On the Electrooxidation and Amperometric Detection of NO Gas at the Pt/Nafion® Electrode
Abstract
Introduction
| Sensing | Operating | NO conc. | Detection | Ref. |
| Electrodes | Condition | range | Limit | |
| Pt/Nafion® | 0.9 V vs. SSCEd | ca. 80~ 480 μM | N. A. | [1] |
| Pt/Nafion®/cellulose acetate | 0.9 V vs. SSCEd | N. A. | N. A. | [1] |
| Pt/Nafion® | 0.86 V vs. Ag/AgCl/sat. CuClc | 0~0.4 μM | N. A. | [2] |
| Pt/Ir | 0.4~0.8 V vs. C | 0.2 nM~1 μM | N. A. | [3] |
| Pt/Poly (4,4’-DHB)a | 0.8 V vs. Ag/AgClf | ca. 0~4.5 μM | 40 nM | [4] |
| C/NiP/Nafion® b | 0.63 V vs. SCE | ca.0~40 μM | 1.5 nM | [5] |
| C/NiP/Nafion® b | 0.64 V vs. SCE | 0~300 μM | 10 μM | [6] |
| C/Nafion® | 0.86 V vs. Ag/AgCl/sat. CuCle | 0~1 μM | N. A. | [2] |
| C/Nafion® (D= 0.1 μm) | 0.86 V vs. Ag/AgCl/sat. CuCle | 50 nM~1 mM | 3 nM | [7] |
| C/Nafion® (D= 7 μm) | 0.86 V vs. Ag/AgCl/sat. CuCle | 10 nM~5 μM | 5 nM | [8] |
| C/Nafion® | 0.86 V vs. Ag/AgC/sat. CuCle | 0~0.4 μM | N. A. | [9] |
