Abstract
The reactions of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in a MeOH/H2O or 2-PrOH/H2O medium in the presence of Na2CO3 afforded 6-alkoxy-2-amino-4-ferrocenylpyridine-3,5-dicarbonitriles 3a,b (multi-component condensation) and 6-alkoxy-2-amino-4-ferrocenyl-3-ferrocenylmethyl-3,4-dihydropyridine-3,5-dicarbonitriles 4a,b (multi-component cyclodimerization). Analogous reactions of 1 with 2 in an MeOH/H2O medium in the presence of NaOH, piperidine, or morpholine gave compounds 3a, 4a and 2-amino-4-ferrocenyl-6-hydroxy-, 6-piperidino- and 6-morpholinopyridine-3,5-dicarbonitriles 3c–e, respectively. The structures of the compounds 3b, 4a and 4b were established by the spectroscopic data and X-ray diffraction analysis. The electrochemical behaviour of compounds 3b, 3d and 4b was investigated by means of cyclic voltammetry.
1. Introduction
Pyridine derivatives have been studied for over a century as an important class of heterocyclic compounds and they still continue to attract considerable attention due to the wide range of medicinal properties they possess, such as vasodilators, anticoagulants, hypolipidemic, tuberculostatic, antihistamine, antihypertensive, cardiovascular and gastrointestinal activities [1,2]. Pyridine systems are also found in important vitamins (PP, B6), alkaloids and herbicides [1].
The incorporation of one or two iron-containing ferrocene substituents into a pyridine molecule will enlarge the spectrum of valuable characteristics. In addition, ferrocene compounds are known to exhibit chemotherapeutic properties [3]. Ferrocenyl-substituted pyridines have been extensively studied as ligands, in the synthesis of non-linear optical materials, etc. [4,5,6]. However, their biological activities have not hitherto been studied. Various methods to prepare ferroceno-containing pyridines have been reported [7,8,9,10]. Syntheses of ferrocenylpyridines are mainly carried out via Negishi cross-coupling reactions of FcZnCl with bromopyridines [7], the condensations of 1,3-diketones with ferrocenecarboxaldehyde in the presence of AcONH4 [8]; the interactions of ferrocenyl-1,2-enones with 3-aminocrotononitrile [9], ethyl 3-aminocrotonate [9] or acetonitrile in the presence of Me3COK [10]. The interest in heterocyclic compounds bearing ferrocenyl substituents in the molecules can be traced back to the discovery of ferrocene [11,12,13,14]. This is determined by a peculiar chemical behavior of such compounds due to mutual influence of the metallocene and heterocyclic moieties. In particular, biological activities of many nitrogen heterocycles, such as quinuclidines, pyrazolines, pyrazoles, pyrimidines, tetrahydropyridazines, bearing ferrocenyl substituents, have been reported [8,15,16,17,18,19,20]. It may be expected that ferrocenylpyridines and cyano(ferrocenyl)pyridines will also prove valuable, because they possess diverse biological activity, find use as potential bio-receptor ligands [13,14,15], new drugs [16,17,18,19,20], and significant intermediates for the synthesis of important materials [21,22,23]. For these reasons, development of new compounds containing cyano and ferrocenyl groups in the pyridines is strongly desired.
Herein we report results from our investigations into reactions of the condensation of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) and of the tandem-transformations of 1 in alcohols/aqueous medium in the presence of bases and nucleophiles. The electrochemical behavior of the 6-alkoxy-2-amino-4-ferrocenylpyridine-3,5-dicarbonitriles and 6-alkoxy-2-amino-4-ferrocenyl-3-ferrocenylmethyl-3,4-dihydropyridine-3,5-dicarbonitriles was studied.
2. Results and Discussion
2.1. Synthesis of 6-Alkoxy-2-amino-4-ferrocenylpyridine-3,5-dicarbonitriles and 6-alkoxy-2-amino-4-ferrocenyl-3-ferrocenylmethyl-3,4-dihydropyridine-3,5-dicarbonitriles
We found that two competitive processes occur upon reaction of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in MeOH/H2O or 2-PrOH/H2O medium in the presence of Na2CO3, viz, formation of 6-alkoxy-2-amino-4-ferrocenylpyridine-3,5-dicarbonitriles 3a,b (multi-component condensation) and of cyclodimeric products 4a,b (multi-component cyclodimerization). In addition, other minor reaction products of the starting compounds were also isolated, however their structures could not be established from their 1H and 13C-NMR spectroscopy and mass spectrometry data (Scheme 1).
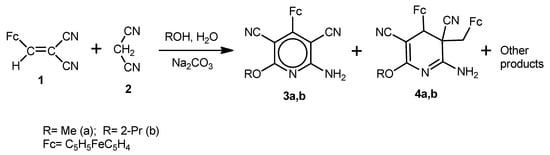
Scheme 1.
Reaction of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in the presence of Na2CO3.
The compounds 3a,b and 4a,b were isolated by column chromatography on alumina and their structures were characterized by IR and NMR spectroscopy, mass spectrometry, and elemental analysis (see Experimental section). According to the 1H- and 13C-NMR data, the cyclodimerization of 1 occurs with high diastereoselectivity, and compounds 4a and 4b were isolated as a single diastereomeric form. One cannot rule out the formation of minor diastereomeric products; however they could not be isolated and characterized.
The molecular structures of compounds 3b, 4a and 4b were determined by X-ray diffraction analysis of their single crystals. The general views of molecules 3b, 4a, and 4b are shown in Figure 1 Figure 2 Figure 3, respectively, while the principal geometric parameters are listed in Table 1.
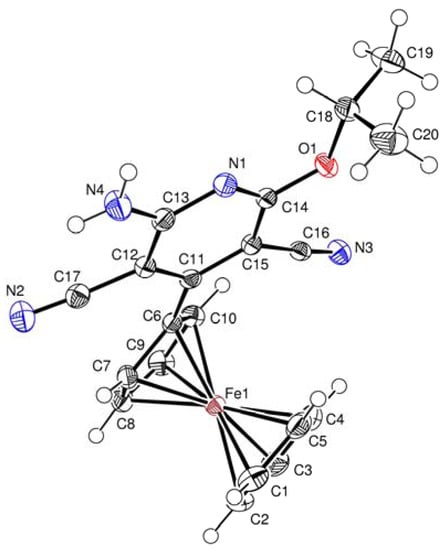
Figure 1.
