Formulation of Multicomponent Chrysin-Hydroxy Propyl β Cyclodextrin-Poloxamer Inclusion Complex Using Spray Dry Method: Physicochemical Characterization to Cell Viability Assessment
Abstract
1. Introduction
2. Results and Discussions
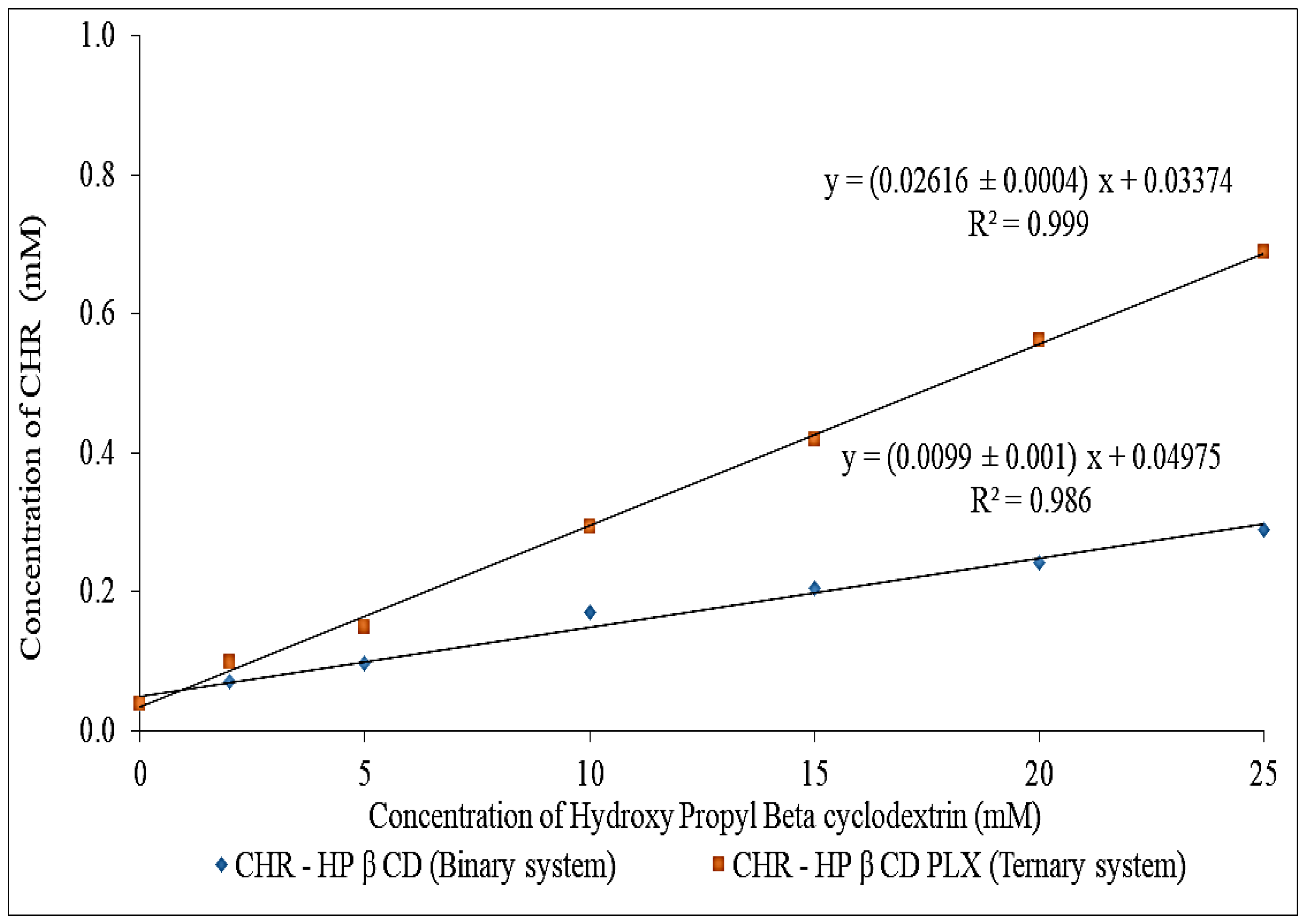
2.1. Phase Solubility Assessment
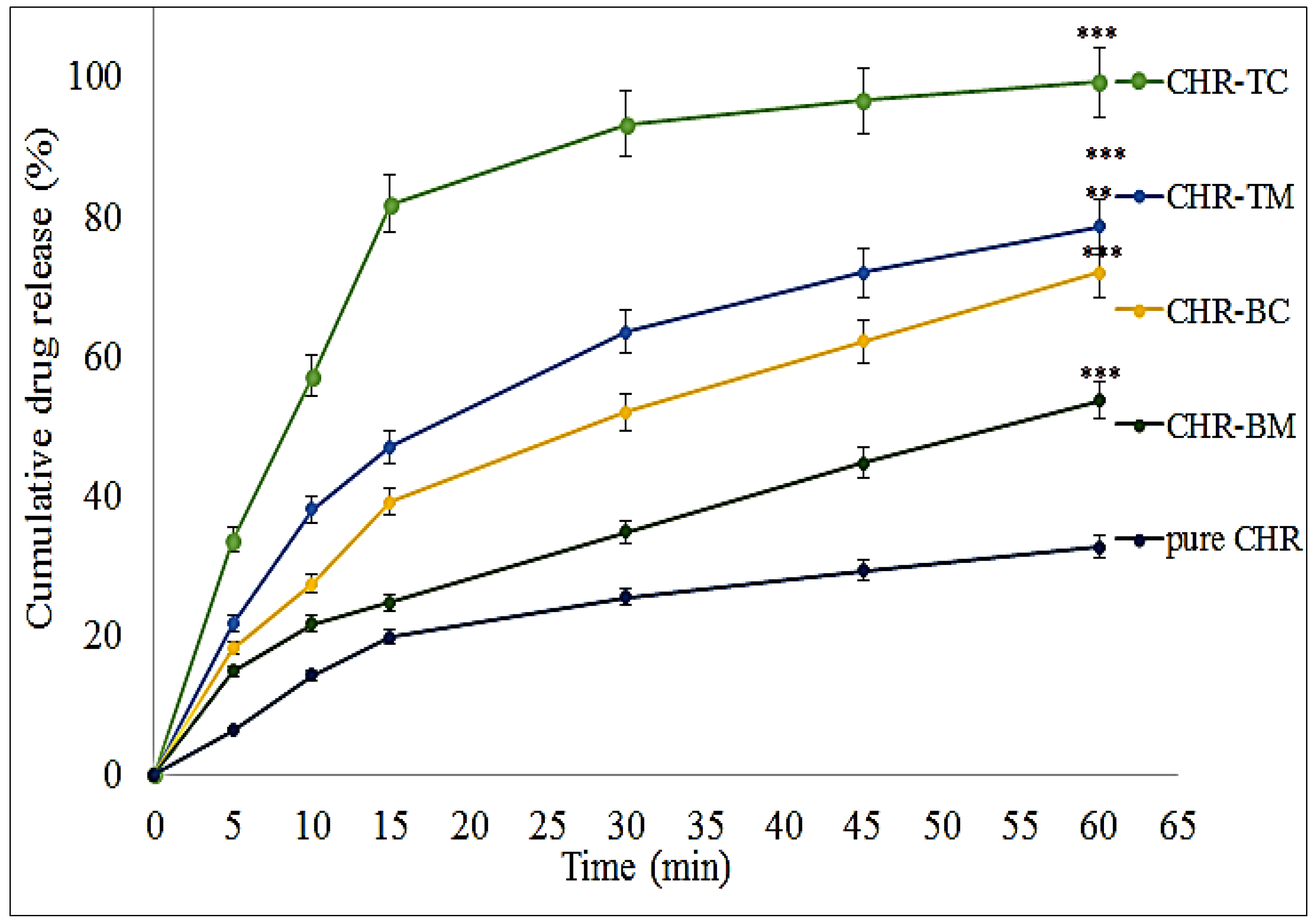
2.2. Dissolution Study
2.3. Particle Characterization
2.4. Surface Morphology
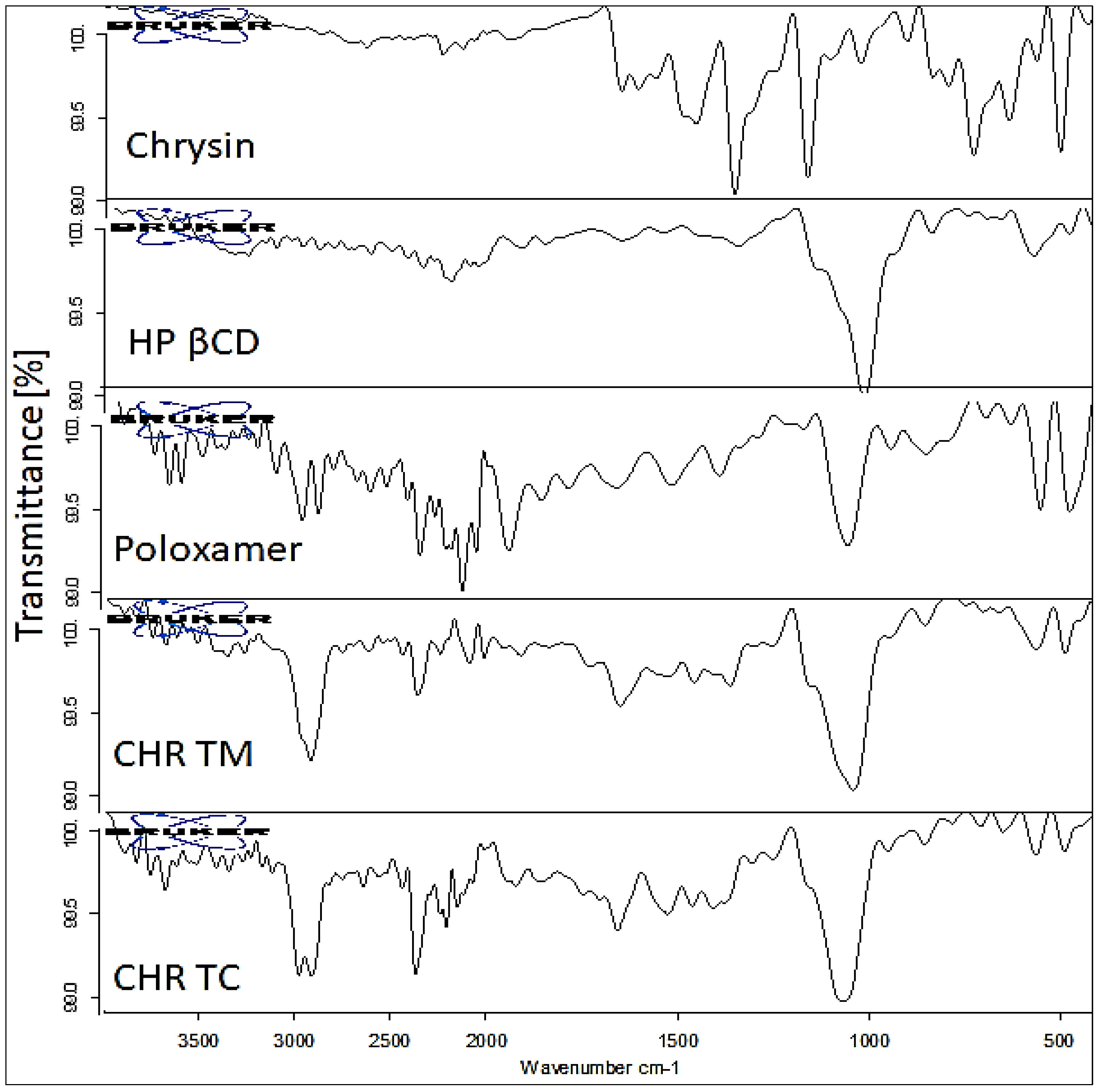
2.5. Fourier-Transformed Infrared Spectroscopy
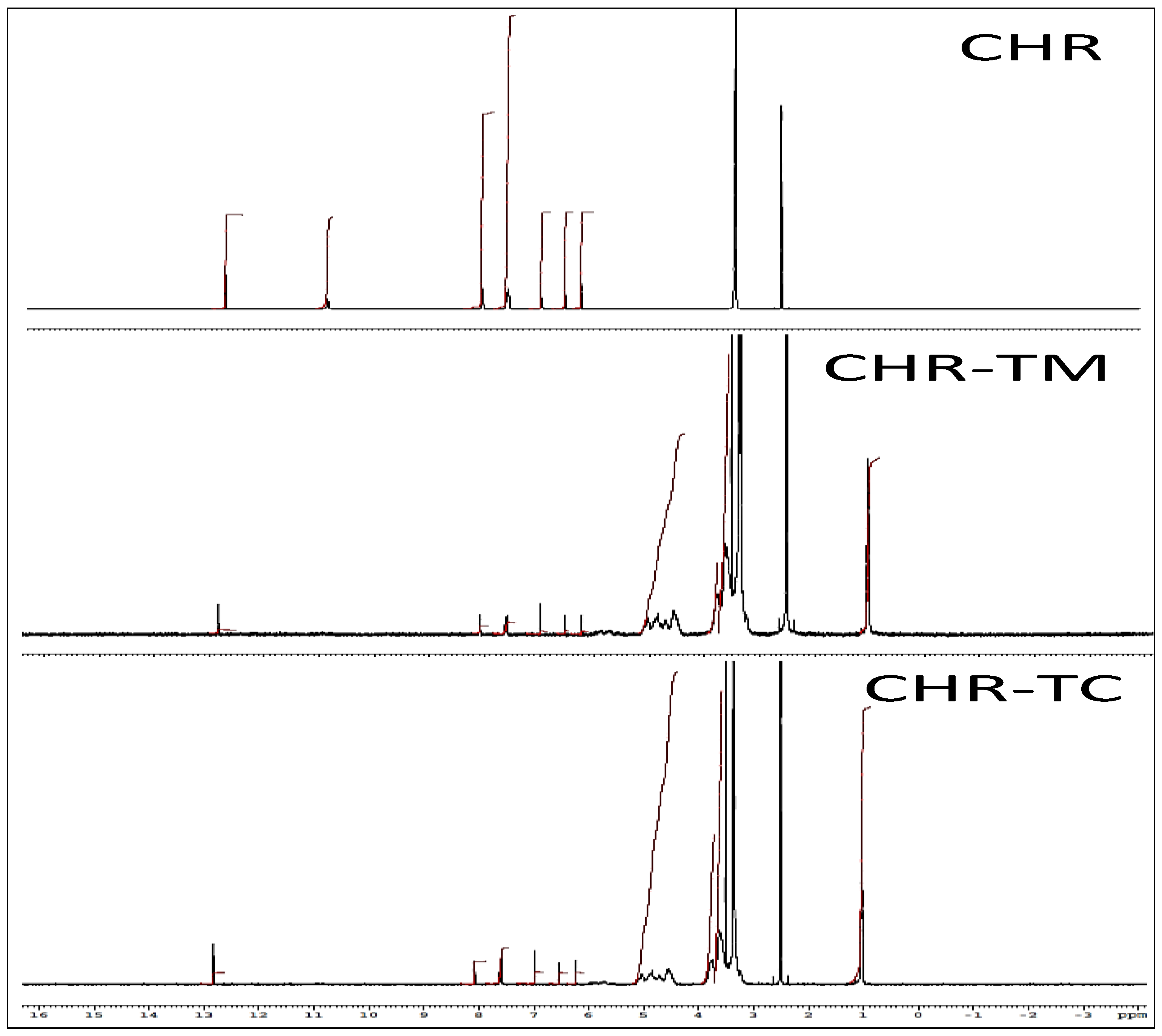
2.6. Nuclear Magnetic Resonance
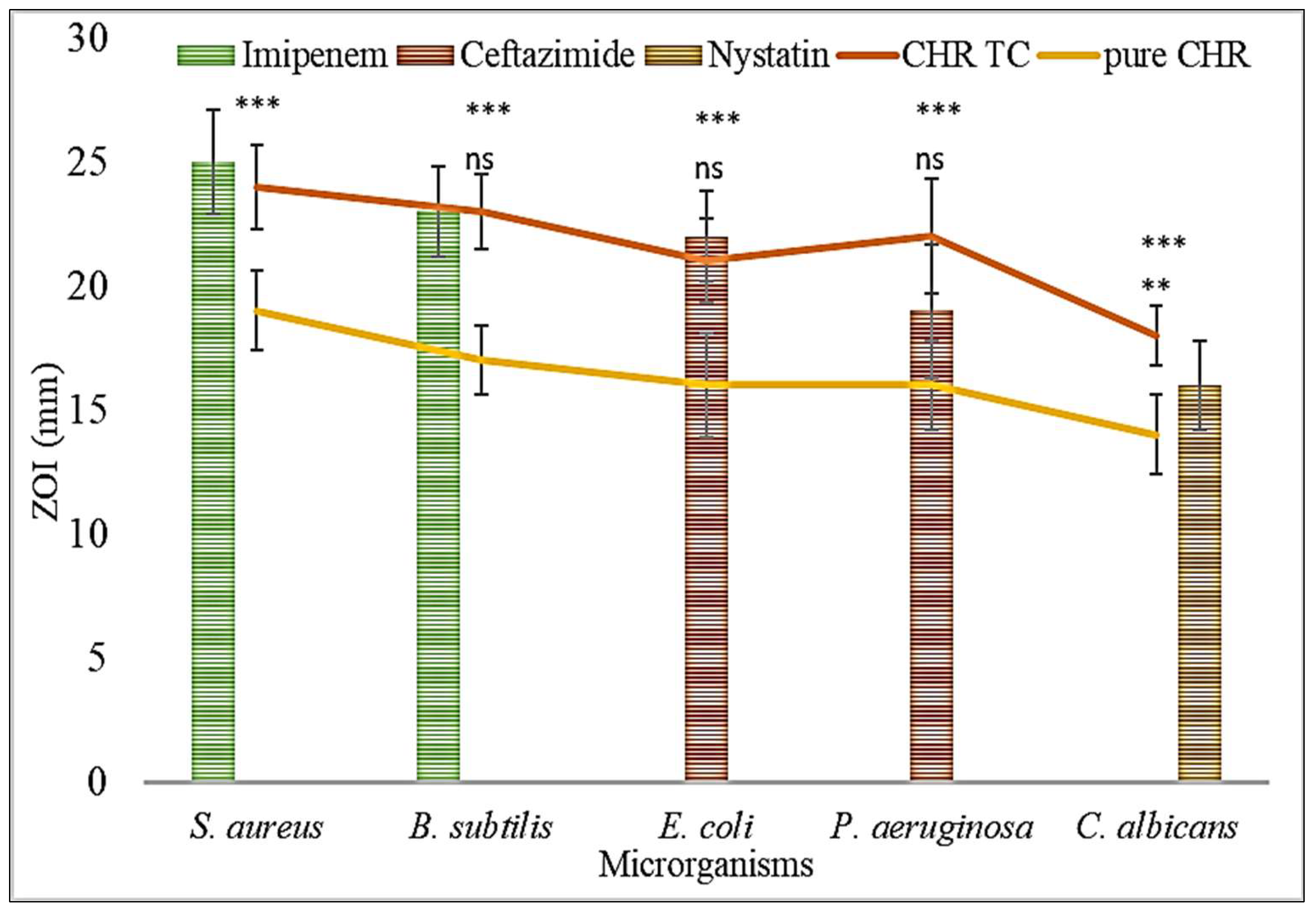
2.7. Antimicrobial Activity
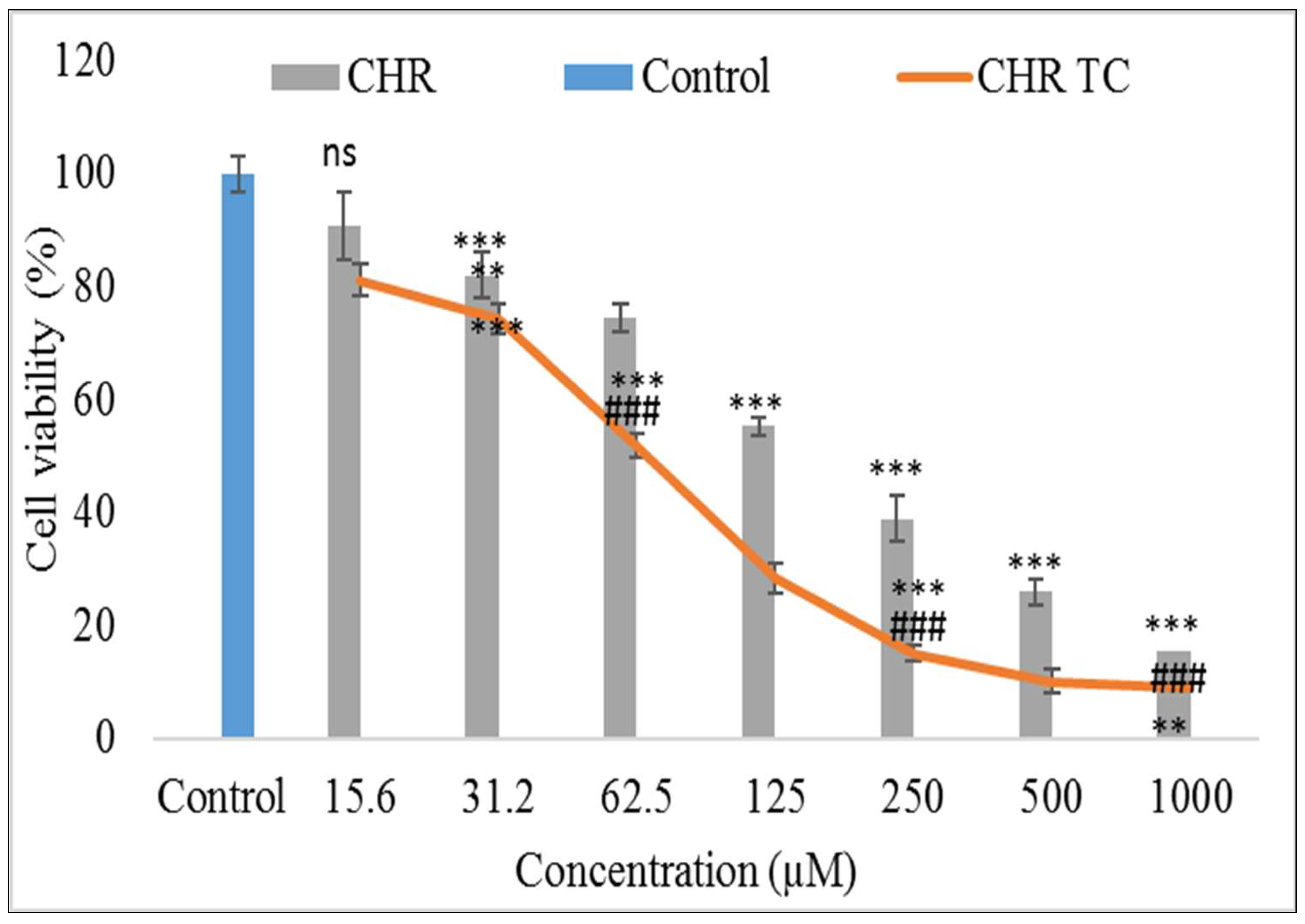
2.8. Cell Viability
3. Materials and Methods
3.1. Phase Solubility Assessement
3.2. Formulation of Inclusion Complex
3.3. Dissolution Study
3.4. Particle Characterization
3.5. Surface Morphology
3.6. Fourier Transform Infrared Spectroscopy
3.7. Nuclear Magnetic Resonance
3.8. Antimicrobial Study
3.9. Cell Viability Assessement
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zarzecki, M.S.; Araujo, S.M.; Bortolotto, V.C.; de Paula, M.T.; Jesse, C.R.; Prigol, M. Hypolipidemic action of chrysin on Triton WR-1339-induced hyperlipidemia in female C57BL/6 mice. Toxicol. Rep. 2014, 1, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Pushpavalli, G.; Kalaiarasi, P.; Veeramani, C.; Pugalendi, K.V. Effect of chrysin on hepatoprotective and antioxidant status in d-galactosamine-induced hepatitis in rats. Eur. J. Pharmacol. 