Retro-Curcuminoids as Mimics of Dehydrozingerone and Curcumin: Synthesis, NMR, X-ray, and Cytotoxic Activity
Abstract
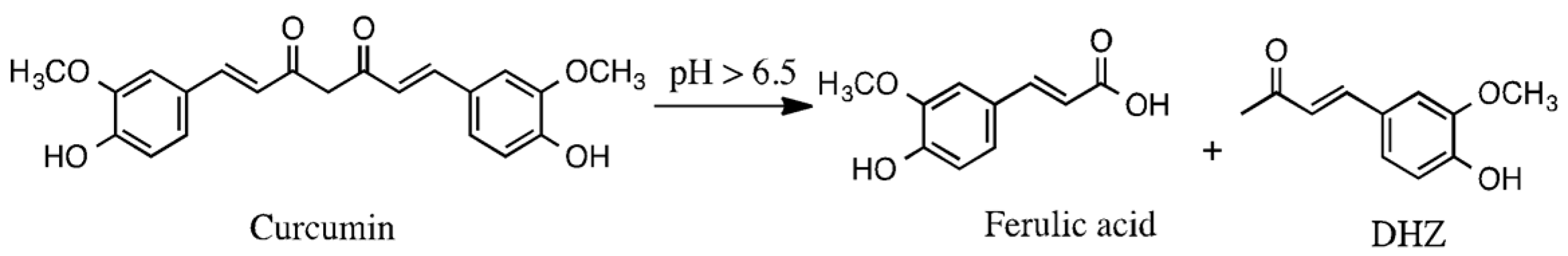
:1. Introduction
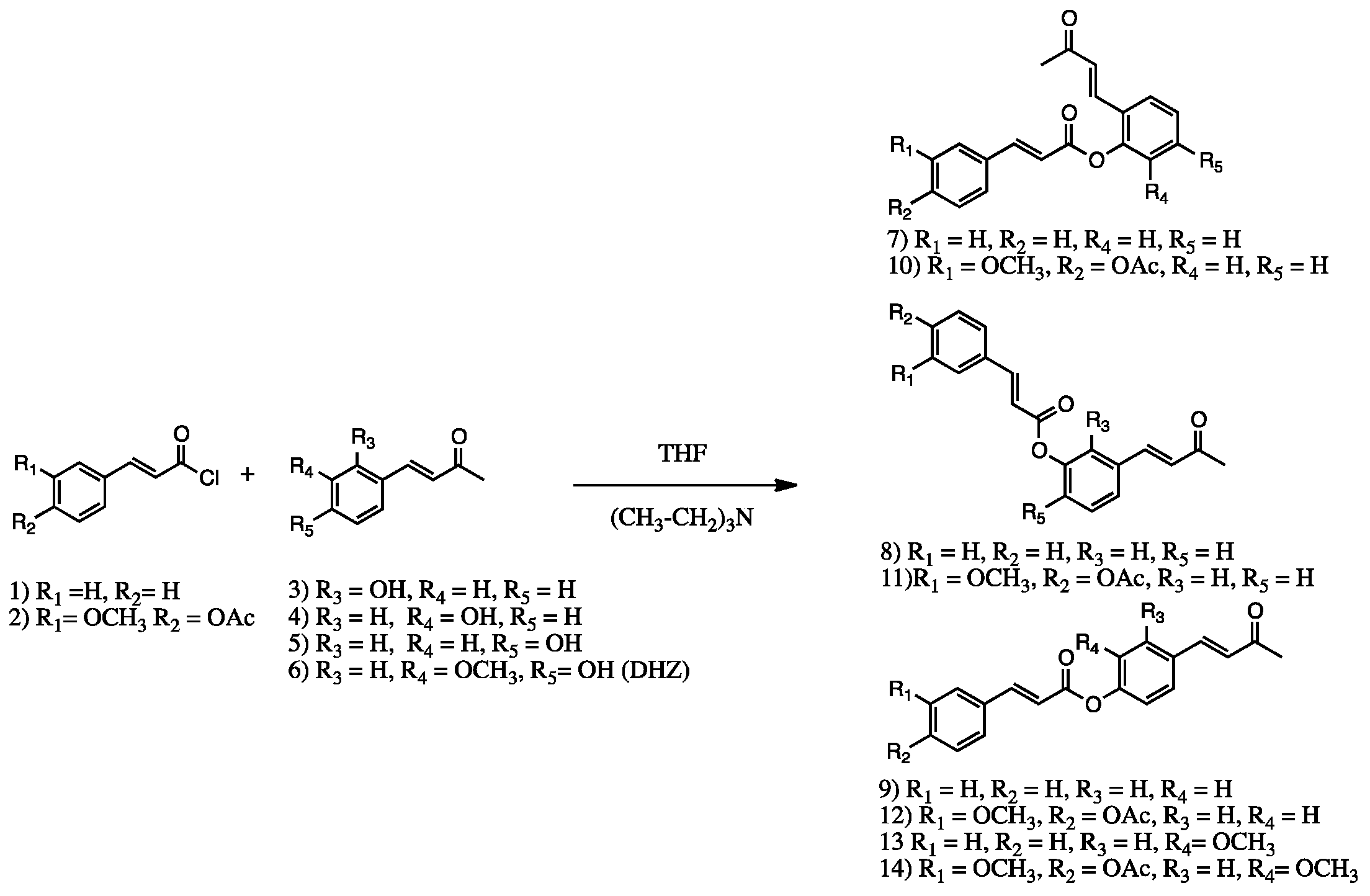
2. Results and Discussion
2.1. Structural Features
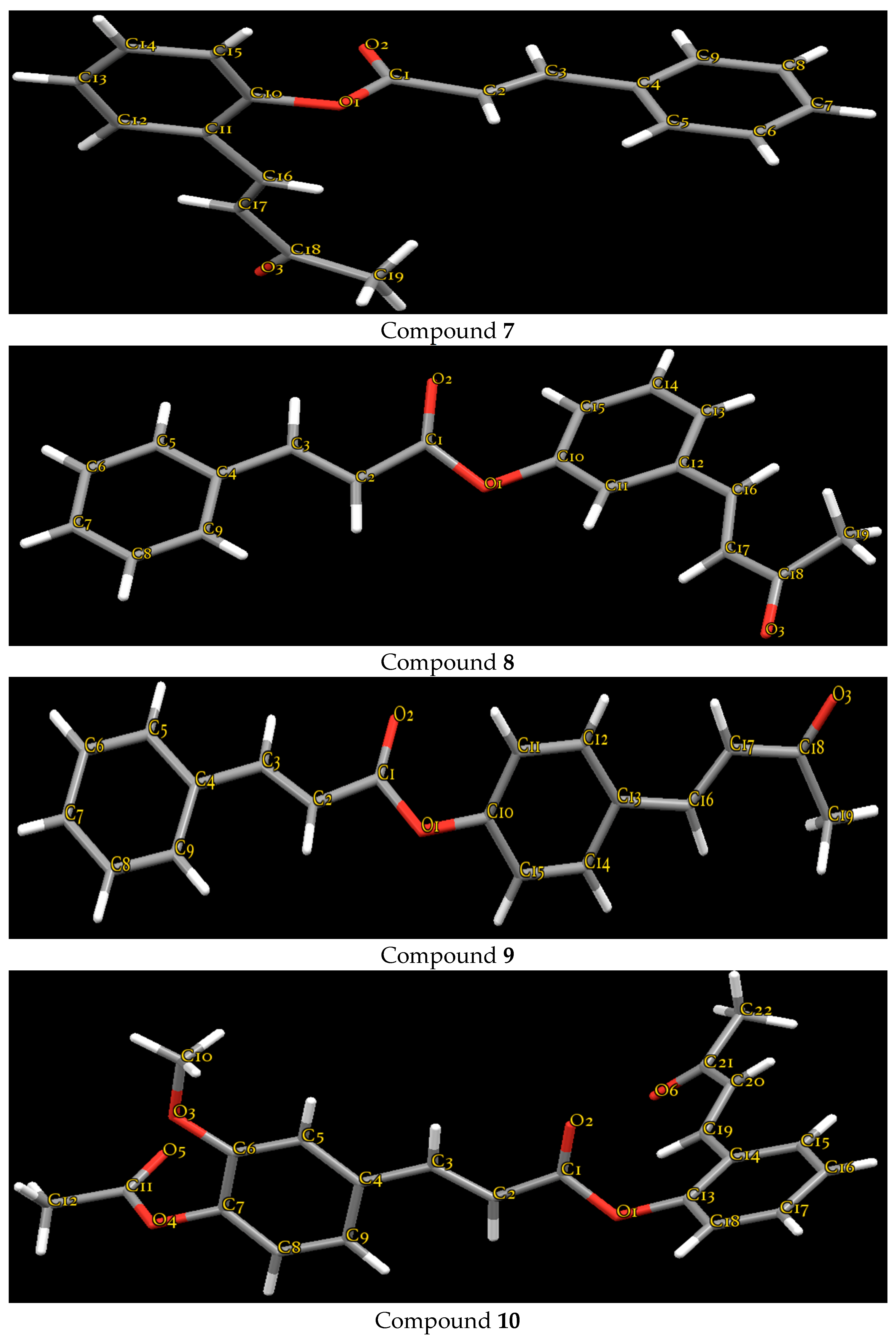
2.2. Crystallography
2.3. Cytotoxicity and Antioxidant Activity
3. Experimental Section
3.1. General Information
3.2. Physical Measurements
3.3. Synthetic Procedures
Retro-Curcuminoids 7–14
3.4. Crystallography
3.5. Cell Lines, Culture Medium, and Cytotoxicity Assay
3.6. Inhibition of Lipid Peroxidation on Rat Brain
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DHZ | Dehydrozingerone |
| TPA | 12-O-tetradecanoylphorbol-13-acetate |
| IC50 | Inhibitory Concentration 50 |
Appendix A
References
- Basnet, P.; Skalko-Basnet, N. Curcumin: An anti-inflammatory molecule from a curry spice on the path to cancer treatment. Molecules 2011, 16, 4567–4598. [Google Scholar] [CrossRef] [PubMed]
- Esatbeyoglu, T.; Huebbe, P.; Ernst, I.M.A.; Chin, D.; Wagner, A.E.; Rimbach, G. Curcumin—From molecule to biological function. Angew. Chem. Int. Ed. 2012, 51, 5308–5332. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, A.B.; Preetha, A.; Aggarwal, B.B. Curcumin inhibites proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008, 269, 199–225. [Google Scholar] [CrossRef] [PubMed]
- Ak, T.; Gülçin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 2008, 174, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Palanivelu, K. The effect of curcumin (turmeric) on Alzheimer disease: An overview. Ann. Indian Acad. Neurol. 