Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
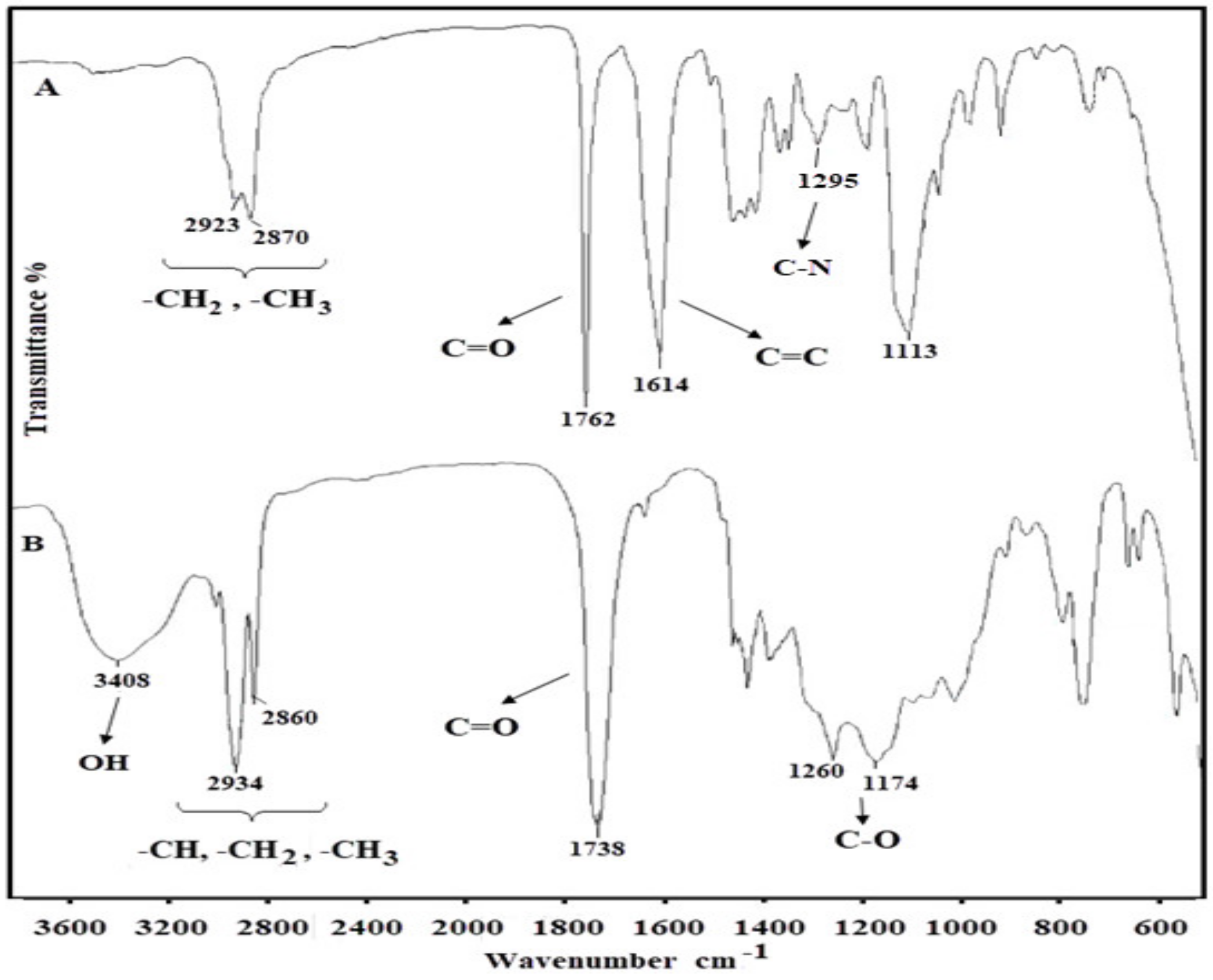
2.2.1. Fourier Transform Infrared (FTIR) Spectra
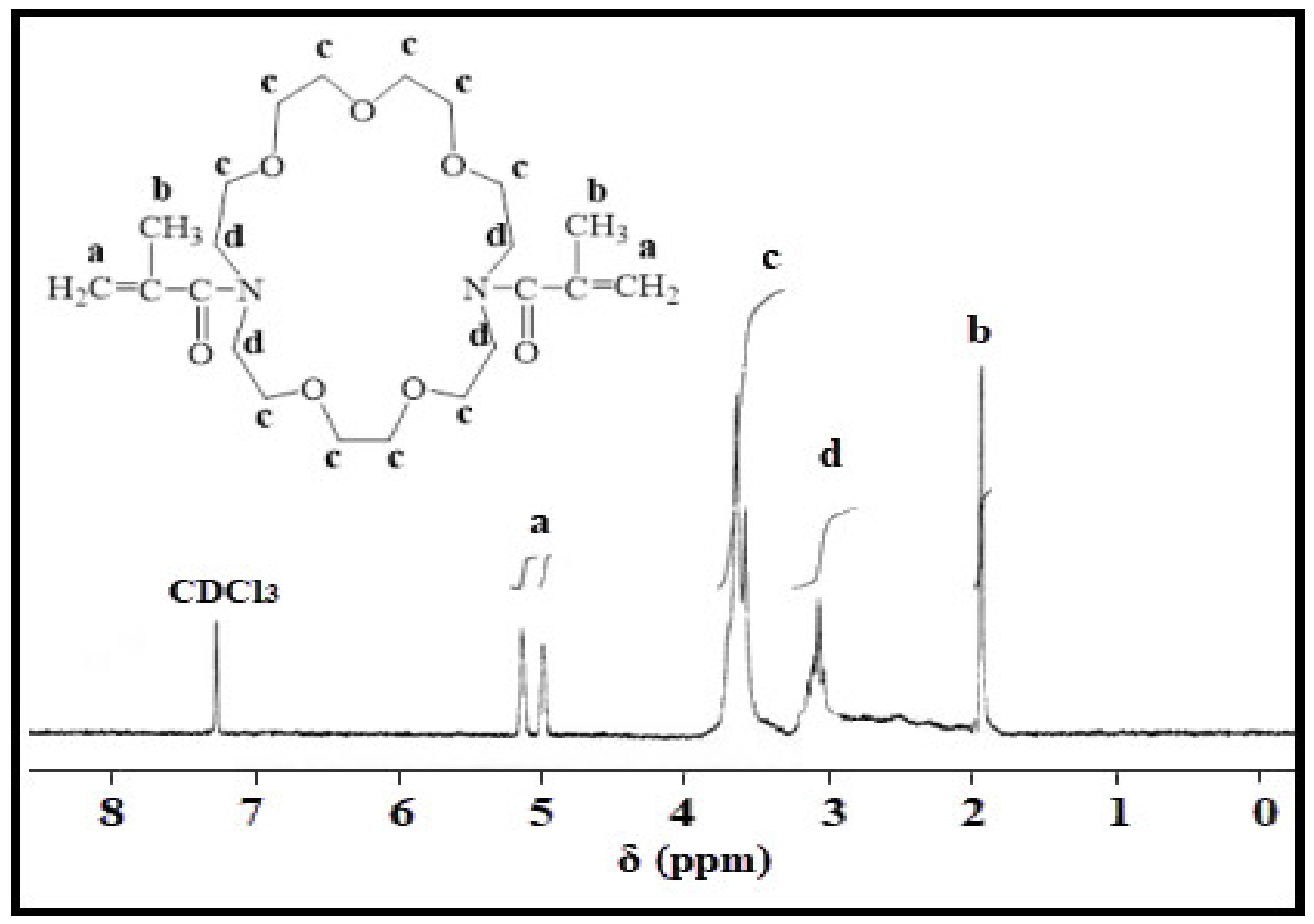
2.2.2. 1H NMR Spectra
2.2.3. Atomic Absorption Spectrometry (AAS)
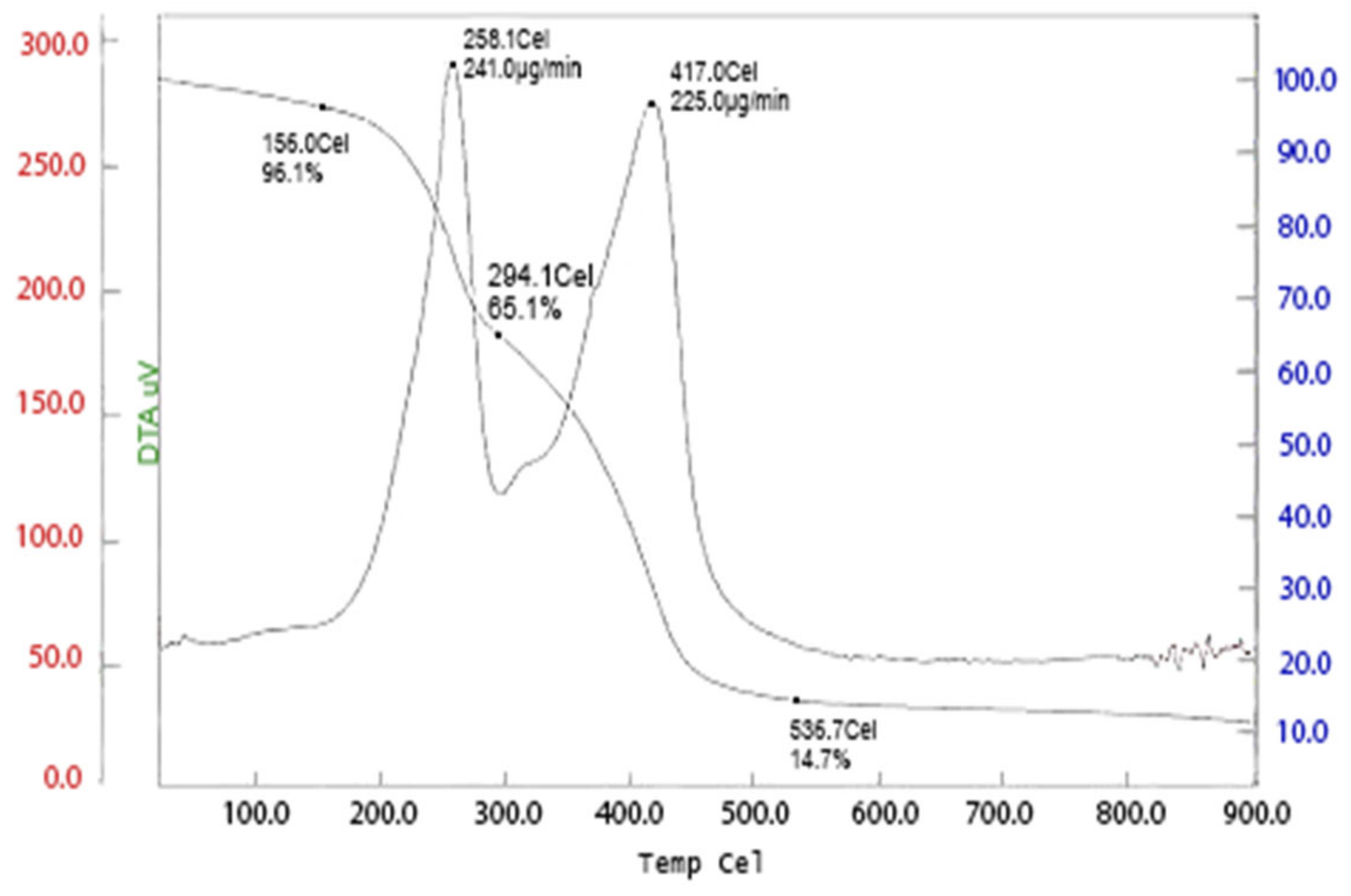
2.2.4. Thermal Gravimetric Analysis (TGA)
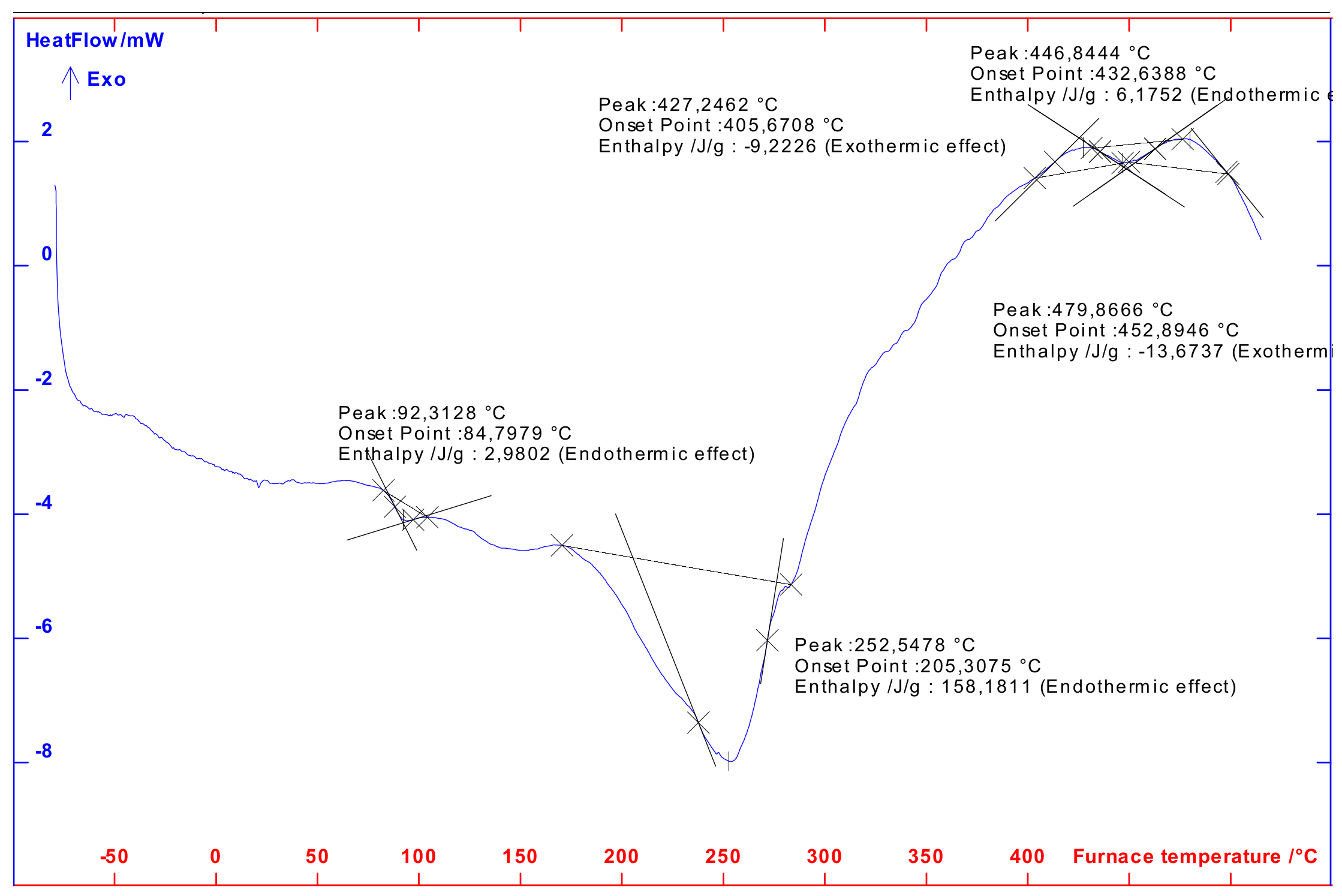
2.2.5. Differential Scanning Calorimeter (DSC)
2.2.6. Scanning Electron Microscopy (SEM)
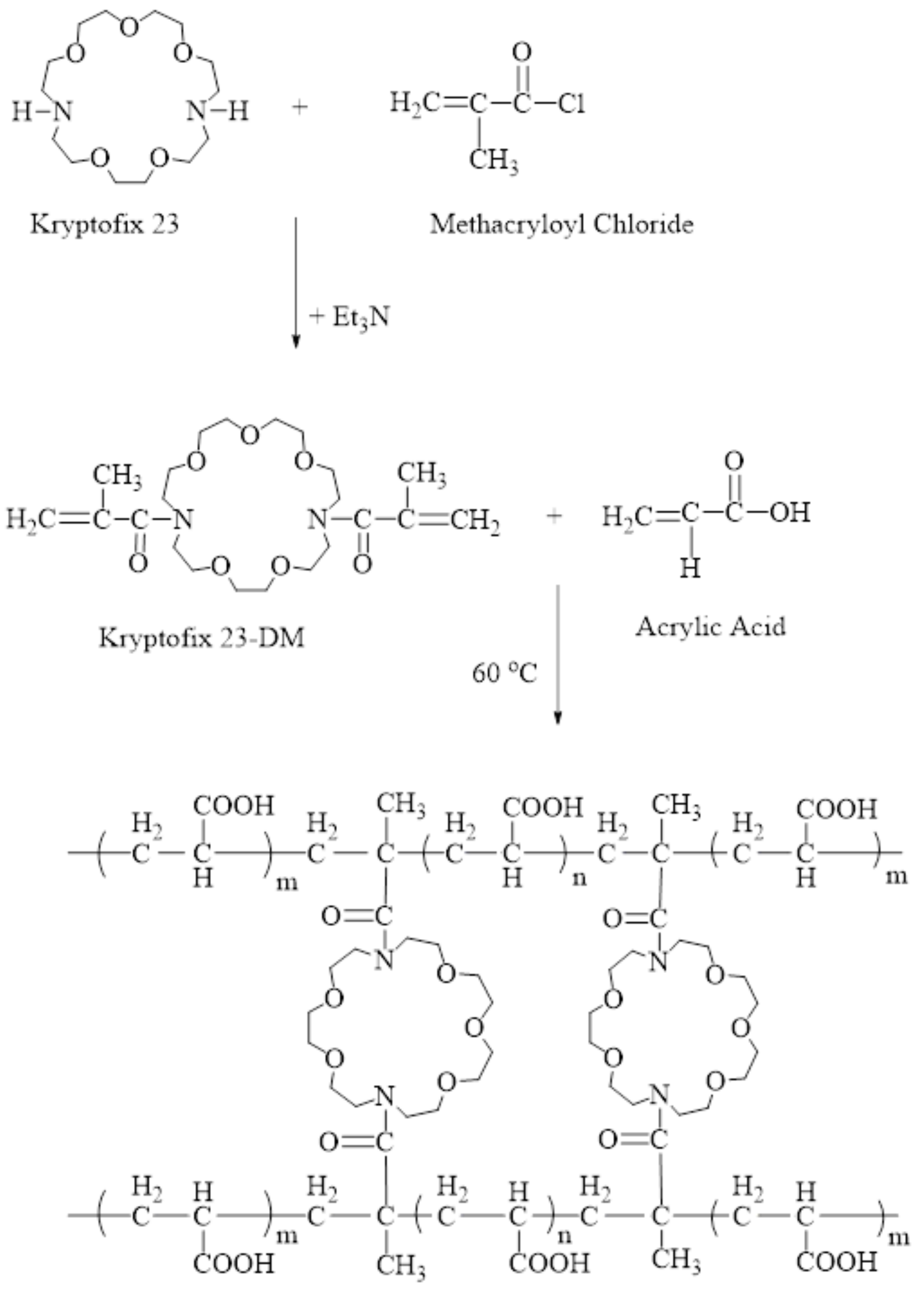
3. Synthesis
3.1. Synthesis of Kry23-DM
3.2. Synthesis of the P (AA/Kry23-DM) SAP
3.3. Computational Method
3.4. Sorption Experiments of the P (AA/Kry23-DM) SAP
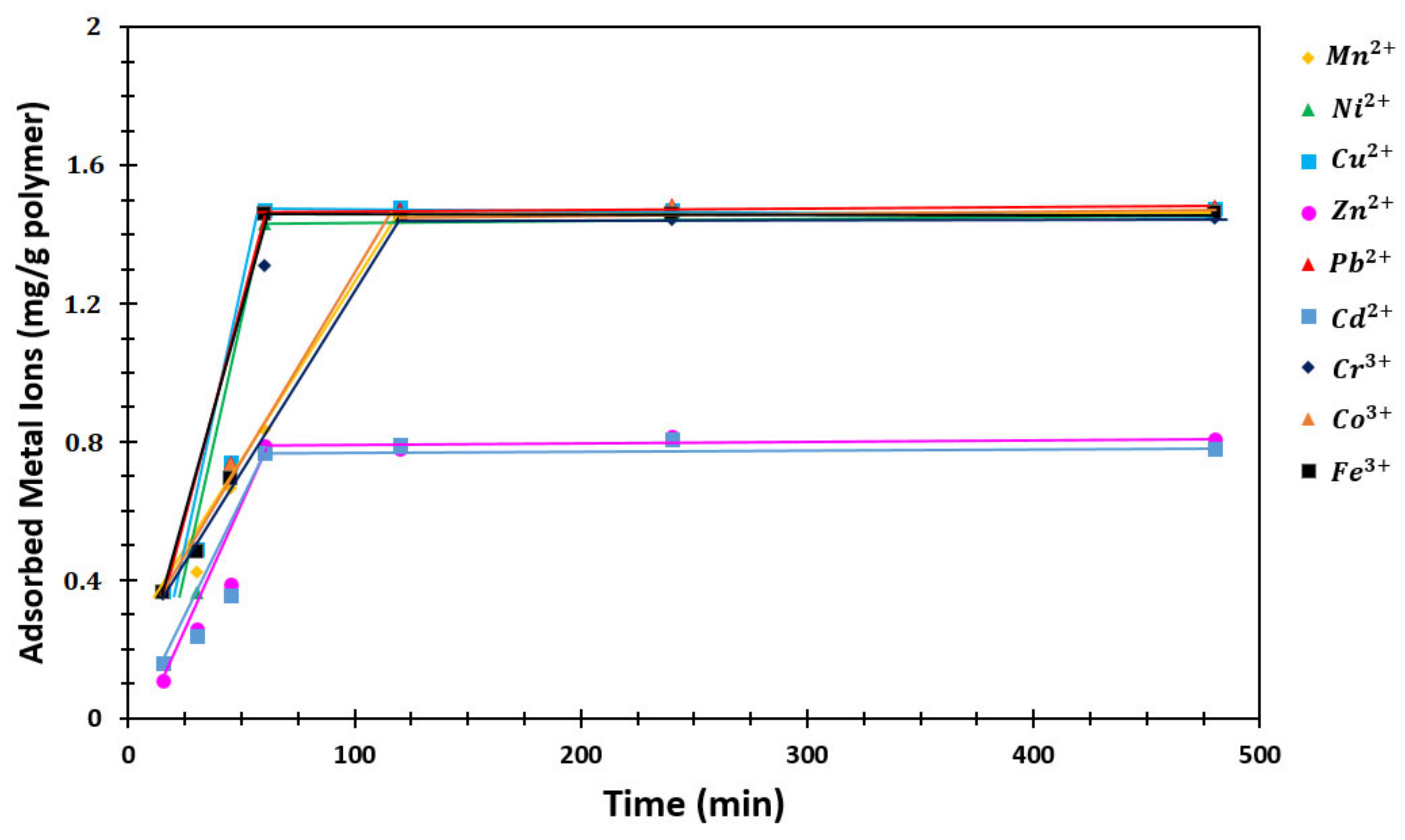
3.4.1. Sorption Rate
3.4.2. Sorption Capacity
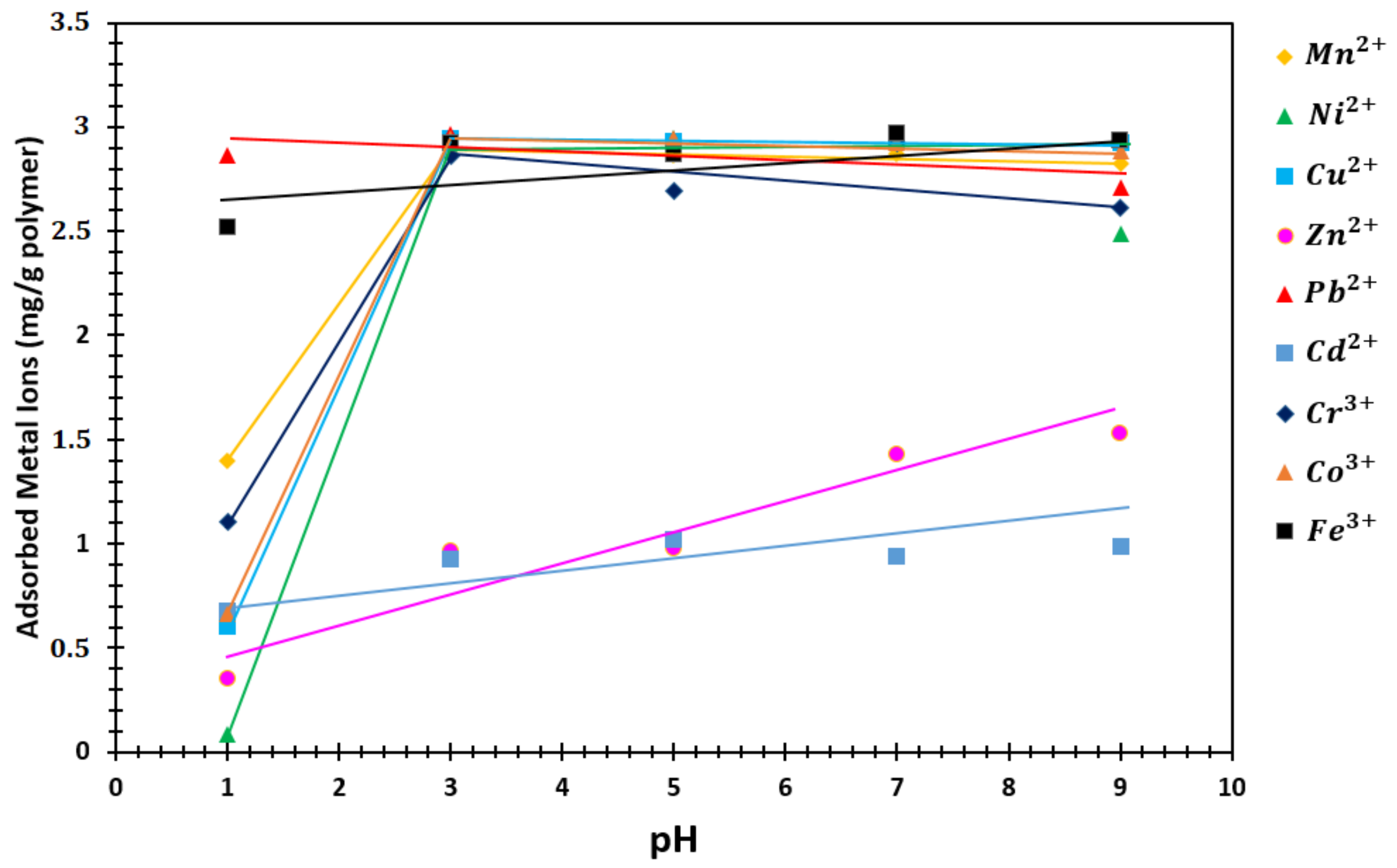
3.4.3. pH Effect on Sorption Capacity
3.4.4. Competitive Sorption
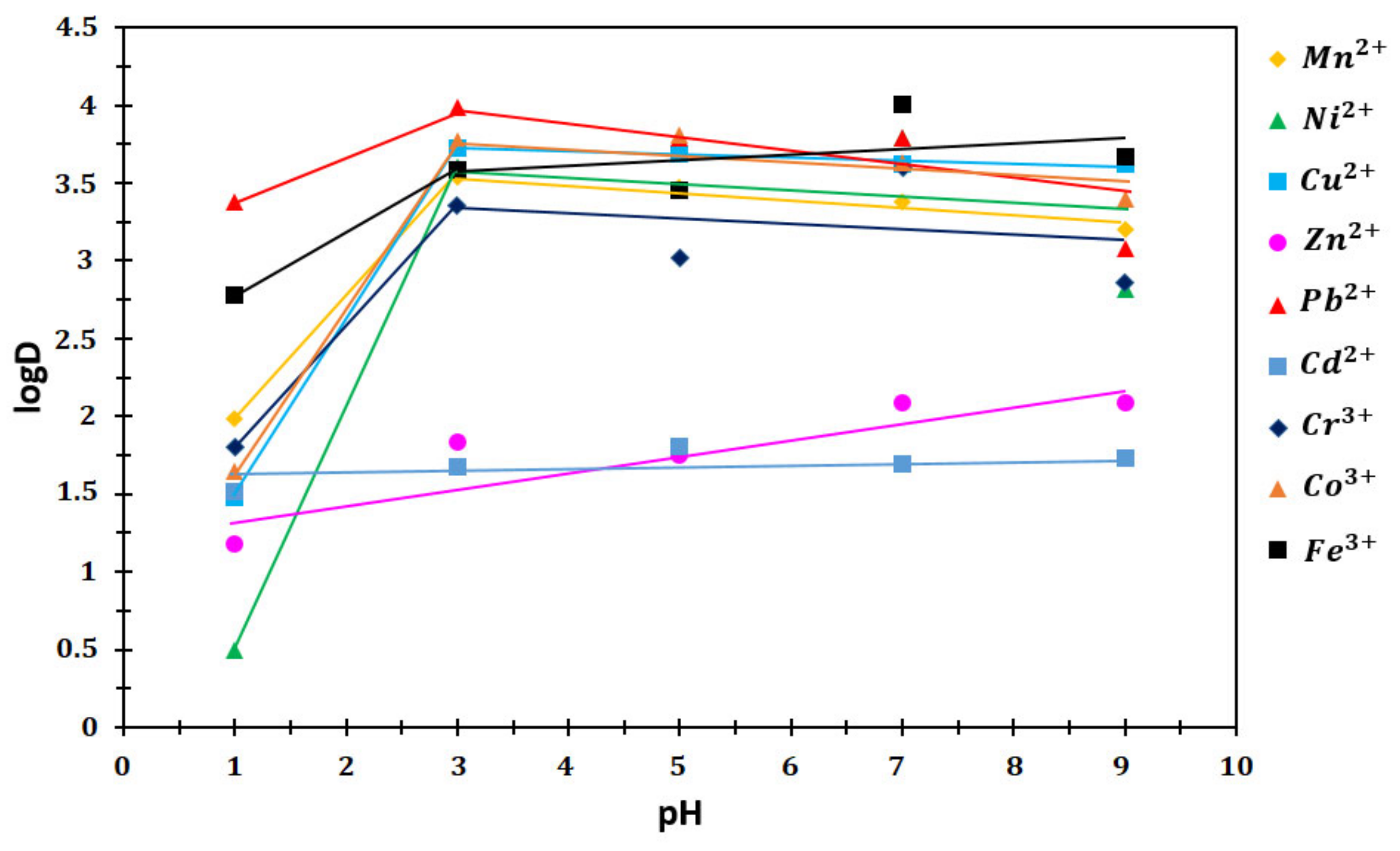
3.4.5. Distribution Ratio (log D)
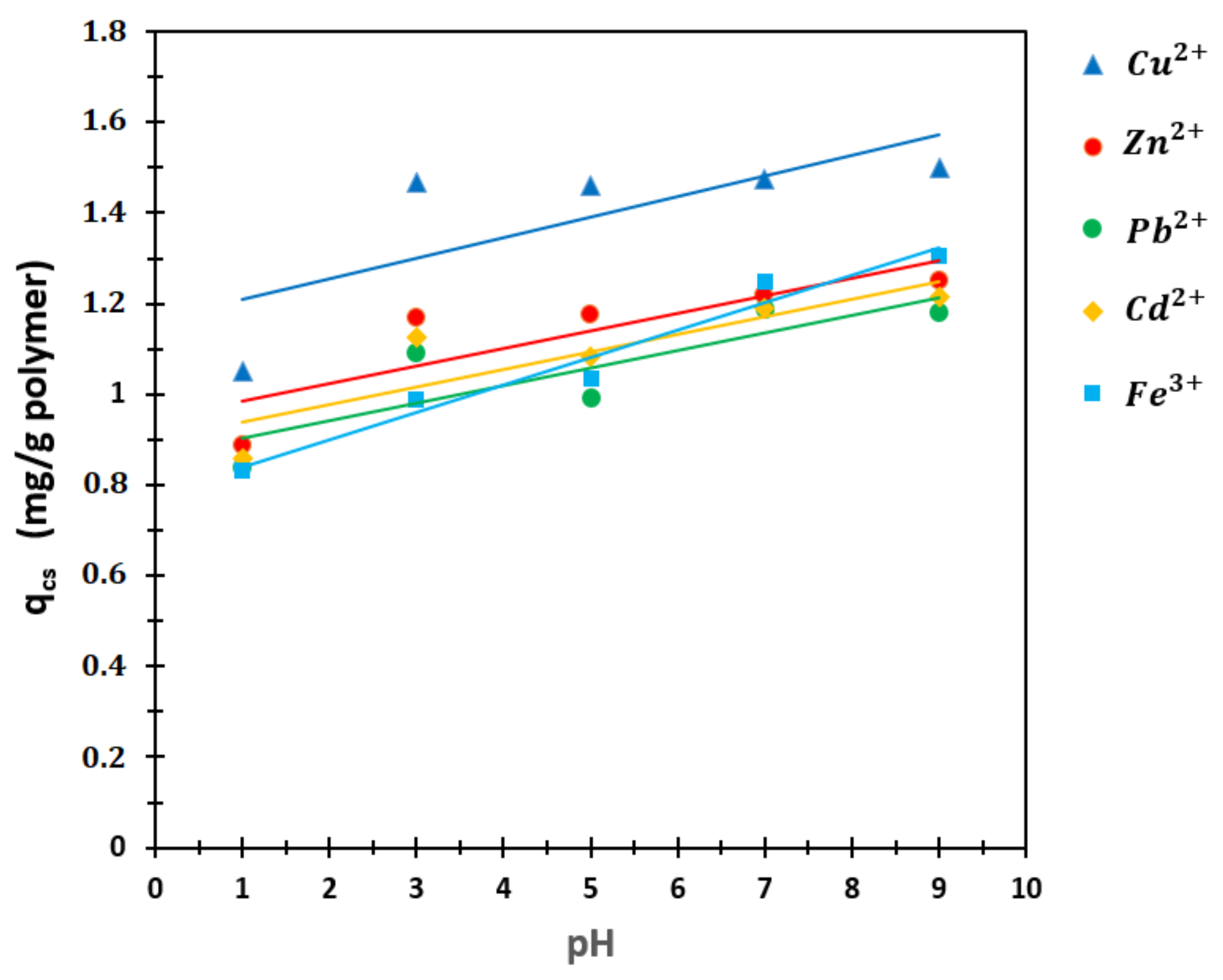
3.4.6. Swelling Ratio, qv
3.4.7. Desorption
3.4.8. Freundlich and Langmuir Isotherm Applications
4. Results and Discussion
4.1. Synthesis of Kry23-DM Cross-Linker and P (AA/Kry23-DM) SAP
4.2. Characterization of Kry23-DM Cross-Linker
4.3. Characterization of P (AA/Kry23-DM) SAP
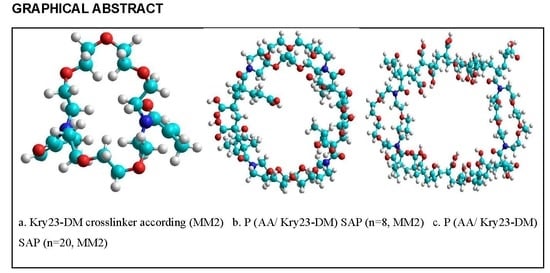
4.4. Computational Method
4.5. Sorption Experiments of the P (AA/Kry23-DM) SAP
4.5.1. Sorption Rate
4.5.2. Sorption Capacity
4.5.3. pH Effect on Sorption Capacity
4.5.4. Competitive Sorption
4.5.5. The Distribution Coefficients (log D)
4.5.6. The Swelling Ratios (qv)
4.5.7. Desorption
4.5.8. Adsorption Isotherms
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sakiyama, T.; Tsutsui, T.; Masuda, E.; Imamura, K.; Nakanishi, K. Ionization Characteristics of Polyelectrolyte Complex Gels: Analysis Based on Their Swelling Behaviors. Macromolecules 2003, 36, 5039–5042. [Google Scholar] [CrossRef]
- Dutkiewicz, J.K. Superabsorbent Materials from Shellfish Waste—A Review. J. Biomed. Mater. Res. 2002, 63, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Shiga, T.; Hirose, Y.; Okada, A.; Kurauchi, T.J. Bending of poly (vinyl alcohol)—Poly (sodium acrylate) composite hydrogel in electric fields. J. Appl. Polym. Sci. 1992, 44, 249–253. [Google Scholar] [CrossRef]
- Gao, L.; Wang, S.; Zhao, X. Synthesis and characterization of agricultural controllable humic acid superabsorbent. J. Environ. Sci. 2013, 25, 69–76. [Google Scholar] [CrossRef]
- Miyajima, T.; Matsubara, Y.; Komatsu, H.; Miyamoto, M.; Suzuki, K. Development of a superabsorbent polymer using iodine transfer polymerization. Polym. J. 2020, 52, 365–373. [Google Scholar] [CrossRef]
