Cholinergic Phenotypes of Acetyl-CoA with ATP-Citrate Lyase Link
Abstract
1. Introduction
2. Brain Cholinergic Neurons
2.1. Regional and Developmental Patterns of Cholinergic Neurons
2.2. Postnatal Development of Cholinergic Neurons
2.3. Intraneuronal Distribution of ChAT
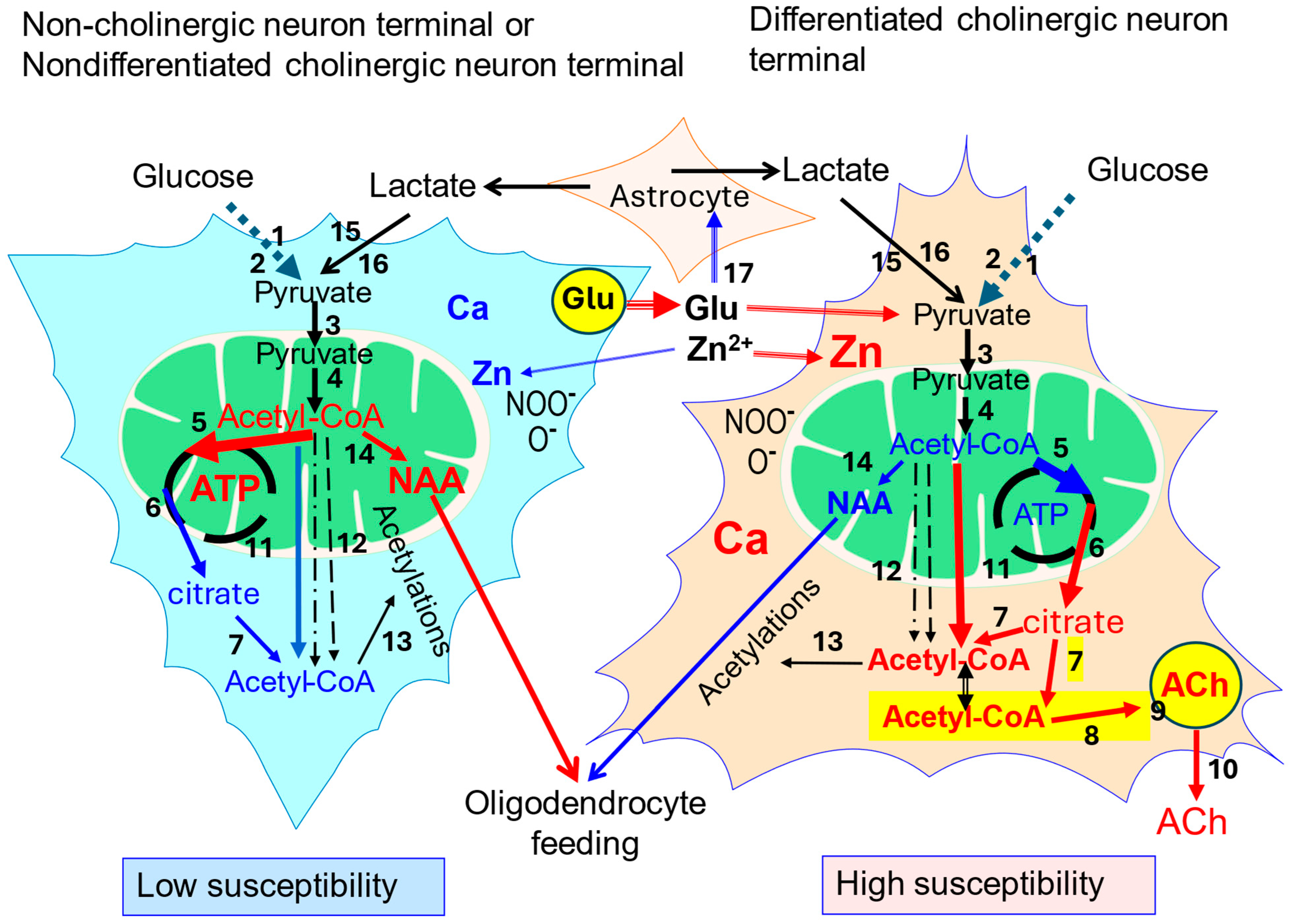
3. Acetyl-CoA Metabolism in Cholinergic Neurons
3.1. Synthesis of Neuronal Acetyl-CoA
3.2. Extramitochondrial Neuronal Acetyl-CoA
3.3. PDHC and Cholinergic Neurons
4. Cholinergic Neurons and ATP-Citrate Lyase
5. Motor Cholinergic Neurons
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Von Bartheld, C.S.; Bahney, J.; Herculano-Houzel, S. The search for true numbers of neurons and glial cells in the human brain: A review of 150 years of cell counting. J. Comp. Neurol. 2016, 524, 3865–3895. [Google Scholar] [CrossRef]
- Druga, R. Neocortical inhibitory system. Folia Biol. 2009, 55, 201–217. [Google Scholar] [CrossRef]
- Edwards, F. Anatomy and electrophysiology of fast central synapses lead to structural model for long-term potentiation. Physiol. Rev. 1995, 75, 759–782. [Google Scholar] [CrossRef]
- Jolivet, R.; Magistretti, P.J.; Weber, B. Deciphering neuron-glia compartmentalization in cortical energy metabolism. Front. Neuroenergetics 2009, 1, 4. [Google Scholar] [CrossRef]
- Jha, M.K.; Lee, I.K.; Suk, K. Metabolic reprogramming by the pyruvate dehydrogenase kinase–lactic acid axis: Linking metabolism and diverse neuropathophysiologies. Neurosci. Biobehav. Rev. 2016, 68, 1–19. [Google Scholar] [CrossRef]
- Jankowska-Kulawy, A.; Klimaszewska-Łata, J.; Gul-Hinc, S.; Ronowska, A.; Szutowicz, A. Metabolic and cellular compartments of acetyl-CoA in the healthy and diseased brain. Int. J. Mol. Sci. 2022, 23, 10073. [Google Scholar] [CrossRef]
- Sarter, M.; Lustig, C. Forebrain cholinergic signalling: Wired and phasic, not tonic, and causing behavior. J. Neurosci. 2022, 40, 712–719. [Google Scholar] [CrossRef]
- Klein, J. The central cholinergic synapses: A primer. Int. J. Mol. Sci. 2025, 26, 9670. [Google Scholar] [CrossRef]
- Gilmor, M.L.; Nash, N.R.; Roghani, A.; Edwards, R.H.; Yi, H.; Hersch, S.M.; Levey, A.I. Expression of the putative vesicular acetylcholine transporter in rat brain and localization in cholinergic synaptic vesicles. J. Neurosci. 1996, 16, 2179–2190. [Google Scholar] [CrossRef]
- Phillis, J.W. Acetylcholine release from the central nervous system: A 50-year retrospective. Crit. Rev. Neurobiol. 2005, 17, 161–217. [Google Scholar] [CrossRef]
- Ahmed, N.Y.; Knowles, R.; Dehorter, N. New Insights into cholinergic neuron diversity. Fron. Mol. Neurosci. 2019, 12, 204. [Google Scholar]
- Szutowicz, A.; Stepień, M.; Bielarczyk, H.; Kabata, J.; Lysiak, W. ATP citrate lyase in cholinergic nerve endings. Neurochem. Res. 1982, 7, 799–810. [Google Scholar] [CrossRef]
- Van der Zee, E.A.; Keijser, J.N. Localization of pre-and postsynaptic cholinergic markers in rodent forebrain: A brief history and comparison of rat and mouse. Behav. Brain Res. 2011, 221, 356–366. [Google Scholar]
- Carrasco, A.; Oorschot, D.E.; Barzaghi, P.; Wickens, J.R. There dimensional spacial analyses of cholinergic neuronal distributions across the mouse septum, nucleus basalis, globus pallidus, nucleus accumbens, and caudate putamen. Neuroinformatics 2022, 20, 1121–1136. [Google Scholar] [CrossRef]
