Abstract
Human immunodeficiency virus (HIV) is associated with neuroendocrine dysfunction which may contribute to co-morbid stress-sensitive disorders. The hypothalamic-pituitary-adrenal (HPA) or -gonadal (HPG) axes are perturbed in up to 50% of HIV patients. The mechanisms are not known, but we have found the HIV-1 trans-activator of transcription (Tat) protein to recapitulate the clinical phenotype in male mice. We hypothesized that HPA and/or HPG dysregulation contributes to Tat-mediated interactions with oxycodone, an opioid often prescribed to HIV patients, in females. Female mice that conditionally-expressed the Tat1–86 protein [Tat(+) mice] or their counterparts that did not [Tat(−) control mice] were exposed to forced swim stress (or not) and behaviorally-assessed for motor and anxiety-like behavior. Some mice had glucocorticoid receptors (GR) or corticotropin-releasing factor receptors (CRF-R) pharmacologically inhibited. Some mice were ovariectomized (OVX). As seen previously in males, Tat elevated basal corticosterone levels and potentiated oxycodone’s psychomotor activity in females. Unlike males, females did not demonstrate adrenal insufficiency and oxycodone potentiation was not regulated by GRs or CRF-Rs. Rather OVX attenuated Tat/oxycodone interactions. Either Tat or oxycodone increased anxiety-like behavior and their combination increased hypothalamic allopregnanolone. OVX increased basal hypothalamic allopregnanolone and obviated Tat or oxycodone-mediated fluctuations. Together, these data provide further evidence for Tat-mediated dysregulation of the HPA axis and reveal the importance of HPG axis regulation in females. HPA/HPG disruption may contribute vulnerability to affective and substance use disorders.
1. Introduction
Human immunodeficiency virus type 1 (HIV-1) infection remains a significant public health concern with ~1.1 million people living with HIV in the U.S. The widespread use of combined antiretroviral therapeutics (cART) has largely increased life expectancy among HIV-infected patients; however, the inability of cART to target latent central nervous system (CNS) reservoirs including microglia and astrocytes [1], likely contributes to the persistence of central viremia and neurological symptomatology [2,3]. Approximately 50% of the HIV+ population contend with a constellation of neurological symptoms (collectively referred to as “neuroHIV”) that include affective disorders (i.e., increase generalized anxiety and major depression), psychomotor deficits and cognitive impairments [4,5,6]. CNS complications also include neuropathic pain resulting in 8–52% of HIV-1 patients being prescribed opioids [7,8,9,10,11,12] which may interact with HIV-1 proteins, complicating outcomes. Thus, despite progress attenuating systemic viremia, the central compartment remains an important target for therapeutic advancement.
While the mechanisms that underlie neuroHIV remain the subject of intense investigation, neurotoxic HIV-1 proteins can persist within in the CNS where cART is poorly retained (indicated by the presence of viral proteins in cerebrospinal fluid [13,14]). In particular, the HIV-1 trans-activator of transcription (Tat) is critical for efficient viral transcription [15] but can also exert neural damage via multiple mechanisms including the promotion of neuroinflammation [16,17], direct excitotoxic injury to neurons [18,19,20] and the promotion of mitochondrial dysregulation and mitotoxicity [18,21,22,23]. In particular, disruption of mitochondrial activity in the CNS may contribute to neuroendocrine dysfunction observed in HIV+ patients given that mitochondria are the rate-limiting organelle necessary for steroidogenesis.
While under-investigated, up to half of HIV-1 patients suffer from neuroendocrine dysfunction that is secondary in etiology (i.e., occurring at the level of the CNS, not the primary steroid-producing glands). HPA dysfunction (elevated basal cortisol levels and paradoxical adrenal insufficiency in response to a stressor) is reported in 14–46% of HIV-1 patient samples [24,25,26,27,28]. Hypogonadism (reduced testosterone production in men and dysregulated estradiol/progestogen levels in women) is reported in 10–50% of HIV-1 samples [29,30,31,32,33,34,35,36]. Perturbation of the hypothalamic-pituitary-adrenal (HPA) or -gonadal (HPG) neuroendocrine axes would be expected to contribute to neuroHIV symptomatology and/or vulnerability. The HPA axis plays a critical role in the recovery from a variety of stressors including immune, psychological or drug of abuse challenges [37,38,39,40]. Prolonged HPA/HPG dysfunction may promote vulnerability to stress-sensitive neuropsychiatric disorders and addiction [41]. Using a transgenic mouse model, we have found conditional expression of HIV-1 Tat in males to disrupt CNS steroidogenesis [23], to elevate basal production of the circulating stress hormone, corticosterone, and to produce adrenal insufficiency in response to a physical or pharmacological HPA challenge [42]. Importantly, HPA dysfunction was observed concurrent with an increased psychomotor response to the clinical opioid, oxycodone [42]. However, the interactions with endocrine function in females may be more complex and is less well-understood. We have observed elevated circulating corticosterone and hypothalamic corticotropin releasing factor (CRF) in Tat- or oxycodone-exposed female mice to be indicative of HPA activation [43]. However, it was not known whether females expressed the adrenally insufficient profile observed in males, what central targets may contribute to these effects, or how their greater estrane/pregnane gonadal steroid capacity would influence outcomes. As such, these potential sex differences in Tat-mediated HPA dysfunction were sought in the present work.
We hypothesized that conditional expression of HIV-1 Tat in female mice would produce HPA dysregulation, potentiate oxycodone’s psychomotor effects and promote anxiety-like behavior. We anticipated these effects to be partly mediated by negative regulators of the HPA axis, glucocorticoid receptors (GR) and/or CRF receptors (CRF-R), as was previously observed in males [42]. We further anticipated that peripherally- or centrally-produced steroids would modify the HPA response.
2. Materials and Methods
The Institutional Animal Care and Use Committee (IACUC) at University of Mississippi preapproved the procedures and protocols for the present study (#18-004 & 21-005; approved on 25 October 2017 & 28 September 2020 respectively). All the experiments were performed in accordance with guidelines defined by the National Institute of Health (NIH; Publication No. 85-23).
2.1. Subjects & Housing
Female, adult, HIV-1 Tat-transgenic mice (n = 313) were bred in the vivarium at the University of Mississippi (Oxford, MS, USA). Mice were housed (2–5/cage) and maintained in a humidity and temperature-controlled environment on a 12:12 h reverse light:dark cycle (lights off at 09:00 h) with ad libitum access to food and water. Briefly, mice expressed the Tat1–86 protein via conditional, GFAP-relegated expression under regulation of a reverse tetracycline-controlled transactivating (rtTA) transcription factor. We and others have previously confirmed mRNA expression of tat in the brain and spinal cord of these mice [44,45,46] and include confirmation in the present tissues via quantitative real-time PCR (see Supplemental Data, Figure S1). Tat(−) control mice lacked the tat transgene but did express the rtTA transcription factor [47]. While rtTA gene expression is notably ‘leaky’ [48], Tat(−) controls lack the tat transgene making them an optimal comparison group. Moreover, we have previously demonstrated that young-adult Tat(+) mice do not demonstrate tat-mediated behavioral interactions with drugs of abuse until doxycycline-induction [23] and include a saline-administered, un-induced control group herein (see Supplemental Data, Figure S2).
2.2. Surgical Manipulation
A subset of mice (n = 32) underwent bilateral ovariectomy under isoflurane anesthesia (2.5–4.0%) per previous methods [49]. Post-surgery, mice were transferred to a clean cage with food, water, and access to acetaminophen (2 mg/mL) for a period of 96 h. Weight, neurological status, surgical site, and food consumption were monitored daily [50]. Two mice failed to recover and were thus excluded from the study. Mice were allowed 7 days of recovery to allow for endogenous hormone washout prior to additional pharmacological manipulations.
2.3. Chemicals
Doxycycline hyclate (Cayman Chemical, Ann Arbor, MI, USA) was made fresh daily to a dose of 30 mg/kg in sterile saline (0.9% w/v) and injected via intraperitoneal (i.p.) route once daily for 5 days. Oxycodone HCl (Sigma-Aldrich, St. Louis, MO, USA) was dissolved to a concentration of 3.0 mg/kg in sterile saline and injected i.p. 15 min prior to behavioral testing (see Figure 1A). The present dose of oxycodone (3.0 mg/kg) was identified as the minimum effective dose to diverge psychomotor responding between Tat(−) and Tat(+) mice [43]. To pharmacologically block CRF-receptors, mice were administered antalarmin (Cayman Chemical) dissolved in 30% Kolliphor (70% sterile saline) to a dose of 20.0 mg/kg (i.p) administered once daily for 7 days [42,51,52]. To block glucocorticoid receptors, mice were administered RU-486 (a.k.a. mifepristone; Cayman Chemical) dissolved in 30% Kolliphor, 1% DMSO and Tween 20 (3 drops) to a dose of 20 mg/kg (i.p.) administered once daily for 8 days [42,53,54].
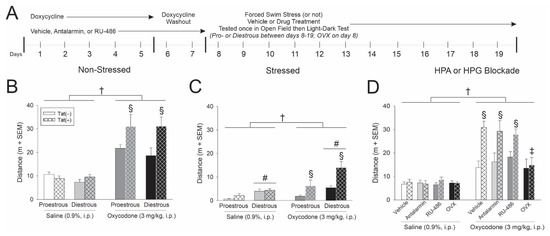
Figure 1.
(A) HIV-1 Tat-transgenic, female mice [hatched bars; Tat(+)] or control counterparts [open bars; Tat(−) controls] had transgene expression induced via doxycycline (30 mg/kg, i.p., once daily for 5 days with 2 days for washout; n = 8–10/group). Some mice were pretreated with either vehicle. antalarmin, RU-486 or were ovariectomized (OVX). On the day of testing, proestrous, diestrous, or OVX mice were exposed to a forced swim stress (or not) and administered saline or oxycodone 15 min prior to assessment in an open field and light-dark transition test. (B) Distance (m) travelled in an open field among (B) non-stressed mice, (C) stressed mice and (D) mice that were pretreated with antalarmin or RU-486 or were OVX. † indicates a main effect for oxycodone-administered mice to differ from saline-administered controls. § indicates an interaction wherein oxycodone-administered Tat(+) mice differ from respective Tat(−) controls and saline-administered controls. # indicates a main effect of estrous cycle wherein diestrous mice differ from proestrous mice. ‡ indicates an interaction wherein oxycodone-administered Tat(+) OVX mice differ from all other oxycodone- administered Tat(+) groups, p < 0.05.
2.4. Determination of Estrous Cycle Phase
Estrous cycle was determined via vaginal lavage conducted daily at 09:00 h. Cytology was assessed to determine the estrous cycle phase as modified from previous descriptions [43,55]. Mice were tested when in the proestrous or diestrous phase of their estrous cycle in order to control for hormonal variations that might influence behavioral responses. In the current study, no significant differences were observed between mice on diestrus I (a.k.a. metestrus) or diestrus II; as such, these groups were combined.
2.5. Behavioral Assays
2.5.1. Forced Swim Stress
To activate the HPA stress axis, mice were exposed to a forced swim stimulus as previously described [42]. Briefly, mice were placed in a container filled with room temperature water (approximately 22 °C) and permitted to swim for 15 min. At the end of the swim stressor, mice were dried and returned to their home cages. Following swim stress mice were assessed in a behavioral battery comprised of an open field and a light-dark transition test (described below).
