Wall Shear Stress Analysis and Optimization in Tissue Engineering TPMS Scaffolds
Abstract
1. Introduction
2. Materials and Methods
2.1. Scaffold Design
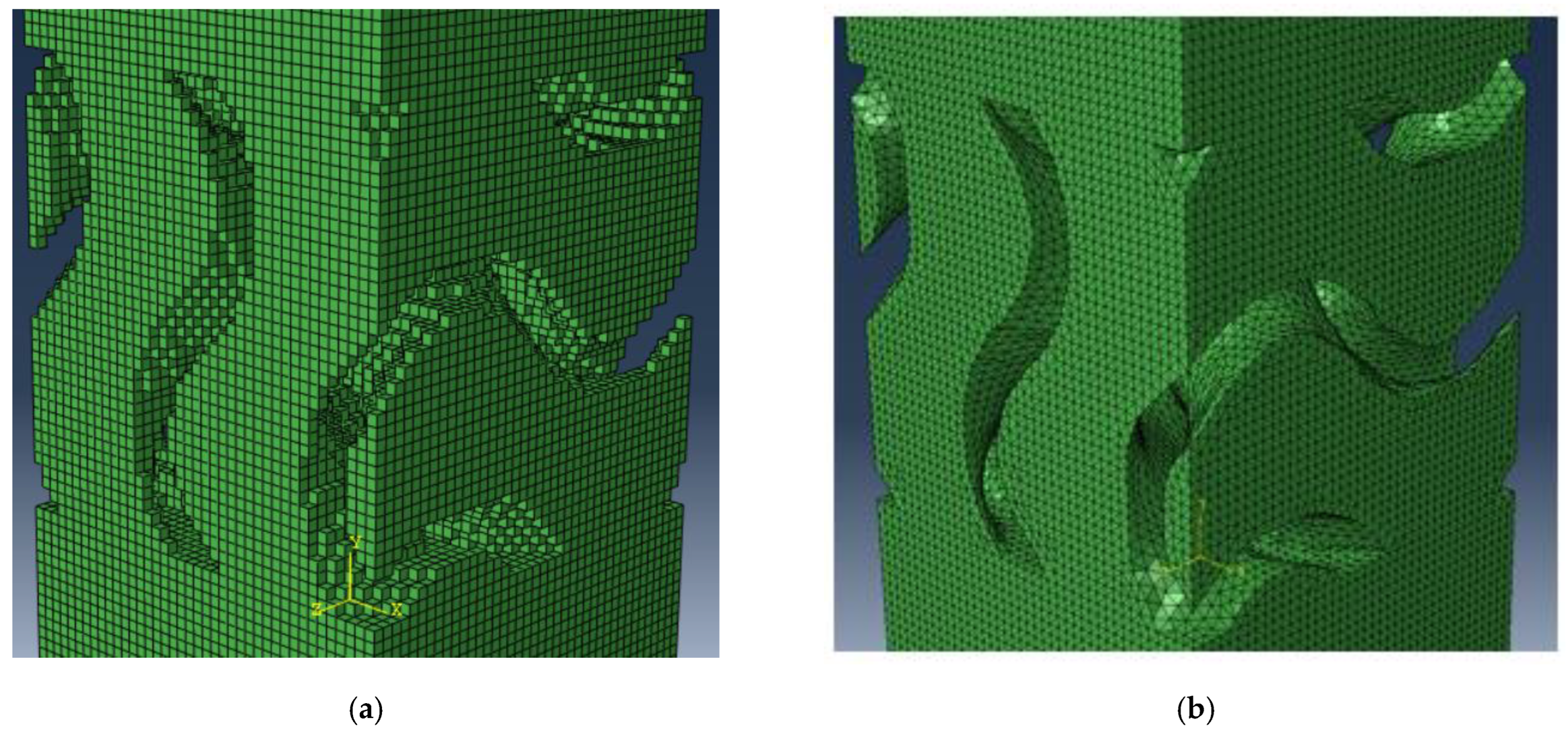
2.2. Surface Topology CFD Analysis
2.3. Structural Optimization Process
3. Results
3.1. Scaffold Design Parameters
3.2. Surface Topology CFD Analysis
3.3. Optimization Method
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Laubach, M.; Suresh, S.; Herath, B.; Wille, M.-L.; Delbrück, H.; Alabdulrahman, H.; Hutmacher, D.W.; Hildebrand, F. Clinical translation of a patient-specific scaffold-guided bone regeneration concept in four cases with large long bone defects. J. Orthop. Transl. 2022, 34, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Perier-Metz, C.; Cipitria, A.; Hutmacher, D.W.; Duda, G.N.; Checa, S. An in silico model predicts the impact of scaffold design in large bone defect regeneration. Acta Biomater. 2022, 145, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Ali, D.; Sen, S. Permeability and fluid flow-induced wall shear stress of bone tissue scaffolds: Computational fluid dynamic analysis using Newtonian and non-Newtonian blood flow models. Comput. Biol. Med. 2018, 99, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Basri, H.; Nasution, J.D.; Syahrom, A.; Sulong, M.A.; Md Saad, A.P.; Prakoso, A.T.; Aminin, F. The effect to flow rate characteristic on biodegradation of bone scaffold. Malays. J. Fundam. Appl. Sci. 2017, 13, 546–552. [Google Scholar] [CrossRef]
- Yu, G.; Li, Z.; Li, S.; Zhang, Q.; Hua, Y.; Liu, H.; Zhao, X.; Dhaidhai, D.T.; Li, W.; Wang, X. The select of internal architecture for porous Ti alloy scaffold: A compromise between mechanical properties and permeability. Mater. Des. 2020, 192, 108754. [Google Scholar] [CrossRef]
- McCoy, R.J.; O’Brien, F.J. Influence of Shear Stress in Perfusion Bioreactor Cultures for the Development of Three-Dimensional Bone Tissue Constructs: A Review. Tissue Eng. Part B Rev. 2010, 16, 587–601. [Google Scholar] [CrossRef]
