Impact of the COVID-19 Lockdown on Patients with Chronic Tinnitus—Preliminary Results
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample and Setting
2.2. Measures
2.2.1. Standardized Questionnaires
- Tinnitus Sample Case History (TSCH)
- Tinnitus Handicap Inventory (THI)
- Beck Anxiety Inventory (BAI)
- Beck Depression Inventory (BDI)
2.2.2. Tonal Audiometry
2.3. Statistical Analyses
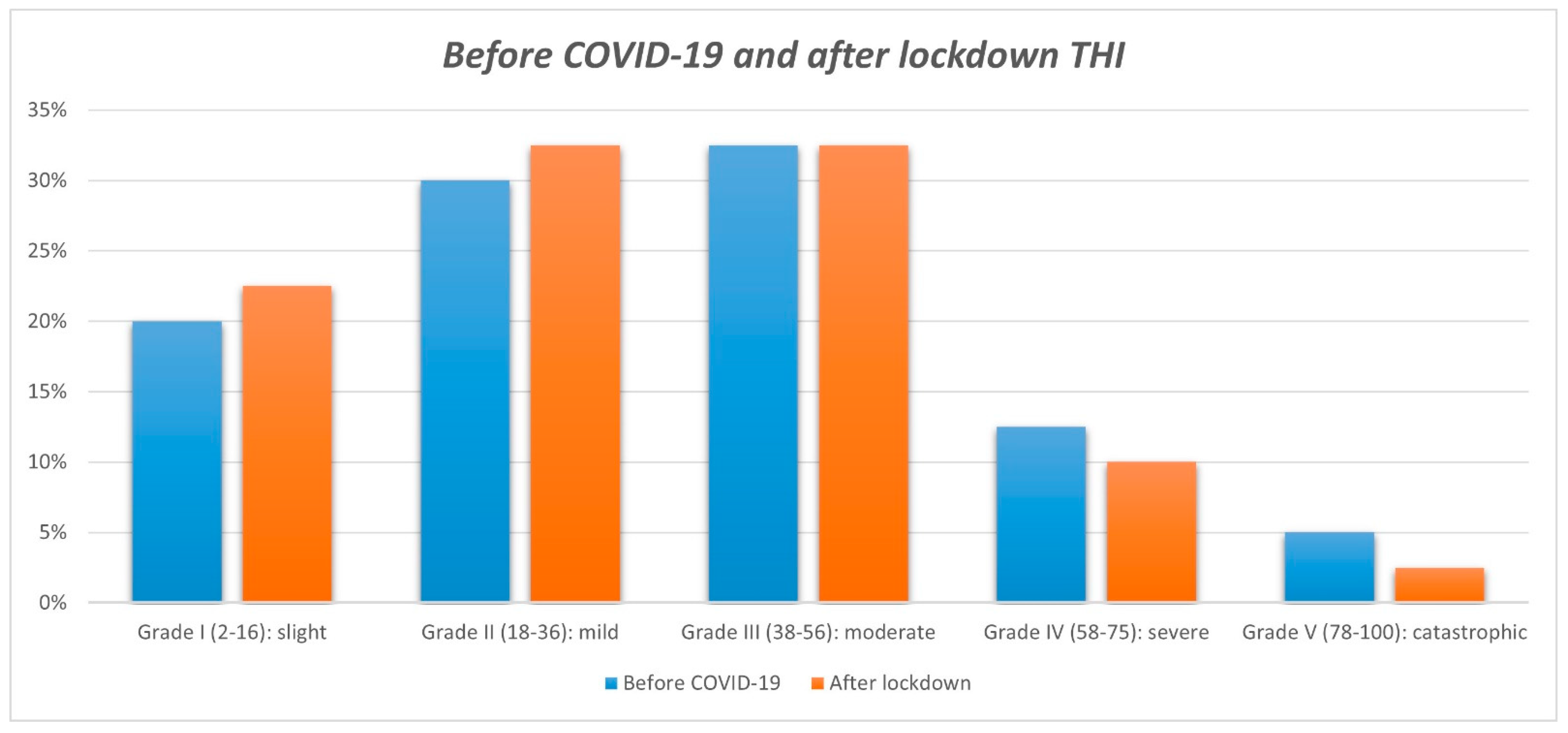
3. Results
3.1. Association of Tinnitus Distress with Depression and Anxiety
3.1.1. Depression
3.1.2. Anxiety
4. Discussion
4.1. Tinnitus Distress and Lifestyle Changes Related to the COVID-19 Pandemic
4.2. No Statistically Significant Change in Depressive Symptoms
4.3. No Statistically Significant Change in Anxiety
4.4. Limits of This Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cima, R.F.F.; Mazurek, B.; Haider, H.; Kikidis, D.; Lapira, A.; Norena, A.; Hoare, D.J. A multidisciplinary European guideline for tinnitus: Diagnostics, assessment, and treatment. HNO 2019, 67, 10–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baguley, D.; McFerran, D.; Hall, D. Tinnitus. Lancet 2013, 382, 1600–1607. [Google Scholar] [CrossRef] [Green Version]
- Biswas, R.; Hall, D.A. Prevalence, Incidence, and Risk Factors for Tinnitus. Curr. Top. Behav. Neurosci. 2021, 51, 3–28. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.A.; Dennis, K.C.; Schechter, M.A. General review of tinnitus: Prevalence, mechanisms, effects, and management. J. Speech Lang. Hear. Res. 2005, 48, 1204–1235. [Google Scholar] [CrossRef]
- Salazar, J.W.; Meisel, K.; Smith, E.R.; Quiggle, A.; Mccoy, D.B.; Amans, M.R. Depression in Patients with Tinnitus: A Systematic Review. Otolaryngol. Head Neck Surg. 2019, 161, 28–35. [Google Scholar] [CrossRef]
- Trevis, K.J.; McLachlan, N.M.; Wilson, S.J. A systematic review and meta-analysis of psychological functioning in chronic tinnitus. Clin. Psychol. Rev. 2018, 60, 62–86. [Google Scholar] [CrossRef]
- Pattyn, T.; Van Den Eede, F.; Vanneste, S.; Cassiers, L.; Veltman, D.J.; Van De Heyning, P.; Sabbe, B.C.G. Tinnitus and anxiety disorders: A review. Hear. Res. 2016, 333, 255–265. [Google Scholar] [CrossRef]
- Durai, M.; Searchfield, G. Anxiety and depression, personality traits relevant to tinnitus: A scoping review. Int. J. Audiol. 2016, 55, 605–615. [Google Scholar] [CrossRef]
- Gomaa, M.A.; Elmagd, M.H.; Elbadry, M.M.; Kader, R.M. Depression, Anxiety and Stress Scale in patients with tinnitus and hearing loss. Eur. Arch. Otorhinolaryngol. 2014, 271, 2177–2184. [Google Scholar] [CrossRef]
