Cruciform Formable Sequences within Pou5f1 Enhancer Are Indispensable for Mouse ES Cell Integrity
Abstract
1. Introduction
2. Results
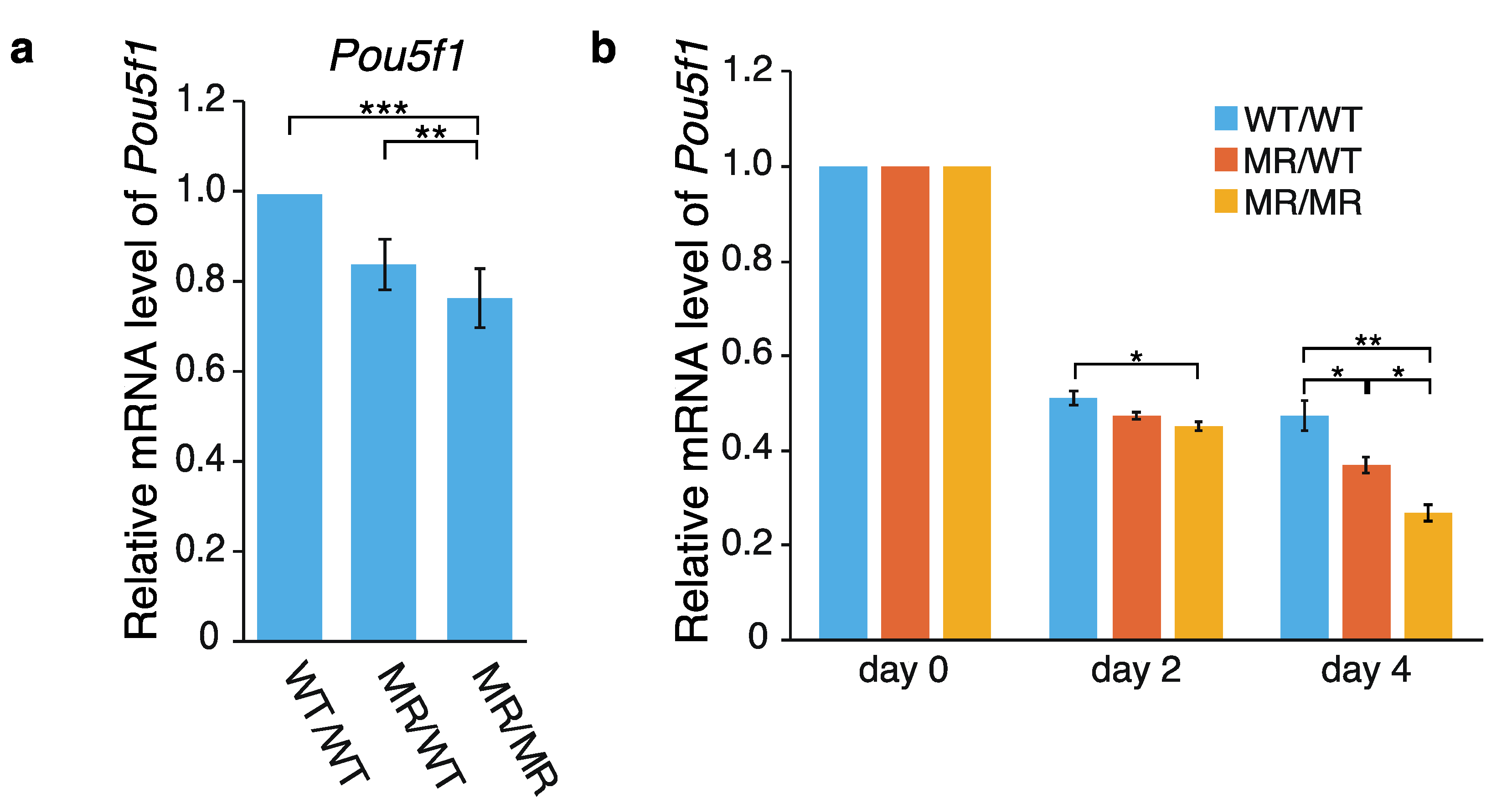
2.1. Effects of the Genome Editing on Pou5f1 and eRNA Transcription
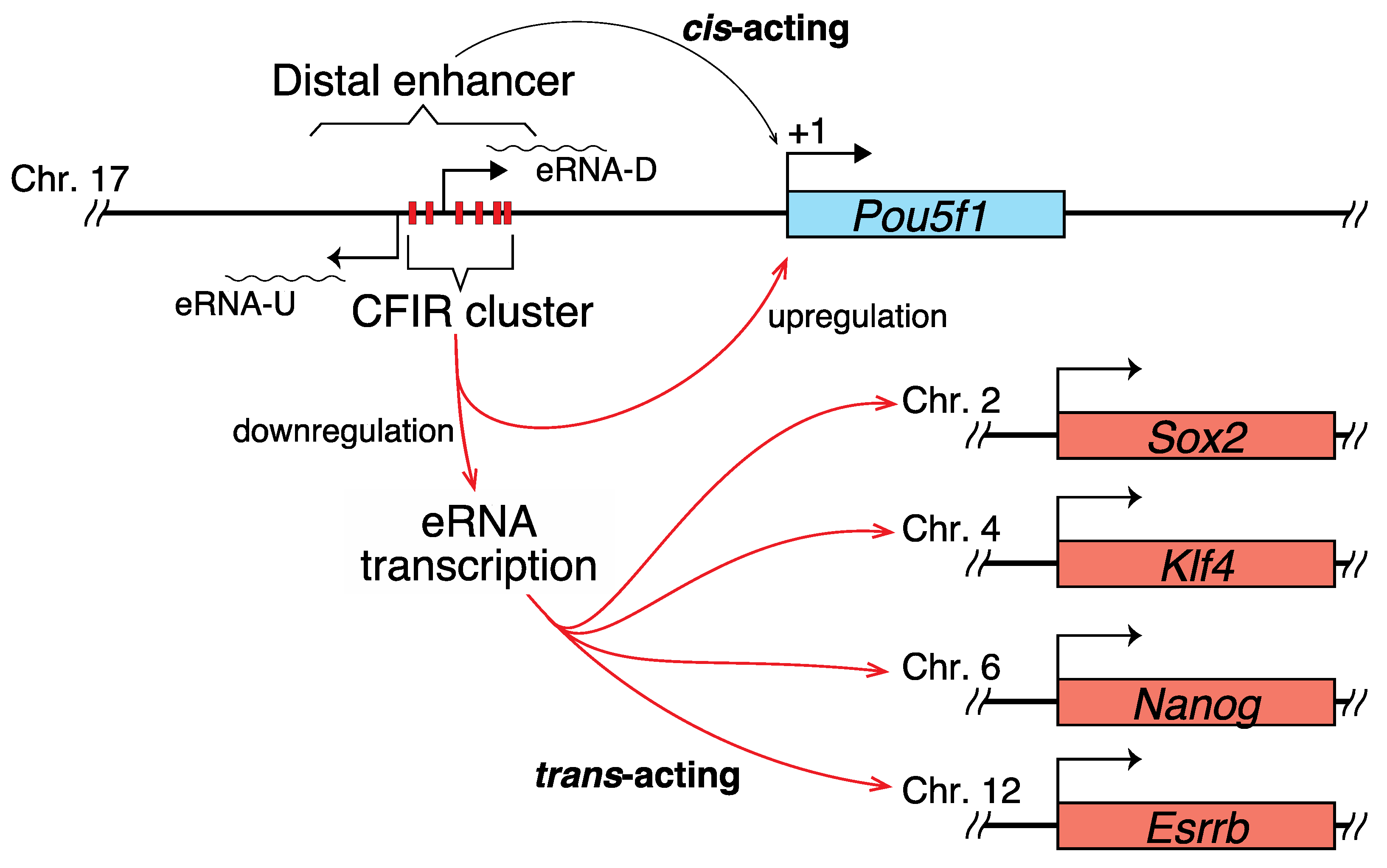
2.2. Correlation between the Transcription of eRNA and That of Genes Controlled by a Super-Enhancer
3. Discussion
3.1. Hypothetical Mechanism Underlying the Editing-Elicited Downregulation of Pou5f1 Expression
3.2. eRNAs Transcribed from DE Are Presumably Implicated in the Transcriptional Regulation of Sox2, Nanog, Klf4, and Esrrb
3.3. The Cluster of CFIRs Can Also Act as an Absorber of Negative Supercoils
4. Materials and Methods
4.1. Cell Culture and Transfection
4.2. Construction of Donor and Px330 Vectors
4.3. Sequence Analysis
4.4. RNA Isolation and Reverse Transcription
4.5. qPCR Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Platt, J.R. Possible separation of intertwined nucleic acid chains by transfer-twist. Proc. Natl. Acad. Sci. USA 1955, 41, 181–183. [Google Scholar] [CrossRef] [PubMed]
- Pearson, C.E.; Zorbas, H.; Price, G.B.; Zannis-Hadjopoulos, M. Inverted repeats, stem-loops, and cruciforms: Significance for initiation of DNA replication. J. Cell. Biochem. 1996, 63, 1–22. [Google Scholar] [CrossRef]
- Zannis-Hadjopoulos, M.; Yahyaoui, W.; Callejo, M. 14-3-3 Cruciform-binding proteins as regulators of eukaryotic DNA replication. Trends Biochem. Sci. 2008, 33, 44–50. [Google Scholar] [CrossRef]
- Brázda, V.; Laister, R.C.; Jagelská, E.B.; Arrowsmith, C. Cruciform structures are a common DNA feature important for regulating biological processes. BMC Mol. Biol. 2011, 12, 33. [Google Scholar] [CrossRef]
- Dai, X.; Greizerstein, M.B.; Nadas-Chinni, K.; Rothman-Denes, L.B. Supercoil-induced extrusion of a regulatory DNA hairpin. Proc. Natl. Acad. Sci. USA 1997, 94, 2174–2179. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Rothman-Denes, L.B. Sequence and DNA structural determinants of N4 virion RNA polymerase-promoter recognition. Genes Dev. 1998, 12, 2782–2790. [Google Scholar] [CrossRef]