2010, 631, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Otake, Y.; Brubaker, J.A.; Walle, U.K.; Halushka, P.V. Disposition and metabolism of the flavonoid chrysin in normal volunteers. Br. J. Clin. Pharmacol. 2001, 51, 143–146. [Google Scholar] [PubMed]
- Timcheh-Hariri, A.; Balali-Mood, M.; Aryan, E.; Sadeghi, M.; Riahi-Zanjani, B. Toxic hepatitis in a group of 20 male body-builders taking dietary supplements. Food Chem. Toxicol. 2012, 50, 3826–3832. [Google Scholar] [CrossRef] [PubMed]
- Ramalho, Í.M.d.M.; Bezerra, G.S.; Ostrosky, E.A.; Ferrari, M.; Oliveira, V.d.S.; Wanderley Neto, A.d.O.; Quintans, J.d.S.S.; Passos, F.R.S.; Heimfarth, L.; Quintans-Júnior, L.J.; et al. Chrysin-Loaded Microemulsion: Formulation Design, Evaluation and Antihyperalgesic Activity in Mice. Appl. Sci. 2022, 12, 477. [Google Scholar] [CrossRef]
- Walle, T. Absorption and metabolism of flavonoids. Free Radic. Biol. Med. 2004, 36, 829–837. [Google Scholar] [CrossRef]
- Walle, T. Methylation of dietary flavones increases their metabolic stability and chemopreventive effects. Int. J. Mol. Sci. 2009, 10, 5002–5019. [Google Scholar] [CrossRef]
- Crini, G. Review: A history of cyclodextrin. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef]
