Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis
Abstract
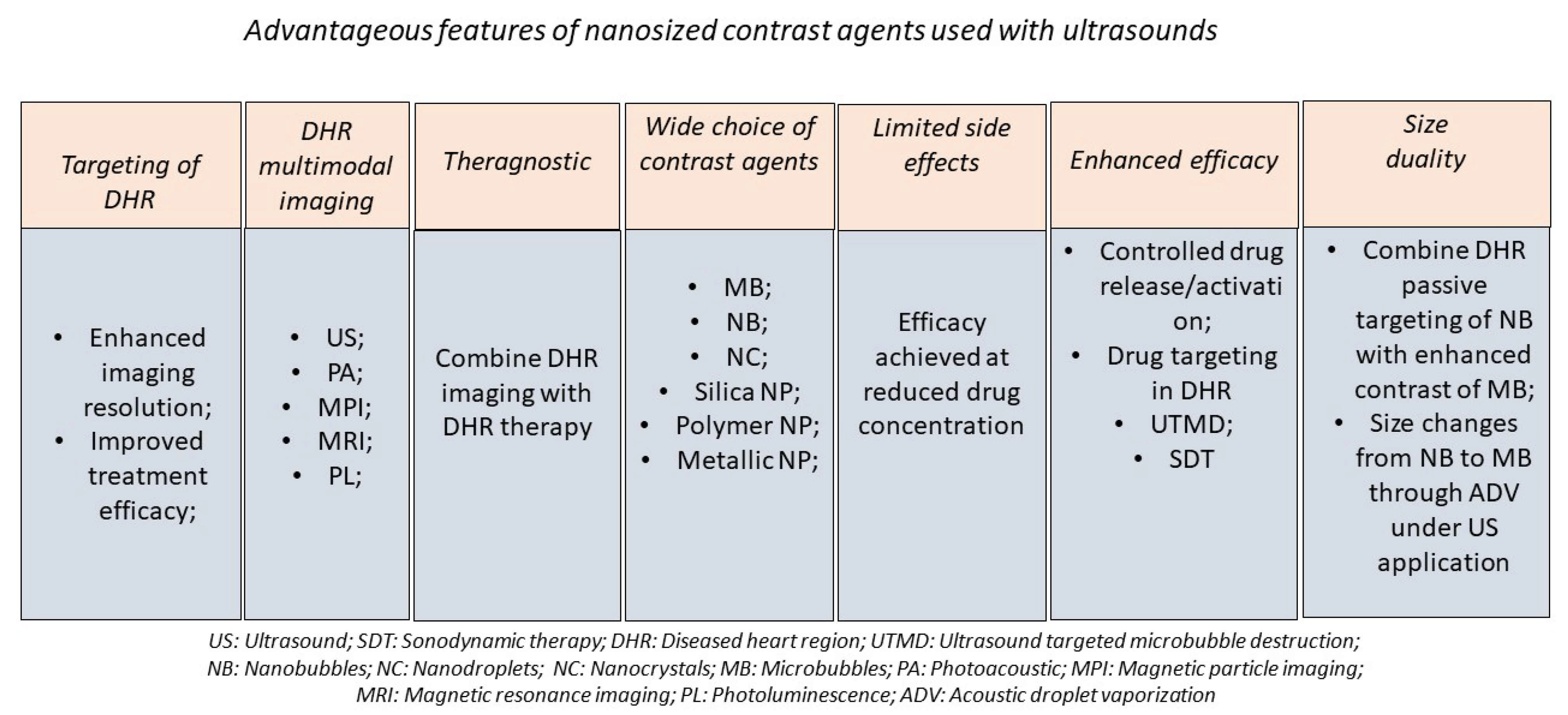
1. Introduction
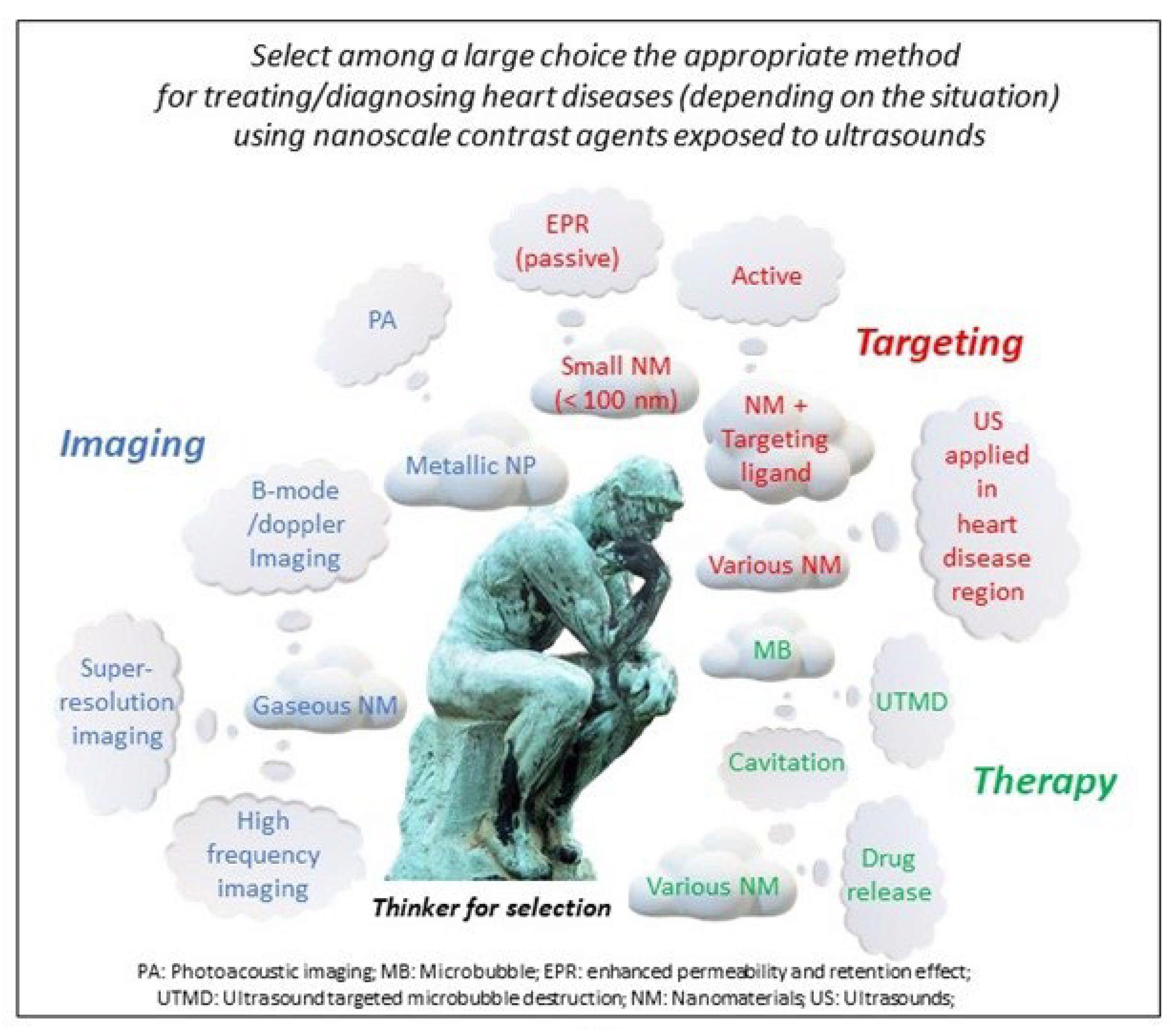
2. Various Types of Nanomaterials Used as Theragnostic Ultrasound Contrast Agents for the Treatment/Diagnosis of Heart Diseases:
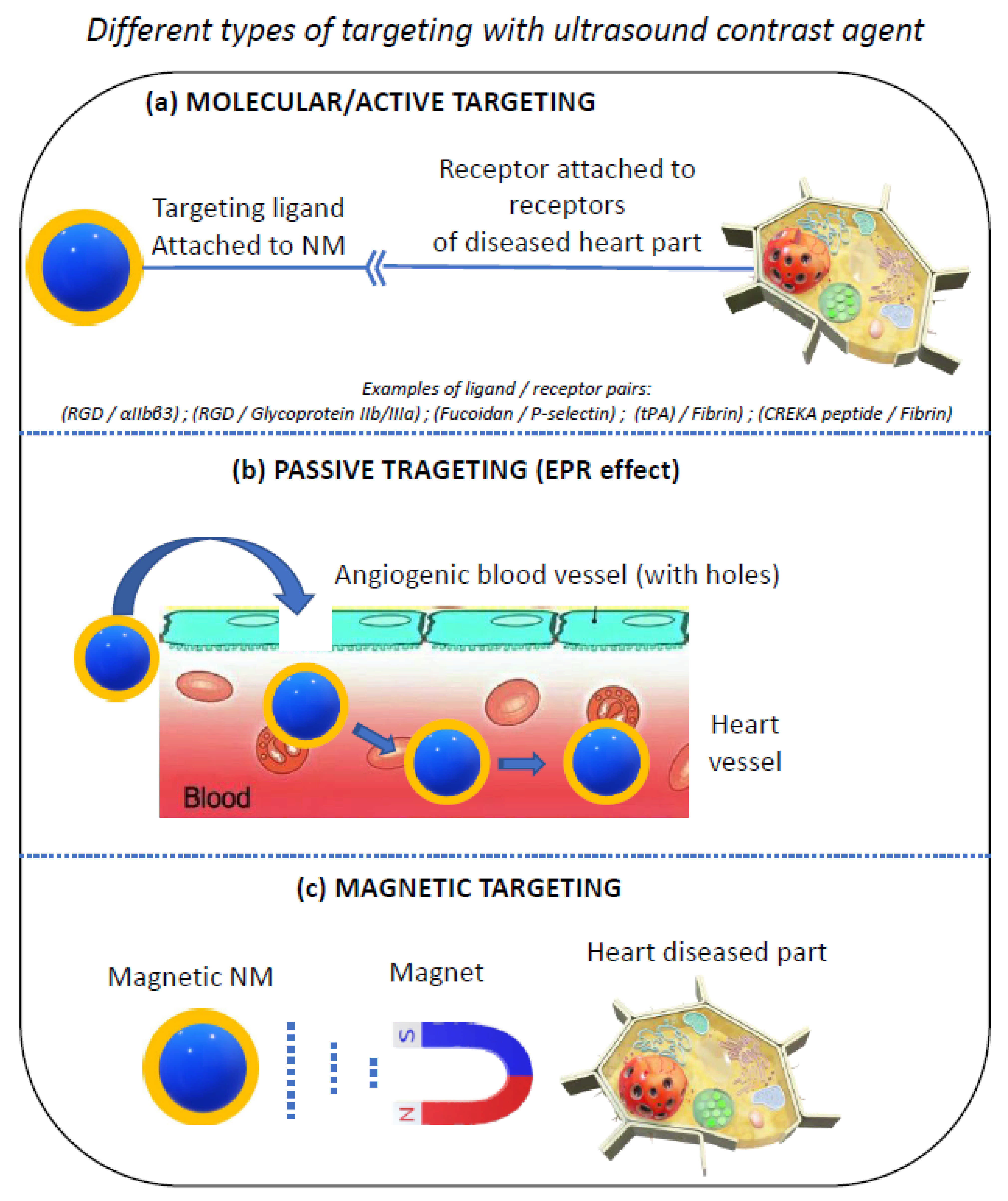
3. Nanoscale Ultrasound Contrast Agents for Targeting Specific Diseased Heart Regions
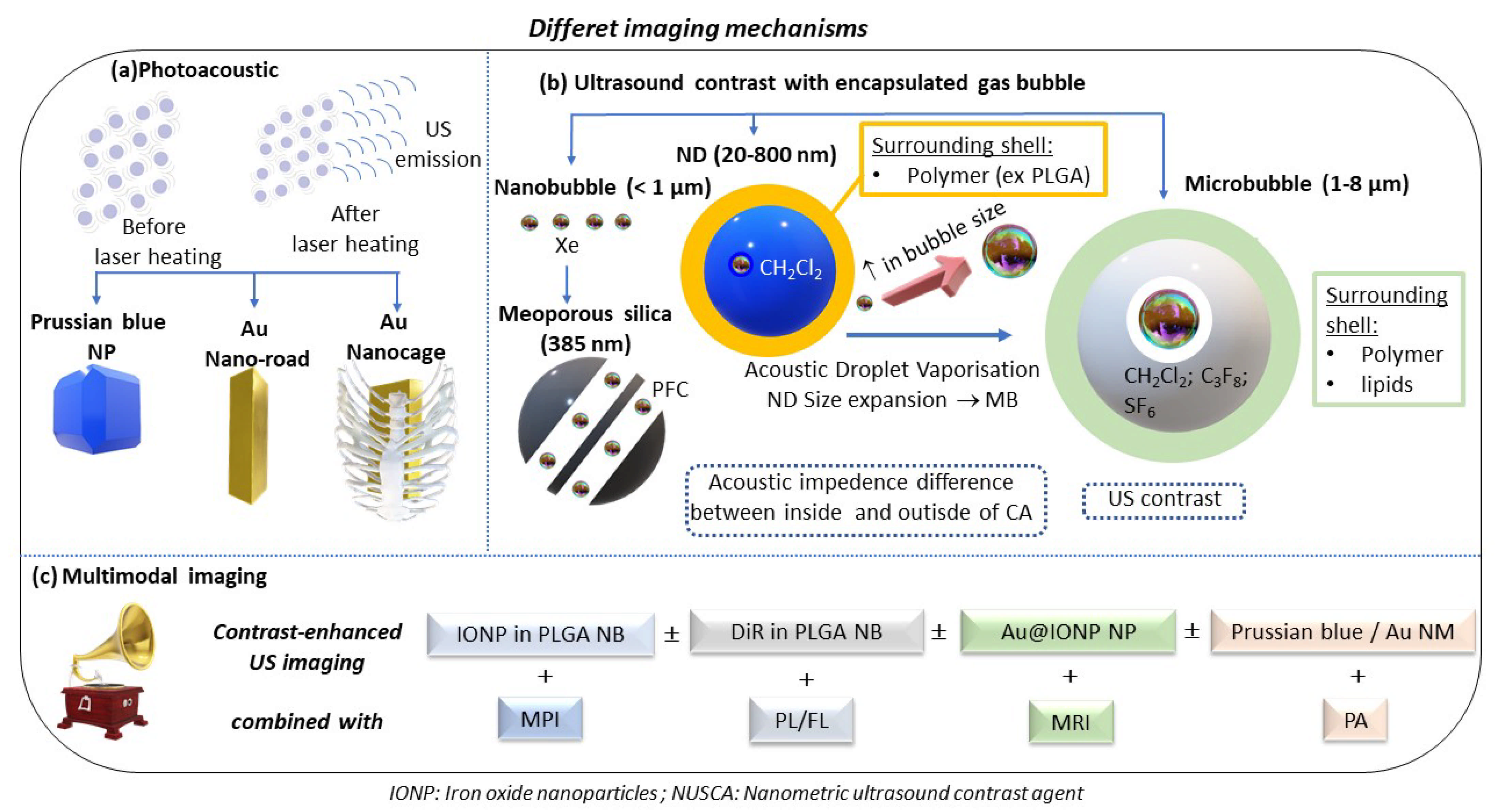
4. Improved Ultrasound Imaging with Nanoscale Contrast Agents
5. Photoacoustic Imaging to Detect Heart Abnormalities
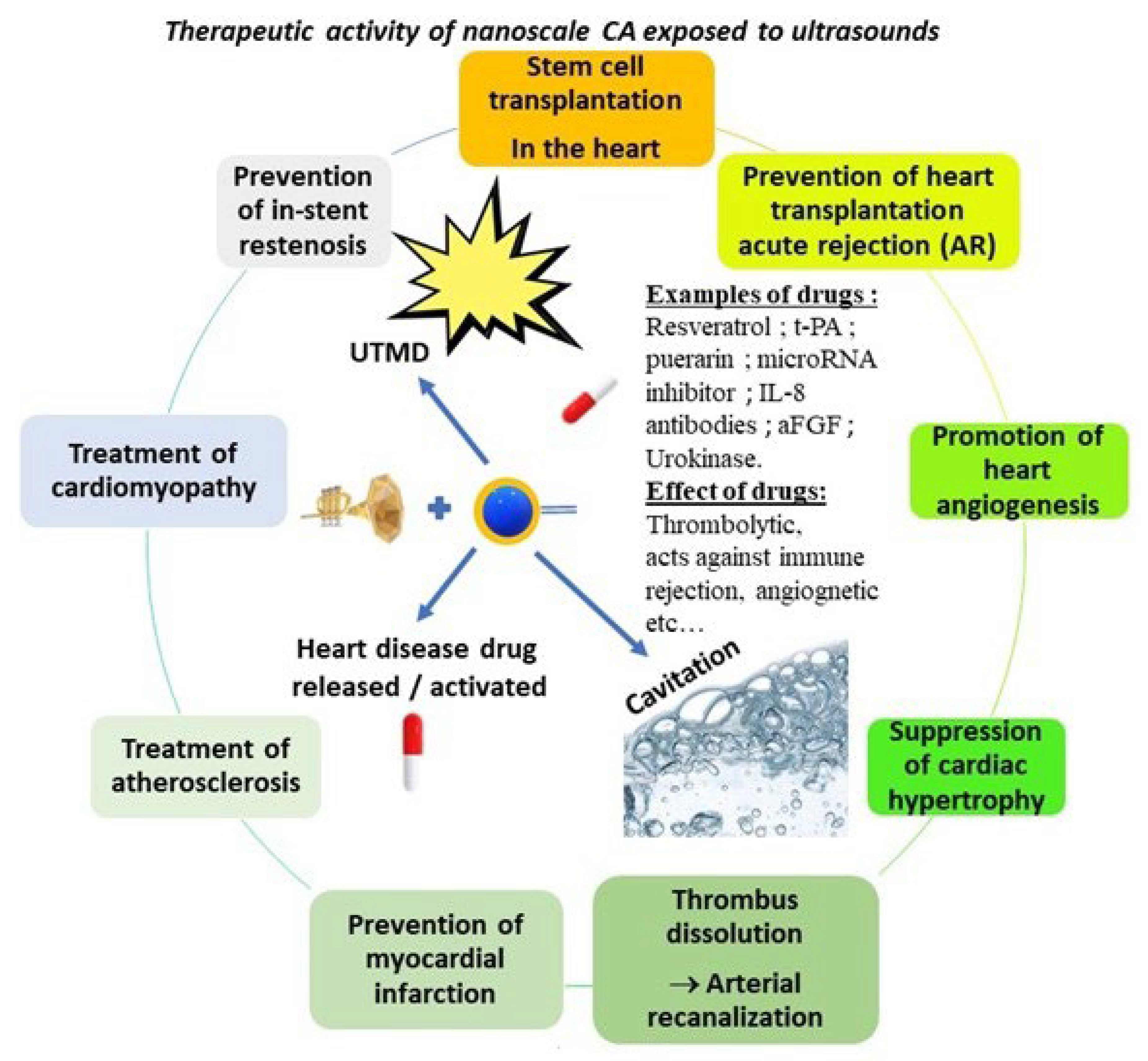
6. Therapeutic Activities of Nanoscale Contrast Agents Exposed to Ultrasound against Heart Diseases
7. The Various Heart Diseases that Can Be Detected/Treated By Using a Combination of Nanoscale Contrast Agents and Ultrasound
8. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Klibanov, A.L.; Hossack, J.A. Ultrasound in Radiology: From Anatomic, Functional, Molecular Imaging to Drug Delivery and Image-Guided Therapy. Investig. Radiol. 2015, 50, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, M.A. Ultrasound-guided Microbubble in the Treatment of Cancer: A Mini Narrative Review. Cureus 2018, 10, e3256. [Google Scholar] [CrossRef]
- Lee, H.; Kim, H.; Han, H.; Lee, M.; Lee, S.; Yoo, H.; Chang, J.H.; Kim, H. Microbubbles used for contrast enhanced ultrasound and theragnosis: A review of principles to applications. Biomed. Eng. Lett. 2017, 7, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Köse, G.; Darguzyte, M.; Kiessling, F. Molecular Ultrasound Imaging. Nanomaterials 2020, 10, 1935. [Google Scholar] [CrossRef] [PubMed]
- Lemaster, J.E.; Jokerst, J.V. What is new in nanoparticle-based photoacoustic imaging? Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1404. [Google Scholar] [CrossRef]
- Duan, L.; Yang, L.; Jin, J.; Yang, F.; Liu, D.; Hu, K.; Wang, Q.; Yue, Y.; Gu, N. Micro/nano-bubble-assisted ultrasound to enhance the EPR effect and potential theranostic applications. Theranostics 2020, 10, 462–483. [Google Scholar] [CrossRef]
- Zhou, Y. Application of acoustic droplet vaporization in ultrasound therapy. J. Ther. Ultrasound 2015, 3, 20. [Google Scholar] [CrossRef]
- Andrews, L.E.; Chan, M.H.; Liu, R.S. Nano-lipospheres as acoustically active ultrasound contrast agents: Evolving tumor imaging and therapy technique. Nanotechnology 2019, 30, 182001. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, Y.; Yu, T.; Guo, Y.; Liu, F.; Yao, Y.; Li, P.; Wang, D.; Wang, Z.; Chen, Y.; et al. Drug Release from Phase-Changeable Nanodroplets Triggered by Low-Intensity Focused Ultrasound. Theranostics 2018, 8, 1327–1339. [Google Scholar] [CrossRef]
- Ji, J.; Ji, S.Y.; Yang, J.A.; He, X.; Yang, X.H.; Ling, W.P.; Chen, X.L. Ultrasound-targeted transfection of tissue-type plasminogen activator gene carried by albumin nanoparticles to dog myocardium to prevent thrombosis after heart mechanical valve replacement. Int. J. Nanomed. 2012, 7, 2911–2919. [Google Scholar] [CrossRef]
- Tian, X.Q.; Ni, X.W.; Xu, H.L.; Zheng, L.; ZhuGe, D.L.; Chen, B.; Lu, C.T.; Yuan, J.J.; Zhao, Y.Z. Prevention of doxorubicin-induced cardiomyopathy using targeted MaFGF mediated by nanoparticles combined with ultrasound-targeted MB destruction. Int. J. Nanomed. 2017, 12, 7103–7119. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Guo, X.; Ren, L.; Wang, B.; Hou, L.; Zhou, H.; Gao, Q.; Gao, Y.; Wang, L. Targeting and deep-penetrating delivery strategy for stented coronary artery by magnetic guidance and ultrasound stimulation. Ultrason.-Sonochem. 2020, 67, 105188. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Aid-Launais, R.; Labour, M.N.; Zenych, A.; Juenet, M.; Choqueux, C.; Ollivier, V.; Couture, O.; Letourneur, D.; Chauvierre, C. Functionalized polymer microbubbles as new molecular ultrasound contrast agent to target P-selectin in thrombus. Biomaterials 2019, 194, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Jin, L.; Feng, L.; Wang, Y.; Yang, R. ICAM-1-carrying targeted nano contrast agent for evaluating inflammatory injury in rabbits with atherosclerosis. Sci. Rep. 2021, 11, 16508. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhao, P.; Zhou, Y.; Xing, C.; Zhao, L.; Li, Z.; Yuan, L. Ultrasound targeted microbubble destruction assisted exosomal delivery of miR-21 protects the heart from chemotherapy associated cardiotoxicity. Biochem. Biophys. Res. Commun. 2020, 532, 60–67. [Google Scholar] [CrossRef]
- Sun, Z.; Xie, Y.; Lee, R.J.; Chen, Y.; Jin, Q.; Lv, Q.; Wang, J.; Yang, Y.; Li, Y.; Cai, Y.; et al. Myocardium-targeted transplantation of PHD shRNA-modified bone mesenchymal stem cells through ultrasound-targeted microbubble destruction protects the heart from acute myocardial infarction. Theranostics 2020, 10, 4967–4982. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, S.; Li, S.; Yu, W.; Chen, J.; Yu, D.; Zhao, C.; Li, Y.; Kang, K.; Wang, R.; et al. Targeted galectin-7 inhibition with ultrasound microbubble targeted gene therapy as a sole therapy to prevent acute rejection following heart transplantation in a Rodent model. Biomaterials 2020, 263, 120366. [Google Scholar] [CrossRef]