- Jagelská, E.B.; Pivoňková, H.; Fojta, M.; Brázda, V. The potential of the cruciform structure formation as an important factor influencing p53 sequence-specific binding to natural DNA targets. Biochem. Biophys. Res. Commun. 2010, 391, 1409–1414. [Google Scholar] [CrossRef]
- Brázda, V.; Coufal, J.; Liao, J.C.C.; Arrowsmith, C.H. Preferential binding of IFI16 protein to cruciform structure and superhelical DNA. Biochem. Biophys. Res. Commun. 2012, 422, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Coufal, J.; Jagelská, E.B.; Liao, J.C.C.; Brázda, V. Preferential binding of p53 tumor suppressor to p21 promoter sites that contain inverted repeats capable of forming cruciform structure. Biochem. Biophys. Res. Commun. 2013, 441, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Miura, O.; Ogake, T.; Ohyama, T. Requirement or exclusion of inverted repeat sequences with cruciform-forming potential in Escherichia coli revealed by genome-wide analyses. Curr. Genet. 2018, 64, 945–958. [Google Scholar] [CrossRef]
- Miura, O.; Ogake, T.; Yoneyama, H.; Kikuchi, Y.; Ohyama, T. A strong structural correlation between short inverted repeat sequences and the polyadenylation signal in yeast and nucleosome exclusion by these inverted repeats. Curr. Genet. 2019, 65, 575–590. [Google Scholar] [CrossRef]
- Lin, C.T.; Lyu, Y.L.; Liu, L.F. A cruciform-dumbbell model for inverted dimer formation mediated by inverted repeats. Nucleic Acids Res. 1997, 25, 3009–3016. [Google Scholar] [CrossRef] [PubMed]
- Shlyakhtenko, L.S.; Hsieh, P.; Grigoriev, M.; Potaman, V.N.; Sinden, R.R.; Lyubchenko, Y.L. A cruciform structural transition provides a molecular switch for chromosome structure and dynamics. J. Mol. Biol. 2000, 296, 1169–1173. [Google Scholar] [CrossRef] [PubMed]
- Lobachev, K.S.; Gordenin, D.A.; Resnick, M.A. The Mre11 complex is required for repair of hairpin-capped double-strand breaks and prevention of chromosome rearrangements. Cell 2002, 108, 183–193. [Google Scholar] [CrossRef]
- Wang, Y.; Leung, F.C.C. Long inverted repeats in eukaryotic genomes: Recombinogenic motifs determine genomic plasticity. FEBS Lett. 2006, 580, 1277–1284. [Google Scholar] [CrossRef]
- Andersson, R.; Gebhard, C.; Miguel-Escalada, I.; Hoof, I.; Bornholdt, J.; Boyd, M.; Chen, Y.; Zhao, X.; Schmidl, C.; Suzuki, T.; et al. An atlas of active enhancers across human cell types and tissues. Nature 2014, 507, 455–461. [Google Scholar] [CrossRef]
