Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils
Abstract
1. Introduction
2. Results
2.1. Identification of Compounds
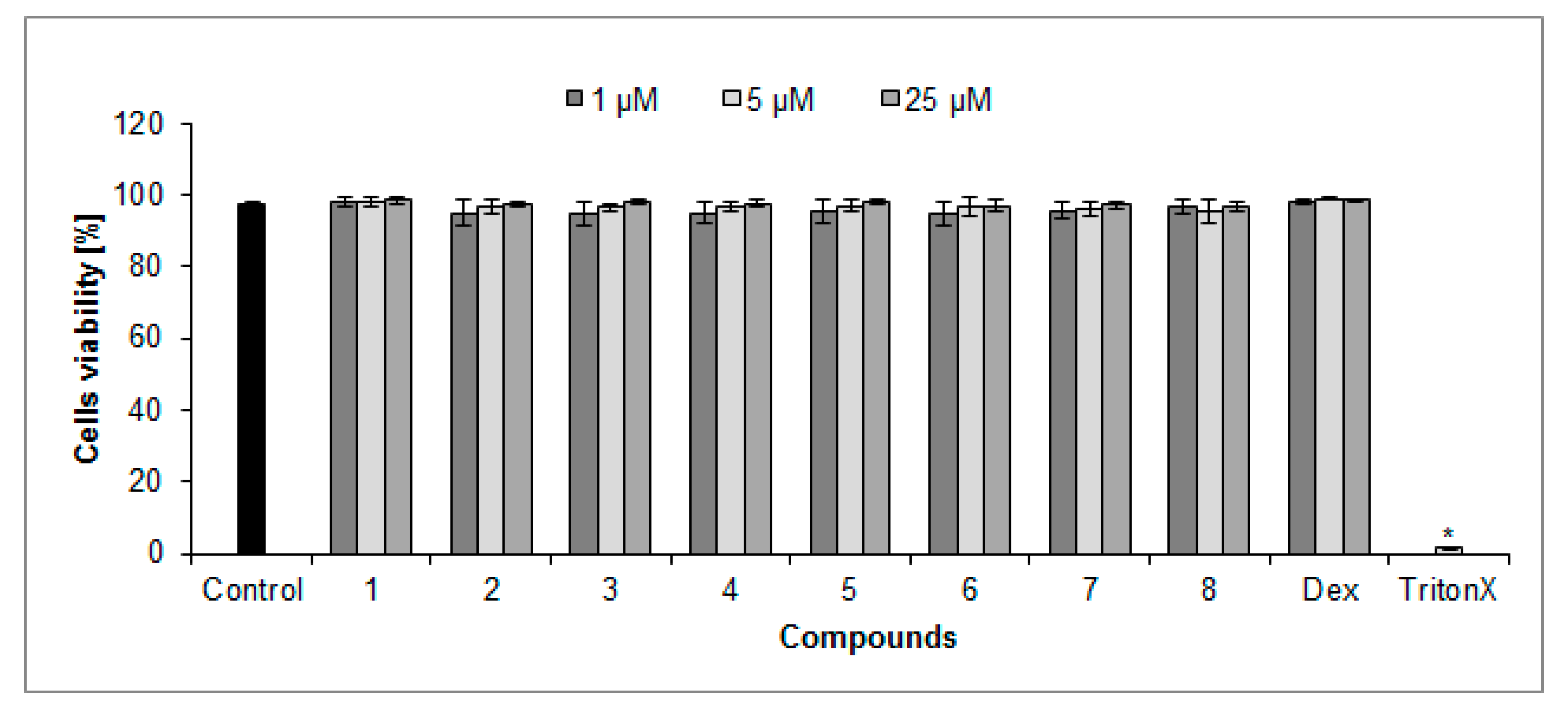
2.2. Cytotoxicity of Compounds
2.3. Inhibition of IL-8 Secretion
2.4. Inhibition of TNF-α Production
2.5. Inhibition of ROS Production
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Material
4.3. Compounds Isolation
4.4. Phytochemical Analysis by HPLC-DAD-MSn Method
4.5. PMNs Isolation
4.6. Cytotoxicity
4.7. TNF-α and IL-8 Production by PMNs
4.8. Evaluation of ROS Production by Human Neutrophils
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yalçin, F.N.; Kaya, D. Ethnobotany, pharmacology and phytochemistry of the genus Lamium (Lamiaceae). Fabad J. Pharm. Sci. 2006, 31, 43–53. [Google Scholar]
- Turner, N.; Łuczaj, Ł.; Migliorini, P.; Pieroni, A.; Dreon, A.; Sacchetti, L. Edible and tended wild plants, traditional ecological knowledge and agroecology. Crit. Rev. Plant Sci. 2011, 30, 198–225. [Google Scholar] [CrossRef]
- Łuczaj, Ł. Archival data on wild food plants used in Poland in 1948. J. Ethnobiol. Ethnomed. 2008, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.; Müller, W.; Galli, C. Local Mediterranean Food Plants and Nutraceuticals; Forum Nutr. Karger: Basel, Switzerland, 2006; Volume 59, pp. 1–17. [Google Scholar]
- Pereira, O.R.; Domingues, M.R.M.; Silva, A.M.S.; Cardoso, S.M. Phenolic constituents of Lamium album: Focus on isoscutellarein derivatives. Food Res. Int. 2012, 48, 330–335. [Google Scholar] [CrossRef]
- Yordanova, Z.P.; Zhiponova, M.K.; Iakimova, E.T.; Dimitrova, M.A.; Kapchina-Toteva, V.M. Revealing the reviving secret of the white dead nettle (Lamium album L.). Phytochem. Rev. 2014, 13, 375–389. [Google Scholar] [CrossRef]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Seigler, D.S. Phenylpropanoids. In Plant Secondary Metabolism; Springer: Boston, MA, USA, 1998; pp. 106–129. [Google Scholar]
- Kurkin, V. Phenylpropanoids from medicinal plants: Distribution, classification, structural analysis, and biological activity. Chem. Nat. Comp. 2003, 39, 123–153. [Google Scholar] [CrossRef]
- Bruneton, J. Pharmacognosy: Phytochemistry of Medicinal Plants; Lavoisier: Paris, France, 1999. [Google Scholar]
- Budzianowski, J.; Skrzypczak, L. Phenylpropanoid esters from Lamium album flowers. Phytochemistry 1995, 38, 997–1001. [Google Scholar] [CrossRef]
- Czerwińska, M.E.; Świerczewska, A.; Woźniak, M.; Kiss, A.K. Bioassay-guided isolation of iridoids and phenylpropanoids from aerial parts of Lamium album and their anti-inflammatory activity in human neutrophils. Planta Med. 2017, 83, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Damtoft, S.; Jensen, S.R.; Nielsen, B.J. Biosynthesis of iridoid glucosides in Lamium album. Phytochemistry 1991, 31, 135–137. [Google Scholar] [CrossRef]