- Szutowicz, A.; Łysiak, W. Regional and subcellular distribution of ATP-citrate lyase and other enzymes of acetyl-CoA metabolism in rat brain. J. Neurochem. 1980, 35, 775–785. [Google Scholar] [CrossRef]
- Szutowicz, A. Regional and developmental correlations between choline acetyltransferase and ATP citrate oxaloacetate lyase in rat brain. In Biological Aspects of Learning, Memory Formation and Ontogeny of the CNS; Mathies, H., Krug, M., Popov, N., Eds.; Akademie Verlag: Berlin, Germany, 1978; pp. 489–499. [Google Scholar]
- Boskovic, Z.; Meier, S.; Wang, Y.; Milne, M.R.; Onraet, T.; Tedoldi, A.; Coulson, E.J. Regulation of cholinergic basal forebrain development, connectivity, and function by neurotrophin receptors. Neuronal Signal. 2019, 3, NS20180066. [Google Scholar] [CrossRef]
- Holler, T.; Berse, B.; Cermak, J.M.; Diebler, M.F.; Blusztajn, J.K. Differences in the developmental expression of the vesicular acetylcholine transporter and choline acetyltransferase in the rat brain. Neurosci. Lett. 1996, 12, 107–110. [Google Scholar] [CrossRef]
- Szutowicz, A.; Kabata, J.; Bielarczyk, H. The contribution of citrate to the synthesis of acetyl units in synaptosomes of developing rat brain. J. Neurochem. 1983, 38, 1196–1204. [Google Scholar] [CrossRef]
- Bruel-Jungerman, E.; Lucassen, P.J.; Francis, F. Cholinergic influences on cortical development and adult neurogenesis. Behav. Brain Res. 2011, 221, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Mallet, J.; Houhou, L.; Pajak, F.; Oda, Y.; Cervini, R.; Bejanin, S.; Berrard, S. The cholinergic locus: ChAT and VAChT genes. J. Physiol. Paris 1998, 92, 145–147. [Google Scholar]
- Ananth, M.R.; Rajebhosale, P.; Kim, R.; Talmage, D.A.; Role, L.W. Basal forebrain cholinergic signalling: Development, connectivity and roles in cognition. Nat. Rev. Neurosci. 2023, 24, 233–251. [Google Scholar] [CrossRef]
- Snyder, P.J.; Maruff, P.T. Cognition and modulation of the cholinergic system. Handb. Clin. Neurol. 2025, 211, 55–61. [Google Scholar] [CrossRef]
- Szutowicz, A.; Frazier, W.A.; Bradshaw, R.A. Subcellular localization of nerve growth factor receptor. J. Biol. Chem. 1976, 251, 1524–1528. [Google Scholar] [CrossRef] [PubMed]
- Barrett, G.L.; Naim, T.; Trieu, J.; Huang, M. In vivo knockdown of basal forebrain p75 neurotrophin receptor stimulates choline acetyltransferase activity in the mature hippocampus. J. Neurosci. Res. 2016, 94, 384–400. [Google Scholar] [CrossRef] [PubMed]
- Szutowicz, A.; Madziar, B.; Pawełczyk, T.; Tomaszewicz, M.; Bielarczyk, H. Effects of NGF on acetylcholine, acetyl-CoA metabolism, and viability of differentiated and non-differentiated cholinergic neuroblastoma cells. J. Neurochem. 2004, 90, 952–961. [Google Scholar] [CrossRef]
- Latina, V.; Caioli, S.; Zona, C.; Ciotti, M.T.; Amadoro, G.; Calissano, P. Impaired NGF/TrkA Signaling Causes Early AD-Linked Presynaptic Dysfunction in Cholinergic Primary Neurons. Front. Cell Neurosci. 2017, 11, 15. [Google Scholar] [CrossRef]
