Abstract
Phytochemical investigation of an ethanolic extract of stems of Schisandra neglecta led to the isolation and identification of two new dibenzocyclooctadiene lignans, designated neglschisandrins E (1) and F (2), and thirteen known lignans. All structures and stereochemistries were determined by spectroscopic methods, including 2D-NMR techniques. The isolates were evaluated for in vitro cytotoxic activity. Among them, compounds 2–6 exhibited moderate to weak cytotoxicity against the human colorectal carcinoma HCT-8 cell line with EC50 values of 7.33~19.8 μg/mL. In addition, compounds 2–4 also exhibited marginal cytotoxicity against the human lung carcinoma A549 cell line with EC50 values of 11.8~15.0 μg/mL.
1. Introduction
Stems or fruits of plants in the Schisandraceae family are used widely in China as tonic and astringent agents for the treatment of rheumatic arthritis, traumatic injury, and related diseases [1]. Schisandraceae plants are rich in lignans, especially dibenzocyclooctadiene lignans, which show beneficial pharmacological effects, including anti-HIV, antitumor-promoting, calcium antagonistic, and anti-lipid peroxidative actions [2,3,4,5,6,7,8,9,10,11,12,13].
In our previous study, we reported four new dibenzocyclooctadiene lignans from the stems of Schisandra neglecta, which is indigenous to the Tibet Autonomous Region of China [14,15]. Our further investigation of the same plant has now led to the isolation and identification of two new dibenzocyclooctadiene lignans, named neglschisandrins E (1) and F (2) (Figure 1), together with thirteen known lignans, 6-O-benzoylgomisin O (3) [16], (+)-γ-rubschisandrin (4) [17], rubschisantherin (5) [17], benzoylisogomisin O (6) [18], schisandrin A (7) [19], schisanhenol (8) [20], angeloylgomisin H (9) [21], gomisin H (10) [21], tigloylgomisin H (11) [21], benzoylgomisin H (12) [21], schisandrin (13) [22], gomisin B (14) [23], and angeloyl-(+)-gomisin K3 (15) [24]. This paper reports the isolation and structural elucidation of these compounds as well as their in vitro cytotoxicity against human lung carcinoma A549 and human colorectal carcinoma HCT-8 cell lines.
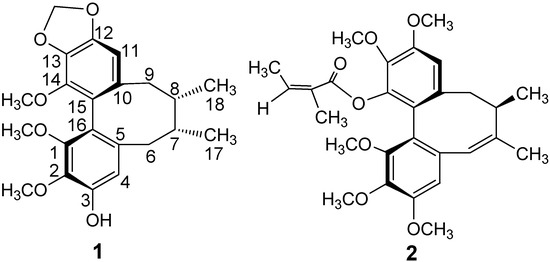
Figure 1.
Structures of Compounds 1–2.
Figure 1.
Structures of Compounds 1–2.
2. Results and Discussion
A 95% alcoholic extract prepared from stems of Schisandra neglecta was partitioned between diethyl ether and water. The diethyl ether extract was subjected to silica gel column chromatography (CC), preparative TLC, and semi-preparative reversed phase HPLC to give the two new and thirteen known dibenzocyclooctadiene lignans named above.
Compound 1, obtained as amorphous powder, had the molecular formula C22H26O6 based on HR-ESI-MS (m/z 387.1810 [M+H]+). The UV and NMR spectra indicated that 1 was a dibenzocyclooctadiene lignan [25]. The 1H-NMR spectrum of 1 (Table 1) showed signals due to two secondary methyl groups (δH 0.97, 0.73, each 3H, d, J = 7.1 Hz), assignable to the cis-oriented Me-7 and Me-8, respectively [26]. The presence of four benzylic methylene signals (δH 2.25, 1H, dd, J = 13.0, 9.6; 2.01 Hz, 1H, d, J = 13.0 Hz; 2.55, 1H, dd, J = 13.6, 7.4; 2.45 Hz, 1H, dd, J = 13.6, 1.5 Hz) indicated that C-6 and C-9 were unsubstituted. The 1H-NMR spectrum also showed signals for two aromatic protons (δH 6.63, 6.49 each 1H, s), assignable to CH-4 and CH-11, respectively. Three methoxy groups [δH 3.50, 3.79 and 3.93, each 3H, s) and a methylenedioxy (–OCH2O–) moiety (δH 5.96, 5.95, each 1H, d, J = 1.3 Hz] were also observed as substituents on the aromatic rings. The proton signal at δH 5.68 (br s) showed no correlations in the HMQC spectrum, and was assigned as an hydroxyl on the aromatic ring. This assignment was supported by an IR band at 3447 cm−1 [27].

Table 1.
1H- (400 MHz) and 13C- (100 MHz) NMR data (measured in CDCl3) for compounds 1 and 2 (δ in ppm, J in Hz).
