The Castagnoli–Cushman Reaction
Abstract
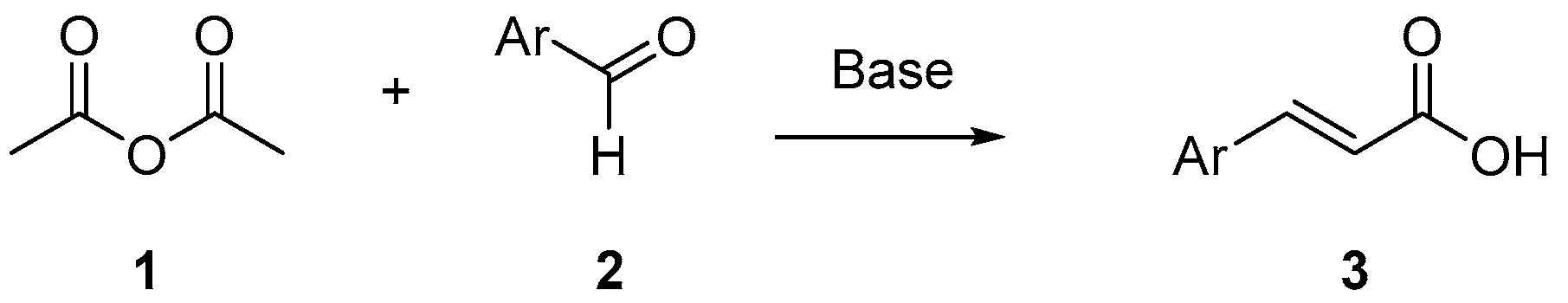
1. Introduction
2. Mechanism
3. Two-Component Castagnoli–Cushman Reactions (2C-CCR)
3.1. Anhydride Substrates
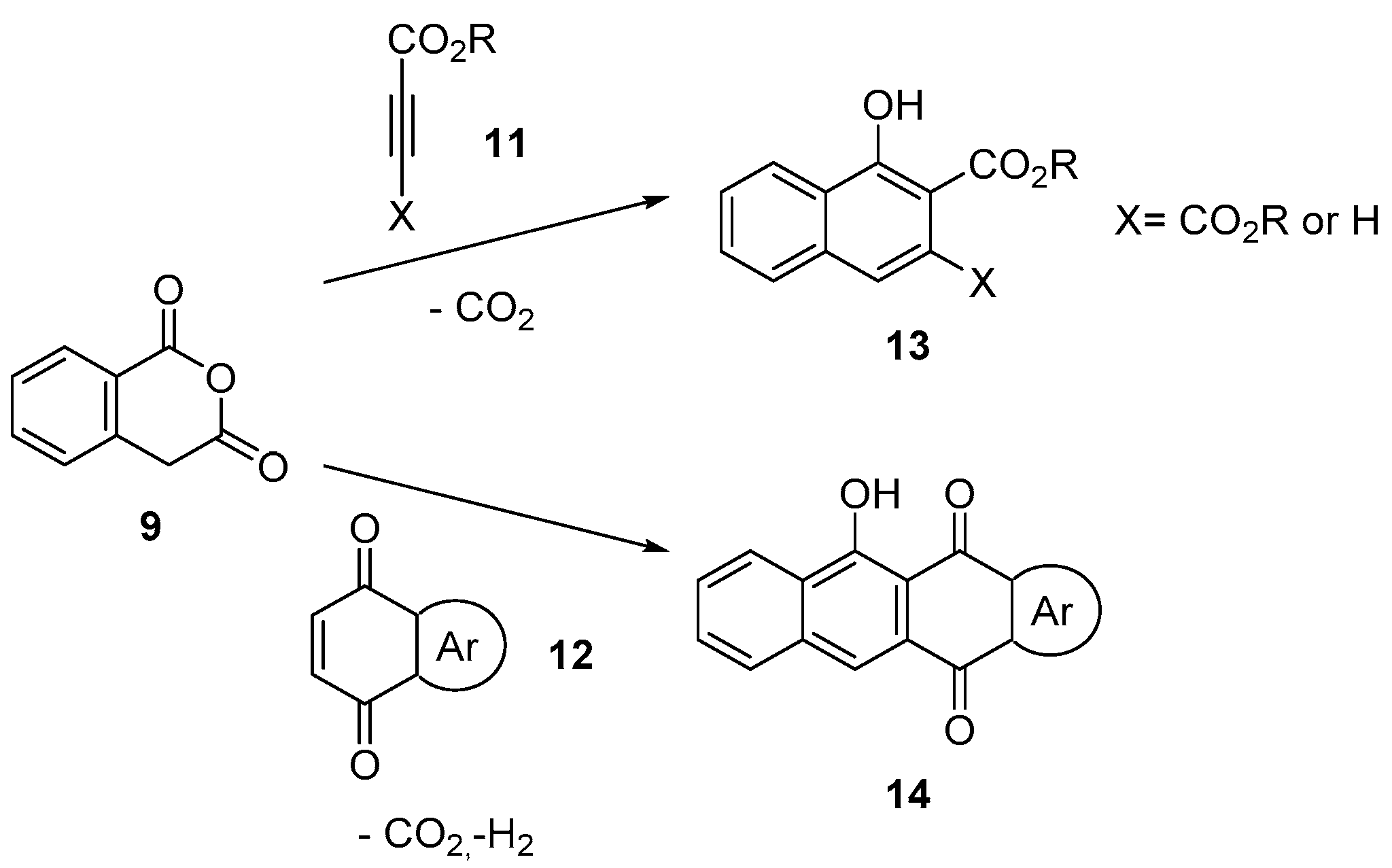
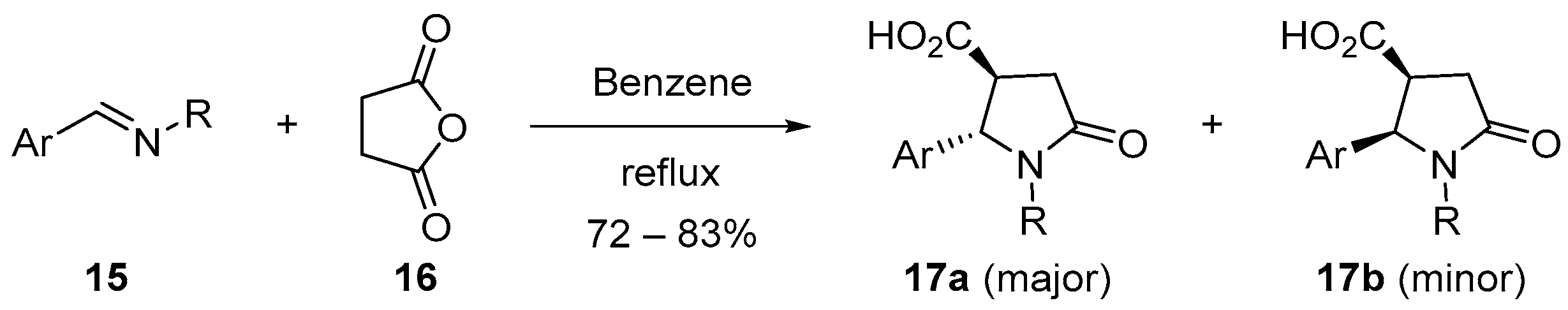
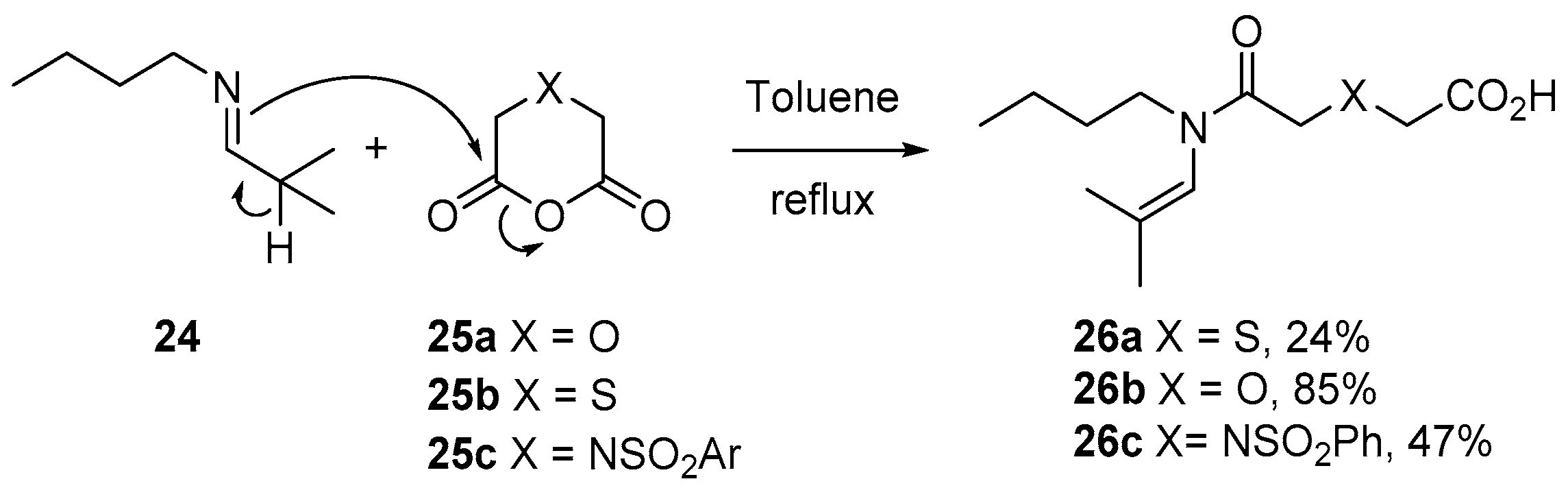
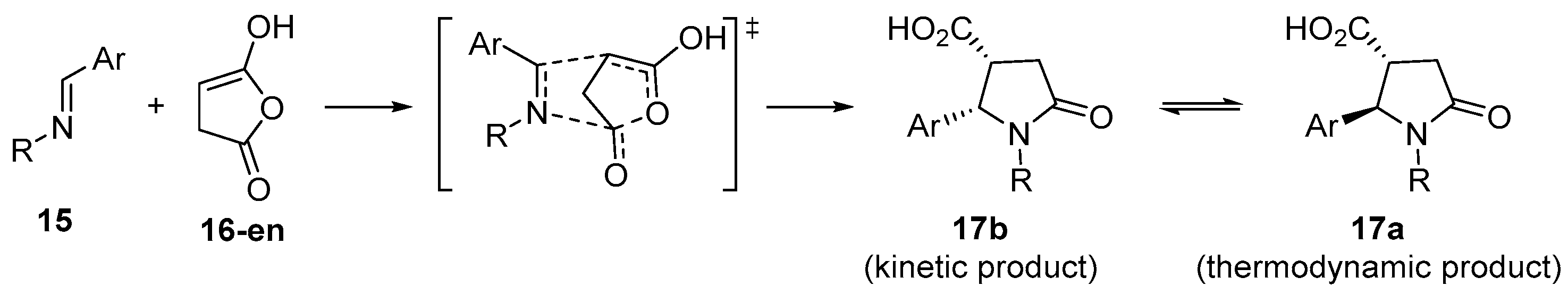
3.1.1. 5-Membered Cyclic Anhydrides
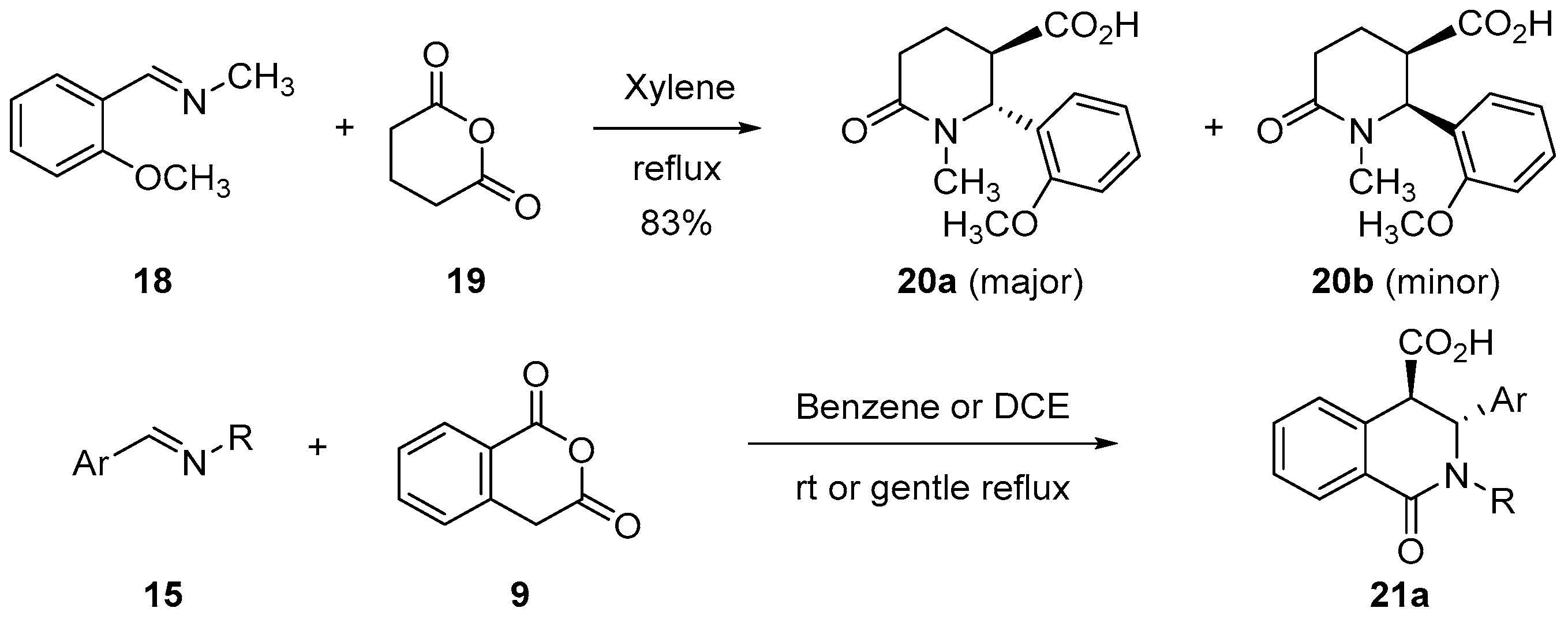
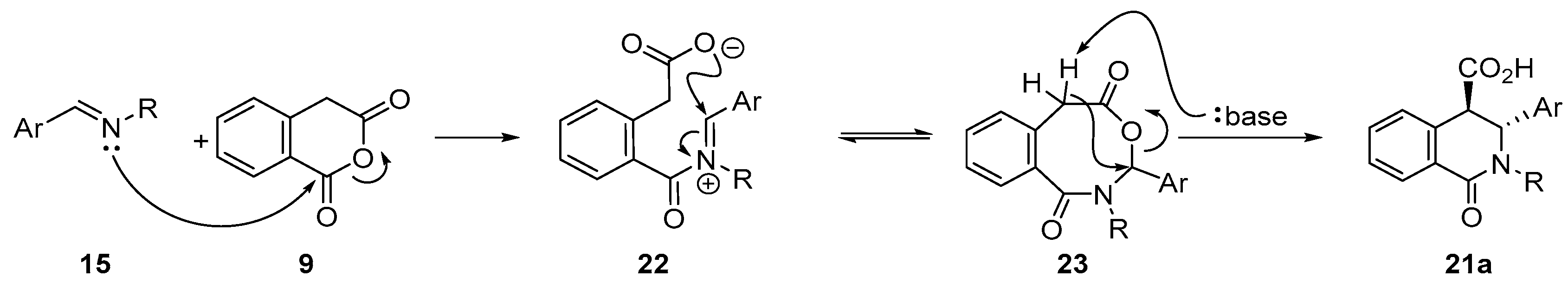
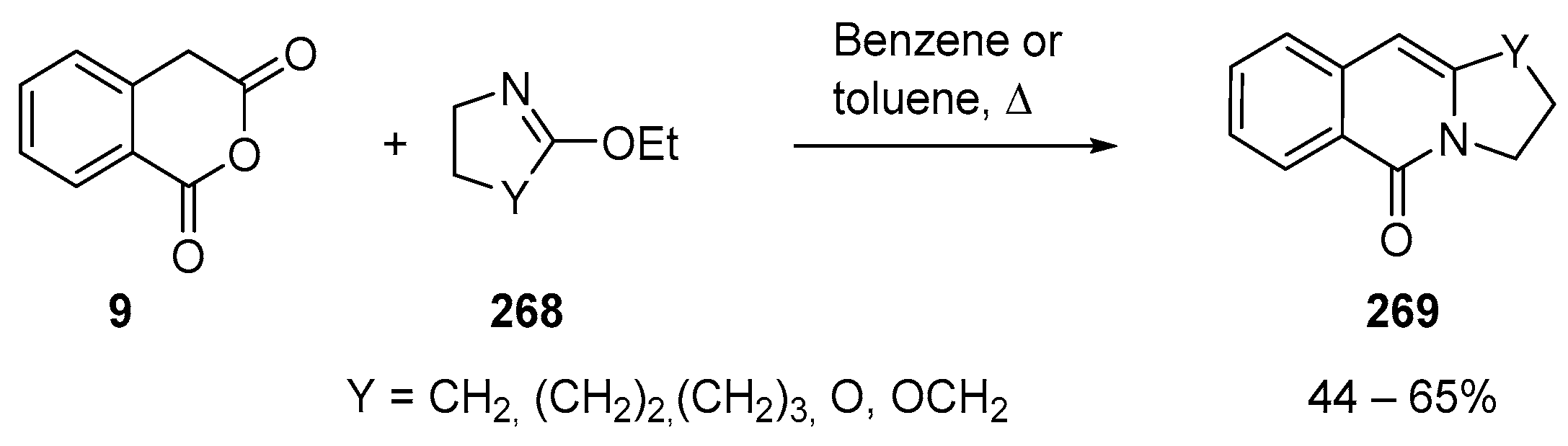
3.1.2. Six-Membered Cyclic Anhydrides
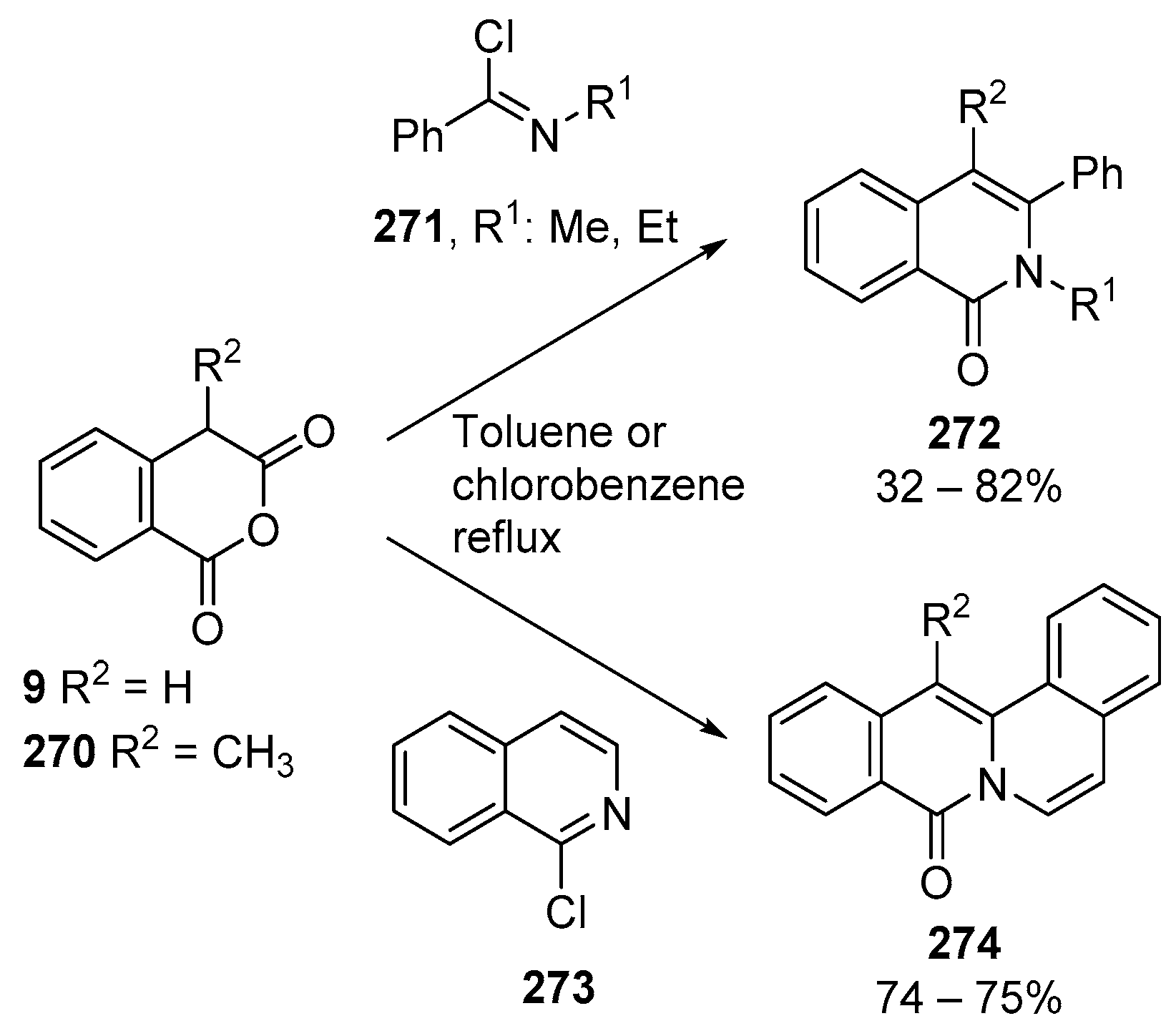
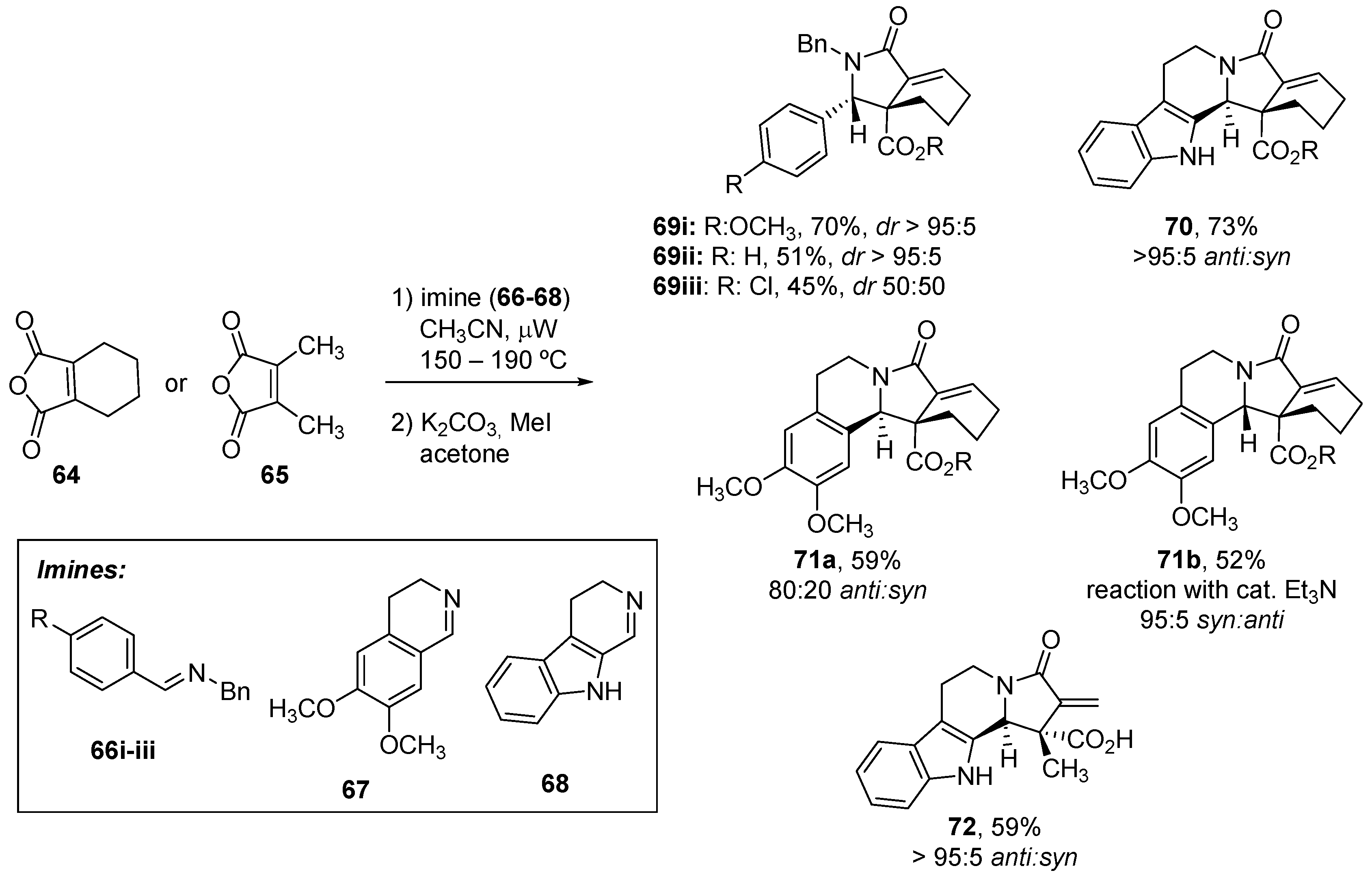
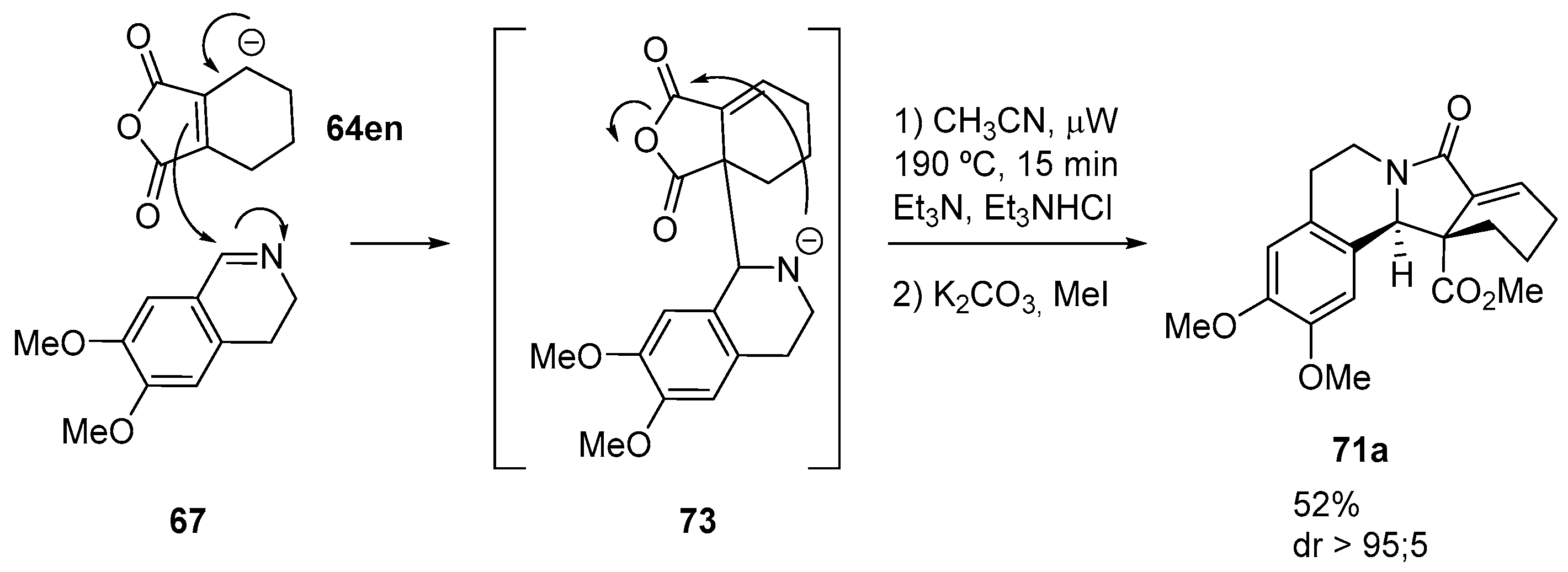
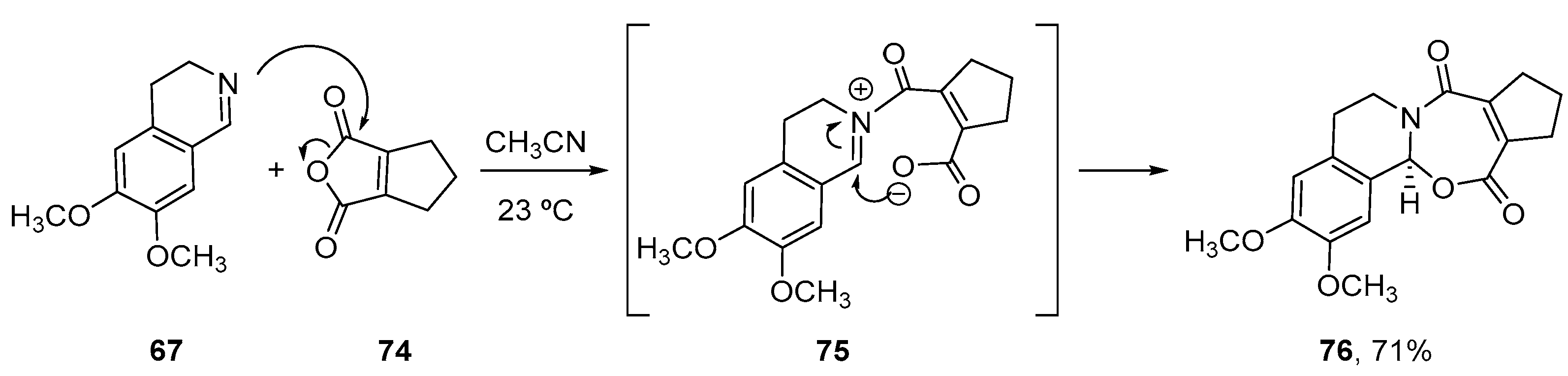
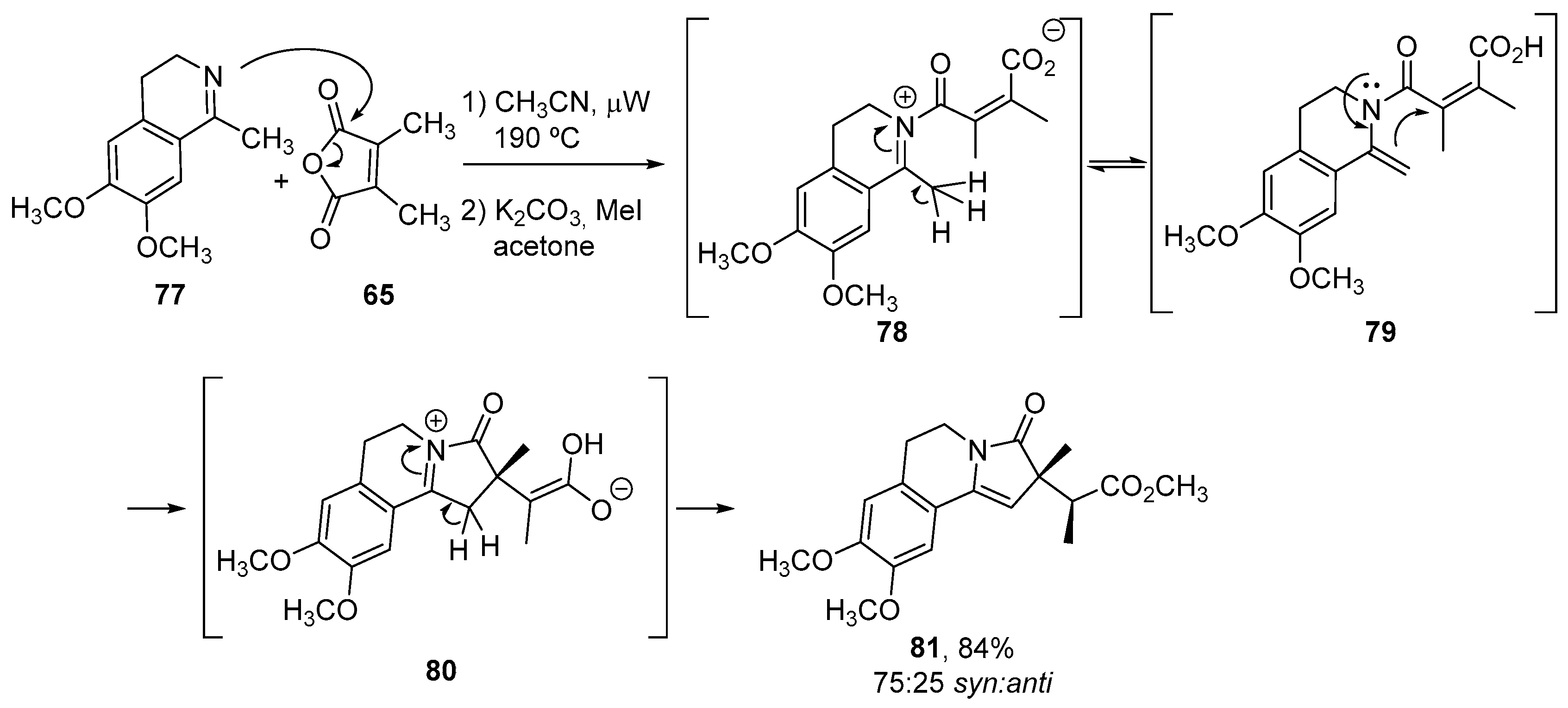
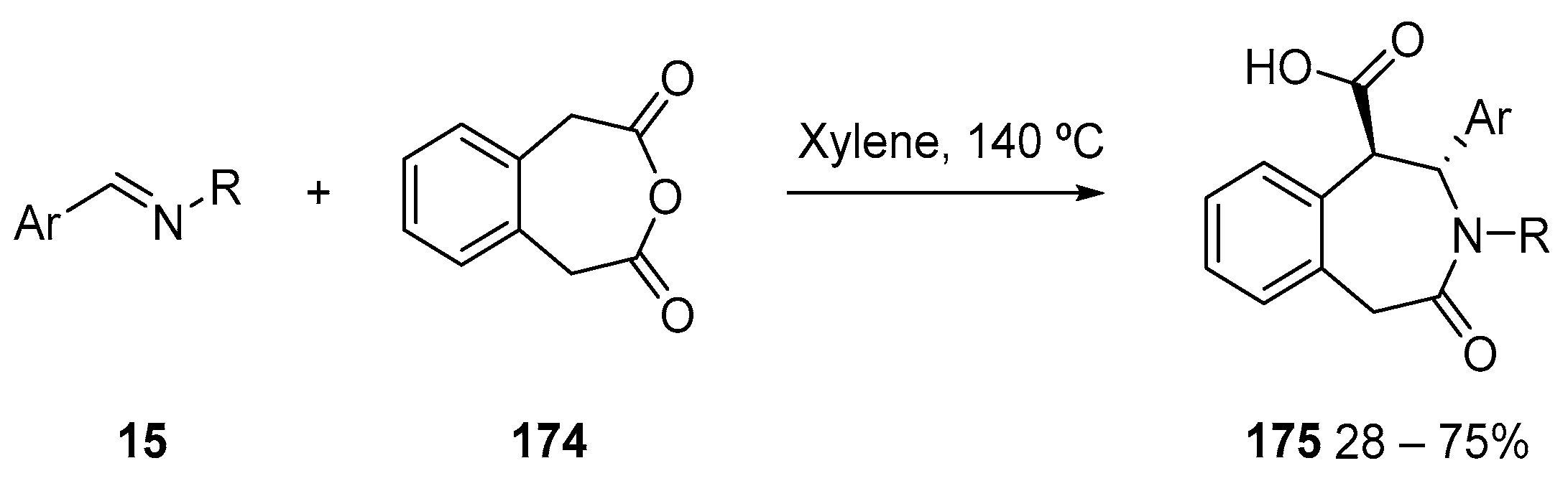
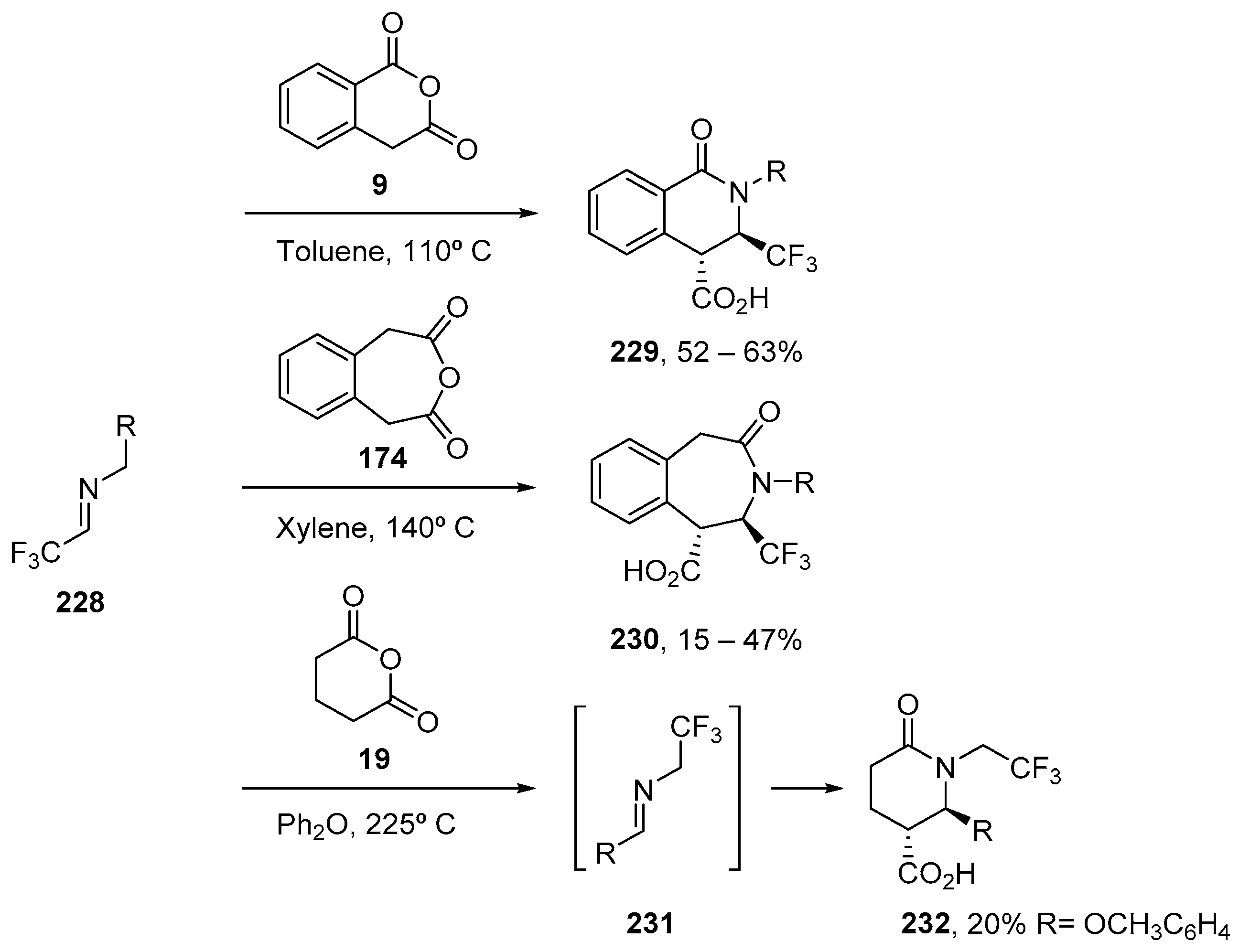
3.1.3. Higher Order Cyclic Anhydrides
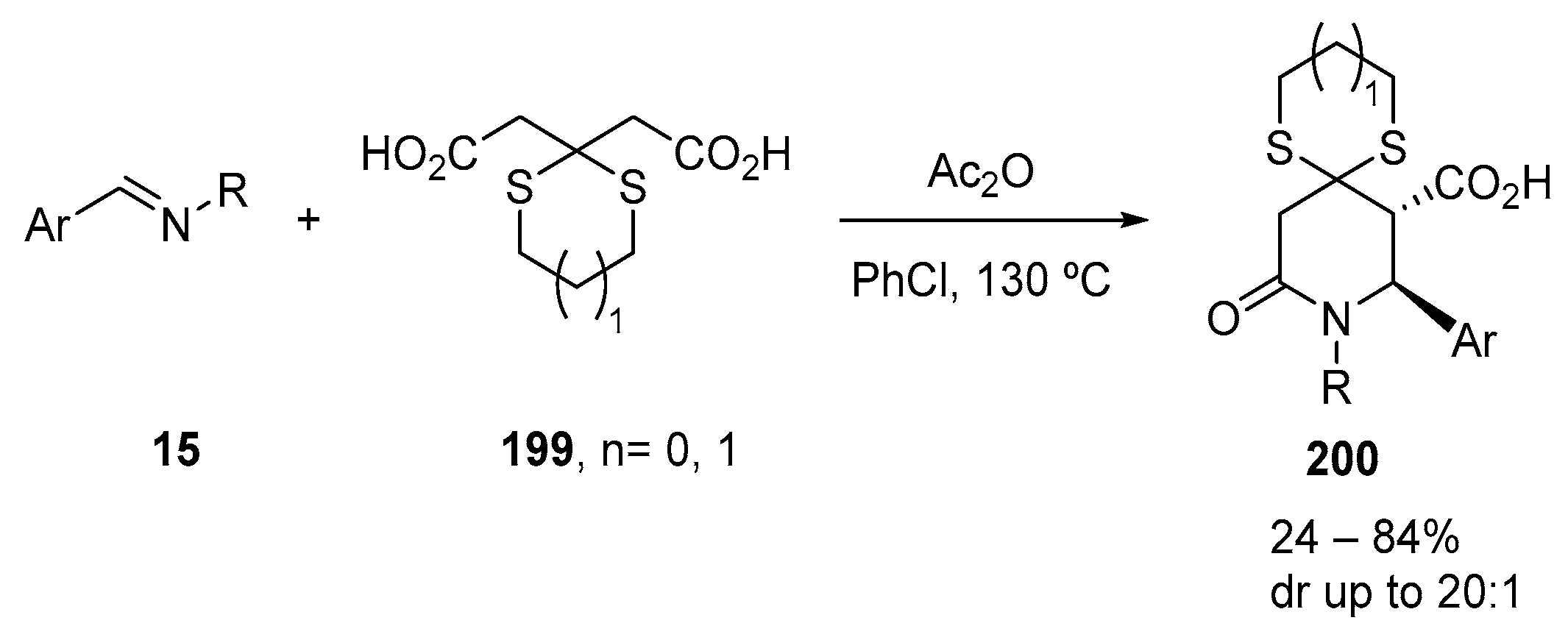
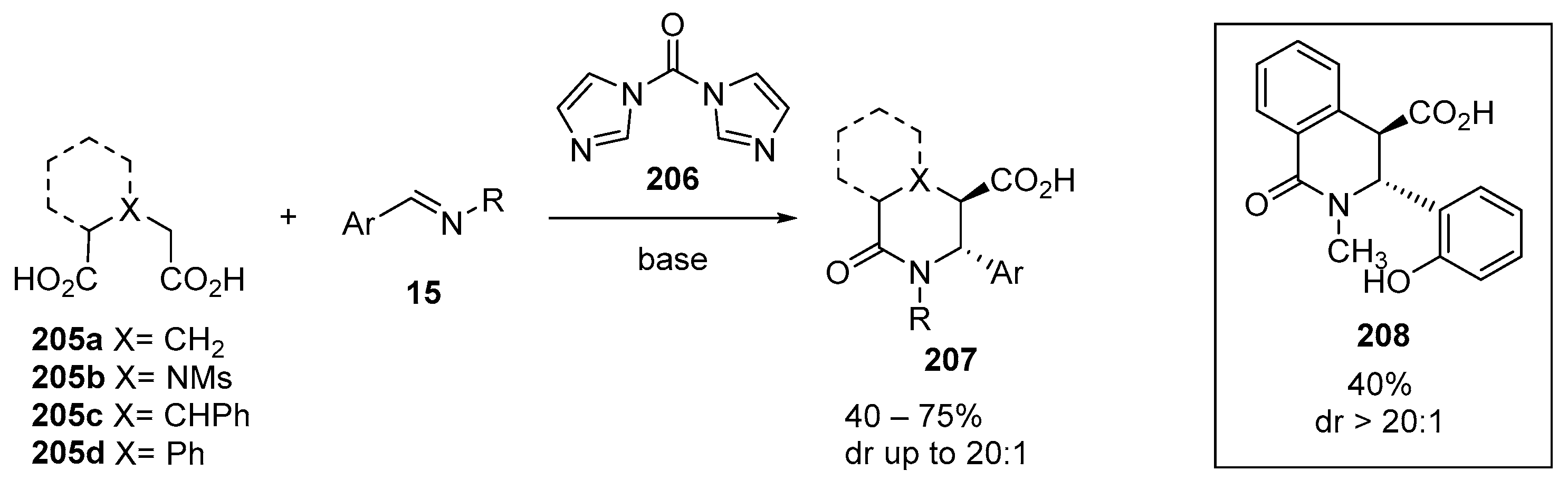
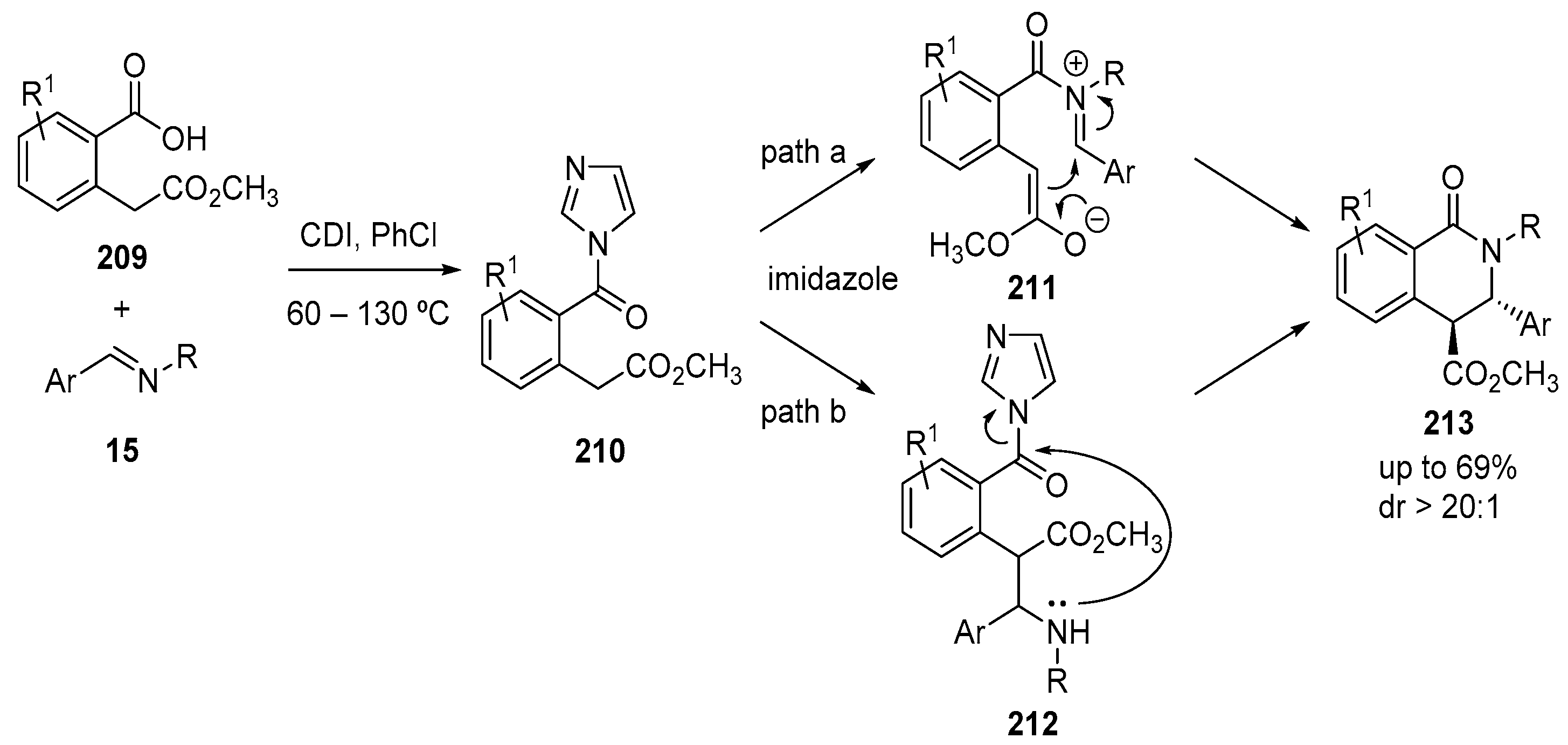
3.1.4. Diacid Anhydride Precursors
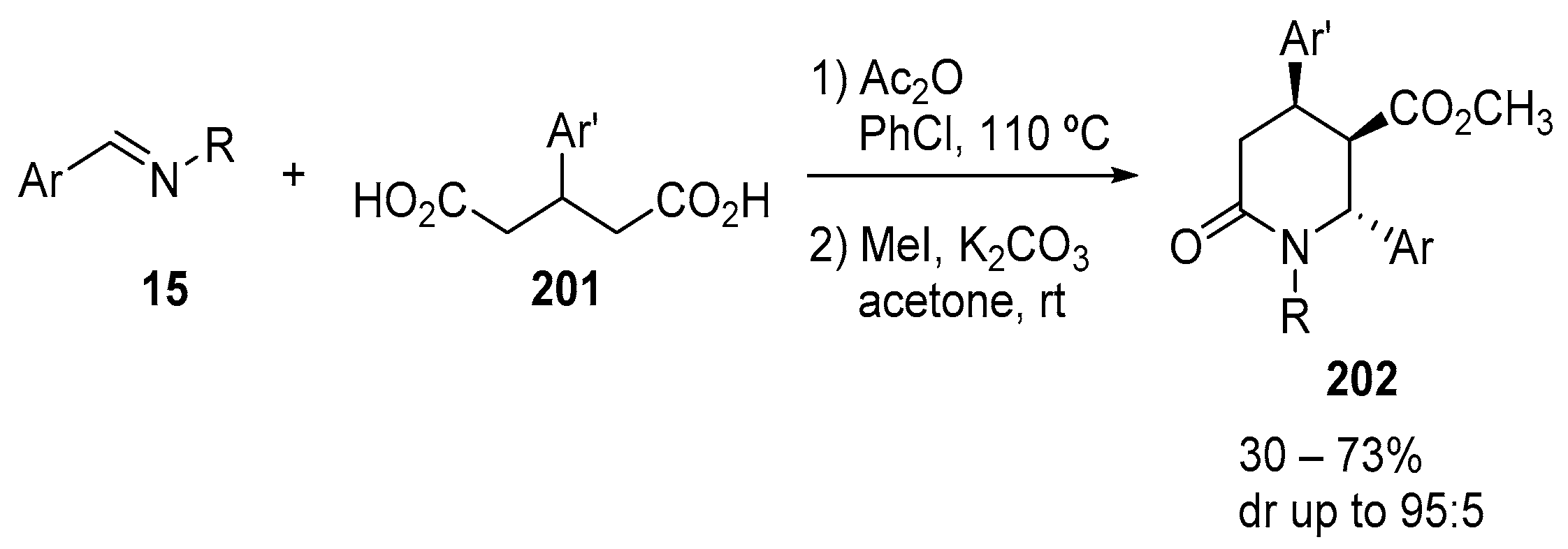
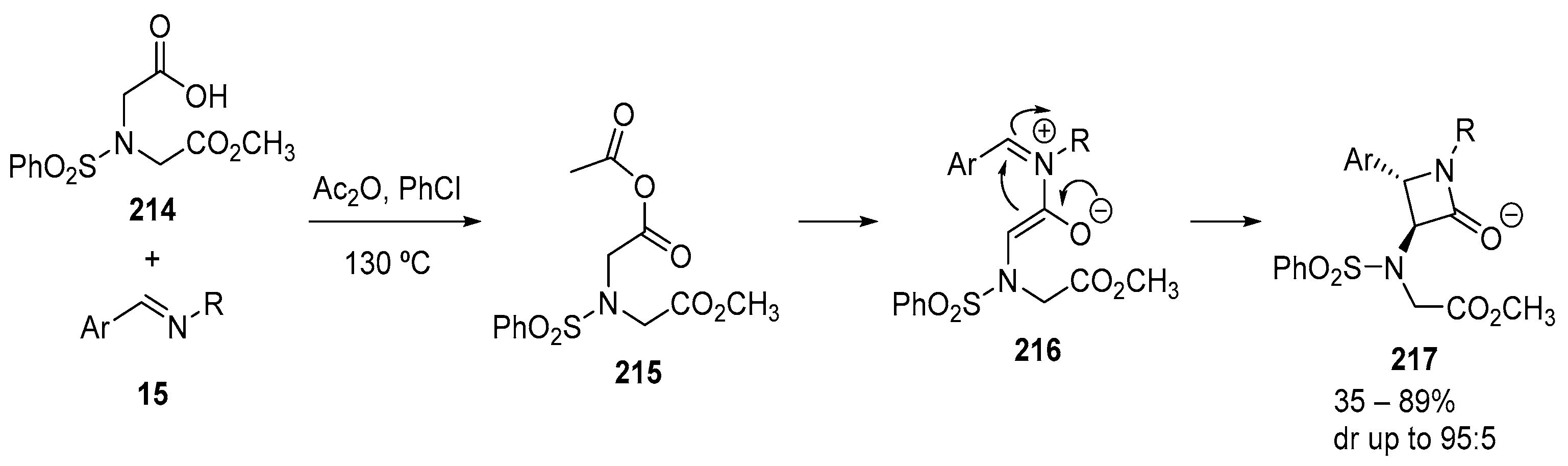
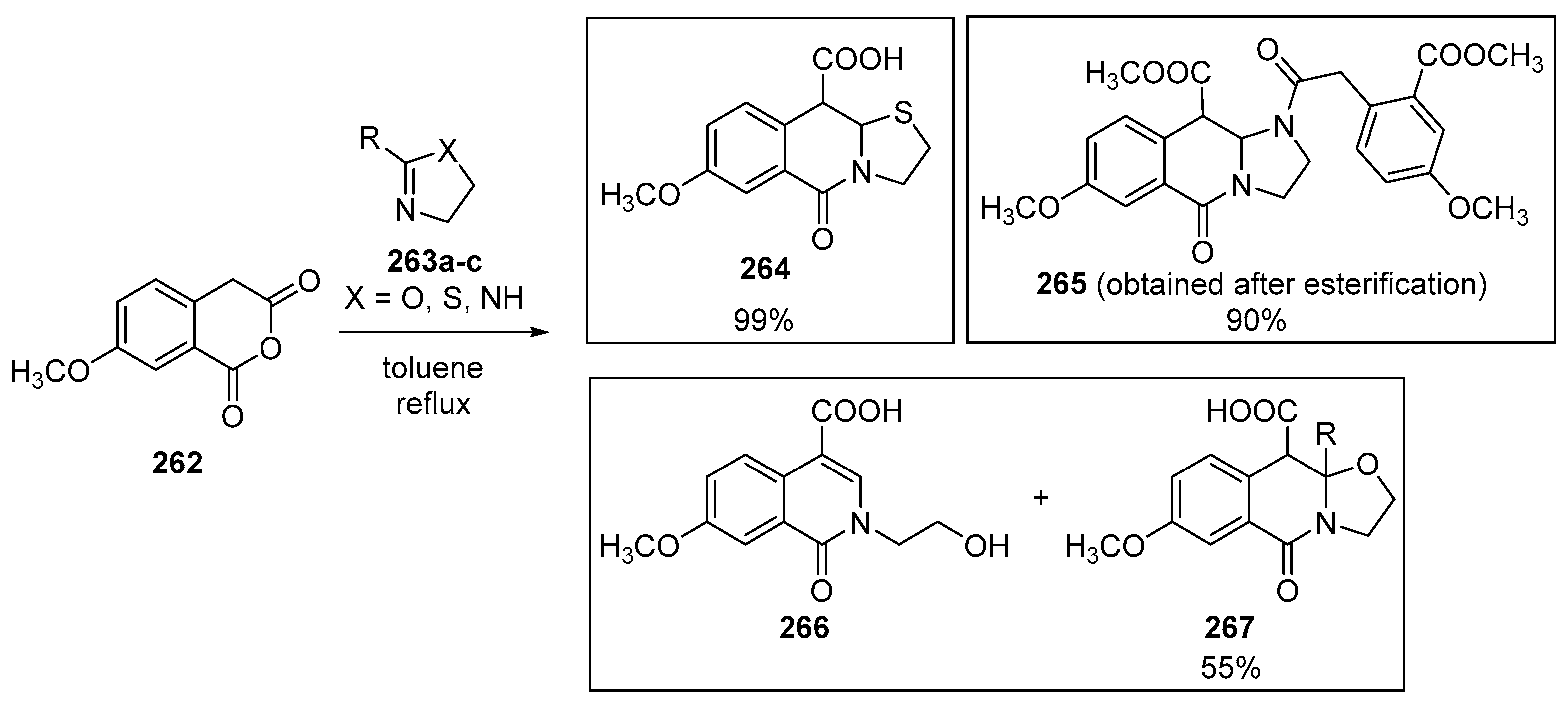
3.1.5. Dicarboxylic Acid Monoesters
3.2. Scope of the Imine Substrates
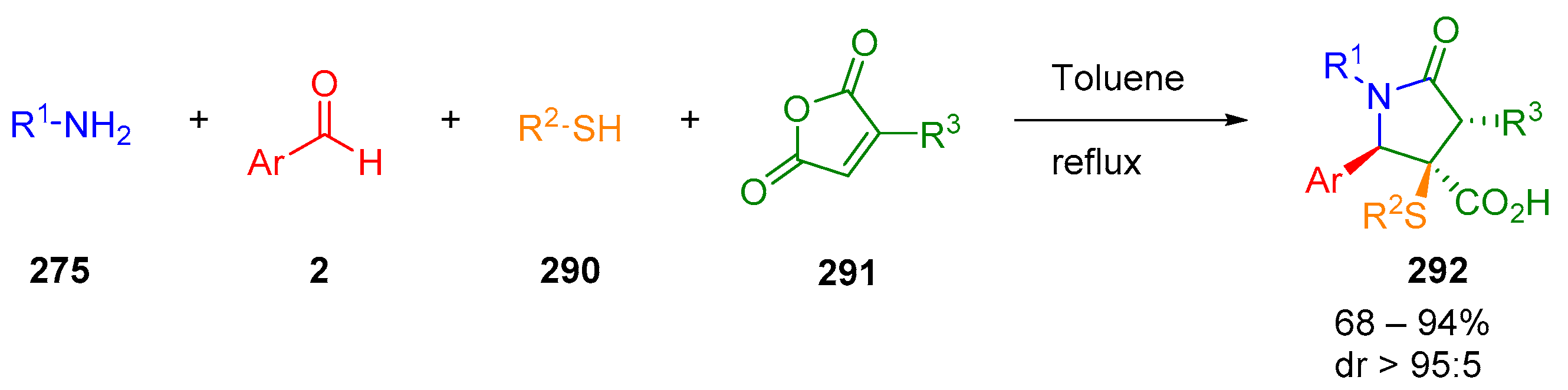
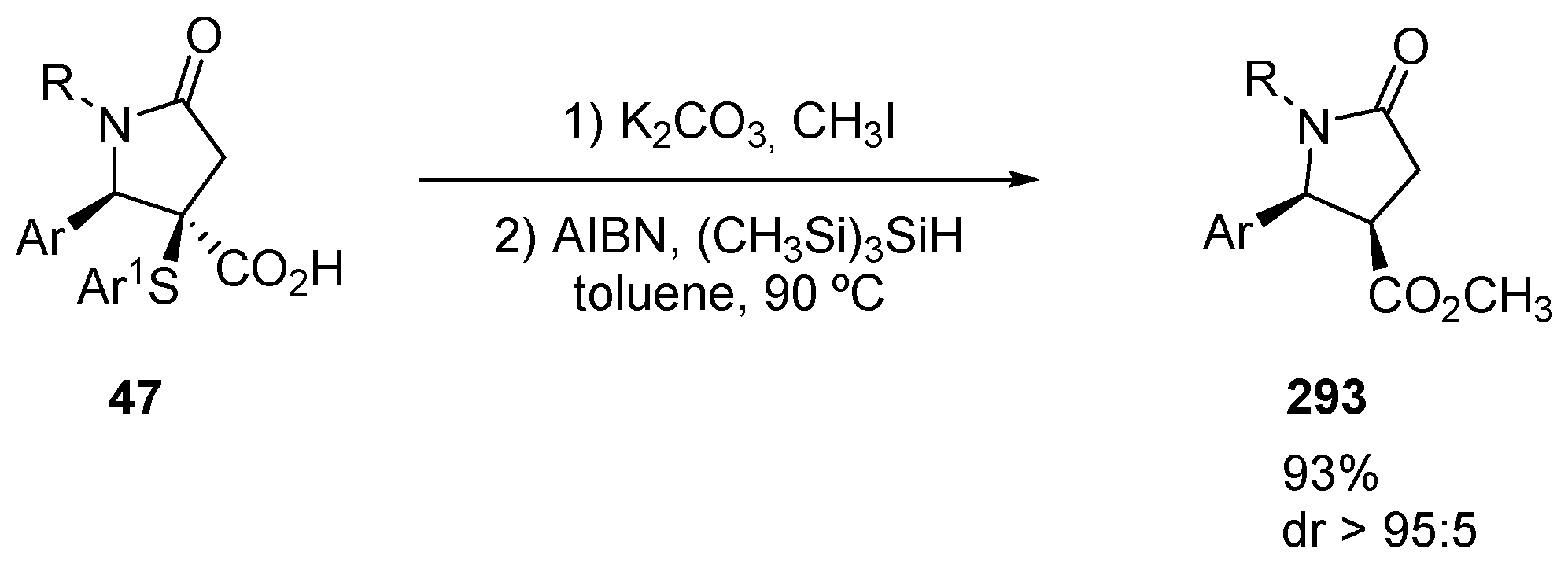
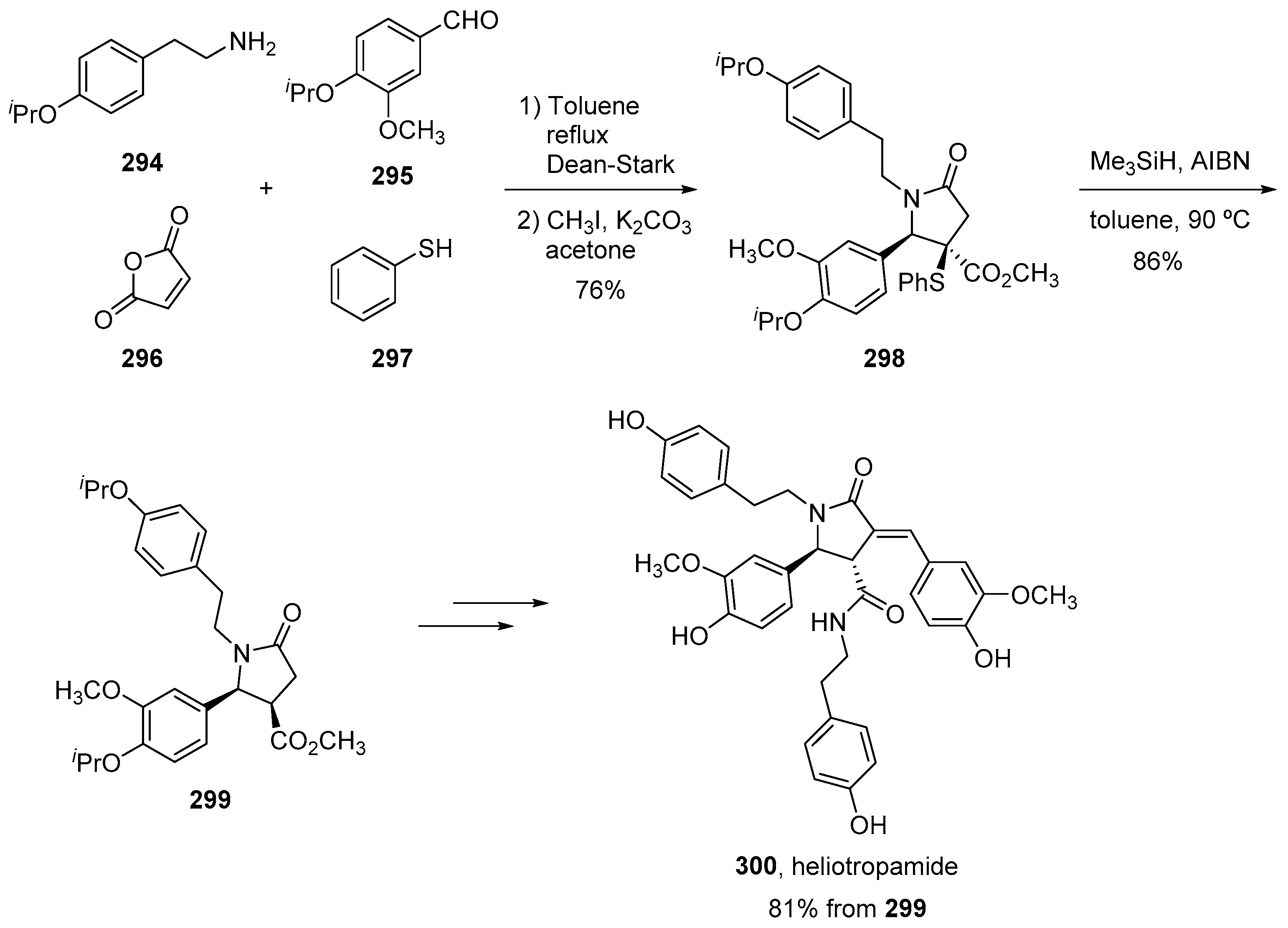
4. Three-Component Castagnoli–Cushman Reactions (3C-CCRs)
4.1. Limitations
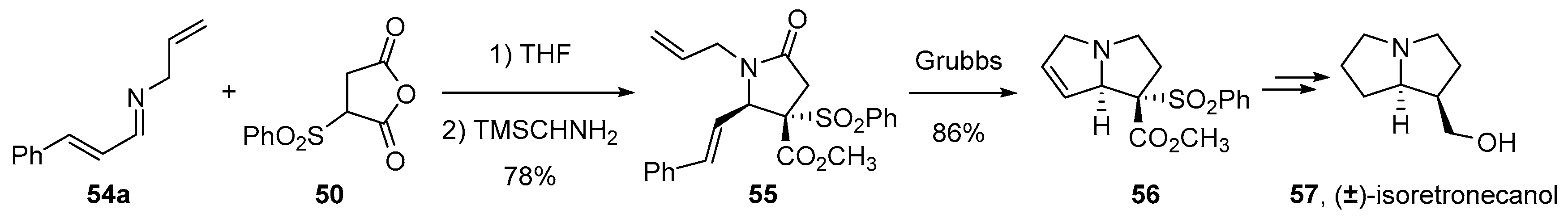
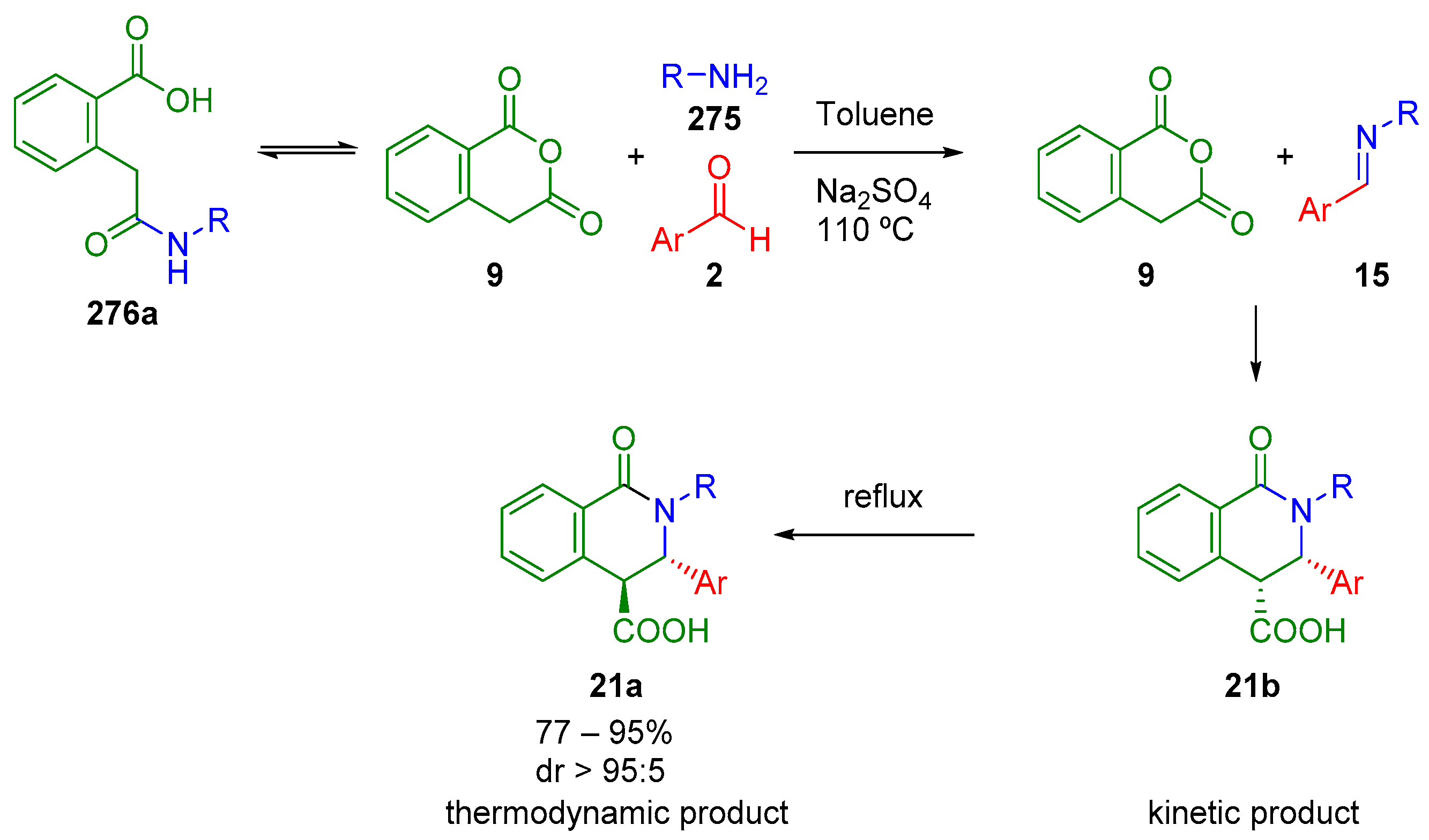
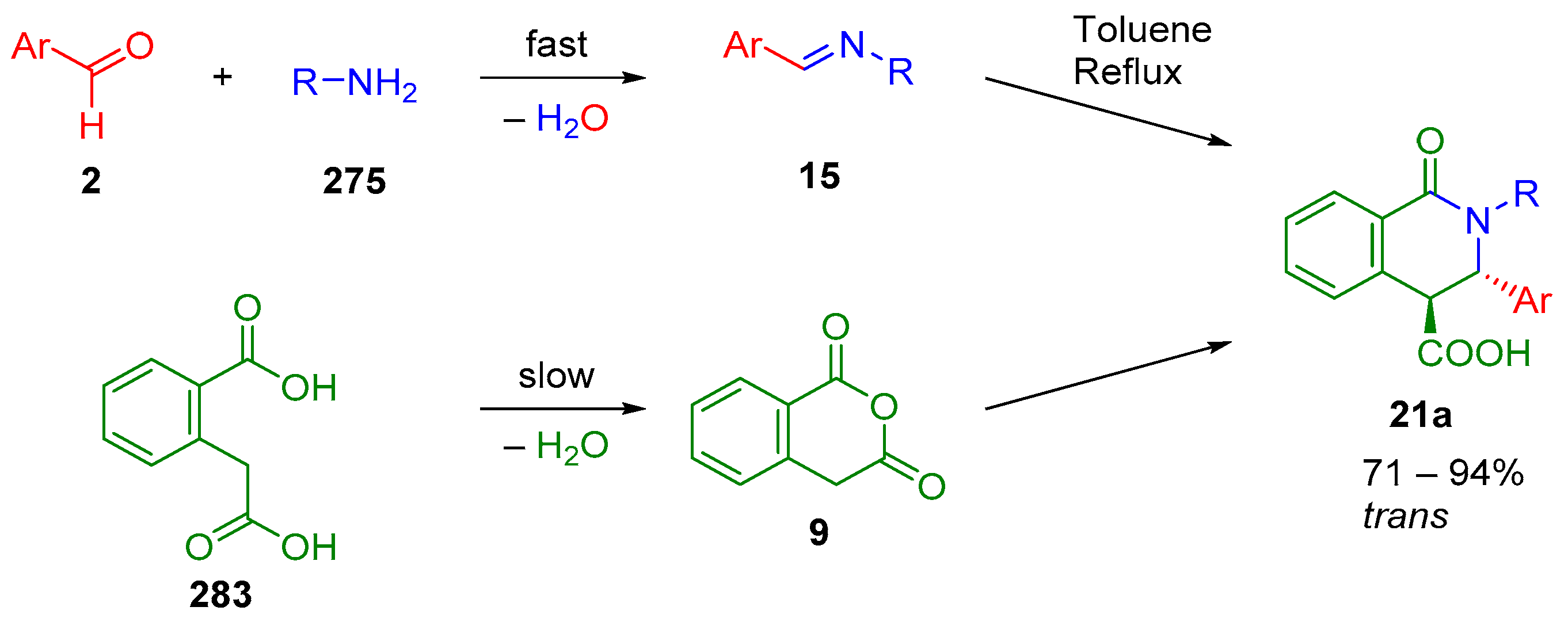
4.2. Sequential Reactions
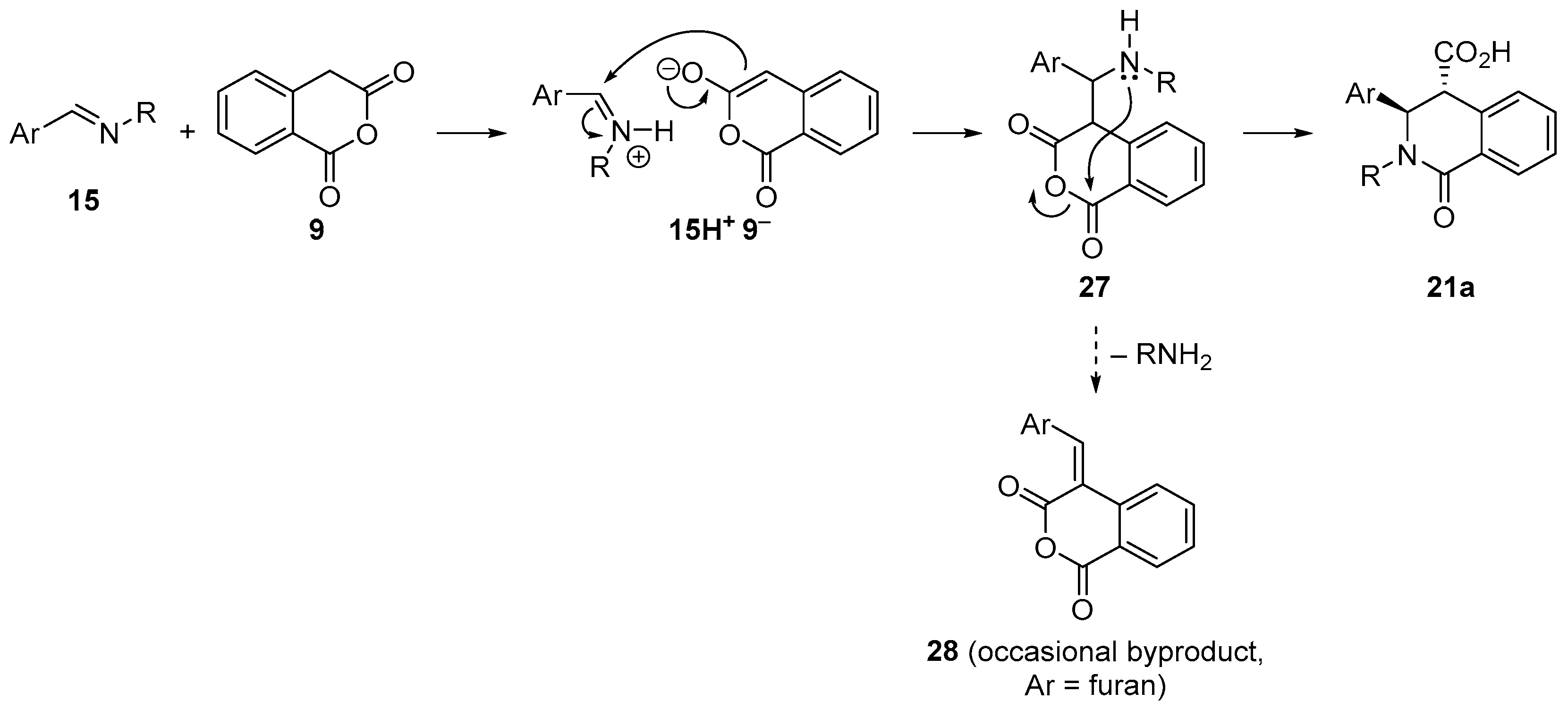
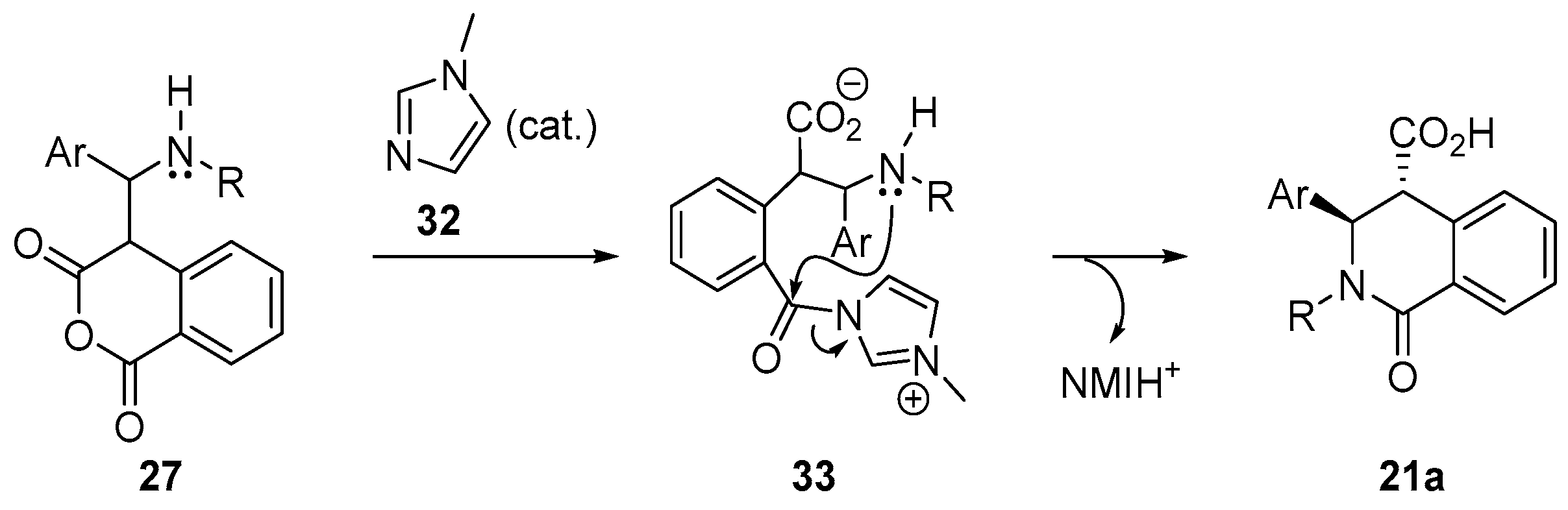
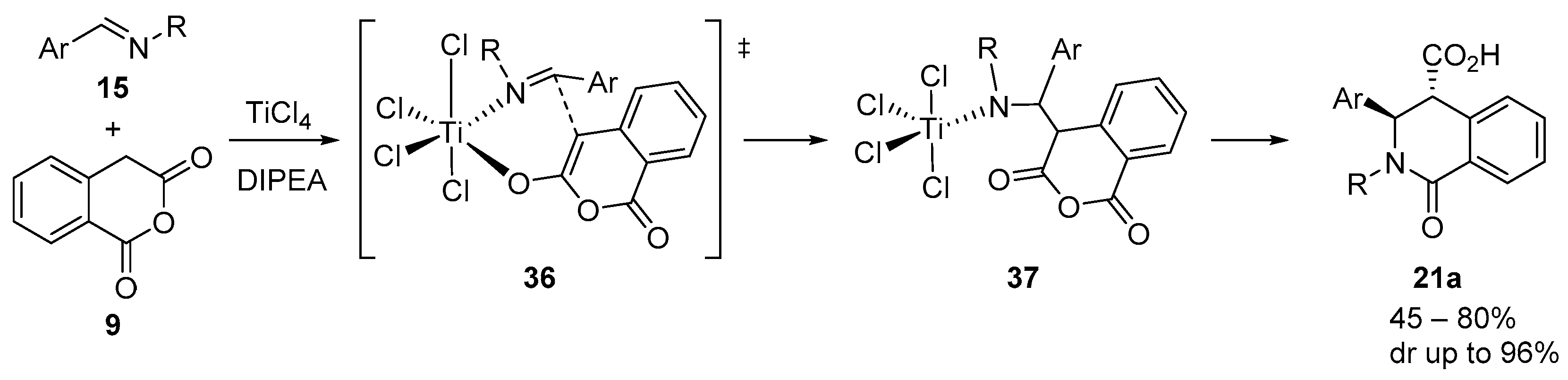
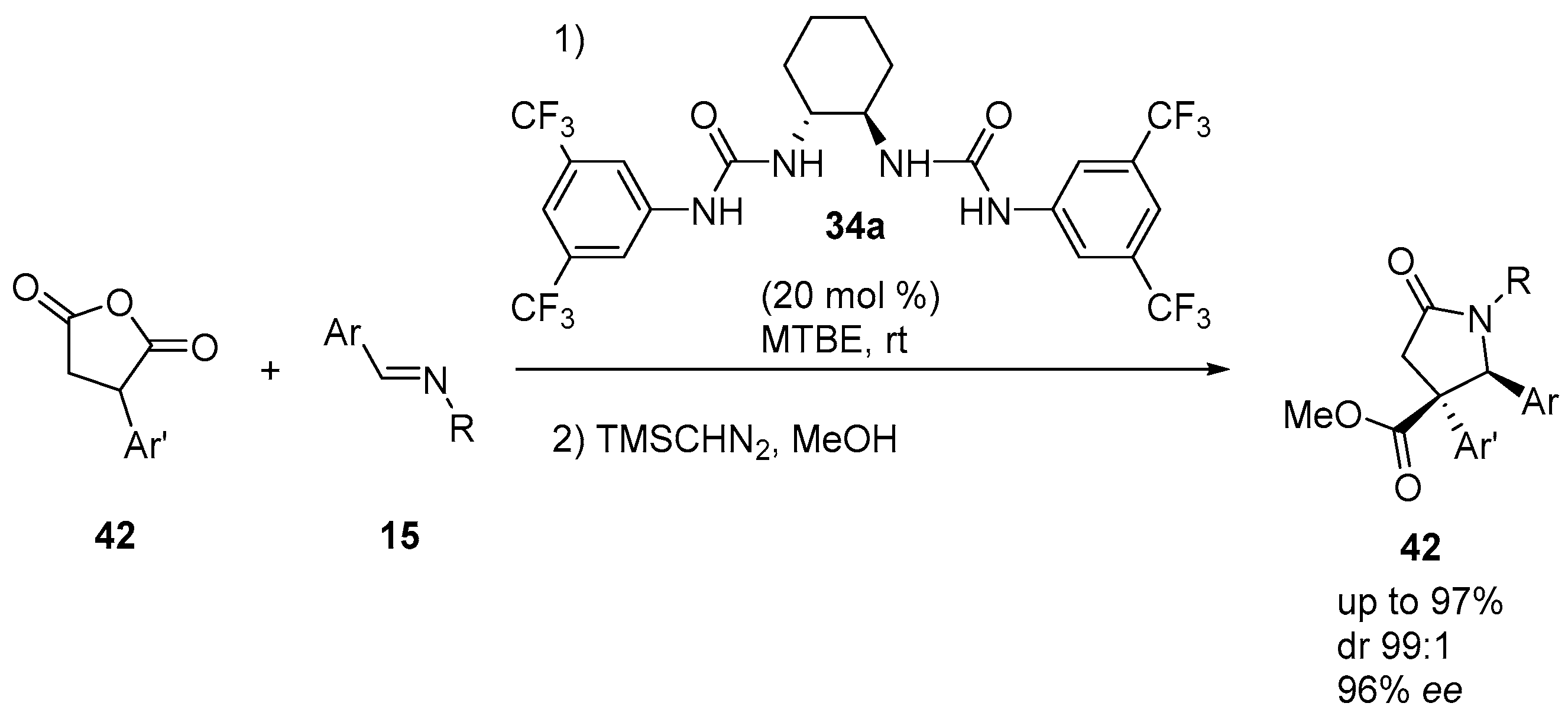
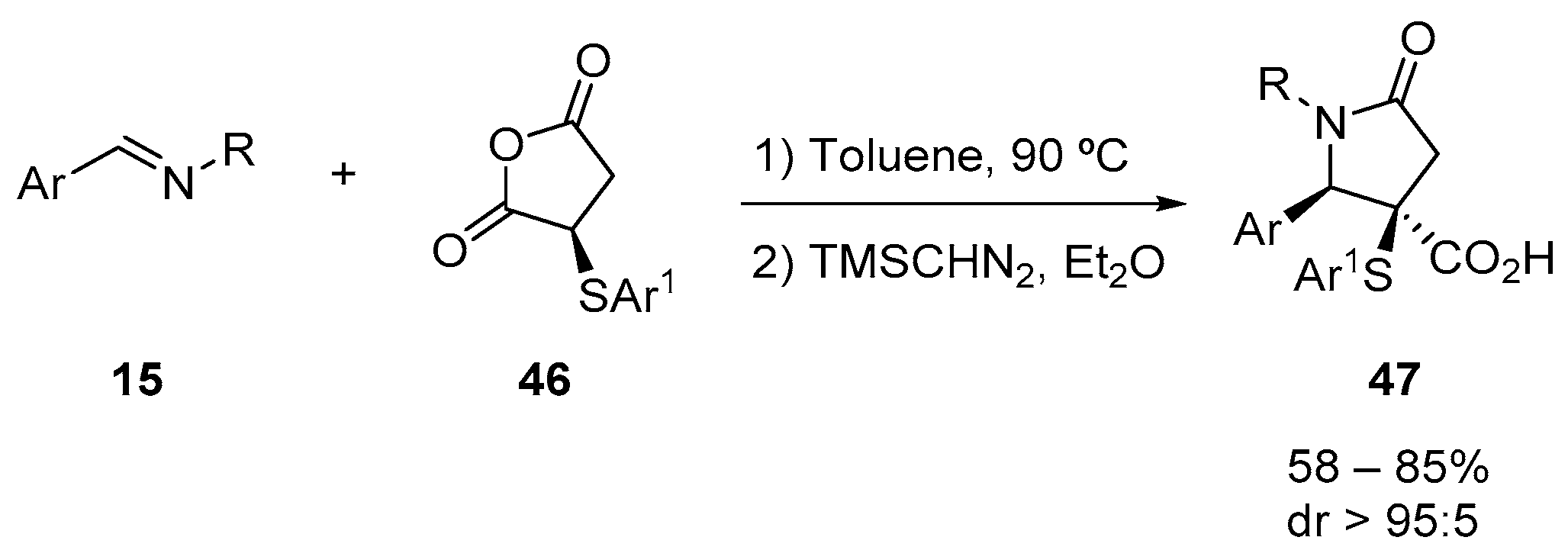
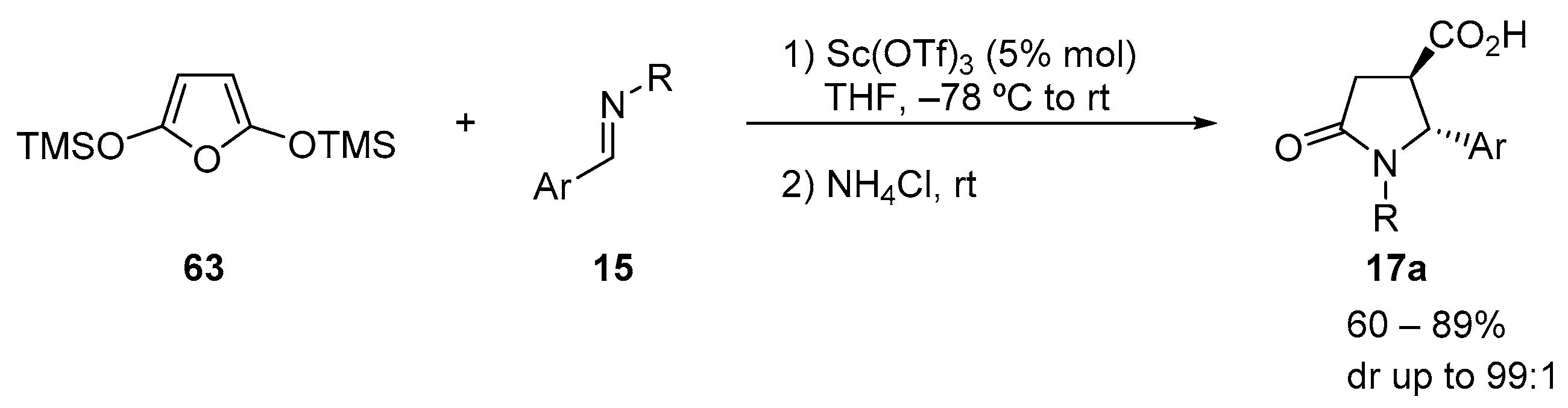
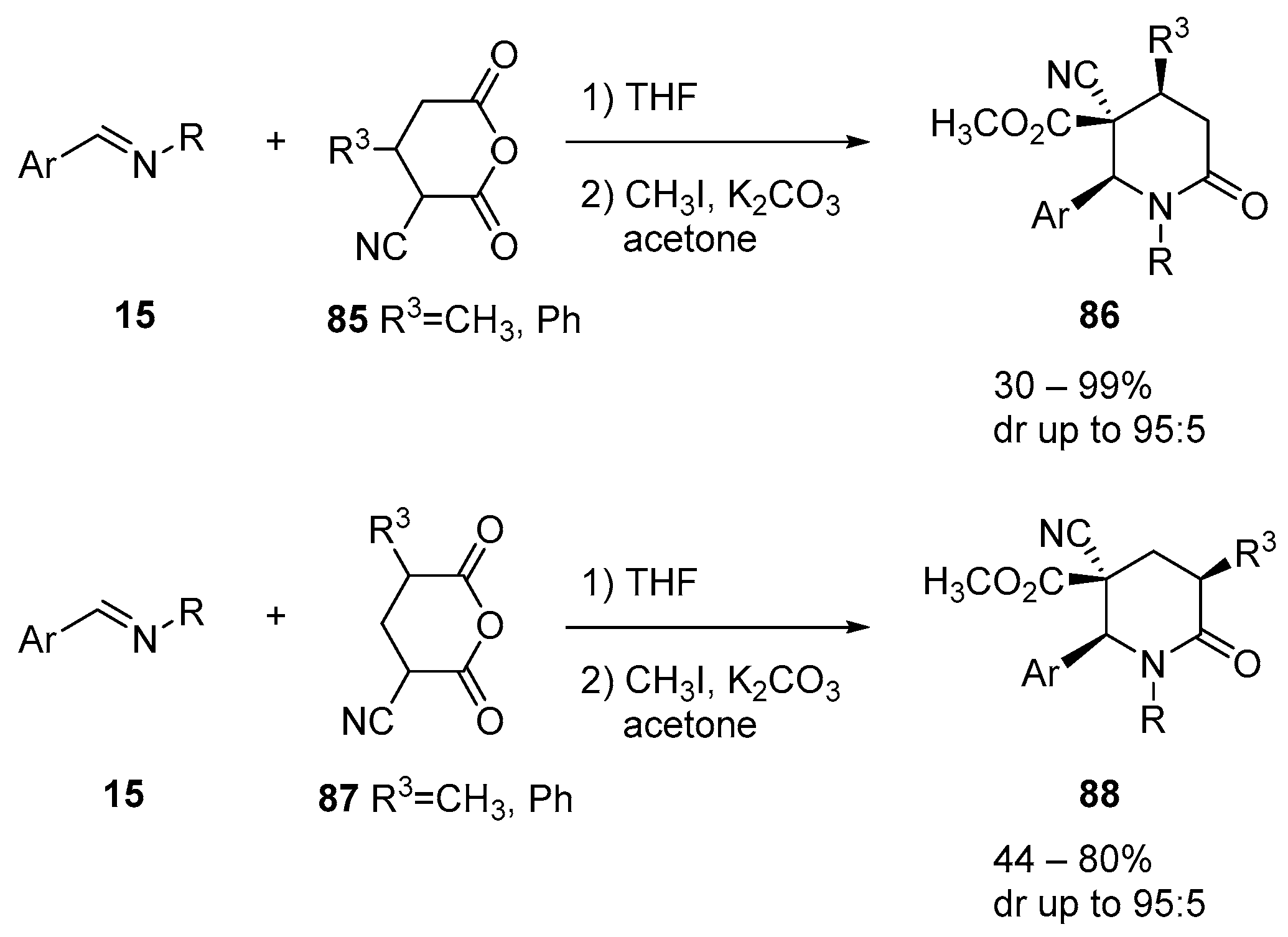
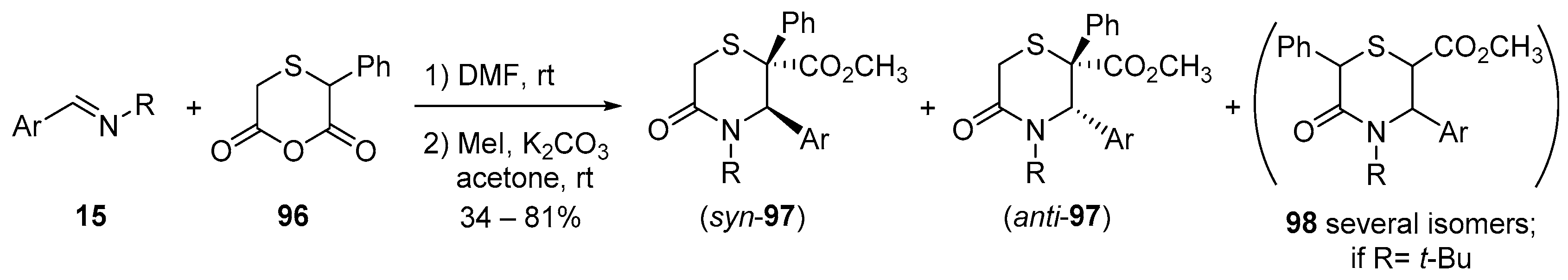
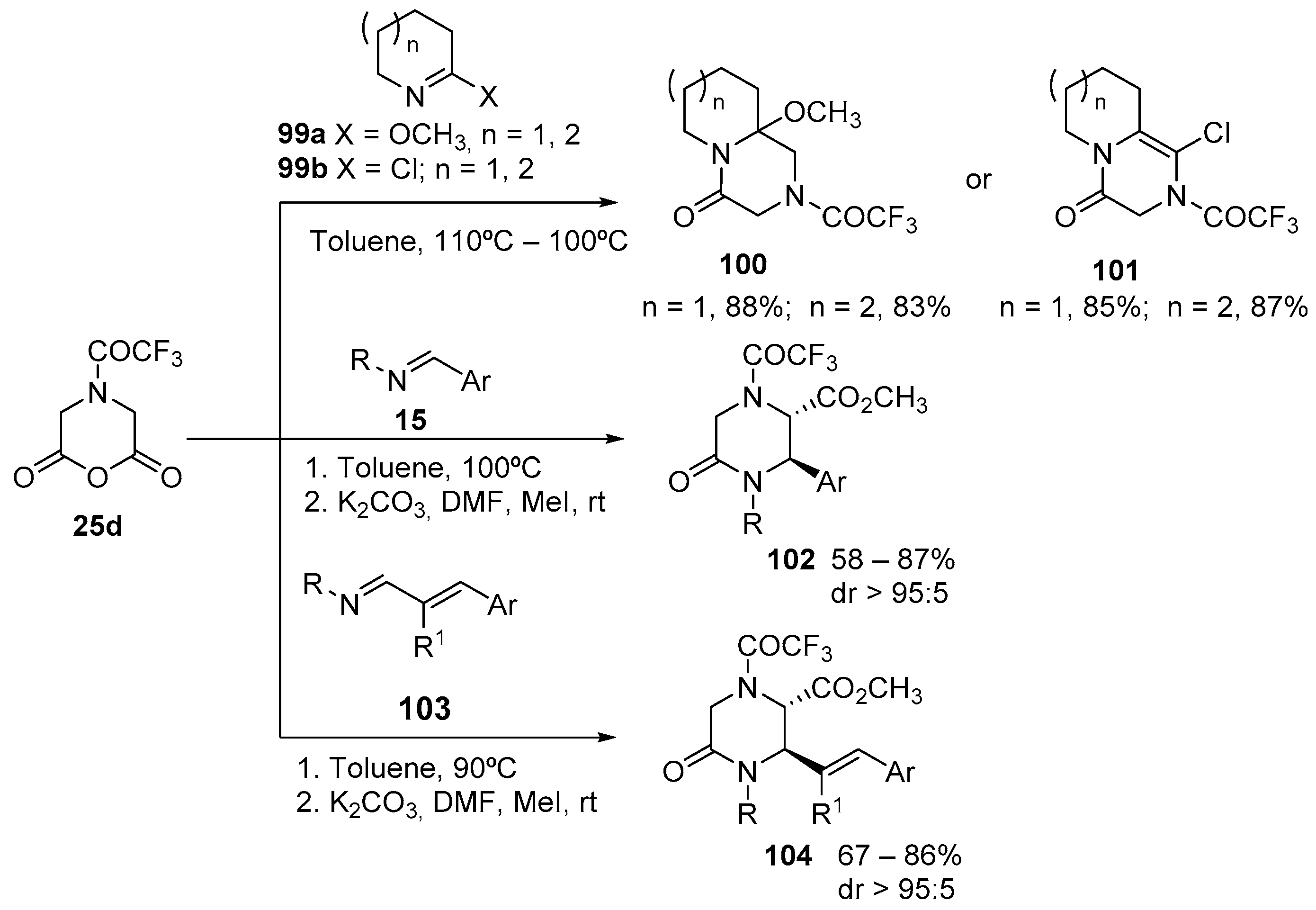
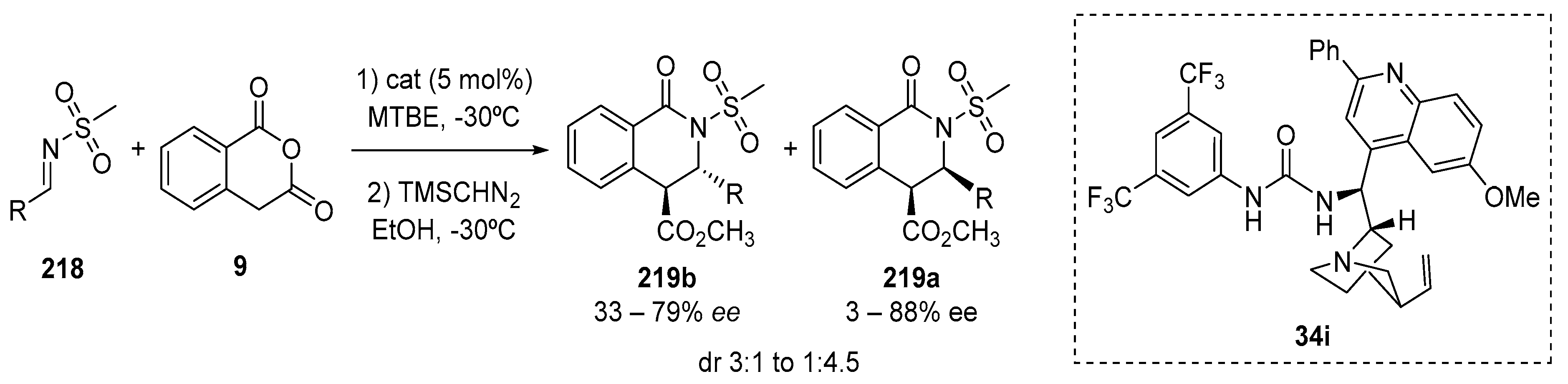
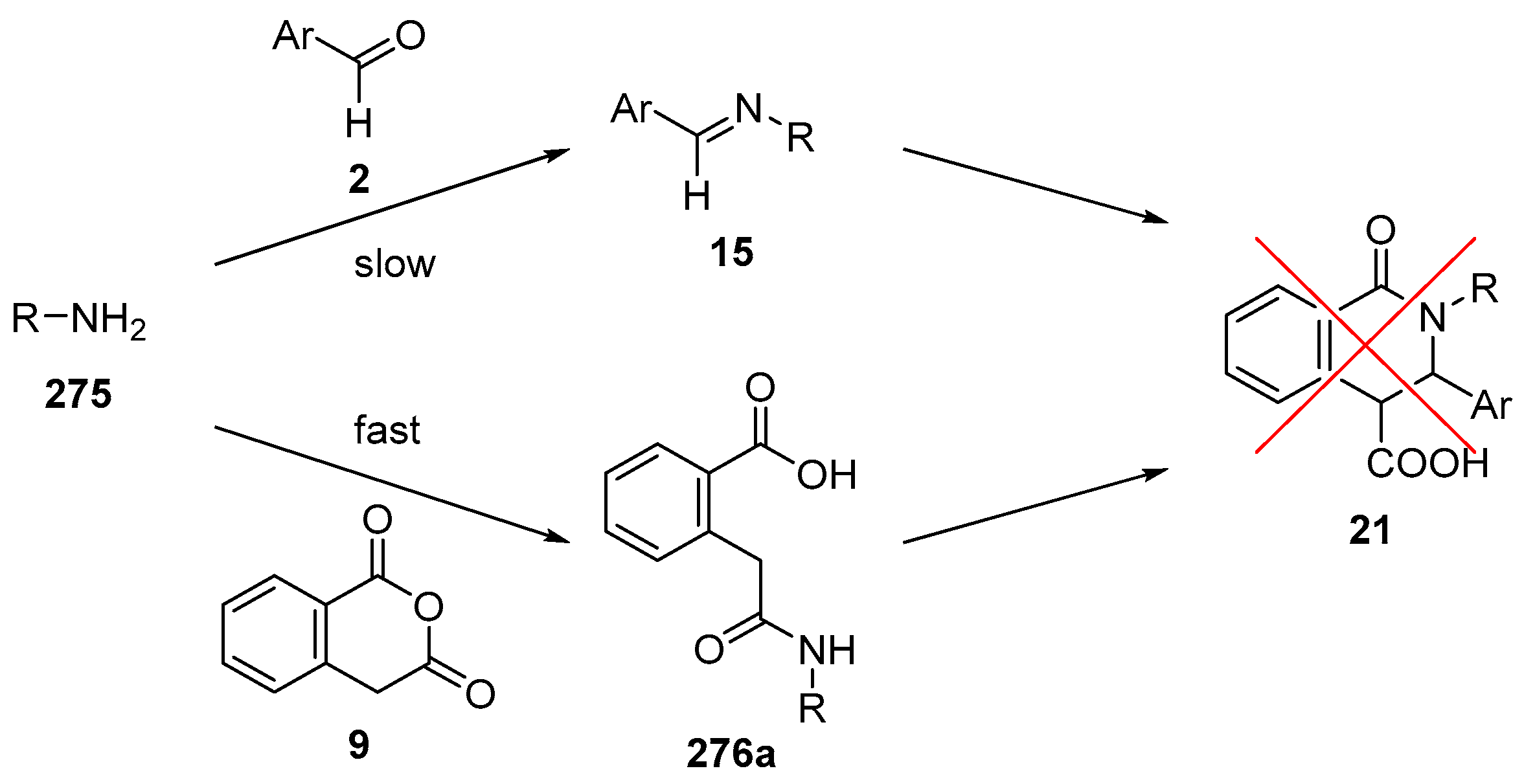
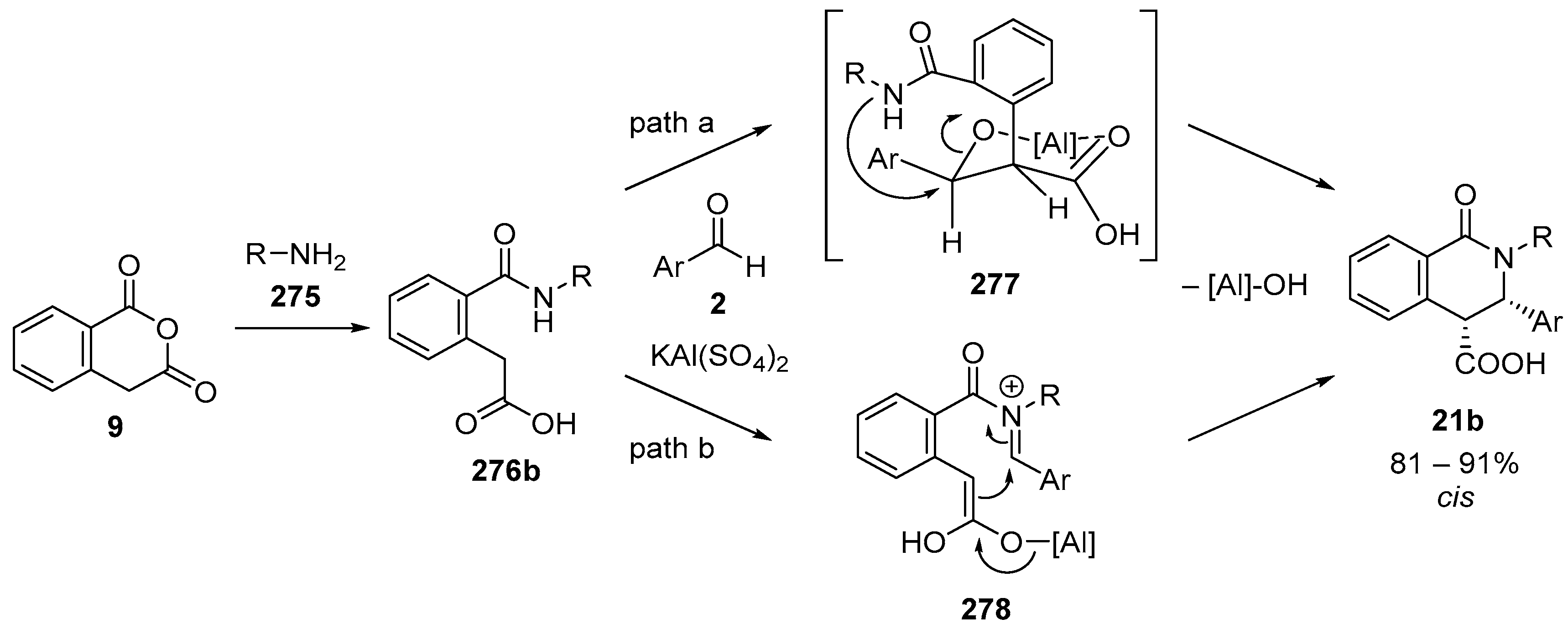
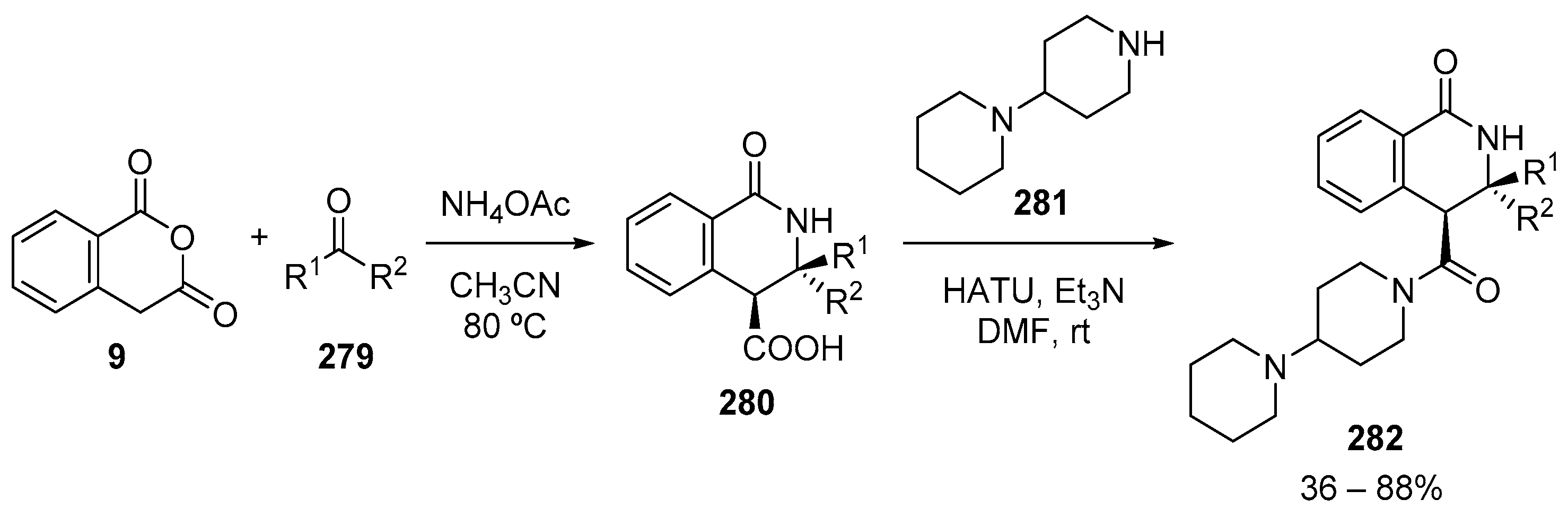
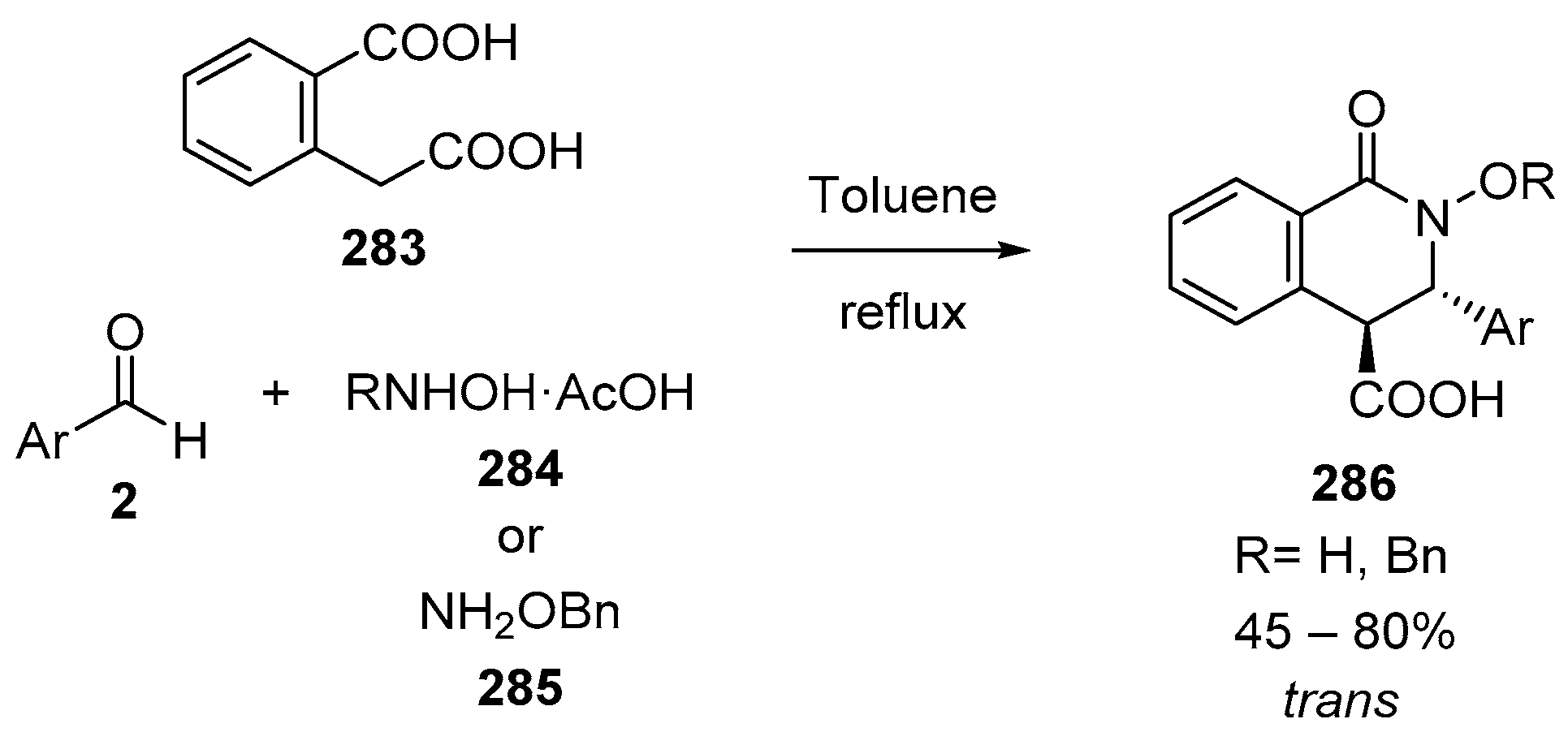
4.3. Catalysed and Thermal 3C-CCRs
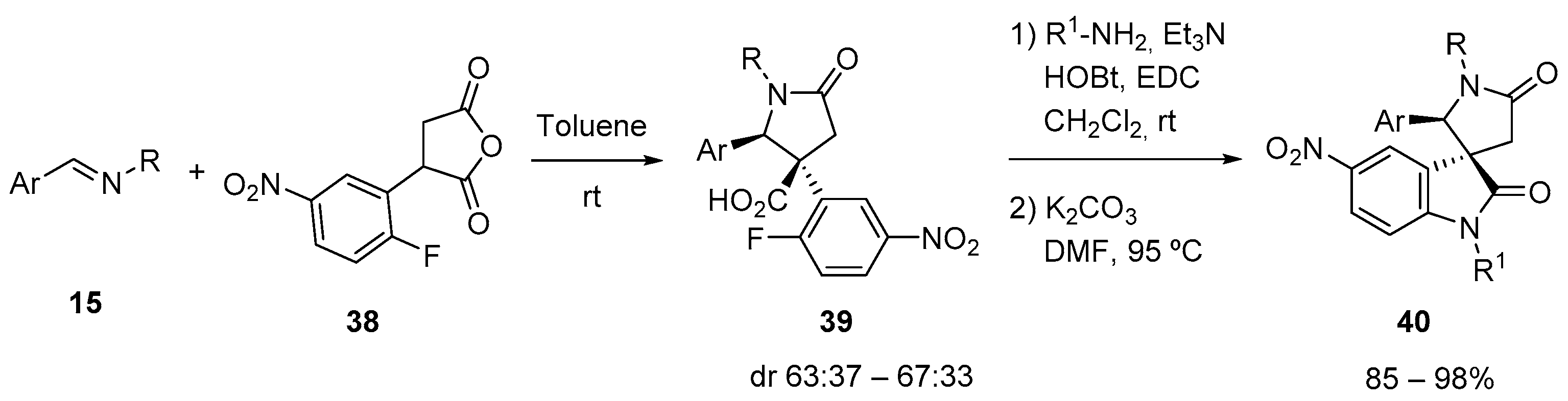
4.4. 3C-CCR with Diacids
5. Four-Component Castagnoli–Cushman Reactions (4C-CCRs)
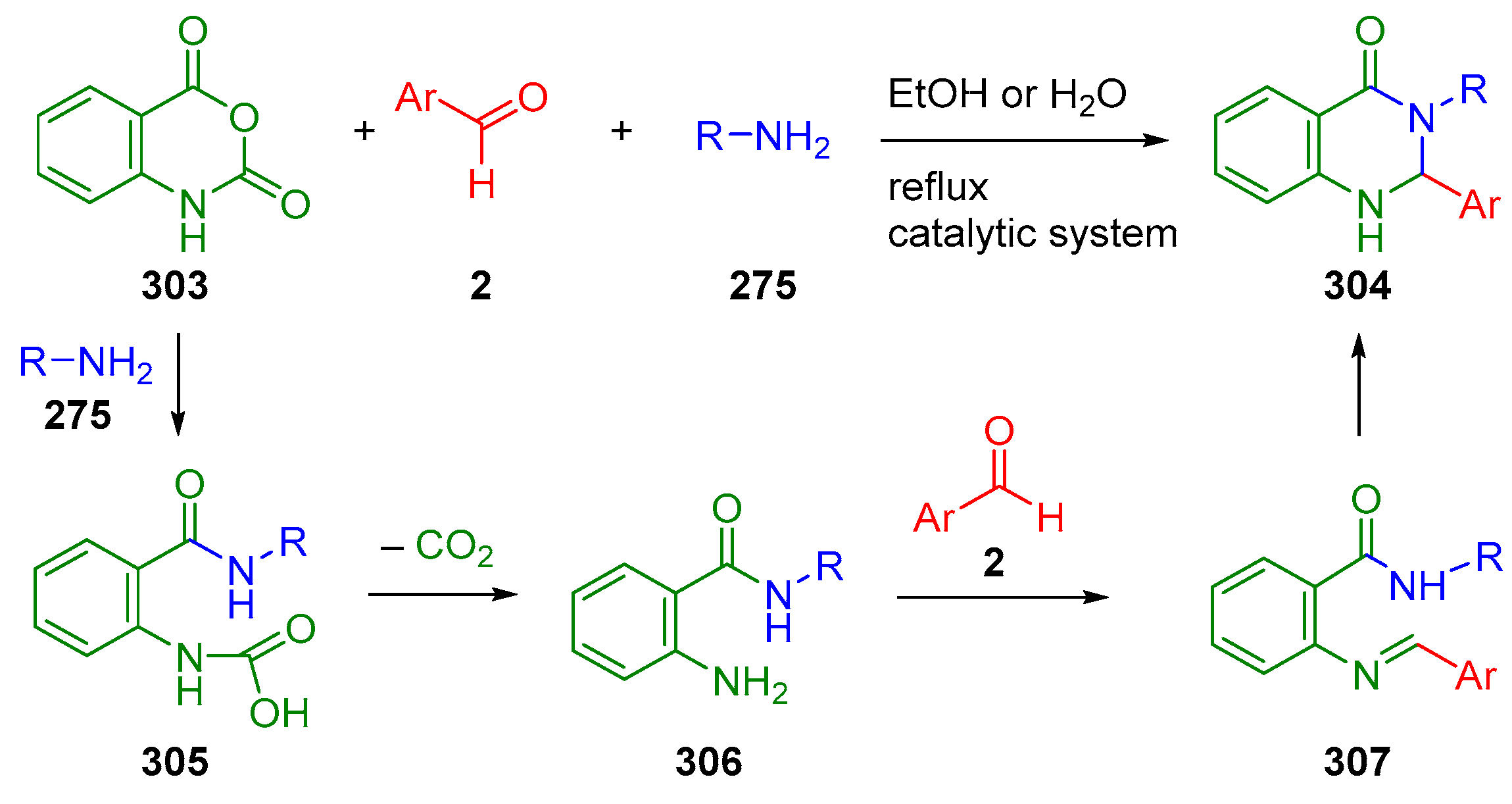
6. Reactions with Isatoic Anhydride
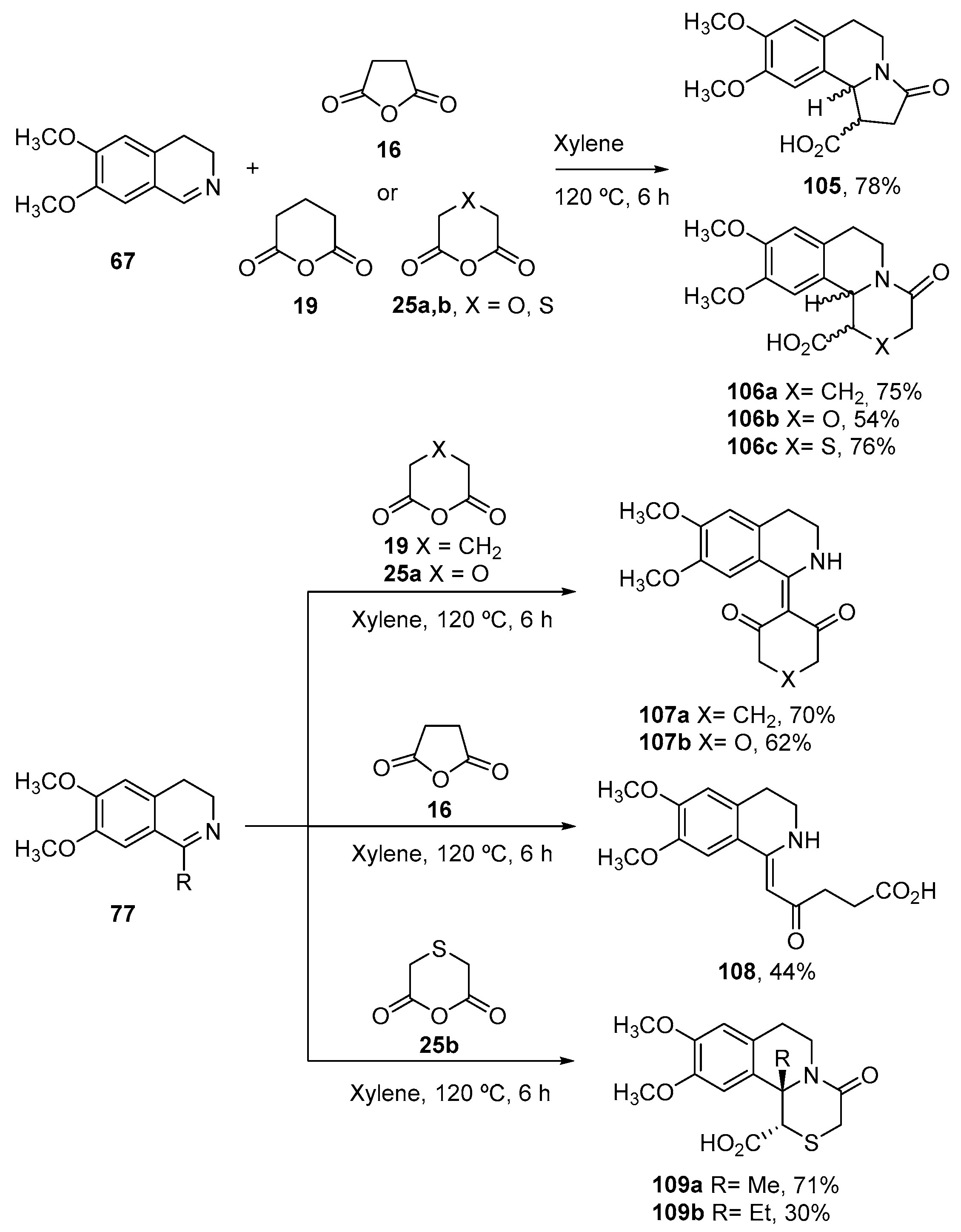
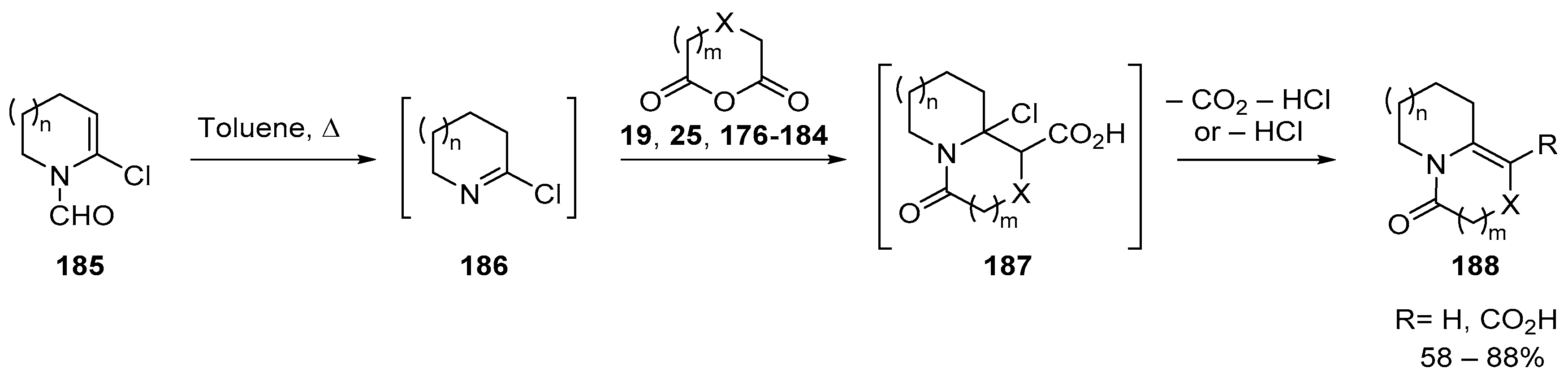
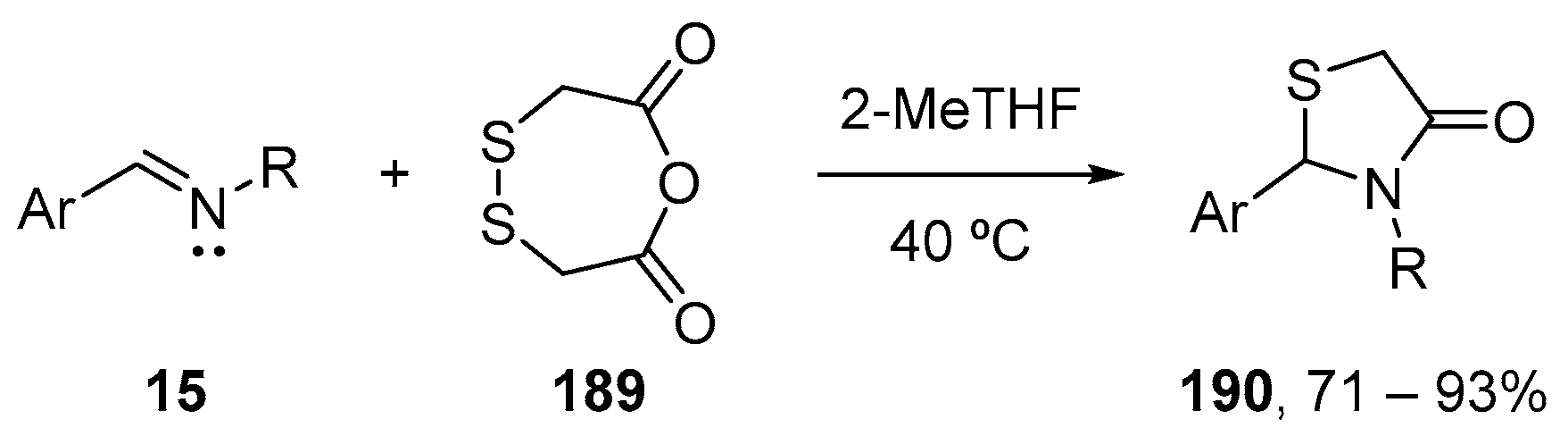
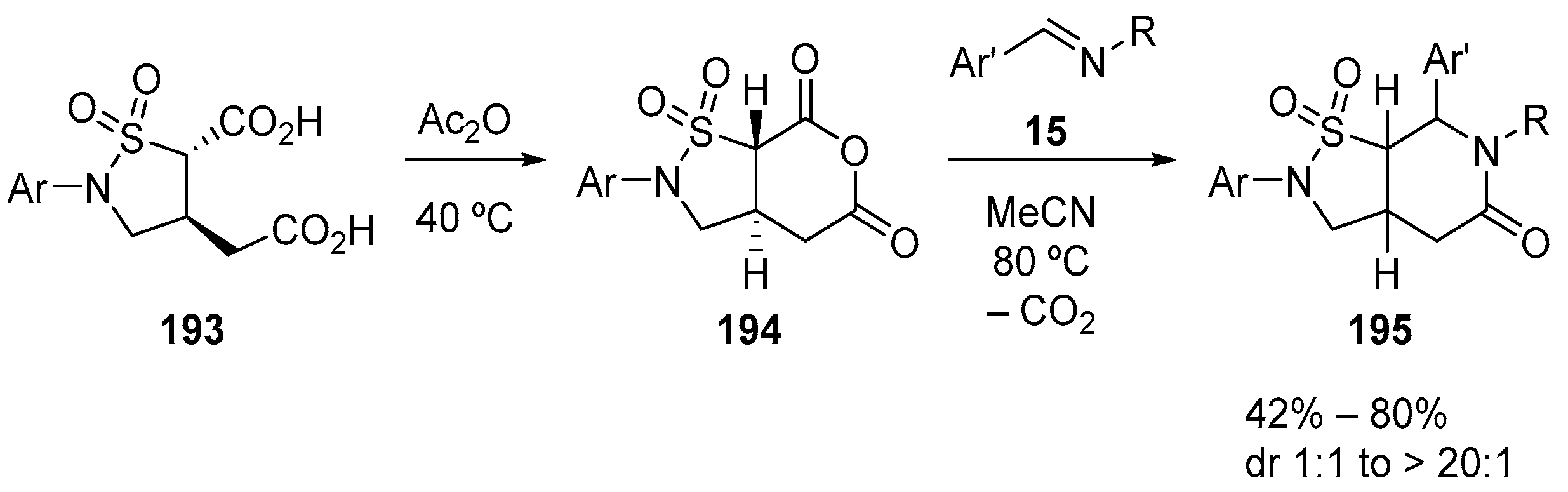
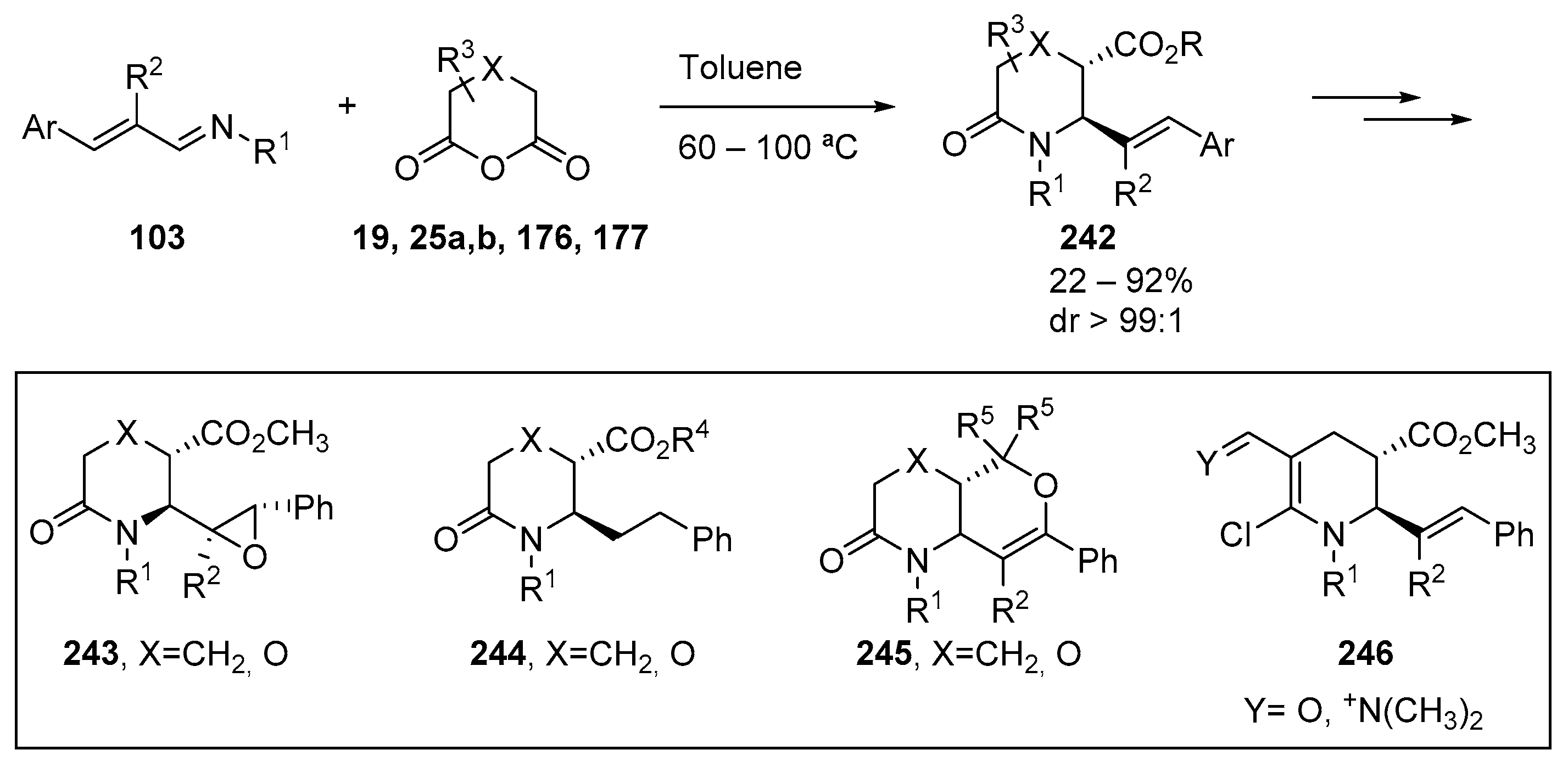
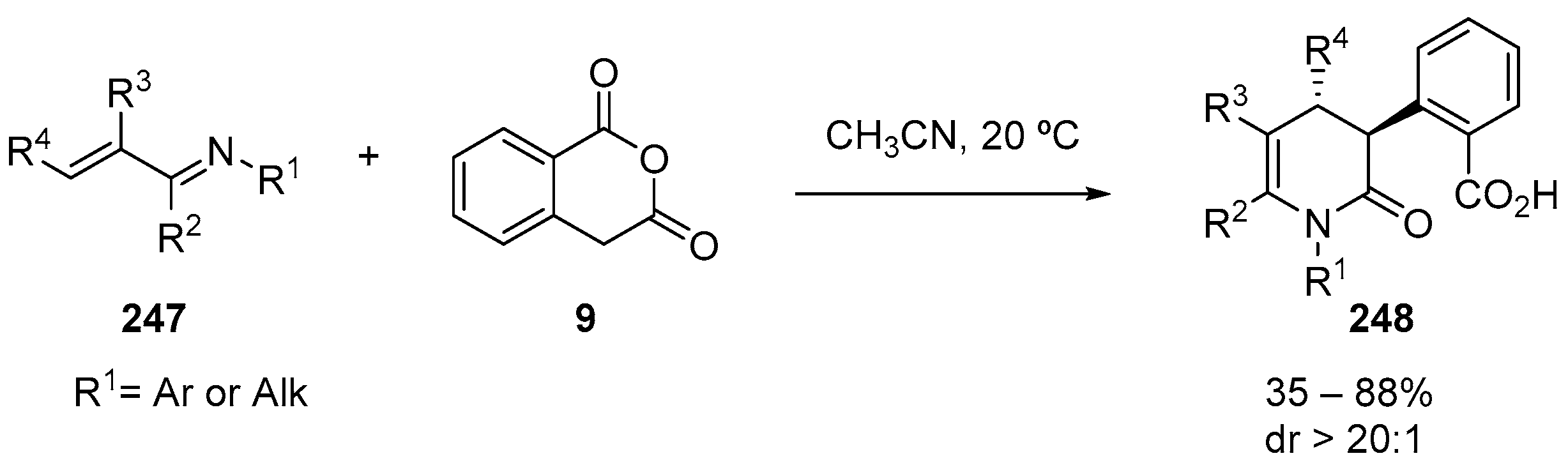
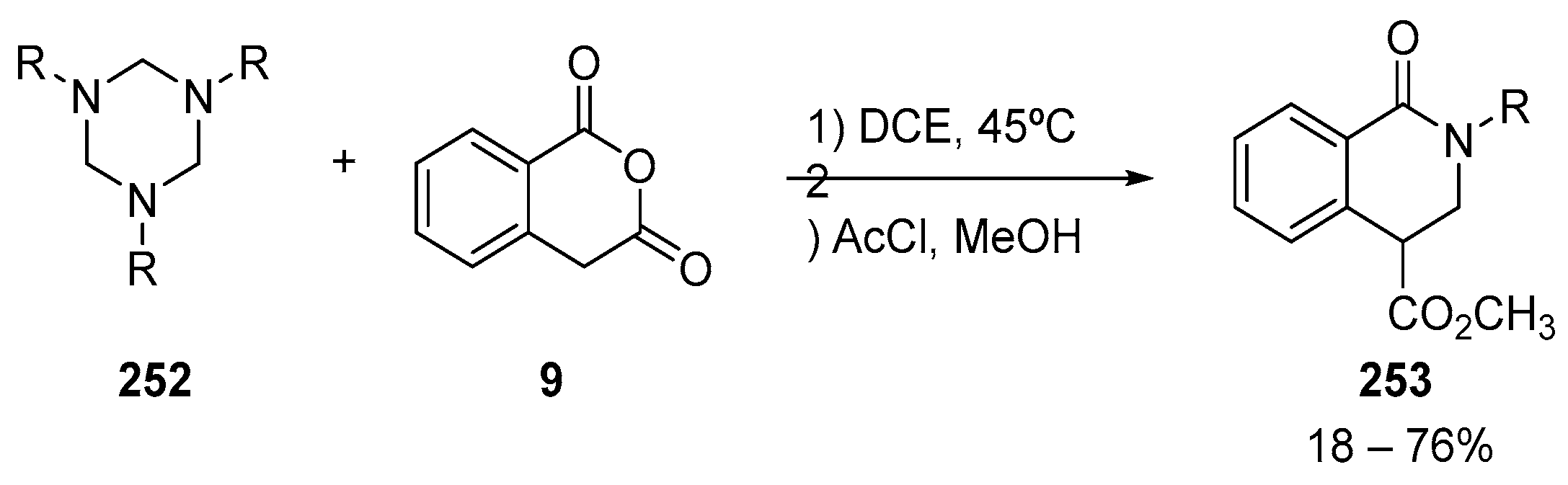
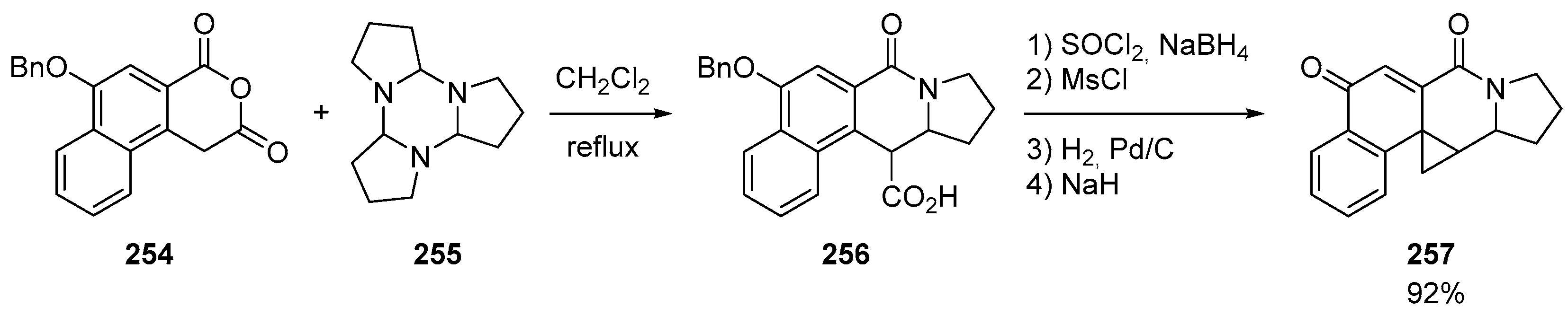
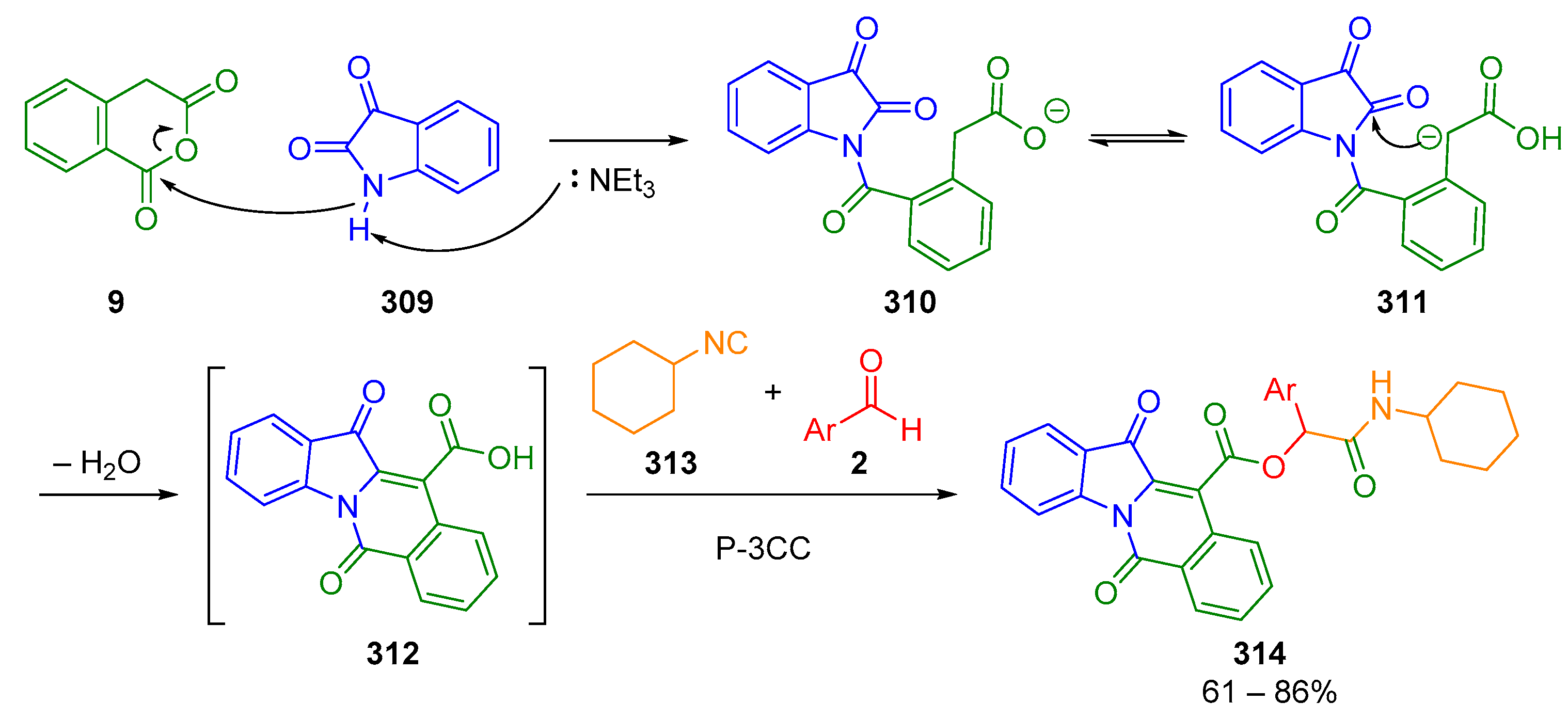
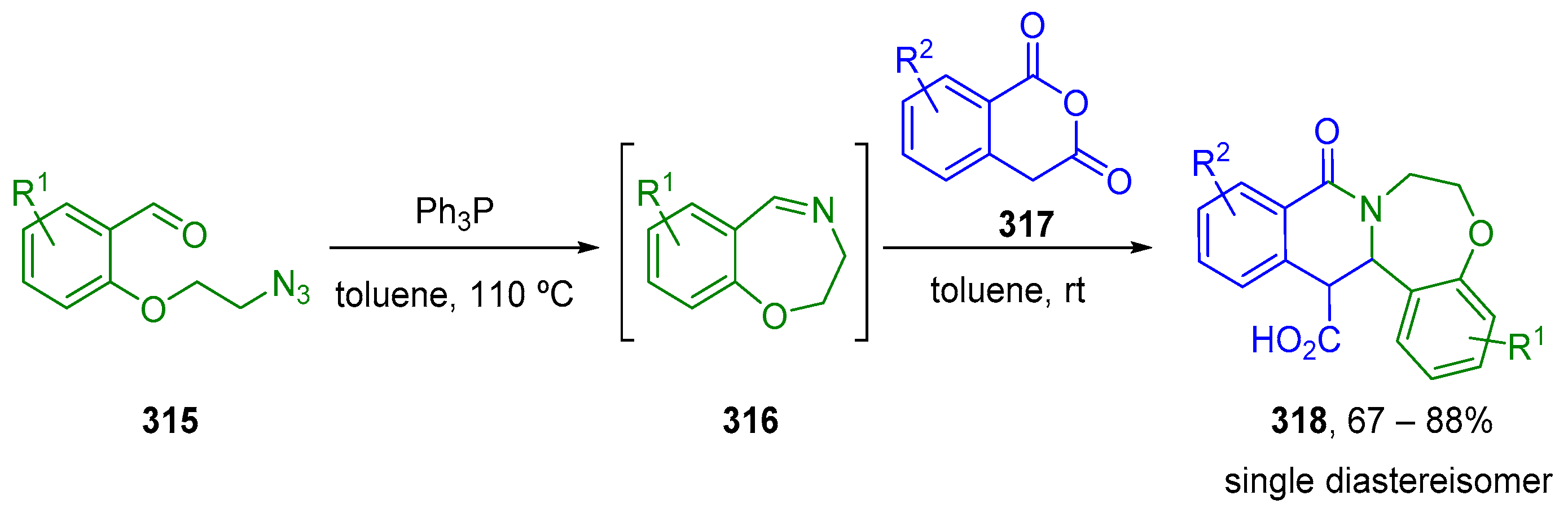
7. Miscellaneous
8. Conclusions and Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Govindachari, T.R.; Chinnasamy, P.; Rajeswari, S.; Chandrasekaran, S.; Premila, M.S.; Natarajan, S.; Nagarajan, K.; Pai, B.R. Some Recent Work on Schiff Bases, Imines and Iminium Salts in Synthetic Heterocyclic Chemistry—A Review. Heterocycles 1984, 22, 585–655. [Google Scholar] [CrossRef]
- González-López, M.; Shaw, J. Cyclic Anhydrides in Formal Cycloadditions and Multicomponent Reactions. Chem. Rev. 2009, 109, 164–189. [Google Scholar] [CrossRef]
- Martin, K.S.; Shaw, J.T.; Younai, A. Anhydride-based multicomponent reactions. In Multicomponent Reactions in Organic Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 379–400. [Google Scholar] [CrossRef]
- Krasavin, M.; Dar’In, D. Current diversity of cyclic anhydrides for the Castagnoli–Cushman-type formal cycloaddition reactions: Prospects and challenges. Tetrahedron Lett. 2016, 57, 1635–1640. [Google Scholar] [CrossRef]
- Mal, D. [4+2] annulation of homophthalic anhydrides. In Anionic Annulations in Organic Synthesis; Elsevier: Amsterdam, The Netherlands, 2019; pp. 223–246. [Google Scholar] [CrossRef]
- Perkin, W.H., VI. —On the artificial production of coumarin and formation of its homologues. J. Chem. Soc. 1868, 21, 53–63. [Google Scholar] [CrossRef]
- Rosen, T. The perkin reaction. In Comprehensive Organic Synthesis; Trost, B.M., Fleming, I., Eds.; Pergamon: Oxford, UK, 1991; pp. 395–408. [Google Scholar] [CrossRef]
- Molnar, M.; Lončarić, M.; Kovač, M. Green Chemistry Approaches to the Synthesis of Coumarin Derivatives. Curr. Org. Chem. 2020, 24, 4–43. [Google Scholar] [CrossRef]
- Sinha, A.K.; Kumar, V.; Sharma, A.; Sharma, A.; Kumar, R. An unusual, mild and convenient one-pot two-step access to (E)-stilbenes from hydroxy-substituted benzaldehydes and phenylacetic acids under microwave activation: A new facet of the classical Perkin reaction. Tetrahedron 2007, 63, 11070–11077. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Karri, P. Revised mechanism and improved methodology for the Perkin condensation. Resuscitation of the mechanism involving benzal acetate and the improbability of the enolate of acetic anhydride. Tetrahedron Lett. 2006, 47, 2249–2251. [Google Scholar] [CrossRef]
- Edwards, M.; Rourk, P.M.; Riby, P.G.; Mendham, A.P. Not quite the last word on the Perkin reaction. Tetrahedron 2014, 70, 7245–7252. [Google Scholar] [CrossRef]
- Fittig, R. Über die ungesättigten säuren. Justus Liebigs Ann. Chem. 1883, 216, 26–29. [Google Scholar] [CrossRef]
- Jayne, H.W. Ueber das Phenyl-Butyrolacton und die Phenyl-Paraconsäure. Justus Liebigs Ann. Chem. 1883, 216, 97–114. [Google Scholar] [CrossRef]