- Vetsch, J.R.; Betts, D.C.; Müller, R.; Hofmann, S. Flow velocity-driven differentiation of human mesenchymal stromal cells in silk fibroin scaffolds: A combined experimental and computational approach. PLoS ONE 2017, 12, e0180781. [Google Scholar] [CrossRef]
- Zhao, F.; Vaughan, T.J.; McNamara, L.M. Quantification of fluid shear stress in bone tissue engineering scaffolds with spherical and cubical pore architectures. Biomech. Model. Mechanobiol. 2016, 15, 561–577. [Google Scholar] [CrossRef]
- Zhao, F.; Rietbergen, B.; Ito, K.; Hofmann, S. Fluid flow-induced cell stimulation in bone tissue engineering changes due to interstitial tissue formation in vitro. Int. J. Numer. Methods Biomed. Eng. 2020, 36, e3342. [Google Scholar] [CrossRef]
- Zhao, F.; Vaughan, T.J.; Mcnamara, L.M. Multiscale fluid–structure interaction modelling to determine the mechanical stimulation of bone cells in a tissue engineered scaffold. Biomech. Model. Mechanobiol. 2015, 14, 231–243. [Google Scholar] [CrossRef]
- Ali, D.; Ozalp, M.; Blanquer, S.B.G.; Onel, S. Permeability and fluid flow-induced wall shear stress in bone scaffolds with TPMS and lattice architectures: A CFD analysis. Eur. J. Mech. B/Fluids 2019, 79, 376–385. [Google Scholar] [CrossRef]
- Melchels, F.P.W.; Barradas, A.M.C.; van Blitterswijk, C.A.; de Boer, J.; Feijen, J.; Grijpma, D.W. Effects of the architecture of tissue engineering scaffolds on cell seeding and culturing. Acta Biomater. 2010, 6, 4208–4217. [Google Scholar] [CrossRef] [PubMed]
- Melchels, F.P.W.; Tonnarelli, B.; Olivares, A.L.; Martin, I.; Lacroix, D.; Feijen, J.; Wendt, D.J.; Grijpma, D.W. The influence of the scaffold design on the distribution of adhering cells after perfusion cell seeding. Biomaterials 2011, 32, 2878–2884. [Google Scholar] [CrossRef] [PubMed]
- Pires, T.; Dunlop, J.W.C.; Fernandes, P.R.; Castro, A.P.G. Challenges in computational fluid dynamics applications for bone tissue engineering. Proc. R. Soc. A Math. Phys. Eng. Sci. 2022, 478. [Google Scholar] [CrossRef]
