Topology of the Electron Density and of Its Laplacian from Periodic LCAO Calculations on f-Electron Materials: The Case of Cesium Uranyl Chloride
Abstract
1. Introduction
2. Computational Details
3. The Implementation
3.1. Basis Functions
3.2. The Electron Density and Its Derivatives
3.3. Old Strategy Based on the Expansion in Hermite Gaussian Type Functions
3.4. New Strategy Based on a Direct Evaluation of the Electron Density in the RSSH-GTF Basis
3.5. The Electron Localization Function
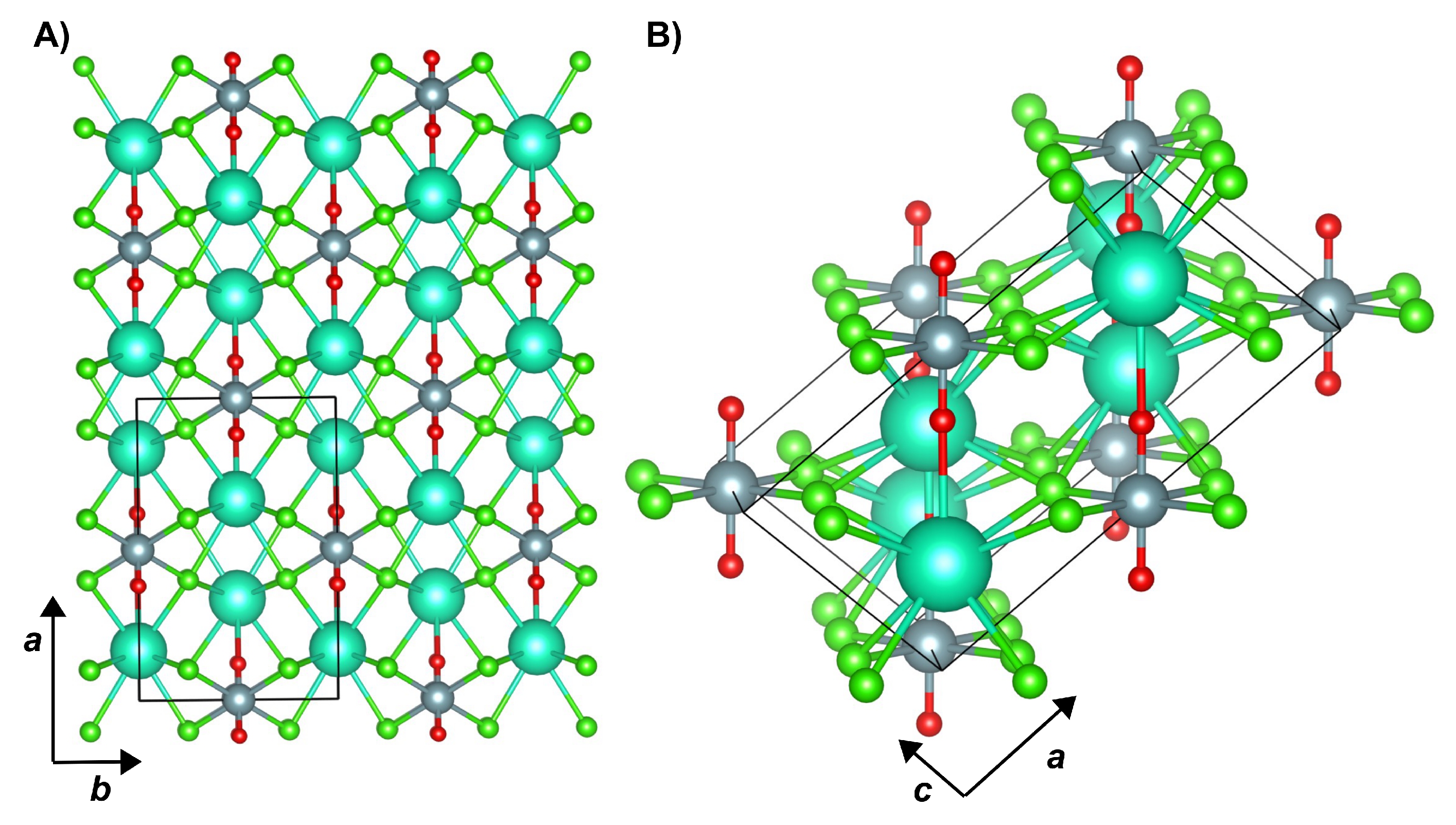
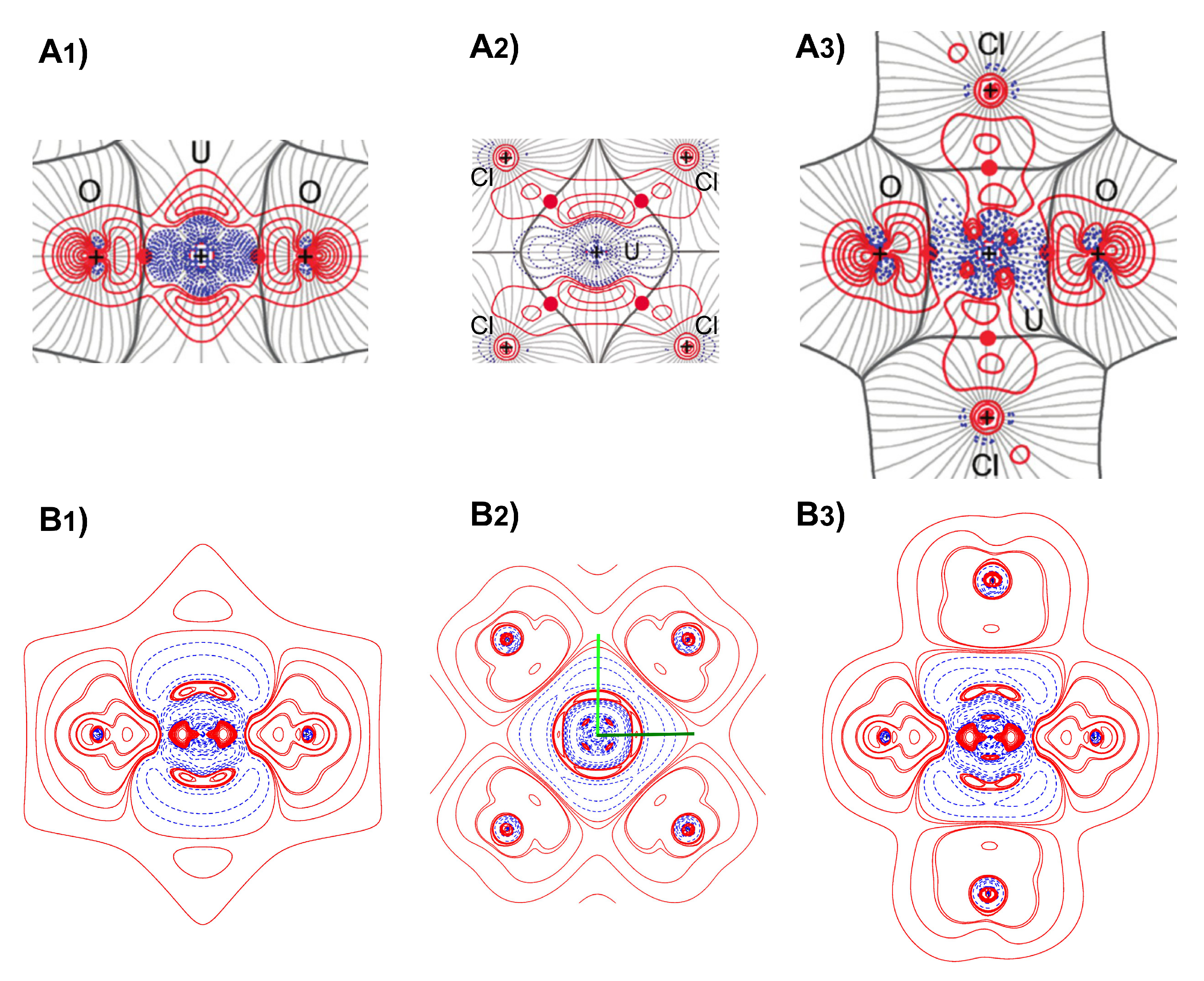
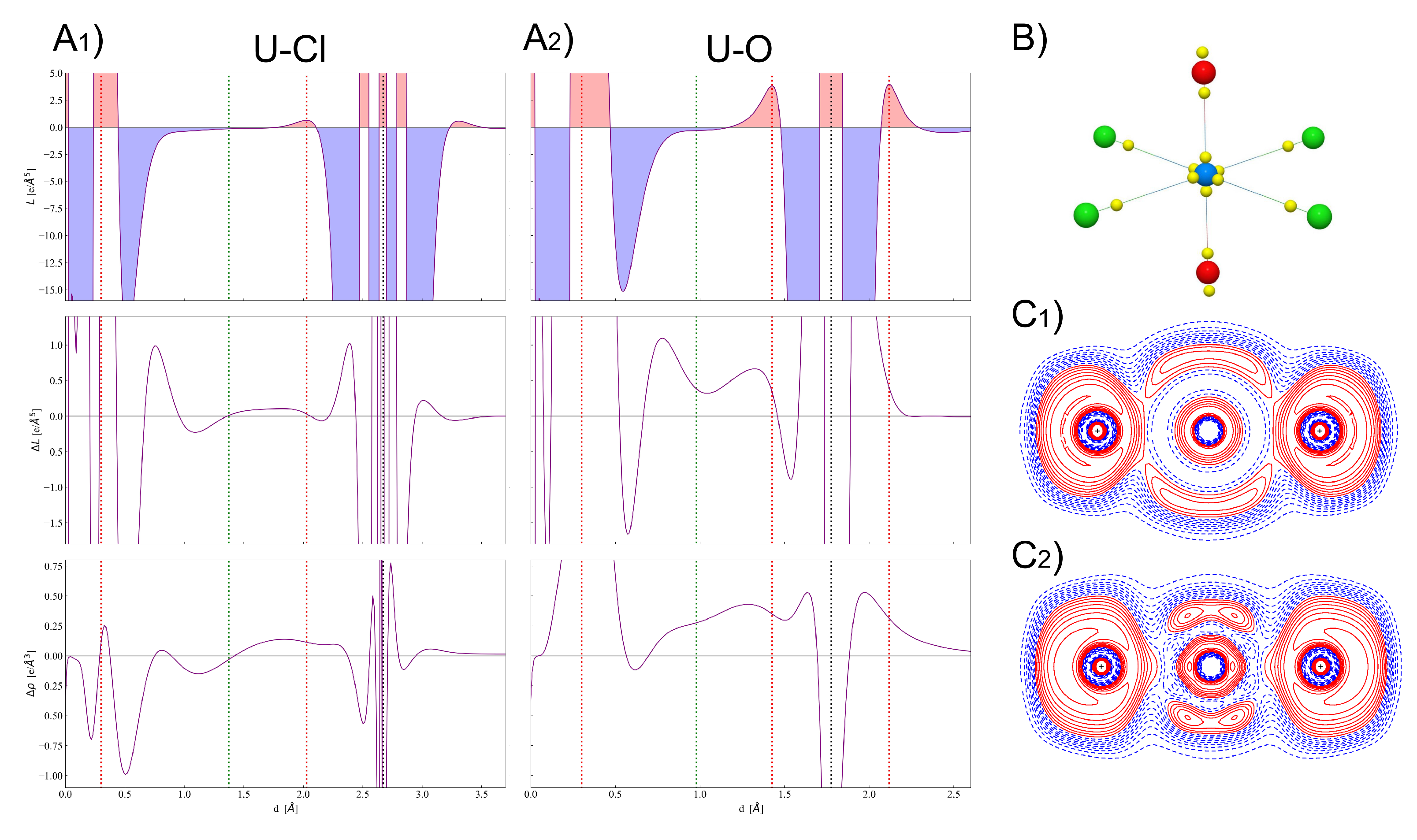
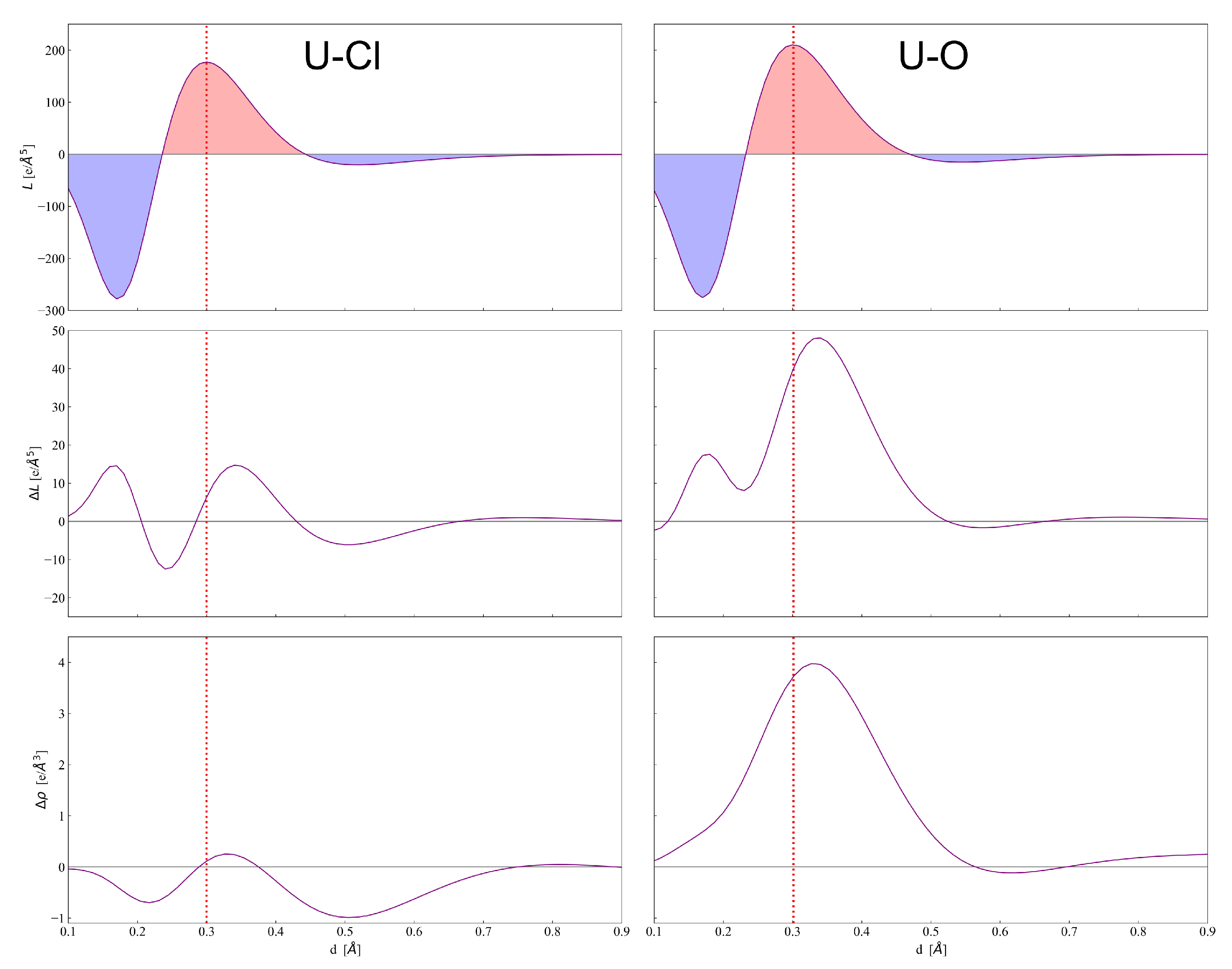
4. Results and Discussion
5. Conclusions and Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Söderlind, P.; Kotliar, G.; Haule, K.; Oppeneer, P.M.; Guillaumont, D. Computational modeling of actinide materials and complexes. MRS Bull. 2010, 35, 883–888. [Google Scholar] [CrossRef]
- Vitova, T.; Pidchenko, I.; Fellhauer, D.; Bagus, P.S.; Joly, Y.; Pruessmann, T.; Bahl, S.; Gonzalez-Robles, E.; Rothe, J.; Altmaier, M.; et al. The role of the 5 f valence orbitals of early actinides in chemical bonding. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef]
- Neidig, M.L.; Clark, D.L.; Martin, R.L. Covalency in f-element complexes. Coord. Chem. Rev. 2013, 257, 394–406. [Google Scholar] [CrossRef]
- Kerridge, A. f-Orbital covalency in the actinocenes (An= Th–Cm): Multiconfigurational studies and topological analysis. RSC Adv. 2014, 4, 12078–12086. [Google Scholar] [CrossRef]
