Pumping Schedule Optimization in Acid Fracturing Treatment by Unified Fracture Design
Abstract
:1. Introduction
2. Methods
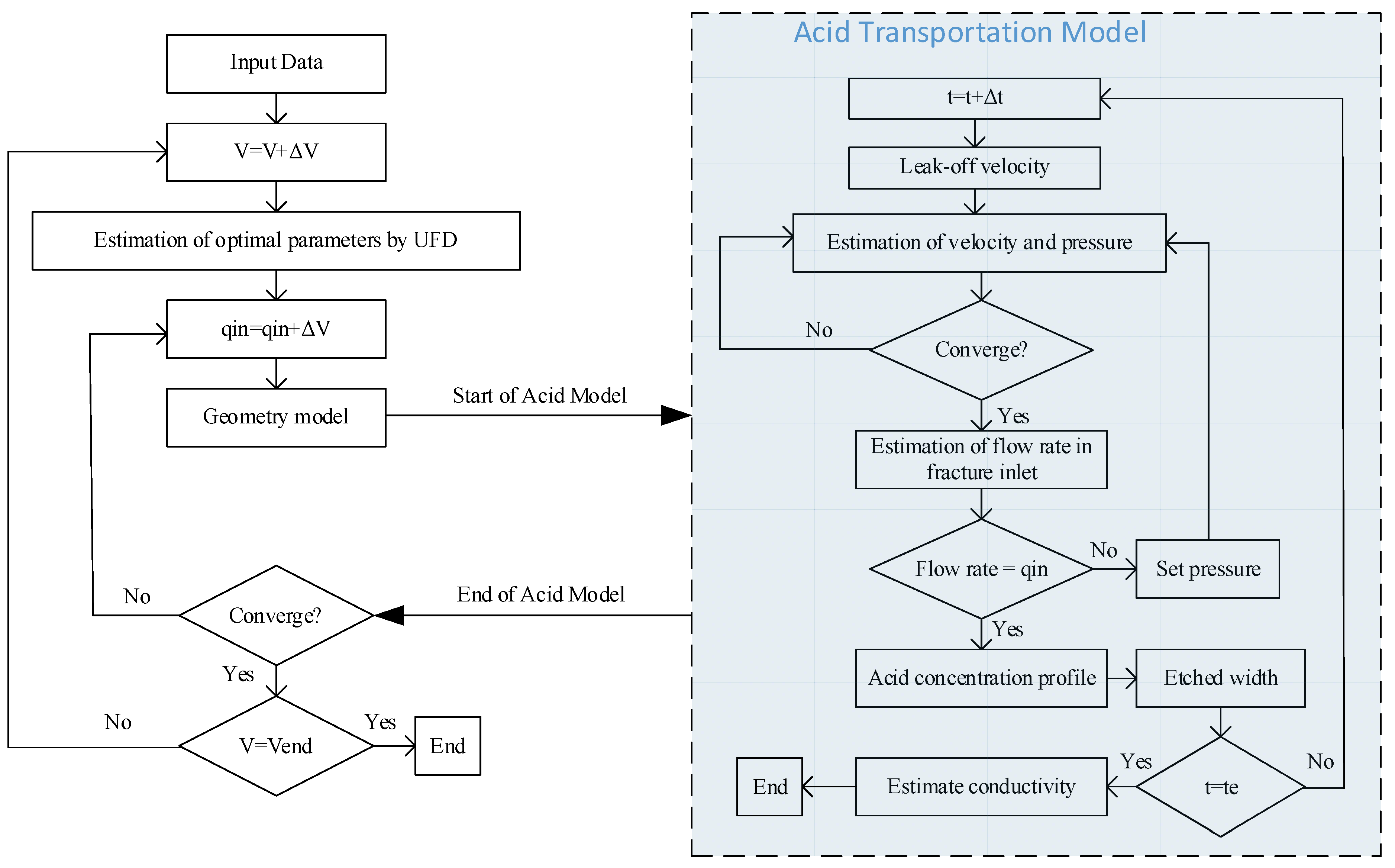
2.1. Workflow
2.2. Optimization of the Fracture Geometry Parameters
- For a specific acid volume and an initial guess for final (), the average ideal fracture width is as [4]:where is porosity, is initial guess optimal fracture half-length, is the fracture height and is equal to the net pay of the formation. is injected acid volume, and is volumetric dissolving power.
