Abstract
Strategically designed carboxylated acyclonucleosides have been probed as a new class of RNase A inhibitors. Several experimental and theoretical studies have been performed to compile relevant qualitative and quantitative information regarding the nature and extent of inhibition. The inhibition constant (Ki) values were determined using a UV-based kinetics experiment. The changes in the secondary structure of the enzyme upon binding with the inhibitors were obtained from circular dichroism studies. The binding constants for enzyme-inhibitor interactions were determined with the help of fluorescence spectroscopy. Docking studies were performed to reveal the possible binding sites of the inhibitors within the enzyme. The cytosine analogues were found to possess better inhibitory properties in comparison to the corresponding uracil derivatives. An increment in the number of carboxylic acid groups (-COOH) in the inhibitor backbone was found to result in better inhibition.
1. Introduction
Ribonucleases are a family of digestive enzymes that degrade RNA [1]. Effective inhibition of their enzymatic activity has become a topic of growing interest with the realization that the detrimental biological activities manifested by certain members of this family [2,3,4,5,6] are critically dependent upon their ribonucleolytic activity [7,8]. Ribonuclease A (RNase A) is a representative member of this family [9,10] that works at the juncture of the transcription and translation processes, thereby maintaining the cellular RNA levels. Structural homology among the various members of this family [11,12,13] permits the use of Ribonuclease A (RNase A) as a model system to explore structure–activity relationships.
Recent reports from our laboratory have revealed that modified nucleoside carboxylic acids manifest RNase A inhibitory properties in a competitive, reversible manner [14,15,16,17,18,19,20,21]. These molecules have an added advantage over the reported phosphate- or pyrophosphate-based nucleotide inhibitors [22,23,24,25,26,27,28,29,30,31] as the polyionic nature of the latter hampers their migration through the cell membrane [32]. The active site of RNase A consists of several subsites made up of polar amino acid residues for specific recognition [9,33] (Figure 1). The cleavage of the phosphodiester bond of RNA at the P1 subsite involves the two His residues (His 12 and His 119) participating in a conjugate acid-base mechanism [34,35]. At physiological pH, the carboxylic acid (-COOH) group(s) in the inhibitor remains deprotonated, and interacts electrostatically with the protonated His and Lys residues present at the ribonucleolytic site [36]. This perturbation of the protonating/deprotonating environment of the P1 subsite results in the inhibition of RNase A.
Following the same hypothesis, it was expected that carboxylated acyclonucleosides may elicit similar inhibitory properties because of their in-built structural features. The absence of the rigid ribose ring further enhances the flexibility of these molecules, and generates additional information on the importance of the furanoside ring. We report the synthesis of several uracil- and cytosine-based modified acyclonucleosides followed by the exploration of their RNase A inhibitory properties. The nucleobases have been carefully selected, as the B1 subsite of RNase A shows preferential recognition towards pyrimidine bases. The acidity of the molecules has been increased via stepwise incorporation of carboxylic acid groups in the molecular framework to study the resulting effect on their inhibition capacities.
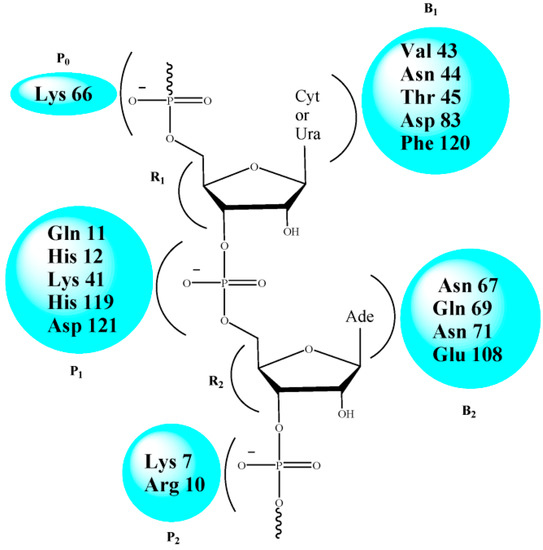
Figure 1.
Key residues of the active site of RNase A.
2. Results and Discussion
2.1. Synthesis of Nucleosides
The uracil-glycine conjugate 3 was synthesized according to a literature reported procedure [37] (Scheme 1). Syntheses of other modified uracil derivatives were achieved via coupling of the known uracil-1-acetic acid 1 [37] with suitable secondary amines, followed by hydrolysis and/or deprotection as required (Scheme 1). The synthesis of the uracil-diethanolamine conjugate 5 [38] was achieved by coupling 1 with the tertbutyldimethyl silyl (TBDMS)-protected diethanolamine, and deprotecting the TBDMS groups of the coupled product 4 with a catalytic amount of CH3COCl in methanol (Scheme 1). TBDMS-protected N-(2-hydroxyethyl)glycine ethyl ester, on EDC-HOBT-mediated coupling with 1, generated compound 6. Deprotection of the TBDMS group produced compound 7, which was converted to the corresponding acid 8 via hydrolysis (Scheme 1). Again, 1 was coupled with diethyl iminodiacetate to afford the corresponding diester 9. The diacid derivative 10 was produced from 9 by base-mediated hydrolysis (Scheme 1).
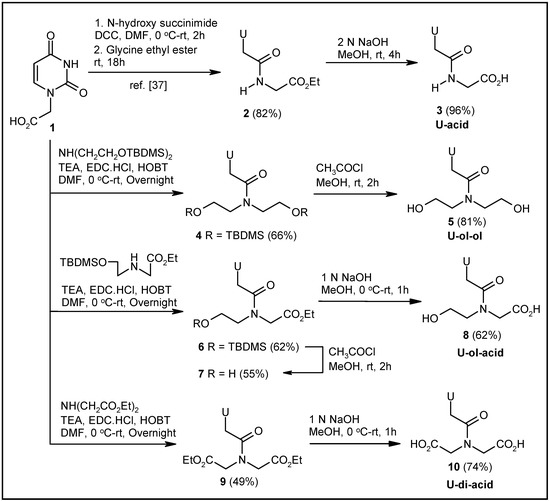
Scheme 1.
Synthesis of uracil-based modified acyclonucleosides.
Syntheses of the corresponding cytosine-based molecules were achieved using the reported cytosine derivative 11 [39] as the starting material (Scheme 2). EDC-HOBT-mediated coupling of glycine ethyl ester to compound 11 produced the coupled product 12. The deprotection of the tertbutyloxy carbonyl (BOC) groups of 12 by TFA resulted in the formation of compound 13. The hydrolysis of the ester group provided the desired acid derivative 14 (Scheme 2). Compound 11, on coupling with TBDMS-protected diethanolamine, generated the coupled derivative 15. TFA treatment of compound 15 afforded the deprotected cytosine-diethanolamine conjugate 16 (Scheme 2). Again, coupling of the TBDMS-protected N-(2-hydroxyethyl)glycine ethyl ester with compound 11, followed by hydrolysis and deprotection of the resulting ester 17, afforded the corresponding hydroxy acid derivative 18 (Scheme 2). Finally, the diacid 21 was obtained from compound 11 following a similar sequence of steps (Scheme 2). Compound 11, on coupling with diethyl iminodiacetate, produced the coupled product 19. Compound 19 was transformed to compound 20 by treatment with TFA. Base-mediated hydrolysis of 20 afforded the desired diacid derivative 21.
