Abstract
In the course of a phytochemical and chemotaxonomical investigation of Castanopsis species (Fagaceae), three new phenolic compounds, (3R,1′S)-[1′-(6″-O-galloyl-β-d-gluco-pyranosyl)oxyethyl]-3-hydroxy-dihydrofuran-2(3H)-one (1), (2R,3S)-2-[2′-(galloyl)oxyethyl]-dihydroxybutanoic acid (2), and (3S,4S)-3-hydroxymethyl-3,4-dihydro-5,6,7-trihydroxy-4-(4′-hydroxy-3′-methoxyphenyl)-1H-[2]-benzopyran-1-one (3) were isolated from the fresh leaves of Castanopsis fargesii. In addition, a known phenolic glycoside, gentisic acid 5-O-α-l-rhamnopyranosyl-(1→2)-β-d-glucopyranoside (4) was also isolated and identified. Their structures were elucidated by means of spectroscopic methods including one- and two-dimensional NMR techniques.
1. Introduction
The genus Castanopsis belongs to the Fagaceae family and is commonly found in the evergreen forests of East Asia. There are about 120 species of Castanopsis, however, the classical plant taxonomy of the species is very complicated and sometimes confusing [], thus, the application of other auxiliary methods and technologies, such as chemotaxonomy and cytotaxonomy, is necessary to identify species within this genus []. Previous phytochemical investigations on the plants of this genus C. fissa, C. cuspidata var. seiboldii, and C. hystrix have led to the isolation of triterpene hexahydroxydiphenoyl (HHDP) esters, HHDP glucoses, galloyl, acylated quinic acids, phenol glucosides, condensed tannins, and flavonol glycosides [,,,,,].
In this study, we investigated C. fargesii, which is widely distributed in southern China, where it is usually used as a traditional medicine for the treatment of diarrhea, hemorrhage, and chronic ulcers []. Our preliminary analysis by HPLC and TLC indicated that the leaves are rich in tannins. Subsequent chromatographic separation of the extract yielded two metabolites 1 and 2, which were identified as 2,3-dihydroxy-2-(2-hydroxyethyl)-butanoic acid derivatives, and two phenolic compounds 3 and 4. This paper reports the isolation and structural characterization of the new compounds 1–3 and an assessment of the cytotoxicity of these molecules.
2. Results and Discussion
The fresh leaves of C. fargesii were extracted with 80% aqueous ethanol, and the extract was partitioned between Et2O and water. The Et2O and aqueous fractions were separated by a combination of Sephadex LH-20, MCI gel CHP 20P, Toyopearl Butyl-650C, Chromatorex ODS, and Diaion HP20SS column chromatography and semi-preparative reverse-phase HPLC, to yield three new compounds 1–3 and one known phenolic compound gentisic acid 5-O-α-l-rhamnopyranosyl-(1→2)-β-d-glucoside [] (4) (Figure 1).
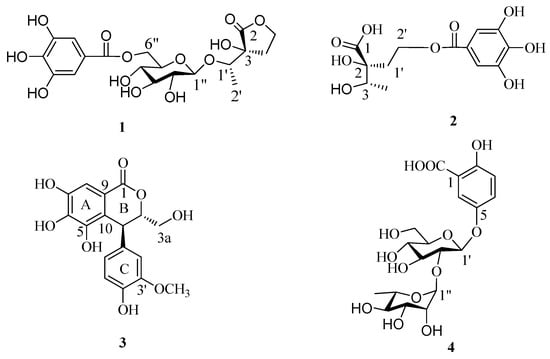
Figure 1.
Structures of compounds 1–4.