- Zhou, N.Q.; Fang, Z.X.; Huang, N.; Zuo, Y.; Qiu, Y.; Guo, L.J.; Song, P.; Xu, J.; Wan, G.R.; Tian, X.Q.; et al. aFGF Targeted Mediated by Novel Nanoparticles-Microbubble Complex Combined With Ultrasound-Targeted Microbubble Destruction attenuates Doxorubicin-Induced Heart Failure via Anti-Apoptosis and Promoting Cardiac Angiogenesis. Front. Pharmacol. 2021, 12, 607785. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, M.; Zhang, Y.; Zhang, J.; Su, J.; Yan, C. Molecular imaging of atherosclerotic plaque with lipid nanobubbles as targeted ultrasound contrast agents. Colloids Surf. B Biointerfaces 2020, 189, 110861. [Google Scholar] [CrossRef]
- Zhu, T.; Zhang, Y.; Tang, J.; Tang, N.; He, Y.; Chen, X.; Gao, S.; Xu, Y.; Liu, Z. Ultrasound-targeted microbubble destruction accelerates angiogenesis and ameliorates left ventricular dysfunction aftermyocardial infarction in mice. Ultrasound Med. Biol. 2021, 47, 2692–2701. [Google Scholar] [CrossRef]
- Liu, J.; Chen, Y.; Wang, G.; Lv, Q.; Yang, Y.; Wang, J.; Zhang, P.; Liu, J.; Xie, Y.; Zhang, L.; et al. Ultrasound molecular imaging of acute cardiac transplantation rejection using nanobubbles targeted to T lymphocytes. Biomaterials 2018, 162, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Lemaster, J.E.; Chen, F.; Kim, T.; Hariri, A.; Jokerst, J.V. Development of a Trimodal Contrast Agent for Acoustic and Magnetic Particle Imaging of Stem Cells. ACS Appl. Nano Mater. 2018, 1, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Li, M.; Li, J.; Li, B.; Duan, L.; Yang, F.; Gu, N. Xenon Nanobubbles for the Image-Guided Preemptive Treatment of Acute Ischemic Stroke via Neuroprotection and Microcirculatory Restoration. ACS Appl. Mater. Interfaces 2021, 13, 43880–43891. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Xie, Y.; Chen, Y.; Deng, C.; Fang, L.; Li, H.; Sun, Z.; Gao, T.; Liu, J.; Yuan, J.; et al. Ultrasound Imaging of Treatment-Needed Cardiac Rejection with CD4-Targeted Nanobubbles. Part. Part. Syst. Charact. 2021, 38, 2100091. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Zhang, H.; Zhang, L.; Liu, L.; Cao, Y.; Ran, H.; Tian, J. A non-invasive nanoparticles for multimodal imaging of ischemic myocardium in rats. J. Nanobiotechnol. 2021, 19, 82. [Google Scholar] [CrossRef]
- Chen, H.; Chen, L.; Liang, R.; Wei, J. Ultrasound and magnetic resonance molecular imaging of atherosclerotic neovasculature with perfluorocarbon magnetic nanocapsules targeted against vascular endothelial growth factor receptor 2 in rats. Mol. Med. Rep. 2017, 16, 5986–5996. [Google Scholar] [CrossRef]
- Li, S.; Gou, T.; Wang, Q.; Chen, M.; Chen, Z.; Xu, M.; Wang, Y.; Han, D.; Cao, R.; Liu, J.; et al. Ultrasound/Optical Dual-Modality Imaging for Evaluation of Vulnerable Atherosclerotic Plaques with Osteopontin Targeted Nanoparticles. Macromol. Biosci. 2020, 20, 1900279. [Google Scholar] [CrossRef]
- Vannozzi, L.; Ricotti, L.; Filippeschi, C.; Sartini, S.; Coviello, V.; Piazza, V.; Pingue, P.; La Motta, C.; Dario, P.; Menciassi, A. Nanostructured ultra-thin patches for ultrasound-modulated delivery of anti-restenotic drug. Int. J. Nanomed. 2016, 11, 69–92. [Google Scholar] [CrossRef]
- Gao, J.; Liu, J.; Meng, Z.; Li, Y.; Hong, Y.; Wang, L.; He, L.; Hu, B.; Zheng, Y.; Li, T.; et al. Ultrasound-assisted C 3 F 8 -filled PLGA nanobubbles for enhanced FGF21 delivery and improved prophylactic treatment of diabetic cardiomyopathy. Acta Biomater. 2021, 130, 395–408. [Google Scholar] [CrossRef]
- Zhong, Y.; Zhang, Y.; Xu, J.; Zhou, J.; Liu, J.; Ye, M.; Zhang, L.; Qiao, B.; Wang, Z.G.; Ran, H.T.; et al. Low-Intensity Focused Ultrasound-Responsive Phase-Transitional Nanoparticles for Thrombolysis without Vascular Damage: A Synergistic Nonpharmaceutical Strategy. ACS Nano 2019, 13, 3387–3403. [Google Scholar] [CrossRef]
- Zhou, Q.; Zeng, Y.; Xiong, Q.; Zhong, S.; Li, P.; Ran, H.; Yin, Y.; Reutelingsperger, C.; Prinze, F.W.; Ling, Z. Construction of CNA35 Collagen-Targeted Phase-Changeable Nanoagents for Low-Intensity Focused Ultrasound-Triggered Ultrasound Molecular Imaging of Myocardial Fibrosis in Rabbits. ACS Appl. Mater. Interfaces 2019, 11, 23006–23017. [Google Scholar] [CrossRef] [PubMed]
- Masuda, S.; Nakano, K.; Funakoshi, K.; Zhao, G.; Meng, W.; Kimura, S.; Matoba, T.; Miyagawa, M.; Iwata, E.; Sunagawa, K.; et al. Imatini Mesylate-Incorporated Nanoparticle-Eluting Stent Attenuates In-Stent Neointimal Formation in Porcine Coronary Arteries. J. Atheroscler. Thromb. 2011, 18, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Du, W.; Lyu, N.; Chen, X.; Du, Y.; Wang, H.; Yang, D.; Wu, S.; Liang, J.; Pan, Y.; et al. Ultra-Early Diagnosis of Acute Myocardial Infarction in Rats Using Ultrasound Imaging of Hollow Double-Layer Silica Nanospheres. Adv. Healthc. Mater. 2020, 9, 1901155. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Wang, L.; Chi, X.; Bao, J.; Yang, L.; Zhao, W.; Chen, Z.; Wang, X.; Chen, X.; Gao, J. Engineered iron-oxide-based nanoparticles as enhanced T1 contrast agents for efficient tumor imaging. ACS Nano 2013, 23, 3287–3296. [Google Scholar] [CrossRef] [PubMed]
- Jokerst, J.V.; Khademi, C.; Gambhir, C.C. Intracellular Aggregation of Multimodal Silica Nanoparticles for Ultrasound-Guided Stem Cell Implantation. Sci. Transl. Med. 2013, 5, 177ra35. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Shen, C.L.; Li, J.M.; Ma, Y.L.; Yan, N.; Tian, X.Q.; Zhao, Y.Z. Assessment of the Preventive Effect Against Diabetic Cardiomyopathy of FGF1-Loaded Nanoliposomes Combined With Microbubble Cavitation by Ultrasound. Front. Pharmacol. 2020, 10, 1535. [Google Scholar] [CrossRef]
