Monkeypox: A Comprehensive Review
Abstract
1. Introduction
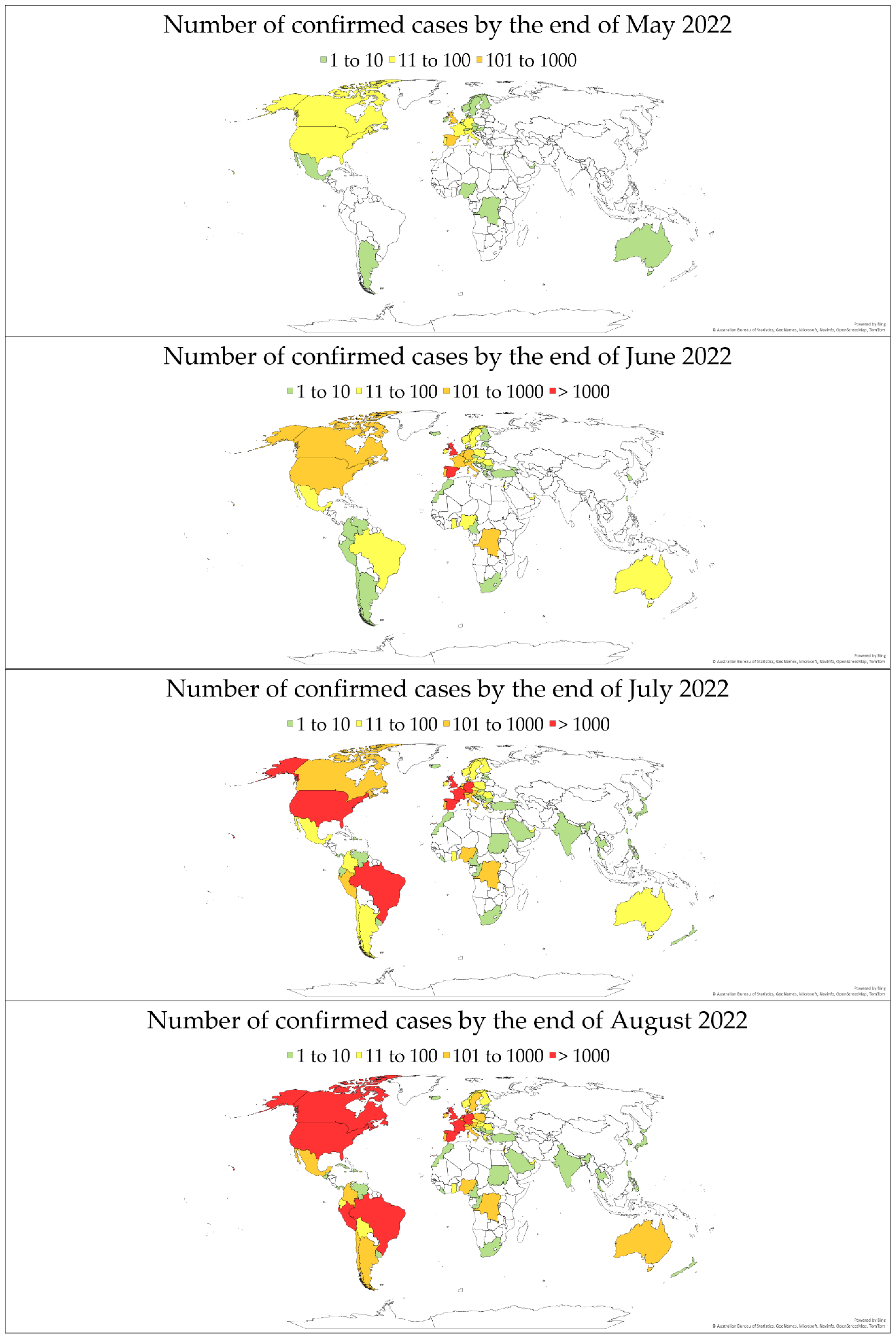
2. An Updated Overview of Confirmed Case Numbers Worldwide
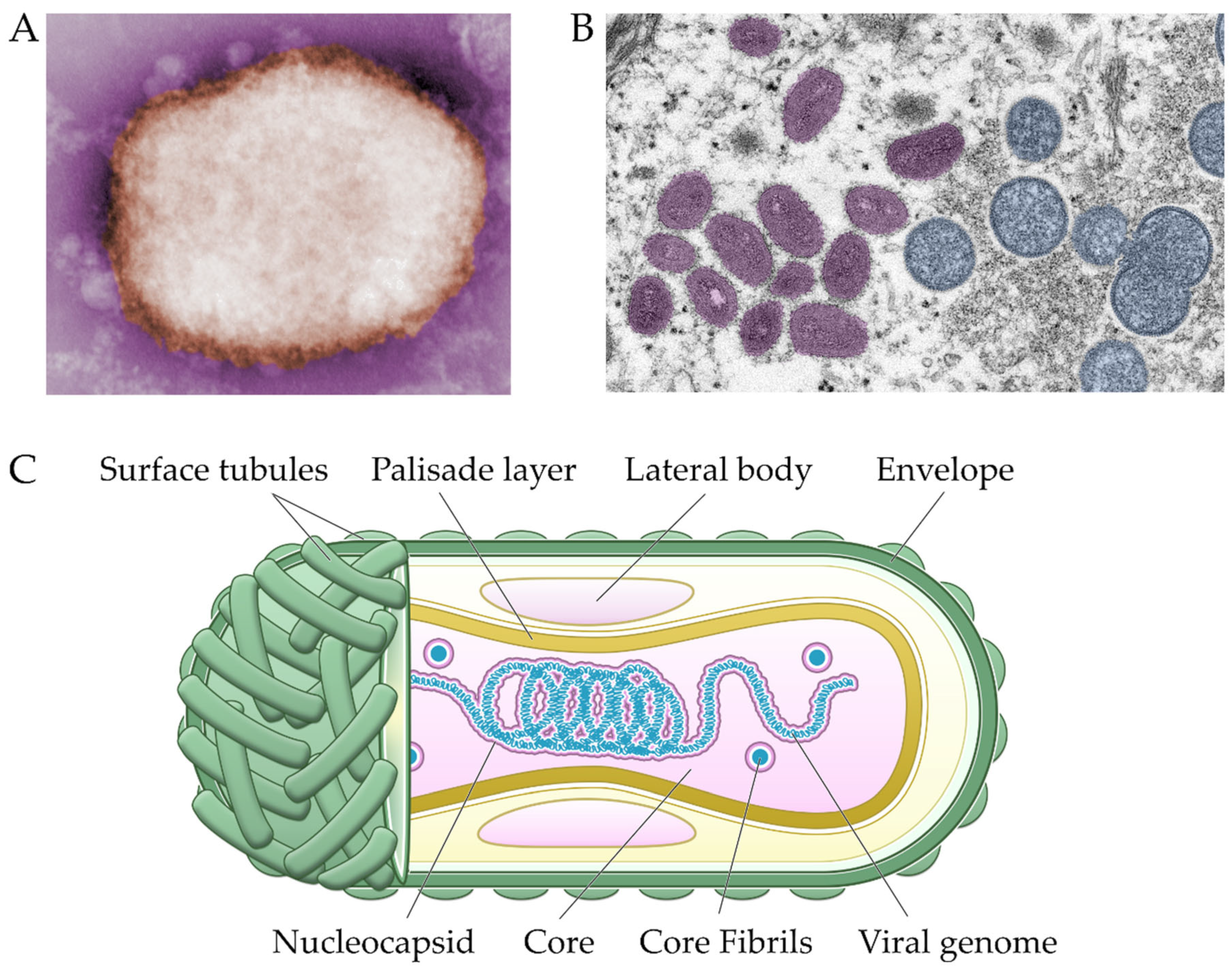
3. Virology and Genomic Classification
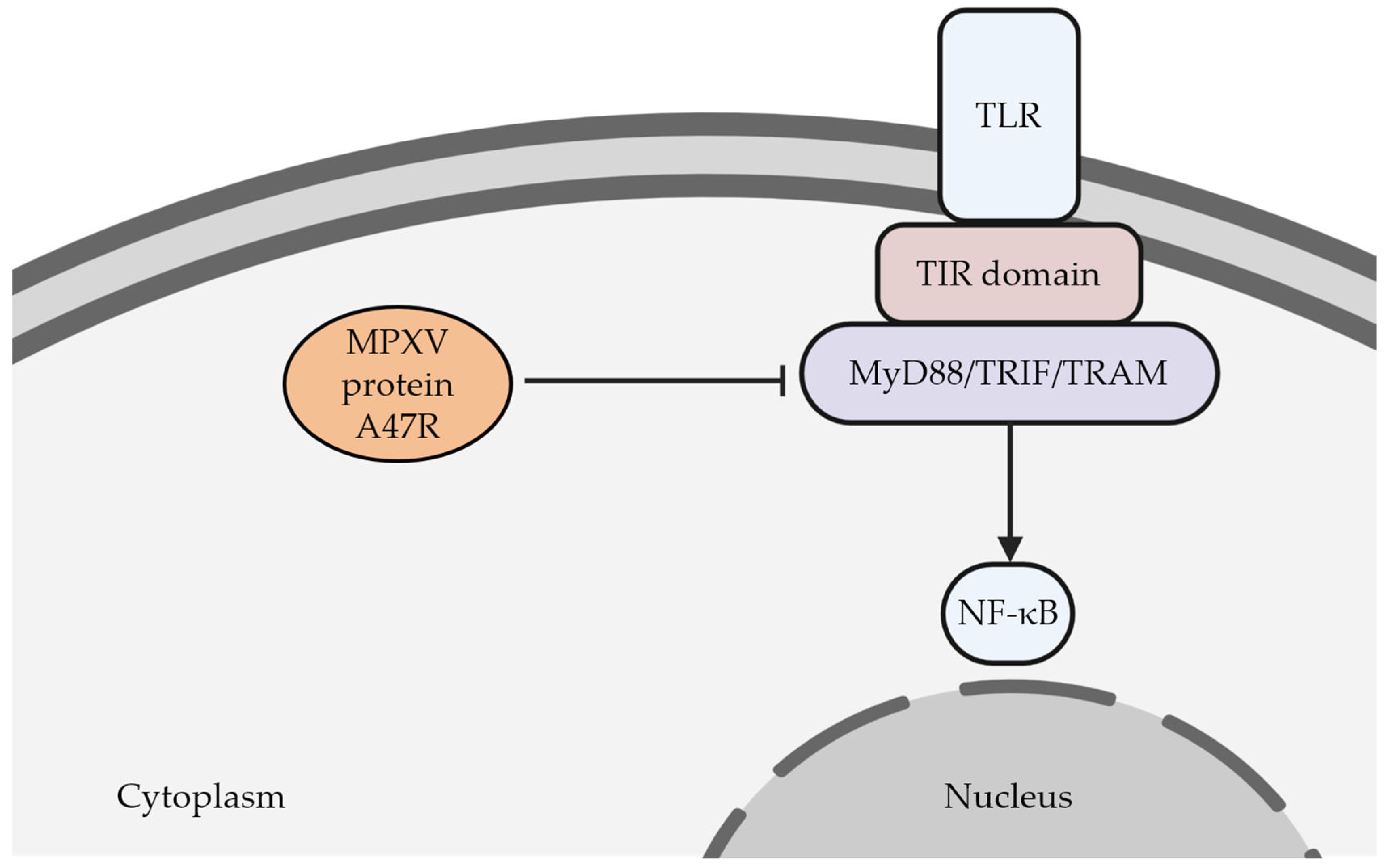
4. Pathophysiology and Immune Evasion
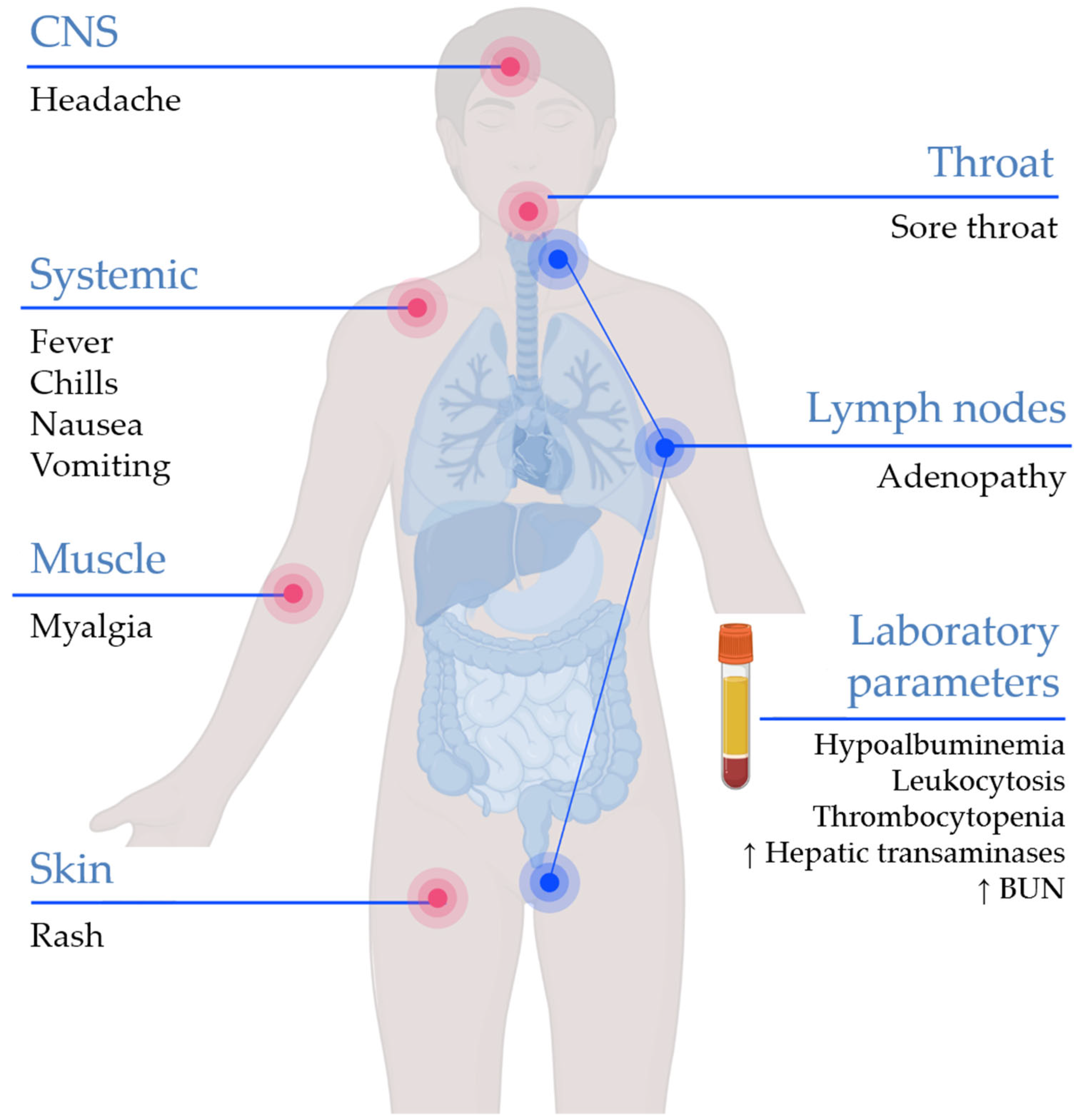
5. Clinical Characteristics and Laboratory Findings
6. Diagnosis
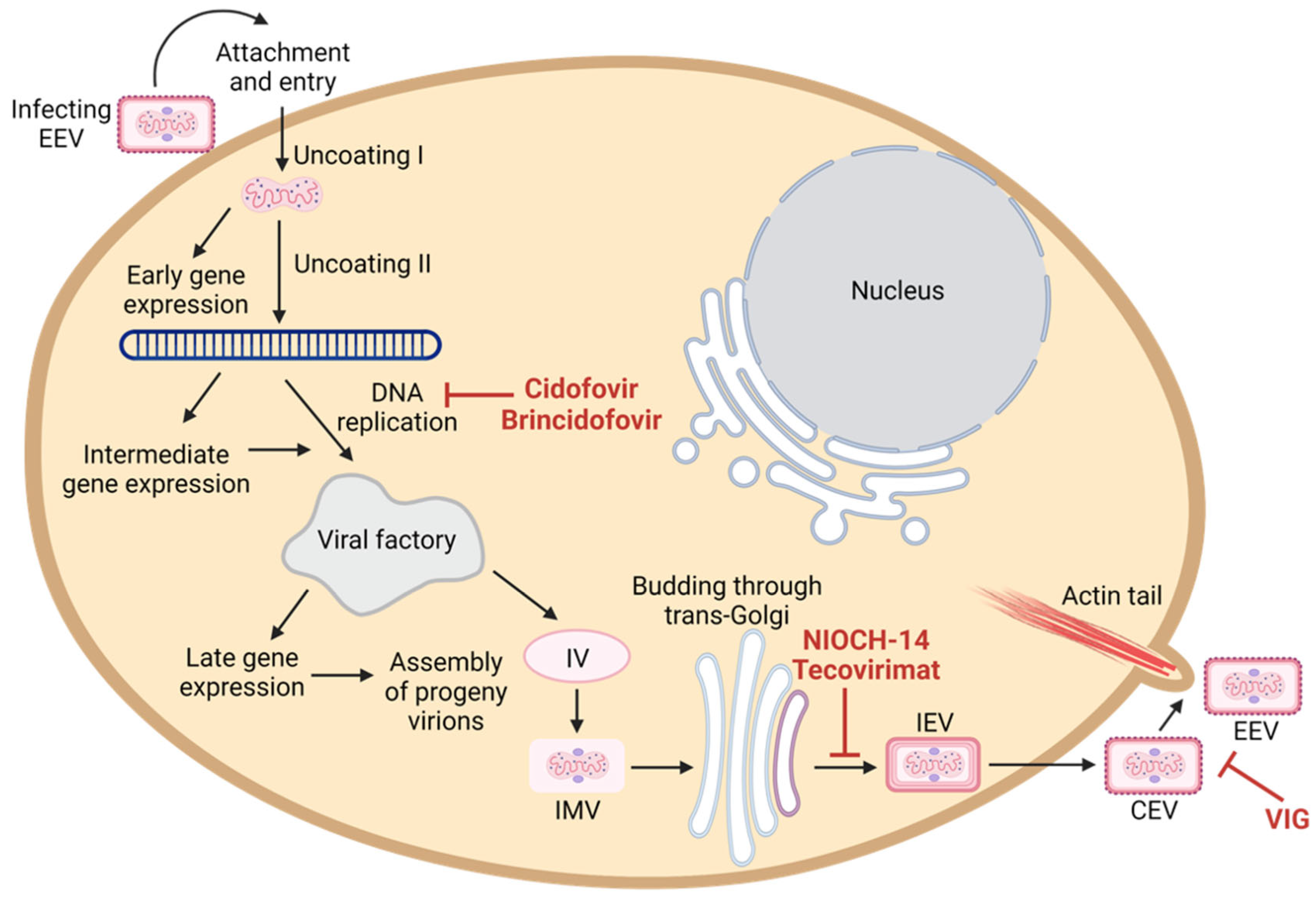
7. Treatment
7.1. Management of Mild or Uncomplicated Monkeypox
7.2. Management of High-Risk Patients and Severe or Complicated Monkeypox
7.3. Antiviral Agents
7.3.1. Tecovirimat
7.3.2. Brincidofovir and Cidofovir
7.3.3. NIOCH-14
7.3.4. Vaccinia Immunoglobulin (VIG)
8. Complications and Sequalae
9. Prevention
9.1. Prevention for Individual, Household, and Community
9.2. Prevention and Control in Healthcare Settings
10. Vaccines and Vaccination
10.1. ACAM2000
10.2. JYNNEOS
11. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO | World Health Organization. Multi-Country Monkeypox Outbreak in Non-Endemic Countries. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON385 (accessed on 30 May 2022).