- Forrest, A.R.R.; Kawaji, H.; Rehli, M.; Baillie, J.K.; De Hoon, M.J.L.; Haberle, V.; Lassmann, T.; Kulakovskiy, I.V.; Lizio, M.; Itoh, M.; et al. A promoter-level mammalian expression atlas. Nature 2014, 507, 462–470. [Google Scholar] [CrossRef] [PubMed]
- De Santa, F.; Barozzi, I.; Mietton, F.; Ghisletti, S.; Polletti, S.; Tusi, B.K.; Muller, H.; Ragoussis, J.; Wei, C.L.; Natoli, G. A large fraction of extragenic RNA Pol II transcription sites overlap enhancers. PLoS Biol. 2010, 8, e1000384. [Google Scholar] [CrossRef] [PubMed]
- Djebali, S.; Davis, C.A.; Merkel, A.; Dobin, A.; Lassmann, T.; Mortazavi, A.; Tanzer, A.; Lagarde, J.; Lin, W.; Schlesinger, F.; et al. Landscape of transcription in human cells. Nature 2012, 489, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Hemberg, M.; Gray, J.M.; Costa, A.M.; Bear, D.M.; Wu, J.; Harmin, D.A.; Laptewicz, M.; Barbara-Haley, K.; Kuersten, S.; et al. Widespread transcription at neuronal activity-regulated enhancers. Nature 2010, 465, 182–187. [Google Scholar] [CrossRef]
- Gil, N.; Ulitsky, I. Production of spliced long noncoding RNAs specifies regions with increased enhancer activity. Cell Syst. 2018, 7, 537–547.e3. [Google Scholar] [CrossRef] [PubMed]
- Vučićević, D.; Corradin, O.; Ntini, E.; Scacheri, P.C.; Ørom, U.A. Long ncRNA expression associates with tissue-specific enhancers. Cell Cycle 2015, 14, 253–260. [Google Scholar] [CrossRef]
- Pan, G.J.; Chang, Z.Y.I.; Schöler, H.R.; Pei, D. Stem cell pluripotency and transcription factor Oct4. Cell Res. 2002, 12, 321–329. [Google Scholar] [CrossRef]
- Niwa, H. How is pluripotency determined and maintained? Development 2007, 134, 635–646. [Google Scholar] [CrossRef]
- Kellner, S.; Kikyo, N. Transcriptional regulation of the Oct4 gene, a master gene for pluripotency. Histol. Histopathol. 2010, 25, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Whyte, W.A.; Orlando, D.A.; Hnisz, D.; Abraham, B.J.; Lin, C.Y.; Kagey, M.H.; Rahl, P.B.; Lee, T.I.; Young, R.A. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Cell 2013, 153, 307–319. [Google Scholar] [CrossRef]
