Exosomal MALAT1 Derived from High Glucose-Treated Macrophages Up-Regulates Resistin Expression via miR-150-5p Downregulation
Abstract
:1. Introduction
2. Results
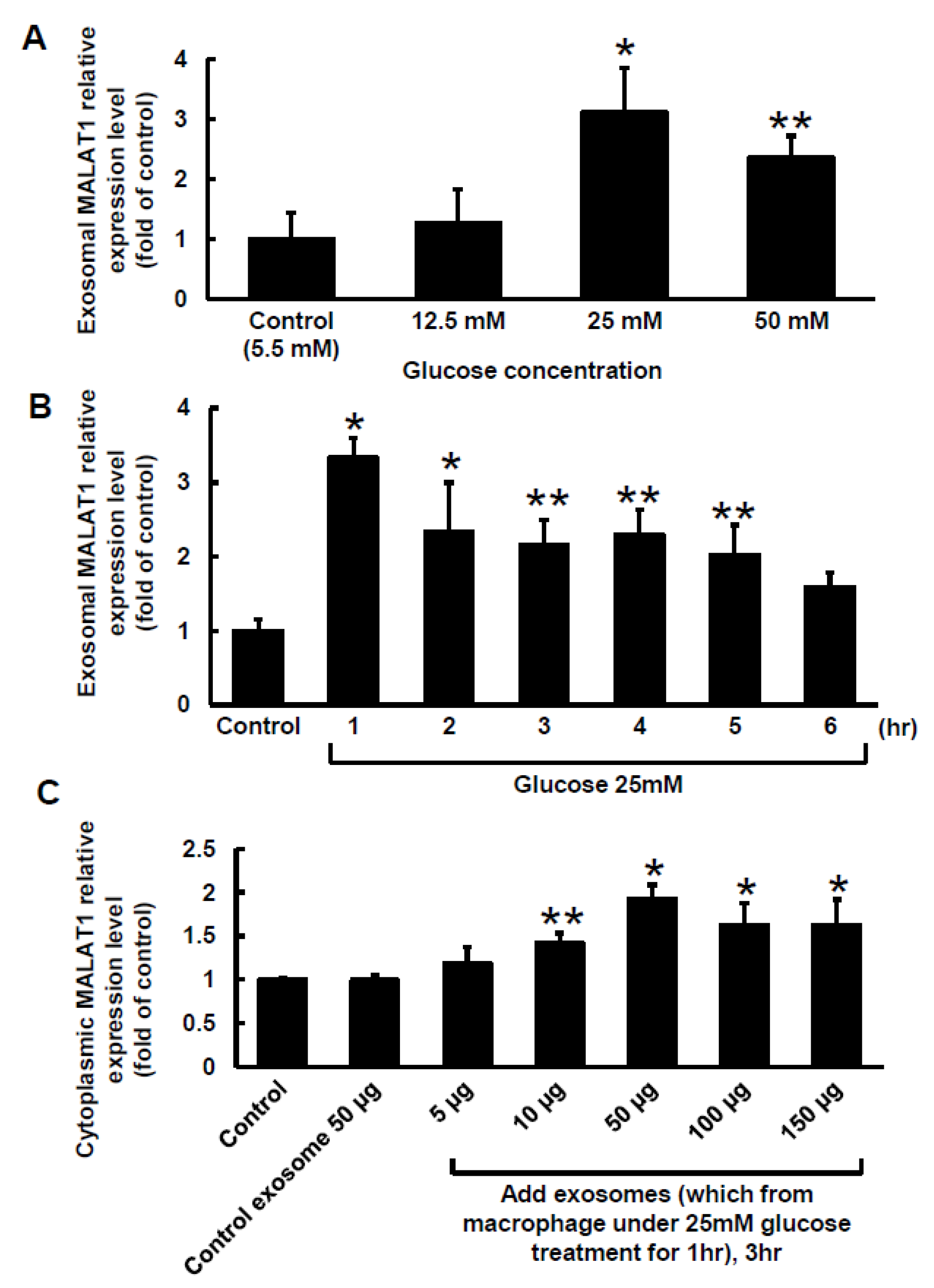
2.1. High Glucose Induced MALAT1 Expression in Macrophage-Derived Exosomes
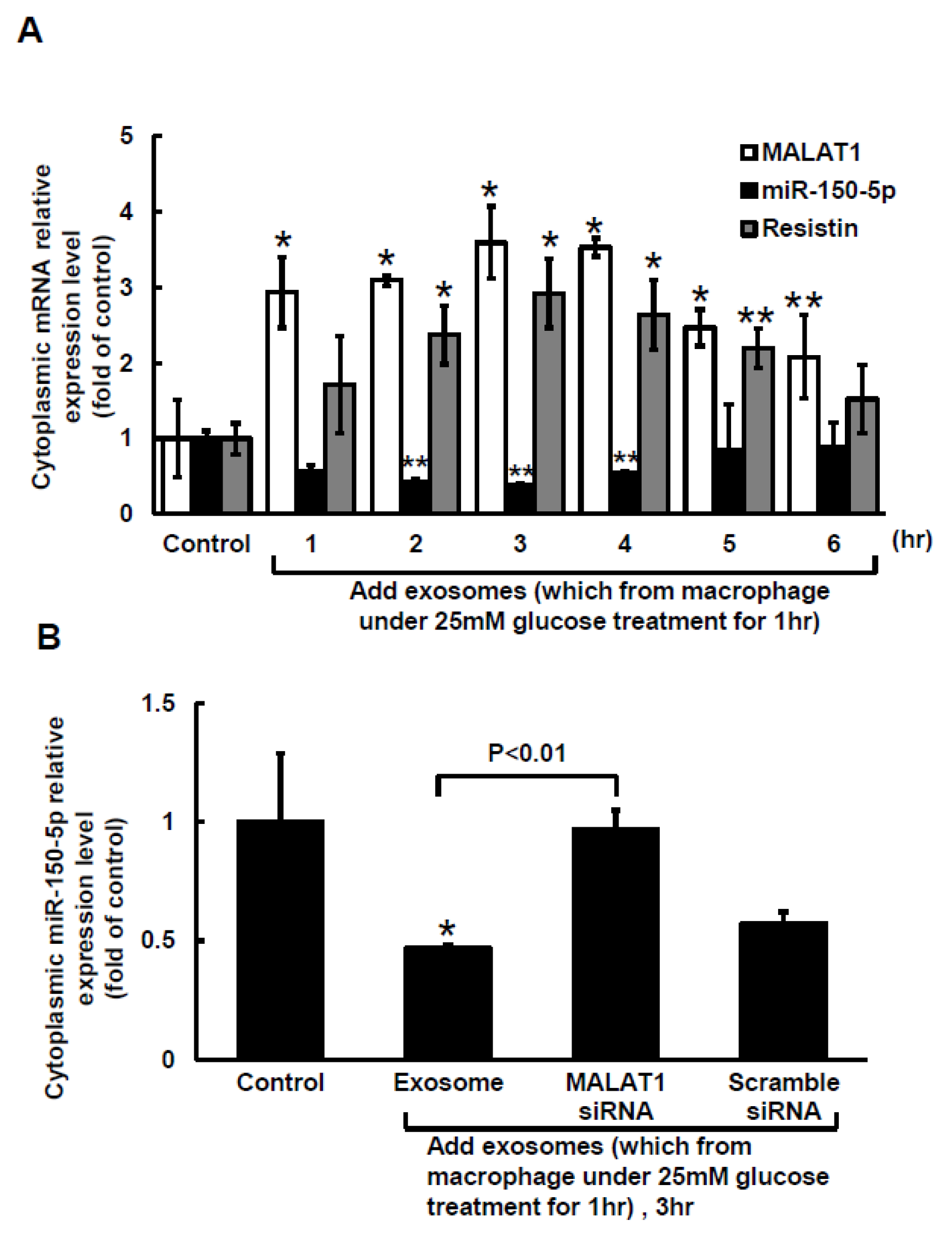
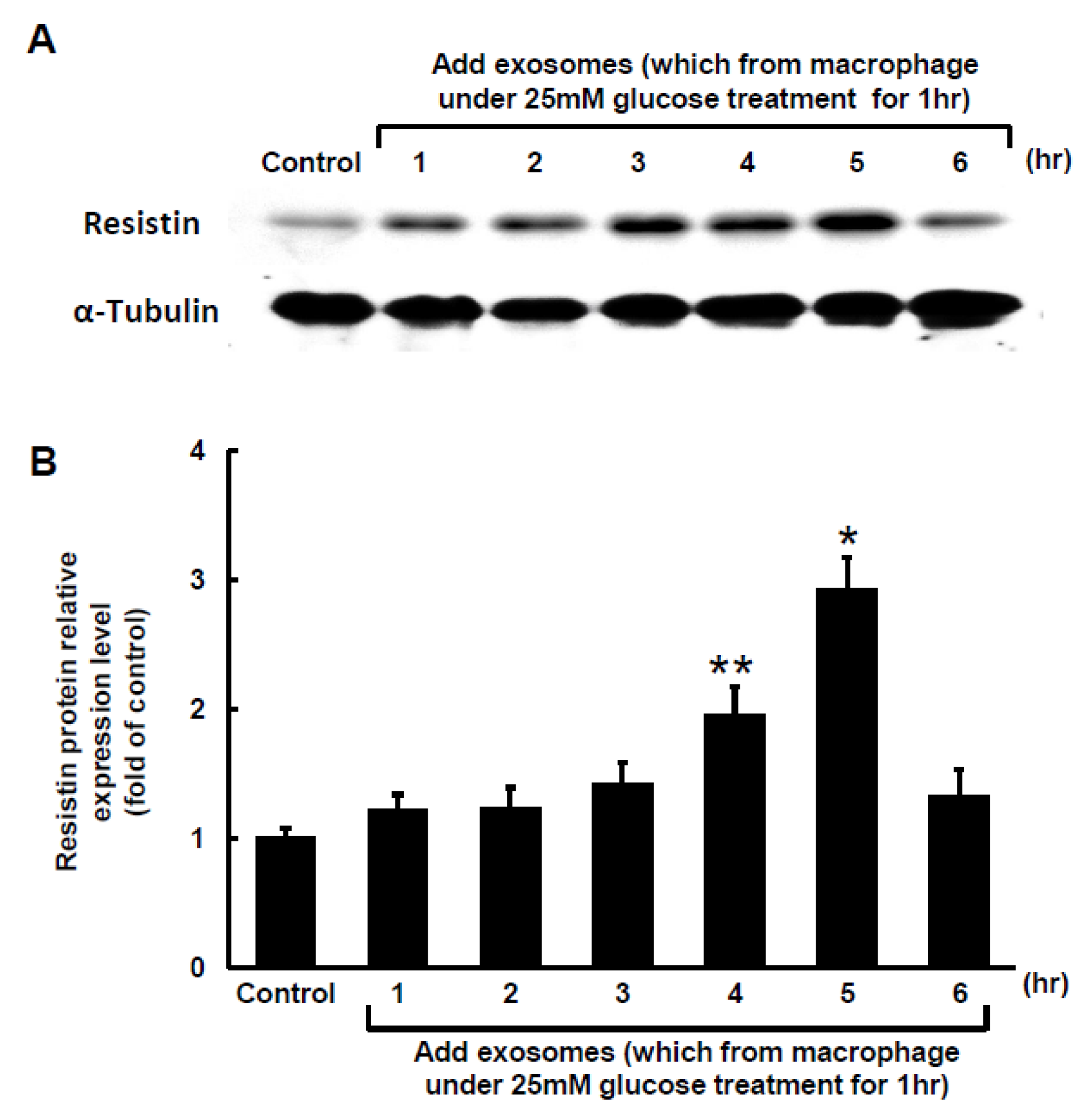
2.2. Macrophage-Derived Exosomes Decreased miR-150-5p and Increased Resistin Expressions in Macrophages
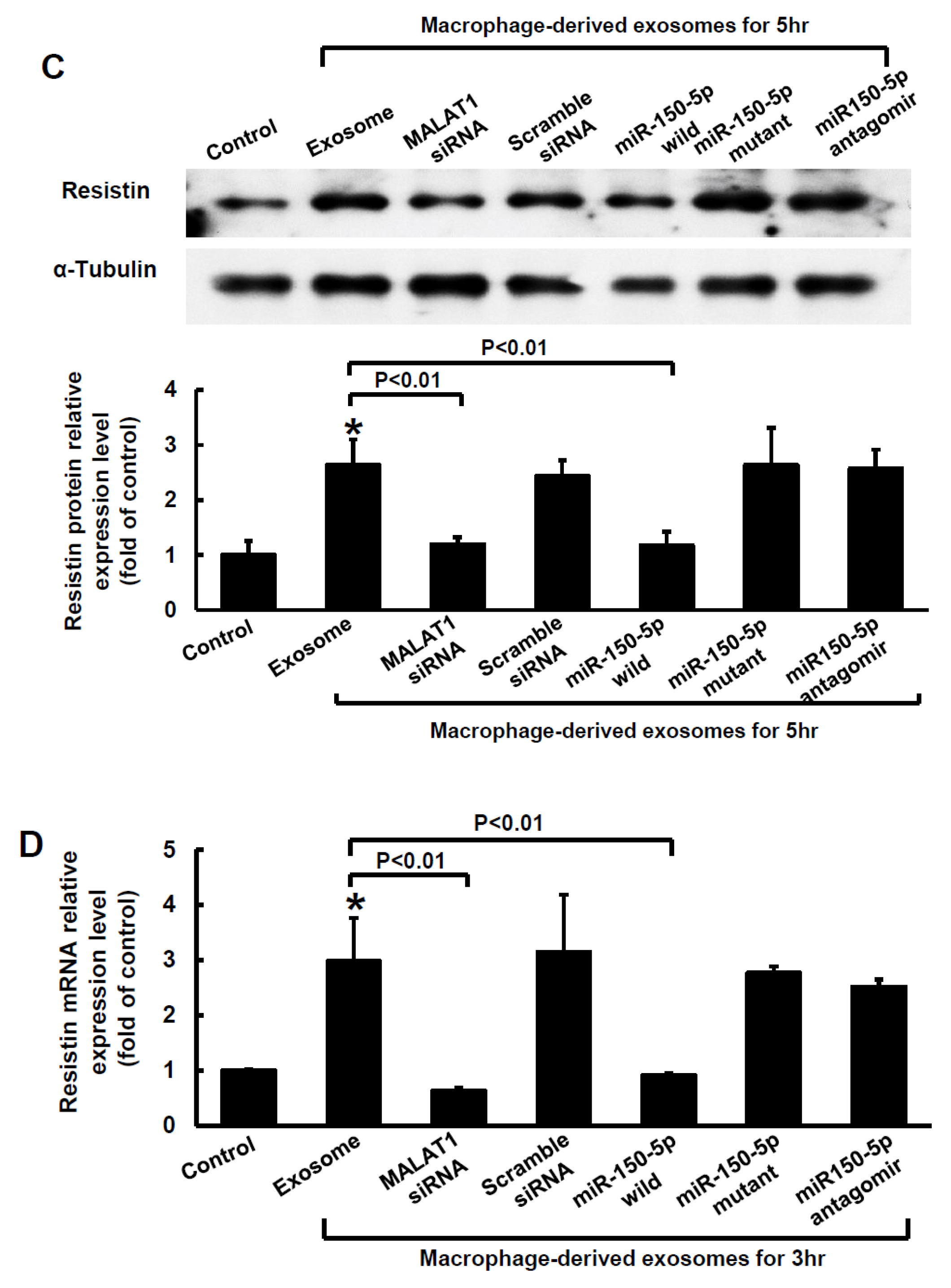
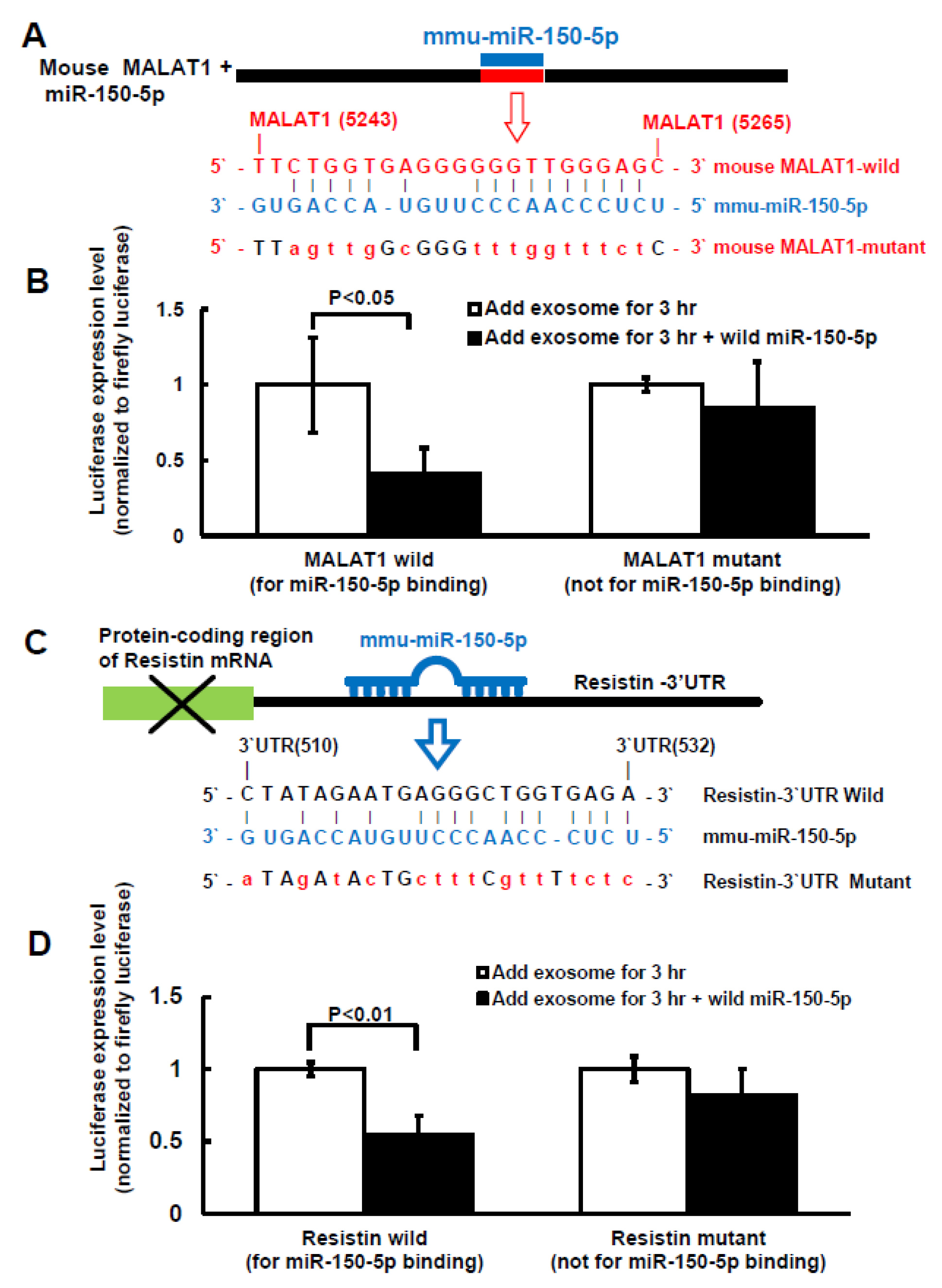
2.3. MiR-150-5p Is the Target of MALAT1 and Resistin Is the Target of miR-150-5 in Macrophage
2.4. Macrophage-Derived Exosomes Reduced Glucose Uptake in Macrophages
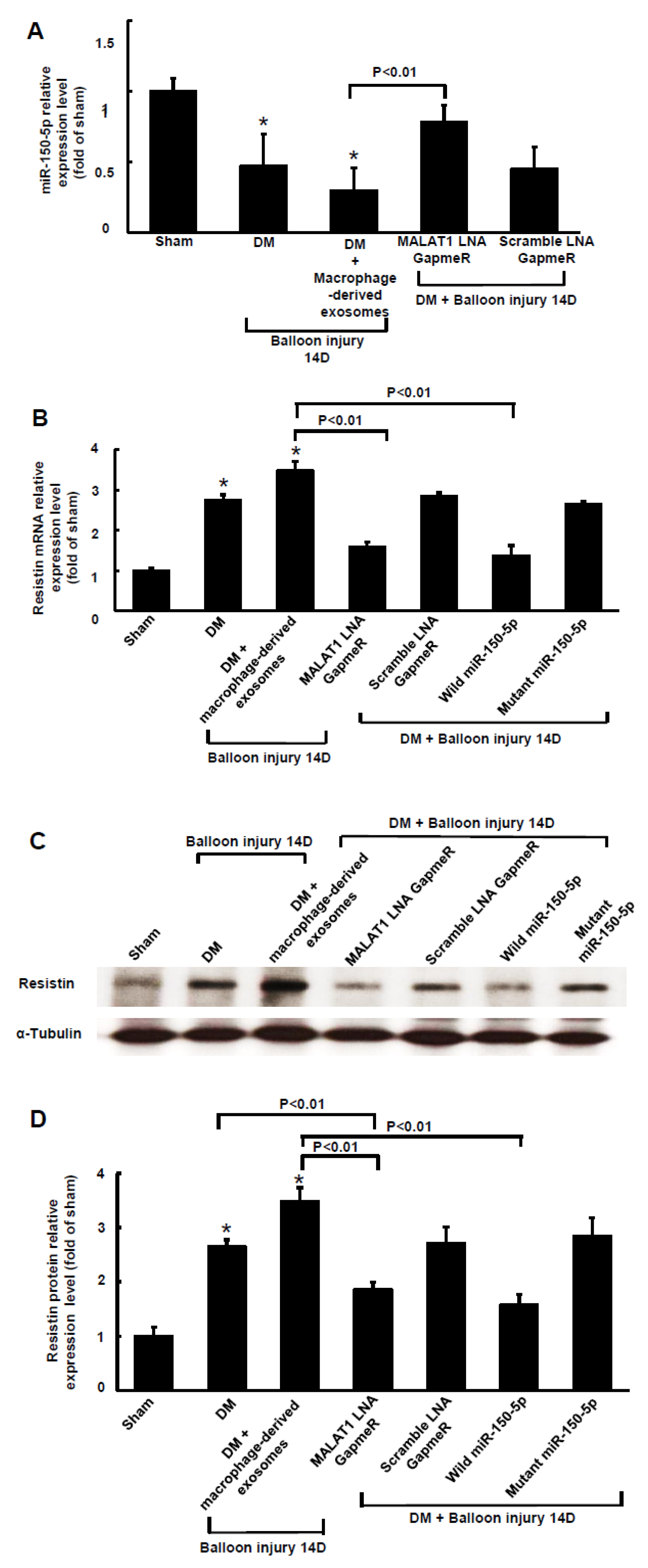
2.5. Balloon Injury Increased MALAT1 to Suppress miR150-5p Expression in Diabetic Rats
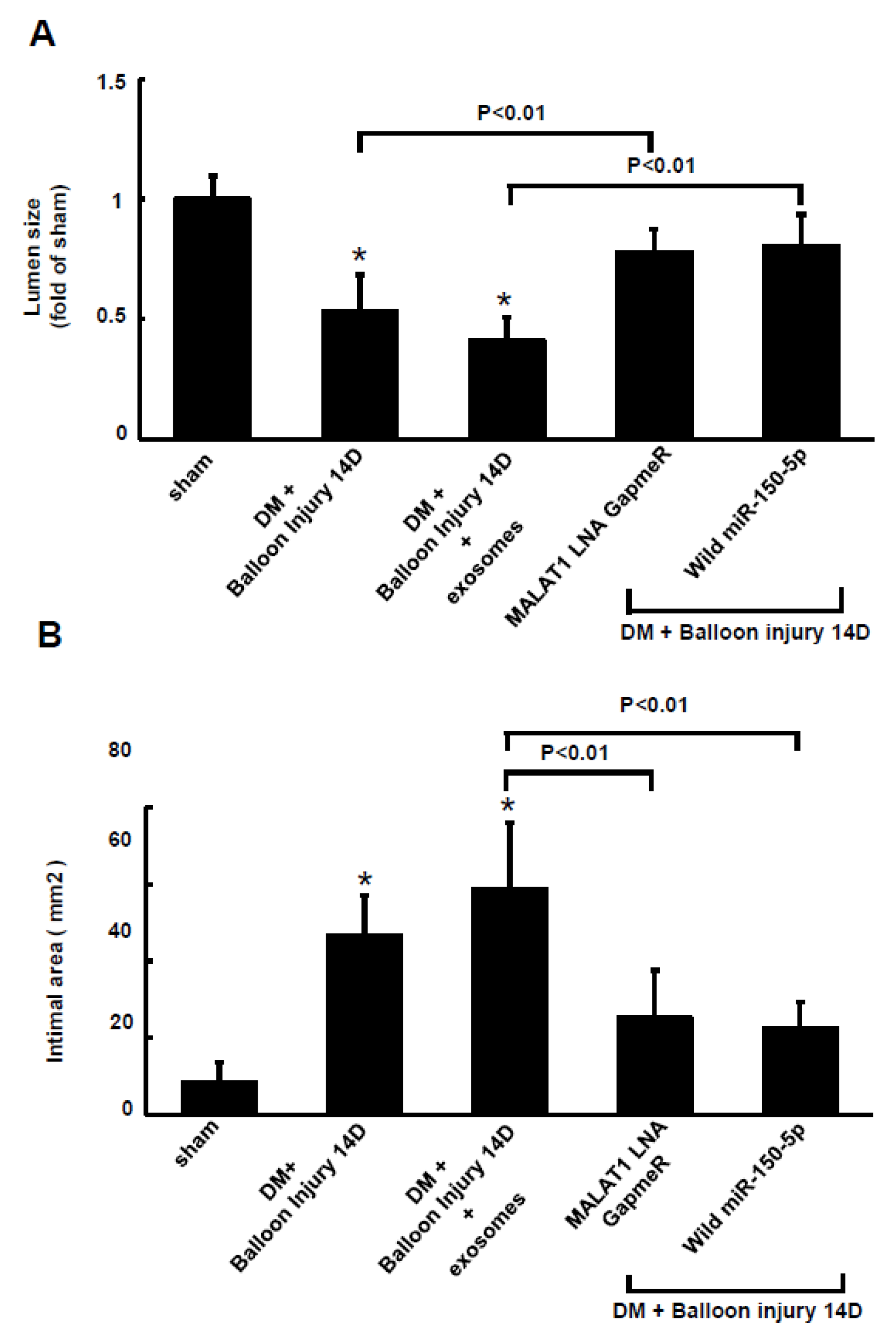
2.6. Macrophages-Derived Exosomes Increased Resistin Protein Expression in the Arterial Tissue of Diabetic Rats after Carotid Artery Balloon Injury
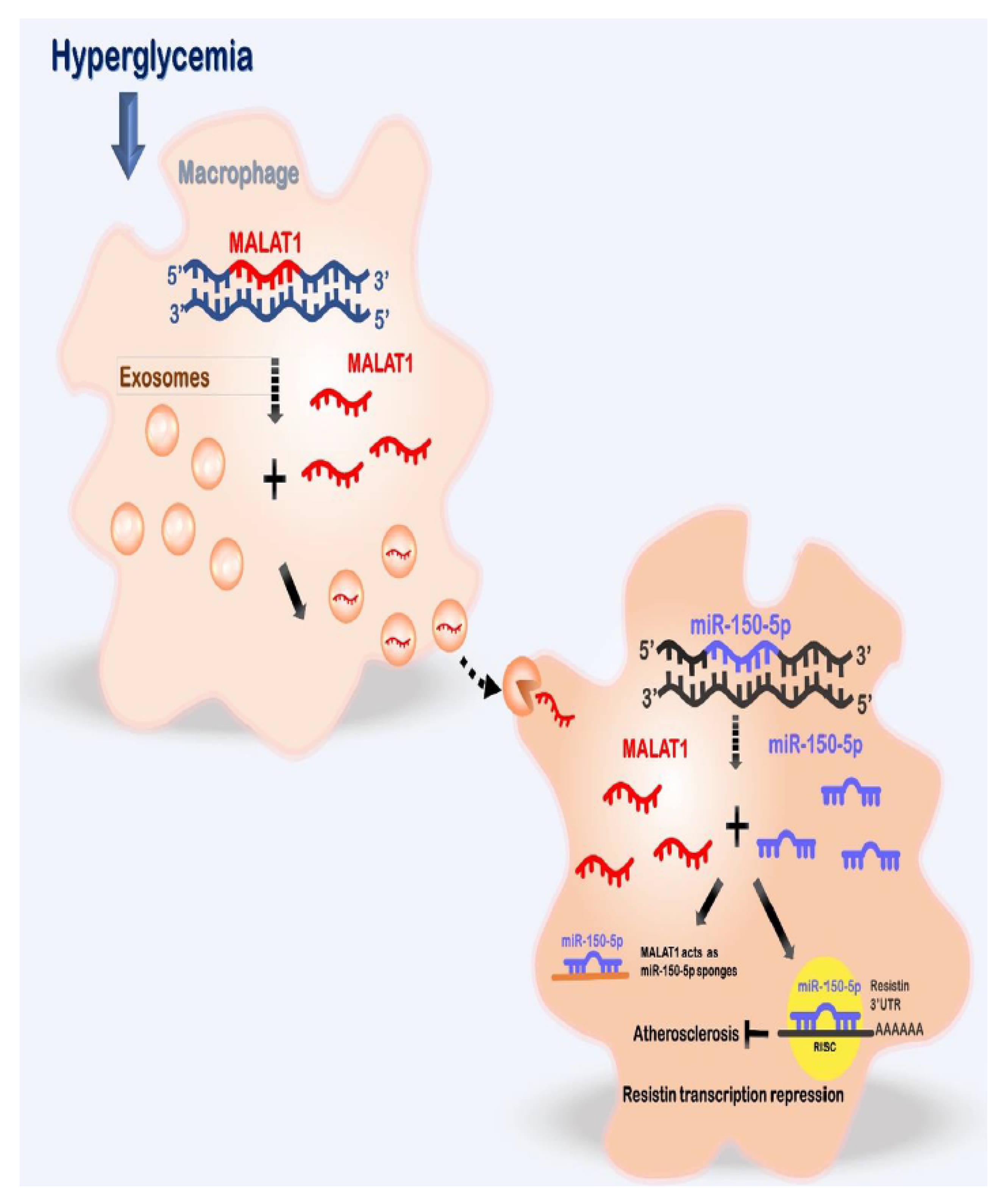
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Hyperglycemic Stress Environment
4.3. Extraction of Exosomes from Cell Media
4.4. RNA Quality Assessment of the Exosomes
4.5. Reverse Transcription and Real-Time Quantitative Polymerase Chain Reaction
4.6. Polymerase Chain Reaction Product Construction and Sequencing
