Enzymatic Cellulose Hydrolysis: Enzyme Reusability and Visualization of β-Glucosidase Immobilized in Calcium Alginate
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of Immobilization Conditions on Residual Activity
| BG (mg/mL) | BSA (mg/mL) | Glutaraldehyde (%) | |
|---|---|---|---|
| A | 7.33 | 0 | 0.75 |
| B | 0.5 | ||
| C | 0.25 | ||
| D | 0 | ||
| E | 3 | 0.75 | |
| F | 0.5 | ||
| G | 0.25 |

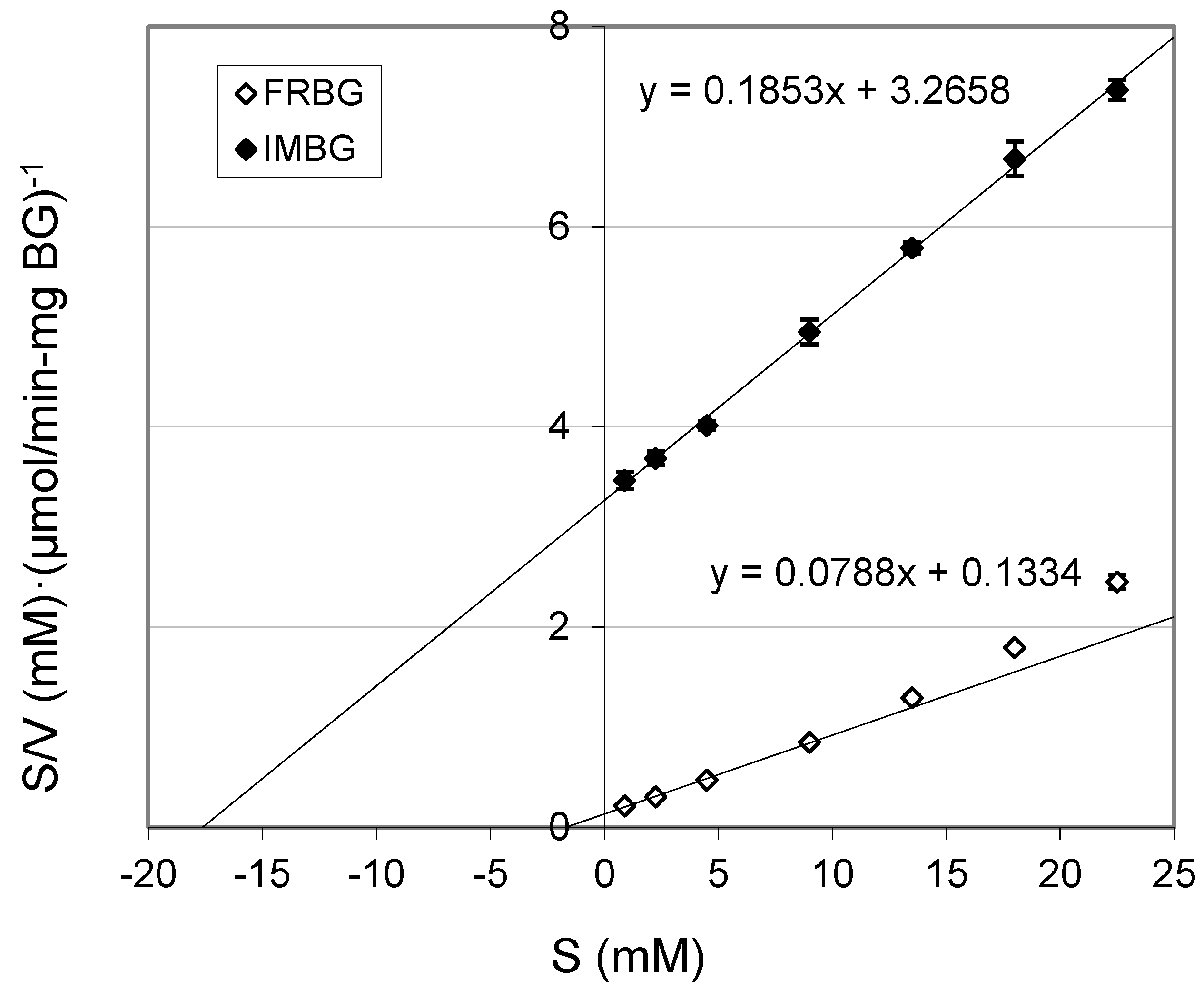
2.2. Km and Vmax of BG
| Km (mM) | Vmax (μmol/(min·mg BG)) | |
|---|---|---|
| FRBG | 1.70 ± 0.12 | 12.71 ± 0.26 |
| IMBG | 17.62 ± 0.11 | 5.39 ± 0.06 |
2.3. Repeated Hydrolysis and Stability of the Recycled, Immobilized Enzyme

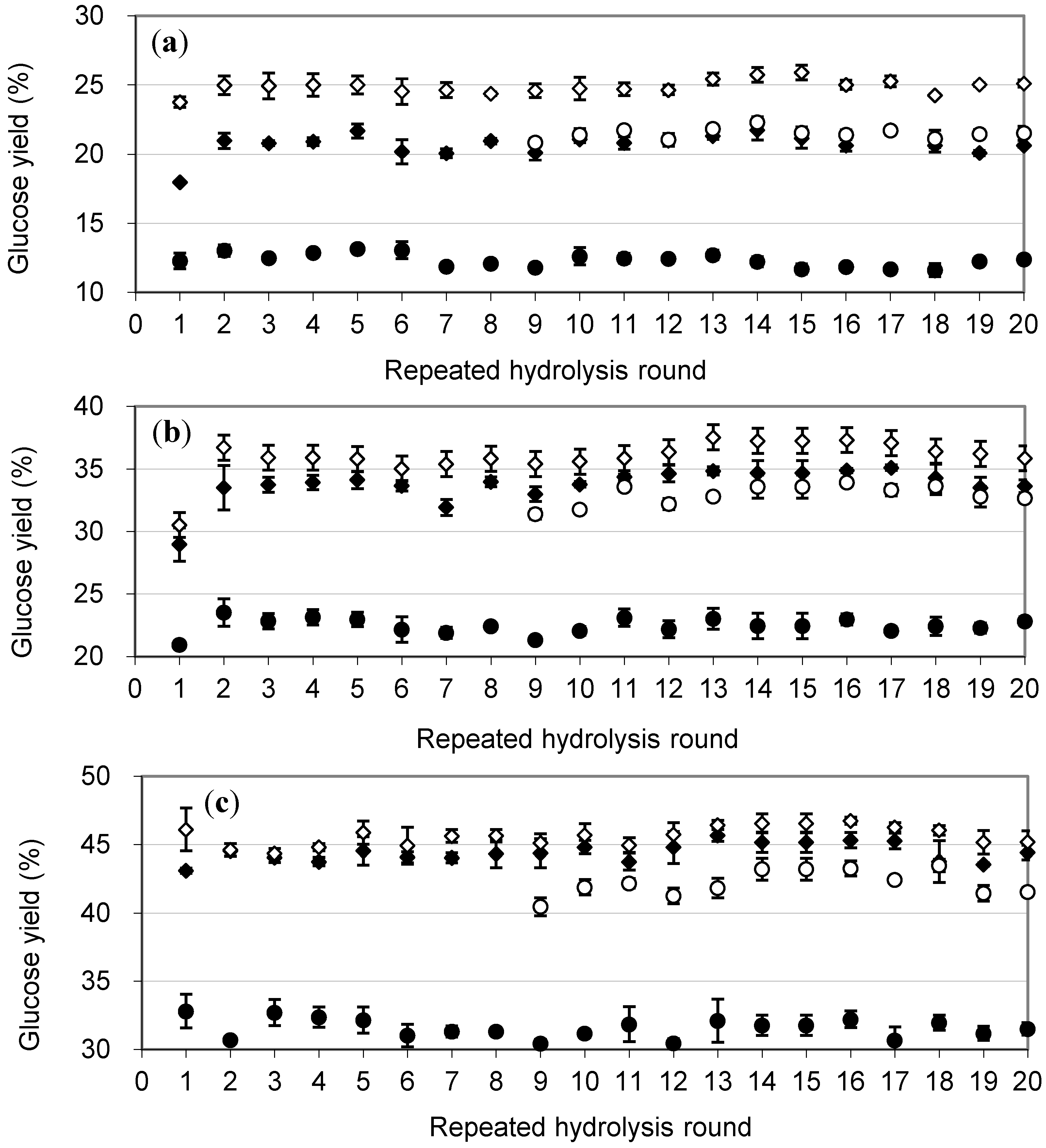
2.4. Enzymatic Hydrolysis of Hydrothermally Pretreated Barley Straw

