Abstract
Colorectal adenoma is linked to metabolic dysfunction. Metabolic dysfunction-associated fatty liver disease (MAFLD) has a precise definition and three subtypes, including non-obese MAFLD. We aimed to investigate the impact of MAFLD on the prevalence of colorectal adenoma by comparing it to non-alcoholic fatty liver disease (NAFLD) in health check-up examinees. This is a multicenter retrospective study. We enrolled 124 consecutive health check-up examinees who underwent colonoscopy. NAFLD and MAFLD were present in 58 and 63 examinees, respectively. Colorectal adenoma was diagnosed by biopsy. The impact of the MAFLD definition on the prevalence of colorectal adenoma was investigated by logistic regression, decision-tree, and random forest analyses. In logistic regression analysis, MAFLD was identified as the only independent factor associated with the presence of colorectal adenoma (OR 3.191; 95% CI 1.494–7.070; p = 0.003). MAFLD was also identified as the most important classifier for the presence of colorectal adenoma in decision-tree and random forest analyses (29 variable importance value). Among the three subtypes of MAFLD, non-obese MAFLD was the sole independent factor associated with the presence of colorectal adenoma (OR 3.351; 95% CI 1.589–7.262; p ≤ 0.001). Non-obese MAFLD was also the most important classifier for the presence of colorectal adenoma in decision-tree and random forest analyses (31 variable importance value). MAFLD, particularly non-obese MAFLD, is the most important factor associated with the presence of colorectal adenoma rather than NAFLD. Colonoscopy examination should be considered in patients with MAFLD, especially those who are non-obese.
1. Introduction
The prevalence of colorectal cancer is increasing, with over 1.8 million new colorectal cancer cases being reported each year [1]. Colorectal cancer ranks second in terms of mortality, and 881,000 deaths were estimated to have occurred in 2018 [1]. Most colorectal cancers develop through the adenoma-carcinoma sequence [2]. A multicenter post-polypectomy surveillance study showed that colonoscopic polypectomy significantly reduces the risk of death from colorectal cancer [3]. Thus, treatment of colorectal adenoma is likely to be an effective strategy to reduce the risk of death from this cancer. This decision can be informed by assessing the risk factors associated with colorectal adenoma.
Aging, gender, and family history increase the risk of colorectal cancer; however, these factors are unchangeable [4]. Many lifestyle-related factors have also been linked to colorectal cancer. These factors are modifiable, and meta-analyses demonstrate that high-risk factors for colorectal cancer are smoking, alcoholic intake, and metabolic dysfunction such as obesity, type 2 diabetes mellitus (T2DM), hypertension, and dyslipidemia [5,6]. NAFLD increases the risk not only for hepatocellular carcinoma but also of extrahepatic cancers including colorectal cancer [7]. Recent studies also showed that NAFLD is associated with an increased risk of colorectal adenoma and screening colonoscopy is recommended for patients with NAFLD [8,9]. However, NAFLD is a heterogeneous disease and can be diagnosed irrespective of the presence of metabolic dysfunction.
Recently, a panel of experts from 22 countries proposed a new definition for the diagnosis of metabolic dysfunction-associated fatty liver disease (MAFLD) [10]. The diagnosis of MAFLD is based on evidence of fatty liver in addition to one of (1) overweight/obesity, (2) the presence of T2DM, or (3) presence of metabolic dysregulation with at least two risk features including central obesity, pre-diabetes, hypertension, hyperlipidemia, and depressed high-density lipoprotein (HDL) cholesterol [10]. Accordingly, MAFLD, rather than NAFLD, is likely to be more associated with metabolic dysregulation-related events. In fact, MAFLD is reported to identify patients with significant hepatic fibrosis better than NAFLD [11,12]. However, it remains unclear whether MAFLD is superior to NAFLD as a factor associated with colorectal adenoma.
The aim of this study is to investigate the impact of MAFLD on colorectal adenoma by comparing it to NAFLD in health check-up examinees.
2. Results
2.1. Patients’ Characteristics
In all subjects, the median age was 59 years and the male ratio was 80.6% (Table 1). The median body mass index was 23.1 and the prevalence of fatty liver was 58.1% (72/124) of subjects. Severe fatty liver (fatty liver index >60) was seen in 19% and advanced hepatic fibrosis (NAFLD fibrosis score > 0.675, corresponding to F3-F4) was seen in 2% (Table 1). In all subjects, 37.9% were diagnosed with colorectal adenoma (Table 1).

Table 1.
Patients’ characteristics.
NAFLD and MAFLD were present in 80.6% and 87.5% of subjects with fatty liver (n = 72), respectively. Patients overlapping NAFLD and MAFLD comprised 69.4% (50/72) of the subjects with fatty liver (Figure 1).
