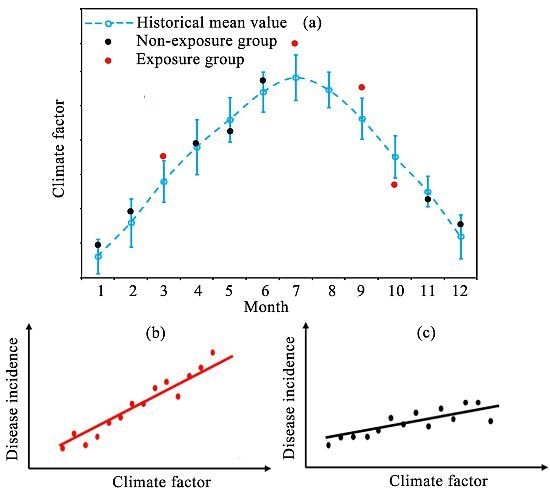
A Method for Screening Climate Change-Sensitive Infectious Diseases
Abstract
:1. Introduction
2. Methodology
| Group | Disease | Disease | Total |
|---|---|---|---|
| Yes | No | ||
| Exposure group | A | B | A+B |
| Non-Exposure group | C | D | C+D |
| Total | A+C | B+D | A+B+ C+D |
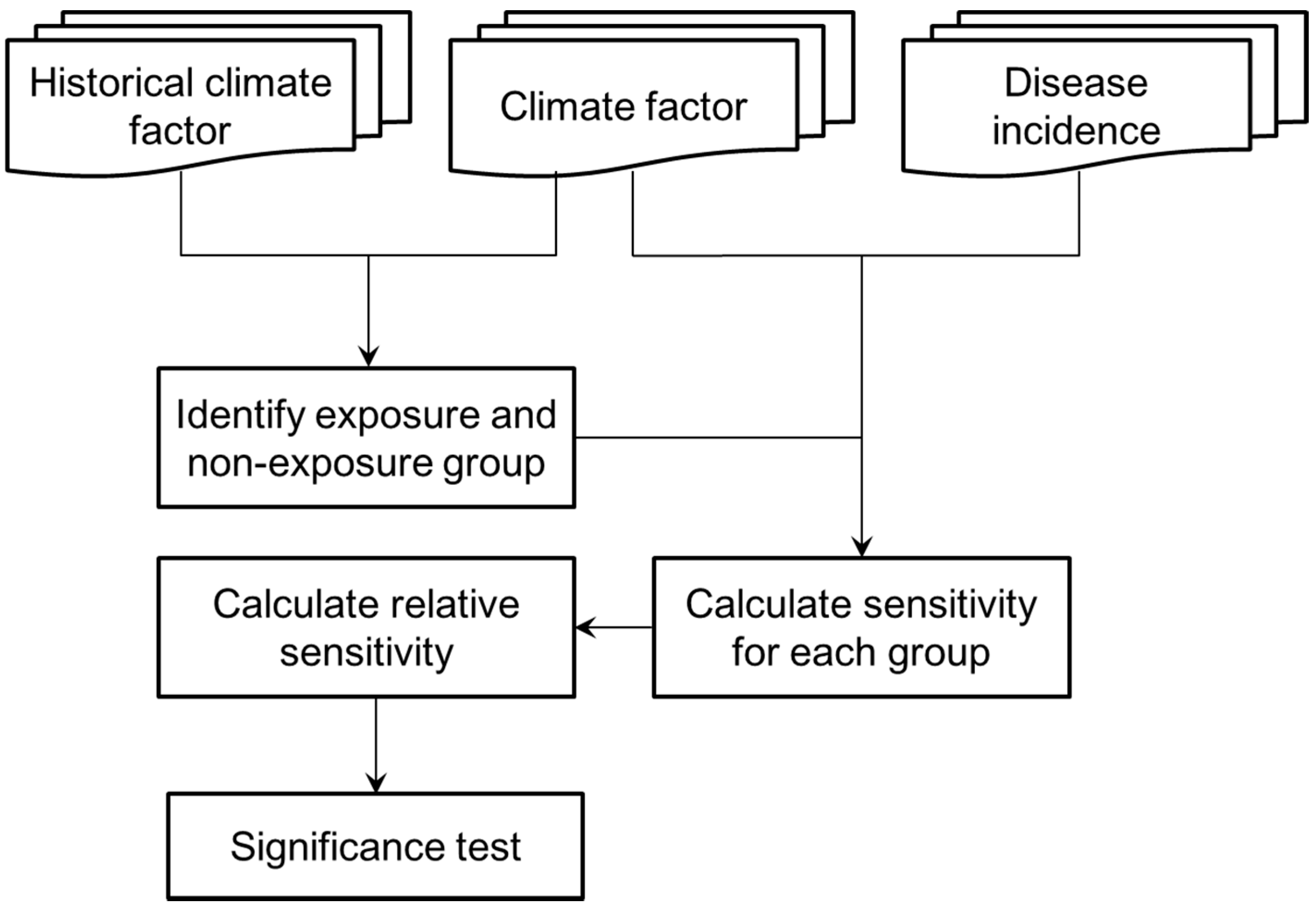

3. Application
3.1. Study Area and Data Processing
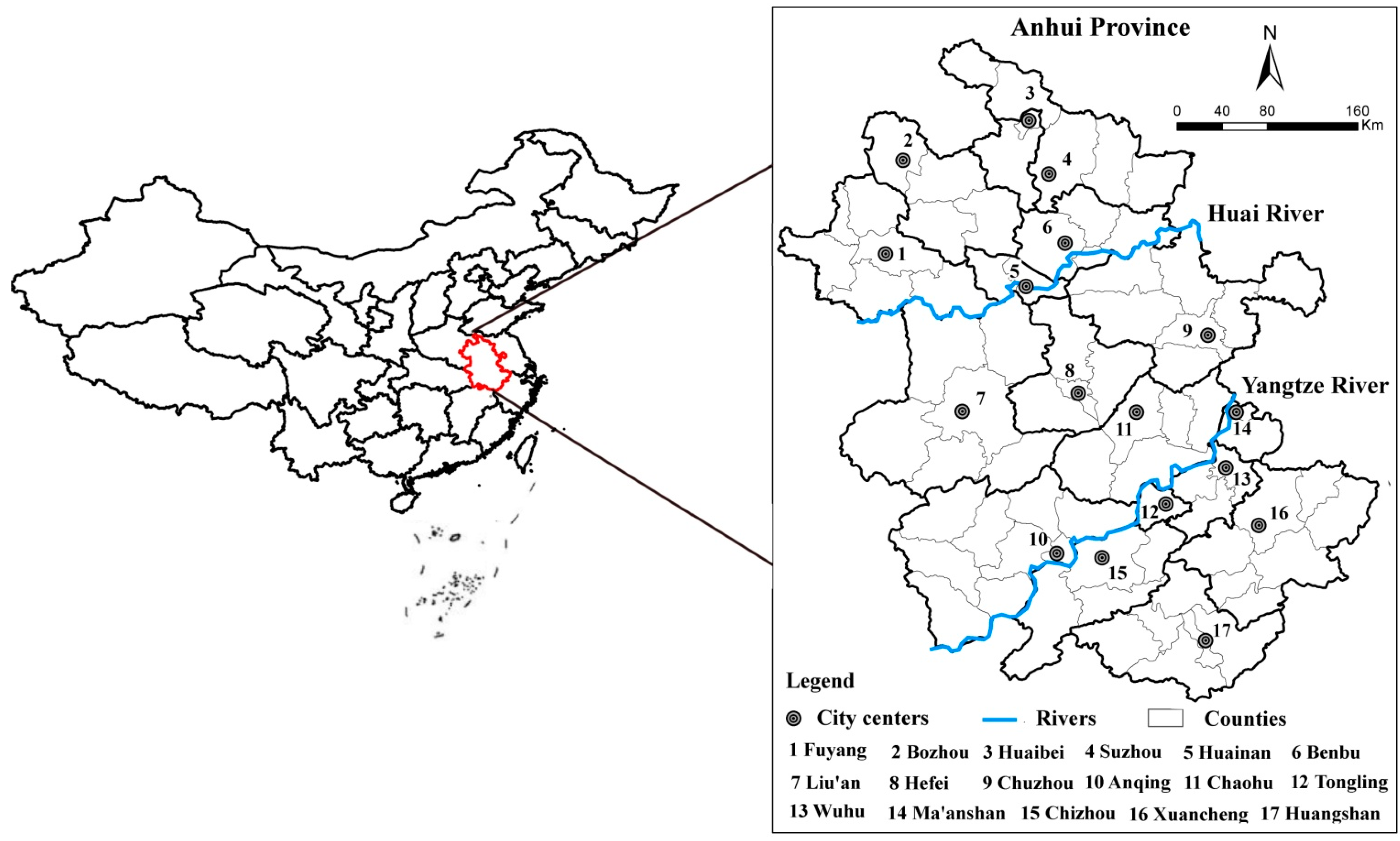
3.2. Results
3.2.1. Characteristics of Climate Variables in Exposure and Non-exposure Group
| Climate Variables | Groups | | PCi − HCimean| | |||
|---|---|---|---|---|---|
| Average | Maximum | Minimum | Standard Deviation | ||
| Temperature (°C) | Exposure | 2.11 | 4.57 | 0.75 | 0.76 |
| Non-exposure | 0.53 | 1.63 | 0.0001 | 0.34 | |
| Precipitation (m) | Exposure | 5.85 | 40.47 | 0.15 | 4.63 |
| Non-exposure | 1.84 | 17.56 | 0.0008 | 1.96 | |
| Absolute Humidity (mg/L) | Exposure | 115.24 | 253.05 | 34.65 | 44.69 |
| Non-exposure | 38.30 | 115.15 | 0.002 | 25.35 | |
3.2.2. Screening Results for Climate Change-Sensitive Disease
| Diseases | Monthly Average Temperature | Monthly Accumulated Precipitation | Monthly Average Absolute Humidity | |||