- Müller, E. Zur Kenntnis der Perkinschen Synthese. Justus Liebigs Ann. Chem. 1931, 491, 251–265. [Google Scholar] [CrossRef]
- Johnson, J.R. The Perkin Reaction and Related Reactions. Org. React. 2011, 1, 210–265. [Google Scholar] [CrossRef]
- Aiello, M.L.; Farid, U.; Trujillo, C.; Twamley, B.; Connon, S.J. Catalytic Asymmetric Cycloadditions between Aldehydes and Enolizable Anhydrides: Cis-Selective Dihydroisocoumarin Formation. J. Org. Chem. 2018, 83, 15499–15511. [Google Scholar] [CrossRef]
- Farid, U.; Aiello, M.L.; Connon, S.J. Highly Enantioselective Catalytic Kinetic Resolution of α-Branched Aldehydes through Formal Cycloaddition with Homophthalic Anhydrides. Chemistry 2019, 25, 10074–10079. [Google Scholar] [CrossRef] [PubMed]
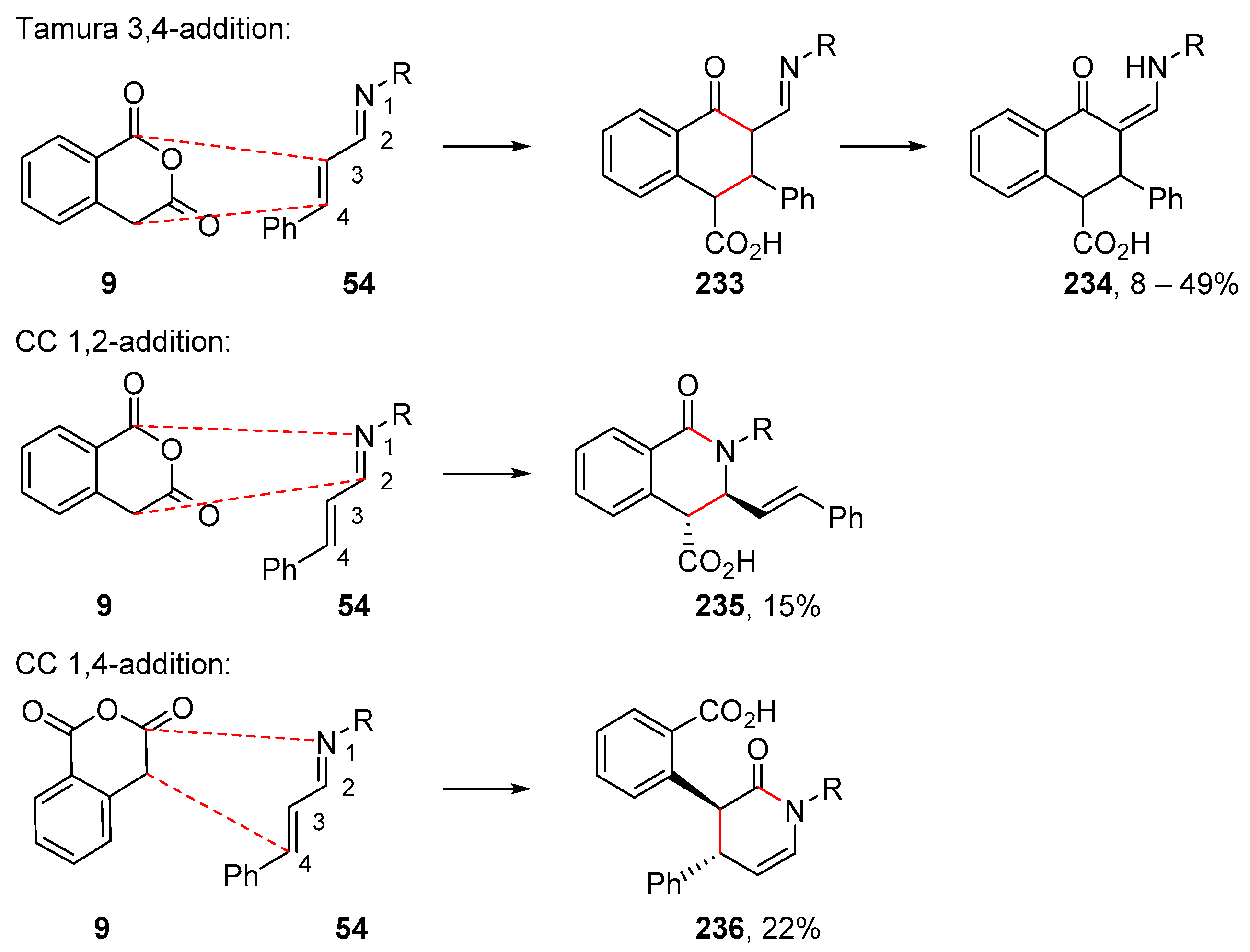
- Tamura, Y.; Wada, A.; Sasho, M.; Kita, Y. Cycloaddition of homophthalic anhydride: A new and simple route to linearly condensed phenolic compoumds. Tetrahedron Lett. 1981, 22, 4283–4286. [Google Scholar] [CrossRef]
- Fujioka, H.; Yamamoto, H.; Kondo, H.; Annoura, H.; Kita, Y. Regio- and stereo-selective construction of anthracyclinones: Total synthesis of (–)-γ-rhodomycinone. J. Chem. Soc. Chem. Commun. 1989, 20, 1509–1511. [Google Scholar] [CrossRef]
- Cox, C.D.; Siu, T.; Danishefsky, S.J. Studies Directed toward the Total Synthesis of Lactonamycin: Control of the Sense of Cycloaddition of a Quinone through Directed Intramolecular Catalysis. Angew. Chem. Int. Ed. 2003, 42, 5625–5629. [Google Scholar] [CrossRef]
- Lockett-Walters, B.; Trujillo, C.; Twamley, B.; Connon, S. The base-catalysed Tamura cycloaddition reaction: Calculation, mechanism, isolation of intermediates and asymmetric catalysis. Chem. Commun. 2019, 55, 11283–11286. [Google Scholar] [CrossRef]
- Manoni, F.; Connon, S.J. Catalytic Asymmetric Tamura Cycloadditions. Angew. Chem. Int. Ed. 2014, 53, 2628–2632. [Google Scholar] [CrossRef]
- Nath, U.; Pan, S.C. Organocatalytic Asymmetric Tamura Cycloaddition with α- Branched Nitroolefins: Synthesis of Functionalized 1-Tetralones. J. Org. Chem. 2017, 82, 3262–3269. [Google Scholar] [CrossRef]
- Xu, H.; Sha, F.; Li, Q.; Wu, X.-Y. Catalytic asymmetric Tamura cycloaddition of homophthalic anhydrides with 2-arylidene-1,3-diones. Org. Biomol. Chem. 2018, 16, 7214–7222. [Google Scholar] [CrossRef] [PubMed]
- Castagnoli, N. Condensation of succinic anhydride with N-benzylidene-N-methylamine. Stereoselective synthesis of trans- and cis-1-methyl-4-carboxy-5-phenyl-2-pyrrolidinone. J. Org. Chem. 1969, 34, 3187–3189. [Google Scholar] [CrossRef] [PubMed]
- Cushman, M.; Castagnoli, N., Jr. Novel approach to the synthesis of nitrogen analogs of the tetrahydrocannabinols. J. Org. Chem. 1973, 38, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Haimova, M.; Mollov, N.; Ivanova, S.; Dimitrova, A.; Ognyanov, V. A highly stereoselective synthesis of 3,4-dihydro-1(2H)-isoquinolinones and 8-oxoberbines from homophthalic anhydrides and azomethines. Tetrahedron 1977, 33, 331–336. [Google Scholar] [CrossRef]
- Dar’In, D.; Bakulina, O.; Nikolskaya, S.; Gluzdikov, I.; Krasavin, M. The rare cis-configured trisubstituted lactam products obtained by the Castagnoli–Cushman reaction in N,N-dimethylformamide. RSC Adv. 2016, 6, 49411–49415. [Google Scholar] [CrossRef]
- Masse, C.E.; Ng, P.Y.; Fukase, Y.; Sánchez-Roselló, M.; Shaw, J.T. Divergent Structural Complexity from a Linear Reaction Sequence: Synthesis of Fused and Spirobicyclic γ-Lactams from Common Synthetic Precursors. J. Comb. Chem. 2006, 8, 293–296. [Google Scholar] [CrossRef]
- Pattawong, O.; Tan, D.Q.; Fettinger, J.C.; Shaw, J.T.; Cheong, P.H.-Y. Stereocontrol in Asymmetric γ-Lactam Syntheses from Imines and Cyanosuccinic Anhydrides. Org. Lett. 2013, 15, 5130–5133. [Google Scholar] [CrossRef]
- Tan, D.Q.; Younai, A.; Pattawong, O.; Fettinger, J.C.; Cheong, P.H.-Y.; Shaw, J.T. Stereoselective Synthesis of γ-Lactams from Imines and Cyanosuccinic Anhydrides. Org. Lett. 2013, 15, 5126–5129. [Google Scholar] [CrossRef]
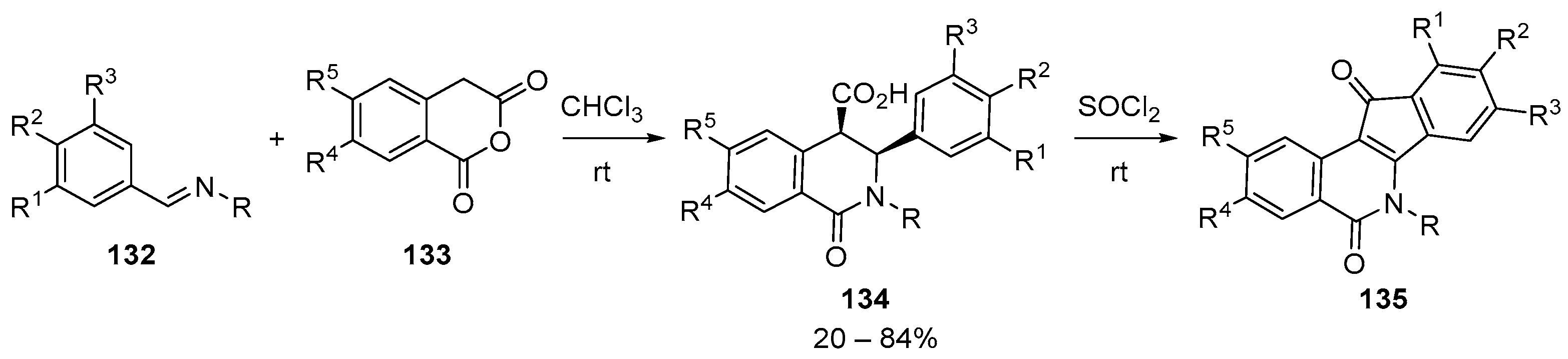
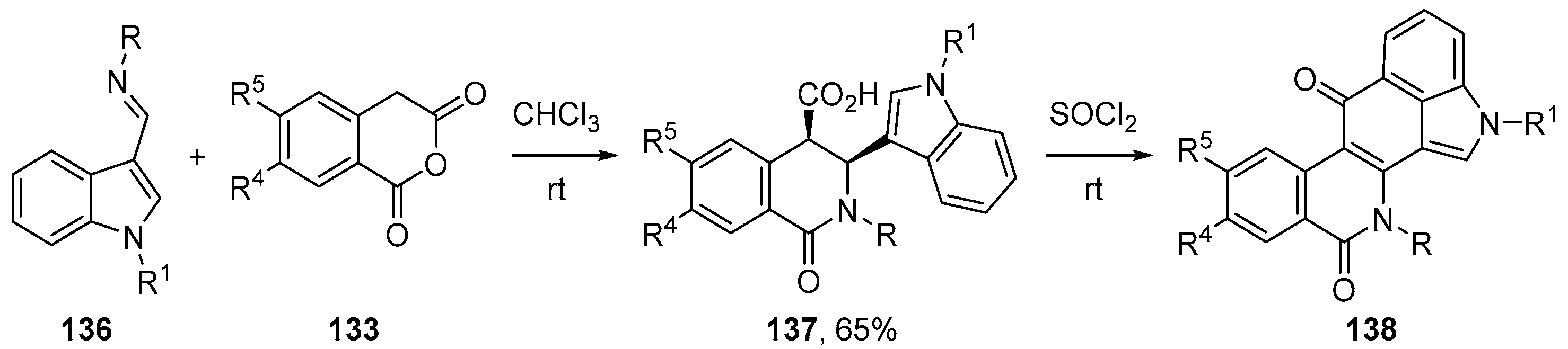
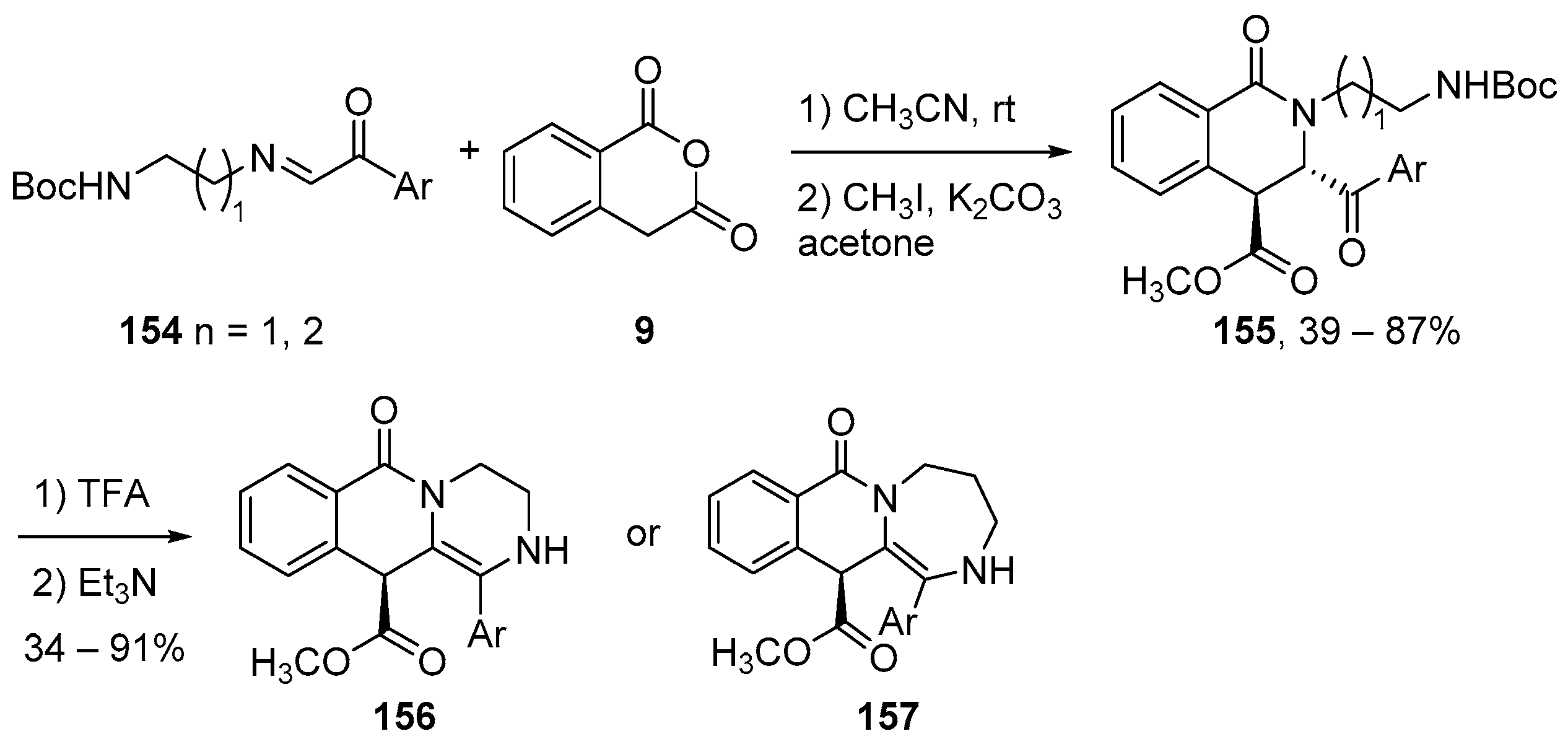
- Cushman, M. Design and Synthesis of Indenoisoquinolines Targeting Topoisomerase I and Other Biological Macromolecules for Cancer Chemotherapy. J. Med. Chem. 2021, 64, 17572–17600. [Google Scholar] [CrossRef]
- Elsayed, M.S.A.; Zeller, M.; Cushman, M. Synthesis of indolo[4,3-bc]phenanthridine-6,11(2H,12H)-diones using the schiff base–homophthalic anhydride cyclization reaction. Synth. Commun. 2016, 46, 1902–1908. [Google Scholar] [CrossRef]
- Bayles, T.; Guillou, C. Trifluoroethanol Promoted Castagnoli–Cushman Cycloadditions of Imines with Homophthalic Anhydride. Molecules 2022, 27, 844. [Google Scholar] [CrossRef] [PubMed]
- Lepikhina, A.; Bakulina, O.; Dar’In, D.; Krasavin, M. The first solvent-free synthesis of privileged γ- and δ-lactams via the Castagnoli–Cushman reaction. RSC Adv. 2016, 6, 83808–83813. [Google Scholar] [CrossRef]
- Tang, Y.; Fettinger, J.C.; Shaw, J.T. One-Step Synthesis of Complex Nitrogen Heterocycles from Imines and Alkyl-Substituted Maleic Anhydrides. Org. Lett. 2009, 11, 3802–3805. [Google Scholar] [CrossRef] [PubMed]
- Pérard-Viret, J.; Souquet, F.; Manisse, M.-L.; Royer, J. An expeditious total synthesis of (±)-jamtine using condensation between imine and acid anhydride. Tetrahedron Lett. 2010, 51, 96–98. [Google Scholar] [CrossRef]
- Vytla, D.; Shaw, P.; Velayuthaperumal, R.; Emmadi, J.; Mathur, A.; Roy, A. Microwave accelerated Castagnoli-Cushman reaction: Synthesis of novel 6,7,8,9-tetrahydropyrido[3′,2′:4,5]pyrrolo[1,2-a]pyrazines. Tetrahedron Lett. 2021, 68, 152943–152947. [Google Scholar] [CrossRef]