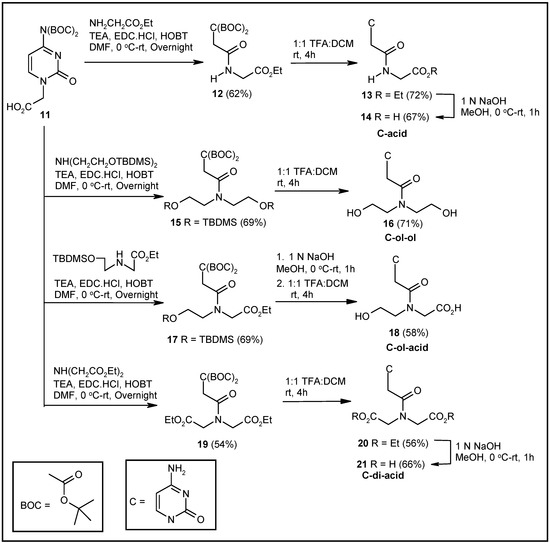
Scheme 2.
Synthesis of cytosine-based modified acyclonucleosides.
2.2. RNase A Inhibition
Qualitative indication of RNase A inhibition by the synthetic inhibitors was obtained from a comparative agarose gel-based assay by monitoring the degradation of RNA by RNase A (Figure 2 and Figure 3). The most intense band observed in lane I in each gel is due to the presence of only RNA. The faint band in lane II is due to the maximum possible degradation of RNA by RNase A. Different intensities of bands from lane III to VI revealed different extents of inhibition by the inhibitors at a fixed concentration (0.5 mM). The histograms obtained by plotting the relative intensities of the bands revealed that compounds U-ol-acid (8), C-ol-acid (18), U-di-acid (10), and C-di-acid (21) were relatively more potent inhibitors in comparison to the others. These experimental observations reaffirmed our assumption that increasing the acidity of the molecules leads to an increase in the inhibitory property of the inhibitor.
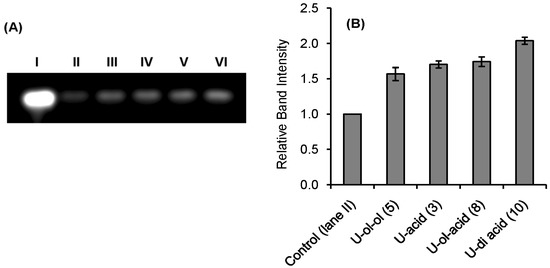
Figure 2.
Agarose gel-based assay for RNase A (1 µM) Inhibition. (A) lane I: RNA (10 mg/mL), lane II: RNA + RNase A, lanes III, IV, V, VI: RNA + RNase A + U-ol-ol (5), U-acid (3), U-ol-acid (8), and U-di-acid (10) (0.5 mmol), respectively. (B) Histogram showing the relative band intensities of agarose gel assay (the data are the mean ± SD).
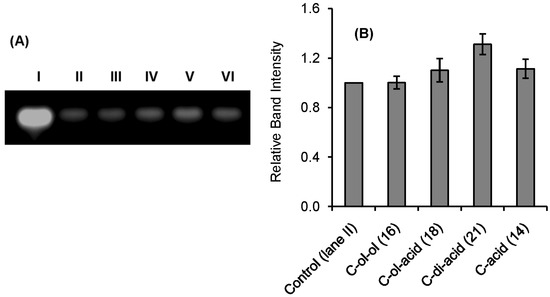
Figure 3.
Agarose gel-based assay for RNase A (1 µM) Inhibition. (A) lane I: RNA (10 mg/mL), lane II: RNA + RNase A, lanes III, IV, V, VI: RNA + RNase A + C-ol-ol (16), C-ol-acid (18), C-di-acid (21), and C-acid (14) (0.5 mmol), respectively. (B) Histogram showing the relative band intensities of agarose gel assay (the data are the mean ± SD).
In order to determine the type of inhibition and the inhibition constant (Ki) values, kinetic experiments were performed with compounds U-ol-acid (8), U-di-acid (10), C-ol-acid (18), and C-di-acid (21). The inhibition constant values are given in Table 1. The competitive nature of inhibition in all the cases was apparent from the nature of the Lineweaver–Burk plots obtained from the kinetic experiments (Figure 4 and Figure S1 (SI)). The numerical order of the inhibition constant (Ki) values indicated that U-di-acid (10) and C-di-acid (21) are the two most potent inhibitors of the series. The results are in good agreement with the results obtained from agarose gel-based assay, suggesting a correlation of inhibitory efficiency with the number of carboxylic acid groups present in the concerned inhibitor. The cytosine analogue C-di-acid (21) was found to possess superior inhibitory property in comparison to the corresponding uracil derivative U-di-acid (10).

Table 1.
Inhibition constants (Ki) of the inhibitors.
| Inhibitor | Ki * (μM) |
|---|---|
| U-ol-acid (8) | 454 ± 9 |
| C-ol-acid (18) | 356 ± 7 |
| U-di-acid (10) | 301 ± 15 |
| C-di-acid (21) | 235 ± 9 |
* The data are the mean ± SD.
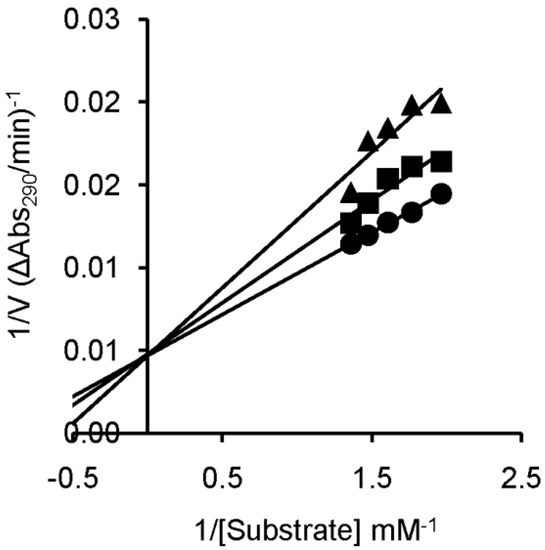
Figure 4.
Lineweaver–Burk plot for inhibition of RNase A by C-di-acid (21) of 0.15 (▲), 0.05 (■), or 0 (●) mM, with 2',3'-cCMP concentrations of 0.75–0.52 mM and RNase A concentration of 9.8 μM.
Inhibitors of RNase A are known to perturb the secondary structure of the enzyme upon binding [40,41,42,43,44]. A model drug–protein interaction study showed that 3'-azido-3'-deoxythymidine increases the α-helix content of RNase A [40]. Similar increments were observed with 3'-O-carboxy esters of thymidine, which inhibited RNase A in reversible competitive mode [42]. Therefore, the probable changes in the secondary structure of the enzyme by the inhibitors were monitored by observing the CD spectra of RNase A in the absence or presence of compounds U-di-acid (10) and C-di-acid (21) (Figure 5). Both the inhibitors induced moderate changes in the secondary structure of the enzyme, which was reflected in the enhanced α-helix content. The α-helix content in native RNase A was 22.7%, which was found to increase upon binding with U-di-acid (10) and C-di-acid (21) to 24.7% and 29.2%, respectively.
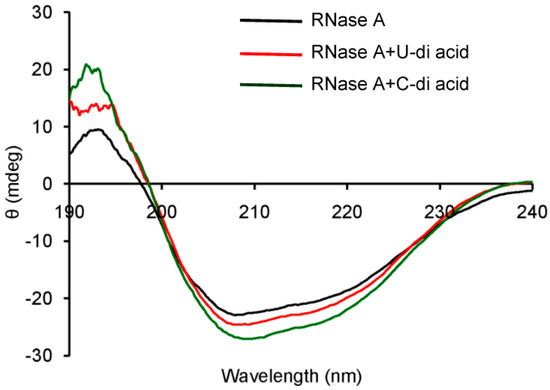
Figure 5.