- One of the important parameters to estimate the fracture conductivity is the average ideal fracture width. A geostatistical method was employed to calculate the fracture conductivity [28] (Equation (3)). In this method, the effect of the spatial behavior of the formation’s permeability and elastic properties are considered in the calculations. Since the fracture conductivity is approximately proportional to , it is important, therefore, that the acid-etched width be accurately determined [29].where is the dimensionless horizontal correlation length, is the dimensionless vertical correlation length, and is the dimensionless standard deviation of permeability. is closure stress, is Young’s modulus, and is the calculated average ideal fracture width in the previous step. and are constant coefficients and erf refers to the error function that is given in the model.
- After that, the proppant number for acid fracturing is determined, as reported in [30]. An equivalent proppant number was used for calculating the optimum dimensionless fracture conductivity.
- In this step, the optimal fracture half-length and optimal fracture width are calculated as in [30]:
- Finally, the calculated fracture half-length and fracture width from Equations (6) and (7) are compared with initial and . This process continues until the estimated values reach a stable condition.
2.3. Fracture Propagation Model
2.4. Acid Model
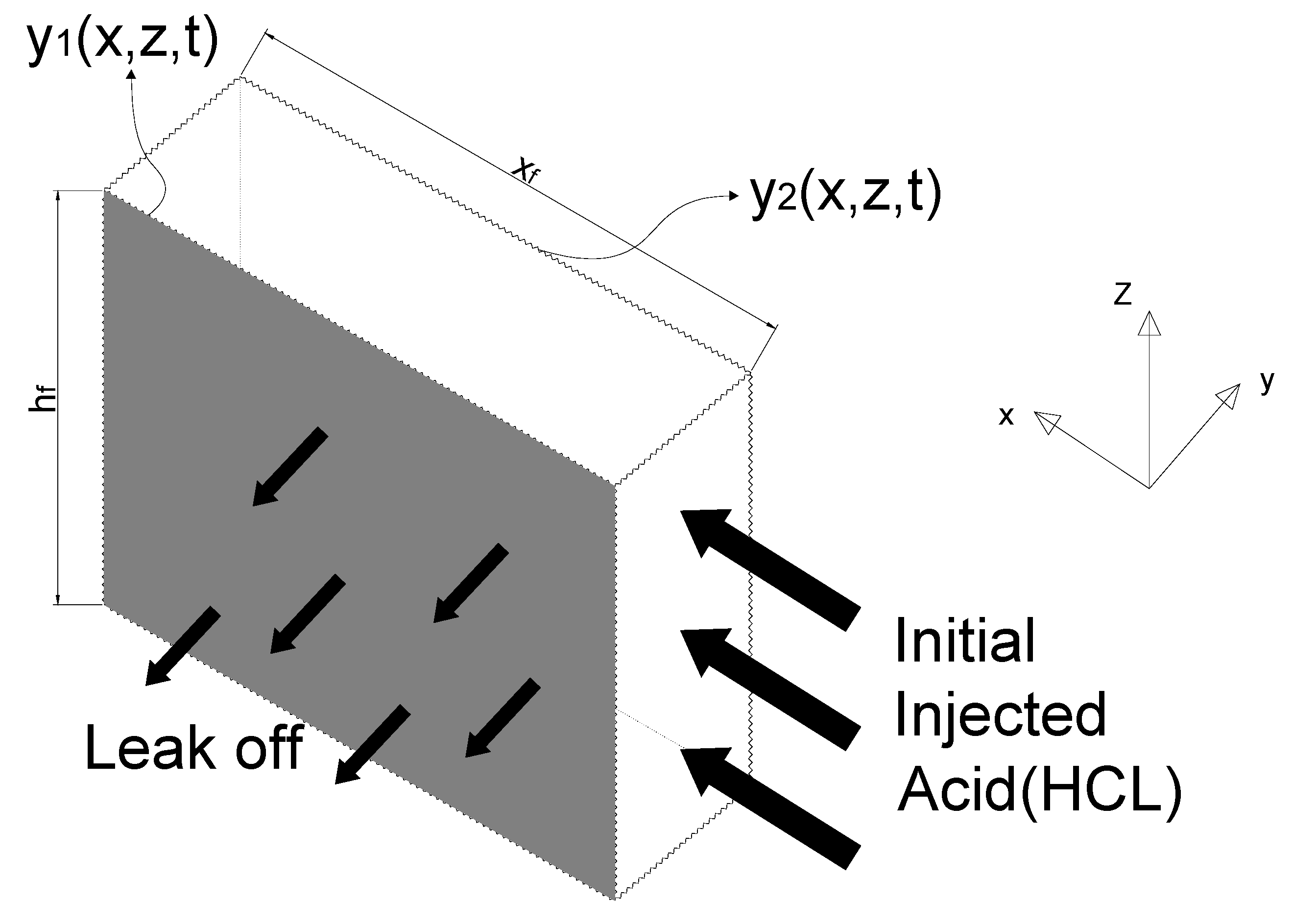
2.4.1. Fluid Velocity Components and Pressure
2.4.2. Calculate the Acid Concentration
2.4.3. Rocks Displacement on the Fracture Surfaces
2.4.4. Boundary Condition
2.5. Leak-Off Model
3. Results and Discussion
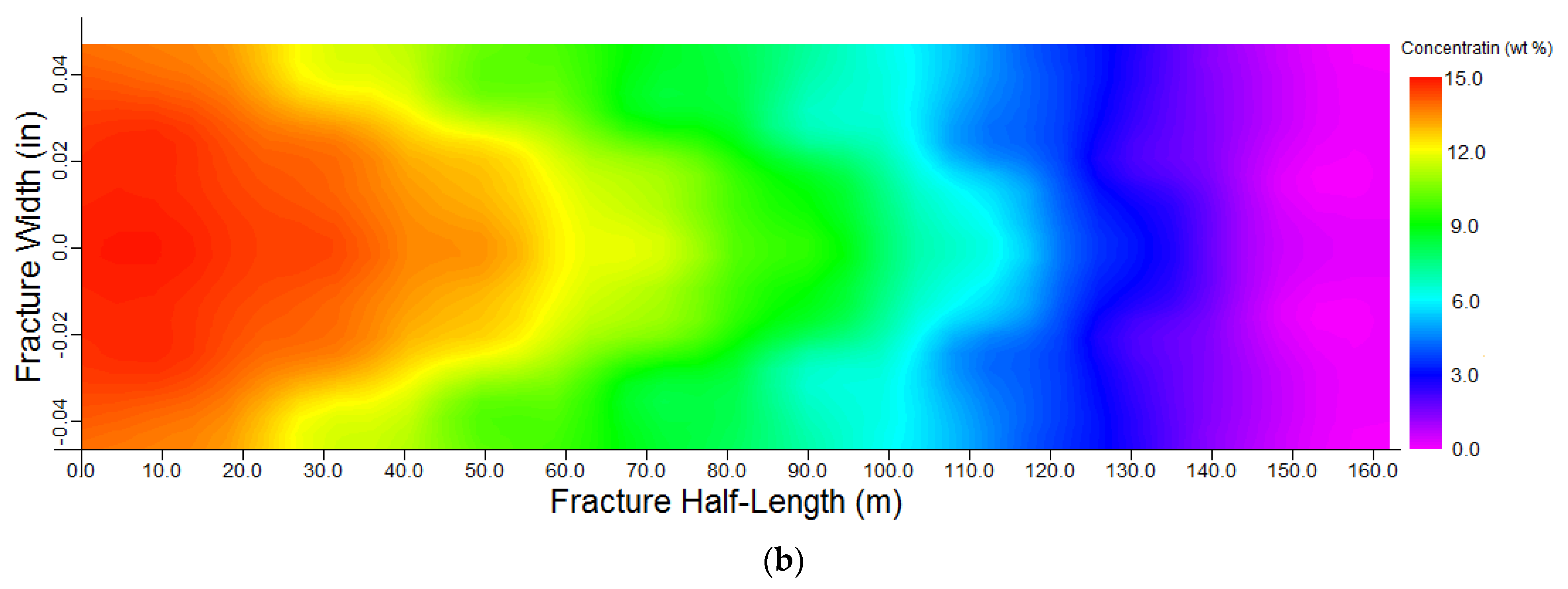
3.1. Acid Model Validation
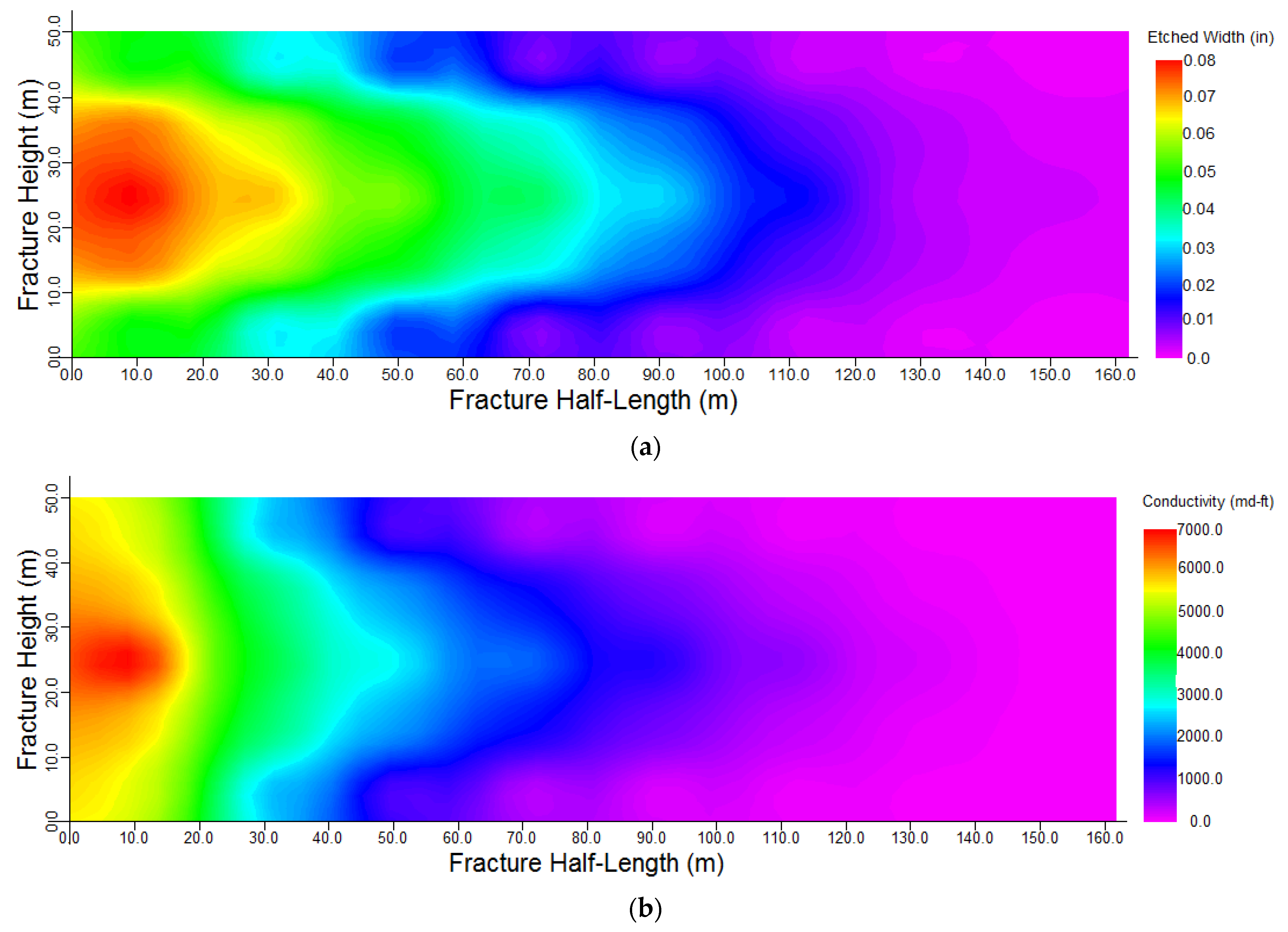
3.2. Model Establishment
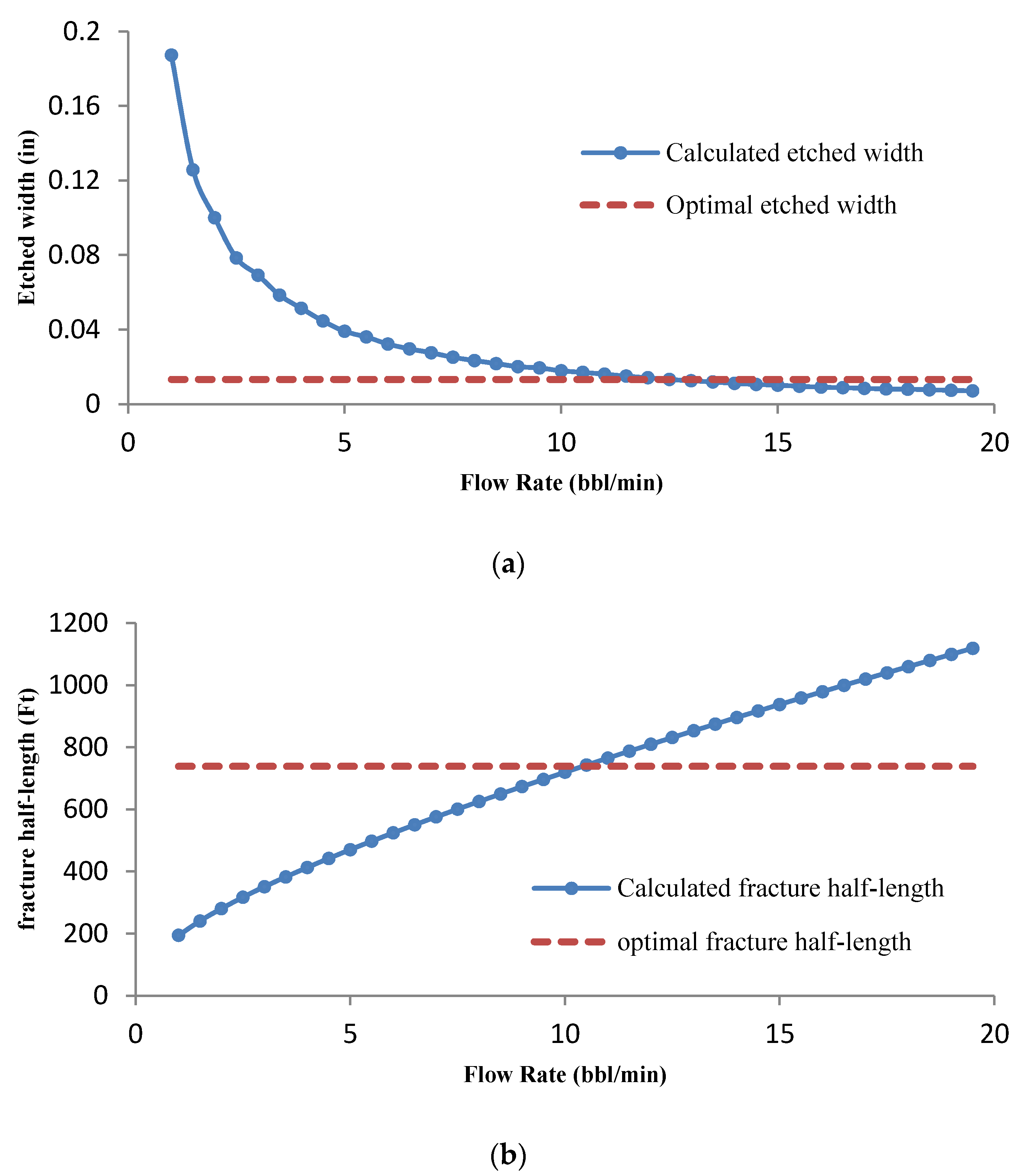
3.3. Optimization of Injection Parameters