- Zhang, M.; Zhu, N.W.; Ma, W.C.; Chen, M.J.; Zheng, L. Combined treatment with ultrasound-targeted microbubble destruction technique and NM-aFGF-loaded PEG-nanoliposomes protects against diabetic cardiomyopathy-induced oxidative stress by activating the AKT/GSK-3b1/Nrf-2 pathway. Drug Deliv. 2020, 27, 938–952. [Google Scholar] [CrossRef]
- Jiang, T.; Hu, B.; Cao, S.; Gao, S.; Cao, Q.; Chen, J.; Zhou, Q.; Guo, R. Stable low-dose oxygen release using H2O2/perfluoropentane phase-change nanoparticles with low-intensity focused ultrasound for coronary thrombolysis. Ultrasound Med. Biol. 2020, 46, 2765–2774. [Google Scholar] [CrossRef]
- Kawata, H.; Uesugi, Y.; Soeda, T.; Takemoto, Y.; Sung, J.H.; Umaki, K.; Kato, K.; Ogiwara, K.; Nogami, K.; Ishigami, K.; et al. A New Drug Delivery System for Intravenous Coronary Thrombolysis With Thrombus Targeting and Stealth Activity Recoverable by Ultrasound. J. Am. Coll. Cardiol. 2012, 60, 2550–2557. [Google Scholar] [CrossRef]
- Zenych, A.; Fournier, L.; Chauvierre, C. Nanomedicine progress in thrombolytic therapy. Biomaterials 2020, 258, 120297. [Google Scholar] [CrossRef]
- Zhao, Y.Z.; Tian, X.Q.; Zhang, M.; Cai, L.; Rud, A.; Shen, X.T.; Jiang, X.; Jin, R.R.; Zheng, L.; Hawkins, K.; et al. Functional and pathological improvements of the hearts in diabetes model by the combined therapy of bFGF-loaded nanoparticles with ultrasound-targeted microbubble destruction. J. Control. Release 2014, 186, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Luo, W.; Hu, J.; Zuo, L.; Wang, J.; Hu, R.; Wang, B.; Xu, L.; Li, J.; Wu, M.; et al. Cardiomyocyte-targeted and 17β-estradiol-loaded acoustic nanoprobes as a theranostic platform for cardiac hypertrophy. J. Nanobiotechnol. 2018, 16, 36. [Google Scholar] [CrossRef]
- Moncion, A.; Lin, M.; O’Neill, E.G.; Franceschi, R.T.; Kripfgans, O.D.; Putnam, A.J.; Fabiilli, M.L. Controlled release of basic fibroblast growth factor for angiogenesis using acoustically-responsive scaffolds. Biomaterials 2017, 140, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Jaafar, F.; Leow, C.H.; Garbin, V.; Sennoga, C.A.; Tang, M.X.; Seddon, J.M. Surface charge measurement of sonovue, definity and optison: A comparison of laser doppler electrophoresis and micro-electrophoresis. Ultrasound Med. Biol. 2015, 41, 2990–3000. [Google Scholar] [CrossRef] [PubMed]
- Delalande, A.; Bastié, C.; Pigeon, L.; Manta, S.; Lebertre, M.; Mignet, N.; Midoux, P.; Pichon, C. Cationic gas-filled microbubbles for ultrasound-based nucleic acids delivery. Biosci. Rep. 2017, 37, BSR20160619. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, X.; Li, P.; Zhu, H.; Zhou, N.; Fang, Z.; Yang, Y.; Jing, Y.; Yuan, J. Ultrasound-targeted microbubble destruction promotes myocardial angiogenesis and functional improvements in rat model of diabetic cardiomyopathy. BMC Cardiovasc. Disord. 2021, 21, 21. [Google Scholar] [CrossRef]
- Stride, E. Physical Principles of Microbubbles for Ultrasound Imaging and Therapy. Cerebrovasc. Dis. 2009, 27 (Suppl. 2), 1–13. [Google Scholar] [CrossRef]
- Disharoon, D.; Marr, D.W.M.; Neeves, K.B. Engineered microparticles and nanoparticles for fibrinolysis. J. Thromb. Haemost. 2019, 17, 2004–2015. [Google Scholar] [CrossRef]
- Sennoga, C.A.; Yeh, J.S.M.; Alter, J.; Stride, E.; Nihoyannopoulos, P.; Seddon, J.M.; Haskard, D.O.; Hajnal, J.V.; Tang, M.X.; Eckersley, R.J. Evaluation of methods for sizing and counting of ultrasound contrast agents. Ultrasound Med. Biol. 2012, 38, 834–845. [Google Scholar] [CrossRef]
- Tay, L.M.; Xu, C. Coating microbubbles with nanoparticles for medical imaging and drug delivery. Nanomedicine 2017, 12, 91–94. [Google Scholar] [CrossRef]
- Kee, A.L.Y.; Teo, B.M. Biomedical applications of acoustically responsive phase shift nanodroplets: Current status and future directions. Ultrason. Sonochem. 2019, 56, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Chen, L.; Fang, Y.; Ma, S. Downregulation of Gsk-3b expression via ultrasound-targeted microbubble destruction enhances atherosclerotic plaque stability in New Zealand rabbits. Ultrasound Med. Biol. 2021, 47, 710–722. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Deng, Q.; Zhou, Q.; Cao, S.; Jiang, N.; Wang, Y.; Chen, J.; Hu, B.; Tan, T. Enhancement of Angiogenesis by Ultrasound-Targeted Microbubble Destruction Combined with Nuclear Localization Signaling Peptides in Canine Myocardial Infarction. BioMed. Res. Int. 2017, 2017, 9390565. [Google Scholar] [CrossRef] [PubMed]
- Voorneveld, J.; Keijzer, L.B.H.; Strachinaru, M.; Bowen, D.J.; Mutluer, F.O.; van der Steen, A.F.W.; Cate, F.J.T.; de Jong, N.; Vos, H.J.; van den Bosch, A.E.; et al. Optimization of Microbubble Concentration and Acoustic Pressure for Left Ventricular High-Frame-Rate EchoPIV in Patients. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 2432–2443. [Google Scholar] [CrossRef] [PubMed]
- Mackman, N.; Bergmeier, W.; Stouffer, G.A.; Weitz, J.I. Therapeutic strategies for thrombosis: New targets and approaches. Nat. Rev. Drug Discov. 2020, 19, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Huang, H.; Chen, Y.; Zheng, Y. Current Strategies for Microbubble-Based Thrombus Targeting: Activation-Specific Epitopes and Small Molecular Ligands. Front. Bioeng. Biotechnol. 2021, 9, 699450. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, X.; Shen, H.; He, Q.; Wu, Z.; Liao, W.; Yuan, M. Application of the Nano-Drug Delivery System in Treatment of Cardiovascular Diseases. Front. Bioeng. Biotechnol. 2020, 7, 489. [Google Scholar] [CrossRef] [PubMed]