- Metz, C.; Duda, G.N.; Checa, S. Towards multi-dynamic mechano-biological optimization of 3D-printed scaffolds to foster bone regeneration. Acta Biomater. 2019, 101, 117–127. [Google Scholar] [CrossRef]
- Boccaccio, A.; Fiorentino, M.; Uva, A.E.; Laghetti, L.N.; Monno, G. Rhombicuboctahedron unit cell based scaffolds for bone regeneration: Geometry optimization with a mechanobiology—Driven algorithm. Mater. Sci. Eng. C 2018, 83, 51–66. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Zhang, L.; Zhang, S.; Lu, W.F.; Hsi Fuh, J.Y. Triply Periodic Minimal Surfaces Sheet Scaffolds for Tissue Engineering Applications: An Optimization Approach toward Biomimetic Scaffold Design. ACS Appl. Bio Mater. 2018, 1, 259–269. [Google Scholar] [CrossRef]
- Uth, N.; Mueller, J.; Smucker, B.; Yousefi, A.-M. Validation of scaffold design optimization in bone tissue engineering: Finite element modeling versus designed experiments. Biofabrication 2017, 9, 015023. [Google Scholar] [CrossRef]
- Boccaccio, A.; Uva, A.E.; Fiorentino, M.; Mori, G.; Monno, G. Geometry design optimization of functionally graded scaffolds for bone tissue engineering: A mechanobiological approach. PLoS ONE 2016, 11, e0146935. [Google Scholar] [CrossRef]
- Boccaccio, A.; Uva, A.E.; Fiorentino, M.; Lamberti, L.; Monno, G. A Mechanobiology-based Algorithm to Optimize the Microstructure Geometry of Bone Tissue Scaffolds. Int. J. Biol. Sci. 2016, 12, 1–17. [Google Scholar] [CrossRef]
- Rodríguez-Montaño, Ó.L.; Cortés-Rodríguez, C.J.; Uva, A.E.; Fiorentino, M.; Gattullo, M.; Monno, G.; Boccaccio, A. Comparison of the mechanobiological performance of bone tissue scaffolds based on different unit cell geometries. J. Mech. Behav. Biomed. Mater. 2018, 83, 28–45. [Google Scholar] [CrossRef] [PubMed]
- Pires, T.; Santos, J.; Ruben, R.B.; Gouveia, B.P.; Castro, A.P.G.; Fernandes, P.R. Numerical-experimental analysis of the permeability-porosity relationship in triply periodic minimal surfaces scaffolds. J. Biomech. 2021, 117, 110263. [Google Scholar] [CrossRef]