- Wu, M.; Shen, J. From super-enhancer non-coding RNA to immune checkpoint: Frameworks to functions. Front. Oncol. 2019, 9, 1–16. [Google Scholar] [CrossRef]
- Yoshimi, K.; Kunihiro, Y.; Kaneko, T.; Nagahora, H.; Voigt, B.; Mashimo, T. ssODN-mediated knock-in with CRISPR-Cas for large genomic regions in zygotes. Nat. Commun. 2016, 7, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Nordhoff, V.; Hübner, K.; Bauer, A.; Orlova, I.; Malapetsa, A.; Schöler, H.R. Comparative analysis of human, bovine, and murine Oct-4 upstream promoter sequences. Mamm. Genome 2001, 12, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.G.; Heath, J.K.; Donaldson, D.D.; Wong, G.G.; Moreau, J.; Stahl, M.; Rogers, D. Inhibition of pluripotential embryonic stem cell differentiation by purified polypeptides. Nature 1988, 336, 688–690. [Google Scholar] [CrossRef]
- Williams, R.L.; Hilton, D.J.; Pease, S.; Willson, T.A.; Stewart, C.L.; Gearing, D.P.; Wagner, E.F.; Metcalf, D.; Nicola, N.A.; Gough, N.M. Myeloid leukaemia inhibitory factor maintains the developmental potential of embryonic stem cells. Nature 1988, 336, 684–687. [Google Scholar] [CrossRef] [PubMed]
- Tippens, N.D.; Vihervaara, A.; Lis, J.T. Enhancer transcription: What, where, when, and why? Genes Dev. 2018, 32, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.; Zevnik, B.; Anastassiadis, K.; Niwa, H.; Klewe-Nebenius, D.; Chambers, I.; Schöler, H.; Smith, A. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 1998, 95, 379–391. [Google Scholar] [CrossRef]
- Yeom, Y.I.; Fuhrmann, G.; Ovitt, C.E.; Brehm, A.; Ohbo, K.; Gross, M.; Hübner, K.; Schöler, H.R. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 1996, 122, 881–894. [Google Scholar]
- Pesce, M.; Schöler, H.R. Oct-4: Control of totipotency and germline determination. Mol. Reprod. Dev. 2000, 55, 452–457. [Google Scholar] [CrossRef]
- Pesce, M.; Schöler, H.R. Oct-4: Gatekeeper in the beginnings of mammalian development. Stem Cells 2001, 19, 271–278. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat. Rev. Mol. Cell Biol. 2016, 17, 183–193. [Google Scholar] [CrossRef]
- Liao, J.; He, Y.; Szabó, P.E. The Pou5f1 distal enhancer is sufficient to drive Pou5f1 promoter-EGFP expression in embryonic stem cells. Int. J. Dev. Biol. 2013, 57, 725–729. [Google Scholar] [CrossRef]