CD spectra of RNase A in absence or presence of U-di-acid (10) and C-di-acid (21).
The fluorescence emission intensity of RNase A due to the presence of six Tyr residues is found to decrease upon interactions with small molecule inhibitors [40,41,42,43,44]. The emission spectrum of RNase A in presence of U-di-acid (2.10) and C-di-acid (2.21) at 25 °C showed quenching of the fluorescence intensity of Tyr residues (Figure S2, (SI)). The fluorescence quenching study was used to calculate the binding parameters for enzyme-inhibitor interactions. Binding constants (Kb) calculated from the experiment were found to be in the order of 105 for C-di-acid (21) and 104 for U-di-acid (10) (Table S1, (SI)), indicating strong binding of the inhibitors with the enzyme.
To gain an insight into the probable binding sites, protein-ligand docking studies were undertaken. The docked conformations shown in Figure 6 revealed that the nucleobase-amino acid conjugates were in close proximity to the amino acid residues of the P1 subsite, resulting in a competitive mode of inhibition, as observed from the kinetic study. The carboxylic acid (-COOH) groups, in each case, were positioned close to His12 and His 119, probably engaged in hydrogen bonding with the residues. For the cytosine-based inhibitors, the N3 and –NH2 groups of the nucleobase were near to several amino acid residues (Arg39, Lys41, Val43), thus increasing the possibility of polar interactions between them. Such interactions were absent for the uracil-derived inhibitors. These extra interactions probably contribute to the experimentally observed better inhibitory potency of the cytosine inhibitors. Apart from the interactions with the active site residues, one of the carboxylic acid (-COOH) groups of U-di-acid (10) was found to be within hydrogen bonding distance of Oε1 of Gln11. Similarly, one –COOH group of C-di-acid (21) was engaged in hydrogen bonding with Phe120. Such favorable interactions of the –COOH groups may possibly result in the better inhibition capacity of the inhibitors. The docked enzyme-inhibitor complexes also revealed that the nucleobase in the acyclic structure is not in close proximity to the amino acid residues of the pyrimidine binding subsite B1 (Thr45, Asp83, Phe120). It can, therefore, be assumed that the ribose ring in the nucleosides may have a role in recognition of the inhibitors by the enzyme. A detailed account of all the interactions between the inhibitors and the enzyme has been provided in Table S2 (SI).
A further clarification regarding the possible binding sites of the inhibitors was obtained by calculating the changes in accessible surface area (ΔASA) of the interacting residues between the free and complexed forms of the enzyme (Table 2). The measurements revealed that the accessible surface area of the amino acid residues of the P1 subsite (His12, His 119 and Lys 41) was largely affected upon binding with the inhibitors. The observed results correlate well with the results of the docking study, suggesting that the inhibitors do bind to the P1 subsite of RNase A.
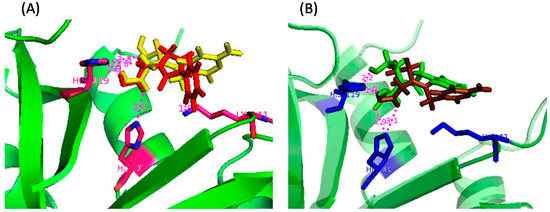
Figure 6.
Docked poses of (A) C-di-acid (21) (yellow) and U-di-acid (10) (red), and (B) U-ol-acid (8) (green) and C-ol-acid (18) (brown) with RNase A (1FS3).

Table 2.
Changes in accessible surface area (ΔASA) of the interacting residues between the uncomplexed and complexed forms of RNase A.
| Amino Acid Residue | ASA (Å2) in RNase A | ΔASA (Å2) for Different Inhibitors | |||
|---|---|---|---|---|---|
| U-ol-Acid (8) | C-ol-Acid (18) | U-di-Acid (10) | C-di-Acid (21) | ||
| Lys 7 | 88.03 | 42.88 | 41.75 | 29.62 | 43.82 |
| His 12 | 12.64 | 7.09 | 7.76 | 9.21 | 7.53 |
| Arg 39 | 142.03 | 25.81 | 29.72 | 5.06 | 30.79 |
| Lys 41 | 36.39 | 26.80 | 31.58 | 33.00 | 31.31 |
| His 119 | 85.05 | 33.62 | 28.29 | 26.69 | 26.64 |
3. Experimental Section
3.1. General Methods
All reagents and fine chemicals were purchased from commercial suppliers and were used without further purification. Column chromatography was performed with silica gel (230–400 mesh). Solvents were dried and distilled following standard methods. TLC was carried out on precoated plates (Merck silica gel 60, f254). 1H and 13C-NMR for the compounds were recorded at 200 and 50 MHz, respectively, using a Bruker NMR instrument. For 1H and 13C-NMR spectra in D2O, CH3CN was used as internal standard. Chemical shifts are reported in parts per million (ppm, δ scale). Methylene carbons have been identified using DEPT spectrum. Melting points were determined in open-end capillary tubes. Bovine pancreatic RNase A, RNA (Torula utilis), and 2',3'-cCMP were purchased from commercial suppliers. UV-vis measurements were made using a UV-vis spectrophotometer (Shimadzu 2450). Concentrations of the solutions were estimated spectrophotometrically using the following data: ε278.5 = 9800 M−1·cm−1 (RNase A) [45] and ε268 = 8500 M−1·cm−1 (2',3'-cCMP) [22]. CD measurements were carried out on a Jasco-810 automatic recording spectrophotometer. Fluorescence measurements were carried out using a Horiba Jobin Yvon Fluoromax-4 Spectrofluorimeter.
3.2. General Procedure for Carboxylic Acid–Amine Coupling Reaction
To a well-stirred solution of carboxylic acid (1, 11) (1 mmol) in DMF (5 mL) at 0 °C was added EDC-HCl (1.2 mmol) followed by triethylamine (1.2 mmol) and the stirring was further continued at this temperature. After 10 min, HOBT (1.2 mmol) was added and the reaction mixture was allowed to warm back to room temperature. Amine (1 mmol) was added to the resulting solution and the stirring was allowed to continue overnight. The reaction mixture was then poured into a brine solution (50 mL) and extracted with EtOAc (3 × 20 mL). Organic extracts were pooled together, dried over anhydrous Na2SO4, filtered, and the filtrate was evaporated under reduced pressure. The resulting residue was purified by column chromatography over silica gel (EtOAc/petroleum ether) to obtain pure coupling products (4, 6, 9, 12, 15, 17, 19).
3.3. General Procedure for Deprotection of –TBDMS Group
To a well-stirred solution of compound (4, 6) (1 mmol) in methanol (10 mL), was added CH3COCl (cat). The resulting solution was allowed to stir for 2 h at room temperature. The solvent was evaporated under reduced pressure and the resulting residue was purified by column chromatography over silica gel (DCM/MeOH) to obtain pure products (5, 7).
3.4. General Procedure for the Deprotection of –BOC Group/Simultaneous Deprotection of –BOC Group and –TBDMS Group
The compound (12, 15, 19) (1 mmol) was dissolved in 1:1 TFA/DCM (5 mL) and the resulting solution was stirred at room temperature for 4 h. The solvent was evaporated under reduced pressure and the resulting residue was purified by column chromatography over silica gel (DCM/MeOH) to obtain pure products (13, 16, 20).