- Ma, X.; Wen, G. Development history and synthesis of super-absorbent polymers: A review. J. Polym. Res. 2020, 27, 373–381. [Google Scholar] [CrossRef]
- Mahon, R.; Balogun, Y.; Oluyemi, G.; Njuguna, J. Swelling performance of sodium polyacrylate and poly (acrylamide-co-acrylic acid) potassium salt. SN Appl. Sci. 2020, 2, 117–132. [Google Scholar] [CrossRef]
- Nnadi, F.; Brave, C. Environmentally friendly superabsorbent polymers for water conservation in agriculture lands. J. Soil Sci. Environ. Manage. 2011, 2, 206–211. [Google Scholar]
- Flory, P.J. Principles of Polymer Chemistry; Cornell University Press: Itaca, NY, USA, 1953; pp. 589–5915. [Google Scholar]
- Lee, W.Y.; Wu, R.I. Studies on the synthesis of water-absorbing polymers. J. Appl. Polym. Sci. 1996, 62, 1009–1017. [Google Scholar] [CrossRef]
- Liu, Z.S.; Rempel, G.L. Preparation of superabsorbent polymers by crosslinking acrylic acid and acrylamide copolymers. J. Appl. Polym. Sci. 1997, 64, 1345–1353. [Google Scholar] [CrossRef]
- Chen, J.; Zhao, Y. Relationship between water absorbency and reaction conditions in aqueous solution polymerization of polyacrylate superabsorbents. J. Appl. Polym. Sci. 2000, 75, 808–814. [Google Scholar] [CrossRef]
- Walker, M.; Hobot, J.A.; Newman, G.R.; Bowler, P.G. Scanning electron microscopic examination of bacterial immobilization in a carboxymethyl cellulose (AQUACEL) and alginate dressings. Biomaterials 2003, 24, 883–890. [Google Scholar] [CrossRef]
- Wang, G.J.; LI, M.; Chen, X.F. Preparation and water-absorbent properties of a water-swellable rubber. J. Appl. Polym. Sci. 1998, 686, 1219–1224. [Google Scholar] [CrossRef]
- Li, A.; Wang, A.Q.; Chen, J.M. Studies on Poly (acrylic acid)/Attapulgite Superabsorbent Composite. I. Synthesis and Characterization. J. Appl. Polym. Sci. 2004, 92, 1596–1603. [Google Scholar] [CrossRef]
- Chiron, N.; Guilet, R.; Deydier, E. Adsorption of Cu (II) and Pb (II) onto a grafted silica: Isotherms and kinetic models. Water Res. 2003, 37, 3079–3086. [Google Scholar] [CrossRef]
- Ngah, W.S.W.; Endud, C.S.; Mayanar, R. Removal of copper (II) ions from aqueous solution onto chitosan and crosslinked chitosan beads. React. Funct. Polym. 2002, 50, 181–190. [Google Scholar] [CrossRef]
- Elbedwehy, A.M.; Abou-Elanwar, A.M.; Ezzat, A.O.; Atta, A.M. Super effective removal of toxic metals water pollutants using multi functionalized polyacrylonitrile and arabic gum grafts. Polymers 2019, 11, 1938–1954. [Google Scholar] [CrossRef]