| 1 | 2 | |||
|---|---|---|---|---|
| Position | δC | δH | δC | δH |
| S | ||||
| 1 | 150.3 (s) | - | 152.3 (s) | - |
| 2 | 137.4 (s) | - | 139.9 (s) | - |
| 3 | 148.8 (s) | - | 152.6 (s) | - |
| 4 | 110.2 (d) | 6.63 (s) | 106.3 (d) | 6.38 (s) |
| 5 | 140.3 (s) | - | 136.5 (s) | - |
| 6 | 35.1 (t) | 2.25 (dd, J = 9.6,13.0) | 125.3 (t) | 6.20 (s) |
| 2.01 (d, J = 13.0) | - | |||
| 7 | 40.9 (d) | 1.79 (m) | 142.2 (s) | - |
| 8 | 33.8 (d) | 1.88 (m) | 34.2 (d) | 2.79 (m) |
| 9 | 38.9 (t) | 2.55 (dd, J = 13.6,7.4) | 42.9 (t) | 3.10 (dd, J = 9,15.1) |
| 2.45 (dd, J = 13.6,1.5) | 2.38 (dd, J = 10.0,15.1) | |||
| 10 | 132.7 (s) | - | 135.3 (s) | - |
| 11 | 106.2 (d) | 6.49 (s) | 111.5 (d) | 6.57 (s) |
| 12 | 147.7 (s) | - | 151.6 (s) | - |
| 13 | 135.1 (s) | - | 139.3 (s) | - |
| 14 | 141.3 (s) | - | 142.5 (s) | - |
| 15 | 122.5 (s) | - | 124.1 (s) | - |
| 16 | 121.3 (s) | - | 122.1 (s) | - |
| 17 | 21.8 (q) | 0.97 (d, J = 7.1) | 18.9 (q) | 1.63 (s) |
| 18 | 12.4 (q) | 0.73 (d, J = 7.1) | 19.4 (q) | 1.03 (d, J = 6.9) |
| MeO-C (1) | 60.1 (q) | 3.50 (s) | 60.7 (q) | 3.61 (s) |
| MeO-C (2) | 61.0 (q) | 3.93 (s) | 60.8 (q) | 3.84 (s) |
| MeO-C (3) | - | - | 55.9 (q) | 3.83 (s) |
| MeO-C (12) | - | - | 55.9 (q) | 3.88 (s) |
| MeO-C (13) | - | - | 60.7 (q) | 3.80 (s) |
| MeO-C (14) | 59.7 (q) | 3.79 (s) | - | - |
| HO-C (3) | 5.68 (brs) | - | ||
| OCH2O | 100.8 (t) | 5.96, 5.95 (d, J = 1.3) | - | - |
| Ang:1' | - | - | 165.2 (s) | - |
| 2' | - | - | 127.8 (s) | - |
| 3' | - | - | 136.8 (d) | 5.86 (m) |
| 4' | - | - | 15.3 (q) | 1.78 (s) |
| 5' | - | - | 20.3 (q) | 1.72 (d, J = 7.1) |
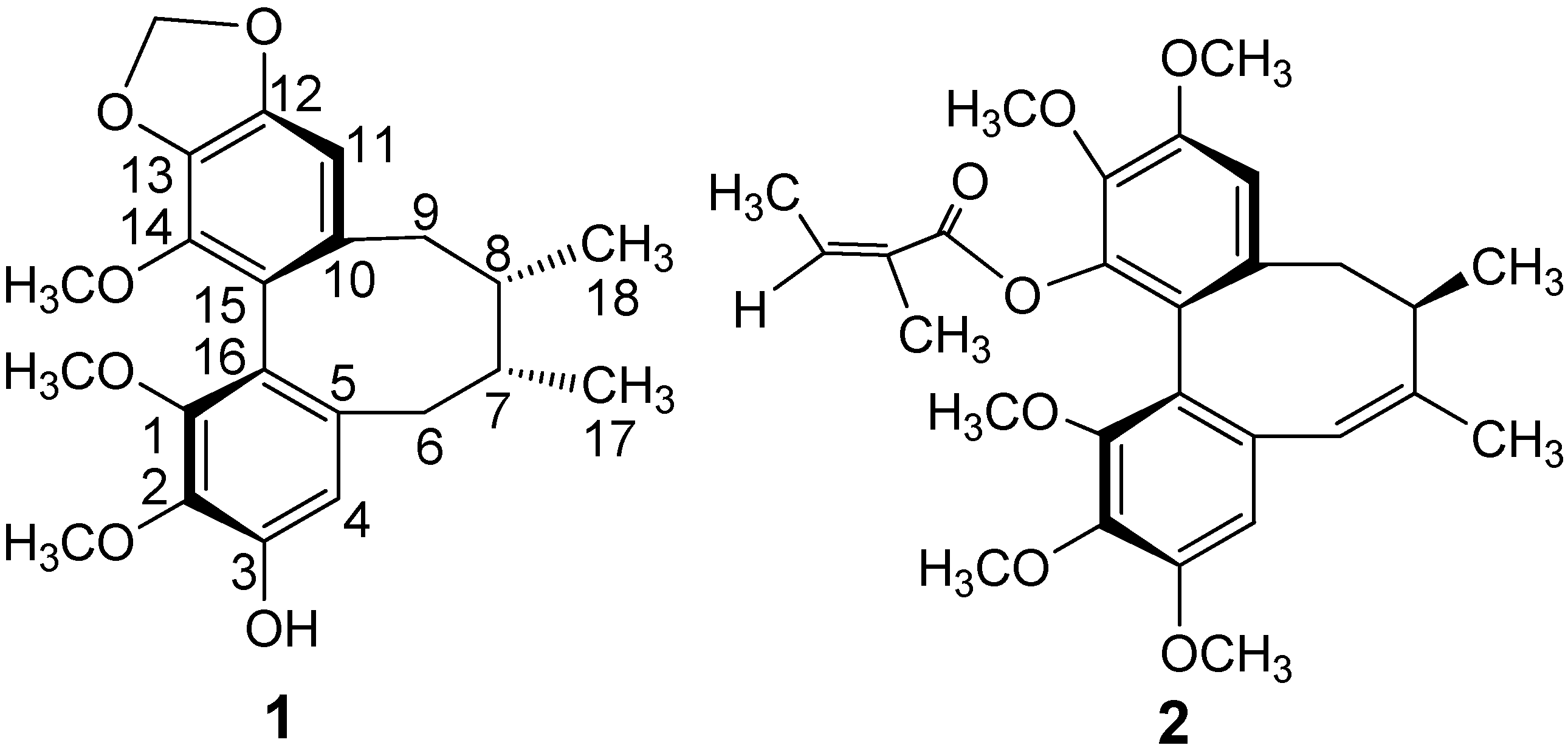
HMBC correlations of CH-11 (δH 6.49) and methylenedioxy (δH 5.96, 5.95) protons with aromatic carbons at δC 147.7, 135.1 indicated that the methylenedioxy group was located at C-12 and C-13. Correlations of the CH-4 proton (δH 6.63) with carbons at δC 148.8 (C-3) and 137.4 (C-2) and of the hydroxyl proton at δH 5.68 with carbons at δC 110.2 (C-4), 148.8 (C-3) and 137.4 (C-2), indicated that the hydroxyl was located at C-3. Thus, the three methoxy groups were located at C-1, C-2, and C-14, based on HMBC correlations of the protons at δH 3.79, 3.93 and 3.50 with aromatic carbons at δC 141.3 (C-14), 137.4 (C-2) and 150.3 (C-1), respectively.