- Alipieva, K.I.; Taskova, R.M.; Jensen, S.R.; Handjieva, N.V. Iridoid glucosides from Lamium album and Lamium maculatum (Lamiaceae). Biochem. Syst. Ecol. 2006, 34, 88–91. [Google Scholar] [CrossRef]
- Mitreski, I.; Stanoeva, J.P.; Stefova, M.; Stefkov, G.; Kulevanova, S. Polyphenols in representative Teucrium species in the flora of R. Macedonia: LC/DAD/ESI-MS(n) profile and content. Nat. Prod. Commun. 2014, 9, 175–180. [Google Scholar] [PubMed]
- Calis, I.; Basaran, A.A.; Saracoglu, I.; Sticher, O.; Ruedi, P. Phlinosides A, B and C, three phenylpropanoid glycosides from Phlomis linearis. Phytochemistry 1990, 29, 1253–1257. [Google Scholar] [CrossRef]
- Calis, I.; Basaran, A.A.; Saracoglu, I.; Sticher, O.; Ruedi, P. Phlinosides D and E, phenylpropanoid glycosides, and iridoids from Phlomis linearis. Phytochemistry 1991, 30, 3073–3075. [Google Scholar] [CrossRef]
- Yalçin, N.F.; Ersöz, T.; Bedir, E.; Şahpaz, S.; Bailleul, F.; Khan, I.A.; Dönmez, A.A.; Çalis, I. Phlinoside F, a new phenylethanoid glycoside from Phlomis angustissima. Turk. J. Chem. 2005, 29, 417–423. [Google Scholar]
- Veitch, N.C.; Regos, I.; Kite, G.C.; Treutter, D. Acylated flavonol glycosides from the forage legume, Onobrychis viciifolia (sainfoin). Phytochemistry 2011, 72, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Yuan, Q.; Liu, E.; Qi, L.; Bi, Z.; Li, P. Fragmentation study of iridoid glycosides and phenylpropanoid glycosides in Radix Scrophulariae by rapid resolution liquid chromatography with diode-array detection and electrospray ionization time-of-flight mass spectrometry. Biomed. Chromatogr. 2010, 24, 808–819. [Google Scholar] [CrossRef] [PubMed]
- Alipieva, K.; Kokubun, T.; Taskova, R.; Evstatieva, L.; Handjieva, N. LC-ESI-MS analysis of iridoid glucosides in Lamium species. Biochem. Syst. Ecol. 2007, 35, 17–22. [Google Scholar] [CrossRef]
- Quade, M.J.; Roth, J.A. A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet. Immunol. Immunopathol. 1997, 58, 239–248. [Google Scholar] [CrossRef]
- Baetta, R.; Corsini, A. Role of polymorphonuclear neutrophils in atherosclerosis: Current state and future perspectives. Atherosclerosis 2010, 210, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [PubMed]
- Palsson-McDermott, E.M.; O’Neill, L.A. Signal transduction by the lipopolysaccharide receptor, Toll-like receptor-4. Immunology 2004, 113, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Bulua, A.C.; Simon, A.; Maddipati, R.; Pelletier, M.; Park, H.; Kim, K.Y.; Sack, M.N.; Kastner, D.L.; Siegel, R.M. Mitochondrial reactive oxygen species promote production of proinflammatory cytokines and are elevated in TNFR1-associated periodic syndrome (TRAPS). J. Exp. Med. 2011, 208, 519–533. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.A.; Smith, M.F.; Sanders, M.K.; Ernst, P.B. Reactive oxygen and nitrogen species differentially regulate Toll-like receptor 4-mediated activation of NF-kappa B and interleukin-8 expression. Infect. Immun. 2004, 72, 2123–2130. [Google Scholar] [CrossRef] [PubMed]