- Song, S.; Gao, K.; Niu, R.; Wang, J.; Zhang, J.; Gao, C.; Yang, B.; Liao, X. Inclusion complexes between chrysin and amino-appended β-cyclodextrins (ACDs): Binding behavior, water solubility, in vitro antioxidant activity and Cytotoxicity. Mat. Sci. Eng. C 2020, 106, 110161. [Google Scholar] [CrossRef]
- Srivalli, K.M.R.; Mishra, B. Improved aqueous solubility and antihypercholesterolemic activity of ezetimibe on formulating with Hydroxypropyl-β-cyclodextrin and hydrophilic auxiliary substances. AAPS PharmSciTech 2016, 17, 272–283. [Google Scholar] [CrossRef]
- Zoeller, T.; Dressman, J.B.; Klein, S. Application of a ternary HP-β-CD-complex approach to improve the dissolution performance of a poorly soluble weak acid under biorelevant conditions. Int. J. Pharm. 2012, 430, 176–183. [Google Scholar] [CrossRef]
- Patel, M.; Hirlekar, R. Multicomponent cyclodextrin system for improvement of solubility and dissolution rate of poorly water soluble drug. Asian J. Pharm. Sci. 2019, 14, 104–115. [Google Scholar] [CrossRef]
- Mennini, N.; Maestrelli, F.; Cirri, M.; Mura, P. Analysis of physicochemical properties of ternary systems ofoxaprozin with randomly methylated-ß-cyclodextrin and l-arginineaimed to improve the drug solubility. J. Pharm. Biomed. Anal. 2016, 129, 350–358. [Google Scholar] [CrossRef]
- Loftsson, T.; Masson, M. The effects of water-soluble polymers oncyclodextrins and cyclodextrin solubilization of drugs. J. Drug Deliv. Sci. Technol. 2004, 14, 35–43. [Google Scholar] [CrossRef]