Crystal structure of 3b.
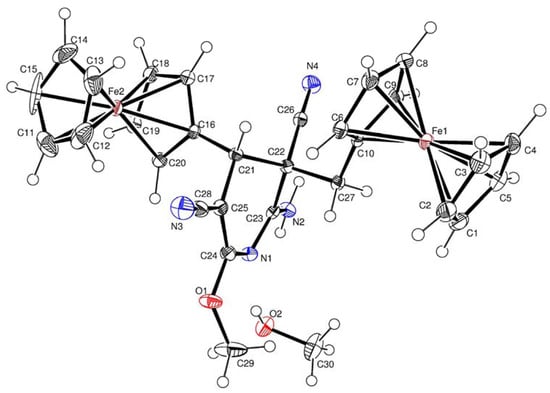
Figure 2.
Crystal structure of 4a.
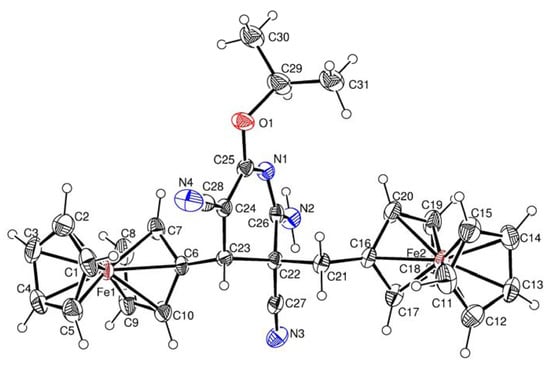
Figure 3.
Crystal structure of 4b.
X-Ray diffraction analysis confirmed the aromatic ferrocenylpyridine structure for compound 3b, and diferrocenyl(dihydro)pyridine structures for compounds 4a and 4b. Central fragment of the molecule 3b is a flat six-membered ring with one nitrogen atom. The N(1)-C(14) bond length in the compound 3b is somewhat shorter [d = 1.318(2) Å] compared to the standard length (cf. d = 1.338 Å [24]). The bond lengths of the C–Fe and C–C bonds in the ferrocenyl substituents as well as the geometric parameters of the ferrocene sandwiches are close to standard values [25].

Table 1.
Selected bond lengths and bond angles for compounds 3b, 4a and 4b.
| Selected bond lengths (Å) | Selected bond angles (°) | |||||
|---|---|---|---|---|---|---|
| 3b | ||||||
| N(1)-C(13) | 1.346(2) | C(15)-C(11)-C(12) | 116.21(15) | |||
| N(1)-C(14) | 1.318(2) | N(1)-C(13)-C(12) | 122.50(16) | |||
| N(4)-C(13) | 1.337(2) | N(1)-C(14)-O(1) | 120.55(15) | |||
| N(3)-C(16) | 1.1150(2) | N(1)-C(14)-C(15) | 124.42(16) | |||
| C(12)-C(13) | 1.422(2) | C(11)-C(15)-C(14) | 119.28(16) | |||
| C(11)-C(12) | 1.406(2) | C(13)-N(1)-C(14) | 117.46(15) | |||
| O(1)-C(14) | 1.338(2) | N(4)-C(13)-N(1) | 116.87(16) | |||
| C(11)-C(15) | 1.404(2) | C(14)-O(1)-C(18) | 119.91(14) | |||
| C(15)-C(14) | 1.417(2) | C(17)-C(12)-C(13) | 116.37(15) | |||
| O(1)-C(18) | 1.466(2) | C(12)-C(11)-C(6) | 120.99(15) | |||
| 4a | ||||||
| N(1)-C(23) | 1.319(3) | N(1)-C(23)-C(22) | 120.62(18) | |||
| C(24)-N(1) | 1.381(3) | N(1)-C(24)-C(25) | 124.48(19) | |||
| C(22)-C(23) | 1.530(3) | N(1)-C(24)-O(1) | 116.55(18) | |||
| C(21)-C(22) | 1.568(3) | C(23)-C(22)-C(21) | 106.96(16) | |||
| C(21)-C(25) | 1.512(3) | C(22)-C(21)-C(25) | 106.41(16) | |||
| C(25)-C(24) | 1.352(3) | C(21)-C(25)-C(24) | 118.82(18) | |||
| C(22)-C(27) | 1.566(3) | N(1)-C(23)-N(2) | 120.29(19) | |||
| C(23)-N(2) | 1.312(3) | N(2)-C(23)-C(22) | 118.99(18) | |||
| C(24)-O(1) | 1.345(2) | C(27)-C (22)-C(26) | 107.67(17) | |||
| 4b | ||||||
| N(1)-C(25) | 1.378(4) | C(24)-C(23)-C(22) | 105.9(3) | |||
| N(1)-C(26) | 1.291(4) | N(4)-C(28)-C(24) | 177.4(4) | |||
| N(2)-C(26) | 1.334(5) | N(2)-C(26)-C(22) | 118.3(3) | |||
| N(3)-C(27) | 1.137(5) | N(3)-C(27)-C(22) | 178.0(4) | |||
| N(4)-C(28) | 1.147(4) | C(26)-C(22)-C(23) | 103.3(3) | |||
| C(22)-C(21) | 1.559(5) | C(24)-C(25)-N(1) | 124.1(3) | |||
| C(22)-C(26) | 1.532(5) | N(2)-C(26)-N(1) | 119.3(3) | |||
| C(23)-C(22) | 1.569(4) | C(21)-C(22)-C(23) | 111.6(3) | |||
| C(24)-C(25) | 1.359(5) | C(26)-N(1)-C(25) | 117.0(3) | |||
| C(23)-H(23) | 0.9800 | C(22)-C(26)-N(1) | 122.4(3) | |||
| C(25)-O(1) | 1.345(4) | O(1)-C(25)-N(1) | 116.8(3) | |||
| C(29)-O(1) | 1.443(5) | C(27)-C(22)-C(23) | 109.9(3) | |||
| C(24)-C(23) | 1.510(5) | C(26)-C(22)-C(23) | 106.3(3) | |||
Key elements of the molecules 4a and 4b are the central six-membered ring with one nitrogen atom in the half-chair conformation. The N(1)-C(23) (for 4a) and N(1)-C(26) (for 4b) bond lengths are equal to d = 1.319(3) Å, and d = 1.291(4) Å, respectively. The ferrocenyl and ferrocenylmethyl substituents at C-4 and C-5 of 4a and 4b are trans oriented relative to the 6-membered cycle.