- Kerridge, A. Quantification of f-element covalency through analysis of the electron density: Insights from simulation. Chem. Commun. 2017, 53, 6685–6695. [Google Scholar] [CrossRef] [PubMed]
- Denning, R.; Green, J.; Hutchings, T.; Dallera, C.; Tagliaferri, A.; Giarda, K.; Brookes, N.; Braicovich, L. Covalency in the uranyl ion: A polarized x-ray spectroscopic study. J. Chem. Phys. 2002, 117, 8008–8020. [Google Scholar] [CrossRef]
- Desmarais, J.K.; Erba, A.; Pan, Y.; Civalleri, B.; John, S.T. Mechanisms for Pressure-Induced Isostructural Phase Transitions in EuO. Phys. Rev. Lett. 2021, 126, 196404. [Google Scholar] [CrossRef] [PubMed]
- Ewing, R.C. Long-term storage of spent nuclear fuel. Nat. Mater. 2015, 14, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Kaltsoyannis, N. Does covalency increase or decrease across the actinide series? Implications for minor actinide partitioning. Inorg. Chem. 2013, 52, 3407–3413. [Google Scholar] [CrossRef]
- Kaltsoyannis, N.; Hay, P.J.; Li, J.; Blaudeau, J.P.; Bursten, B.E. Theoretical studies of the electronic structure of compounds of the actinide elements. In The Chemistry of the Actinide and Transactinide Elements; Springer: Berlin, Germany, 2008; pp. 1893–2012. [Google Scholar]
- Denning, R.G. Electronic structure and bonding in actinyl ions and their analogs. J. Phys. Chem. A 2007, 111, 4125–4143. [Google Scholar] [CrossRef]
- O’Grady, E.; Kaltsoyannis, N. On the inverse trans influence. Density functional studies of [MOX 5] n-(M= Pa, n= 2; M= U, n= 1; M= Np, n= 0; X= F, Cl or Br). J. Chem. Soc. Dalton Trans. 2002, 1233–1239. [Google Scholar] [CrossRef]