- Zhang, Y.; Hong, Y.; Bounhar, Y.; Blacker, M.; Roucou, X.; Tounekti, O.; Vereker, E.; Bowers, W.J.; Federoff, H.J.; Goodyer, C.G.; et al. p75 neurotrophin receptor protects primary cultures of human neurons against extracellular amyloid beta peptide cytotoxicity. J. Neurosci. 2003, 23, 7385–7394. [Google Scholar] [CrossRef]
- Chao, M.V. Neurotrophins and their receptors: Convergence point for many signaling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef]
- Pathak, A.; Carter, B.D. Retrograde apoptotic signaling by the p75 neurotrophin receptor. Neuronal Signal. 2017, 1, NS20160007. [Google Scholar] [CrossRef] [PubMed]
- Kewitz, H.; Rost, K.L.; Pleul, O.; Handke, A. Dose-related effects of nerve growth factor (NGF) on choline acetyltransferase (ChAT), acetylcholine (ACh) content and ACh turnover in the brain of newborn rats. Neurochem. Int. 1990, 17, 239–244. [Google Scholar] [CrossRef]
- Fahnestock, M.; Shekari, A. ProNGF and Neurodegeneration in Alzheimer’s Disease. Front. Neurosci. 2019, 13, 129. [Google Scholar] [CrossRef]
- Whittaker, V.P. The application of subcellular fractionation techniques to the study of brain function. Progr Biophys. Mol. Biol. 1965, 15, 39–96. [Google Scholar] [CrossRef]
- Bina, K.G.; Rusak, B.; Semba, K. Localization of cholinergic neurons in the forebrain and brainstem that project to the suprachiasmatic nucleus of the hypothalamus in rat. J. Comp. Neurol. 1993, 335, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Simpson, I.A.; Carruthers, A.; Vannucci, J. Supply and demand in cerebral energy metabolism: The role of nutrient transporters. J. Cereb. Blood Flow. Metab. 2007, 27, 1766–1791. [Google Scholar] [CrossRef]
- Nithya, U.; Theijeswini, R.C.; Karthick, R.R.; Gayathri, M. Chapter One—Glucose transporters and their homeostasic function in varius organs. Vitam. Horm. 2025, 128, 1–47. [Google Scholar]
- Peréz-Escuredo, J.; Van Hée, V.F.; Sboarina, M.; Falces, J.; Payen, V.L.; Pellerin, L.; Sonveaux, P. Monocarboxylate transporters in the brain and in cancer. Biochim. Biophys. Acta 2016, 1863, 2481–2486. [Google Scholar] [CrossRef]
- Roosterman, D.; Cottrell, G.S. Astrocytes and neurons communicate via a monocarboxylic acid shuttle. Neurosci. 2020, 7, 94–106. [Google Scholar] [CrossRef]
- Bolaños, J.P.; Magistretti, P.J. The neuron-astrocyte metabolic unit as a cornerstone of brain energy metabolism in health and disease. Nat. Metab. 2025, 7, 2414–2423. [Google Scholar] [CrossRef]
- Magistretti, P.J.; Allaman, I. A cellular perspective on brain energy metabolism and functional imaging. Neuron 2015, 86, 883–901. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, Y.; Srere, P.A. Kinetic studies of citrate synthase from rat kidney and rat brain. J. Biol. Chem. 1973, 248, 8022–8030. [Google Scholar] [CrossRef] [PubMed]
- Guynn, R.W.; Gelberg, H.J.; Veech, R.L. Equilibrium constants of the malate dehydrogenase, citrate synthase, citrate lyase, and acetyl coenzyme A hydrolysis reactions under physiological conditions. J. Biol. Chem. 1973, 248, 6957–6965. [Google Scholar] [CrossRef]
- Dominicis, A.; Giovane, A.D.; Torreggiani, R.; Recchia, A.D.; Ciccarone, F.; Ciriolo, M.R.; Ragnini-Wilson, A. N-acetylaspartate drivers oligodendroglial differentiation via histone deacetylase activation. Cells 2023, 12, 1861. [Google Scholar] [CrossRef] [PubMed]