We found further that 2-cyano-3-ferrocenylacrylonitrile (1) reacted analogously with malononitrile (2) in MeOH/H2O medium in the presence of NaOH, piperidine or morpholine to give compounds 3a,c–e and 4a (Scheme 2 and Scheme 3). Cyclodimeric products 4c,d,e with hydroxy-, piperidino- or morpholino-substituents were not detected (Scheme 2 and Scheme 3). As in the case of reaction of 1 with 2 in the presence Na2CO3, the polymerization products of the starting compounds were also present in minor quantities.
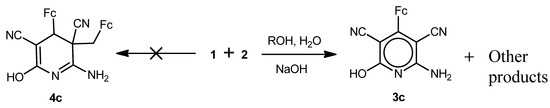
Scheme 2.
Reaction of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in the presence of NaOH.
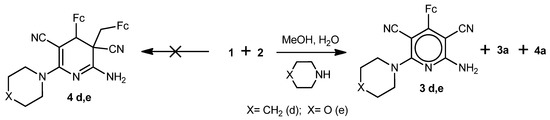
Scheme 3.
Reaction of 2-cyano-3-ferrocenyl-acrylonitrile (1) with malononitrile (2) in the presence of piperidine or morpholine.
Both reaction mixtures were separated by column chromatography on alumina, and the structures of the isolated products were characterized by IR, 1H and 13C-NMR spectroscopy, mass spectrometry, and elemental analysis. These physicochemical characterizations of compounds 3c–e corroborate completely their structures.
The formation of 2-amino-4-ferrocenylpyridine-3,5-dicarbonitriles 3a-e in the presence of bases and nucleophiles proceeds, in our opinion, via multi-component condensation reaction [26] (Scheme 4). Possibly the intermediate 5 is generated in one step and then transformed into pyridines 3a–e.
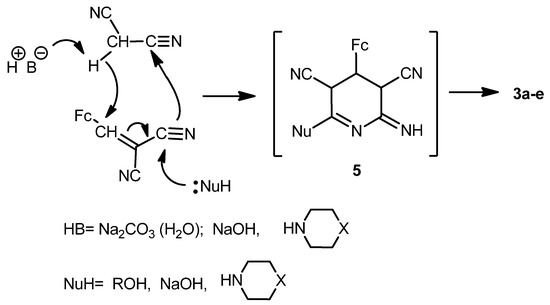
Scheme 4.
Possible mechanism for the formation of compounds 3a–e.
A tentative mechanism for the formation of the diferrocenyl(dihydro)pyridine-3,5-dicarbonitriles 4a,b is represented in Scheme 5.
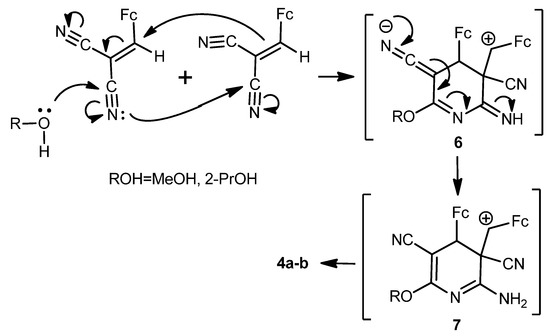
Scheme 5.
Possible mechanism for the formation of compounds 4a,b.
To verify the mechanism described in Scheme 5 above, the cyclodimerization of 2-cyano-3-ferrocenylacrylonitrile (1) was carried out under identical conditions in 2-propanol in the presence of water and Na2CO3. The product of the cyclodimerization, 2-amino-4-ferrocenyl-3-ferrocenylmethyl-6-isopropoxy-3,4-dihydropyridine-3,5-dicarbonitrile (4b), was obtained with ~27% yield. Thus, cyclodimerization of compound 1 represents a novel type of the three-component anomalous reaction of [4+2]-cycloadition, absolutely different from the Diels-Alder reaction.
2.2. Electrochemistry
Figure 4 shows a typical voltammogram of compound 3b recorded from open circuit potential to positive direction using a platinum electrode. It was observed one oxidation signal Ia with anodic peak potential value Epa(Ia) = 0.247 V/Fc-Fc+ and, one reduction signal Ic, with cathodic peak potential value Epc(Ic) = 0.184 V/Fc-Fc+. The ΔEp = 0.063 was independent of scan rate (from 0.1 to 1 V·s−1). The cathodic peak current and the anodic peak current were proportional to v1/2, indicating that I is a diffusion-controlled process [27]. The evidence presented above suggests that process I can be attributed to the reversible electron transfer for the ferrocene moiety Fc-Fc+. The formal potential electrode value was E0' = 0.215 V/Fc-Fc+, estimated as E0' = 1/2(Epa + Epc). The electrochemical behaviour of compound 3d is very similar to that observed for 3b. There are slight changes in peak potential values: Epa(Ia) = 0.241 V/Fc-Fc+, Epc(Ic) = 0.174 V/Fc-Fc+, ΔEp = 0.067 V and E0' = 0.207 V/Fc-Fc+.
Figure 5 shows a cyclic voltammogram of compound 4b. When the scan was started from open circuit potential to positive direction two oxidation signals (Ia) and (IIa) were observed. The anodic peak potentials values for these signals are Epa(Ia) = 0.198 V/Fc-Fc+ and Epa(IIa) = 1.149 V/Fc-Fc+. When the cycle was complete only one reduction signal Ic (related to the oxidation process Ia) was observed. The estimated cathodic peak potential value was Epc(Ic) = 0.099 V/Fc-Fc+. Despite the use of different scan rates (0.1 V·s−1–1.0 V·s−1) in the voltammetric experiments, the product of the electrochemical reduction in the process IIa was not detected. This result points out the absence of electronic communication between the two proximal ferrocenyl centres, which is contrary to the observations reported recently [28], where the communication between ferrocenyl fragments was detected in 3,5-diferrocenylpyridine. The electrochemical process I is attributed to the ferrocene moiety at the para position to the nitrogen atom of the heterocycle, Fcpara−/Fcpara+. The estimated formal potential electrode value was E0' = 0.1485 V/Fc-Fc+.