4.7. Western Blot Analysis
4.8. Transfection of MALAT1 Locked Nucleic Acid (LNA) GapmeR
4.9. Luciferase Activity Assay
4.10. Construction of the mmu-miR-150-5p Expression Vector
4.11. Balloon Injury of Carotid Artery in Diabetic Rat and Macrophage-Derived MALAT1-Contained Exosomes Delivery
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moore, K.J.; Tabas, I. Macrophages in the pathogenesis of atherosclerosis. Cell 2011, 145, 341–355. [Google Scholar] [CrossRef] [Green Version]
- de Bruin, R.G.; Shiue, L.; Prins, J.; de Boer, H.C.; Singh, A.; Fagg, W.S.; van Gils, J.M.; Duijs, J.M.G.; Katzman, S.; Kraaijeveld, A.O.; et al. Quaking promotes monocyte differentiation into pro-atherogenic macrophages by controlling pre-mRNA splicing and gene expression. Nat. Commun. 2016, 7, 10846. [Google Scholar] [CrossRef] [PubMed]
- Uszczynska-Ratajczak, B.; Lagarde, J.; Frankish, A.; Guigó, R.; Johnson, R. Towards a complete map of the human long non-coding RNA transcriptome. Nat. Rev. Genet. 2018, 19, 535–548. [Google Scholar] [CrossRef]
- Zhou, T.; Ding, J.W.; Wang, X.A.; Zheng, X.X. Long noncoding RNAs and atherosclerosis. Atherosclerosis 2016, 248, 51–61. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.Y.; Yao, J.; Li, X.M.; Song, Y.C.; Wang, X.Q.; Li, Y.J. Pathogenic role of lncRNA-MALAT1 in endothelial cell dysfunction in diabetes mellitus. Cell Death Dis. 2014, 