Interpreting the Benefit and Risk Data in Between-Drug Comparisons: Illustration of the Challenges Using the Example of Mefenamic Acid versus Ibuprofen
Abstract
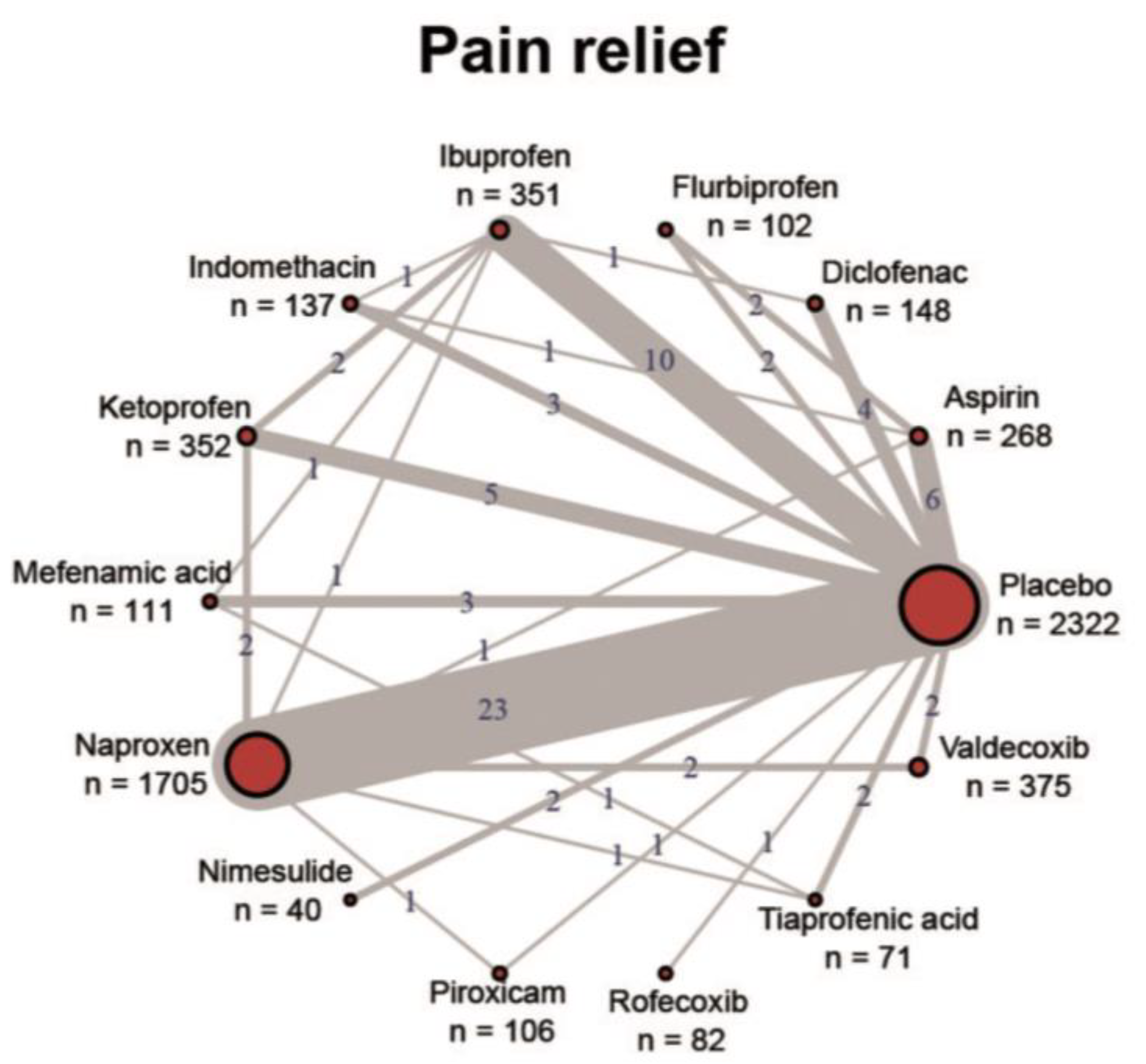
1. Introduction
2. Pain Management in the Course of Time
3. History of NSAIDs and Its Impact in Data Quality Today
4. Mechanism of Action of NSAIDs
5. Ibuprofen and Mefenamic Acid
5.1. Pharmacokinetics
5.1.1. Different Formulations of Ibuprofen
5.1.2. Ibuprofen and Dexibuprofen
5.1.3. Comparative Pharmacology of S(+)-Ibuprofen and R(−)-Ibuprofen
5.2. Therapeutic Indications
5.3. Safety Profiles
5.3.1. Gastrointestinal and Cardiovascular Side Effects
5.3.2. Neurotoxic and Psychiatric Effects
5.4. Selection of the Individually Suitable NSAID
5.5. Consideration of Concomitant Medication
5.6. The Special Case of Acetylsalicylic Acid
5.7. Administration during Pregnancy
5.8. Administration during Lactation
6. Summary of Considerations
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Österreichischer Patientenbericht. Chronischer Schmerz. Periskop 2009, 39, 13. Available online: http://www.patientenbericht.at/files/pb/Patientenbericht_Chronischer_Schmerz_Periskop_39.pdf (accessed on 19 October 2022).
- Stadt Wien. Schmerzbericht Wien 2018. Available online: https://goeg.at/sites/goeg.at/files/inline-files/schmerzbericht-2018.pdf (accessed on 1 March 2022).