| C/silicon | 0.86 V vs. Ag/AgCl/sat. CuCle | 0~100 μM | 0.3 nM | [10] |
| M(salen)/Nafion® c | DPAg | ca. 19.6 nM~2.8 μM | ca. 10~20 nM | [11] |
| Sensing | Operating | Output | Ref. |
| Electrodes | Condition | Signal | |
| Noble metal/membranea | 0.96~1.03 V vs. SHE | Current | [12] |
| Au/Teflon® | 1.5 V vs. SHE | Current | [13] |
| Au/hydrophobic plasticb | 1.6 V vs. SHE | Current | [14,15] |
| Au/C | 0.9~1.5 V vs. SHE | Current | [16] |
| Au/Nafion® | 0.7 V vs. MSEd | Current | [17] |
| Pt/Nafion® | 0.7 V vs. MSEd | Current | [17] |
| Pt/Nafion® | 1.15 V vs. sat. Ag/AgCl | Current | This work |
| Pt/CdCr2O4 | 0.1 V vs. Pt (500 °C) | Current | [18] |
| Pt/Zr2O | -0.55 V vs. C (e.g. 650 °C) | Current | [19] |
| NaNO2/Au | 150 °C | Potential | [20] |
| Cr2O3/Nasicon | 250 °C | Potential | [21] |
| Au/pyrochlore-type oxidec | 400 °C | Potential | [22] |
| Au/Pb2Ru1.9V0.1O7-z | 400 °C | Potential | [23] |
| Pt/CdMn2O4 | 600 °C | Potential | [24] |
| Pt/Zr2O | 300~400 °C | Potential | [25] |