- Baslow, M.H.; Guilfoyle, D.N. Using proton magnetic resonance imaging and spectroscopy to understand brain “activation”. Brain Lang. 2007, 102, 153–154. [Google Scholar] [CrossRef]
- Gao, S.; X, H.; Luo, H.; Tang, X.; Zeng, Y. Histone acetylation: A key regulator of inflammatory responses. Life Sci. 2025, 380, 123936. [Google Scholar] [CrossRef]
- Ronowska, A.; Gul-Hinc, S.; Michno, A.; Bizon-Zygmańska, D.; Zyśk, M.; Bielarczyk, H.; Szutowicz, A.; Gapys, B.; Jankowska-Kulawy, A. Aggravated effects of coexisting marginal thiamine deficits and zinc excess on SN56 neuronal cells. Nutr. Neurosci. 2021, 6, 432–442. [Google Scholar] [CrossRef]
- Lefresne, P.; Guyenet, P.; Glowinski, J. Acetylcholine synthesis from (214C)pyruvate in rat striatal slices. J. Neurochem. 1973, 20, 1083–1097. [Google Scholar] [CrossRef]
- Li, B.; Xia, M.; Zorec, R.; Parpura, V.; Alexei Verkhratsky, A. Astrocytes in heavy metal neurotoxicity and neurodegeneration. Brain Res. 2021, 1752, 147234. [Google Scholar] [CrossRef] [PubMed]
- Flores, A.; Moyano, P.; Sola, E.; García, J.M.; García, J.; Frejo, M.T.; Guerra-Menéndez, L.; Labajo, E.; Lobo, I.; Abascal, L.; et al. Bisphenol-A Neurotoxic Effects on Basal Forebrain Cholinergic Neurons In Vitro and In Vivo. Biology 2023, 12, 782. [Google Scholar] [CrossRef]
- Mudyanselage, A.W.; Wijamunige, B.C.; Kocon, A.; Carter, W.G. Differentiated neurons are more vulnerable to organophosphate and carbamate neurotoxicity than undifferentiated neurons due to the induction of redox stress and accumulate oxidatively-damaged proteins. Brain Sci. 2023, 13, 728. [Google Scholar] [CrossRef]
- Ronowska, A.; Szutowicz, A.; Bielarczyk, H.; Gul-Hinc, S.; Klimaszewska-Łata, J.; Dyś, A.; Zyśk, M.; Jankowska-Kulawy, A. The Regulatory Effects of Acetyl-CoA Distribution in the Healthy and Diseased Brain. Front. Cell Neurosci. 2018, 12, 169. [Google Scholar] [CrossRef] [PubMed]
- Nakai, N.; Obayashi, M.; Nagasaki, M.; Sato, Y.; Fujitsuka, N.; Yoshimura, A.; Miyazaki, Y.; Sugiyama, S.; Shimomura, Y. The abundance of mRNAs for pyruvate dehydrogenase kinase isoenzymes in brain regions of young and aged rats. Life Sci. 2000, 68, 497–503. [Google Scholar] [CrossRef]
- Zhang, C.; Rissman, R.A.; Feng, J. Characterization of ATP alternations in an Alzheimer’s transgenic mouse model. J. Alzheimers’s Dis. 2015, 44, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, R.F.; Giguère, J.F. Pyruvate dehydrogenase activity in regions of the rat brain during postnatal development. J. Neurochem. 1984, 43, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Bubber, P.; Ke, Z.; Gibson, G.E. Tricarboxylic acid cycle enzymes following thiamine deficiency. Neurochem. Int. 2004, 45, 1021–1028. [Google Scholar] [CrossRef]
- Bubber, P.; Haroutunian, V.; Fisch, G.; Blass, J.P.; Gibson, G.E. Mitochondrial abnormalities in Alzheimer brain: Mechanistic implications. Ann. Neurol. 2005, 57, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, R.F. Hepatic encephalopathy in alcoholic cirrhosis. In Handbook of Clinical Neurology Alcohol and the Nervous System; Sullivan, E.V., Pfefferbaum, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 125, pp. 589–601. [Google Scholar]