2.5.2. Open Field Test
The open field test was used to assess spontaneous motor behavior as previously described [42,43]. Briefly, mice were placed gently in a corner of a square-shaped open field box (40 × 40 × 35 cm3; Stoelting Co., Wood Dale, IL, USA) with a brightly-lit center (inner 20 × 20 cm2) and allowed to behave for 5 min. Dependent measures included overall distance travelled (in meters) and mean velocity (m/s) as indices of spontaneous motor behavior.
2.5.3. Light-Dark Transition Test
Following testing in the open field, mice were assessed for anxiety-like behavior in the light-dark transition test. Given the psychostimulatory effects of opioids, the light-dark test was preferred given that anxiety-like indices are less confounded by manipulations that affect motor behavior [56]. The light-dark apparatus consisted of two compartments, one brightly-lit (20 × 20 × 35 cm3) and the other dark and enclosed (20 × 20 × 35 cm3; Stoelting Co., Wood Dale, IL, USA). Briefly, mice were placed in a corner of the brightly-lit side of the apparatus and permitted to behave for 5 min. The latency to enter the dark chamber and the total time spent in the light zone were considered indices of anti-anxiety-like behavior.
2.6. Biochemical Assays
2.6.1. Tissue Collection
Following behavior testing, mice underwent cervical dislocation followed by rapid decapitation. Whole brains and trunk blood were collected. Brains were immediately flash-frozen on dry ice. Blood was collected in a chilled aliquot tube and serum was separated via centrifugation at 13,500× g. Tissues and serum were stored at −80 °C. Hypothalamus was later grossly dissected on ice as previously described [43] and frozen at −80 °C until assay.
2.6.2. Enzyme-Linked Immunosorbant Assay (ELISA)
Serum steroid extraction was achieved via ether-snap freezing. Briefly, 1 mL of anhydrous ethyl ether (ice-cold) was added to serum samples which were vortexed and snap frozen using a dry ice/acetone mixture as described previously [43]. Ether supernatant was collected and evaporated to dryness overnight in a chemical fume hood. Crystalline steroids were reconstituted 5× (for estradiol), 25× (for progesterone) or 50× (for corticosterone) their initial volume in assay extraction buffer (Neogen Life Sciences, Lexington, KY, USA).
Circulating levels of estradiol, progesterone and corticosterone were measured via ELISA per manufacturer-suggested methods (#402110, Estradiol; #402310, Progesterone; #402810, Corticosterone; Neogen Life Sciences) and as previously described [42,43]. All assays were read at an absorbance of 650 nm using a CLARIOstar microplate reader (BMG Labtech Inc., Cary, NC, USA). Respective intra-assay variances were: Estradiol: 9%, Progesterone: 8.3% and Corticosterone: 6.1%; inter-assay variances were: Estradiol: 26.7%, Progesterone: 29.1% and Corticosterone: 21.9%.
2.6.3. Ultra-Performance Liquid Chromatography (UPLC)-Mass Spectrometry (MS)
For UPLC-MS/MS, charcoal-stripped tissue (brain tissue derived from Tat-tg mice) was utilized to prepare both the calibration curve and quality control samples for analysis. A simple protein precipitation method was used for steroid extraction. Samples were homogenized (100 μL of PBS pH 7.4) and precipitated with 100 µL of acetonitrile followed by vortexing (2 min) and centrifugation (10 min at 14,000 rpm). After centrifugation, the supernatant solution was mixed with 50 μL of derivatizing solution (20 mg/mL of 2-hydrazinopyridine solution prepared in 0.5% trifluoroacetic acid ethanol solution) and incubated at 60 °C for 1 h. Following incubation, 20 μL of the internal standard solution (1 μg/mL) was added and vortex mixed. For sample analysis, aliquots of 2 μL were injected onto the UPLC-MS/MS instrument.
2.7. Procedure
All manipulations (apart from genotype and cycle phase) were randomly assigned. HIV-1 Tat was conditionally induced in female adult transgenic mice via doxycycline administration for 5 days (i.p.). Given that the anti-inflammatory effects of doxycycline could potentially mask some Tat-mediated effects, two days of doxycycline washout were included prior to testing [57]. Estrous cycle was determined daily, and mice were assessed when in the proestrous or diestrous phases. Given that effects of Tat are found to be stable for at least 3 weeks [45,58], mice were behaviorally assessed no more than 14 days from Tat induction (Figure 1A,B). Some mice underwent forced swim stress for 15 min (stressed paradigm) or not (non-stressed paradigm). Some mice were pretreated with vehicle, the CRF-R antagonist, antalarmin, or the GR antagonist, RU-486. Some mice were ovariectomized to remove the primary source of gonadal hormones and administered a daily vehicle injection (to account for potential injection stress). All mice received either acute oxycodone or vehicle saline 15 min prior to assessment in the open field and light-dark transition tests. Notably, mice that received vehicle, antalarmin, or RU-486 were administered a final dose 30 min prior to behavioral testing. To assess the restoration of the HPA axis response, all mice were sacrificed approximately 2 h after the initiation of behavior testing (a time when circulating stress steroids are resolving) as opposed to prior observations that were conducted at 30 min post-testing (a time of peak stress steroid response [42]).
2.8. Statistical Analyses
Biochemical endpoints (i.e., circulating and central steroid measures) were initially utilized as covariates in multivariate analyses of variance (MANOVA) but were not found to explain significant variance in primary dependent measures. Thus, behavioral and biochemical dependent measures were assessed via separate two to three-way analyses of variance (ANOVA) with estrous cycle phase (proestrous or diestrous), pretreatment (vehicle, oxycodone, antalarmin, RU-486, or OVX), and/or genotype [Tat(−) or Tat(+)] as between-subject factors. Group differences following main effects were determined via Fisher’s Protected Least Significant Difference post hoc tests. Significant interactions were delineated via simple main effects and main effect contrasts with α controlled for family-wise error. All analyses were considered significant when p < 0.05.
3. Results
3.1. HIV-1 Tat and Oxycodone-Mediated Psychostimulation Is Moderated by Stress and Estrous Cycle
In order to assess combined HIV-1 Tat interactions with oxycodone, Tat was induced via systemic administration of doxycycline for 5 days (with two days of washout; Figure 1A). Estrous cycle was assessed daily over the next 12 days and mice were behaviorally tested when in the proestrus or diestrus phase of their estrous cycle (whichever came first). A 15 min forced swim was used to activate the HPA axis in the stressed paradigm (or not in the non-stressed paradigm). On the day of testing, all mice received saline or oxycodone (3 mg/kg, i.p.) 15 min prior to behavioral assessment (Figure 1A).
In the non-stressed paradigm, oxycodone significantly increased the distance [F(1,70) = 69.07, p < 0.05] (see †, Figure 1B) and velocity [F(1,70) = 69.42, p < 0.05] (see †, Table 1) travelled by the mice in an open field test compared to saline administered controls. There was an interaction wherein oxycodone-administered Tat(+) mice travelled a significantly greater distance (p < 0.0001; see §, Figure 1B) and speed (p < 0.0001–0.0002; see §, Table 1) than any other group, irrespective of estrous cycle phase. Oxycodone-administered Tat(−) controls also travelled a greater distance than their saline-administered counterparts (p = 0.0001–0.0002). When anxiety-like behavior was assessed in a light-dark transition test, estrous cycle, oxycodone, and Tat exposure interacted to influence the time spent in light zone [F(1,67) = 5.34, p < 0.05]. Diestrous Tat(+) mice administered saline demonstrated the least anxiety-like behavior, spending significantly more time in the light zone than any other group with the exception of proestrous Tat(+) mice administered saline (p = 0.0003–0.0478; Table 1). Diestrous Tat(+) mice administered oxycodone demonstrated the most anxiety-like behavior on this test, significantly differing from proestrous, Tat(+) controls (p = 0.0329; Table 1). As expected, oxycodone also influenced motor behavior in the light-dark test, significantly increasing the number of chamber transitions [F(1,69) = 16.10, p < 0.05] (see †, Table 1); whereas Tat(+) mice made significantly fewer transitions than Tat(−) controls [F(1,69) = 4.10, p < 0.05] (see *, Table 1). Estrous cycle also influenced motor/exploratory behavior in this test, with diestrous mice rearing more than proestrous mice [F(1,69) = 6.60, p < 0.05] (see #, Table 1).

Table 1.
Motor and anxiety-like measures for Tat(−) and Tat(+) female mice assessed in open field and light-dark transition tests. Proestrus or diestrous mice were administered saline or oxycodone prior to behavioral assessment. * indicates a main effect of genotype wherein Tat(+) mice differ from Tat(−) controls. † indicates a main effect of drug condition wherein oxycodone-administered mice differs from saline-administered controls. # indicates a main effect of estrous cycle wherein diestrous mice differ from proestrous mice. § indicates an interaction wherein oxycodone-administered Tat(+) mice differ from Tat(−) controls and saline-administered controls. ^ indicates an interaction wherein saline-administered, diestrous Tat(+) mice differ from all other groups except for their respective proestrous counterparts, p < 0.05.
In the stressed paradigm, motor activity was notably reduced compared to the non-stressed paradigm; however, the influence of estrous cycle phase became apparent after forced swim stress. As anticipated, oxycodone-administered mice travelled a significantly greater distance [F(1,71) = 16.51, p < 0.05] (see †, Figure 1C) and velocity [F(1,71) = 16.46, p < 0.05] (Table 2) than saline-administered controls. Following forced swim, diestrous mice travelled a significantly greater distance than their proestrous counterparts [F(1,71) = 17.83, p < 0.05] (see #, Figure 1C). There was an interaction wherein Tat exposure potentiated oxycodone’s psychomotor effects, irrespective of estrous cycle phase [F(1,71) = 7.40, p < 0.05]. Oxycodone-administered Tat(+) mice travelled a significantly greater distance than did any other group (p < 0.0001–0.0001; see §, Figure 1C). Expression of Tat also influenced rearing time such that Tat(+) mice significantly spent more time rearing than their respective Tat(−) controls [F(1,69) = 7.50, p < 0.05] (see *, Figure 1C). When assessed in the light-dark transition test, significant differences in anxiety-like behavior were not observed (Table 2). However, oxycodone administration interacted with Tat expression to influence motor/exploratory behavior [F(1,70) = 5.15, p < 0.05] such that oxycodone-administered Tat(+) mice made more transitions than Tat(−) oxycodone- and Tat(+) saline-administered mice (p = 0.0200–0.0313; see §, Table 2). Estrous cycle also interacted with Tat expression [F(1,70) = 8.52, p < 0.05] such that Tat(−) mice in the proestrous phase made significantly fewer transitions than did Tat(+) mice in the proestrous phase or Tat(−) mice in the diestrous phase (p < 0.0079–0.0407; Table 2).

Table 2.
Motor and anxiety-like measures for Tat(−) and Tat(+) female mice assessed in open field and light-dark transition tests. Proestrus or diestrous mice were exposed to a forced swim stress prior to administration of saline or oxycodone. * indicates a main effect of genotype wherein Tat(+) mice differ from Tat(−) controls. # indicates a main effect of estrous cycle wherein diestrous mice differ from proestrous mice. § indicates an interaction wherein oxycodone-administered Tat(+) mice differ from respective Tat(−) controls and saline-administered controls. ‡ indicates an interaction wherein proestrous Tat(−) mice differ from respective Tat(+) and diestrous Tat(−) controls, p < 0.05.