- Castro, A.P.G.; Pires, T.; Santos, J.; Gouveia, B.P.; Fernandes, P.R. Permeability versus Design in TPMS Scaffolds. Materials 2019, 12, 1313. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.; Pires, T.; Gouveia, B.P.; Castro, A.P.G.; Fernandes, P.R. On the permeability of TPMS scaffolds. J. Mech. Behav. Biomed. Mater. 2020, 110, 103932. [Google Scholar] [CrossRef] [PubMed]
- Cignoni, P.; Callieri, M.; Corsini, M.; Dellepiane, M.; Ganovelli, F.; Ranzuglia, G. MeshLab: An open-source mesh processing tool. In Proceedings of the 6th Eurographics Italian Chapter Conference, Salerno, Italy, 2–4 July 2008; pp. 129–136. [Google Scholar] [CrossRef]
- Arjunan, A.; Demetriou, M.; Baroutaji, A.; Wang, C. Mechanical performance of highly permeable laser melted Ti6Al4V bone scaffolds. J. Mech. Behav. Biomed. Mater. 2019, 102, 103517. [Google Scholar] [CrossRef] [PubMed]
- Campos Marin, A.; Grossi, T.; Bianchi, E.; Dubini, G.; Lacroix, D. µ-Particle tracking velocimetry and computational fluid dynamics study of cell seeding within a 3D porous scaffold. J. Mech. Behav. Biomed. Mater. 2017, 75, 463–469. [Google Scholar] [CrossRef]
- Campos Marin, A.; Brunelli, M.; Lacroix, D. Flow perfusion rate modulates cell deposition onto scaffold substrate during cell seeding. Biomech. Model. Mechanobiol. 2017, 17, 675–687. [Google Scholar] [CrossRef]
- Chor, M.V.; Li, W. A permeability measurement system for tissue engineering scaffolds. Meas. Sci. Technol. 2007, 18, 208–216. [Google Scholar] [CrossRef]
- Ochoa, I.; García-Aznar, J.M.; Boccaccini, A.R.; Doblaré, M.; Sanz-Herrera, J.A.; Yunos, D.M. Permeability evaluation of 45S5 Bioglass®-based scaffolds for bone tissue engineering. J. Biomech. 2008, 42, 257–260. [Google Scholar] [CrossRef]
- Zhao, F.; Lacroix, D.; Ito, K.; van Rietbergen, B.; Hofmann, S. Changes in scaffold porosity during bone tissue engineering in perfusion bioreactors considerably affect cellular mechanical stimulation for mineralization. Bone Rep. 2020, 12, 100265. [Google Scholar] [CrossRef]