- Branquinho, R.T.; Roy, J.; Farah, C.; Garcia, G.M.; Aimond, F.; Le Guennec, J.Y.; Saude-Guimarães, D.A.; Grabe-Guimaraes, A.; Mosqueira, V.C.F.; de Lana, M.; et al. Biodegradable Polymeric Nanocapsules Prevent Cardiotoxicity of Anti-Trypanosomal Lychnopholide. Sci. Rep. 2017, 7, 44998. [Google Scholar] [CrossRef]
- Savojiac, H.; Hossein, M.; Naimeh, M.; Masood, R.; Toroghi, K.; Yan, E.; Yimu, W.; Anastasia, Z.; Samad, K.; Radisi, M.A. Cardiovascular disease models: A game changing paradigm in drug discovery and screening. Biomaterials 2019, 198, 3–26. [Google Scholar] [CrossRef]
- Chen, F.; Zhao, E.R.; Hableel, G.; Hu, T.; Kim, T.; Li, J.; Gonzalez-Pech, N.I.; Cheng, D.J.; Lemaster, J.E.; Xie, Y.; et al. Increasing the Efficacy of Stem Cell Therapy via Triple-Function Inorganic Nanoparticles. ACS Nano 2019, 13, 6605–6617. [Google Scholar] [CrossRef]
- Maeda, H.; Bharate, G.Y.; Daruwalla, J. Polymeric drugs for efficient tumor-targeted drug delivery based on EPR-effect. Eur. J. Pharm. Biopharm. 2009, 71, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Grishenkov, D.; Kari, L.; Brodin, L.A.; Brismar, T.B.; Paradossi, G. In vitro contrast enhanced ultrasound measurements of capillary microcirculation: Comparison between polymer-and phospholipid-shelled microbubbles. Ultrasonics 2011, 51, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Yeh, J.S.M.; Sennoga, C.A.; McConnell, E.; Eckersley, R.; Tang, M.X.; Nourshargh, S.; Seddon, J.M.; Haskard, D.O.; Nihoyannopoulos, P.A. Targeting Microbubble for Ultrasound Molecular Imaging. PLoS ONE 2015, 10, e0129681. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Weil, B.R.; Neelamegham, S. Selectins and Immune Cells in Acute Myocardial Infarction and Post-infarction Ventricular Remodeling: Pathophysiology and Novel Treatments. Front. Immunol. 2019, 10, 300. [Google Scholar] [CrossRef]
- Kopechek, J.A.; McTiernan, C.A.; Chen, X.; Zhu, J.; Mburu, M.; Feroze, R.; Whitehurst, D.A.; Lavery, L.; Cyriac, J.; Villanueva, F.S. Ultrasound and Microbubble-targeted Delivery of a microRNA Inhibitor to the Heart Suppresses Cardiac Hypertrophy and Preserves Cardiac Function. Theranostics 2019, 9, 7088–7098. [Google Scholar] [CrossRef]
- Wang, X.; Gkanatsas, Y.; Palasubramaniam, J.; Hohmann, J.D.; Chen, C.Y.; Lim, B.; Hagemeyer, C.E.; Peter, K. Thrombus-Targeted Theranostic Microbubbles: A New Technology towards Concurrent Rapid Ultrasound Diagnosis and Bleeding-free Fibrinolytic Treatment of Thrombosis. Theranostics 2016, 6, 726–738. [Google Scholar] [CrossRef]
- Gu, M.; Wang, X.; Toh, T.B.; Chow, E.K.H. Applications of stimuli-responsive nanoscale drug delivery systems in translational research. Drug Discov. Today 2018, 23, 1043–1052. [Google Scholar] [CrossRef]
- Kun, Z.; Ming, L.; Yan, S. Preparation of Magnetic Targeting Microbubble Ultrasound Contrast Material and Its Performance in Myocardial Infarction. Sci. Adv. Mater. 2019, 11, 1604–1613. [Google Scholar]
- Shah, B.N.; Chahal, N.S.; Kooner, J.S. Contrast-enhanced ultrasonography vs B-mode ultrasound for visualization of intima-media thickness and detection of plaques in human carotid arteries. Echocardiogr. J. Cardiovasc. Imaging Interv. 2017, 34, 723–730. [Google Scholar] [CrossRef]
- Şen, T.; Tüfekçioğlu, O.; Koza, Y. Mechanical index. Anatol. J. Cardiol. 2015, 15, 334–336. [Google Scholar] [CrossRef]
- Lucas, V.S.; Burk, R.S.; Creehan, S.; Jo Grap, M. Utility of High-Frequency Ultrasound: Moving Beyond the Surface to Detect Changes in Skin Integrity. Plast. Surg. Nurs. 2014, 34, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Dave, J.K.; Kulkarni, S.V.; Pangaonkar, P.P.; Stanczak, M.; McDonald, M.E.; Cohen, I.S.; Mehrotra, P.; Savage, M.P.; Walinsky, P.; Ruggiero, N.J.; et al. Noninvasive intracardiac pressure measurements using subharmonic aided pressure estimation: Proof of concept in humans. Ultrasound Med. Biol. 2017, 43, 2718–2724. [Google Scholar] [CrossRef] [PubMed]
- Mendizabal, M.; Goldberg, D.S.; Piñero, F.; Arufe, D.T.; de la Fuente, M.J.; Testa, P.; Coronel, M.; Baratta, S.; Podestá, L.G.; Fallon, M.B.; et al. Isolated Intrapulmonary Vascular Dilatations and the Risk of Developing Hepatopulmonary Syndrome in Liver Transplant Candidates. Ann. Hepatol. 2017, 16, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Goertz, D.E.; Cherin, E.; Needles, A.; Karshafian, R.; Brown, A.S.; Burns, P.N. High frequency nonlinear B-scan imaging of microbubble contrast agents. IEEE Trans. Ultrason. Ferroelectr. Freq. 2005, 52, 65–79. [Google Scholar] [CrossRef]
- Bezugly, A.; Rembielak, A. The use of high frequency skin ultrasound in non-melanoma skin cancer. J. Contemp. Brachyther. 2021, 13, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Izzetti, R.; Vitali, S.; Aringhieri, G.; Nisi, M.; Oranges, T.; Dini, V.; Ferro, F.; Baldini, C.; Romanelli, M.; Caramella, D.; et al. Ultra-High Frequency Ultrasound, A Promising Diagnostic Technique: Review of the Literature and Single-Center Experience. Can. Assoc. Radiol. J. 2021, 72, 418–431. [Google Scholar] [CrossRef] [PubMed]