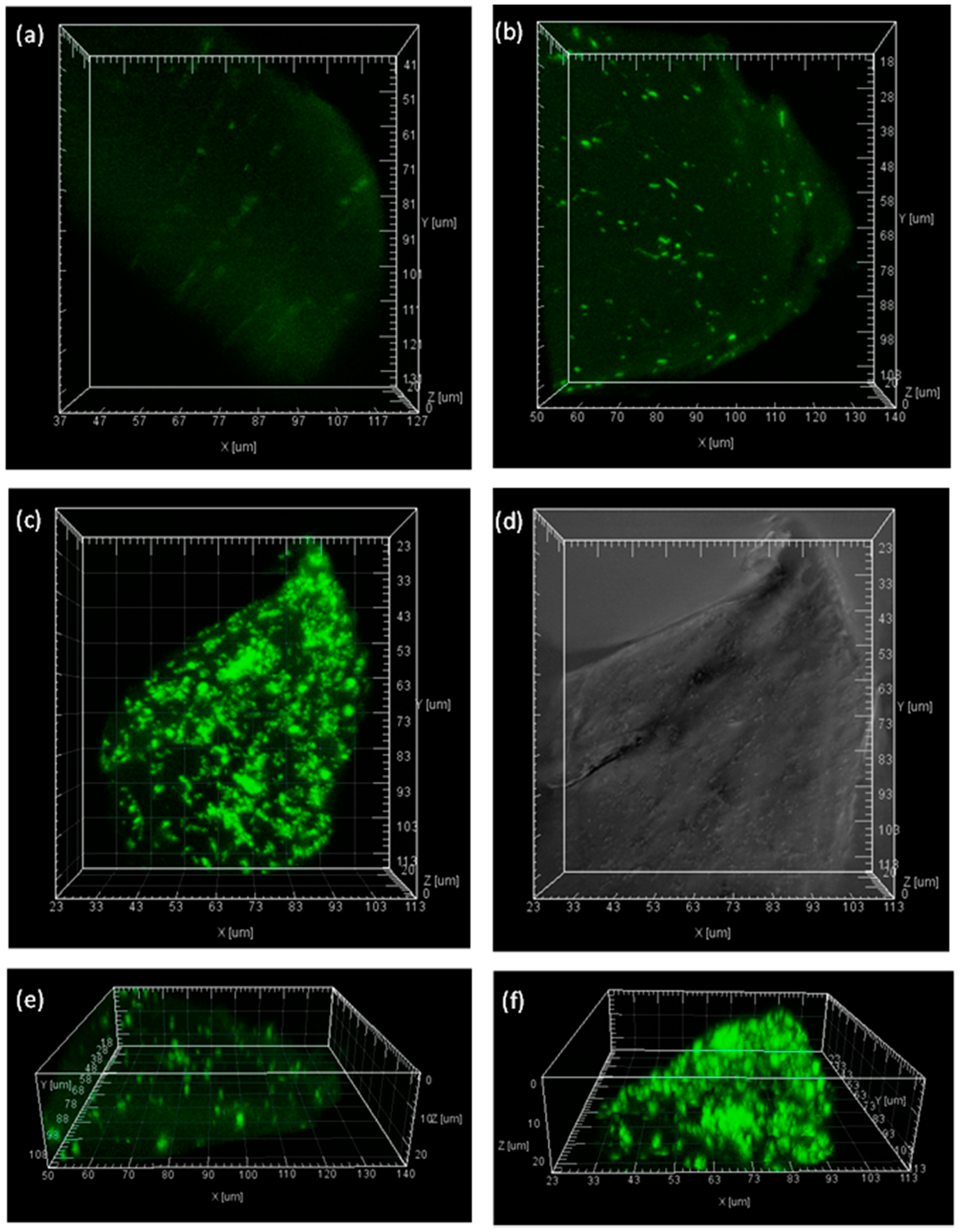
2.5. Visualization of the Immobilized BG inside Calcium Alginate Particles
3. Experimental Section
3.1. Chemicals and Enzymes
3.2. Immobilization of β-Glucosidase
3.3. Analysis of Residual Activity
3.4. Determination of Km and Vmax of BG
3.5. Repeated Hydrolysis for Evaluating the Stability of the Recycled Enzyme
3.6. Hydrothermal Pre-Treatment of Barley Straw
3.7. Enzymatic Hydrolysis of Hydrothermally Pre-Treated Barley Straw
3.8. Visualization of Enzyme Distribution in Calcium Alginate by Confocal Laser Scanning Microscopy
4. Conclusions
Acknowledgments
Author Contributions
Abbreviations
| BG | (β-glucosidase) |
| IMBG | (immobilized β-glucosidase) |
| FRBG | (free β-glucosidase) |