- Bader, R.; Nguyen-Dang, T. Quantum theory of atoms in molecules—Dalton revisited. In Advances in Quantum Chemistry; Elsevier: Amsterdam, The Netherlands, 1981; Volume 14, pp. 63–124. [Google Scholar]
- Gatti, C. Chemical bonding in crystals: New directions. Z. Krist. Cryst. Mater. 2005, 220, 399–457. [Google Scholar] [CrossRef]
- Popelier, P. On the full topology of the Laplacian of the electron density. Coord. Chem. Rev. 2000, 197, 169–189. [Google Scholar] [CrossRef]
- Iversen, B.B.; Larsen, F.K.; Pinkerton, A.A.; Martin, A.; Darovsky, A.; Reynolds, P.A. Characterization of actinide bonding in Th (S2PMe2) 4 by synchrotron x-ray diffraction. Inorg. Chem. 1998, 37, 4559–4566. [Google Scholar] [CrossRef]
- Iversen, B.; Larsen, F.; Pinkerton, A.; Martin, A.; Darovsky, A.; Reynolds, P. Accurate charge densities in days-use of synchrotrons, image plates and very low temperatures. Acta Crystallogr. Sect. B Struct. Sci. 1999, 55, 363–374. [Google Scholar] [CrossRef]
- Zhurov, V.V.; Zhurova, E.A.; Pinkerton, A.A. Chemical Bonding in Cesium Uranyl Chloride Based on the Experimental Electron Density Distribution. Inorg. Chem. 2011, 50, 6330–6333. [Google Scholar] [CrossRef] [PubMed]
- Zhurov, V.V.; Zhurova, E.A.; Stash, A.I.; Pinkerton, A.A. Characterization of bonding in cesium uranyl chloride: Topological analysis of the experimental charge density. J. Phys. Chem. A 2011, 115, 13016–13023. [Google Scholar] [CrossRef]
- Gianopoulos, C.G.; Zhurov, V.V.; Minasian, S.G.; Batista, E.R.; Jelsch, C.; Pinkerton, A.A. Bonding in uranium (V) hexafluoride based on the experimental electron density distribution measured at 20 K. Inorg. Chem. 2017, 56, 1775–1778. [Google Scholar] [CrossRef] [PubMed]