- Hachimi-Idrissi, S. Guidelines for the Management of Acute Pain in Emergency Situations; The European Society for Emergency Medicine: Antwerp, Belgium, 2020. [Google Scholar]
- Schlereth, T. Diagnose und nicht Interventionelle Therapie Neuropathischer Schmerzen. 2019. Available online: https://www.awmf.org/uploads/tx_szleitlinien/030-114l_S2k_Diagnose-nicht-interventionelle-Therapie-neuropathischer-Schmerzen_2020-04_1.pdf (accessed on 2 March 2022).
- Bonezzi, C.; Fornasari, D.; Cricelli, C.; Magni, A.; Ventriglia, G. Not All Pain is Created Equal: Basic Definitions and Diagnostic Work-Up. Pain Ther. 2020, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, M.; Zed, P.J.; Ensom, M.H. Opioid Pharmacokinetics-Pharmacodynamics: Clinical Implications in Acute Pain Man-agement in Trauma. Ann. Pharmacother. 2016, 50, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.; Olofsen, E.; Yassen, A.; Aarts, L.; Dahan, A. Pharmacokinetic-pharmacodynamic modeling in acute and chronic pain: An overview of the recent literature. Expert Rev. Clin. Pharmacol. 2011, 4, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Antman, E.M.; Bennett, J.S.; Daugherty, A.; Roberts, H.; Taubert, K.A.; Association, A.H. Use of nonsteroidal antiinflammatory drugs: An update for clinicians: A scientific statement from the American Heart Association. Circulation 2007, 115, 1634–1642. [Google Scholar] [CrossRef]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States. JAMA 2016, 315, 1624–1645. [Google Scholar] [CrossRef]
- Martinez Alonso, E.; Frydrych, V.; Gonzales, V.; Lake, J.; Fiander, M.; LaFleur, J. Non-Selective Oral Nonsteroidal Anti-Inflammatory Drugs: University of Utah College of Pharmacy, Drug Regimen Review Center. Available online: https://medicaid.utah.gov/pharmacy/ptcommittee/files/Criteria%20Review%20Documents/2018/2018.04%20Oral%20Nonselective%20NSAIDs.pdf (accessed on 7 March 2022).
- Bundesamt für Sicherheit im Gesundheitswesen—Medizinmarktaufsicht. Arzneispezialitätenregister—Online Suche Arzneispezialitäten 2022. Available online: https://aspregister.basg.gv.at/aspregister/faces/aspregister.jspx (accessed on 15 March 2022).
- Bundesamt für Sicherheit im Gesundheitswesen und AGES Medizinmarktaufsicht. Arzneispezialitätenregister 2022. Available online: https://aspregister.basg.gv.at/aspregister/faces/aspregister.jspx;jsessionid=aN5KOhBvXzDh8_uNB6-mLunSDrv7rrMDzdBvjCx80XMJRqIHc0oY!211392629 (accessed on 2 March 2022).
- Hartmann, C. Arzneimittelrückstände in der Umwelt 2016. Available online: https://www.umweltbundesamt.at/fileadmin/site/publikationen/REP0573.pdf (accessed on 2 March 2022).
- Sattelberger, R. Arzneimittelrückstände in der Umwelt—Bestandsaufnahme und Problemdarstellung 1999. Available online: https://www.umweltbundesamt.at/fileadmin/site/publikationen/R162.pdf (accessed on 2 March 2022).
- The Martindale Editorial Team. Martindale—The Complete Drug Reference, 28th ed.; Pharmaceutical Press: London, UK, 2014. [Google Scholar]
- Cimolai, N. The potential and promise of mefenamic acid. Expert Rev. Clin. Pharmacol. 2013, 6, 289–305. [Google Scholar] [CrossRef]
- Connelly, D. A brief history of ibuprofen. Pharm. J. 2017, 299, 28–29. [Google Scholar]
- Flower, R.J. The development of COX2 inhibitors. Nat. Rev. Drug Discov. 2003, 2, 179–191. [Google Scholar] [CrossRef]
- Vane, J.R. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat. New Biol. 1971, 231, 232–235. [Google Scholar] [CrossRef]
- Fu, J.Y.; Masferrer, J.L.; Seibert, K.; Raz, A.; Needleman, P. The induction and suppression of prostaglandin H2 synthase (cyclooxygenase) in human monocytes. J. Biol. Chem. 1990, 265, 16737–16740. [Google Scholar] [CrossRef]
- O’Banion, M.K.; Sadowski, H.B.; Winn, V.; Young, D.A. A serum- and glucocorticoid-regulated 4-kilobase mRNA encodes a cyclooxy-genase-related protein. J. Biol. Chem. 1991, 266, 23261–23267. [Google Scholar] [CrossRef]
- Vargesson, N. Thalidomide-induced teratogenesis: History and mechanisms. Birth Defects Res. Part C Embryo Today Rev. 2015, 105, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Grosser, T.; Smyth, E.; FitzGerald, G. Pharmacotherapy of Inflammation, Fever, Pain, and Gout. In Goodman & Gilman’s: The Pharmacological Basis of Therapeutics, 13th ed.; Brunton, L.L., Hilal-Dandan, R., Knollmann, B.C., Eds.; McGraw-Hill Education: New York, NY, USA, 2017. [Google Scholar]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biology. 2011, 31, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Rainsford, K.D. Ibuprofen: Pharmacology, efficacy and safety. Inflammopharmacology 2009, 17, 275–342. [Google Scholar] [CrossRef]