Compound 1 was isolated as a brown amorphous powder and gave a positive FeCl3 test (dark blue), which suggested the presence of phenol moieties in the molecule. The molecular formula C19H24O13 was determined based on the liquid chromatography-mass spectrometry IT-TOF (LC-MS/IT-TOF), which showed [M − H]− and [M + Na]+ ion peaks at m/z 459.1143 (calcd. for C19H23O13, 459.1144) and 483.1118 (calcd. for C19H24O13Na, 483.1109), respectively. In the 1H- and 13C-NMR spectra (Table 1), two proton singlets at δH 7.13 (2H, s) and four aromatic carbon signals at δC 109.1 and 145.2, along with an ester carbonyl signal at δC 166.4 suggested the presence of a galloyl group []. The hydrolysis of 1 produced d-glucose, which was identified by GC analysis. The coupling constant of the anomeric sugar proton was 7.8 Hz, indicating that the sugar moiety was in the β configuration. The large downfield shift of the glucose H-6″ protons (δH 4.32 and 4.58) suggested esterification with the galloyl moiety at this position. This was confirmed by the HMBC correlation between H-6″ and the carboxy carbon (δC 166.4) (Figure 2). A second HMBC correlation between H-1″ and C-1′ suggested that the glycosyl group is linked to C-1′. HSQC experiment showed that the remaining moiety was composed of six carbons: a carboxy carbon (δC 178.6, C-2), an oxygenated methine (δC 77.8, C-1′), an oxygenated quaternary carbon (δC 76.6, C-3), an oxygenated methylene (δC 65.8, C-5), a methylene (δC 29.9, C-4), and a methyl (δC 15.6, C-2′) carbon. The presence of a γ-lactone structure in the remaining moiety was suggested by the lower field shift of the carboxy carbon (δC 178.6) and the unsaturation index (eight). This was also confirmed by HMBC correlations shown in Figure 2, in which the oxygenated methylene protons (δH 4.31–4.34, H-5) correlated with C-2. Furthermore, the 1H-1H COSY correlations of H-4 (δH 2.10) with H-5 (δH 4.32) and H-1′ with H-2′ and the HMBC correlations of H-1′ with C-3 and C-4 and H-4 with C-2, C-3, and C-5 indicated that the remaining moiety is 3-hydroxy-3-(1′-hydroxyethyl)dihydrofuran-2(3H)-one. Consequently, the structure of compound 1 was established as 3-[1′-(6″-O-galloyl-β-d-glucopyranosyl)oxyethyl]-3-hydroxy-dihydrofuran-2(3H)-one.

Table 1.
1H (500 MHz) and 13C (125 MHz) NMR data of compounds 1 and 2 in acetone-d6.
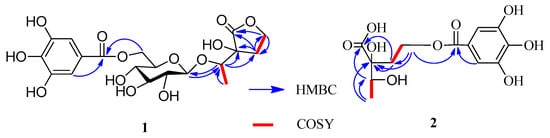
Figure 2.
Key HMBC and 1H-1H COSY correlations of 1 and 2.
Compound 2 was isolated as a yellow amorphous powder and gave a positive FeCl3 test (dark blue). The presence of a galloyl group was deduced from the 13C-NMR signals (Table 1) []. The molecular formula of C13H16O9 was established based on the LC-MS/IT-TOF (m/z 339.0672 [M + Na]+, calcd. for C13H16O9Na, 339.0687) and 13C-NMR data. The 13C-NMR and DEPT spectra showed signals attributable to a methyl (δC 16.9), a methylene (δC 34.2), an oxymethine (δC 71.4), an oxymethylene (δC 60.2), an oxy quaternary (δC 77.7), and a carboxyl (δC 175.4) carbons. The NMR data of 2 (Table 1) were similar to those of 1, except for the absence of the signals for one glucosyl moiety, which was supported by its MS data. A 2,3-dihydroxy-2-(2′-oxyethyl)-butanoic acid moiety could be constructed by 1H-1H COSY correlations (Figure 2) of H-4 with H-3 and H-1′a with H-2′a and the HMBC correlations of H-4 with C-3 (δC 71.4), H-3 with C-1, H-1′ with C-1, and H-2′ with C-2 (Figure 2). The HMBC correlation of H-2′ with the carboxy carbon (δC 165.9) indicated that the galloyl group was attached to C-2′. Based on these results, the structure of 2 was determined to be 2-[2′-(galloyl)-oxyethyl]-2,3-dihydroxybutanoic acid.
