The Bone-Protective Effect of Genistein in the Animal Model of Bilateral Ovariectomy: Roles of Phytoestrogens and PTH/PTHR1 Against Post-Menopausal Osteoporosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
Medicine
Materials
2.2. Experimental Protocols
2.3. Determination of Bone Mineral Density
2.4. Femoral Mechanical Testing
2.5. Serum and Urine Mineral Analysis
2.6. Serum Biochemistry
2.7. RNA Isolation and Reverse Transcription-PCR
2.8. Western Blot
2.9. Immunohistochemistry
2.10. Statistical Analysis
3. Results
3.1. Body Weights
3.2. Bone Mineral Density
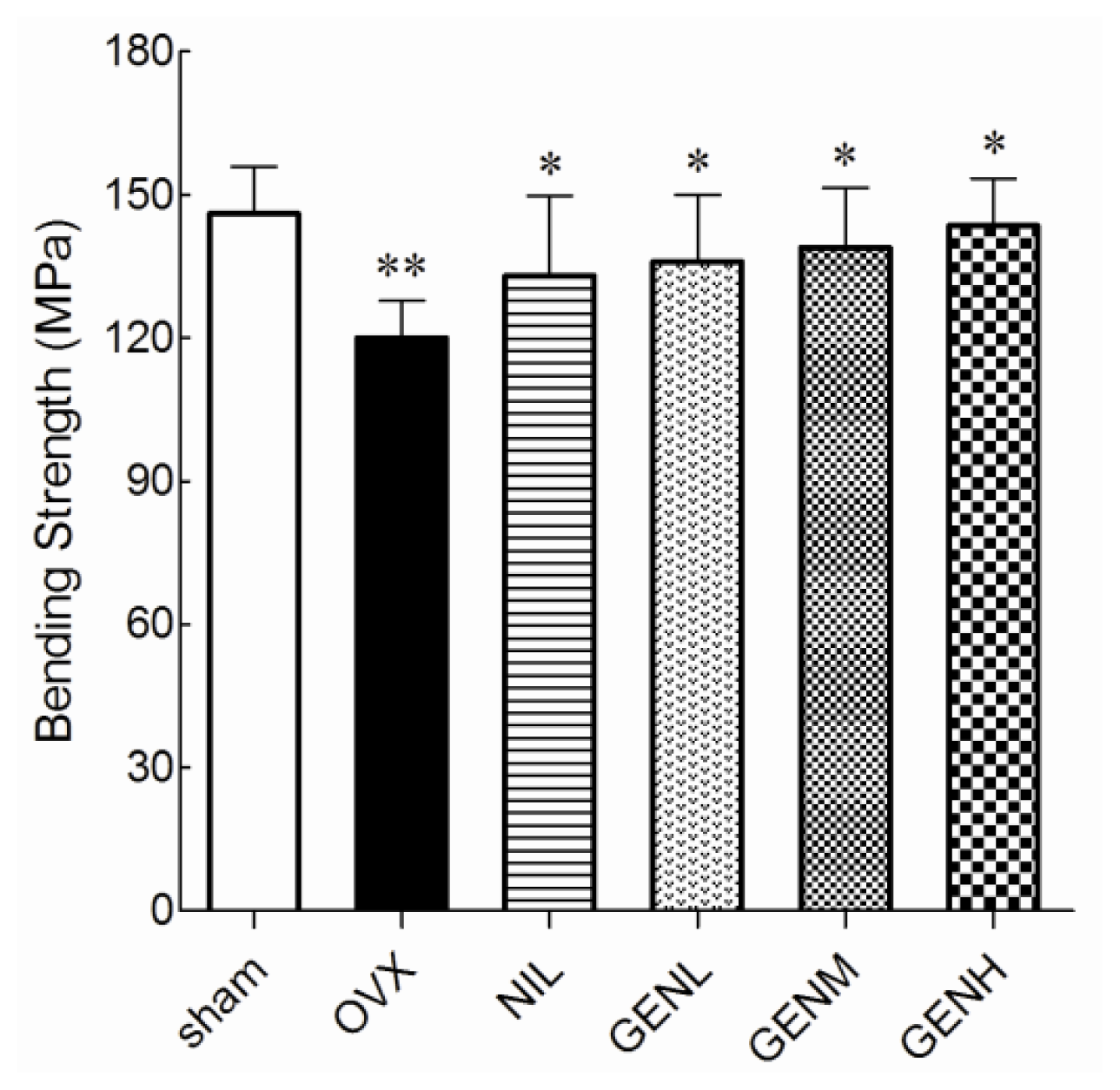
3.3. Mechanical Properties
3.4. Serum and Urine Minerals and PTH Levels
3.5. Effects of Genistein on Serum Bone Markers
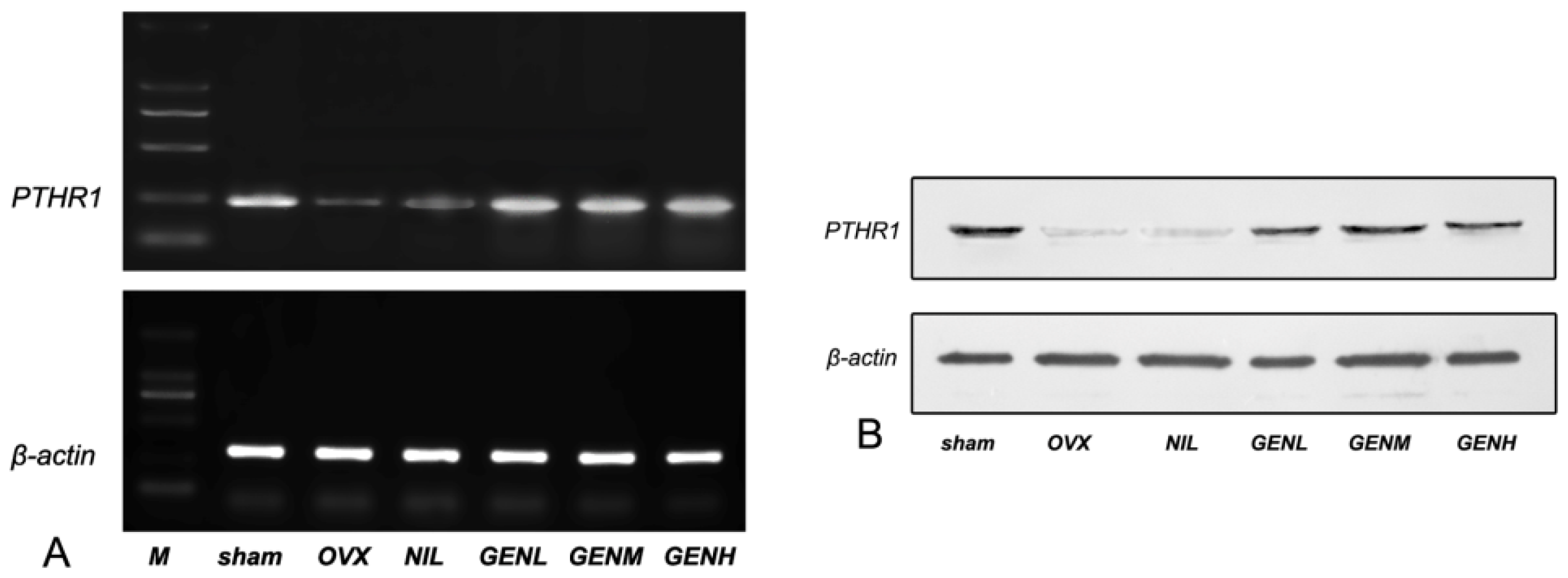
3.6. PTHR1 Expressions in Femoral Tissues
3.7. Localization and Distribution of PTHR1 in the Kidney
4. Discussion
5. Conclusion
References
- Cummings, S.R.; Melton, L.J. Epidemiology and outcomes of osteoporotic fractures. Lancet 2002, 359, 1761–1767. [Google Scholar]
- Paoletti, A.M.; Floris, S.; Mannias, M.; Orrù, M.; Crippa, D.; Orlandi, R.; del Zompo, M.M.; Melis, G.B. Evidence that cyproterone acetate improves psychological symptoms and enhances the activity of the dopaminergic system in postmenopause. J. Clin. Endocrinol. Metab 2001, 86, 608–612. [Google Scholar]
- Nelson, H.D. Menopause. Lancet 2008, 37, 760–770. [Google Scholar]
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of oestrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 2002, 288, 321–323.