- Donnerer, J. Die Bedeutung der NSAR in der Schmerztherapie. JATROS Orthopädie Traumatol. Rheumatol. 2017, 4, 62–65. [Google Scholar]
- Shin, D.; Lee, S.J.; Ha, Y.-M.; Choi, Y.S.; Kim, J.W.; Park, S.-R.; Park, M.K. Pharmacokinetic and pharmacodynamic evaluation according to absorption differences in three formulations of ibuprofen. Drug Des. Dev. Ther. 2017, 11, 135–141. [Google Scholar] [CrossRef]
- Whitehouse, M.W.; Rainsford, K.D. Why are non-steroidal anti-inflammatory drugs so gastrotoxic, even when given orally as solubilized salt formulations or parenterally? In Side-Effects of Anti-Inflammatory Drugs: Part Two Studies in Major Organ Systems; Rainsford, K.D., Velo, G.P., Eds.; Springer: Dordrecht, The Netherlands, 1987; pp. 55–65. [Google Scholar]
- Seibel, K.; Schaffler, K.; Reeh, P.; Reitmeir, P. Comparison of two different preparations of ibuprofen with regard to the time course of their analgesic effect. A randomised, placebo-controlled, double-blind cross-over study using laser somatosensory evoked potentials obtained from UW-irritated skin in healthy volunteers. Arzneimittelforschung 2004, 54, 444–451. [Google Scholar]
- Müller, P.; Simon, B. Effects of ibuprofen lysinate and acetylsalicylic acid on gastric and duodenal mucosa. Randomized single-blind placebo-controlled endoscopic study in healthy volunteers. Arzneimittelforschung 1994, 44, 840–843. [Google Scholar]
- Kyselovič, J.; Koscova, E.; Lampert, A.; Weiser, T. A Randomized, Double-Blind, Placebo-Controlled Trial of Ibuprofen Lysinate in Comparison to Ibuprofen Acid for Acute Postoperative Dental Pain. Pain Ther. 2020, 9, 249–259. [Google Scholar] [CrossRef]
- Tracy, T.S.; Hall, S.D. Metabolic inversion of (R)-ibuprofen. Epimerization and hydrolysis of ibuprofenyl-coenzyme A. Drug Metab. Dispos. Biol. Fate Chem. 1992, 20, 322–327. [Google Scholar] [PubMed]
- Evans, A.M. Comparative Pharmacology of S(+)-Ibuprofen and (RS)-Ibuprofen. Clin. Rheumatol. 2001, 20, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Rogers, J.D.; Demetriades, J.L.; Holland, S.D.; Seibold, J.R.; DePuy, E. Pharmacokinetics and Bioinversion of Ibuprofen Enantiomers in Humans. Pharm. Res. 1994, 11, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Williams, K.; Day, R.; Graham, G.; Champion, D. Stereoselective disposition of ibuprofen enantiomers in man. Br. J. Clin. Pharmacol. 1985, 19, 669–674. [Google Scholar] [CrossRef]
- Hall, S.D.; Rudy, A.C.; Knight, P.M.; Brater, D.C. Lack of presystemic inversion of (R)- to (S)-ibuprofen in humans. Clin. Pharmacol. Ther. 1993, 53, 393–400. [Google Scholar] [CrossRef]
- Gudis, K.; Sakamoto, C. The Role of Cyclooxygenase in Gastric Mucosal Protection. Am. J. Dig. Dis. 2005, 50, S16–S23. [Google Scholar] [CrossRef]
- Jeffrey, P.; Tucker, G.T.; Bye, A.; Crewe, H.K.; Wright, P.A. The site of inversion of R(-)-ibuprofen: Studies using rat in-situ isolated perfused intes-tine/liver preparations. J. Pharm. Pharmacol. 1991, 43, 715–720. [Google Scholar] [CrossRef]
- Rainsford, K.D. (Ed.) Ibuprofen: A Critical Bibliographic Review, 1st ed.; CRC Press: London, UK, 2019. [Google Scholar]
- Gliszczyńska, A.; Sánchez-López, E. Dexibuprofen Therapeutic Advances: Prodrugs and Nanotechnological Formulations. Pharmaceutics 2021, 13, 414. [Google Scholar] [CrossRef]
- Tucker, G.; Lennard, M. Enantiomer specific pharmacokinetics. Pharmacol. Ther. 1990, 45, 309–329. [Google Scholar] [CrossRef]
- Rudy, A.C.; Knight, P.M.; Brater, D.C.; Hall, S.D. Stereoselective metabolism of ibuprofen in humans: Administration of R-, S- and racemic ibuprofen. J. Pharmacol. Exp. Ther. 1991, 259, 1133–1139. [Google Scholar]
- Food and Drug Administration. FDA Strengthens Warning that Non-Aspirin Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Can Cause Heart Attacks or Strokes 2015. Available online: https://www.fda.gov/media/92768/download (accessed on 22 March 2022).
- European Medicines Agency. Updated Advice on Use of High-Dose Ibuprofen 2015. Available online: https://www.ema.europa.eu/en/medicines/human/referrals/ibuprofen-dexibuprofen-containing-medicines (accessed on 22 March 2022).
- Bruno, A.; Tacconelli, S.; Patrignani, P. Variability in the Response to Non-Steroidal Anti-Inflammatory Drugs: Mechanisms and Perspectives. Basic Clin. Pharmacol. Toxicol. 2013, 114, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Bateman, D.; Kennedy, J.G. Non-steroidal anti-inflammatory drugs and elderly patients. BMJ 1995, 310, 817–818. [Google Scholar] [CrossRef] [PubMed]
- Therapeutics Initiative. Selective COX-2 Inhibitors: Are They Safer NSAIDs? The University of British Columbia. 2001. Available online: https://www.ti.ubc.ca/PDF/39.PDF (accessed on 7 March 2022).