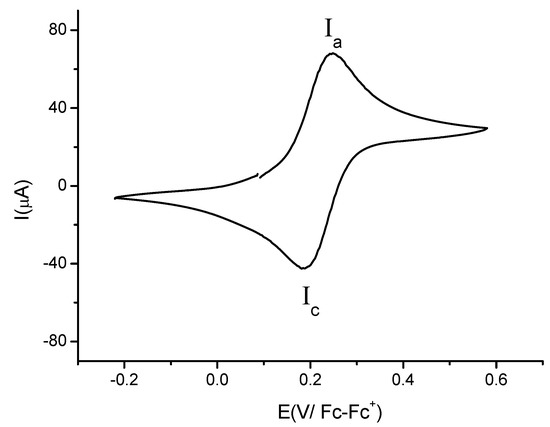
Figure 4.
Cyclic voltammogram of compound 3b in the presence of 0.1 M TBABF4 in MeCN. Scan rate 0.1 V·s−1. The working electrode used was platinum.
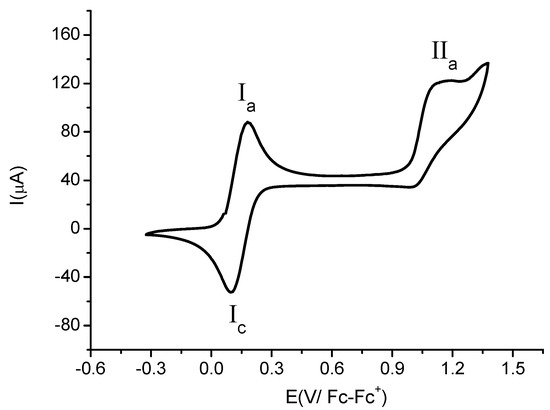
Figure 5.
Cyclic voltammogram of compound 4b in the presence of 0.1 M TBABF4 in MeCN. The scan rate 0.10 V·s−1. The working electrode used was platinum.
The second oxidation process (IIa) is related to the ferrocene moiety at the meta position to the nitrogen atom of the heterocycle (Fcmeta/Fcmeta+) with high positive electronic density due to its proximity to the CN group. The absence of the reduction signal in the process II could be attributed to a low stabilization of the electro-generated dication (Fc+para-Fcmeta+) by the solvent [29,30]. This fact was confirmed when the experiment was performed in a coordinative solvent such as DMSO, where electrochemical response becomes more irreversible.
3. Experimental
3.1. General
All the solvents were dried according to the standard procedures and were freshly distilled before use [31]. Column chromatography was carried out on alumina (Brockmann activity III). The 1H and 13C-NMR spectra were recorded on a Unity Inova Varian spectrometer (300 and 75 MHz, Palo Alto, CA, USA) for solutions in CDCl3, with Me4Si as the internal standard. The IR spectra were measured on a Spectrophotometer FT-IR (Spectrum RXI Perkin Elmer instruments, Waltham, MA, USA) using KBr pellets. The mass spectra were obtained on a Varian MAT CH-6 instrument (EI MS, 70 eV). Elementar Analysensysteme LECO CHNS-900 (St. Joseph, MI, USA) was used for elemental analysis.
The electrochemical behavior of compounds 3b, 3d and 4b was explored with a Biologic SP-50 (Grenoble, France) potentiostat/galvanostat. The current interrupt method was used for iR compensation during all the experiments. The sample concentration employed was ca. 1 mM in acetonitrile in the presence of 0.1 M tetra-N-butylammonium tetrafluoroborate (TBABF4). A platinum disk and a platinum wire were used as working electrode and counter-electrode, respectively. A silver wire was used as a pseudo reference electrode. All solutions were bubbled with nitrogen 5 minutes prior each measurement. Cyclic voltammetry experiments were initiated from open circuit potential (Eocp) to positive direction, using scan rates from 0.1 to 1.0 V·s−1. All potentials were reported versus the couple Fc/Fc+ according to IUPAC convention [32]. The following reagents were purchased from Aldrich (Toluca, Mexico): ferrocenecarboxaldehyde, 99%; malononitrile, 99%; methyl alcohol, 99.9%; 2-propanol, 99.9%; morpholine, 99+%; piperidine, 99%. 2-Cyano-3-ferrocenylacrylonitrile (1) was prepared by condensation of ferrocenecarbaldehyde with malononitrile in benzene in the presence of piperidinium acetate [33]. The physical and 1H-NMR spectroscopic characteristics of compound 1 were in accordance with the literature data [34].
Reactions of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in the presence of Na2CO3. A mixture of compound 1 (1.13 g, 5.0 mmol), malononitrile 2 (0.4 g, 6.0 mmol), methanol or 2-propanol (100 mL), H2O (10 mL) and Na2CO3 (0.5 g, 5.0 mmol) was stirred and refluxed for 8 h. The solvents were removed in vacuo and the residue was dissolved in dichloromethane (50 mL). The solution was mixed with Al2O3 (activity III, 20 g) and the solvent was evaporated in air. This sorbent was applied onto a column with Al2O3 (the height of alumina is ca. 20 cm) and the reaction products were eluted from the column first with petroleum ether, then with a 2:1 hexane–dichloromethane to give compounds 3a,b, 4a,b and polymeric compounds.
2-Amino-4-ferrocenyl-6-methoxypyridine-3,5-dicarbonitrile (3a). Red crystals, yield 0.93 g (52%), m.p. 162–163 °C; IR (KBr): 425, 497, 509, 575, 812, 843, 911, 1004, 1044, 1107, 1185, 1223, 1259, 1295, 1321, 1340, 1386, 1424, 1468, 1482, 1541, 1557, 1613, 2212, 2216, 2321, 2982, 3101, 3226, 3372, 3462 cm−1; 1H-NMR: 4.00 (s, 3H, CH3), 4.28 (s, 5H, C5H5), 4.59 (m, 2H, C5H4), 5.21 (m, 2H, C5H4), 5.57 (bs, 2H, NH2); 13C-NMR: 55.43 (CH3), 71.00 (C5H5), 70.49, 71.17 (C5H4), 84.91 (CipsoFc), 116.27, 117.60 (2CN), 160.17, 160.43, 160.52, 164.88, 167.50 (5C); MS: m/z 358 [M]+. Anal. Calcd. for C18H14FeN4O: C 60.36, H 3.94, Fe 15.60, N 15.63. Found: C 60.45, H 4.04, Fe 15.46, N 15.49.