- Shore, S.E.; Roberts, L.E.; Langguth, B. Maladaptive plasticity in tinnitus—Triggers, mechanisms and treatment. Nat. Rev. Neurol. 2016, 12, 150–160. [Google Scholar] [CrossRef] [Green Version]
- Munro, K.J.; Uus, K.; Almufarrij, I.; Chaudhuri, N.; Yioe, V. Persistent self-reported changes in hearing and tinnitus in post-hospitalisation COVID-19 cases. Int. J. Audiol. 2020, 59, 889–890. [Google Scholar] [CrossRef] [PubMed]
- Almufarrij, I.; Munro, K.J. One year on: An updated systematic review of SARS-CoV-2, COVID-19 and audio-vestibular symptoms. Int. J. Audiol. 2021, 60, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Viola, P.; Ralli, M.; Pisani, D.; Malanga, D.; Sculco, D.; Messina, L.; Laria, C.; Aragona, T.; Leopardi, G.; Ursini, F.; et al. Tinnitus and equilibrium disorders in COVID-19 patients: Preliminary results. Eur. Arch. Otorhinolaryngol. 2020, 278, 3725–3730. [Google Scholar] [CrossRef] [PubMed]
- Beukes, E.; Ulep, A.; Eubank, T.; Manchaiah, V. The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review. J. Clin. Med. 2021, 10, 2763. [Google Scholar] [CrossRef] [PubMed]
- Mazza, C.; Ricci, E.; Biondi, S.; Colasanti, M.; Ferracuti, S.; Napoli, C.; Roma, P.A. Nationwide Survey of Psychological Distress among Italian People during the COVID-19 Pandemic: Immediate Psychological Responses and Associated Factors. Int. J. Environ. Res. Public Health 2020, 17, 3165. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Favieri, F.; Tambelli, R.; Forte, G. The enemy who sealed the world: Effects quarantine due to the COVID-19 on sleep quality, anxiety, and psychological distress in the Italian population. Sleep Med. 2020, 75, 12–20. [Google Scholar] [CrossRef]
- Ferrucci, R.; Averna, A.; Marino, D.; Reitano, M.R.; Ruggiero, F.; Mameli, F.; Dini, M.; Poletti, B.; Barbieri, S.; Priori, A.; et al. Psychological Impact During the First Outbreak of COVID-19 in Italy. Front. Psychiatry 2020, 11, 559266. [Google Scholar] [CrossRef]
- Huang, Y.; Zhao, N. Generalized anxiety disorder, depressive symptoms and sleep quality during COVID-19 outbreak in China: A web-based cross-sectional survey. Psychiatry Res. 2020, 288, 112954. [Google Scholar] [CrossRef]
- Ozamiz-Etxebarria, N.; Dosil-Santamaria, M.; Picaza-Gorrochategui, M.; Idoiaga-Mondragon, N. Stress, anxiety, and depression levels in the initial stage of the COVID-19 outbreak in a population sample in the northern Spain. Cad. Saude Publica 2020, 36, e00054020. [Google Scholar] [CrossRef]
- Roy, D.; Tripathy, S.; Kar, S.K.; Sharma, N.; Verma, S.K.; Kaushal, V. Study of knowledge, attitude, anxiety & perceived mental healthcare need in Indian population during COVID-19 pandemic. Asian J. Psychiatr. 2020, 51, 102083. [Google Scholar] [CrossRef]