- Higuchi, T.; Connors, K.A. Phase solubility techniques. Adv. Anal. Chem. Instr. 1965, 4, 117–212. [Google Scholar]
- Zheng, Y.; Chow, A.H.L. Production and characterization of a spray-dried hydroxypropyl-β-cyclodextrin/quercetin complex. Drug Dev. Ind. Pharm. 2009, 35, 727–734. [Google Scholar] [CrossRef]
- Ezawa, T.; Inoue, Y.; Murata, I.; Takao, K.; Sugita, Y.; Kanamoto, I. Characterization of the Dissolution Behavior of Piperine/Cyclodextrins Inclusion Complexes. AAPS PharmSciTech 2018, 19, 923–933. [Google Scholar] [CrossRef]
- Kulkarni, A.; Belgamwar, V. Inclusion complex of chrysin with sulfobutyl ether-β-cyclodextrin (Captisol®): Preparation, characterization, molecular modeling and in vitro anticancer activity. J. Mol. Str. 2016, 1128, 563–571. [Google Scholar] [CrossRef]
- Bera, H.; Chekuri, S.; Sarkar, S.; Kumar, S.; Muvva, N.B.; Mothe, S.; Nadimpalli, J. Novel pimozide-β-cyclodextrin-polyvinylpyrrolidone inclusion complexes for Tourette syndrome treatment. J. Mol. Liq. 2016, 215, 135–143. [Google Scholar] [CrossRef]
- Wang, D.; Li, H.; Gu, J.; Guo, T.; Yang, S.; Guo, Z.; Zhang, X.; Zhu, W.; Zhang, J. Ternary system of dihydroartemisinin with hydroxypropyl-β-cyclodextrin and lecithin: Simultaneous enhancement of drug solubility and stability in aqueous solutions. J. Pharm. Biomed. 2013, 83, 141–148. [Google Scholar] [CrossRef]
