Small-Cell Lung Cancer—An Update on Targeted and Immunotherapies
Abstract
1. Introduction
2. Pathological and Genomic Profiles of SCLC
3. Current Standard Treatment
3.1. Limited Stage SCLC
3.2. Extensive Stage SCLC
| Study | Year | Agents | Phase; Line (n) | Key Results |
|---|---|---|---|---|
| First Line Treatment | ||||
| IMpower133 [5] | 2018 | EP +/− atezolizumab | III; 1 (403) | EP + atezolizumab: mOS 12.3 mo EP + placebo: mOS 10.3 mo (p = 0.0154) |
| CASPIAN [22] | 2019 | EP + durvalumab + tremelimumab or EP + durvalumab or EP alone | III; 1 (805) | EP + durva + trem: mOS 10.4 mo EP + durva: mOS 12.9 mo EP alone: mOS 10.5 mo |
| KEYNOTE-604 [23] | 2020 | EP +/− pembrolizumab | III; 1 (453) | EP + pembrolizumab: mOS 10.8 mo EP + placebo: 9.7 mo (p = 0.0164 1) |
| CheckMate 451 [24] | 2021 | EP -> ipilimumab + nivolumab followed by nivolumab, or EP -> nivolumab, or EP -> placebo | III; 1 (849) | EP -> ipi/nivo: mOS 9.2 mo EP -> nivo: mOS 10.4 mo EP -> placebo: 9.6 mo |
| CAPSTONE-1 [25] | 2022 | EP +/− adebrelimab | III; 1 (462) | EP + adebrelimab: mOS 15.3 mo EP + placebo: mOS 12.8 mo (p = 0.0017) |
| ASTRUM-005 [26] | 2022 | EP +/− serplulimab | III; 1 (585) | EP + serplulimab: mOS 15.4 mo EP + placebo: mOS 10.9 mo (p < 0.001) |
| Recurrent Disease | ||||
| CheckMate 032 [28] | 2016 | Nivolumab 2 | I/II; ≥3 (109) | mPFS 1.4 mo; mOS 5.6 mo |
| KEYNOTE-028, KEYNOTE-158 [29] | 2020 3 | Pembrolizumab (10 mg/kg q2 weeks, n = 19, 200 mg q3 weeks n = 64) | Ib/II; ≥2 (83) | mPFS 2.0 mo, mOS 7.7 mo (mOS 14.6 mo for PD-L1 CPS ≥ 1) |
4. Recurrent Disease
4.1. Chemotherapy
4.2. Immunotherapy
4.3. Alkylating Agent—Lurbinectedin
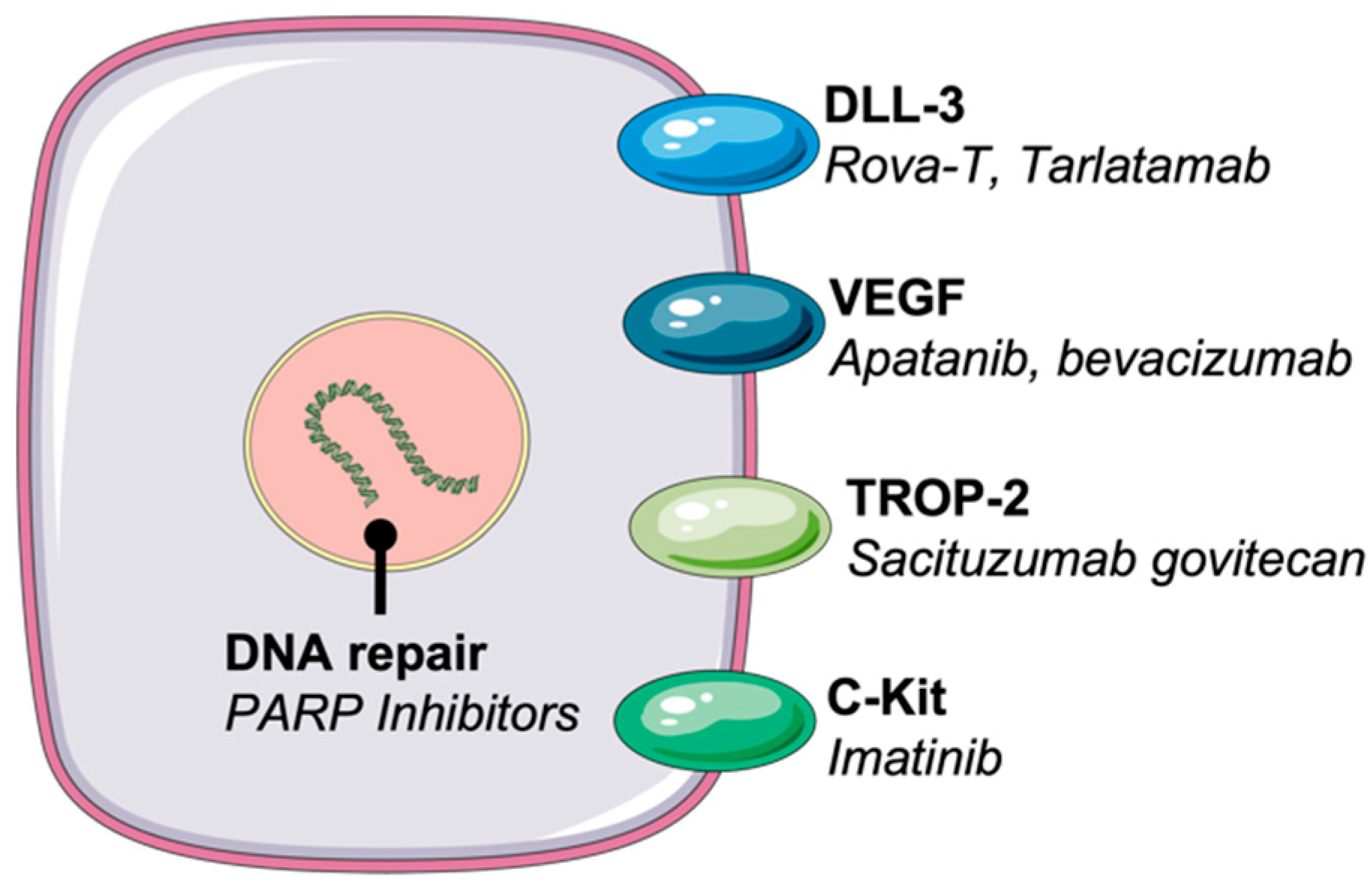
5. Emerging Therapies
5.1. Antibody–Drug Conjugates (ADCs)
5.1.1. DLL-3 Targeting ADCs
5.1.2. TROP-2 Directed ADCs
5.2. PARP Inhibitors
| Agents | Study Design | Key Results |
|---|---|---|
| Veliparib + temozolomide [58] | Phase II, randomized, placebo-controlled N = 104 | TMZ/veliparib mOS 8.2 mo; ORR 39% TMZ/placebo mOS 7.0 mo; ORR 14% SLFN11 predicted improved PARPi response |
| Olaparib + temozolomide [59] | Phase I/II, single-arm N = 48 | mOS 6.7 mo; ORR 41% * |
| Fuzuloparib + SHR-1316 (PD-L1 inhibitor) [63] | Phase Ib, multi-stage N = 23 | mOS 5.6 mo; ORR 6.3% * |
| Olaparib + durvalumab [62] | Phase II, single-arm N = 20 | mOS 4.1 mo; ORR 10.5% 1 CR in BRCA-mutant disease |
5.3. Anti-Angiogenesis
5.4. Tyrosine Kinase Inhibitors
5.5. Bispecific T-Cell Engagers
6. Discussion
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gazdar, A.F.; Bunn, P.A.; Minna, J.D. Small-cell lung cancer: What we know, what we need to know and the path forward. Nat. Rev. Cancer 2017, 17, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Seute, T.; Leffers, P.; ten Velde, G.P.M.; Twijnstra, A. Neurologic disorders in 432 consecutive patients with small cell lung carcinoma. Cancer 2004, 100, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Skarlos, D.; Samantas, E.; Kosmidis, P.; Fountzilas, G.; Angelidou, M.; Palamidas, P.; Mylonakis, N.; Provata, A.; Papadakis, E.; Klouvas, G.; et al. Randomized comparison of etoposide-cisplatin vs. etoposide-carboplatin and irradiation in small-cell lung cancer: Hellenic Co-operative Oncology Group. Ann. Oncol. 1995, 12, 601–607. [Google Scholar] [CrossRef]
- Stahel, R.A. Lung Cancer Therapy Annual, 7th ed.; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar] [CrossRef]
- Liu, S.V.; Reck, M.; Mansfield, A.S.; Mok, T.; Scherpereel, A.; Reinmuth, N.; Garassino, M.C.; De Castro Carpeno, J.; Califano, R.; Nishio, M.; et al. Updated Overall Survival and PD-L1 Subgroup Analysis of Patients with Extensive-Stage Small-Cell Lung Cancer Treated with Atezolizumab, Carboplatin, and Etoposide (IMpower133). J. Clin. Oncol. 2021, 39, 619–630. [Google Scholar] [CrossRef]
