9 | 96.5 | 3.6 | 12.5 | 4.7 | 0.2 | 15.8 |
| Adenostoma | 1 | 6.7 | 122.7 | 4.3 | 187.5 | 6.9 | 14.1 | 5.3 | 0.1 | 8.1 |
| Adenostoma fasciculatum | 1 | 6.7 | 78.5 | 2.8 | 177.2 | 6.6 | 13.3 | 5.0 | 0.2 | 10.2 |
| Carya x schneckii | 1 | 6.7 | 213.8 | 7.6 | 89.4 | 3.3 | 6.0 | 2.3 | 0.1 | 4.0 |
| Dovyalis abyssinica | 1 | 6.7 | 314.2 | 11.1 | 728.9 | 27.0 | 62.3 | 23.5 | 0.1 | 6.2 |
| Hakea francisiana | 1 | 6.7 | 4.3 | 4.3 | 73.6 | 2.7 | 9.0 | 3.4 | <0.1 | 2.0 |
| Lagarostrobos franklinii | 1 | 6.7 | 122.7 | 4.3 | 11.5 | 0.4 | 1.4 | 0.5 | <0.1 | 1.1 |
| Salix fragilis | 1 | 6.7 | 122.7 | 4.3 | 5.5 | 0.2 | 0.3 | 0.1 | <0.1 | 0.5 |
| Total | 15 | 100 | 2822.7 | 100 | 2698.6 | 100 | 265.0 | 100 | 1.6 | 100 |
| Summary | Ecosystem Service Values (Approx Values) |
|---|---|
| The number of trees | 563,500,000 (an average of 3,700,000 in each sample frame) |
| Tree Cover | 63.0% |
| Most common species of trees | Adenaria floribunda, Crack willow, Huon Pine connoted species. |
| Percentage of trees less than 15.2 cm diameter | 13.3% |
| Pollution Removal | N/A—data not collected |
| Carbon Storage | 519.4 million metric tons (40.1 trillion NGN) |
| Carbon Sequestration | 10.44 million metric tons (806 billion NGN /year) |
| Oxygen Production | 12.6 million metric tons/year |
| Avoided Runoff | 52.5 million cubic meters/year (50.9 billion NGN/year). |
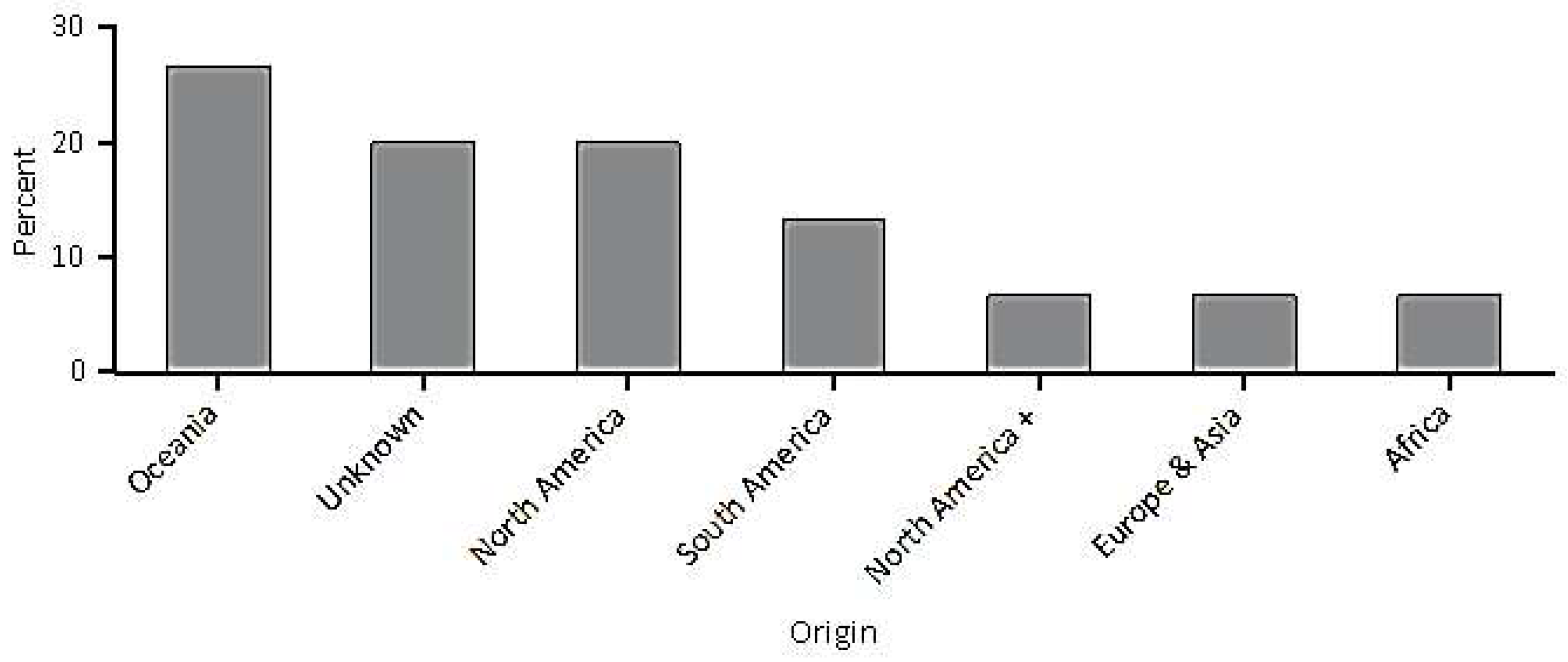
| Stratum | Africa | Europe and Asia | North America | North America + | Oceania | South America | Unknown |
|---|---|---|---|---|---|---|---|
| Urban | 6.7 | 6.7 | 20.0 | 6.7 | 26.7 | 13.3 | 20.0 |