2008, 11, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Zhang, X.; Tautiva, N.A.; Nyabera, A.N.; Owa, O.O.; Baidya, M.H.C.; Taunk, P.S.; Abdollahi, S.; Charles, S.; Gonnella, R.G.; et al. Small molecules and Alzheimer’s disease: Misfolding, metabolism and imaging. Curr. Alzheimer Res. 2016, 12, 445–461. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, Y.; Zhang, C.; Tian, X.; Ross, A.W.; Moir, R.D.; Sun, H.; Tanzi, R.E.; Moore, A.; Ran, C. Near-infrared fluorescence molecular imaging of amyloid beta species and monitoring therapy in animal models of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2015, 112, 9734–9739. [Google Scholar] [CrossRef] [PubMed]
- Tatsuzaki, J.; Nakagawa-Goto, K.; Tokuda, H.; Lee, K.H. Cancer preventive agents 10. Prenylated dehydrozingerone analogs as potent chemopreventive agents. J. Asian Nat. Prod. Res. 2010, 12, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef] [PubMed]
- Roughley, P.J.; Whiting, D.A. Experiments in the biosynthesis of curcumin. J. Chem. Soc. 1973, 2379–2388. [Google Scholar] [CrossRef]
- Sardjiman, S.S.; Reksohadiprodjo, M.S.; Hakim, L.; Van der Goot, H.; Timmerman, H. 1,5-Diphenyl-1,4-pentadiene-3-ones and cyclic analogues as antioxidative agents. Synthesis and structure-activity relationship. Eur. J. Med. Chem. 1997, 32, 625–630. [Google Scholar] [CrossRef]
- Muthusamy, G.; Balupillai, A.; Ramasamy, K.; Shanmugam, M.; Gunaseelan, S.; Mary, B.; Prasad, N.R. Ferulic acid reverses ABCB1-mediated paclitaxel resistance in MDR cell lines. Eur. J. Pharmacol. 2016, 786, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Yogosawa, S.; Yamada, S.; Yasuda, S.; Sun, Q.; Takizawa, K.; Sakai, T. Dehydrozingerone, a structural analogue of curcumin, induces cell-cycle arrest at the G2/M phase and accumulates intracellular ROS in HT-29 human colon cancer cells. J. Nat. Prod. 2012, 75, 2088–2093. [Google Scholar] [CrossRef] [PubMed]