2-Amino-4-ferrocenyl-6-isopropoxypyridine-3,5-dicarbonitrile (3b). Red crystals, yield 0.9 g (48%), m.p. 176–177 °C; IR (KBr): 425, 502, 541, 584, 813, 845, 912, 1003, 1044, 1106, 1185, 1253, 1296, 1322, 1334, 1365, 1383, 1425, 1477, 1483, 1542, 1556, 1612, 2200, 2217, 2325, 2979, 3103, 3224, 3369, 3459 cm−1; 1H-NMR: 1.39 (d, 6H, 2CH3, J = 6.3 Hz), 4.28 (s, 5H, C5H5), 4.57 (m, 2H, C5H4), 5.20 (m, 2H, C5H4), 5.32 (m, 1H, CH, J = 6.3 Hz), 5.53 (bs, 2H, NH2); 13C-NMR: 21.95 (2CH3), 71.16 (CH), 71.01 (C5H5), 70.51, 71.07 (C5H4), 82.05 (CipsoFc), 116.32, 117.77 (2CN), 160.14, 160.21, 161.56, 165.38, 166.86 (5C); MS: m/z 386 [M]+. Anal. Calcd. for C20H18FeN4O: C 62.20, H 4.70, Fe 14.46, N 14.50. Found: C 62.31, H 4.63, Fe 14.58, N 14.67.
2-Amino-4-ferrocenyl-3-ferrocenylmethyl-6-methoxy-3,4-dihydropyridine-3,5-dicarbonitrile (4a). Yellow crystals, yield 0.25 g (18%), m.p. dec. ca. 272 °C; IR (KBr): 484, 559, 691, 799, 811, 1002, 1029, 1041, 1103, 1190, 1235, 1282, 1321, 1371, 1387, 1452, 1472, 1534, 1550, 1597, 1641, 1663, 2217, 2225, 3090, 3321, 3429 cm−1; 1H-NMR: 2.91 (d, 1H, CH2, J = 14.1 Hz), 3.10 (d, 1H, CH2, J = 14.1 Hz), 3.47 (s, 1H, CH), 3.91 (s, 3H, CH3), 4.18 (s, 5H, C5H5), 4.27 (s, 5H, C5H5), 3.87 (m, 1H, C5H4), 4.08 (m, 1H, C5H4), 4.12 (m, 1H, C5H4), 4.17 (m, 1H, C5H4), 4.22 (m, 2H, C5H4), 4.23 (m, 1H, C5H4), 4.41 (m, 1H, C5H4), 5.59 (bs, 2H, NH2); 13C-NMR: 42.19 (CH2), 55.13 (CH3), 63.61 (CH), 69.23, 69.79 (2C5H5), 67.84, 68.36, 68.75, 68.99, 69.12, 69.56, 69.97, 70.44 (2C5H4), 80.21, 82.45 (2CipsoFc), 119.21, 120.50 (2CN), 64.45, 160.62, 164.86, 165.72 (4C); MS: m/z 558 [M]+. Anal. Calcd. for C29H26Fe2N4O: C 62.40, H 4.70, Fe 20.01, N 10.03. Found: C 62.29, H 4.61, Fe 19.89, N 10.12.
2-Amino-4-ferrocenyl-3-ferrocenylmethyl-6-isopropoxy-3,4-dihydropyridine-3,5-dicarbonitrile (4b). Yellow crystals, yield 0.29 g (19%), m.p. dec. ca. 302 °C; IR (KBr): 483, 553, 682, 721, 783, 821, 915, 1001, 1026, 1042, 1106, 1142, 1181, 1249, 1294, 1316, 1355, 1371, 1383, 1423, 1475, 1544, 1585, 1629, 2191, 2300, 2930, 2978, 3095, 3241, 3335, 3466 cm−1; 1H-NMR: 1.39 (d, 6H, 2CH3, J = 6.0 Hz), 2.90 (d, 1H, CH2, J = 13.8 Hz), 3.09 (d, 1H, CH2, J = 13.8 Hz), 3.81 (s, 1H, CH), 4.13 (s, 5H, C5H5), 4.28 (s, 5H, C5H5), 4.08 (m, 1H, C5H4), 4.16 (m, 2H, C5H4), 4.23 (m, 2H, C5H4), 4.28 (m, 2H, C5H4), 4.42 (m, 1H, C5H4), 5.07 (m, 1H, CH, J = 6.0 Hz), 5.54 (bs, 2H, NH2). 13C-NMR: 21.94 (2CH3), 36.96 (CH2), 41.40, 66.96 (2CH), 69.04, 69.56 (2C5H5), 68.20, 68.54, 68.90, 68.94, 69.09, 69.84, 70.38, 71.52 (2C5H4), 79.25, 83.68 (2CipsoFc), 119.16, 120.49 (2CN), 51.28, 160.43, 165.37, 166.82 (4C); MS: m/z 586 [M]+. Anal. Calcd. for C31H30Fe2N4O: C 63.51, H 5.16, Fe 19.05, N 9.55. Found C 63.67, H 5.03, Fe 19.13, N 9.41.
Reaction of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in the presence of NaOH. The reaction of compound 1 (1.13 g, 5.0 mmol) with malononitrile 2 (0.4 g, 6.0 mmol) and 0.4 g NaOH in methanol (100 mL) and H2O (10 mL) was carried out under conditions described above; subsequent chromatography afforded 3a (15%), 3c and 4a (12%).