- Canon, E.; Jouneau, L.; Blachère, T.; Peynot, N.; Daniel, N.; Boulanger, L.; Maulny, L.; Archilla, C.; Voisin, S.; Jouneau, A.; et al. Progressive methylation of POU5F1 regulatory regions during blastocyst development. Reproduction 2018, 156, 145–161. [Google Scholar] [CrossRef]
- Chew, J.L.; Loh, Y.H.; Zhang, W.; Chen, X.; Tam, W.L.; Yeap, L.S.; Li, P.; Ang, Y.S.; Lim, B.; Robson, P.; et al. Reciprocal transcriptional regulation of Pou5f1 and Sox2 via the Oct4/Sox2 complex in embryonic stem cells. Mol. Cell. Biol. 2005, 25, 6031–6046. [Google Scholar] [CrossRef]
- Okumura-Nakanishi, S.; Saito, M.; Niwa, H.; Ishikawa, F. Oct-3/4 and Sox2 regulate Oct-3/4 gene in embryonic stem cells. J. Biol. Chem. 2005, 280, 5307–5317. [Google Scholar] [CrossRef]
- Feldman, N.; Gerson, A.; Fang, J.; Li, E.; Zhang, Y.; Shinkai, Y.; Cedar, H.; Bergman, Y. G9a-mediated irreversible epigenetic inactivation of Oct-3/4 during early embryogenesis. Nat. Cell Biol. 2006, 8, 188–194. [Google Scholar] [CrossRef]
- Hattori, N.; Nishino, K.; Ko, Y.G.; Hattori, N.; Ohgane, J.; Tanaka, S.; Shiota, K. Epigenetic control of mouse Oct-4 gene expression in embryonic stem cells and trophoblast stem cells. J. Biol. Chem. 2004, 279, 17063–17069. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Pu, M.T.; Hirasawa, R.; Li, B.Z.; Huang, Y.N.; Zeng, R.; Jing, N.H.; Chen, T.; Li, E.; Sasaki, H.; et al. Synergistic function of DNA methyltransferases Dnmt3a and Dnmt3b in the methylation of Oct4 and Nanog. Mol. Cell. Biol. 2007, 27, 8748–8759. [Google Scholar] [CrossRef]
- Tachibana, M.; Sugimoto, K.; Nozaki, M.; Ueda, J.; Ohta, T.; Ohki, M.; Fukuda, M.; Takeda, N.; Niida, H.; Kato, H.; et al. G9a histone methyltransferase plays a dominant role in euchromatic histone H3 lysine 9 methylation and is essential for early embryogenesis. Genes Dev. 2002, 16, 1779–1791. [Google Scholar] [CrossRef]
- Ben-Shushan, E.; Pikarsky, E.; Klar, A.; Bergman, Y. Extinction of Oct-3/4 gene expression in embryonal carcinoma x fibroblast somatic cell hybrids is accompanied by changes in the methylation status, chromatin structure, and transcriptional activity of the Oct-3/4 upstream region. Mol. Cell. Biol. 1993, 13, 891–901. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Minucci, S.; Botquin, V.; Yeom, Y.I.; Dey, A.; Sylvester, I.; Zand, D.J.; Ohbo, K.; Ozato, K.; Schöler, H.R. Retinoic acid-mediated down-regulation of Oct3/4 coincides with the loss of promoter occupancy in vivo. EMBO J. 1996, 15, 888–899. [Google Scholar] [CrossRef]