In summary, Tat expression potentiated oxycodone-mediated psychomotor behavior. Stress enhanced this effect among diestrous, compared to proestrous, mice implicating factors associated with the HPG axis.
3.2. Gonadal Steroids Are Necessary for Tat to Potentiate Oxycodone-Mediated Psychostimulation
In order to determine the importance of the HPA and HPG axes in Tat-potentiated psychomotor and/or anxiety-like behavior, HPA axis receptor sites were pharmacologically blocked and circulating gonadal hormones were surgically attenuated. To achieve this, some mice were administered vehicle, antalarmin (CRF-R blocker), or RU-486 (GR blocker) concurrent with Tat induction for seven days. Some mice were OVX to remove the primary source of circulating gonadal hormones. Gonadally-intact mice were tested when in proestrus. All mice received an acute injection of saline or oxycodone (3 mg/kg) 15 min prior to behavior testing (Figure 1A).
Oxycodone significantly increased the distance travelled for all mice compared to saline administration [F(3,128) = 2.73, p > 0.05] (see †, Figure 1D). HIV-1 Tat exposure interacted with oxycodone administration to influence psychomotor behavior as assessed by the distance [F(1,128) = 14.42, p < 0.05] and velocity [F(1,128) = 14.56, p < 0.05] travelled in an open field (Figure 1D; Table 3). Irrespective of treatment with vehicle, antalarmin or RU-486, oxycodone-administered Tat(+) mice travelled a significantly greater distance (p < 0.0001; see §, Figure 1D) and speed (p < 0.0001; Table 3) than did Tat(−) controls or their saline administered counterparts. As well, there was an interaction for OVX to attenuate the Tat-potentiated increase in oxycodone-mediated distance [F(3,128) = 2.66, p < 0.05] and velocity [F(3,128) = 2.66, p < 0.05] travelled (see ‡, Figure 1D). OVX/Tat(+) mice administered oxycodone demonstrated a significant attenuation in the distance (p < 0.0023–0.0033; see ‡, Figure 1D) and speed (p < 0.0023–0.0033; Table 3) of travel compared to vehicle- or inhibitor-treated Tat(+) mice administered oxycodone, indicating the significant role ovarian hormones play modulating these effects.

Table 3.
Motor and anxiety-like measures for Tat(−) and Tat(+) female mice assessed in open field and light-dark transition tests. Proestrus mice were pretreated with vehicle, antalarmin, RU-486 or were ovariectomized (OVX) prior to administration of saline or oxycodone. * indicates a main effect of genotype wherein Tat(+) mice differ from Tat(−) controls. † indicates a main effect of drug condition wherein oxycodone-administered mice differ from saline-administered controls. # indicates a main effect for any HPA or HPG manipulation to differ from vehicle-pretreated mice. ^ indicates a main effect for RU-486 to differ from antalarmin or OVX. § indicates an interaction wherein oxycodone-administered Tat(+) mice differ from respective Tat(−) and saline-administered controls. @ indicates an interaction wherein antalarmin treated Tat(−) mice differ from respective Tat(+) mice and vehicle-treated Tat(−) controls. ‡ indicates an interaction wherein oxycodone-administered OVX mice differ from oxycodone-administered mice pretreated with vehicle, RU-486 and Antalarmin, p < 0.05.
In the light-dark transition test, either oxycodone [F(1,124) = 4.29, p < 0.05] (see †, Table 3) or Tat expression [F(1,124) = 16.66, p < 0.05] (see *, Table 3) significantly increased anxiety-like behavior by reducing the amount of time spent in the light zone. Pretreatment with RU-486 also increased anxiety-like behavior, reducing the amount of time spent in the light zone compared to antalarmin administration or OVX [F(3,124) = 2.70, p < 0.05] (see #, Table 3). Any pharmacological pretreatment or OVX significantly reduced the time spent time rearing compared to vehicle pretreatment [F(3,123) = 6.30, p < 0.05] (see #, Table 3). When transitions were assessed, oxycodone administration, estrous cycle phase and Tat expression interacted [F(3,126) = 2.75, p < 0.05] (see #, Table 3) such that antalarmin-treated Tat(+) mice made significantly fewer transitions than their respective Tat(−) control group (p = 0.046; Table 3). Conversely, antalarmin-treated Tat(−) mice made significantly more transitions than their respective Tat(−) control group (p = 0.0151; Table 3).
In summary, OVX attenuated Tat’s capacity to potentiate oxycodone-mediated psychostimulation. Neither CRF nor GR blockade influenced these effects, further supporting the influence of HPG factors in female mice.
3.3. Tat, Oxycodone, and Gonadal Steroids Interact to Influence Circulating Steroids
Circulating steroid concentrations were assessed in serum ~2 h following saline or opioid injection when HPA activation is expected to be resolving. In non-stressed mice, expression of Tat interacted with estrous cycle phase to influence resolving corticosterone concentrations [F(1,69) = 4.60, p < 0.05] (see ^, Figure 2B). Irrespective of oxycodone administration, either Tat(+) or diestrous mice demonstrated significantly greater corticosterone than did proestrous Tat(−) controls (p = 0.0035–0.0101; Figure 2B). No significant differences were observed in circulating estradiol (Figure 2B’). Circulating progesterone was significantly greater among diestrous, compared to proestrous, mice [F(1,70) = 14.84, p < 0.05] (see #, Figure 2B’’).
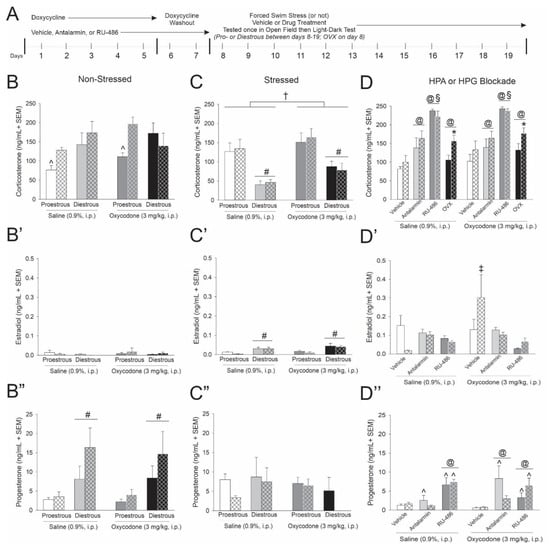
Figure 2.
(A) Circulating steroid concentrations obtained from Tat-transgenic, female mice [hatched bars; Tat(+)] or control counterparts [open bars; Tat(−) controls] described in Figure 1 (n = 6–10/group). Corticosterone, estradiol and progesterone were assessed in (B–B’’) non-stressed, (C–C’’) stressed and (D–D’’) HPA or HPG manipulated mice. ^ indicates an interaction wherein the denoted group differs from all other groups in panelsB and D”. † indicates a main effect for oxycodone to differ from saline administered mice in panel C. § indicates an interaction wherein mice pretreated with RU-486 differ from all other groups in panel D. @ indicates an interaction wherein the denoted group differs from their respective vehicle controls in panel D and D”. * indicates a main effect of genotype wherein Tat(+) mice differ from Tat(−) controls in panel D. # indicates a main effect for proestrous mice to differ from diestrous mice in panel C, C’ and B”. ‡ indicates an interaction wherein the denoted group differs from all other groups in panel D’, p < 0.05.
Among swim stress-exposed mice, circulating corticosterone was significantly greater in oxycodone-administered mice [F(1,70) = 6.30, p < 0.05] (see †, Figure 2C) and was significantly reduced among diestrous mice [F(1,70) = 37.05, p < 0.05] (see #, Figure 2C) compared to their respective saline-administered or proestrous counterparts. Concurrently, diestrous mice had significantly greater estradiol levels than did proestrous mice [F(1,71) = 24.60, p < 0.05] (see #, Figure 2C’). No significant differences were observed in circulating progesterone among stressed mice (Figure 2C’’).
These data recapitulate the clinical endophenotype of elevated basal cortisol observed in HIV-infected individuals and further reveal HPA/HPG interactions for stress to influence circulating estradiol and progesterone.
Pharmacologically antagonizing receptors that mediate HPA feedback or removing the primary source of gonadal steroids influenced circulating corticosterone. Either pretreating mice with antalarmin or RU-486, or conducting OVX, significantly increased circulating corticosterone [F(3,125) = 39.65, p < 0.05] (see @, Figure 2D). However, only OVX Tat(+) mice demonstrated a significant corticosterone increase compared to their respective Tat(−) controls [F(1,125) = 6.1, p < 0.05] (see *, Figure 2D). Among pretreatments, GR inhibition via RU-486 increased circulating corticosterone to a greater degree than other manipulations [F(3,125) = 39.65, p < 0.05] (see §, Figure 2D). When circulating estradiol was assessed, oxycodone-administered Tat(+) mice demonstrated greater circulating concentrations than did any other group [F(2,97) = 3.23, p < 0.0001–0.0239] (see ‡, Figure 2D’). Tat exposure and HPA receptor antagonism interacted to alter circulating progesterone [F(2,97) = 3.43, p < 0.05] (see ^, Figure 2D’’). Blocking GRs via RU-486 increased circulating progesterone, irrespective of Tat exposure. However, blocking CRF-Rs via antalarmin only increased progesterone among Tat(−) control mice (p = 0.0002–0.0020; see ^, Figure 2D’’). Additionally, HPA receptor antagonism and oxycodone administration interacted [F(2,97) = 4.45, p < 0.05] (see @, Figure 2D’’). Blocking GRs via RU-486 increased progesterone, irrespective of oxycodone administration. Blocking CRF-Rs via antalarmin only increased progesterone among oxycodone-treated mice (p < 0.0001–0.0493; see @, Figure 2D’’).
In summary, OVX prompted greater circulating corticosterone among Tat(+) mice compared to Tat(−) controls, the endocrine phenotype associated with an attenuated psychomotor response to oxycodone. While CRF-R or GR blockade altered total glucocorticoid levels, no differences between Tat(−) and Tat(+) mice were observed. These data support the notion that reinstatement of HPA responsivity may involve HPG manipulation and may rescue the Tat-mediated psychomotor response to oxycodone.
3.4. Acute Oxycodone Interacted with Tat Exposure to Influence Hypothalamic Allopregnanolone
Hypothalamic allopregnanolone content was greater among diestrous, compared to proestrous mice, in the non-stressed [F(1,56) = 36.02, p < 0.05] (see #, Figure 3A) and stressed paradigms [F(1,56) = 21.42, p < 0.05] (see #, Figure 3B). Moreover, Tat exposure interacted with oxycodone and estrous cycle phase to influence hypothalamic allopregnanolone content among non-stressed [F(1,56) = 4.02, p < 0.05] (see §, Figure 3A) and stressed mice [F(1,56) = 4.09, p < 0.05] (see § and ^, Figure 3B). Among non-stressed mice, oxycodone-administered Tat(−) controls demonstrated greater allopregnanolone in the diestrous phase of their cycle than did their saline-administered counterparts or any proestrous group (p < 0.0001–0.0066, see §, Figure 3A). Among stressed mice, Tat(+) saline-administered mice demonstrated greater allopregnanolone in the diestrous phase of their cycle than did any other proestrous group (p < 0.0001–0.0298, see §, Figure 3B) or their oxycodone-administered diestrous counterparts (p = 0.03, see ^, Figure 3B). No differences in hypothalamic allopregnanolone were observed among OVX mice (Figure 3C), despite an apparent basal increase compared to naturally-cycling mice.