The absolute configurations at C-2 and C-3 of 2 were established using the modified Mosher’s method [,]. Treatment of 2 with CH3I, then with (R)-(−)- and (S)-(+)-2-methoxy-2-trifluoromethyl-2-phenylacetyl (MTPA) chloride to get the C-3 (S)- and (R)-MTPA ester derivatives, respectively. Δδ values obtained from the 1H-NMR data of the C-3 (R)- and (S)-MTPA ester derivative indicated that the absolute configuration at C-3 of 2 was S (Figure 3). Compound 2 reacted with 2,2-dimethoxypropane (DMP) and pyridinium p-toluene sulfonate (PPTS) to form the 2,3-O-isopropylidene derivative. The C-2 and C-3 relative configuration of 2,3-O-isopropylidene derivative was determined based on the NOE correlation of H-1′ with H3-4 (Figure 4). Thus, the absolute configurations at C-2 and C-3 of 2 were assigned as R and S, respectively.
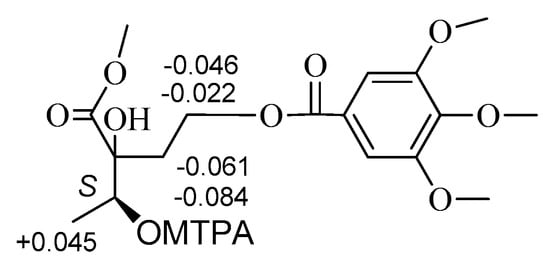
Figure 3.
Δδ(S-R) values of MTPA ester derivative of 2.
Figure 4.
Key NOE correlations of 2,3-O-isopropylidene derivative of 2.
Compounds 1 and 2 both contain a 2,3-dihydroxy-2-(2-hydroxyethyl)-butanoic acid moiety. The hydrolysis of 1 in 1 M HCl yielded 3-hydroxy-3-(1-hydroxyethyl)dihydrofuran-2(3H)-one that was identified to have the same absolute configuration as 2 by comparing their and CD data. Hence, the absolute configurations of 1 were assigned as 3R,1′S.
Compound 3 was obtained as a brown amorphous powder, which gave a dark blue color with FeCl3. The molecular formula C17H16O8 was deduced from the [M − H]− peak at m/z 347.0768 in the LC-MS/IT-TOF (calcd. for C17H15O8, 347.0772). Comparison of the 1H- and 13C-NMR data of 3 (Table 2) and (3S,4S)-3-[(β-d-glucopyranosyl)oxymethyl]-3,4-dihydro-5,6,7-trihydroxy-4-(4′-hydroxy-3′-methoxyphenyl)-1H-[2]-benzopyran-1-one [] revealed that the methyl at C-3 in the known compound was replaced by a hydroxymethyl in 3. This was confirmed by the MS data and the correlations of the methine proton (δH 4.67) with the methylene carbon (δC 62.4) in the HMBC spectrum (Figure 5), as well as 1H-1H COSY correlations of H-3 (δH 4.67) with H-3a (δH 3.54) and H-4 (δH 4.50) (Figure 5). Comparison of the CD and the optical rotation ( +18.3°) data of 3 with those of similar compounds suggested that the absolute configuration is 3S,4S []. Based on the above evidences, the structure of compound 3 was concluded to be (3S,4S)-3-hydroxymethyl-3,4-dihydro-5,6,7-trihydroxy-4-(4′-hydroxy-3′-methoxyphenyl)-1H-[2]-benzopyran-1-one.

Table 2.
1H-NMR (500 MHz) and 13C-NMR (125 MHz) data of compound 3 in DMSO-d6.