- Travis, W.D.; Brambilla, E.; Nicholson, A.G.; Yatabe, Y.; Austin, J.H.M.; Beasley, M.B.; Chirieac, L.R.; Dacic, S.; Duhig, E.; Flieder, D.B.; et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J. Thorac. Oncol. 2015, 10, 1243–1260. [Google Scholar] [CrossRef]
- George, J.; Lim, J.S.; Jang, S.J.; Cun, Y.; Ozretić, L.; Kong, G.; Leenders, F.; Lu, X.; Fernández-Cuesta, L.; Bosco, G. Comprehensive genomic profiles of small cell lung cancer. Nature 2015, 524, 47–53. [Google Scholar] [CrossRef]
- Rudin, C.M.; Durinck, S.; Stawiski, E.W.; Poirier, J.T.; Modrusan, Z.; Shames, D.S.; Bergbower, E.A.; Guan, Y.; Shin, J.; Guillory, J. Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat. Genet. 2012, 44, 1111–1116. [Google Scholar] [CrossRef]
- Gay, C.M.; Stewart, C.A.; Park, E.M.; Diao, L.; Groves, S.M.; Heeke, S.; Nabet, B.Y.; Fujimoto, J.; Solis, L.M.; Lu, W.; et al. Patterns of transcription factor programs and immune pathway activation define four major subtypes of SCLC with distinct therapeutic vulnerabilities. Cancer Cell 2021, 39, 346–360.e7. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The eighth edition AJCC cancer staging manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef]
- Micke, P.; Faldum, A.; Metz, T.; Beeh, K.M.; Bittinger, F.; Hengstler, J.G.; Buhl, R. Staging small cell lung cancer: Veterans Administration Lung Study Group versus International Association for the Study of Lung Cancer—What limits limited disease? Lung Cancer 2002, 37, 271–276. [Google Scholar] [CrossRef]
- Pignon, J.-P.; Arriagada, R.; Ihde, D.C.; Johnson, D.H.; Perry, M.C.; Souhami, R.L.; Brodin, O.; Joss, R.A.; Kies, M.S.; Lebeau, B.; et al. A Meta-Analysis of Thoracic Radiotherapy for Small-Cell Lung Cancer. N. Engl. J. Med. 1992, 327, 1618–1624. [Google Scholar] [CrossRef] [PubMed]
- Fukuoka, M.; Furuse, K.; Saijo, N.; Nishiwaki, Y.; Ikegami, H.; Tamura, T.; Shimoyama, M.; Suemasu, K. Randomized Trial of Cyclophosphamide, Doxorubicin, and Vincristine Versus Cisplatin and Etoposide Versus Alternation of These Regimens in Small-Cell Lung Cancer. JNCI J. Natl. Cancer Inst. 1991, 83, 855–861. [Google Scholar] [CrossRef]
- Kosmidis, P.A.; Samantas, E.; Fountzilas, G.; Pavlidis, N.; Apostolopoulou, F.; Skarlos, D. Cisplatin/etoposide versus carboplatin/etoposide chemotherapy and irradiation in small cell lung cancer: A randomized phase III study. Lung Cancer 1995, 12, 146–147. [Google Scholar] [CrossRef]
- Wakeam, E.; Acuna, S.A.; Leighl, N.B.; Giuliani, M.E.; Finlayson, S.R.G.; Varghese, T.K.; Darling, G.E. Surgery Versus Chemotherapy and Radiotherapy For Early and Locally Advanced Small Cell Lung Cancer: A Propensity-Matched Analysis of Survival. Lung Cancer 2017, 109, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.-F.J.; Chan, D.Y.; Speicher, P.J.; Gulack, B.C.; Wang, X.; Hartwig, M.G.; Onaitis, M.W.; Tong, B.C.; D’Amico, T.A.; Berry, M.F.; et al. Role of Adjuvant Therapy in a Population-Based Cohort of Patients with Early-Stage Small-Cell Lung Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Saji, H.; Miyazawa, T.; Marushima, H.; Nakamura, H. Pathological upstaging and treatment strategy of clinical stage I small cell lung cancer following surgery. J. Thorac. Dis. 2017, 9, E285–E289. [Google Scholar] [CrossRef]
- Stahel, R.; Thatcher, N.; Früh, M.; Le Péchoux, C.; Postmus, P.E.; Sorensen, J.B.; Felip, E. 1st ESMO Consensus Conference in lung cancer; Lugano 2010: Small-cell lung cancer. Ann. Oncol. 2011, 22, 1973–1980. [Google Scholar] [CrossRef]
- Govindan, R.; Page, N.; Morgensztern, D.; Read, W.; Tierney, R.; Vlahiotis, A.; Spitznagel, E.L.; Piccirillo, J. Changing Epidemiology of Small-Cell Lung Cancer in the United States over the Last 30 Years: Analysis of the Surveillance, Epidemiologic, and End Results Database. J. Clin. Oncol. 2006, 24, 4539–4544. [Google Scholar] [CrossRef]