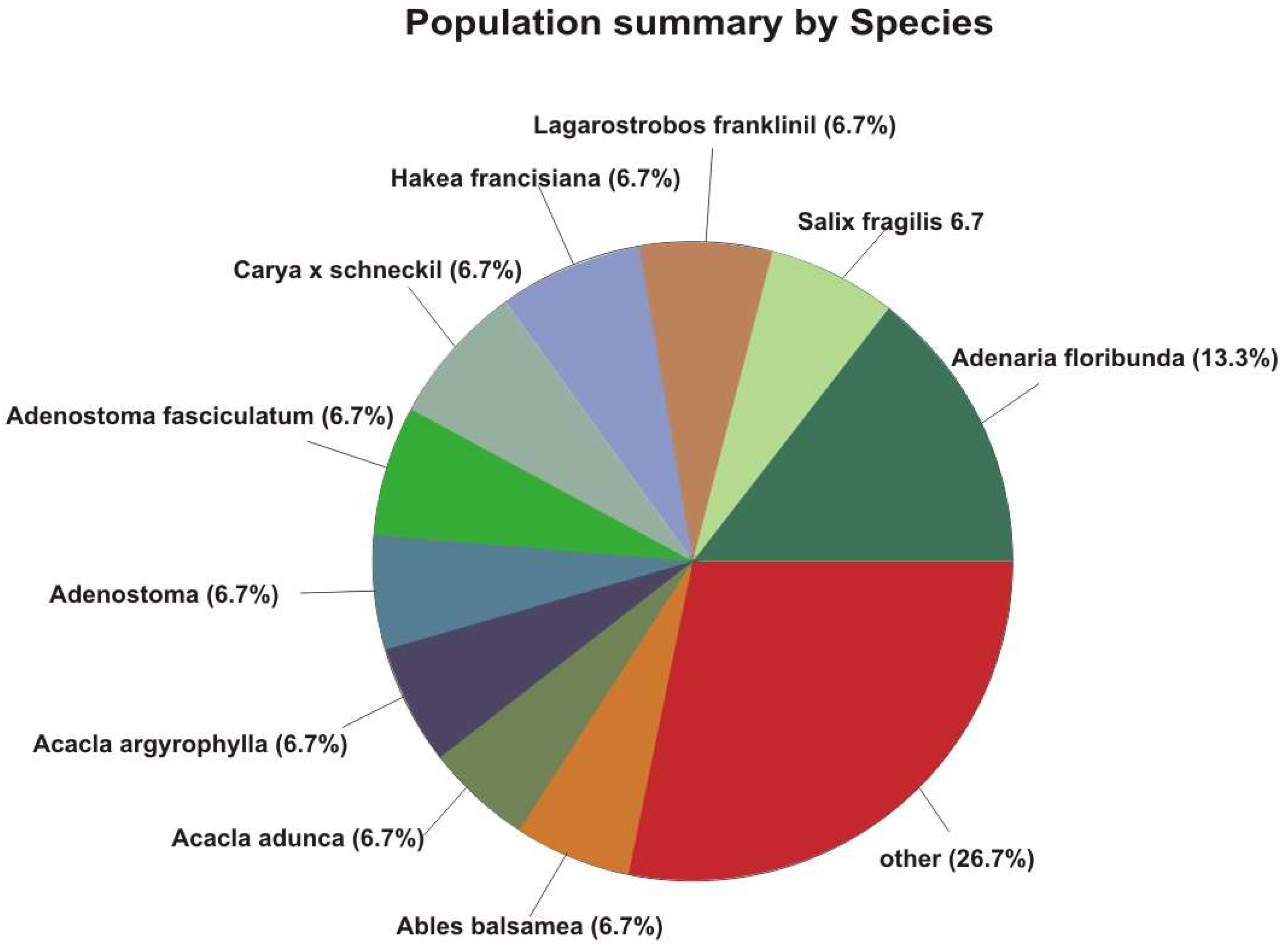
| Species | Number of Trees | Percentage of Population |
|---|---|---|
| Adenaria floribunda | 75,128,717 | 13.3% |
| Salix fragilis | 37,564,359 | 6.7% |
| Lagarostrobos franklinii | 37,564,359 | 6.7% |
| Hakea francisiana | 37,564,359 | 6.7% |
| Carya x schneckii | 37,564,359 | 6.7% |
| Adenostoma fasciculatum | 37,564,359 | 6.7% |
| Adenostoma | 37,564,359 | 6.7% |
| Acacia argyrophylla | 37,564,359 | 6.7% |
| Acacia adunca | 37,564,359 | 6.7% |
| Abies balsamea | 37,564,359 | 6.7% |
| Dovyalis abyssinica | 37,564,359 | 6.7% |
| Abarema | 37,564,359 | 6.7% |
| Abelia | 37,564,359 | 6.7% |
| Abies | 37,564,359 | 6.7% |
| Total | 563,465,378 | 100% |
| Plot ID | Tree ID | Species Name | Carbon Storage (kg) | % of Total |
|---|---|---|---|---|
| 1 | 1 | Abies | 851.1 | 6.2 |
| 1 | 2 | Abelia | 598.0 | 4.3 |
| 1 | 3 | Dovyalis abyssinica | 1605.9 | 11.6 |
| 1 | 4 | Abarema | 980.1 | 7.1 |
| 1 | 5 | Abies balsamea | 592.5 | 4.3 |
| 1 | 6 | Acacia adunca | 1128.4 | 8.2 |
| 1 | 7 | Lagarostrobos franklinii | 88.5 | 0.6 |
| 1 | 8 | Adenostoma | 2206.9 | 16.0 |
| 1 | 9 | Adenaria floribunda | 459.5 | 3.3 |
| 1 | 10 | Salix fragilis | 28.5 | 0.2 |
| 1 | 11 | Hakea francisiana | 147.9 | 1.1 |
| 1 | 12 | Acacia argyrophylla | 1251.8 | 9.1 |
| 1 | 13 | Adenaria floribunda | 2129.1 | 15.4 |
| 1 | 14 | Carya x schneckii | 565.5 | 4.1 |
| 1 | 15 | Adenostoma fasciculatum | 1193.4 | 8.6 |
| Total | 13,827.0 | 100% |
| Plot ID | Tree ID | Species Name | Carbon Sequestration (kg/year) | % of Total |
|---|---|---|---|---|