- Wang, C.; Pan, R.; Wan, X.; Tan, Y.; Xu, L.; Ho, C.S.; Ho, R.C. Immediate psychological responses and associated factors during the initial stage of the 2019 coronavirus disease (COVID-19) epidemic among the general population in China. Int. J. Environ. Res. Public Health 2020, 17, 1729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langguth, B.; Goodey, R.; Azevedo, A.; Bjorne, A.; Cacace, A.; Crocetti, A.; Del Bo, L.; De Ridder, D.; Diges, I.; Elbert, T.; et al. Consensus for tinnitus patient assessment and treatment outcome measurement: Tinnitus Research Initiative meeting, Regensburg, July 2006. Prog. Brain Res. 2007, 166, 525–536. [Google Scholar] [PubMed] [Green Version]
- Newman, C.W.; Jacobson, G.P.; Spitzer, J.B. Development of the Tinnitus Handicap Inventory. Arch. Otolaryngol. Head Neck Surg. 1996, 122, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Passi, S.; Ralli, G.; Capparelli, E.; Mammone, A.; Scacciatelli, D.; Cianfrone, G. The THI questionnaire: Psychometric data for reliability and validity of the Italian version. Int. Tinnitus J. 2008, 14, 26–33. [Google Scholar] [PubMed]
- Salviati, M.; Macri, F.; Terlizzi, S.; Melcore, C.; Provenzano, A.; Capparelli, E.; Altissimi, G.; Cianfrone, G. The Tinnitus Handicap Inventory as a screening test for psychiatric comorbidity in patients with tinnitus. Psychosomatics 2013, 54, 248–256. [Google Scholar] [CrossRef]
- Altissimi, G.; Salviati, M.; Turchetta, R.; Orlando, M.P.; Greco, A.; De Vincentiis, M.; Ciofalo, A.; Marinelli, C.; Testugini, V.; Mazzei, F.; et al. When alarm bells ring: Emergency tinnitus. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2955–2973. [Google Scholar]
- Beck, A.T.; Epstein, N.; Brown, G.; Steer, R.A. An inventory for measuring clinical anxiety: Psychometric properties. J. Consult. Clin. Psychol. 1988, 56, 893–897. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory—II; Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Schlee, W.; Hølleland, S.; Bulla, J.; Simoes, J.; Neff, P.; Schoisswohl, S.; Woelflick, S.; Schecklmann, M.; Schiller, A.; Staudinger, S.; et al. The Effect of Environmental Stressors on Tinnitus: A Prospective Longitudinal Study on the Impact of the COVID-19 Pandemic. J. Clin. Med. 2020, 9, 2756. [Google Scholar] [CrossRef]
- Anzivino, R.; Sciancalepore, P.I.; Petrone, P.; D’Elia, A.; Petrone, D.; Quaranta, N. Tinnitus revival during COVID-19 lockdown: How to deal with it? Eur. Arch. Otorhinolaryngol. 2021, 278, 295–296. [Google Scholar] [CrossRef]