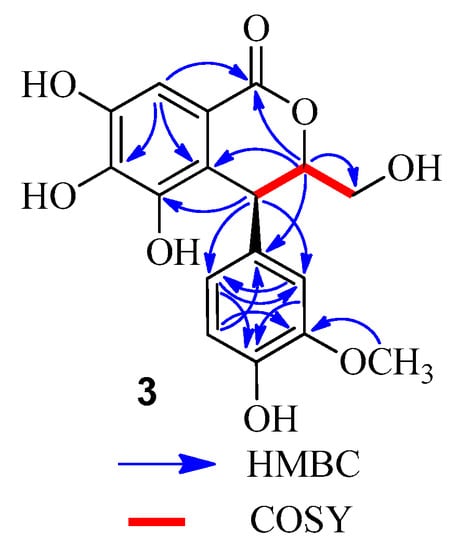
Figure 5.
Key HMBC and COSY correlations of 3.
All isolates were subjected to a cytotoxicity assay in vitro against human lung epithelial A549, human hepatocellular carcinoma SMMC-7721 cell, human gastric carcinoma MGC-803 cell, liver hepatocellular HepG2 cell, and human breast adenocarcinoma MCF-7 tumour cell. Unfortunately, none of the isolates showed inhibitions of those tumour cells at the highest concentration tested (IC50 value > 10 μM).
3. Experimental Section
3.1. Materials
The leaves of C. fargesii were collected at Guangxi Institute of Botany, Guangxi, China, in August 2014, and were identified by Prof. Shi-Hong Lu. A voucher specimen (20140627) was deposited in the Guangxi Key Laboratory of Functional Phytochemicals Research and Utilization, Guangxi Institute of Botany, China.
3.2. General Experimental Procedures
Optical rotations were measured with a 341 digital polarimeter (Perkin-Elmer Corp., Waltham, MA, USA). 1H- and 13C-NMR spectra were measured in acetone at 27 °C, using an Avance 500 spectrometer (500 MHz for 1H and 125 MHz for 13C, Bruker Biospin AG, Fällanden, Switzerland). Coupling constants and chemical shifts were given in Hz and on a δ (ppm) scale, respectively. GC was performed on a 6890N instrument equipped with a FID detector (Agilent Technologies, Santa Clara, CA, USA) operated at 280 °C (column: 28 m × 0.32 mm i.d. HP-5, column temp. 160 °C). LC-MS/IT-TOF was recorded on a LCMS-IT-TOF spectrometer (Shimadzu, Kyoto, Japan). Semi-preparative HPLC was performed on an Agilent 1200 apparatus equipped with a UV detector and a Zorbax SB-C-18 (9.4 × 250 mm) column (Agilent). Column chromatography (CC) was performed using Sephadex LH-20 (25–100 μm; GE Healthcare Bio-Science AB, Uppsala, Sweden), MCI gel CHP 20P (75–150 μm; Mitsubishi Chemical, Tokyo, Japan), Diaion HP20SS (Mitsubishi Chemical), Chromatorex ODS (100–200 mesh; Fuji Silysia Chemical, Aichi, Japan), and Toyopearl Butyl-650C (TOSOH, Tokyo, Japan) columns. TLC was performed on precoated Kieselgel 60 F254 plates (0.2 mm thick; Merck, Darmstadt, Germany) with toluene–HCO2Et–HCO2H (1:7:1, v/v) as the solvent, and spots were detected by spraying with a 2% ethanolic FeCl3.