5, e1506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puthanveetil, P.; Chen, S.; Feng, B.; Gautam, A.; Chakrabarti, S. Long non-coding RNA MALAT1 regulates hyperglycemia induced inflammatory process in the endothelial cells. J. Cell. Mol. Med. 2015, 19, 1418–1425. [Google Scholar] [CrossRef]
- Liu, S.X.; Zheng, F.; Xie, K.L.; Xie, M.R.; Jiang, L.J.; Cai, Y. Exercise reduces insulin resistance in type 2 diabetes mellitus via mediating the lncRNA MALAT1/microRNA-382-3p/resistin axis. Mol. Ther. Nucleic Acids 2019, 18, 34–44. [Google Scholar] [CrossRef] [Green Version]
- Yan, B.; Tao, Z.F.; Li, X.M.; Zhang, H.; Yao, J.; Jiang, Q. Aberrant expression of long noncoding RNAs in early diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2014, 55, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Moccetti, T.; Marban, E.; Vassalli, G. Roles of exosomes in cardioprotection. Eur. Heart J. 2017, 38, 1372–1379. [Google Scholar] [CrossRef]
- Qian, B.; Yang, Y.; Tang, N.; Wang, J.; Sun, P.; Yang, N.; Chen, F.; Wu, T.; Sun, T.; Li, Y.; et al. M1 macrophage-derived exosomes impaired beta cell insulin secretion via miR-212-5p by targeting SIRT2 and inhibiting Akt/GSK-3β/β-catenin pathway in mice. Diabetologia 2021, 64, 2037–2051. [Google Scholar] [CrossRef]
- Haffner, S.M.; Lehto, S.; Rönnemaa, T.; Pyörälä, K.; Laakso, M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N. Engl. J. Med. 1998, 339, 229–234. [Google Scholar] [CrossRef]