- Hampannavar, G.A.; Karpoormath, R.; Palkar, M.B.; Shaikh, M.S. An appraisal on recent medicinal perspective of curcumin degradant: Dehydrozingerone (DZG). Bioorg. Med. Chem. 2016, 24, 501–520. [Google Scholar] [CrossRef] [PubMed]
- Ratković, Z.; Muškinja, J.; Burmudžija, A.; Ranković, B.; Kosanić, M.; Bogdanović, G.A.; Simović Marković, B.; Nikolić, A.; Arsenijević, N.; Đorđevic, S.; et al. Dehydrozingerone based 1-acetyl-5-aryl-4,5-dihydro-1H-pyrazoles: Synthesis, characterization and anticancer activity. J. Mol. Struct. 2016, 1109, 82–88. [Google Scholar] [CrossRef]
- Marchiani, A.; Mammi, S.; Siligardi, G.; Hussain, R.; Tessari, I.; Bubacco, L.; Delogu, G.; Fabbri, D.; Dettori, M.A.; Sanna, D.; et al. Small molecules interacting with α-synuclein: Antiaggregating and cytoprotective properties. Amino Acids 2013, 45, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Wei, D.; Wang, J.; Jiang, B.; Wang, M.; Xue, X.; Zhou, S.; Wu, B.; Jiang, M. Crystal structure, optical properties and biological imaging of two curcumin derivatives. Dyes Pigments 2014, 101, 312–317. [Google Scholar] [CrossRef]
- Kumar, C.S.; Jagadeesan, G.; Dhamodaran, S.; Ananth, K.; Aravindhan, S. 2,6-Dibromo-4-formylphenyl 3-phenylprop-2-enoate. Acta Cryst. 2012, E68, o2873. [Google Scholar]
- Sachitanand, M.M.; Anupam, B.; Sandip, V.J.; Mothukuri, G.K.; Hosahudya, N.G. Synthesis of α,β-unsaturated γ-amino esters with unprecedented high (E)-stereoselectivity and their conformational analysis in peptides. Org. Biomol. Chem. 2011, 9, 6566–6574. [Google Scholar]
- Nissa, M.N.; Aravindan, P.G.; Kasinath, V.; Gopalakrishnan, G.; Merazig, H.; Velmurugan, D. Crystal structures of 2-chloro cinnamoyl phenolate (I) and 3-chloro cinnamanilide (II). Cryst. Res. Technol. 2004, 39, 643–649. [Google Scholar] [CrossRef]