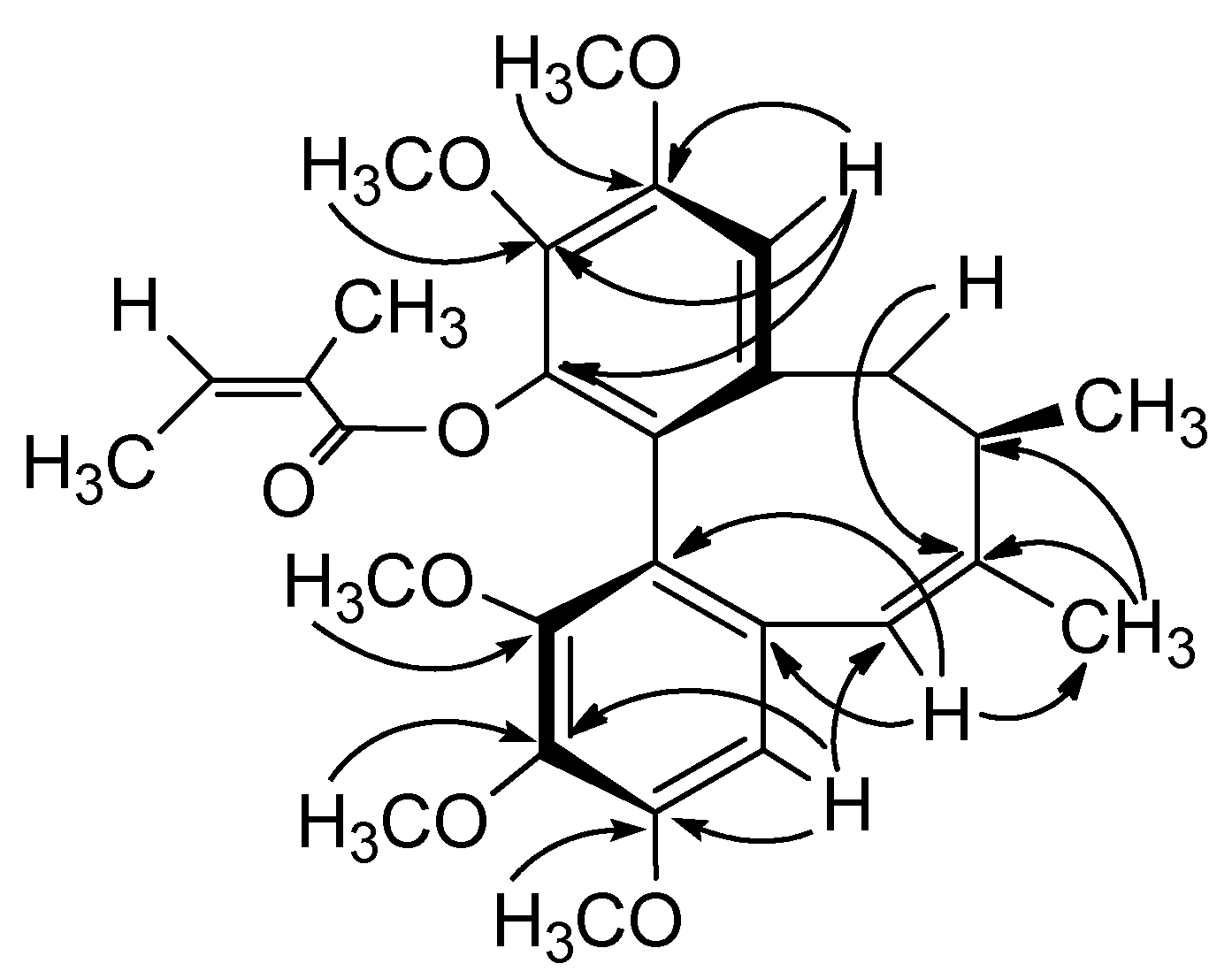
The circular dichroism (CD) spectrum of 1 showed a negative Cotton effect at 216 nm and a positive Cotton effect at 254 nm, which indicated an R-biphenyl configuration in 1 [28]. NOESY correlations between CH-11/Me-18, CH-4/CH-6α and CH-11/CH-9β indicated a twist-boat-chair (TBC) conformation for the cyclooctadiene ring [29] (Figure 2). The substituent positions and stereochemical assignments in the cyclooctadiene ring of 1 were further supported by NOESY correlations of MeO-1/MeO-14, Me-17/Me-18, CH-6β/Me-18 and CH-4/CH-7. Thus, the structure of 1 was determined as that shown in Figure 1. Compound 1 and (−)-gomisin L2 have the same planar structure, but different stereochemistry [30].
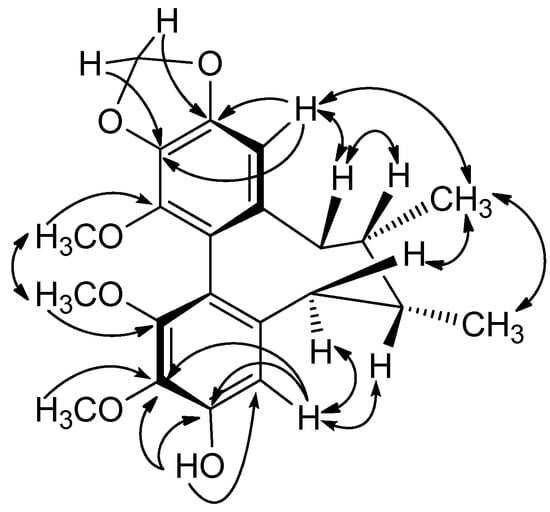
Figure 2.
Key HMBC (→) and NOESY (↔) Correlations of 1.
Figure 2.
Key HMBC (→) and NOESY (↔) Correlations of 1.
Compound 2, obtained as colorless powder, has the molecular formula C28H34O7 according to HR-ESI-MS (m/z 505.2187 [M+Na]+). Its IR, UV, CD and NMR data indicated that 2 is a also a dibenzocyclooctadiene lignan. The 1H-NMR spectrum of 2 (Table 1) showed the presence of five methoxy groups at δH 3.61, 3.80, 3.83, 3.84 and 3.88 (each 3H, s), and an angeloyl group at δH 5.86 (1H, m), 1.78 (3H, s), 1.72 (3H, d, J = 7.1 Hz) on aromatic rings [24].