- Ali, D.; Sen, S. Computational Fluid Dynamics Study of the Effects of Surface Roughness on Permeability and Fluid Flow-Induced Wall Shear Stress in Scaffolds. Ann. Biomed. Eng. 2018, 46, 2023–2035. [Google Scholar] [CrossRef] [PubMed]
- Noordin, M.A.; Rahim, R.A.A.; Roslan, A.N.H.; Ali, I.A.; Syahrom, A.; Saad, A.P.M. Controllable Macroscopic Architecture of Subtractive Manufactured Porous Iron for Cancellous Bone Analogue: Computational to Experimental Validation. J. Bionic Eng. 2020, 17, 357–369. [Google Scholar] [CrossRef]
- Egan, P.F.; Gonella, V.C.; Engensperger, M.; Ferguson, S.J.; Shea, K. Computationally designed lattices with tuned properties for tissue engineering using 3D printing. PLoS ONE 2017, 12, e0182902. [Google Scholar] [CrossRef] [PubMed]

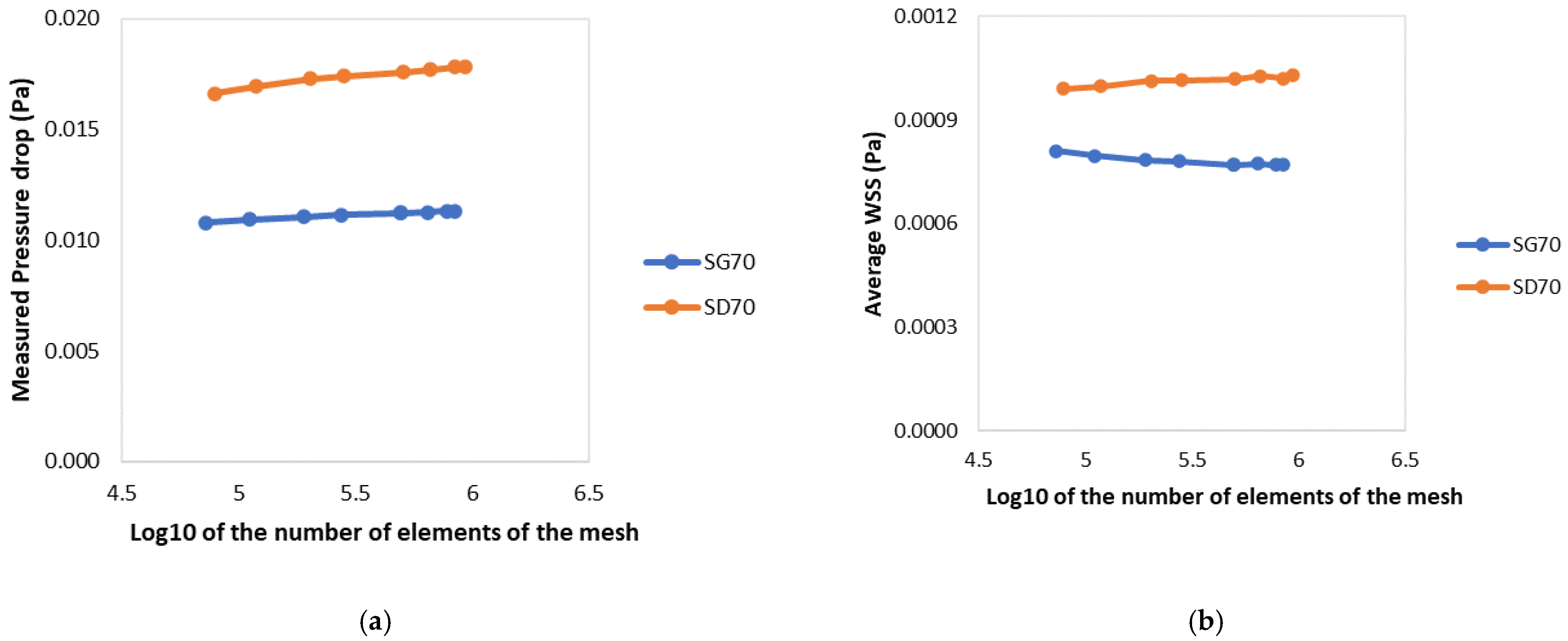

| World Unit | 0.095 | 0.1 | 0.125 | 0.15 | 0.175 | 0.2 | 0.25 | 0.3 |
| # Elements | 936246 | 840421 | 662816 | 503793 | 282630 | 203490 | 118248 | 78767 |
| Pressure Drop (mPa) | 17.846 | 17.817 | 17.713 | 17.587 | 17.401 | 17.283 | 16.937 | 16.635 |
| WSS (mPa) | 1.029 | 1.021 | 1.028 | 1.018 | 1.016 | 1.013 | 0.998 | 0.990 |
| World Unit | 0.095 | 0.1 | 0.125 | 0.15 | 0.175 | 0.2 | 0.25 | 0.3 |
| # Elements | 844429 | 777951 | 645069 | 499543 | 275998 | 190338 | 110911 | 72858 |
| Pressure Drop (mPa) | 11.319 | 11.305 | 11.260 | 11.210 | 11.131 | 11.061 | 10.922 | 10.790 |
| WSS (mPa) | 0.770 | 0.770 | 0.774 | 0.768 | 0.781 | 0.785 | 0.797 | 0.811 |
| SG70 Half Chamber | SG70 Full Chamber | SD70 Half Chamber | SD70 Full Chamber | |
|---|---|---|---|---|
| Pressure Drop (mPa) | 11.212 | 11.187 | 17.587 | 17.555 |
| Relative Difference (%) | −0.224 | −0.172 | ||
| Average WSS (mPa) | 0.768 | 0.768 | 1.018 | 1.021 |
| Relative Difference (%) | −0.028 | 0.287 | ||
| SG60 Original | SG60 Smoothed | SG70 Original | SG70 Smoothed | SG80 Original | SG80 Smoothed | |
|---|---|---|---|---|---|---|
| Average WSS (mPa) | 0.700 | 0.930 | 0.568 | 0.768 | 0.455 | 0.625 |
| Relative Difference (%) | 32.843 | 35.221 | 37.320 | |||