- Foster, F.S.; Mehi, J.; Lukac, M.; Hirson, D.; White, C.; Chaggares, C.; Needles, A. A NEW 15–50 MHz, Array-based micro-ultrasound scanner for preclinical imaging. Ultrasound Med. Biol. 2009, 35, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Chen, P.Y.; Peng, P.H.; Lee, P.Y. 40 MHz high-frequency ultrafast ultrasound imaging. Med. Phys. 2017, 44, 2185–2195. [Google Scholar] [CrossRef]
- Ho-Chiang, C.; Huang, H.; Huang, C.C. High-frequency ultrasound deformation imaging for adult zebrafish during heart regeneration. Quant. Imaging Med. Surg. 2020, 10, 66–75. [Google Scholar] [CrossRef]
- Daeichin, V.; Rooij, T.V.; Skachkov, I.; Ergin, B.; Specht, P.A.C.; Lima, A.; Ince, C.; Bosch, J.G.; van der Steen, A.F.W.; de Jong, N.; et al. Microbubble Composition and Preparation for High-Frequency Contrast-Enhanced Ultrasound Imaging: In Vitro and In Vivo Evaluation. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2017, 64, 555–567. [Google Scholar] [CrossRef]
- Yu, J.; Lavery, L.; Kim, K. Super-resolution ultrasound imaging method for microvasculature in vivo with a high temporal accuracy. Sci. Rep. 2018, 8, 13918. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Zhu, R.; Song, J.; Yang, H.; Chen, X. Photoacoustic Imaging: Contrast Agents and Their Biomedical Applications. Adv. Mater. 2019, 31, 1805875. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, A.; Grajo, J.R.; Dhyani, M.; Anthony, B.W.; Samir, A.E. Principles of ultrasound elastography. Abdom. Radiol. 2018, 43, 773–785. [Google Scholar] [CrossRef]
- Fan, Q.; Cheng, K.; Hu, X.; Ma, X.; Zhang, R.; Yang, M.; Lu, X.; Xing, L.; Huang, W.; Gambhir, S.S.; et al. Transferring Biomarker into Molecular Probe: Melanin Nanoparticle as a Naturally Active Platform for Multimodality Imaging. J. Am. Chem. Soc. 2014, 136, 15185–15194. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Maslov, K.; Wang, L.V. Second-generation optical-resolution photoacoustic microscopy with improved sensitivity and speed. Opt. Lett. 2011, 36, 1134–1136. [Google Scholar] [CrossRef] [PubMed]
- Sangha, G.S.; Phillips, E.H.; Goergen, C.J. In vivo photoacoustic lipid imaging in mice using the second near-infrared window. Biomed. Opt. Express 2017, 8, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wang, P.; Wang, H.W.; Cheng, J.X. Mapping lipid and collagen by multispectral photoacoustic imaging of chemical bond vibration. J. Biomed. Opt. 2012, 17, 096010. [Google Scholar] [CrossRef]
- Jokerst, J.; Cole, A.J.; van de Sompel, D.; Gambhir, S.S. Gold Nanorods for Ovarian Cancer Detection with Photoacoustic Imaging and Resection Guidance via Raman Imaging in Living Mice. ACS Nano 2012, 6, 10366–10377. [Google Scholar] [CrossRef]
- Wei, C.W.; Lombardo, M.; Larson-Smith, K.; Pelivanov, I.; Perez, C.; Xia, J.; Matula, T.; Pozzo, D.; O’Donnell, M. Nonlinear contrast enhancement in photoacoustic molecular imaging with gold nanosphere encapsulated nanoemulsions. Appl. Phys. Lett. 2014, 104, 033701. [Google Scholar] [CrossRef]
- Wi, J.S.; Park, J.; Kang, H.; Jung, D.; Lee, S.W.; Lee, T.G. Stacked Gold Nanodisks for Bimodal Photoacoustic and Optical Coherence Imaging. ACS Nano 2017, 11, 6225–6232. [Google Scholar] [CrossRef]
- Lipka, J.; Semmler-Behnke, M.; Sperling, R.A.; Wenk, A.; Takenaka, S.; Schleh, C.; Kissel, T.; Parak, W.J.; Kreyling, W.G. Biodistribution of PEG-modified gold nanoparticles following intratracheal instillation and intravenous injection. Biomaterials 2010, 31, 6574–6581. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Tang, S.; Guo, Z.; Wang, X.; Mo, S.; Huang, X.; Liu, G.; Zheng, N. Core-shell Pd@Au nanoplates as theranostic agents for in-vivo photoacoustic imaging, CT imaging, and photothermal therapy. Adv. Mater. 2014, 26, 8210–8216. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Liu, L.; Cai, Z.; Xu, J.; Xu, Z.; Zhang, D.; Hu, X. Plasmonic nanoparticles tuned thermal sensitive photonic polymer for biomimetic chameleon. Sci. Rep. 2016, 6, 31328. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Chen, M.; Sun, X.; Rong, P.; Zheng, N.; Chen, X. Palladium nanosheets as highly stable and effective contrast agents for in vivo photoacoustic molecular imaging. Nanoscale 2014, 6, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Shen, S.; Liu, T.; Cheng, L.; Liu, Z. Two-dimensional TiS2 nanosheets for in vivo photoacoustic imaging and photothermal cancer therapy. Nanoscale 2015, 7, 6380–6387. [Google Scholar] [CrossRef]
- Liu, Z.; Tabakman, S.; Welsher, K.; Dai, H. Carbon Nanotubes in Biology and Medicine: In vitro and in vivo Detection, Imaging and Drug Delivery. Nano Res. 2009, 2, 85–120. [Google Scholar] [CrossRef]
- Kim, T.; Lemaster, J.E.; Chen, F.; Li, D.J.; Jokerst, J.V. Photoacoustic Imaging of Human Mesenchymal Stem Cells Labeled with Prussian Blue–Poly(L-lysine) Nanocomplexes. ACS Nano 2017, 11, 9022–9032. [Google Scholar] [CrossRef]
- Adjei, I.M.; Yang, H.; Plumton, G.; Maldonado-Camargo, L.; Dobson, J.; Rinaldi, C.; Jiang, H.; Sharma, B. Multifunctional nanoparticles for intracellular drug delivery and photoacoustic imaging of mesenchymal stem cells. Drug. Deliv. Transl. Res. 2019, 3, 652–666. [Google Scholar] [CrossRef]
- James, S.; Neuhaus, K.; Murphy, M.; Leahy, M. Contrast agents for photoacoustic imaging: A review of stem cell tracking. Stem Cell Res. Ther. 2021, 12, 511. [Google Scholar] [CrossRef]
- Qin, Z.; Li, Y.; Gu, N. Progress in Applications of Prussian Blue Nanoparticles in Biomedicine. Adv. Healthc. Mater. 2018, 10, 1800347. [Google Scholar] [CrossRef]