- Centers for Disease Control and Prevention (CDC). 2022 Monkeypox Outbreak Global Map. Available online: https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html (accessed on 10 September 2022).
- WHO | World Health Organization. WHO Director-General’s Statement at the Press Conference Following IHR Emergency Committee Regarding the Multi-Country Outbreak of Monkeypox, 23 July 2022. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-statement-on-the-press-conference-following-IHR-emergency-committee-regarding-the-multi--country-outbreak-of-monkeypox--23-july-2022 (accessed on 25 July 2022).
- Sklenovská, N.; Van Ranst, M. Emergence of Monkeypox as the Most Important Orthopoxvirus Infection in Humans. Front. Public Health 2018, 6, 241. [Google Scholar] [CrossRef]
- Magnus, P.v.; Andersen, E.K.; Petersen, K.B.; Birch-Andersen, A. A pox-like disease in cynomolgus monkeys. Pathol. Microbiol. Scand. 1959, 46, 156–176. [Google Scholar] [CrossRef]
- Arita, I.; Gispen, R.; Kalter, S.S.; Wah, L.T.; Marennikova, S.S.; Netter, R.; Tagaya, I. Outbreaks of monkeypox and serological surveys in nonhuman primates. Bull. World Health Organ. 1972, 46, 625–631. [Google Scholar]
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A human infection caused by monkeypox virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593–597. [Google Scholar]
- Rezza, G. Emergence of human monkeypox in west Africa. Lancet Infect. Dis. 2019, 19, 797–799. [Google Scholar] [CrossRef]
- Bunge, E.M.; Hoet, B.; Chen, L.; Lienert, F.; Weidenthaler, H.; Baer, L.R.; Steffen, R. The changing epidemiology of human monkeypox—A potential threat? A systematic review. PLoS Negl. Trop. Dis. 2022, 16, e0010141. [Google Scholar] [CrossRef]
- Nolen, L.D.; Osadebe, L.; Katomba, J.; Likofata, J.; Mukadi, D.; Monroe, B.; Doty, J.; Hughes, C.M.; Kabamba, J.; Malekani, J.; et al. Extended Human-to-Human Transmission during a Monkeypox Outbreak in the Democratic Republic of the Congo. Emerg. Infect. Dis. 2016, 22, 1014–1021. [Google Scholar] [CrossRef]
- Hammarlund, E.; Lewis, M.W.; Carter, S.V.; Amanna, I.; Hansen, S.G.; Strelow, L.I.; Wong, S.W.; Yoshihara, P.; Hanifin, J.M.; Slifka, M.K. Multiple diagnostic techniques identify previously vaccinated individuals with protective immunity against monkeypox. Nat. Med. 2005, 11, 1005–1011. [Google Scholar] [CrossRef]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The Detection of Monkeypox in Humans in the Western Hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef]
- Beer, E.M.; Rao, V.B. A systematic review of the epidemiology of human monkeypox outbreaks and implications for outbreak strategy. PLoS Negl. Trop. Dis. 2019, 13, e0007791. [Google Scholar] [CrossRef]
- Rimoin Anne, W.; Mulembakani Prime, M.; Johnston Sara, C.; Lloyd Smith James, O.; Kisalu Neville, K.; Kinkela Timothee, L.; Blumberg, S.; Thomassen Henri, A.; Pike Brian, L.; Fair Joseph, N.; et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the Democratic Republic of Congo. Proc. Natl. Acad. Sci. USA 2010, 107, 16262–16267. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.W.; Kabamba, J.; McCollum, A.M.; Lushima, R.S.; Wemakoy, E.O.; Muyembe Tamfum, J.-J.; Nguete, B.; Hughes, C.M.; Monroe, B.P.; Reynolds, M.G. Vaccinating against monkeypox in the Democratic Republic of the Congo. Antivir. Res. 2019, 162, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Khodakevich, L.; Szczeniowski, M.; Nambu-ma, D.; Jezek, Z.; Marennikova, S.; Nakano, J.; Meier, F. Monkeypox virus in relation to the ecological features surrounding human settlements in Bumba zone, Zaire. Trop. Geogr. Med. 1987, 39, 56–63. [Google Scholar] [PubMed]
- Haider, N.; Guitian, J.; Simons, D.; Asogun, D.; Ansumana, R.; Honeyborne, I.; Velavan, T.P.; Ntoumi, F.; Valdoleiros, S.R.; Petersen, E.; et al. Increased outbreaks of monkeypox highlight gaps in actual disease burden in Sub-Saharan Africa and in animal reservoirs. Int. J. Infect. Dis. 2022, 122, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, E.; Dattani, S.; Ritchie, H.; Max, R.; Fiona, S. Monkeypox: Cumulative Confirmed Cases, by Date of Confirmation. 2022. Available online: https://ourworldindata.org/monkeypox (accessed on 9 September 2000).
- Falendysz, E.A.; Lopera, J.G.; Doty, J.B.; Nakazawa, Y.; Crill, C.; Lorenzsonn, F.; Kalemba, L.s.N.; Ronderos, M.D.; Mejia, A.; Malekani, J.M.; et al. Characterization of Monkeypox virus infection in African rope squirrels (Funisciurus sp.). PLoS Negl. Trop. Dis. 2017, 11, e0005809. [Google Scholar] [CrossRef] [PubMed]
- Radonić, A.; Metzger, S.; Dabrowski, P.W.; Couacy-Hymann, E.; Schuenadel, L.; Kurth, A.; Mätz-Rensing, K.; Boesch, C.; Leendertz, F.H.; Nitsche, A. Fatal monkeypox in wild-living sooty mangabey, Côte d’Ivoire, 2012. Emerg. Infect. Dis. 2014, 20, 1009–1011. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Doty, J.B.; McCollum, A.M.; Olson, V.A.; Nakazawa, Y. Monkeypox re-emergence in Africa: A call to expand the concept and practice of One Health. Expert Rev. Anti. Infect. Ther. 2019, 17, 129–139. [Google Scholar] [CrossRef]
- Khodakevich, L.; Jezek, Z.; Kinzanzka, K. Isolation of monkeypox virus from wild squirrel infected in nature. Lancet 1986, 1, 98–99. [Google Scholar] [CrossRef]
- Gruber, C.E.M.; Giombini, E.; Mazzotta, V.; Vita, S.; Carletti, F.; Rueca, M.; Gramigna, G.; D’Abramo, A.; Cicalini, S.; Lapa, D.; et al. First Available Monkeypox Genome Sequence From the First Case in Italy Related to Multi-Country Outbreak in May 2022. Available online: https://virological.org/t/first-available-monkeypox-genome-sequence-from-the-first-case-in-italy-related-to-multi-country-outbreak-in-may-2022/833 (accessed on 20 September 2022).
- Happi, C.; Adetifa, I.; Mbala, P.; Njouom, R.; Nakoune, E.; Happi, A.; Ndodo, N.; Ayansola, O.; Mboowa, G.; Bedford, T.; et al. Urgent Need for a Non-Discriminatory and Non-Stigmatizing Nomenclature for Monkeypox Virus. Available online: https://virological.org/t/urgent-need-for-a-non-discriminatory-and-non-stigmatizing-nomenclature-for-monkeypox-virus/853 (accessed on 20 September 2022).