- Ramdani, A.; Kadeche, A.; Adjdir, M.; Taleb, Z.; Ikhou, D.; Taleb, S.; Deratani, A. Lead and cadmium removal by adsorption process using hydroxyapatite porous materials. Water Pract. Technol. 2020, 15, 130–141. [Google Scholar] [CrossRef]
- Aalaie, J.; Rahmatpour, A. Preparation and swelling behavior of partially hydrolyzed polyacrylamide nanocomposite hydrogels in electrolyte solutions. J. Macromol. Sci. Phys. 2008, 47, 98–108. [Google Scholar] [CrossRef]
- Kasgoz, H.; Ozgumus, S.; Orbay, M. Modified polyacrylamide hydrogels and their application in removal of heavy metal ions. Polymer 2003, 44, 1785–1793. [Google Scholar] [CrossRef]
- Xie, J.J.; Liu, X.R.; Liang, J.F. Absorbency and adsorption poly (acrylic acid-co-acryl-amide) hydrogel. J. Appl. Polym. Sci. 2007, 106, 1606–1613. [Google Scholar] [CrossRef]
- Rivas, B.L.; Maturana, H.A.; Molina, M.J.; Gómez-Aantón, M.R.; Piérola, L.F. Metal ion binding properties of poly (N-vinylimidazole) hydrogels. J. Appl. Polym. Sci. 1998, 67, 1109–1118. [Google Scholar] [CrossRef]
- Kasgoz, H.; Kasgoz, A.; Sahin, U.; Temelli, T.Y.; Bayat, C. Hydrogels with acid groups for removal of copper (II) and lead (II) ions. Polym. Plast. Technol. Eng. 2006, 45, 117–124. [Google Scholar] [CrossRef]
- Rivas, B.L.; Seguel, G.V.; Geckeler, K.E. Synthesis, Characterization, and Properties of Polychelates of Poly (Styrene Sulfonic Acid-co-Maleic Acid) with Co (II), Cu (II), Ni (II), and Zn (II). J. Appl. Polym. Sci. 2002, 85, 2546–2551. [Google Scholar] [CrossRef]
- Rivas, B.L.; Pooley, S.A.; Maturana, H.A.; Villegas, S. Metal ion uptake properties of acrylamide derivative resins. Macromol. Chem. Phys. 2001, 202, 443–447. [Google Scholar] [CrossRef]
- Fonseca, R.J.D.A.; Segatelli, M.G.; Borges, K.B.; Tarley, C.R.T. Synthesis and evaluation of different adsorbents based on poly (methacrylic acid–trimethylolpropane trimethacrylate) and poly (vinylimidazole–trimethylolpropane trimethacrylate) for the adsorption of tebuthiuron from aqueous medium. React Funct. Polym. 2015, 93, 1–9. [Google Scholar] [CrossRef]
- Dragan, E.S.; Dinu, M.V. Advances in porous chitosan-based composite hydrogels: Synthesis and applications. React Funct. Polym. 2020, 146, 104372. [Google Scholar] [CrossRef]
- Rosa, F.; Bordado, J.; Casquilho, M. Kinetics of water absorbency in AA-AMPS copolymers: Applications of a diffusion-relaxation model. Polymer 2002, 43, 63–70. [Google Scholar] [CrossRef]