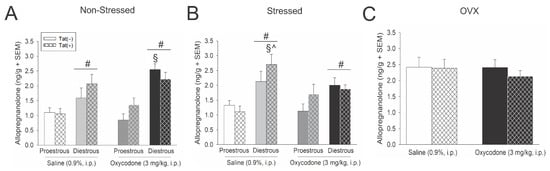
Figure 3.
Proestrous, diestrous or ovariectomized (OVX) Tat(−) and Tat(+) mice described in Figure 1 had allopregnanolone content (ng/g) assessed in the hypothalamus in (A) non-stressed, (B) stressed, and (C) OVX mice (n = 8/group). # indicates a main effect for diestrous mice to differ from proestrous mice. § indicates an interaction wherein the denoted group differs from Tat(−) or Tat(+) proestrous mice. ^ indicates an interaction wherein the denoted group differs from oxycodone-administered, Tat(+) diestrous mice, p < 0.05.
In summary, hypothalamic allopregnanolone was greater among diestrous, compared to proestrous, mice and OVX apparently increased basal allopregnanolone content and obviated oxycodone- or Tat-mediated fluctuations.
4. Discussion
The hypotheses that the HPA and/or HPG axes contribute to Tat- and oxycodone-mediated psychomotor and affective behavior were upheld; albeit the HPA phenotype produced by Tat in females and the mechanisms contributing to these effects differed from those previously observed in males. Exposure to Tat activated the HPA stress axis, recapitulating the clinical phenotype of elevated basal cortisol seen in up to 46% of HIV+ patients [26,28,59,60,61]. Moreover, either Tat expression and/or oxycodone exposure, increased anxiety-like behavior in the light-dark transition test. These data recapitulate our prior findings in male Tat-tg mice [42]; however, important sex differences were also revealed. Females exposed to Tat did not demonstrate adrenal insufficiency, nor did pharmacological blockade of the HPA feedback loop attenuate Tat’s capacity to potentiate oxycodone’s psychomotor effects. One potential mechanism underlying these sex differences may involve the contribution of HPA-mediating gonadal steroid hormones. Pregnane steroids fluctuate to greater levels in females, compared to males and may help offset HPA insults. In support, we observed a full attenuation of Tat’s capacity to potentiate oxycodone’s motor effects when mice were OVX, which was accompanied by a compensatory increase in adrenal glucocorticoid. Moreover, neurosteroids such as allopregnanolone are well-characterized to play a compensatory role to curtail stress-induced HPA-activation [62,63,64]. We assessed the hypothalamic levels of allopregnanolone and observed that OVX or combined Tat and oxycodone exposure promoted an increase in endogenous hypothalamic allopregnanolone. This may serve as a central adaptive response to stress similar to our prior findings in males exposed to Tat and morphine [23]. Together, these data suggest that Tat can dysregulate glucocorticoid neuroendocrine function and that endogenous neurosteroids like allopregnanolone may be upregulated in response. These data extend findings to implicate dysregulated HPA/HPG axes in the susceptibility to neuropsychiatric complications, opioid effects, and potential abuse liability.
Sex/gender differences in HIV are understudied, contributing to some discrepancies within the extant literature. While some studies find HIV+ women to have an improved immunological response to cART [65,66] and slower viral progression [67] compared to HIV+ men, studies assessing neuroHIV outcomes reveal women to be more vulnerable to HIV-associated neurocognitive disorders [68,69,70]. However, neuroHIV vulnerability may differ based on the dimensions assessed. While women are more prone to affective disorders in the general population, large clinical studies find HIV+ women to have a lower prevalence of major depression or any anxiety disorder [71,72] compared to HIV+ men. Unfortunately, these studies are often not stratified by gender and many are not controlled by route of HIV acquisition or endocrine factors, adding heterogeneity to findings. Some studies find no gender differences in HIV-related affect [73,74]. The potential mechanisms that underlie these discrepancies in cognitive vs. affective gender differences, may involve vulnerability to stress. Domains that involve HPA axis mediation, such as generalized anxiety and depressive symptomatology, may favor women [72,75,76]. HPA axis function provides a biological basis for sex differences in stress-related psychiatric disorders [77] and may interact with HPG factors. Gonadal hormones in premenopausal women contribute to the central responsivity of the stress response [78]. In animal models, females also have a particularly robust neuroendocrine response to stress [79,80,81,82,83] and demonstrate increased hypothalamic release of CRF and downstream circulating glucocorticoids [79,83,84]. Dysregulation of HPG and HPA axes may promote neurological behavioral deficits [37,85,86]. In support, we found stressed mice to be more vulnerable to Tat/oxycodone behavioral interactions when in the diestrous phase of their cycle (higher estradiol:progesterone ratio). These data are consistent with higher estradiol levels promoting psychostimulant response to drugs of abuse in rodent models [87,88,89,90].
Psychomotor locomotion is a robust assay to quantify the response of opioids in rodents [91] and may inform abuse liability. Opioid prescriptions for HIV+ patients are rising [7,8,9] and opioids exert long-term capacity to promote neuroendocrine dysfunction in people [92,93]. Given the potential vulnerability that neuroendocrine dysregulation confers to substance use and affective disorders [94,95,96,97], it is important to understand the effects that HIV and opioids exert on the HPA and HPG axes. We and others find that HIV-1 Tat facilitates the rewarding and sensitizing effects of opioids [42,43,98,99]. Indeed, HPA dysregulation confers vulnerability to substance use disorders [100]. Together, these data support the notion that maintenance of neuroendocrine function may help counteract HIV-related opioid abuse liability.
There are several mechanisms that may underlie endocrine abnormalities observed in HIV patients. Hypercortisolemia is reported in a considerable proportion of HIV+ patients [61,101,102]. Tat may be a contributor given that the current animal model recapitulates this phenotype. There may be several mechanisms for the hypercortisolemia observed. Tat has been proposed to promote GR transcription partly via its capacity to drive positive transcription elongation factor-b accumulation on GR-responsive promoters [103]. As well, Tat (alone or in conjunction with other proinflammatory HIV proteins) may promote GR activation indirectly via cytokine activation. Elevated levels of IL-2 and IL-4 decrease the affinity of the GR for cortisol and lead to a glucocorticoid resistant state [104]. As well, the β isoform of the GR, which inhibits the active α isoform of the receptor, may be increased by proinflammatory cytokines, thereby reducing GR signaling and promoting an increase in adrenal glucocorticoid [105,106,107]. Importantly, affective neuroHIV symptoms such as depression are associated with impaired GR signaling in HIV+ women [108]. Another aspect of HPA dysregulation observed in HIV patients is adrenal insufficiency (observed in up to 46% patients [24,25,26,27,28,61]); however, we did not observe this in Tat-exposed females. Adrenal insufficiency based on sex as biological variable has not been well characterized in the HIV clinical population due to its multifactorial nature [24,61,109,110,111]. There may be several explanations for the lack of adrenal insufficiency in females including lower CRF-R internalization, thereby increasing sensitivity to CRF [112]. Other reasons may include lower GR receptor density and GR translocation at the level of the hypothalamus in females, thereby reducing negative feedback [113,114]. Additionally, male mice demonstrate a robust increase in hypothalamic, GR-mediated negative feedback compared to females [114], which may increase their vulnerability to HPA insults. Females may also have higher corticosterone binding globulin, reducing bioavailable corticosterone and potentially the reserve for negative HPA feedback [115]. Lastly, both circulating and central pregnane steroids fluctuate to greater levels in females compared to males and may confer protection.
The HPA axis is modulated by non-traditional-acting steroid hormones that are produced de novo in the brain (i.e., neurosteroids [62]). Neurosteroids, such as allopregnanolone, are rapidly produced in response to stress and reinstate HPA-axis homeostasis, partly via potent positive allosteric modulation of inhibitory GABAA receptors [116,117,118,119]. The paraventricular nucleus of the hypothalamus (PVN) is rich in GABAergic neurons [120] and GABAA receptor expression [121]. We have previously observed Tat to promote the selective loss of GABAergic interneurons within the hippocampus, particularly those that are nNOS+, SST+ or PV+ [122]. Increased AlloP content in hypothalamus may compensate for inhibitory deficits promoted by the loss of GABAergic interneurons. AlloP’s capacity to prevent upregulation of CRF mRNA in the PVN following adrenalectomy, attenuates CRF release in response to stress and inhibits the firing of PVN CRF neurons [121,123,124,125]. Moreover, downstream ACTH release and subsequent corticosterone secretion in rodents are attenuated by neurosteroids [62,123,124,126,127,128]. These effects are dynamic; while neurosteroids dampen the HPA response, a dysregulated HPA axis may alter neurosteroidogenesis [129,130,131,132]. We have found that HIV-1 Tat expression in mice increases neurosteroidogenesis of pregnenolone and 5α-reduced steroid metabolites, including allopregnanolone [23]. These demonstrations of upregulated allopregnanolone content in the brain are similar to those observed following central trauma and may indicate a central adaptive response to stress. In support, CNS allopregnanolone is increased following traumatic brain injury, spinal cord injury, ischemic stroke and in neurodegenerative disease models where it exerts a neuroprotective functions (reviewed in [133]). Herein, we find that OVX mice have an increase in basal, hypothalamic AlloP content concurrent with a reduction of oxycodone’s psychomotor effects, supporting its role to maintain homeostasis. Beyond its capacity to modulate GABAA receptors [134], allopregnanolone is an inhibitor of L-type calcium channels [135,136] which may also attenuate Tat-mediated excitotoxicity [23,137]. Greater lability and fluctuations of pregnane steroids, such as allopregnanolone, in females may confer resilience to HPA insults such as those produced by Tat exposure.
It must be noted that Tat is not the only HIV-1 protein that may be expressed in the CNS. Other proteins including gp120 and viral protein R (VPR) may act alone or in concert with Tat to influence the HPA axis. In support, HIV-1 gp120 is observed to increase pituitary ACTH as well as circulating corticosterone [138]. Moreover, exogenous gp120 stimulates hypothalamic CRH and arginine vasopressin secretion in rats [139]. HIV-1 VPR promotes glucocorticoid hypersensitivity and was found to bind GRs directly and potentiate their signaling, in part by acting as a GR co-factor [140,141,142]. Effects of VPR to promote GR transcription may coincide with Tat’s potential capacity to co-activate GRs [61]. Future studies should assess the endocrine interactions between these proteins.
5. Conclusions
Together, these data demonstrate the capacity for HIV-1 Tat to dysregulate the HPA stress axis and promote vulnerability to the psychostimulant effects of oxycodone. The HPG axis likely plays an important regulatory role in these effects. Tat and oxycodone exposure altered steroid hormone production in circulation and neurosteroid formation in the hypothalamus. These effects occurred concurrent with increased psychomotor and anxiety-like behavior. Thus, maintenance of the neuroendocrine axes may exert beneficial effects on combined HIV-1 and oxycodone mediated pathology.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/v13050813/s1, Figure S1: Fold changes of tat mRNA expression using Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR), Figure S2: Motor behavioral measures acquired in open field, Table S1: Primers used for qRT-PCR, Table S2: Summary of findings for primary dependent measures.
