Advances in Pyranopyrazole Scaffolds’ Syntheses Using Sustainable Catalysts—A Review
Abstract
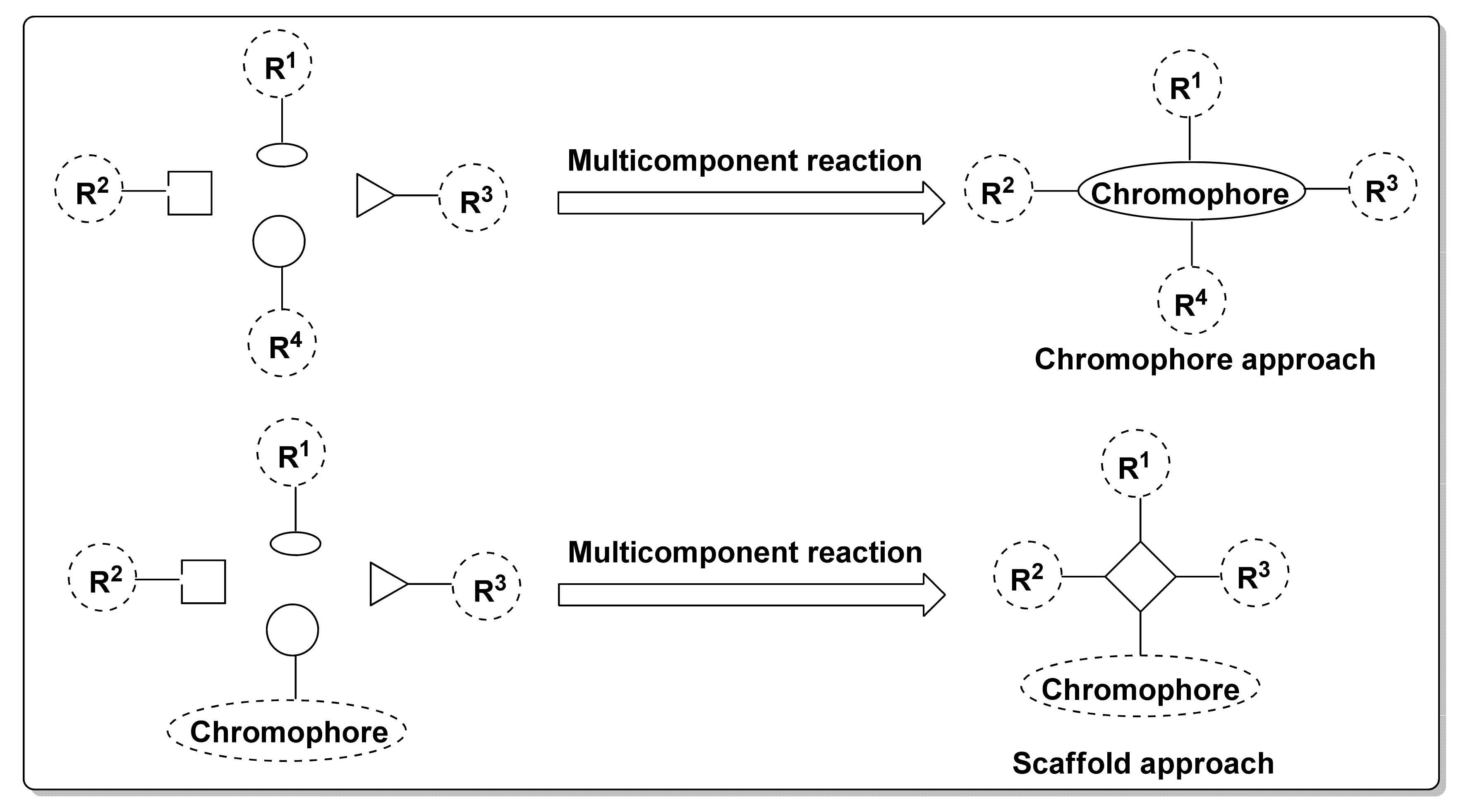
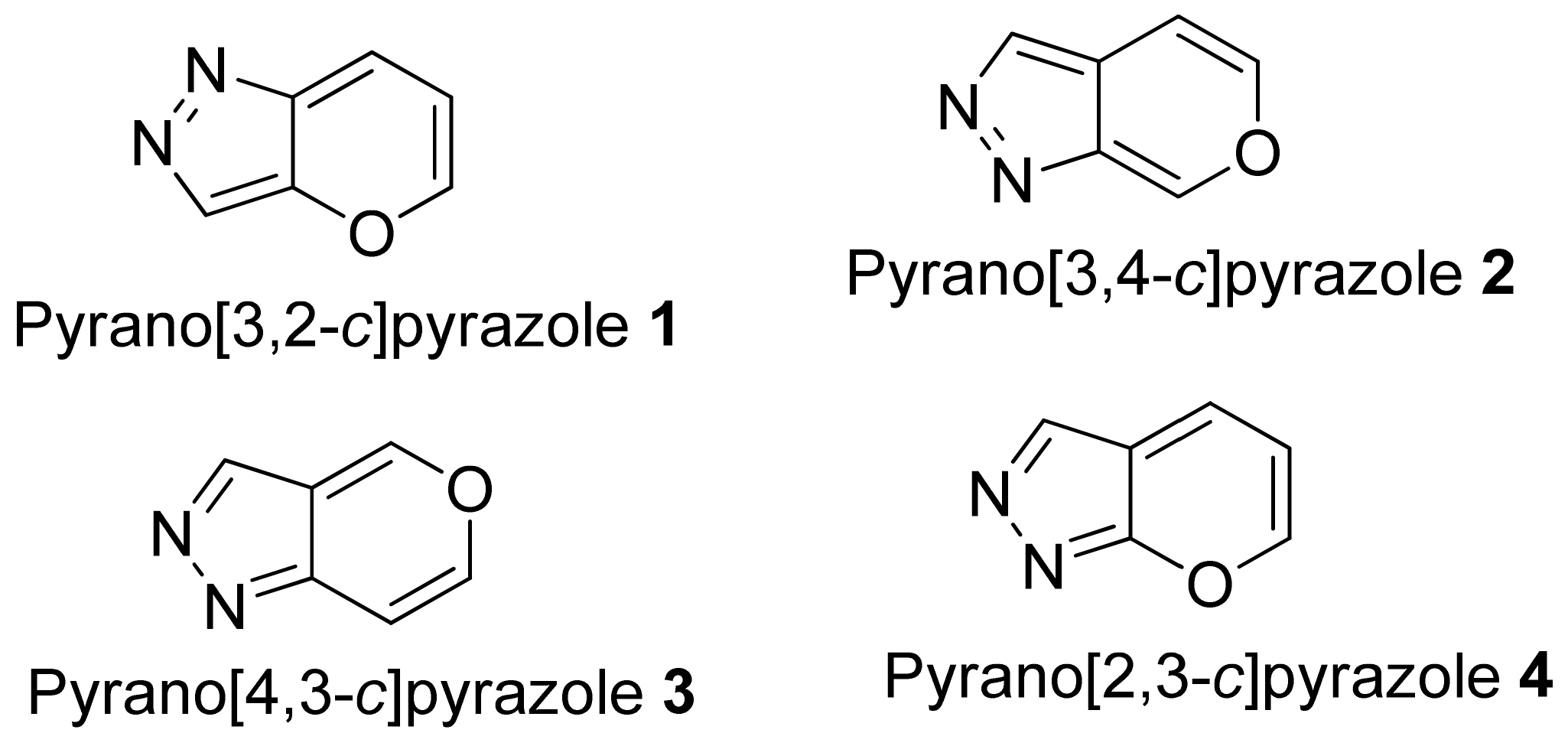
1. Introduction
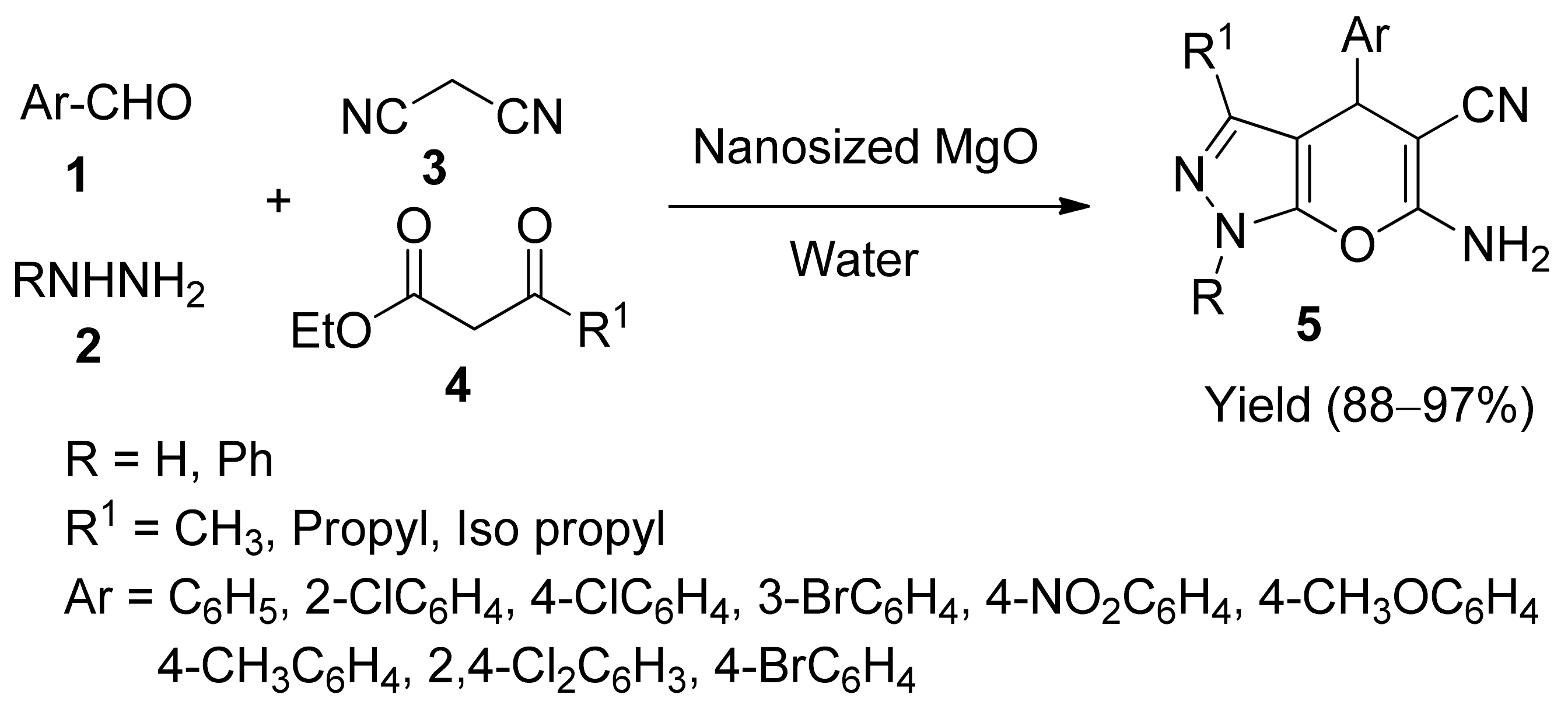
2. Single Metal-Containing Catalysts
3. With Bi-Metallic Catalysts
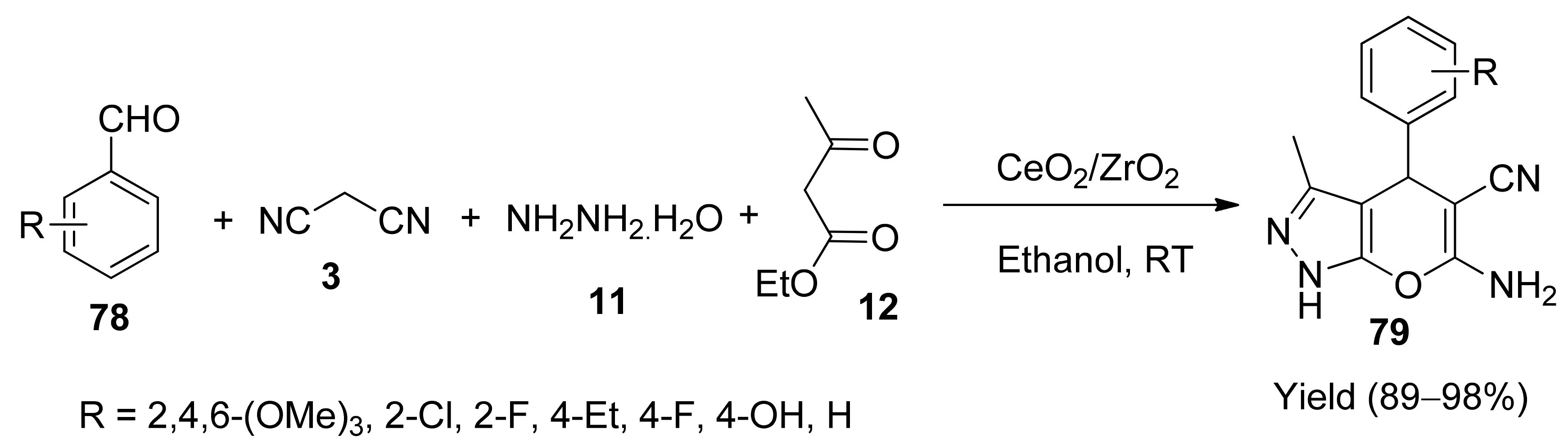
4. With Tri-Metallic Catalysts
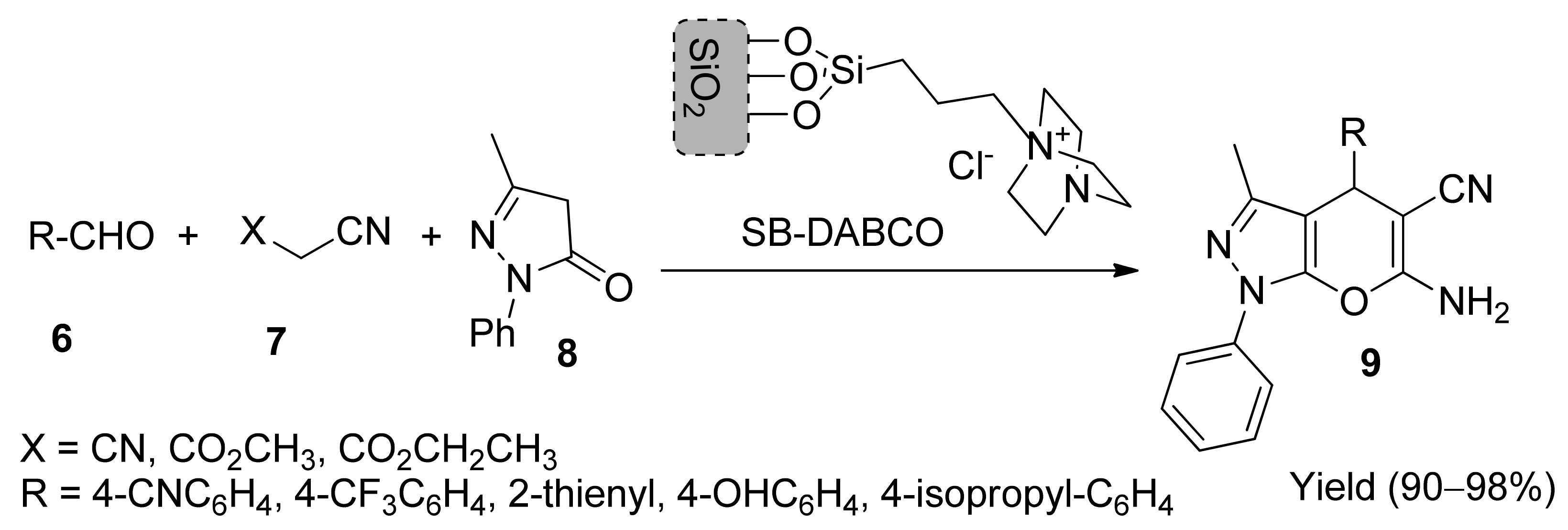
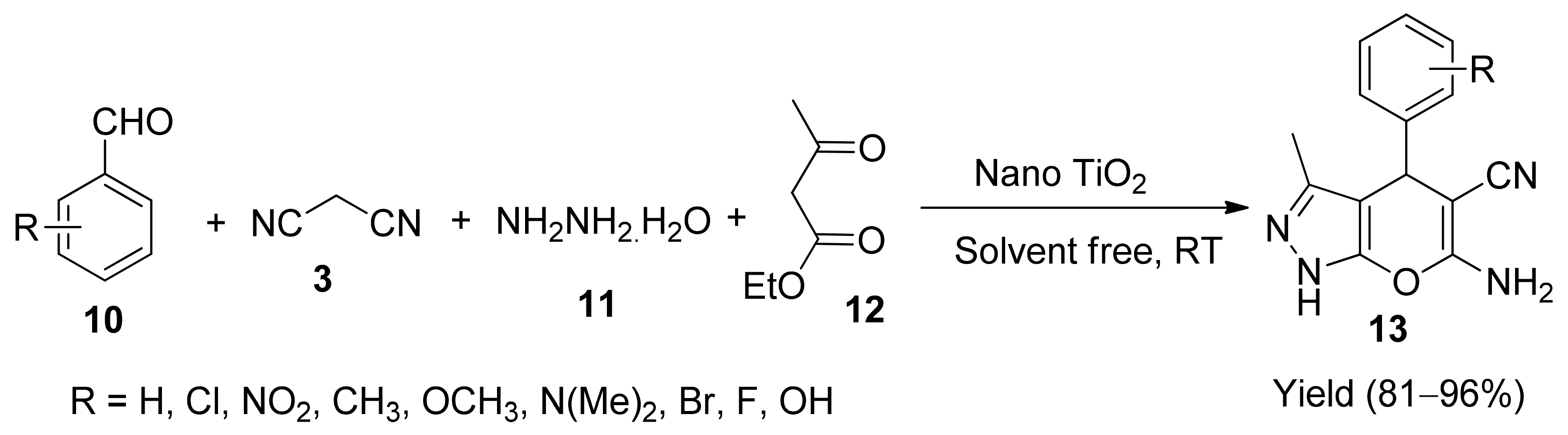
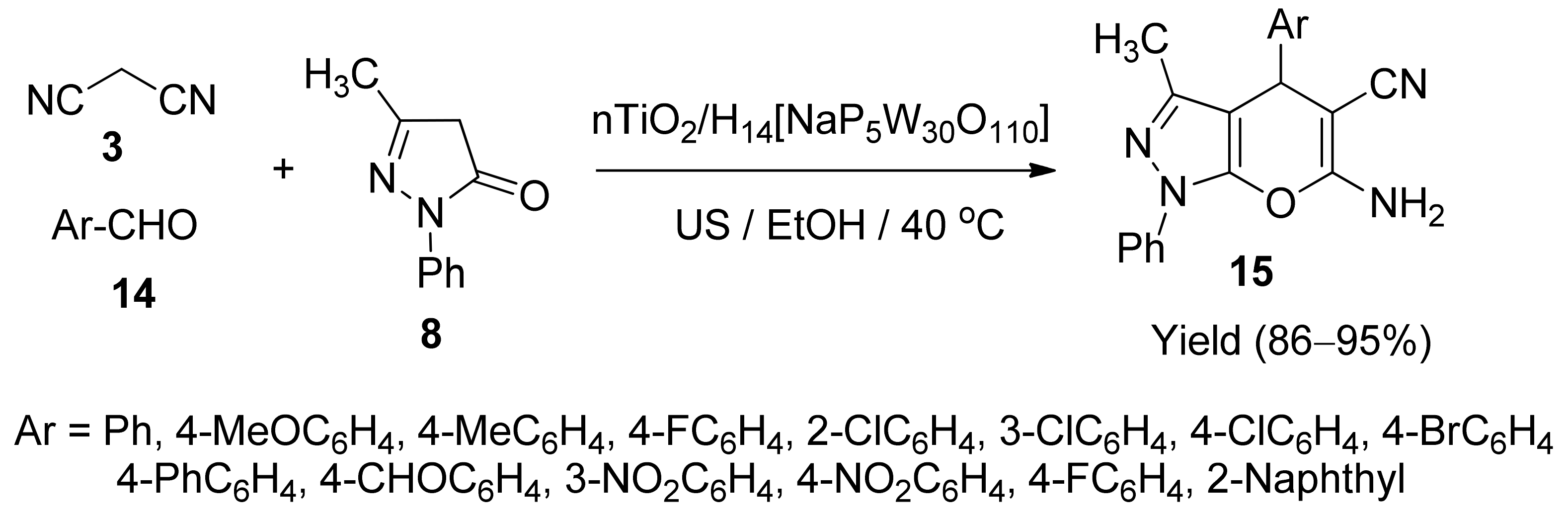
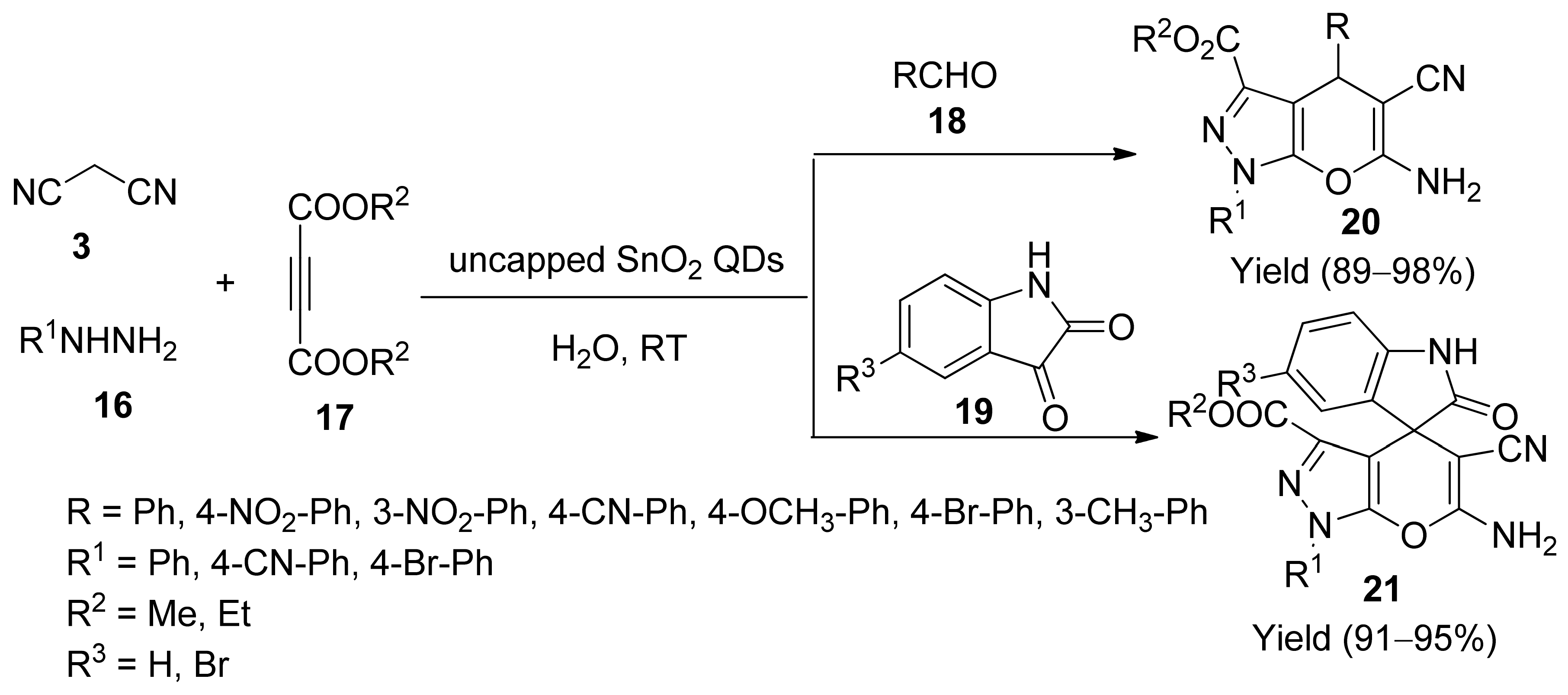
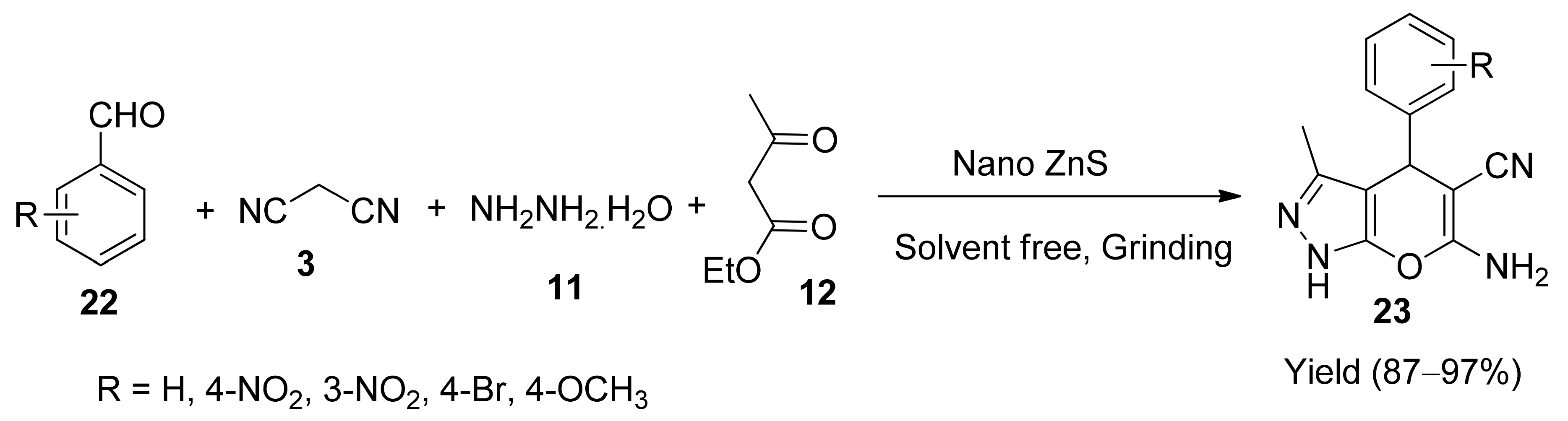
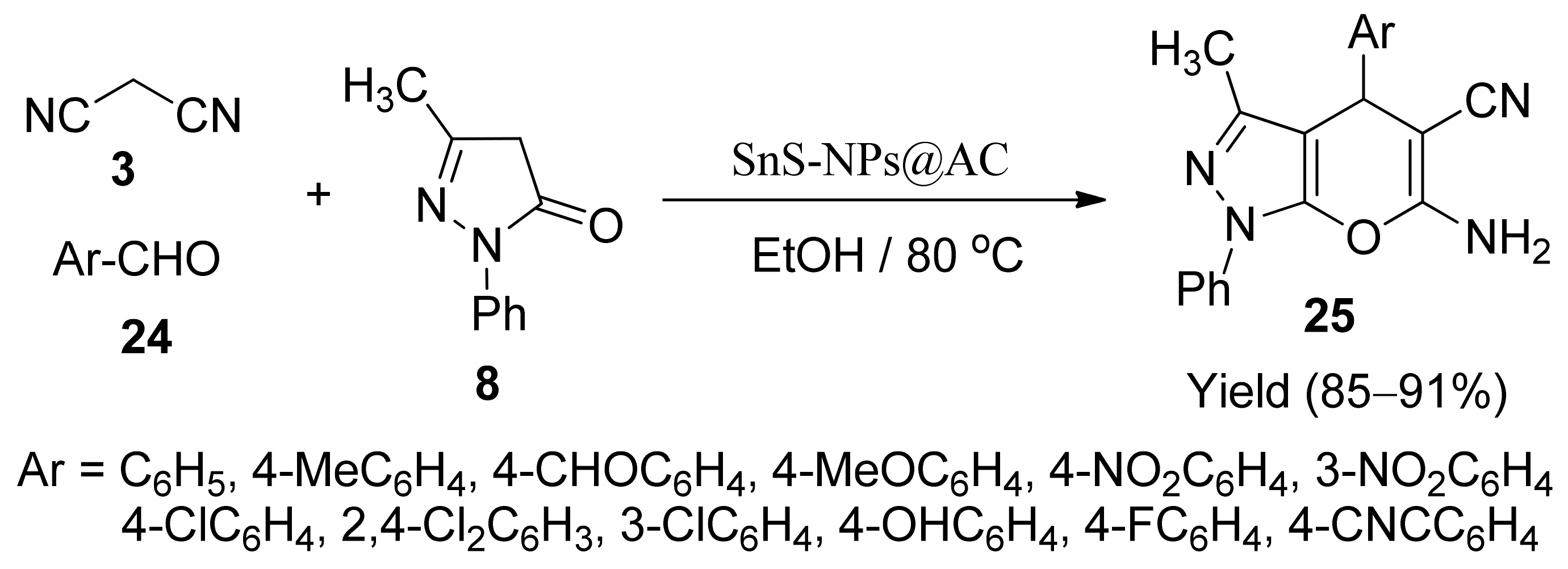
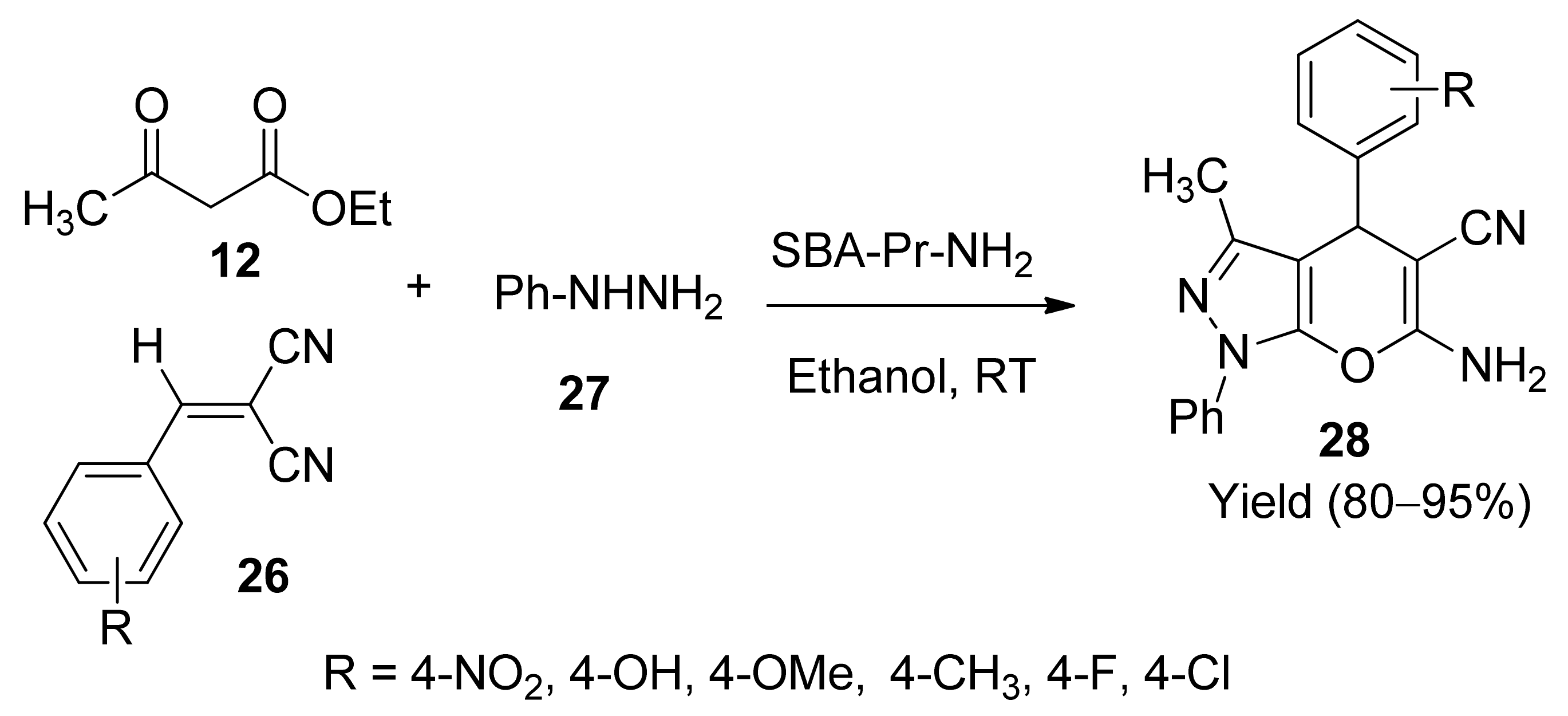
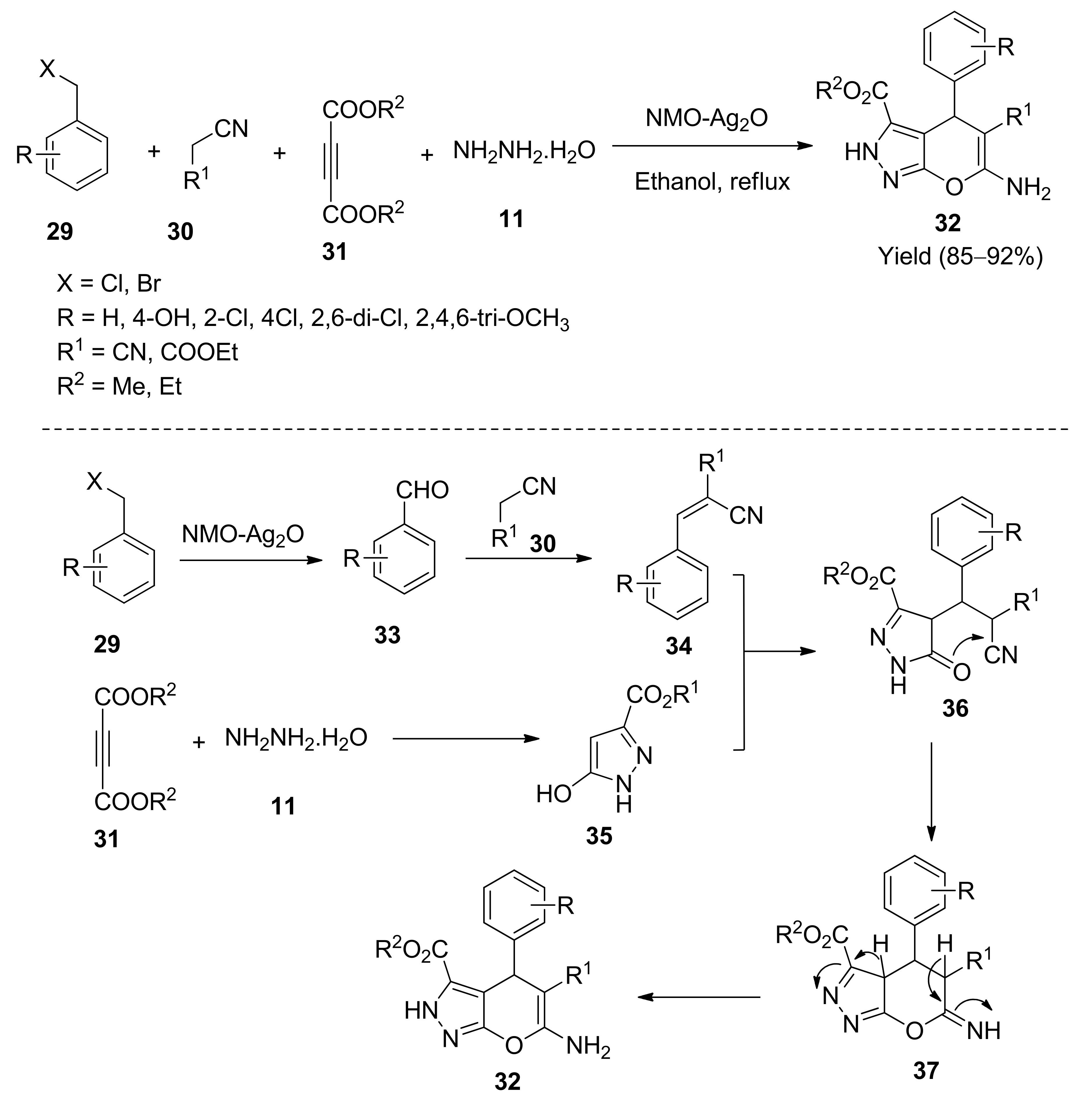
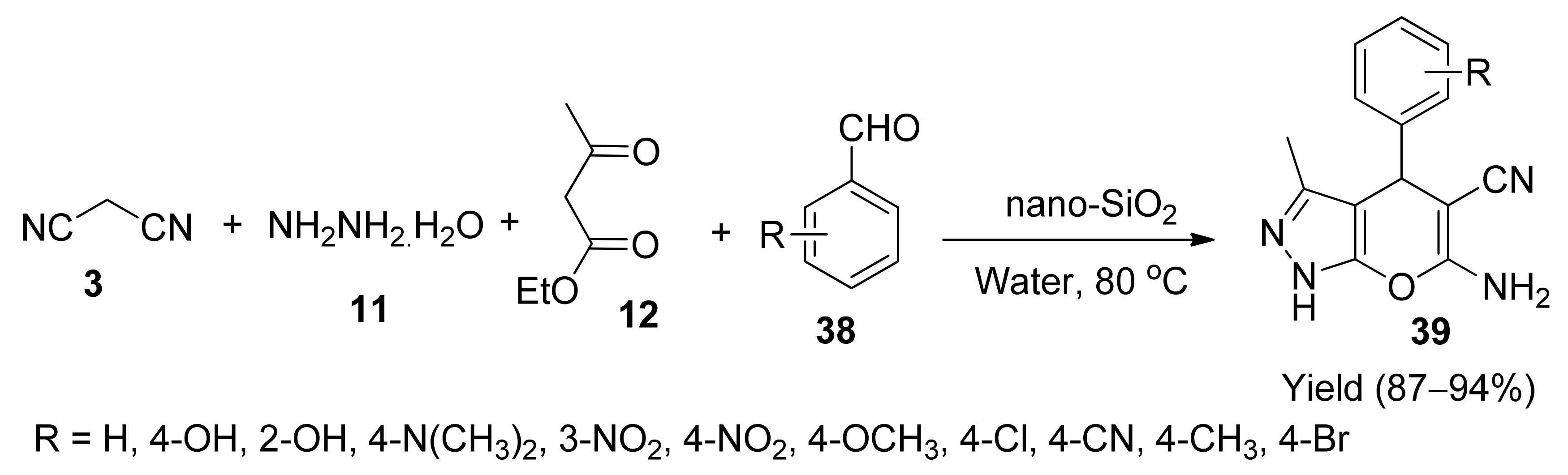
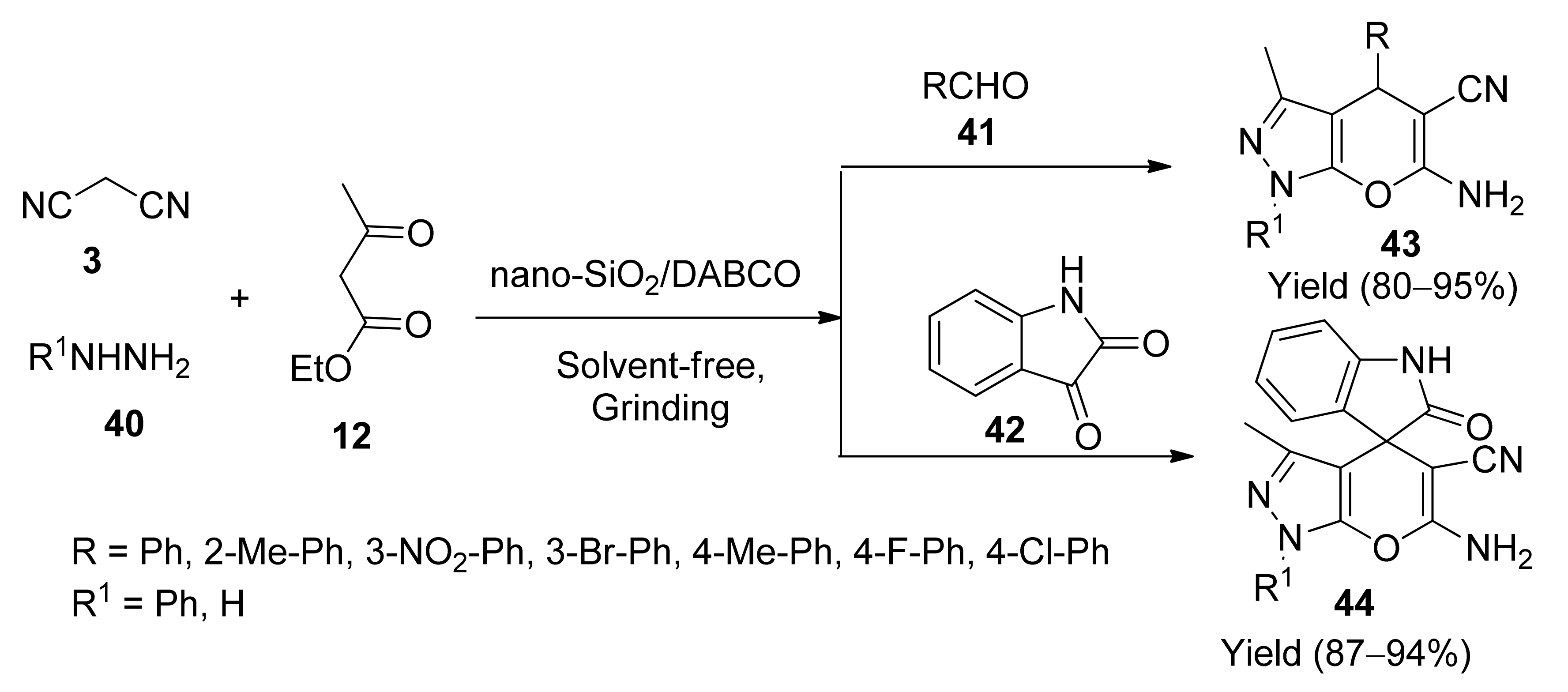
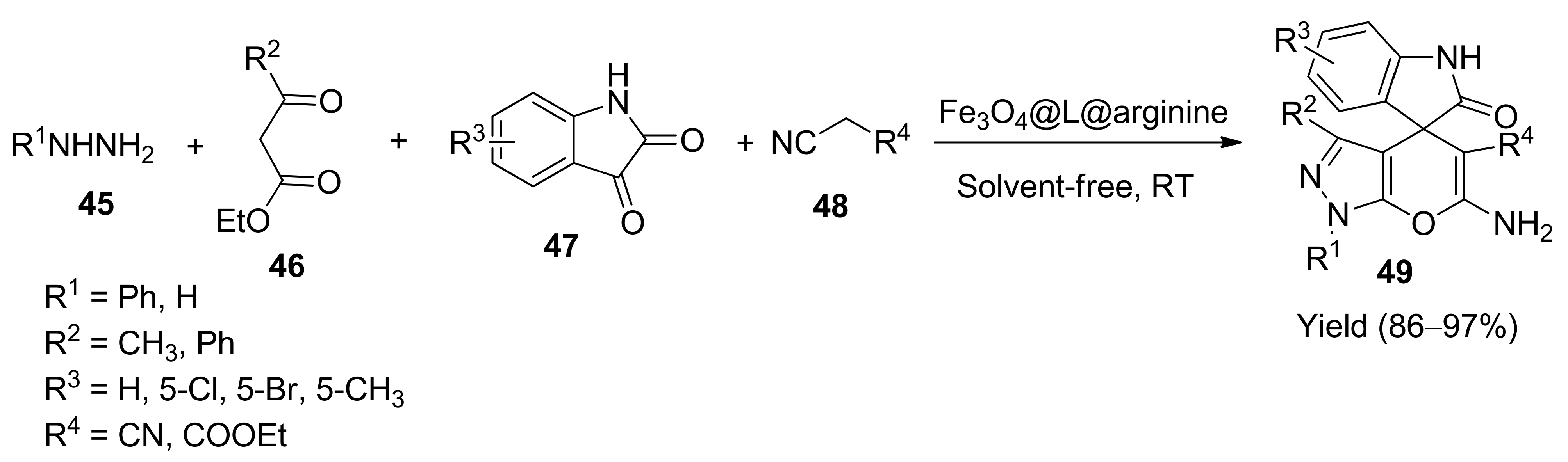
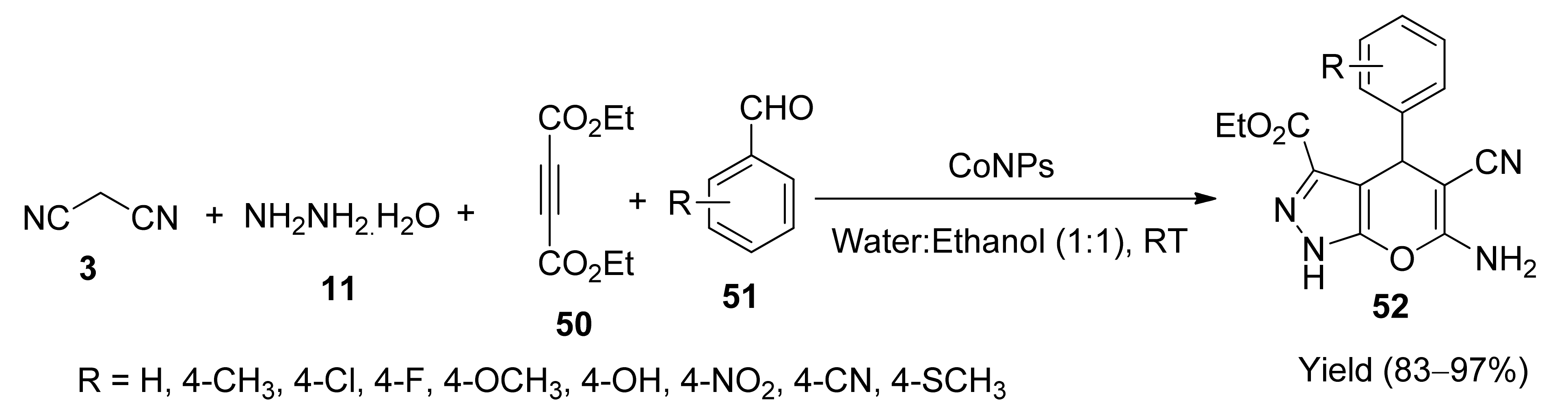
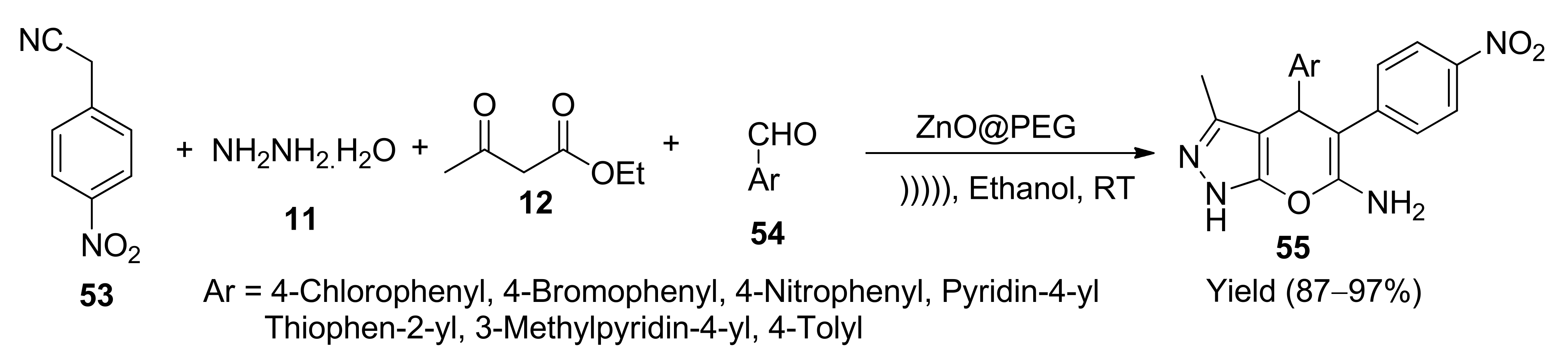
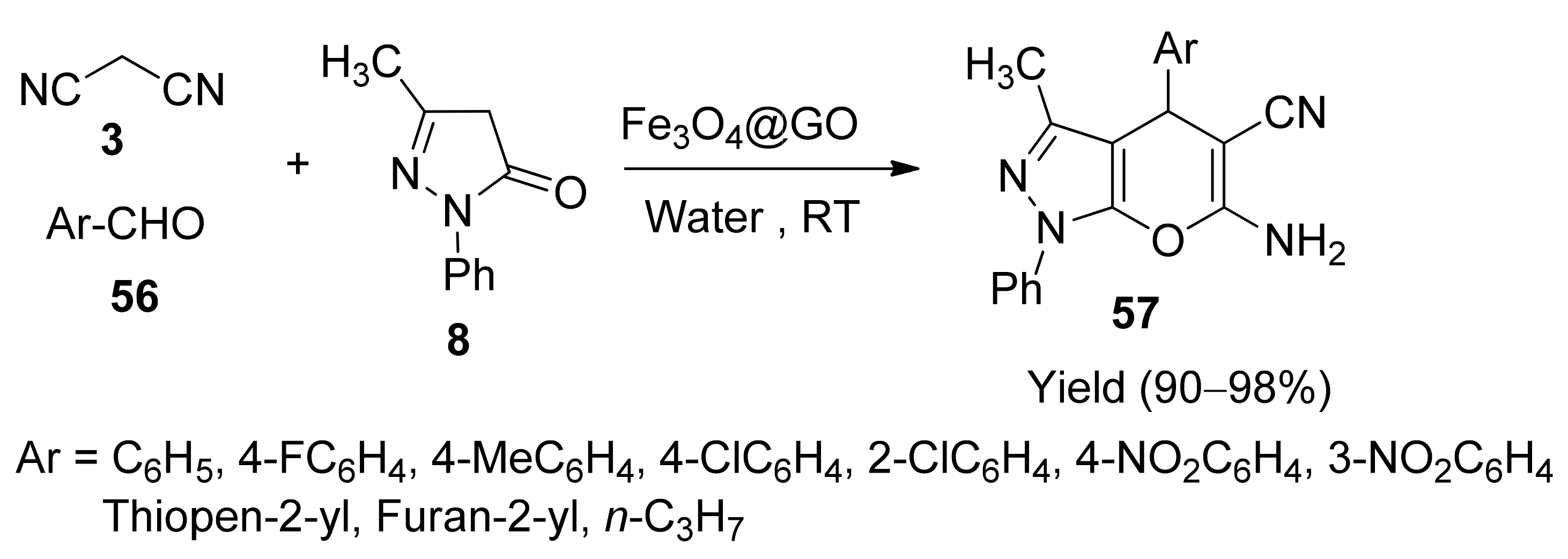
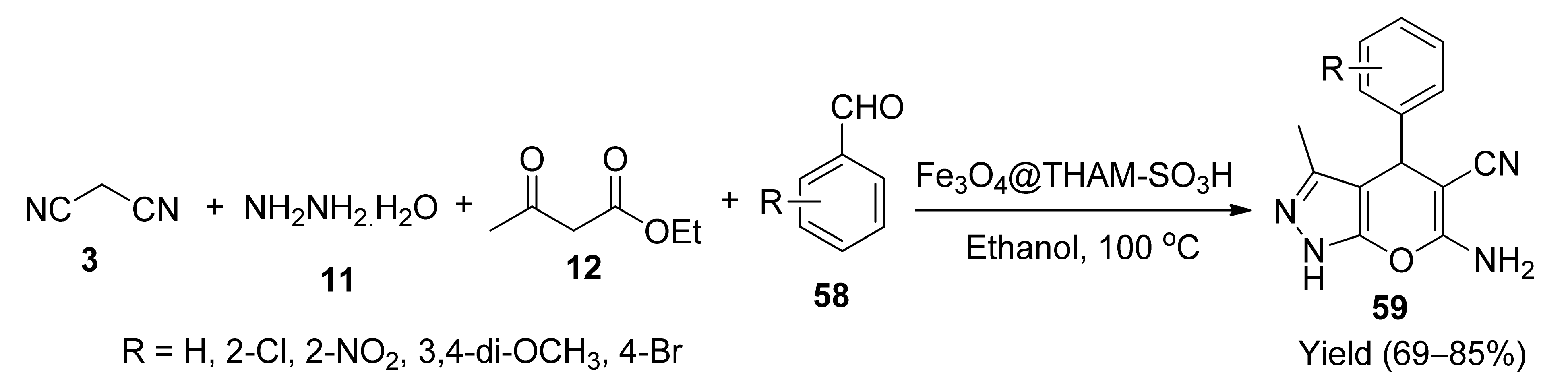
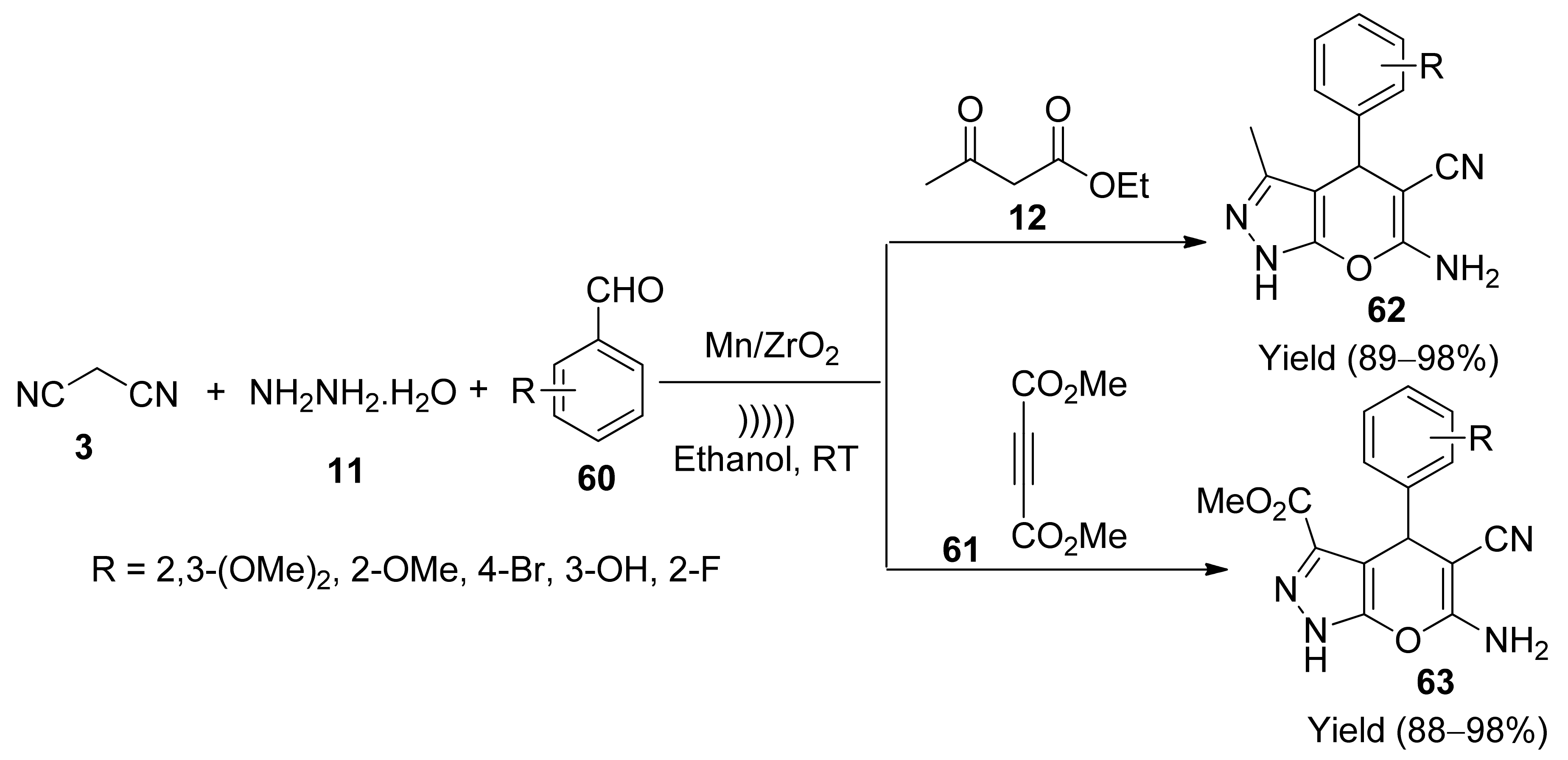
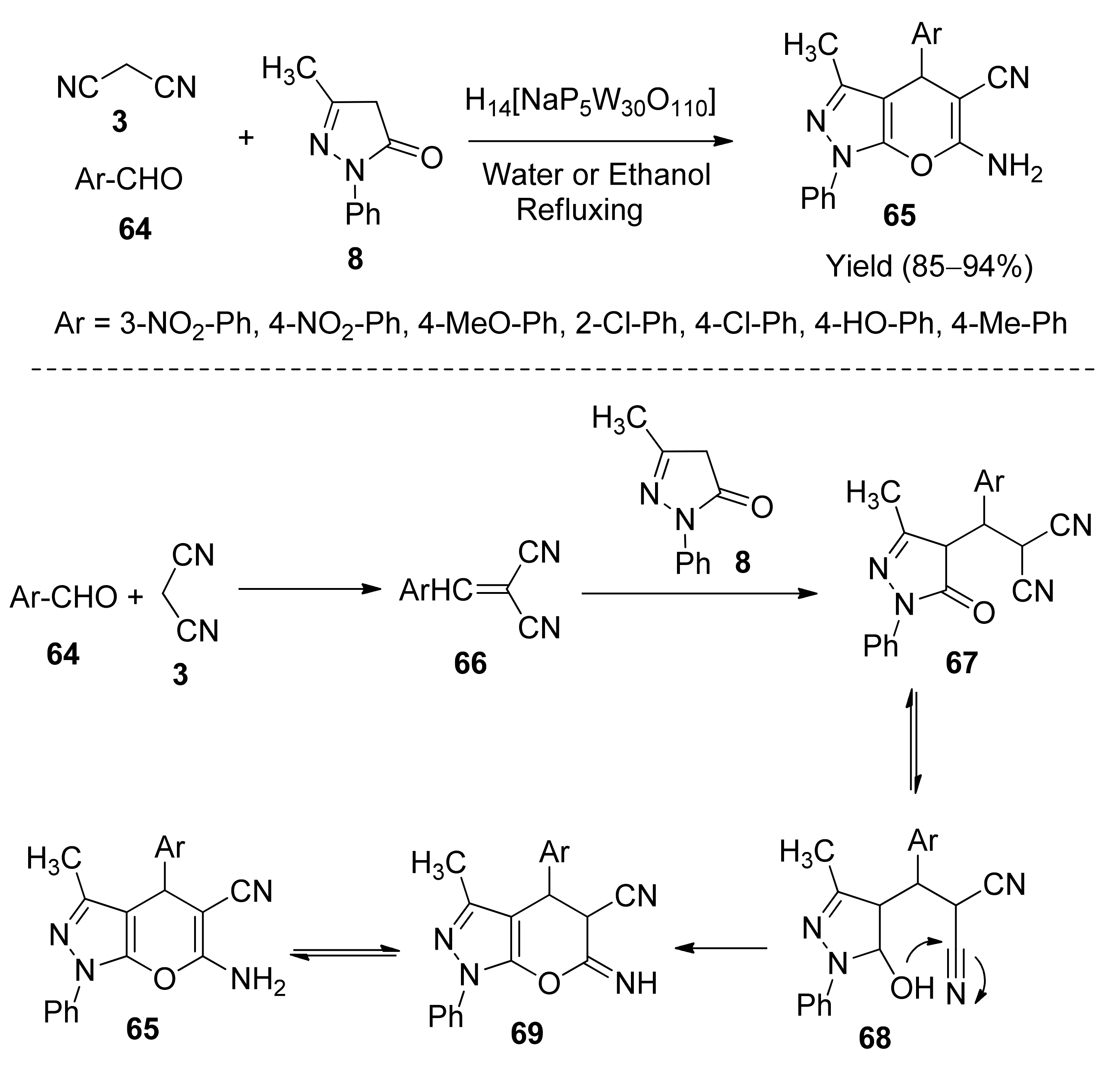
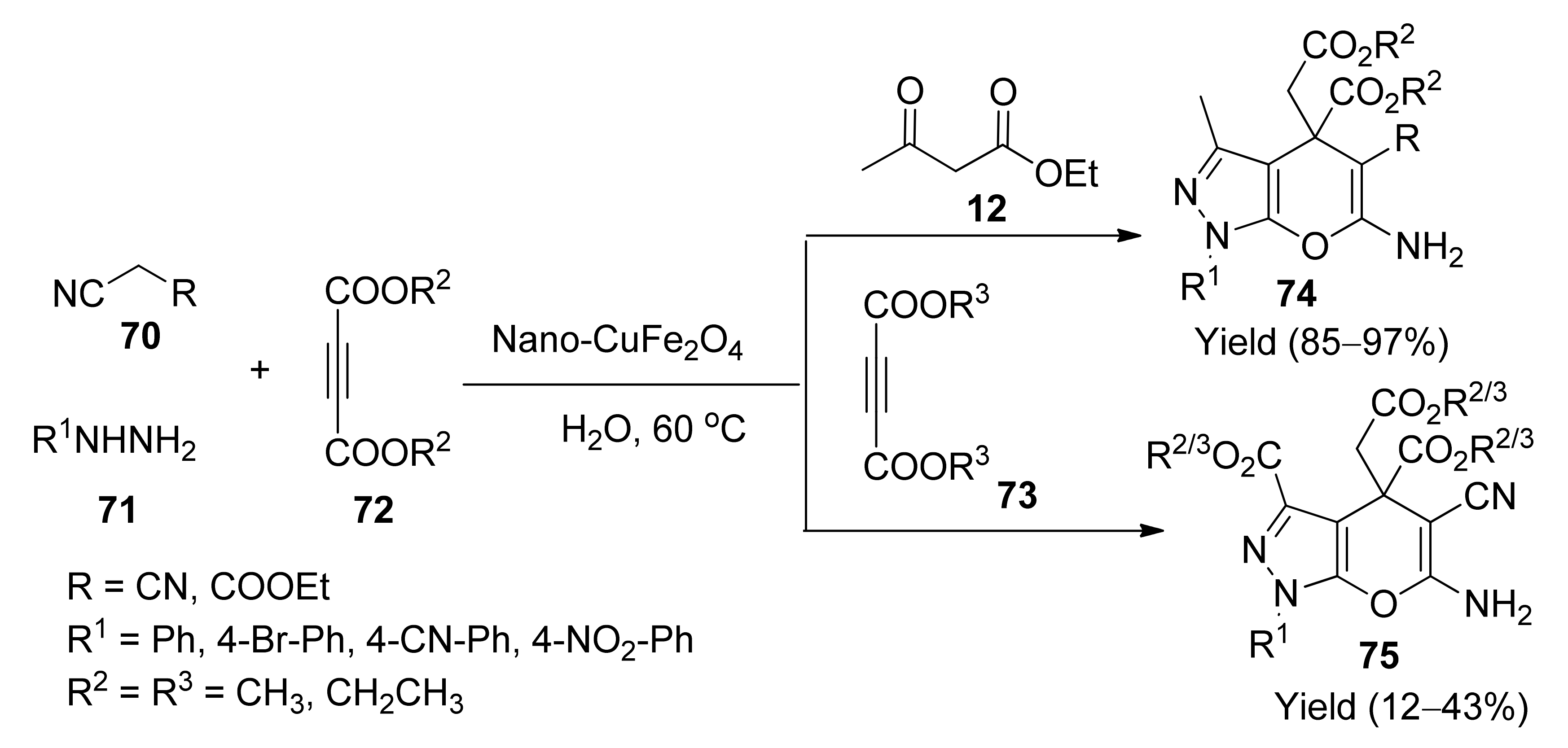
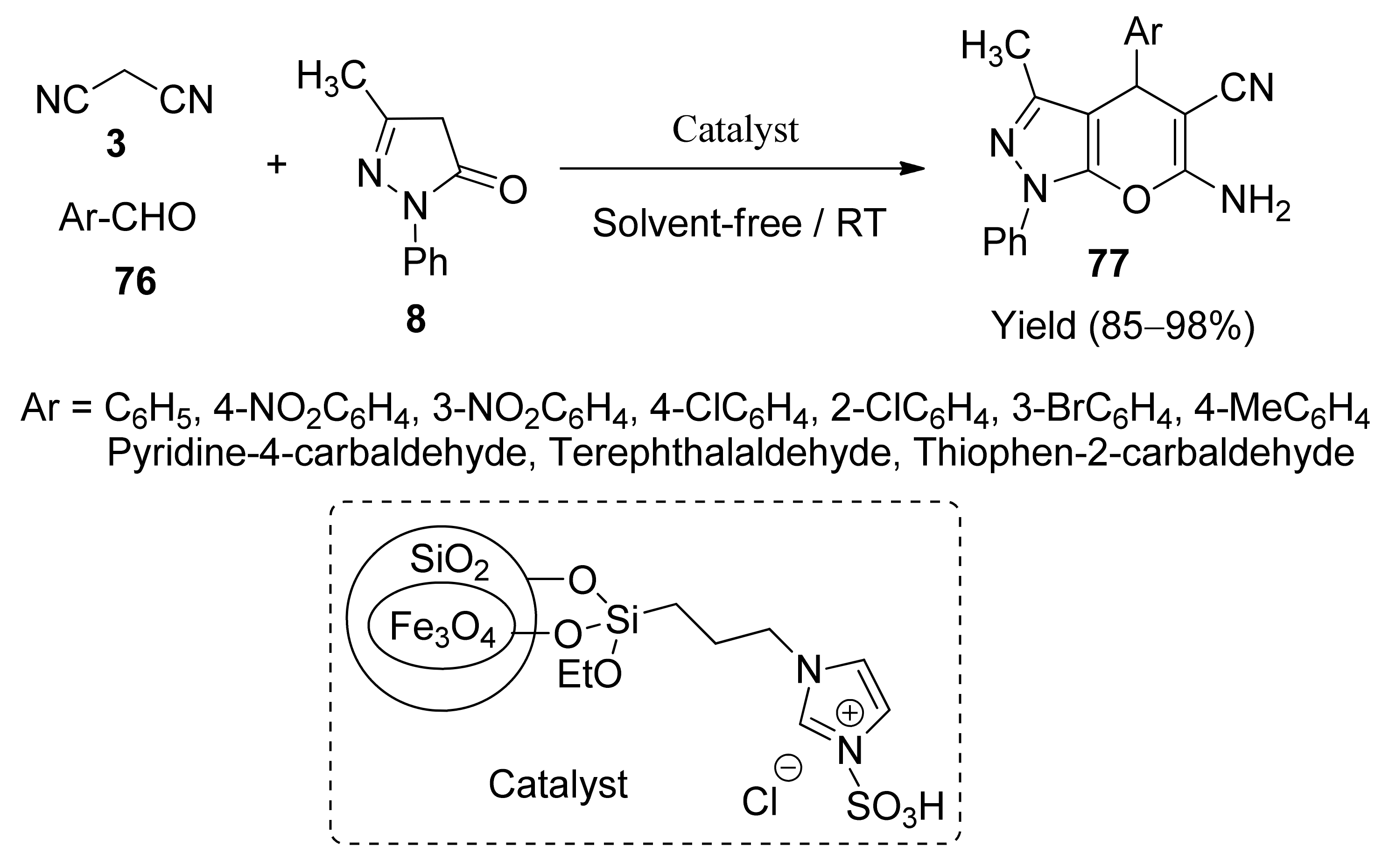
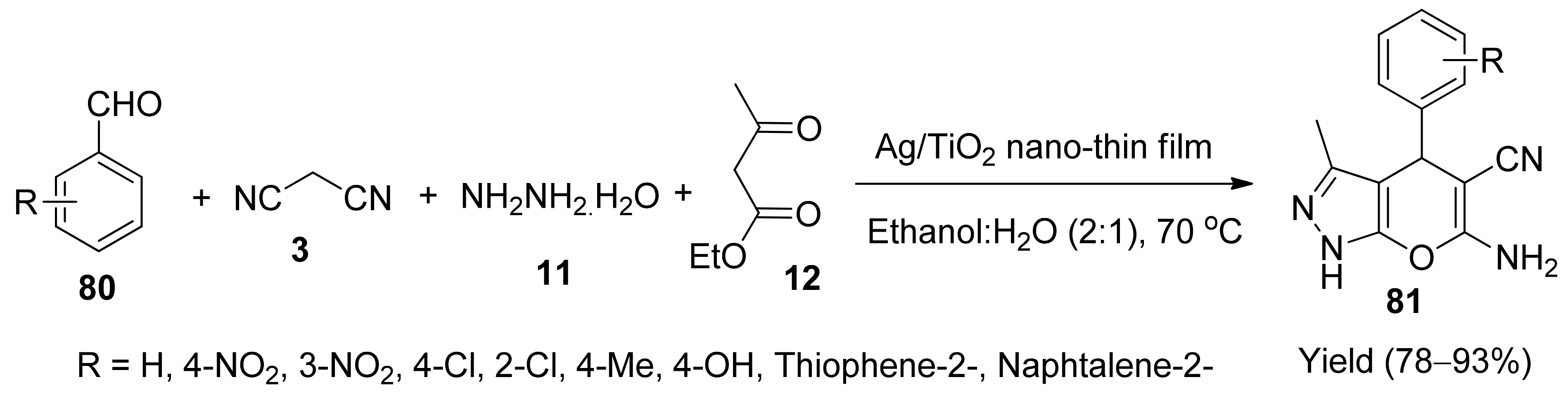
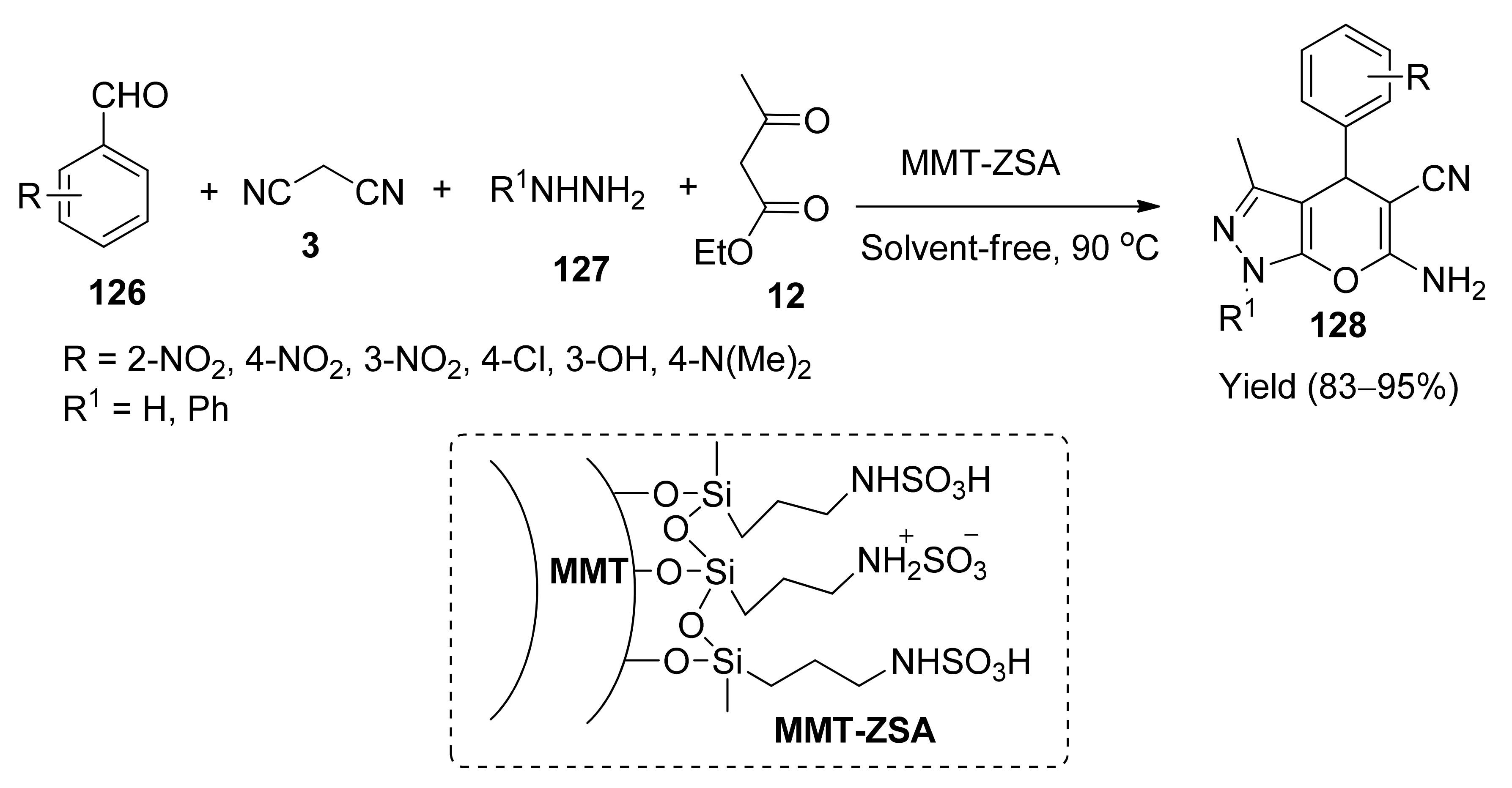
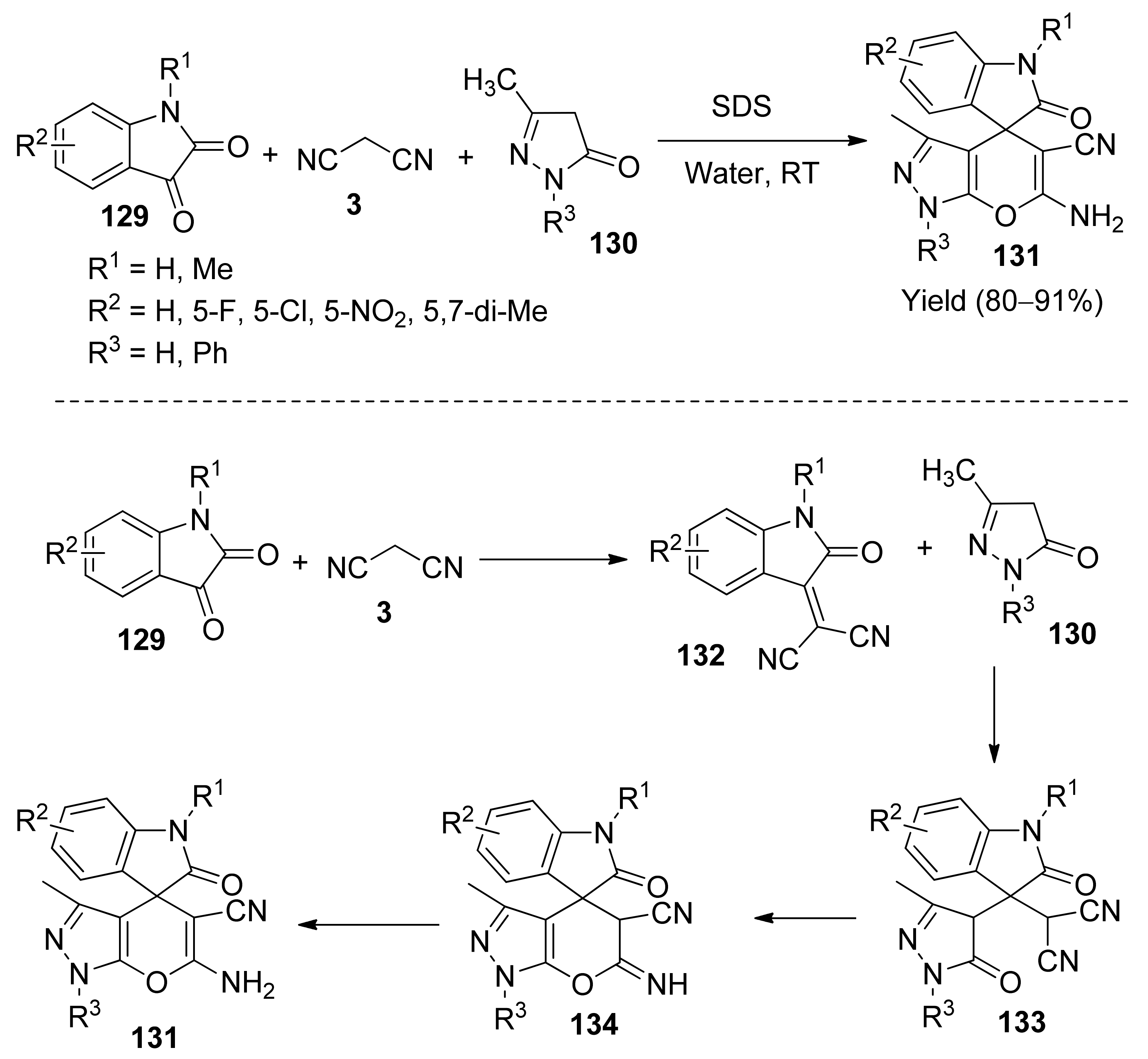
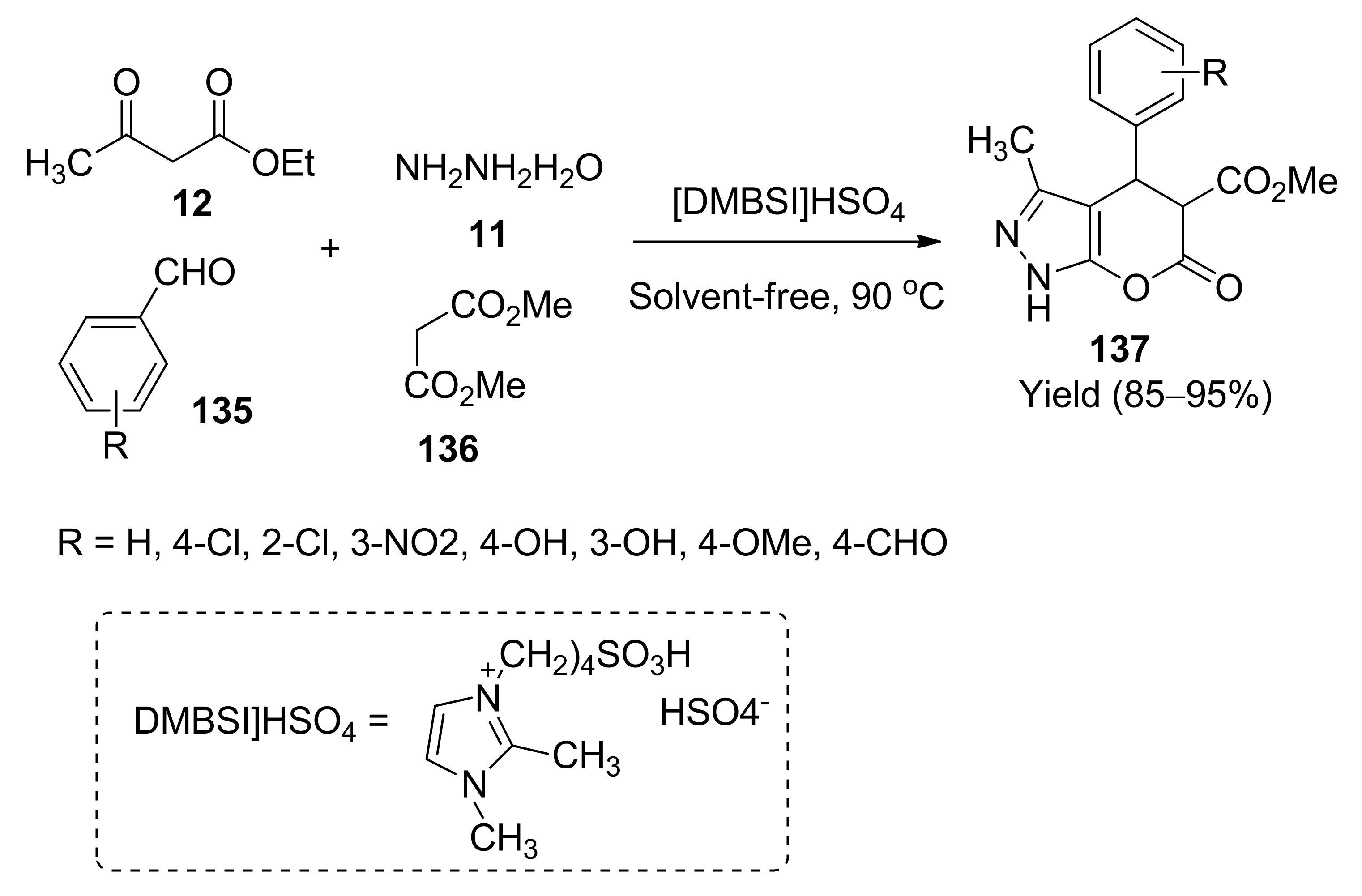
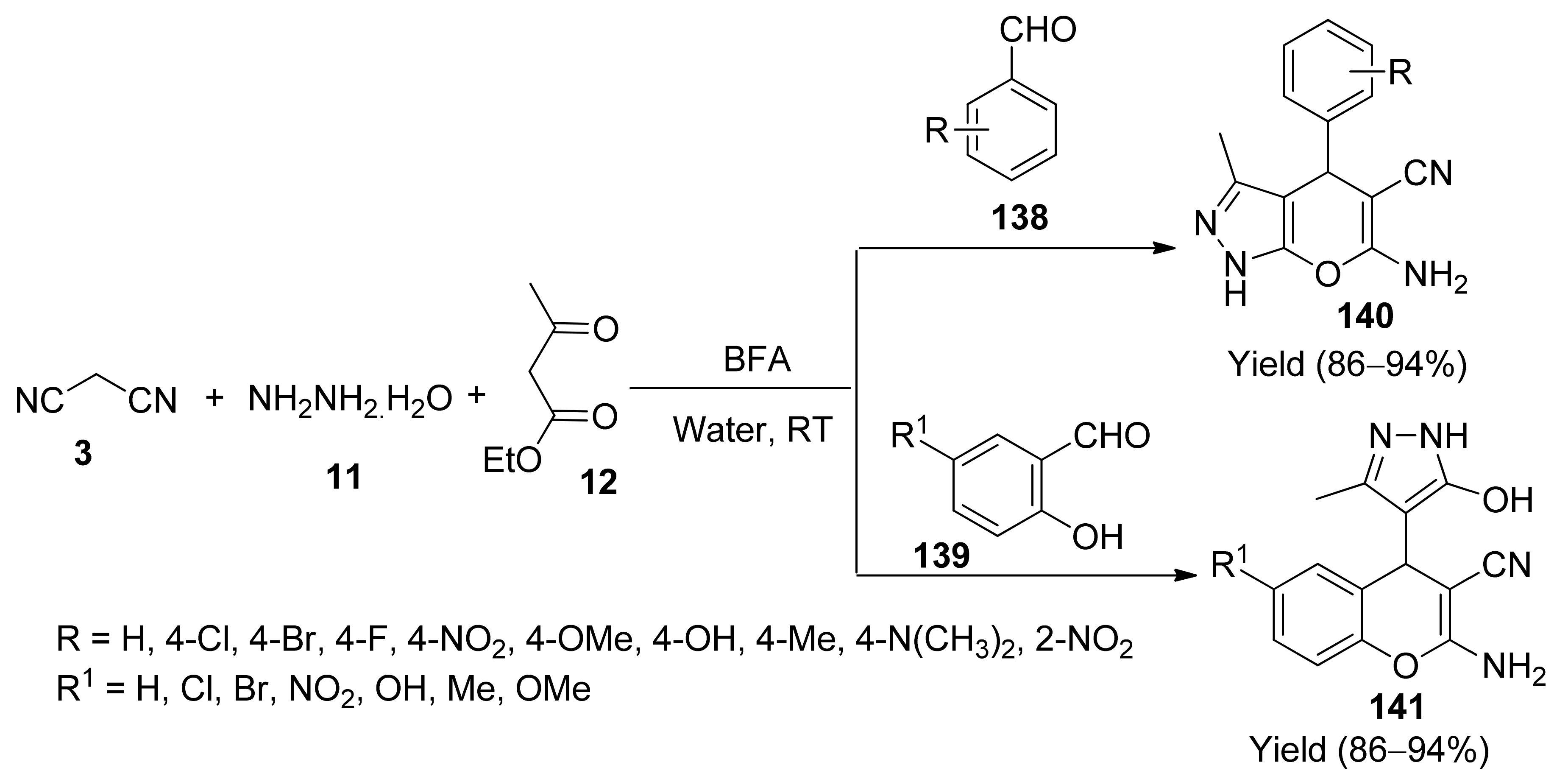
5. With Miscellaneous Materials and Composites as Catalysts
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vedrine, J.C. Heterogeneous catalysis on metal oxides. Catalysts 2017, 7, 341. [Google Scholar] [CrossRef]
- Hutchings, G.J. Heterogeneous catalysts-discovery and design. J. Mater. Chem. 2009, 19, 1222–1235. [Google Scholar] [CrossRef]
- Zecchina, A.; Lamberti, C.; Bordiga, S. Surface acidity and basicity: General concepts. Catal. Today 1998, 41, 169–177. [Google Scholar] [CrossRef]
- Qiu, R.; Chen, Y.; Yin, S.F.; Xu, X.; Au, C.T. A mini-review on air-stable organometallic Lewis acids: Synthesis, characterisation, and catalytic application in organic synthesis. RSC Adv. 2012, 2, 10774–10793. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.; Jonnalagadda, S.B. A review of recent advances in the green synthesis of azole- and pyran-based fused heterocycles using MCRs and sustainable catalysts. Curr. Org. Chem. 2021, 25, 4–39. [Google Scholar] [CrossRef]
- Kerru, N.; Bhaskaruni, S.V.H.S.; Gummidi, L.; Maddila, S.N.; Maddila, S.; Jonnalagadda, S.B. Recent advances in heterogeneous catalysts for the synthesis of imidazole derivatives. Syn. Commun. 2019, 49, 2437–2459. [Google Scholar] [CrossRef]
- Climent, M.J.; Corma, A.; Iborra, S. Homogeneous and heterogeneous catalysts for multicomponent reactions. RSC Adv. 2012, 2, 16–58. [Google Scholar] [CrossRef]
- Bhaskaruni, S.V.H.S.; Maddila, S.; Gangu, K.K.; Jonnalagadda, S.B. A review on multicomponent green synthesis of N-containing heterocycles using mixed oxides as heterogeneous catalysts. Arab. J. Chem. 2020, 13, 1142–1178. [Google Scholar] [CrossRef]
- Ruban, A.; Hammer, B.; Stoltze, P.; Skriver, H.L.; Norskov, J.K. Surface electronic structure and reactivity of transition and noble metals. J. Mol. Catal. Chem. 1997, 115, 421–429. [Google Scholar] [CrossRef]
- Maddila, S.N.; Maddila, S.; Bhaskaruni, S.V.H.S.; Kerru, N.; Jonnalagadda, S.B. MnO2 on hydroxyapatite: A green heterogeneous catalyst and synthesis of pyran-carboxamide derivatives. Inorg. Chem. Commun. 2020, 112, 107706. [Google Scholar] [CrossRef]