- Dadhaniya, P.V.; Patel, M.P.; Patel, R.G. Removal of anionic dyes from aqueous solution using poly [N-vinyl pyrrolidone/2-(methacryloyloxyethyl) trimethyl ammonium chloride] superswelling hydrogels. Polym. Bull. 2007, 58, 359–369. [Google Scholar] [CrossRef]
- Kasgoz, H. New sorbent hydrogels for removal of acidic dyes and metal ions from aqueous solutions. Polym. Bull. 2006, 56, 517–528. [Google Scholar] [CrossRef]
- Devine, M.D.; Higginbotham, C.L. Synthesis and characterisation of chemically crosslinked NVP based hydrogel. Eur. Polym. J. 2005, 41, 1272–1279. [Google Scholar] [CrossRef]
- Ponder, J.W.; Case, D.A. Force fıelds for proteın simulations. In Advances in Protein Chemistry; Elsevier Inc.: Berkeley, CA, USA, 2003; pp. 27–78. [Google Scholar]
- Yilmaz, S.S.; Kul, D.; Erdöl, M.; Özdemir, M.; Abbasoğlu, R. Synthesis of a novel crosslinked superabsorbent copolymer with diazacyclooctadecane crown ether and its sorption capability. Eur. Polym. J. 2007, 43, 1923–1932. [Google Scholar] [CrossRef]
- Allinger, N.; Kollman, P.; Jorgensen, W.; Karplus, M.; Poljack, J.R.; Huber, R.; Sieker, L.C.; Blundell, T.L.; Kim, S.H.; Teeter, M.M.; et al. Hyperchem, Version 8.0.; Hypercube Inc.: Gainesville, FL, USA, 2000. [Google Scholar]
- Dewar, M.S.J.; Zoebisch, E.G.; Healy, E.F.; Stewart, J.J.P. Development and use of quantum mechanical molecular models. AM1: A new general purpose quantum mechanical molecular model. J. Am. Chem. Soc. 1985, 107, 3902–3909. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; et al. Gaussian 03, revision D.02; Gaussian Inc.: Wallingford, CT, USA, 2004. [Google Scholar]
- Rigaudy, J.; Klesney, S.P. Nomenclature of Organic and Polymer Chemistry, Sections D, E, F and H, 1st ed.; Pergamon Press: Oxford, UK, 1979; pp. 386–390. [Google Scholar]
- Yilmaz, S.S.; Abbasoglu, R.; Hazer, B. A molecular mechanics and semiempirical molecular orbital study on the conformation of polynorbornene chains. J. Molec. Modell. 2003, 9, 230–234. [Google Scholar] [CrossRef]
- Ozturk, T.; Yılmaz, S.S.; Hazer, B.; Menceloglu, Y.Z. ATRP of methyl methacrylate initiated with a bifunctional initiator bearing bromomethyl functional groups: Synthesis of the block and graft copolymers. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 1364–1373. [Google Scholar] [CrossRef]
- Yılmaz, S.S. Theoretical investigation on the conformation of ethylene-norbornene copolymer chains. Indian J. Chem. 2007, 46A, 923–992. [Google Scholar]
- Wang, X.; Jing, S.; Hou, Z.; Liu, Y.; Qiu, X.; Liu, Y.; Tan, Y. Permeable, robust and magnetic hydrogel beads: Water droplet templating synthesis and utilization for heavy metal ions removal. J. Mater. Sci. 2018, 53, 15009–15024. [Google Scholar] [CrossRef]