- Bielarczyk, H.; Jankowska-Kulawy, A.; Höfling, C.; Ronowska, A.; Gul-Hinc, S.; Rößner, S.; Schliebs, R.; Pawełczyk, T.; Szutowicz, A. AβPP-transgenic 2576 mice mimic cell type-specific aspects of acetyl-CoA-linked metabolic deficits in Alzheimer’s disease. J. Alzheimer’s Dis. 2015, 48, 1083–1094. [Google Scholar] [CrossRef]
- Belkacemi, A.; Ramasamy, C. Time sequence of oxidative stress in the brain from transgenic mouse model of Alzheimer’s disease related to the amyloid- β cascade. Free Radic. Biol. Med. 2012, 52, 593–600. [Google Scholar] [CrossRef]
- Du, H.; Guo, L.; Yan, S.; Sosunov, A.A.; McKhann, G.M.; ShiDu Yan, S. Early deficits in synaptic mitochondria in an Alzheimer’s disease mouse model. Proc. Natl. Acad. Sci. USA 2010, 107, 18670–18675. [Google Scholar] [CrossRef]
- Esquerda-Canals, G.; Montoliu- Gaya, L.; Güell-Bosch, J.; Villegas, S. Mouse models of Alzheimer’s disease. J. Alzheimer’s Dis. 2017, 57, 1171–1183. [Google Scholar] [CrossRef]
- Chhimpa, N.; Singh, N.; Puri, N.; Kayath, H.P. The Novel Role of Mitochondrial Citrate Synthase and Citrate in the Pathophysiology of Alzheimer’s Disease. J. Alzheimer’s Dis. 2023, 94, 453–472. [Google Scholar] [CrossRef]
- Mukhejee, A.G.; Mishra, S.; Gopalakrishnan, A.V.; Kannampuzha, S.; Murali, R.; Wanjari, U.R.; Stany, B.; Vellingiri, B.; Madhyastha, H.; Kanagavel, D.; et al. Unraveling the mystery of citrate transporters in Alzheimer’s disease: An update review. Ageing Res. Rev. 2025, 107, 102726. [Google Scholar] [CrossRef]
- Szutowicz, A.; Łysiak, W.; Angielski, S. The effect of (-) hydroxycitrate on pyruvate metabolism in rat brain synaptosomes. J. Neurochem. 1977, 29, 375–378. [Google Scholar] [PubMed]
- Rícný, J.; Tuček, S. Acetylcoenzyme A and acetylcholine in slices of rat caudate nuclei incubated with (-)-hydroxycitrate, citrate, and EGTA. J. Neurochem. 1982, 39, 668–673. [Google Scholar] [CrossRef]
- Burton, M.D.; Nouri, M.; Kazemi, H. Acetylcholine and central respiratory control: Perturbations of acetylcholine synthesis in the isolated brainstem of the neonatal rat. Brain Res. 1995, 670, 39–47. [Google Scholar] [CrossRef]
- Szutowicz, A.; Tomaszewicz, M.; Jankowska, A.; Kisielevski, Y. Acetylcholine synthesis in nerve terminals of diabetic rats. NeuroReport 1994, 18, 2421–2424. [Google Scholar] [CrossRef]
- Fernandez-Fuente, G.; Overmyer, K.A.; Lawton, A.J.; Kasza, I.; Shapiro, S.L.; Gallego-Muñoz, P.; Coon, J.J.; Denu, J.M.; Alexander, C.M.; Puglielli, L. The citrate transporters SLC13A5 and SLC25A1 elicit different metabolic responses and phenotypes in the mouse. Commun. Biol. 2023, 6, 926. [Google Scholar] [CrossRef]
- Feng, M.-H.; Li, Z.-X.; Wang, Q.; Manyande, A.; Li, Y.-J.; Li, S.-Y.; Xu, W.; Xiang, H.-B. Neurochemical alterations of different cerebral regions in rats with myocardial ischemia-reperfusion injury based on proton nuclear magnetic spectroscopy analysis. Aging 2021, 13, 2294–2309. [Google Scholar] [CrossRef] [PubMed]
- Tuček, S.; Doležal, V.; Sullivan, A.C. Inhibition of the synthesis of acetylcholine in rat brain slices by (-)-hydroxycitrate and citrate. J. Neurochem. 1981, 36, 1331–1337. [Google Scholar] [CrossRef]
- Gibson, G.E.; Peterson, C. Acetylcholine and oxidative metabolism in septum and hippocampus in vitro. J. Biol. Chem. 1983, 25, 1142–1145. [Google Scholar] [CrossRef]