- Maddila, S.N.; Maddila, S.; Kerru, N.; Bhaskaruni, S.V.H.S.; Jonnalagadda, S.B. Facile one-pot synthesis of arylsulfonyl-4H-pyrans catalysed by Ru loaded fluorapatite. Chem. Sel. 2020, 5, 1786–1791. [Google Scholar]
- Sun, J.; Zhu, K.; Gao, F.; Wang, C.; Liu, J.; Peden, C.H.F.; Wang, Y. Direct conversion of bio-ethanol to isobutene on nanosized Zn(x)Zr(y)O(z) mixed oxides with balanced acid-base sites. J. Am. Chem. Soc. 2011, 133, 11096–11099. [Google Scholar] [CrossRef]
- Cioc, R.C.; Ruijter, E.; Orru, R.V.A. Multicomponent reactions: Advanced tools for sustainable organic synthesis. Green Chem. 2014, 16, 2958–2975. [Google Scholar] [CrossRef]
- Gu, Y. Multicomponent reactions in unconventional solvents: State of the art. Green Chem. 2012, 14, 2091–2128. [Google Scholar] [CrossRef]
- Elwahy, A.H.M.; Shaaban, M.R. Synthesis of heterocycles and fused heterocycles catalysed by nanomaterials. RSC Adv. 2015, 5, 75659–75710. [Google Scholar] [CrossRef]
- Levi, L.; Muller, T.J.J. Multicomponent syntheses of functional chromophores. Chem. Soc. Rev. 2016, 45, 2825–2846. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Gangu, K.K.; Maddila, S.; Jonnalagadda, S.B. Synthesis of novel furo[3,2-c]coumarin derivatives through multicomponent [4+1] cycloaddition reaction using ZnO/FAp as a sustainable catalyst. Chem. Sel. 2020, 5, 4104–4110. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.; Jonnalagadda, S.B. Polyethylene glycol (PEG-400) mediated one-pot green synthesis of 4,7-dihydro-2H-pyrazolo[3,4-b]pyridines under catalyst-free conditions. Chem. Sel. 2020, 5, 12407–12410. [Google Scholar]
- Kerru, N.; Gummidi, L.; Maddila, S.; Jonnalagadda, S.B. Efficient synthesis of novel functionalised dihydro-pyrazolo[3,4-d] pyridines via the three-component reaction using MgO/HAp as a sustainable catalyst. Inorg. Chem. Commun. 2021, 123, 108321. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Bhaskaruni, S.V.H.S.; Maddila, S.N.; Jonnalagadda, S.B. Green synthesis and characterisation of novel 1,2,4,5-tetrasubstituted imidazole derivatives with eco-friendly red brick clay as efficacious catalyst. Mol. Divers. 2020, 24, 889–901. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.; Jonnalagadda, S.B. Gadolinium oxide loaded zirconia and multicomponent synthesis of novel dihydro-pyrazolo[3,4-d]pyridines under green conditions. Sustain. Chem. Pharmacy 2020, 18, 100316. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.N.; Bhaskaruni, S.V.H.S.; Jonnalagadda, S.B. Bi2O3/FAp, a sustainable catalyst for synthesis of dihydro- [1,2,4]triazolo[1,5-a]pyrimidine derivatives through green strategy. Appl. Organometal. Chem. 2020, 34, 5590. [Google Scholar] [CrossRef]
- Kerru, N.; Bhaskaruni, S.V.H.S.; Gummidi, L.; Maddila, S.N.; Rana, S.; Singh, P.; Jonnalagadda, S.B. Synthesis of novel pyrazole-based triazolidin-3-one derivatives by using ZnO/ZrO2 as a reusable catalyst under green conditions. Appl. Organometal. Chem. 2019, 33, e4722. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.N.; Bhaskaruni, S.V.H.S.; Maddila, S.; Jonnalagadda, S.B. Green synthesis and characterisation of novel [1,3,4]thiadiazolo/benzo[4,5]thiazolo[3,2-a] pyrimidines via multicomponent reaction using vanadium oxide loaded on fluorapatite as a robust and sustainable catalyst. RSC Adv. 2020, 10, 19803–19810. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Bhaskaruni, S.V.H.S.; Maddila, S.N.; Jonnalagadda, S.B. One-pot green synthesis of novel 5,10-dihydro-1H-pyrazolo[1,2-b]phthalazine derivatives with eco-friendly biodegradable eggshell powder as efficacious catalyst. Res. Chem. Intermed. 2020, 46, 3067–3083. [Google Scholar] [CrossRef]