- Gianopoulos, C.G.; Zhurov, V.V.; Pinkerton, A.A. Charge densities in actinide compounds: Strategies for data reduction and model building. IUCrJ 2019, 6, 895–908. [Google Scholar] [CrossRef]
- Tassell, M.J.; Kaltsoyannis, N. Covalency in AnCp4 (An= Th–Cm): A comparison of molecular orbital, natural population and atoms-in-molecules analyses. Dalton Trans. 2010, 39, 6719–6725. [Google Scholar] [CrossRef]
- Huang, Q.R.; Kingham, J.R.; Kaltsoyannis, N. The strength of actinide—Element bonds from the quantum theory of atoms-in-molecules. Dalton Trans. 2015, 44, 2554–2566. [Google Scholar] [CrossRef]
- Tanti, J.; Lincoln, M.; Kerridge, A. Decomposition of d-and f-shell contributions to uranium bonding from the quantum theory of atoms in molecules: Application to uranium and uranyl halides. Inorganics 2018, 6, 88. [Google Scholar] [CrossRef]
- Ebenezer, C.; Solomon, R.V. Insights into the Extraction of Actinides from Lanthanides Using 3, 3’-Dimethoxy-phenyl-bis-1, 2, 4-triazinyl-2, 6-pyridine Ligand–A DFT Study. Chem. Select. 2020, 5, 13895–13901. [Google Scholar]
- Chen, Y.M.; Wang, C.Z.; Wu, Q.Y.; Lan, J.H.; Chai, Z.F.; Nie, C.M.; Shi, W.Q. Complexation of trivalent lanthanides and actinides with diethylenetriaminepentaacetic acid: Theoretical unraveling of bond covalency. J. Mol. Liq. 2020, 299, 112174. [Google Scholar] [CrossRef]
- Du, J.; Jiang, G. Adsorption of actinide ion complexes on C60O: An all-electron ZORA-DFT-D3 study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 223, 117375. [Google Scholar] [CrossRef] [PubMed]