| 1 | 1 | Abies | 44.9 | 16.2 |
| 1 | 2 | Abelia | 27.9 | 10.0 |
| 1 | 3 | Dovyalis abyssinica | 45.1 | 16.2 |
| 1 | 4 | Abarema | 29.6 | 10.6 |
| 1 | 5 | Abies balsamea | 8.9 | 3.2 |
| 1 | 6 | Acacia adunca | 0.9 | 0.3 |
| 1 | 7 | Lagarostrobos franklinii | 9.0 | 3.2 |
| 1 | 8 | Adenostoma | 34.4 | 12.4 |
| 1 | 9 | Adenaria floribunda | 25.0 | 9.0 |
| 1 | 10 | Salix fragilis | 5.3 | 1.9 |
| 1 | 11 | Hakea francisiana | 9.2 | 3.3 |
| 1 | 12 | Acacia argyrophylla | 14.6 | 5.3 |
| 1 | 13 | Adenaria floribunda | 0.1 | 0.0 |
| 1 | 14 | Carya x schneckii | 20.2 | 7.3 |
| 1 | 15 | Adenostoma fasciculatum | 2.7 | 1.0 |
| Total | 277.8 | 100% |
| Plot ID | Tree ID | Species Name | Isoprene (g/year) | Monoterpene (g/year) | VOCs (g/year) |
|---|---|---|---|---|---|
| 1 | 1 | Abies | 118.1 | 1875.7 | 1993.9 |
| 1 | 2 | Abelia | 0.0 | 0.0 | 0.0 |
| 1 | 3 | Dovyalis abyssinica | 4.1 | 49.6 | 53.6 |
| 1 | 4 | Abarema | 90.4 | 38.6 | 128.9 |
| 1 | 5 | Abies balsamea | 32.7 | 519.7 | 552.4 |
| 1 | 6 | Acacia adunca | 0.1 | 54.6 | 54.6 |
| 1 | 7 | Lagarostrobos franklinii | 0.0 | 0.0 | 0.0 |
| 1 | 8 | Adenostoma | 0.0 | 40.4 | 40.4 |
| 1 | 9 | Adenaria floribunda | 25.8 | 27.9 | 53.7 |
| 1 | 10 | Salix fragilis | 13.2 | 0.9 | 14.1 |
| 1 | 11 | Hakea francisiana | 0.6 | 14.2 | 14.8 |
| 1 | 12 | Acacia argyrophylla | 0.2 | 204.7 | 205.0 |
| 1 | 13 | Adenaria floribunda | 15.6 | 16.8 | 32.4 |
| 1 | 14 | Carya x schneckii | 0.0 | 33.3 | 33.3 |
| 1 | 15 | Adenostoma fasciculatum | 0.0 | 38.2 | 38.2 |
| Total | 300.7 | 2914.7 | 3215.4 |
| Plot ID | Tree ID | Species Name | Oxygen Production (kg/year) |