- Boutin, R.C.; Alsahafi, Z.; Pagliardini, S.J. Cholinergic modulation of the parafacial respiratory group. J. Physiol. 2017, 595, 1377–1392. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Ghosh, P.; Wan, R.; Ouyang, X.; Cheng, H.; Mattson, M.P.; Cheng, A. Permeability transition pore-mediated mitochondrial superoxide flashes mediate an early inhibitory effect of amyloid beta1-42 on neural progenitor cell proliferation. Neurobiol. Aging 2014, 35, 975–987. [Google Scholar] [CrossRef]
- Hettiarachchi, N.T.; Dallas, M.L.; Pearson, H.A.; Bruce, G.; Deuchars, S.; Boyle, J.P.; Peers, C. Gap junction-mediated spontaneous Ca(2+) waves in differentiated cholinergic SN56 cells. Biochem. Biophys. Res. Commun. 2010, 397, 644–648. [Google Scholar] [CrossRef]
- Ronowska, A.; Dyś, A.; Jankowska-Kulawy, A.; Klimaszewska-Łata, J.; Bielarczyk, H.; Romianowski, P.; Pawełczyk, T.; Szutowicz, A. Short-term effects of zinc on acetylcholine metabolism and viability of SN56 cholinergic neuroblastoma cells. Neurochem. Int. 2010, 56, 143–151. [Google Scholar] [CrossRef]
- Sonkusare, S.; Srinivasan, K.; Kaul, C.; Ramarao, P. Effect of donepezil and lercanidipine on memory impairment induced by intracerebroventricular streptozotocin in rats. Life Sci. 2005, 77, 1–14. [Google Scholar] [CrossRef]
- Szutowicz, A.; Srere, P.A.; Allen, C.N.; Crawford, I.L. Effects of septal lesions on enzymes of acetyl-CoA metabolism in the cholinergic system of the rat hippocampus. J. Neurochem. 1982, 39, 458–463. [Google Scholar] [CrossRef]
- Tomaszewicz, M.; Rossner, S.; Schliebs, R.; Cwikowska, J.; Szutowicz, A. Changes in cortical acetyl-CoA metabolism after selective basal forebrain cholinergic degeneration by 192IgG-saporin. J. Neurochem. 2003, 87, 318–324. [Google Scholar] [CrossRef]
- Szutowicz, A.; Stepień, M.; Lysiak, W.; Angielski, S. Purificational and kinetic properties of ATP: Citrate oxaloacetate lyase from rat brain. J. Neurochem. 1974, 25, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Meher, M.M.; Afrin, M.; Jahan, M.R.; Nozaki, K.; Masumoto, K.H.; Yanai, A.; Islam, M.N. Neurochemical heterogeneity of ChAT-immunoreactive neurons in the basal forebrain cholinergic nuclei and striatum in reference to CGRP, CCK, and calcium-binding proteins. Acta Histochem. 2025, 127, 152291. [Google Scholar] [CrossRef] [PubMed]
- Ueno, M.; Chiba, Y.; Murakami, R.; Miyai, Y.; Matsumoto, K.; Wakamatsu, K.; Takebayashi, G.; Uemura, N.; Yanase, K. Distribution of monocarboxylate transporters in brain and choroid plexus epithelium. Pharmaceutics 2023, 15, 2062. [Google Scholar] [CrossRef] [PubMed]
- Le Ray, D.; Bertrand, S.S.; Dubuc, R. Cholinergic modulation cirquits in vertebrates. Int. J. Mol. Sci. 2022, 23, 10738. [Google Scholar] [CrossRef]
- Kobayashi, M.M.; Brownstein, M.; Saavedra, J.M.; Palkovits, M. Choline acetyltransferase content in discrete regions of the rat brain. J. Neurochem. 1975, 24, 637–640. [Google Scholar] [CrossRef]
- Simon, J.R.; Oderfeld-Nowak, B.; Felten, D.L.; Aprison, M.H. Distribution of choline acetyltransferase, acetylcholinesterase, muscarinic receptor binding, and choline uptake, in discrete areas of the rat medulla oblongata. Neurochem. Res. 1981, 6, 497–505. [Google Scholar] [CrossRef]