2-Amino-4-ferrocenyl-6-hydroxypyridine-3,5-dicarbonitrile (3c). Red crystals, yield 0.98 g (57%), m.p. 146–147 °C; IR (KBr): 423, 495, 508, 580, 814, 839, 910, 1004, 1042, 1103, 1181, 1242, 1290, 1321, 1340, 1378, 1420, 1468, 1481, 1540, 1554, 1612 2211, 2221, 2327, 2985, 3109, 3219, 3371, 3489, 3670 cm−1; 1H-NMR: 4.27 (s, 5H, C5H5), 4.58 (m, 2H, C5H4), 5.21 (m, 2H, C5H4), 5.60 (bs, 2H, NH2), 5.73 (bs, 1H, OH); 13C-NMR: 71.05 (C5H5), 70.54, 71.22 (C5H4), 82.50 (CipsoFc), 116.31, 117.61 (2CN), 157.70, 160.26, 160.82, 161.52, 167.58 (5C); MS: m/z 344 [M]+. Anal. Calcd. for C17H12FeN4O: C 59.33, H 3.52, Fe 16.23, N 16.27. Found: C 59.24, H 3.47, Fe 16.09, N 16.18.
Reactions of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in the presence of amines. A solution of compounds 1 (5.0 mmol) and 2 (6.0 mmol), piperidine or morpholine (2.0 mL) in methanol (100 mL) was stirred for 6 h at 60 °C. The reaction mixture was evaporated in vacuo, and the residue was subjected to TLC on SiO2 (hexane–dichloromethane, 2:1) to give compounds 3a (~20%, Rf = 0.78), 4a (~9%, Rf = 0.67) and 3d,e (58–61%, Rf = 0.35–0.54).
2-Amino-4-ferrocenyl-6-piperidinopyridine-3,5-dicarbonitrile (3d). Red crystals, yield 1.25 g (61%), m.p. 182–183 °C; IR (KBr): 416, 481, 503, 585, 814, 912, 1001, 1019, 1100, 1177, 1251, 1289, 1311, 1343, 1392, 1425, 1464, 1472, 1546, 1553, 1612, 2217, 2226, 2334, 2973, 3101, 3239, 3388, 3469 cm−1; 1H-NMR: 1.70 (m, 2H, CH2), 1.83 (m, 4H, 2 CH2), 3.18 (m, 4H, 2CH2), 4.32 (s, 5H, C5H5), 4.56 (m, 2H, C5H4), 5.05 (m, 2H, C5H4), 5.34 (bs, 2H, NH2); 13C-NMR: 24.55 (CH2), 26.05 (2CH2), 49.83 (2CH2), 70.86 (C5H5), 70.66, 70.90 (C5H4), 83.00 (CipsoFc), 118.52, 119.29 (2CN), 160.51 (2C), 155.51, 160.74, 163.32 (3C); MS: m/z 411 [M]+. Anal. Calcd. for C22H21FeN5: C 64.25, H 5.15, Fe 13.58, N 17.02. Found: C 64.33, H 5.07, Fe 13.61, N 16.89.
2-Amino-4-ferrocenyl-6-morpholinopyridine-3,5-dicarbonitrile (3e). Red crystals, yield 1.20 g (58%), m.p. 190–192 °C; IR (KBr): 432, 491, 512, 591, 815, 861, 908, 1002, 1041, 1101, 1120, 1215, 1251, 1299, 1312, 1340, 1396, 1442, 1470, 1510, 1567, 1621, 1692, 2212, 2227, 2989, 3138, 3278, 3363, 3476 cm−1; 1H-NMR: 3.26 (m, 4H, 2CH2), 3.80 (m, 4H, 2CH2), 4.37 (s, 5H, C5H5), 4.68 (m, 2H, C5H4), 5.01 (m, 2H, C5H4), 5.65 (bs, 2H, NH2); 13C-NMR: 47.09 (2CH2), 50.98 (2CH2), 70.99 (C5H5), 67.17, 70.66 (C5H4), 80.35 (CipsoFc), 116.03, 118.31 (2CN), 157.68 (2C), 155.25, 161.21. 168.65 (3C); MS: m/z 413 [M]+. Anal. Calcd. for C21H19FeN5O: C 61.04, H 4.63, Fe 13.52, N 16.94. Found: C 60.94, H 4.48, Fe 13.44, N 17.08.
Chemical transformations of 2-cyano-3-ferrocenylacrylonitrile (1) in the presence of 2-PrOH, H2O and Na2CO3. A mixture of compound 1 (1.13 g, 5.0 mmol), 2-propanol (60 mL), H2O (10 mL) and Na2CO3 (1.0 g, 10mmol) was stirred for 12 h at 80 °C. The reaction mixture was worked up as described above, subsequent chromatography on Al2O3 gave compounds 3b (25%) and 4b (27%), respectively, and polimeric compounds.
3.2. Crystal Structures of 3b, 4a and 4b
Single crystals of 3b and 4b were obtained by crystallization from chloroform, while crystals of 4a were obtained by crystallization from methanol. The unit cell parameters and the X-ray diffraction intensities were recorded on a Gemini (detector Atlas CCD, Cryojet N2, Loveland, CO, USA) diffractometer. The structures of compounds 3b, 4a and 4b were solved by the direct method (SHELXS-97 [35]) and refined using full-matrix least-squares on F2.
Crystal data for C20H18FeN4O (3b): M = 386.23 g·mol−1, orthorhombic P bca, a = 12.4557(8), b = 14.9714(6), c = 18.4217(7) Å, α = 90, β = 90, γ = 90°, V = 3435.3(3) Å3, T = 130(2) K, Z = 8, ρ = 1.494 Mg/m3, wavelength 1.71073 Å, F(000) = 1,600, absorption coefficient 0.895 mm−1, index ranges −15 ≤ h ≤ 15, −18 ≤ k ≤ 17, −23 ≤ l ≤ 23, scan range 3.54 ≤ θ ≤ 26.73°, 3633 independent reflections, Rint = 0.0326, 26385 total reflections, 243 refinable parameters, final R indices [I > 2σ(I)] R1 = 0.0301, wR2 = 0.0705, R indices (all data) R1 = 0.0398, wR2 = 0.0775, goodness-of-fit on F2 1.074, largest difference peak and hole 0.555/−0.310 eÅ−3.