| CLSM | (Confocal laser scanning microscopy) |
| GA | (glutaraldehyde) |
| BSA | (bovine serum albumin) |
Conflicts of Interest
References and Notes
- Merino, S.T.; Cherry, J. Progress and challenges in enzyme development for biomass utilization. Adv. Biochem. Eng. Biotechnol. 2007, 108, 95–120. [Google Scholar] [PubMed]
- Vinzant, T.B.; Adney, W.S.; Decker, S.R.; Baker, J.O.; Kinter, M.T.; Sherman, N.E.; Fox, J.W.; Himmel, M.E. Fingerprinting Trichoderma reesei hydrolases in a commercial cellulase preparation. Appl. Biochem. Biotechnol. 2001, 91–93, 99–107. [Google Scholar] [CrossRef]
- Häkkinen, M.; Arvas, M.; Oja, M.; Aro, N.; Penttilä, M.; Saloheimo, M.; Pakula, T.M. Re-annotation of the CAZy genes of Trichoderma reesei and transcription in the presence of lignocellulosic substrates. Microb. Cell Fact. 2012, 11, 134–159. [Google Scholar] [CrossRef]
- Martinez, D.; Berka, R.M.; Henrissat, B.; Saloheimo, M.; Arvas, M.; Baker, S.E.; Chapman, J.; Chertkov, O.; Coutinho, P.M.; Cullen, D.; et al. Genome sequencing and analysis of the biomass-degrading fungus Trichoderma reesei (syn. Hypocrea jecorina). Nat. Biotechnol. 2008, 26, 553–560. [Google Scholar] [CrossRef]
- Levasseur, A.; Drula, E.; Lombard, V.; Coutinho, P.M.; Henrissat, B. Expansion of the enzymatic repertoire of the CAZy database to integrate auxiliary redox enzymes. Biotechnol. Biofuels 2013, 6, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Carbohydrate-Active enZYmes Database. Available online: www.cazy.org (accessed on 21 November 2014).