- Shigeta, J.; Takahashi, S.; Okabe, S. Role of cyclooxygenase-2 in the healing of gastric ulcers in rats. J. Pharmacol. Exp. Ther. 1998, 286, 1383–1390. [Google Scholar]
- Gunter, B.R.; Butler, K.A.; Wallace, R.L.; Smith, S.M.; Harirforoosh, S.; Harirforoosh, S. Non-steroidal anti-inflammatory drug-induced cardiovascular adverse events: A meta-analysis. J. Clin. Pharm. Ther. 2016, 42, 27–38. [Google Scholar] [CrossRef]
- Davies, N.M.; Jamali, F. COX-2 selective inhibitors cardiac toxicity: Getting to the heart of the matter. J. Pharm. Pharm. Sci. 2004, 7, 332–336. [Google Scholar]
- Harirforoosh, S.; Jamali, F. Effect of nonsteroidal anti-inflammatory drugs with varying extent of COX-2–COX-1 selectivity on urinary sodium and potassium excretion in the rat. Can. J. Physiol. Pharmacol. 2005, 83, 85–90. [Google Scholar] [CrossRef]
- Harirforoosh, S.; Aghazadeh-Habashi, A.; Jamali, F. Extent of renal effect of cyclo-oxygenase-2-selective inhibitors is pharma-cokinetic dependent. Clin. Exp. Pharmacol. Physiol. 2006, 33, 917–924. [Google Scholar] [CrossRef]
- Mason, R.P.; Walter, M.F.; McNulty, H.P.; Lockwood, S.F.; Byun, J.; Day, C.A.; Jacob, R.F. Rofecoxib Increases Susceptibility of Human LDL and Membrane Lipids to Oxidative Damage: A Mechanism of Cardiotoxicity. J. Cardiovasc. Pharmacol. 2006, 47, S7–S14. [Google Scholar] [CrossRef]
- Liu, J.-Y.; Li, N.; Yang, J.; Li, N.; Qiu, H.; Ai, D.; Chiamvimonvat, N.; Zhu, Y.; Hammock, B.D. Metabolic profiling of murine plasma reveals an unexpected biomarker in rofecoxib-mediated cardiovascular events. Proc. Natl. Acad. Sci. USA 2010, 107, 17017–17022. [Google Scholar] [CrossRef]
- Schmassmann, A.; Zoidl, G.; Peskar, B.M.; Waser, B.; Schmassmann-Suhijar, D.; Gebbers, J.-O.; Reubi, J.C. Role of the different isoforms of cyclooxygenase and nitric oxide synthase during gastric ulcer healing in cyclooxygenase-1 and -2 knockout mice. Am. J. Physiol. Liver Physiol. 2006, 290, G747–G756. [Google Scholar] [CrossRef]
- Wongrakpanich, S.; Wongrakpanich, A.; Melhado, K.; Rangaswami, J. A Comprehensive Review of Non-Steroidal Anti-Inflammatory Drug Use in The Elderly. Aging Dis. 2018, 9, 143–150. [Google Scholar] [CrossRef]
- Knights, K.M.; Mangoni, A.A.; Miners, J.O. Defining the COX inhibitor selectivity of NSAIDs: Implications for understanding toxicity. Expert Rev. Clin. Pharmacol. 2010, 3, 769–776. [Google Scholar] [CrossRef]
- Cryer, B.; Feldman, M. Cyclooxygenase-1 and Cyclooxygenase-2 Selectivity of Widely Used Nonsteroidal Anti-Inflammatory Drugs. Am. J. Med. 1998, 104, 413–421. [Google Scholar] [CrossRef]
- Rampal, P.; Moore, N.; VAN Ganse, E.; Le Parc, J.-M.; Wall, R.; Schneid, H.; Verrière, F. Gastrointestinal Tolerability of Ibuprofen Compared with Paracetamol and Aspirin at Over-the-Counter Doses. J. Int. Med. Res. 2002, 30, 301–308. [Google Scholar] [CrossRef]
- Sikes, D.H.; Agrawal, N.M.; Zhao, W.W.; Kent, J.D.; Recker, D.P.; Verburg, K.M. Incidence of gastroduodenal ulcers associated with valdecoxib compared with that of ibuprofen and diclofenac in patients with osteoarthritis. Eur. J. Gastroenterol. Hepatol. 2002, 14, 1101–1111. [Google Scholar] [CrossRef]
- García Rodríguez, L.A.; Jick, H. Risk of upper gastrointestinal bleeding and perforation associated with individual non-steroidal anti-inflammatory drugs. Lancet 1994, 343, 769–772. [Google Scholar] [CrossRef]
- Langman, M.J.; Weil, J.; Wainwright, P.; Lawson, D.H.; Rawlins, M.D.; Logan, R.F.; Murphy, M.; Vessey, M.P.; Colin-Jones, D.G. Risks of bleeding peptic ulcer associated with individual non-steroidal an-ti-inflammatory drugs. Lancet 1994, 343, 1075–1078. [Google Scholar] [CrossRef]
- Hawkins, C.; Hanks, G.W. The Gastroduodenal Toxicity of Nonsteroidal Anti-Inflammatory Drugs. A Review of the Literature. J. Pain Symptom Manag. 2000, 20, 140–151. [Google Scholar] [CrossRef]
- Henry, D.; Lim, L.L.-Y.; Rodriguez, L.A.G.; Perez-Gutthann, S.; Carson, J.L.; Griffin, M.; Savage, R.; Logan, R.; Moride, Y.; Hawkey, C.; et al. Variability in risk of gastrointestinal complications with individual non-steroidal anti-inflammatory drugs: Results of a collaborative meta-analysis. BMJ 1996, 312, 1563–1566. [Google Scholar] [CrossRef]
- Bundesinstitut für Arzneimittel und Medizinprodukte—Sachverständigen-Ausschuss für Verschreibungspflicht. Dexibu-profen 2021. Available online: https://www.bfarm.de/SharedDocs/Downloads/DE/Arzneimittel/Pharmakovigilanz/Gremien/Verschreibungspflicht/84Sitzung/anlage5.pdf?__blob=publicationFile (accessed on 5 April 2022).