Experimental
Sensing electrode characterization and experimental setup
Pretreating or cleansing Pt/Nafion® electrode by cyclic voltammetric (CV) activation
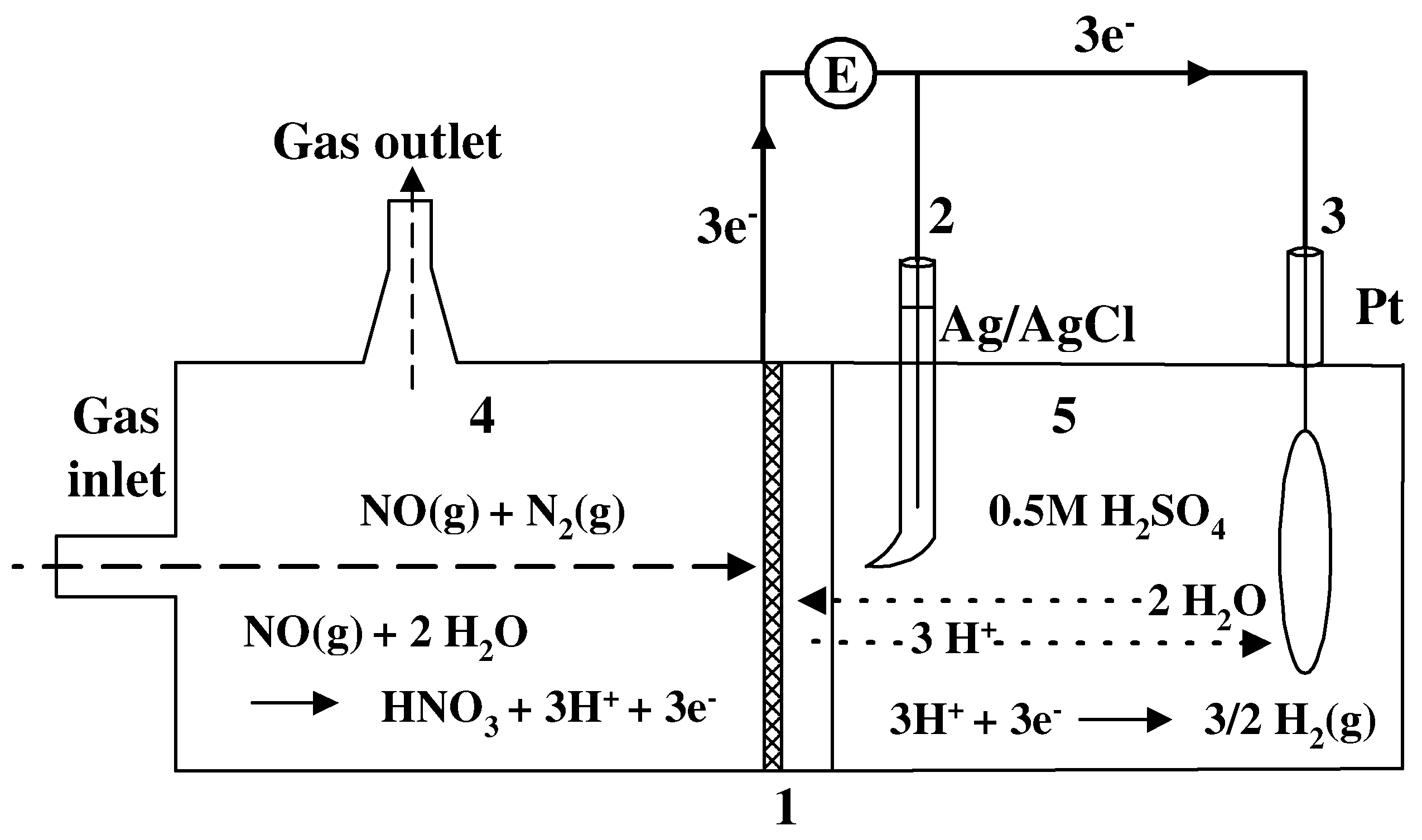
Results and Discussion
Real surface area of a fresh Pt/Nafion® electrode
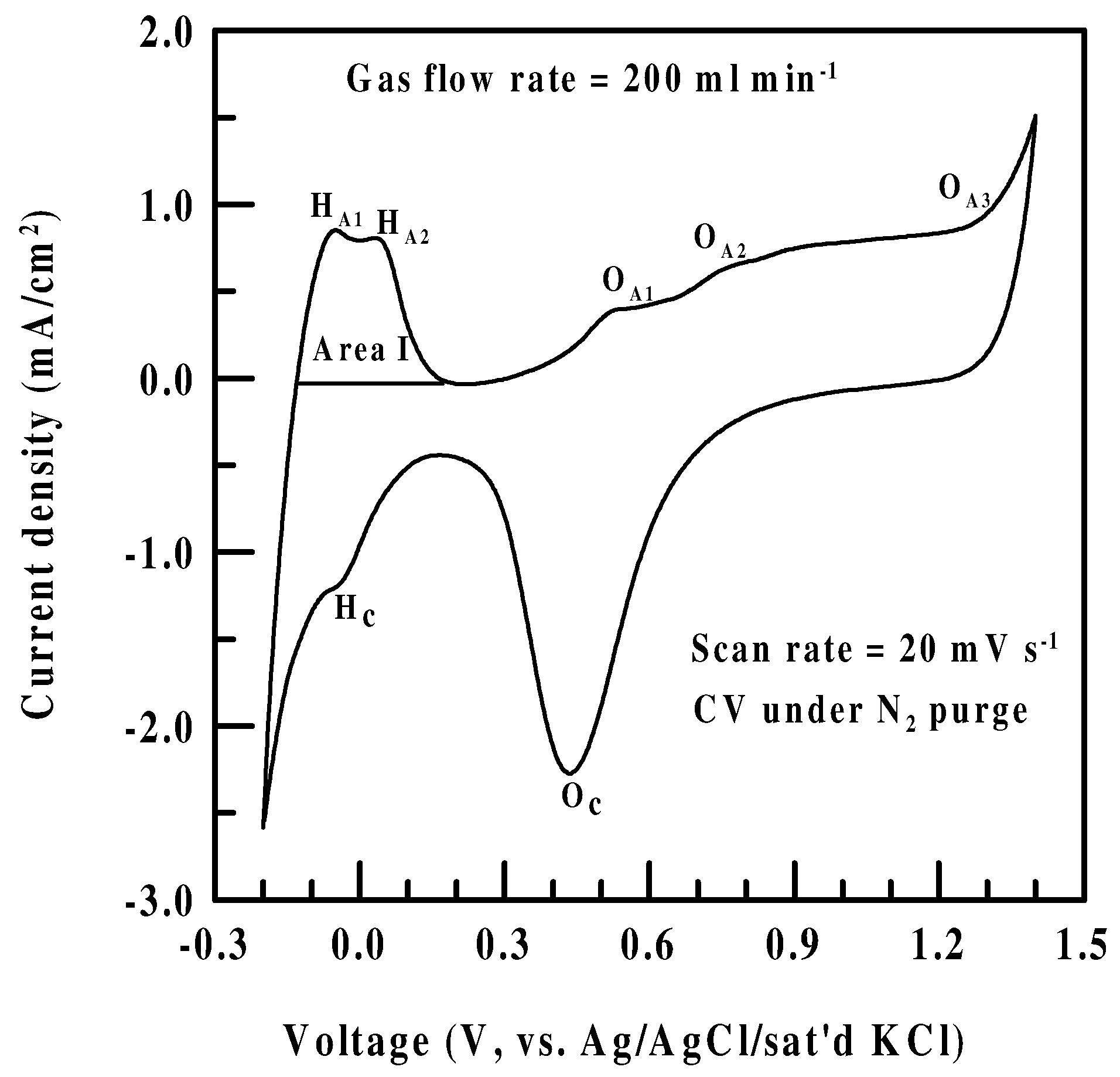
CV of a Pt/Nafion® electrode in N2 and NO
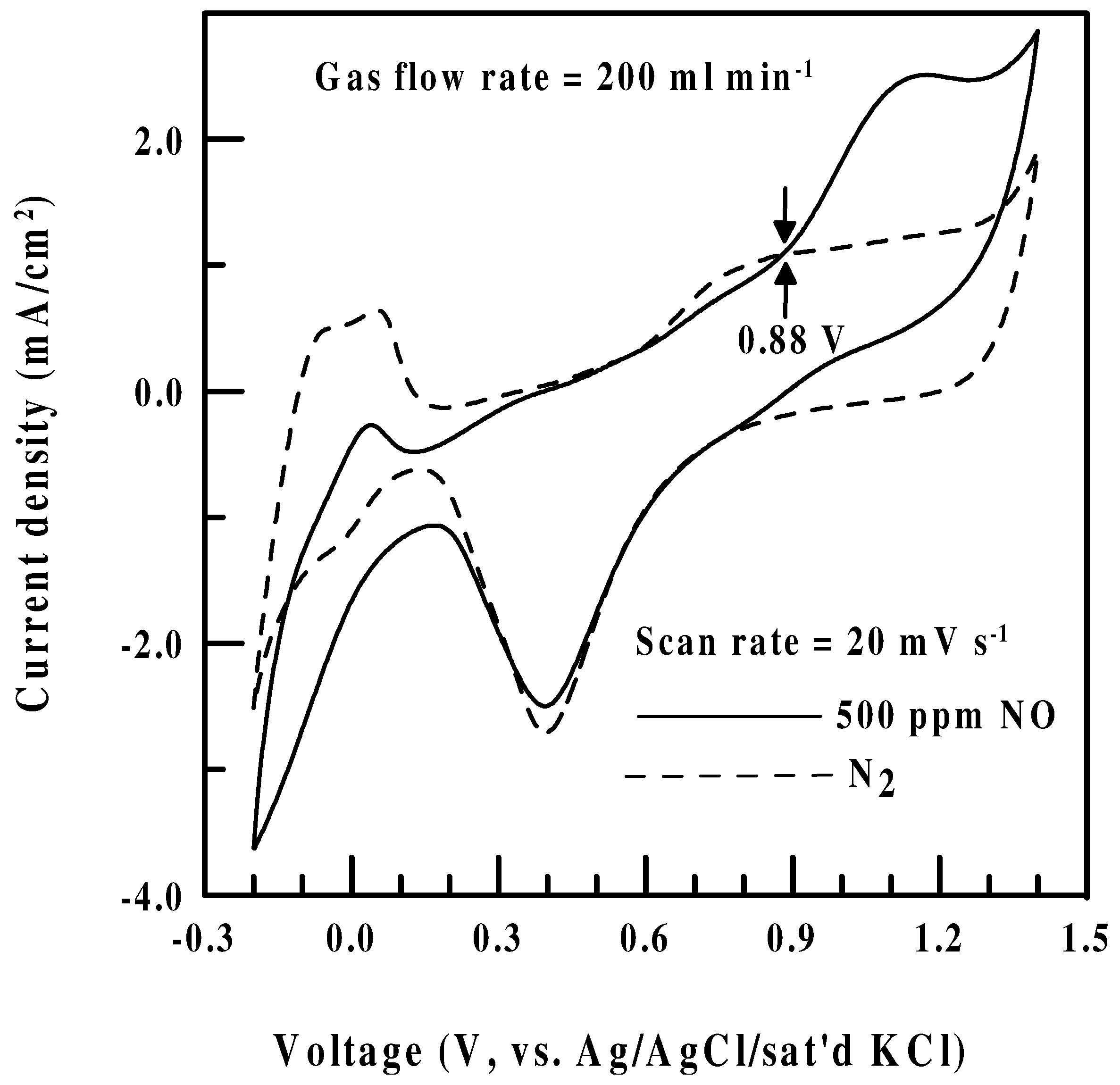
Effect of sensing potential-Polarization data
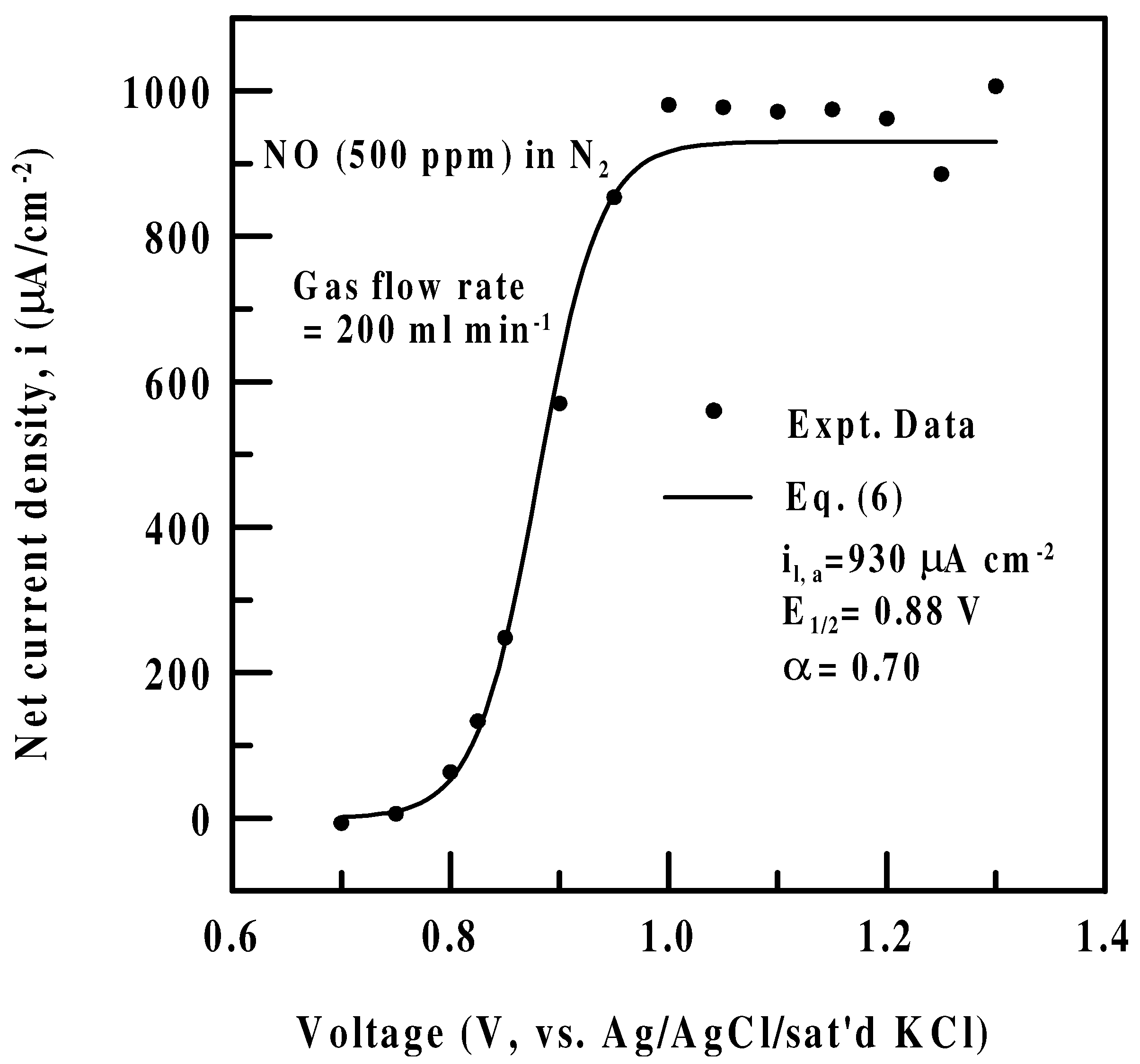