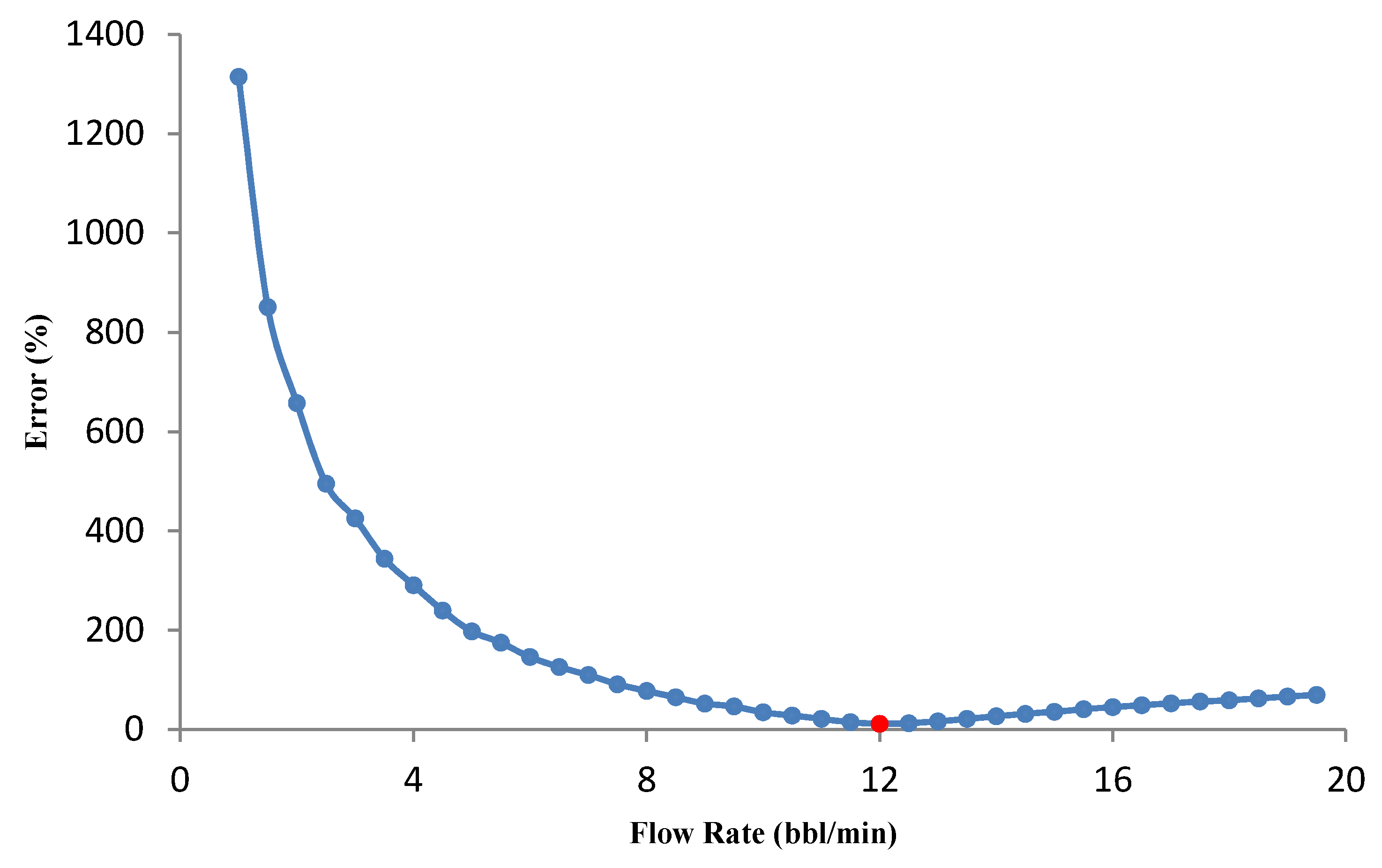
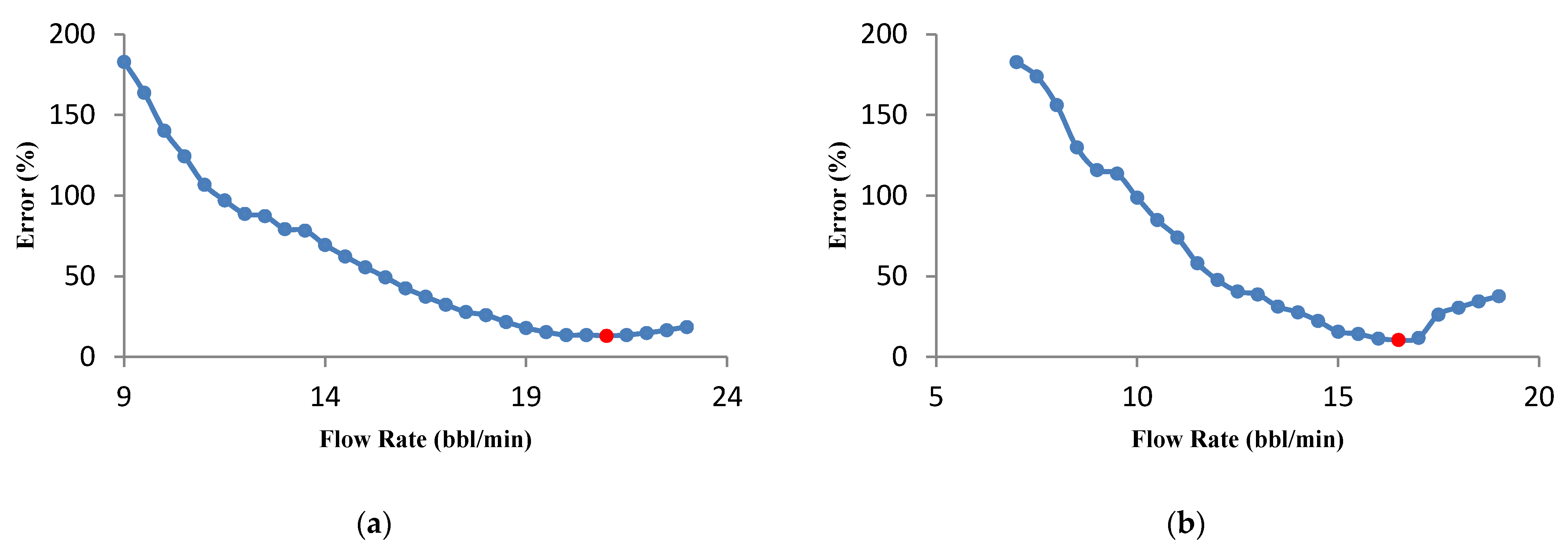
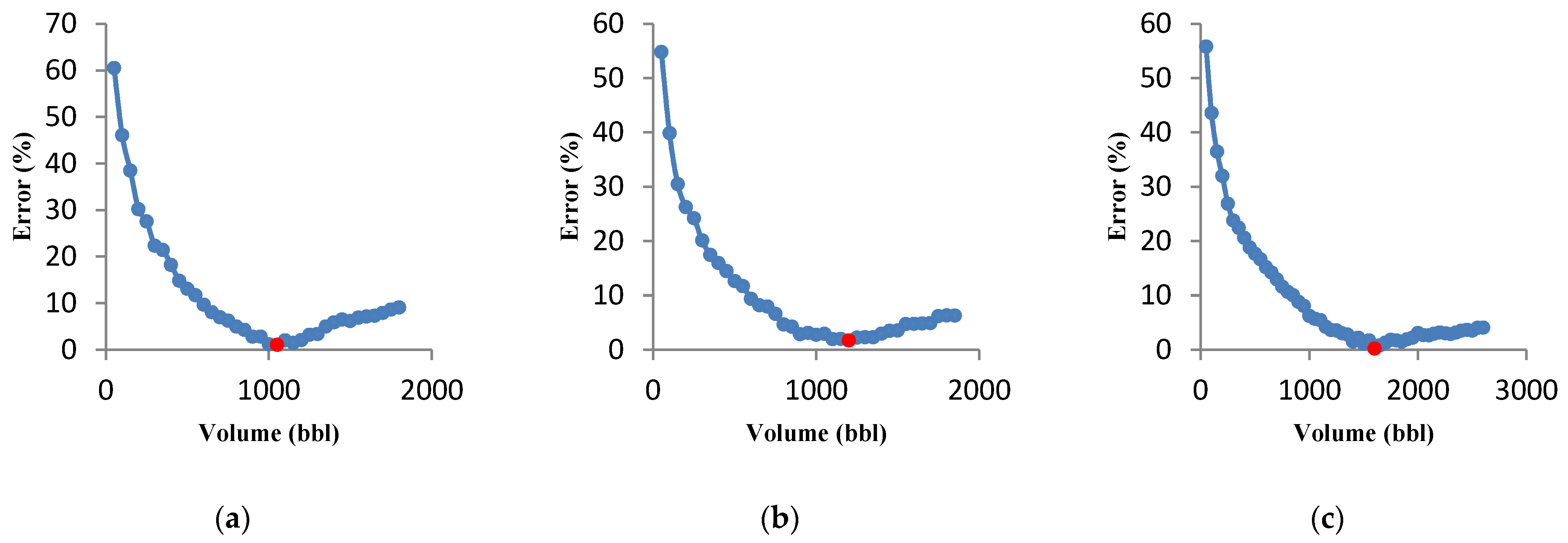
3.3.1. Effect of Flow Rate on Optimization Results
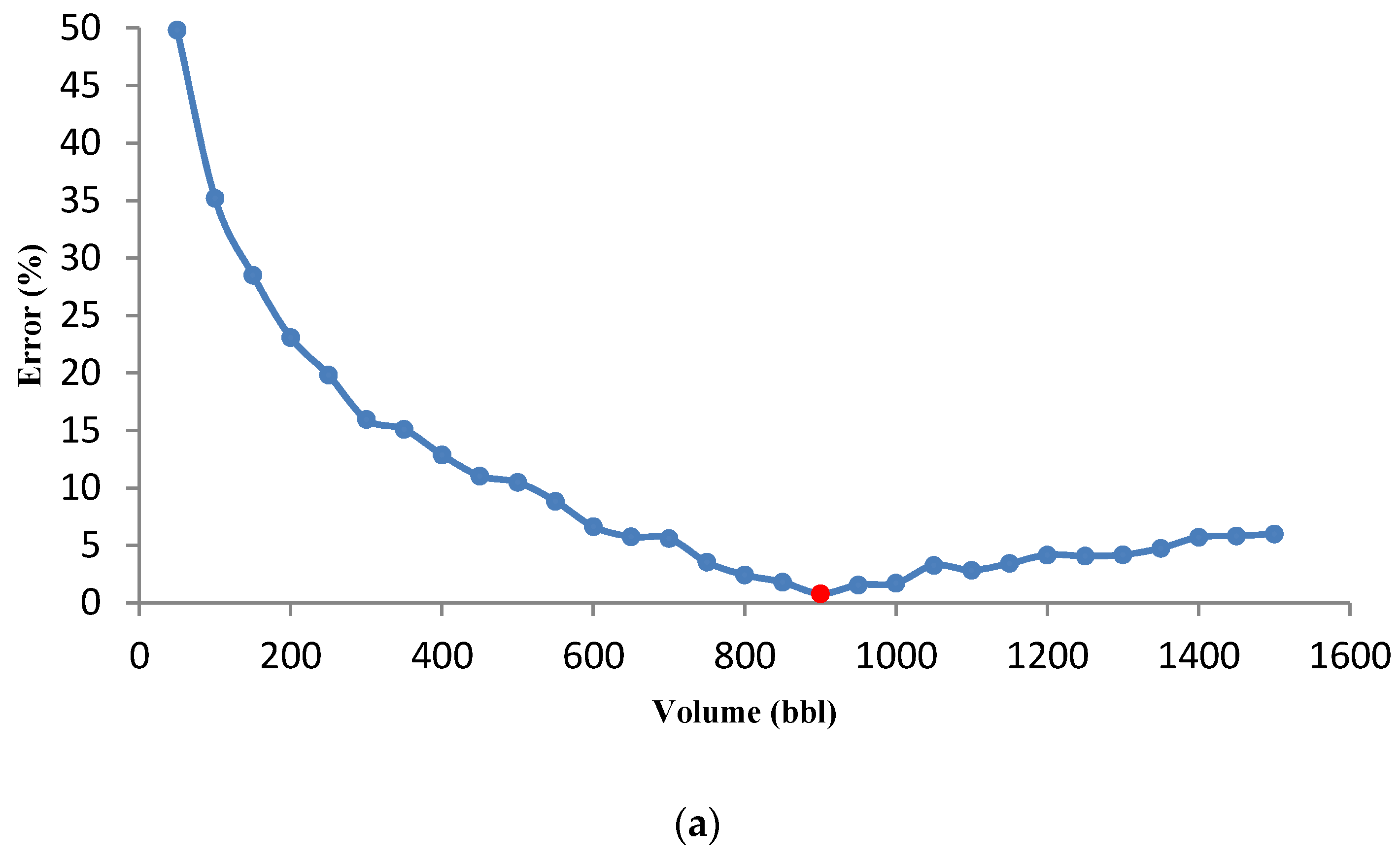
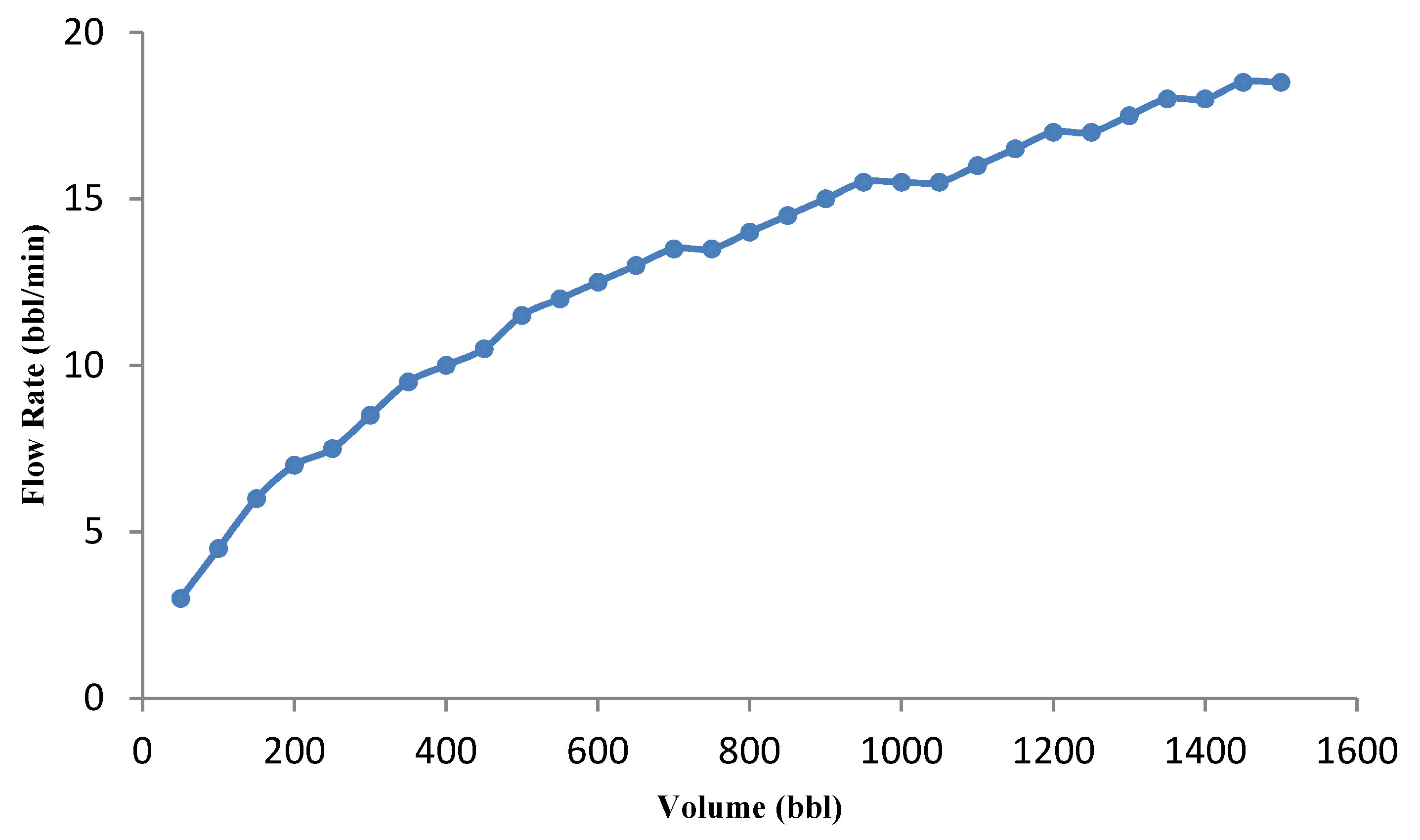
3.3.2. Effect of Acid Volume on Optimization Results
3.4. Parametric Study
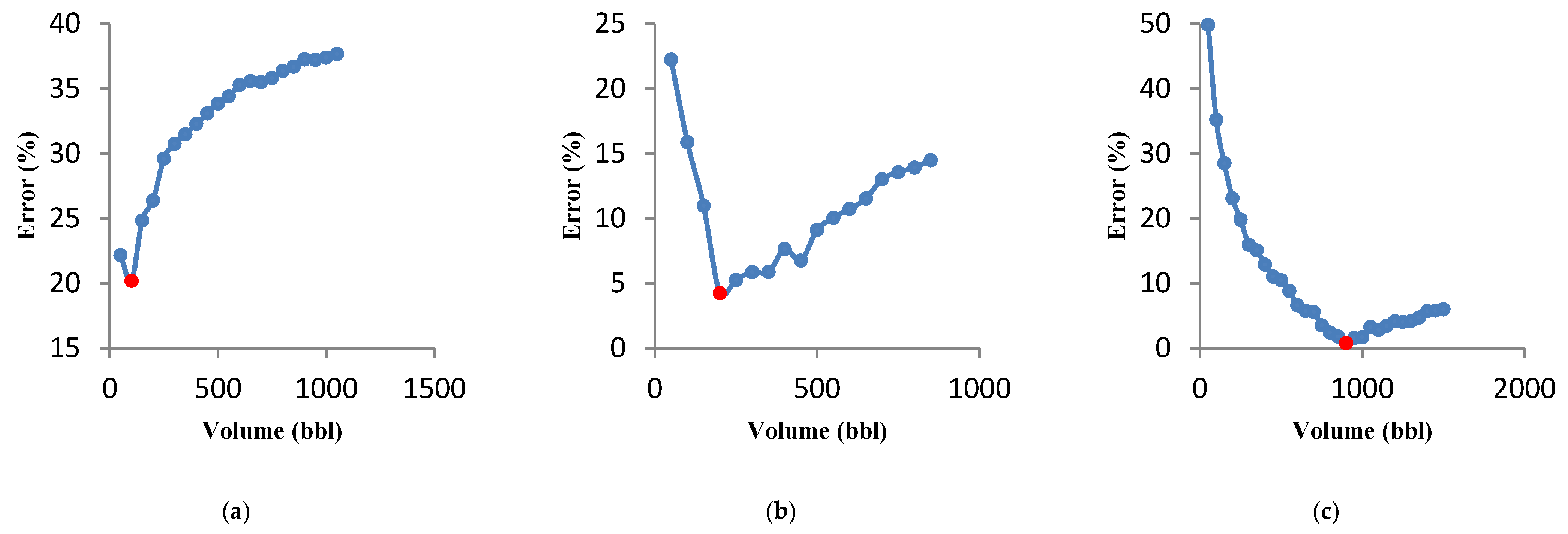
3.4.1. Effect of Formation Permeability
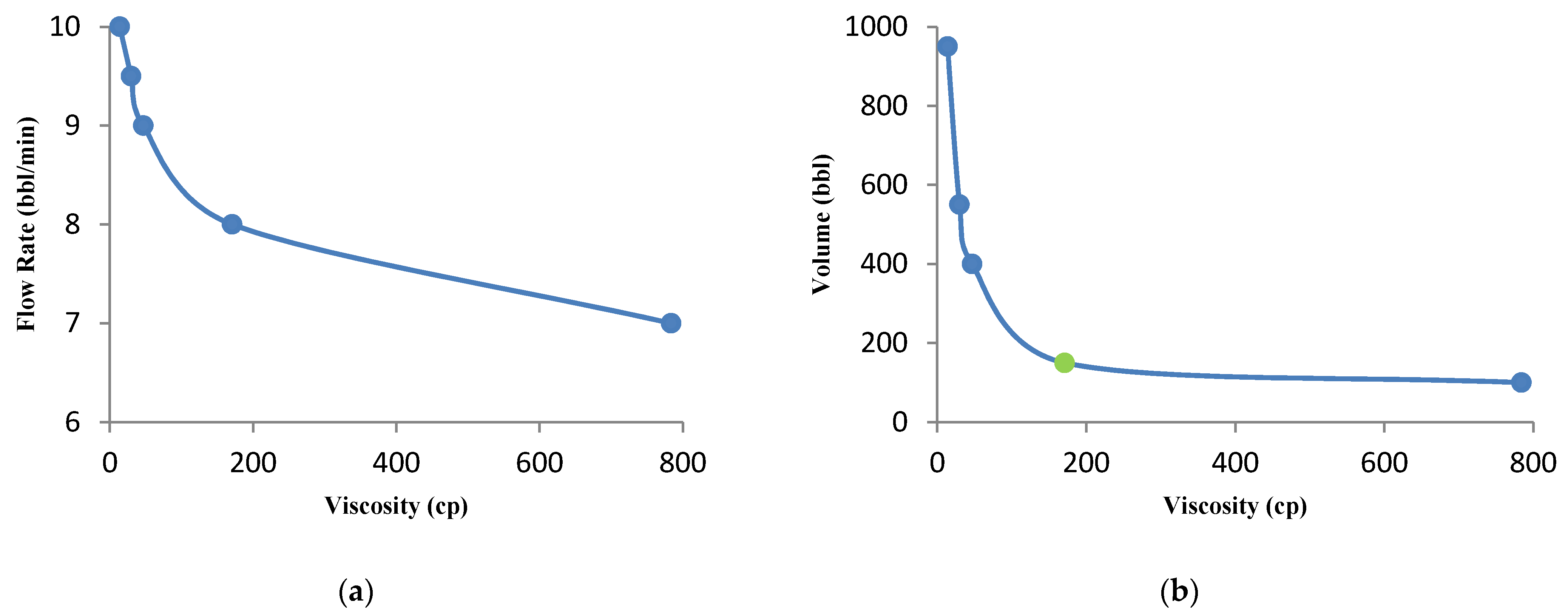
3.4.2. Effect of Injection Fluid Rheology
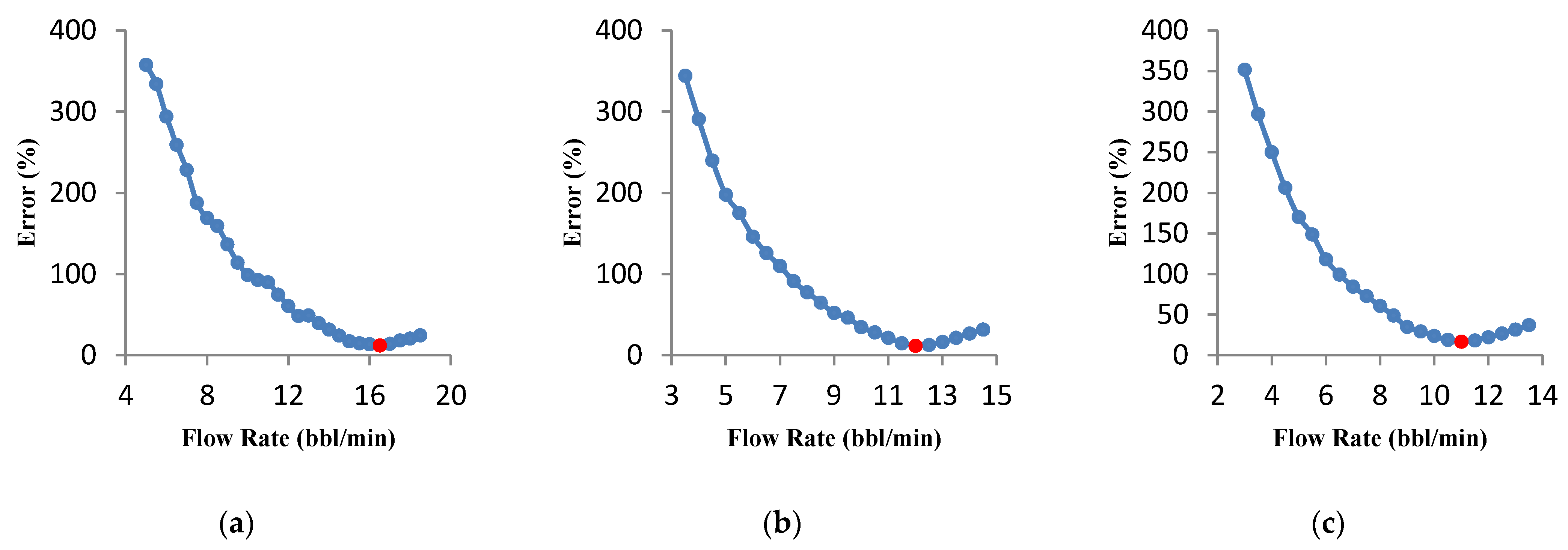
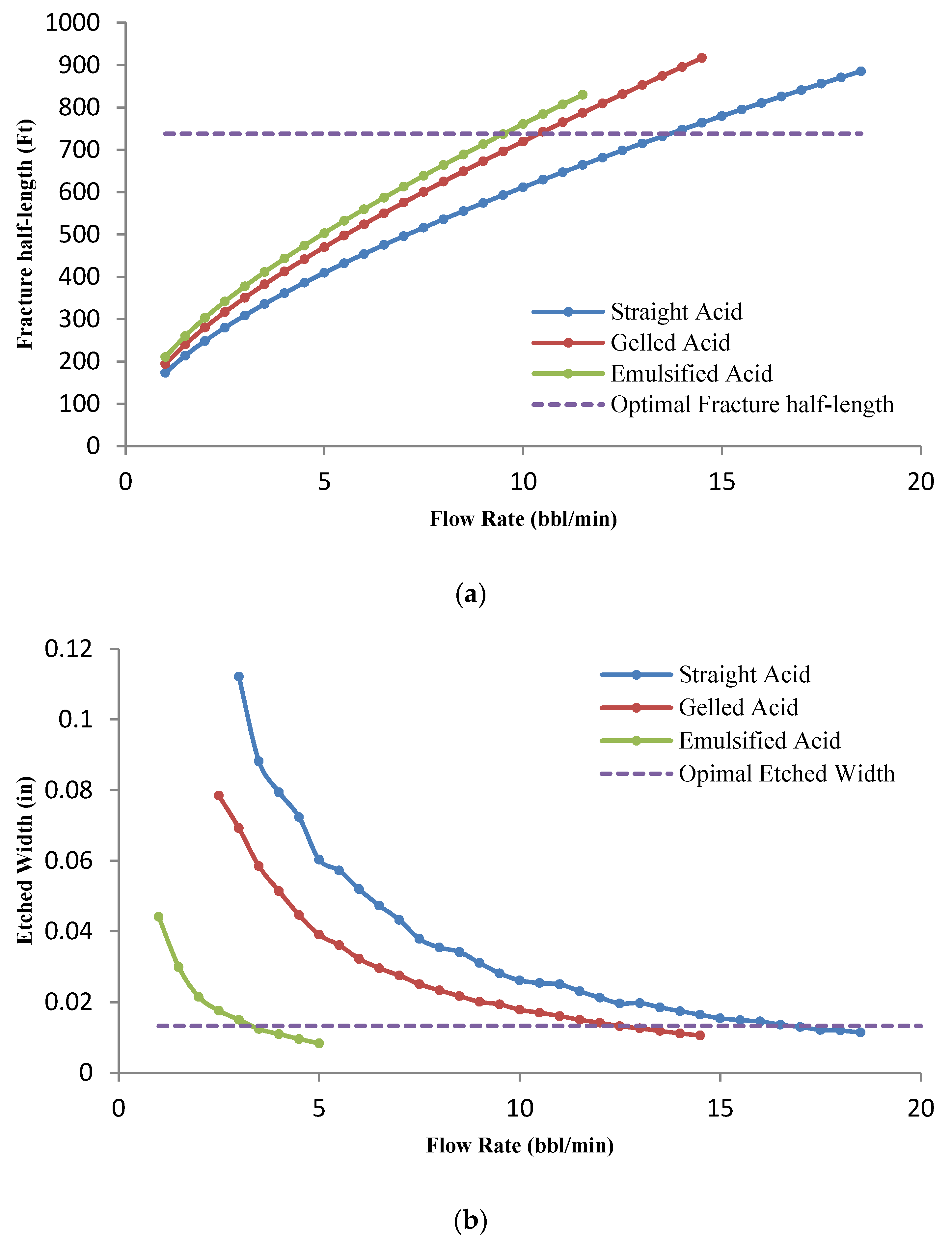
3.4.3. Effect of the Acid Type