- Cushman, M.; Madaj, E.J. A study and mechanistic interpretation of the electronic and steric effects that determine the stereochemical outcome of the reaction of Schiff bases with homophthalic anhydride and 3-phenylsuccinic anhydride. J. Org. Chem. 1987, 52, 907–915. [Google Scholar] [CrossRef]
- Castagnoli, N., Jr.; Cushman, M. Condensation of succinic anhydrides with Schiff bases. Scope and mechanism. J. Org. Chem. 1971, 36, 3404–3406. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.Q.; Atherton, A.L.; Smith, A.J.; Soldi, C.; Hurley, K.; Fettinger, J.C.; Shaw, J.T. Synthesis of a γ-Lactam Library via Formal Cycloaddition of Imines and Substituted Succinic Anhydrides. ACS Comb. Sci. 2012, 14, 218–223. [Google Scholar] [CrossRef]
- Wei, J.; Shaw, J.T. Diastereoselective Synthesis of γ-Lactams by a One-Pot, Four-Component Reaction. Org. Lett. 2007, 9, 4077–4080. [Google Scholar] [CrossRef]
- Dar’In, D.; Bakulina, O.; Chizhova, M.; Krasavin, M. New Heterocyclic Product Space for the Castagnoli–Cushman Three-Component Reaction. Org. Lett. 2015, 17, 3930–3933. [Google Scholar] [CrossRef]
- Kaneti, J.; Bakalova, S.M.; Pojarlieff, I.G. Schiff Base Addition to Cyclic Dicarboxylic Anhydrides: An Unusual Concerted Reaction. An MO and DFT Theoretical Study. J. Org. Chem. 2003, 68, 6824–6827. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Z.; Levin, A.; Emge, T.J.; Rablen, P.R.; Floyd, D.M.; Knapp, S. N-Methylimidazole Promotes the Reaction of Homophthalic Anhydride with Imines. J. Org. Chem. 2014, 79, 7593–7599. [Google Scholar] [CrossRef]
- Kozekov, I.D.; Koleva, R.I.; Palamareva, M.D. Newtrans/cistetrahydroisoquinolines. 1.trans-2-benzyl-3-(l-methyl-1h-pyrrol-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolin-1-ones and corresponding tetrahydroisoquinolines. J. Heterocycl. Chem. 2002, 39, 229–236. [Google Scholar] [CrossRef]
- Hong, J.; Wang, Z.; Levin, A.; Emge, T.J.; Floyd, D.M.; Knapp, S. Dimerization and comments on the reactivity of homophthalic anhydride. Tetrahedron Lett. 2015, 56, 3001–3004. [Google Scholar] [CrossRef]
- Kandinska, M.I.; Kozekov, I.D.; Palamareva, M.D. Synthesis of new trans-2-benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones. Molecules 2006, 11, 403–414. [Google Scholar] [CrossRef]
- Polyak, D.; Phung, N.; Liu, J.; Barrows, R.; Emge, T.J.; Knapp, S. Stereochemistry and reactivity of the HPA-imine Mannich intermediate. Tetrahedron Lett. 2017, 58, 3879–3883. [Google Scholar] [CrossRef] [PubMed]
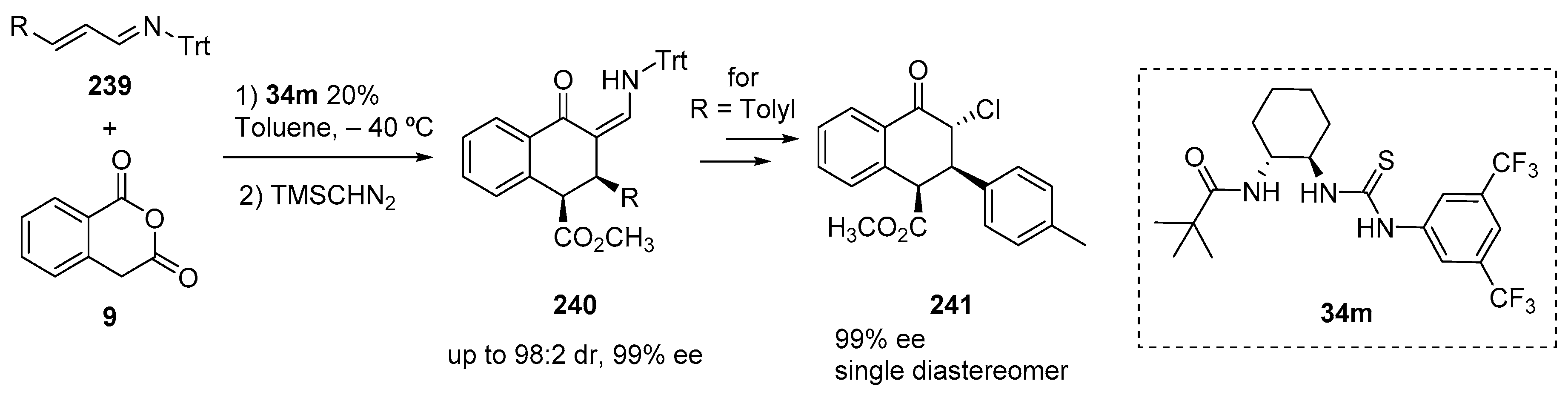
- Jarvis, C.L.; Hirschi, J.S.; Vetticatt, M.J.; Seidel, D. Catalytic Enantioselective Synthesis of Lactams through Formal [4+2] Cycloaddition of Imines with Homophthalic Anhydride. Angew. Chem. Int. Ed. 2017, 56, 2670–2674. [Google Scholar] [CrossRef]
- Cronin, S.A.; Collar, A.G.; Gundala, S.; Cornaggia, C.; Torrente, E.; Manoni, F.; Botte, A.; Twamley, B.; Connon, S.J. The first catalytic asymmetric cycloadditions of imines with an enolisable anhydride. Org. Biomol. Chem. 2016, 14, 6955–6959. [Google Scholar] [CrossRef]
- Collar, A.G.; Trujillo, C.; Lockett-Walters, B.; Twamley, B.; Connon, S.J. Catalytic Asymmetric γ-Lactam Synthesis from Enolisable Anhydrides and Imines. Chem. A Eur. J. 2019, 25, 7275–7279. [Google Scholar] [CrossRef]
- Vara, Y.; Bello, T.; Aldaba, E.; Arrieta, A.; Pizarro, J.L.; Arriortua, M.I.; Lopez, X.; Cossío, F.P. Trans-Stereoselectivity in the Reaction between Homophthalic Anhydride and Imines. Org. Lett. 2008, 10, 4759–4762. [Google Scholar] [CrossRef]
- Phae-Nok, S.; Pohmakotr, M.; Kuhakarn, C.; Reutrakul, V.; Soorukram, D. Site-Specific Synthesis of β-Fluorinated γ-Butyrolactams via Decarboxylative Fluorination of β-Carboxyl-γ-Butyrolactams. Eur. J. Org. Chem. 2019, 2019, 4710–4720. [Google Scholar] [CrossRef]
- Ng, P.Y.; Masse, C.E.; Shaw, J. Cycloaddition Reactions of Imines with 3-Thiosuccinic Anhydrides: Synthesis of the Tricyclic Core of Martinellic Acid. Org. Lett. 2006, 8, 3999–4002. [Google Scholar] [CrossRef] [PubMed]
- Sorto, N.A.; Di Maso, M.J.; Muñoz, M.A.; Dougherty, R.J.; Fettinger, J.C.; Shaw, J.T. Diastereoselective Synthesis of γ- and δ-Lactams from Imines and Sulfone-Substituted Anhydrides. J. Org. Chem. 2014, 79, 2601–2610. [Google Scholar] [CrossRef]
- Di Maso, M.J.; Nepomuceno, G.M.; Peter, M.A.S.; Gitre, H.H.; Martin, K.S.; Shaw, J.T. Synthesis of (±)-Bisavenanthramide B-6 by an Anionic Anhydride Mannich Reaction. Org. Lett. 2016, 18, 1740–1743. [Google Scholar] [CrossRef]
- Pohmakotr, M.; Yotapan, N.; Tuchinda, P.; Kuhakarn, C.; Reutrakul, V. Highly Diastereoselective Synthesis of β-Carboxy-γ-lactams and Their Ethyl Esters via Sc(OTf)3-Catalyzed Imino Mukaiyama-Aldol Type Reaction of 2,5-Bis(trimethylsilyloxy)furan with Imines. J. Org. Chem. 2007, 72, 5016–5019. [Google Scholar] [CrossRef]
- Howard, S.Y.; Di Maso, M.J.; Shimabukuro, K.; Burlow, N.P.; Tan, D.Q.; Fettinger, J.C.; Malig, T.C.; Hein, J.E.; Shaw, J.T. Mechanistic Investigation of Castagnoli–Cushman Multicomponent Reactions Leading to a Three-Component Synthesis of Dihydroisoquinolones. J. Org. Chem. 2021, 86, 11599–11607. [Google Scholar] [CrossRef] [PubMed]
- Sucu, B.O.; Ocal, N.; Erden, I. Direct synthesis of imidazolidin-4-ones via cycloadditions of imines with a Leuchs’ anyhdride. Tetrahedron Lett. 2015, 56, 2590–2592. [Google Scholar] [CrossRef]