- Tree, J.A.; Hall, G.; Pearson, G.; Rayner, E.; Graham, V.A.; Steeds, K.; Bewley, K.R.; Hatch, G.J.; Dennis, M.; Taylor, I.; et al. Sequence of pathogenic events in cynomolgus macaques infected with aerosolized monkeypox virus. J. Virol. 2015, 89, 4335–4344. [Google Scholar] [CrossRef]
- Osorio, J.E.; Iams, K.P.; Meteyer, C.U.; Rocke, T.E. Comparison of monkeypox viruses pathogenesis in mice by in vivo imaging. PLoS ONE 2009, 4, e6592. [Google Scholar] [CrossRef]
- Sklenovská, N. Monkeypox Virus. In Animal-Origin Viral Zoonoses; Malik, Y.S., Singh, R.K., Dhama, K., Eds.; Springer: Singapore, 2020; pp. 39–68. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). CDC Public Health Image Library (PHIL). Available online: https://phil.cdc.gov (accessed on 20 September 2022).
- Zhao, K.; Wohlhueter, R.M.; Li, Y. Finishing monkeypox genomes from short reads: Assembly analysis and a neural network method. BMC Genom. 2016, 17 (Suppl. 5), 497. [Google Scholar] [CrossRef] [PubMed]
- Remichkova, M. Poxviruses: Smallpox vaccine, its complications and chemotherapy. Virus Adaptation Treat. 2010, 2010, 41–46. [Google Scholar] [CrossRef]
- Fenner, F. The Orthopoxviruses; Frank, F., Ed.; Elsevier: Amsterdam, The Netherlands, 2012; p. 442. [Google Scholar]
- Haller, S.L.; Peng, C.; McFadden, G.; Rothenburg, S. Poxviruses and the evolution of host range and virulence. Infect. Genet. Evol. 2014, 21, 15–40. [Google Scholar] [CrossRef]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-Rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A tale of two clades: Monkeypox viruses. J. Gen. Virol. 2005, 86 Pt 10, 2661–2672. [Google Scholar] [CrossRef]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, F.M.; Pozzobon, D.; Di Meglio, A.; Mercatelli, D. Genomic characterization of the recent monkeypox outbreak. bioRxiv 2022. [Google Scholar] [CrossRef]
- Happi, C.; Adetifa, I.; Mbala, P.; Njouom, R.; Nakoune, E.; Happi, A.; Ndodo, N.; Ayansola, O.; Mboowa, G.; Bedford, T.; et al. Urgent need for a non-discriminatory and non-stigmatizing nomenclature for monkeypox virus. PLoS Biol. 2022, 20, e3001769. [Google Scholar] [CrossRef]
- Knight, J.C.; Goldsmith, C.S.; Tamin, A.; Regnery, R.L.; Regnery, D.C.; Esposito, J.J. Further analyses of the orthopoxviruses volepox virus and raccoon poxvirus. Virology 1992, 190, 423–433. [Google Scholar] [CrossRef]
- Liszewski, M.K.; Leung, M.K.; Hauhart, R.; Buller, R.M.L.; Bertram, P.; Wang, X.; Rosengard, A.M.; Kotwal, G.J.; Atkinson, J.P. Structure and regulatory profile of the monkeypox inhibitor of complement: Comparison to homologs in vaccinia and variola and evidence for dimer formation. J. Immunol. 2006, 176, 3725–3734. [Google Scholar] [CrossRef]
- Estep, R.D.; Messaoudi, I.; O’Connor, M.A.; Li, H.; Sprague, J.; Barron, A.; Engelmann, F.; Yen, B.; Powers, M.F.; Jones, J.M.; et al. Deletion of the monkeypox virus inhibitor of complement enzymes locus impacts the adaptive immune response to monkeypox virus in a nonhuman primate model of infection. J. Virol. 2011, 85, 9527–9542. [Google Scholar] [CrossRef]
- Chen, N.; Li, G.; Liszewski, M.K.; Atkinson, J.P.; Jahrling, P.B.; Feng, Z.; Schriewer, J.; Buck, C.; Wang, C.; Lefkowitz, E.J.; et al. Virulence differences between monkeypox virus isolates from West Africa and the Congo basin. Virology 2005, 340, 46–63. [Google Scholar] [CrossRef] [PubMed]
- WHO | World Health Organization. Multi-Country Monkeypox Outbreak: Situation Update. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON393 (accessed on 20 September 2022).
- Hutson, C.L.; Carroll, D.S.; Gallardo-Romero, N.; Drew, C.; Zaki, S.R.; Nagy, T.; Hughes, C.; Olson, V.A.; Sanders, J.; Patel, N.; et al. Comparison of Monkeypox Virus Clade Kinetics and Pathology within the Prairie Dog Animal Model Using a Serial Sacrifice Study Design. Biomed. Res Int. 2015, 2015, 965710. [Google Scholar] [CrossRef] [PubMed]
- McCollum, A.M.; Damon, I.K. Human monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Ogoina, D.; Izibewule, J.H.; Ogunleye, A.; Ederiane, E.; Anebonam, U.; Neni, A.; Oyeyemi, A.; Etebu, E.N.; Ihekweazu, C. The 2017 human monkeypox outbreak in Nigeria—Report of outbreak experience and response in the Niger Delta University Teaching Hospital, Bayelsa State, Nigeria. PLoS ONE 2019, 14, e0214229. [Google Scholar] [CrossRef]
- Durski, K.N.; McCollum, A.M.; Nakazawa, Y.; Petersen, B.W.; Reynolds, M.G.; Briand, S.; Djingarey, M.H.; Olson, V.; Damon, I.K.; Khalakdina, A. Emergence of monkeypox—West and central Africa, 1970–2017. Morb. Mortal. Wkly. Rep. 2018, 67, 306. [Google Scholar] [CrossRef]
- Di Giulio, D.B.; Eckburg, P.B. Human monkeypox: An emerging zoonosis. Lancet. Infect. Dis. 2004, 4, 15–25. [Google Scholar] [CrossRef]
- Weinstein, R.A.; Nalca, A.; Rimoin, A.W.; Bavari, S.; Whitehouse, C.A. Reemergence of monkeypox: Prevalence, diagnostics, and countermeasures. Clin. Infect. Dis. 2005, 41, 1765–1771. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; McCollum, A.M.; Nguete, B.; Shongo Lushima, R.; Petersen, B.W. Improving the care and treatment of monkeypox patients in low-resource settings: Applying evidence from contemporary biomedical and smallpox biodefense research. Viruses 2017, 9, 380. [Google Scholar] [CrossRef]
- Amarante-Mendes, G.P.; Adjemian, S.; Branco, L.M.; Zanetti, L.C.; Weinlich, R.; Bortoluci, K.R. Pattern Recognition Receptors and the Host Cell Death Molecular Machinery. Front. Immunol. 2018, 9, 2379. [Google Scholar] [CrossRef]
- Shchelkunov, S.N. Orthopoxvirus genes that mediate disease virulence and host tropism. Adv. Virol. 2012, 2012, 524743. [Google Scholar] [CrossRef]
- O’Neill, L.A.J.; Bowie, A.G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat. Rev. Immunol. 2007, 7, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Albensi, B.C. What Is Nuclear Factor Kappa B (NF-κB) Doing in and to the Mitochondrion? Front. Cell Dev. Biol. 2019, 7, 154. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.S.; Ghosh, S. Shared principles in NF-κB signaling. Cell 2008, 132, 344–362. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y. Caspase activation, inhibition, and reactivation: A mechanistic view. Protein. Protein Sci. 2004, 13, 1979–1987. [Google Scholar] [CrossRef]
- Youle, R.J.; Strasser, A. The BCL-2 protein family: Opposing activities that mediate cell death. Nat. Rev. Mol. Cell. Biol. 2008, 9, 47–59. [Google Scholar] [CrossRef]
- Li, Z.W.; Chu, W.; Hu, Y.; Delhase, M.; Deerinck, T.; Ellisman, M.; Johnson, R.; Karin, M. The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J. Exp. Med. 1999, 189, 1839–1845. [Google Scholar] [CrossRef]
- Li, X.; Massa, P.E.; Hanidu, A.; Peet, G.W.; Aro, P.; Savitt, A.; Mische, S.; Li, J.; Marcu, K.B. IKKα, IKKβ, and NEMO/IKKγ Are Each Required for the NF-κB-mediated Inflammatory Response Program*. J. Biol. Chem. 2002, 277, 45129–45140. [Google Scholar] [CrossRef]
- Goetzke, C.C.; Ebstein, F.; Kallinich, T. Role of Proteasomes in Inflammation. J. Clin. Med. 2021, 10, 1783. [Google Scholar] [CrossRef]
- Howard, J.; Justus, D.E.; Totmenin, A.V.; Shchelkunov, S.; Kotwal, G.J. Molecular mimicry of the inflammation modulatory proteins (IMPs) of poxviruses: Evasion of the inflammatory response to preserve viral habitat. J. Leukoc. Biol. 1998, 64, 68–71. [Google Scholar] [CrossRef]
- Miller, C.G.; Shchelkunov, S.N.; Kotwal, G.J. The cowpox virus-encoded homolog of the vaccinia virus complement control protein is an inflammation modulatory protein. Virology 1997, 229, 126–133. [Google Scholar] [CrossRef]