- Rossi, A.; Maio, M.D.; Chiodini, P.; Rudd, R.M.; Okamoto, H.; Skarlos, D.V.; Früh, M.; Qian, W.; Tamura, T.; Samantas, E.; et al. Carboplatin- or Cisplatin-Based Chemotherapy in First-Line Treatment of Small-Cell Lung Cancer: The COCIS Meta-Analysis of Individual Patient Data. J. Clin. Oncol. 2012, 30, 1692–1698. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Özgüroğlu, M.; Ji, J.H. Durvalumab plus platinum–etoposide versus platinum–etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): A randomised, controlled, open-label, phase 3 trial. Lancet 2019, 394, 1929–1939. [Google Scholar] [CrossRef]
- Goldman, J.W.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Özgüroğlu, M.; Ji, J.H. Durvalumab, with or without tremelimumab, plus platinum–etoposide versus platinum–etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): Updated results from a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M.; Awad, M.M.; Navarro, A.; Gottfried, M.; Peters, S.; Csőszi, T.; Cheema, P.K.; Rodriguez-Abreu, D.; Wollner, M.; Yang, J.C.-H.; et al. Pembrolizumab or Placebo Plus Etoposide and Platinum as First-Line Therapy for Extensive-Stage Small-Cell Lung Cancer: Randomized, Double-Blind, Phase III KEYNOTE-604 Study. J. Clin. Oncol. 2020, 38, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Owonikoko, T.K.; Park, K.; Govindan, R.; Ready, N.; Reck, M.; Peters, S.; Dakhil, S.R.; Navarro, A.; Rodríguez-Cid, J.; Schenker, M.; et al. Nivolumab and Ipilimumab as Maintenance Therapy in Extensive-Disease Small-Cell Lung Cancer: CheckMate 451. J. Clin. Oncol. 2021, 39, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhou, C.; Yao, W.; Wang, Q.; Min, X.; Chen, G.; Xu, X.; Li, X.; Xu, F.; Fang, Y. Adebrelimab or placebo plus carboplatin and etoposide as first-line treatment for extensive-stage small-cell lung cancer (CAPSTONE-1): A multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022, 23, 739–747. [Google Scholar] [CrossRef]
- Cheng, Y.; Han, L.; Wu, L.; Chen, J.; Sun, H.; Wen, G.; Ji, Y.; Dvorkin, M.; Shi, J.; Pan, Z.; et al. Effect of First-Line Serplulimab vs Placebo Added to Chemotherapy on Survival in Patients With Extensive-Stage Small Cell Lung Cancer: The ASTRUM-005 Randomized Clinical Trial. JAMA 2022, 328, 1223–1232. [Google Scholar] [CrossRef]
- Horn, L.; Mansfield, A.S.; Szczęsna, A.; Havel, L.; Krzakowski, M.; Hochmair, M.J.; Huemer, F.; Losonczy, G.; Johnson, M.L.; Nishio, M.; et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2220–2229. [Google Scholar] [CrossRef]
- Ready, N.; Farago, A.F.; de Braud, F.; Atmaca, A.; Hellmann, M.D.; Schneider, J.G.; Spigel, D.R.; Moreno, V.; Chau, I.; Hann, C.L.; et al. Third-Line Nivolumab Monotherapy in Recurrent SCLC: CheckMate 032. J. Thorac. Oncol. 2019, 14, 237–244. [Google Scholar] [CrossRef]
- Chung, H.C.; Piha-Paul, S.A.; Lopez-Martin, J.; Schellens, J.H.M.; Kao, S.; Miller, W.H.; Delord, J.-P.; Gao, B.; Planchard, D.; Gottfried, M.; et al. Pembrolizumab After Two or More Lines of Previous Therapy in Patients with Recurrent or Metastatic SCLC: Results From the KEYNOTE-028 and KEYNOTE-158 Studies. J. Thorac. Oncol. 2020, 15, 618–627. [Google Scholar] [CrossRef]
- Spigel, D.; Vicente, D.; Ciuleanu, T.; Gettinger, S.; Peters, S.; Horn, L.; Audigier-Valette, C.; Aranda, N.P.; Juan-Vidal, O.; Cheng, Y. Second-line nivolumab in relapsed small-cell lung cancer: CheckMate 331☆. Ann. Oncol. 2021, 32, 631–641. [Google Scholar] [CrossRef]
- Aix, S.P.; Ciuleanu, T.E.; Navarro, A.; Cousin, S.; Bonanno, L.; Smit, E.F.; Chiappori, A.; Olmedo, M.E.; Horvath, I.; Grohé, C.; et al. Combination lurbinectedin and doxorubicin versus physician’s choice of chemotherapy in patients with relapsed small-cell lung cancer (ATLANTIS): A multicentre, randomised, open-label, phase 3 trial. Lancet Respir. Med. 2023, 11, 74–86. [Google Scholar] [CrossRef]
- Lung Cancer Research Foundation. FDA Approvals in Lung Cancer Treatment. Available online: https://www.lungcancerresearchfoundation.org/research/why-research/treatment-advances/ (accessed on 29 March 2022).