- Di Maso, M.J.; Snyder, K.M.; Fernandes, F.D.S.; Pattawong, O.; Tan, D.Q.; Fettinger, J.C.; Cheong, P.H.; Shaw, J.T. Diastereoselective Synthesis of and Mechanistic Understanding for the Formation of 2-Piperidinones from Imines and Cyano-Substituted Anhydrides. Chem. A Eur. J. 2016, 22, 4794–4801. [Google Scholar] [CrossRef]
- Firsov, A.; Bakulina, O.; Dar’In, D.V.; Guranova, N.; Krasavin, M. Further Insight into the Castagnoli–Cushman-type Synthesis of 1,4,6-Trisubstituted 1,6-Dihydropyridin-2-(3H)-ones from 3-Arylglutaconic Acid Anhydrides. J. Org. Chem. 2020, 85, 6822–6829. [Google Scholar] [CrossRef]
- Levashova, E.; Firsov, A.; Bakulina, O.; Peshkov, A.; Kanov, E.; Gainetdinov, R.R.; Krasavin, M. Rare cis-configured 2,4-disubstituted 1-alkylpiperidines: Synthesized and tested against trace-amine-associated receptor 1 (TAAR1). Mendeleev Commun. 2021, 31, 488–489. [Google Scholar] [CrossRef]
- Bannykh, A.; Levashova, E.; Bakulina, O.; Krasavin, M. New reagent space and new scope for the Castagnoli–Cushman reaction of oximes and 3-arylglutaconic anhydrides. Org. Biomol. Chem. 2022, 20, 8643–8648. [Google Scholar] [CrossRef] [PubMed]
- Burdzhiev, N.; Stanoeva, E.; Shivachev, B.; Nikolova, R. Synthesis and transformations of polysubstituted diastereomeric 5-oxomorpholin-2-carboxylic acids. Comptes Rendus Chim. 2014, 17, 420–430. [Google Scholar] [CrossRef]
- Beng, T.K.; Farah, A.O.; Shearer, V. Modular synthesis and transition metal-free alkynylation/alkenylation of Castagnoli–Cushman-derived N,O- and N,S-heterocyclic vinyl chlorides. RSC Adv. 2020, 10, 37153–37160. [Google Scholar] [CrossRef] [PubMed]
- Chizhova, M.; Bakulina, O.; Dar’In, D.; Krasavin, M. New Dicarboxylic Acid Anhydride for Ambient-Temperature Castagnoli-Cushman Reactions. Chemistryselect 2016, 1, 5487–5492. [Google Scholar] [CrossRef]
- Moreno, A.; Beng, T.K. Direct access to vicinally functionalized and N-trifluoroacetylated (bicyclic)ketopiperazines using a readily affordable N-heterocyclic anhydride. Org. Biomol. Chem. 2020, 18, 1577–1581. [Google Scholar] [CrossRef]
- Pashev, A.; Burdzhiev, N.; Stanoeva, E. One-step route to tricyclic fused 1,2,3,4-tetrahydroisoquinoline systems via the Castagnoli–Cushman protocol. Beilstein J. Org. Chem. 2020, 16, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Usmanova, L.; Dar’In, D.; Novikov, M.S.; Gureev, M.; Krasavin, M. Bicyclic Piperazine Mimetics of the Peptide β-Turn Assembled via the Castagnoli–Cushman Reaction. J. Org. Chem. 2018, 83, 5859–5868. [Google Scholar] [CrossRef]
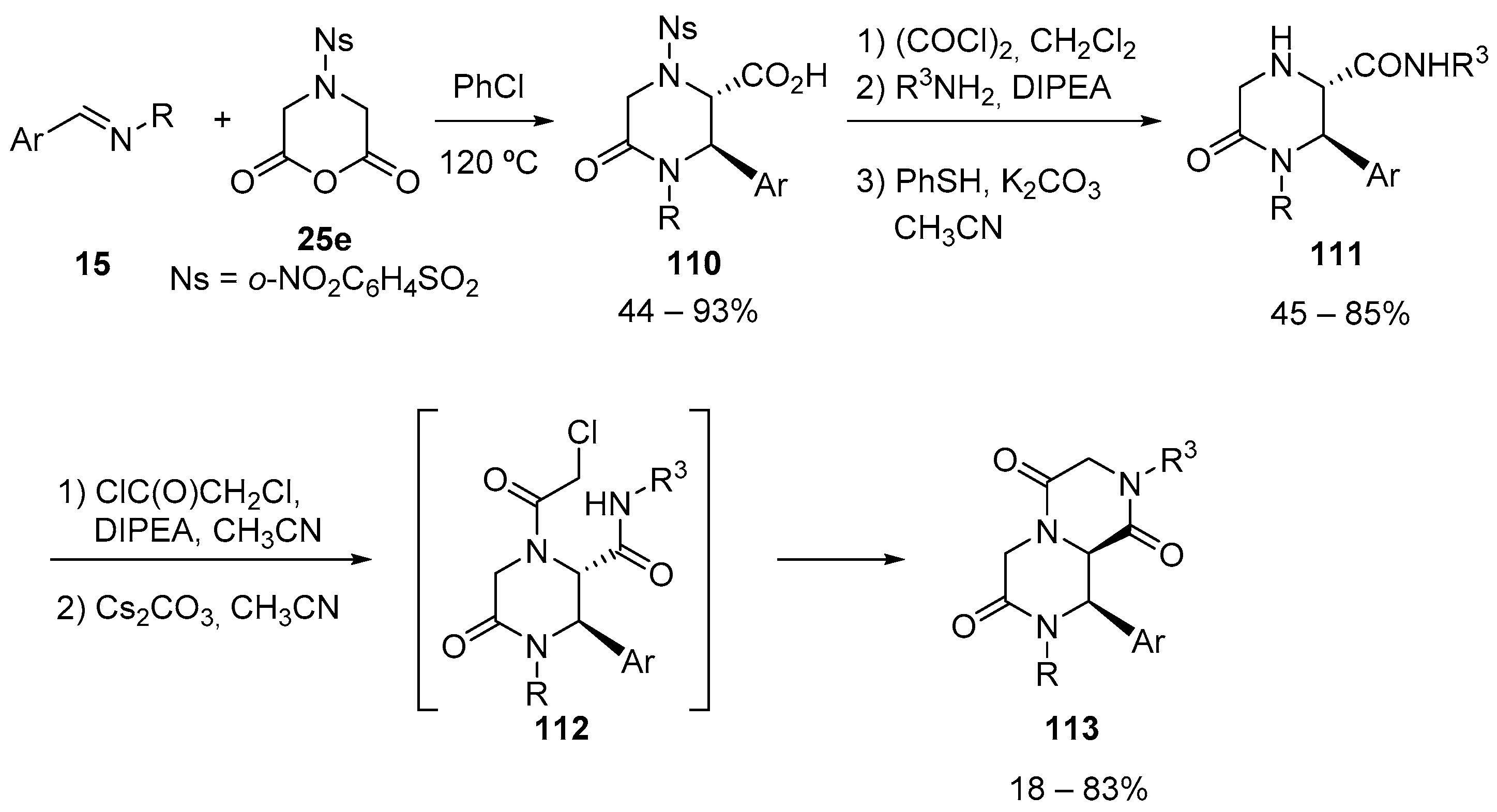
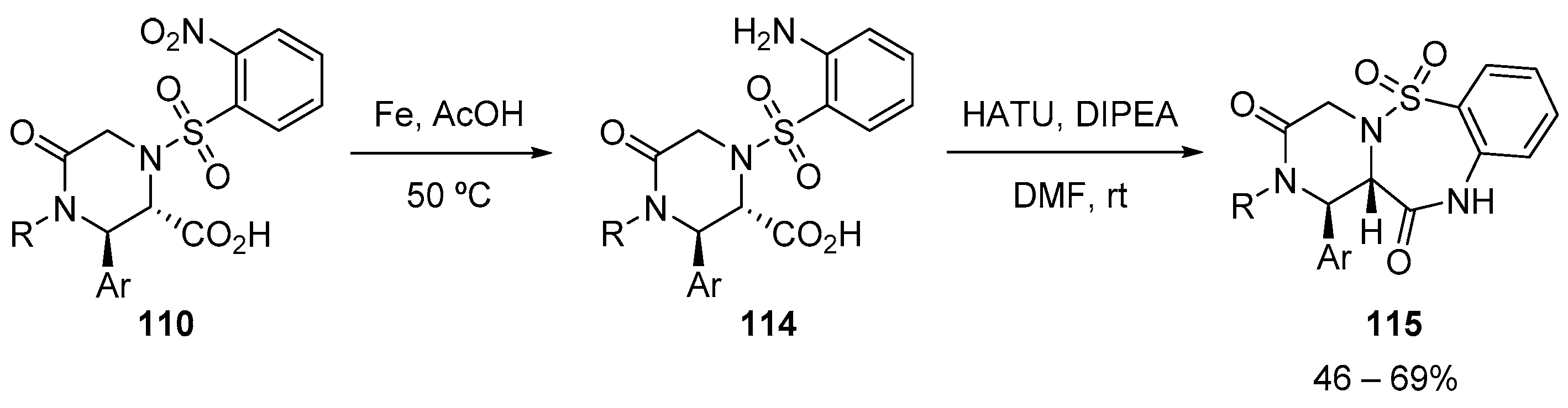
- Usmanova, L.; Dar’In, D.; Krasavin, M. A novel variant of the medicinally relevant 1,2,5-benzothiadiazepin-4-one-1,1-dioxide scaffold accessed via the downstream modification of Castagnoli-Cushman lactams. Tetrahedron Lett. 2019, 60, 151003–151005. [Google Scholar] [CrossRef]
- Usmanova, L.; Bakulina, O.; Dar’In, D.; Krasavin, M. Spontaneous formation of tricyclic lactones following the Castagnoli–Cushman reaction. Chem. Heterocycl. Compd. 2017, 53, 474–479. [Google Scholar] [CrossRef]
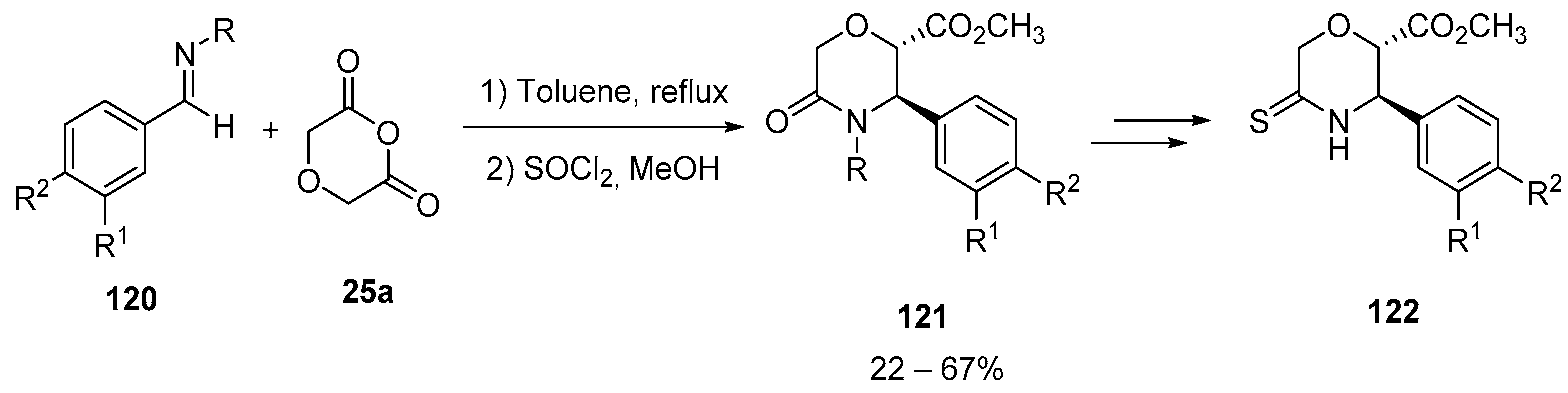
- Calugi, L.; Lenci, E.; Innocenti, R.; Trabocchi, A. Synthesis of morpholine derivatives using the Castagnoli-Cushman reaction as BACE1 inhibitors: Unexpected binding activity of cyclic thioamides. Bioorg. Med. Chem. Lett. 2020, 30, 127211. [Google Scholar] [CrossRef]
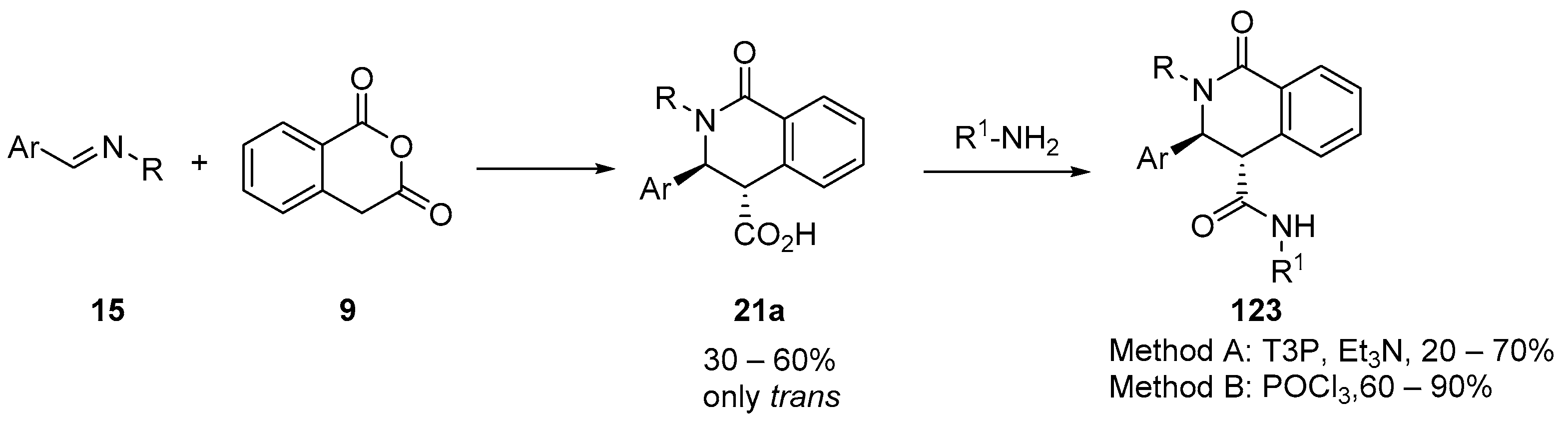
- Floyd, D.M.; Stein, P.; Wang, Z.; Liu, J.; Castro, S.; Clark, J.A.; Connelly, M.; Zhu, F.; Holbrook, G.; Matheny, A.; et al. Hit-to-Lead Studies for the Antimalarial Tetrahydroisoquinolone Carboxanilides. J. Med. Chem. 2016, 59, 7950–7962. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Díaz, M.B.; Ebert, D.; Salinas, Y.; Pradhan, A.; Lehane, A.M.; Myrand-Lapierre, M.-E.; O’Loughlin, K.G.; Shackleford, D.M.; de Almeida, M.J.; Carrillo, A.K.; et al. (+)-SJ733, a clinical candidate for malaria that acts through ATP4 to induce rapid host-mediated clearance of Plasmodium. Proc. Natl. Acad. Sci. USA 2014, 111, E5455–E5462. [Google Scholar] [CrossRef] [PubMed]
- Potowski, M.; Kunig, V.B.K.; Losch, F.; Brunschweiger, A. Synthesis of DNA-coupled isoquinolones and pyrrolidines by solid phase ytterbium- and silver-mediated imine chemistry. MedChemComm 2019, 10, 1082–1093. [Google Scholar] [CrossRef]
- Yuen, J.; Chai, J.; Ding, Y. Condensation of DNA-Conjugated Imines with Homophthalic Anhydride for the Synthesis of Isoquinolones on DNA. Bioconjug. Chem. 2020, 31, 2712–2718. [Google Scholar] [CrossRef]
- Cushman, M.; Castagnoli, N. Synthesis of pharmacologically active nitrogen analogs of the tetrahydrocannabinols. J. Org. Chem. 1974, 39, 1546–1550. [Google Scholar] [CrossRef] [PubMed]
- Cushman, M.; Gentry, J.; Dekow, F.W. Condensation of imines with homophthalic anhydrides. A convergent synthesis of cis- and trans-13-methyltetrahydroprotoberberines. J. Org. Chem. 1977, 42, 1111–1116. [Google Scholar] [CrossRef]
- Burdzhiev, N.T.; Stanoeva, E.R. Reaction between glutaric anhydride and N-benzylidenebenzylamine, and further transformations to new substituted piperidin-2-ones. Tetrahedron 2006, 62, 8318–8326. [Google Scholar] [CrossRef]
- Strumberg, D.; Pommier, Y.; Paull, K.; Jayaraman, M.; Nagafuji, P.; Cushman, M. Synthesis of Cytotoxic Indenoisoquinoline Topoisomerase I Poisons. J. Med. Chem. 1999, 42, 446–457. [Google Scholar] [CrossRef]
- Ahn, G.; Schifano-Faux, N.; Goossens, J.-F.; Baldeyrou, B.; Couture, A.; Grandclaudon, P.; Lansiaux, A.; Ryckebusch, A. Synthesis, cytotoxicity and topoisomerase inhibition properties of multifarious aminoalkylated indeno[1,2-c]isoquinolin-5,11-diones. Bioorg. Med. Chem. Lett. 2011, 21, 2259–2263. [Google Scholar] [CrossRef]