- Huhn, G.D.; Bauer, A.M.; Yorita, K.; Graham, M.B.; Sejvar, J.; Likos, A.; Damon, I.K.; Reynolds, M.G.; Kuehnert, M.J. Clinical characteristics of human monkeypox, and risk factors for severe disease. Clin. Infect. Dis. 2005, 41, 1742–1751. [Google Scholar] [CrossRef]
- Pittman, P.R.; Martin, J.W.; Kingebeni, P.M.; Tamfum, J.-J.M.; Wan, Q.; Reynolds, M.G.; Quinn, X.; Norris, S.; Townsend, M.B.; Satheshkumar, P.S. Clinical characterization of human monkeypox infections in the Democratic Republic of the Congo. medRxiv 2022. [Google Scholar] [CrossRef]
- Adler, H.; Gould, S.; Hine, P.; Snell, L.B.; Wong, W.; Houlihan, C.F.; Osborne, J.C.; Rampling, T.; Beadsworth, M.B.; Duncan, C.J.; et al. Clinical features and management of human monkeypox: A retrospective observational study in the UK. Lancet. Infect. Dis. 2022, 22, 1153–1162. [Google Scholar] [CrossRef]
- Yinka-Ogunleye, A.; Aruna, O.; Dalhat, M.; Ogoina, D.; McCollum, A.; Disu, Y.; Mamadu, I.; Akinpelu, A.; Ahmad, A.; Burga, J. Outbreak of human monkeypox in Nigeria in 2017–18: A clinical and epidemiological report. Lancet. Infect. Dis. 2019, 19, 872–879. [Google Scholar] [CrossRef]
- Guagliardo, S.A.J.; Monroe, B.; Moundjoa, C.; Athanase, A.; Okpu, G.; Burgado, J.; Townsend, M.B.; Satheshkumar, P.S.; Epperson, S.; Doty, J.B. Asymptomatic orthopoxvirus circulation in humans in the wake of a monkeypox outbreak among chimpanzees in Cameroon. Am. J. Trop. Med. Hyg. 2020, 102, 206. [Google Scholar] [CrossRef] [PubMed]
- De Baetselier, I.; Van Dijck, C.; Kenyon, C.; Coppens, J.; Michiels, J.; de Block, T.; Smet, H.; Coppens, S.; Vanroye, F.; Bugert, J.J.; et al. Retrospective detection of asymptomatic monkeypox virus infections among male sexual health clinic attendees in Belgium. Nat. Med. 2022; Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Grant, R.; Nguyen, L.-B.L.; Breban, R. Modelling human-to-human transmission of monkeypox. Bull. World Health Organ. 2020, 98, 638. [Google Scholar] [CrossRef]
- Hobson, G.; Adamson, J.; Adler, H.; Firth, R.; Gould, S.; Houlihan, C.; Johnson, C.; Porter, D.; Rampling, T.; Ratcliffe, L. Family cluster of three cases of monkeypox imported from Nigeria to the United Kingdom, May 2021. Eurosurveillance 2021, 26, 2100745. [Google Scholar] [CrossRef]
- Petersen, E.; Kantele, A.; Koopmans, M.; Asogun, D.; Yinka-Ogunleye, A.; Ihekweazu, C.; Zumla, A. Human monkeypox: Epidemiologic and clinical characteristics, diagnosis, and prevention. Infect. Dis. Clin. 2019, 33, 1027–1043. [Google Scholar] [CrossRef]
- Di Gennaro, F.; Veronese, N.; Marotta, C.; Shin, J.I.; Koyanagi, A.; Silenzi, A.; Antunes, M.; Saracino, A.; Bavaro, D.F.; Soysal, P.; et al. Human Monkeypox: A Comprehensive Narrative Review and Analysis of the Public Health Implications. Microorganisms 2022, 10, 1633. [Google Scholar] [CrossRef]
- WHO | World Health Organization. Monkeypox: Key Facts. Available online: https://www.who.int/news-room/fact-sheets/detail/monkeypox (accessed on 22 September 2022).
- Saijo, M.; Ami, Y.; Suzaki, Y.; Nagata, N.; Iwata, N.; Hasegawa, H.; Ogata, M.; Fukushi, S.; Mizutani, T.; Iizuka, I. Diagnosis and assessment of monkeypox virus (MPXV) infection by quantitative PCR assay: Differentiation of Congo Basin and West African MPXV strains. Jpn. J. Infect. Dis. 2008, 61, 140. [Google Scholar] [PubMed]
- Polcz, M.E.; Barbul, A. The Role of Vitamin A in Wound Healing. Nutr. Clin. Pract. 2019, 34, 695–700. [Google Scholar] [CrossRef] [PubMed]
- WHO | World Health Organization. Clinical Management and Infection Prevention and Control for Monkeypox: Interim Rapid Response Guidance, 10 June 2022. Available online: https://www.who.int/publications/i/item/WHO-MPX-Clinical-and-IPC-2022.1 (accessed on 24 June 2022).
- Cono, J.; Cragan, J.D.; Jamieson, D.J.; Rasmussen, S.A. Prophylaxis and treatment of pregnant women for emerging infections and bioterrorism emergencies. Emerg. Infect. Dis. 2006, 12, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Mbala, P.K.; Huggins, J.W.; Riu-Rovira, T.; Ahuka, S.M.; Mulembakani, P.; Rimoin, A.W.; Martin, J.W.; Muyembe, J.T. Maternal and Fetal Outcomes Among Pregnant Women With Human Monkeypox Infection in the Democratic Republic of Congo. J. Infect. Dis. 2017, 216, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Fenner, F.; Henderson, D.A.; Arita, I.; Jezek, Z.; Ladnyi, I.D.; World Health, O. Smallpox and Its Eradication; World Health Organization: Geneva, Switzerland, 1988. [Google Scholar]
- Grosenbach, D.W.; Honeychurch, K.; Rose, E.A.; Chinsangaram, J.; Frimm, A.; Maiti, B.; Lovejoy, C.; Meara, I.; Long, P.; Hruby, D.E. Oral Tecovirimat for the Treatment of Smallpox. N. Engl. J. Med. 2018, 379, 44–53. [Google Scholar] [CrossRef]
- Grosenbach, D.W.; Jordan, R.; Hruby, D.E. Development of the small-molecule antiviral ST-246 as a smallpox therapeutic. Future Virol. 2011, 6, 653–671. [Google Scholar] [CrossRef]
- Yang, G.; Pevear, D.C.; Davies, M.H.; Collett, M.S.; Bailey, T.; Rippen, S.; Barone, L.; Burns, C.; Rhodes, G.; Tohan, S.; et al. An orally bioavailable antipoxvirus compound (ST-246) inhibits extracellular virus formation and protects mice from lethal orthopoxvirus Challenge. J. Virol. 2005, 79, 13139–13149. [Google Scholar] [CrossRef]
- Duraffour, S.; Snoeck, R.; de Vos, R.; van Den Oord, J.J.; Crance, J.M.; Garin, D.; Hruby, D.E.; Jordan, R.; De Clercq, E.; Andrei, G. Activity of the anti-orthopoxvirus compound ST-246 against vaccinia, cowpox and camelpox viruses in cell monolayers and organotypic raft cultures. Antivir Ther. 2007, 12, 1205–1216. [Google Scholar] [CrossRef]
- Smith, S.K.; Olson, V.A.; Karem, K.L.; Jordan, R.; Hruby, D.E.; Damon, I.K. In vitro efficacy of ST246 against smallpox and monkeypox. Antimicrob. Agents Chemother. 2009, 53, 1007–1012. [Google Scholar] [CrossRef]
- Sbrana, E.; Jordan, R.; Hruby, D.E.; Mateo, R.I.; Xiao, S.Y.; Siirin, M.; Newman, P.C.; AP, D.A.R.; Tesh, R.B. Efficacy of the antipoxvirus compound ST-246 for treatment of severe orthopoxvirus infection. Am. J. Trop. Med. Hyg. 2007, 76, 768–773. [Google Scholar] [CrossRef]
- Smith, S.K.; Self, J.; Weiss, S.; Carroll, D.; Braden, Z.; Regnery, R.L.; Davidson, W.; Jordan, R.; Hruby, D.E.; Damon, I.K. Effective antiviral treatment of systemic orthopoxvirus disease: ST-246 treatment of prairie dogs infected with monkeypox virus. J. Virol. 2011, 85, 9176–9187. [Google Scholar] [CrossRef] [PubMed]
- Huggins, J.; Goff, A.; Hensley, L.; Mucker, E.; Shamblin, J.; Wlazlowski, C.; Johnson, W.; Chapman, J.; Larsen, T.; Twenhafel, N.; et al. Nonhuman primates are protected from smallpox virus or monkeypox virus challenges by the antiviral drug ST-246. Antimicrob. Agents Chemother. 2009, 53, 2620–2625. [Google Scholar] [CrossRef] [PubMed]
- Merchlinsky, M.; Albright, A.; Olson, V.; Schiltz, H.; Merkeley, T.; Hughes, C.; Petersen, B.; Challberg, M. The development and approval of tecoviromat (TPOXX(®)), the first antiviral against smallpox. Antivir. Res. 2019, 168, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Jordan, R.; Tien, D.; Bolken, T.C.; Jones, K.F.; Tyavanagimatt, S.R.; Strasser, J.; Frimm, A.; Corrado, M.L.; Strome, P.G.; Hruby, D.E. Single-dose safety and pharmacokinetics of ST-246, a novel orthopoxvirus egress inhibitor. Antimicrob. Agents Chemother. 2008, 52, 1721–1727. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). TPOXX (tecovirimat) Capsules for Oral Use Initial, U.S. Approval: 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/208627s000lbl.pdf (accessed on 22 June 2022).