- Marcus, Y. Ionic radii in aqueous solutions. Chem. Rev. 1988, 88, 98–1475. [Google Scholar] [CrossRef]
- Pearson, I. Hydrated metal ions in aqueous solution: How regular are their structures. Pure Appl. Chem. 2010, 82, 1901–1917. [Google Scholar] [CrossRef]
- Weber, E.; Toner, J.L.; Patai, S.; Rappoport, Z. Crown Ethers and Analogs; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 1989. [Google Scholar]
- Franks, F. Water; RSC Publishing: London, UK, 1983. [Google Scholar]
- Brian, A.O.; Hirotaka, O.; Yoshiki, H.; Kazuhiro, H. Poly (1, 4-diazocane-5, 8-dione) macrocyclic-functionalized hydrogel for high selectivity transition metal ion adsorption. React. Func. Polym. 2018, 125, 11–19. [Google Scholar] [CrossRef]
- Zuming, J.; Xulong, C.; Lanlei, G. Synthesis and swelling behavior of poly (acrylic acid-acryl amide2-acrylamido-2-methyl-propansulfonic acid) superabsorbent copolymer. J. Petrol. Explor. Prod. Technol. 2017, 7, 69–75. [Google Scholar] [CrossRef]
- Burugapalli, K.; Bhatia, D.; Koul, V.; Choudhary, V. Interpenetrating polymer networks based on poly (acrylic acid) and gelatin. I: Swelling and thermal behavior. J. Appl. Polym. Sci. 2001, 82, 217–227. [Google Scholar] [CrossRef]
- Moharram, M.A.; Mousa, A.A. Study of the interaction of poly (acrylic acid) and poly (acrylic acid-poly acrylamide) complex with bone powders and hydroxyapatite by using TGA and DSC. J. Appl. Polym. Sci. 2007, 105, 3220–3227. [Google Scholar] [CrossRef]
- Cardenas, G.; Munfoz, C.; Carbacho, H. Thermal properties and TGA-FTIR studies of polyacrylic and polymethacrylic acid doped with metal clusters. Euro. Polym. J. 2000, 36, 1091–1099. [Google Scholar] [CrossRef]
- Krusic, M.K.; Dzunuzovic, E.; Trifunovic, S.; Filipovic, S. Polyacrylamide and poly (ita-conic acid) complexes. Euro. Polym. J. 2004, 40, 793. [Google Scholar] [CrossRef]
- Ouyang, D.; Zhuo, Y.; Hu, L.; Zeng, Q.; Hu, Y.; He, Z. Research on the adsorption behavior of heavy metal ıons by porous material prepared with silicate tailings. Minerals 2019, 9, 291. [Google Scholar] [CrossRef]
- Boomhower, A.E. The Removal of Cd (Ii) from Water by Activated Carbon Process. Master’s. Thesis, University of Delaware, Newark, DE, USA, 1982. [Google Scholar]
- Caban, R.T.; Carmen, A.; Vega, O.; Camilde, N.M. Adsorption of Ni2+ and Cd2+ from Water by Calcium Alginate/Spent Coffee Grounds Composite. Appl. Sci. 2019, 9, 4531. [Google Scholar] [CrossRef]