- Conda-Sheridan, M.; Park, E.-J.; Beck, D.E.; Reddy, P.V.N.; Nguyen, T.X.; Hu, B.; Chen, L.; White, J.J.; van Breemen, R.B.; Pezzuto, J.M.; et al. Design, Synthesis, and Biological Evaluation of Indenoisoquinoline Rexinoids with Chemopreventive Potential. J. Med. Chem. 2013, 56, 2581–2605. [Google Scholar] [CrossRef]
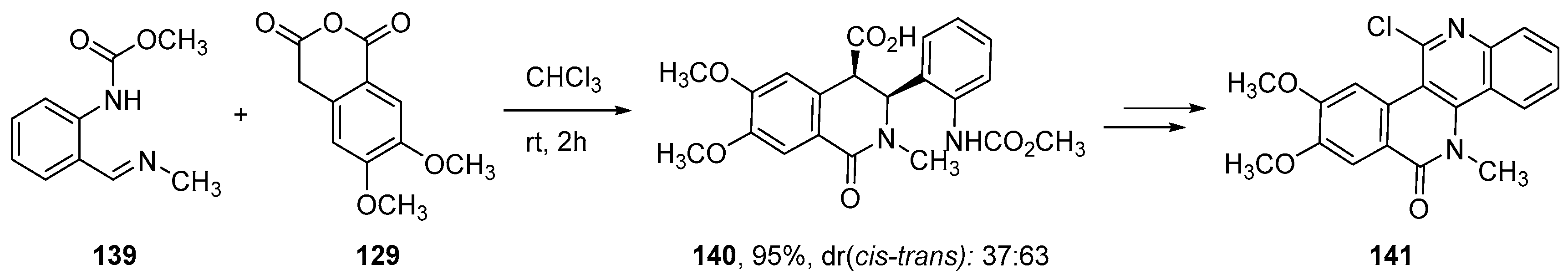
- Kiselev, E.; Dexheimer, T.S.; Pommier, Y.; Cushman, M. Design, Synthesis, and Evaluation of Dibenzo[c,h][1,6]naphthyridines as Topoisomerase I Inhibitors and Potential Anticancer Agents. J. Med. Chem. 2010, 53, 8716–8726. [Google Scholar] [CrossRef] [PubMed]
- Kiselev, E.; Empey, N.; Agama, K.; Pommier, Y.; Cushman, M. Dibenzo[c,h][1,5]naphthyridinediones as Topoisomerase I Inhibitors: Design, Synthesis, and Biological Evaluation. J. Org. Chem. 2012, 77, 5167–5172. [Google Scholar] [CrossRef]
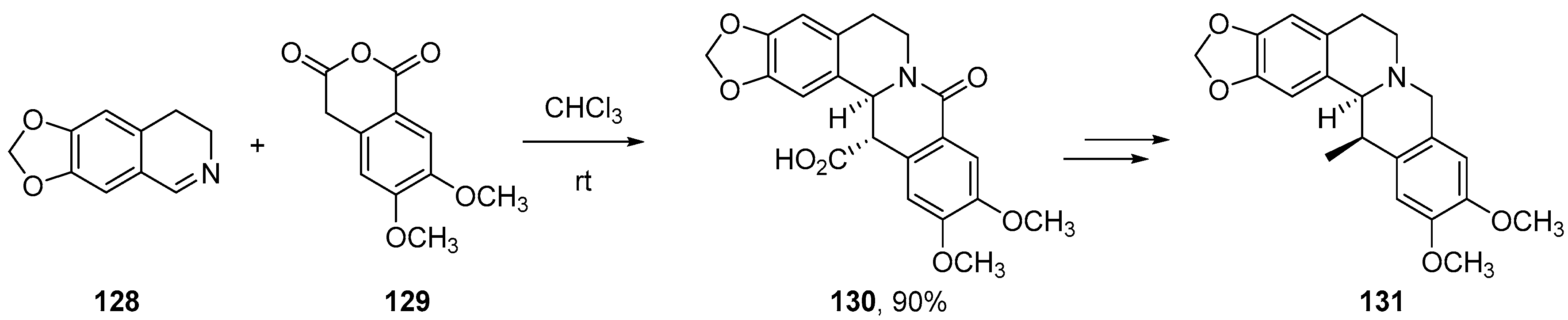
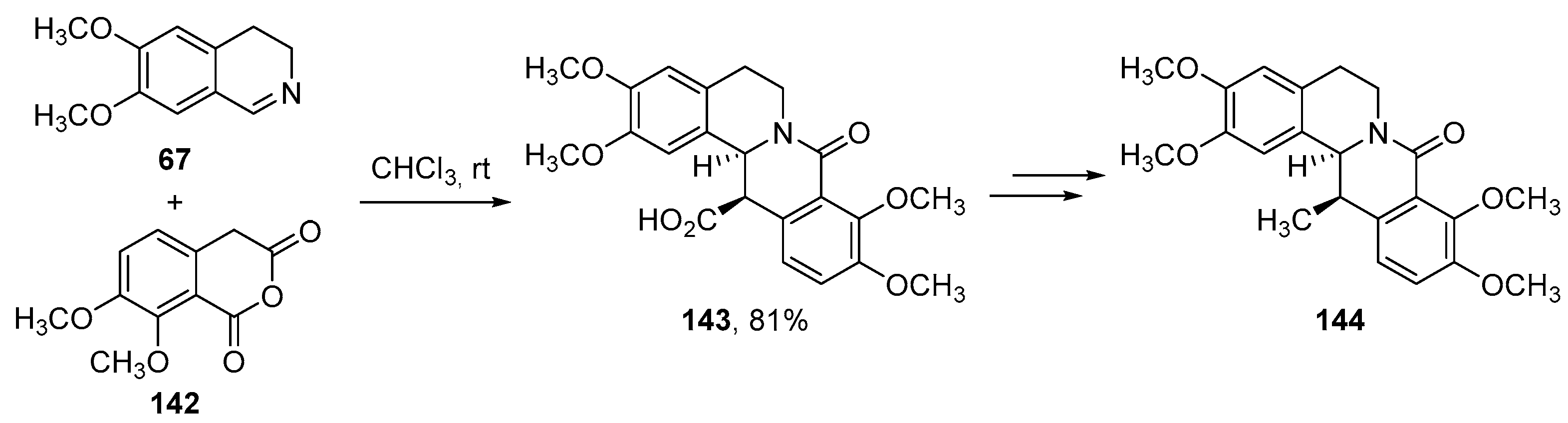
- Cushman, M.; Dekow, F.W. A total synthesis of corydaline. Tetrahedron 1978, 34, 1435–1439. [Google Scholar] [CrossRef]
- Karchuganova, E.; Bakulina, O.; Dar’In, D.; Krasavin, M. Two Annulated Azaheterocyclic Cores Readily Available from a Single Tetrahydroisoquinolonic Castagnoli–Cushman Precursor. Molecules 2020, 25, 2049. [Google Scholar] [CrossRef] [PubMed]
- Cushman, M.; Dikshit, D.K. Formation of the 5-benzo[d]naphtho[2,3-b]pyran system during an attempted benzophenanthridine synthesis. J. Org. Chem. 1980, 45, 5064–5067. [Google Scholar] [CrossRef]
- Sarnpitak, P.; Krasavin, M. Convenient access to novel functionalized pyrazino[1,2-b]isoquinolin-6-one and diazepino[1,2-b]isoquinolin-7-one scaffolds via the Cushman multicomponent reaction followed by post-condensation. Tetrahedron Lett. 2014, 55, 2299–2303. [Google Scholar] [CrossRef]
- Bakulina, O.; Ivanov, A.; Suslonov, V.; Dar’In, D.; Krasavin, M. A speedy route to sterically encumbered, benzene-fused derivatives of privileged, naturally occurring hexahydropyrrolo[1,2-b]isoquinoline. Beilstein J. Org. Chem. 2017, 13, 1413–1424. [Google Scholar] [CrossRef]
- Jarvis, C.L.; Jemal, N.M.; Knapp, S.; Seidel, D. Formal [4 + 2] cycloaddition of imines with alkoxyisocoumarins. Org. Biomol. Chem. 2018, 16, 4231–4235. [Google Scholar] [CrossRef]
- Kita, Y.; Mohri, S.; Tsugoshi, T.; Maeda, H.; Tamura, Y. Reaction of heteroaromatic analogs of homophthalic anhydride: Synthesis of hetero analogs of peri-hydroxy polycyclic aromatic compounds, isocoumarins, isoquinolinones, and related compounds. Chem. Pharm. Bull. 1985, 33, 4723–4731. [Google Scholar] [CrossRef]
- Moreau, E.; Dar’In, D.; Krasavin, M. The First Example of Azole-Fused Cyclic Anhydride Reacting in the Castagnoli–Cushman Way. Synlett 2018, 29, 890–893. [Google Scholar] [CrossRef]
- Chizhova, M.; Khoroshilova, O.; Dar’In, D.; Krasavin, M. Unusually Reactive Cyclic Anhydride Expands the Scope of the Castagnoli–Cushman Reaction. J. Org. Chem. 2018, 83, 12722–12733. [Google Scholar] [CrossRef]
- Chizhova, M.E.; Dar’In, D.V.; Krasavin, M. The Castagnoli–Cushman reaction of bicyclic pyrrole dicarboxylic anhydrides bearing electron-withdrawing substituents. Mendeleev Commun. 2020, 30, 496–497. [Google Scholar] [CrossRef]
- Adamovskyi, M.I.; Ryabukhin, S.V.; Sibgatulin, D.A.; Rusanov, E.; Grygorenko, O.O. Beyond the Five and Six: Evaluation of Seven-Membered Cyclic Anhydrides in the Castagnoli–Cushman Reaction. Org. Lett. 2016, 19, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Bakulina, O.; Dar’In, D.; Krasavin, M. o-Phenylenediacetic Acid Anhydride in the Castagnoli–Cushman Reaction: Extending the Product Space to ε-Lactams. Synlett 2017, 28, 1165–1169. [Google Scholar] [CrossRef]
- Beng, T.K.; Langevin, S.; Farah, A.O.; Goodsell, J.; Wyatt, K. One-shot access to isoquinolone and (hetero)izidinone architectures using cyclic α-chloro eneformamides and cyclic anhydrides. New J. Chem. 2019, 43, 5282–5286. [Google Scholar] [CrossRef]
- Beng, T.K.; Sax, M.; Borg, C. Serendipitous synthesis of 2-alkenyl- and 2-aryl-4-thiazolidinones using dithiodiglycolic anhydride. New J. Chem. 2022, 46, 18505–18511. [Google Scholar] [CrossRef]
- Chizhova, M.; Khoroshilova, O.; Dar’In, D.; Krasavin, M. Acetic anhydride to the rescue: Facile access to privileged 1,2,3,4-tetrahydropyrazino[1,2-a]indole core via the Castagnoli-Cushman reaction. Tetrahedron Lett. 2018, 59, 3612–3615. [Google Scholar] [CrossRef]
- Firsov, A.; Bakulina, O.; Dar’In, D.; Sokolov, V.V.; Krasavin, M. Synthesis of γ-Sultam-Annelated δ-Lactams via the Castagnoli–Cushman Reaction of Sultam-Based Dicarboxylic Acids. J. Org. Chem. 2022, 87, 1537–1540. [Google Scholar] [CrossRef]
- Moshnenko, N.; Kazantsev, A.; Bakulina, O.; Dar’In, D.; Krasavin, M. The Use of Aryl-Substituted Homophthalic Anhydrides in the Castagnoli–Cushman Reaction Provides Access to Novel Tetrahydroisoquinolone Carboxylic Acid Bearing an All-Carbon Quaternary Stereogenic Center. Molecules 2022, 27, 8462. [Google Scholar] [CrossRef]
- Rashevskii, A.; Bakulina, O.; Novikov, M.S.; Dar’In, D.; Krasavin, M. When periphery matters: Enhanced reactivity of 8-oxa-1,4-dithiaspiro[4.5]decane-7,9-dione and 9-oxa-1,5-dithiaspiro[5.5]undecane-8,10-dione in the Castagnoli-Cushman reaction with imines. Tetrahedron Lett. 2020, 61, 152658–152662. [Google Scholar] [CrossRef]
- Paramonova, P.; Bakulina, O.; Nabiyev, A.; Dar’In, D.; Krasavin, M. Castagnoli-Cushman Reaction of 3-Aryl Glutaric Acids: A Convenient, Diastereoselective Reaction for 6-Oxo-2,4-diarylpiperidine-3-carboxylic Acid Scaffold. Chemistryselect 2022, 7, e202104011. [Google Scholar] [CrossRef]