|---|---|---|---|---|---|---|
| Effective Counties | Counties with RS ≠ 0 * (Proportion) | Effective Counties | Counties with RS ≠ 0 * (Proportion) | Effective Counties | Counties with RS ≠ 0 * (Proportion) | |
| Dysentery | 77 | 35 (45%) | 77 | 51 (66%) | 77 | 73 (95%) |
| Hand, foot and mouth | 76 | 48 (63%) | 76 | 33 (43%) | 76 | 72 (95%) |
| Hepatitis A | 69 | 33 (48%) | 68 | 40 (59%) | 68 | 37 (54%) |
| Malaria | 36 | 21 (59%) | 36 | 10 (28%) | 36 | 30 (83%) |
| Influenza | 29 | 20 (69%) | 29 | 10 (34%) | 29 | 18 (62%) |
| Typhoid fever | 16 | 10 (63%) | 15 | 9 (60%) | 16 | 10 (63%) |
| Hemorrhagic fever | 10 | 1 (10%) | 10 | 2 (20%) | 10 | 6 (60%) |
| Meningitis | 8 | 4(50%) | 8 | 3 (38%) | 8 | 7 (88%) |
| Schistosomiasis | 9 | 6 (67%) | 7 | 4 (57%) | 9 | 5 (56%) |
3.2.3. Spatial Distribution for RS of the Climate Change-Sensitive Disease
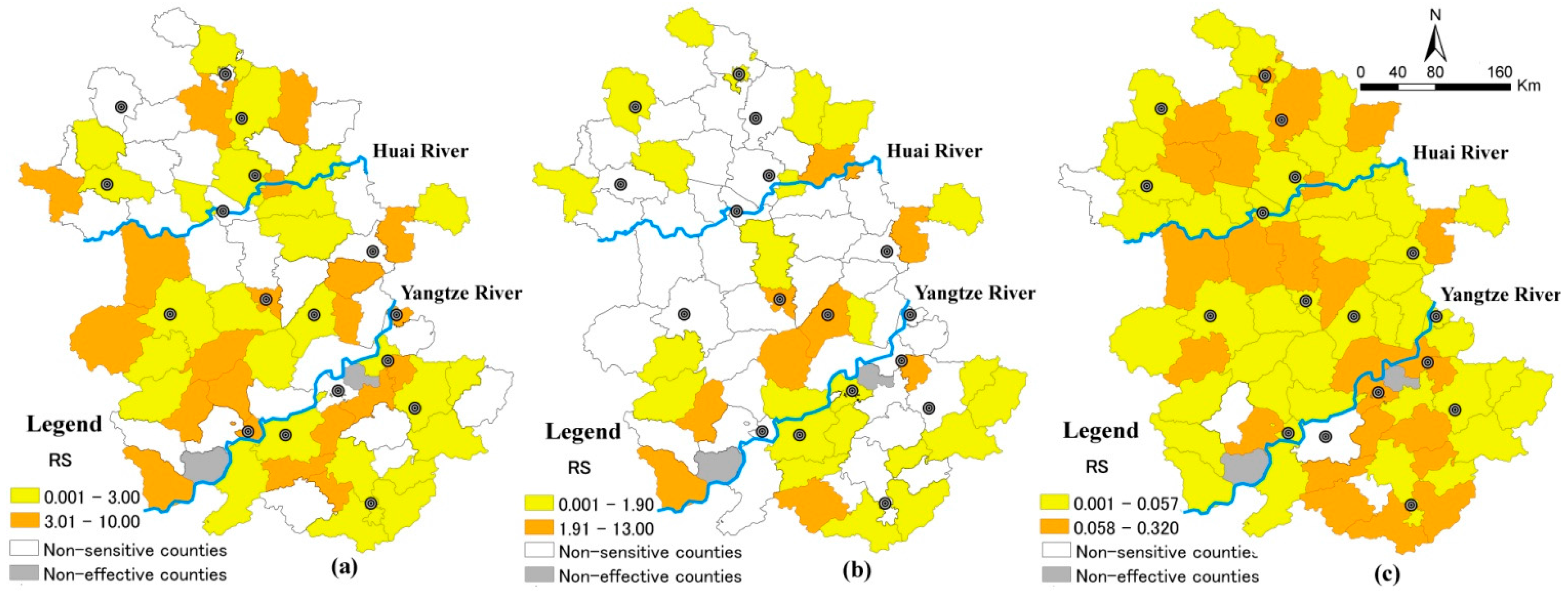
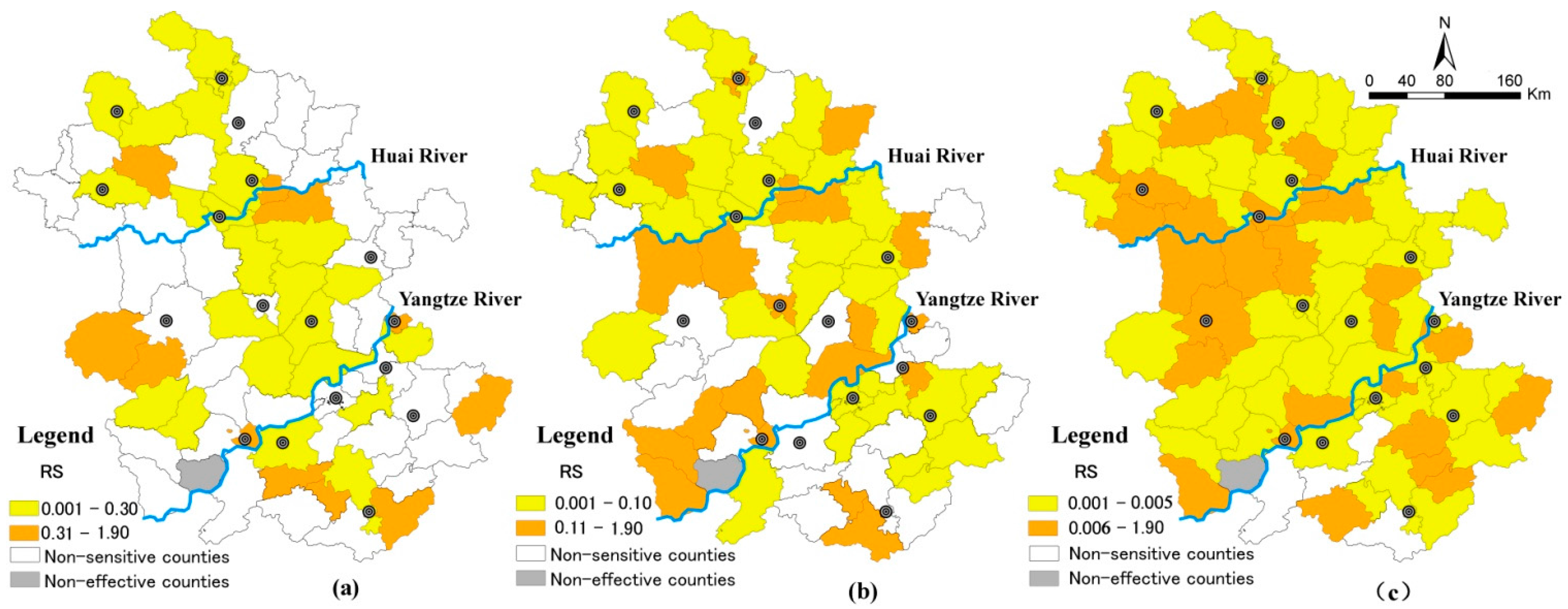
3.3. Discussions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- IPCC. Climate Change 2013: The Physical Science Basis; Stocker, T.F., Qin, D., Plattner, G.K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK and New York, NY, USA, 2013. [Google Scholar]