3.3. Extraction and Separation
The fresh leaves of C. fargesii (5.20 kg) were cut into small pieces and extracted three times with EtOH/H2O (8:2, v/v, 36 L) by maceration at room temperature for 7 days. The extracts were combined and concentrated under reduced pressure to give an aqueous solution. The solution was partitioned with Et2O four times to give the Et2O fraction (32.4 g). The aqueous layer was subjected to Sephadex LH-20 CC (8 cm i.d. × 40 cm) with 0%–100% MeOH–H2O (20% stepwise elution, each 1.5 L) to give 9 fractions: frs 1 (15.6 g), 2 (84.5 g), 3 (26.6 g), 4 (27.0 g), 5 (130.0 g), 6 (12.9 g), 7 (3.3 g), 8 (2.3 g), and 9 (2.2 g). Fraction fr. 2 (84.5 g) was separated by MCI gel CHP 20PCC (6 cm i.d. × 40 cm) with MeOH–H2O (10% stepwise elution, each 1.0 L) to yield seven fractions, and fraction fr. 2-2 (5.3 g) was further fractionated by Diaion HP20SS CC (4 cm i.d. × 30 cm) with H2O containing increasing proportions of MeOH (0%–100%, 10% stepwise elution, each 0.5 L) to give 4 (105 mg). The Et2O fraction was subjected to MCI gel CHP 20PCC (5 cm i.d. × 50 cm) with 0%–100% MeOH in H2O (10% stepwise elution, each 0.5 L) to yield 10 fractions: frs E-1 (5.3 g), 2 (7.5 g), 3 (1.6 g), 4 (3.2 g), 5 (4.3 g), 6(1.5 g), 7 (2.3 g), 8 (1.0 g), 9 (9.9 g) and 10 (3.5 g). Fr. E-3 was fractionated by Toyopearl Butyl-650C CC (3 cm i.d. × 30 cm) with 0%–100% MeOH–H2O containing 0.1% CF3CO2H (TFA) (10% stepwise elution, each 0.3 L) to give fr. E-31 (1.3 g) and fr. E-32 (122 mg). Fr. E-32 was further purified by Chromatorex ODS CC (3 cm i.d. × 30 cm) with 0%–80% MeOH in H2O (5% stepwise elution, each 0.2 L) to give 3 (12 mg). Fraction E-4 was separated by Sephadex LH-20 CC (4 cm i.d. × 40 cm) with H2O containing increasing amounts of MeOH (0%–100%, 10% stepwise elution, each 0.5 L) to yield Fr. E-41 (250 mg), Fr. E-42 (150 mg), Fr. E-43 (296 mg) and Fr. E-44 (1.7 g). The Fr. E-42 and Fr. E-43 were further purified by semi-preparative HPLC (MeCN/H2O, 20:80, 2.5 mL/min) to give 1 (46 mg, tR 14.5 min) and 2 (68 mg, tR 13.2 min), respectively.
3.4. Spectroscopic Data
(3R,1'S)-[1'-(6"-O-Galloyl-β-d-glucopyranosyl)oxyethyl]-3-hydroxy-dihydrofuran-2(3H)-one (1): Brown amorphous powder; +52.1° (c = 0.12, MeOH); UV (MeOH) λmax nm (log ε): 272 (4.32); CD (MeOH) λmax (Δε) 278 (8.4), 254 (4.5), 209 (2.7). 1H- and 13C-NMR data, see Table 1; LC-MS/IT-TOF m/z [M − H]− 459.1143 (calcd. for C19H23O13, 459.1144) and [M + Na]+ 483.1118 (calcd. for C19H24O13Na, 483.1109).
(3R,1'S)-3-Hydroxy-1'-hydroxyethyl-dihydrofuran-2(3H)-one (Hydrochloride of 1): −17.0° (c = 0.15, MeOH); UV (MeOH) λmax nm (log ε): 265 (3.16); CD (MeOH) λmax (Δε) 267 (5.7), 251 (3.2), 211 (1.2). 1H-NMR (MeOH-d4, 500 MHz) δ 4.36 (1H, m, H-5a), 4.25 (1H, m, H-5b), 2.11 (1H, m, H-4a), 2.33 (1H, m, H-4b), 1.21 (3H, d, J = 6.5 Hz, H-4), 4.12 (1H, q, J = 6.5 Hz, H-1′), 1.21 (3H, d, J = 6.5 Hz, H-2′); LC-MS/IT-TOF m/z 169.0475 [M + Na]+ (calcd. for C6H10O4Na, 169.0477).