- Bakulina, O.; Chizhova, M.; Dar’In, D.; Krasavin, M. A General Way to Construct Arene-Fused Seven-Membered Nitrogen Heterocycles. Eur. J. Org. Chem. 2018, 2018, 362–371. [Google Scholar] [CrossRef]
- Chupakhin, E.G.; Bakulina, O.Y.; Dar’In, D.V.; Krasavin, M. 1,1′-Carbonyldiimidazole as a cyclodehydrating agent for the Castagnoli–Cushman reaction of dicarboxylic acids and imines. Mendeleev Commun. 2019, 29, 292–293. [Google Scholar] [CrossRef]
- Guranova, N.; Bakulina, O.; Dar’In, D.; Kantin, G.; Krasavin, M. Homophthalic Esters: A New Type of Reagents for the Castagnoli-Cushman Reaction. Eur. J. Org. Chem. 2022, 2022, e202101281. [Google Scholar] [CrossRef]
- Guranova, N.; Yakovleva, L.; Bakulina, O.; Dar’In, D.; Krasavin, M. Extending the Scope of the New Variant of the Castagnoli–Cushman Cyclocondensation onto o-Methyl Benzoic Acids Bearing Various Electron-Withdrawing Groups in the α-Position. Molecules 2022, 27, 7211. [Google Scholar] [CrossRef]
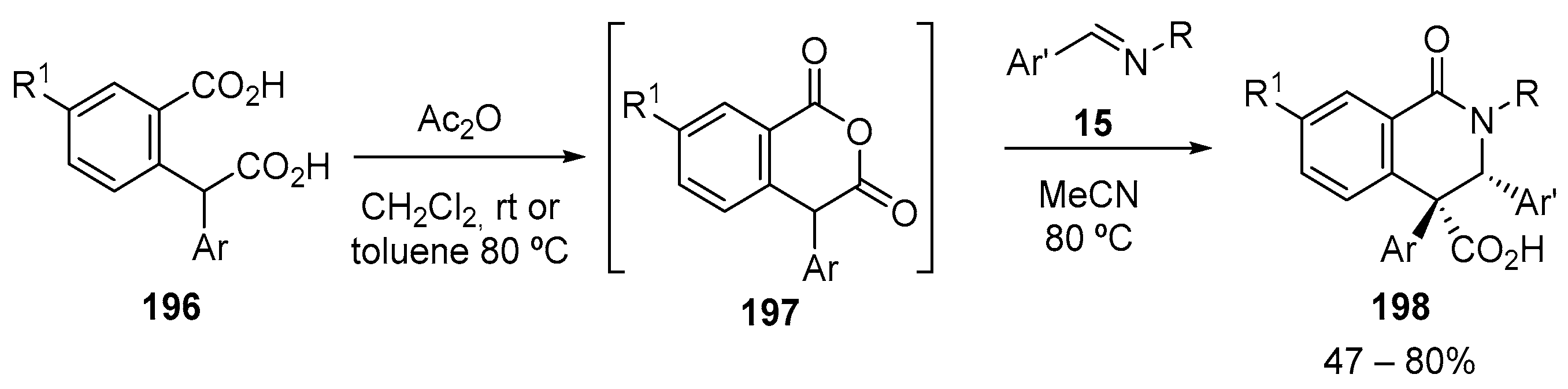
- Ananeva, A.; Bakulina, O.; Dar’In, D.; Kantin, G.; Krasavin, M. Dicarboxylic Acid Monoesters in β- and δ-Lactam Synthesis. Molecules 2022, 27, 2469. [Google Scholar] [CrossRef]
- Laws, S.W.; Moore, L.C.; Di Maso, M.; Nguyen, Q.N.N.; Tantillo, D.J.; Shaw, J.T. Diastereoselective Base-Catalyzed Formal [4 + 2] Cycloadditions of N-Sulfonyl Imines and Cyclic Anhydrides. Org. Lett. 2017, 19, 2466–2469. [Google Scholar] [CrossRef]
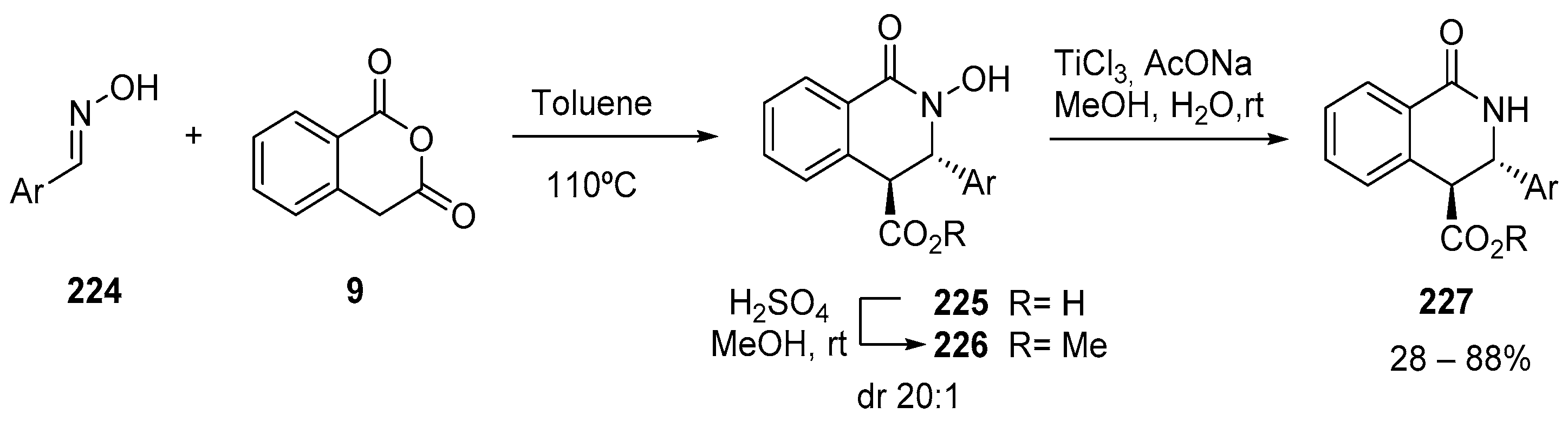
- Bannykh, A.V.; Bakulina, O.Y.; Dar’In, D.V.; Krasavin, M. Hydroxylamine as an ammonia equivalent: Access to NH-tetrahydroisoquinolonic derivatives from aldoximes by the Castagnoli–Cushman reaction followed by reduction. Mendeleev Commun. 2019, 29, 337–338. [Google Scholar] [CrossRef]
- Bakulina, O.; Bannykh, A.; Dar’In, D.; Krasavin, M. Cyclic Hydroxamic Acid Analogues of Bacterial Siderophores as Iron-Complexing Agents prepared through the Castagnoli-Cushman Reaction of Unprotected Oximes. Chem. A Eur. J. 2017, 23, 17667–17673. [Google Scholar] [CrossRef]
- Tutov, A.; Bakulina, O.; Dar’In, D.; Krasavin, M. Concise synthesis of 2-N-hydroxy-3,4-dihydroisoquinol-2-one: A bacterial siderophore and human 5-lipooxygenase inhibitor. Tetrahedron Lett. 2018, 59, 1511–1512. [Google Scholar] [CrossRef]
- Bakulina, O.; Bannykh, A.; Levashova, E.; Krasavin, M. Conjugates of Iron-Transporting N-Hydroxylactams with Ciprofloxacin. Molecules 2022, 27, 3910. [Google Scholar] [CrossRef] [PubMed]
- Adamovskyi, M.I.; Avramenko, M.M.; Volochnyuk, D.M.; Ryabukhin, S.V. Fluoral Hydrate: A Perspective Substrate for the Castagnoli–Cushman Reaction. ACS Omega 2020, 5, 20932–20942. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, A.; Stanoeva, E.; Spassov, S.; Haimova, M.; De Kimpe, N.; Boelens, M.; Keppens, M.; Kemme, A.; Mishnev, A. Cyclocondensations of homophthalic anhydrides with 1-aza-1,3-dienes. Tetrahedron 1995, 51, 6099–6114. [Google Scholar] [CrossRef]
- Georgieva, A.; Stanoeva, E.; Spassov, S.; Haimova, M.; De Kimpe, N.; Boelens, M.; Keppens, M.; Kemme, A.; Mishnev, A. Corrigenda to “cyclocondensations of homophthalic anhydrides with 1-aza-1,3-dienes”. Tetrahedron 1995, 51, 8407. [Google Scholar] [CrossRef]
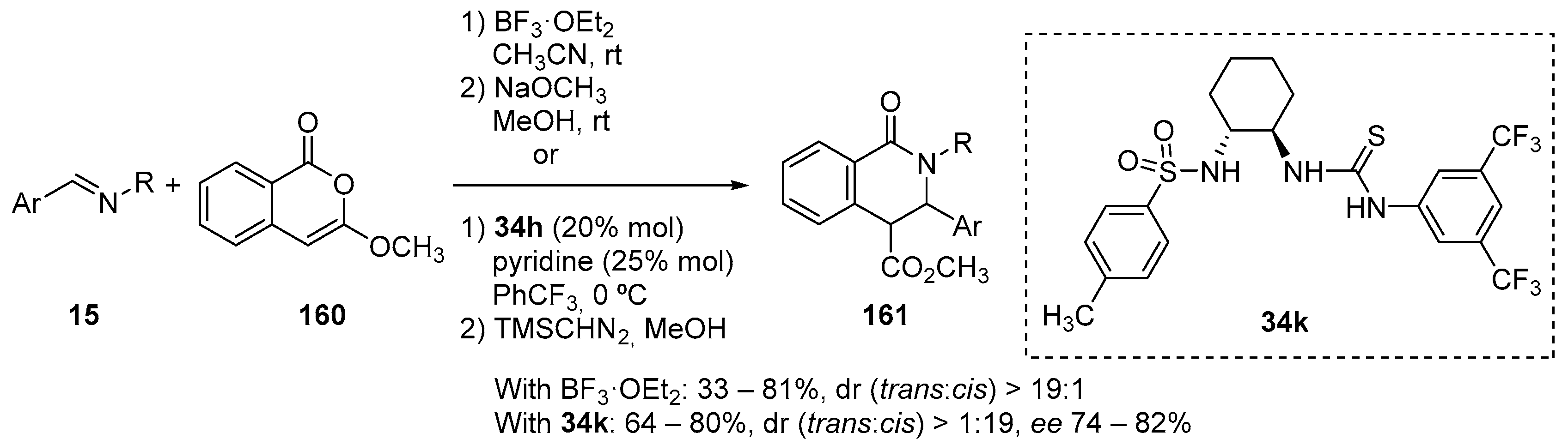
- Burlow, N.P.; Howard, S.Y.; Saunders, C.M.; Fettinger, J.C.; Tantillo, D.J.; Shaw, J.T. Formal [4 + 2] Cycloadditions of Anhydrides and α,β-Unsaturated N-Tosyl Ketimines. Org. Lett. 2019, 21, 1046–1049. [Google Scholar] [CrossRef]
- Collar, A.G.; Trujillo, C.; Connon, S.J. Highly Enantio- and Diastereoselective Catalytic Asymmetric Tamura Cycloaddition Reactions. Chem. A Eur. J. 2019, 25, 7270–7274. [Google Scholar] [CrossRef] [PubMed]
- Braunstein, H.; Langevin, S.; Khim, M.; Adamson, J.; Hovenkotter, K.; Kotlarz, L.; Mansker, B.; Beng, T.K. Modular access to vicinally functionalized allylic (thio)morpholinonates and piperidinonates by substrate-controlled annulation of 1,3-azadienes with hexacyclic anhydrides. Org. Biomol. Chem. 2016, 14, 8864–8872. [Google Scholar] [CrossRef]
- Beng, T.K.; Moreno, A. Copper-catalyzed alkenylation of novel N-iodoarylated allylic ketopiperazinonates with unactivated alkenes. New J. Chem. 2020, 44, 4257–4261. [Google Scholar] [CrossRef]
- Guranova, N.; Golubev, P.; Bakulina, O.; Dar’In, D.; Kantin, G.; Krasavin, M. Unexpected formal [4 + 2]-cycloaddition of chalcone imines and homophthalic anhydrides: Preparation of dihydropyridin-2(1H)-ones. Org. Biomol. Chem. 2021, 19, 3829–3833. [Google Scholar] [CrossRef]
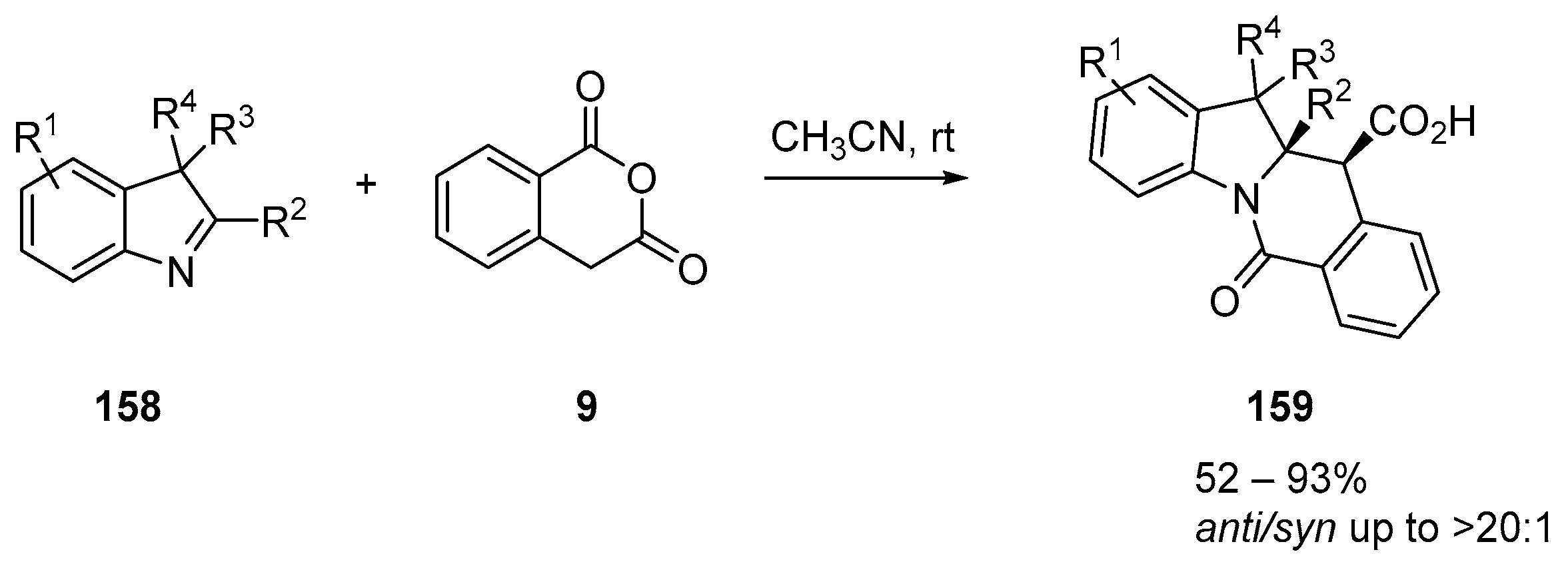
- Chizhova, M.; Dar’In, D.; Krasavin, M. Complications in the Castagnoli-Cushman reaction: An unusual course of reaction between cyclic anhydrides and sterically hindered indolenines. Tetrahedron Lett. 2017, 58, 3470–3473. [Google Scholar] [CrossRef]
- Guranova, N.; Dar’In, D.; Krasavin, M. Facile Access to 3-Unsubstituted Tetrahydroisoquinolonic Acids via the Castagnoli–Cushman Reaction. Synthesis 2018, 50, 2001–2008. [Google Scholar] [CrossRef]
- Venkatram, A.; Colley, T.; Deruiter, J.; Smith, F. Synthesis of 1,2,3,12a,12b-Hexahydrocyclopropa-[1,2-d]benzo[f]pyrrolo[1,2-b]isoquinolin-5,7-dione related to duocarmycins and anthramycin. J. Heterocycl. Chem. 2005, 42, 297–301. [Google Scholar] [CrossRef]
- Mikheyev, A.; Kantin, G.; Krasavin, M. Aldazines in the Castagnoli–Cushman Reaction. Synthesis 2018, 50, 2076–2086. [Google Scholar] [CrossRef]