3.4.4. Effect of the Acid Percentage
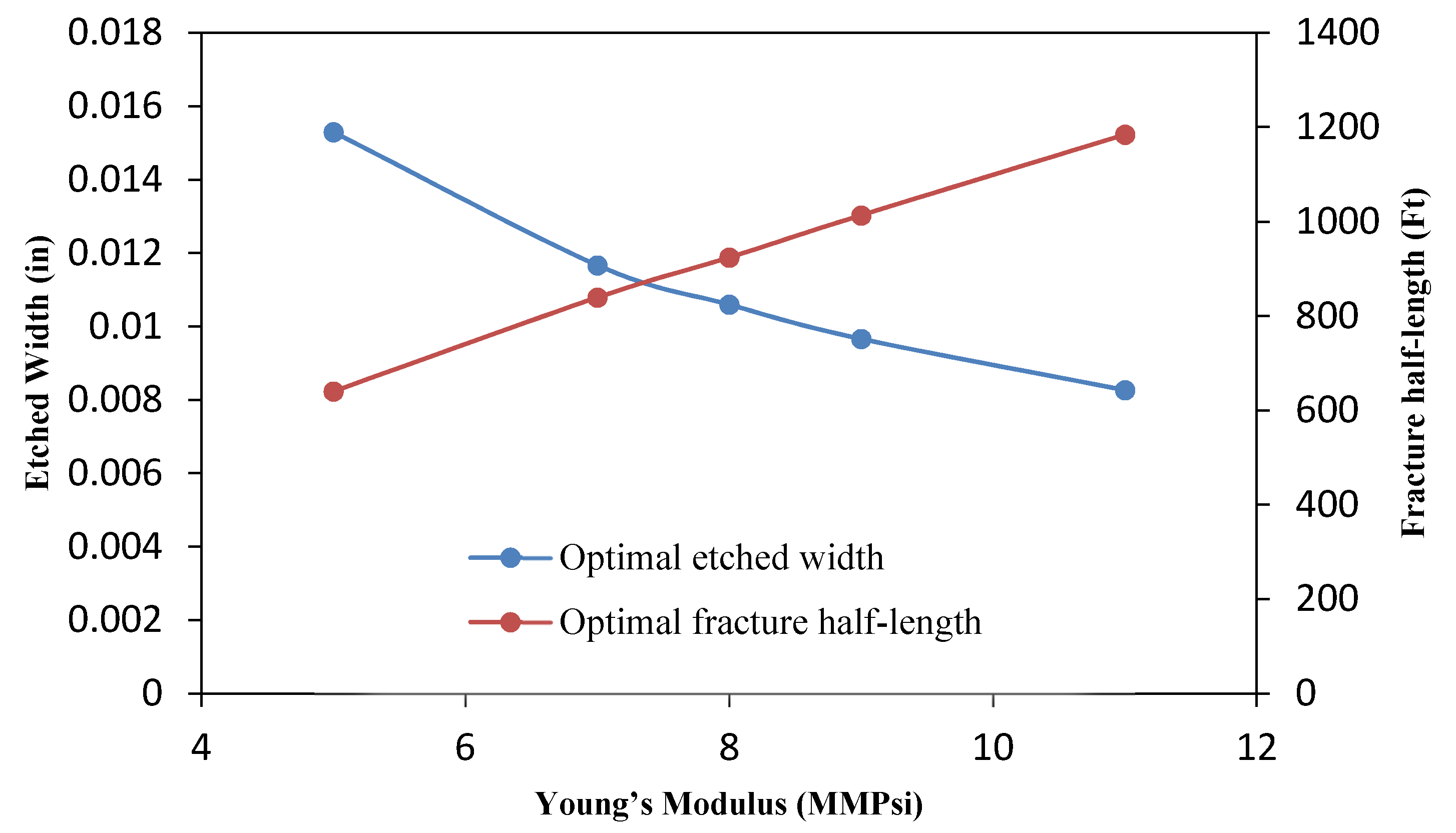
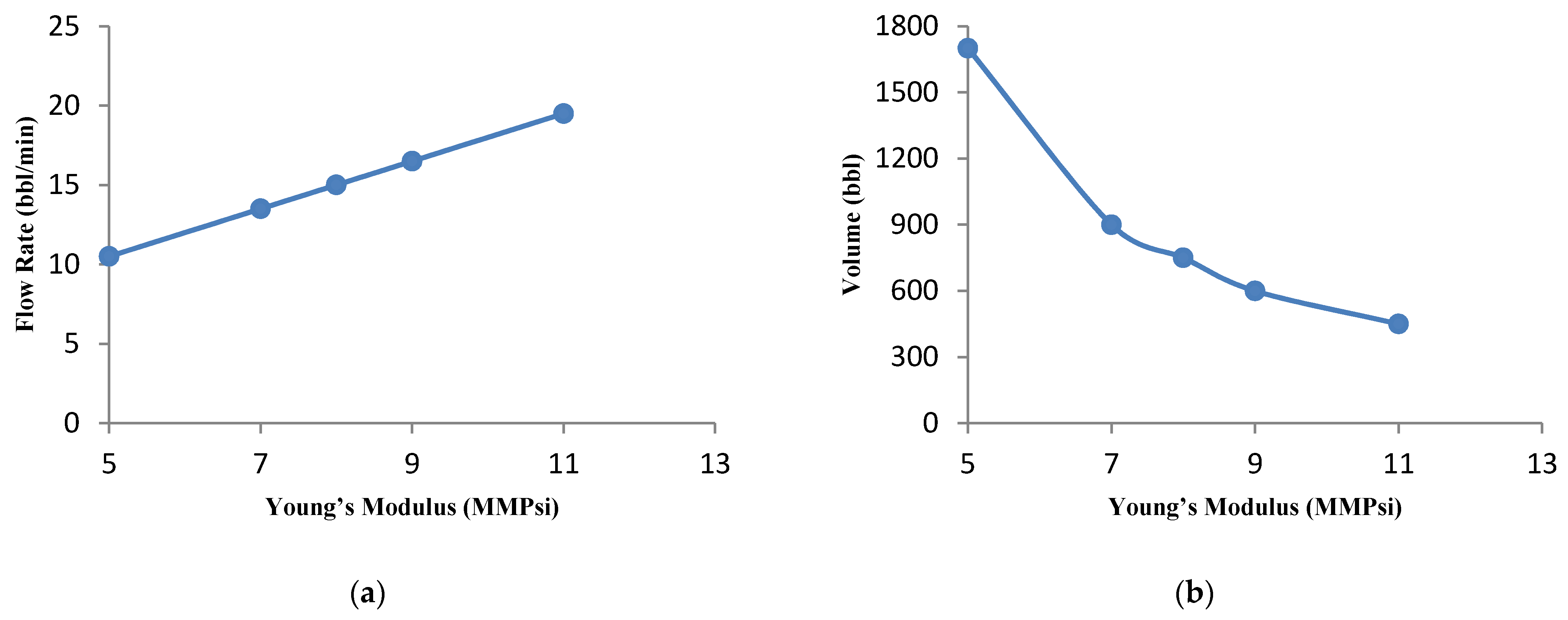
3.4.5. Effect of the Young’s Modulus
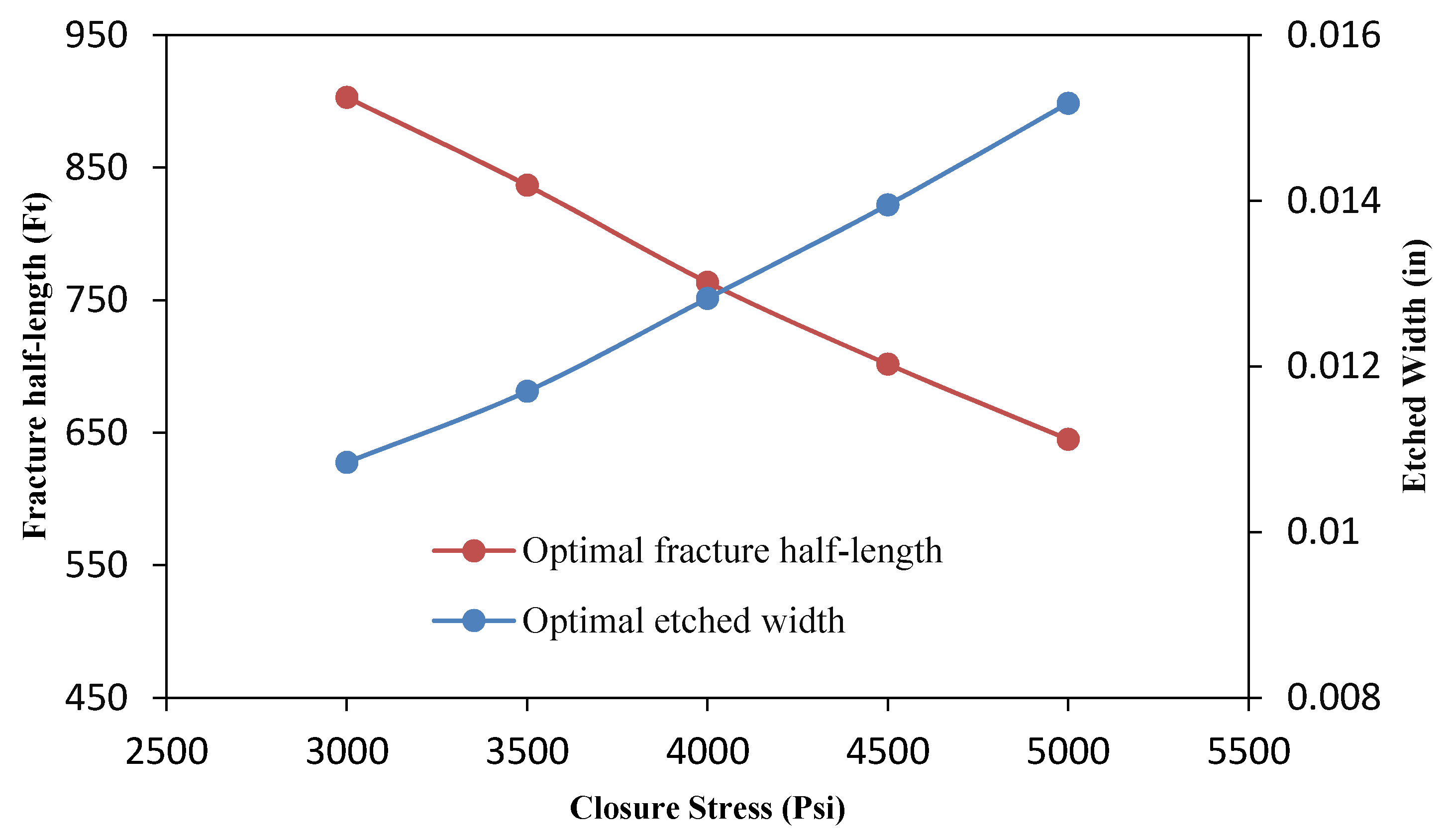
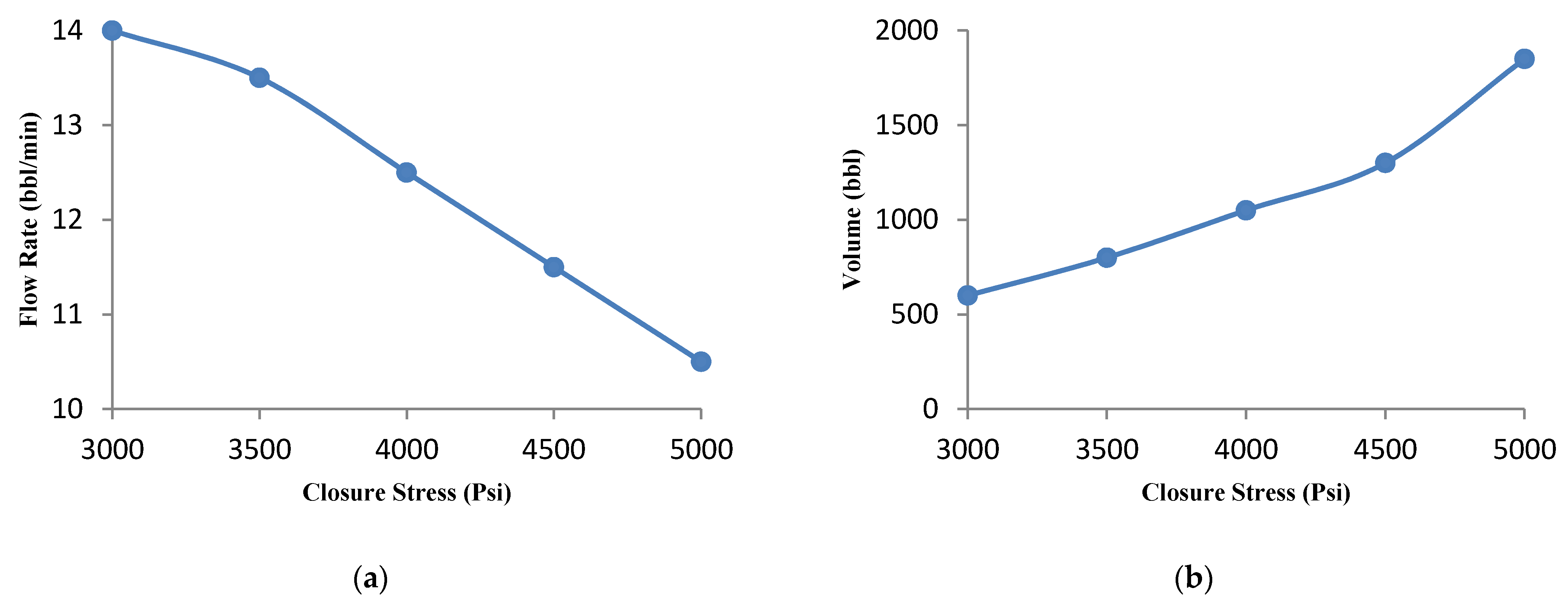
3.4.6. Effect of the Formation Closure Stress
4. Conclusions
- Due to the large number of calculations, the simulations were performed with the proposed method for a specific case. The results have shown that the concentration of acid decreases along the fracture length and fracture walls. Acid-etched width and consequently, conductivity, decrease along the fracture length.