The following proton-carbon HMBC correlations were observed: CH-4 (δH 6.38) with C-3 (δC 152.6) and C-2 (δC 139.9), CH-11 (δH 6.57) with C-12 (δC 151.6), C-13 (δC 139.3) and C-14 (δC 142.5), and the five methoxy signals at δH 3.61, 3.84, 3.83, 3.88 and 3.80 with aromatic carbon signals at δC 152.3 (C-1), 139.9 (C-2), 152.6 (C-3), 151.6 (C-12) and 139.3 (C-13), respectively. These correlations indicated that the five methoxy groups were attached to C-1, C-2, C-3, C-12, and C-13, and the angeloyl group was located at C-14 (Figure 3).
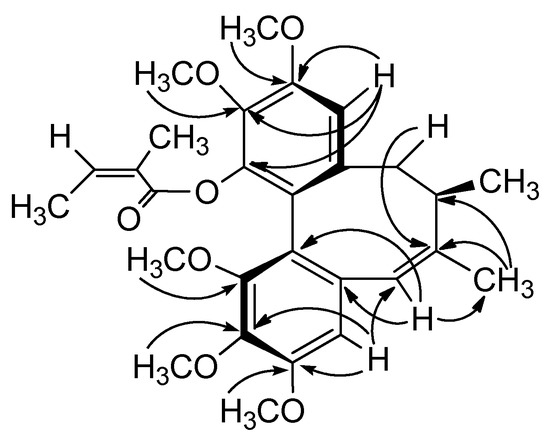
Figure 3.
Key HMBC correlations of 2.
Figure 3.
Key HMBC correlations of 2.
Furthermore, the 1H-NMR spectrum of 2 showed a proton signal at δH 6.20 (1H, s) and the 13C-NMR spectrum showed a quaternary olefinic carbon at δC 142.2 and a methine olefinic carbon at δC 125.3, suggesting the formation of a double bond. A singlet methyl signal at δH 1.63 (3H, s, Me-17) in the 1H-NMR spectrum indicated that the double bond was located between C-6 and C-7 and was substituted with a methyl group. HMBC correlations of δH 6.38 (CH-4) with δC 125.3 (C-6), δH 6.20 (CH-6) with δC 136.5 (C-5), 122.1 (C-16) and 18.9 (C-17), δH1.63 (Me-17) with δC 142.2 (C-7) and 34.2 (C-8), δH 2.38 and 3.10 (CH2-9) with δC 142.2 (C-7) confirmed the substructure.
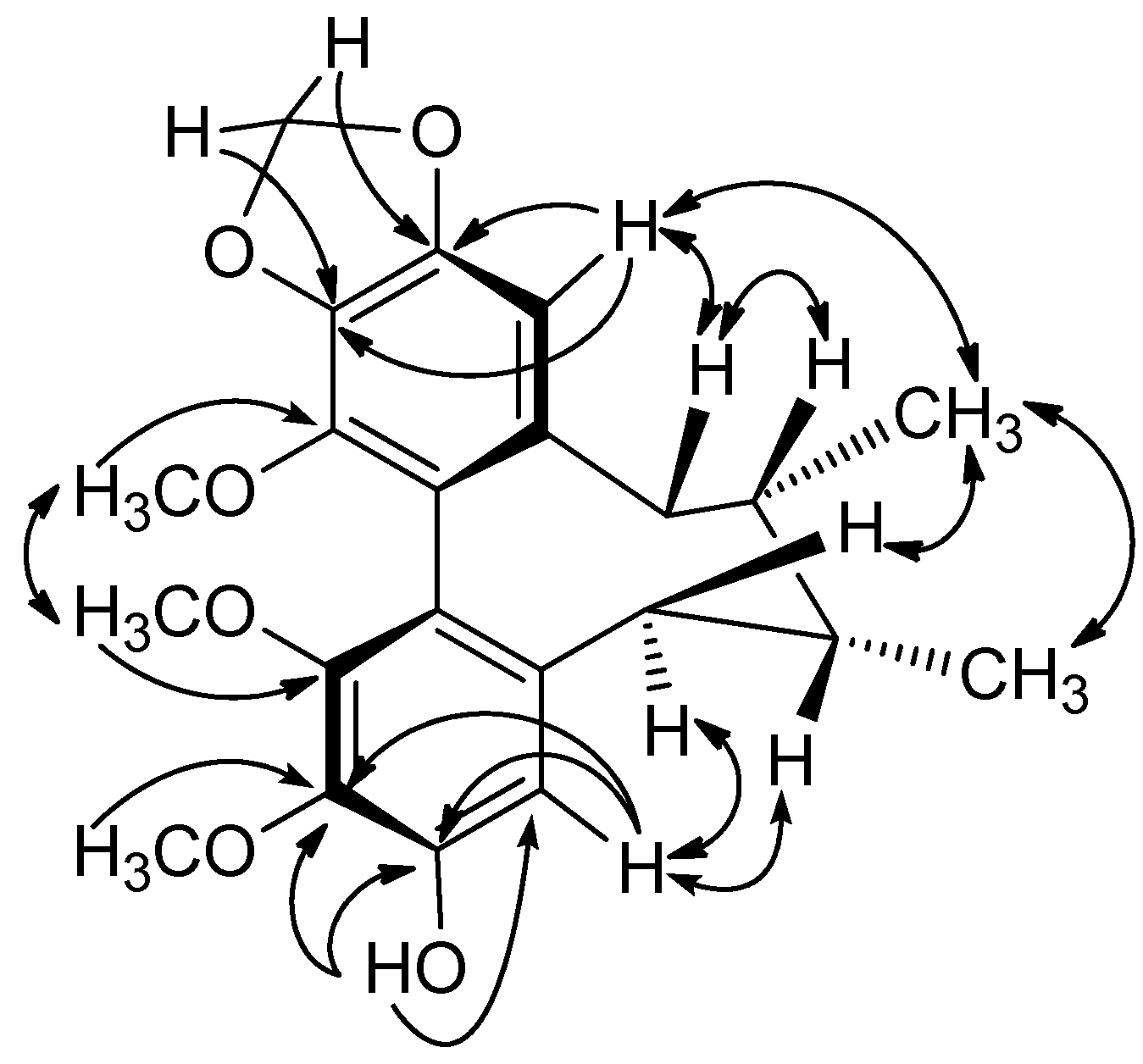
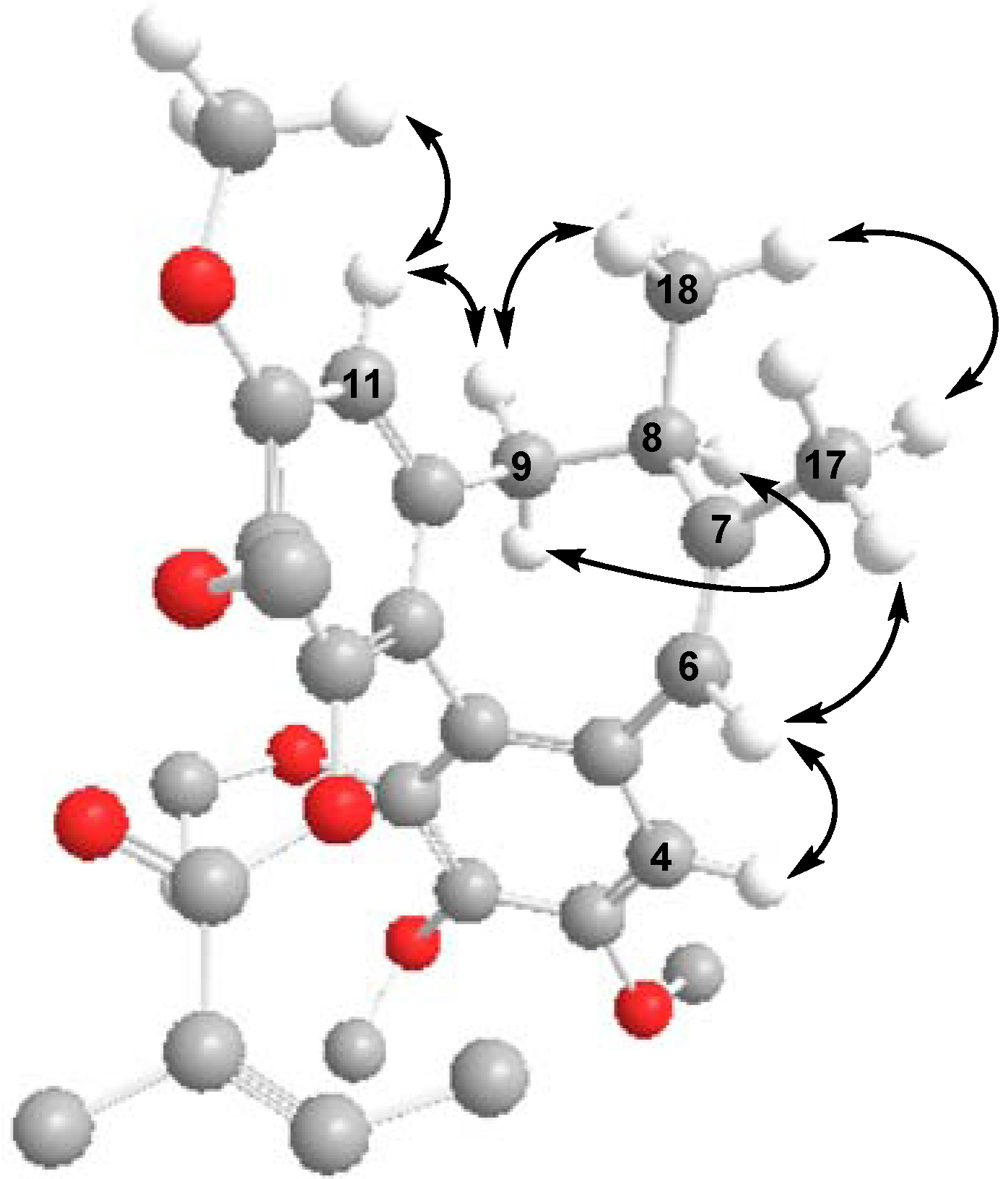