- European Medicines Agency (EMA). Tecovirimat SIGA. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/tecovirimat-siga (accessed on 24 June 2022).
- Vora, S.; Damon, I.; Fulginiti, V.; Weber, S.G.; Kahana, M.; Stein, S.L.; Gerber, S.I.; Garcia-Houchins, S.; Lederman, E.; Hruby, D.; et al. Severe eczema vaccinatum in a household contact of a smallpox vaccinee. Clin. Infect. Dis. 2008, 46, 1555–1561. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Progressive vaccinia in a military smallpox vaccinee-United States, 2009. MMWR Morb Mortal Wkly Rep. 2009, 58, 532–536. [Google Scholar]
- Lederman, E.R.; Davidson, W.; Groff, H.L.; Smith, S.K.; Warkentien, T.; Li, Y.; Wilkins, K.A.; Karem, K.L.; Akondy, R.S.; Ahmed, R.; et al. Progressive vaccinia: Case description and laboratory-guided therapy with vaccinia immune globulin, ST-246, and CMX001. J. Infect. Dis. 2012, 206, 1372–1385. [Google Scholar] [CrossRef]
- Whitehouse, E.R.; Rao, A.K.; Yu, Y.C.; Yu, P.A.; Griffin, M.; Gorman, S.; Angel, K.A.; McDonald, E.C.; Manlutac, A.L.; de Perio, M.A.; et al. Novel Treatment of a Vaccinia Virus Infection from an Occupational Needlestick-San Diego, California, 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 943–946. [Google Scholar] [CrossRef]
- Wendt, R.; Tittelbach, J.; Schrick, L.; Kellner, N.; Kalbitz, S.; Ruehe, B.; Michel, J.; Schliemann, S.; Elsner, P.; Lübbert, C.; et al. Generalized cowpox virus infection in an immunosuppressed patient. Int. J. Infect. Dis. 2021, 106, 276–278. [Google Scholar] [CrossRef]
- Rao, A.K.; Schulte, J.; Chen, T.H.; Hughes, C.M.; Davidson, W.; Neff, J.M.; Markarian, M.; Delea, K.C.; Wada, S.; Liddell, A.; et al. Monkeypox in a Traveler Returning from Nigeria-Dallas, Texas, July 2021. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 509–516. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). Smallpox Preparedness and Response Updates from FDA. Available online: https://www.fda.gov/emergency-preparedness-and-response/mcm-issues/smallpox-preparedness-and-response-updates-fda (accessed on 20 September 2022).
- Andrei, G.; Snoeck, R. Cidofovir Activity against Poxvirus Infections. Viruses 2010, 2, 2803–2830. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Smith, J.L.; Chen, M.S. Effect of incorporation of cidofovir into DNA by human cytomegalovirus DNA polymerase on DNA elongation. Antimicrob. Agents Chemother. 1997, 41, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Izzedine, H.; Launay-Vacher, V.; Deray, G. Antiviral drug-induced nephrotoxicity. Am. J. Kidney Dis. 2005, 45, 804–817. [Google Scholar] [CrossRef] [PubMed]
- Beadle, J.R. Synthesis of cidofovir and (S)-HPMPA ether lipid prodrugs. Curr Protoc Nucleic Acid Chem 2007, 15, 2–15. [Google Scholar] [CrossRef]
- Wei, H.; Huang, D.; Fortman, J.; Wang, R.; Shao, L.; Chen, Z.W. Coadministration of cidofovir and smallpox vaccine reduced vaccination side effects but interfered with vaccine-elicited immune responses and immunity to monkeypox. J. Virol. 2009, 83, 1115–1125. [Google Scholar] [CrossRef]
- Lanier, R.; Trost, L.; Tippin, T.; Lampert, B.; Robertson, A.; Foster, S.; Rose, M.; Painter, W.; O’Mahony, R.; Almond, M.; et al. Development of CMX001 for the Treatment of Poxvirus Infections. Viruses 2010, 2, 2740–2762. [Google Scholar] [CrossRef]
- Tippin, T.K.; Morrison, M.E.; Brundage, T.M.; Momméja-Marin, H. Brincidofovir Is Not a Substrate for the Human Organic Anion Transporter 1: A Mechanistic Explanation for the Lack of Nephrotoxicity Observed in Clinical Studies. Ther. Drug Monit. 2016, 38, 777–786. [Google Scholar] [CrossRef]
- Chan-Tack, K.; Harrington, P.; Bensman, T.; Choi, S.Y.; Donaldson, E.; O’Rear, J.; McMillan, D.; Myers, L.; Seaton, M.; Ghantous, H.; et al. Benefit-risk assessment for brincidofovir for the treatment of smallpox: U.S. Food and Drug Administration’s Evaluation. Antivir. Res. 2021, 195, 105182. [Google Scholar] [CrossRef]
- Trost, L.C.; Rose, M.L.; Khouri, J.; Keilholz, L.; Long, J.; Godin, S.J.; Foster, S.A. The efficacy and pharmacokinetics of brincidofovir for the treatment of lethal rabbitpox virus infection: A model of smallpox disease. Antivir. Res. 2015, 117, 115–121. [Google Scholar] [CrossRef]
- Grossi, I.M.; Foster, S.A.; Gainey, M.R.; Krile, R.T.; Dunn, J.A.; Brundage, T.; Khouri, J.M. Efficacy of delayed brincidofovir treatment against a lethal rabbitpox virus challenge in New Zealand White rabbits. Antivir. Res. 2017, 143, 278–286. [Google Scholar] [CrossRef]
- Hutson, C.L.; Kondas, A.V.; Mauldin, M.R.; Doty, J.B.; Grossi, I.M.; Morgan, C.N.; Ostergaard, S.D.; Hughes, C.M.; Nakazawa, Y.; Kling, C.; et al. Pharmacokinetics and Efficacy of a Potential Smallpox Therapeutic, Brincidofovir, in a Lethal Monkeypox Virus Animal Model. mSphere 2021, 6, e00927-20. [Google Scholar] [CrossRef] [PubMed]
- Grimley, M.S.; Chemaly, R.F.; Englund, J.A.; Kurtzberg, J.; Chittick, G.; Brundage, T.M.; Bae, A.; Morrison, M.E.; Prasad, V.K. Brincidofovir for Asymptomatic Adenovirus Viremia in Pediatric and Adult Allogeneic Hematopoietic Cell Transplant Recipients: A Randomized Placebo-Controlled Phase II Trial. Biol. Blood Marrow Transpl. 2017, 23, 512–521. [Google Scholar] [CrossRef] [PubMed]
- Marty, F.M.; Winston, D.J.; Chemaly, R.F.; Mullane, K.M.; Shore, T.B.; Papanicolaou, G.A.; Chittick, G.; Brundage, T.M.; Wilson, C.; Morrison, M.E.; et al. A Randomized, Double-Blind, Placebo-Controlled Phase 3 Trial of Oral Brincidofovir for Cytomegalovirus Prophylaxis in Allogeneic Hematopoietic Cell Transplantation. Biol. Blood Marrow Transpl. 2019, 25, 369–381. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration (FDA). Food and Drug Administration, FDA Approves Drug to Treat Smallpox. Available online: https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-drug-treat-smallpox (accessed on 24 June 2022).
- U.S. Food and Drug Administration (FDA). TEMBEXA (Brincidofovir) Tablets, for Oral Use. TEMBEXA (Brincidofovir) Oral Suspension. Available online: https://www.chimerix.com/wp-content/uploads/2021/06/TEMBEXA-USPI-and-PPI-04June2021.pdf (accessed on 24 June 2022).