- O’Brien, M.E.; Ciuleanu, T.E.; Tsekov, H.; Shparyk, Y.; Cuceviá, B.; Juhasz, G.; Thatcher, N.; Ross, G.A.; Dane, G.C.; Crofts, T. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J. Clin. Oncol. 2006, 24, 5441–5447. [Google Scholar] [CrossRef] [PubMed]
- von Pawel, J.; Schiller, J.H.; Shepherd, F.A.; Fields, S.Z.; Kleisbauer, J.P.; Chrysson, N.G.; Stewart, D.J.; Clark, P.I.; Palmer, M.C.; Depierre, A.; et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J. Clin. Oncol. 1999, 17, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Baize, N.; Monnet, I.; Greillier, L.; Geier, M.; Lena, H.; Janicot, H.; Vergnenegre, A.; Crequit, J.; Lamy, R.; Auliac, J.-B.; et al. Carboplatin plus etoposide versus topotecan as second-line treatment for patients with sensitive relapsed small-cell lung cancer: An open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2020, 21, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Care Network. Small Cell Lung Cancer (Version 2.2022). Available online: https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf (accessed on 29 March 2022).
- Chung, H.C.; Lopez-Martin, J.A.; Kao, S.C.-H.; Miller, W.H.; Ros, W.; Gao, B.; Marabelle, A.; Gottfried, M.; Zer, A.; Delord, J.-P.; et al. Phase 2 study of pembrolizumab in advanced small-cell lung cancer (SCLC): KEYNOTE-158. J. Clin. Oncol. 2018, 36, 8506. [Google Scholar] [CrossRef]
- Calvo, E.; Spira, A.; Miguel, M.; Kondo, S.; Gazzah, A.; Millward, M.; Prenen, H.; Rottey, S.; Warburton, L.; Alanko, T.; et al. Safety, pharmacokinetics, and efficacy of budigalimab with rovalpituzumab tesirine in patients with small cell lung cancer. Cancer Treat. Res. Commun. 2021, 28, 100405. [Google Scholar] [CrossRef]
- Farago, A.F.; Drapkin, B.J.; Lopez-Vilarino de Ramos, J.A.; Galmarini, C.M.; Núñez, R.; Kahatt, C.; Paz-Ares, L. ATLANTIS: A Phase III study of lurbinectedin/doxorubicin versus topotecan or cyclophosphamide/doxorubicin/vincristine in patients with small-cell lung cancer who have failed one prior platinum-containing line. Future Oncol. 2019, 15, 231–239. [Google Scholar] [CrossRef]
- Singh, S.; Jaigirdar, A.A.; Mulkey, F.; Cheng, J.; Hamed, S.S.; Li, Y.; Liu, J.; Zhao, H.; Goheer, A.; Helms, W.S. FDA approval summary: Lurbinectedin for the treatment of metastatic small cell lung cancer. Clin. Cancer Res. 2021, 27, 2378–2382. [Google Scholar] [CrossRef]
- Trigo, J.; Subbiah, V.; Besse, B.; Moreno, V.; López, R.; Sala, M.A.; Peters, S.; Ponce, S.; Fernández, C.; Alfaro, V.; et al. Lurbinectedin as second-line treatment for patients with small-cell lung cancer: A single-arm, open-label, phase 2 basket trial. Lancet Oncol. 2020, 21, 645–654. [Google Scholar] [CrossRef]
- Huang, R.S.P.; Holmes, B.F.; Powell, C.; Marati, R.V.; Tyree, D.; Admire, B.; Streator, A.; Newell, A.E.H.; Perez, J.; Dalvi, D.; et al. Delta-like Protein 3 Prevalence in Small Cell Lung Cancer and DLL3 (SP347) Assay Characteristics. Arch. Pathol. Lab. Med. 2019, 143, 1373–1377. [Google Scholar] [CrossRef]
- Saunders, L.; Bankovich, A.; Anderson, W.; Aujay, M.; Bheddah, S.; Black, K.; Desai, R.; Escarpe, P.; Hampl, J.; Laysang, A.; et al. A DLL3-targeted antibody-drug conjugate eradicates high-grade pulmonary neuroendocrine tumor-initiating cells in vivo. Sci. Transl. Med. 2015, 7, 302ra136. [Google Scholar] [CrossRef]
- Giffin, M.J.; Cooke, K.; Lobenhofer, E.K.; Estrada, J.; Zhan, J.; Deegen, P.; Thomas, M.; Murawsky, C.M.; Werner, J.; Liu, S.; et al. AMG 757, a Half-Life Extended, DLL3-Targeted Bispecific T-Cell Engager, Shows High Potency and Sensitivity in Preclinical Models of Small-Cell Lung Cancer. Clin. Cancer Res. 2021, 27, 1526–1537. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M.; Pietanza, M.C.; Bauer, T.M.; Ready, N.; Morgensztern, D.; Glisson, B.S.; Byers, L.A.; Johnson, M.L.; Burris, H.A., 3rd; Robert, F.; et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: A first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol. 2017, 18, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.L.; Zvirbule, Z.; Laktionov, K.; Helland, A.; Cho, B.C.; Gutierrez, V.; Colinet, B.; Lena, H.; Wolf, M.; Gottfried, M.; et al. Rovalpituzumab Tesirine as a Maintenance Therapy After First-Line Platinum-Based Chemotherapy in Patients with Extensive-Stage–SCLC: Results From the Phase 3 MERU Study. J. Thorac. Oncol. 2021, 16, 1570–1581. [Google Scholar] [CrossRef] [PubMed]