The CD spectrum of 2 had a negative Cotton effect at 219 nm and a positive Cotton effect at 251 nm, indicating that 2 has an R-biphenyl configuration. The NOESY correlations between CH-4/CH-6, CH-6/Me-17, and CH-11/CH-9β, CH-9α/CH-8, CH-11/MeO-12, CH-9β/Me-18, and Me-17/Me-18 fully supported the assigned structure and stereochemistry (Figure 4). Compound 2 has an endocylic double bond rather than the exocyclic double bond found in the previously reported neglschisandrin B [14].
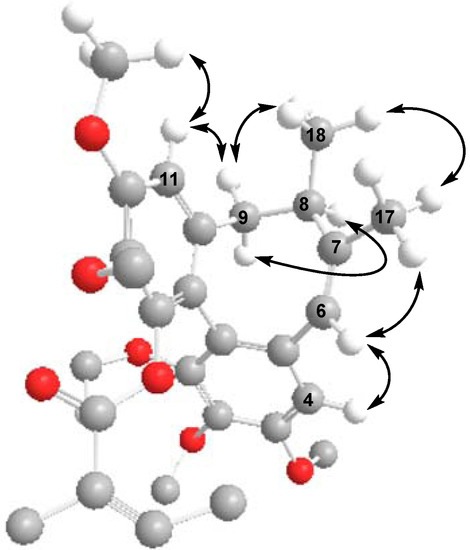
Figure 4.
Key NOESY correlations of 2.
Figure 4.
Key NOESY correlations of 2.
The lignans were evaluated in vitro for cytotoxicity against human lung carcinoma A549 and human colorectal carcinoma HCT-8 cell lines employing a MTT-assay with paclitaxel as the positive control (Table 2). Five lignans exhibited moderate cytotoxicity, while the remaining lignans showed no activity. Against HCT-8, compounds 3 and 6 showed moderate cytotoxicity (EC50 9.58 and 7.33 μg/mL, respectively); compounds 2 and 4 exhibited marginal cytotoxicity (EC50 13.8 and 12.6 μg/mL, respectively), and compound 5 showed weak cytotoxicity (EC50 19.6 μg/mL). Against A549, compounds 2–4 exhibited marginal cytotoxicity with EC50 values ranging from 11.8 to 15.0 μg/mL.

Table 2.
Cytotoxicity data of lignans from Schisandra neglecta.
| Compound | Cytotoxicity (EC50, μg/mL) | |
|---|---|---|
| HCT-8 | A549 | |
| 2 | 13.8 | 11.8 |
| 3 | 9.58 | 13.8 |
| 4 | 12.6 | 15.09 |
| 5 | 19.8 | NA a |
| 6 | 7.33 | NA a |
| Paclitaxel | 0.21 | <0.005 |
a NA (not active)--test compound (20 μg/mL) did not reach 50% inhibition.