- Andric, P.; Meyer, A.S.; Jensen, P.A.; Dam-Johansen, K. Reactor design for minimizing product inhibition during enzymatic lignocellulose hydrolysis: I. Significance and mechanism of cellobiose and glucose inhibition on cellulolytic enzymes. Biotechnol. Adv. 2010, 28, 308–324. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Fan, L.T. Kinetic studies of enzymatic hydrolysis of insoluble cellulose II. Analysis of extended hydrolysis times. Biotechnol. Bioeng. 1983, 25, 939–966. [Google Scholar] [CrossRef] [PubMed]
- Rosgaard, L.; Pedersen, S.; Langston, J.; Akerhielm, D.; Cherry, J.R.; Meyer, A.S. Evaluation of minimal Trichoderma reesei cellulase mixtures on differently pretreated barley straw substrates. Biotechnol. Prog. 2007, 23, 1270–1276. [Google Scholar] [CrossRef]
- Aden, A.; Faust, T. Technoeconomic analysis of the dilute sulfuric acid and enzymatic hydrolysis process for the conversion of corn stover to ethanol. Cellulose 2009, 16, 535–545. [Google Scholar] [CrossRef]
- Morales-Rodriguez, R.; Meyer, A.S.; Gernaey, K.V.; Sin, G. A framework for modelbased optimization of bioprocesses under uncertainty: Lignocellulosic ethanol production case. Comput. Chem. Eng. 2012, 42, 115–129. [Google Scholar] [CrossRef]
- Billard, H.; Faraj, A.; Ferreira, N.L.; Menir, S.; Heiss-Blanquet, S. Optimization of a synthetic mixture composed of major Trichoderma reesei enzymes for the hydrolysis of steam-exploded wheat straw. Biotechnol. Biofuels 2012, 5, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Qin, Y.; Li, Z.; Qu, Y. Development of highly efficient, low-cost lignocellulolytic enzyme systems in the post-genomic era. Biotechnol. Adv. 2013, 31, 962–975. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.; Zhang, X.; Paice, M.; MacFarlane, P.; Saddler, J.N. The potential of enzyme recycling during the hydrolysis of a mixed softwood feedstock. Bioresour. Technol. 2009, 100, 6407–6415. [Google Scholar] [CrossRef] [PubMed]
- Weiss, N.; Börjesson, J.; Pedersen, L.H.; Meyer, A.S. Enzymatic lignocellulose hydrolysis: Improved cellulase productivity by insoluble solids recycling. Biotechnol. Biofuels 2013, 6, 5–19. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Fernandez-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galan, C.; Berenguer-Murcia, A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Syn. Cat. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Guzik, U.; Hupert-Kocurek, K.; Wojcieszynska, D. Immobilization as a strategy for improving enzyme properties—Application to oxidoreductases. Molecules 2014, 19, 8995–9018. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, D.B.; da Rocha-Leão, M.H.M. Immobilization of β-glucosidase aggregates in calcium alginate. Biomass Bioenergy 1991, 1, 213–216. [Google Scholar] [CrossRef]
- Busto, M.D.; Ortega, N.; Perez-Mateos, M. Studies of microbial β-d-glucosidase immobilized in alginate gel beads. Process Biochem. 1995, 30, 421–426. [Google Scholar]
- Shen, X.; Xia, L. Production and immobilization of cellobiase from Aspergillus niger ZU-07. Process Biochem. 2004, 39, 1363–1367. [Google Scholar] [CrossRef]
- Ortega, N.; Busto, M.D.; Perez-Mateos, M. Optimisation of β-glucosidase entrapment in alginate and polyacrylamide gels. Bioresour. Technol. 1998, 64, 105–111. [Google Scholar] [CrossRef]
- Busto, M.D.; Ortega, N.; Perez-Mateos, M. Stabilisation of cellulases by cross-linking with glutaraldehyde and soil humates. Bioresour. Technol. 1997, 60, 27–33. [Google Scholar] [CrossRef]
- Calsavara, L.P.V.; Moraes, F.F.D.; Zanin, G.M. Comparison of catalytic properties of free and immobilized cellobiase Novozym 188. Appl. Biochem. Biotechnol. 2001, 91–93, 615–626. [Google Scholar] [CrossRef]