- Levis, S.; Strickman-Stein, N.; Ganjei-Azar, P.; Xu, P.; Doerge, D.R.; Krischer, J. Soy isoflavones in the prevention of menopausal bone loss and menopausal symptoms: A randomized, double-blind trial. Arch. Intern. Med 2011, 171, 1363–1369. [Google Scholar]
- Duncan, A.M.; Phipps, W.R.; Kurzer, M.S. Phyto-oestrogens. Best Pract. Res. Clin. Endocrinol. Metab 2003, 17, 253–271. [Google Scholar]
- Zhang, X.; Shu, X.O.; Li, H.; Yang, G.; Li, Q.; Gao, Y.T.; Zheng, W. Prospective cohort study of soy food consumption and risk of bone fracture among postmenopausal women. Arch. Intern. Med 2005, 165, 1890–1895. [Google Scholar]
- Gutendorf, B.; Westendorf, J. Comparison of an array of in vitro assays for the assessment of the estrogenic potential of natural and synthetic estrogens, phytoestrogens and xenoestrogens. Toxicology 2001, 166, 79–89. [Google Scholar]
- Wang, Z.L.; Sun, J.Y.; Wang, D.N.; Xie, Y.H.; Wang, S.W.; Zhao, W.M. Pharmacological studies of the large-scaled purified genistein from Huaijiao (Sophora japonica-Leguminosae) on anti-osteoporosis. Phytomedicine 2006, 13, 718–723. [Google Scholar]
- Wang, J.; Shang, F.; Mei, Q.; Wang, J.; Zhang, R.; Wang, S. NO-donating genistein prodrug alleviates bone loss in ovariectomised rats. Swiss Med. Wkly 2008, 138, 602–607. [Google Scholar]
- Bitto, A.; Burnett, B.P.; Polito, F.; Marini, H.; Levy, R.M.; Armbruster, M.A.; Minutoli, L.; di Stefano, V.; Irrera, N.; Antoci, S.; et al. Effects of genistein aglycone in osteoporotic, ovariectomized rats: A comparison with alendronate, raloxifene and oestradiol. Br. J. Pharmacol 2008, 155, 896–905. [Google Scholar]
- Bitto, A.; Burnett, B.P.; Polito, F.; Levy, R.M.; Marini, H.; di Stefano, V.; Irrera, N.; Armbruster, M.A.; Minutoli, L.; Altavilla, D.; et al. Genistein aglycone reverses glucocorticoid-induced osteoporosis and increases bone breaking strength in rats: A comparative study with alendronate. Br. J. Pharmacol 2009, 156, 1287–1295. [Google Scholar]
- Bitto, A.; Polito, F.; Burnett, B.; Levy, R.; di Stefano, V.; Armbruster, M.A.; Marini, H.; Minutoli, L.; Altavilla, D.; Squadrito, F. Protective effect of genistein aglycone on the development of osteonecrosis of the femoral head and secondary osteoporosis induced by methylprednisolone in rats. J. Endocrinol 2009, 201, 321–328. [Google Scholar]
- Marini, H.; Minutoli, L.; Polito, F.; Bitto, A.; Altavilla, D.; Atteritano, M.; Gaudio, A.; Mazzaferro, S.; Frisina, A.; Frisina, N.; et al. Effects of the phytoestrogen genistein on bone metabolism in osteopenic postmenopausal women: A randomized trial. Ann. Intern. Med 2007, 146, 839–847. [Google Scholar]
- Adlercreutz, H.; Honjo, H.; Higashi, A. Urinary excretion of lignans and isoflavomoids phytoesrogens in Japanese men and women consuming a traditional Japanese diet. Am. J. Clin. Nutr 1991, 54, 1093–1100. [Google Scholar]
- Kimira, M.; Arai, Y.; Shimoi, K.; Watanabe, S. Japanese intake of flavonoids and isoflavonoids from food. J. Epidemiol 1998, 8, 168–175. [Google Scholar]
- Nakamura, Y.; Tsuji, S.; Tonogai, Y. Determination of the levels of isoflavonoids in soybeans and soy-derived foods and estimation of isoflavonoids in the Japanese daily intake. J. AOAC Int 2000, 83, 635–650. [Google Scholar]