- Settypalli, T.; Chunduri, V.R.; Kerru, N.; Nallapaneni, H.K.; Chintha, V.R.; Wudayagiri, R.; Daggupati, T.; Yeguvapalli, S. Design, synthesis, neuroprotective, antibacterial activities and docking studies of novel thieno[2,3-d]pyrimidine-alkyne mannich base and oxadiazole hybrids. Bioorg. Med. Chem. Lett. 2018, 28, 1663–1669. [Google Scholar]
- Gummidi, L.; Kerru, N.; Awolade, P.; Ibeji, C.; Karpoormath, R.; Singh, P. N-Phenyl substituent controlled diastereoselective synthesis of β-lactam-isatin conjugates. Tetrahed. Lett. 2020, 61, 151602. [Google Scholar] [CrossRef]
- Gummidi, L.; Kerru, N.; Awolade, P.; Raza, A.; Sharma, A.; Singh, P. Synthesis of indole-tethered [1,3,4]thiadiazolo and [1,3,4]oxadiazolo[3,2-a]pyrimidin-5-one hybrids as anti-pancreatic cancer agents. Bioorg. Med. Chem. Lett. 2020, 30, 127544. [Google Scholar] [CrossRef]
- Slobbe, P.; Ruijter, E.; Orru, R.V.A. Recent applications of multicomponent reactions in medicinal chemistry. Med. Chem. Comm. 2012, 3, 1189–1218. [Google Scholar] [CrossRef]
- Rotstein, B.H.; Zaretsky, S.; Rai, V.; Yudin, A.K. Small heterocycles in multicomponent reactions. Chem. Rev. 2014, 114, 8323–8359. [Google Scholar] [CrossRef]
- Afshari, R.; Shaabani, A. Materials functionalisation with multicomponent reactions: State of the art. ACS Comb. Sci. 2018, 20, 499–528. [Google Scholar] [CrossRef] [PubMed]
- Bhaskaruni, S.V.H.S.; Gangu, K.K.; Maddila, S.; Jonnalagadda, S.B. Our contributions in synthesis of diverse heterocyclic scaffolds by using mixed oxides as heterogeneous catalysts. Chem. Rec. 2019, 19, 1793–1812. [Google Scholar] [CrossRef] [PubMed]
- Kerru, N.; Gummidi, L.; Maddila, S.N.; Gangu, K.K.; Jonnalagadda, S.B. Four-component rapid protocol with nickel oxide loaded on fluorapatite as a sustainable catalyst for the synthesis of novel imidazole analogs. Inorg. Chem. Commun. 2020, 116, 107935. [Google Scholar] [CrossRef]
- Kerru, N.; Bhaskaruni, S.V.H.S.; Gummidi, L.; Maddila, S.N.; Singh, P.; Jonnalagadda, S.B. Efficient synthesis of novel pyrazole-linked 1,2,4-triazolidine-3-thiones using bismuth on zirconium oxide as a recyclable catalyst in aqueous medium. Mol. Divers. 2020, 24, 345–354. [Google Scholar] [CrossRef]
- Domling, A.; Wang, W.; Wang, K. Chemistry and biology of multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef]
- Constable, D.J.C.; Dunn, P.J.; Hayler, J.D.; Humphrey, G.R.; Leazer, J.L.; Linderman, R.J.; Lorenz, K.; Manley, J.; Pearlman, B.A.; Wells, A.; et al. Key green chemistry research areas-a perspective from pharmaceutical manufacturers. Green Chem. 2007, 9, 411–420. [Google Scholar] [CrossRef]
- Kreuder, A.D.; Knight, T.H.; Whitford, J.; Ponnusamy, E.; Miller, P.; Jesse, N.; Rodenborn, R.; Sayag, S.; Gebel, M.; Aped, I.; et al. A method for assessing greener alternatives between chemical products following the 12 principles of green chemistry. ACS Sustain. Chem. Eng. 2017, 5, 2927–2935. [Google Scholar] [CrossRef]
