Design of a Gene Panel to Expose the Versatile Role of Hepatic Stellate Cells in Human Liver Fibrosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Precision-Cut Liver Slices (PCLS)
2.3. Quantitative Real-Time Polymerase Chain Reaction (PCR)
2.4. Statistical Analyses
3. Results
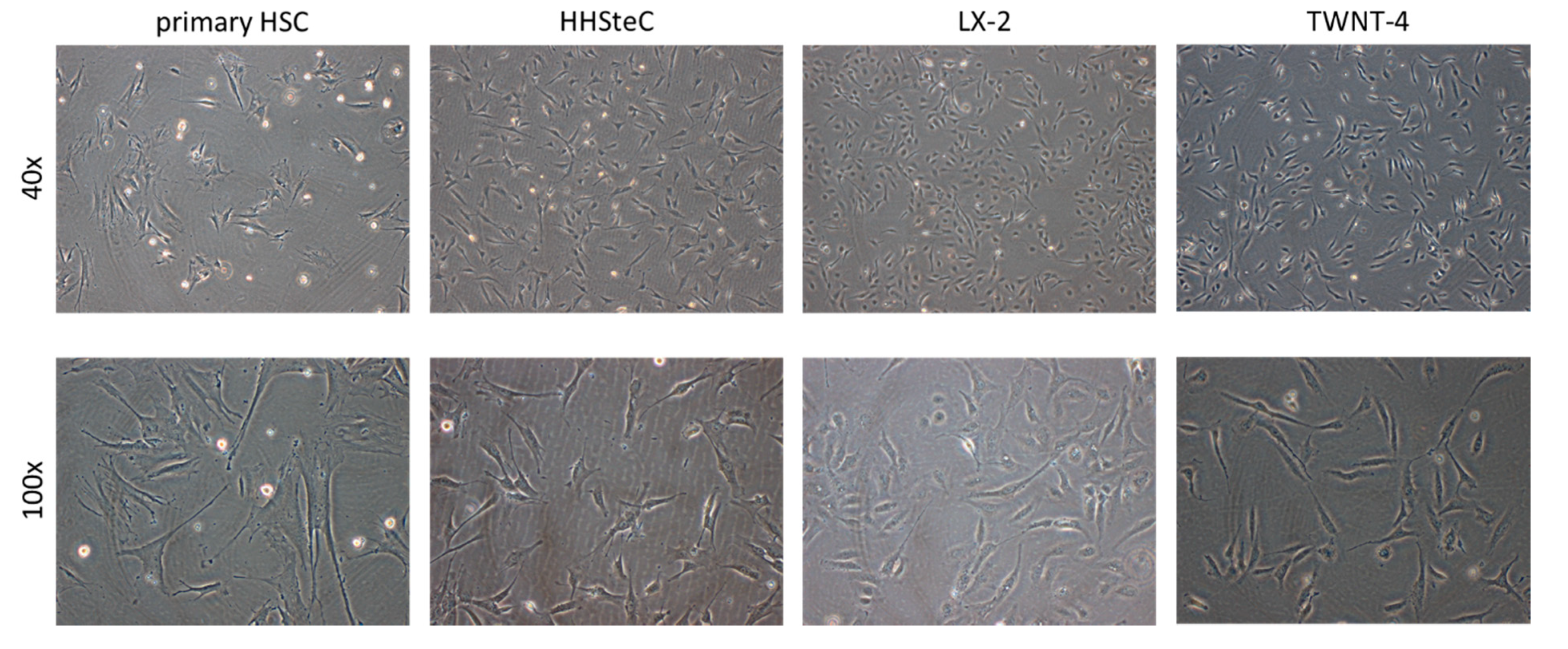
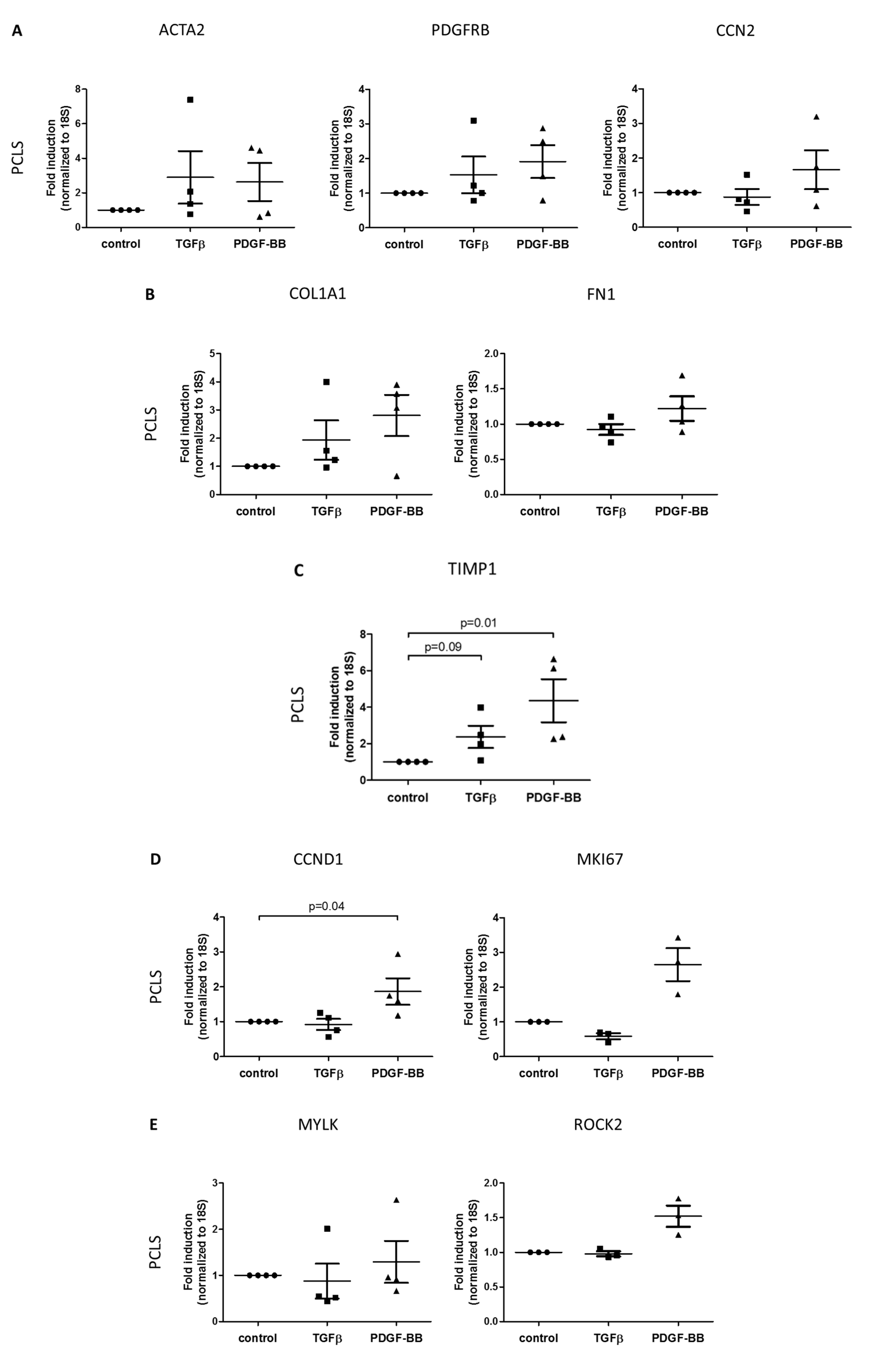
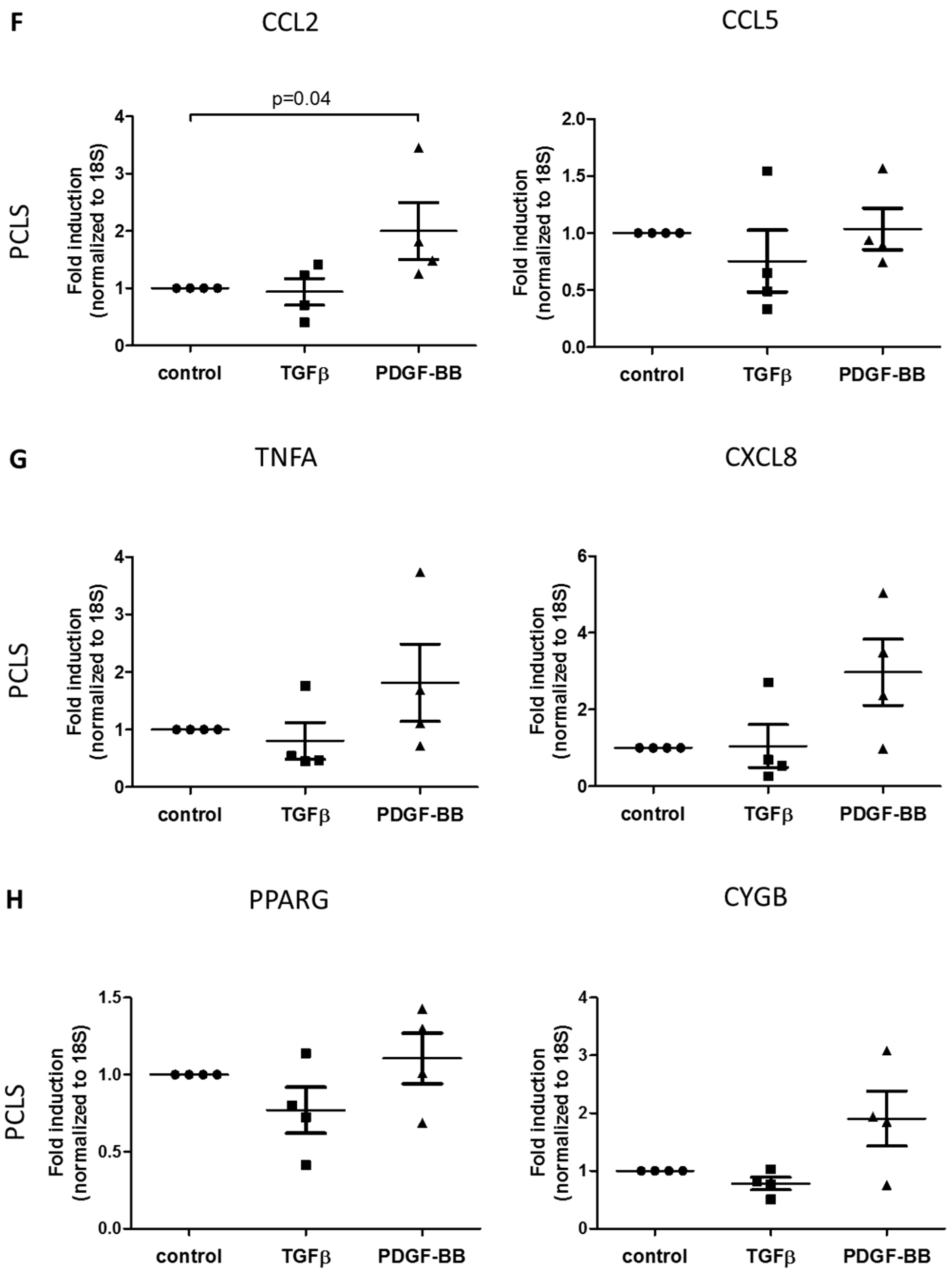
3.1. Morphology of the in Vitro Models
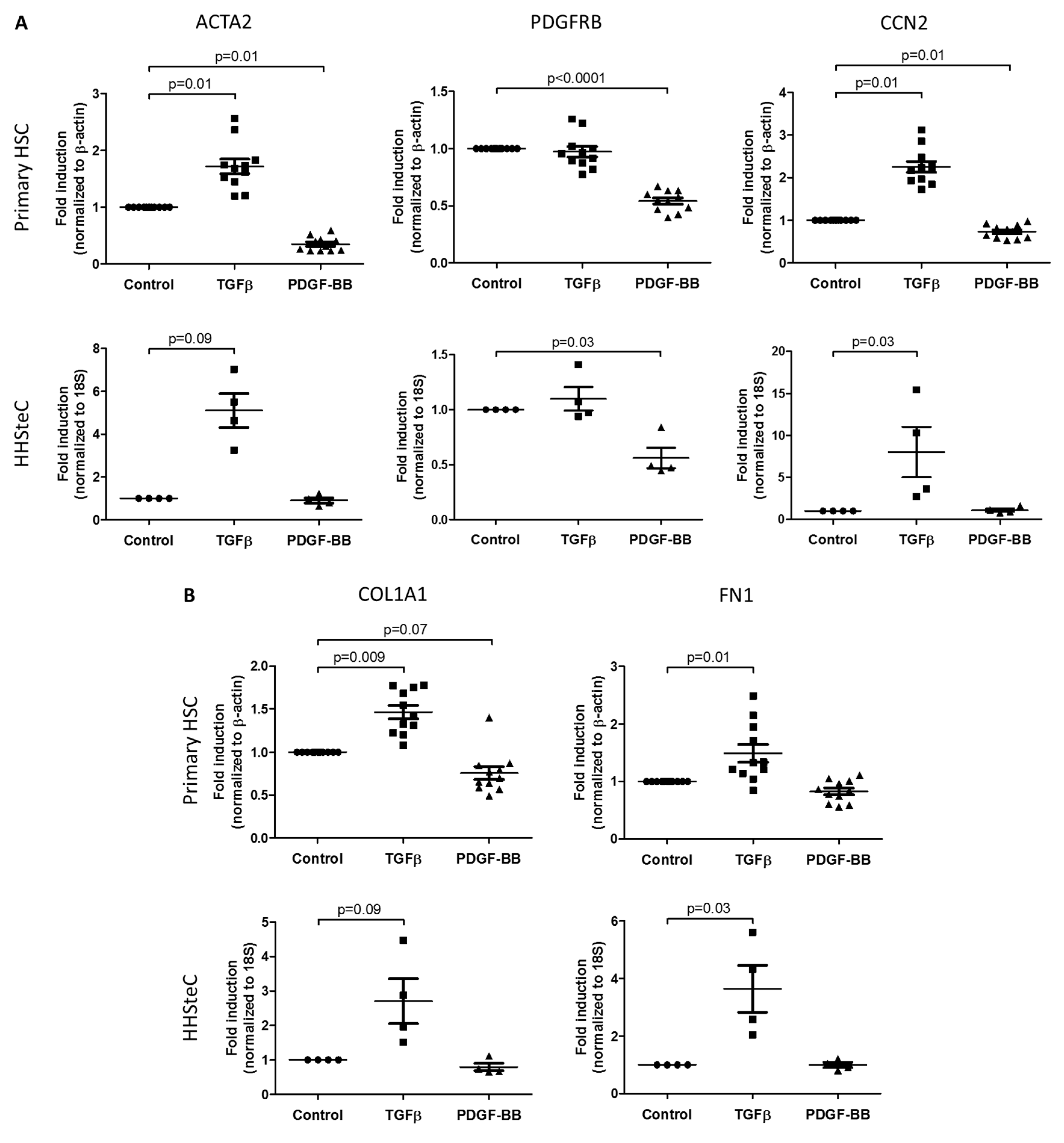
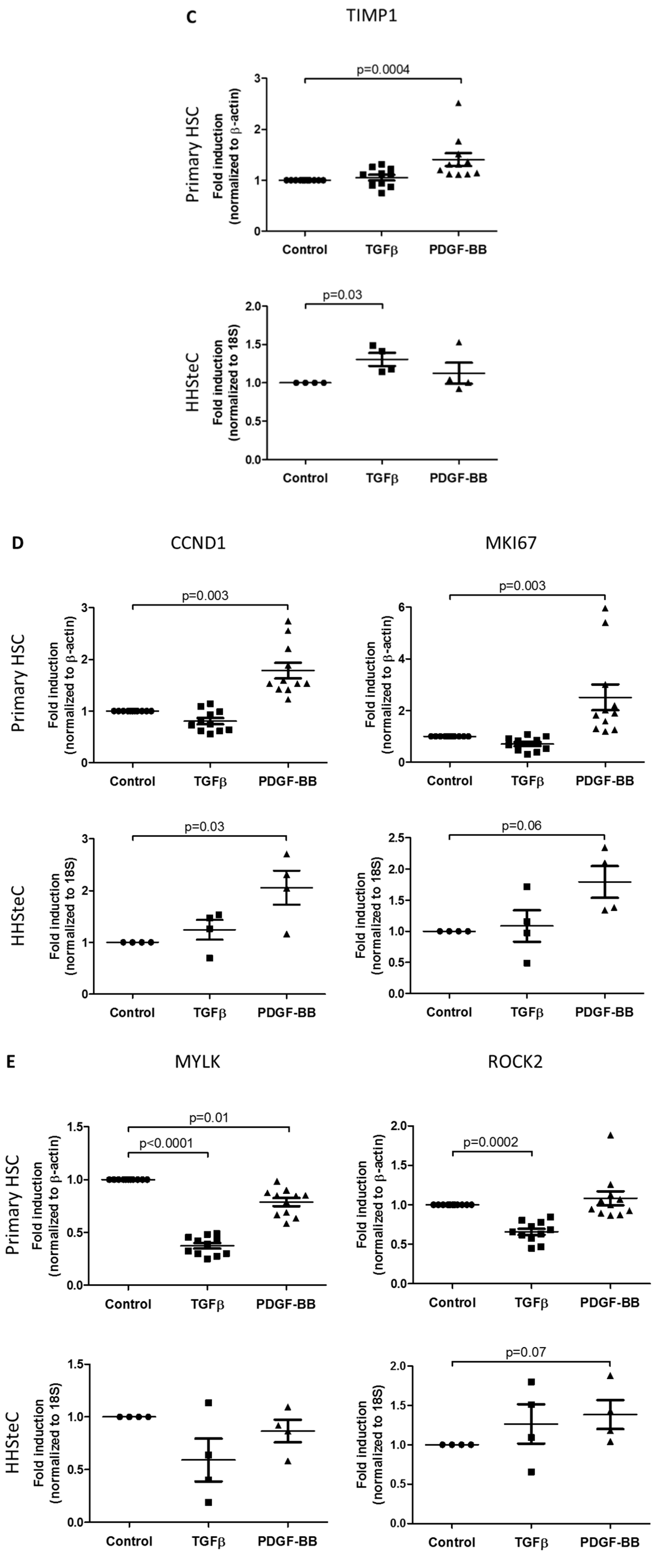
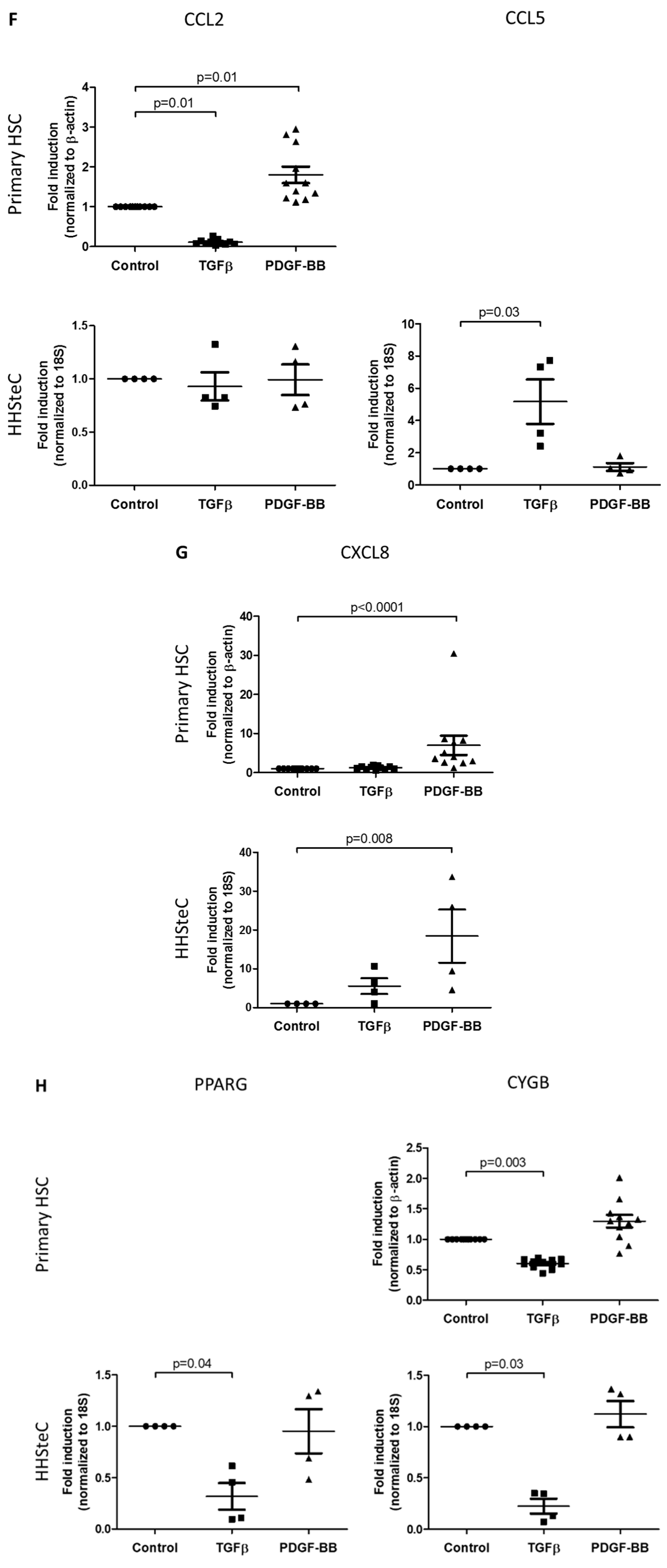
3.2. TGFβ- and PDGF-BB-Related Fibrotic Responses in Primary Human Hepatic Stellate Cells
3.3. TGFβ- and PDGF-BB-Related Fibrotic Responses in Human Precision-Cut Liver Slices
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Friedman, S.L. Hepatic Stellate Cells: Protean, Multifunctional, and Enigmatic Cells of the Liver. Physiol. Rev. 2007, 88, 125–172. [Google Scholar] [CrossRef] [PubMed]
- Nallagangula, K.S.; Nagaraj, S.K.; Venkataswamy, L.; Chandrappa, M. Liver fibrosis: A compilation on the biomarkers status and their significance during disease progression. Future Sci. OA 2017, 4, FSO250. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Friedman, S.L. Mechanisms of hepatic stellate cell activation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 397–411. [Google Scholar] [CrossRef]