- Kovats, R.S.; Menne, B.; McMichael, A.J.; Corvalan, C.; Bertollini, R. Climate Change and Human Health: Impact and Adaptation; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Altizer, S.; Ostfeld, R.S.; Johnson, P.T.; Kutz, S.; Harvell, C.D. Climate change and infectious diseases: From evidence to a predictive framework. Science 2013, 341, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Epstein, P.R. Climate change and emerging infectious diseases. Microbes Infect. 2001, 3, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, K.; Campbell–Lendrum, D.; Haines, A.; Cox, J. Using Climate to Predict Infectious Disease Epidemics; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Wu, X.; Tian, H.; Zhou, S.; Chen, L.; Xu, B. Impact of global change on transmission of human infectious diseases. Sci. China. Earth. Sci. 2014, 57, 189–203. [Google Scholar] [CrossRef]
- Leeson, H. Longevity of anopheles maculipennis race atroparvus, van thiel, at controlled temperature and humidity after one blood meal. Bull. Entomol. Res. 1939, 30, 103–301. [Google Scholar] [CrossRef]
- Patz, J.A.; Epstein, P.R.; Burke, T.A.; Balbus, J.M. Global climate change and emerging infectious diseases. JAMA 1996, 275, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Beck-Johnson, L.M.; Nelson, W.A.; Paaijmans, K.P.; Read, A.F.; Thomas, M.B. The effect of temperature on anopheles mosquito population dynamics and the potential for malaria transmission. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Bunyavanich, S.; Landrigan, C.P.; McMichael, A.J.; Epstein, P.R. The impact of climate change on child health. Ambul. Pediatr. 2003, 3, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhuang, J.; Yang, M.; Zhang, Z.; Wei, J.; Peng, W.; Jiang, Q. Effects of low temperature on the schistosome-transmitting snail Oncomelania hupensis and the implications of global climate change. Molluscan. Res. 2010, 30, 102–108. [Google Scholar]
- Lloyd, J.S.; Kovats, R.S.; Armstrong, B.G. Global diarrhoea morbidity, weather and climate. Climate Res. 2007, 34, 119–127. [Google Scholar] [CrossRef]
- Chou, W.; Wu, J.; Wang, Y.; Huang, H.; Sung, F.; Chuang, C. Modeling the impact of climate variability on diarrhea-associated diseases in Taiwan (1996–2007). Sci. Total. Environ. 2010, 409, 43–51. [Google Scholar] [CrossRef]
- Stenseth, N.C.; Mysterud, A. Climate, changing phenology, and other life history traits: Nonlinearity and match-mismatch to the environment. Proc. Natl. Acad. Sci. 2002, 99, 13379–13381. [Google Scholar] [CrossRef] [PubMed]
- Iler, A.M.; Høye, T.T.; Inouye, D.W.; Schmidt, N.M. Nonlinear flowering responses to climate: Are species approaching their limits of phenological change? Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2013, 368. [Google Scholar] [CrossRef] [PubMed]
- Woolf, R. On estimating the relation between blood group and disease. Ann. Hum. Genet. 1955, 19, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Falk, C.T.; Rubinstein, P. Haplotype relative risks: An easy reliable way to construct a proper control sample for risk calculations. Ann. Hum. Genet. 1987, 51, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yu, K.F. What’s relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 1998, 280, 1690–1691. [Google Scholar] [CrossRef] [PubMed]
- Last, J.M. A Dictionary of Epidemiology,, 4th ed.; Oxford University Press/International Epidemiological Association: New York, NY, USA, 2001. [Google Scholar]
- WikiHow. http://www.wikihow.com/Calculate-Relative-Risk (accessed on 22 May 2013).