- Fukutake, F.; Takahashi, M.; Ishida, K.; Kawamura, H.; Sugimura, T.; Wakabayashi, K. Quantification of genistein and genistin in soybeans and soybean products. Food Chem. Toxicol 1996, 34, 457–461. [Google Scholar]
- Foord, S.M.; Bonner, T.I.; Neubig, R.R.; Rosser, E.M.; Pin, J.P.; Davenport, A.P.; Spedding, M.; Harmar, A.J. International union of pharmacology. XLVI. G protein-coupled receptor list. Pharmacol. Rev 2005, 57, 279–288. [Google Scholar]
- Potts, J.T. Parathyroid hormone: Past and present. J. Endocrinol 2005, 187, 311–325. [Google Scholar]
- Neer, R.M.; Arnaud, C.D.; Zanchetta, J.R. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N. Engl. J. Med 2001, 344, 1434–1441. [Google Scholar]
- Dempster, D.W.; Cosman, F.; Kurland, E.S.; Zhou, H.; Nieves, J.; Woelfert, L.; Shane, E.; Plavetić, K.; Müller, R.; Bilezikian, J.; et al. Effects of daily treatment with parathyroid hormone on bone microarchitecture and turnover in patients with osteoporosis: A paired biopsy study. J. Bone Miner. Res 2001, 16, 1846–1853. [Google Scholar]
- Zhou, H.; Shen, V.; Dempster, D.W.; Lindsay, R. Continuous parathyroid hormone and estrogen administration increases vertebral cancellous bone volume and cortical width in the estrogen-deficient rat. J. Bone Miner. Res 2001, 16, 1300–1307. [Google Scholar]
- Yang, D.; Singh, R.; Divieti, P.; Guo, J.; Bouxsein, M.L.; Bringhurst, F.R. Contributions of parathyroid hormone (PTH)/PTH-related peptide receptor signaling pathways to the anabolic effect of PTH on bone. Bone 2007, 40, 1453–1461. [Google Scholar]
- SPSS software, version 13.0; SPSS, Inc: Chicago, IL, USA, 2004.
- Beral, V. Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet 2003, 362, 419–427. [Google Scholar]
- Messina, M.J. Legumes and soybeans: overview of their nutritional profiles and health effects. Am. J. Clin. Nutr 1999, 70, 439–450. [Google Scholar]
- D’Anna, R.; Cannata, M.L.; Atteritano, M.; Cancellieri, F.; Corrado, F.; Baviera, G.; Triolo, O.; Antico, F.; Gaudio, A.; Frisina, N.; et al. Effects of the phytoestrogen genistein on hot flushes, endometrium, and vaginal epithelium in postmenopausal women: A 1-year randomized, double-blind, placebo controlled study. Menopause 2007, 14, 648–655. [Google Scholar]
- Sapronov, N.S.; Kasakova, S.B. Effects of synthetic and plant-derived selective modulators of estrogen receptors on depression-like behavior of female rats. Bull. Exp. Biol. Med 2008, 146, 73–76. [Google Scholar]
- Atteritano, M.; Pernice, F.; Mazzaferro, S.; Mantuano, S.; Frisina, A.; D’Anna, R.; Cannata, M.L.; Bitto, A.; Squadrito, F.; Frisina, N.; et al. Effects of phytoestrogen genistein on cytogenetic biomarkers in postmenopausal women: 1 year randomized, placebo-controlled study. Eur. J. Pharmacol 2008, 589, 22–26. [Google Scholar]
- Taylor, C.K.; Levy, R.M.; Elliott, J.C.; Burnett, B.P. The effect of genistein aglycone on cancer and cancer risk: A review of in vitro, preclinical, and clinical studies. Nutr. Rev 2009, 67, 398–415. [Google Scholar]