- Reed, B.E.; Nonavinakere, S.K. Metal adsorption by activated carbon: Effect of complexing ligands, competitive adsorbates, ionic strength and background electrolyte. Sep. Sci. Technol. 1992, 27, 1985–1998. [Google Scholar] [CrossRef]
- Mohan, Y.M.; Murthy, P.S.K.; Raju, K.M. Synthesis, characterization and effect of reaction parameters on swelling properties of acrylamide–sodium methacrylate superabsorbent copolymers. React. Func. Polym. 2005, 63, 11–26. [Google Scholar] [CrossRef]
- Huang, D.; Wang, W.; Yuru Kang, Y.; Wang, A. Efficient adsorption and recovery of Pb(II) from aqueous solution by a granular pH-sensitive chitosan-based semi-IPN hydrogel. J. Macromol. Part A Pure Appl. Chem. 2012, 49, 971–979. [Google Scholar] [CrossRef]
- Mignon, G.; Graulus, J.; Snoeck, D.; Martins, J.; Belie, N.D.; Dubruel, P.; Van Vlierberghe, S. PH-sensitive superabsorbent polymers: A potential candidate material for self-healing concrete. J. Mater. Sci. 2015, 50, 970–979. [Google Scholar] [CrossRef]
- Reed, B.E.; Matsumoto, M.R. Modeling cadmium adsorption by activated carbon using the Langmuir and Freundlich Isotherm Expressions. Sep. Sci. Technol. 1993, 28, 2179. [Google Scholar] [CrossRef]
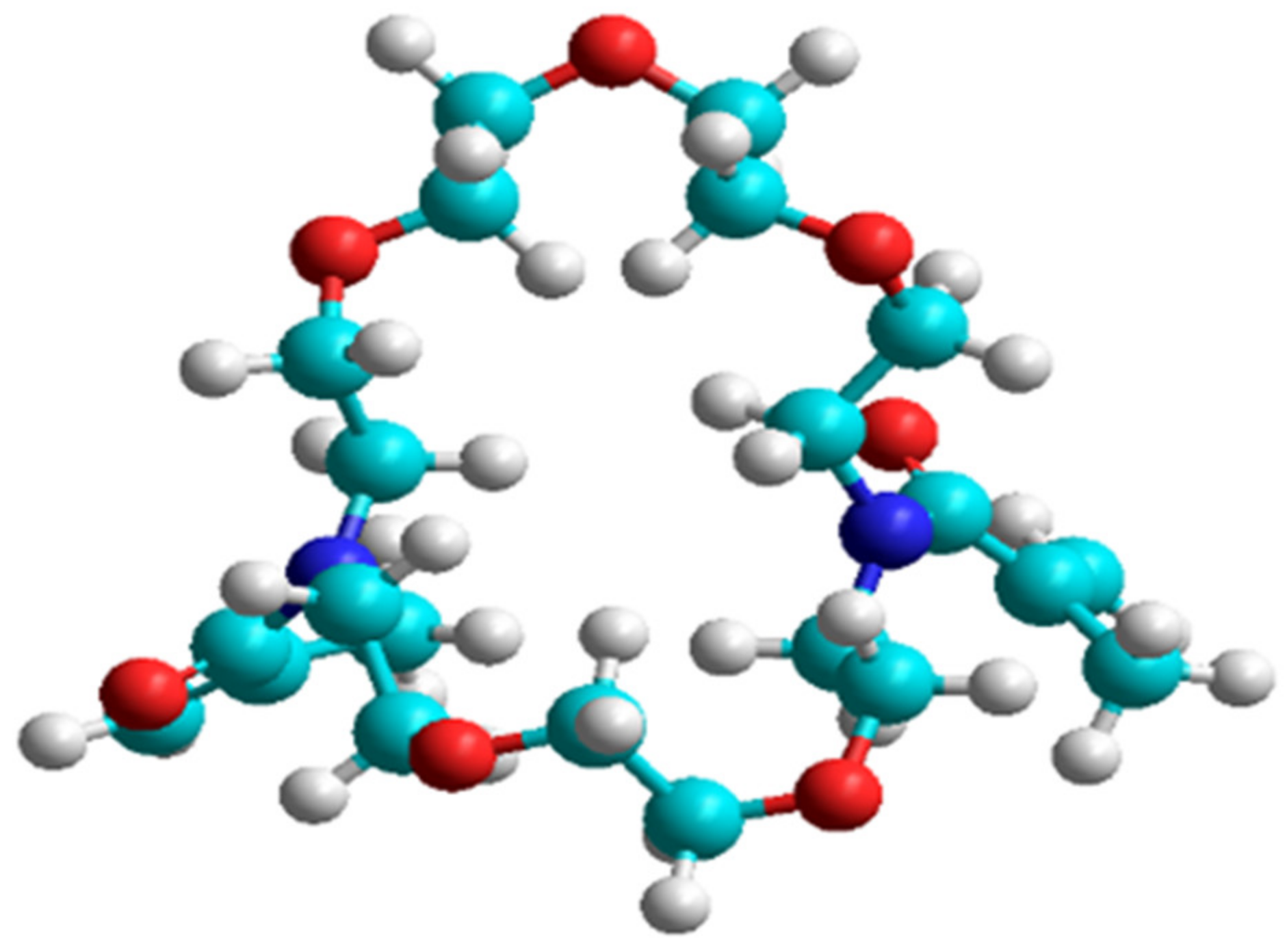

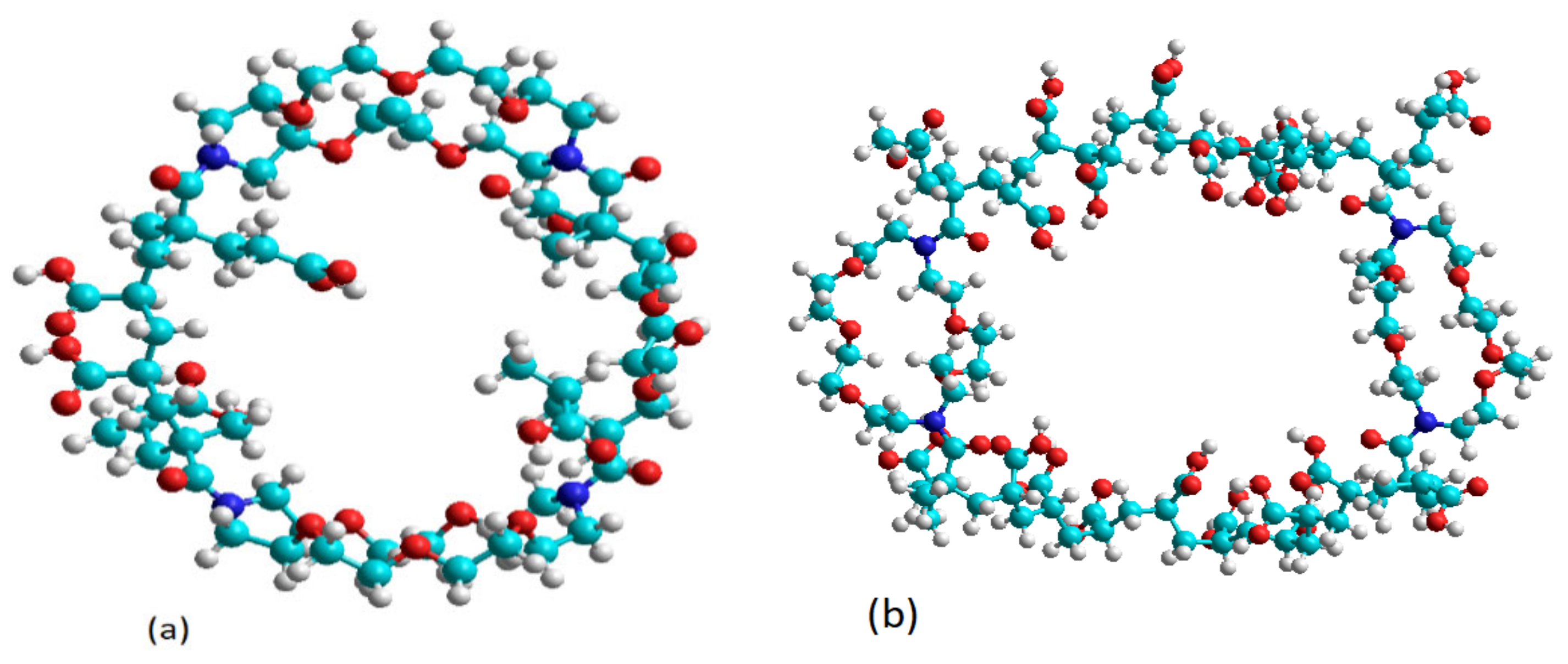

| Calculation Parameters | Quantum Method | Cross-linker | PAA (n = 8) | PAA (n = 20) |
|---|---|---|---|---|
| ΔfH0(kcal mol−1) | AM1 | −6580.002 | −21,083.115 | −32,872.784 |
| Estr (kcal mol−1) | MM2 | 45.465 | 110.954 | 156.202 |
| AMBER | 44.434 | 162.305 | 207.171 | |
| OPLS | 31.802 | 90.816 | 110.439 | |
| φ (°) | MM2 | 173.038 | 175.209 | 174.394 |
| AMBER | 171.574 | 168.767 | 171.372 | |
| OPLS | 172.267 | 170.765 | 169.816 | |
| d (Å) | MM2 | – | 1.549 | 1.547 |
| AMBER | – | 1.535 | 1.544 | |
| OPLS | – | 1.533 | 1.522 | |
| ℓ(Å) | MM2 | – | 7.832 | 27.868 |
| AMBER | – | 7.773 | 24.796 | |
| OPLS | – | 7.394 | 26.2141 | |
| ΔE (kcal mol−1) | MM2 | −2.376 | 75.693 | 133.363 |
| AMBER | 7.8059 | 80.481 | 66.881 | |
| OPLS | 6.7407 | 55.867 | 47.064 |
| Time (min) | Mn2+ | Ni2+ | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Cr3+ | Co3+ | Fe3+ |
|---|---|---|---|---|---|---|---|---|---|
| 15 | 0.3598 | 0.3664 | 0.3684 | 0.1109 | 0.3674 | 0.1587 | 0.3592 | 0.3673 | 0.3668 |
| 30 | 0.4243 | 0.3663 | 0.4918 | 0.2593 | 0.4922 | 0.2385 | 0.4892 | 0.4932 | 0.4857 |
| 45 | 0.6673 | 0.7367 | 0.7395 | 0.3877 | 0.7423 | 0.3556 | 0.6990 | 0.7365 | 0.6945 |
| 60 | 0.8340 | 1.4297 | 1.4705 | 0.7870 | 1.4635 | 0.7692 | 1.3094 | 1.4645 | 1.4621 |
| 120 | 1.4545 | 1.4602 | 1.4772 | 0.7792 | 1.4737 | 0.7938 | 1.4629 | 1.4647 | 1.4539 |