Crystal data for C29H26Fe2N4O·CH3OH (4a): M = 590.28 g·mol−1, monoclinic P21/n, a = 13.1816(4), b = 10.0587(2), c = 20.4566(6) Å, α = 90, β = 107.323(3), γ = 90°, V = 2589.31(12) Å3, T = 130(2) K, Z = 4, ρ = 1.514 Mg/m3, wavelength 1.71073 Å, F(000) = 1,224, absorption coefficient 1.157 mm−1, index ranges −13 ≤ h ≤ 16, −12 ≤ k ≤ 12, −25 ≤ l ≤ 25, scan range 3.62 ≤ θ ≤ 26.05°, 5113 independent reflections, Rint = 0.0387, 18671 total reflections, 354 refinable parameters, final R indices [I > 2σ(I)] R1 = 0.0323, wR2 = 0.0720, R indices (all data) R1 = 0.0441, wR2 = 0.0781, goodness-of-fit on F2 1.034, largest difference peak and hole 0.419/−0.337 eÅ−3.
Crystal data for C31H30Fe2N4O (4b): M = 586.29 g·mol−1, triclinic P-1, a = 10.5168(8), b = 11.7533(9), c = 12.4547(10) Å, α = 90.551(6), β = 111.455(7), γ = 107.402(7)°, V = 1354.90(18) Å3, T = 293(2) K, Z = 2, ρ = 1.437 Mg/m3, wavelength 1.71073 Å, F(000) = 608, absorption coefficient 1.102 mm−1, index ranges −12 ≤ h ≤ 11, −14 ≤ k ≤ 14, −11 ≤ l ≤ 15, scan range 3.55 ≤ θ ≤ 26.06°, 5346 independent reflections, Rint = 0.0500, 9894 total reflections, 344 refinable parameters, final R indices [I > 2σ(I)] R1 = 0.0558, wR2 = 0.1336, R indices (all data) R1 = 0.0785, wR2 = 0.1551, goodness-of-fit on F2 1.050, largest difference peak and hole 0.877/−0.748 eÅ−3. CCDC-878738 (for 3b), CCDC–878739 (for 4a) and CCDC-878741 (for 4b) contain the supplementary crystallographic data for this paper. These data can be obtained free of charge at www.ccdc.cam.ac.uk/const/retrieving.html [or from the Cambridge Crystallographic Data Centre, 12, Union Road, Cambridge DB2 1EZ, UK; Fax: (internat.) +44-1223-336-033; Email: deposit@ccdc.cam.ac.uk].
4. Conclusions
The reaction of 2-cyano-3-ferrocenylacrylonitrile (1) with malononitrile (2) in a MeOH/H2O or 2-PrOH/H2O medium in the presence of Na2CO3, NaOH, piperidine or morpholine affords products of multi-component condensation: 6-alkoxy-2-amino-, 2-amino-6-hydroxy-, 2,6-diamino-4-ferrocenylpiridine-3,5-dicarbonitriles 3a–e, respectively, as well as products of multi-component cyclodimerization: 6-alkoxy-2-amino-4-ferrocenyl-3-ferrocenylmethyl-3,4-dihydropyridine-3,5-dicarbonitriles 4a,b. This method can be widely used in the synthesis of various pyridine derivatives with ferrocenyl substituents. The reactions described in this study should be of interest to synthetic, theoretical and practical organic chemists seeking ways to prepare functionalized ferrocenylpyridines. The electrochemical behavior of compounds 3b, 3d and 4b was investigated by means of cyclic voltammetry. For 3b and 3d two electrochemical processes (Ia,Ic), attributed to the oxidation and reduction of the ferrocene moieties were found. On the other hand, for compound 4b a double electron transfer for both ferrocene groups (Ia,IIa) and the electrochemical monogeneration of the dication species (Ic) were detected.
Acknowledgments
This work was supported by CONACyT (Mexico, Grant 100970) and DGAPA-UNAM (Mexico, Grant IN 211112). Thanks are due to Eduardo Arturo Vázquez López and Vanessa Ramírez-Delgado for their technical assistance.
Conflict of Interest
The authors declare no conflict of interest.
References
- Eicher, T.; Hauptmann, S. The Chemistry of Heterocycles, Structures, Reactions, Synthesis and Applications; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2006; pp. 269–316. [Google Scholar]
- Abdel-Latif, N.A.; Sabry, N.M.; Mohamed, A.M.; Abdulla, M.M. Synthesis, analgesic, and antiparkinsonianprofiles of some pyridine, pyrazoline, and thiopy-rimidine derivatives. Monatsh. Chem. 2007, 138, 715–724. [Google Scholar] [CrossRef]
- Kopf-Maier, P.; Kopf, H. Non-platinum-group metal antitumor agents: History, current status, and perspectives. Chem. Rev. 1987, 87, 1137–1152. [Google Scholar] [CrossRef]
- Miller, T.M.; Ahmed, K.J.; Wrighton, M.S. Complexes of rhenium carbonyl containing ferrocenyl-derived ligands: Tunable electron density at rhenium by control of the redox state of the ferrocenyl ligand. Inorg. Chem. 1989, 28, 2347–2355. [Google Scholar] [CrossRef]
- Rajput, J.; Hutton, A.T.; Moss, J.R.; Su, H.; Imrie, C. Ferrocenyl-nitrogen donor ligands. Synthesis and characterization of Rhodium(I) complexes of ferrocenylpyridine and related ligands. J. Organomet. Chem. 2006, 691, 4573–4588. [Google Scholar]
- Beletskaya, I.P.; Tsvetkov, A.V.; Latyshev, G.V.; Tafeenko, V.A.; Lukashev, N.V. Bis(ferrocenyl)mercury as a source of ferrocenyl moiety in Pd-catalyzed reactions of carbon-carbon bond formation. J. Organomet. Chem. 2001, 637–639, 653–663. [Google Scholar]
- Mamane, V. Metal-catalyzed cross-coupling reactions for ferrocene functionalization: Recent applications in synthesis, material science and asymmetric catalysis. Mini-Rev. Org. Chem. 2008, 5, 303–312. [Google Scholar]
- Schvekhgeimer, M.-G.A. Heterylferrocenes. Russ. Chem. Rev. 1996, 65, 66–69. [Google Scholar]
- Shibata, K.; Katsuyama, I.; Izoe, H.; Matsui, M.; Muramutsu, H. Synthesis of 4,6-disubstituted 2-methylpyridines and their 3-carboxamides. J. Heterocycl. Chem. 1993, 30, 277–281. [Google Scholar]