- Kerru, N.; Maddila, S.; Jonnalagadda, S.B. Design of carbon-carbon and carbon-heteroatom bond formation reactions under green conditions. Curr. Org. Chem. 2019, 23, 3156–3192. [Google Scholar] [CrossRef]
- Clarke, C.J.; Tu, W.C.; Levers, O.; Brohl, A.; Hallett, J.P. Green and sustainable solvents in chemical processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green solvents for sustainable organic synthesis: State of theart. Green Chem. 2005, 7, 267–278. [Google Scholar] [CrossRef]
- Taylor, A.P.; Robinson, R.P.; Fobian, Y.M.; Blakemore, D.C.; Jones, L.H.; Fadeyi, O. Modern advances in heterocyclic chemistry in drug discovery. Org. Biomol. Chem. 2016, 14, 6611–6637. [Google Scholar] [CrossRef]
- Taylor, R.D.; MacCoss, M.; Lawson, A.D.G. Rings in drugs. J. Med. Chem. 2014, 57, 5845–5859. [Google Scholar] [CrossRef]
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef]
- Kerru, N.; Singh, P.; Koorbanally, N.; Raj, R.; Kumar, V. Recent advances (2015–2016) in anticancer hybrids. Eur. J. Med. Chem. 2017, 142, 179–212. [Google Scholar] [CrossRef]
- Gomtsyan, A. Heterocycles in drugs and drug discovery. Chem. Heterocycl. Compd. 2012, 48, 7–10. [Google Scholar] [CrossRef]
- Jampilek, J. Heterocycles in medicinal chemistry. Molecules 2019, 24, 3839. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.; Gangu, K.K.; Jonnalagadda, S.B. A review on recent advances in nitrogen- containing molecules and their biological applications. Molecules 2020, 25, 1909. [Google Scholar] [CrossRef]
- Kerru, N.; Singh-Pillay, A.; Awolade, P.; Singh, P. Current anti-diabetic agents and their molecular targets: A review. Eur. J. Med. Chem. 2018, 152, 436–488. [Google Scholar] [CrossRef]
- Li, M.; Zhao, B.X. Progress of the synthesis of condensed pyrazole derivatives (from 2010 to mid-2013). Eur. J. Med. Chem. 2014, 85, 311–340. [Google Scholar] [CrossRef]
- Mamaghani, M.; Nia, R.H. A review on the recent multicomponent synthesis of pyranopyrazoles. Polycycl. Arom. Comp. 2021, 41, 223–291. [Google Scholar] [CrossRef]
- Aslam, N.; White, J.M.; Zafar, A.M.; Jabeen, M.; Ghafoor, A.; Sajjid, N.; Noreen, S.; Khan, M.A. 4H-Pyrano[2,3-c]pyrazoles: A review. Arkivoc 2018, vi, 139–203. [Google Scholar] [CrossRef]
- Kumar, A.; Lohan, P.; Aneja, D.K.; Gupta, G.K.; Kaushik, D.; Prakash, O. Design, synthesis, computational and biological evaluation of some new hydrazino derivatives of DHA and pyranopyrazoles. Eur. J. Med. Chem. 2012, 50, 81–89. [Google Scholar] [CrossRef]
- Kashtoh, H.; Muhammad, M.T.; Khan, J.J.A.; Rasheed, S.; Khan, A.; Perveen, S.; Javaid, K.; Wahab, A.T.; Khan, K.M.; Choudhary, M.I. Dihydropyrano [2,3-c] pyrazole: Novel in vitro inhibitors of yeast α-glucosidase. Bioorg. Chem. 2016, 65, 61–72. [Google Scholar] [CrossRef]
- Reddy, G.M.; Kumari, A.K.; Reddy, V.H.; Garcia, J.R. Novel pyranopyrazole derivatives comprising a benzoxazole core as antimicrobial inhibitors: Design, synthesis, microbial resistance and machine aided results. Bioorg. Chem. 2020, 100, 103908. [Google Scholar] [CrossRef]
- Derabli, C.; Boualia, I.; Abdelwahab, A.B.; Boulcina, R.; Bensouici, C.; Kirsch, G.; Debache, A. A cascade synthesis, in vitro cholinesterases inhibitory activity and docking studies of novel Tacrine-pyranopyrazole derivatives. Bioorg. Med. Chem. Lett. 2018, 28, 2481–2484. [Google Scholar] [CrossRef]