(2R,3S)-2-[2'-(Galloyl)oxyethyl]-dihydroxybutanoic acid (2): Yellow amorphous powder; −16.7° (c = 0.12, MeOH); UV (MeOH) λmax nm (log ε): 267 (4.26); CD (MeOH) λmax (Δε) 268 (10.6), 252 (4.6), 212 (1.4). 1H- and 13C-NMR data, see Table 1; LC-MS/IT-TOF m/z 339.0672 [M + Na]+ (calcd. for C13H16O9Na, 339.0687).
(3S,4S)-3-Hydroxymethyl-3,4-dihydro-5,6,7-trihydroxy-4-(4'-hydroxy-3'-methoxyphenyl)-1H-[2]-benzopyran-1-one (3): Brown amorphous powder; +18.3° (c = 0.11, MeOH); UV (MeOH) λmax nm (log ε): 220 (4.26), 278 (2.35); CD (MeOH) λmax (Δε) 288 (11.4), 242 (6.5), 218 (2.7). 1H- and 13C-NMR data, see Table 2; LC-MS/IT-TOF m/z 347.0768 [M − H]− (calcd. for C17H15O8, 347.0772).
3.5. Preparation of MTPA Esters Derivatives
CH3I (30 mg) and K2CO3 (15 mg) were added to a solution of 2 (10 mg) in DMF (5 mL). After stirring for 24 h at room temperature (r.t.), the reaction mixture was suspended in H2O and extracted with CHCl3. The CHCl3 layer was vacuum dried to afford a residue (6.2 mg). Then, DMAP (3.8 mg), Et3N (4.0 μL), and (R)-(−)-MTPACl (3.0 μL) were added to a solution of the residue (3.1 mg) in CH2Cl2 (1.0 mL) and stirred for 4 h at r.t. The reaction mixture was dried under a stream of N2. Separation of the residue was done by a silica gel column (hexane/EtOAc, 4:1) to afford the (S)-MTPA ester derivative (2.1 mg). The (R)-MTPA ester derivative (2.3 mg) was obtained according to the same procedure using (S)-(+) MTPACl.
(3S)-MTPA Ester derivative of 2: Colorless oil; 1H-NMR (MeOH-d4, 500 MHz) δ 7.0512–7.3901 (7H), 4.8212 (1H, q, J = 6.6 Hz, H-3), 1.3648 (3H, d, J = 6.6 Hz, H-4), 2.2511 (1H, m, H-1′a), 2.3004 (1H, m, H-1′b), 4.2526 (1H, m, H-2′a), 4.2812 (1H, m, H-2′b), 3.3206–3.8516 (-OCH3 × 5). LC-MS/IT-TOF m/z 611.17147 [M + Na]+ (calcd. for C27H31F3O11Na, 611.17162).
(3R)-MTPA Ester derivative of 2: Colorless oil; 1H-NMR (MeOH-d4, 500 MHz) δ 7.0510–7.3902 (7H), 4.8210 (1H, q, J = 6.6 Hz, H-3), 1.3603 (3H, d, J = 6.6 Hz, H-4), 2.2572 (1H, m, H-1′a), 2.3088 (1H, m, H-1′b), 4.2548 (1H, m, H-2′a), 4.2858 (1H, m, H-2′b), 3.3312–3.8716 (-OCH3 × 5). LC-MS/IT-TOF m/z 611.1710 [M + Na]+ (calcd. for C27H31F3O11Na, 611.1716).
3.6. Preparation of Acetonide Derivative of 2
Compound 2 (10.2 mg) was dissolved in acetone (1.0 mL) and treated with DMP (0.2 mL) and PPTS (6.5 mg) at r.t. After 4 h, Et3N (7.5 μL) was added and the mixture was concentrated by N2 blowing. The residue was separated on a silica gel column (CH2Cl2/EtOAc, 4:1–2:1) to afford the 2,3-O-isopropylidene derivative (3.2 mg) of 2.