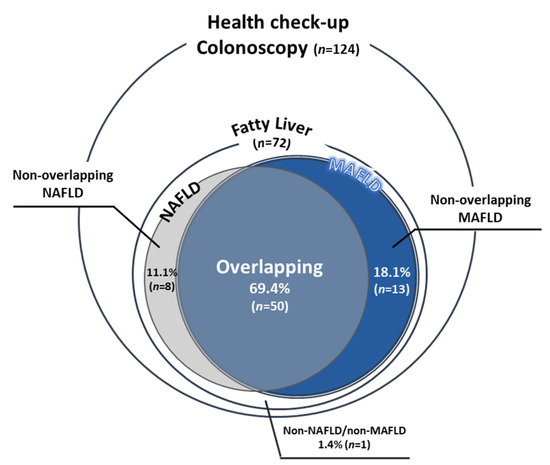
Figure 1.
The population of MAFLD and NAFLD. The Venn diagram indicates the proportion of patients with NAFLD (gray) and patients with MAFLD (blue).
Patients’ characteristics are summarized in Table 1. There was no significant difference in age, sex, and body mass index (BMI) between the NAFLD and MAFLD groups. In the MAFLD group, the prevalence of alcohol drinkers (men ≥30 gms/day, women ≥20 gms/day) was 20.6%. No significant difference was seen in the prevalence of ever-smoker, central obesity, T2DM, hypertension, dyslipidemia, and colorectal cancer in first-degree relatives between the 2 groups. There was no significant difference between the 2 groups in FIB-4 index, hemoglobin A1c (HbA1c) level, and serum levels of HDL cholesterol, triglycerides, and CRP (Table 1).
2.2. Logistic Regression Analysis for Colorectal Adenoma
We analyzed 6 factors including MAFLD, NAFLD, age, sex, alcohol intake, and smoking in the stepwise procedure. Although MAFLD and age were selected as explanatory variables for the logistic regression analysis, MAFLD was identified as the only independent factor associated with colorectal adenoma (odds ratios (OR) 3.191; 95% confidence intervals (CI) 1.494–7.070; p = 0.003; Figure 2A). On the other hand, NAFLD was not an independent factor associated with colorectal adenoma.
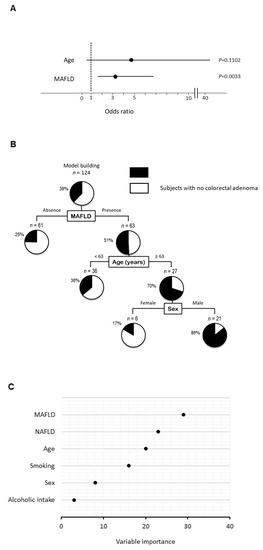
Figure 2.
Independent factors and profiles associated with the presence of colorectal adenoma. (A) Independent factors for the presence of colorectal adenoma analyzed by logistic regression analysis, (B) profiles for the presence of colorectal adenoma analyzed by decision-tree analysis. The pie graphs indicate the proportion of patients with colorectal adenoma (black) and patients with no colorectal adenoma (white), (C) distinguishing factors for the presence of colorectal adenoma analyzed by random forest analysis. The relative contributions of each variable to the presence of colorectal adenoma is expressed by variable importance.
2.3. Decision-Tree Analysis for Colorectal Adenoma
In a decision-tree algorithm, MAFLD was identified as the most important classifier for the presence of colorectal adenoma. Colorectal adenoma was observed in 51% of subjects with MAFLD, while colorectal adenoma was observed in 25% of subjects in the absence of MAFLD (Figure 2B). The second and third classifiers were age and sex, respectively. Colorectal adenoma was observed in 86% of subjects with MAFLD, ≥63 years old, and men. On the other hand, NAFLD was not identified as a classifier for the presence of colorectal adenoma.
2.4. Random Forest Analysis for Colorectal Adenoma
In a random forest analysis for the presence of colorectal adenoma, MAFLD was identified as the distinguishing factor with the highest variable importance. NAFLD, age, and sex were the second, third, and fourth distinguishing factors, respectively (Figure 2C).
2.5. Patients’ Characteristics among Subtypes of MALFD
Patients’ characteristics for subtypes of MALFD are summarized in Table 2. There was no significant difference in age, sex, and BMI among the obese-MAFLD, non-obese MAFLD, and T2DM-MAFLD groups. No significant difference was seen in the prevalence of daily alcohol intake, ever-smoker, central obesity, hypertension, dyslipidemia, and colorectal cancer in first-degree relatives among the 3 groups. In the T2DM-MAFLD group, HbA1c level was significantly higher than that in the obese and non-obese MAFLD groups. There was no significant difference in FIB-4 index, serum HDL cholesterol, triglycerides, and CRP levels among the 3 groups (Table 2).

Table 2.
Patients’ characteristics in subtypes of MAFLD.
2.6. Logistic Regression Analysis for Colorectal Adenoma Using Subtypes of MALFD
We analyzed 7 factors, namely, obese-MAFLD, non-obese MAFLD, T2DM-MAFLD, age, sex, alcoholic intake, and smoking in the stepwise procedure. In the stepwise procedure, non-obese MAFLD was only selected as an explanatory variable for the logistic regression analysis. Non-obese MAFLD was identified as the only independent factor associated with colorectal adenoma (OR 3.351; 95% CI 1.589–7.262; p ≤ 0.001; Figure 3A). On the other hand, obese- and T2DM-MALFD were not independent factors associated with colorectal adenoma.