- Seber, G.A.F.; Lee, A.J. Linear Regression Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 154–159. [Google Scholar]
- Hu, M.; Li, Z.; Wang, J.; Jia, L.; Liao, Y. Determinants of the incidence of hand, foot and mouth disease in China using geographically weighted regression models. PLoS One 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zheng, X.; Dai, Y.; Yang, C.; Chen, Z.; Zhang, S.; Wu, G.; Wang, Z.; Huang, C.; Shen, Y.; Liao, R. Mapping near-surface air temperature, pressure, relative humidity and wind speed over mainland China with high spatiotemporal resolution. Adv. Atmos. Sci. 2014, 31, 1127–1135. [Google Scholar] [CrossRef]
- Zhang, Y.; Bi, P.; Hiller, J.E.; Sun, Y.; Ryan, P. Climate variations and bacillary dysentery in northern and southern cities of China. J. Infect. 2007, 55, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Guo, Y.; Christakos, G.; Yang, W.; Liao, Y.; Li, Z.; Li, X.; Lai, S.; Chen, H. Hand, foot and mouth disease: Spatiotemporal transmission and climate. Int. J. Health. Geogr. 2011, 25, 1–10. [Google Scholar]
- Hii, Y.L.; Rock lÖv, J.; Ng, N. Short term effects of weather on hand, foot and mouth disease. PLoS One 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Ma, E.; Lam, T.; Wong, C.; Chuang, S.K. Is hand, foot and mouth disease associated with meteorological parameters? Epidemiol. Infect. 2010, 138, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Onozuka, D.; Hashizume, M.; Hagihara, A. Effects of weather variability on infectious gastroenteritis. Epidemiol. Infect. 2010, 138, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Q.; Luan, R.; Liu, X.; Zhou, G.; Jiang, J.; Li, H.; Li, Z. Spatial-temporal analysis of malaria and the effect of environmental factors on its incidence in Yongcheng, China, 2006–2010. BMC Public Health 2012, 544, 1–13. [Google Scholar]
- Parham, P.E.; Michael, E. Modeling climate change and malaria transmission. Adv. Exp. Med. Biol. 2010, 673, 184–199. [Google Scholar] [PubMed]
- Barreca, A.I.; Shimshack, J.P. Absolute humidity, temperature, and influenza mortality: 30 years of county-level evidence from the United States. Am. J. Epidemiol. 2012, 176, 114–122. [Google Scholar] [CrossRef]
- Jaakkola, K.; Saukkoriipi, A.; Jokelainen, J.; Juvonen, R.; Kauppila, J.; Vainio, O.; Ziegler, T.; Rönkkö, E.; Jaakkola Jouni, J.K.; Ikäheimo, T.M.; KIAS-Study Group. Decline in temperature and humidity increases the occurrence of influenza in cold climate. Environ. Health 2014, 22, 1–8. [Google Scholar]
- Zhang, K.; Huang, S.; Shi, C. Effect of meteorological and geological factors on epidemic of typhoid fever/paratyphoid fever in Guilin. Chin. J. Dis. Control Prevent. 2009, 5, 520–523. [Google Scholar]
- Qu, B.; Huang, D.; Guo, H.; Zhou, B.; Dong, C.; Lu, J. The model of back-propagation neural network about meteorological factors and Typhoid Fever, Paratyphoid Fever in a drought area. Chin. Health Stat. 2004, 11, 333–337. [Google Scholar]
- Abdussalam, A.F.; Monaghan, A.J.; Steinhoff, D.F.; Dukic, V.M.; Hayden, M.H.; Hopson, T.M.; Thornes, J.E.; Leckebusch, G.C. The impact of climate change on meningitis in Northwest Nigeria: An assessment using CMIP5 climate model simulations. Wea. Climate. Soc. 2014, 6, 371–379. [Google Scholar] [CrossRef]
- Yang, G.; Vounatsou, P.; Zhou, X.; Tanner, M.; Utzinger, J. A potential impact of climate change and water resource development on the transmission of Schistosoma japonicum in China. Parassitologia 2005, 47, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Ding, G.; Zhang, Y.; Gao, L.; Ma, W.; Li, X. Quantitative analysis of burden of infectious diarrhea associated with floods in northwest of Anhui province, China: A mixed method evaluation. PLoS One 2013, 8, 1–8. [Google Scholar]
- Li, Z.; Wang, L.; Sun, W.; Hou, X.; Yang, H.; Sun, L.; Xu, S.; Sun, Q.; Zhang, J.; Song, H.; Lin, H. Identifying high-risk areas of bacillary dysentery and associated meteorological factors in Wuhan, China. Sci. Rep. 2013, 21, 1–5. [Google Scholar]
- Morand, S.; Owers, K.A.; Waret-Szkuta, A.; McIntyre, K.M.; Baylis, M. Climate variability and outbreaks of infectious diseases in Europe. Sci. Rep. 2013, 3, 1–6. [Google Scholar] [CrossRef]
- Huang, Y.; Ren, Z.; Hang, D.; Hong, Q.; Gao, Y.; Guo, J.; Sun, D.; Zuo, Y. Potential effect of climate changes on schistosomiasis japonica transmission in east route of South-to-North Water Diversion Project. Chin. J. Schist. Control 2009, 21, 197–204. [Google Scholar]
- Oregon State University. http://oregonstate.edu/ua/ncs/archives/2010/feb/absolute-humidity-temperature-anomalies-tied-seasonal-outbreaks-influenza-0 (accessed on 5 May 2014).