- Mandha, S.R.; Siliveri, S.; Alla, M.; Bommena, V.R.; Bommineni, M.R.; Balasubramanian, S. Eco-friendly synthesis and biological evaluation of substituted pyrano[2,3-c]pyrazoles. Bioorg. Med. Chem. Lett. 2012, 22, 5272–5278. [Google Scholar] [CrossRef]
- Gorle, S.; Gangu, K.K.; Maddila, S.; Jonnalagadda, S.B. Synthesis and anticancer activity of novel pyrazolo[4′,3′:5,6]pyrano[2,3-d] pyrimidin-5(2H)-one derivatives. Chem. Data Collect. 2020, 28, 100471. [Google Scholar] [CrossRef]
- Otto, H.H. Darstellung einiger 4H-pyrano[2.3-c]Pyrazolderivate. Arch. Pharm. 1974, 307, 444–447. [Google Scholar] [CrossRef]
- Babaie, M.; Sheibani, H. Nanosized magnesium oxide as a highly effective heterogeneous base catalyst for the rapid synthesis of pyranopyrazoles via a tandem four-component reaction. Arab. J. Chem. 2011, 4, 159–162. [Google Scholar] [CrossRef]
- Alireza, H.; Mohsen, S.; Nooshin, G.; Abdolkarim, Z.; Mohammad, M.D. Silica bonded n-propyl-4-aza-1 azoniabicyclo [2.2.2]octane chloride (SB-DABCO): A highly efficient, reusable and new heterogeneous catalyst for the synthesis of 4H-benzo[b]pyran derivatives. Appl. Catal. A Gen. 2011, 402, 11–22. [Google Scholar]
- Hamid Reza, S.; Kobra, A. Mild, four-component synthesis of 6-amino-4-aryl-3-methyl-1,4-dihydropyrano[2,3-c]pyrazole-5-carbonitriles catalysed by titanium dioxide nanosized particles. Res. Chem. Intermed. 2014, 40, 661–667. [Google Scholar]
- Davood, A.; Sayeed, M.K.; Razieh, N.Y. Nano-titania-supported preyssler-type heteropolyacid: An efficient and reusable catalyst in ultrasound-promoted synthesis of 4H-chromenes and 4H-pyrano[2,3-c]pyrazoles. J. Chem. Sci. 2014, 126, 95–101. [Google Scholar]
- Paul, S.; Prdhan, K.; Ghosh, S.; De, S.K.; Das, A.R. Uncapped SnO2 quantum dot catalysed cascade assembling of four components: A rapid and green approach to the pyrano[2,3-c]pyrazole and spiro-2-oxindole derivatives. Tetrahedron 2014, 70, 6088–6099. [Google Scholar] [CrossRef]
- Borhade, A.V.; Uphade, B.K. ZnS nanoparticles as an efficient and reusable catalyst for synthesis of 4H-pyrano[2,3-c]pyrazoles. J. Iran. Chem. Soc. 2014, 12, 1107–1113. [Google Scholar] [CrossRef]
- Nasir, I.; Mosadegh, K.; Hossein, A.S.K.; Rasool, P. Tin sulfide nanoparticles supported on activated carbon as an efficient and reusable Lewis acid catalyst for three-component one-pot synthesis of 4H-pyrano[2,3-c]pyrazole derivatives. Chin. J. Catal. 2015, 36, 626–633. [Google Scholar]
- Mahnaz, Z.; Mohammad, A.A. SBA-Pr-NH2 catalysed preparation of pyrano[2,3-c] pyrazolesunder solvent-free conditions. Rev. Roum. Chim. 2018, 63, 31–37. [Google Scholar]
- Mallappa, B.; Kalegowda, S. Four component synthesis of highly functionalised pyrano[2,3-c]pyrazoles from benzyl halides. Syn. Commun. 2018, 48, 146–154. [Google Scholar]
- Patel, K.G.; Misra, N.M.; Venkariya, R.H.; Shettigar, R.R. One-pot multicomponent synthesis in aqueous medium of 1,4-dihydropyrano[2,3-c]pyrazole-5-carbonitrile and derivatives using a green and reusable nano-SiO2 catalyst from agricultural waste. Res. Chem. Intermed. 2018, 44, 289–304. [Google Scholar] [CrossRef]
- Salehi, N.; Mirjalili, B.B.F. Green Synthesis of Pyrano[2,3-c]pyrazoles and spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles] using nano-silica supported 1,4- Diazabicyclo[2.2.2]octane as a novel catalyst. Org. Prep. Proced. Int. 2018, 50, 578–587. [Google Scholar] [CrossRef]