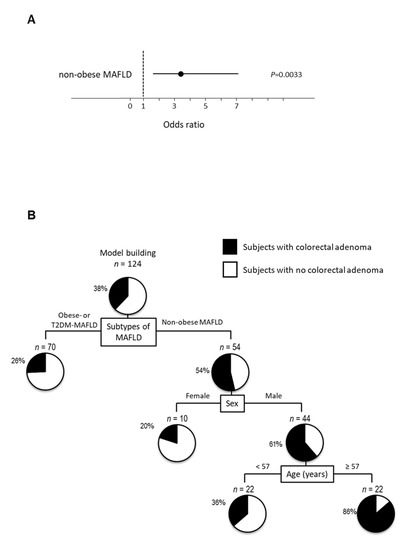
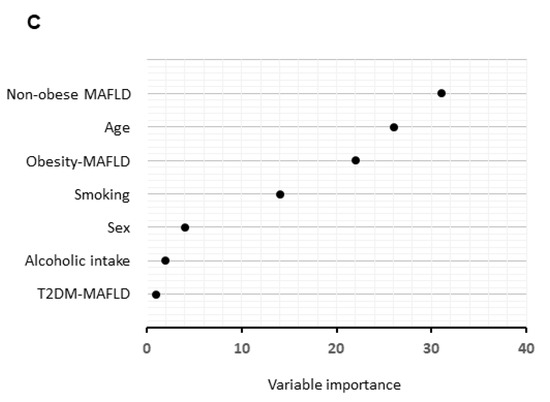
Figure 3.
Independent factors and profiles associated with the presence of colorectal adenoma according to subtypes of MAFLD. (A) Independent factors for the presence of colorectal adenoma analyzed by logistic regression analysis, (B) profiles for the presence of colorectal adenoma analyzed by decision-tree analysis. The pie graphs indicate the proportion of patients with colorectal adenoma (black) and patients with no colorectal adenoma (white), (C) distinguishing factors for the presence of colorectal adenoma analyzed by random forest analysis. The relative contributions of each variable to the presence of colorectal adenoma is expressed by variable importance.
2.7. Decision-Tree Analysis for Colorectal Adenoma Using Subtypes of MALFD
In a decision-tree algorithm, non-obese MAFLD was identified as the most important classifier for the presence of colorectal adenoma. Colorectal adenoma was observed in 54% of subjects with MAFLD, while colorectal adenoma was observed in 26% of subjects with obese- or T2DM-MAFLD (Figure 3B). The second and third classifiers were sex and age, respectively. Colorectal adenoma was observed in 86% of subjects with non-obese MAFLD, men, and ≥57 years old. On the other hand, both obesity and T2DM-MAFLD were not identified as a classifier for the presence of colorectal adenoma.
2.8. Random Forest Analysis for Colorectal Adenoma
In a random forest analysis for the presence of colorectal adenoma in MAFLD, only non-obese MAFLD was identified as a distinguishing factor with the highest variable importance. Age and obese MAFLD were the second and third factors, respectively (Figure 3C).
3. Discussion
In this study, we demonstrate that MAFLD is the only independent factor associated with the presence of colorectal adenoma. We also show that MAFLD, but not NAFLD, is the most important factor associated with the presence of colorectal adenoma in decision-tree and random forest analyses. Furthermore, non-obese MAFLD was associated with the presence of colorectal adenoma among the three subtypes of MAFLD.
In this study, the prevalence of NAFLD was 46.8% among subjects who underwent colonoscopy examination (58/124). The prevalence of NAFLD has been reported to be between 40 and 52% in subjects who undergo colonoscopy [13,14,15]. Metabolic disorders including NAFLD are risk factors for colorectal adenoma [16,17]. In this study, the prevalence of colorectal adenoma was 50.0%. Wong et al. reported the prevalence of colorectal adenoma as 51.6% among subjects who underwent colonoscopy examination [9]. Furthermore, systematic reviews and meta-analyses reported that the prevalence of colorectal adenoma ranged from 30 to 50% in asymptomatic adults [8,18]. Thus, in this study, the characteristics of enrolled patients with NAFLD and the prevalence of colorectal adenoma were similar to previous reports.
MAFLD was an independent factor for colorectal adenoma in this study. Decision-tree and random forest analyses also revealed that MAFLD, but not NAFLD, was the most important factor for the presence of colorectal adenoma. Recently, several studies have shown the superiority of MAFLD over NAFLD for the identification of patients with significant hepatic fibrosis, cardiovascular event, and chronic kidney disease [11,12,19,20]. Moreover, we now demonstrate the superiority of MAFLD over NAFLD to identify patients with colorectal adenoma. Although the reason for the superiority of MAFLD remains unclear, a possible explanation is a difference in the diagnostic criteria between the two definitions. NAFLD can be diagnosed regardless of the presence of metabolic dysregulation [21]. However, the presence of metabolic dysfunction is a necessary inclusion criterion for the diagnosis of MAFLD [10,22]. Metabolic dysfunction including obesity and T2DM are well-known risk factors for colorectal adenoma [23,24]. Furthermore, the diagnosis of MAFLD is independent of alcoholic intake, which is also a risk factor for colorectal adenoma [25]. Thus, MAFLD includes risk factors for colorectal adenoma, and, therefore, MAFLD may identify patients with colorectal adenoma better than NAFLD.