- Smith, F.T.; Atigadda, R.V. Condensation of homophthalic anhydrides with heterocyclic imines and DMAD under mild conditions. J. Heterocycl. Chem. 1991, 28, 1813–1815. [Google Scholar] [CrossRef]
- Christov, P.P.; Palamareva, M.D. Synthesis and a Configurational Correlation within cis- and trans- Oxazolotetrahydroisoquinolinones with an Angular Substituent. Z. Für Nat. B 2007, 62, 1305–1312. [Google Scholar] [CrossRef]
- Christov, P.P.; Kozekov, I.D.; Palamareva, M.D. NewTRANS/CIStetrahydroisoquinolines. 3. [1,3]-oxazolo-[3,2-B]-tetrahydroisoquinolinones having an angular aryl substituent. J. Heterocycl. Chem. 2006, 43, 1015–1019. [Google Scholar] [CrossRef]
- Coppola, G.M. Novel heterocycles.8. Fused isoquinolines derived from the reaction of homophthalic anhydride with cyclic imino ethers. J. Heterocycl. Chem. 1981, 18, 767–770. [Google Scholar] [CrossRef]
- Stanoeva, E.; Haimova, M.; Ognyanov, V. Reactions of Glutaconic and Homophthalic Anhydrides with Imidates, Imidoyl Chlorides, and 1-Chloroisoquinoline. Liebigs Ann. Chem. 1984, 1984, 389–394. [Google Scholar] [CrossRef]
- Ognyanov, V.I.; Haimova, M.A.; Mollov, N.M. Synthesis of 5,7,11 b,12-tetrahydro-isoindolo[2,1-b]isoquinolinium methiodides and theirStevens rearrangement. Mon. Chem. 1982, 113, 993–998. [Google Scholar] [CrossRef]
- Haimova, M.A.; Ognyanov, V.I.; Mollov, N.M. One-Pot Synthesis of 5,6-Dihydro-8H-dibenzo[a,g]quinolizine-8-ones and Related Isoquinolines; A New Synthesis of Xylopinine. Synthesis 1980, 1980, 845–847. [Google Scholar] [CrossRef]
- Ryabukhin, S.V.; Panov, D.M.; Granat, D.S.; Ostapchuk, E.N.; Kryvoruchko, D.V.; Grygorenko, O.O. Toward Lead-Oriented Synthesis: One-Pot Version of Castagnoli Condensation with Nonactivated Alicyclic Anhydrides. ACS Comb. Sci. 2014, 16, 146–153. [Google Scholar] [CrossRef]
- Lepikhina, A.; Dar’In, D.; Bakulina, O.; Chupakhin, E.; Krasavin, M. Skeletal Diversity in Combinatorial Fashion: A New Format for the Castagnoli–Cushman Reaction. ACS Comb. Sci. 2017, 19, 702–707. [Google Scholar] [CrossRef]
- Azizian, J.; Mohammadi, A.A.; Karimi, A.R.; Mohammadizadeh, M.R. A Stereoselective Three-Component Reaction: KAl(SO4)2·12H2O, an Efficient and Reusable Catalyst for the One-Pot Synthesis of cis-Isoquinolonic Acids. J. Org. Chem. 2005, 70, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Chupakhin, E.; Dar’In, D.; Krasavin, M. The Castagnoli-Cushman reaction in a three-component format. Tetrahedron Lett. 2018, 59, 2595–2599. [Google Scholar] [CrossRef]
- Azizian, J.; Mohammadi, A.A.; Soleimani, E.; Karimi, A.R.; Mohammadizadeh, M.R. A stereoselective three-component reaction: One-pot synthesis ofcis-isoquinolonic acids catalyzed by silica sulfuric acid under mild and heterogeneous conditions. J. Heterocycl. Chem. 2006, 43, 187–190. [Google Scholar] [CrossRef]
- Mohammadi, M.H.; Mohammadi, A.A. One-Pot, Three-Component Synthesis of Cis-Isoquinolonic Acids Using ZnCl2, AlCl3-SiO2 as Catalyst. Synth. Commun. 2011, 41, 523–527. [Google Scholar] [CrossRef]
- Karimi, A.; Pashazadeh, R. Sulfonic Acid Functionalized Silica: A Mild, Reusable and Efficient Heterogeneous Catalyst for the Highly Diastereoselective Synthesis of cis-Isoquinolonic Acids. Synthesis 2009, 2010, 437–442. [Google Scholar] [CrossRef]
- Yadav, J.; Reddy, B.; Raj, K.S.; Prasad, A. Room temperature ionic liquids promoted three-component coupling reactions: A facile synthesis of cis-isoquinolonic acids. Tetrahedron 2003, 59, 1805–1809. [Google Scholar] [CrossRef]
- Wang, L.; Liu, J.; Tian, H.; Qian, C.; Sun, J. One-Pot Synthesis ofcis-Isoquinolonic Acid Derivativesvia Three-Component Reaction of Homophthalic Anhydride with Aldehydes and Amines using Ytterbium(III) Triflate as Catalyst. Adv. Synth. Catal. 2005, 347, 689–694. [Google Scholar] [CrossRef]
- Yadav, J.; Reddy, B.; Reddy, A.; Narsaiah, A. Iodine as a Mild, Efficient, and Cost-Effective Reagent for the Synthesis of cis-1-Oxo-1,2,3,4-tetrahydroisoquinoline-4-carboxylic Acids. Synthesis 2007, 2007, 3191–3194. [Google Scholar] [CrossRef]
- Yu, N.; Poulain, R.; Gesquiere, J.-C. A One-Pot Synthesis of Tetrahydroisoquinolonic Acids from Aldehydes and Amines in Trimethylorthoformate. Synlett 2000, 2000, 355–356. [Google Scholar] [CrossRef]
- Karimi, A.R.; Momeni, H.R.; Pashazadeh, R. l-Proline-catalyzed diastereoselective synthesis of cis-isoquinolonic acids and evaluation of their neuroprotective effects. Tetrahedron Lett. 2012, 53, 3440–3443. [Google Scholar] [CrossRef]
- Yu, N.; Bourel, L.; Deprez, B.; Gesquiere, J.-C. Lewis acid-induced reaction of homophthalic anhydride with imines: A convenient synthesis of trans-isoquinolonic acids. Tetrahedron Lett. 1998, 39, 829–832. [Google Scholar] [CrossRef]
- Ghorbani-Choghamarani, A.; Hajjami, M.; Norouzi, M.; Abbasityula, Y.; Eigner, V.; Dušek, M. Diastereoselective and one-pot synthesis of trans-isoquinolonic acids via three-component condensation of homophthalic anhydride, aldehydes, and ammonium acetate catalyzed by aspartic acid. Tetrahedron 2013, 69, 6541–6544. [Google Scholar] [CrossRef]
- Krasavin, M.; Safrygin, A.; Bakulina, O.; Dar’In, D. Three-Component Reaction of Homophthalic Anhydride with Carbonyl Compounds and Ammonium Acetate: New Developments. Synthesis 2020, 52, 2190–2195. [Google Scholar] [CrossRef]
- Safrygin, A.; Dar’In, D.; Bakulina, O.; Krasavin, M. Synthesis of spirocyclic tetrahydroisoquinolines (spiroTHIQs) via the Castagnoli-Cushman reaction. Tetrahedron Lett. 2020, 61, 152408–152410. [Google Scholar] [CrossRef]