- Blackhall, F.; Jao, K.; Greillier, L.; Cho, B.C.; Penkov, K.; Reguart, N.; Majem, M.; Nackaerts, K.; Syrigos, K.; Hansen, K.; et al. Efficacy and Safety of Rovalpituzumab Tesirine Compared with Topotecan as Second-Line Therapy in DLL3-High SCLC: Results From the Phase 3 TAHOE Study. J. Thorac. Oncol. 2021, 16, 1547–1558. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, J.; Nikolinakos, P.; Leal, T.; Lehman, J.; Morgensztern, D.; Patel, J.D.; Wrangle, J.M.; Curigliano, G.; Greillier, L.; Johnson, M.L.; et al. A Phase 1–2 Study of Rovalpituzumab Tesirine in Combination with Nivolumab Plus or Minus Ipilimumab in Patients With Previously Treated Extensive-Stage SCLC. J. Thorac. Oncol. 2021, 16, 1559–1569. [Google Scholar] [CrossRef] [PubMed]
- Hann, C.L.; Burns, T.F.; Dowlati, A.; Morgensztern, D.; Ward, P.J.; Koch, M.M.; Chen, C.; Ludwig, C.; Patel, M.; Nimeiri, H.; et al. A Phase 1 Study Evaluating Rovalpituzumab Tesirine in Frontline Treatment of Patients with Extensive-Stage SCLC. J. Thorac. Oncol. 2021, 16, 1582–1588. [Google Scholar] [CrossRef]
- Bardia, A.; Hurvitz, S.A.; Tolaney, S.M.; Loirat, D.; Punie, K.; Oliveira, M.; Brufsky, A.; Sardesai, S.D.; Kalinsky, K.; Zelnak, A.B.; et al. Sacituzumab Govitecan in Metastatic Triple-Negative Breast Cancer. N. Engl. J. Med. 2021, 384, 1529–1541. [Google Scholar] [CrossRef]
- Gray, J.E.; Heist, R.S.; Starodub, A.N.; Camidge, D.R.; Kio, E.A.; Masters, G.A.; Purcell, W.T.; Guarino, M.J.; Misleh, J.; Schneider, C.J.; et al. Therapy of Small Cell Lung Cancer (SCLC) with a Topoisomerase-I-inhibiting Antibody-Drug Conjugate (ADC) Targeting Trop-2, Sacituzumab Govitecan. Clin. Cancer Res. 2017, 23, 5711–5719. [Google Scholar] [CrossRef]
- TGA. Trodelvy. 2021. Available online: https://www.tga.gov.au/apm-summary/trodelvy (accessed on 30 August 2022).
- U.S. FDA Grants Accelerated Approval to Trodelvy® for the Treatment of Metastatic Urothelial; Gilead Sciences, Inc.: Foster City, CA, USA, 2021.
- Satoh, M.S.; Lindahl, T. Role of poly(ADP-ribose) formation in DNA repair. Nature 1992, 356, 356–358. [Google Scholar] [CrossRef]
- Allison Stewart, C.; Tong, P.; Cardnell, R.J.; Sen, T.; Li, L.; Gay, C.M.; Masrorpour, F.; Fan, Y.; Bara, R.O.; Feng, Y.; et al. Dynamic variations in epithelial-to-mesenchymal transition (EMT), ATM, and SLFN11 govern response to PARP inhibitors and cisplatin in small cell lung cancer. Oncotarget 2017, 8, 28575–28587. [Google Scholar] [CrossRef]
- Woll, P.; Gaunt, P.; Steele, N.; Ahmed, S.; Mulatero, C.; Shah, R.; Danson, S.; Hodgkinson, E.; James, K.; Watkins, B.; et al. P1.07-015 STOMP: A UK National Cancer Research Network Randomised, Double Blind, Multicentre Phase II Trial of Olaparib as Maintenance Therapy in SCLC. J. Thorac. Oncol. 2017, 12, S704–S705. [Google Scholar] [CrossRef]
- Knelson, E.H.; Patel, S.A.; Sands, J.M. PARP Inhibitors in Small-Cell Lung Cancer: Rational Combinations to Improve Responses. Cancers 2021, 13, 727. [Google Scholar] [CrossRef] [PubMed]
- Pietanza, M.C.; Waqar, S.N.; Krug, L.M.; Dowlati, A.; Hann, C.L.; Chiappori, A.; Owonikoko, T.K.; Woo, K.M.; Cardnell, R.J.; Fujimoto, J.; et al. Randomized, Double-Blind, Phase II Study of Temozolomide in Combination with Either Veliparib or Placebo in Patients with Relapsed-Sensitive or Refractory Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 2386–2394. [Google Scholar] [CrossRef] [PubMed]
- Farago, A.F.; Yeap, B.Y.; Stanzione, M.; Hung, Y.P.; Heist, R.S.; Marcoux, J.P.; Zhong, J.; Rangachari, D.; Barbie, D.A.; Phat, S.; et al. Combination Olaparib and Temozolomide in Relapsed Small-Cell Lung Cancer. Cancer Discov. 2019, 9, 1372–1387. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Lin, C.-C.; Bai, L.-Y.; Chung, W.-P.; Kao, W.-C.; Zielinski, R.; Shim, B.Y.; Hong, M.H.; Kim, S.-W.; Liu, C.-Y.; et al. A study of senaparib in combination with temozolomide for the treatment of patients with advanced solid tumors and extensive-stage small cell lung cancer. J. Clin. Oncol. 2022, 40, 3102. [Google Scholar] [CrossRef]
- Sen, T.; Rodriguez, B.L.; Chen, L.; Corte, C.M.D.; Morikawa, N.; Fujimoto, J.; Cristea, S.; Nguyen, T.; Diao, L.; Li, L.; et al. Targeting DNA Damage Response Promotes Antitumor Immunity through STING-Mediated T-cell Activation in Small Cell Lung Cancer. Cancer Discov. 2019, 9, 646–661. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Vilimas, R.; Trindade, C.; Erwin-Cohen, R.; Roper, N.; Xi, L.; Krishnasamy, V.; Levy, E.; Mammen, A.; Nichols, S.; et al. Durvalumab in Combination with Olaparib in Patients with Relapsed SCLC: Results from a Phase II Study. J. Thorac. Oncol. 2019, 14, 1447–1457. [Google Scholar] [CrossRef]