- Derry, S.; Best, J.; Moore, R.A. Single dose oral dexibuprofen [S(+)-ibuprofen] for acute postoperative pain in adults. Cochrane Database Syst. Rev. 2013, 2013, CD007550. [Google Scholar] [CrossRef]
- European Medicines Agency—Pharmacovigilance Risk Assessment Committee. Assessment Report: Ibuprofen and Dexibu-profen Containing Medicinal Products. Available online: https://www.ema.europa.eu/en/documents/referral/ibuprofen-dexibuprofen-article-31-referral-assessment-report_en.pdf (accessed on 5 April 2022).
- Panel BtAGSBCUE. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults. J. Am. Geriatr. Soc. 2019, 67, 674–694. [Google Scholar] [CrossRef] [PubMed]
- Auriel, E.; Regev, K.; Korczyn, A.D. Nonsteroidal anti-inflammatory drugs exposure and the central nervous system. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2014; Volume 119, pp. 577–584. [Google Scholar]
- Kamour, A.; Crichton, S.; Cooper, G.; Lupton, D.J.; Eddleston, M.; Vale, J.A.; Thompson, J.P.; Thomas, S.H.L. Central nervous system toxicity of mefenamic acid overdose compared with other NSAIDs: An analysis of cases reported to the United Kingdom National Poisons Information Service. Br. J. Clin. Pharmacol. 2016, 83, 855–862. [Google Scholar] [CrossRef]
- Hall, A.H.; Smolinske, S.C.; Conrad, F.L.; Wruk, K.M.; Kulig, K.W.; Dwelle, T.L.; Rumack, B.H. Ibuprofen overdose: 126 cases. Ann. Emerg. Med. 1986, 15, 1308–1313. [Google Scholar] [CrossRef]
- Rodríguez, S.C.; Olguín, A.M.; Miralles, C.P.; Viladrich, P.F. Characteristics of meningitis caused by Ibuprofen: Report of 2 cases with re-current episodes and review of the literature. Medicine 2006, 85, 214–220. [Google Scholar] [CrossRef]
- Sánchez-Borges, M. Clinical Management of Nonsteroidal Anti-inflammatory Drug Hypersensitivity. World Allergy Organ. J. 2008, 1, 29–33. [Google Scholar] [CrossRef]
- Morís, G.; Garcia-Monco, J.C. The Challenge of Drug-Induced Aseptic Meningitis Revisited. JAMA Intern. Med. 2014, 174, 1511–1512. [Google Scholar] [CrossRef] [PubMed]
- Seaton, R.A.; France, A.J. Recurrent aseptic meningitis following non- steroidal anti-inflammatory drugs—A reminder. Postgrad. Med. J. 1999, 75, 771–772. [Google Scholar] [CrossRef][Green Version]
- Nguyen, H.T.; Juurlink, D.N. Recurrent Ibuprofen-Induced Aseptic Meningitis. Ann. Pharmacother. 2004, 38, 408–410. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. Concomitant Use of Ibuprofen and Aspirin: Potential for Attenuation of the Anti-Platelet Effect of Aspirin 2006. Available online: https://www.fda.gov/media/76636/download (accessed on 18 March 2022).
- Oswald, S. Gecheckt: ASS und NSAR—Wann die thrombozytenaggregationshemmende Wirkung aufgehoben wird. DAZOnline 2020, 41. Available online: https://www.deutsche-apotheker-zeitung.de/daz-az/2020/daz-41-2020/gecheckt-ass-und-nsar (accessed on 19 October 2022).