| 240 | 1.4516 | 1.4700 | 1.4720 | 0.8156 | 1.4882 | 0.8063 | 1.4415 | 1.4817 | 1.4598 |
| 480 | 1.4614 | 1.4641 | 1.4730 | 0.8063 | 1.4807 | 0.7818 | 1.4472 | 1.4746 | 1.4640 |
| C0 | Mn2+ | Ni2+ | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Cr3+ | Co3+ | Fe3+ |
|---|---|---|---|---|---|---|---|---|---|
| 15 | 1.4537 | 1.4623 | 1.4791 | 0.8318 | 1.4972 | 0.7928 | 1.4293 | 1.4856 | 1.4585 |
| 20 | 1.9031 | 1.9467 | 1.9502 | 0.9675 | 1.9549 | 0.8055 | 1.9269 | 1.9637 | 1.9327 |
| 25 | 2.4053 | 2.4415 | 2.4418 | 1.1907 | 2.4939 | 0.8250 | 2.1855 | 2.4606 | 2.4130 |
| 30 | 2.8863 | 2.9345 | 2.9332 | 1.4037 | 2.9487 | 0.9071 | 2.9190 | 2.9246 | 2.9531 |
| pH | Mn2+ | Ni2+ | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Cr3+ | Co3+ | Fe3+ |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.4005 | 0.0823 | 0.6057 | 0.3518 | 2.8651 | 0.6805 | 1.1067 | 0.6669 | 2.5167 |
| 3 | 2.9035 | 2.9029 | 2.9421 | 0.9627 | 2.9676 | 0.9268 | 2.8612 | 2.9476 | 2.9225 |
| 5 | 2.8643 | 2.9451 | 2.9342 | 0.9746 | 2.9406 | 1.0188 | 2.6955 | 2.9482 | 2.8647 |
| 7 | 2.8683 | 2.9454 | 2.9296 | 1.4291 | 2.9473 | 0.9390 | 2.9220 | 2.9267 | 2.9693 |
| 9 | 2.8236 | 2.4858 | 2.9246 | 1.5315 | 2.7092 | 0.9892 | 2.6105 | 2.8848 | 2.9339 |
| pH | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Fe3+ |
|---|---|---|---|---|---|
| 1 | 1.0523 | 0.8874 | 0.8394 | 0.8577 | 0.8330 |
| 3 | 1.4679 | 1.1680 | 1.0931 | 1.1260 | 0.9881 |
| 5 | 1.4606 | 1.1771 | 0.9930 | 1.0840 | 1.0345 |
| 7 | 1.4745 | 1.2190 | 1.1875 | 1.1897 | 1.2479 |
| 9 | 1.5000 | 1.2507 | 1.1802 | 1.2149 | 1.3070 |
| pH | Mn2+ | Ni2+ | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Cr3+ | Co3+ | Fe3+ |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.9876 | 0.4915 | 1.4786 | 1.1802 | 3.3801 | 1.5166 | 1.8042 | 1.6436 | 2.7773 |
| 3 | 3.5392 | 3.6025 | 3.7228 | 1.8388 | 3.9824 | 1.6797 | 3.3517 | 3.7714 | 3.5849 |
| 5 | 3.4707 | 3.7295 | 3.6864 | 1.7468 | 3.7917 | 1.8080 | 3.0226 | 3.8087 | 3.4562 |
| 7 | 3.3756 | 3.7926 | 3.6236 | 2.0892 | 3.7893 | 1.6960 | 3.6030 | 3.6305 | 4.0027 |
| 9 | 3.2043 | 2.8147 | 3.6260 | 2.0864 | 3.0832 | 1.7333 | 2.8637 | 3.3987 | 3.6681 |
| Ion | Mn2+ | Ni2+ | Cu2+ | Zn2+ | Pb2+ | Cd2+ | Cr3+ | Co3+ | Fe3+ |
|---|---|---|---|---|---|---|---|---|---|
| log D/pH | 0.57 | 0.48 | 0.30 | 0.54 | 0.52 | 0.24 | 0.51 | 0.54 | 0.52 |
| pH | 1 | 3 | 5 | 7 | 9 |
|---|---|---|---|---|---|
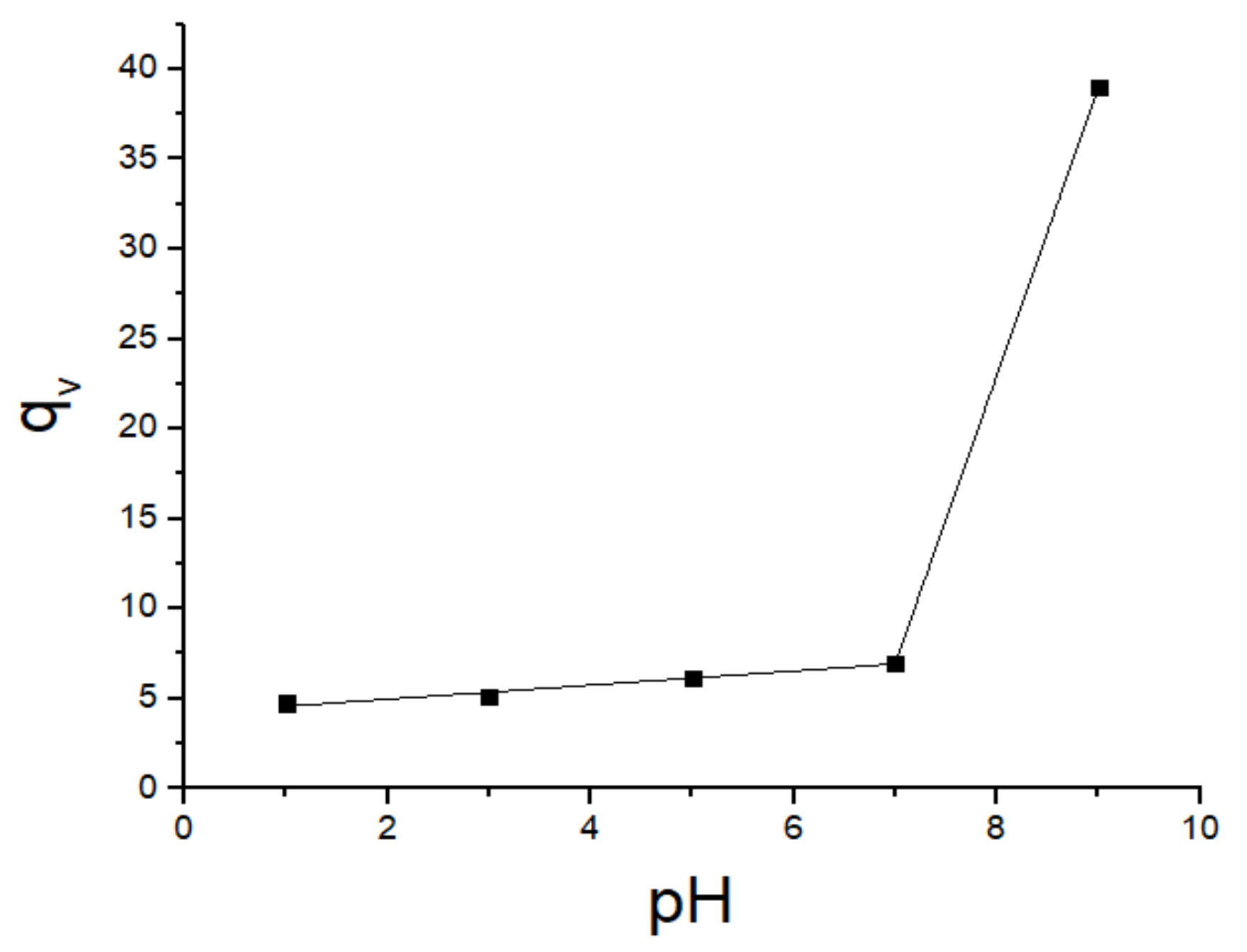
| Swollen polymer (g) | 0.4165 | 0.4472 | 0.5373 | 0.6084 | 0.7891 |
| qv | 4.74 | 5.10 | 6.16 | 7.00 | 38.98 |