- The flow rate optimization for 16% gelled acid shows that the optimization results are influenced by the acid transport behavior within the fracture.
- The behavior of acid volume against minimum error (for a given volume) was investigated, and it was observed that the optimal flow rate increases with increasing acid volume.
- The parametric study shows that when formation permeability is decreased, the optimal fracture half-length and average fracture width increase and decrease, respectively. In this case, the optimal flow rate increases.
- Fluid viscosity is a controllable parameter during acid fracturing operations. As the fluid viscosity increases, the optimal flow rate and volume of the injected acid decrease. Therefore, increasing the fluid viscosity, to a certain extent, can improve the results of the acid fracturing treatment optimization. On the other hand, its excessive increase has no economic justification.
- Sensitivity analyses on three types of acid systems show that the optimal flow rate for straight acid is higher than for the other two types of acid. It was also observed that for retarded acids, the optimal conditions are reached only at a high acid volume.
- The acid percentage is an influential parameter on the results. For a 15% acid concentration, the required flow rate is higher than 5% and 10%. The acid volume must be increased to achieve optimal conditions for a sample with a high acid concentration.
- The parametric study shows that the optimal flow rate and acid volume increase and decrease, respectively, for high Young’s modulus. In addition, the effect of closure stress was also investigated and it was observed that for a sample with high closure stress, low flow rate and high acid volume are required.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| Dimensionless acid concentration, dimensionless | |
| Optimum fracture conductivity, dimensionless | |
| Leak-off coefficient, Ft/ [m/] | |
| Compressibility fluid-loss coefficient, Ft/ [m/] | |
| Acid equilibrium concentration, | |
| Injected-acid concentration, | |
| Total compressibility, 1/psi [m.s2/kg] | |
| Viscous fluid-loss coefficient, Ft/ [m/] | |
| Viscous fluid-loss coefficient with wormhole, Ft/ [m/] | |
| Wall-building fluid-loss coefficient, Ft/ [m/] | |
| Mean acid concentration | |
| Effective acid diffusion coefficient, Ft2/min [m2/s] | |
| Young’s modulus, psi [kg/m.s2] | |
| Reaction rate coefficient, | |
| Fraction of acid to react before leaking off, dimensionless | |
| Constant for the mean acid concentration profile | |
| Fracture height, Ft [m] | |
| Formation permeability, md [m2] | |
| Consistency index, lb.sn/Ft2 [Pa.sn] | |
| Fracture permeability, md [m2] | |
| Molecular weight of the acid, | |
| Power in the power-law, dimensionless | |
| Reaction order, dimensionless | |
| Acid number, dimensionless | |
| Peclet number | |
| Reynolds number | |
| Fluid net pressure, psi [kg/m.s2] | |
| Pressure difference between fracture and formation, psi [kg/m.s2] | |
| Injected flow rate, bbl/min [m3/s] | |
| Number of PV’s injected at wormhole breakthrough, dimensionless | |
| Spurt loss coefficient, Ft [m] | |
| Injection time, s | |
| Time for acid to reach a particular point in the fracture, min [s] | |
| Velocity in the direction, Ft/min [m/s] | |
| Velocity in the direction, Ft/min [m/s] | |
| Acid volume, bbl [m3] | |
| Induced fracture volume, Ft3 [m3] | |
| Velocity vector, Ft/min [m/s] | |
| Leak-off velocity, Ft/min [m/s] | |
| Reservoir drainage volume, Ft3 [m3] | |
| Velocity in the direction, Ft/min [m/s] | |
| Averaged fracture width in pad stage, in [m] | |
| Estimated average acid-etched width, in [m] | |
| Average ideal fracture width, in [m] | |
| Optimal fracture width, in [m] | |
| Fracture conductivity, md-ft [m3] | |
| Estimated fracture half-length, Ft [m] | |
| Initial guess for final , Ft [m] | |
| Optimal fracture half-length, Ft [m] |
Greek
| Gravitational dissolving power, dimensionless | |
| Constant in Equation (8), dimensionless | |
| Dimensionless horizontal correlation length, dimensionless | |
| Dimensionless vertical correlation length, dimensionless | |
| Eigenvalues for the mean acid concentration profile | |
| Closure stress, MMpsi [kg/m.s2] | |
| Dimensionless standard deviation of permeability, dimensionless | |
| Formation porosity, dimensionless | |
| Fluid density, lbm/Ft3 [kg/m3] | |
| Formation rock density, lbm/Ft3 [kg/m3] | |
| Poisson ratio, dimensionless | |
| Fracture-fluid viscosity of Newtonian fluid, cp [kg/m.s] | |
| Acid viscosity, cp [kg/m.s] | |
| Oil viscosity, cp [kg/m.s] | |
| Volumetric dissolving power, dimensionless |
References
- Roberts, L.D. The Effect of Surface Kinetics in Fracture Acidizing. Soc. Pet. Eng. J. 1974, 14, 385–395. [Google Scholar] [CrossRef]
- Williams, B.B.; Nierode, D.E. Design of Acid Fracturing Treatments. J. Pet. Technol. 1972, 24, 849–859. [Google Scholar] [CrossRef]
- Coulter, A.W.; Alderman, E.N.; Cloud, J.E.; Crowe, C.W. Mathematical Model Simulates Actual Well Conditions In Fracture Acidizing Treatment Design. In Proceedings of the Fall Meeting of the Society of Petroleum Engineers of AIME, Houston, TX, USA, 6–9 October 1974. [Google Scholar] [CrossRef]
- Schechter, R.S. Oil Well Stimulation; Prentice-Hall, Inc.: Englewood Cliffs, NJ, USA, 1992. [Google Scholar]
- Berman, A.S. Laminar Flow in Channels with Porous Walls. J. Appl. Phys. 1953, 24, 1232–1235. [Google Scholar] [CrossRef]
- Hill, A.D.; Zhu, D.; Wang, Y. The Effect of Wormholing on the Fluid Loss Coefficient in Acid Fracturing. SPE Prod. Facil. 1995, 10, 257–264. [Google Scholar] [CrossRef]
- Gdanski, A.D.; Lee, W.S. On the Design of Fracture Acidizing Treatments. In Proceedings of the SPE Production Operations Symposium, Oklahoma City, OK, USA, 13–14 March 1989. [Google Scholar] [CrossRef]
- Navarrete, R.; Miller, M.; Gordon, J. Laboratory and Theoretical Studies for Acid Fracture Stimulation Optimization. In Proceedings of the 1998 SPE Permian Basin Oil and Gas Recovery Conference, Midland, TX, USA, 23–26 March 1998. [Google Scholar] [CrossRef]
- Lo, K.K.; Dean, R.H. Modeling of Acid Fracturing. SPE Prod. Eng. 1989, 4, 194–200. [Google Scholar] [CrossRef]
- Settari, A.; Sullivan, R.; Hansen, C. A New Two-Dimensional Model for Acid-Fracturing Design. SPE Prod. Facil. 2001, 16, 200–209. [Google Scholar] [CrossRef]
- Romero, J.; Gu, H.; Gulrajani, S.N. 3D Transport in Acid-Fracturing Treatments: Theoretical Development and Consequences for Hydrocarbon Production. SPE Prod. Facil. 2001, 16, 122–130. [Google Scholar] [CrossRef]
- Mou, J.; Zhu, D.; Hill, A.D. Acid-Etched Channels in Heterogeneous Carbonates—A Newly Discovered Mechanism for Creating Acid-Fracture Conductivity. SPE J. 2009, 15, 404–416. [Google Scholar] [CrossRef]
- Oeth, C.V.; Hill, A.D.; Zhu, D. Acid Fracture Treatment Design with Three-Dimensional Simulation. In Proceedings of the SPE Hydraulic Fracturing Technology Conference, The Woodlands, TX, USA, 4–6 February 2014. [Google Scholar] [CrossRef]
- Aljawad, M.S.; Zhu, D.; Hill, A.D. Modeling Study of Acid Fracture Fluid System Performance. In Proceedings of the SPE Hydraulic Fracturing Technology Conference, The Woodlands, TX, USA, 9–11 February 2016. [Google Scholar] [CrossRef]
- Alhubail, M.M.; Misra, A.; Barati, R. A Novel Acid Transport Model with Robust Finite Element Discretization. In Proceedings of the SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition, Dammam, Saudi Arabia, 24–27 April 2017. [Google Scholar] [CrossRef]