- Quenelle, D.C.; Prichard, M.N.; Keith, K.A.; Hruby, D.E.; Jordan, R.; Painter, G.R.; Robertson, A.; Kern, E.R. Synergistic efficacy of the combination of ST-246 with CMX001 against orthopoxviruses. Antimicrob. Agents Chemother. 2007, 51, 4118–4124. [Google Scholar] [CrossRef]
- Mazurkov, O.Y.; Shishkina, L.N.; Bormotov, N.I.; Skarnovich, M.O.; Serova, O.A.; Mazurkova, N.A.; Chernonosov, A.A.; Tikhonov, A.Y.; Selivanov, B.A. Estimation of Absolute Bioavailability of the Chemical Substance of the Anti-Smallpox Preparation NIOCH-14 in Mice. Bull. Exp. Biol. Med. 2020, 170, 207–210. [Google Scholar] [CrossRef]
- Kempe, C.H. Studies smallpox and complications of smallpox vaccination. Pediatrics 1960, 26, 176–189. [Google Scholar] [CrossRef]
- Hopkins, R.J.; Kramer, W.G.; Blackwelder, W.C.; Ashtekar, M.; Hague, L.; Winker-La Roche, S.D.; Berezuk, G.; Smith, D.; Leese, P.T. Safety and pharmacokinetic evaluation of intravenous vaccinia immune globulin in healthy volunteers. Clin. Infect. Dis. 2004, 39, 759–766. [Google Scholar] [CrossRef]
- Kesson, A.M.; Ferguson, J.K.; Rawlinson, W.D.; Cunningham, A.L. Progressive vaccinia treated with ribavirin and vaccinia immune globulin. Clin. Infect. Dis. 1997, 25, 911–914. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Monitoring and Risk Assessment for Persons Exposed in the Community. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/monitoring.html (accessed on 21 June 2022).
- Ježek, Z.; Szczeniowski, M.; Paluku, K.; Mutombo, M. Human monkeypox: Clinical features of 282 patients. J. Infect. Dis. 1987, 156, 293–298. [Google Scholar] [CrossRef]
- Mahase, E. Monkeypox: What do we know about the outbreaks in Europe and North America? BMJ 2022, 377, o1274. [Google Scholar] [CrossRef]
- Antinori, A.; Mazzotta, V.; Vita, S.; Carletti, F.; Tacconi, D.; Lapini, L.E.; D’Abramo, A.; Cicalini, S.; Lapa, D.; Pittalis, S.; et al. Epidemiological, clinical and virological characteristics of four cases of monkeypox support transmission through sexual contact, Italy, May 2022. Eur. Surveill. 2022, 27, 2200421. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Disinfecting Home and Other Non-Healthcare Settings. Available online: https://www.cdc.gov/poxvirus/monkeypox/if-sick/home-disinfection.html (accessed on 21 June 2022).
- Khalil, A.; Samara, A.; O’Brien, P.; Morris, E.; Draycott, T.; Lees, C.; Ladhani, S. Monkeypox and pregnancy: What do obstetricians need to know? Ultrasound Obstet. Gynecol. 2022, 60, 22–27. [Google Scholar] [CrossRef]
- Alsanafi, M.; Al-Mahzoum, K.; Sallam, M. Monkeypox Knowledge and Confidence in Diagnosis and Management with Evaluation of Emerging Virus Infection Conspiracies among Health Professionals in Kuwait. Pathogens 2022, 11, 994. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Setiawan, A.M.; Yufika, A.; Anwar, S.; Wahyuni, S.; Asrizal, F.W.; Sufri, M.R.; Putra, R.P.; Wijayanti, N.P.; Salwiyadi, S.; et al. Knowledge of human monkeypox viral infection among general practitioners: A cross-sectional study in Indonesia. Pathogens Glob. Health 2020, 114, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Riccò, M.; Ferraro, P.; Camisa, V.; Satta, E.; Zaniboni, A.; Ranzieri, S.; Baldassarre, A.; Zaffina, S.; Marchesi, F. When a Neglected Tropical Disease Goes Global: Knowledge, Attitudes and Practices of Italian Physicians towards Monkeypox, Preliminary Results. Trop. Med. Infect. Dis. 2022, 7, 135. [Google Scholar] [CrossRef]
- Sallam, M.; Al-Mahzoum, K.; Dardas, L.A.; Al-Tammemi, A.a.B.; Al-Majali, L.; Al-Naimat, H.; Jardaneh, L.; AlHadidi, F.; Al-Salahat, K.; Al-Ajlouni, E.; et al. Knowledge of Human Monkeypox and Its Relation to Conspiracy Beliefs among Students in Jordanian Health Schools: Filling the Knowledge Gap on Emerging Zoonotic Viruses. Medicina 2022, 58, 924. [Google Scholar] [CrossRef]
- Harapan, H.; Setiawan, A.M.; Yufika, A.; Anwar, S.; Wahyuni, S.; Asrizal, F.W.; Sufri, M.R.; Putra, R.P.; Wijayanti, N.P.; Salwiyadi, S.; et al. Confidence in managing human monkeypox cases in Asia: A cross-sectional survey among general practitioners in Indonesia. Acta Trop. 2020, 206, 105450. [Google Scholar] [CrossRef]
- Sallam, M.; Al-Mahzoum, K.; Al-Tammemi, A.a.; Alkurtas, M.; Mirzaei, F.; Kareem, N.; Al-Naimat, H.; Jardaneh, L.; Al-Majali, L.; AlHadidi, A.; et al. Assessing Healthcare Workers’ Knowledge and Their Confidence in the Diagnosis and Management of Human Monkeypox: A Cross-Sectional Study in a Middle Eastern Country. Healthcare 2022, 10, 1722. [Google Scholar] [CrossRef]
- Henderson, D.A. Surveillance of smallpox. Int. J. Epidemiol. 1976, 5, 19–28. [Google Scholar] [CrossRef]
- WHO | World Health Organization. Post–Eradication. Available online: https://www.who.int/news-room/feature-stories/detail/post-eradication (accessed on 20 September 2022).
- Rao, A.K.; Petersen, B.W.; Whitehill, F.; Razeq, J.H.; Isaacs, S.N.; Merchlinsky, M.J.; Campos-Outcalt, D.; Morgan, R.L.; Damon, I.; Sánchez, P.J.; et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices-United States, 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 734–742. [Google Scholar] [CrossRef]
- Yen, C.; Hyde, T.B.; Costa, A.J.; Fernandez, K.; Tam, J.S.; Hugonnet, S.; Huvos, A.M.; Duclos, P.; Dietz, V.J.; Burkholder, B.T. The development of global vaccine stockpiles. Lancet. Infect. Dis. 2015, 15, 340–347. [Google Scholar] [CrossRef]
- Nalca, A.; Zumbrun, E.E. ACAM2000: The new smallpox vaccine for United States Strategic National Stockpile. Drug Des Devel Ther. 2010, 4, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.W.; Harms, T.J.; Reynolds, M.G.; Harrison, L.H. Use of Vaccinia Virus Smallpox Vaccine in Laboratory and Health Care Personnel at Risk for Occupational Exposure to Orthopoxviruses-Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2015. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 257–262. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). ACAM2000 Package Insert. Available online: https://www.fda.gov/media/75792/download (accessed on 20 September 2022).
- Hatch, G.J.; Graham, V.A.; Bewley, K.R.; Tree, J.A.; Dennis, M.; Taylor, I.; Funnell, S.G.; Bate, S.R.; Steeds, K.; Tipton, T.; et al. Assessment of the protective effect of Imvamune and Acam2000 vaccines against aerosolized monkeypox virus in cynomolgus macaques. J. Virol. 2013, 87, 7805–7815. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.T.; Berhanu, A.; Bigger, C.B.; Prigge, J.; Silvera, P.M.; Grosenbach, D.W.; Hruby, D. Co-administration of tecovirimat and ACAM2000™ in non-human primates: Effect of tecovirimat treatment on ACAM2000 immunogenicity and efficacy versus lethal monkeypox virus challenge. Vaccine 2020, 38, 644–654. [Google Scholar] [CrossRef]
- Keckler, M.S.; Salzer, J.S.; Patel, N.; Townsend, M.B.; Nakazawa, Y.J.; Doty, J.B.; Gallardo-Romero, N.F.; Satheshkumar, P.S.; Carroll, D.S.; Karem, K.L.; et al. IMVAMUNE(®) and ACAM2000(®) Provide Different Protection against Disease When Administered Postexposure in an Intranasal Monkeypox Challenge Prairie Dog Model. Vaccines 2020, 8, 396. [Google Scholar] [CrossRef]
- Artenstein, A.W.; Johnson, C.; Marbury, T.C.; Morrison, D.; Blum, P.S.; Kemp, T.; Nichols, R.; Balser, J.P.; Currie, M.; Monath, T.P. A novel, cell culture-derived smallpox vaccine in vaccinia-naive adults. Vaccine 2005, 23, 3301–3309. [Google Scholar] [CrossRef]
- Cassimatis, D.C.; Atwood, J.E.; Engler, R.M.; Linz, P.E.; Grabenstein, J.D.; Vernalis, M.N. Smallpox vaccination and myopericarditis: A clinical review. J. Am. Coll. Cardiol. 2004, 43, 1503–1510. [Google Scholar] [CrossRef]
- Halsell, J.S.; Riddle, J.R.; Atwood, J.E.; Gardner, P.; Shope, R.; Poland, G.A.; Gray, G.C.; Ostroff, S.; Eckart, R.E.; Hospenthal, D.R.; et al. Myopericarditis following smallpox vaccination among vaccinia-naive US military personnel. JAMA 2003, 289, 3283–3289. [Google Scholar] [CrossRef]
- Cono, J.; Casey, C.G.; Bell, D.M. Smallpox vaccination and adverse reactions. Guidance for clinicians. MMWR Morb. Mortal. Wkly. Rep. 2003, 52, 1–28. [Google Scholar]
- Earl, P.L.; Americo, J.L.; Wyatt, L.S.; Eller, L.A.; Whitbeck, J.C.; Cohen, G.H.; Eisenberg, R.J.; Hartmann, C.J.; Jackson, D.L.; Kulesh, D.A.; et al. Immunogenicity of a highly attenuated MVA smallpox vaccine and protection against monkeypox. Nature 2004, 428, 182–185. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration (FDA). JYNNEOS Package Insert. Available online: https://www.fda.gov/media/131078/download (accessed on 20 September 2022).