MAFLD consists of three subtypes, namely, obese-MAFLD, non-obese MAFLD, and T2DM-MAFLD [10,22]. We found that non-obese MAFLD was the independent factor for the presence of colorectal adenoma, while obese-MAFLD and T2DM-MAFLD were not. There was no significant difference in the number of complicating metabolic abnormalities or in the alcohol intake among the three subgroups of MAFLD. Thus, it remains unclear which factor is responsible for colorectal adenoma in the non-obese MAFLD group. Recently, non-obese NAFLD has been reported as a major subtype of NAFLD [26]. Sarcopenia and alterations in gut microbiota are pathophysiological factors associated with the development of non-obese NAFLD [27,28]. These factors are also known as risks for colorectal adenoma [29,30,31]. In addition, the transmembrane 6 superfamily member 2 (TM6SF2) gene polymorphism is associated with lean NAFLD [32], while it is also reported to be associated with colorectal adenoma [25]. Taken together, possible factors for an association between non-obese MAFLD and colorectal adenoma include alterations in skeletal muscle mass, gut microbiota, and/or TM6SF2 gene polymorphism.
There are several limitations to this study. First, this study is retrospective, with a small sample size. Second, all participants were of Asian ancestry. Third, we did not evaluate factors associated with colorectal cancer, including dietary habits and physical activity. Fourth, since all subjects were health check-up examinees, the prevalence of severe steatosis and advanced fibrosis was low in this cohort. Therefore, we could not evaluate the impact of severity of hepatic steatosis and fibrosis on the prevalence of colorectal adenomas. Fifth, we also could not evaluate the impact of MAFLD on colorectal cancer, because of the small number of patients with colorectal cancer in this cohort (2/126). Accordingly, further studies should be designed in an international prospective study with a large sample size to more comprehensively evaluate the effects of MAFLD on the development of colorectal adenomas/adenocarcinomas using various factors including lifestyle habits and hepatic steatosis/fibrosis using FibroMax indexes [33,34,35].
In conclusion, we found that MAFLD was the only independent factor associated with the presence of colorectal adenoma. In both decision-tree and random forest analyses, these results were confirmed, while NAFLD was not an independent factor. In addition, we identified that non-obese MAFLD was associated with the presence of colorectal adenoma among the three subtypes of MAFLD. Since the identification of colorectal adenoma is an effective strategy to reduce the death rate from colorectal cancer, we suggest that colonoscopy examination is better considered in patients with MAFLD, particularly those who are non-obese.
4. Materials and Methods
4.1. Study Design and Ethics
This study was designed as a multicenter cross-sectional retrospective study in Japan. The protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as reflected by prior approval from the institutional review board of Kurume University School of Medicine (ID 20114). This research was performed in accordance with relevant guidelines and regulations. An opt-out approach was used to obtain informed consent from patients, and personal information was protected during data collection.
4.2. Study Population and Selection of Patients for Analysis
We enrolled health check-up examinees who met the following inclusion criteria at the Kurume University Hospital, Kumamoto Central Hospital, and Kurate Hospital in Japan from April 2018 to March 2020 (n = 130): (1) subjects who were 20 years old or greater, and (2) subjects who underwent colonoscopy examination from 2018 to 2020. Of these, we have excluded subjects with (1) incomplete colonoscopy examination (n = 1), (2) positive results of hepatitis B virus surface antigen (n = 1) or hepatitis C virus antibody (n = 2), and (3) colorectal cancer (n = 2). Thus, a total of 124 subjects were analyzed in this study (Figure 4). All patients were Asian. Colonoscopy examination was performed as a part of their clinical review. We excluded patients with colorectal cancer (n = 2) or with an incomplete examination, defined as an endoscope not reaching the cecum as documented by a picture of the ileocecal valve (n = 1). Finally, 124 health check-up examinees were analyzed in this study.
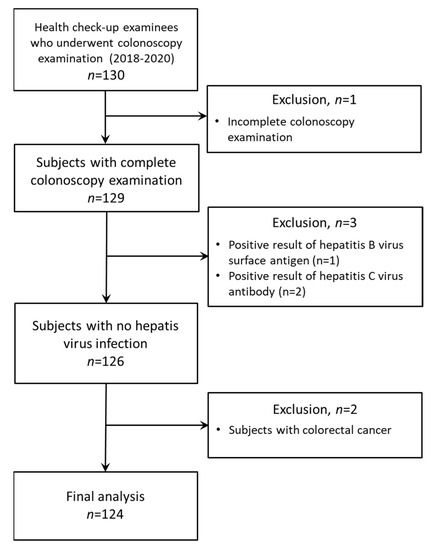
Figure 4.
A flow chart for study populations.