- Han, Y.; Qiu, H.; Pei, X.; Fan, Y.; Tian, H.; Geng, J. Low-dose sinapic acid abates the pyroptosis of macrophages by downregulation of lncRNA-MALAT1 in rats with diabetic atherosclerosis. J. Cardiovasc. Pharmacol. 2018, 71, 104–112. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Chen, G.; He, R.; Yang, L. LncRNA MALAT1 promotes osteoarthritis by modulating miR-150-5p/AKT 3 axis. Cell Biosci. 2019, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Liu, Q. Long noncoding RNA MALAT1 modulates sepsis-induced cardiac inflammation through the miR-150-5p/NF-kB axis. Int. J. Clin. Exp. Pathol. 2019, 12, 3311–3319. [Google Scholar] [PubMed]
- Liu, L.; Yan, L.N.; Sui, Z. MicroRNA-150 affects endoplasmic reticulum stress vai MALAT1-miR-150 axis-mediated NF-kB pathway in LPS-challenged HUVECs and septic mice. Life Sci. 2021, 265, 118744. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.Y.; Zhang, W.H.; Ma, W.T.; Liu, Q.H.; Xing, L.H.; Zhao, G.F. Long non-coding RNA MALAT1 exacerbates acute respiratory distress syndrome by upregulating ICAM-1 expression via microRNA-150-5p downregulation. Aging 2020, 12, 6570–6585. [Google Scholar] [CrossRef] [PubMed]
- Qatanani, M.; Szwergold, N.; Greaves, D.; Ahima, R.S.; Lazar, M.A. Macrophages-derived human resistin exacerbates adipose tissue inflammation and insulin resistance in mice. J. Clin. Investig. 2009, 119, 531–539. [Google Scholar] [CrossRef] [Green Version]