- Aiken, C.R.; Fisher, E.W.; Gumport, R.I. The specific binding, bending, and unwinding of DNA by RsrI endonuclease, an isoschizomer of EcoRI endonuclease. J. Biol. Chem. 1991, 266, 19063–19069. [Google Scholar] [CrossRef]
- Giese, K.; Pagel, J.; Groosshedl, R. Functional analysis of DNA bending and unwinding by the high mobility group domain of LEF-1. Proc. Natl. Acad. Sci. USA 1997, 94, 12845–12850. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.L.; Nikolov, D.B.; Burley, S.K. Co-crystal structure of TBP recognizing the minor groove of a TATA element. Nature 1993, 365, 520–527. [Google Scholar] [CrossRef]
- Kim, Y.; Geiger, J.H.; Hahn, S.; Sigler, P.B. Crystal structure of a yeast TBP/TATA-box complex. Nature 1993, 365, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Missura, M.; Buterin, T.; Hindges, R.; Hübscher, U.; Kaspárková, J.; Brabec, V.; Naegeli, H. Double-check probing of DNA bending and unwinding by XPA-RPA: An architectural function in DNA repair. EMBO J. 2001, 20, 3554–3564. [Google Scholar] [CrossRef] [PubMed]
- Coulombe, B.; Burton, Z.F. DNA bending and wrapping around RNA polymerase: A “revolutionary” model describing transcriptional Mmechanisms. Microbiol. Mol. Biol. Rev. 1999, 63, 457–478. [Google Scholar] [CrossRef] [PubMed]
- Nazarov, I.B.; Krasnoborova, V.A.; Mitenberg, A.G.; Chikhirzhina, E.V.; Davidov-Sinitzin, A.P.; Liskovykh, M.A.; Tomilin, A.N. Transcription regulation of Oct4 (Pou5F1) gene by its distal enhancer. Cell Tissue Biol. 2014, 8, 27–32. [Google Scholar] [CrossRef]
- Hnisz, D.; Abraham, B.J.; Lee, T.I.; Lau, A.; Saint-André, V.; Sigova, A.A.; Hoke, H.A.; Young, R.A. Super-enhancers in the control of cell identity and disease. Cell 2013, 155, 934. [Google Scholar] [CrossRef]