3.5. General Procedure for Ester Hydrolysis
To a well-stirred solution of the compound (7, 9, 13, 20) (1 mmol) in methanol (5 mL) at 0 °C was added 1 N NaOH solution (2 mL) dropwise. The resulting solution was allowed to warm back up to room temperature and stirred for 1 h. After evaporation of methanol under reduced pressure, the resulting residue was dissolved in water (10 mL) and neutralized with acidic amberlyte. The resulting mixture was filtered, and the filtrate was evaporated under reduced pressure. The resulting residue was purified by column chromatography over silica gel (DCM/MeOH) to obtain pure products (8, 10, 14, 21).
N,N-Bis-[2-(tert-butyldimethylsilyloxy)-ethyl]-2-(2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-acetamide (4): Compound 1 (0.68 g, 3.99 mmol) was converted to compound 4 (1.28 g, 66%) following the general procedure in Section 3.2. Rf = 0.4 [30% EtOAc in pet ether]. White solid. M.P: 122–124 °C. 1H-NMR (200 MHz, CDCl3): δ 0.02 (s, 6H), 0.06 (s, 6H), 0.86 (s, 9H), 0.88 (s, 9H), 3.47 (t, J = 5.5 Hz, 2H), 3.59 (t, J = 5.1 Hz, 2H), 3.71–3.80 (m, 4H), 4.66 (s, 2H), 5.69 (d, J = 8.0 Hz, 1H), 7.11 (d, J = 7.8 Hz, 1H), 9.28 (bs, 1H). 13C-NMR (50 MHz, CDCl3): δ −5.3, −5.2, 18.3, 18.5, 26.0, 26.1, 48.1 (CH2), 49.2 (CH2), 51.1 (CH2), 61.0(CH2), 61.5 (CH2), 102.1, 145.4, 151.2, 163.9, 167.0. HRMS (ESI+): m/z calcd for C22H44N3O5Si2 (M+H)+: 486.2820; found: 486.2827.
2-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-N,N-bis-(2-hydroxy-ethyl) acetamide (5): Compound 5 [38] (0.39 g, 81%) was obtained from compound 4 (0.9 g, 1.85 mmol) following the general procedure in Section 3.3. White solid. Rf = 0.4 [15% MeOH in DCM]. 1H-NMR (200 MHz, DMSO-d6): δ 3.31–3.58 (m, 8H), 4.67 (s, 2H), 4.75–4.80 (m, 1H), 5.01–5.05 (m, 1H), 5.54 (d, J = 7.8 Hz, 1H), 7.47 (d, J = 7.8 Hz, 1H), 11.23 (bs, 1H).
{[2-(tert-Butyldimethylsilyloxy) ethyl]-[2-(2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl-acetyl] amino}acetic acid ethyl ester (6): Compound 1 (0.93 g, 5.47 mmol) was converted to compound 6 (1.40 g, 62%) following the general procedure in Section 3.2. Rf = 0.3 [40% EtOAc in pet ether]. White solid. M.P: 171–172 °C. The compound was obtained as a mixture of rotamers as indicated by the NMR spectra. 1H-NMR (200 MHz, CDCl3): δ 0.01 (s, 6H), 0.06 (s, 6H), 0.86 (s, 9H), 0.87 (s, 9H), 1.20–1.32 (m, 3H), 3.52–3.56 (m, 2H), 3.70–3.81 (m, 2H), 4.10–4.24 (m, 4H), 4.30 (s, 2H), 4.46 (s, 2H), 4.71 (s, 2H), 5.68–5.73 (m, 1H), 7.13–7.26 (m, 1H), 9.39 (bs, 1H), 9.51 (bs, 1H). 13C-NMR (50 MHz, CDCl3): δ −5.3, 14.3, 18.4, 26.0, 26.1, 47.9 (CH2), 48.6 (CH2), 50.8 (CH2), 61.4 (CH2), 61.6 (CH2), 62.2 (CH2), 102.3, 145.4, 151.1, 163.9, 167.5, 168.9, 172.9. HRMS (ESI+): m/z calcd for C18H32N3O6Si (M+H)+: 414.2060; found: 414.2088.
[[2-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl-acetyl]-(2-hydroxyethyl)-amino] acetic acid ethyl ester (7): Compound 7 (0.48 g, 55%) was generated from compound 6 (1.21 g, 2.90 mmol) following the general procedure in Section 3.3. Rf = 0.4 [10% MeOH in DCM]. Light yellow gum. The compound was obtained as a mixture of rotamers as indicated by the NMR spectra. 1H-NMR (200 MHz, DMSO-d6): δ 1.14–1.26 (m, 3H), 3.34–3.58 (m, 4H), 4.01–4.18 (m, 4H), 4.34 (s, 2H), 4.53 (s, 2H), 4.74 (s, 2H), 5.53–5.57 (m, 1H), 7.34–7.45 (m, 1H), 11.25 (bs, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 14.0, 48.0 (CH2), 49.9 (CH2), 50.7 (CH2), 58.3 (CH2), 58.9 (CH2), 60.5 (CH2), 61.03(CH2), 100.6, 146.5, 151.0, 163.9, 167.4, 167.5, 169.1, 169.4. HRMS (ESI+): m/z calcd for C12H17N3O6Na (M+Na)+: 322.1015; found: 322.1003.
[[2-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl-acetyl]-(2-hydroxyethyl) amino] acetic acid (8): Compound 7 (0.4 g, 1.34 mmol) was converted to compound 8 (0.22 g, 62%) following the general procedure in Section 3.5. Rf = 0.2 [20% MeOH in DCM]. White solid. M.P: 177–179 °C. The compound was obtained as a mixture of rotamers as indicated by the NMR spectra. 1H-NMR (200 MHz, D2O): δ 3.54–3.82 (m, 4H), 4.19 (s, 2H), 4.37 (s, 2H), 4.57 (s, 2H), 4.90 (s, 2H), 5.80–5.87 (m, 1H), 7.50–7.56 (m, 1H). 13C-NMR (50 MHz, D2O): δ 49.2 (CH2), 50.0 (CH2), 50.6 (CH2), 51.0 (CH2), 59.3 (CH2), 59.4 (CH2), 102.2, 148.1, 152.6, 167.2, 170.0, 173.3. HRMS (ESI+): m/z calcd for C10H13N3O6Na (M+Na)+: 294.0702; found: 294.0707.
{[2-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl-acetyl] ethoxycarbonylmethylamino}acetic acid ethyl ester (9): Compound 1 (0.52 g, 3.05 mmol) was transformed to compound 9 (0.51 g, 49%) following the general procedure in Section 3.2. Rf = 0.5 [70% EtOAc in pet ether]. White solid. M.P: 140–142 °C. 1H-NMR (200 MHz, CDCl3): δ 1.22–1.34 (m, 6H), 4.12–4.31 (m, 8H), 4.61 (s, 2H), 5.73 (d, J = 8.0 Hz, 1H), 7.20 (d, J = 7.8 Hz, 1H), 9.09 (bs, 1H). 13C-NMR (50 MHz, CDCl3): δ 14.3, 47.7 (CH2), 49.0 (CH2), 50.2 (CH2), 61.8 (CH2), 62.5 (CH2), 102.6, 145.2, 151.0, 163.7, 167.6, 168.6. HRMS (ESI+): m/z calcd for C14H19N3O7Na (M+Na)+: 364.1121; found: 364.1109.
{Carboxymethyl-[2-(2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl) acetyl] amino}acetic acid (10): Compound 9 (0.42 g, 1.23 mmol) was converted to compound 10 (0.26 g, 74%) following the general procedure in Section 3.5. Rf = 0.2 [30% MeOH in DCM]. White solid. M.P: 118–120 °C 1H-NMR (200 MHz, DMSO-d6): δ 4.00 (s, 2H), 4.25 (s, 2H), 4.62 (s, 2H), 5.55 (d, J = 7.2 Hz, 1H), 7.43 (d, J = 7.6 Hz, 1H), 11.28 (s, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 47.7 (CH2), 48.4 (CH2), 49.2 (CH2), 100.7, 146.4, 151.0, 163.8, 167.7, 170.3. HRMS (ESI+): m/z calcd for C10H11N3O7Na (M+Na)+: 308.0495; found: 308.0475.