- Zhou, L.; Li, J.Y.; He, P.P.; Yu, X.H.; Tang, C.K. Resistin: Potential biomarker and therapeutic target in atherosclerosis. Clin. Chim. Acta 2021, 123, 84–91. [Google Scholar] [CrossRef]
- Jung, H.S.; Park, K.H.; Cho, Y.M.; Chong, S.S.; Cho, H.J.; Cho, S.Y.; Kim, S.J.; Kim, S.Y.; Lee, H.K.; Park, K.S. Resistin is secreted from macrophages in atheromas and promotes atherosclerosis. Cardiovasc. Res. 2006, 69, 76–85. [Google Scholar] [CrossRef]
- Gast, M.; Rauch, B.; Nakagawa, S.; Haghikia, A.; Jasina, A.; Haas, J.; Nath, N.; Jensen, L.; Stroux, A.; Böhm, A.; et al. Immune system-mediated atherosclerosis caused by deficiency of long-coding RNA MALAT1 in ApoE−/− mice. Cardiovasc. Res. 2019, 115, 302–314. [Google Scholar] [CrossRef]
- Cremer, S.; Michalik, K.M.; Fischer, A.; Pfisterer, L.; Jae, N.; Winter, C.; Boon, R.A.; Muhly-Reinholz, M.; John, D.; Uchida, S.; et al. Hematopoietic deficiency of the long noncoding RNA MALAT1 promotes atherosclerosis and plaque inflammation. Circulation 2019, 139, 1320–1334. [Google Scholar] [CrossRef]
- Chen, L.; Hu, L.; Zhu, X.; Li, Q.; Ma, J.; Li, H. MALAT1 overexpression attenuates AS by inhibiting ox-LDL-stimulated dendritic cell maturation via miR155-5p/NFIA axis. Cell Cycle 2020, 19, 2472–2485. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Yang, L.; Guo, R.; Lu, N.; Shi, Y.; Wang, X. Long noncoding RNA MALAT1 promotes high glucose-induced human endothelial cells pyroptosis by affecting NLRP3 expression through competitively binding miR-22. Biochem. Biophys. Res. Commun. 2019, 509, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Qi, Y.; Wang, Y.; Tang, H.; Li, Z.; Wang, Y.; Tang, S.; Zhu, H. LncRNA MALAT1 suppression protects endothelium against oxLDL-induced inflammation via inhibiting expression of miR-181b target gene TOX. Oxidative Med. Cell. Longev. 2019, 2019, 8245810. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Yang, T.; Duan, J.; Mu, N.; Zhang, T. MALAT1/miR-15b-5p/MAPK1 mediates endothelial progenitor cells autophagy and affects coronary atherosclerotic heart disease via mTOR signaling pathway. Aging 2019, 114, 1089–1109. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Wang, X.; Lin, C.; An, Z.; Yu, J.; Cao, H.; Fan, Y.; Liang, X. Exosomal MALAT1 derived from ox-LDL-treated endothelial cells induce neutrophil extracellular traps to aggravate atherosclerosis. Biol. Chem. 2020, 401, 367376. [Google Scholar] [CrossRef]
- Lu, X. The role of exosomes and exosome-derived micro-RNA in atherosclerosis. Curr. Pharm. Des. 2017, 23, 6182–6193. [Google Scholar] [CrossRef]
- Wang, Y.; Jia, L.; Xie, Y.J.; Cai, Z.; Liu, Z.; Shen, J.; Lu, Y.; Wang, Y.; Su, S.; Ma, Y.; et al. Involvement of macrophage-derived exosomes in abdominal aortic aneurysm development. Atherosclerosis 2019, 289, 64–72. [Google Scholar] [CrossRef]