4.3. Data Collection
All data were collected retrospectively from medical records at the time of colonoscopy. The following information was obtained using a self-reported questionnaire: age, sex, comorbidity, medication use, and colorectal cancer in first-degree relatives. At the clinical review, we obtained the following data: BMI, waist circumference, blood pressure, presence or absence of T2DM, hypertension, and dyslipidemia; these were diagnosed according to standard criteria [10,36,37,38]. We obtained the data for current alcohol intake and smoking. Daily alcohol intake habit was defined as men ≥30 gms/day or women ≥20 gms/day.
4.4. Biochemical Analysis
Patients fasted overnight before collection of blood samples for the following tests: complete blood count, aspartate aminotransferase, alanine aminotransferase, lactate dehydrogenase, alkaline phosphatase, γ-glutamyl transpeptidase, albumin, total bilirubin, HDL cholesterol, triglycerides, fasting glucose, HbA1c, and C-reactive protein. FIB-4 index was calculated using age, serum levels of AST, ALT, and platelet count as previously described [39].
4.5. Diagnosis of Fatty Liver and Assessment for the Severity of Steatosis and Hepatic Fibrosis
The diagnosis of fatty liver was based on the presence of any of the following findings on abdominal ultrasonography: increased hepato-renal contrast, increased echogenicity of liver parenchyma, unclear visualization of the intrahepatic vessels, and/or impaired visualization of the diaphragm as previously described [40].
The severity of hepatic steatosis was assessed by fatty liver index as previously described [41]. The severity of hepatic fibrosis was assessed by FIB-4 index and NAFLD fibrosis score as previously described [12].
4.6. Diagnosis of NAFLD
The diagnosis of NAFLD was according to the EASL-EASD-EASO and American Association for the Study of Liver Diseases Clinical Practice Guidelines for the Management of NAFLD [42,43]: (1) fatty liver by abdominal ultrasonography, (2) alcohol intake no greater than 30 gms/day for men and 20 gms/day for women, and (3) no competing etiologies for fatty liver or coexisting causes of chronic liver disease [42,43].
4.7. Diagnosis of MAFLD
MAFLD was diagnosed according to the criteria proposed in 2020 by an international expert panel [10]. The criteria include evidence of fatty liver, in addition to one of the following: obesity, presence of T2DM, or non-obesity with evidence of metabolic dysregulation. Since all patients were Asian, BMI and waist circumstance were evaluated using cut-off values for Asians [10]. Obesity was defined as BMI ≥23 kg/m2 in this Asian cohort and T2DM was defined as HbA1c ≥6.5% or specific drug treatment. Metabolic dysregulation was defined as the presence of at least two metabolic risk abnormalities: (1) Central obesity (waist circumstance ≥90/80 cm in men and women), (2) blood pressure ≥130 mmHg or specific drug treatment, (3) plasma triglycerides ≥150 mg/dL or specific drug treatment, (4) plasma HDL-cholesterol <40 mg/dL for men and <50 mg/dL for women or specific drug treatment, and (5) prediabetes (fasting glucose levels 100 to 125 mg/dL or HbA1c 5.7–6.4%) [10]. Although the homeostasis model assessment of insulin resistance score and plasma high-sensitivity C-reactive protein level are metabolic risk abnormalities in the MAFLD criteria [10], these were not available in our dataset.
4.8. Colonoscopy Examination and Diagnosis of Colorectal Adenoma
Colonoscopy examination (CF H260AZI/PCF H290ZI; Olympus, Tokyo, Japan) was performed by endoscopists with experience of performing more than 1000 procedures. Subjects were given polyethylene glycol for bowel preparation according to the manufacturer’s instructions. A complete examination was defined as an endoscope reaching the cecum as documented by a picture of the ileocecal valve. All identified colorectal polyps were removed and were tubular adenomas, which were diagnosed by pathological findings. This is a retrospective study and diagnosis of colorectal adenoma had already been made at the entry of this study.
In all colonoscopy examinations, the quality of bowel preparation was graded as good (no or small volume of clear liquid, with >95% of the surface seen). The withdrawal time of the colonoscopy procedure was at least 6 min to minimize the chance of missing lesions [44]. Incomplete examinations were excluded from the analysis.
4.9. Statistical Analysis
Continuous variables are expressed as median and range or number. Categorical variables are expressed as frequencies and percentages. The differences between groups were analyzed using the Wilcoxon rank-sum test for continuous variables and the Fisher’s exact test for categorical variables. A logistic regression model was used to identify independent factors associated with the presence of colorectal adenoma. Data are expressed as OR and 95% CI.
A decision-tree algorithm was constructed to reveal profiles associated with the presence of colorectal adenoma as previously described [12,40]. A random forest analysis was used to identify factors that distinguished for the presence of colorectal adenoma as previously described [45,46]. The variable importance value which reflects the relative contribution of each variable to the model was estimated by randomly permuting its values and recalculating the predictive accuracy of the model. p < 0.05 was considered to indicate statistical significance. Data were analyzed using the JMP Pro15 (SAS Institute Inc., Cary, NC, USA).
5. Conclusions
MAFLD, particularly non-obese MAFLD, is the most important factor associated with the presence of colorectal adenoma rather than NAFLD. Colonoscopy examination should be considered in patients with MAFLD, especially those who are non-obese.