- Xu, Y.; Huang, Z.; Fang, J.; Liu, A.; Lu, H.; Yu, X.; Chen, K.; Xu, X.; Ma, X.; Shi, W.; et al. Tolerability, safety, and preliminary antitumor activity of fuzuloparib in combination with SHR-1316 in patients with relapsed small cell lung cancer: A multicenter, open-label, two-stage, phase Ib trial. Transl. Lung Cancer Res. 2021, 11, 1069–1078. [Google Scholar] [CrossRef]
- Lok, B.H.; Gardner, E.E.; Schneeberger, V.E.; Ni, A.; Desmeules, P.; Rekhtman, N.; de Stanchina, E.; Teicher, B.A.; Riaz, N.; Powell, S.N.; et al. PARP Inhibitor Activity Correlates with SLFN11 Expression and Demonstrates Synergy with Temozolomide in Small Cell Lung Cancer. Clin. Cancer Res. 2017, 23, 523–535. [Google Scholar] [CrossRef]
- Zauderer, M.G.; Drilon, A.; Kadota, K.; Huberman, K.; Sima, C.S.; Bergagnini, I.; Sumner, D.K.; Travis, W.D.; Heguy, A.; Ginsberg, M.S.; et al. Trial of a 5-day dosing regimen of temozolomide in patients with relapsed small cell lung cancers with assessment of methylguanine-DNA methyltransferase. Lung Cancer 2014, 86, 237–240. [Google Scholar] [CrossRef]
- He, J.; Pan, H.; He, J.; Li, S. Effectiveness and safety of bevacizumab in extensive-disease small cell lung cancer: A systemic review and meta-analysis. Ann. Transl. Med. 2021, 9, 1285. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wu, T.; Jing, L.; Li, M.-J.; Tian, T.; Ruan, Z.-P.; Liang, X.; Nan, K.-J.; Liu, Z.-Y.; Yao, Y.; et al. Angiogenesis inhibitors for the treatment of small cell lung cancer (SCLC): A meta-analysis of 7 randomized controlled trials. Medicine 2017, 96, e6412. [Google Scholar] [CrossRef] [PubMed]
- Ready, N.E.; Pang, H.H.; Gu, L.; Otterson, G.A.; Thomas, S.P.; Miller, A.A.; Baggstrom, M.; Masters, G.A.; Graziano, S.L.; Crawford, J.; et al. Chemotherapy with or without Maintenance Sunitinib for Untreated Extensive-Stage Small-Cell Lung Cancer: A Randomized, Double-Blind, Placebo-Controlled Phase II Study-CALGB 30504 (Alliance). J. Clin. Oncol. 2015, 33, 1660–1665. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, S.S.; Belani, C.P.; Mack, P.C.; Vokes, E.E.; Longmate, J.; Govindan, R.; Koczywas, M.; Ivy, S.P.; Gandara, D.R. Phase II Study of Cediranib (AZD 2171), an Inhibitor of the Vascular Endothelial Growth Factor Receptor, for Second-Line Therapy of Small Cell Lung Cancer (National Cancer Institute #7097). J. Thorac. Oncol. 2010, 5, 1279–1284. [Google Scholar] [CrossRef]
- Gitlitz, B.J.; Moon, J.; Glisson, B.S.; Reimers, H.J.; Bury, M.J.; Floyd, J.D.; Schulz, T.K.; Sundaram, P.K.; Ho, C.; Gandara, D.R. Sorafenib in Platinum-Treated Patients with Extensive Stage Small Cell Lung Cancer: A Southwest Oncology Group (SWOG 0435) Phase II Trial. J. Thorac. Oncol. 2010, 5, 1835–1840. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, J.-Y.; Xu, Y.-H.; Chen, M.; Deng, L.-C.; Wu, J.-P.; Zhou, T.; Zhang, L.-Q.; Tan, J.; Pu, X.-X.; et al. Efficacy and safety of apatinib as second or later-line therapy in extensive-stage small cell lung cancer: A prospective, exploratory, single-arm, multi-center clinical trial. Transl. Lung Cancer Res. 2022, 11, 832–844. [Google Scholar] [CrossRef]
- Gao, X.; Peng, L.; Zhang, L.; Huang, K.; Yi, C.; Li, B.; Meng, X.; Li, J. Real-world efficacy and safety of anlotinib as third- or further-line treatment in refractory small cell lung cancer. J. Cancer Res. Clin. Oncol. 2022, 148, 2661–2671. [Google Scholar] [CrossRef]
- Koinis, F.; Agelaki, S.; Karavassilis, V.; Kentepozidis, N.; Samantas, E.; Peroukidis, S.; Katsaounis, P.; Hartabilas, E.; Varthalitis, I.I.; Messaritakis, I.; et al. Second-line pazopanib in patients with relapsed and refractory small-cell lung cancer: A multicentre phase II study of the Hellenic Oncology Research Group. Br. J. Cancer 2017, 117, 8–14. [Google Scholar] [CrossRef]
- Burger, H.; den Bakker, M.A.; Stoter, G.; Verweij, J.; Nooter, K. Lack of c-kit exon 11 activating mutations in c-KIT/CD117-positive SCLC tumour specimens. Eur. J. Cancer 2003, 39, 793–799. [Google Scholar] [CrossRef]
- Krug, L.M.; Crapanzano, J.P.; Heelan, R.T.; Azzoli, C.G.; Miller, V.A.; Rizvi, N.; Gomez, J.; Kris, M.G.; Pizzo, B.; Tyson, L.; et al. Imatinib mesylate lacks activity in small cell lung carcinoma expressing c-kit protein: A phase II clinical trial. Cancer 2005, 103, 2128–2131. [Google Scholar] [CrossRef]