- Roumie, C.L.; Choma, N.N.; Ms, L.K.; Mitchel, J.M.E.F.; Arbogast, P.G.; Griffin, M.R. Non-aspirin NSAIDs, cyclooxygenase-2 inhibitors and risk for cardiovascular events-stroke, acute myocardial infarction, and death from coronary heart disease. Pharmacoepidemiol. Drug Saf. 2009, 18, 1053–1063. [Google Scholar] [CrossRef]
- Khansari, P.S.; Halliwell, R.F. Mechanisms Underlying Neuroprotection by the NSAID Mefenamic Acid in an Experimental Model of Stroke. Front. Neurosci. 2019, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- Bavry, A.; Park, K. Risk of stroke associated with nonsteroidal anti-inflammatory drugs. Vasc. Heal. Risk Manag. 2014, 10, 25–32. [Google Scholar] [CrossRef]
- Mandrekar-Colucci, S.; Landreth, G.E. Microglia and inflammation in Alzheimer’s disease. CNS Neurol. Disord. Drug Targets 2010, 9, 156–167. [Google Scholar] [CrossRef] [PubMed]
- McGeer, P.L.; McGeer, E.G. Anti-inflammatory drugs in the fight against Alzheimer’s disease. Ann. N. Y. Acad. Sci. 1996, 777, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Doğan, N.; Çaylak, S.T.; Yılmaz, S. Central nervous system toxicity due to mefenamic acid. Am. J. Emerg. Med. 2019, 37, 561.e1–561.e2. [Google Scholar] [CrossRef]
- Onder, G.; Pellicciotti, F.; Gambassi, G.; Bernabei, R. NSAID-related psychiatric adverse events: Who is at risk? Drugs 2004, 64, 2619–2627. [Google Scholar] [CrossRef]
- Steinhauer, H.B.; Hertting, G. Lowering of the convulsive threshold by non-steroidal anti-inflammatory drugs. Eur. J. Pharmacol. 1981, 69, 199–203. [Google Scholar] [CrossRef]
- Novakova, I.; Subileau, E.-A.; Toegel, S.; Gruber, D.; Lachmann, B.; Urban, E.; Chesne, C.; Noe, C.R.; Neuhaus, W. Transport Rankings of Non-Steroidal Antiinflammatory Drugs across Blood-Brain Barrier In Vitro Models. PLoS ONE 2014, 9, e86806. [Google Scholar] [CrossRef]
- Noe, C.R.; Noe-Letschnig, M.; Handschuh, P.; Noe, C.A.; Lanzenberger, R. Dysfunction of the Blood-Brain Barrier—A Key Step in Neurodegeneration and Dementia. Front. Aging Neurosci. 2020, 12, 185. [Google Scholar] [PubMed]
- Feng, X.; Wang, X. Comparison of the efficacy and safety of non-steroidal anti-inflammatory drugs for patients with primary dysmenorrhea: A network meta-analysis. Mol. Pain 2018, 14, 1744806918770320. [Google Scholar] [CrossRef] [PubMed]
- Konijnenbelt-Peters, J.; Van Der Heijden, C.; Ekhart, C.; Bos, J.; Bruhn, J.; Kramers, C. Metamizole (Dipyrone) as an Alternative Agent in Postoperative Analgesia in Patients with Contraindications for Nonsteroidal Anti-Inflammatory Drugs. Pain Pract. 2016, 17, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Wertli, M.M.; Flury, J.S.; Streit, S.; Limacher, A.; Schuler, V.; Ferrante, A.-N.; Rimensberger, C.; Haschke, M. Efficacy of metamizole versus ibuprofen and a short educational intervention versus standard care in acute and subacute low back pain: A study protocol of a randomised, multicentre, factorial trial (EMISI trial). BMJ Open 2021, 11, e048531. [Google Scholar] [CrossRef] [PubMed]
- Lupu, M.N.; Miulescu, M.; Dobre, M.; Vesa, D. Metamizole for Postoperative Pain in Pediatric Patients Undergoing Subarachnoid Anesthesia. Am. J. Ther. 2020, 27, e338–e345. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.K.; Bijur, P.E.; Esses, D.; Barnaby, D.P.; Baer, J. Effect of a Single Dose of Oral Opioid and Nonopioid Analgesics on Acute Extremity Pain in the Emergency Department: A Randomized Clinical Trial. JAMA 2017, 318, 1661–1667. [Google Scholar] [CrossRef]
- Krebs, E.E.; Gravely, A.; Nugent, S.; Jensen, A.C.; DeRonne, B.; Goldsmith, E.S.; Kroenke, K.; Bair, M.J.; Noorbaloochi, S. Effect of Opioid vs Nonopioid Medications on Pain-Related Function in Patients with Chronic Back Pain or Hip or Knee Osteoarthritis Pain: The SPACE Randomized Clinical Trial. JAMA 2018, 319, 872–882. [Google Scholar] [CrossRef]
- Teater, D. NSAIDs are Stronger Pain Medications than Opioids: A Summary of Evidence: National Safety Council. Available online: https://www.mndental.org/files/NSAIDs-are-stronger-pain-medications-than-opioids-A-Summary-of-Evidence.pdf (accessed on 20 April 2022).