3. Experimental
3.1. General
Optical rotations: P-1020 digital spectropolarimeter (JASCO) with MeOH as solvent. UV spectra: Hitachi U-3010 spectrophotometer in MeOH; λmax (log ε) in nm. 1H-, 13C-, 2D-NMR spectra: Bruker DRX400 (400 MHz for 1H, 100 MHz for 13C) spectrometer in CDCl3; δ in ppm rel. to Me4Si as internal references. J in Hz. ESI mass: Bio TOF Q spectrometer; in m/z (rel. %). HR-ESI-MS: Bruker Daltonics BioToF-ШQ mass spectrometer in positive-ion mode (Bremen, Germany). IR spectra: Avatar Thermo Nicolet 360-ESP spectrophotometer. TLC was performed on silica gel plates GF254 (Yantai Institute of Chemical Technology, Yantai, China). The TLC spots were visualized by UV light (254 nm) and sprayed with 10% H2SO4, followed by heating. Column chromatography (CC) was carried out on silica gel (200–300 mesh or 300–400 mesh Qingdao Marine Chemical Factory, Qingdao, China). Semi-prep HPLC was carried out on an octadecylsilane column (RP-18, 250 × 10 mm, 10 μm, YMC, detector: Amersham UV-900) with a flow rate of 3.0 mL/min.
3.2. Plant Material
Stems of Schisandra neglecta were collected in Lin-zhi County, Tibet Autonomous Region, People’s Republic of China in September of 2004, and identified by Professor Hong-ping Deng, School of Life Sciences, Southwest University. A voucher specimen (MC-LZ-040901) is deposited in the Herbarium of Medicinal Plant, School of Life Sciences, Southwest University, Chongqing, China.
3.3. Extraction and Isolation
Powdered air-dried stems (5.0 kg) of Schisandra neglecta were extracted exhaustively with 95% EtOH (5 × 10 L, each three days) at room temperature. The alcoholic extract was evaporated in vacuo to yield a semisolid (430 g), which was suspended in water (1,000 mL) and extracted five times with diethyl ether. The organic solution was concentrated to yield 112 g of residue, which was subjected to silica gel CC eluted with petroleum ether/acetone mixtures of increasing polarity (99:1 to 3:7) to obtain ten fractions. Fraction 3 (8.3 g), eluted with petroleum ether–acetone (95:5), gave 7 (1.2 g), and then was further chromatographed with silica gel CC eluting with petroleum ether/CHCl3 (9:2) to obtain six subfractions. Subfraction 3–3 (1.3 g) was subjected to preparative TLC with petroleum ether/CHCl3 (1:1) to yield 4 (14 mg). Fraction 4 (7.4 g), eluted with petroleum ether/acetone (9:1), was subjected to CC with petroleum ether/EtOAc (15:1~4:1) to obtain eight subfractions. Subfraction 4–3 (0.5 g) was purified by preparative HPLC with MeOH/H2O (70:30) to yield 5 (4 mg, RT 25.3 min) and 6 (10 mg, RT 31.1 min). Subfraction 4–4 (0.7 g) was subjected to preparative TLC with petroleum ether/CHCl3 (1:1) to yield 3 (17 mg). Subfraction 4–5 (0.3 g) was purified by semi-preparative HPLC with MeOH-H2O (70:30) to yield 1 (2 mg, RT 33.4 min). Subfraction 4–6 (3.2 g) was subjected to silica gel CC with petroleum ether-EtOAc (5:1) to yield 8 (2.3 g). Fraction 5 (6.7 g) eluted with petroleum ether-acetone (8:2) was subjected to silica gel CC with petroleum ether/EtOAc (10:2–5:2). Subfraction 5–2 (0.6 g) was purified by semi-preparative HPLC with MeOH/H2O (75:25) to yield 9 (32 mg, RT 37.5) and 15 (46 mg, RT 40.2). Subfraction 5–3 (1.7 g) was subjected to silica gel CC with petroleum ether/EtOAc (9:2), then purified by semi-preparative HPLC with MeOH/H2O (64:36–80:20) to yield 10 (4 mg, RT 25.6 min), 11 (10 mg, RT, 28.7 min), and 12 (43 mg, RT 37.9 min), Subfraction 5–5 (0.9 g) was subjected to silica gel CC with petroleum ether/CHCl3 (5:6), and further purified by semi-preparative HPLC with MeOH/H2O (7:3) to yield 13 (24 mg, RT 40.3 min) and 14 (19 mg, RT 44.5 min). Fraction 6 (5.2 g) eluted with petroleum ether-acetone (7:3) was subjected to silica gel CC with petroleum ether/EtOAc (10:3). Subfraction 6–3 (0.7 g) was purified by semi-preparative HPLC with MeOH-H2O (75:25 to 85:15) to yield 2 (10 mg, RT 24. 7 min).
6R,7S,R-biar-[5,6,7,8-Tetrahydro-1,2,13-trimethoxy-6,7-dimethyl-benzo-[3',4']cycloocta[1',2':4,5]-benzo[1,2-d][1,3]dioxol-3-ol (neglschisandrin E, 1). White amorphous powder; [α]22D +26.9° (c = 0.26, MeOH). UV (MeOH): 216 (4.67), 250 (4.22), 280 (3.97). CD (c = 0.08, MeOH), [θ]15 (nm): −28275 (216), +28636 (254). IR (KBr): 3447, 2925, 1615, 1585, 1476, 737. 1H-NMR and 13C-NMR: see Table 1. HR-ESI-MS: found 387.1810 ([M+H]+, C22H27O6+, calc. 387.1808).
5Z,7R,R-biar-7,8-Dihydro-1,2,3,10,11-pentamethoxy-13-(2Z)-methylbut-2-enoyl-6,7-dimethyl-dibenzo[a,c]cycloocten-5(6H)-one (neglschisandrin F, 2). White amorphous powder; [α]22D +16.9° (c = 0.31, MeOH). UV (MeOH): 216 (4.78), 250 (4.37), 279 (3.97). CD (c = 0.052, MeOH), [θ]15 (nm): −295132 (219), +155445 (251). IR (KBr): 3404, 2936, 1737, 1636, 1591, 1488, 734. 1H-NMR and 13C-NMR: see Table 1. HR-ESI-MS: found 505.2187 ([M+Na]+, C28H34NaO7+, calc. 505.2197).