- Woodward, J.; Krasniak, S.R.; Smith, R.D.; Spielberg, F. Kinetic properties of β-glucosidase preparations immobilized in calcium alginate gel spheres. Biotechnol. Bioeng. Symp. 1982, 12, 485–489. [Google Scholar]
- Tanaka, H.; Matsumura, M.; Veliky, A. Diffusion characteristics of substrates in Ca-alginate gel beads. Biotechnol. Bioeng. 1984, 26, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: Behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 2004, 37, 790–802. [Google Scholar] [PubMed]
- Wine, Y.; Cohen-Hadar, N.; Freeman, A.; Frolow, F. Elucidation of the mechanism and end products of glutaraldehyde crosslinking reaction by X-ray structure analysis. Biotechnol. Bioeng. 2007, 98, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Adv. 2014, 4, 1583–1600. [Google Scholar]
- Woodward, J.; Capps, K.M. Cellobiose hydrolysis by glutaraldehyde-treated β-glucosidase entrapped in propylene glycol alginate/bone gelatin spheres. Appl. Biochem. Biotechnol. 1992, 34/35, 341–347. [Google Scholar] [CrossRef]
- Broun, G.B. Chemically aggregated enzymes. In Methods in Enzymology; Mosbach, K., Ed.; Academic Press: New York, NY, USA, 1976; pp. 263–280. [Google Scholar]
- Busto, M.D.; Ortega, N.; Perez-Mateos, M. Effect of immobilization on the stability of bacterial and fungal β-d-glucosidase. Process Biochem. 1997, 32, 441–449. [Google Scholar] [CrossRef]
- Baker, J.O.; Oh, K.K.; Grohmann, K.; Himmel, M.E. Thermal stabilization of fungal β-glucosidase through glutaraldehyde crosslinking. Biotechnol. Lett. 1988, 10, 325–330. [Google Scholar] [CrossRef]
- Chaplin, M.F.; Bucke, C. Enzyme Technology; Cambridge University Press: Cambridge, UK, 1990; pp. 80–137. [Google Scholar]
- Rosgaard, L.; Pedersen, S.; Cherry, J.R.; Harris, P.; Meyer, A.S. Efficiency of new fungal cellulose systems in boosting enzymatic degradation of barley straw lignocellulose. Biotechnol. Prog. 2006, 22, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Ghose, T.K. Measurement of cellulase activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar]
- Rosgaard, L.; Pedersen, S.; Meyer, A.S. Comparison of different pretreatment strategies for enzymatic hydrolysis of wheat and barley straw. Appl. Biochem. Biotechnol. 2007, 143, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; NREL Technical Report 2011, NREL/TP-510-42618 (Version 07.08.2011); National Renewable Energy Laboratory: Boulder, Colorado, 2011. [Google Scholar]
- Tsai, C.T.; Morales-Rodriguez, R.; Sin, G.; Meyer, A.S. A dynamic model for cellulosic biomass hydrolysis: A comprehensive analysis and validation of hydrolysis and product inhibition mechanisms. Appl. Biochem. Biotechnol. 2014, 172, 2815–2837. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not applicable.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, C.-T.; Meyer, A.S. Enzymatic Cellulose Hydrolysis: Enzyme Reusability and Visualization of β-Glucosidase Immobilized in Calcium Alginate. Molecules 2014, 19, 19390-19406. https://doi.org/10.3390/molecules191219390
Tsai C-T, Meyer AS. Enzymatic Cellulose Hydrolysis: Enzyme Reusability and Visualization of β-Glucosidase Immobilized in Calcium Alginate. Molecules. 2014; 19(12):19390-19406. https://doi.org/10.3390/molecules191219390
Chicago/Turabian StyleTsai, Chien-Tai, and Anne S. Meyer. 2014. "Enzymatic Cellulose Hydrolysis: Enzyme Reusability and Visualization of β-Glucosidase Immobilized in Calcium Alginate" Molecules 19, no. 12: 19390-19406. https://doi.org/10.3390/molecules191219390
APA StyleTsai, C.-T., & Meyer, A. S. (2014). Enzymatic Cellulose Hydrolysis: Enzyme Reusability and Visualization of β-Glucosidase Immobilized in Calcium Alginate. Molecules, 19(12), 19390-19406. https://doi.org/10.3390/molecules191219390