- Bickel, M. The role of interleukin-8 in inflammation and mechanisms of regulation. J. Periodontol. 1993, 64, 456–460. [Google Scholar] [PubMed]
- Harada, A.; Sekido, N.; Akahoshi, T.; Wada, T.; Mukaida, N.; Matsushima, K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J. Leukoc. Biol. 1994, 56, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Yang, B. Phenylethanoid glycosides: Research advances in their phytochemistry, pharmacological activity and pharmacokinetics. Molecules 2016, 21, 991. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.R.; Menichini, F.; Statti, G.A.; Menichini, F. Biological and pharmacological activities of iridoids: Recent developments. Mini Rev. Med. Chem. 2008, 8, 399–420. [Google Scholar] [CrossRef] [PubMed]
- Speranza, L.; Franceschelli, S.; Pesce, M.; Reale, M.; Menghini, L.; Vinciguerra, I.; De Lutiis, M.A.; Felaco, M.; Grilli, A. Antiinflammatory effects in THP-1 cells treated with verbascoside. Phytother. Res. 2010, 24, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, M.; Obermeier, F.; Paper, D.H.; Balan, K.; Dunger, N.; Menzel, K.; Falk, W.; Schoelmerich, J.; Herfarth, H.; Rogler, G. In vivo treatment with the herbal phenylethanoid acteoside ameliorates intestinal inflammation in dextran sulphate sodium-induced colitis. Clin. Exp. Immunol. 2007, 148, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.L.; Chang, H.S.; Tseng, Y.F.; Shi, L.S. Suppression of IL-8 release by sweet olive ethanolic extract and compounds in WiDr colon adenocarcinoma cells. J. Food Sci. 2017, 82, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Serreli, G.; Incani, A.; Atzeri, A.; Angioni, A.; Campus, M.; Cauli, E.; Zurru, R.; Deiana, M. Antioxidant effect of natural table olives phenolic extract against oxidative stress and membrane damage in enterocyte-like cells. J. Food Sci. 2017, 82, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Kupeli, E.; Harput, U.S.; Varel, M.; Yesilada, E.; Saracoglu, I. Bioassay-guided isolation of iridoid glucosides with antinociceptive and anti-inflammatory activities from Veronica anagallis-aquatica L. J. Ethnopharmacol. 2005, 102, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.J.; Lim, K.H.; Jung, H.J.; Park, E.H. Anti-inflammatory evaluation of gardenia extract, geniposide and genipin. J. Ethnopharmacol. 2006, 103, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Gyurkovska, V.; Alipieva, K.; Maciuk, A.; Dimitrova, P.; Ivanovska, N.; Haas, C.; Bley, T.; Georgiev, M. Anti-inflammatory activity of Devil’s claw in vitro systems and their active constituents. Food Chem. 2011, 125, 171–178. [Google Scholar] [CrossRef]