- Dahlström, A.; Heiwall, P.O. Intra-axonal transport of transmitters in mammalian neurons. J. Neur. Trans. 1975, 12, 97–114. [Google Scholar]
- Ishijima, T.; Nakajima, K. Restoration of injured motoneurons reduce microglial proliferation in the adult rat facial nucleus. J. Neuropath. Exp. Neurol. 2024, 83, 166–180. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, R.A.D. Axonal transport of acetylcholine, choline acetyltransferase an cholinersterase in regenerating peripheral nerve. J. Physiol. 1978, 282, 91–103. [Google Scholar] [CrossRef]
- Lee, J.M.; Yoo, M.C.; Kim, Y.J.; Kim, S.S.; Yeo, S.G. Expression of ChAT, Iba-1, and NOS in the central nervous system following facial nerve injury. Antioxidants 2024, 13, 595. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Lu, Y.; Zhu, R.; Zong, Y.; Huang, Y.; Wang, D.; Da, Z.; Yu, B.; Shen, L.; Cao, Q. Pyruvate dehydrogenase beat subunit (Pdhb) promotes peripheral axon regeneration by regulating energy supply and gene expression. Exp. Neurol. 2023, 363, 114360. [Google Scholar] [CrossRef]
- Kato, T.; Murashima, Y.L. Choline acetyltransferase activities in single motor neurons from vertebrate spinal cords. J. Neurochem. 1985, 44, 675–679. [Google Scholar] [CrossRef]
- Zahavi, E.E.; Hoogenraad, C.C. Multiple layers of spatial regulation coordinate axonal cargo transport. Curr. Opin. Neurobiol. 2021, 69, 241–246. [Google Scholar] [CrossRef]
- Roney, J.C.; Cheng, X.T.; Sheng, Z.H. Neuronal endolysosomal transport and lysosomal functionality in maintaining axonostasis. J. Cell Biol. 2022, 221, e202111077. [Google Scholar] [CrossRef]
- Doerksen, A.H.; Herath, N.N.; Sanders, S.S. Fat traffic control: S-acylation in axonal transport. Mol. Pharmacol. 2025, 107, 100039. [Google Scholar] [CrossRef]
- Henn, R.E.; Noureldein, M.H.; Elzinga, S.E.; Kim, B.; Savelieff, M.G.; Feldman, E.L. Glial-neuron crosstalk in health and disease: A focus on metabolism, obesity, and cognitive impairment. Neurobiol. Dis. 2022, 170, 105766. [Google Scholar] [CrossRef]
- Guillaud, L.; Garanzini, A.; Zakhia, S.; De la Fuente, S.; Dimitrov, D.; Boerner, S.; Terenzio, M. Loss of intracellular ATP affects axoplasmic viscosity and pathological protein aggregation in mammalian neurons. Sci. Adv. 2025, 11, eadq6077. [Google Scholar] [CrossRef] [PubMed]
- Torregrosa-Muñumer, R.; Turkla, J.; Ermis, R.; Kvist, J.; Harjuhaahto, S.; Pennonen, J.; Hietakangas, V.; Ylikallio, E.; Tynismaa, H. Metabolic cost and trade-off hypermetabolism in human moor neurons with ATP synthase deficiency. Commun. Biol. 2025, 8, 1759. [Google Scholar] [CrossRef] [PubMed]
- Murray-Cors, S.; Owens, M.; Kiew, Y.T.; Day, M.; Cairns, W.; Prokop, A. Impacts of mitochondrial dysfunction on axonal microtubule bundles as a potential mechanism of neurodegeneration. Front. Neurosci. 2025, 19, 1631752. [Google Scholar] [CrossRef] [PubMed]
- Even, A.; Morelli, G.; Tuchetto, S.; Shilian, M.; Le Bail, R.; Laguesse, S.; Krusy, N.; Brisker, A.; Brandis, A.; Inbar, S.; et al. ATP citrate lyase promotes axonal transport across species. Nat. Commun. 2021, 12, 5878, Erratum in Nat Commun. 2021, 12, 6678. [Google Scholar] [CrossRef]