- Sharmila, P.; Kumar, C.S.; Ananth, K.; Narasimhanb, S.; Aravindhan, S. Diethyl 2,6-dimethyl-4-[4-(3-phenyl-acryloyloxy)phenyl]-1,4-dihydropyridine-3,5-di carboxylate hemihydrate. Acta Cryst. 2013, E69, o389. [Google Scholar]
- Momin, M.I.K.; Pawar, S.; Koorbanally, N.A.; Su, H.; Ramjugernath, D. 2-Acetylphenyl (2E)-3-(4-fluorophenyl)acrylate. Acta Cryst. 2012, E68, o3049. [Google Scholar] [CrossRef] [PubMed]
- Schultz, T.W.; Yarbrough, J.W. Trends in structure-toxicity relationships for carbonyl-containing α,β-unsaturated compounds. SAR QSAR Environ. Res. 2004, 15, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Armarego, W.L.F.; Perrin, D.D. Purification of Laboratory Chemicals, 4th ed.; Butterworth Heinemann: Oxford, UK, 1997. [Google Scholar]
- Hosoda, A.; Nomura, E.; Mizuno, K.; Taniguchi, H. Preparation of a (±)-1,6-di-O-feruloyl-myo-inositol derivative: An efficient method for introduction of ferulic acid to 1,6-vicinal hydroxyl groups of myo-inositol. J. Org. Chem. 2001, 66, 7199–7201. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.Y.; Cui, C.; Wang, Z.P.; Li, Y.Q.; Xiong, L.Z.; Wang, L.Z.; Yu, S.J.; Li, Z.M.; Zhao, W.G. Synthesis and characteristics of (Hydrogenated) ferulic acid derivatives as potential antiviral agents with insecticidal activity. Chem. Cent. J. 2013, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Vashchenko, V.; Kutulya, L.; Krivoshey, A. Simple and effective protocol for Claisen–Schmidt condensation of hindered cyclic ketones with aromatic aldehydes. Synthesis 2007, 14, 2125–2134. [Google Scholar] [CrossRef]
- CrysAlis PRO, v. 1.171.36.28; Agilent Technologies: Yarnton, UK, 2014.