{[4-[Bis(1,1-dimethylethoxy)carbonyl]amino-2-oxo-2H-pyrimidin-1-yl] acetylamino}acetic acid ethyl ester (12): Compound 11 (0.39 g, 1.05 mmol) was converted to compound 12 (0.30 g, 62%) following the general procedure in Section 3.2. Rf = 0.5 [60% EtOAc in pet ether]. White solid. M.P: 118–120 °C. 1H-NMR (200 MHz, CDCl3): δ 1.18–1.25 (m, 3H), 1.52 (s, 18H), 3.95 (d, J = 5.6 Hz, 2H), 4.12 (q, J = 7.2 Hz, 2H), 4.58 (s, 2H), 7.08 (d, J = 7.2 Hz, 1H), 7.69–7.76 (m, 1H). 13C-NMR (50 MHz, CDCl3): δ 14.1, 27.7, 41.4 (CH2), 52.8 (CH2), 61.4 (CH2), 85.0, 96.7, 149.0, 149.4, 152.6, 155.5, 162.7, 167.1, 169.4. HRMS (ESI+): m/z calcd for C20H30N4O8Na (M+Na)+: 477.1961; found: 477.1975.
[2-(4-Amino-2-oxo-2H-pyrimidin-1-yl) acetylamino] acetic acid ethyl ester (13): Compound 12 (0.28 g, 0.62 mmol) was converted to compound 13 (0.11 g, 72%) following the general procedure in Section 3.4. Rf = 0.3 [5% MeOH in DCM]. White solid. M.P: 155–158 °C. 1H-NMR (200 MHz, DMSO-d6): δ 1.17 (t, J = 7.2 Hz, 3H), 3.86 (d, J = 5.8 Hz, 2H), 4.07 (q, J = 7.2 Hz, 2H), 4.42 (s, 2H), 5.82 (d, J = 7.2 Hz, 1H), 7.65 (d, J = 7.4 Hz, 1H), 7.90 (bs, 1H), 8.09 (bs, 1H), 8.61 (t, J = 5.8 Hz, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 14.5, 41.2 (CH2), 50.9 (CH2), 61.0 (CH2), 93.8, 148.8, 153.5, 164.4, 168.0, 170.0. HRMS (ESI+): m/z calcd for C10H15N4O4 (M+H)+: 255.1093; found: 255.1076.
[2-(4-Amino-2-oxo-2H-pyrimidin-1-yl) acetylamino] acetic acid (14): Compound 13 (0.09 g, 0.35 mmol) was transformed to compound 14 (0.05 g, 67%) following the general procedure in Section 3.5. Rf = 0.3 [20% MeOH in DCM]. Yellowish white solid. M.P: > 200 °C. 1H-NMR (200 MHz, DMSO-d6): δ 3.61–3.95 (m, 2H), 4.54 (s, 2H), 6.22 (d, J = 7.2 Hz, 1H), 7.99 (d, J = 7.4 Hz, 1H), 8.77 (s, 1H), 8.94 (s, 1H), 10.17 (s, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 40.8 (CH2), 50.4 (CH2), 93.1, 147.4, 150.6, 160.1, 166.4, 170.8. HRMS (ESI+): m/z calcd for C8H11N4O4 (M+H)+: 227.0780; found: 227.0784.
[4-[Bis(1,1-dimethylethoxy)carbonyl]amino-2-oxo-2H-pyrimidin-1-yl]-N,N-bis-[2(tertbutyldimethyl silyloxy)ethyl]acetamide (15): Compound 15 (0.55 g, 69%) was obtained from compound 11 (0.43 g, 1.16 mmol) following the general procedure in Section 3.2. Rf = 0.3 [25% EtOAc in pet ether]. Colourless gum. 1H-NMR (200 MHz, CDCl3): δ −0.03 (s, 6H), 0.01 (s, 6H), 0.81 (s, 9H), 0.83 (s, 9H), 1.49 (s, 18H), 3.43 (t, J = 5.6 Hz, 2H), 3.64–3.74 (m, 6H), 4.70 (s, 2H), 7.00 (d, J = 7.4 Hz, 1H), 7.52 (d, J = 7.4 Hz, 1H). 13C-NMR (50 MHz, CDCl3): δ −5.4, 18.2, 18.3, 26.0, 27.7, 49.3 (CH2), 49.5 (CH2), 51.1 (CH2), 61.0 (CH2), 61.2 (CH2), 84.8, 96.1, 149.1, 149.6, 155.1, 162.6, 167.1. HRMS (ESI+): m/z calcd for C32H61N4O8Si2 (M+H)+: 685.4028; found: 685.4038.
2-(4-Amino-2-oxo-2H-pyrimidin-1-yl)-N,N-bis-(2-hydroxyethyl) acetamide (16): Compound 15 (0.48 g, 0.70 mmol) was transformed to compound 16 (0.13 g, 71%) following the general procedure in Section 3.4. Rf = 0.3 [15% MeOH in DCM]. White solid. M.P: 150 °C (decomposed). 1H-NMR (200 MHz, DMSO-d6): δ 2.99–3.04 (m, 1H), 3.34–3.68 (m, 8H), 4.65 (s, 2H), 5.78 (d, J = 7.2 Hz, 1H), 7.51 (d, J = 7.2 Hz, 1H), 7.75 (bs, 2H). 13C-NMR (50 MHz, DMSO-d6): δ 48.8 (CH2), 49.3 (CH2), 49.9 (CH2), 58.7 (CH2), 59.0 (CH2), 93.2, 148.1, 154.4, 164.8, 167.3. HRMS (ESI+): m/z calcd for C10H17N4O4 (M+H)+: 257.1250; found: 257.1255.
{[4-[Bis(1,1-dimethylethoxy)carbonyl]amino-2-oxo-2H-pyrimidin-1-yl-acetyl]-[2-(tert-butyldimethylsilyloxy) ethyl] acetic acid ethyl ester (17): Compound 11 (0.46 g, 1.24 mmol) was converted to compound 17 (0.52 g, 69%) following the general procedure in Section 3.2. Rf = 0.4 [40% EtOAc in pet ether]. Colorless gum. The compound was obtained as a mixture of rotamers as indicated by the NMR spectra. 1H-NMR (200 MHz, CDCl3): δ −0.03–0.01 (m, 6H), 0.81 (s, 9H), 0.83 (s, 9H), 1.15–1.27 (m, 3H), 1.49 (s, 18H), 3.33–3.80 (m, 4H), 4.05–4.18 (m, 4H), 4.40 (s, 2H), 4.49 (s, 2H), 4.75 (s, 2H), 6.97–7.03 (m, 1H), 7.54–7.62 (m, 1H). 13C-NMR (50 MHz, CDCl3): δ −5.4, 14.2, 18.2, 25.9, 27.7, 49.0 (CH2), 49.3 (CH2), 51.1 (CH2), 51.2 (CH2), 61.2 (CH2), 61.6 (CH2), 61.8 (CH2), 62.0 (CH2), 84.8, 96.2, 149.0, 149.1, 149.5, 155.0, 162.7, 162.8, 167.4, 167.5, 168.8, 169.7. HRMS (ESI+): m/z calcd for C28H49N4O9Si (M+H)+: 613.3269; found: 613.3278.