3.4. Cytotoxicity Assay
Freshly trypsinized cell suspensions were seeded in 96-well microtiter plates at densities of 5,000–10,000 cells per well with compounds added from DMSO-diluted stock. After three days in culture, attached cells were incubated with cold 10% trichloroacetic acid (MTT, 0.5 mg/mL, 2 h) and subsequently solubilized in DMSO, and the isolates were tested against A549 and HCT-8 cancer cell lines using established colorimetric MTT assay protocols [31]. Paclitaxel was used as a positive control. All stock cultures were grown in T-25 flasks. The mean EC50 is the concentration of agent that reduces cell growth by 50% under the experimental conditions and is the average from at least three independent determinations that were reproducible and statistically significant. The absorbance was measured at 550 nm using a microplate reader.
4. Conclusions
Two new lignans, neglschisandrins E and F (1 and 2, respectively), and thirteen known lignans were isolated from stems of Schisandra neglecta. Their structures and stereochemistries were established by means of NMR and ESI-MS analyses, including 2D-NMR techniques. The isolates were evaluated for in vitro cytotoxic activity. Compounds 2–6 showed cytotoxicity against the human HCT-8 colorectal carcinoma cell line with EC50 values of 7.33~19.8 μg/mL, while compounds 2–4 also exhibited cytotoxicity against the human A549 lung carcinoma cell line with EC50 values of 11.8~15.0 μg/mL, respectively. Since other dibenzocyclooctadiene lignans in the genus Schisandra exhibit proven antitumor activities [32], further research to identify related potent cytotoxic compounds or explore detailed structure-activity relationships is merited.
Acknowledgments
This investigation was supported in part by grants from the National Natural Science Foundation of China (81102894), the State Key Program (2010ZX09401-306-1-4) from the Ministry of Science and Technology, China and the Fundamental Research Funds for the Central Universities (XBJK2009C171) awarded to M.C., the National Natural Science Foundation of China (30925042) and the State Key Program (2009ZX09502-013) from the Ministry of Science and Technology, China, awarded to D.F.C., and by Grant CA-17625 from the National Cancer Institute, NIH awarded to K.H.L.
References
- Chinese Pharmacopoeia Commission, Pharmacopoeia of the People’s Republic of China; People’s Medical Publishing House: Beijing, China, 2010; Volume 1, pp. 61, 227.
- Peng, H.L.; Chen, D.F.; Lan, H.X.; Zhang, X.M.; Gu, Z.; Jiang, M.H. Anti-lipid Peroxidation of Gomisin J on Liver Mitochondria and Cultured Myocardial Cells. Acta Pharmacol. Sin. 1996, 17, 538–541. [Google Scholar]
- Chen, D.F.; Zhang, S.X.; Xie, L.; Xie, J.X.; Chen, K.; Kashiwada, Y.; Zhou, B.N.; Wang, P.; Cosentino, L.M.; Lee, K.H. Anti-AIDS Agents—XXVI. Structure-Activity Correlations of Gomisin-G-related Anti-HIV Lignans from Kadsura interior and of Related Synthetic Analogues. Bioorg. Med. Chem. 1997, 5, 1715–1723. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Huang, R.L.; Kuo, Y.L.K.; Chen, C. F Novel C19 Homolignans, Taiwanschirin A, B, and Cytotoxic Taiwanschirin C, and a New C18 Lignan, Schizanrin A, from Schizandra arisan. J. Org. Chem. 1999, 64, 7023–7027. [Google Scholar] [CrossRef]
- Zhang, X.M.; Chen, D.F.; He, X.J.; Yang, S.; Zheng, P.; Jiang, M.H. Blocking Effects of Heteroclitin D and Gomisin J on L-type Calcium Channels in Ventricular Cells of Guinea Pig. Acta Pharmacol. Sin. 2000, 21, 373–376. [Google Scholar]
- Kuo, Y.H.; Li, S.Y.; Huang, R.L.; Wu, M.D.; Huang, H.C.; Lee, K.H. Schizarin B, C, D, and E, Four New Lignans from Kadsura matsudai and Their Antihepatitis Activities. J. Nat. Prod. 2001, 64, 487–490. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, D.F. Analysis of Schisandra chinensis and Schisandra sphenantera. J. Chromatogr. A 2009, 1216, 1980–1990. [Google Scholar] [CrossRef]
- Yang, G.Y.; Li, Y.K.; Wang, R.R.; Xiao, W.L.; Yang, L.M.; Pu, J.X.; Zheng, Y.T.; Sun, H.D. Dibenzocyclooctadiene Lignans from the Fruits of Schisandra wilsoniana and Their Anti-HIV-1 Activities. J. Asian Nat. Prod. 2010, 12, 470–476. [Google Scholar] [CrossRef]
- Liu, C.C.; Abd El-Razek, M.H.; Liaw, C.C.; Cheng, Y.B.; Chen, C.K.; Chien, C.T.; Kuo, Y.H.; Liou, S.S.; Shen, Y.C. Arisanschinins A-E, Lignans from Schisandra arisanensis hay. Planta Med. 2010, 74, 1605–1610. [Google Scholar]
- Yang, J.M.; Ip, P.S.; Che, C.T.; Yeung, J.H. Relaxant Effects of Schisandra chinensis and Its Major Lignans on Agonists-induced Contraction in Guinea Pig Ileum. Phytomedicine 2011, 18, 1153–1160. [Google Scholar] [CrossRef]
- Mu, H.X.; Li, X.S.; Fan, P.; Yang, G.Y.; Pu, J.X.; Sun, H.D.; Hu, Q.F.; Xiao, W.L. Dibenzocyclooctadiene Lignans from the Fruits of Schisandra rubriflora and Their Anti-HIV-1 Activities. J. Asian Nat. Prod. 2011, 13, 393–399. [Google Scholar] [CrossRef]