- Sadhu, B.; Dolg, M.; Kulkarni, M.S. Periodic trends and complexation chemistry of tetravalent actinide ions with a potential actinide decorporation agent 5-LIO (Me-3, 2-HOPO): A relativistic density functional theory exploration. J. Comput. Chem. 2020, 41, 1427–1435. [Google Scholar] [CrossRef]
- Niu, S.; Cai, H.X.; Zhao, H.B.; Li, L.; Pan, Q.J. Redox and structural properties of accessible actinide (ii) metallocalixarenes (Ac to Pu): A relativistic DFT study. RSC Adv. 2020, 10, 26880–26887. [Google Scholar] [CrossRef]
- Dovesi, R.; Erba, A.; Orlando, R.; Zicovich-Wilson, C.M.; Civalleri, B.; Maschio, L.; Rérat, M.; Casassa, S.; Baima, J.; Salustro, S.; et al. Quantum-Mechanical Condensed Matter Simulations with CRYSTAL. WIREs Comput. Mol. Sci. 2018, 8, e1360. [Google Scholar] [CrossRef]
- Erba, A.; Baima, J.; Bush, I.; Orlando, R.; Dovesi, R. Large Scale Condensed Matter DFT Simulations: Performance and Capabilities of the Crystal Code. J. Chem. Theory Comput. 2017, 13, 5019–5027. [Google Scholar] [CrossRef]
- Desmarais, J.; Erba, A.; Dovesi, R. Generalization of the Periodic LCAO Approach in the CRYSTAL Code to g-type Orbitals. Theor. Chem. Acc. 2018, 137, 28. [Google Scholar] [CrossRef]
- El-Kelany, K.E.; Ravoux, C.; Desmarais, J.K.; Cortona, P.; Pan, Y.; Tse, J.; Erba, A. Spin Localization, Magnetic Ordering and Electronic Properties of Strongly Correlated Ln2O3 Sesquioxides (Ln=La, Ce, Pr, Nd). Phys. Rev. B 2018, 97, 245118. [Google Scholar] [CrossRef]
- Gatti, C.; Casassa, S.M. TOPOND14 User’s Manual. 2013. Available online: http://crystal.unito.it/topond.php (accessed on 29 May 2021).