Author Contributions
M.F.S.; conceptualization, funding acquisition, data curation, formal analysis, writing-original draft, writing—review & editing. F.M.; data curation. S.P.S.; data curation. J.J.P.; conceptualization, funding acquisition, data curation, formal analysis, writing—review & editing. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the National Institutes of Health [R00 DA039791 to J.J.P.; R01 DA052851 to J.J.P.; P30 GM122733 (PI: SoumyaJit Majumdar) pilot project to J.J.P.], a student grant-in-aid of research from the University of Mississippi, Graduate Student Council (to M.F.S.; Award #001408) and the University of Mississippi, School of Pharmacy. The content is solely the responsibility of the authors and does not necessarily represent the official views of funding agencies. We thank Dr. John Rimoldi for his guidance conducting the present allopregnanolone assays.
Institutional Review Board Statement
The study was conducted according to the ethical guidelines defined by National Institute of Health (NIH Publication No. 85–23) and approved by the Institutional Animal Care and Use Committee (IACUC) of University of Mississippi (Protocol #18-004 and 21-005; approved October 2017; September 2020).
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available upon request.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Wallet, C.; De Rovere, M.; Van Assche, J.; Daouad, F.; De Wit, S.; Gautier, V.; Mallon, P.W.G.; Marcello, A.; Van Lint, C.; Rohr, O.; et al. Microglial Cells: The Main HIV-1 Reservoir in the Brain. Front. Cell. Infect. Microbiol. 2019, 9, 362. [Google Scholar] [CrossRef] [PubMed]
- Nath, A.; Anderson, C.; Jones, M.; Maragos, W.; Booze, R.; Mactutus, C.; Bell, J.; Hauser, K.F.; Mattson, M. Neurotoxicity and dysfunction of dopaminergic systems associated with AIDS dementia. J. Psychopharmacol. 2000, 14, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Nath, A.; Hauser, K.F.; Wojna, V.; Booze, R.M.; Maragos, W.; Prendergast, M.; Cass, W.; Turchan, J.T. Molecular basis for interactions of HIV and drugs of abuse. J. Acquir. Immune Defic. Syndr. 2002, 31, S62–S69. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.B.; Kaul, M. Neuronal Stress and Injury Caused by HIV-1, cART and Drug Abuse: Converging Contributions to HAND. Brain Sci. 2017, 7, 25. [Google Scholar] [CrossRef]
- Saylor, D.; Dickens, A.M.; Sacktor, N.; Haughey, N.; Slusher, B.; Pletnikov, M.; Mankowski, J.L.; Brown, A.; Volsky, D.J.; McArthur, J.C. HIV-associated neurocognitive disorder-pathogenesis and prospects for treatment. Nat. Rev. Neurol. 2016, 12, 309. [Google Scholar] [CrossRef]
- McArthur, J.C.; Steiner, J.; Sacktor, N.; Nath, A. Human immunodeficiency virus-associated neurocognitive disorders: Mind the gap. Ann. Neurol. 2010, 67, 699–714. [Google Scholar] [CrossRef]
- Jeevanjee, S.; Penko, J.; Guzman, D.; Miaskowski, C.; Bangsberg, D.R.; Kushel, M.B. Opioid analgesic misuse is associated with incomplete antiretroviral adherence in a cohort of HIV-infected indigent adults in San Francisco. AIDS Behav. 2014, 18, 1352–1358. [Google Scholar] [CrossRef]
- Merlin, J.S.; Tamhane, A.; Starrels, J.L.; Kertesz, S.; Saag, M.; Cropsey, K. Factors Associated with Prescription of Opioids and Co-prescription of Sedating Medications in Individuals with HIV. AIDS Behav. 2016, 20, 687–698. [Google Scholar] [CrossRef]
- Silverberg, M.J.; Ray, G.T.; Saunders, K.; Rutter, C.M.; Campbell, C.I.; Merrill, J.O.; Sullivan, M.D.; Banta-Green, C.J.; Von Korff, M.; Weisner, C. Prescription long-term opioid use in HIV-infected patients. Clin. J. Pain 2012, 28, 39–46. [Google Scholar] [CrossRef]
- Canan, C.E.; Chander, G.; Monroe, A.K.; Gebo, K.A.; Moore, R.D.; Agwu, A.L.; Alexander, G.C.; Lau, B.; HIV Research Network. High-Risk Prescription Opioid Use among People Living with HIV. J. Acquir. Immune Defic. Syndr. 2018, 78, 283–290. [Google Scholar] [CrossRef]
- Brunet, L.; Napravnik, S.; Heine, A.D.; Leone, P.A.; Eron, J.J. Brief Report: Longitudinal Opioid Use among HIV-Infected Patients, 2000 to 2014. J. Acquir. Immune Defic. Syndr. 2017, 75, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Peters, P.J.; Pontones, P.; Hoover, K.W.; Patel, M.R.; Galang, R.R.; Shields, J.; Blosser, S.J.; Spiller, M.W.; Combs, B.; Switzer, W.M.; et al. Indiana HIV Outbreak Investigation Team. HIV Infection Linked to Injection Use of Oxymorphone in Indiana, 2014–2015. N. Engl. J. Med. 2016, 375, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.J.; Johnson, T.P.; Smith, B.R.; Reoma, L.B.; Santamaria, U.A.; Bachani, M.; Demarino, C.; Barclay, R.A.; Snow, J.; Sacktor, N.; et al. Presence of Tat and transactivation response element in spinal fluid despite antiretroviral therapy. AIDS 2019, 33, S145–S157. [Google Scholar] [CrossRef] [PubMed]
- Hudson, L.; Liu, J.; Nath, A.; Jones, M.; Raghavan, R.; Narayan, O.; Male, D.; Everall, I. Detection of the human immunodeficiency virus regulatory protein tat in CNS tissues. J. Neurovirol. 2000, 6, 145–155. [Google Scholar] [CrossRef]
- Mousseau, G.; Valente, S.T. Role of Host Factors on the Regulation of Tat-Mediated HIV-1 Transcription. Curr. Pharm. Des. 2017, 23, 4079–4090. [Google Scholar] [CrossRef] [PubMed]
- Fitting, S.; Zou, S.; Chen, W.; Vo, P.; Hauser, K.F.; Knapp, P.E. Regional heterogeneity and diversity in cytokine and chemokine production by astroglia: Differential responses to HIV-1 Tat, gp120, and morphine revealed by multiplex analysis. J. Proteome Res. 2010, 9, 1795–1804. [Google Scholar] [CrossRef]
- King, J.E.; Eugenin, E.A.; Buckner, C.M.; Berman, J.W. HIV tat and neurotoxicity. Microbes Infect. 2006, 8, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Fitting, S.; Knapp, P.E.; Zou, S.; Marks, W.D.; Bowers, M.S.; Akbarali, H.I.; Hauser, K.F. Interactive HIV-1 Tat and morphine-induced synaptodendritic injury is triggered through focal disruptions in Na+ influx, mitochondrial instability, and Ca2+ overload. J. Neurosci. 2014, 34, 12850–12864. [Google Scholar] [CrossRef]
- Haughey, N.J.; Nath, A.; Mattson, M.P.; Slevin, J.T.; Geiger, J.D. HIV-1 Tat through phosphorylation of NMDA receptors potentiates glutamate excitotoxicity. J. Neurochem. 2001, 78, 457–467. [Google Scholar] [CrossRef]
- Hu, X.T. HIV-1 Tat-Mediated Calcium Dysregulation and Neuronal Dysfunction in Vulnerable Brain Regions. Curr. Drug. Targets 2016, 17, 4–14. [Google Scholar] [CrossRef]
- Norman, J.P.; Perry, S.W.; Kasischke, K.A.; Volsky, D.J.; Gelbard, H.A. HIV-1 trans activator of transcription protein elicits mitochondrial hyperpolarization and respiratory deficit, with dysregulation of complex IV and nicotinamide adenine dinucleotide homeostasis in cortical neurons. J. Immunol. 2007, 178, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Thangaraj, A.; Periyasamy, P.; Liao, K.; Bendi, V.S.; Callen, S.; Pendyala, G.; Buch, S. HIV-1 TAT-mediated microglial activation: Role of mitochondrial dysfunction and defective mitophagy. Autophagy 2018, 14, 1596–1619. [Google Scholar] [CrossRef] [PubMed]
- Paris, J.J.; Liere, P.; Kim, S.; Mahdi, F.; Buchanan, M.E.; Nass, S.R.; Qrareya, A.N.; Salahuddin, M.F.; Pianos, A.; Fernandez, N.; et al. Pregnane steroidogenesis is altered by HIV-1 Tat and morphine: Physiological allopregnanolone is protective against neurotoxic and psychomotor effects. Neurobiol. Stress. 2020, 12, 100211. [Google Scholar] [CrossRef] [PubMed]
- González-González, J.G.; de la Garza-Hernández, N.E.; Garza-Morán, R.A.; Rivera-Morales, I.M.; Montes-Villarreal, J.; Valenzuela-Rendón, J.; Villarreal-Pérez, J.Z. Prevalence of abnormal adrenocortical function in human immunodeficiency virus infection by low-dose cosyntropin test. Int. J. STD AIDS 2001, 12, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Prasanthai, V.; Sunthornyothin, S.; Phowthongkum, P.; Suankratay, C. Prevalence of adrenal insufficiency in critically ill patients with AIDS. J. Med. Assoc. Thai. 2007, 90, 1768–1774. [Google Scholar]
- Marik, P.E.; Kiminyo, K.; Zaloga, G.P. Adrenal insufficiency in critically ill patients with human immunodeficiency virus. Crit. Care Med. 2002, 30, 1267–1273. [Google Scholar] [CrossRef]
- Afreen, B.; Khan, K.A.; Riaz, A. Adrenal Insufficiency in Pakistani HIV Infected Patients. J. Ayub. Med. Coll. Abbottabad 2017, 29, 428–431. [Google Scholar]
- Sharma, N.; Sharma, L.K.; Anand, A.; Gadpayle, A.K.; Gaurav, K.; Mukherjee, S.; Kulshreshtha, B.; Dutta, D. Presence, patterns & predictors of hypocortisolism in patients with HIV infection in India. Indian J. Med. Res. 2018, 147, 142–150. [Google Scholar] [CrossRef]
- Dobs, A.S.; Dempsey, M.A.; Ladenson, P.W.; Polk, B.F. Endocrine disorders in men infected with human immunodeficiency virus. Am. J. Med. 1988, 84, 611–616. [Google Scholar] [CrossRef]
- Dutta, D.; Sharma, L.K.; Sharma, N.; Gadpayle, A.K.; Anand, A.; Gaurav, K.; Gupta, A.; Poondla, Y.; Kulshreshtha, B. Occurrence, patterns & predictors of hypogonadism in patients with HIV infection in India. Indian J. Med. Res. 2017, 145, 804–814. [Google Scholar] [CrossRef]
- Grinspoon, S.; Corcoran, C.; Miller, K.; Biller, B.M.; Askari, H.; Wang, E.; Hubbard, J.; Anderson, E.J.; Basgoz, N.; Heller, H.M.; et al. Body composition and endocrine function in women with acquired immunodeficiency syndrome wasting. J. Clin. Endocrinol. Metab. 1997, 82, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.S.; Lo, Y.; Santoro, N.; Dobs, A.S. Androgen levels in older men who have or who are at risk of acquiring HIV infection. Clin. Infect. Dis. 2005, 41, 1794–1803. [Google Scholar] [CrossRef]