- Zhang, Y.S.; Wang, Y.; Wang, L.; Wang, Y.; Cai, X.; Zhang, C.; Wang, L.V.; Xia, Y. Labeling Human Mesenchymal Stem Cells with Gold Nanocages for in vitro and in vivo Tracking by Two-Photon Microscopy and Photoacoustic Microscopy. Theranostics 2013, 3, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Brüßler, J.; Strehlow, B.; Becker, A.; Schubert, R.; Schümmelfeder, J.; Nimsky, C.; Bakowsky, U. Nanoscaled ultrasound contrast agents for enhanced sonothrombolysis. Colloids Surf. B Biointerfaces 2018, 172, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Guan, L.; Wang, C.; Yan, X.; Liu, L.; Li, Y.; Mu, Y. A thrombolytic therapy using diagnostic ultrasound combined with RGDS-targeted microbubbles and urokinase in a rabbit model. Sci. Rep. 2020, 10, 12511. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Zhou, L.; Liu, P.; He, Y.; Tan, K.; Chen, Q.; Gao, Y.; Gao, Y. In vivo thrombolysis with targeted microbubbles loading tissue plasminogen activator in a rabbit femoral artery thrombus model. J. Thromb. Thrombol. 2014, 38, 57–64. [Google Scholar] [CrossRef]
- Kandadai, M.A.; Mukherjee, P.; Shekhar, H.; Shaw, G.J.; Papautsky, I.; Holland, C.K. Microfluidic manufacture of rt-PA—Loaded echogenic liposomes. Biomed. Microdevices 2016, 18, 48. [Google Scholar] [CrossRef]
- Correa-Paza, C.; Poupard, M.F.N.; Rodríguez-Pérez, M.; Taboa, P.; Iglesias-Rey, R.; Hervella, P.; Sobrino, T.; Vivien, D.; Castillo, J.; Del Pino, P.; et al. In vivo ultrasound-activated delivery of recombinant tissue plasminogen activator from the cavity of sub-micrometric capsules. J. Control. Release 2019, 308, 162–171. [Google Scholar] [CrossRef]
- Liu, S.; Feng, X.; Jin, R.; Li, G. Tissue plasminogen activator-based nanothrombolysis for ischemic stroke. Expert Opin. Drug Deliv. 2018, 15, 173–184. [Google Scholar] [CrossRef]
- De Saint Victor, M.; Barnsley, L.C.; Carugo, D.; Owen, J.; Coussios, C.C.; Stridetagedend, E. Sonothrombolysis with magnetically targeted microbubbles. Ultrasound Med. Biol. 2019, 45, 1151–1163. [Google Scholar] [CrossRef]
- Rengeng, L.; Qianyu, Z.; Yuehong, L.; Zhongzhong, P.; Libo, L. Sonodynamic therapy, a treatment developing from photodynamic therapy. Photodiagnosis Photodyn. Ther. 2017, 19, 159–166. [Google Scholar] [CrossRef]
- Geng, C.; Zhang, Y.; Hidru, T.H.; Zhi, L.; Tao, M.; Zou, L.; Chen, C.; Lia, H.; Liu, Y. Sonodynamic therapy: A potential treatment for Atherosclerosis. Life Sci. 2018, 207, 304–313. [Google Scholar] [CrossRef]
- Jintao, X.; Nanqian, Z.; Yuping, Y.; Yun, J.; Yue, Q.; Yanhua, L.; Junxiu, L.; Xinqiao, T.; Yaling, Y.; Peng, L. Puerarin-loaded ultrasound microbubble contrast agent used as sonodynamic therapy for diabetic cardiomyopathy rats. Colloids Surf. B Biointerfaces 2020, 190, 110887. [Google Scholar] [CrossRef] [PubMed]
- Gnecchi, M.; Danieli, P.; Cervio, E. Mesenchymal stem cell therapy for heart disease. Vasc. Pharmacol. 2012, 57, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wu, S.; Liu, Z.; Zhuo, Z.; Tan, K.; Xia, H.; Zhuo, L.; Deng, X.; Gao, Y.; Xu, Y. Ultrasound-Targeted Microbubble Destruction Improves the Migration and Homing of Mesenchymal Stem Cells after Myocardial Infarction by Upregulating SDF-1/CXCR4: A Pilot Study. Stem Cells Int. 2015, 2015, 691310. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Zheng, Y.; Zhang, W.; Tan, J.; Zheng, H. Ultrasound-targeted microbubble destruction-mediated Galectin-7-siRNA promotes the homing of bone marrow mesenchymal stem cells to alleviate acute myocardial infarction in rats. Int. J. Mol. Med. 2021, 47, 677–687. [Google Scholar] [CrossRef]
- Leong-Poi, H. Molecular imaging using contrast-enhanced ultrasound: Evaluation of angiogenesis and cell therapy. Cardiovascular Res. 2009, 84, 190–200. [Google Scholar] [CrossRef]
- Yao, M.; Ma, M.; Zhang, H.; Zhang, Y.; Wan, G.; Shen, J.; Chen, H.; Wu, R. Mesopore-Induced Aggregation of Cobalt Protoporphyrin for Photoacoustic Imaging and Antioxidant Protection of Stem Cells. Adv. Funct. Mater. 2018, 28, 1804497. [Google Scholar] [CrossRef]
- Oduk, Y.; Zhu, W.; Kannappan, R.; Zhao, M.; Borovjagin, A.V.; Oparil, S.; Zhang, J.J. VEGF nanoparticles repair the heart after myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2018, 314, H278–H284. [Google Scholar] [CrossRef]
- Weller, G.E.R.; Lu, E.; Csikari, M.M.; Klibanov, A.L.; Fischer, D.; Wagner, W.R.; Villanueva, F.S. Ultrasound Imaging of Acute Cardiac Transplant Rejection With Microbubbles Targeted to Intercellular Adhesion Molecule-1. Circulation 2003, 108, 218–224. [Google Scholar] [CrossRef]
- Punjabi, M.; Xu, L.; Ochoa-Espinos, A.; Kosarev, A.; Wolff, T.; Murtaja, A.; Broisat, A.; Devoogdt, N.; Kaufmann, B.A. Ultrasound Molecular Imaging of Atherosclerosis With Nanobodies. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 2520–2530. [Google Scholar] [CrossRef]
- Yuan, H.; Hu, H.; Sun, J.; Shi, M.; Yu, H.; Li, C.; Sun, Y.; Yang, Z.; Hoffman, R.M. Ultrasound microbubble delivery targeting intraplaque neovascularization inhibits atherosclerotic plaque in an apoe-deficient mouse model. In Vivo 2018, 32, 1025–1032. [Google Scholar] [CrossRef]
- Shen, M.; Wang, Y.; Hu, F.; Lv, L.; Chen, K.; Xing, G. Thrombolytic Agents: Nanocarriers in Targeted Release. Molecules 2021, 26, 6776. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Website. Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 1 December 2021).