- Gatti, C.; Saunders, V.; Roetti, C. Crystal field effects on the topological properties of the electron density in molecular crystals: The case of urea. J. Chem. Phys. 1994, 101, 10686–10696. [Google Scholar] [CrossRef]
- Casassa, S.; Erba, A.; Baima, J.; Orlando, R. Electron Density Analysis of Large (Molecular and Periodic) Systems: A Parallel Implementation. J. Comput. Chem. 2015, 36, 1940–1946. [Google Scholar] [CrossRef]
- Cossard, A.; Desmarais, J.K.; Casassa, S.; Gatti, C.; Erba, A. Charge Density Analysis of Actinide Compounds from the Quantum Theory of Atoms in Molecules and Crystals. J. Phys. Chem. Lett. 2021, 12, 1862–1868. [Google Scholar] [CrossRef]
- Vallet, V.; Wahlgren, U.; Grenthe, I. Probing the nature of chemical bonding in uranyl (VI) complexes with quantum chemical methods. J. Phys. Chem. A 2012, 116, 12373–12380. [Google Scholar] [CrossRef] [PubMed]
- Wellington, J.P.; Kerridge, A.; Kaltsoyannis, N. Should environmental effects be included when performing QTAIM calculations on actinide systems? A comparison of QTAIM metrics for Cs2UO2Cl4, U(Se2PPh2)4 and Np(Se2PPh2)4 in gas phase, COSMO and PEECM. Polyhedron 2016, 116, 57–63. [Google Scholar] [CrossRef][Green Version]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648. [Google Scholar] [CrossRef]
- Ponec, R.; Bucinsky, L.; Gatti, C. Relativistic Effects on Metal- Metal Bonding: Comparison of the Performance of ECP and Scalar DKH Description on the Picture of Metal- Metal Bonding in Re2Cl82-. J. Chem. Theory Comput. 2010, 6, 3113–3121. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Dolg, M.; Stoll, H. Valence basis sets for relativistic energy-consistent small-core actinide pseudopotentials. J. Chem. Phys. 2003, 118, 487–496. [Google Scholar] [CrossRef]
- Dolg, M.; Cao, X. Accurate relativistic small-core pseudopotentials for actinides. Energy adjustment for uranium and first applications to uranium hydride. J. Phys. Chem. A 2009, 113, 12573–12581. [Google Scholar] [CrossRef] [PubMed]
- Desmarais, J.K.; Flament, J.P.; Erba, A. Spin-orbit coupling from a two-component self-consistent approach. I. Generalized Hartree-Fock theory. J. Chem. Phys. 2019, 151, 074107. [Google Scholar] [CrossRef]
- Desmarais, J.K.; Flament, J.P.; Erba, A. Fundamental Role of Fock Exchange in Relativistic Density Functional Theory. J. Phys. Chem. Lett. 2019, 10, 3580–3585. [Google Scholar] [CrossRef]
- Desmarais, J.K.; Flament, J.P.; Erba, A. Spin-orbit coupling in periodic systems with broken time-reversal symmetry: Formal and computational aspects. Phys. Rev. B 2020, 101, 235142. [Google Scholar] [CrossRef]