[[2-(4-Amino-2-oxo-2H-pyrimidin-1-yl) acetyl]-(2-hydroxyethyl) amino] acetic acid (18): Compound 17 (0.40 g, 0.65 mmol) was hydrolyzed to corresponding acid following the general procedure in Section 3.5. The crude residue obtained was subjected to TFA treatment as mentioned in the general procedure in Section 3.4, and purified by column chromatography over silica gel to obtain 18 (0.10 g, 58%). Rf = 0.2 [30% MeOH in DCM]. White solid. M.P: 184–188 °C. The compound was obtained as a mixture of rotamers, as indicated by the NMR spectra. 1H-NMR (200 MHz, DMSO-d6): δ 3.35–3.59 (m, 4H), 4.01 (s, 2H), 4.24 (s, 2H), 4.54 (s, 2H) 4.75 (s, 2H), 5.84 (d, J = 7.0 Hz, 1H), 7.55 (d, J = 7.0 Hz, 1H), 7.97 (bs, 2H). 13C-NMR (50 MHz, DMSO-d6): δ 47.9 (CH2), 49.1 (CH2), 49.9 (CH2), 58.9 (CH2), 93.4, 148.6, 152.8, 163.6, 167.6, 170.7, 171.1. HRMS (ESI+): m/z calcd for C10H15N4O5 (M+H)+: 271.1042; found: 271.1039.
{[4-[Bis(1,1-dimethylethoxy)carbonyl]amino-2-oxo-2H-pyrimidin-2-yl-acetyl] ethoxycarbonylmethylamino}acetic acid ethyl ester (19): Compound 19 (0.29 g, 54%) was generated from compound 11 (0.37 g, 1.00 mmol) following the general procedure in Section 3.2. Rf = 0.4 [50% EtOAc in pet ether]. Colorless gum. 1H-NMR (200 MHz, CDCl3): δ 1.17–1.30 (m, 6H), 1.51 (s, 18H), 4.07–4.25 (m, 6H), 4.32 (s, 2H), 4.67 (s, 2H), 7.05 (d, J = 7.4 Hz, 1H), 7.62 (d, J = 7.4 Hz, 1H). 13C-NMR (50 MHz, CDCl3): δ 14.2, 27.8, 49.1 (CH2), 49.4 (CH2), 50.5 (CH2), 61.6 (CH2), 62.1 (CH2), 85.0, 96.5, 149.0, 149.5, 155.1, 162.8, 167.9, 168.7, 168.9. HRMS (ESI+): m/z calcd for C24H37N4O10 (M+H)+: 541.2510; found: 541.2516.
{[2-(4-Amino-2-oxo-2H-pyrimidin-1-yl) acetyl] ethoxycarbonylmethylamino}acetic acid ethyl ester (20): Compound 19 (0.27 g, 0.50 mmol) was converted to compound 20 (0.09 g, 56%) following the general procedure in Section 3.4. Rf = 0.3 [3% MeOH in DCM]. White solid. M.P: 152–154 °C. 1H-NMR (200 MHz, DMSO-d6): δ 1.14–1.27 (m, 6H), 4.00–4.22 (m, 6H), 4.39 (s, 2H), 4.80 (s, 2H), 6.10 (d, J = 7.6 Hz, 1H), 7.81 (d, J = 7.6 Hz, 1H), 9.52 (bs, 1H), 9.67 (bs, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 14.0, 48.5 (CH2), 49.1 (CH2), 60.6 (CH2), 61.1 (CH2), 93.5, 148.2, 150.2, 160.6, 167.3, 168.5, 168.8. HRMS (ESI+): m/z calcd for C14H21N4O6 (M+H)+: 341.1461; found: 341.1452.
{[2-(4-Amino-2-oxo-2H-pyrimidin-1-yl)-acetyl] carboxymethylamino}acetic acid (21): Compound 20 (0.07 g, 0.20 mmol) was transformed to compound 21 (0.04 g, 66%) following the general procedure in Section 3.5. Rf = 0.15 [40% MeOH in DCM]. Eluent: 30%–50% MeOH in DCM. White solid. M.P: 190–193 °C. 1H-NMR (200 MHz, D2O): δ 4.23 (s, 2H), 4.39 (s, 2H), 4.89 (s, 2H), 6.24 (d, J = 7.4 Hz, 1H), 7.79 (d, J = 7.8 Hz, 1H). 13C-NMR (50 MHz, DMSO-d6): δ 48.8 (CH2), 49.3 (CH2), 49.9 (CH2), 93.4, 148.4, 152.2, 163.5, 167.8, 170.4, 170.7. HRMS (ESI+): m/z calcd for C10H13N4O6 (M+H)+: 285.0835; found: 285.0852.
3.6. Comparative Agarose Gel-Based Assay
Inhibition of RNase A was assayed qualitatively by the degradation of RNA in an agarose gel. In this method, 20 μL of RNase A (1 μM) was mixed with 20 μL (0.5 mM) of compounds U-ol-ol (5), C-ol-ol (16), U-acid (3), C-acid (14), U-ol-acid (8), C-ol-acid (18), U-di-acid (10), and C-di-acid (21) separately to a final volume of 50 μL and the resulting solutions incubated for 3 h. Twenty-microliter aliquots of the incubated mixtures were then mixed with 20 μL of RNA solution (10.0 mg/mL RNA, freshly dissolved in RNase free water) and incubated for another 30 min. Then 10 μL of sample buffer (containing 10% glycerol and 0.025% bromophenol blue) were added to this mixture and 15 μL from each solution were extracted and loaded onto a 1.1% agarose gel. The gel was run using a 0.04-M Tris-Acetic acid-EDTA (TAE) buffer (pH 8.0). The residual RNA was visualized by ethidium bromide staining under UV light.
3.7. Inhibition Kinetics with RNase A
A quantitative account of RNase A inhibition by the individual inhibitors was obtained by a UV spectroscopic method described by Anderson and co-workers [22]. The assay was performed in a 0.1-M Mes-NaOH buffer, pH 6.0 containing 0.1 M NaCl using 2',3'-cCMP as the substrate. The inhibition constants were calculated from initial velocity data using a Lineweaver–Burk plot. The slopes from the Lineweaver–Burk double reciprocal plot were plotted against the corresponding inhibitor concentrations to get inhibition constants (Ki).
3.8. Circular Dichroism Measurements
Circular Dichroism (CD) was performed in order to monitor the changes in the secondary structure of the enzyme as a result of interaction with the inhibitors. In this method, 200 µL of RNase A (30 µM) were mixed separately with 200 µL of U-di-acid (10) (30 µM) and C-di-acid (21) (30 µM) and incubated for 3 h. From the resulting solutions, an aliquot of 300 µL was used for CD measurements, taking a 1 mm path length quartz cell. The spectra were recorded in the range of 190–240 nm with a scan rate of 50 nm/min. Three scans were accumulated for each spectrum. The secondary structure was determined using an online server, DICHROWEB [46].
3.9. Fluorescence Spectroscopy
Fluorescence quenching study was performed to garner an idea of the binding affinities of U-di-acid (10) and C-di-acid (21) towards RNase A. The emission spectra were recorded from 290 to 400 nm with excitation at 275 nm [47] using a 5 nm slit width. The interaction between the ligands and RNase A was investigated by titration of 3 mL solution of RNase A with successive addition of the respective ligands (0–15 μM) in a 20-mM phosphate buffer of pH 7.0. Binding constants (Kb) were calculated using double-logarithm plot [48].