- Tatematsu, A.; Shimizu, J.; Murakami, Y.; Horio, Y.; Nakamura, S.; Hida, T.; Mitsudomi, T.; Yatabe, Y. Epidermal growth factor receptor mutations in small cell lung cancer. Clin. Cancer Res. 2008, 14, 6092–6096. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Xu, J.; Wang, S.; Zhang, Y.; Qian, J.; Qiao, R.; Lu, J.; Zhang, L.; Zhao, Y.; Han, B. Characteristics and outcome of small cell lung cancer patients (SCLC) transformed from adenocarcinoma after tyrosine kinase inhibitor treatment. Ann. Oncol. 2018, 29, ix157. [Google Scholar] [CrossRef]
- Moore, A.M.; Einhorn, L.H.; Estes, D.; Govindan, R.; Axelson, J.; Vinson, J.; Breen, T.E.; Yu, M.; Hanna, N.H. Gefitinib in patients with chemo-sensitive and chemo-refractory relapsed small cell cancers: A Hoosier Oncology Group phase II trial. Lung Cancer 2006, 52, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Shibata, T.; Kokubu, A.; Tsuta, K.; Hirohashi, S. Oncogenic mutation of PIK3CA in small cell lung carcinoma: A potential therapeutic target pathway for chemotherapy-resistant lung cancer. Cancer Lett. 2009, 283, 203–211. [Google Scholar] [CrossRef]
- Walls, M.; Baxi, S.M.; Yin, M.-J.; Mehta, P.P.; Liu, K.K.C.; Jinjiang, Z.H.U.; Estrella, H.; Chunze, L.I.; Zientek, M.; Qing, Z.; et al. Targeting Small Cell Lung Cancer Harboring PIK3CA Mutation with a Selective Oral PI3K Inhibitor PF-4989216. Clin. Cancer Res. 2014, 20, 631–643. [Google Scholar] [CrossRef]
- Koprivnikar, J.; Marcotulli, D.; Jones, E.; Perry, G.; Kuo, Y.-H.; Gagnon, J.; Aviador, M.; Stanislaus, G.; McCloskey, J. Blinatumumab Induces Responses in Extramedulary B-Cell Acute Lymphoid Leukemia (B-ALL) and Lymphoid Blast Crisis Chronic Myelogenous Leukemia (CML), and Rarely Results in CD19 Negative Relapse. Blood 2018, 132, 2703. [Google Scholar] [CrossRef]
- Huehls, A.M.; Coupet, T.A.; Sentman, C.L. Bispecific T-cell engagers for cancer immunotherapy. Immunol. Cell Biol. 2015, 93, 290–296. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Champiat, S.; Lai, W.V.; Izumi, H.; Govindan, R.; Boyer, M.; Hummel, H.-D.; Borghaei, H.; Johnson, M.L.; Steeghs, N.; et al. Tarlatamab, a First-In-Class DLL3-Targeted Bispecific T-Cell Engager, in Recurrent Small Cell Lung Cancer: An Open-Label, Phase I Study. J. Clin. Oncol. 2023. [Google Scholar] [CrossRef]
- Byers, L.A.; Chiappori, A.; Smit, M.-A.D. Phase 1 study of AMG 119, a chimeric antigen receptor (CAR) T cell therapy targeting DLL3, in patients with relapsed/refractory small cell lung cancer (SCLC). J. Clin. Oncol. 2019, 37, TPS8576. [Google Scholar] [CrossRef]
- Amgen. A Phase 1 Study Evaluating the Safety, Tolerability and Effeciacy of AMPG 119 in Subjects with RR SCLC USA: U.S. National Library of Medicine. 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT03392064 (accessed on 30 March 2023).
- Hamilton, G.; Rath, B. Immunotherapy for small cell lung cancer: Mechanisms of resistance. Expert Opin. Biol. Ther. 2019, 19, 423–432. [Google Scholar] [CrossRef]
- Xie, M.; Chugh, P.; Broadhurst, H.; Lai, Z.; Whitston, D.; Paz-Ares, L.; Gay, C.; Byers, L.; Rudin, C.M.; Stewart, R.; et al. Abstract CT024: Durvalumab (D) + platinum-etoposide (EP) in 1L extensive-stage small-cell lung cancer (ES-SCLC): Exploratory analysis of SCLC molecular subtypes in CASPIAN. Cancer Res. 2022, 82, CT024. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Antonia, S.J. Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2019, 380, 989–990. [Google Scholar] [CrossRef] [PubMed]
- Senan, S.; Shire, N.; Mak, G.; Yao, W.; Jiang, H. ADRIATIC: A phase III trial of durvalumab ± tremelimumab after concurrent chemoradiation for patients with limited stage small cell lung cancer. Ann. Oncol. 2019, 30, ii25. [Google Scholar] [CrossRef]
- Ross, H.J.; Hu, C.; Higgins, K.A.; Jabbour, S.K.; Kozono, D.E.; Owonikoko, T.K.; Movsas, B.; Solberg, T.; Xiao, C.; Williams, T.M.; et al. NRG Oncology/Alliance LU005: A phase II/III randomized clinical trial of chemoradiation versus chemoradiation plus atezolizumab in limited stage small cell lung cancer. J. Clin. Oncol. 2020, 38, TPS9082. [Google Scholar] [CrossRef]
- Burris, H.A.; Tibbitts, J.; Holden, S.N.; Sliwkowski, M.X.; Lewis Phillips, G.D. Trastuzumab Emtansine (T-DM1): A Novel Agent for Targeting HER2+ Breast Cancer. Clin. Breast Cancer 2011, 11, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Miles, D.; Gianni, L.; Krop, I.E.; Welslau, M.; Baselga, J.; Pegram, M.; Oh, D.-Y.; Diéras, V.; Guardino, E.; et al. Trastuzumab Emtansine for HER2-Positive Advanced Breast Cancer. N. Engl. J. Med. 2012, 367, 1783–1791. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Huang, C.-S.; Mano, M.S.; Loibl, S.; Mamounas, E.P.; Untch, M.; Wolmark, N.; Rastogi, P.; Schneeweiss, A.; Redondo, A.; et al. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. N. Engl. J. Med. 2019, 380, 617–628. [Google Scholar] [CrossRef]