- Mohammad, A.G.; Boshra, M.E.; Mohammad, H.A. Green synthesis of spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles] using Fe3O4@ l-arginine as a robust and reusable catalyst. BMC Chem. 2019, 13, 119. [Google Scholar]
- Robabeh, S.M.; Mohammad, A.G.; Mohammad, R.Z.M. Green fabrication of cobalt NPs using aqueous extract of antioxidant rich Zingiber and their catalytic applications for the synthesis of pyrano[2,3-c]pyrazoles. Comb. Chem. High Throughput Screen. 2019, 22, 18–26. [Google Scholar]
- Tabassum, S.; Devi, K.R.; Govindaraju, S. An insight into the superior performance of ZnO@PEG nanocatalyst for the synthesis of 1,4-dihydropyrano[2,3-c]pyrazoles under ultrasound. Mater. Today Proc. 2021, 45, 3898–3903. [Google Scholar] [CrossRef]
- Masoud, K.A.; Davood, A.; Hamid, R.E.Z. Sulfonic acid-functionalised Fe3O4-supported magnetised graphene oxide quantum dots: A novel organic-inorganic nanocomposite as an efficient and recyclable nanocatalyst for the synthesis of dihydropyrano[2,3-c]pyrazole and 4H-chromene derivatives. Appl. Organomet. Chem. 2020, 34, e6004. [Google Scholar]
- Homayoun, F.N.; Nourallah, H.; Malek, T.M. Synthesis and characterisation of Fe3O4@THAM-SO3H as a highly reusable nanocatalyst and its application for the synthesis of dihydropyrano[2,3-c]pyrazole derivatives. Appl. Organometal. Chem. 2020, 34, e5472. [Google Scholar]
- Maddila, S.; Gorle, S.; Shabalala, S.; Oyetade, O.; Maddila, S.N.; Palakondu, L.; Jonnalagadda, S.B. Ultrasound mediated green synthesis of pyrano[2,3-c]pyrazoles by using Mn doped ZrO2. Arab. J. Chem. 2019, 12, 671–679. [Google Scholar] [CrossRef]
- Heravia, M.M.; Ghodsa, A.; Derikvanda, F.; Bakhtiaria, K.; Bamoharramb, F.F. H14[NaP5W30O110] Catalysed one-pot three-component synthesis of dihydropyrano[2,3-c]pyrazole and pyrano[2,3-d]pyrimidine derivatives. J. Iran. Chem. Soc. 2020, 7, 615–620. [Google Scholar] [CrossRef]
- Pradhan, K.; Paul, S.; Das, A.R. Magnetically retrievable nano crystalline CuFe2O4 catalysed multicomponent reaction: A facile and efficient synthesis of functionalised dihydropyrano[2,3-c]pyrazole, pyrano[3,2-c]coumarin and 4H-chromene derivatives in aqueous media. Catal. Sci. Technol. 2014, 4, 822–831. [Google Scholar] [CrossRef]
- Mohammad, A.Z.; Roya, A.N.; Saeed, B.; Vahid, K.; Saeid, A. Applications of a novel nano magnetic catalyst in the synthesis of 1,8-dioxo-octahydroxanthene and dihydropyrano[2][2,3-c]pyrazole derivatives. J. Mol. Catal. A Chem. 2016, 418–419, 54–67. [Google Scholar]
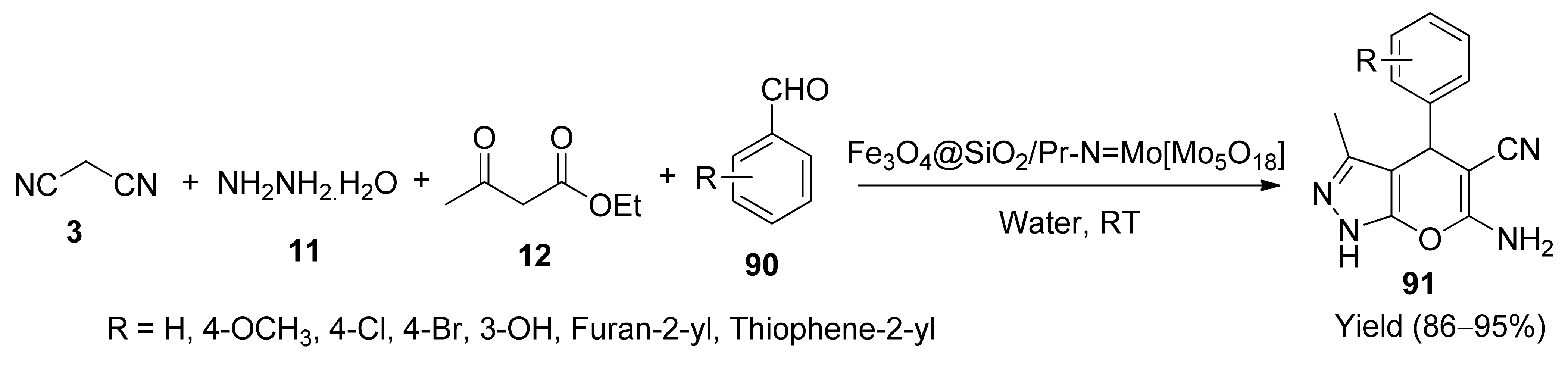
- Maddila, S.N.; Maddila, S.; van Zyl, W.E.; Jonnalagadda, S.B. CeO2/ZrO2 as green catalyst for one-pot synthesis of new pyrano[2,3-c]-pyrazoles. Res. Chem. Intermed. 2017, 43, 4313–4325. [Google Scholar] [CrossRef]
- Maryam, F.; Fatemeh, N.S.; Nourallah, H.; Malek, T.M.; Mohammad, S.H.; Sahar, M. Ag/TiO2 nano-thin films as robust heterogeneous catalyst for one-pot, multicomponent synthesis of bis (pyrazol-5-ol) and dihydropyrano[2,3-c]pyrazole analogs. J. Saudi Chem. Soc. 2017, 21, 998–1006. [Google Scholar]
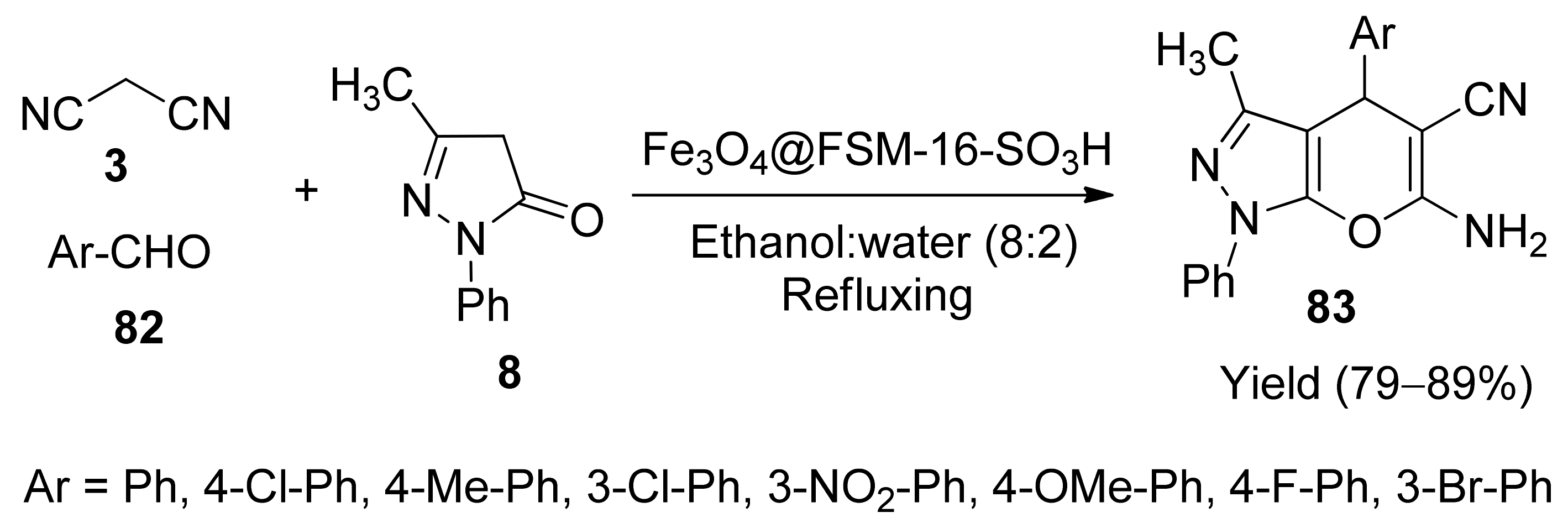
- Somayeh, H.-U.; Mohammad, A.-A.; Reza, R.-K. Fe3O4@FSM-16-SO3H as a new magnetically recyclable nanostructured catalyst: Synthesis, characterisation and catalytic application for the synthesis of pyrano[2,3-c]pyrazoles. Iran. J. Catal. 2018, 8, 311–323. [Google Scholar]