- Bruker. APEX2, SAINT and SADABS, Bruker AXS Inc.: Madison, WI, USA, 2009.
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New features for the visualization and investigation of crystal structures. J. Appl. Cryst. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Nardelli, M. PARST95. An update to PARST: A system of Fortran routines for calculating molecular structure parameters from the results of crystal structure analyses. J. Appl. Cryst. 1995, 28, 659. [Google Scholar] [CrossRef]
- Monks, A.; Scudiero, D.; Skehan, P.; Shoemaker, R.; Paul, K.; Vistica, D.; Hose, C.; Langley, J.; Cronise, P.; Vaigro-Wolff, A.; et al. Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J. Natl. Cancer Inst. 1991, 38, 757–766. [Google Scholar] [CrossRef]
- Sumantran, V.N. Cellular chemosensitivity assays: An overview. In Cancer Cell Culture: Methods and Protocols, 2nd ed.; Cree, I.A., Ed.; Humana Press: New York, NY, USA, 2011; Chapter 19; pp. 219–236. [Google Scholar]
- Domínguez, M.; Nieto, A.; Marín, J.C.; Keck, A.S.; Jeffery, E.; Céspedes, C.L. Antioxidant activities of extracts from Barkleyanthus salicifolius (Asteraceae) and Penstemon gentianoides (Scrophulariaceae). J. Agric. Food Chem. 2005, 53, 5889–5895. [Google Scholar] [CrossRef] [PubMed]
- Rossato, J.I.; Ketzer, L.A.; Centuriao, F.B.; Silva, S.J.; Lüdtke, D.S.; Zeni, G.; Braga, A.L.; Rubin, M.A.; Rocha, B.T. Antioxidant properties of new chalcogenides against lipid peroxidation in rat brain. Neurochem. Res. 2002, 27, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Ng, T.B.; Liu, F.; Wang, Z.T. Antioxidative activity of natural products from plants. Life Sci. 2000, 66, 709–723. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehyde lipid peroxidation products. Methods Enzymol. 1990, 186, 407–421. [Google Scholar] [PubMed]
- Sample Availability: Some samples of the compounds are available from the authors.

| Compound | 7 | 8 | 9 | 10 | 12 | 13 | 14 |
|---|---|---|---|---|---|---|---|
| CCDC deposit No. | 1452007 | 1451557 | 1451621 | 1451555 | 1451617 | 1451556 | 1056387 |
| Crystal size (mm) | 0.37 × 0.05 × 0.03 | 0.30 × 0.20 × 0.09 | 0.39 × 0.19 × 0.13 | 0.37 × 0.11 × 0.11 | 0.31 × 0.12 × 0.05 | 0.35 × 0.21 × 0.15 | Not determined |
| Color/shape | Light green/needles | Light yellow/needles | Colorless/prism | Light green/needles | Colorless/prism | Light yellow/needles | Colorless/prism |
| Empirical formula | C19H16O3 | C19H16O3 | C19H16O3 | C22H20O6 | C22H20O6 | C20H18O4 | C23H22O7 |