- Stittelaar, K.J.; van Amerongen, G.; Kondova, I.; Kuiken, T.; van Lavieren, R.F.; Pistoor, F.H.; Niesters, H.G.; van Doornum, G.; van der Zeijst, B.A.; Mateo, L.; et al. Modified vaccinia virus Ankara protects macaques against respiratory challenge with monkeypox virus. J. Virol. 2005, 79, 7845–7851. [Google Scholar] [CrossRef] [PubMed]
- Keckler, M.S.; Carroll, D.S.; Gallardo-Romero, N.F.; Lash, R.R.; Salzer, J.S.; Weiss, S.L.; Patel, N.; Clemmons, C.J.; Smith, S.K.; Hutson, C.L.; et al. Establishment of the black-tailed prairie dog (Cynomys ludovicianus) as a novel animal model for comparing smallpox vaccines administered preexposure in both high- and low-dose monkeypox virus challenges. J. Virol. 2011, 85, 7683–7698. [Google Scholar] [CrossRef] [PubMed]
- Frey, S.E.; Winokur, P.L.; Salata, R.A.; El-Kamary, S.S.; Turley, C.B.; Walter, E.B., Jr.; Hay, C.M.; Newman, F.K.; Hill, H.R.; Zhang, Y.; et al. Safety and immunogenicity of IMVAMUNE® smallpox vaccine using different strategies for a post event scenario. Vaccine 2013, 31, 3025–3033. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, R.N.; Hay, C.M.; Stapleton, J.T.; Marbury, T.C.; Wagner, E.; Kreitmeir, E.; Röesch, S.; von Krempelhuber, A.; Young, P.; Nichols, R.; et al. A Randomized, Double-Blind, Placebo-Controlled Phase II Trial Investigating the Safety and Immunogenicity of Modified Vaccinia Ankara Smallpox Vaccine (MVA-BN®) in 56-80-Year-Old Subjects. PLoS ONE 2016, 11, e0157335. [Google Scholar] [CrossRef]
- Von Krempelhuber, A.; Vollmar, J.; Pokorny, R.; Rapp, P.; Wulff, N.; Petzold, B.; Handley, A.; Mateo, L.; Siersbol, H.; Kollaritsch, H.; et al. A randomized, double-blind, dose-finding Phase II study to evaluate immunogenicity and safety of the third generation smallpox vaccine candidate IMVAMUNE. Vaccine 2010, 28, 1209–1216. [Google Scholar] [CrossRef]
- Walsh, S.R.; Wilck, M.B.; Dominguez, D.J.; Zablowsky, E.; Bajimaya, S.; Gagne, L.S.; Verrill, K.A.; Kleinjan, J.A.; Patel, A.; Zhang, Y.; et al. Safety and immunogenicity of modified vaccinia Ankara in hematopoietic stem cell transplant recipients: A randomized, controlled trial. J. Infect. Dis. 2013, 207, 1888–1897. [Google Scholar] [CrossRef]
- Von Sonnenburg, F.; Perona, P.; Darsow, U.; Ring, J.; von Krempelhuber, A.; Vollmar, J.; Roesch, S.; Baedeker, N.; Kollaritsch, H.; Chaplin, P. Safety and immunogenicity of modified vaccinia Ankara as a smallpox vaccine in people with atopic dermatitis. Vaccine 2014, 32, 5696–5702. [Google Scholar] [CrossRef]
- Overton, E.T.; Stapleton, J.; Frank, I.; Hassler, S.; Goepfert, P.A.; Barker, D.; Wagner, E.; von Krempelhuber, A.; Virgin, G.; Meyer, T.P.; et al. Safety and Immunogenicity of Modified Vaccinia Ankara-Bavarian Nordic Smallpox Vaccine in Vaccinia-Naive and Experienced Human Immunodeficiency Virus-Infected Individuals: An Open-Label, Controlled Clinical Phase II Trial. Open Forum Infect. Dis. 2015, 2, ofv040. [Google Scholar] [CrossRef]
- Overton, E.T.; Lawrence, S.J.; Wagner, E.; Nopora, K.; Rösch, S.; Young, P.; Schmidt, D.; Kreusel, C.; De Carli, S.; Meyer, T.P.; et al. Immunogenicity and safety of three consecutive production lots of the non replicating smallpox vaccine MVA: A randomised, double blind, placebo controlled phase III trial. PLoS ONE 2018, 13, e0195897. [Google Scholar] [CrossRef]
- Kozlov, M. Monkeypox Goes Global: Why Scientists Are on Alert. Available online: https://www.nature.com/articles/d41586-022-01421-8 (accessed on 11 June 2022).
- Vivancos, R.; Anderson, C.; Blomquist, P.; Balasegaram, S.; Bell, A.; Bishop, L.; Brown, C.S.; Chow, Y.; Edeghere, O.; Florence, I.; et al. Community transmission of monkeypox in the United Kingdom, April to May 2022. Eurosurveillance 2022, 27, 2200422. [Google Scholar] [CrossRef] [PubMed]
- Bragazzi, N.L.; Khamisy-Farah, R.; Tsigalou, C.; Mahroum, N.; Converti, M. Attaching a stigma to the LGBTQI+ community should be avoided during the monkeypox epidemic. J. Med. Virol. 2022; Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, M. Monkeypox Outbreaks: 4 Key Questions Researchers Have. Available online: https://www.nature.com/articles/d41586-022-01493-6 (accessed on 11 June 2022).
- Velavan, T.P.; Meyer, C.G. Monkeypox 2022 outbreak: An update. Trop. Med. Int. Health 2022, 27, 604–605. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Risk Assessment: Monkeypox Multi-Country Outbreak. Available online: https://www.ecdc.europa.eu/en/publications-data/risk-assessment-monkeypox-multi-country-outbreak (accessed on 11 June 2022).
- Van Seventer, J.M.; Hochberg, N.S. Principles of Infectious Diseases: Transmission, Diagnosis, Prevention, and Control. Int. Encycl. Pub. Health 2017, 22–39. [Google Scholar] [CrossRef]

| Publication | Huhn et al. [61] | Pittman et al. [62] | Adler et al. [63] | Yinka-Ogunleye et al. [64] |
|---|---|---|---|---|
| Country | U.S. | Democratic Republic of the Congo | U.K. | Nigeria |
| Number of patients | 37 | 216 | 7 | 122 |
| Fever | 87% | 18.5% | 42% | 79% |
| Rash | 97% | 99.5% | 100% | 88% |
| Malaise | - | 85.2% | - | 50% |
| Myalgia | 56% | 6.9% | - | 58% |
| Chill | 71% | 44.9% | - | 65% |
| Adenopathy | 71% | 57.4% | 71% | 69% |
| Headache | 65% | 23.6% | - | 79% |
| Sore throat | 60% | 78.2% | - | 58% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harapan, H.; Ophinni, Y.; Megawati, D.; Frediansyah, A.; Mamada, S.S.; Salampe, M.; Bin Emran, T.; Winardi, W.; Fathima, R.; Sirinam, S.; et al. Monkeypox: A Comprehensive Review. Viruses 2022, 14, 2155. https://doi.org/10.3390/v14102155
Harapan H, Ophinni Y, Megawati D, Frediansyah A, Mamada SS, Salampe M, Bin Emran T, Winardi W, Fathima R, Sirinam S, et al. Monkeypox: A Comprehensive Review. Viruses. 2022; 14(10):2155. https://doi.org/10.3390/v14102155
Chicago/Turabian StyleHarapan, Harapan, Youdiil Ophinni, Dewi Megawati, Andri Frediansyah, Sukamto S. Mamada, Mirnawati Salampe, Talha Bin Emran, Wira Winardi, Raisha Fathima, Salin Sirinam, and et al. 2022. "Monkeypox: A Comprehensive Review" Viruses 14, no. 10: 2155. https://doi.org/10.3390/v14102155
APA StyleHarapan, H., Ophinni, Y., Megawati, D., Frediansyah, A., Mamada, S. S., Salampe, M., Bin Emran, T., Winardi, W., Fathima, R., Sirinam, S., Sittikul, P., Stoian, A. M., Nainu, F., & Sallam, M. (2022). Monkeypox: A Comprehensive Review. Viruses, 14(10), 2155. https://doi.org/10.3390/v14102155