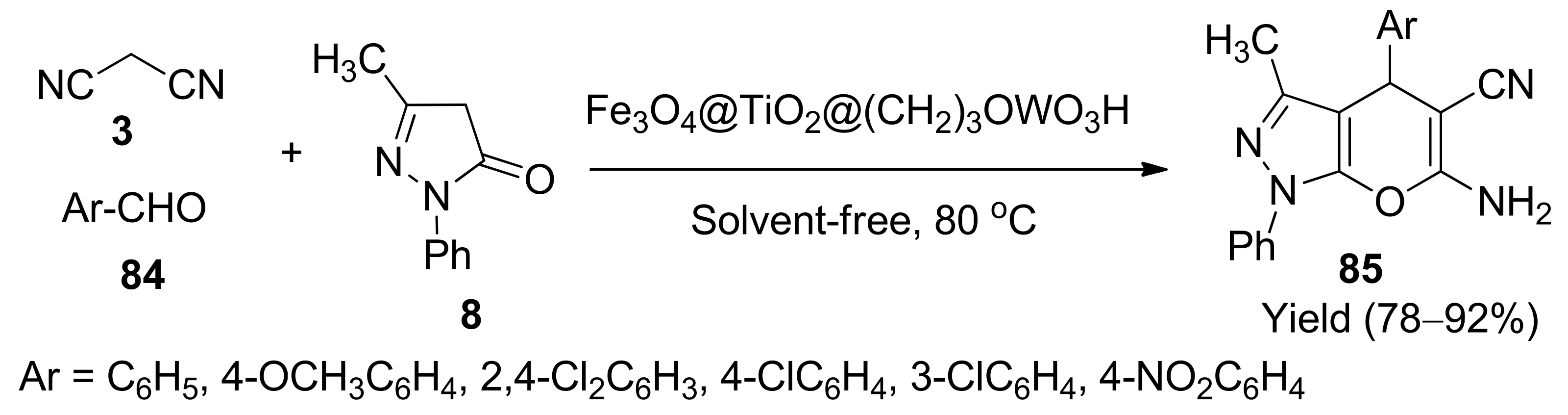
- Gholtash, J.E.; Farahi, M. Tungstic acid-functionalised Fe3O4@TiO2: Preparation, characterisation and its application for the synthesis of pyrano[2,3-c]pyrazole derivatives as a reusable magnetic nanocatalyst. RSC Adv. 2018, 8, 40962–40967. [Google Scholar] [CrossRef]
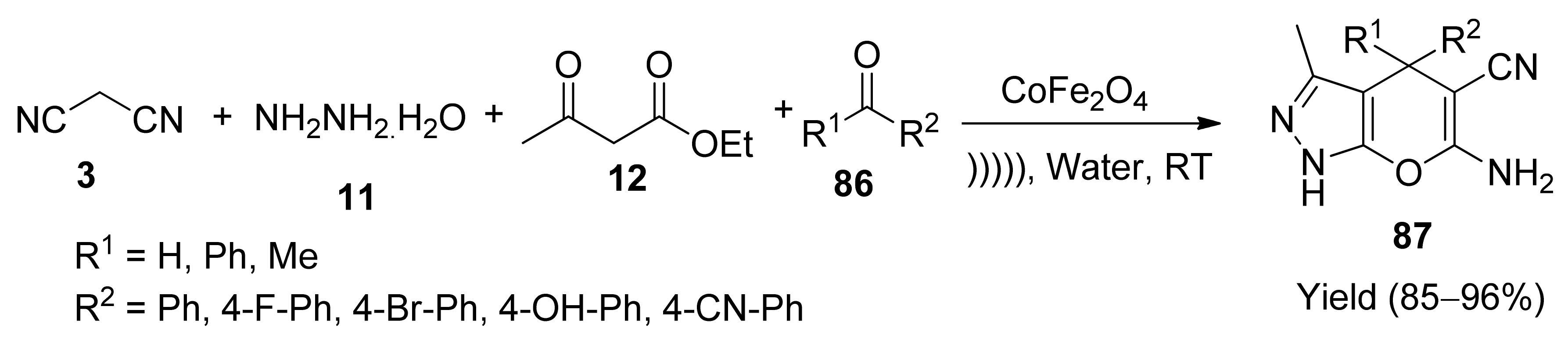
- Mishra, M.; Nizam, A.; Jomon, K.J.; Tadaparthi, K. A new facile ultrasound-assisted magnetic nano-[CoFe2O4]-catalysed one-pot synthesis of pyrano[2,3-c]pyrazoles. Rus. J. Org. Chem. 2019, 55, 1925–1928. [Google Scholar] [CrossRef]
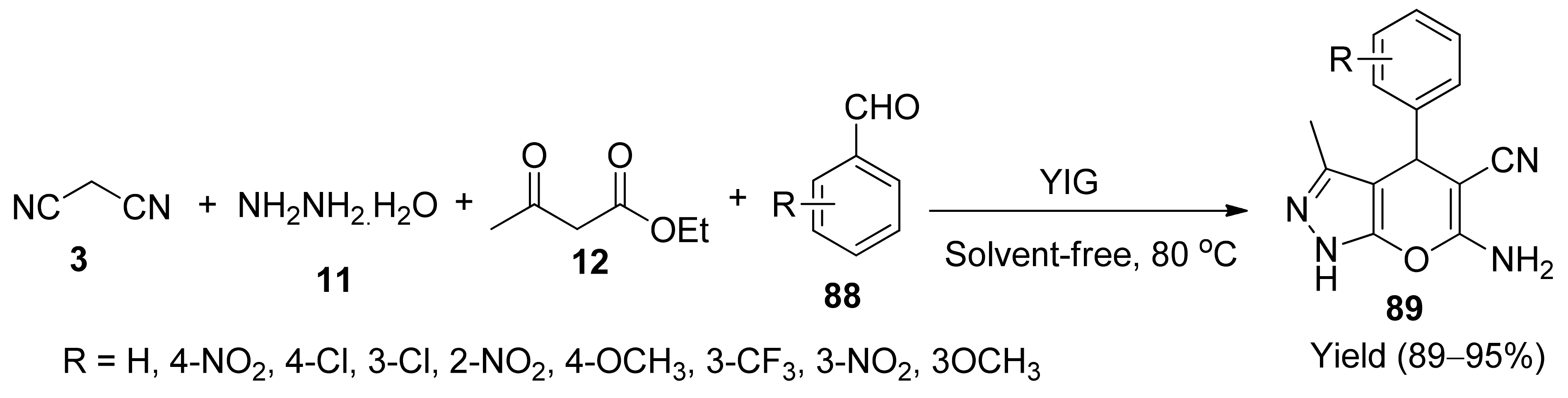
- Sedighini, E.; Badri, R.; Kiasat, A.R. Application of yttrium iron garnet as a powerful and recyclable nanocatalyst for one-pot synthesis of pyrano[2,3-c]pyrazole derivatives under solvent-free conditions. Russ. J. Org. Chem. 2019, 55, 1755–1763. [Google Scholar] [CrossRef]
- Maryam, N.; Ali, Z.; Dawood, E. Core-shell structured magnetic silica supported propylamine/molybdate complex: An efficient and magnetically recoverable nanocatalyst. New J. Chem. 2019, 43, 12283–12291. [Google Scholar]
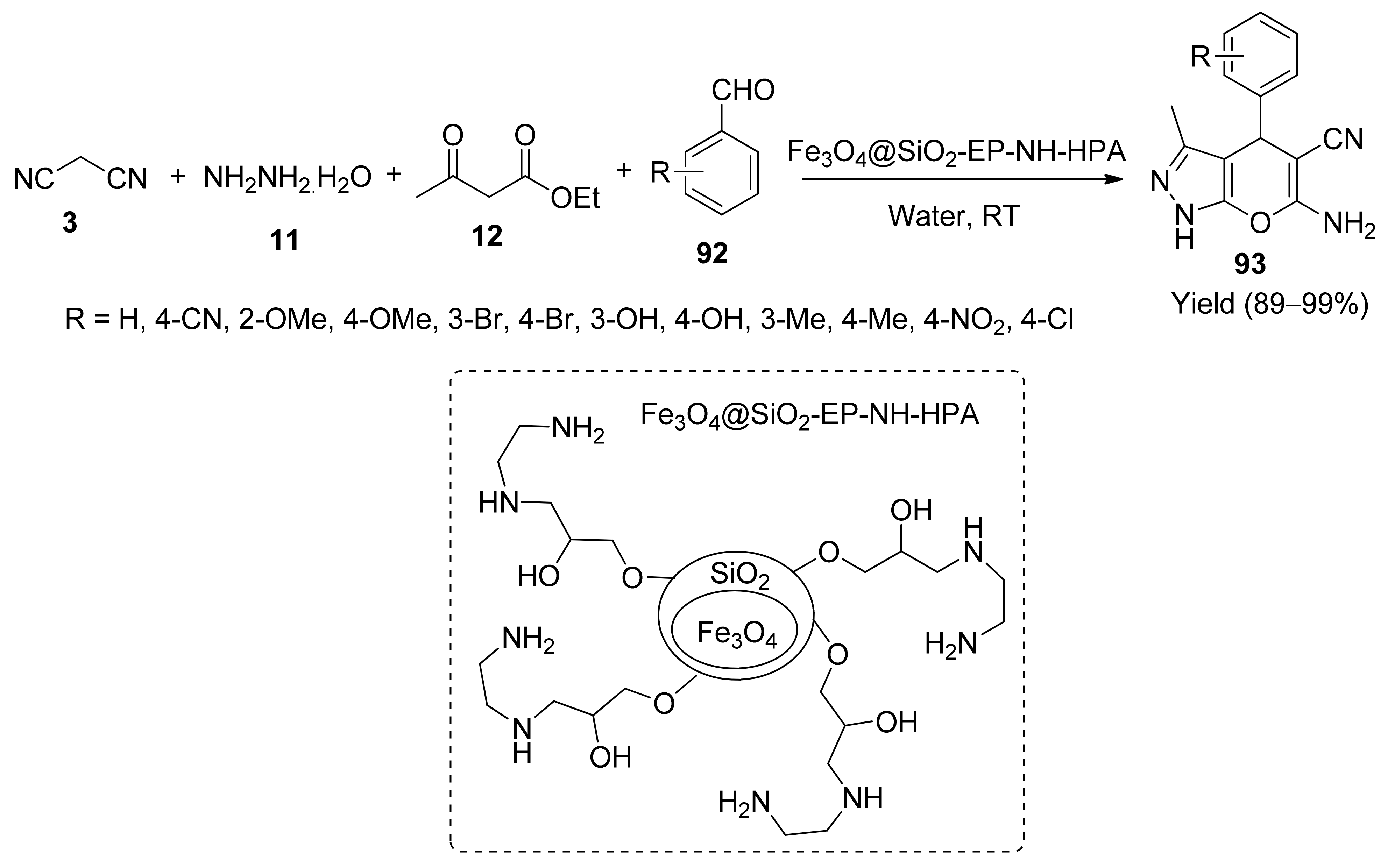
- Nina, H.M.; Mostafa, G. Nano silica extracted from horsetail plant as a natural silica support for the synthesis of H3PW12O40 immobilised on aminated magnetic nanoparticles (Fe3O4@ SiO2-EP-NH-HPA): A novel and efficient heterogeneous nanocatalyst for the green one-pot synthesis of pyrano[2,3-c]pyrazole derivatives. Res. Chem. Intermed. 2020, 46, 3037–3066. [Google Scholar]
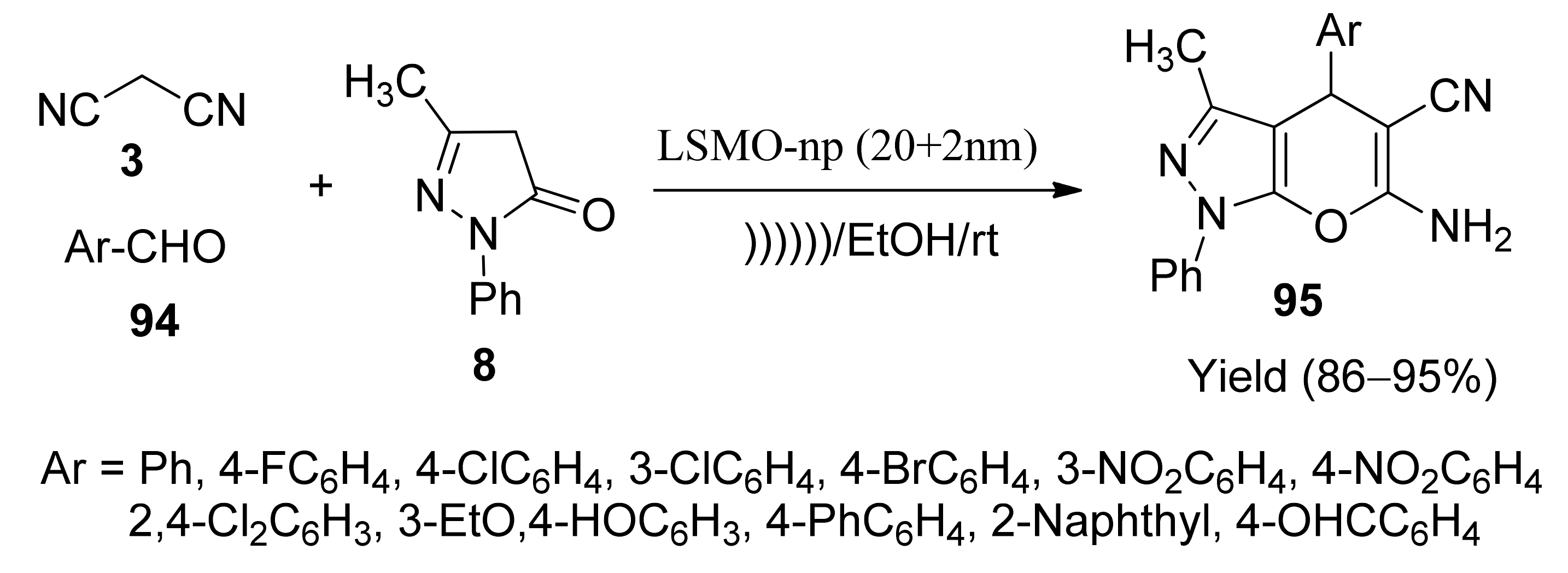
- Ali, A.; Razieh, N.-Y.; Mohammad, A.K.; Davood, A. Magnetic La0.7Sr0.3MnO3 nanoparticles: Recyclable and efficient catalyst for ultrasound-accelarated synthesis of 4H-chromenes, and 4H-pyrano[2,3-c]pyrazoles. J. Iran. Chem. Soc. 2013, 10, 439–446. [Google Scholar]
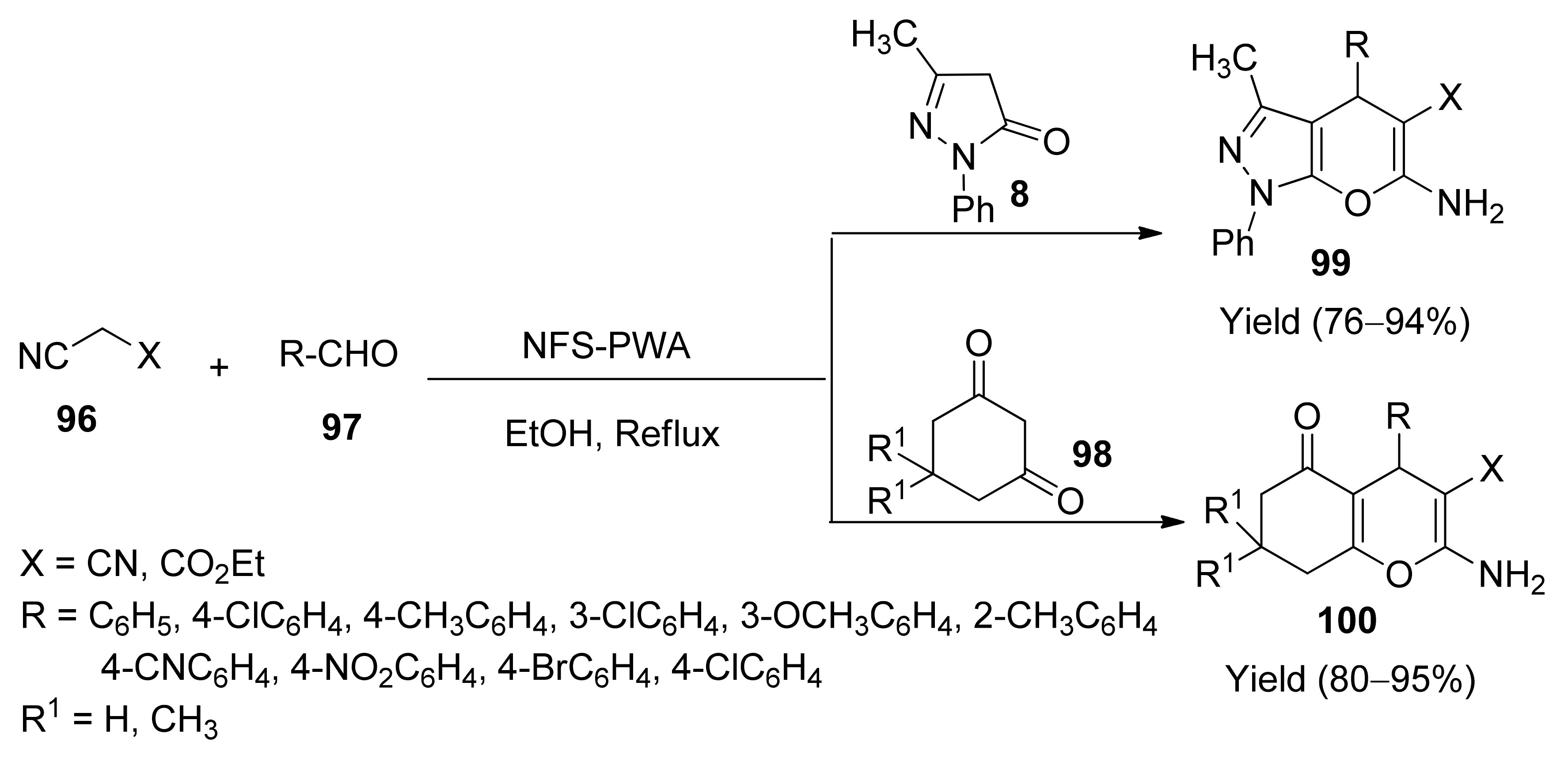
- Behrooz, M.; Hossein, E.; Mohammad, B.; Negar, N.; Amir, K.; Samaneh, S.A.; Omid, P. Silica-coated magnetic NiFe2O nanoparticlessupported H3PW12O40; synthesis, preparation, and application as an efficient, magnetic, green catalyst for one-pot synthesis of tetrahydrobenzo[b]pyran and pyrano[2,3-c]pyrazole derivatives. Res. Chem. Intermed. 2015, 42, 3071–3093. [Google Scholar]
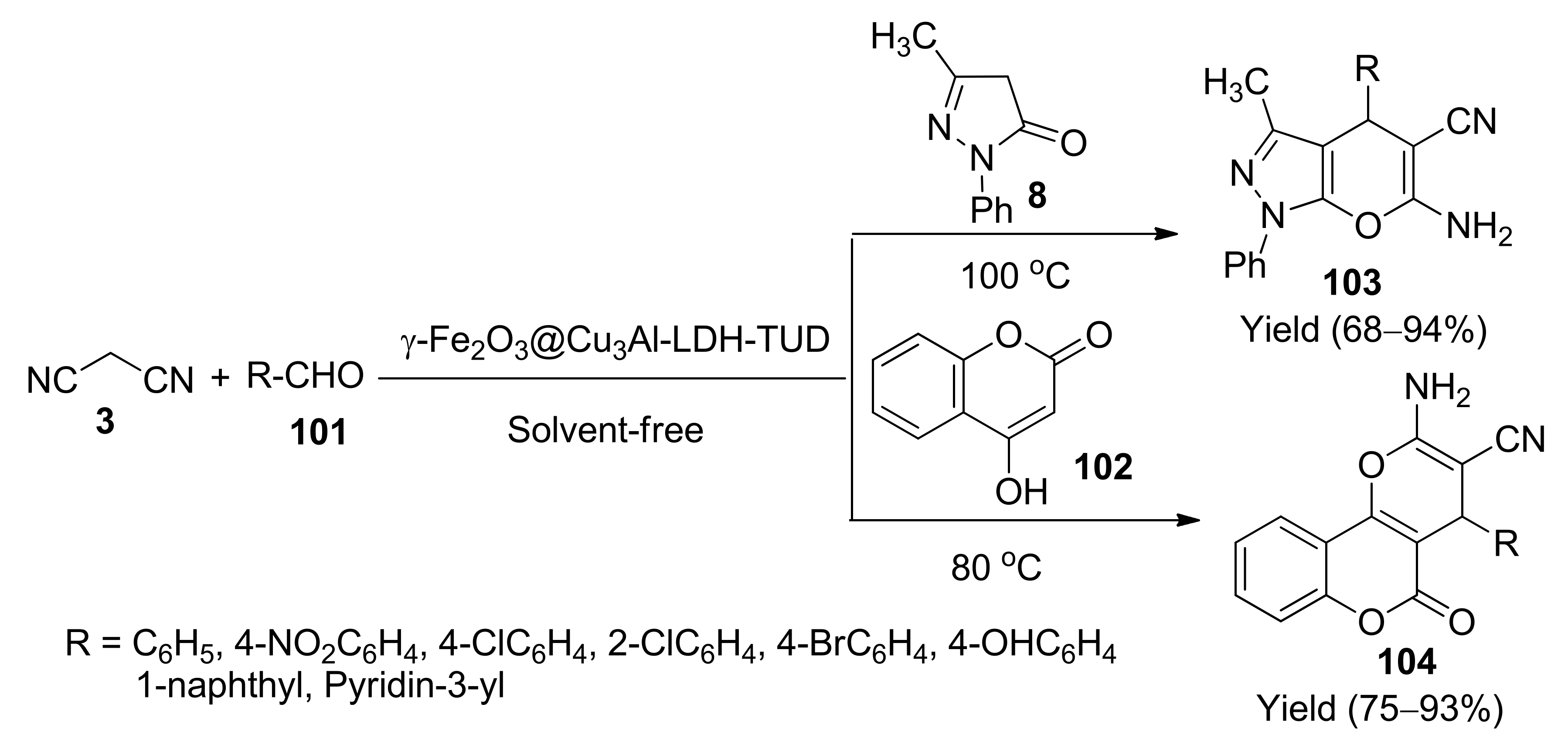
- Davood, A.; Mehrdad, T.; Masoumeh, G. γ-Fe2O3@Cu3Al-LDH-TUD as a new amphoteric, highly efficient and recyclable heterogeneous catalyst for the solvent-free synthesis of dihydropyrano[3,2-c]pyrazoles and dihydropyrano [3,2-c]chromens. Appl. Organometal. Chem. 2018, 32, e4293. [Google Scholar]
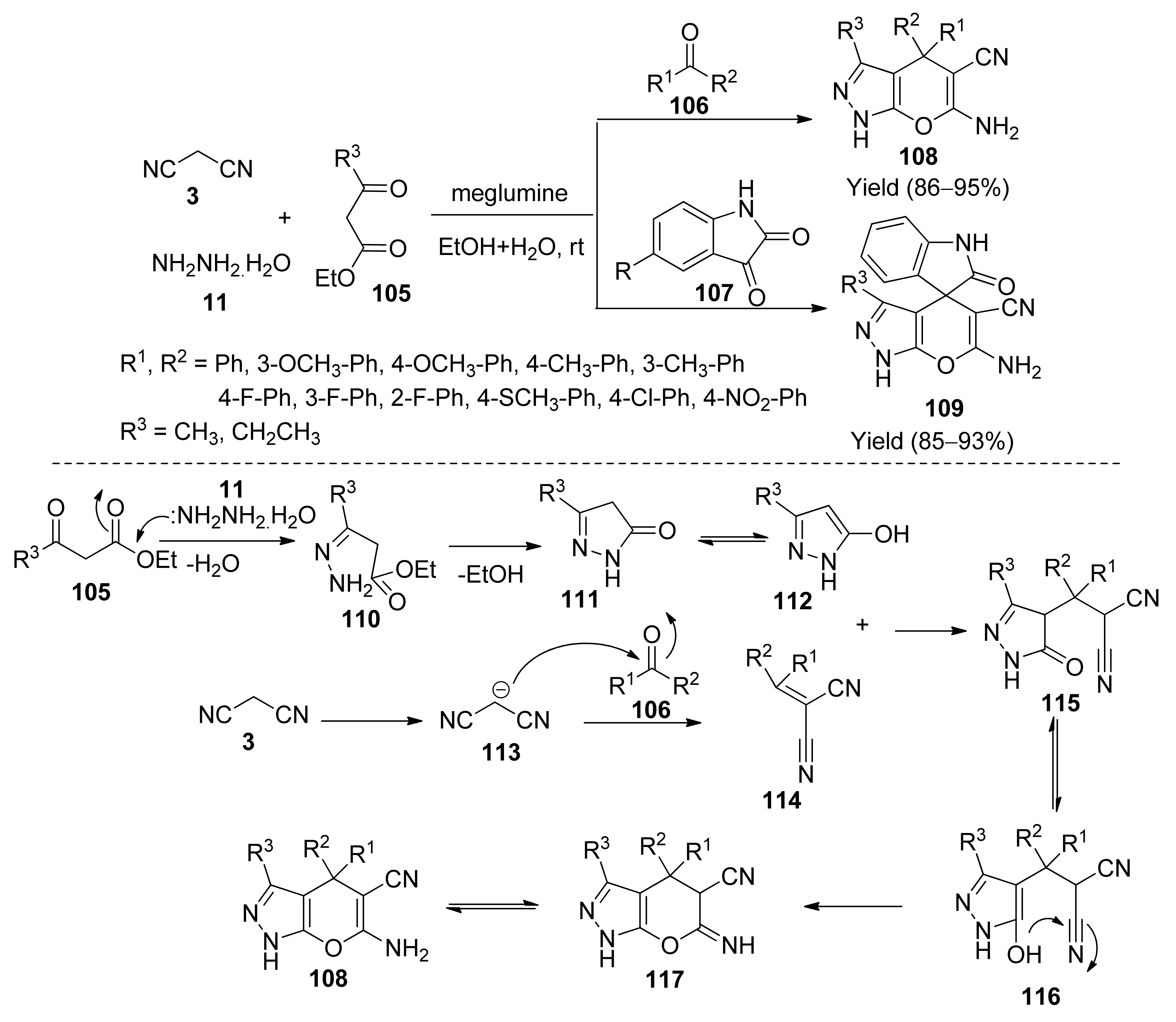
- Rui-Yun, G.; Zhi-Min, A.; Li-Ping, M.; Shu-Tao, Y.; Hong-Xia, L.; Shu-Xia, W.; Zhan-Hui, Z. Meglumine promoted one-pot, four-component synthesis of pyranopyrazole derivatives. Tetrahedron 2013, 69, 9931–9938. [Google Scholar]
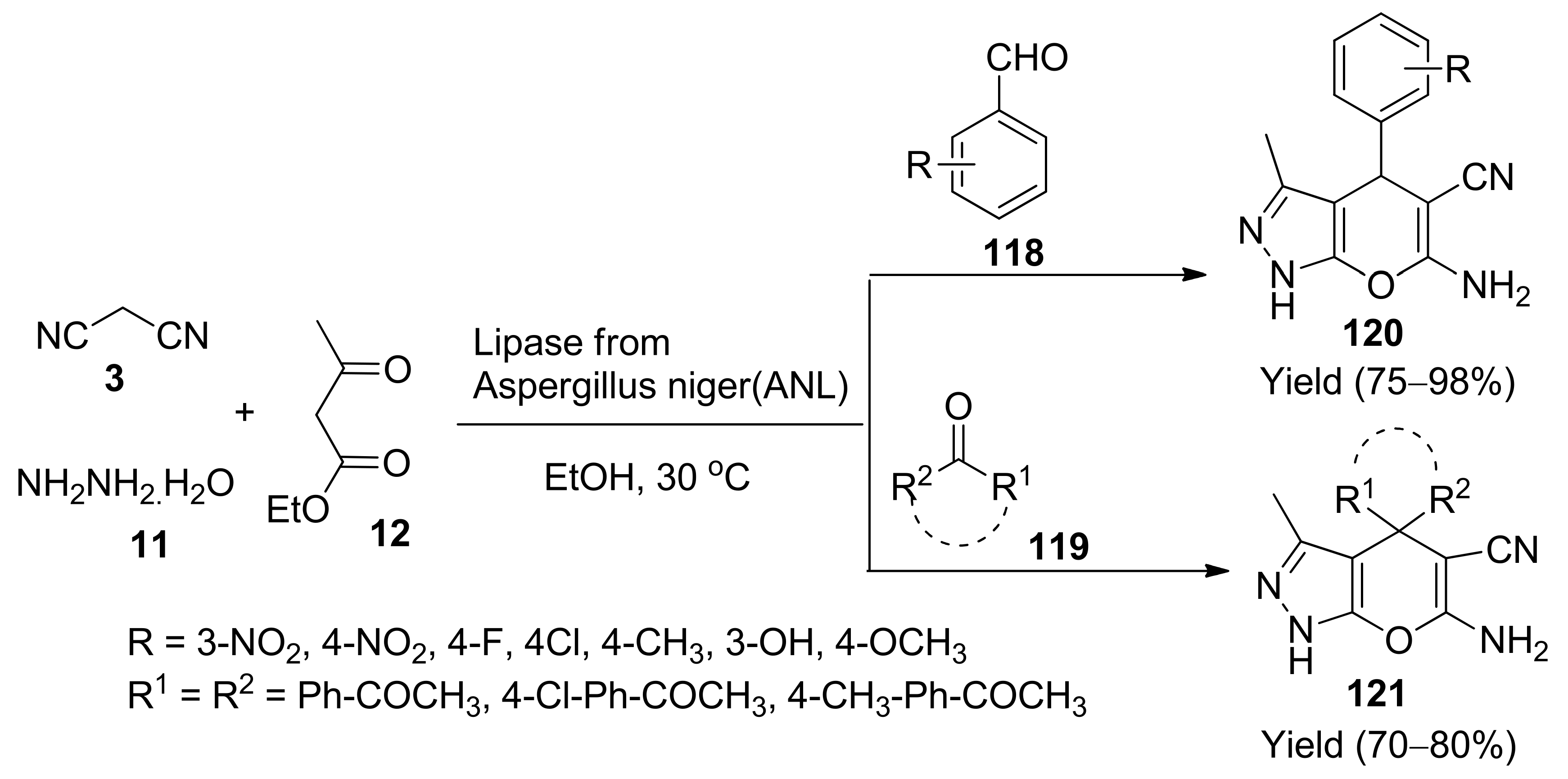
- Bora, P.; Bihani, M.; Bez, G. Multicomponent synthesis of dihydropyrano[2,3-c]pyrazoles catalysed by lipase from aspergillus niger. J. Mol. Catal. B Enzym. 2013, 92, 24–33. [Google Scholar] [CrossRef]
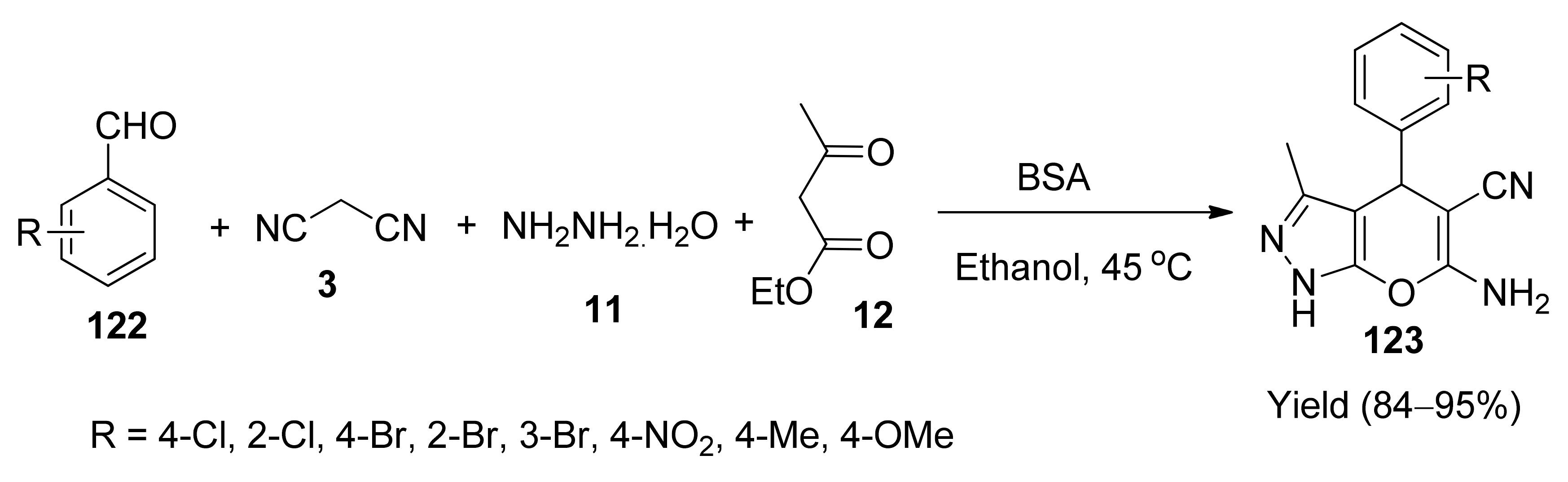
- Xingtian, H.; Zhipeng, L.; Dongyang, W.; Yiqun, L. Bovine serum albumin: An efficient and green biocatalyst for the one-pot four-component synthesis of pyrano[2,3-c]pyrazoles. Chin. J. Catal. 2016, 37, 1461–1467. [Google Scholar]