Author Contributions
Conceptualization, S.F., H.A., T.T., and D.N.; methodology, D.N., and K.K.; software, A.O., T.K., and H.A.; validation, M.E., J.G., T.T., and H.K. (Hidefumi Kuroki); formal analysis, H.K. (Hidefumi Kuroki), K.M., and K.K.; investigation, S.F. and H.K. (Hidemichi Kawata); resources, R.N., K.M., and T.N.; data curation, S.F., D.N., T.K., and H.K. (Hidemichi Kawata); writing—original draft preparation, M.E., J.G., K.M., and T.T.; writing—review and editing, M.E., J.G., K.M., and T.T.; visualization, R.N.; supervision, M.E., J.G., K.M., and T.T.; project administration, A.O., J.G., and T.N.; funding acquisition, T.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (C) JP20K08395.
Institutional Review Board Statement
The protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as reflected by prior approval from the institutional review board of Kurume University School of Medicine (ID 20114).
Informed Consent Statement
An opt-out approach was used to obtain informed consent from patients and personal information was protected during data collection.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
We thank Toshiaki Tanaka (Kurate Hospital) and Masafumi Inoue (Kumamoto Central Hospital) for providing health check-up data.
Conflicts of Interest
T.K. received lecture fees from Mitsubishi Tanabe Pharma Corporation and Otsuka Pharmaceutical Co., Ltd. The other authors have no conflicts of interest relevant to this publication.
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Hill, M.J.; Morson, B.C.; Bussey, H.J. Aetiology of adenoma—Carcinoma sequence in large bowel. Lancet 1978, 1, 245–247. [Google Scholar] [CrossRef]
- Zauber, A.G.; Winawer, S.J.; O’Brien, M.J.; Lansdorp-Vogelaar, I.; van Ballegooijen, M.; Hankey, B.F.; Shi, W.; Bond, J.H.; Schapiro, M.; Panish, J.F.; et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N. Engl. J. Med. 2012, 366, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Dekker, E.; Tanis, P.J.; Vleugels, J.L.A.; Kasi, P.M.; Wallace, M.B. Colorectal cancer. Lancet 2019, 394, 1467–1480. [Google Scholar] [CrossRef]
- Esposito, K.; Chiodini, P.; Colao, A.; Lenzi, A.; Giugliano, D. Metabolic syndrome and risk of cancer: A systematic review and meta-analysis. Diabetes Care 2012, 35, 2402–2411. [Google Scholar] [CrossRef] [PubMed]
- Ben, Q.; An, W.; Jiang, Y.; Zhan, X.; Du, Y.; Cai, Q.C.; Gao, J.; Li, Z. Body mass index increases risk for colorectal adenomas based on meta-analysis. Gastroenterology 2012, 142, 762–772. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhao, X.; Chen, S.; Wang, Y.; Cao, L.; Liao, W.; Sun, Y.; Wang, X.; Zheng, Y.; Wu, S.; et al. Associations between nonalcoholic fatty liver disease and cancers in a large cohort in China. Clin. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef]
- Chen, W.; Wang, M.; Jing, X.; Wu, C.; Zeng, Y.; Peng, J.; Cai, X. High risk of colorectal polyps in men with non-alcoholic fatty liver disease: A systematic review and meta-analysis. J. Gastroenterol. Hepatol. 2020, 35, 2051–2065. [Google Scholar] [CrossRef]
- Wong, V.W.; Wong, G.L.; Tsang, S.W.; Fan, T.; Chu, W.C.; Woo, J.; Chan, A.W.; Choi, P.C.; Chim, A.M.; Lau, J.Y.; et al. High prevalence of colorectal neoplasm in patients with non-alcoholic steatohepatitis. Gut 2011, 60, 829–836. [Google Scholar] [CrossRef]
- Eslam, M.; Newsome, P.N.; Sarin, S.K.; Anstee, Q.M.; Targher, G.; Romero-Gomez, M.; Zelber-Sagi, S.; Wong, V.W.-S.; Dufour, J.-F.; Schattenberg, J.M.; et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 2020, 73, 202–209. [Google Scholar] [CrossRef]
- Lin, S.; Huang, J.; Wang, M.; Kumar, R.; Liu, Y.; Liu, S.; Wu, Y.; Wang, X.; Zhu, Y. Comparison of MAFLD and NAFLD diagnostic criteria in real world. Liver Int. 2020, 40, 2082–2089. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, S.; Eslam, M.; Kawaguchi, T.; Tsutsumi, T.; Nakano, D.; Yoshinaga, S.; Takahashi, H.; Anzai, K.; George, J.; Torimura, T. MAFLD identifies patients with significant hepatic fibrosis better than NAFLD. Liver Int. 2020, 40, 3018–3030. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.W.; Leu, H.B.; Wang, Y.J.; Luo, J.C.; Lin, H.C.; Lee, F.Y.; Chan, W.L.; Lin, J.K.; Chang, F.Y. Patients with nonalcoholic fatty liver disease have higher risk of colorectal adenoma after negative baseline colonoscopy. Colorectal Dis. 2013, 15, 830–835. [Google Scholar] [CrossRef]