- Choi, S.S.; Diehl, A.M. Epithelial-to-mesenchymal transitions in the liver. Hepatology 2009, 50, 2007–2013. [Google Scholar] [CrossRef] [PubMed]
- Kisseleva, T.; Brenner, D.A. Fibrogenesis of parenchymal organs. Proc. Am. Thorac. Soc. 2008, 5, 338–342. [Google Scholar] [CrossRef]
- Zeisberg, E.M.; Potenta, S.E.; Sugimoto, H.; Zeisberg, M.; Kalluri, R. Fibroblasts in kidney fibrosis emerge via endothelial-to-mesenchymal transition. J. Am. Soc. Nephrol. 2008, 19, 2282–2287. [Google Scholar] [CrossRef] [PubMed]
- Hempel, F.; Roderfeld, M.; Savai, R.; Sydykov, A.; Irungbam, K.; Schermuly, R.; Voswinckel, R.; Köhler, K.; Churin, Y.; Kiss, L.; et al. Depletion of Bone Marrow-Derived Fibrocytes Attenuates TAA-Induced Liver Fibrosis in Mice. Cells 2019, 8, 1210. [Google Scholar] [CrossRef]
- Mederacke, I.; Hsu, C.C.; Troeger, J.S.; Huebener, P.; Mu, X.; Dapito, D.H.; Pradere, J.P.; Schwabe, R.F. Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nat. Commun. 2013, 4, 2823. [Google Scholar] [CrossRef]
- Hasegawa, D.; Wallace, M.C.; Friedman, S.L. Stellate cells and hepatic fibrosis. In Stellate Cells in Health and Disease; Gandhi, C.R., Pinzani, M., Eds.; Academic Press: Cambridge, MA, USA, 2015; pp. 41–62. [Google Scholar]
- Mallat, A.; Lotersztajn, S. Reversion of hepatic stellate cell to a quiescent phenotype: From myth to reality? J. Hepatol. 2013, 59, 383–386. [Google Scholar] [CrossRef]