- Dube, M.P.; Parker, R.A.; Mulligan, K.; Tebas, P.; Robbins, G.K.; Roubenoff, R.; Grinspoon, S.K. Effects of potent antiretroviral therapy on free testosterone levels and fat-free mass in men in a prospective, randomized trial: A5005s, a substudy of AIDS Clinical Trials Group Study 384. Clin. Infect. Dis. 2007, 45, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Wunder, D.M.; Bersinger, N.A.; Fux, C.A.; Mueller, N.J.; Hirschel, B.; Cavassini, M.; Elzi, L.; Schmid, P.; Bernasconi, E.; Mueller, B.; et al. Swiss HIV Cohort Study. Hypogonadism in HIV-1-infected men is common and does not resolve during antiretroviral therapy. Antivir. Ther. 2007, 12, 261–265. [Google Scholar]
- Rietschel, P.; Corcoran, C.; Stanley, T.; Basgoz, N.; Klibanski, A.; Grinspoon, S. Prevalence of hypogonadism among men with weight loss related to human immunodeficiency virus infection who were receiving highly active antiretroviral therapy. Clin. Infect. Dis. 2000, 31, 1240–1244. [Google Scholar] [CrossRef]
- Crum-Cianflone, N.F.; Bavaro, M.; Hale, B.; Amling, C.; Truett, A.; Brandt, C.; Pope, B.; Furtek, K.; Medina, S.; Wallace, M.R. Erectile dysfunction and hypogonadism among men with HIV. AIDS Patient Care STDS 2007, 21, 9–19. [Google Scholar] [CrossRef]
- Herman, J.P.; McKlveen, J.M.; Ghosal, S.; Kopp, B.; Wulsin, A.; Makinson, R.; Scheimann, J.; Myers, B. Regulation of the Hypothalamic-Pituitary-Adrenocortical Stress Response. Compr. Physiol. 2016, 6, 603–621. [Google Scholar] [CrossRef]
- Frye, C.A.; Paris, J.J.; Osborne, D.M.; Campbell, J.C.; Kippin, T.E. Prenatal Stress Alters Progestogens to Mediate Susceptibility to Sex-Typical, Stress-Sensitive Disorders, such as Drug Abuse: A Review. Front. Psychiatry 2011, 2, 52. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Vale, W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006, 8, 383–395. [Google Scholar] [CrossRef]
- Tomaselli, G.; Vallée, M. Stress and drug abuse-related disorders: The promising therapeutic value of neurosteroids focus on pregnenolone-progesterone-allopregnanolone pathway. Front. Neuroendocrinol. 2019, 55, 100789. [Google Scholar] [CrossRef]
- Koob, G.F.; Volkow, N.D. Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry 2016, 3, 760–773. [Google Scholar] [CrossRef]
- Salahuddin, M.F.; Mahdi, F.; Paris, J.J. HIV-1 Tat Dysregulates the Hypothalamic-Pituitary-Adrenal Stress Axis and Potentiates Oxycodone-Mediated Psychomotor and Anxiety-Like Behavior of Male Mice. Int. J. Mol. Sci. 2020, 21, 8212. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, M.F.; Qrareya, A.N.; Mahdi, F.; Jackson, D.; Foster, M.; Vujanovic, T.; Box, J.G.; Paris, J.J. Combined HIV-1 Tat and oxycodone activate the hypothalamic-pituitary-adrenal and -gonadal axes and promote psychomotor, affective, and cognitive dysfunction in female mice. Horm. Behav. 2020, 119, 104649. [Google Scholar] [CrossRef]
- Paris, J.J.; Chen, X.; Anderson, J.; Qrareya, A.N.; Mahdi, F.; Du, F.; McLaughlin, J.P.; Kaufman, M.J. In vivo proton magnetic resonance spectroscopy detection of metabolite abnormalities in aged Tat-transgenic mouse brain. Geroscience 2021. [Google Scholar] [CrossRef]
- Fitting, S.; Scoggins, K.L.; Xu, R.; Dever, S.M.; Knapp, P.E.; Dewey, W.L.; Hauser, K.F. Morphine efficacy is altered in conditional HIV-1 Tat transgenic mice. Eur. J. Pharmacol. 2012, 689, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Wodarski, R.; Bagdas, D.; Paris, J.J.; Pheby, T.; Toma, W.; Xu, R.; Damaj, M.I.; Knapp, P.E.; Rice, A.S.C.; Hauser, K.F. Reduced intraepidermal nerve fibre density, glial activation, and sensory changes in HIV type-1 Tat-expressing female mice: Involvement of Tat during early stages of HIV-associated painful sensory neuropathy. Pain Rep. 2018, 3, e654. [Google Scholar] [CrossRef] [PubMed]
- Bruce-Keller, A.J.; Turchan-Cholewo, J.; Smart, E.J.; Geurin, T.; Chauhan, A.; Reid, R.; Xu, R.; Nath, A.; Knapp, P.E.; Hauser, K.F. Morphine causes rapid increases in glial activation and neuronal injury in the striatum of inducible HIV-1 Tat transgenic mice. Glia 2008, 56, 1414–1427. [Google Scholar] [CrossRef] [PubMed]
- Dickens, A.M.; Yoo, S.W.; Chin, A.C.; Xu, J.; Johnson, T.P.; Trout, A.L.; Hauser, K.F.; Haughey, N.J. Chronic low-level expression of HIV-1 Tat promotes a neurodegenerative phenotype with aging. Sci. Rep. 2017, 7, 7748. [Google Scholar] [CrossRef] [PubMed]
- Paris, J.J.; Fenwick, J.; McLaughlin, J.P. Progesterone protects normative anxiety-like responding among ovariectomized female mice that conditionally express the HIV-1 regulatory protein, Tat, in the CNS. Horm. Behav. 2014, 65, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Crawley, J.N.; Paylor, R. A proposed test battery and constellations of specific behavioral paradigms to investigate the behavioral phenotypes of transgenic and knockout mice. Horm. Behav. 1997, 31, 197–211. [Google Scholar] [CrossRef]
- Dong, H.; Wang, S.; Zeng, Z.; Li, F.; Montalvo-Ortiz, J.; Tucker, C.; Akhtar, S.; Shi, J.; Meltzer, H.Y.; Rice, K.C.; et al. Effects of corticotrophin-releasing factor receptor 1 antagonists on amyloid-β and behavior in Tg2576 mice. Psychopharmacology 2014, 231, 4711–4722. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, S.; Rice, K.C.; Munro, C.A.; Wand, G.S. Restraint stress and ethanol consumption in two mouse strains. Alcohol. Clin. Exp. Res. 2008, 32, 840–852. [Google Scholar] [CrossRef] [PubMed]
- Hofford, R.S.; Prendergast, M.A.; Bardo, M.T. Pharmacological manipulation of glucocorticoid receptors differentially affects cocaine self-administration in environmentally enriched and isolated rats. Behav. Brain Res. 2015, 283, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, E.M.; Medley, G.A.; Reeks, T.; Alexander, S.; Burne, T.H.J.; Eyles, D.W. Effect of the glucocorticoid receptor antagonist RU486 on MK-801 induced behavioural sensitisation. PLoS ONE 2017, 12, e0176156. [Google Scholar] [CrossRef] [PubMed]
- Paris, J.J.; Fenwick, J.; McLaughlin, J.P. Estrous cycle and HIV-1 Tat protein influence cocaine-conditioned place preference and induced locomotion of female mice. Curr. HIV Res. 2014, 12, 388–396. [Google Scholar] [CrossRef]
- Ferraz-de-Paula, V.; Stankevicius, D.; Ribeiro, A.; Pinheiro, M.L.; Rodrigues-Costa, E.C.; Florio, J.C.; Lapachinske, S.F.; Moreau, R.L.; Palermo-Neto, J. Differential behavioral outcomes of 3,4-methylenedioxymethamphetamine (MDMA-ecstasy) in anxiety-like responses in mice. Braz. J. Med. Biol. Res. 2011, 44, 428–437. [Google Scholar] [CrossRef]
- Leite, L.M.; Carvalho, A.G.; Ferreira, P.L.; Pessoa, I.X.; Gonçalves, D.O.; Lopes Ade, A.; Góes, J.G.; Alves, V.C.; Leal, L.K.; Brito, G.A.; et al. Anti-inflammatory properties of doxycycline and minocycline in experimental models: An in vivo and in vitro comparative study. Inflammopharmacology 2011, 19, 99–110. [Google Scholar] [CrossRef]
- Paris, J.J.; Singh, H.D.; Ganno, M.L.; Jackson, P.; McLaughlin, J.P. Anxiety-like behavior of mice produced by conditional central expression of the HIV-1 regulatory protein, Tat. Psychopharmacology 2014, 231, 2349–2360. [Google Scholar] [CrossRef]
- Christeff, N.; Gherbi, N.; Mammes, O.; Dalle, M.T.; Gharakhanian, S.; Lortholary, O.; Melchior, J.C.; Nunez, E.A. Serum cortisol and DHEA concentrations during HIV infection. Psychoneuroendocrinology 1997, 22, S11–S18. [Google Scholar] [CrossRef]
- Christeff, N.; Nunez, E.A.; Gougeon, M.L. Changes in cortisol/DHEA ratio in HIV-infected men are related to immunological and metabolic perturbations leading to malnutrition and lipodystrophy. Ann. N. Y. Acad. Sci. 2000, 917, 962–970. [Google Scholar] [CrossRef]
- Chrousos, G.P.; Zapanti, E.D. Hypothalamic-pituitary-adrenal axis in HIV infection and disease. Endocrinol. Metab. Clin. N. Am. 2014, 43, 791–806. [Google Scholar] [CrossRef] [PubMed]
- Crowley, S.K.; Girdler, S.S. Neurosteroid, GABAergic and hypothalamic pituitary adrenal (HPA) axis regulation: What is the current state of knowledge in humans? Psychopharmacology 2014, 231, 3619–3634. [Google Scholar] [CrossRef]
- Girdler, S.S.; Klatzkin, R. Neurosteroids in the context of stress: Implications for depressive disorders. Pharmacol. Ther. 2007, 116, 125–139. [Google Scholar] [CrossRef]
- Gunn, B.G.; Cunningham, L.; Mitchell, S.G.; Swinny, J.D.; Lambert, J.J.; Belelli, D. GABAA receptor-acting neurosteroids: A role in the development and regulation of the stress response. Front. Neuroendocrinol. 2015, 36, 28–48. [Google Scholar] [CrossRef] [PubMed]
- Finkel, D.G.; John, G.; Holland, B.; Slim, J.; Smith, S.M. Women have a greater immunological response to effective virological HIV-1 therapy. AIDS 2003, 17, 2009–2011. [Google Scholar] [CrossRef] [PubMed]
- Grinsztejn, B.; Smeaton, L.; Barnett, R.; Klingman, K.; Hakim, J.; Flanigan, T.; Kumarasamy, N.; Campbell, T.; Currier, J.; PEARLS study team of the ACTG. Sex-associated differences in pre-antiretroviral therapy plasma HIV-1 RNA in diverse areas of the world vary by CD4(+) T-cell count. Antivir. Ther. 2011, 16, 1057–1062. [Google Scholar] [CrossRef]
- Jarrin, I.; Geskus, R.; Bhaskaran, K.; Prins, M.; Perez-Hoyos, S.; Muga, R.; Hernández-Aguado, I.; Meyer, L.; Porter, K.; del Amo, J.; et al. Gender differences in HIV progression to AIDS and death in industrialized countries: Slower disease progression following HIV seroconversion in women. Am. J. Epidemiol. 2008, 168, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Maki, P.M.; Rubin, L.H.; Springer, G.; Seaberg, E.C.; Sacktor, N.; Miller, E.N.; Valcour, V.; Young, M.A.; Becker, J.T.; Martin, E.M.; et al. Differences in Cognitive Function Between Women and Men with HIV. J. Acquir. Immune Defic. Syndr. 2018, 79, 101–107. [Google Scholar] [CrossRef]