| Formula weight | 292.32 | 292.32 | 292.32 | 380.38 | 380.38 | 322.34 | 410.40 |
| Crystal system | Orthorhombic | Monoclinic | Monoclinic | Monoclinic | Monoclinic | Triclinic | Monoclinic |
| Space group | P 212121 | P 21/c | P 21/c | P 21/c | P 21/c | P-1 | P 21/c |
| Unit cell dimensions | |||||||
| a, Å | 5.7012(2) | 33.3788(14) | 19.2677(6) | 6.07840(10) | 22.6222(13) | 7.9343(10) | 5.9945(4) |
| b, Å | 15.1134 | 5.7488(2) | 4.10240(10) | 17.3377(4) | 7.6843(4) | 10.0440(14) | 36.2260(19) |
| c, Å | 18.1035 | 8.0128(4) | 21.2526(6) | 18.3150(5) | 10.9077(7) | 11.9990(16) | 9.6239(8) |
| α, deg | 90.00 | 90.00 | 90.00 | 90.00 | 90.00 | 70.652(4) | 90.00 |
| β, deg | 90.00 | 95.736(2) | 115.0910(10) | 90.5980(10) | 95.314(2) | 71.023(4) | 91.083(6) |
| γ, deg | 90.00 | 90.00 | 90.00 | 90.00 | 90.00 | 89.865(5) | 90.00 |
| Volume, Å3 | 1559.88(10) | 1529.86(11) | 1521.36 | 1930.03(8) | 1888.00(19) | 847.2(2) | 2089.5(2) |
| Z | 4 | 4 | 4 | 4 | 4 | 2 | 4 |
| Density (calculated), Mg/m3 | 1.245 | 1.269 | 1.276 | 1.309 | 1.338 | 1.264 | 1.305 |
| Absorption coefficient, mm−1 | 0.084 | 0.085 | 0.086 | 0.095 | 0.098 | 0.088 | 0.806 |
| F(000) | 616 | 616 | 616 | 800 | 800 | 340 | 864 |
| Θ range for data collection (°) | 2.25 to 25.39 | 2.45 to 25.31 | 1.94 to 25.40 | 2.22 to 25.50 | 2.71 to 25.32 | 2.16 to25.33 | Not determined |
| Index ranges | −6 ≤ η ≤ 6, −17 ≤ κ ≤ 17, −21 ≤ λ ≤ 21 | −39 ≤ η ≤ 39, −5 ≤ κ ≤ 5, −9 ≤ λ ≤ 9 | −23 ≤ η ≤ 23, −4 ≤ κ ≤ 4, −25 ≤ λ ≤ 25 | −7 ≤ h ≤ 7, −20 ≤ k ≤ 20, −22 ≤ l ≤ 22 | −27 ≤ h ≤ 27, −9 ≤ k ≤ 9, −13 ≤ l ≤ 13 | −9 ≤ h ≤ 9, −12 ≤ k ≤ 12, −14 ≤ l ≤ 14 | −7 ≤ h ≤ 6, −44 ≤ k ≤ 44, −11 ≤ l ≤ 11 |
| Reflections collected | 13,670 | 20,778 | 16,473 | 26,381 | 22,547 | 12,126 | 20,379 |
| Independent reflections | 2857 (Rint = 0.1503) | 2764 (Rint = 0.1323) | 2781 (Rint = 0.0559) | 3585 (Rint = 0.0925) | 3433 (Rint = 0.1724) | 3065 (Rint = 0.0631) | 4128 |
| Data/restraints/parameters | 2857/154/246 | 2764/0/200 | 2781/162/246 | 3585/0/257 | 3433/0/256 | 3065/0/219 | 4128/0/275 |
| Goodness-of-fit on F2 | 0.749 | 1.028 | 1.052 | 0.963 | 1.010 | 1.046 | 1.020 |
| Final R indices [(I) > 2σ(I)] | R1 = 0.0472, wR2 = 0.0486 | R1 = 0.0494, wR2 = 0.1162 | R1 = 0.0489, wR2 = 0.1271 | R1 = 0.0461, wR2 = 0.1072 | R1 = 0.0606, wR2 = 0.1255 | R1 = 0.0632, wR2 = 0.1629 | R1 = 0.0620, wR2 = 0.1162 |
| R indices (all data) | R1 = 0.2148, wR2 = 0.0749 | R1 = 0.0816, wR2 = 0.1361 | R1 = 0.0676, wR2 = 0.1422 | R1 = 0.0851, wR2 = 0.1267 | R1 = 0.1493, wR2 = 0.11618 | R1 = 0.0888, wR2 = 0.1831 | R1 = 0.1574, wR2 = 0.1945 |