- Mohamed, N.M.; Makboul, M.A.; Farag, S.F.; Tarawenh, A.H.; Khan, S.I.; Brooks, T.A.; Wang, Y.-H.; Roos, S.A. Iridoid and phenylpropanoid glycosides from the roots of Lantana montevidensis. Med. Chem. Res. 2017, 26, 1117–1126. [Google Scholar] [CrossRef]
- Tutin, T.; Heywood, V.; Burges, N.; Moore, D.; Valentine, D.; Walters, S.; Webb, D. Flora Europaea; Cambridge at the University Press: Cambridge, UK, 1972; Volume 3. [Google Scholar]
- Rutkowski, L. Klucz Do Oznaczania Roślin Naczyniowych Polski Niżowej, 2nd ed.; Wydawnictwo Naukowe PWN: Warszawa, Poland, 2011. [Google Scholar]
- Böyum, A. A one-stage procedure for isolation of granulocytes and lymphocytes from human blood. General sedimentation properties of white blood cells in a 1g gravity field. Scand. J. Clin. Lab. Invest. 1968, 97, 51–76. [Google Scholar]
Sample Availability: Samples of the compounds 2–7 are available from the authors. |



| Analyte No. | Compound | UV λmax [nm] | [M − H]− m/z | Fragmentary Ions |
|---|---|---|---|---|
| 1 | Apigenin 7-O-β-d-(6′′-trans-p-coumaroyl)-glucoside | 269, 317 | 577 | 307, 269 |
| 2 | Kaempferol 3-O-glucoside (astragalin) | 235sh, 265, 342 | 447 | 327, 285, 255 |
| 3 | Quercetin 3-O-[(4′′′-O-E-feruloyl)-α-rhamnopyranosyl-(1→6)]-β-glucopyranoside | 251, 333 | 785 | 623, 609, 591, 477, 301, 271 |
| 4 | Verbascoside | 245, 329 | 623 | 461, 315 |
| 5 | Lamalbid (lamiridoside) | 241 | 467 * | 421, 259 |
| 6 | Shanzhiside methyl ester | 221 | 451 * | 405, 283, 225, 179, 143 |
| 7 | Phlinoside D | 216, 328 | 769 | 637, 607, 593, 461 |
| 8 | Quercetin 3-O-α-rhamnopyranosyl-(1→2)[(4′′′′-O-E-feruloyl)-α-rhamnopyranosyl-(1→6)]-β-glucopyranoside | 251, 332 | 931 | 887, 785, 769, 755, 738, 702, 609, 562, 490, 301 |
| Quercetin | Compound 3 | Compound 8 | ||
|---|---|---|---|---|
| 1H | 13C | 1H | 13C | |
| 2 | - | 159.1 | - | 158.8 |
| 3 | - | 135.3 | - | 134.2 |
| 4 | - | 179.4 | - | 179.4 |
| 4a | - | 105.6 | - | 105.8 |
| 5 | - | 163.1 | - | 163.1 |
| 6 | 6.19 (d, J = 2.0 Hz) | 99.9 | 6.17 (d, J = 2.1 Hz) | 99.7 |
| 7 | - | 166.0 | - | 165.6 |
| 8 | 6.33 (d, J = 2.0 Hz) | 94.8 | 6.30 (d, J = 2.1 Hz) | 94.6 |
| 8a | - | 158.5 | - | 158.5 |
| 1′ | - | 123.2 | - | 123.3 |
| 2′ | 7.65 (d, J = 2.1 Hz) | 117.5 | 7.58 (d, J = 2.1 Hz) | 117.5 |
| 3′ | - | 146.0 | - | 146.1 |
| 4′ | - | 149.7 | - | 149.4 |
| 5′ | 6.92 (d, J = 8.4 Hz) | 116.1 | 6.94 (d, J = 8.2 Hz) | 116.1 |