- Johnson, T.; Zhao, L.; Manuel, G.; Taylor, H.; Liu, D. Approaches to therapeutic angiogenesis for ischemic heart disease. J. Mol. Med. 2019, 97, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, J.; Navarro-Marquez, M.; Morales-Zavala, F.; Morales, J.O.; Garcia-Carvajal, I.; Araya-Fuentes, E.; Flores, Y.; Verdejo, H.E.; Castro, P.F.; Lavandero, S.; et al. Nanoparticles for diagnosis and therapy of atherosclerosis and myocardial infarction: Evolution toward prospective theranostic approaches. Theranostics 2018, 8, 4710–4732. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, C.; Liu, Q.; Xu, W.; Zhou, X. Molecular biomarkers in cardiac hypertrophy. J. Cell. Mol. Med. 2019, 23, 1671–1677. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Sogo, Y.; Wang, X.; Ito, A. Biosafety of mesoporous silica nanoparticles: A combined experimental and literature study. J. Mater. Sci. Mater. Med. 2021, 32, 102. [Google Scholar] [CrossRef]
- Guimaraes, D.; Cavaco-Paulo, A.; Nogueira, E. Design of liposomes as drug delivery system for therapeutic applications. Int. J. Pharm. 2021, 601, 120571. [Google Scholar] [CrossRef]
- Sonin, D.; Pochkaeva, E.; Zhuravskii, S.; Postnov, V.; Korolev, D.; Vasina, L.; Kostina, D.; Mukhametdinova, D.; Zelinskaya, I.; Skorik, Y.; et al. Biological Safety and Biodistribution of Chitosan Nanoparticles. Nanomaterials 2020, 10, 810. [Google Scholar] [CrossRef]
- Alphandéry, E. Biodistribution and targeting properties of iron oxide nanoparticles for treatments of cancer and iron anemia disease. Nanotoxicology 2019, 13, 573–596. [Google Scholar] [CrossRef]
| Nanosystem | Size (nm) | Origin of Echogenicity | US Parameters | Targeting/Activity | Imaging Results | Therapeutic Results | Applications | Ref |
|---|---|---|---|---|---|---|---|---|
| MICROBUBBLES (MB) | ||||||||
| nanopackaged tissue-type plasminogen activator (t-PA) gene plasmid cross-linked to ultrasonic MB composed of sucrose and bovine serum albumin | 2000 to 5000 | MB inner gas | Therapeutic ultrasound: f = 1 MHz; i = 1.5 W/cm2; t = 10 minutes; | t-PA: serine protease cleaving plasminogen into active plasmin for fibrin digestion in thrombus; | Following iv injection of MB/t-PA: Before therapeutic ultrasound applied: MB/t-PA observed by US in heart; After therapeutic ultrasound applied: MB/t-PA disappear from US imaging in heart; | Treatment steps:
| Avoiding thrombosis after mechanical heart valve replacement | [10] |
| MaFGF-loaded NP (MaFGF-NP) + MB | 132 | MB inner gas | f = 12–14 MHz; | None | None | (MaFGF-NP) + MB destruction: ↓ left ventricular dysfunction, myocardial fibrosis, cardiomyocytes apoptosis and oxidative stress. | Prevention of DOX-induced cardiomopathy | [11] |
| PTA-PTX NP encapsulated in shell of magnetic microbubble (MMB-PLGA- PTX) | 6000 | MB inner gas | f = 10–900 kHz; t = 4 sec.; | Drug targeting stent under magnetic field application; Drug released from stent under US application; | none | MMB-PLGA-PTX: magnetic targeting of stent; low intensity focused ultrasound (LIFU): production of stable microbubble oscillations → release of PLGA-PTX; | in-stent restenosis treatment | [12] |
| Polymer (PIBC) MB conjugated with fucoidan (Fucoidan-MB) | 2000 to 6000 | Air inside MB | f = 40 MHz; | Fucoidan target target P-selectin in thrombus | Fucoidan-MB: localize/image rat thrombotic wall; Fucoidan-MB: Not present in healthy vein; | None | Imaging of thrombus | [13] |
| nano lipid MB (DPPC + DSPE-PEG) conjugated to anti-ICAM-1 (MB-anti-ICAM-1) | 683 | C3F8 | f = 14 MHz | ICAM-1 (marker of atherosclerosis) | MB-anti-ICAM-1 located/imaged in vascular wall of abdominal aorta | None | identifyi inflammatory injury due to atherosclerosis | [14] |
| miRNA in exosomes + UTMD with MB (sonovue) | 30–150 | MB inner gas | f = 40 MHz (US imaging); f = 0.7 MHz t = 1 min (in target area for UTMD); | UTMD for targeted delivery of exosomes to the heart | none | Exosome + miRNA delivered in mouse heart using UTMD → restores cardiac function (following doxorubicin induced cardiotoxicity) | Protects the heart from chemotherapy related cardiotoxicity | [15] |
| MB+ bone marrow stem cell (BMSC) transduced with lentiviral PHD2 shRNA | 1000 | C3F8 | f = 10 MHz (echography) f = 1 MHz; i = 2 W/cm2; (UTMD) | UTMD | Imaging of MB in heart | Transplantion of PHD2 shRNA-modified BMSC in presence of UTMD:
| Improve stem cell therapy following acute myocardial infarction. | [16] |
| Galectin-7-siRNA (siGal-7) bound to cationic MB (CMB) | 424 (CMB) | C3F8 | f = 1 MHz; t = 2 min; i = 2 W/cm2; (UTMD) | siRNA stops translation of Gal-7 (Gal-7: responsible for rejection of grafted heart); | none | galectin-7-siRNA-CMB + UTMD: stops acute cellular rejection following allograft heart transplantation | acute cellular rejection (AR) after heart transplantation (HT) | [17] |
| Complex of: aFGF–NP + cationic lipid microbubbles (CPMB) | 4390 | SF6 gas | f = 4 MHz; Pulsed mode; (UTMD) | Through UTMD in heart aFGF: targets myocardial tissue + | M-mode echocardiography of heart with Nano-complex + UTMD | aFGF–NP + CPMB + UTMD prevents left ventricular dysfunction due to DOX. aFGF ↑ vascular endothelial/smooth muscle cells proliferation → treatment of HF; NP + aFGF: ↑ half–life, stability, efficacy of free aFGF; | Treatment of Doxorubicin-Induced Heart failure | [18] |
| DSPE-PEG2000 NB conjugated with anti-VEGF-2 (NB-VEGF2) | 320 | SF6 | NA | Active targeting: anti-VEGF-2: targeting angiogenesis (dominant in atherosclerosis) Passive targeting: EPR effect | US detection of rabbit abdominal aorta atherosclerotic plaquet using NB-VEGF2 as targeted contrast agent | None | Detection of atherosclerosis | [19] |
| NANOBUBBLES (NB) | ||||||||
| NB (Nanobubbles) + anti-CD25 antibody | 420 | Perfluoropropane | f = 7–14 MHz; | Anti-CD25 antibody targeting T cells in in myocardium | MCE + NB + antibody → imaging of acute rejection (AR) after heart transplantation | None | Detection of Acute rejection after heart transplantation | [20] |
| NB + anti-CD3 antibody (NB-CD3) | 460 | C3F8 inside NB | NA | NB-CD3 target T lymphocytes | US signal of NB-CD3 proportional to number of T lymphocytes | None | Detection of acute rejection detection after cardiac transplantation (through detection of T lymphocyte infiltration) | [21] |
| (PLGA)-IONP NB conjugated with (DiR) | 185 (IONP: 4) | NA | f = 21 MHz | None |
NB injected in myocardium: Signal of stem cells in cardiac tissue increased by 4 (US), 10 (PA), 20 (MPI). | None | Stem cell therapy | [22] |
| NB encapsulating Xe (Xe-NB) | 225 | Xe gas | f = 18 MHz; | accumulation or aggregation of NB to ischemic lesion | Xe-NB in ischemic lesion:
| Xe-NB therapeutic effects:
Restore neurological function; | Treatment of Acute Ischemic Stroke | [23] |
| NB conjugated with anti-CD4+ (NBCD4) | 545 | NA | f = 5.6 MHz; MI = 0.085; | NBCD4: Targeting of CD4+ activated by ACAR | US signal of NBCD4: ↑ in animals prone to ACAR due to infiltrating CD4+ lymphocytes; Detect the presence of ACAR | None | Acute cardiac allograft rejection (ACAR) after heart transplantation | [24] |
| NANODROPLETS (ND) | ||||||||