- Aoyama, T.; Hafa, S.; Nakao, T.; Tanigawa, Y.; Oka, C.; Kawaichi, M. Cayman ataxia protein caytaxin is transported by kinesin along neurites through binding to kinesin light chains. J. Cell Sci. 2009, 122, 4177–4185. [Google Scholar] [CrossRef]
- Sun, J.; Pan, C.Q.; Chew, T.W.; Liang, F.; Burmeister, M.; Low, B.C. BNIP-H recruits the cholinergic machinery to neurite terminals to promote acetylcholine signaling and neuritogenesis. Dev. Cell 2015, 34, 555–568. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, M.; Guo, Y.; Wang, L.; Guo, W. D-2-hydroxyglutarate dehydrogenase governs adult neural stem cell activation and promotes histone acetylation via ATP-citrate lyase. Cell Rep. 2023, 42, 112067. [Google Scholar] [CrossRef]
- Chittaranjan, S.; Chan, S.; Yang, C.; Yang, K.C.; Chen, V.; Moradian, A.; Firme, M.; Song, J.; Go, N.E.; Blough, M.D.; et al. Mutations in CIC and IDH1 cooperativaly regulate 2-hydroxyglutarate levels and cell clongenicity. Oncotarget 2014, 5, 7960–7979. [Google Scholar] [CrossRef]
- Angelova, P.R.; Abramov, A.Y. Interplay of mitochondrial calcium signalling and reactive oxygen species production in the brain. Biochem. Soc. Trans. 2024, 52, 1939–1946. [Google Scholar] [CrossRef]
- Murphy, M.; Wilson, Y.M.; Vargas, E.; Munro, K.M.; Smith, B.; Huang, A.; Li, Q.X.; Xiao, J.; Masters, C.L.; Reid, C.A.; et al. Reduction of p75 neurotrophin receptor ameliorates the cognitive deficits in a model of Alzheimer’s disease. Neurobiol. Aging 2015, 36, 740–752. [Google Scholar] [CrossRef] [PubMed]
- Sutendra, G.; Kinnaird, A.; Dromparis, P.; Paulin, R.; Stenson, T.H.; Haromy, A.; Hashimoto, K.; Zhang, N.; Flaim, E.; Michelakis, E.D. A nuclear pyruvate dehydrogenase complex is important for the generation of acetyl-CoA and histone acetylation. Cell 2014, 158, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Zervopoulos, S.D.; Boukouris, A.E.; Saleme, B.; Haromy, A.; Tejay, S.; Sutendra, G.; Michelakis, E.D. MFN2-driven mitochondria-to-nucleus tethering allows a non-canonical nuclear entry pathway of the mitochondrial pyruvate dehydrogenase complex. Mol. Cell. 2022, 82, 1066–1077. [Google Scholar] [CrossRef]
- Kagawa, Y.; Umaru, B.A.; Shima, H.; Ito, R.; Zama, R.; Islam, A.; Kanno, S.-I.; Yasui, A.; Sato, S.; Jozaki, K.; et al. FABP7 Regulates Acetyl-CoA Metabolism Through the Interaction with ACLY in the Nucleus of Astrocytes. Mol. Neurobiol. 2020, 57, 4891–4910. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Gul-Hinc, S.; Jankowska-Kulawy, A.; Szutowicz, A. Cholinergic Phenotypes of Acetyl-CoA with ATP-Citrate Lyase Link. Int. J. Mol. Sci. 2026, 27, 782. https://doi.org/10.3390/ijms27020782
Gul-Hinc S, Jankowska-Kulawy A, Szutowicz A. Cholinergic Phenotypes of Acetyl-CoA with ATP-Citrate Lyase Link. International Journal of Molecular Sciences. 2026; 27(2):782. https://doi.org/10.3390/ijms27020782
Chicago/Turabian StyleGul-Hinc, Sylwia, Agnieszka Jankowska-Kulawy, and Andrzej Szutowicz. 2026. "Cholinergic Phenotypes of Acetyl-CoA with ATP-Citrate Lyase Link" International Journal of Molecular Sciences 27, no. 2: 782. https://doi.org/10.3390/ijms27020782
APA StyleGul-Hinc, S., Jankowska-Kulawy, A., & Szutowicz, A. (2026). Cholinergic Phenotypes of Acetyl-CoA with ATP-Citrate Lyase Link. International Journal of Molecular Sciences, 27(2), 782. https://doi.org/10.3390/ijms27020782