- Beukes, E.W.; Baguley, D.M.; Jacquemin, L.; Lourenco, M.P.C.G.; Allen, P.M.; Onozuka, J.; Stockdale, D.; Kaldo, V.; Andersson, G.; Manchaiah, V. Changes in Tinnitus Experiences during the COVID-19 Pandemic. Front. Public Health 2020, 8, 592878. [Google Scholar] [CrossRef]
- Li, S.; Wang, Y.; Xue, J.; Zhao, N. The impact of COVID-19 epidemic declaration on psychological consequences: A study on active Weibo users. Int. J. Environ. Res. Public Health 2020, 17, 2032. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, L.; He, G.; Feng, Y.; Yu, X.; Zhao, X.; Yin, S.; Chen, Z.; Wang, J.; Fan, J.; Dong, C. COVID-19 associated anxiety enhances tinnitus. PLoS ONE 2021, 16, e0246328. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Shen, B.; Zhao, M.; Wang, Z.; Xie, B.; Xu, Y. A nationwide survey of psychological distress among Chinese people in the COVID-19 epidemic: Implication and policy recommendations. Gen. Psychiatr. 2020, 33, e100213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Stefano, V.; Ornello, R.; Gagliardo, A.; Torrente, A.; Illuminato, E.; Caponnetto, V.; Frattale, I.; Golini, R.; Di Felice, C.; Graziano, F.; et al. Social Distancing in Chronic Migraine during the COVID-19 Outbreak: Results from a Multicenter Observational Study. Nutrients 2021, 13, 1361. [Google Scholar] [CrossRef]
- Asensio, C.; Aumond, P.; Can, A.; Gascó, L.; Lercher, P.; Wunderli, J.-M.; Lavandier, C.; de Arcas, G.; Ribeiro, C.; Muñoz, P.; et al. A Taxonomy Proposal for the Assessment of the Changes in Soundscape Resulting from the COVID-19 Lockdown. Int. J. Environ. Res. Public Health 2020, 17, 4205. [Google Scholar] [CrossRef]
- Muzet, A. Environmental noise, sleep and health. Sleep Med. Rev. 2007, 11, 135–142. [Google Scholar] [CrossRef]
- Zacarías, F.F.; Molina, R.H.; Ancela, J.L.C.; López, S.L.; Ojembarrena, A.A. Noise exposure in preterm infants treated with respiratory support using neonatal helmets. Acta Acust. United Acust. 2013, 99, 590–597. [Google Scholar] [CrossRef]
- Erickson, L.C.; Newman, R.S. Influences of background noise on infants and children. Curr. Dir. Psychol. Sci. 2017, 26, 451–457. [Google Scholar] [CrossRef]
- Dratva, J.; Phuleria, H.C.; Foraster, M.; Gaspoz, J.-M.; Keidel, D.; Künzli, N.; Liu, L.-J.S.; Pons, M.; Zemp, E.; Gerbase, M.W.; et al. Transportation noise and blood pressure in a population-based sample of adults. Environ. Health Perspect. 2012, 120, 50–55. [Google Scholar] [CrossRef]
- Babisch, W.; Beule, B.; Schust, M.; Kersten, N.; Ising, H. Traffic noise and risk of myocardial infarction. Epidemiology 2005, 16, 33–40. [Google Scholar] [CrossRef]