- Higashi, T.; Friedman, S.L.; Hoshida, Y. Hepatic stellate cells as key target in liver fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 27–42. [Google Scholar] [CrossRef]
- Kisseleva, T.; Cong, M.; Paik, Y.; Scholten, D.; Jiang, C.; Benner, C.; Iwaisako, K.; Moore-Morris, T.; Scott, B.; Tsukamoto, H.; et al. Myofibroblasts revert to an inactive phenotype during regression of liver fibrosis. Proc. Natl. Acad. Sci. USA 2012, 109, 9448–9453. [Google Scholar] [CrossRef] [PubMed]
- Troeger, J.S.; Mederacke, I.; Gwak, G.Y.; Dapito, D.H.; Mu, X.; Hsu, C.C.; Pradere, J.P.; Friedman, R.A.; Schwabe, R.F. Deactivation of hepatic stellate cells during liver fibrosis resolution in mice. Gastroenterology 2012, 143, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Popov, Y.; Schuppan, D. Targeting liver fibrosis: Strategies for development and validation of antifibrotic therapies. Hepatology 2009, 50, 1294–1306. [Google Scholar] [CrossRef] [PubMed]
- Olinga, P.; Schuppan, D. Precision-cut liver slices: A tool to model the liver ex vivo. J. Hepatol. 2013, 58, 1252–1253. [Google Scholar] [CrossRef]