- Desmarais, J.K.; Flament, J.P.; Erba, A. Adiabatic Connection in Spin-Current Density Functional Theory. Phys. Rev. B 2020, 102, 235118. [Google Scholar] [CrossRef]
- Eickerling, G.; Mastalerz, R.; Herz, V.; Scherer, W.; Himmel, H.J.; Reiher, M. Relativistic effects on the topology of the electron density. J. Chem. Theory Comput. 2007, 3, 2182–2197. [Google Scholar] [CrossRef] [PubMed]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef]
- Wadt, W.R.; Hay, P.J. Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi. J. Chem. Phys. 1985, 82, 284–298. [Google Scholar] [CrossRef]
- Saunders, V. Molecular integrals for Gaussian type functions. In Methods in Computational Molecular Physics; Springer Netherlands: Dordrecht, The Netherlands, 1983; pp. 1–36. [Google Scholar]
- Pisani, C.; Dovesi, R.; Roetti, C. Hartree-Fock ab Initio Treatment of Crystalline Systems; Springer & Business Media: Berlin, Germany, 2012; Volume 48. [Google Scholar]
- Becke, A.D.; Edgecombe, K.E. A simple measure of electron localization in atomic and molecular systems. J. Chem. Phys. 1990, 92, 5397–5403. [Google Scholar] [CrossRef]
- Savin, A.; Jepsen, O.; Flad, J.; Andersen, O.K.; Preuss, H.; von Schnering, H.G. Electron localization in solid-state structures of the elements: The diamond structure. Angew. Chem. Int. Ed. Engl. 1992, 31, 187–188. [Google Scholar] [CrossRef]
- Espinosa, E.; Alkorta, I.; Elguero, J.; Molins, E. From weak to strong interactions: A comprehensive analysis of the topological and energetic properties of the electron density distribution involving X–H? F–Y systems. J. Chem. Phys. 2002, 117, 5529–5542. [Google Scholar] [CrossRef]
- Clark, A.E.; Sonnenberg, J.L.; Hay, P.J.; Martin, R.L. Density and wave function analysis of actinide complexes: What can fuzzy atom, atoms-in-molecules, Mulliken, Löwdin, and natural population analysis tell us? J. Chem. Phys. 2004, 121, 2563–2570. [Google Scholar] [CrossRef] [PubMed]
- Pyykko, P.; Li, J.; Runeberg, N. Quasirelativistic pseudopotential study of species isoelectronic to uranyl and the equatorial coordination of uranyl. J. Phys. Chem. 1994, 98, 4809–4813. [Google Scholar] [CrossRef]
- Savin, A.; Nesper, R.; Wengert, S.; Fässler, T.F. ELF: The electron localization function. Angew. Chem. Int. Ed. Engl. 1997, 36, 1808–1832. [Google Scholar] [CrossRef]
- Fuentealba, P.; Chamorro, E.; Santos, J.C. Understanding and using the electron localization function. Theor. Comput. Chem. 2007, 19, 57–85. [Google Scholar]
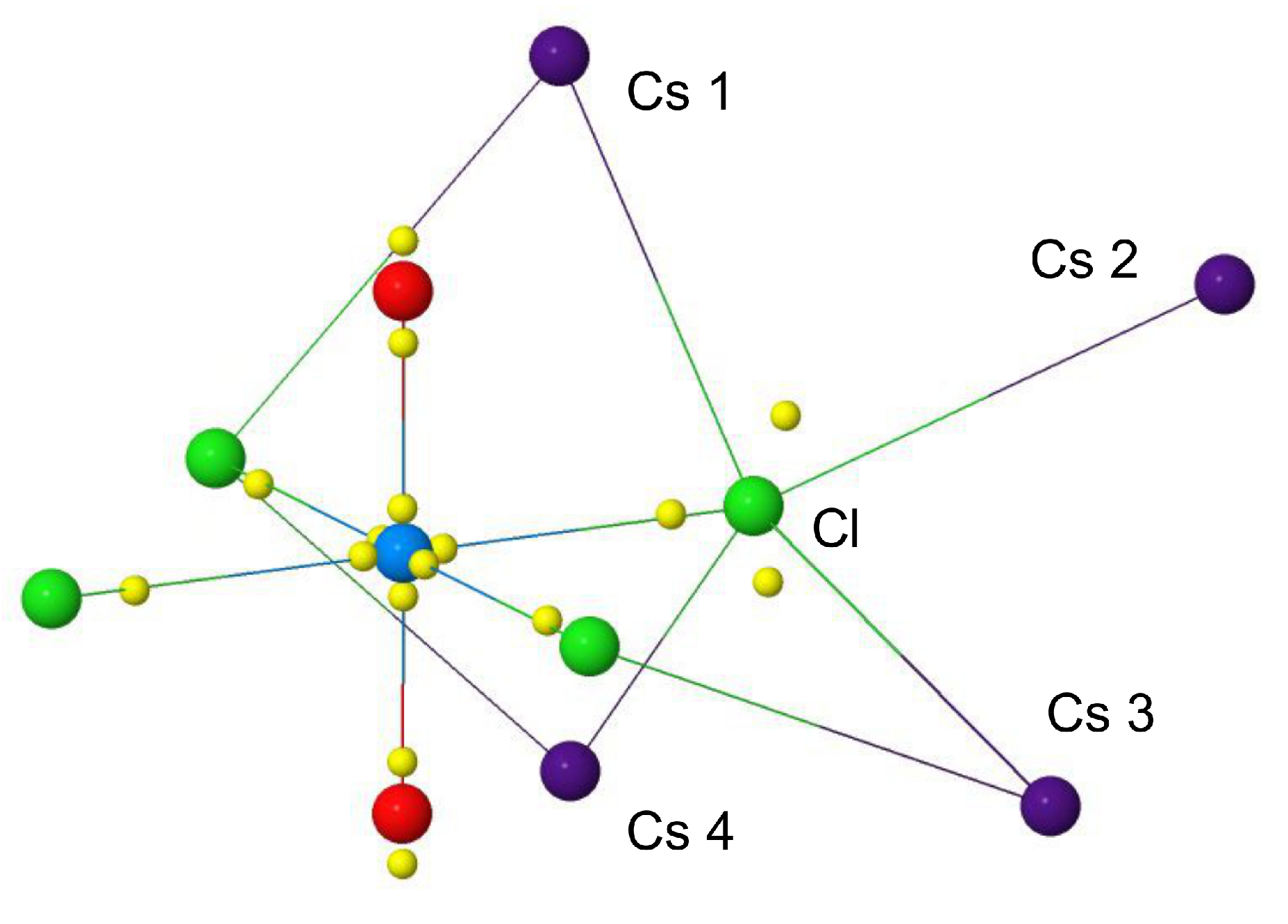