- Bose, D.A.; Donahue, G.; Reinberg, D.; Shiekhattar, R.; Bonasio, R.; Berger, S.L. RNA binding to CBP stimulates histone acetylation and transcription. Cell 2017, 168, 135–149.e22. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.L.; Fei, T.; Chen, Y.; Li, T.; Gao, Y.; Wang, X.; Sun, T.; Sweeney, C.J.; Lee, G.S.M.; Chen, S.; et al. Enhancer RNAs participate in androgen receptor-driven looping that selectively enhances gene activation. Proc. Natl. Acad. Sci. USA 2014, 111, 7319–7324. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.; Orom, U.A.; Cesaroni, M.; Beringer, M.; Taatjes, D.J.; Blobel, G.A.; Shiekhattar, R. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature 2013, 494, 497–501. [Google Scholar] [CrossRef]
- Mousavi, K.; Zare, H.; Dell’Orso, S.; Grontved, L.; Gutierrez-Cruz, G.; Derfoul, A.; Hager, G.L.; Sartorelli, V. eRNAs promote transcription by establishing chromatin accessibility at defined genomic loci. Mol. Cell 2013, 51, 606–617. [Google Scholar] [CrossRef]
- Schaukowitch, K.; Joo, J.Y.; Liu, X.; Watts, J.K.; Martinez, C.; Kim, T.K. Enhancer RNA facilitates NELF release from immediate early genes. Mol. Cell 2014, 56, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Spurlock, C.F.; Shaginurova, G.; Tossberg, J.T.; Hester, J.D.; Chapman, N.; Guo, Y.; Crooke, P.S.; Aune, T.M. Profiles of long noncoding RNAs in human naive and memory T cells. J. Immunol. 2017, 199, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Sinden, R.R. DNA Structure and Function; Elsevier: Amsterdam, The Netherlands, 1994; ISBN 9780080571737. [Google Scholar]
- Panayotatos, N.; Wells, R.D. Cruciform structures in supercoiled DNA. Nature 1981, 289, 466–470. [Google Scholar] [CrossRef]
- Hsieh, T.-S.; Wang, J.C. Thermodynamic properties of superhelical DNAs. Biochemistry 1975, 14, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, T.; Nishikawa, K.; Takemura, S. Studies on T. Utilis tRNATyr variants with enzymatically altered D-loop sequences. I. deletion of the conserved sequence Gm-G and its effects on aminoacylation and conformation. J. Biochem. 1985, 97, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, T. New aspects of magnesium function: A key regulator in nucleosome self-assembly, chromatin folding and phase separation. Int. J. Mol. Sci. 2019, 20, 4232. [Google Scholar] [CrossRef] [PubMed]