- Huang, C.C.; Hsu, B.Y.; Wu, N.L.; Tsui, W.H.; Lin, T.J.; Su, C.C.; Hung, C.F. Anti-photoaging effects of soy isoflavone extract (aglycone and acetylglucoside form) from soybean cake. Int. J. Mol. Sci 2010, 12, 4782–4795. [Google Scholar]
- Lynch, M.P.; Capparelli, C.; Stein, J.L.; Stein, G.S.; Lian, J.B. Apoptosis during bone-like tissue development in vitro. J. Cell. Biochem 1998, 68, 31–49. [Google Scholar]




| 0 week | 1 week | 2 weeks | 4 weeks | 8 weeks | 12 weeks | |
|---|---|---|---|---|---|---|
| sham | 229.6 ± 7.1 | 248.3 ± 9.7 | 242.5 ± 10.3 | 252.8 ± 14.8 | 303.3 ± 23.7 | 324.3 ± 35.2 |
| OVX | 223.4 ± 19.1 | 254.7 ± 28.2 | 273.4 ± 15.3 a | 298.0 ± 32.6 a | 310.7 ± 23.0 | 325.3 ± 31.7 |
| NIL | 226.3 ± 15.3 | 246.2 ± 12.9 | 250.1 ± 4.8 b | 264.9 ± 21.9 b | 277.2 ± 14.7 b | 297.1 ± 27.5 b |
| GENL | 228.7 ± 11.2 | 250.9 ± 10.3 | 265.2 ± 25.3 | 276.1 ± 24.2 b | 280.7 ± 35.9 b | 312.8 ± 26.7 |
| GENM | 225.5 ± 13.3 | 262.2 ± 13.4 | 268.0 ± 14.7 | 284.6 ± 17.5 b | 329.7 ± 20.5 b | 347.7 ± 31.2 b |
| GENH | 227.4 ± 9.8 | 255.0 ± 18.1 | 261.4 ± 8.7 | 275.2 ± 10.5 b | 312.1 ± 22.4 | 328.8 ± 36.3 |
| Group | Serum Ca (mmol/L) | Serum P (mmol/L) | Urine Ca/Cr | Urine P/Cr | PTH (pg/mL) |
|---|---|---|---|---|---|
| Sham | 2.15 ± 0.53 | 2.39 ± 0.24 | 0.30 ± 0.03 | 4.47 ± 0.23 | 108.54 ± 12.36 |
| OVX | 1.72 ± 0.36 a | 2.00 ± 0.39 a | 0.58 ± 0.02 a | 5.85 ± 0.30 a | 5.14 ± 17.49 a |
| NIL | 1.90 ± 0.63 b | 2.21 ± 0.32 b | 0.35 ± 0.01 b | 4.82 ± 0.43 b | 83.43 ± 20.78 |
| GENL | 1.83 ± 0.40 b | 2.45 ± 0.38 b | 0.49 ± 0.06 b | 5.48 ± 0.14 b | 78.62 ± 18.95 |
| GENM | 2.09 ± 0.31 b | 2.63 ± 0.84 b | 0.37 ± 0.05 b | 5.00 ± 0.36 b | 80.23 ± 22.54 |
| GENH | 2.12 ± 0.42 b | 2.71 ± 0.75 b | 0.32 ± 0.02 b | 4.64 ± 0.20 b | 95.37 ± 21.57 b |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Miao, Q.; Li, J.-G.; Miao, S.; Hu, N.; Zhang, J.; Zhang, S.; Xie, Y.-H.; Wang, J.-B.; Wang, S.-W. The Bone-Protective Effect of Genistein in the Animal Model of Bilateral Ovariectomy: Roles of Phytoestrogens and PTH/PTHR1 Against Post-Menopausal Osteoporosis. Int. J. Mol. Sci. 2012, 13, 56-70. https://doi.org/10.3390/ijms13010056
Miao Q, Li J-G, Miao S, Hu N, Zhang J, Zhang S, Xie Y-H, Wang J-B, Wang S-W. The Bone-Protective Effect of Genistein in the Animal Model of Bilateral Ovariectomy: Roles of Phytoestrogens and PTH/PTHR1 Against Post-Menopausal Osteoporosis. International Journal of Molecular Sciences. 2012; 13(1):56-70. https://doi.org/10.3390/ijms13010056
Chicago/Turabian StyleMiao, Qing, Jing-Ge Li, Shan Miao, Nan Hu, Jin Zhang, Song Zhang, Yan-Hua Xie, Jian-Bo Wang, and Si-Wang Wang. 2012. "The Bone-Protective Effect of Genistein in the Animal Model of Bilateral Ovariectomy: Roles of Phytoestrogens and PTH/PTHR1 Against Post-Menopausal Osteoporosis" International Journal of Molecular Sciences 13, no. 1: 56-70. https://doi.org/10.3390/ijms13010056
APA StyleMiao, Q., Li, J.-G., Miao, S., Hu, N., Zhang, J., Zhang, S., Xie, Y.-H., Wang, J.-B., & Wang, S.-W. (2012). The Bone-Protective Effect of Genistein in the Animal Model of Bilateral Ovariectomy: Roles of Phytoestrogens and PTH/PTHR1 Against Post-Menopausal Osteoporosis. International Journal of Molecular Sciences, 13(1), 56-70. https://doi.org/10.3390/ijms13010056