- Petri, D.; Licitra, G.; Vigotti, M.A.; Fredianelli, L. Effects of Exposure to Road, Railway, Airport and Recreational Noise on Blood Pressure and Hypertension. Int. J. Environ. Res. Public Health 2021, 18, 9145. [Google Scholar] [CrossRef] [PubMed]
- Vukić, L.; Fredianelli, L.; Plazibat, V. Seafarers’ Perception and Attitudes towards Noise Emission on Board Ships. Int. J. Environ. Res. Public Health 2021, 18, 6671. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Prato, A.; Lesina, L.; Schiavi, A. Effects of low-frequency noise on human cognitive performances in laboratory. Build. Acoust. 2018, 25, 17–33. [Google Scholar] [CrossRef]
- Miedema, H.M.E.; Oudshoorn, C.G.M. Annoyance from transportation noise: Relationships with exposure metrics DNL and DENL and their confidence intervals. Environ. Health Perspect. 2001, 109, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Cueto, J.L.; Petrovici, A.M.; Hernández, R.; Fernández, F. Analysis of the Impact of Bus Signal Priority on Urban Noise. Acta Acust. United Acust. 2017, 103, 561–573. [Google Scholar] [CrossRef]
- Morley, D.W.; de Hoogh, K.; Fecht, D.; Fabbri, F.; Bell, M.; Goodman, P.S.; Elliott, P.; Hodgson, S.; Hansell, A.L.; Gulliver, J. International scale implementation of the CNOSSOS-EU road traffic noise prediction model for epidemiological studies. Environ. Pollut. 2015, 206, 332–341. [Google Scholar] [CrossRef]
- Ruiz-Padillo, A.; Ruiz, D.P.; Torija, A.J.; Ramos-Ridao, Á. Selection of suitable alternatives to reduce the environmental impact of road traffic noise using a fuzzy multi-criteria decision model. Environ. Impact Assess. Rev. 2016, 61, 8–18. [Google Scholar] [CrossRef] [Green Version]
- Bunn, F.; Trombetta Zannin, P.H. Assessment of railway noise in an urban setting. Appl. Acoust. 2016, 104, 16–23. [Google Scholar] [CrossRef]
- Iglesias-Merchan, C.; Diaz-Balteiro, L.; Soliño, M. Transportation planning and quiet natural areas preservation: Aircraft overflights noise assessment in a National Park. Transp. Res. Part D Transp. Environ. 2015, 41, 1–12. [Google Scholar] [CrossRef]
- Nastasi, M.; Fredianelli, L.; Bernardini, M.; Teti, L.; Fidecaro, F.; Licitra, G. Parameters affecting noise emitted by ships moving in port areas. Sustainability 2020, 12, 8742. [Google Scholar] [CrossRef]
- Tong, H.; Aletta, F.; Mitchell, A.; Oberman, T.; Kang, J. Increases in noise complaints during the COVID-19 lockdown in Spring 2020: A case study in Greater London, UK. Sci. Total Environ. 2021, 785, 147213. [Google Scholar] [CrossRef]
- Mishra, A.; Das, S.; Singh, D.; Maurya, A.K. Effect of COVID-19 lockdown on noise pollution levels in an Indian city: A case study of Kanpur. Environ. Sci. Pollut. Res. 2021, 28, 46007–46019. [Google Scholar] [CrossRef] [PubMed]