| Largest diff. peak and hole (e/A−3) | 0.168 and −0.141 | 0.140 and −0.171 | 0.156 and −0.227 | 0.279 and −0.258 | 0.195 and −0.185 | 0.435 and −0.190 | 0.305 and −0.160 |
| Compound | D-H···A | D-H | H···A | D···A | D-H···A | Symmetry Code | ||
|---|---|---|---|---|---|---|---|---|
| 7 | C3-H3•••O3 | 0.930(5) | 2.790(2) | 3.486(2) | 132.5(4) | −x + 1 | +y + 1/2, | −z + 1/2 |
| C9-H9•••O3 | 0.930(5) | 2.749(1) | 3.463(2) | 134.3(4) | −x + 1 | +y + 1/2 | −z + 1/2 | |
| C14-H14•••O2 | 0.930(5) | 2.902(3) | 3.571(6) | 130.0(3) | x − 1/2 | −y + 1/2 + 1 | −z | |
| C17-H17•••O1 | 0.931(11) | 2.740(3) | 3.356(12) | 124.5(7) | x + 1 | +y | +z | |
| 8 | C2-H2•••O2 | 0.930(2) | 2.735(2) | 3.665(2) | 179.9(1) | x | +y − 1 | +z |
| C9-H9•••O2 | 0.929(2) | 2.957(2) | 3.590(3) | 126.7(2) | x | −y + 1/2 | +z + 1/2 | |
| C15-H15•••O2 | 0.931(2) | 2.761(2) | 3.543(2) | 142.1(1) | x | −y + 1/2 + 1 | +z − 1/2 | |
| 9 | C5-H5•••O3 | 0.930(2) | 2.672(5) | 3.539(5) | 155.3(2) | −x | +y − 1/2 | −z + 1/2 |
| C3-H3•••O3 | 0.930(2) | 2.719(4) | 3.588(4) | 155.8(1) | −x | +y − 1/2 | −z + 1/2 | |
| C12-H12•••O2 | 0.930(2) | 2.634(2) | 3.554(3) | 169.9(1) | −x | +y − 1/2 | −z + 1/2 | |
| C17-H17•••O2 | 0.930(3) | 2.605(1) | 3.534(3) | 177.2(2) | −x | +y − 1/2 | −z + 1/2 | |
| 10 | C10-H10A•••O2 | 0.981(2) | 2.597(1) | 3.124(2) | 113.8(1) | −x + 2 | −y + 2 | −z |
| C12-H12A•••O6 | 0.980(2) | 2.661(2) | 3.414(3) | 133.9(2) | x − 1 | −y + 1/2+1 | z − 1/2 | |
| C17-H17•••O6 | 0.951(2) | 2.410(2) | 3.183(3) | 138.2(1) | −x + 2 | +y + 1/2 | −z + 1/2 | |
| C20-H20•••O5 | 0.951(2) | 2.484(2) | 3.246(3) | 137.1(2) | −x + 2 | +y + 1/2 | −z + 1/2 | |
| 12 | C18-H18•••O1 | 0.930(3) | 2.358(2) | 2.846(4) | 112.5(2) | x | y | z |
| C5-H5•••O6 | 0.931(3) | 2.376(2) | 3.267(4) | 160.2(2) | −x + 1 | −y | −z + 1 | |
| C20-H20•••O1 | 0.929(3) | 2.714(3) | 3.431(4) | 134.7(2) | −x + 1 | −y | −z + 1 | |
| C12-H12A•••O3 | 0.961(3) | 2.680(2) | 3.531(4) | 147.9(2) | −x + 2 | +y + 1/2 | z + 1/2 + 2 | |
| C12-H12B•••O5 | 0.959(3) | 2.429(2) | 3.372(4) | 167.6(2) | −x + 2 | +y + 1/2 | z + 1/2 + 2 | |
| 13 | C6-H6•••O1 | 0.930(3) | 2.417(2) | 3.328(4) | 166.0(2) | −x | −y + 1 | −z |
| C11-H11B•••O4 | 0.960(3) | 2.825(2) | 3.475(4) | 125.8(2) | x − 1 | +y | +z | |
| C18-H18•••O2 | 0.929(2) | 2.718(2) | 3.332(3) | 124.3(1) | −x | −y | −z + 1 | |
| C19-H19•••O2 | 0.930(4) | 2.846(2) | 3.397(4) | 119.1(2) | −x | −y | −z + 1 | |
| 14 | C13-H13•••O11 | 0.929(3) | 2.496(3) | 3.413(4) | 169.2(2) 1 | −x + 1 | −y + 1 | −z |