- Qiao, X.; Lin, H.; Chen, X.; Ning, C.; Wang, K.; Shen, W.; Xu, X.; Xu, X.; Liu, X.; He, N.; et al. Sex differences in neurocognitive screening among adults living with HIV in China. J. Neurovirol. 2019, 25, 363–371. [Google Scholar] [CrossRef]
- Rubin, L.H.; Neigh, G.N.; Sundermann, E.E.; Xu, Y.; Scully, E.P.; Maki, P.M. Sex Differences in Neurocognitive Function in Adults with HIV: Patterns, Predictors, and Mechanisms. Curr. Psychiatry Rep. 2019, 21, 94. [Google Scholar] [CrossRef]
- Bing, E.G.; Burnam, M.A.; Longshore, D.; Fleishman, J.A.; Sherbourne, C.D.; London, A.S.; Turner, B.J.; Eggan, F.; Beckman, R.; Vitiello, B.; et al. Psychiatric disorders and drug use among human immunodeficiency virus-infected adults in the United States. Arch. Gen. Psychiatry 2001, 58, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.; Olfson, M.; Rabkin, J.; Hasin, D.S.; Alegría, A.A.; Lin, K.H.; Grant, B.F.; Blanco, C. Gender, HIV status, and psychiatric disorders: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. J. Clin. Psychiatry 2012, 73, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Tsao, J.C.; Dobalian, A.; Moreau, C.; Dobalian, K. Stability of anxiety and depression in a national sample of adults with human immunodeficiency virus. J. Nerv. Ment. Dis. 2004, 192, 111–118. [Google Scholar] [CrossRef]
- Sewell, M.C.; Goggin, K.J.; Rabkin, J.G.; Ferrando, S.J.; McElhiney, M.C.; Evans, S. Anxiety syndromes and symptoms among men with AIDS: A longitudinal controlled study. Psychosomatics 2000, 41, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Orlando, M.; Burnam, M.A.; Beckman, R.; Morton, S.C.; London, A.S.; Bing, E.G.; Fleishman, J.A. Re-estimating the prevalence of psychiatric disorders in a nationally representative sample of persons receiving care for HIV: Results from the HIV Cost and Services Utilization Study. Int. J. Methods Psychiatr. Res. 2002, 11, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Goggin, K.; Engelson, E.S.; Rabkin, J.G.; Kotler, D.P. The relationship of mood, endocrine, and sexual disorders in human immunodeficiency virus positive (HIV+) women: An exploratory study. Psychosom. Med. 1998, 60, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Andreano, J.M.; Touroutoglou, A.; Dickerson, B.; Barrett, L.F. Hormonal Cycles, Brain Network Connectivity, and Windows of Vulnerability to Affective Disorder. Trends Neurosci. 2018, 41, 660–676. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.M.; Jerram, M.; Abbs, B.; Whitfield-Gabrieli, S.; Makris, N. Sex differences in stress response circuitry activation dependent on female hormonal cycle. J. Neurosci. 2010, 30, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Viau, V.; Bingham, B.; Davis, J.; Lee, P.; Wong, M. Gender and puberty interact on the stress-induced activation of parvocellular neurosecretory neurons and corticotropin-releasing hormone messenger ribonucleic acid expression in the rat. Endocrinology 2005, 146, 137–146. [Google Scholar] [CrossRef]
- MacLusky, N.J.; Yuan, H.; Elliott, J.; Brown, T.J. Sex differences in corticosteroid binding in the rat brain: An in vitro autoradiographic study. Brain Res. 1996, 708, 71–81. [Google Scholar] [CrossRef]
- Handa, R.J.; Burgess, L.H.; Kerr, J.E.; O′Keefe, J.A. Gonadal steroid hormone receptors and sex differences in the hypothalamo-pituitary-adrenal axis. Horm. Behav. 1994, 28, 464–476. [Google Scholar] [CrossRef] [PubMed]
- Babb, J.A.; Masini, C.V.; Day, H.E.; Campeau, S. Sex differences in activated corticotropin-releasing factor neurons within stress-related neurocircuitry and hypothalamic-pituitary-adrenocortical axis hormones following restraint in rats. Neuroscience 2013, 234, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki-Sekino, A.; Mano-Otagiri, A.; Ohata, H.; Yamauchi, N.; Shibasaki, T. Gender differences in corticotropin and corticosterone secretion and corticotropin-releasing factor mRNA expression in the paraventricular nucleus of the hypothalamus and the central nucleus of the amygdala in response to footshock stress or psychological stress in rats. Psychoneuroendocrinology 2009, 34, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Duncko, R.; Kiss, A.; Skultétyová, I.; Rusnák, M.; Jezová, D. Corticotropin-releasing hormone mRNA levels in response to chronic mild stress rise in male but not in female rats while tyrosine hydroxylase mRNA levels decrease in both sexes. Psychoneuroendocrinology 2001, 26, 77–89. [Google Scholar] [CrossRef]
- Oyola, M.G.; Handa, R.J. Hypothalamic-pituitary-adrenal and hypothalamic-pituitary-gonadal axes: Sex differences in regulation of stress responsivity. Stress 2017, 20, 476–494. [Google Scholar] [CrossRef] [PubMed]
- Young, E.A. Sex differences and the HPA axis: Implications for psychiatric disease. J. Gend. Specif. Med. 1998, 1, 21–27. [Google Scholar]
- Hu, M.; Becker, J.B. Effects of sex and estrogen on behavioral sensitization to cocaine in rats. J. Neurosci. 2003, 23, 693–699. [Google Scholar] [CrossRef]
- Calipari, E.S.; Juarez, B.; Morel, C.; Walker, D.M.; Cahill, M.E.; Ribeiro, E.; Roman-Ortiz, C.; Ramakrishnan, C.; Deisseroth, K.; Han, M.H.; et al. Dopaminergic dynamics underlying sex-specific cocaine reward. Nat. Commun. 2017, 8, 13877. [Google Scholar] [CrossRef]
- Vandegrift, B.J.; You, C.; Satta, R.; Brodie, M.S.; Lasek, A.W. Estradiol increases the sensitivity of ventral tegmental area dopamine neurons to dopamine and ethanol. PLoS ONE 2017, 12, e0187698. [Google Scholar] [CrossRef]
- Ramôa, C.P.; Doyle, S.E.; Naim, D.W.; Lynch, W.J. Estradiol as a mechanism for sex differences in the development of an addicted phenotype following extended access cocaine self-administration. Neuropsychopharmacology 2013, 38, 1698–1705. [Google Scholar] [CrossRef]
- Zhang, J.J.; Kong, Q. Locomotor activity: A distinctive index in morphine self-administration in rats. PLoS ONE 2017, 12, e0174272. [Google Scholar] [CrossRef] [PubMed]
- Daniell, H.W. Hypogonadism in men consuming sustained-action oral opioids. J. Pain 2002, 3, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Daniell, H.W. Opioid endocrinopathy in women consuming prescribed sustained-action opioids for control of nonmalignant pain. J. Pain 2008, 9, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.; Kreek, M.J. Stress, dysregulation of drug reward pathways, and the transition to drug dependence. Am. J. Psychiatry 2007, 164, 1149–1159. [Google Scholar] [CrossRef] [PubMed]
- Porter, R.J.; Gallagher, P. Abnormalities of the HPA axis in affective disorders: Clinical subtypes and potential treatments. Acta Neuropsychiatr. 2006, 18, 193–209. [Google Scholar] [CrossRef]
- Brown, T.T.; Wisniewski, A.B.; Dobs, A.S. Gonadal and Adrenal Abnormalities in Drug Users: Cause or Consequence of Drug Use Behavior and Poor Health Outcomes. Am. J. Infect. Dis. 2006, 2, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Blaine, S.K.; Milivojevic, V.; Fox, H.; Sinha, R. Alcohol Effects on Stress Pathways: Impact on Craving and Relapse Risk. Can. J. Psychiatry 2016, 61, 145–153. [Google Scholar] [CrossRef]
- Gonek, M.; McLane, V.D.; Stevens, D.L.; Lippold, K.; Akbarali, H.I.; Knapp, P.E.; Dewey, W.L.; Hauser, K.F.; Paris, J.J. CCR5 mediates HIV-1 Tat-induced neuroinflammation and influences morphine tolerance, dependence, and reward. Brain Behav. Immun. 2018, 69, 124–138. [Google Scholar] [CrossRef]
- Kesby, J.P.; Najera, J.A.; Romoli, B.; Fang, Y.; Basova, L.; Birmingham, A.; Marcondes, M.C.G.; Dulcis, D.; Semenova, S. HIV-1 TAT protein enhances sensitization to methamphetamine by affecting dopaminergic function. Brain Behav. Immun. 2017, 65, 210–221. [Google Scholar] [CrossRef]
- Lovallo, W.R. The hypothalamic-pituitary-adrenocortical axis in addiction. Int. J. Psychophysiol. 2006, 59, 193–194. [Google Scholar] [CrossRef]
- Hoshino, Y.; Yamashita, N.; Nakamura, T.; Iwamoto, A. Prospective examination of adrenocortical function in advanced AIDS patients. Endocr. J. 2002, 49, 641–647. [Google Scholar] [CrossRef][Green Version]
- Membreno, L.; Irony, I.; Dere, W.; Klein, R.; Biglieri, E.G.; Cobb, E. Adrenocortical function in acquired immunodeficiency syndrome. J. Clin. Endocrinol. Metab. 1987, 65, 482–487. [Google Scholar] [CrossRef]
- Kino, T.; Chrousos, G.P. Human immunodeficiency virus type-1 accessory protein Vpr: A causative agent of the AIDS-related insulin resistance/lipodystrophy syndrome? Ann. N. Y. Acad. Sci. 2004, 1024, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Norbiato, G.; Bevilacqua, M.; Vago, T.; Baldi, G.; Chebat, E.; Bertora, P.; Moroni, M.; Galli, M.; Oldenburg, N. Cortisol resistance in acquired immunodeficiency syndrome. J. Clin. Endocrinol. Metab. 1992, 74, 608–613. [Google Scholar] [CrossRef]
- Leung, D.Y.; Hamid, Q.; Vottero, A.; Szefler, S.J.; Surs, W.; Minshall, E.; Chrousos, G.P.; Klemm, D.J. Association of glucocorticoid insensitivity with increased expression of glucocorticoid receptor beta. J. Exp. Med. 1997, 186, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Bamberger, C.M.; Bamberger, A.M.; de Castro, M.; Chrousos, G.P. Glucocorticoid receptor beta, a potential endogenous inhibitor of glucocorticoid action in humans. J. Clin. Investig. 1995, 95, 2435–2441. [Google Scholar] [CrossRef]