| [UOCl] | cry-CsUOCl | ||||
|---|---|---|---|---|---|
| Q | Q | Q | Q | Q | |
| U | +2.18 | +2.90 | +2.14 | +2.94 | +2.75 |
| O | −0.61 | −0.93 | −0.60 | −1.02 | −0.92 |
| Cl | −0.74 | −0.76 | −0.74 | −0.74 | −0.60 |
| Cs | - | - | +1.00 | +0.98 | +0.77 |
| O–U | Cl–U | O–Cs | Cl–Cs | Cl–Cs | Cl–Cs | Cl–Cs | ||
|---|---|---|---|---|---|---|---|---|
| l (Å) | Exp | 1.776 | 2.670 | 3.259 | 3.502 | 3.518 | 3.544 | 3.624 |
| Mol | 0.797 | 1.298 | - | - | - | - | - | |
| (Å) | Cry | 0.797 | 1.298 | 1.458 | 1.693 | 1.715 | 1.734 | 1.773 |
| Exp | 0.818 | 1.279 | 1.494 | 1.729 | 1.703 | 1.757 | 1.771 | |
| Mol | 2.059 | 0.421 | - | - | - | - | - | |
| (e/Å) | Cry | 2.058 | 0.425 | 0.057 | 0.070 | 0.069 | 0.066 | 0.058 |
| Exp | 1.695 | 0.486 | 0.053 | 0.067 | 0.071 | 0.076 | 0.068 | |
| Mol | 7.47 | 3.44 | - | - | - | - | - | |
| (e/Å) | Cry | 7.55 | 3.42 | 0.88 | 0.81 | 0.79 | 0.76 | 0.65 |
| Exp | 15.77 | 3.28 | 0.72 | 0.65 | 0.73 | 0.64 | 0.63 | |
| Mol | 1.780 | 1.256 | - | - | - | - | - | |
| Cry | 1.778 | 1.264 | 0.740 | 0.799 | 0.825 | 0.823 | 0.806 | |
| Exp | 1.587 | 1.417 | 0.724 | 0.845 | 0.831 | 0.902 | 0.860 | |
| Mol | −0.903 | −0.196 | - | - | - | - | - | |
| (a.u.) | Cry | −0.902 | −0.202 | 0.224 | 0.141 | 0.119 | 0.121 | 0.128 |
| Exp | −0.926 | −0.340 | 0.204 | 0.091 | 0.105 | 0.053 | 0.079 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cossard, A.; Casassa, S.; Gatti, C.; Desmarais, J.K.; Erba, A. Topology of the Electron Density and of Its Laplacian from Periodic LCAO Calculations on f-Electron Materials: The Case of Cesium Uranyl Chloride. Molecules 2021, 26, 4227. https://doi.org/10.3390/molecules26144227
Cossard A, Casassa S, Gatti C, Desmarais JK, Erba A. Topology of the Electron Density and of Its Laplacian from Periodic LCAO Calculations on f-Electron Materials: The Case of Cesium Uranyl Chloride. Molecules. 2021; 26(14):4227. https://doi.org/10.3390/molecules26144227
Chicago/Turabian StyleCossard, Alessandro, Silvia Casassa, Carlo Gatti, Jacques K. Desmarais, and Alessandro Erba. 2021. "Topology of the Electron Density and of Its Laplacian from Periodic LCAO Calculations on f-Electron Materials: The Case of Cesium Uranyl Chloride" Molecules 26, no. 14: 4227. https://doi.org/10.3390/molecules26144227
APA StyleCossard, A., Casassa, S., Gatti, C., Desmarais, J. K., & Erba, A. (2021). Topology of the Electron Density and of Its Laplacian from Periodic LCAO Calculations on f-Electron Materials: The Case of Cesium Uranyl Chloride. Molecules, 26(14), 4227. https://doi.org/10.3390/molecules26144227