| SD60 Original | SD60 Smoothed | SD70 Original | SD70 Smoothed | SD80 Original | SD80 Smoothed | |
|---|---|---|---|---|---|---|
| Average WSS (mPa) | 0.955 | 1.278 | 0.749 | 1.018 | 0.607 | 0.821 |
| Relative Difference (%) | 33.826 | 35.915 | 35.180 | |||
| G60 | G70 | G80 | SD60 | SD70 | SD80 | |
|---|---|---|---|---|---|---|
| Original | 599.5 | 700.5 | 799.8 | 600.8 | 699.1 | 800.5 |
| Smoothed | 599.4 | 701.4 | 801.6 | 599.1 | 699.2 | 801.71 |
| Relative Difference (%) | −0.012 | 0.128 | 0.216 | −0.280 | 0.026 | 0.155 |
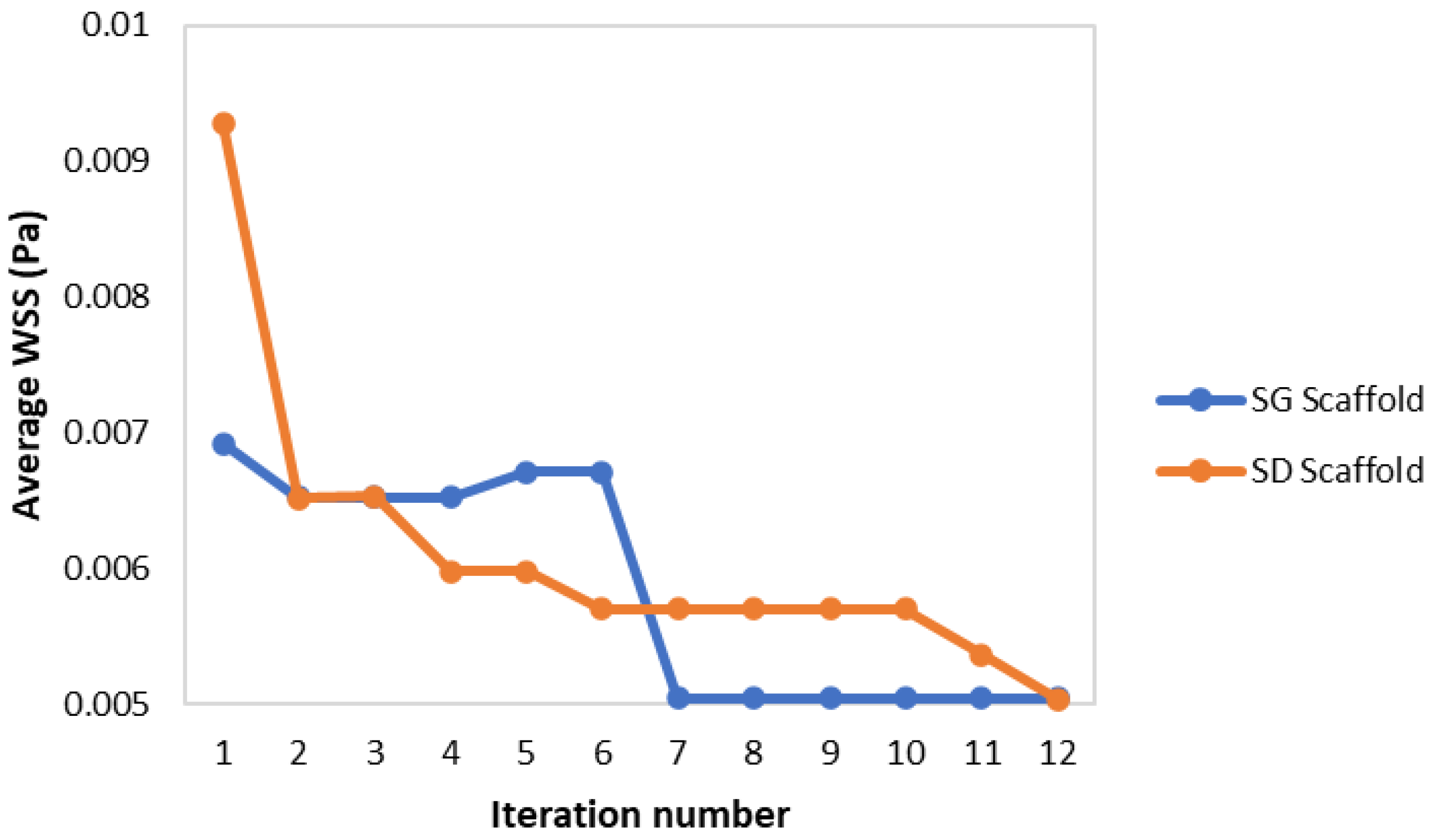
| Optimization Iteration | Cubic Unit Length (mm) | Porosity (%) | Average WSS (mPa) |
|---|---|---|---|
| 1 | 5.500 | 70 | 9.277 |
| 2 | 7.375 | 73.1 | 6.515 |
| 3 | 7.735 | 70.8 | 6.531 |
| 4 | 9.523 | 65.4 | 5.978 |
| 6 | 10.000 | 65.4 | 5.697 |
| 11 | 10.000 | 68.2 | 5.361 |
| 12 | 10.000 | 71.2 | 5.039 |
| Optimization Iteration | Cubic Unit Length (mm) | Porosity (%) | Average WSS (mPa) |
|---|---|---|---|
| 1 | 5.500 | 70 | 6.916 |
| 2 | 6.183 | 67.2 | 6.522 |
| 5 | 6.081 | 66.5 | 6.710 |
| 7 | 7.320 | 71.6 | 5.041 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pires, T.H.V.; Dunlop, J.W.C.; Castro, A.P.G.; Fernandes, P.R. Wall Shear Stress Analysis and Optimization in Tissue Engineering TPMS Scaffolds. Materials 2022, 15, 7375. https://doi.org/10.3390/ma15207375
Pires THV, Dunlop JWC, Castro APG, Fernandes PR. Wall Shear Stress Analysis and Optimization in Tissue Engineering TPMS Scaffolds. Materials. 2022; 15(20):7375. https://doi.org/10.3390/ma15207375
Chicago/Turabian StylePires, Tiago H. V., John W. C. Dunlop, André P. G. Castro, and Paulo R. Fernandes. 2022. "Wall Shear Stress Analysis and Optimization in Tissue Engineering TPMS Scaffolds" Materials 15, no. 20: 7375. https://doi.org/10.3390/ma15207375
APA StylePires, T. H. V., Dunlop, J. W. C., Castro, A. P. G., & Fernandes, P. R. (2022). Wall Shear Stress Analysis and Optimization in Tissue Engineering TPMS Scaffolds. Materials, 15(20), 7375. https://doi.org/10.3390/ma15207375