2,3-O-Isopropylidene Derivative of 2: Colorless oil; 1H-NMR (MeOH-d4, 500 MHz) δ 6.98 (2H, s), 4.36 (1H, q, J = 6.5 Hz, H-3), 1.21 (3H, d, J = 6.5 Hz, H-4), 2.11 (1H, m, H-1′a), 2.23 (1H, m, H-1′b), 4.25 (1H, m, H-2′a), 4.29 (1H, m, H-2′b), 1.27 (3H, s, acetonide-CH3), 1.31 (3H, s, acetonide-CH3); LC-MS/IT-TOF m/z 379.1007 [M + Na]+ (calcd. for C18H20O9Na, 379.1005).
3.7. Acid Hydrolysis and Sugar Analysis by GC
Compound 1 (6 mg) was dissolved in MeOH (4.0 mL) and 1 M H2SO4 (2.0 mL) and refluxed for 2 h on a H2O bath. After the hydrolysate was cool, H2O (8.0 mL) was added, then extracted with EtOAc (3 × 10.0 mL). The EtOAc layer was vacuum dried and chromatographed on semi-preparative HPLC eluting with a gradient of MeOH–H2O (5:95–25:75, v/v) to afford 3-hydroxy-1′-hydrooxyethyl-dihydrofuran-2(3H)-one. The aqueous layer was neutralized with aqueous Ba(OH)2 and evaporated under reduced pressure to give a residue. The residue was dissolved in pyridine (100 μL), subsequent treated with 0.1 mL cysteine methyl ester hydrochloride (150 μL; Sigma, St. Louis, MO, USA) and warmed at 60 °C for 1 h, then the trimethysilylation reagent HMDS/TMCS (hexamethyldisilazane/trimethylchlorosilane/pyridine 2:1:10; Acros Organics, Geel, Belgium) was added and warmed at 60 °C for 30 min. The reaction mixture was partitioned between water and hexane. The hexane extract was analyzed by GC [] (detector temperature: 280 °C; injector temperature: 250 °C; temperature gradient: start at 160 °C, hold for 5 min, increase to 280 °C at 5 °C/min, hold for 10 min). The authentic samples were analyzed in the same way. The tR values of d-glucose and l-glucose were 13.25 min and 15.32 min, respectively. The thiazolidine derivatives of the samples were confirmed by comparison with authentic standards.
3.8. Cytotoxicity Assay
All isolates were tested for cytotoxicity in vitro against A549, SMMC-7721, MGC-803, HepG2, and MCF-7 tumour cells via the MTT assay [,] with hydroxycamptothecine as a positive control.
4. Conclusions
In this study, we separated and identified three new compounds 1–3 and a known compound 4 from the leaves of C. fargesii. The 2,3-dihydroxy-2-(2-hydroxyethyl)-butanoic acid moiety in 1 and 2 is an unusual carboxylic acid in Nature, thus, these compounds might be recognized as chemotaxonomic markers. Our preliminary examination also suggested this plant contains triterpene HHDP esters, which are important chemotaxonomical markers of Castanopsis sp.; therefore, further phytochemical investigations of the leaves of C. fargesii are in progress.
Acknowledgments
We gratefully acknowledge support from the Guangxi Natural Science Foundation (2014GXNSFCB118001), National Natural Science Foundation China (21562008), “Western Light” Talent Training Plan of Chinese Academy of Sciences (No. [2013]165), Science Research Foundation of Guangxi Academy of Sciences (15YJ22ZWS22) and Program for Bagui Scholars of Guangxi Institute of Botany (The third batch).