- Sapte, S.; Pore, Y. Inclusion complexes of cefuroxime axetil with cyclodextrin: Physicochemical characterization, molecular modeling and effect of l-arginine on complexation. J. Pharm. Anal. 2016, 6, 300–306. [Google Scholar] [CrossRef]
- Kurniawansyah, I.S.; Gozali, D.; Sopyan, I.; Iqbal, M.; Subarnas, A. Physical study of chloramphenicol in situ gel with base hydroxypropyl methylcellulose and poloxamer 188. J. Pharm. Bioallied Sci. 2019, 11, S547. [Google Scholar] [CrossRef] [PubMed]
- Nogueiras-Nieto, L.; Sobarzo-Sánchez, E.; Gomez-Amoza, J.L.; Otero, F.J. Competitive displacement of drugs from cyclodextrin inclusion complex by polypseudorotaxane formation with poloxamer: Implications in drug solubilization and delivery. Eur. J. Pharm. Biopharm. 2012, 80, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, X.; Yang, Q.; Zhang, N.; Du, Y.; Zhu, H. Preparation and characterization of inclusion complex of benzyl isothiocyanate extracted from papaya seed with β-cyclodextrin. Food Chem. 2015, 184, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Mashaqbeh, H.; Obaidat, R.; Al-Shar’i, N. Evaluation and Characterization of Curcumin-β-Cyclodextrin and Cyclodextrin-Based Nanosponge Inclusion Complexation. Polymers 2021, 13, 4073. [Google Scholar] [CrossRef]
- Venuti, V.; Crupi, V.; Fazio, B.; Majolino, D.; Acri, G.; Testagrossa, B.; Stancanelli, R.; De Gaetano, F.; Gagliardi, A.; Paolino, D.; et al. Physicochemical characterization and antioxidant activity evaluation of idebenone/hydroxypropyl-β-cyclodextrin inclusion complex. Biomolecules 2019, 9, 531. [Google Scholar] [CrossRef]