- Lee, U.E.; Friedman, S.L. Mechanisms of hepatic fibrogenesis. Best Pract. Res. Clin. Gastroenterol. 2011, 25, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Rombouts, K.; Carloni, V. Determination and characterization of tetraspanin-associated phosphoinositide-4 kinases in primary and neoplastic liver cells. In Lipid Signaling Protocols; Waugh, M., Ed.; Springer: Berlin, Germany, 2016; pp. 203–212. [Google Scholar]
- Xu, L.; Hui, A.Y.; Albanis, E.; Arthur, M.J.; O’Byrne, S.M.; Blaner, W.S.; Mukherjee, P.; Friedman, S.L.; Eng, F.J. Human hepatic stellate cell lines, LX-1 and LX-2: New tools for analysis of hepatic fibrosis. Gut 2005, 54, 142–151. [Google Scholar] [CrossRef]
- Shibata, N.; Watanabe, T.; Okitsu, T.; Sakaguchi, M.; Takesue, M.; Kunieda, T.; Omoto, K.; Yamamoto, S.; Tanaka, N.; Kobayashi, N. Establishment of an immortalized human hepatic stellate cell line to develop antifibrotic therapies. Cell Transpl. 2003, 12, 499–507. [Google Scholar] [CrossRef]
- De Graaf, I.A.; Olinga, P.; de Jager, M.H.; Merema, M.T.; de Kanter, R.; van de Kerkhof, E.G.; Groothuis, G.M.M. Preparation and incubation of precision-cut liver and intestinal slices for application in drug metabolism and toxicity studies. Nat. Protoc. 2010, 5, 1540–1551. [Google Scholar] [CrossRef]
- Farrell, G.C.; Larter, C.Z. Nonalcoholic fatty liver disease: From steatosis to cirrhosis. Hepatology 2006, 43, S99–S112. [Google Scholar] [CrossRef]