- González, E.L.M.; Patrignani, P.; Tacconelli, S.; Rodríguez, L.A.G. Variability among nonsteroidal antiinflammatory drugs in risk of upper gastrointestinal bleeding. Arthritis Rheum. 2010, 62, 1592–1601. [Google Scholar] [CrossRef]
- García Rodríguez, L.A.; Barreales Tolosa, L. Risk of Upper Gastrointestinal Complications Among Users of Traditional NSAIDs and COXIBs in the General Population. Gastroenterology 2007, 132, 498–506. [Google Scholar] [CrossRef]
- MacDonald, T.M.; Morant, S.V.; Robinson, G.C.; Shield, M.J.; McGilchrist, M.M.; Murray, F.E.; McDevitt, D.G. Association of upper gastrointestinal toxicity of non-steroidal an-ti-inflammatory drugs with continued exposure: Cohort study. BMJ 1997, 315, 1333–1337. [Google Scholar] [CrossRef][Green Version]
- Coxib and traditional NSAID Trialists’ (CNT) Collaboration; Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.A.; Bombardier, C.; Cannon, C.; Farkouh, M.E. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 2013, 382, 769–779. [Google Scholar]
- Motov, S.; Masoudi, A.; Drapkin, J.; Sotomayor, C.; Kim, S.; Butt, M.; Likourezos, A.; Fassassi, C.; Hossain, R.; Brady, J.; et al. Comparison of Oral Ibuprofen at Three Single-Dose Regimens for Treating Acute Pain in the Emergency Department: A Randomized Controlled Trial. Ann. Emerg. Med. 2019, 74, 530–537. [Google Scholar] [CrossRef]
- Brook, P.; Pickering, T.; Connell, J. Oxford Handbook of Pain Management; OUP Oxford: Oxford, UK, 2011. [Google Scholar]
- Nunes, A.P.; Costa, I.M.; Costa, F.A. Determinants of self-medication with NSAIDs in a Portuguese community pharmacy. Pharm. Pract. (Granada) 2016, 14, 648. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A. Insight into Interactions between Aspirin and Non-Steriodal Anti-Inflammatory Drugs. Ph.D. Thesis, Heinrich Heine University Düsseldorf, Düsseldorf, Germany, 2013. [Google Scholar]
- Richter, S.; Schrör, K.; Rassaf, T.; Merx, M.W.; Kelm, M.; Hohlfeld, T.; Zeus, T.; Polzin, A. Prevention of dipyrone (metamizole) induced inhibition of aspirin antiplatelet effects. Thromb. Haemost. 2015, 114, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, C.; Spielmann, H.; Vetter, K.; Weber-Schöndorfer, C. Arzneimittel in Schwangerschaft und Stillzeit, 8th ed.; Urban & Fischer Verlag, Elsevier GmbH: Munich, Germany, 2011. [Google Scholar]
- Bloor, M.; Paech, M. Nonsteroidal Anti-Inflammatory Drugs During Pregnancy and the Initiation of Lactation. Anesthesia Analg. 2013, 116, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Menahem, S. Prostaglandin inhibitors during pregnancy and the effect on the fetus. Aust. N. Z. J. Obstet. Gynaecol. 1991, 31, 190–191. [Google Scholar] [PubMed]
- Food and Drug Administration. Mefenamic Acid Capsules, US Prescribing Information 2021. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/015034s045lbl.pdf (accessed on 29 March 2022).
- Dathe, K.; Fietz, A.-K.; Pritchard, L.W.; Padberg, S.; Hultzsch, S.; Meixner, K.; Meister, R.; Schaefer, C. No evidence of adverse pregnancy outcome after exposure to ibuprofen in the first trimester—Evaluation of the national Embryotox cohort. Reprod. Toxicol. 2018, 79, 32–38. [Google Scholar] [CrossRef]
- Zafeiri, A.; Mitchell, R.T.; Hay, D.C.; Fowler, P.A. Over-the-counter analgesics during pregnancy: A comprehensive review of global prevalence and offspring safety. Hum. Reprod. Updat. 2020, 27, 67–95. [Google Scholar] [CrossRef]
- Burdan, F. Developmental toxicity evaluation of ibuprofen and tolmetin administered in triple daily doses to Wistar CRL: (WI) WUBR rats. Birth Defects Res. Part B Dev. Reprod. Toxicol. 2004, 71, 321–330. [Google Scholar] [CrossRef]
- Ofori, B.; Oraichi, D.; Blais, L.; Rey, E.; Bérard, A. Risk of congenital anomalies in pregnant users of non-steroidal anti-inflammatory drugs: A nested case-control study. Birth Defects Res. Part B Dev. Reprod. Toxicol. 2006, 77, 268–279. [Google Scholar] [CrossRef]
- Li, D.K.; Liu, L.; Odouli, R. Exposure to non-steroidal anti-inflammatory drugs during pregnancy and risk of miscarriage: Pop-ulation based cohort study. BMJ 2003, 327, 368. [Google Scholar] [CrossRef]
- Jensen, M.S.; Rebordosa, C.; Thulstrup, A.M.; Toft, G.; Sørensen, H.T.; Bonde, J.P.; Henriksen, T.B.; Olsen, J. Maternal Use of Acetaminophen, Ibuprofen, and Acetylsalicylic Acid During Pregnancy and Risk of Cryptorchidism. Epidemiology 2010, 21, 779–785. [Google Scholar] [CrossRef]
- Nezvalová-Henriksen, K.; Spigset, O.; Nordeng, H. Effects of ibuprofen, diclofenac, naproxen, and piroxicam on the course of pregnancy and pregnancy outcome: A prospective cohort study. BJOG: Int. J. Obstet. Gynaecol. 2013, 120, 948–959. [Google Scholar] [CrossRef]
- Nakhai-Pour, H.R.; Broy, P.; Sheehy, O.; Bérard, A. Use of nonaspirin nonsteroidal anti-inflammatory drugs during pregnancy and the risk of spontaneous abortion. Can. Med. Assoc. J. 2011, 183, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Townsend, R.; Benedetti, T.; Erickson, S.; Cengiz, C.; Gillespie, W.; Gschwend, J.; Albert, K. Excretion of ibuprofen into breast milk. Am. J. Obstet. Gynecol. 1984, 149, 184–186. [Google Scholar] [CrossRef]
- Weibert, R.T.; Townsend, R.J.; Kaiser, D.G.; Naylor, A.J. Lack of ibuprofen secretion into human milk. Clin. Pharm. 1982, 1, 457–458. [Google Scholar] [PubMed]
- Walter, K.; Dilger, C. Ibuprofen in human milk. Br. J. Clin. Pharmacol. 1997, 44, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Blajchman, A.; Stephenson, M.; Eliopoulos, C.; Koren, G. Prospective follow-up of adverse reactions in breast-fed infants exposed to maternal medication. Am. J. Obstet. Gynecol. 1993, 168, 1393–1399. [Google Scholar] [CrossRef]
- Spigset, O.; Hägg, S. Analgesics and breast-feeding: Safety considerations. Paediatr. Drugs 2000, 2, 223–238. [Google Scholar] [CrossRef]
- Sachs, H.C.; Frattarelli, D.A.C.; Galinkin, J.L.; Green, T.P.; Johnson, T.; Neville, K.; Paul, I.M.; Anker, J.V.D.; COMMITTEE ON DRUGS. The Transfer of Drugs and Therapeutics into Human Breast Milk: An Update on Selected Topics. Pediatrics 2013, 132, e796–e809. [Google Scholar] [CrossRef]
- Buchanan, R.A.; Eaton, C.J.; Koeff, S.T.; Kinkel, A.W. The breast milk excretion of mefenamic acid. Curr. Ther. Res. 1968, 10, 592–597. [Google Scholar]
- Drugs and Lactation Database (LactMed): Mefenamic Acid. National Library of Medicine (US). 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK501012/ (accessed on 29 March 2022).