- Charmandari, E.; Chrousos, G.P.; Ichijo, T.; Bhattacharyya, N.; Vottero, A.; Souvatzoglou, E.; Kino, T. The human glucocorticoid receptor (hGR) beta isoform suppresses the transcriptional activity of hGRalpha by interfering with formation of active coactivator complexes. Mol. Endocrinol. 2005, 19, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Bekhbat, M.; Mehta, C.C.; Kelly, S.D.; Vester, A.; Ofotokun, I.; Felger, J.; Wingood, G.; Anastos, K.; Gustafson, D.R.; Kassaye, S.; et al. HIV and symptoms of depression are independently associated with impaired glucocorticoid signaling. Psychoneuroendocrinology 2018, 96, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Eledrisi, M.S.; Verghese, A.C. Adrenal insufficiency in HIV infection: A review and recommendations. Am. J. Med. Sci. 2001, 321, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Freda, P.U.; Bilezikian, J.P. The hypothalamus-pituitary-adrenal axis in HIV disease. AIDS Read. 1999, 9, 43–50. [Google Scholar]
- Mayo, J.; Collazos, J.; Martínez, E.; Ibarra, S. Adrenal Function in the Human Immunodeficiency Virus–Infected Patient. Arch. Intern. Med. 2002, 162, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
- Bangasser, D.A.; Curtis, A.; Reyes, B.A.; Bethea, T.T.; Parastatidis, I.; Ischiropoulos, H.; Van Bockstaele, E.J.; Valentino, R.J. Sex differences in corticotropin-releasing factor receptor signaling and trafficking: Potential role in female vulnerability to stress-related psychopathology. Mol. Psychiatry 2010, 877, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.B.; Weaver, D.A. Sexual dimorphism of glucocorticoid binding in rat brain. Brain Res. 1985, 343, 16–23. [Google Scholar] [CrossRef]
- Solomon, M.B.; Loftspring, M.; de Kloet, A.D.; Ghosal, S.; Jankord, R.; Flak, J.N.; Wulsin, A.C.; Krause, E.G.; Zhang, R.; Rice, T.; et al. Neuroendocrine Function After Hypothalamic Depletion of Glucocorticoid Receptors in Male and Female Mice. Endocrinology 2015, 156, 2843–2853. [Google Scholar] [CrossRef] [PubMed]
- Tannenbaum, B.; Rowe, W.; Sharma, S.; Diorio, J.; Steverman, A.; Walker, M.; Meaney, M.J. Dynamic variations in plasma corticosteroid-binding globulin and basal HPA activity following acute stress in adult rats. J. Neuroendocrinol. 1997, 9, 163–168. [Google Scholar] [CrossRef]
- Majewska, M.D.; Harrison, N.L.; Schwartz, R.D.; Barker, J.L.; Paul, S.M. Steroid hormone metabolites are barbiturate-like modulators of the GABA receptor. Science 1986, 232, 1004–1007. [Google Scholar] [CrossRef]
- Morrow, A.L.; Suzdak, P.D.; Paul, S.M. Steroid hormone metabolites potentiate GABA receptor-mediated chloride ion flux with nanomolar potency. Eur. J. Pharmacol. 1987, 142, 483–485. [Google Scholar] [CrossRef]
- Lambert, J.J.; Cooper, M.A.; Simmons, R.D.; Weir, C.J.; Belelli, D. Neurosteroids: Endogenous allosteric modulators of GABA(A) receptors. Psychoneuroendocrinology 2009, 34, S48–S58. [Google Scholar] [CrossRef]
- Reddy, D.S.; Rogawski, M.A. Stress-induced deoxycorticosterone-derived neurosteroids modulate GABA(A) receptor function and seizure susceptibility. J. Neurosci. 2002, 22, 3795–3805. [Google Scholar] [CrossRef]
- Miklós, I.H.; Kovács, K.J. GABAergic innervation of corticotropin-releasing hormone (CRH)-secreting parvocellular neurons and its plasticity as demonstrated by quantitative immunoelectron microscopy. Neuroscience 2002, 113, 581–592. [Google Scholar] [CrossRef]
- Cullinan, W.E. GABA(A) receptor subunit expression within hypophysiotropic CRH neurons: A dual hybridization histochemical study. J. Comp. Neurol. 2000, 419, 344–351. [Google Scholar] [CrossRef]
- Marks, W.D.; Paris, J.J.; Schier, C.J.; Denton, M.D.; Fitting, S.; McQuiston, A.R.; Knapp, P.E.; Hauser, K.F. HIV-1 Tat causes cognitive deficits and selective loss of parvalbumin, somatostatin, and neuronal nitric oxide synthase expressing hippocampal CA1 interneuron subpopulations. J. Neurovirol. 2016, 22, 747–762. [Google Scholar] [CrossRef] [PubMed]
- Patchev, V.K.; Shoaib, M.; Holsboer, F.; Almeida, O.F. The neurosteroid tetrahydroprogesterone counteracts corticotropin-releasing hormone-induced anxiety and alters the release and gene expression of corticotropin-releasing hormone in the rat hypothalamus. Neuroscience 1994, 62, 265–271. [Google Scholar] [CrossRef]
- Patchev, V.K.; Hassan, A.H.; Holsboer, D.F.; Almeida, O.F. The neurosteroid tetrahydroprogesterone attenuates the endocrine response to stress and exerts glucocorticoid-like effects on vasopressin gene transcription in the rat hypothalamus. Neuropsychopharmacology 1996, 15, 533–540. [Google Scholar] [CrossRef]
- Gunn, B.G.; Cunningham, L.; Cooper, M.A.; Corteen, N.L.; Seifi, M.; Swinny, J.D.; Lambert, J.J.; Belelli, D. Dysfunctional astrocytic and synaptic regulation of hypothalamic glutamatergic transmission in a mouse model of early-life adversity: Relevance to neurosteroids and programming of the stress response. J. Neurosci. 2013, 33, 19534–19554. [Google Scholar] [CrossRef] [PubMed]
- Bitran, D.; Dugan, M.; Renda, P.; Ellis, R.; Foley, M. Anxiolytic effects of the neuroactive steroid pregnanolone (3 alpha-OH-5 beta-pregnan-20-one) after microinjection in the dorsal hippocampus and lateral septum. Brain Res. 1999, 850, 217–224. [Google Scholar] [CrossRef]
- Carboni, E.; Wieland, S.; Lan, N.C.; Gee, K.W. Anxiolytic properties of endogenously occurring pregnanediols in two rodent models of anxiety. Psychopharmacology 1996, 126, 173–178. [Google Scholar] [CrossRef]
- Owens, M.J.; Ritchie, J.C.; Nemeroff, C.B. 5 alpha-pregnane-3 alpha, 21-diol-20-one (THDOC) attenuates mild stress-induced increases in plasma corticosterone via a non-glucocorticoid mechanism: Comparison with alprazolam. Brain Res. 1992, 573, 353–355. [Google Scholar] [CrossRef]
- Romeo, E.; Ströhle, A.; Spalletta, G.; di Michele, F.; Hermann, B.; Holsboer, F.; Pasini, A.; Rupprecht, R. Effects of antidepressant treatment on neuroactive steroids in major depression. Am. J. Psychiatry 1998, 155, 910–913. [Google Scholar] [CrossRef]
- Uzunova, V.; Sheline, Y.; Davis, J.M.; Rasmusson, A.; Uzunov, D.P.; Costa, E.; Guidotti, A. Increase in the cerebrospinal fluid content of neurosteroids in patients with unipolar major depression who are receiving fluoxetine or fluvoxamine. Proc. Natl. Acad. Sci. USA 1998, 95, 3239–3244. [Google Scholar] [CrossRef] [PubMed]
- Serra, M.; Pisu, M.G.; Littera, M.; Papi, G.; Sanna, E.; Tuveri, F.; Usala, L.; Purdy, R.H.; Biggio, G. Social isolation-induced decreases in both the abundance of neuroactive steroids and GABA(A) receptor function in rat brain. J. Neurochem. 2000, 75, 732–740. [Google Scholar] [CrossRef]
- Matsumoto, K.; Pinna, G.; Puia, G.; Guidotti, A.; Costa, E. Social isolation stress-induced aggression in mice: A model to study the pharmacology of neurosteroidogenesis. Stress 2005, 8, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Guennoun, R.; Labombarda, F.; Gonzalez Deniselle, M.C.; Liere, P.; De Nicola, A.F.; Schumacher, M. Progesterone and allopregnanolone in the central nervous system: Response to injury and implication for neuroprotection. J. Steroid. Biochem. Mol. Biol. 2015, 146, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Callachan, H.; Cottrell, G.A.; Hather, N.Y.; Lambert, J.J.; Nooney, J.M.; Peters, J.A. Modulation of the GABAA receptor by progesterone metabolites. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1987, 231, 359–369. [Google Scholar] [CrossRef]
- Earl, D.E.; Tietz, E.I. Inhibition of recombinant L-type voltage-gated calcium channels by positive allosteric modulators of GABAA receptors. J. Pharmacol. Exp. Ther. 2011, 337, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Hu, A.Q.; Wang, Z.M.; Lan, D.M.; Fu, Y.M.; Zhu, Y.H.; Dong, Y.; Zheng, P. Inhibition of evoked glutamate release by neurosteroid allopregnanolone via inhibition of L-type calcium channels in rat medial prefrontal cortex. Neuropsychopharmacology 2007, 32, 1477–1489. [Google Scholar] [CrossRef][Green Version]
- Paris, J.J.; Zou, S.; Hahn, Y.K.; Knapp, P.E.; Hauser, K.F. 5α-reduced progestogens ameliorate mood-related behavioral pathology, neurotoxicity, and microgliosis associated with exposure to HIV-1 Tat. Brain Behav. Immun. 2016, 55, 202–214. [Google Scholar] [CrossRef]
- Raber, J.; Toggas, S.M.; Lee, S.; Bloom, F.E.; Epstein, C.J.; Mucke, L. Central nervous system expression of HIV-1 Gp120 activates the hypothalamic-pituitary-adrenal axis: Evidence for involvement of NMDA receptors and nitric oxide synthase. Virology 1996, 226, 362–373. [Google Scholar] [CrossRef]
- Costa, A.; Nappi, R.E.; Polatti, F.; Poma, A.; Grossman, A.B.; Nappi, G. Stimulating effect of HIV-1 coat protein gp120 on corticotropin-releasing hormone and arginine vasopressin in the rat hypothalamus: Involvement of nitric oxide. Exp. Neurol. 2000, 166, 376–384. [Google Scholar] [CrossRef]
- Kino, T.; Gragerov, A.; Kopp, J.B.; Stauber, R.H.; Pavlakis, G.N.; Chrousos, G.P. The HIV-1 virion-associated protein Vpr is a coactivator of the human glucocorticoid receptor. J. Exp. Med. 1999, 189, 51–61. [Google Scholar] [CrossRef]
- Kino, T.; Gragerov, A.; Slobodskaya, O.; Tsopanomichalou, M.; Chrousos, G.P.; Pavlakis, G.N. Human immunodeficiency virus type 1 (HIV-1) accessory protein Vpr induces transcription of the HIV-1 and glucocorticoid-responsive promoters by binding directly to p300/CBP coactivators. J. Virol. 2002, 76, 9724–9734. [Google Scholar] [CrossRef] [PubMed]
- Sherman, M.P.; de Noronha, C.M.; Pearce, D.; Greene, W.C. Human immunodeficiency virus type 1 Vpr contains two leucine-rich helices that mediate glucocorticoid receptor coactivation independently of its effects on G(2) cell cycle arrest. J. Virol. 2000, 74, 8159–8165. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).