- Xue, J.J.; Cao, J.L.; Yang, G.Y.; Pu, J.X.; Sun, H.D.; Hu, Q.F.; Xiao, W.L. Lignans from Schisandra lancifolia. J. Asian Nat. Prod. 2011, 13, 492–497. [Google Scholar] [CrossRef]
- Kang, K.; Lee, K.M.; Yoo, J.H.; Lee, H.J.; Kim, C.Y.; Nho, C.W. Dibenzocyclooctadiene Lignans, Gomisins J and N Inhibit the Wnt/β-catenin Signaling Pathway in HCT116 Cells. Biochem. Biophys. Res. Commun. 2012, 428, 285–291. [Google Scholar] [CrossRef]
- Chen, M.; Xu, X.M.; Liao, Z.H.; Dong, L.; Li, L.; Huang, C.Z. Neglschisandrins A-B: Two New Dibenzocyclooctene Lignans from Schisandra neglecta. Molecules 2008, 13, 548–555. [Google Scholar] [CrossRef]
- Chen, M.; Liao, Z.H.; Xu, X.M.; Wen, Y.; Sun, M.; Zhang, H.X.; Ma, W.H. Neglschisandrins C-D: Two New Dibenzocyclooctene Lignans from Schisandra neglecta. Molecules 2008, 13, 1148–1155. [Google Scholar] [CrossRef]
- Chen, C.C.; Shen, C.C.; Shih, Y.Z.; Pan, T.M. 6-O-Benzoylgomisin, a New Lignan from the Fruits of Schizandra chinensis. J. Nat. Prod. 1994, 57, 1164–1165. [Google Scholar] [CrossRef]
- Wang, H.J.; Chen, Y.Y. Studies of Lignans from Schisandra rubriflora Rhed et Wils. Acta Pharm. Sin. 1985, 20, 832–841. [Google Scholar]
- Ikeya, Y.; Ookawa, N.; Taguchi, H.; Yosioka, I. The Constituents of Schizandra chinensis Baill. II. The Structures of 3 New Lignans, Angeloylgomisin-O, and Angeloylisogomisin-O and Benzoylisogomisin-O. Chem. Pharm. Bull. 1982, 30, 3202–3206. [Google Scholar]
- Ikeya, Y.; Taguchi, H.; Yosioka, I.; Kobayashi, H. Constituents of Schizandra chinensis Baill. 5. Structures of 4 New Lignans, Gomisin-N, Gomisin-O, Epigomisin-O and Gomisin-E, and Transformation of Gomisin-N to Deangeloylgomisin-B. Chem. Pharm. Bull. 1979, 27, 2695–2709. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Yang, Y.Q. Studies on the SGPT-lowering Active Component of the Fruits of Schisandra rubriflora Rhed et Wils. Acta Pharm. Sin. 1982, 17, 312–313. [Google Scholar]
- Ikeya, Y.; Taguchi, H.; Yosioka, I.; Kobayashi, H. Constituents of Schizandra-chinensis Baill. 3. Structures of 4 New Lignans, Gomisin-H and Its Derivatives, Angeloyl-gomisin-H, Tigloyl-gomisin-H and Benzoyl-gomisin-H. Chem. Pharm. Bull. 1979, 27, 1576–1582. [Google Scholar] [CrossRef]
- Ikeya, Y.; Taguchi, H.; Yosioka, I. Constituents of Schizandra-chinensis Baill. 1. Isolation and Structure Determination of 5 New Lignans, Gomisin-A, Gomisin-B, Gomisin-C, Gomisin-F and Gomisin-G and the Absolute Structure of Schizandrin. Chem. Pharm. Bull. 1979, 27, 1383–1394. [Google Scholar] [CrossRef]
- Han, G.Q.; Dai, P.; Xue, R.; Arison, B.H.; Lanki, D.C.; Hwang, S.B. Dibenzocyclooctadiene Lignans with Platelet-activating Factor (PAF) Antagonist Activity from Kadsura heteroclita. J. Chin. Pharm. Sci. 1992, 1, 20–27. [Google Scholar]
- Lei, C.; Huang, S.X.; Chen, J.J.; Pu, J.X.; Yang, L.B.; Zhao, Y.; Liu, J.P.; Gao, X.M.; Xiao, W.L.; Sun, H. Lignans from Schisandra propinqua var. propinqua. Chem. Pharm. Bull. 2007, 55, 1281–1283. [Google Scholar] [CrossRef]
- Chen, M.; Kilgore, N.; Lee, K.H.; Chen, D.F. Rubrisandrins A and B, Lignans and Related Anti-HIV Compounds from Schisandra rubriflora. J. Nat. Prod. 2006, 69, 1697–1901. [Google Scholar] [CrossRef]
- Ghera, E.; Ben-David, Y.; Becker, D. Deoxyschizandrin, Stereochemistry and Total Synthesis. Tetrahedron Lett. 1977, 18, 463–467. [Google Scholar] [CrossRef]
- Chen, M.; Liao, Z.X.; Chen, D.F. Four New Dibenzocyclooctene Lignans from Kadsura renchangiana. Helv. Chim. Acta 2004, 87, 1368–1376. [Google Scholar] [CrossRef]
- Li, H.R.; Feng, Y.L.; Yang, Z.G.; Wang, J.; Daikonya, A.; Kitanaka, S.; Xu, L.Z.; Yang, S.L. New Lignans from Kadsura coccinea and Their Nitric Oxide Inhibitory Activities. Chem. Pharm. Bull. 2006, 54, 1022–1025. [Google Scholar] [CrossRef]
- Liu, J.S.; Li, L. Schisantherins L-O and Acetylschisantherin L from Kadsura coccinea. Phytochemistry 1993, 32, 1293–1296. [Google Scholar] [CrossRef]
- Ikeya, Y.; Taguchi, H.; Yosioka, I. The Constituents of Schizandra chinensis Baill. 10. The Structures of γ-Schizandrin and 4 New Lignans, (−)-Gomisins L1 and L2, (±)-Gomisin M1 and (+)-Gomisin M2. Chem. Pharm. Bull. 1982, 30, 132–139. [Google Scholar] [CrossRef]
- Wu, J.H.; Zhao, M.; Qian, K.D.; Lee, K.H.; Morris-Natschke, S.; Peng, S.Q. Novel N-(3-Carboxyl-9-benzyl-beta-carboline-1-yl)ethylamino Acids: Synthesis, Anti-tumor Evaluation, Intercalating Determination, 3D QSAR Analysis and Docking Investigation. Eur. J. Med. Chem. 2009, 44, 4153–4161. [Google Scholar] [CrossRef]
- Xu, L.J.; Liu, H.T.; Peng, Y.; Xiao, P.G. A Preliminary Pharmacophylogenetic Investigation in Schisandraceae. J. Syst. Evol. 2008, 46, 692–723. [Google Scholar]
- Sample Availability: Samples of the compounds 1 and 2 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