| Fe3O4 NP + PFH inside nano-capsule (DSPE-PEG2000-IMTP + DPPC + Cholesterol) | 348 | Gas (Perfluorohexane) Liquid vaporizes to gas phase under US activation (ADV); | f = 1 MHz; i = 1–4 W/cm2; t = 1–4 min; pulsed wave (low intensity ultrasound) | Targeting of ischemic region of myocardium:
Active targeting: Ischemic myocardium-targeted peptide (IMTP) |
Targeting of rat heart; | None | Targeting/imaging of ischemic/hypoxia injured heart cells | [25] |
| POLYMER NP | ||||||||
| Perfluorocarbon + SPIO in NP (polymer: PLA+PLGA-COOH+PFOB) + VEGFR-2 antibody | 404 | Gas (Perfluorocarbon) | MI=1.6; f = 15 MHz; | Endothelial VEGFR-2. |
| None | prediction of plaque vulnerability | [26] |
| NP (PLA) encapsulating PFOB surrounded by OPN + Cy5.5 | 360 | perfluorooctyl Bromide (PFOB) | f = 40 MHz (high frequency) | Osteopontin (OPN) to target VSMC | Bimodal imaging: ultrasound + optical Imaging of vascular smooth muscle cells (VSMC)
| None | Detection of Atherosclerotic Plaques | [27] |
| BaTiO3 NP + PLLA + PE + anti-restenotic drug sandwitched between PE/PLLA layers | 100 (BaTiO3) | None | i = 20 W; f = 40 kHz; t = 10 sec; | Drug released from PE → antiproliferative effect on human smooth muscle cells yielding restenosis. | None | Anti-restenotic drug released from nano-patches under ultrasound application | Anti-restenotic treatment | [28] |
| PLGA NB bound to FGF21 (NB-FGF21) | 880 | C3F8 gas | f = 40 MHz; (imaging) f = 500 kHz; i = 2 W; t = 5 min; (LFUS) | LFUS enables controlled release of FGF21 FGF21: ↓ hypertensive cardiac remodelling, ↓ cardiac hypertrophy, ↓ inflammation ↓ oxidative stress caused by DOX. | Echocardiography: increase of contrast of cardiac chamber between before and after NB-FGF21 injection; | LFUS on NB-FGF21:
↓ of myocardial hypertrophy, interstitial fibrosis in diabetic mice. | Treatment of diabetic cardiomyopathy | [29] |
| Fe3O4-poly(lactic-co-glycolic acid)-PFH-CREKA nanoparticles (NP) | 311 | PFH Perfluoro-hexane | i = 1 W/cm2; | CREKA peptide: targets fibrin of thrombus; | NP phase transition monitored by photoacoustic imaging; (Fe3O4 for MRI imaging) | NP phase transition (PT) under low-intensity focused ultrasound → vaporization of PFH to yield thrombolysis. | Treatment of thrombus | [30] |
| CNA35-PFP polymer NP (DPPC+DSPE+DSPG) | 295 | PFP Perfluoro-pentane (liquid−gas phase transition) | f = 5−9 MHz; i = 1−2 W/cm2; (Low Intensity Focused Ultrasound) | Passive targeting: CNA35-PFP NP diffuse through endothelial cell gap (EPR effect); Active targeting: CNA35-PFP NP target fibrosis in ischemic myocardium; → CNA35-PFP NP adhere to surface of fibroblasts in fibrotic myocardium; | iv injection of CNA35-PFP NP in animal model of myocardial infarction Followed by LIUF application: → Transform PFP from liquid to gaseous MB + ↑ US in fibrotic region; | None | Detection of Myocardial Fibrosis | [31] |
| Imatinib mesylate encapsulated in bioadsorbable polymeric NP | 200 | Contrast media: Iopamidol | f = 40 MHz; | Drug-eluting stents (DES) targeting vascular smooth cells | Ultrasound imaging to assess the extend of neointima formation | Imatinib-NP eluting stent: ↓ in-stent neointima + stenosis | Supression of neointima formation; Prevention of in-stent restenosis; | [32] |
| SILICA NP | ||||||||
| Silica nanosphere | 338 | perfluorodecyl Silane inside nanosphere | MI=1.3 | Active targeting with anti-Cardiac Troponin I Antibody at surface of nanosphere | delineation of myocardial necrosis sites | None | Early Diagnosis of Acute Myocardial Infarction | [33] |
| Fe3O4 + IGF in pores of mesocellular foam silica NP | 383 (16: pore) | mesocellular foam silica NP (MCS) | f = 40 MHz; | IGF: insulin-like growth factor → improve cell viability | Enhanced ultrasound signal in the presence of MCS | Release of IGF from nano-system: ↑ efficacy of stem cell therapy; | Stem cell therapy in heart disease | [34] |
| silica-based NP | 300 | Aggregated NP | F = 16–40 MHz; | None |
↑ resolution with US (down to 70 000 cells) than MRI (down to 250 000 cells) |
| Stem cell therapy in heart disease | [35] |
| NANOLIPOSOME | ||||||||
| FGF1-loaded nanoliposomes (FGF1-nlip) | 80 | NA | f = 12 to 14 MHz | Acidic fibroblast growth factor (FGF1) → prevents diabetic cardiomyopathy. |
|
| Treatment of diabetic cardiomyopathy | [36] |
| Non-mitogenic acidic fibroblast growth factor (NM-aFGF) in PEGylated nanoliposomes (NM-aFGF-PEG-lips) | 125 | None | f = 12–14 MHz; MI = 1.9; T = 10 sec; (MB destruction) | combination of NM-aFGFPEG- lips and UTMD could achieve cardiac-targeted delivery | None |
| Treat cardiac abnormailities due to diabete | [37] |
| OTHER TYPES OF NANOMATERIALS | ||||||||
| hydrogen peroxide (H2O2)/perfluoropentane (PFP) phase-change NP | 457 | Perfluoropentane | Therapeutic ultrasound: f = 1 MHz; i = 1–8 W; | None | US applied on H2O2/PFP NP → Acoustic signal
|
| Treatmment of coronary thrombolysis | [38] |
| NP with t-PA + gelatin + zinc ions | 100 | None | transthoracic US f = 1 MHz; i = 1 W/cm2; | t-PA for thrombus destruction | None |
| intracoronary thrombolysis | [39] |
| Basic fibroblast growth factor (bFGF) + NP (NP-bFGF) | 128 | MB | f = 14 MHz; MI = 1.9; t = 10 sec; (MB destruction) | Destruction of MB + NP in heart by ultrasound application | MB imaged to control MB destruction. |
| Treatment of Diabetic cardiomyopathy | [40] |
| albumin-bound particle form of paclitaxel (nab-PTX) | 130 | None | NA | PTX to reduce restenosis | Ultrasound used for placing stent in coronary lesion |
| reducing in-stent restenosis | [41] |
| Nano-probe encapsulating PFP and E2 conjugated with PCM; | 418 nm | PFP (Perfluoro-carbon) | f = 5–12 MHz; i = 3.2 W/cm2 t = 10 min (low-intensity focused ultrasound imaging/therapy) | Targeting: primary cardiomyocyte (PCM) targeting primary cardiomyocyte; Activity: 17β-estradiol (E2) as anti-hypertrophic drug; | PCM-E2/PFP: imaging contrast agent. | PCM-E2/PFP + LIFUS:
| Treatment of cardiac hypertrophy | [42] |
| Acoustically-responsive fibrin scaffold (ARS) containing basic fibroblast growth factor (bFGF) | 14,000 | PFH | F = 2.5 MHz; Pressure = 2 Mpa; | bFGF: pro-angiogenic growth factor to stimulate blood vessel formation and restore perfusion; | Imaging of gas bubbles generated by ADV (pressure > 2 Mpa) in ARS. | Release from ARS of bFGF under ultrasound application: perfusion and blood vessel density; | treatment of vascular disease | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alphandéry, E. Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis. Int. J. Mol. Sci. 2022, 23, 1683. https://doi.org/10.3390/ijms23031683
Alphandéry E. Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis. International Journal of Molecular Sciences. 2022; 23(3):1683. https://doi.org/10.3390/ijms23031683
Chicago/Turabian StyleAlphandéry, Edouard. 2022. "Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis" International Journal of Molecular Sciences 23, no. 3: 1683. https://doi.org/10.3390/ijms23031683
APA StyleAlphandéry, E. (2022). Nanomaterials as Ultrasound Theragnostic Tools for Heart Disease Treatment/Diagnosis. International Journal of Molecular Sciences, 23(3), 1683. https://doi.org/10.3390/ijms23031683