- Chen, Z.; Jain, A.; Liu, H.; Zhao, Z.; Cheng, K. Targeted Drug Delivery to Hepatic Stellate Cells for the Treatment of Liver Fibrosis. J. Pharmacol. Exp. Ther. 2019, 370, 695–702. [Google Scholar] [CrossRef]
- Gandhi, C.R. Hepatic stellate cell activation and pro-fibrogenic signals. J. Hepatol. 2017, 67, 1104–1105. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, Y.; Okazaki, I. Emerging insights into Transforming growth factor beta Smad signal in hepatic fibrogenesis. Gut 2007, 56, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Marra, F.; Caligiuri, A. Cytokine production and signaling in stellate cells. In Stellate Cells in Health and Disease; Gandhi, C.R., Pinzani, M., Eds.; Academic Press: Cambridge, MA, USA, 2015; pp. 63–86. [Google Scholar]
- Pinzani, M. PDGF and signal transduction in hepatic stellate cells. Front. Biosci. 2002, 7, d1720–d1726. [Google Scholar] [CrossRef] [PubMed]
- Fallowfield, J.A.; Kendall, T.J.; Iredale, J.P. Reversal of fibrosis: No longer a pipe dream? Clin. Liver Dis. 2006, 10, 481–497. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.H.; Pinzani, M. Reversal of hepatic fibrosis: Pathophysiological basis of antifibrotic therapies. Hepat. Med. 2011, 3, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Sato-Matsubara, M.; Matsubara, T.; Daikoku, A.; Okina, Y.; Longato, L.; Rombouts, K.; Thuy, L.T.; Adachi, J.; Tomonaga, T.; Ikeda, K.; et al. Fibroblast growth factor 2 (FGF2) regulates cytoglobin expression and activation of human hepatic stellate cells via JNK signaling. J. Biol. Chem. 2017, 292, 18961–18972. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, J.; Brenner, D.A.; Kisseleva, T. Reversibility of Liver Fibrosis and Inactivation of Fibrogenic Myofibroblasts. Curr. Pathobiol. Rep. 2013, 1, 209–214. [Google Scholar] [CrossRef]
- Ramachandran, P.; Henderson, N.C. Antifibrotics in chronic liver disease: Tractable targets and translational challenges. Lancet Gastroenterol. Hepatol. 2016, 1, 328–340. [Google Scholar] [CrossRef]
| Gene Symbol | Full Gene Name | Sequence Forward | Sequence Reverse |
|---|---|---|---|
| ACTA2 | Alpha smooth muscle actin | CCCCATCTATGAGGGCTATG | CAGTGGCCATCTCATTTTCA |
| PDGFRB | Platelet-derived growth factor receptor beta | GTAAGGTGCCAACCTGCAAT | CATGGGGGTATGGTTTTGTC |
| CCN2 | Cellular communication network factor 2 | GACTGGAAGACACGTTTGGC | CTTCCAGGTCAGCTTCGCAA |
| COL1A1 | Collagen type I alpha 1 chain | GTACTGGATTGACCCCAACC | CGCCATACTCGAACTGGAAT |
| FN1 | Fibronectin 1 | AGGCTTGAACCAACCTACGGATGA | GCCTAAGCACTGGCACAACAGTTT |
| TIMP1 | Tissue inhibitor of metalloproteinases 1 | AATTCCGACCTCGTCATCAG | TGCAGTTTTCCAGCAATGAG |
| CCND1 | Cyclin D1 | GACCCCGCACGATTTCATTG | AAGTTGTTGGGGCTCCTCAG |
| MKI67 | Marker of proliferation Ki-67 | CGTCCCAGTGGAAGAGTTGT | CCCCTTCCAAACAAGCAGGT |