3.10. FlexX Docking
The crystal structure of RNase A (PDB entry 1FS3) was downloaded from the Protein Data Bank [49]. The 3D structures of the inhibitors were generated in Sybyl6.92 (Tripos Inc., St. Louis, MO, USA). Minimum energy conformations were obtained with the help of the MMFF94 force field using MMFF94 charges with a gradient of 0.005 kcal/mole by 1000 iterations with all other default parameters. The ligands were docked with the protein using FlexX software. The ranking of the generated solutions was performed using a scoring function that estimates the free binding energy (ΔG) of the protein-ligand complex considering various types of molecular interactions [50]. Docked conformations were visualized using PyMol [51].
3.11. Accessible Surface Area Calculations
Accessible surface area of uncomplexed RNase A and its docked complexes were calculated using the program NACCESS. The structures obtained from the FlexX analysis were used for the calculation. The change in ASA for a particular residue X was calculated using: ΔASAX = ASAXRNase A − ASAXRNase A + inhibitor.
4. Conclusions
Strategically designed carboxylated acyclonucleosides have been established as moderate RNase A inhibitors. The experimental outcome points towards the possible contribution of a sugar ring in RNase A inhibition. Cytosine analogues have been proven to have better inhibitory properties than the corresponding uracil derivatives. C-di-acid (21) emerged as the most potent inhibitor of the series, having an inhibition constant (Ki) value of 235 μM. It was observed that an increment in the number of carboxylic acid groups resulted in better inhibitory properties. However, the absence of the rigid ribose ring in the molecules significantly affects the inhibition properties of the synthetic nucleosides. These findings should act as a guideline for future design of inhibitors for RNase A and other members of the ribonuclease superfamily.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/20/04/5924/s1.
Acknowledgments
The authors thank the Department of Biotechnology, Ministry of Science and Technology, New Delhi for funding (project no. SB/S1/OC-30/2014). Kaustav Chakraborty thanks D. Tripathy and S. Ghosh for their help in some of the experiments and the Council for Scientific and Industrial Research, New Delhi for a fellowship.
Author Contributions
S.D.G and T.P conceived the idea and designed the study. K.C performed the synthetic and biological experiments, and analyzed the data. K.C, S.D.G, and T.P wrote the manuscript together. All authors read and approved the final version of the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- D’Alessio, G. The superfamily of vertebrate-secreted ribonucleases. In Ribonucleases; Nicholson, A.W., Ed.; Springer: Heidelberg, Germany, 2011; pp. 1–34. [Google Scholar]
- Loverix, S.; Steyaert, J. Ribonucleases: from prototypes to therapeutic targets. Curr. Med. Chem. 2003, 10, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Viola, M.; Libra, M.; Callari, D.; Sinatra, F.; Spada, D.; Noto, D.; Emmanuele, G.; Romano, F.; Averna, M.; Pezzino, F.M.; et al. Bovine seminal ribonuclease is cytotoxic for both malignant and normal telomerase-positive cells. Int. J. Oncol. 2005, 27, 1071–1077. [Google Scholar] [PubMed]
- Zrinski, R.T.; Dodig, S. Eosinophil cationic protein-current concept and controversies. Biochem. Med. 2011, 21, 111–121. [Google Scholar] [CrossRef]
- Li, S.; Ibaragi, S.; Hu, G. Angiogenin as a molecular target for the treatment of prostate cancer. Curr. Cancer Ther. Rev. 2011, 7, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Fang, E.F.; Ng, T.B. Ribonucleases of different origins with a wide spectrum of medicinal applications. Biochim. Biophys. Acta 2011, 1815, 65–74. [Google Scholar] [PubMed]
- Leland, P.A.; Schultz, L.W.; Kim, B.; Raines, R.T. Ribonuclease A variants with potent cytotoxic activity. Proc. Natl. Acad. Sci. USA 1998, 95, 10407–10412. [Google Scholar] [CrossRef] [PubMed]
- Leland, P.A.; Staniszewski, K.E.; Park, C.; Kelemen, B.R.; Raines, R.T. The ribonucleolytic activity of angiogenin. Biochemistry 2002, 41, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Raines, R.T. Ribonuclease A. Chem. Rev. 1998, 98, 1045–1065. [Google Scholar] [CrossRef] [PubMed]
- Marshall, G.; Feng, J.A.; Kuster, D.J. Back to the future: ribonuclease A. Biopolymers 2007, 90, 259–277. [Google Scholar] [CrossRef]
- Shapiro, R.; Weremowicz, S.; Riordan, J.F.; Vallee, B.L. Ribonucleolytic activity of angiogenin: Essential histidine, lysine, and arginine residues. Proc. Natl. Acad. Sci. USA 1987, 84, 8783–8787. [Google Scholar] [CrossRef] [PubMed]
- Hamann, K.J.; Barker, R.L.; Loegering, D.A.; Pease, L.R.; Gleich, G.L. Sequence of human eosinophil-derived neurotoxin cDNA: Identity of deduced amino acid sequence with human nonsecretory ribonucleases. Gene 1989, 83, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Gagné, D.; Charest, L.; Morin, S.; Kovrigin, E.L.; Doucet, N. Conservation of flexible residue clusters among structural and functional enzyme homologues. J. Biol. Chem. 2012, 287, 44289–44300. [Google Scholar] [CrossRef] [PubMed]
- Maiti, T.K.; De, S.; Dasgupta, S.; Pathak, T. 3'-N-Alkylamino-3'-deoxy-ara-uridines: A new class of potential inhibitors of ribonuclease A and angiogenin. Bioorg. Med. Chem. 2006, 14, 1221–1228. [Google Scholar] [CrossRef]
- Leonidas, D.D.; Maiti, T.K.; Samanta, A.; Dasgupta, S.; Pathak, T.; Zographos, S.E.; Oikonomakos, N.G. The binding of 3'-N-piperidine-4-carboxyl-3'-deoxy-ara-uridine to ribonuclease A in the crystal. Bioorg. Med. Chem. 2006, 14, 6055–6066. [Google Scholar] [CrossRef] [PubMed]
- Debnath, J.; Dasgupta, S.; Pathak, T. Inhibition of ribonuclease A by nucleoside-dibasic acid conjugates. Bioorg. Med. Chem. 2009, 17, 6491–6496. [Google Scholar] [CrossRef] [PubMed]
- Debnath, J.; Dasgupta, S.; Pathak, T. Comparative inhibitory activity of 3'- and 5'-functionalized nucleosides on ribonuclease A. Bioorg. Med. Chem. 2010, 18, 8257–8263. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Dasgupta, S.; Pathak, T. 5'-modified pyrimidine nucleosides as inhibitors of ribonuclease A. Bioorg. Med. Chem. 2009, 17, 6491–6496. [Google Scholar] [CrossRef] [PubMed]
- Datta, D.; Samanta, A.; Dasgupta, S.; Pathak, T. 3'-Oxo-, amino-, thio-, and sulfone-acetic acid modified thymidines: Effect of increased acidity on ribonuclease A inhibition. Bioorg. Med. Chem. 2013, 21, 4634–4645. [Google Scholar] [CrossRef] [PubMed]
- Datta, D.; Samanta, A.; Dasgupta, S.; Pathak, T. Synthesis of 5'-carboxymethylsulfonyl-5'-deoxyribonucleosides under mild hydrolytic conditions: A new class of acidic nucleosides as inhibitors of ribonuclease A. RSC Adv. 2014, 4, 2214–2218. [Google Scholar] [CrossRef]