- Tunde, A.M.; Adeleke, E.A.; Adeniyi, E.E. Impact of climate variability on human health in Ilorin, Nigeria. Environ. Natl. Resour. Res. 2013, 3, 127–134. [Google Scholar]
- Shaman, J.; Kohn, M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. USA 2009, 106, 3243–3248. [Google Scholar] [CrossRef] [PubMed]
- Shaman, J.; Pitzer, V.E.; Viboud, C.; Grenfell, B.T.; Lipsitch, M. Absolute humidity and the seasonal onset of influenza in the continental United States. PLoS Biol. 2010, 8, 1–13. [Google Scholar]
- Shaman, J.; Goldstein, E.; Lipsitch, M. Absolute humidity and pandemic versus epidemic influenza. Am. J. Epidemiol. 2011, 173, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Kurane, I. The effect of global warming on infectious diseases. Public Health Res. Perspect. 2010, 1, 4–9. [Google Scholar] [CrossRef]
- Adu-Prah, S.; Tetteh, E.K. Spatiotemporal analysis of climate variability impacts on malaria prevalence in Ghana. Appl. Geogr. 2014, 12, 1–8. [Google Scholar]
- Yeager, J.G.; O’Brien, R.T. Enterovirus inactivation in soil. Appl. Environ. Microbiol. 1979, 38, 694–701. [Google Scholar] [PubMed]
- Kung, C.; King, C.; Lee, C.; Huang, L.; Lee, P. Differences in replication capacity between enterovirus 71 isolates obtained from patients with encephalitis and those obtained from patients with herpangina in Taiwan. J. Med. Virol. 2007, 79, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Onozuka, D.; Hashizume, M. The influence of temperature and humidity on the incidence of hand, foot, and mouth disease in Japan. Sci. Total Environ. 2011, 410–411, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Deng, T.; Yu, S.; Gu, J.; Huang, C.; Xiao, G. Effect of meteorological variables on the incidence of hand, foot, and mouth disease in children: a time-series analysis in Guangzhou, China. BMC Infect. Dis. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Lin, H.; Li, X.; Lang, L.; Xiao, X.; Ding, P. Short-term effects of meteorological factors on children hand, foot and mouth disease in Guangzhou, China. Int. J. Biometeorol. 2014, 58, 1605–1614. [Google Scholar] [CrossRef] [PubMed]
- Anhui Statistical Bureau. Available online: http://www.ahtjj.gov.cn/tjj/web/tjnjview.jsp?strColId=13787135717978521&_index=1 (accessed on 20 March 2014).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Rao, Y.; Wu, X.; Zhao, H.; Chen, J. A Method for Screening Climate Change-Sensitive Infectious Diseases. Int. J. Environ. Res. Public Health 2015, 12, 767-783. https://doi.org/10.3390/ijerph120100767
Wang Y, Rao Y, Wu X, Zhao H, Chen J. A Method for Screening Climate Change-Sensitive Infectious Diseases. International Journal of Environmental Research and Public Health. 2015; 12(1):767-783. https://doi.org/10.3390/ijerph120100767
Chicago/Turabian StyleWang, Yunjing, Yuhan Rao, Xiaoxu Wu, Hainan Zhao, and Jin Chen. 2015. "A Method for Screening Climate Change-Sensitive Infectious Diseases" International Journal of Environmental Research and Public Health 12, no. 1: 767-783. https://doi.org/10.3390/ijerph120100767
APA StyleWang, Y., Rao, Y., Wu, X., Zhao, H., & Chen, J. (2015). A Method for Screening Climate Change-Sensitive Infectious Diseases. International Journal of Environmental Research and Public Health, 12(1), 767-783. https://doi.org/10.3390/ijerph120100767