| MYLK | Myosin light-chain kinase | GGTGACATGGCACAGAAACG | AGCTGCTTCGCAAAACTTCC |
| ROCK2 | Rho-associated coiled coil-containing protein kinase 2 | AACGTCAGGATGCAGATGGG | CAGCCAAAGAGTCCCGTTCA |
| CCL2 | C-C motif chemokine ligand 2 | CTCGCTCAGCCAGATGCAAT | TCCATGGAATCCTGAACCCAC |
| CCL5 | C-C motif chemokine ligand 5 | TGCTGCTTTGCCTACATTGC | CACACTTGGCGGTTCTTTCG |
| TNF | Tumor necrosis factor alpha | CGTCTCCTACCAGACCAAGG | CCAAAGTAGACCTGCCCAGA |
| CXCL8 | C-X-C motif chemokine ligand 8 | TGCAGTTTTGCCAAGGAGTG | CAACCCTCTGCACCCAGTTT |
| PPARG | Peroxisome proliferator activated receptor gamma | AGGAAGGGGCCTTAACCTCT | CACGGAGCTGATCCCAAAGT |
| CYGB | Cytoglobin | GTCATTCTGGAGGTGGTCGC | GTGGAGTTAGGGGTCCTACG |
| ACTB | Beta actin | CCTCGCCTTTGCCGATCC | AGGAATCCTTCTGACCCATGC |
| RNA18S5 | RNA, 18S ribosomal N5 | CGGCTACCCACATCCAAGGA | CCAATTACAGGGCCTCGAAA |
| HSC Activity | Gene | TGF-β | PDGF-BB | ||||||
|---|---|---|---|---|---|---|---|---|---|
| pHSC | HHSteC | LX-2 | TWNT-4 | pHSC | HHSteC | LX-2 | TWNT-4 | ||
| Activation | ACTA2 | ||||||||
| PDGFRB | |||||||||
| CCN2 | |||||||||
| Fibrogenesis | COL1A1 | ||||||||
| FN1 | |||||||||
| Matrix degradation | TIMP1 | ||||||||
| Proliferation | CCND1 | ||||||||
| MKI67 | |||||||||
| Contractility | MYLK | ||||||||
| ROCK2 | |||||||||
| Chemotaxis | CCL2 | ||||||||
| CCL5 | |||||||||