| 6′ | 7.64 (dd, J = 8.4, 2.1 Hz) | 123.4 | 7.56 (m) | 123.5 |
| d-Glucose | ||||
| 1′′ | 5.31 (d, J = 7.7 Hz) | 103.8 | 5.78 (d, J = 7.7 Hz) | 100.1 |
| 2′′ | 3.50 (m) | 75.7 | 3.70 (m) | 79.6 |
| 3′′ | 3.45 (m) | 78.1 | 3.58 (m) | 79.1 |
| 4′′ | 3.39 (m) | 71.1 | 3.49 (m) | 71.1 |
| 5′′ | 3.40 (m) | 76.9 | 3.37 (m) | 76.6 |
| 6′′ | 3.85, 3.53 (m) | 68.0 | 3.83, 3.55 (m) | 67.6 |
| l-Rhamnose I | ||||
| 1′′′ | 4.61 (d, J = 1.6 Hz) | 102.1 | 4.61 (d, J = 1.6 Hz) | 101.7 |
| 2′′′ | 3.75 (dd, J = 3.4, 1.6 Hz) | 72.2 | 3.75 (m) | 72.2 |
| 3′′′ | 3.80 (dd, J = 9.8, 3.4 Hz) | 70.3 | 3.73 (m) | 72.2 |
| 4′′′ | 4.92 (t, J = 9.8 Hz) | 75.2 | 4.88 (t, J = 9.8 Hz) | 75.1 |
| 5′′′ | 3.66 (m) | 67.7 | 3.61 (m) | 67.6 |
| 6′′′ | 0.90 (d, J = 6.3 Hz) | 17.6 | 0.81 (d, J = 6.3 Hz) | 17.4 |
| l-Rhamnose II | ||||
| 1′′′′ | 5.24 (d, J = 1.6 Hz) | 102.3 | ||
| 2′′′′ | 3.98 (dd, J = 3.4, 1.7 Hz) | 72.3 | ||
| 3′′′′ | 3.80 (m) | 70.3 | ||
| 4′′′′ | 3.29 (m) | 74.0 | ||
| 5′′′′ | 3.95 (m) | 69.8 | ||
| 6′′′′ | 0.86 (d, J = 6.3 Hz) | 17.3 | ||
| Feruloyl (*/#) | ||||
| α | 6.26 (d, J = 15.9 Hz) | 116.3 | 6.16 (d, J = 15.9 Hz) | 116.3 |
| β | 7.56 (d, J = 15.9 Hz) | 146.8 | 7.53 (d, J = 15.9 Hz) | 146.8 |
| γ | - | 169.0 | - | 169.0 |
| 1′′′′/1′′′′′ | - | 129.9 | - | 129.0 |
| 2′′′′/2′′′′′ | 7.13 (d, J = 2.1 Hz) | 115.0 | 7.14 (d, J = 2.1 Hz) | 115.2 |
| 3′′′′/3′′′′′ | - | 147.8 | - | 147.9 |
| 4′′′′/4′′′′′ | - | 151.4 | - | 151.4 |
| 5′′′′/5′′′′′ | 6.98 (d, J = 8.4 Hz) | 112.5 | 7.00 (d, J = 8.4 Hz) | 112.6 |
| 6′′′′/6′′′′′ | 7.10 (dd, J = 8.4, 2.1 Hz) | 122.9 | 7.12 (dd, J = 8.3, 2.1 Hz) | 122.8 |
| OCH3 | 3.90 (s) | 56.3 | 3.92 (s) | 56.4 |
| Analyte | ROS Production [%] † |
|---|---|
| (−) f-MLP | 31.3 ± 2.6 |
| (+) f-MLP | 104.5 ± 8.3 # |
| 1 | 42.6 ± 5.1 * |
| 2 | 92.7 ± 6.2 |
| 3 | 16.9 ± 1.2 ** |
| 4 | 42.6 ± 4.2 * |
| 5 | 68.6 ± 5.2 * |
| 6 | 45.7 ± 1.4 * |
| 7 | 21.0 ± 3.4 ** |
| 8 | 10.0 ± 2.6 ** |
| Quercetin | 8.8 ± 1.8 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czerwińska, M.E.; Świerczewska, A.; Granica, S. Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils. Molecules 2018, 23, 2770. https://doi.org/10.3390/molecules23112770
Czerwińska ME, Świerczewska A, Granica S. Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils. Molecules. 2018; 23(11):2770. https://doi.org/10.3390/molecules23112770
Chicago/Turabian StyleCzerwińska, Monika E., Anita Świerczewska, and Sebastian Granica. 2018. "Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils" Molecules 23, no. 11: 2770. https://doi.org/10.3390/molecules23112770
APA StyleCzerwińska, M. E., Świerczewska, A., & Granica, S. (2018). Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils. Molecules, 23(11), 2770. https://doi.org/10.3390/molecules23112770