- Liu, Y.; Song, X.; He, J. Synthetic derivatives of chrysin and their biological activities. Med. Chem. Res. 2014, 23, 555–563. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Cui, H.; Wu, J.; Lin, L. Improving anti-listeria activity of cheese packaging via nanofiber containing nisin-loaded nanoparticles. LWT—Food Sci. Technol. 2016, 81, 233–240. [Google Scholar] [CrossRef]
- Cui, H.; Zhou, H.; Lin, L. The specific antibacterial effect of the Salvia oil nanoliposomes against Staphylococcus aureus biofilms on milk container. Food Cont. 2016, 61, 92–98. [Google Scholar] [CrossRef]
- Parajuli, P.; Joshee, N.; Rimando, A.M.; Mittal, S.; Yadav, A.K. In vitro antitumor mechanisms of various Scutellaria extracts and constituent flavonoids. Planta Med. 2009, 75, 41–48. [Google Scholar] [CrossRef]
- Asadzadeh Vostakolaei, F.; Broeders, M.J.; Mousavi, S.M.; Kiemeney, L.A.; Verbeek, A.L. The effect of demographic and lifestyle changes on the burden of breast cancer in Iranian women: A projection to 2030. Breast 2013, 22, 277–281. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Borji, A.; Hasanzadeh, M.; Jabbari, F.; Farkhondeh, T.; Samini, M. Inhibitory and cytotoxic activities of chrysin on human breast adenocarcinoma cells by induction of apoptosis. Phcog Mag. 2016, 12, 436–440. [Google Scholar]