- Nazari, S.; Keshavarz, M. Amberlite-supported L-Prolinate: A novel heterogeneous organocatalyst for the three-component synthesis of 4H-Pyrano[2,3-c]pyrazole derivatives. Russ. J. Gen. Chem. 2017, 87, 539–545. [Google Scholar] [CrossRef]
- Safari, J.; Ahmadzadeh, M. Zwitterionic sulfamic acid functionalised nanoclay: A novel nanocatalyst for the synthesis of dihydropyrano[2,3-c]pyrazoles and spiro[indoline-3,4´-pyrano[2,3-c]pyrazole] derivatives. J. Taiwan Institu. Chem. Eng. 2017, 74, 14–24. [Google Scholar] [CrossRef]
- Jutika, D.; Subarna, J.K.; Dibakar, C.D. A quick micelle-catalysed one-pot synthesis of spiro [indoline-3,4’-pyrano[2, 3-c]pyrazoles] in water at room temperature. Chem. Sel. 2018, 3, 1512–1516. [Google Scholar]
- Pegah, F.; Manouchehr, M.; Nosrat, O.M.; Khalil, T. A green and practical method for the synthesis of novel pyrano[2,3-c]pyrazoles and bis-pyrano[2,3-c]pyrazoles using sulfonic acid-functionalised ionic liquid. J. Iran. Chem. Soc. 2018, 15, 11–16. [Google Scholar]
- Shinde, S.K.; Patil, M.U.; Damate, S.A.; Patil, S.S. Synergetic effects of naturally sourced metal oxides in organic synthesis: A greener approach for the synthesis of pyrano[2,3-c]pyrazoles and pyrazolyl-4H-chromenes. Res. Chem. Intermed. 2018, 44, 1775–1795. [Google Scholar] [CrossRef]
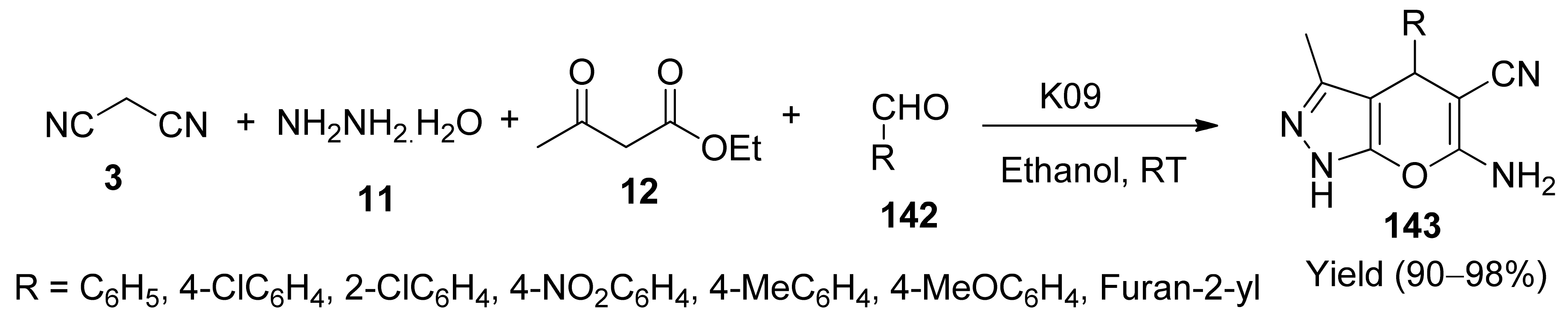
- Mejdoubi, K.E.; Sallek, B.; Digua, K.; Chaair, H.; Oudadessec, H. Natural phosphate K09 as a new reusable catalyst for the synthesis of dihydropyrano[2,3-c]pyrazole derivatives at room temperature. Kinet. Catal. 2019, 60, 536–542. [Google Scholar] [CrossRef]
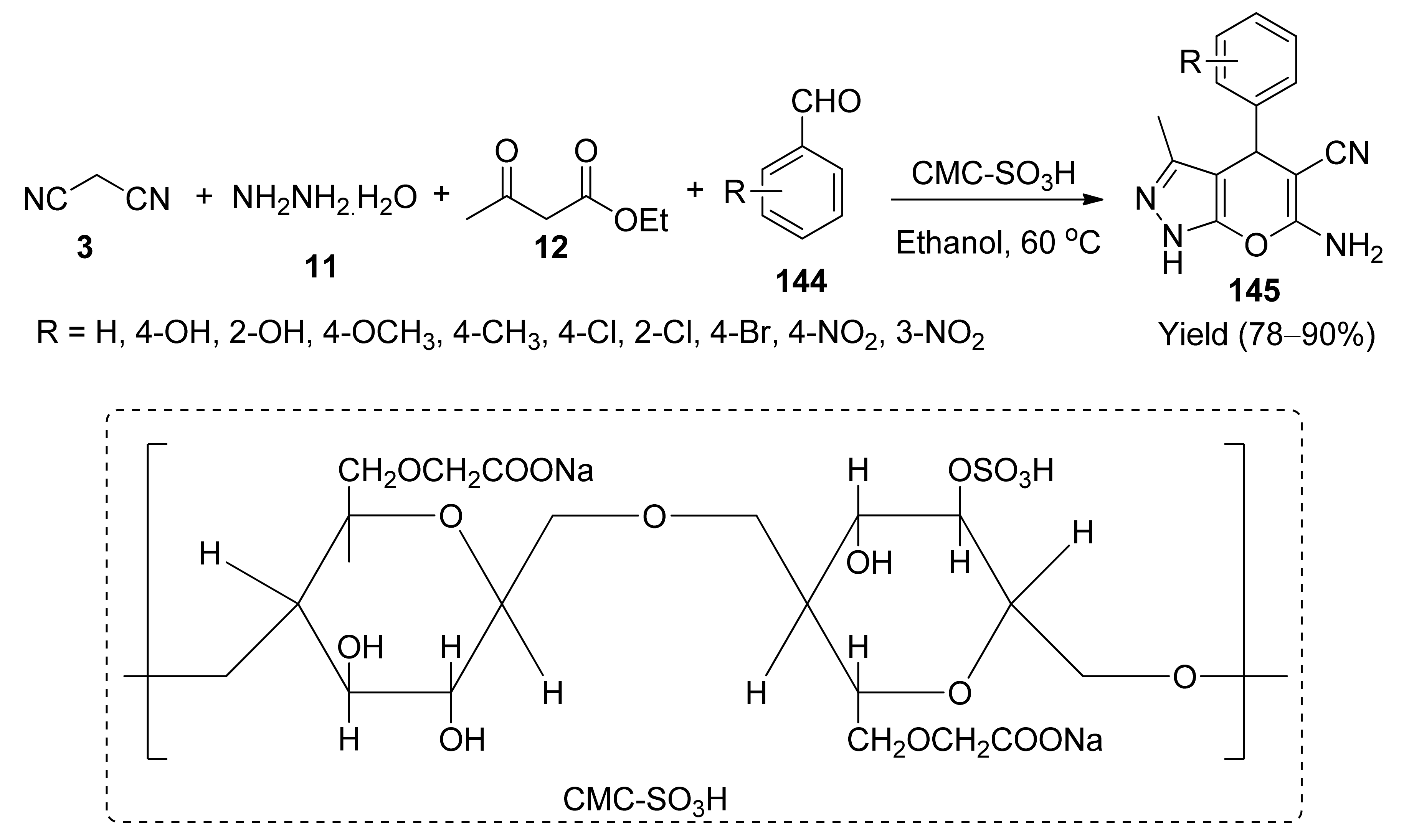
- Elham, A.; Naimi-Jamal, M.R.; Ramin, G. One-pot multicomponent synthesis of pyrano[2,3 c] pyrazole derivatives using CMCSO3H as a green catalyst. Chem. Sel. 2019, 4, 9033–9039. [Google Scholar]
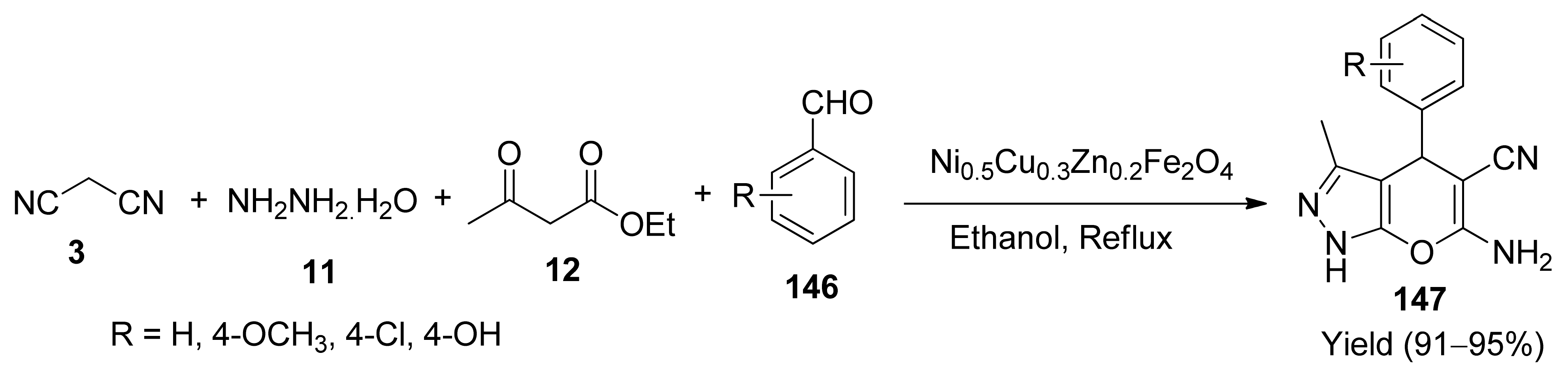
- Mandle, U.M.; Pachpinde, A.M.; Kulkarni, D.R.; Shinde, B.L. An efficient one pot multicomponent synthesis of pyrano pyrazoles using Cu2+ doped Ni-Zn nano ferrite catalyst. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
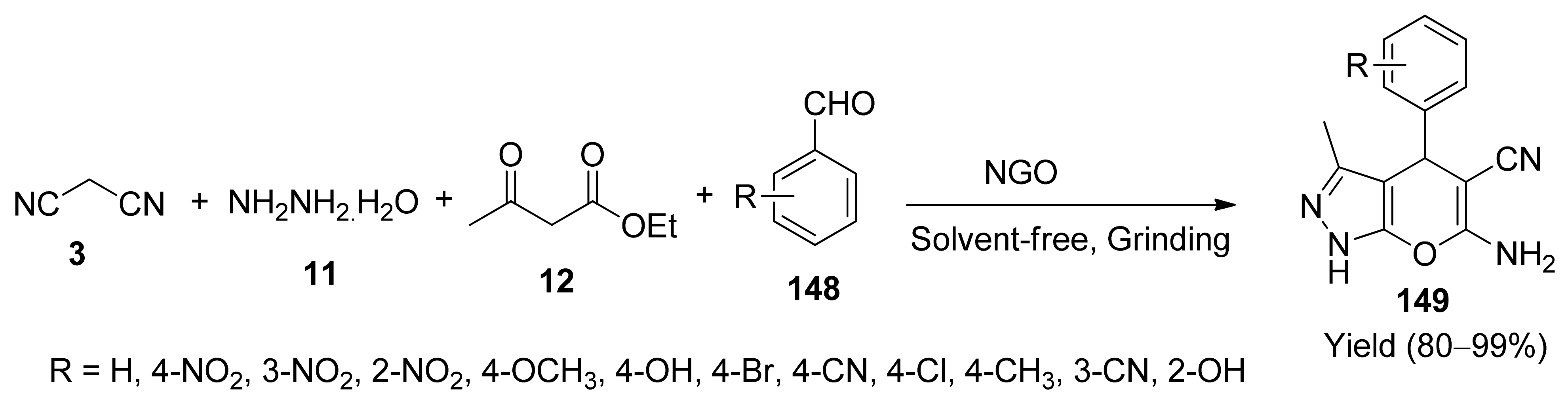
- Ganasan, N.S.; Suresh, P. Nitrogen-doped graphene oxide as a sustainable carbonaceous catalyst for greener synthesis: Benign and solvent-free synthesis of pyranopyrazoles. Chem. Sel. 2020, 5, 4988–4993. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganta, R.K.; Kerru, N.; Maddila, S.; Jonnalagadda, S.B. Advances in Pyranopyrazole Scaffolds’ Syntheses Using Sustainable Catalysts—A Review. Molecules 2021, 26, 3270. https://doi.org/10.3390/molecules26113270
Ganta RK, Kerru N, Maddila S, Jonnalagadda SB. Advances in Pyranopyrazole Scaffolds’ Syntheses Using Sustainable Catalysts—A Review. Molecules. 2021; 26(11):3270. https://doi.org/10.3390/molecules26113270
Chicago/Turabian StyleGanta, Ravi Kumar, Nagaraju Kerru, Suresh Maddila, and Sreekantha B. Jonnalagadda. 2021. "Advances in Pyranopyrazole Scaffolds’ Syntheses Using Sustainable Catalysts—A Review" Molecules 26, no. 11: 3270. https://doi.org/10.3390/molecules26113270
APA StyleGanta, R. K., Kerru, N., Maddila, S., & Jonnalagadda, S. B. (2021). Advances in Pyranopyrazole Scaffolds’ Syntheses Using Sustainable Catalysts—A Review. Molecules, 26(11), 3270. https://doi.org/10.3390/molecules26113270