- Shibata, K.; Katsuyama, I.; Matsui, M.; Muramutsu, H. Synthesis of ferrocenyl-substituted 3-cyano-2-methylpyridines. Bull. Chem. Soc. Jpn. 1990, 63, 3710–3712. [Google Scholar] [CrossRef]
- Zhou, W.-J.; Ji, S.-J.; Shen, Z.-L. An efficient synthesis of ferrocenyl substituted 3-cyanopyridine derivatives under ultrasound irradiation. J. Organomet. Chem. 2006, 691, 1356–1360. [Google Scholar]
- Zhuang, Q.; Jia, R.; Tu, S.; Zhang, J.; Jiang, B.; Zhang, Y.; Yao, C. Green chemistry approach to the synthesis of 2-amino-4-aryl-6-ferrocenylpyridine derivatives by a one-pot reaction in aqueous medium. J. Heterocycl. Chem. 2007, 44, 895–900. [Google Scholar] [CrossRef]
- Zhu, H.; Lin, H.; Guo, H.; Yu, L. Microwave absorbing property of Fe-filled nanotubes synthesized by a practical route. Mat. Sci. Eng. B 2007, 138, 101–104. [Google Scholar]
- Biot, C.; Chavain, N.; Dubar, F.; Pradines, B.; Trivelli, X.; Brocard, J.; Forfar, I.; Dive, D. Structure-activity relation-ships of 4-N-substituted ferroquine analogues: Time to re-evaluate the mechanism of action of ferroquine. J. Organomet. Chem. 2009, 694, 845–854. [Google Scholar] [CrossRef]
- Yao, T.; Rechnitz, G.A. Amperometric enzyme-immunosensor based on ferrocene-mediated amplification. Biosensors 1987, 3, 307–312. [Google Scholar] [CrossRef]
- Epton, R.; Marr, G.; Regers, G.K. The ferrocene analogues of salicylic acid and aspirin. J. Organomet. Chem. 1976, 110, C42–C44. [Google Scholar]
- Biot, C.; Delhaes, L.; N’Diaye, C.M.; Maciejewski, L.A.; Camus, D.; Dive, D. Synthesis and antimalarial activity in vitro of potential metabolites of ferrochloro-quine and related compounds. Bioorg. Med. Chem. 1999, 7, 2843–2847. [Google Scholar] [CrossRef]
- Hartinger, C.G.; Dyson, P.J. Bioorganometallic chemistry—From teaching paradigms to medicinal applications. Chem. Soc. Rev. 2009, 38, 391–401. [Google Scholar] [CrossRef]
- Gasser, G.; Ott, I.; Metzler-Nolte, N. Organometallic anticancer compounds. J. Med. Chem. 2011, 54, 3–25. [Google Scholar] [CrossRef]
- Chavain, N.; Vezin, V.; Dive, D.; Touati, N.; Paul, J.-F.; Buisine, E.; Biot, C. Investigation of the redox behavior of ferroquine, a new antimalarial. Mol. Pharm. 2008, 5, 510–516. [Google Scholar]
- Kaifer, A.E.; de Mendoza, J. Comprehensive Supramolecular Chemistry; Elsevier: Oxford, UK, 1996; Volume 1, pp. 701–725. [Google Scholar]
- Kowalski, K.; Winter, R.F. The synthesis and electrochemistry of 2,5-dimethylazaferrocenes with heteroaryl bridges. J. Organomet. Chem. 2012, 700, 58–68. [Google Scholar] [CrossRef]
- Kowalski, K.; Koceva-Chyla, A.; Pieniazek, K.; Bernasinska, J.; Skiba, J.; Rybarczyk-Pirek, A.J.; Józwiak, Z. The synthesis, structure, electrochemistry and in vitro anticancer activity studies of ferrocenyl-thymine conjugates. J. Organomet. Chem. 2009, 694, 1041–1048. [Google Scholar]
- Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Guy Orpen, A.; Taylor, R. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lengths in organic compounds. J. Chem. Soc. Perkin Trans. II 1987, S1–S19. [Google Scholar]
- Postnov, V.N.; Klimova, E.I.; Pushin, A.N.; Meleshonkova, N.N. The interaction of the 1,3-bis(p-methoxyphenyl)allylic cation with ferrocenyl-1,3-butadienes. Metalloorg. Chem. 1992, 5, 564–569. [Google Scholar]
- Tietze, L.F.; Brasche, G.; Gerike, K.M. Domino Reactions in Organic Synthesis; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods, Fundamentals and Applications, 2nd ed; John Wiley and Sons: New York, NY, USA, 2001; Chapter 5. [Google Scholar]
- Wright, J.R.; Shaffer, K.J.; McAdam, C.J.; Crowley, J.D. 3,5-Diferrocenylpyridine: Synthesis, characterisation, palladium(II) dichloride complex and electrochemistry. Polyhedron 2012, 36, 73–78. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods, Fundamentals and Applications; John Wiley and Sons: New York, NY, USA, 1980. [Google Scholar]
- Zanello, P. Inorganic Electrochemistry, Theory, Practice and Application; The Royal Society of Chemistry: Cambridge, UK, 2003. [Google Scholar]
- Robin, M.B.; Day, P. Mixed valence chemistry. A survey and classification. Adv. Inorg. Chem. Radiochem. 1967, 10, 247–422. [Google Scholar] [CrossRef]
- Gritzner, G.; Küta, J. Recommendations on reporting electrode potencials in nonaqueous solvents. Pure Appl. Chem. 1984, 4, 461–466. [Google Scholar] [CrossRef]
- Postnov, V.N.; Polivin, Y.N.; Sazonova, V.A. Fragmentation of β-dicarbonyl-compounds with ferrocenyl group. Dokl. Akad. Nauk SSSR 1983, 271, 1402–1404. [Google Scholar]
- Toma, S.; Putala, M.; Salisava, M. Ultrasound-accelerated synthesis of ferrocene-containing pyrimidine derivatives. Collect. Czech. Chem. Commun. 1987, 52, 395–398. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS-97, Program for the Refinement of Crystal Structures; University of Göttingen: Göttingen, Germany, 1994. [Google Scholar]
- Sample Availability: Samples of the compounds 3a,b,d and 4a,b are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).