| Before COVID-19 | After Lockdown | ||||
|---|---|---|---|---|---|
| (n = 40) | (n = 40) | ||||
| n | % | n | % | p-value * | |
| Tinnitus perception | |||||
| Constant | 27 | 67.5% | 32 | 80% | 0.00019 |
| Intermittent | 11 | 27.5% | 8 | 20% | |
| Missing | 2 | 5% | - | - | |
| Tinnitus location | |||||
| Only one ear (right or left) | 14 | 35% | 9 | 22.5% | 0.0041 |
| Both ears and inside the head | 26 | 65% | 31 | 77.5% | |
| Missing | - | - | - | - | |
| Tinnitus loudness varying from day to day Yes | 27 | 67.5% | 29 | 72,5% | 0.0136 |
| No | 13 | 32.5% | 11 | 27.5% | |
| Missing | - | - | - | - | |
| Stress effect | |||||
| Increased tinnitus | 26 | 65% | 31 | 77.5% | 0.0005 |
| No effect | 9 | 22.5% | 9 | 22.5% | |
| Missing | 5 | 12.5% | - | - | |
| Tinnitus pitch | |||||
| Very high and high | 29 | 72.5% | 24 | 60% | 0.0090 |
| Medium and low | 9 | 22.5% | 16 | 40% | |
| Missing | 2 | 5% | - | - | |
| Intolerance to sound | |||||
| Never rarely sometimes | 28 | 70% | 29 | 72.5% | 0.0012 |
| Often always | 9 | 22.5% | 11 | 27.5% | |
| Missing | 3 | 7.5% | - | - | |
| Headache Yes | 19 | 47.5% | 17 | 42.5% | ns |
| No | 19 | 47.5% | 23 | 57.5% | |
| Missing | 2 | 5% | - | - | |
| Vertigo/dizziness Yes | 10 | 25% | 13 | 32.5% | 0.026 |
| No | 27 | 67.5% | 27 | 67.5% | |
| Missing | 3 | 7.5% | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fioretti, A.; Natalini, E.; Triggianese, G.; Eibenstein, R.; Angelone, A.M.; Lauriello, M.; Eibenstein, A. Impact of the COVID-19 Lockdown on Patients with Chronic Tinnitus—Preliminary Results. Audiol. Res. 2022, 12, 327-336. https://doi.org/10.3390/audiolres12030034
Fioretti A, Natalini E, Triggianese G, Eibenstein R, Angelone AM, Lauriello M, Eibenstein A. Impact of the COVID-19 Lockdown on Patients with Chronic Tinnitus—Preliminary Results. Audiology Research. 2022; 12(3):327-336. https://doi.org/10.3390/audiolres12030034
Chicago/Turabian StyleFioretti, Alessandra, Eleonora Natalini, Gianluigi Triggianese, Rebecca Eibenstein, Anna Maria Angelone, Maria Lauriello, and Alberto Eibenstein. 2022. "Impact of the COVID-19 Lockdown on Patients with Chronic Tinnitus—Preliminary Results" Audiology Research 12, no. 3: 327-336. https://doi.org/10.3390/audiolres12030034
APA StyleFioretti, A., Natalini, E., Triggianese, G., Eibenstein, R., Angelone, A. M., Lauriello, M., & Eibenstein, A. (2022). Impact of the COVID-19 Lockdown on Patients with Chronic Tinnitus—Preliminary Results. Audiology Research, 12(3), 327-336. https://doi.org/10.3390/audiolres12030034