| Inflammatory signaling | TNF | ||||||||
| CXCL8 | |||||||||
| Reversion | PPARG | ||||||||
| CYGB | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Dijk, F.; Hazelhoff, C.M.; Post, E.; Prins, G.G.H.; Rombouts, K.; Poelstra, K.; Olinga, P.; Beljaars, L. Design of a Gene Panel to Expose the Versatile Role of Hepatic Stellate Cells in Human Liver Fibrosis. Pharmaceutics 2020, 12, 278. https://doi.org/10.3390/pharmaceutics12030278
van Dijk F, Hazelhoff CM, Post E, Prins GGH, Rombouts K, Poelstra K, Olinga P, Beljaars L. Design of a Gene Panel to Expose the Versatile Role of Hepatic Stellate Cells in Human Liver Fibrosis. Pharmaceutics. 2020; 12(3):278. https://doi.org/10.3390/pharmaceutics12030278
Chicago/Turabian Stylevan Dijk, Fransien, Christa M. Hazelhoff, Eduard Post, Gerian G. H. Prins, Krista Rombouts, Klaas Poelstra, Peter Olinga, and Leonie Beljaars. 2020. "Design of a Gene Panel to Expose the Versatile Role of Hepatic Stellate Cells in Human Liver Fibrosis" Pharmaceutics 12, no. 3: 278. https://doi.org/10.3390/pharmaceutics12030278
APA Stylevan Dijk, F., Hazelhoff, C. M., Post, E., Prins, G. G. H., Rombouts, K., Poelstra, K., Olinga, P., & Beljaars, L. (2020). Design of a Gene Panel to Expose the Versatile Role of Hepatic Stellate Cells in Human Liver Fibrosis. Pharmaceutics, 12(3), 278. https://doi.org/10.3390/pharmaceutics12030278