- Li, Y.; Liu, S.; Gao, Y.; Ma, H.; Zhan, S.; Yang, Y.; Xin, Y.; Xuan, S. Association between NAFLD and risk of colorectal adenoma in Chinese han population. J. Clin. Transl. Hepatol. 2019, 7, 99–105. [Google Scholar] [CrossRef]
- Stadlmayr, A.; Aigner, E.; Steger, B.; Scharinger, L.; Lederer, D.; Mayr, A.; Strasser, M.; Brunner, E.; Heuberger, A.; Hohla, F.; et al. Nonalcoholic fatty liver disease: An independent risk factor for colorectal neoplasia. J. Intern. Med. 2011, 270, 41–49. [Google Scholar] [CrossRef]
- Kim, B.C.; Shin, A.; Hong, C.W.; Sohn, D.K.; Han, K.S.; Ryu, K.H.; Park, B.J.; Nam, J.H.; Park, J.W.; Chang, H.J.; et al. Association of colorectal adenoma with components of metabolic syndrome. Cancer Causes Control 2012, 23, 727–735. [Google Scholar] [CrossRef]
- Kim, J.H.; Lim, Y.J.; Kim, Y.H.; Sung, I.K.; Shim, S.G.; Oh, S.O.; Park, S.S.; Yang, S.; Son, H.J.; Rhee, P.L.; et al. Is metabolic syndrome a risk factor for colorectal adenoma? Cancer Epidemiol. Biomark. Prev. 2007, 16, 1543–1546. [Google Scholar] [CrossRef]
- Mantovani, A.; Dauriz, M.; Byrne, C.D.; Lonardo, A.; Zoppini, G.; Bonora, E.; Targher, G. Association between nonalcoholic fatty liver disease and colorectal tumours in asymptomatic adults undergoing screening colonoscopy: A systematic review and meta-analysis. Metabolism 2018, 87, 1–12. [Google Scholar] [CrossRef]
- Lee, H.; Lee, Y.H.; Kim, S.U.; Kim, H.C. Metabolic dysfunction-associated fatty liver disease and incident cardiovascular disease risk: A nationwide cohort study. Clin. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef]
- Sun, D.Q.; Jin, Y.; Wang, T.Y.; Zheng, K.I.; Rios, R.S.; Zhang, H.Y.; Targher, G.; Byrne, C.D.; Yuan, W.J.; Zheng, M.H. MAFLD and risk of CKD. Metabolism 2021, 115, 154433. [Google Scholar] [CrossRef]
- Watanabe, S.; Hashimoto, E.; Ikejima, K.; Uto, H.; Ono, M.; Sumida, Y.; Seike, M.; Takei, Y.; Takehara, T.; Tokushige, K.; et al. Evidence-based clinical practice guidelines for nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatol. Res. 2015, 45, 363–377. [Google Scholar] [CrossRef]
- Eslam, M.; Sarin, S.K.; Wong, V.W.; Fan, J.G.; Kawaguchi, T.; Ahn, S.H.; Zheng, M.H.; Shiha, G.; Yilmaz, Y.; Gani, R.; et al. The Asian Pacific association for the study of the liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol. Int. 2020, 14, 889–919. [Google Scholar] [CrossRef]
- Ottaviano, L.F.; Li, X.; Murray, M.; Frye, J.T.; Lung, B.E.; Zhang, Y.Y.; Yang, J.; Taub, E.M.; Bucobo, J.C.; Buscaglia, J.M.; et al. Type 2 diabetes impacts colorectal adenoma detection in screening colonoscopy. Sci. Rep. 2020, 10, 7793. [Google Scholar] [CrossRef]
- Kim, T.J.; Kim, J.E.; Choi, Y.H.; Hong, S.N.; Kim, Y.H.; Chang, D.K.; Rhee, P.L.; Kim, M.J.; Jung, S.H.; Son, H.J. Obesity-related parameters and colorectal adenoma development. J. Gastroenterol. 2017, 52, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.Z.; Wang, Y.M.; Zhou, Q.Y.; Zhu, K.F.; Yu, C.H.; Li, Y.M. Systematic review with meta-analysis: Alcohol consumption and the risk of colorectal adenoma. Aliment. Pharmacol. Ther. 2014, 40, 325–337. [Google Scholar] [CrossRef]
- Ye, Q.; Zou, B.; Yeo, Y.H.; Li, J.; Huang, D.Q.; Wu, Y.; Yang, H.; Liu, C.; Kam, L.Y.; Tan, X.X.E.; et al. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 739–752. [Google Scholar] [CrossRef]
- Shida, T.; Oshida, N.; Suzuki, H.; Okada, K.; Watahiki, T.; Oh, S.; Kim, T.; Isobe, T.; Okamoto, Y.; Ariizumi, S.I.; et al. Clinical and anthropometric characteristics of non-obese non-alcoholic fatty liver disease subjects in Japan. Hepatol. Res. 2020, 50, 1032–1046. [Google Scholar] [CrossRef]
- Duarte, S.M.B.; Stefano, J.T.; Miele, L.; Ponziani, F.R.; Souza-Basqueira, M.; Okada, L.; de Barros, F.G.C.; Toda, K.; Mazo, D.F.C.; Sabino, E.C.; et al. Gut microbiome composition in lean patients with NASH is associated with liver damage independent of caloric intake: A prospective pilot study. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 369–384. [Google Scholar] [CrossRef]