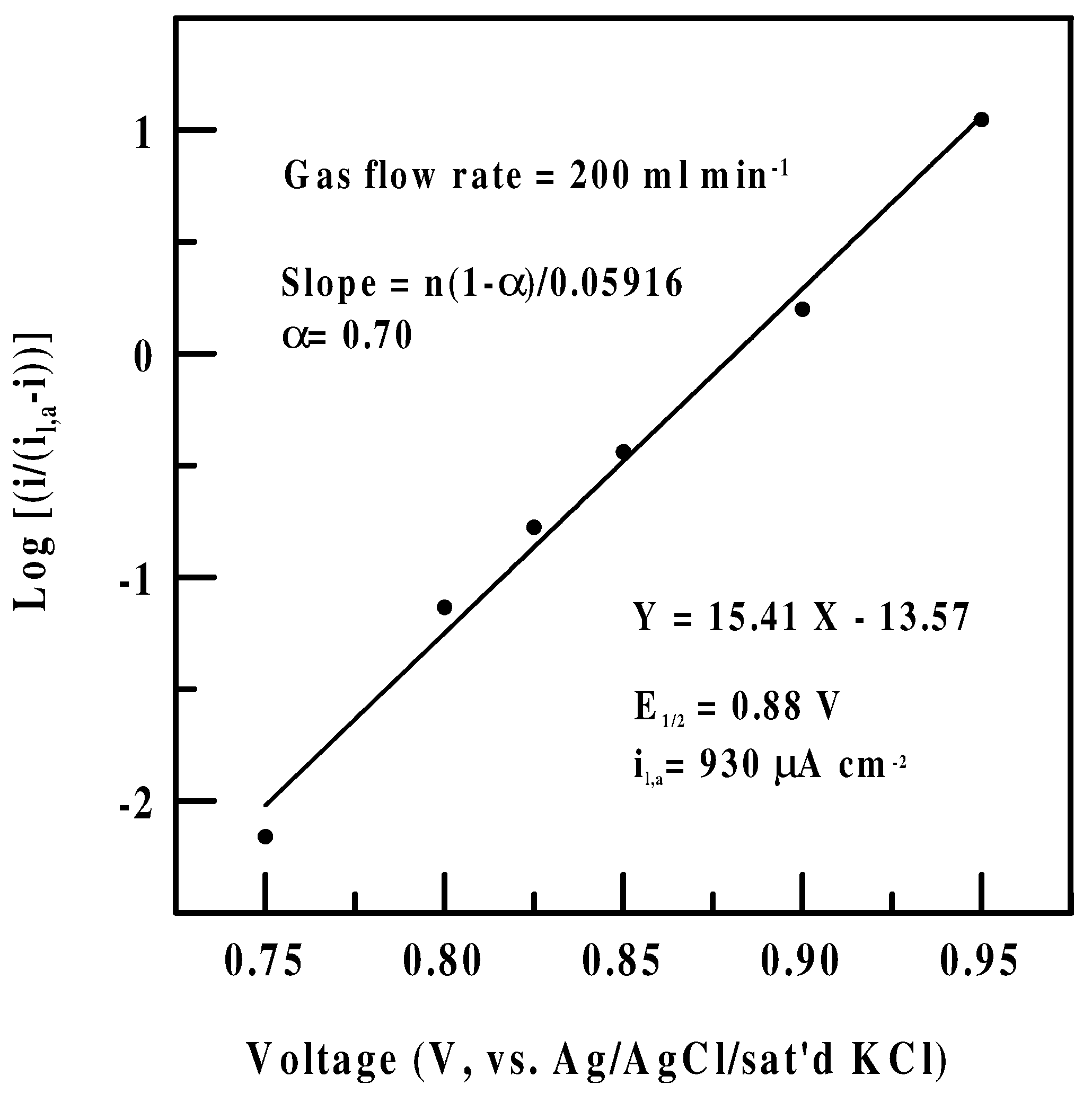
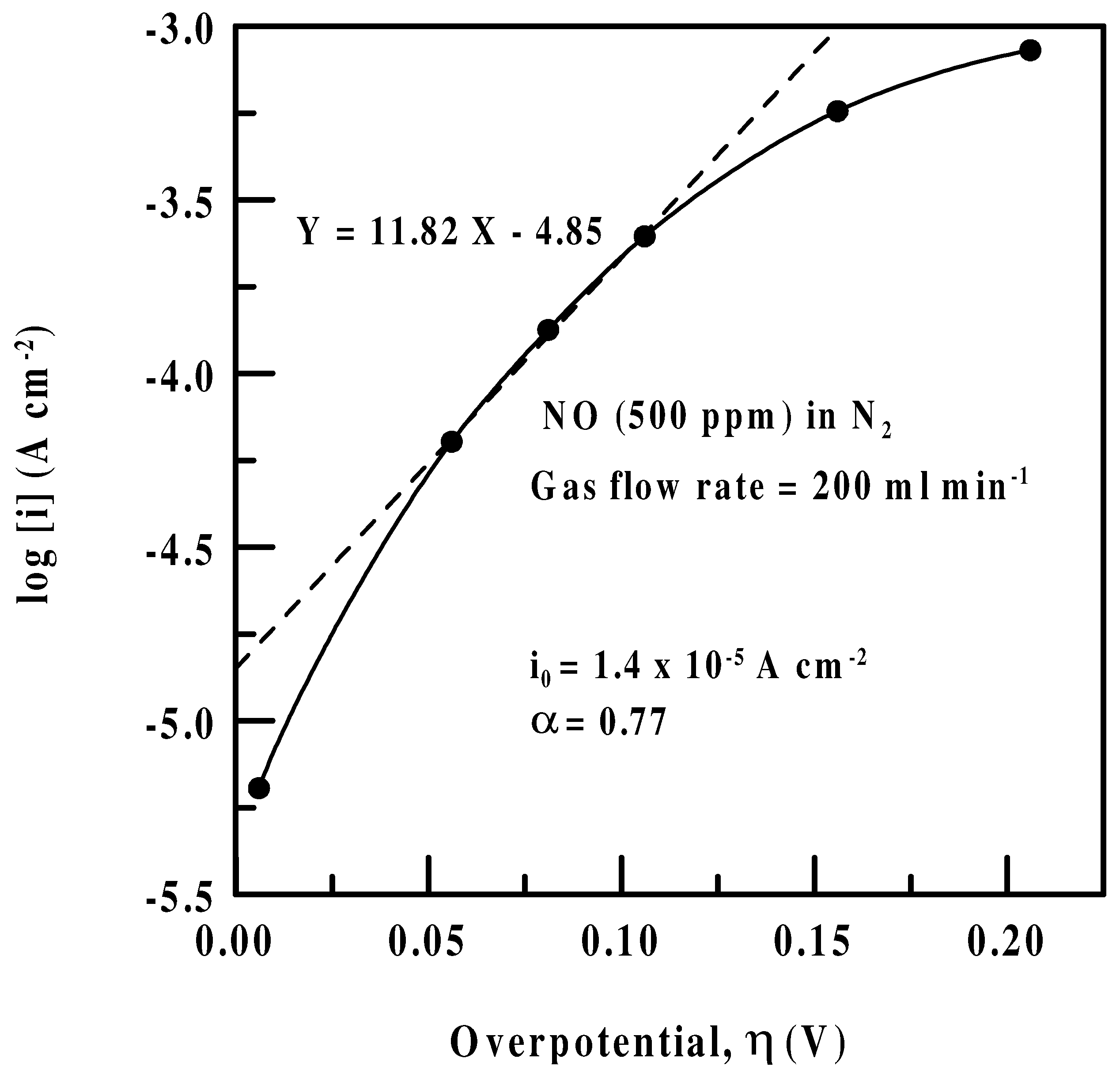
Tafel kinetics
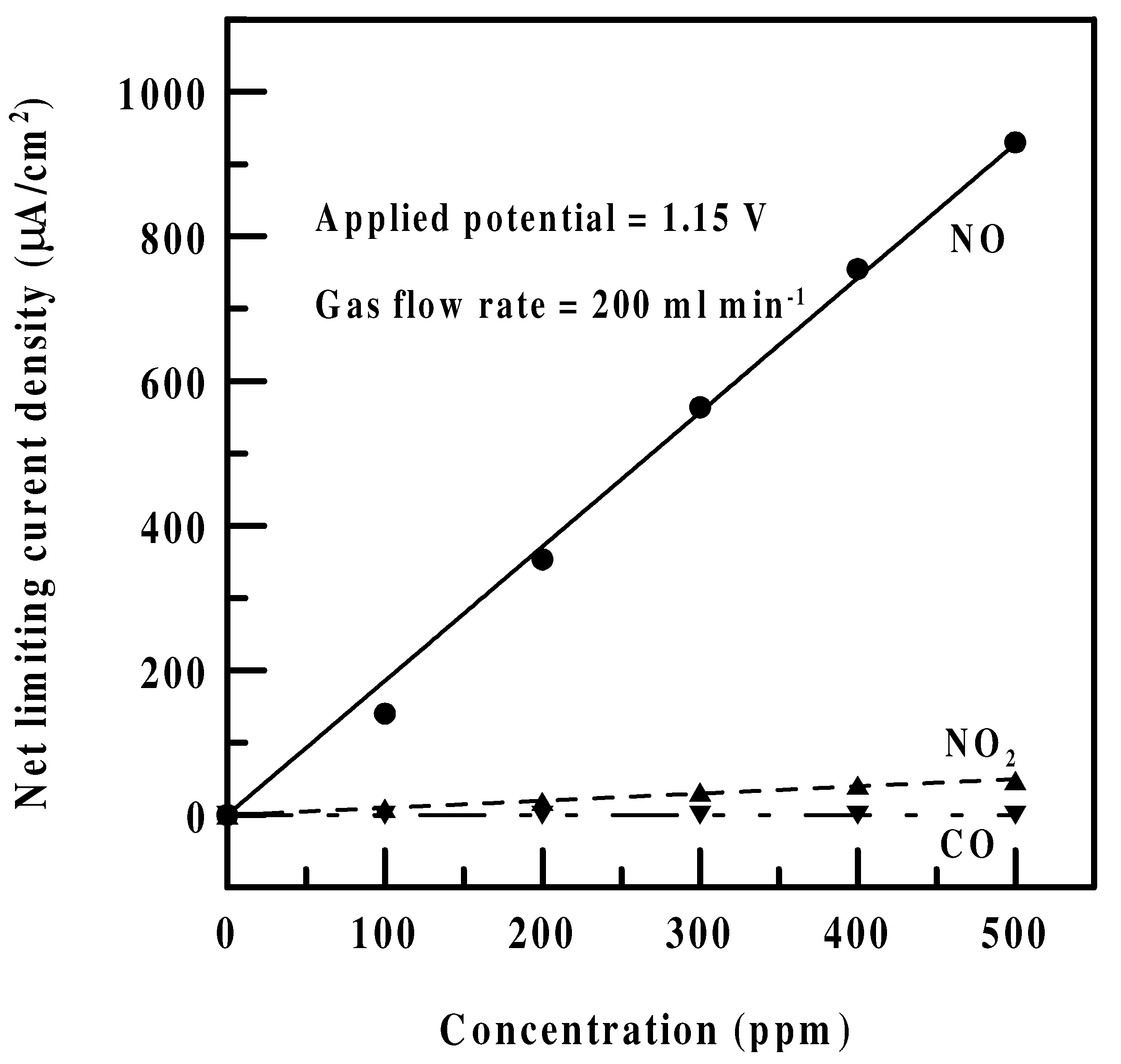
Interferences and sensitivities
Conclusion
Acknowledgement
References
- Pariente, F.; Alonso, J. L.; Abruna, H. D. J. Electroanal. Chem. 1994, 379, 191–197.
- Zhang, X.; Cardosa, L.; Broderick, M.; Fein, H.; Davies, I.R. Eelctroanalysis 2000, 12, 425–428.
- Ichimori, K.; Ishida, H.; Fukahori, M.; Nakazawa, H.; Murakami, E. Rev. Sci. Instrum. 1994, 65, 2714–2718.
- Pallini, M.; Curulli, A.; Amine, A.; Palleschi, G. Electroanalysis 1998, 10, 1010–1016.
- Lantoine, F.; Trevin, S.; Bedioui, F.; Devynck, J. J. Electroanal. Chem. 1995, 392, 85–89.
- Malinski, T.; Taha, Z. Nature 1992, 358, 676–679. [PubMed]
- Zhang, X.; Kislyak, Y.; Cardosa, L.; Broderick, M.; Fein, H. Free Radic. Biol. Med. 2000, 29, S78.
- Zhang, X.; Cardosa, L.; Broderick, M.; Fein, H.; Lin, J. Electroanalysis 2000, 12, 1113–1117.
- Zhang, X.; Broderick, M. Mod. Asp. Immunobiol. 2000, 1, 160–165.