- Datta, D.; Dasgupta, S.; Pathak, T. Ribonuclease A inhibition by carboxymethylsulfonyl-modified xylo- and arabinopyrimidines. ChemMedChem 2014, 9, 2138–2149. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.G.; Hammes, G.G.; Walz, F.G. Binding of phosphate ligands to ribonuclease A. Biochemistry 1968, 7, 1637–1645. [Google Scholar] [CrossRef] [PubMed]
- Walz, F.G. Kinetic and equilibrium studies on the interaction of ribonuclease A and 2'-deoxyuridine 3'-phosphate. Biochemistry 1971, 10, 2156–2162. [Google Scholar] [CrossRef] [PubMed]
- Russo, N.; Shapiro, R.; Vallee, B.L. 5'-Diphosphoadenosine-3'-phosphate is a potent inhibitor of bovine pancreatic ribonuclease A. Biochem. Biophys. Res. Commun. 1997, 231, 671–674. [Google Scholar] [CrossRef] [PubMed]
- Leonidas, D.D.; Shapiro, R.; Irons, L.I.; Russo, N.; Acharya, K.R. Crystal structures of ribonuclease A complexes with 5'-diphosphoadenosine 3'-phosphate and 5'-diphosphoadenosine 2'-phosphate at 1.7 Å resolution. Biochemistry 1997, 36, 5578–5588. [Google Scholar] [CrossRef] [PubMed]
- Leonidas, D.D.; Shapiro, R.; Irons, L.I.; Russo, N.; Acharya, K.R. Towards rational design of ribonuclease inhibitors: High resolution crystal structure of a ribonuclease A complex with a potent 3',5'-pyrophosphate-linked dinucleotide inhibitor. Biochemistry 1999, 38, 10287–10297. [Google Scholar] [CrossRef] [PubMed]
- Russo, N.; Shapiro, R. Potent inhibition of mammalian ribonucleases by 3',5'-pyrophosphate-linked nucleotides. J. Biol. Chem. 1999, 274, 14902–14908. [Google Scholar] [CrossRef] [PubMed]
- Leonidas, D.D.; Chavali, G.B.; Oikonomakos, N.G.; Chrysina, E.D.; Kosmopoulou, M.N.; Vlassi, M.; Frankling, C.; Acharya, K.R. High-resolution crystal structures of ribonuclease A complexed with adenylic and uridylic nucleotide inhibitors. Implications for structure-based design of ribonucleolytic inhibitors. Protein Sci. 2003, 12, 2559–2574. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Jenkins, J.L.; Jardine, A.M.; Shapiro, R. Inhibition of mammalian ribonucleases by endogenous adenosine dinucleotides. Biochem. Biophys. Res. Commun. 2003, 300, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, C.L.; Thiyagarajan, N.; Sweeney, R.Y.; Guy, M.P.; Kelemen, B.R.; Acharya, K.R.; Raines, R.T. Binding of non-natural 3'-nucleotides to ribonuclease A. FEBS J. 2005, 272, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Hatzopoulos, G.N.; Leonidas, D.D.; Kardakaris, R.; Kobe, J.; Oikonomakos, N.G. The binding of IMP to ribonuclease A. FEBS J. 2005, 272, 3988–4001. [Google Scholar] [CrossRef] [PubMed]
- Yakovlev, G.I.; Mitkevich, V.A.; Makarov, A.A. Ribonuclease inhibitors. Mol. Biol. 2006, 40, 867–874. [Google Scholar] [CrossRef]
- Nogués, M.V.; Vilanova, M.; Cuchillo, C.M. Bovine pancreatic ribonuclease A as a model of an enzyme with multiple substrate binding sites. Biochim. Biophys. Acta 1995, 1253, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Findlay, D.; Herries, D.G.; Mathias, A.P.; Rabin, B.R.; Ross, C.A. The active site and mechanism of action of bovine pancreatic ribonuclease. Nature 1961, 190, 781–784. [Google Scholar] [CrossRef] [PubMed]
- Cuchillo, C.M.; Nogués, M.V.; Raines, R.T. Bovine pancreatic ribonuclease: Fifty years of the first enzymatic reaction mechanism. Biochemistry 2011, 50, 7835–7841. [Google Scholar] [CrossRef] [PubMed]
- Silverman, R.B. The Organic Chemistry of Drug Design and Drug Action, 2nd ed.; Elsevier: San Diego, CA, USA, 2004; p. 126. [Google Scholar]
- Liu, X.; Chen, R. Synthesis of novel phosphonotripeptides containing uracil or thymine group. Phosphorus Sulfur Silicon Relat. Elem. 2001, 176, 19–28. [Google Scholar] [CrossRef]
- Rezazgui, O.; Boëns, B.; Teste, K.; Vergaud, J.; Trouillas, P.; Zerrouki, R. One-pot and catalyst-free amidation of ester: A matter of non-bonding interactions. Tetrahedron Lett. 2011, 52, 6796–6799. [Google Scholar] [CrossRef]
- Porcheddu, A.; Giacomelli, G.; Piredda, I.; Carta, M.; Nieddu, G. A practical and efficient approach to PNA monomers compatible with Fmoc-mediated solid-phase synthesis protocols. Eur. J. Org. Chem. 2008, 34, 5786–5797. [Google Scholar] [CrossRef]
- Gaudreau, S.; Novetta-dellen, A.; Neault, J.F.; Diamantoglou, S.; Tajmir-riahi, H.A. 3'-azido-3'-deoxythymidine binding to ribonuclease A: Model for drug-protein interaction. Biopolymers 2003, 72, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.S.; Maiti, T.K.; Mandal, A.; Dasgupta, S. Copper complexes of (-)-epicatechin gallate and (-)-epigallactocatechin gallate act as inhibitors of ribonuclease A. FEBS Lett. 2006, 580, 4703–4708. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.S.; Debnath, J.; Dutta, P.; Sahoo, B.K.; Dasgupta, S. Exploring the potential of 3'-O-carboxy esters of thymidine as inhibitors of ribonuclease A and angiogenin. Bioorg. Med. Chem. 2008, 16, 2819–2828. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Basak, A.; Dasgupta, S. Synthesis and ribonuclease A inhibition activity of resorcinol and phloroglucinol derivatives of catechin and epicatechin: Importance of hydroxyl groups. Bioorg. Med. Chem. 2010, 18, 6538–6546. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, D.R.; Roy, A.S.; Dasgupta, S. Complex formation of rutin and quercetin with copper alters the mode of inhibition of ribonuclease A. FEBS Lett. 2011, 585, 3270–3276. [Google Scholar] [CrossRef] [PubMed]
- Sela, M.; Anfinsen, C.B. Some spectrophotometric and polarimetric experiments with ribonucleases. Biochim. Biophys. Acta 1957, 24, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, L.; Wallace, B.A. DICHROWEB, an online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic Acids Res. 2004, 32, 668–673. [Google Scholar] [CrossRef]
- Garcia-Borron, J.C.; Escribano, J.; Jimenez, M.; Iborra, J.L. Quantitative determination of tryptophanyl and tyrosyl residues of proteins by second-derivative fluorescence spectroscopy. Anal. Biochem. 1982, 125, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Xie, M.X.; Zheng, D.; Liu, Y.; Li, X.Y.; Cheng, X. Spectroscopic studies on the interaction of cinnamic acid and its hydroxyl derivatives with human serum albumin. J. Mol. Struct. 2004, 692, 71–80. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The protein data bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Rarey, M.; Kramer, B.; Lengauer, T.; Klebe, G. A fast flexible docking method using an incremental construction algorithm. J. Mol. Biol. 1996, 261, 470–489. [Google Scholar] [CrossRef] [PubMed]
- DeLano, W.L. The PyMOL Molecular Graphics System; DeLano Scientific: San Carlos, CA, USA, 2004. Available online: http://pymol.sourceforge.net/.
- Sample Availability: Samples are not available from authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).