Author Contributions
In this work, Yong-Lin Huang designed the experiments and the manuscript writing; Ya-Feng Wang, Jin-Lei Liu, Yue-Yuan Chen, and Feng-Lai Lu performed the extraction, isolation; Lei Wang and Dian-Peng Li carried out the structural elucidations; Takashi Tanak are viewed the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- CAS. Flora Republicae Popularis Sinicae; China Science Publishing & Media Ltd.: Beijing, China, 1998; Volume 13–14, p. 55. [Google Scholar]
- Suranto. Application of modern experimental technique to solve morphological complexity in plants taxonomy. Biodiversitas 2000, 10, 80–84. [Google Scholar]
- Ageta, M.; Nonaka, G.; Nishioka, I. Tannins and related compounds. LXVII. Isolation and characterization of castanopsinins A–H, novel ellagitannins containing a triterpenoid core, from Castanopsis cuspidata var. sieboldii NAKAI (3). Chem. Pharm. Bull. 1988, 36, 1646–1663. [Google Scholar] [CrossRef]
- Chen, H.F.; Tanaka, T.; Nonaka, G.; Fujoka, T.; Mihsahi, K. Hydrolysable tannins based on a triterpenoid glycoside core, from Castanopsis hystrix. Phytochemistry 1993, 32, 1457–1460. [Google Scholar] [CrossRef]
- Huang, Y.L.; Tsujita, T.; Tanaka, T.; Matsuo, Y.; Kouno, I.; Li, D.P.; Nonaka, G.I. Triterpene hexahydroxydiphenoyl esters and a quinic acid purpurogallin carbonyl ester from the leaves of Castanopsis fissa. Phytochemistry 2011, 72, 2006–2014. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.; Matsuo, Y.; Tanaka, T.; Kouno, I.; Li, D.P.; Nonaka, G.I. New phenylpropanoid-substituted flavan-3-ols from the leaves of Castanopsis sclerophylla. Heterocycles 2011, 83, 2321–2328. [Google Scholar]
- Huang, Y.L.; Tanaka, T.; Matsuo, Y.; Kouno, I.; Li, D.P.; Nonaka, G.I. Two new phenolic glucosides and an ellagitannin from the leaves of Castanopsis sclerophylla. Phytochem. Lett. 2012, 5, 158–161. [Google Scholar] [CrossRef]
- Huang, Y.L.; Tanaka, T.; Matsuo, Y.; Kouno, I.; Li, D.P.; Nonaka, G.I. Isolation of ellagitannin monomer and macrocyclic dimer from Castanopsis carlesii leaves. Heterocycles 2012, 86, 381–389. [Google Scholar]
- Fujii, S.; Aoki, H.; Kômoto, M. Fluorescent glycosides accumulated in Taphrina weisneri-infected Cherry stems. Agric. Biol. Chem. 1971, 35, 1526–1534. [Google Scholar] [CrossRef]
- Yoshi, T.; Fenga, W.S.; Okuda, T. Two polyphenol glycosides and tannins from Rosa cymosa. Phytochemistry 1993, 32, 1033–1036. [Google Scholar] [CrossRef]
- Ohtani, I.; Kusumi, T.; Kashman, Y.; Kakisawa, H. A new aspect of the high-field NMR application of Mosher’s method. The absolute configuration of marine triterpene sipholenol A. J. Am. Chem. Soc. 1991, 113, 4092–4095. [Google Scholar] [CrossRef]
- Kusumi, T.; Ooi, T.; Ohkubo, Y.; Yabuuchi, T. The Modified Mosher’s Method and the Sulfoximine Method. ChemInform 2006, 37, 965–980. [Google Scholar] [CrossRef]
- Duan, W.J.; Jin, X.; Chen, L.X. Four new compounds from Paeonia albiflora. J. Asian Nat. Prod. Res. 2009, 11, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Magid, A.A.; Voutquenne-Nazabadioko, L.; Moroy, G.; Moretti, C.; Lavaud, C. Dihydroisocoumarin glucosides from stem bark of Caryocar glabrum. Phytochemistry 2007, 68, 2439–2443. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Pan, Q.D.; Li, D.P.; Liu, J. L.; Wen, Y.X.; Huang, Y.L.; Lu, F.L. New pregnane glycosides from Brucea javanica and their antifeedant activity. Chem. Biodivers. 2011, 8, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar] [PubMed]
- Ge, Y.Z.; Zhang, H.; Liu, H.C.; Dong, L.; Ding, J.; Yue, J.M. Cytotoxic dinorditer penoids from Drypetes perreticulata. Phytochemistry 2014, 100, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1–4 are available from the authors.
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).