- Hooper, M.; Hardy, K.; Handyside, A.; Hunter, S.; Monk, M. HPRT-deficient (Lesch–Nyhan) mouse embryos derived from germline colonization by cultured cells. Nature 1987, 326, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Tanase, J.; Mitani, T.; Udagawa, K.; Nishikawa, J.; Ohyama, T. Competence of an artificial bent DNA as a transcriptional activator in mouse ES cells. Mol. Biol. Rep. 2011, 38, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Naito, Y.; Hino, K.; Bono, H.; Ui-Tei, K. CRISPRdirect: Software for designing CRISPR/Cas guide RNA with reduced off-target sites. Bioinformatics 2015, 31, 1120–1123. [Google Scholar] [CrossRef]
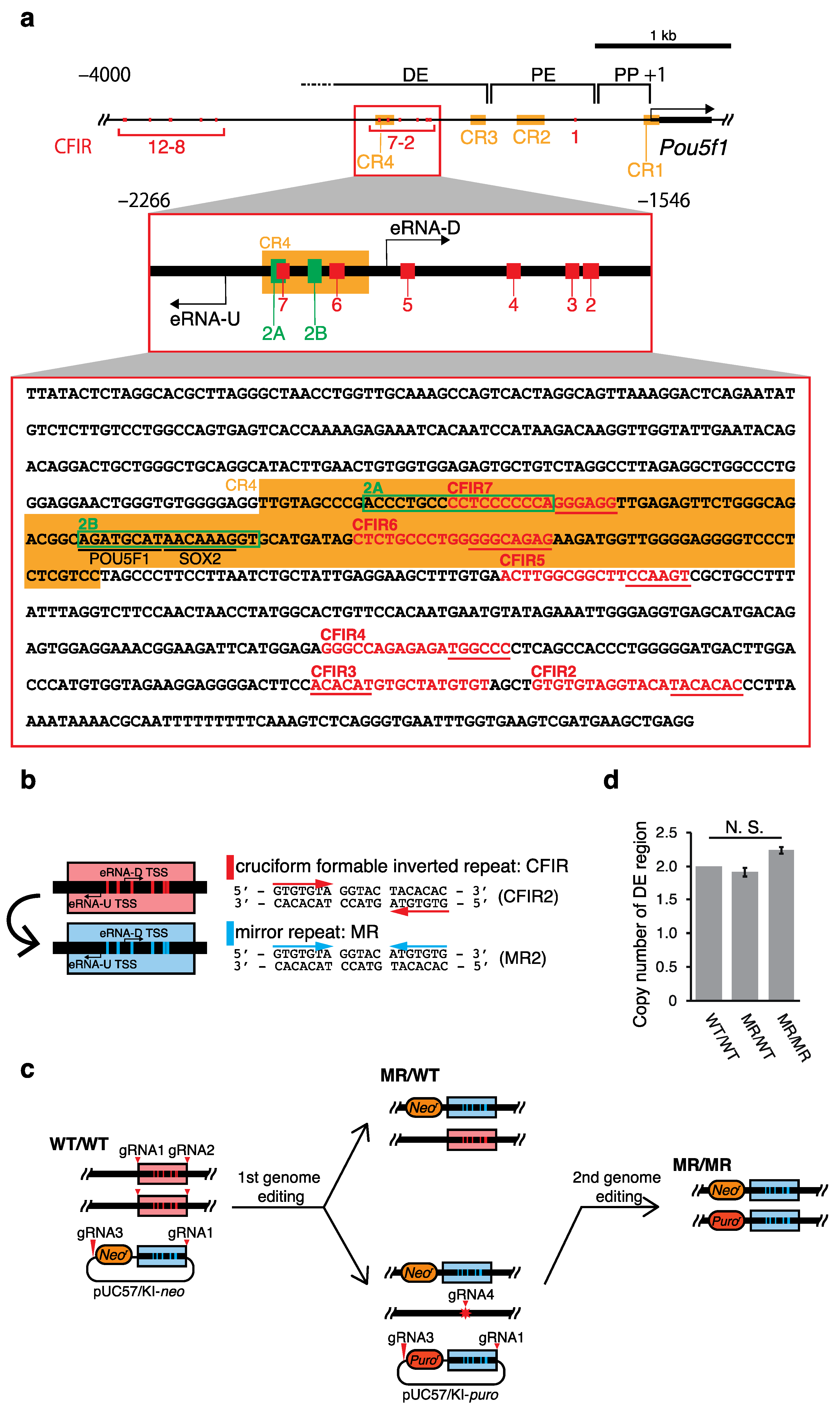

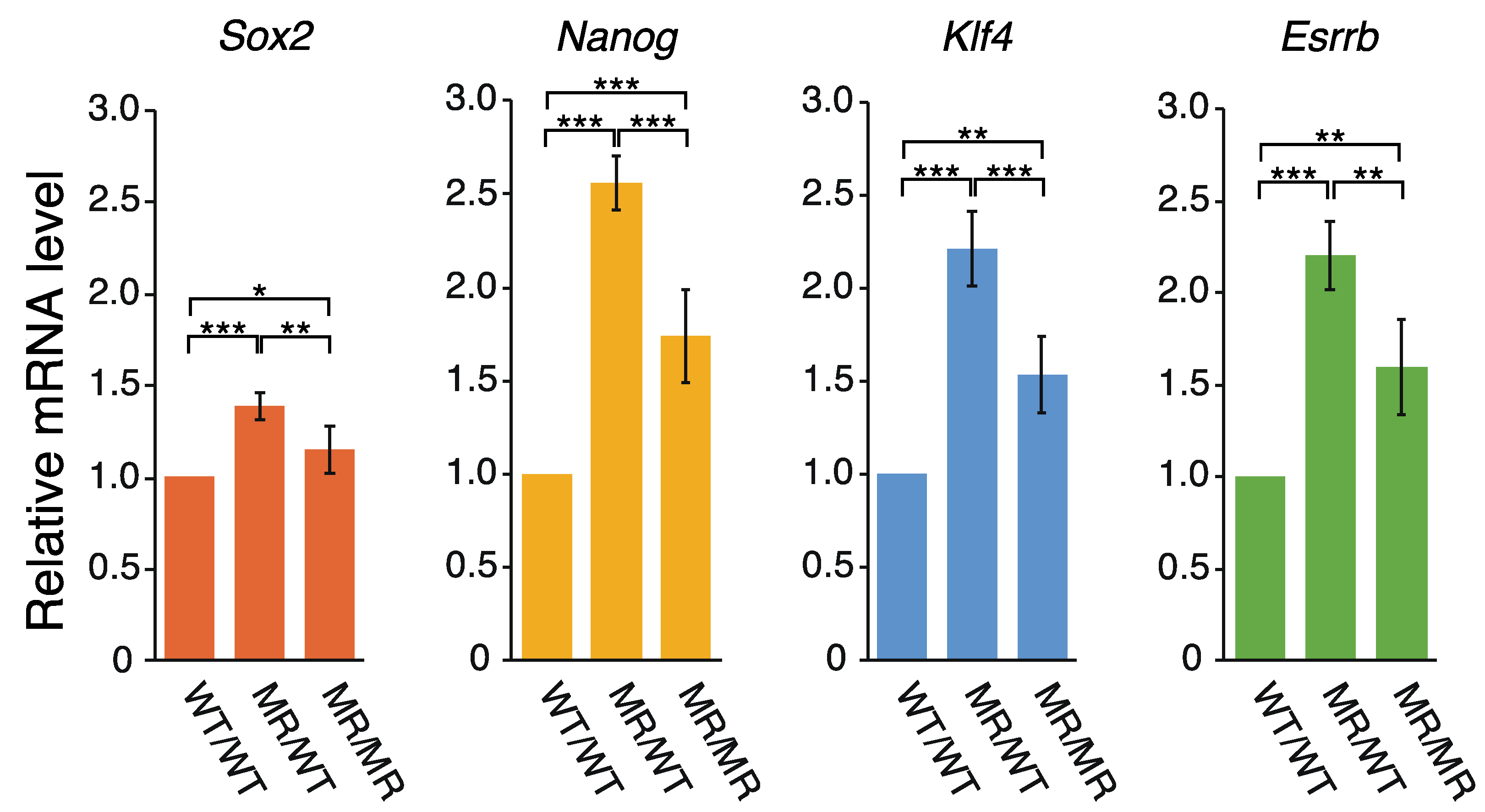
| Name | Position Relative to TSS (+1) | Structure (bp) | Sequence (Watson Strand) 1 | |||
|---|---|---|---|---|---|---|
| Start | End | Repeat Unit | Spacer | Total | ||
| CFIR2 | −1615 | −1634 | 7 | 6 | 20 | GTGTGTAGGTACATACACAC |
| atgtgtg | ||||||
| CFIR3 | −1639 | −1655 | 6 | 5 | 17 | ACACATGTGCTATGTGT |
| tgtgta | ||||||
| CFIR4 | −1710 | −1727 | 6 | 6 | 18 | GGGCCAGAGAGATGGCCC |
| accggg | ||||||
| CFIR5 | −1839 | −1856 | 6 | 6 | 18 | ACTTGGCGGCTTCCAAGT |
| ggttca | ||||||
| CFIR6 | −1925 | −1943 | 8 | 3 | 19 | CTCTGCCCTGGGGGCAGAG |
| cccgtctc | ||||||
| CFIR7 | −1992 | −2007 | 6 | 4 | 16 | CCTCCCCCCAGGGAGG |
| ccctcc | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamamoto, Y.; Miura, O.; Ohyama, T. Cruciform Formable Sequences within Pou5f1 Enhancer Are Indispensable for Mouse ES Cell Integrity. Int. J. Mol. Sci. 2021, 22, 3399. https://doi.org/10.3390/ijms22073399
Yamamoto Y, Miura O, Ohyama T. Cruciform Formable Sequences within Pou5f1 Enhancer Are Indispensable for Mouse ES Cell Integrity. International Journal of Molecular Sciences. 2021; 22(7):3399. https://doi.org/10.3390/ijms22073399
Chicago/Turabian StyleYamamoto, Yu, Osamu Miura, and Takashi Ohyama. 2021. "Cruciform Formable Sequences within Pou5f1 Enhancer Are Indispensable for Mouse ES Cell Integrity" International Journal of Molecular Sciences 22, no. 7: 3399. https://doi.org/10.3390/ijms22073399
APA StyleYamamoto, Y., Miura, O., & Ohyama, T. (2021). Cruciform Formable Sequences within Pou5f1 Enhancer Are Indispensable for Mouse ES Cell Integrity. International Journal of Molecular Sciences, 22(7), 3399. https://doi.org/10.3390/ijms22073399