| C5-H5•••O20 | 0.930(3) | 2.581(3) | 3.504(4) | 172.0(2) 2 | −x + 1 | −y + 1 | −z + 1 | |
| C25-H25•••O20 | 0.931(3) | 2.600(2) | 3.499(4) | 162.6(2) 2 | −x + 1 | −y + 1 | −z + 1 | |
| C2-H2•••O22 | 0.930(3) | 2.672(2) | 3.485(4) | 146.3(2) 4 | −x | −y + 1 | −z | |
| C23-H23C•••O18 | 0.960(4) | 2.550(2) | 3.475(4) | 161.9(2) 5 | −x + 1 | +y | +z | |
| IC50 (µM) on Cell Line | |||
|---|---|---|---|
| Retro-Curcuminoid | K562 | MCF-7 | SKLU-1 |
| Curcumin | 9.75 ± 0.6 | 10.43 ± 1.8 | 7.78 ± 0.64 |
| 7 | 10.74 ± 0.5 | 13.19 ± 1.4 | 11.59 ± 1.2 |
| 8 | 12.8 ± 1.9 | 18.68 ± 2.5 | 12.85 ± 0.9 |
| 9 | 28.15 ± 2.5 * | 63.46 ± 10.8 * | 63.81 ± 2.7 * |
| 10 | 8.60 ± 0.44 | 12.01 ± 0.62 | 8.07 ± 0.55 |
| Retro-Curcuminoid | Concentration (µg/mL) | % Inhibition |
|---|---|---|
| 7 | 50 | 4.33 ± 1.06 * |
| 5 | 1.97 ± 0.77 * | |
| 8 | 50 | 1.77 ± 0.22 * |
| 5 | 0.27 ± 0.06 * | |
| 9 | 50 | 3.30 ± 0.78 * |
| 5 | 3.19 ± 0.53 * | |
| 10 | 50 | 96.18 ± 6.53 |
| 5 | 14.81 ± 2.33 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obregón-Mendoza, M.A.; Estévez-Carmona, M.M.; Hernández-Ortega, S.; Soriano-García, M.; Ramírez-Apan, M.T.; Orea, L.; Pilotzi, H.; Gnecco, D.; Cassani, J.; Enríquez, R.G. Retro-Curcuminoids as Mimics of Dehydrozingerone and Curcumin: Synthesis, NMR, X-ray, and Cytotoxic Activity. Molecules 2017, 22, 33. https://doi.org/10.3390/molecules22010033
Obregón-Mendoza MA, Estévez-Carmona MM, Hernández-Ortega S, Soriano-García M, Ramírez-Apan MT, Orea L, Pilotzi H, Gnecco D, Cassani J, Enríquez RG. Retro-Curcuminoids as Mimics of Dehydrozingerone and Curcumin: Synthesis, NMR, X-ray, and Cytotoxic Activity. Molecules. 2017; 22(1):33. https://doi.org/10.3390/molecules22010033
Chicago/Turabian StyleObregón-Mendoza, Marco A., María Mirian Estévez-Carmona, Simón Hernández-Ortega, Manuel Soriano-García, María Teresa Ramírez-Apan, Laura Orea, Hugo Pilotzi, Dino Gnecco, Julia Cassani, and Raúl G. Enríquez. 2017. "Retro-Curcuminoids as Mimics of Dehydrozingerone and Curcumin: Synthesis, NMR, X-ray, and Cytotoxic Activity" Molecules 22, no. 1: 33. https://doi.org/10.3390/molecules22010033
APA StyleObregón-Mendoza, M. A., Estévez-Carmona, M. M., Hernández-Ortega, S., Soriano-García, M., Ramírez-Apan, M. T., Orea, L., Pilotzi, H., Gnecco, D., Cassani, J., & Enríquez, R. G. (2017). Retro-Curcuminoids as Mimics of Dehydrozingerone and Curcumin: Synthesis, NMR, X-ray, and Cytotoxic Activity. Molecules, 22(1), 33. https://doi.org/10.3390/molecules22010033