- Ogitani, Y.; Hagihara, K.; Oitate, M.; Naito, H.; Agatsuma, T. Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody–drug conjugate, in tumors with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci. 2016, 107, 1039–1046. [Google Scholar] [CrossRef]
- Cortés, J.; Kim, S.B.; Chung, W.P.; Im, S.A.; Park, Y.H.; Hegg, R.; Kim, M.H.; Tseng, L.M.; Petry, V.; Chung, C.F.; et al. Trastuzumab Deruxtecan versus Trastuzumab Emtansine for Breast Cancer. N. Engl. J. Med. 2022, 386, 1143–1154. [Google Scholar] [CrossRef]
- Ohba, A.; Morizane, C.; Kawamoto, Y.; Komatsu, Y.; Ueno, M.; Kobayashi, S.; Ikeda, M.; Sasaki, M.; Furuse, J.; Okano, N.; et al. Trastuzumab deruxtecan (T-DXd; DS-8201) in patients (pts) with HER2-expressing unresectable or recurrent biliary tract cancer (BTC): An investigator-initiated multicenter phase 2 study (HERB trial). J. Clin. Oncol. 2022, 40, 4006. [Google Scholar] [CrossRef]
- Uprety, D.; Remon, J.; Adjei, A.A. All That Glitters Is Not Gold: The Story of Rovalpituzumab Tesirine in SCLC. J. Thorac. Oncol. 2021, 16, 1429–1433. [Google Scholar] [CrossRef] [PubMed]
- Meador, C.B.; Digumarthy, S.; Yeap, B.Y.; Farago, A.F.; Heist, R.S.; Marcoux, J.P.; Rangachari, D.; Barbie, D.A.; Piotrowska, Z. Phase I/II investigator-initiated study of olaparib and temozolomide in SCLC: Updated analysis and CNS outcomes. J. Clin. Oncol. 2022, 40, 8565. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Callahan, M.K.; Awad, M.M.; Calvo, E.; Ascierto, P.A.; Atmaca, A.; Rizvi, N.A.; Hirsch, F.R.; Selvaggi, G.; Szustakowski, J.D.; et al. Tumor Mutational Burden and Efficacy of Nivolumab Monotherapy and in Combination with Ipilimumab in Small-Cell Lung Cancer. Cancer Cell 2018, 33, 853–861.e4. [Google Scholar] [CrossRef] [PubMed]
- Almodovar, K.; Iams, W.T.; Meador, C.B.; Zhao, Z.; York, S.; Horn, L.; Yan, Y.; Hernandez, J.; Chen, H.; Shyr, Y.; et al. Longitudinal Cell-Free DNA Analysis in Patients with Small Cell Lung Cancer Reveals Dynamic Insights into Treatment Efficacy and Disease Relapse. J. Thorac. Oncol. 2018, 13, 112–123. [Google Scholar] [CrossRef] [PubMed]
| Subtype (Key Gene; % of Sample 1) | Key Characteristics | Potential Therapeutic Vulnerabilities |
|---|---|---|
| SCLC-A (ASCL1; 51%) | Neuroendocrine, epithelial subtype; TTF1 expression | BCL2 inhibitors |
| SCLC-N (NEUROD1; 23%) | Neuroendocrine, lacks TTF1 expression, cMYC expression | Aurora kinase inhibitors (AURKi) |
| SCLC-P (POU2F3; 7%) | Less neuroendocrine (NE) expression | PARP inhibitors, antimetabolites, AURKi |
| SCLC-I (inflamed; 17%) 2 | Less NE expression, mesenchymal type | Immune checkpoint inhibitors |
| First line | Carboplatin or cisplatin, etoposide + atezolizumab or durvalumab |
| Second line | CAV (cyclophosphamide, doxorubicin, vincristine) Topotecan Lurbinectedin * Nivolumab + Pembrolizumab |
| Agents | Study Design | Key Results |
|---|---|---|
| Cediranib [69] | Phase II, single-arm N = 25 | mPFS 2 mo; mOS 6 mo; No confirmed objective responses |
| Sorafenib [70] | Phase II, single-arm N = 89 | Platinum-sensitive: mOS 6.7 mo; ORR 11% Platinum-resistant: mOS 5.3 mo; ORR 2% |
| Apatinib [71] | Phase II, single-arm N = 57 | mOS 11.2 mo; ORR 14.3% |
| Anlotinib [72] | Retrospective N = 40 | mOS 7.8 mo; ORR 10% |
| Pazopanib [73] | Phase II, single-arm N = 58 | Platinum-sensitive: mOS 8.0 mo; ORR 17.9% Platinum-resistant: mOS 4.0 mo; ORR 5.3% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McNamee, N.; da Silva, I.P.; Nagrial, A.; Gao, B. Small-Cell Lung Cancer—An Update on Targeted and Immunotherapies. Int. J. Mol. Sci. 2023, 24, 8129. https://doi.org/10.3390/ijms24098129
McNamee N, da Silva IP, Nagrial A, Gao B. Small-Cell Lung Cancer—An Update on Targeted and Immunotherapies. International Journal of Molecular Sciences. 2023; 24(9):8129. https://doi.org/10.3390/ijms24098129
Chicago/Turabian StyleMcNamee, Nicholas, Ines Pires da Silva, Adnan Nagrial, and Bo Gao. 2023. "Small-Cell Lung Cancer—An Update on Targeted and Immunotherapies" International Journal of Molecular Sciences 24, no. 9: 8129. https://doi.org/10.3390/ijms24098129
APA StyleMcNamee, N., da Silva, I. P., Nagrial, A., & Gao, B. (2023). Small-Cell Lung Cancer—An Update on Targeted and Immunotherapies. International Journal of Molecular Sciences, 24(9), 8129. https://doi.org/10.3390/ijms24098129