- Yoo, K.M.; Yang, H.J.; Park, S.K.; Jung, Y.S.; Park, J.H.; Park, D.I.; Sohn, C.I. Skeletal muscle mass and risk of advanced adenoma in surveillance colonoscopy. J. Gastroenterol. Hepatol. 2020, 35, 2088–2095. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Vogtmann, E.; Ahlquist, D.A.; Devens, M.E.; Kisiel, J.B.; Taylor, W.R.; White, B.A.; Hale, V.L.; Sung, J.; Chia, N.; et al. Fecal metabolomic signatures in colorectal adenoma patients are associated with gut microbiota and early events of colorectal cancer pathogenesis. mBio 2020, 11. [Google Scholar] [CrossRef]
- Saito, K.; Koido, S.; Odamaki, T.; Kajihara, M.; Kato, K.; Horiuchi, S.; Adachi, S.; Arakawa, H.; Yoshida, S.; Akasu, T.; et al. Metagenomic analyses of the gut microbiota associated with colorectal adenoma. PLoS ONE 2019, 14, e0212406. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Esmaili, S.; Rogers, G.B.; Bugianesi, E.; Petta, S.; Marchesini, G.; Bayoumi, A.; Metwally, M.; Azardaryany, M.K.; Coulter, S.; et al. Lean NAFLD: A distinct entity shaped by differential metabolic adaptation. Hepatology 2020, 71, 1213–1227. [Google Scholar] [CrossRef]
- Morra, R.; Munteanu, M.; Imbert-Bismut, F.; Messous, D.; Ratziu, V.; Poynard, T. FibroMAX: Towards a new universal biomarker of liver disease? Expert Rev. Mol. Diagn. 2007, 7, 481–490. [Google Scholar] [CrossRef]
- Munteanu, M.; Ratziu, V.; Morra, R.; Messous, D.; Imbert-Bismut, F.; Poynard, T. Noninvasive biomarkers for the screening of fibrosis, steatosis and steatohepatitis in patients with metabolic risk factors: FibroTest-fibromax experience. J. Gastrointestin. Liver Dis. 2008, 17, 187–191. [Google Scholar]
- Gudowska, M.; Wojtowicz, E.; Cylwik, B.; Gruszewska, E.; Chrostek, L. The distribution of liver steatosis, fibrosis, steatohepatitis and inflammation activity in alcoholics according to FibroMax test. Adv. Clin. Exp. Med. 2015, 24, 823–827. [Google Scholar] [CrossRef]
- Anonymous. 2018 practice guidelines for the management of arterial hypertension of the European society of hypertension and the European society of cardiology. ESH/ESC task force for the management of arterial hypertension. Erratum. J. Hypertens. 2019, 37, 456. [Google Scholar] [CrossRef]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; Hansen, T.B.; et al. 2019 ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur. Heart J. 2020, 41, 255–323. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; de Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Yamamura, S.; Kawaguchi, T.; Nakano, D.; Tomiyasu, Y.; Yoshinaga, S.; Doi, Y.; Takahashi, H.; Anzai, K.; Eguchi, Y.; Torimura, T.; et al. Profiles of advanced hepatic fibrosis evaluated by FIB-4 index and shear wave elastography in health checkup examinees. Hepatol. Res. 2020, 50, 199–213. [Google Scholar] [CrossRef]
- Hirata, A.; Sugiyama, D.; Kuwabara, K.; Hirata, T.; Tsutatani, H.; Funamoto, M.; Watanabe, K.; Miyamatsu, N.; Okamura, T. Fatty liver index predicts incident diabetes in a Japanese general population with and without impaired fasting glucose. Hepatol. Res. 2018, 48, 708–716. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American association for the study of liver diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Barclay, R.L.; Vicari, J.J.; Doughty, A.S.; Johanson, J.F.; Greenlaw, R.L. Colonoscopic withdrawal times and adenoma detection during screening colonoscopy. N. Engl. J. Med. 2006, 355, 2533–2541. [Google Scholar] [CrossRef] [PubMed]
- Noda, Y.; Kawaguchi, T.; Korenaga, M.; Yoshio, S.; Komukai, S.; Nakano, M.; Niizeki, T.; Koga, H.; Kawaguchi, A.; Kanto, T.; et al. High serum interleukin-34 level is a predictor of poor prognosis in patients with non-viral hepatocellular carcinoma. Hepatol. Res. 2019, 49, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, T.; Tokushige, K.; Hyogo, H.; Aikata, H.; Nakajima, T.; Ono, M.; Kawanaka, M.; Sawada, K.; Imajo, K.; Honda, K.; et al. A data mining-based prognostic algorithm for NAFLD-related hepatoma patients: A nationwide study by the Japan study group of NAFLD. Sci. Rep. 2018, 8, 10434. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).