- Safrygin, A.; Zhmurov, P.; Dar’In, D.; Silonov, S.; Kasatkina, M.; Zonis, Y.; Gureev, M.; Krasavin, M. Three-component Castagnoli-Cushman reaction with ammonium acetate delivers 2-unsubstituted isoquinol-1-ones as potent inhibitors of poly(ADP-ribose) polymerase (PARP). J. Enzym. Inhib. Med. Chem. 2021, 36, 1916–1921. [Google Scholar] [CrossRef]
- Chupakhin, E.; Bakulina, O.; Dar’In, D.; Krasavin, M. Facile Access to Fe(III)-Complexing Cyclic Hydroxamic Acids in a Three-Component Format. Molecules 2019, 24, 864. [Google Scholar] [CrossRef]
- Firsov, A.; Chupakhin, E.; Dar’In, D.; Bakulina, O.; Krasavin, M. Three-Component Castagnoli–Cushman Reaction of 3-Arylglutaconic Acids with Aromatic Aldehydes and Amines Delivers Rare 4,6-Diaryl-1,6-dihydropyridin-2(3H)-ones. Org. Lett. 2019, 21, 1637–1640. [Google Scholar] [CrossRef]
- Tan, D.Q.; Martin, K.S.; Fettinger, J.C.; Shaw, J.T. Ammonia synthons for the multicomponent assembly of complex γ-lactams. Proc. Natl. Acad. Sci. USA 2011, 108, 6781–6786. [Google Scholar] [CrossRef]
- Younai, A.; Chin, G.F.; Shaw, J.T. Diastereoselective Synthesis of (±)-Heliotropamide by a One-Pot, Four-Component Reaction. J. Org. Chem. 2010, 75, 8333–8336. [Google Scholar] [CrossRef]
- Biggs-Houck, J.E.; Davis, R.L.; Wei, J.; Mercado, B.Q.; Olmstead, M.M.; Tantillo, D.J.; Shaw, J.T. Carbon–Carbon Bond-Forming Reactions of α-Thioaryl Carbonyl Compounds for the Synthesis of Complex Heterocyclic Molecules. J. Org. Chem. 2012, 77, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Salehi, P.; Dabiri, M.; Zolfigol, M.A.; Baghbanzadeh, M. A Novel Method for the One-Pot Three-Component Synthesis of 2,3-Dihydroquinazolin-4(1H)-ones. Synlett 2005, 2005, 1155–1157. [Google Scholar] [CrossRef]
- Dabiri, M.; Salehi, P.; Otokesh, S.; Baghbanzadeh, M.; Kozehgary, G.; Mohammadi, A.A. Efficient synthesis of mono- and disubstituted 2,3-dihydroquinazolin-4(1H)-ones using KAl(SO4)2·12H2O as a reusable catalyst in water and ethanol. Tetrahedron Lett. 2005, 46, 6123–6126. [Google Scholar] [CrossRef]
- Azizi, S.; Soleymani, J.; Hasanzadeh, M. KCC-1/Pr-SO3H: An efficient heterogeneous catalyst for green and one-pot synthesis of 2,3-dihydroquinazolin-4(1H)-one. Nanocomposites 2020, 6, 31–40. [Google Scholar] [CrossRef]
- Dabiri, M.; Salehi, P.; Baghbanzadeh, M. Ionic Liquid Promoted Eco-friendly and Efficient Synthesis of 2,3-Dihydroquinazolin-4(1H)-ones. Mon. Chem. 2007, 138, 1191–1194. [Google Scholar] [CrossRef]
- Khandebharad, A.U.; Sarda, S.R.; Gill, C.H.; Agrawal, B.R. Synthesis of Quinazolinone Derivatives Catalyzed by Triethanolamine/NaCl in Aqueous Media. Polycycl. Aromat. Compd. 2020, 40, 437–445. [Google Scholar] [CrossRef]
- Dutta, A.; Trivedi, P.; Kulshrestha, A.; Kumar, A.; Chaturvedi, V.; Sarma, D. Sustainable parts-per-million level catalysis with FeIII: One-pot cascade synthesis of 2,3-dihydroquinazolin-4(1H)-ones in water. Appl. Organomet. Chem. 2021, 35, e6116. [Google Scholar] [CrossRef]
- Baghbanzadeh, M.; Dabiri, M.; Salehi, P. A New Efficient Method for the Three-Component Synthesis of 4(3H)-Quinazolinones. Heterocycles 2008, 75, 2809–2815. [Google Scholar] [CrossRef]
- Dabiri, M.; Salehi, P.; Bahramnejad, M.; Alizadeh, M. A practical and versatile approach toward a one-pot synthesis of 2,3-disubstituted 4(3H)-quinazolinones. Mon. Chem. 2010, 141, 877–881. [Google Scholar] [CrossRef]
- Bui, H.T.B.; Do, K.M.; Nguyen, H.T.D.; Van Mai, H.; Danh, T.L.D.; Tran, D.Q.; Morita, H. Efficient one-pot tandem synthesis and cytotoxicity evaluation of 2,3-disubstituted quinazolin-4(3H)-one derivatives. Tetrahedron 2021, 98, 132426–132432. [Google Scholar] [CrossRef]
- Rao, K.R.; Mekala, R.; Raghunadh, A.; Meruva, S.B.; Kumar, S.P.; Kalita, D.; Laxminarayana, E.; Prasad, B.; Pal, M. A catalyst-free rapid, practical and general synthesis of 2-substituted quinazolin-4(3H)-ones leading to luotonin B and E, bouchardatine and 8-norrutaecarpine. RSC Adv. 2015, 5, 61575–61579. [Google Scholar] [CrossRef]
- Abbas, S.Y.; El-Bayouki, K.; Basyouni, W.M. Utilization of isatoic anhydride in the syntheses of various types of quinazoline and quinazolinone derivatives. Synth. Commun. 2016, 46, 993–1035. [Google Scholar] [CrossRef]
- Kenarkoohi, T.; Rahmati, A. An efficient solvent-free synthesis of 2-(alkylamino)-2-oxo-1-arylethyl-6,12-dioxo-6,12-dihydroindolo[1,2-b]isoquinoline-11-carboxylate derivatives via four-component reaction. Mol. Divers. 2019, 23, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, R.; Dar’In, D.; Kantin, G.; Bakulina, O.; Krasavin, M. One-Pot Sequence of Staudinger/aza-Wittig/Castagnoli–Cushman Reactions Provides Facile Access to Novel Natural-like Polycyclic Ring Systems. Molecules 2022, 27, 8130. [Google Scholar] [CrossRef]






















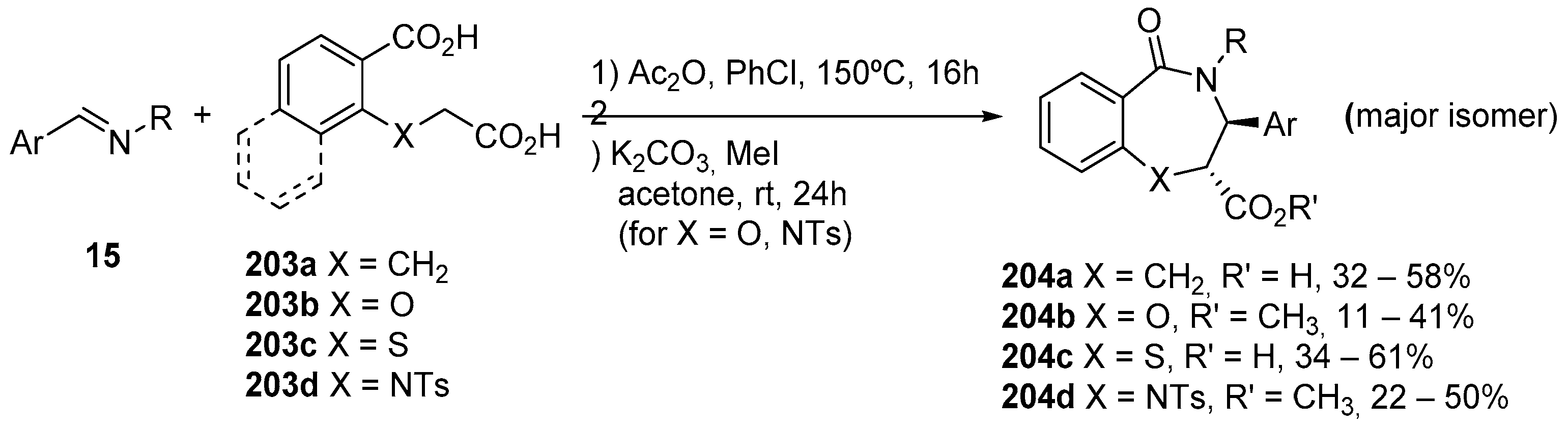







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramiro, J.L.; Martínez-Caballero, S.; Neo, A.G.; Díaz, J.; Marcos, C.F. The Castagnoli–Cushman Reaction. Molecules 2023, 28, 2654. https://doi.org/10.3390/molecules28062654
Ramiro JL, Martínez-Caballero S, Neo AG, Díaz J, Marcos CF. The Castagnoli–Cushman Reaction. Molecules. 2023; 28(6):2654. https://doi.org/10.3390/molecules28062654
Chicago/Turabian StyleRamiro, José Luis, Sonia Martínez-Caballero, Ana G. Neo, Jesús Díaz, and Carlos F. Marcos. 2023. "The Castagnoli–Cushman Reaction" Molecules 28, no. 6: 2654. https://doi.org/10.3390/molecules28062654
APA StyleRamiro, J. L., Martínez-Caballero, S., Neo, A. G., Díaz, J., & Marcos, C. F. (2023). The Castagnoli–Cushman Reaction. Molecules, 28(6), 2654. https://doi.org/10.3390/molecules28062654