| Metal Ion | Desorbed Metal Ion (mg) | Sorbed Metal Ion (mg) | Desorption Ratio (%) | ID (Hydrated) (Å) | ID (Å) |
|---|---|---|---|---|---|
| Mn2+ | 0.1125 | 0.1524 | 73.8 | 0.86 | 1.60 |
| Ni2+ | 0.1107 | 0.1687 | 65.6 | 0.72 | 1.34 |
| Cu2+ | 0.0849 | 0.1144 | 74.2 | 0.62 | 1.44 |
| Zn2+ | 0.0536 | 0.0738 | 72.7 | 0.74 | 1.40 |
| Pb2+ | 0.0912 | 0.1066 | 85.6 | 1.20 | 2.42 |
| Cd2+ | 0.1047 | 0.1325 | 79.0 | 0.96 | 1.82 |
| Cr3+ | 0.0880 | 0.1261 | 69.8 | 0.65 | 1.26 |
| Co3+ | 0.0748 | 0.1085 | 68.9 | 0.745 | 0.69 |
| Fe3+ | 0.0341 | 0.1436 | 23.7 | 0.645 | 0.63 |
| Ion | Langmuir Isotherm | Freundlich Isotherm | |||||
|---|---|---|---|---|---|---|---|
| Kb | As | R2 | k | n | 1/n | R2 | |
| Cr3+ | 0.7583 | 0.0439 | 0.9599 | 0.0563 | 10.8342 | 0.0504 | 0.0504 |
| Ni2+ | −0.4554 | −0.1425 | 0.4138 | 11.6547 | 0.8163 | 0.9448 | 0.9448 |
| Mn2 | 0.4259 | 0.1101 | 0.3716 | 0.6046 | 1.5152 | 0.7922 | 0.7922 |
| Zn2+ | 0.0070 | 0.0439 | 0.8257 | 0.0479 | 1.6745 | 0.9485 | 0.9485 |
| Cd2+ | 0.0041 | 0.0086 | 0.9863 | 0.0093 | 9.3985 | 0.6939 | 0.6939 |
| Pb2+ | 1.7774 | 0.0121 | 0.9013 | 0.0249 | 8.3542 | 0.3465 | 0.3465 |
| Cu2+ | 0.6626 | 0.0737 | 0.6999 | 0.4560 | 1.8982 | 0.8698 | 0.8698 |
| Co3+ | 0.9605 | 0.0663 | 0.9459 | 0.3029 | 2.41255 | 0.9285 | 0.9285 |
| Fe3+ | 0.5979 | 0.0558 | 0.5211 | 0.1313 | 3.6738 | 0.0926 | 0.0926 |
| Sample | TI (°C) | Tf (°C) | Tmax (°C) | Weight Loss (%) | Char Yield (%) | ||
|---|---|---|---|---|---|---|---|
| PAA45 | 154 | 225 | 409.0 | 6.82 | 11.16 | 17.53 | 44.59 at 542.65 °C |
| 323 | |||||||
| PAA SAP | 156.0 | 258.1 | 417.0 | 3.90 | 19.0 | 39.90 | 14.7 at 536.70 °C |
| 294.1 | 34.9 | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savaskan Yilmaz, S.; Yildirim, N.; Misir, M.; Misirlioglu, Y.; Celik, E. Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation. Materials 2020, 13, 4390. https://doi.org/10.3390/ma13194390
Savaskan Yilmaz S, Yildirim N, Misir M, Misirlioglu Y, Celik E. Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation. Materials. 2020; 13(19):4390. https://doi.org/10.3390/ma13194390
Chicago/Turabian StyleSavaskan Yilmaz, Sevil, Nuri Yildirim, Murat Misir, Yasin Misirlioglu, and Emre Celik. 2020. "Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation" Materials 13, no. 19: 4390. https://doi.org/10.3390/ma13194390
APA StyleSavaskan Yilmaz, S., Yildirim, N., Misir, M., Misirlioglu, Y., & Celik, E. (2020). Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation. Materials, 13(19), 4390. https://doi.org/10.3390/ma13194390