- Khoury, M.K.; Yang, H.; Liu, B. Macrophage biology in cardiovascular diseases. Arter. Thromb. Vasc. Biol. 2021, 41, e77–e81. [Google Scholar] [CrossRef]
- Kim, H.; Wang, S.Y.; Kwak, G.; Yang, Y.; Kwon, I.C.; Kim, S.H. Exosome-guided phenotypic switch of M1 to M2 macrophages for cutanous woundhealing. Adv. Sci. 2019, 6, 1900513. [Google Scholar] [CrossRef] [Green Version]
- Park, G.M.; Lee, C.H.; Lee, S.W.; Yun, S.C.; Kim, Y.H.; Kim, Y.G.; Won, K.B.; Ann, S.H.; Kim, S.J.; Yang, D.H.; et al. Impact of diabetes control on subclinical atherosclerosis: Analysis from coronary computed tomographic angiography registry. Diabetes Mtab. J. 2020, 44, 470–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shyu, K.G.; Wang, B.W.; Pan, C.M.; Fang, W.J.; Lin, C.M. Hyperbaric oxygen boosts long noncoding RNA MALAT1 exosome secretion to suppress microRNA-92a expression in therapeutic angiogenesis. Int. J. Cardiol. 2019, 274, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Qinna, N.A.; Badwan, A.A. Impact of streptozotocin on altering normal glucose homeostasis during insulin testing in diabetic rats compared to normoglycemic rats. Drug Des. Dev. Ther. 2015, 9, 2515–2525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.M.; Hou, S.W.; Wang, B.W.; Ong, J.R.; Chang, H.; Shyu, K.G. Molecular mechanism of (−)-epigallocatechin-3-gallate on balloon injury-induced neointimal formation and leptin expression. J. Agric. Food Chem. 2014, 62, 1213–1220. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shyu, K.-G.; Wang, B.-W.; Fang, W.-J.; Pan, C.-M.; Lin, C.-M. Exosomal MALAT1 Derived from High Glucose-Treated Macrophages Up-Regulates Resistin Expression via miR-150-5p Downregulation. Int. J. Mol. Sci. 2022, 23, 1095. https://doi.org/10.3390/ijms23031095
Shyu K-G, Wang B-W, Fang W-J, Pan C-M, Lin C-M. Exosomal MALAT1 Derived from High Glucose-Treated Macrophages Up-Regulates Resistin Expression via miR-150-5p Downregulation. International Journal of Molecular Sciences. 2022; 23(3):1095. https://doi.org/10.3390/ijms23031095
Chicago/Turabian StyleShyu, Kou-Gi, Bao-Wei Wang, Wei-Jen Fang, Chun-Ming Pan, and Chiu-Mei Lin. 2022. "Exosomal MALAT1 Derived from High Glucose-Treated Macrophages Up-Regulates Resistin Expression via miR-150-5p Downregulation" International Journal of Molecular Sciences 23, no. 3: 1095. https://doi.org/10.3390/ijms23031095
APA StyleShyu, K.-G., Wang, B.-W., Fang, W.-J., Pan, C.-M., & Lin, C.-M. (2022). Exosomal MALAT1 Derived from High Glucose-Treated Macrophages Up-Regulates Resistin Expression via miR-150-5p Downregulation. International Journal of Molecular Sciences, 23(3), 1095. https://doi.org/10.3390/ijms23031095