- Zhang, X.; Lin, J.; Cardosa, L.; Broderick, M.; Darley-Usmar, V. Electroanalysis 2002, 14, 697–703.
- Mao, L.; Yamamoto, K.; Zhou, W.; Jin, L. Electroanalysis 2000, 12, 72–77.
- Chand, R. U.S. Patent No. 3,622,487, 1971.
- Sedlak, J. M.; Blurton, K. F. J. Electrochem. Soc. 1976, 123, 1476–1478.
- Blurton, K.F.; Sedlak, J. M. U.S. Patent No. 4,001,103, 1977.
- Blurton, K. F.; Sedlak, J. M. U.S. Patent No. 4,052,268, 1977.
- Blurton, K. F.; Sedlak, J. M. U.S. Patent No. 4,042,464, 1977.
- Jacquinot, P.; Hodgson, A. W. E.; Hauser, P. C. Anal. Chim. Acta 2001, 443, 53–61.
- Miura, N.; Lu, G.; Yamazeo, N. Sens. Actuators B 1998, 52, 169–178.
- Somov, S.; Reinhardt, G.; Guth, U.; Gopel, W. Sens. Actuators B 1996, 35–36, 409–418.
- Yao, S.; Shimizu, Y.; Miura, N.; Yamazoe, N. Chem. Letter 1992, 587–590.
- Shimizu, Y.; Maeda, K. Chem. Letter 1996, 117–118.
- Shimizu, Y.; Maeda, K. Sens. Actuators B 1998, 52, 84–89.
- Shimizu, Y.; Nishi, H.; Suzuki, H.; Maeda, K. Sens. Actuators B 2000, 65, 141–143.
- Morio, N.; Kurosawa, H.; Hasei, M.; Lu, G.; Yamazoe, N. Solid State Ionics 1996, 86–88, 1069–1073.
- Takashi, O.; Masaharu, H.; Akira, K. EP Patent No. 1,167,957, 2002.
- Ho, K. C.; Hung, W. T. Sens. Actuators B 2001, 79, 11–16.
- Bilmes, S. A.; De Tacconi, N. R.; Arvia, A. J. J. Electroanal. Chem. 1984, 164, 129–143.
- Parthasarathy, A.; Srinivasan, S.; Appleby, A. J.; Martin, C. R. J. Electrochem. Soc. 1992, 139, 2530–2537.
- Parthasarathy, A.; Dave, B.; Srinivasan, S.; Appleby, A. J.; Martin, C. R. J. Electrochem. Soc. 1992, 139, 1634–1641.
- Basura, I.; Beattie, P. D.; Holocoft, S. J. Electroanal. Chem. 1998, 458, 1–5.
- Will, F. G. J. Electrochem. Soc. 1965, 112, 451–455.
- Biegler, T.; Rand, D. A. J.; Woods, R. J. Electroanal. Chem. 1971, 29, 269–277.
- Woods, R. J. Electroanal. Chem. Interfacial Electrochem. 1974, 49, 217–226.
- Angerstein-Kozlowska, H.; Conway, B. E.; Sharp, W. B. A. J. Electroanal. Chem. Interfacial Electrochem. 1973, 43, 9–36.
- Opekar, F.; Stulik, K. Anal. Chim. Acta 1999, 385, 151–162.
- Bard, A. J.; Faulkner, L. R. Electrochemical Methods, 2nd Ed. ed; John Wiley & Sons: New York, 2001; Chapter 1 and 3. [Google Scholar]
- Sample Availability: Available from the authors.
© 2003 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Ho, K.-C.; Hung, W.-T.; Yang, J.-C. On the Electrooxidation and Amperometric Detection of NO Gas at the Pt/Nafion® Electrode. Sensors 2003, 3, 290-303. https://doi.org/10.3390/s30800290
Ho K-C, Hung W-T, Yang J-C. On the Electrooxidation and Amperometric Detection of NO Gas at the Pt/Nafion® Electrode. Sensors. 2003; 3(8):290-303. https://doi.org/10.3390/s30800290
Chicago/Turabian StyleHo, Kuo-Chuan, Wen-Tung Hung, and Jin-Cherng Yang. 2003. "On the Electrooxidation and Amperometric Detection of NO Gas at the Pt/Nafion® Electrode" Sensors 3, no. 8: 290-303. https://doi.org/10.3390/s30800290
APA StyleHo, K.-C., Hung, W.-T., & Yang, J.-C. (2003). On the Electrooxidation and Amperometric Detection of NO Gas at the Pt/Nafion® Electrode. Sensors, 3(8), 290-303. https://doi.org/10.3390/s30800290