|---|---|---|---|
| 1 | 1 | Abies | 119.8 |
| 1 | 2 | Abelia | 74.3 |
| 1 | 3 | Dovyalis abyssinica | 120.3 |
| 1 | 4 | Abarema | 78.9 |
| 1 | 5 | Abies balsamea | 23.7 |
| 1 | 6 | Acacia adunca | 2.3 |
| 1 | 7 | Lagarostrobos franklinii | 24.0 |
| 1 | 8 | Adenostoma | 91.8 |
| 1 | 9 | Adenaria floribunda | 66.7 |
| 1 | 10 | Salix fragilis | 14.3 |
| 1 | 11 | Hakea francisiana | 24.7 |
| 1 | 12 | Acacia argyrophylla | 38.9 |
| 1 | 13 | Adenaria floribunda | 0.3 |
| 1 | 14 | Carya x schneckii | 53.7 |
| 1 | 15 | Adenostoma fasciculatum | 7.2 |
| Total | 740.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, X.; Adesina, J.A. Biodiversity Conservation of National Parks and Nature-Protected Areas in West Africa: The Case of Kainji National Park, Nigeria. Sustainability 2022, 14, 7322. https://doi.org/10.3390/su14127322
Tang X, Adesina JA. Biodiversity Conservation of National Parks and Nature-Protected Areas in West Africa: The Case of Kainji National Park, Nigeria. Sustainability. 2022; 14(12):7322. https://doi.org/10.3390/su14127322
Chicago/Turabian StyleTang, Xiaolan, and John Adekunle Adesina. 2022. "Biodiversity Conservation of National Parks and Nature-Protected Areas in West Africa: The Case of Kainji National Park, Nigeria" Sustainability 14, no. 12: 7322. https://doi.org/10.3390/su14127322
APA StyleTang, X., & Adesina, J. A. (2022). Biodiversity Conservation of National Parks and Nature-Protected Areas in West Africa: The Case of Kainji National Park, Nigeria. Sustainability, 14(12), 7322. https://doi.org/10.3390/su14127322