| NSAIDs | Absorption | Distribution | Metabolism | Excretion | Half-Life |
|---|---|---|---|---|---|
| Mefenamic acid | BA: easily absorbed Tmax: 2–4 h | Vd: 1.06 L/kg PB: >90% | Hepatic; mainly via CYP2C9 | Renal: 52% Fecal: 20% | 2–4 h |
| Ibuprofen | BA: 80% Tmax: 1–2 h | Vd: 0.12-2 L/kg PB: 90–99% | Hepatic; rapidly metabolized via CYP2C9 | Renal: 45–79% | 1.8–2.44 h |
| Medication | Interaction |
|---|---|
| Antiplatelets (aspirin, clopidogrel) | Increases risk of gastrointestinal bleeding |
| Angiotensin-converting-enzyme inhibitor (ACEI) and Angiotensin Receptor Blockers (ARB) | Increases in blood pressure by attenuating antihypertensive effects |
| Beta blockers | Increases in blood pressure by attenuating antihypertensive effects |
| Calcium antagonists | Increases in blood pressure by attenuating antihypertensive effects |
| Corticosteroids | Increases risk of gastrointestinal bleeding |
| Digitalis glycosides | Increase serum digoxin level |
| Diuretics | Increases in blood pressure by attenuating antihypertensive effects |
| Methotrexate | NSAIDs reduce renal excretion of methotrexate, causing methotrexate toxicity. |
| Selective serotonin reuptake inhibitors (SSRIs) | Increases risk of gastrointestinal bleeding |
| Step | Consideration | Comment |
|---|---|---|
| 1 | Indication to be treated | The approved indication for NSAIDs may differ by country, formulation and brand. There is a larger number of oral formulations and generic brands for ibuprofen than for mefenamic acid |
| 2 | Special populations | Pregnancy: only if absolutely necessary during the first and second trimester, not to be used during the third trimester; ibuprofen is well studied in pregnancy; mefenamic acid has mostly evidence from animal models Lactation: Ibuprofen is safe to be used during lactation; mefenamic acid is not well studied During pregnancy and lactation mefenamic acid is not recommended Elderly patients: the chronic, regular use of NSAIDs in elderly patients should be avoided |
| 3 | Patient age | NSAIDs are generally used in children and adults, differences in indications may apply for special populations such as newborns or preterm babies, and the elderly |
| 4 | Comorbidities | Older patients tend to have a higher risk of relevant comorbidities such as cardiovascular disease, renal disease, or gastrointestinal bleeding, which should be assessed |
| 5 | Concomitant medications | The risk of gastrointestinal bleeding is increased with antiplatelets, corticosteroids, and SSRIs. Blood pressure may be increased with ACEI/ARB, betablockers, calcium antagonists, and diuretics |
| 6 | Side effects | Gastrointestinal and cardiovascular: the risk should not be assessed based on COX-selectivity alone. A warning of gastrointestinal and cardiovascular side effects is present in the package leaflets of both ibuprofen and mefenamic acid; ibuprofen has been shown in trials to have a low gastrointestinal and cardiovascular toxicity at doses up to 1200 mg |
| 7 | Other considerations | Ibuprofen has a racemic and a pure enantiomer formulation available with different properties that may be considered in an individual patient |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farkouh, A.; Hemetsberger, M.; Noe, C.R.; Baumgärtel, C. Interpreting the Benefit and Risk Data in Between-Drug Comparisons: Illustration of the Challenges Using the Example of Mefenamic Acid versus Ibuprofen. Pharmaceutics 2022, 14, 2240. https://doi.org/10.3390/pharmaceutics14102240
Farkouh A, Hemetsberger M, Noe CR, Baumgärtel C. Interpreting the Benefit and Risk Data in Between-Drug Comparisons: Illustration of the Challenges Using the Example of Mefenamic Acid versus Ibuprofen. Pharmaceutics. 2022; 14(10):2240. https://doi.org/10.3390/pharmaceutics14102240
Chicago/Turabian StyleFarkouh, André, Margit Hemetsberger, Christian R. Noe, and Christoph Baumgärtel. 2022. "Interpreting the Benefit and Risk Data in Between-Drug Comparisons: Illustration of the Challenges Using the Example of Mefenamic Acid versus Ibuprofen" Pharmaceutics 14, no. 10: 2240. https://doi.org/10.3390/pharmaceutics14102240
APA StyleFarkouh, A., Hemetsberger, M., Noe, C. R., & Baumgärtel, C. (2022). Interpreting the Benefit and Risk Data in Between-Drug Comparisons: Illustration of the Challenges Using the Example of Mefenamic Acid versus Ibuprofen. Pharmaceutics, 14(10), 2240. https://doi.org/10.3390/pharmaceutics14102240