- Ugursal, A.; Schwalbert, M.P.; Zhu, D.; Hill, A.D. Acid Fracturing Productivity Model for Naturally Fractured Carbonate Reservoirs. In Proceedings of the SPE International Hydraulic Fracturing Technology Conference and Exhibition, Muscat, Oman, 16–18 October 2018. [Google Scholar] [CrossRef]
- Dang, L.; Zhou, C.; Huang, M.; Jiang, D. Simulation of effective fracture length of prepad acid fracturing considering multiple leak-off effect. Nat. Gas Ind. B 2019, 6, 64–70. [Google Scholar] [CrossRef]
- Ben-Naceur, K.; Economides, M.J. Design and Evaluation of Acid Fracturing Treatments. In Proceedings of the SPE Joint Rocky Mountain Regional/Low Permeability Reservoirs Symposium and Exhibition, Denver, Colorado, 6–8 March 1989. [Google Scholar] [CrossRef]
- Guo, J.; Li, Y.; Zhao, J.; Luo, J. Research of Three-Dimensional Model for Acid Fracturing and Optimum Design For the Treatments. In Proceedings of the Canadian International Petroleum Conference, Calgary, Alberta, 8–10 June 2004. [Google Scholar] [CrossRef]
- Ravikumar, A.; Marongiu-Porcu, M.; Morales, A. Optimization of Acid Fracturing with Unified Fracture Design. In Proceedings of the Abu Dhabi International Petroleum Exhibition and Conference, Abu Dhabi, United Arab Emirates, 9–12 November 2015. [Google Scholar] [CrossRef]
- Ai, K.; Duan, L.; Gao, H.; Jia, G. Hydraulic Fracturing Treatment Optimization for Low Permeability Reservoirs Based on Unified Fracture Design. Energies 2018, 11, 1720. [Google Scholar] [CrossRef] [Green Version]
- Aljawad, M.S.; Schwalbert, M.P.; Zhu, D.; Hill, A.D. Optimizing Acid Fracture Design in Calcite Formations: Guidelines Using a Fully Integrated Model. SPE Prod. Oper. 2020, 35, 161–177. [Google Scholar] [CrossRef]
- Aljawad, M.S.; Zhu, D.; Hill, A.D. Temperature and Geometry Effects on the Fracture Surfaces Dissolution Patterns in Acid Fracturing. In Proceedings of the SPE Europec featured at 80th EAGE Conference and Exhibition, Copenhagen, Denmark, 11–14 June 2018. [Google Scholar] [CrossRef]
- Aljawad, M.S. Impact of Diversion on Acid Fracturing of Laminated Carbonate Formations: A Modeling Perspective. ACS Omega 2020, 5, 6153–6162. [Google Scholar] [CrossRef] [PubMed]
- Al-Ameri, A.; Gamadi, T. Optimization of acid fracturing for a tight carbonate reservoir. Petroleum 2020, 6, 70–79. [Google Scholar] [CrossRef]
- Hassan, A.; Aljawad, M.S.; Mahmoud, M. An Artificial Intelligence-Based Model for Performance Prediction of Acid Fracturing in Naturally Fractured Reservoirs. ACS Omega 2021, 6, 13654–13670. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.J.; Economides, P. Hydraulic Fracturing Mechanics; John Wiley and Sons: New York, NY, USA, 1995. [Google Scholar]
- Deng, J.; Mou, J.; Hill, A.D.; Zhu, D. A New Correlation of Acid-Fracture Conductivity Subject to Closure Stress. SPE Prod. Oper. 2012, 27, 158–169. [Google Scholar] [CrossRef]
- Oeth, C.V. Three-dimensional Modeling of Acid Transport and Etching in a Fracture. Ph.D. Thesis, Texas A & M University, College Station, TX, USA, 2013. Available online: https://hdl.handle.net/1969.1/151892 (accessed on 30 November 2021).
- Economides, M.J.; Oligney, R.E.; Valko, P.P. Unified Fracture Design; Orsa Press: Alvin, TX, USA, 2002. [Google Scholar]
- Crank, J. Free and Moving Boundary Problems; Clarendon Press: New York, NY, USA, 1984. [Google Scholar]
- Mou, J. Modeling Acid Transport and Non-Uniform Etching in a Stochastic Domain in Acid Fracturing. Ph.D. Thesis, Texas A&M University, College Station, TX, USA, 2009. [Google Scholar]
- Acharya, S.; Moukalled, F.H. Improvements to incompressible flow calculation on a nonstaggered curvilinear grid. Numer. Heat Transfer Part B Fundam. 1989, 15, 131–152. [Google Scholar] [CrossRef]
- Penny, G.S.; Conway, M.W. Fluid Leak-off. In Recent Advances in Hydraulic Fracturing; Gidley, J.L., Holditch, S.A., Nierode, D.E., Veatch, R.W., Jr., Eds.; Society of Petroleum: Richardson, TX, USA, 1989; pp. 147–176. [Google Scholar]
- Settari, A. Modeling of Acid-Fracturing Treatments. SPE Prod. Facil. 1993, 8, 30–38. [Google Scholar] [CrossRef]
- Terrill, R.M. Heat transfer in laminar flow between parallel porous plates. Int. J. Heat Mass Transf. 1965, 8, 1491–1497. [Google Scholar] [CrossRef]
- De Rozieres, J. Measuring Diffusion Coefficients in Acid Fracturing Fluids and Their Application to Gelled and Emulsified Acids. In Proceedings of the SPE Annual Technical Conference and Exhibition, New Orleans, Louisiana, 25–28 September 1994. [Google Scholar] [CrossRef]
- Valko, P.; Norman, L.; Daneshy, A. Petroleum Well Construction (Book)|Etdeweb; Well Stimulation; Wiley: Hoboken, NJ, USA, 1998; Chapter 17; p. 506. Available online: https://www.osti.gov/Etdeweb/Biblio/300026 (accessed on 30 November 2021).





| Parameter | Value | Unit |
|---|---|---|
| Formation properties | ||
| Young’s Modulus (E) | 6 | MMPsi |
| Poisson Ratio () | 0.25 | - |
| Porosity () | 0.071 | - |
| Permeability (k) | 0.4 | md |
| Wormhole breakthrough pore volume () | 1.5 | - |
| Layer Thickness (H) | 50 | m |
| Closure Stress () | 4200 | Psi |
| Total compressibility () | 1.983 × 10−5 | |
| Reservoir Oil Viscosity () | 1.66 | cp |
| Formation Rock Density () | 2600 | |
| Reservoir Temperature (T) | 246 | °F |
| Reservoir Pressure () | 3000 | Psi |
| Fracturing Pressure () | 4300 | Psi |
| Acid Properties | ||
| Density () | 1000 | |
| Acid initial concentration () | 4.4 (16%) | |
| Spurt loss () | 0 | m |
| Fraction of acid to react before leaking off () | 0.3 | - |
| Reaction order () | 0.63 | - |
| Reaction rate coefficient () | 0.3263 | |
| Parameters of UFD method | ||
| Volumetric dissolving power () | 0.082 | - |
| Drainage radius () | 1900 | Ft |
| Dimensionless horizontal correlation length () | 1 | - |
| Dimensionless vertical correlation length () | 0.05 | - |
| Dimensionless standard deviation of permeability () | 0.4 | - |
| 0 | 1.68231 | −2.26693 | 6.7544 | −1.8408 | 6.7593 | −4.6274 |
| 1 | 5.67053 | −0.696 | 17.2931 | −2.9304 | 1.0032 | −3.4376 |
| 2 | 9.66842 | −0.39587 | 10.7745 | −0.5564 | −5.7028 | −0.4705 |
| 3 | 13.66772 | −0.27662 | 7.9375 | −0.1358 | −9.15 | −0.5668 |
| 4 | 17.6674 | −0.21305 | 6.34331 | −0.0373 | −12.4496 | −0.71196 |
| 0 | 9.10378 | −2.38279 | 14.9298 | −8.97017 | −7.08188 | −1.18392 |
| 1 | 0.53126 | 1.88909 | −12.5375 | 8.13482 | 4.01538 | 0.35148 |
| 2 | 0.15272 | 0.39035 | −1.6607 | 0.68079 | 1.0394 | 0.5154 |
| 3 | 0.06807 | 0.0733 | −0.4172 | 0.11131 | 0.58639 | 0.14123 |
| 4 | 0.03737 | 0.01901 | −0.1503 | 0.02756 | 0.35277 | 0.05623 |
| Acid Types | n | |||
|---|---|---|---|---|
| Straight | 1 | 0.0000213 | 1.0 | 0.00109 |
| Gelled | 15 | 0.000008 | 0.65 | 0.05 |
| Emulsified | 30 | 2.64 × 10−8 | 0.675 | 0.315 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lotfi, R.; Hosseini, M.; Aftabi, D.; Baghbanan, A.; Xu, G. Pumping Schedule Optimization in Acid Fracturing Treatment by Unified Fracture Design. Energies 2021, 14, 8185. https://doi.org/10.3390/en14238185
Lotfi R, Hosseini M, Aftabi D, Baghbanan A, Xu G. Pumping Schedule Optimization in Acid Fracturing Treatment by Unified Fracture Design. Energies. 2021; 14(23):8185. https://doi.org/10.3390/en14238185
Chicago/Turabian StyleLotfi, Rahman, Mostafa Hosseini, Davood Aftabi, Alireza Baghbanan, and Guanshui Xu. 2021. "Pumping Schedule Optimization in Acid Fracturing Treatment by Unified Fracture Design" Energies 14, no. 23: 8185. https://doi.org/10.3390/en14238185
APA StyleLotfi, R., Hosseini, M., Aftabi, D., Baghbanan, A., & Xu, G. (2021). Pumping Schedule Optimization in Acid Fracturing Treatment by Unified Fracture Design. Energies, 14(23), 8185. https://doi.org/10.3390/en14238185