- Grebogi, A.V.T.; ABarison, C.P.S.G.; Ferraz, H.; Rodrigues, L.C. Binary and ternary inclusion complexes of dapsone in cyclodextrins and polymers: Preparation, characterization and evaluation. J. Incl. Phenom. Macrocycl. Chem. 2012, 73, 467–474. [Google Scholar] [CrossRef]
- Hirlekar, R.S.; Sonawane, S.N.; Kadam, V.J. Studies on the effect of water-soluble polymers on drug-cyclodextrin complex solubility. AAPS PharmSciTech 2009, 10, 858–863. [Google Scholar] [CrossRef]
- Suvarna, V.; Kajwe, A.; Murahari, M.; Pujar, G.V.; Inturi, B.K.; Sherje, A.P. Inclusion Complexes of Nateglinide with HP–β–CD and L-Arginine for Solubility and Dissolution Enhancement: Preparation, Characterization, and Molecular Docking Study. J. Pharm. Innov. 2017, 12, 168–181. [Google Scholar] [CrossRef]
- Munir, R.; Hadi, A.; Khan, S.-U.-D.; Asghar, S.; Irfan, M.; Khan, I.U.; Hameed, M.; Inam, S.; Islam, N.; Hassan, S.F.; et al. Solubility and Dissolution Enhancement of Dexibuprofen with Hydroxypropyl beta cyclodextrin (HPβCD) and Poloxamers (188/407) Inclusion Complexes: Preparation and In Vitro Characterization. Polymers 2022, 14, 579. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Luo, Y.; Liu, Y.; Wang, X.T.; Liu, F.; Guo, M.Z.; Wang, Z.; Liu, A.J.; Zhang, Y.M. Inclusion of chrysin in b-cyclodextrin and its biological activities. J. Drug Del. Sci. Technol. 2016, 31, 176–186. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imam, S.S.; Alshehri, S.; Mahdi, W.A.; Alotaibi, A.M.; Alhwaifi, M.H.; Hussain, A.; Altamimi, M.A.; Qamar, W. Formulation of Multicomponent Chrysin-Hydroxy Propyl β Cyclodextrin-Poloxamer Inclusion Complex Using Spray Dry Method: Physicochemical Characterization to Cell Viability Assessment. Pharmaceuticals 2022, 15, 1525. https://doi.org/10.3390/ph15121525
Imam SS, Alshehri S, Mahdi WA, Alotaibi AM, Alhwaifi MH, Hussain A, Altamimi MA, Qamar W. Formulation of Multicomponent Chrysin-Hydroxy Propyl β Cyclodextrin-Poloxamer Inclusion Complex Using Spray Dry Method: Physicochemical Characterization to Cell Viability Assessment. Pharmaceuticals. 2022; 15(12):1525. https://doi.org/10.3390/ph15121525
Chicago/Turabian StyleImam, Syed Sarim, Sultan Alshehri, Wael A. Mahdi, Ahmed M. Alotaibi, Moath H. Alhwaifi, Afzal Hussain, Mohammad A. Altamimi, and Wajhul Qamar. 2022. "Formulation of Multicomponent Chrysin-Hydroxy Propyl β Cyclodextrin-Poloxamer Inclusion Complex Using Spray Dry Method: Physicochemical Characterization to Cell Viability Assessment" Pharmaceuticals 15, no. 12: 1525. https://doi.org/10.3390/ph15121525
APA StyleImam, S. S., Alshehri, S., Mahdi, W. A., Alotaibi, A. M., Alhwaifi, M. H., Hussain, A., Altamimi, M. A., & Qamar, W. (2022). Formulation of Multicomponent Chrysin-Hydroxy Propyl β Cyclodextrin-Poloxamer Inclusion Complex Using Spray Dry Method: Physicochemical Characterization to Cell Viability Assessment. Pharmaceuticals, 15(12), 1525. https://doi.org/10.3390/ph15121525









