Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruit Samples using High Performance Liquid Chromatography
Abstract
:1. Introduction
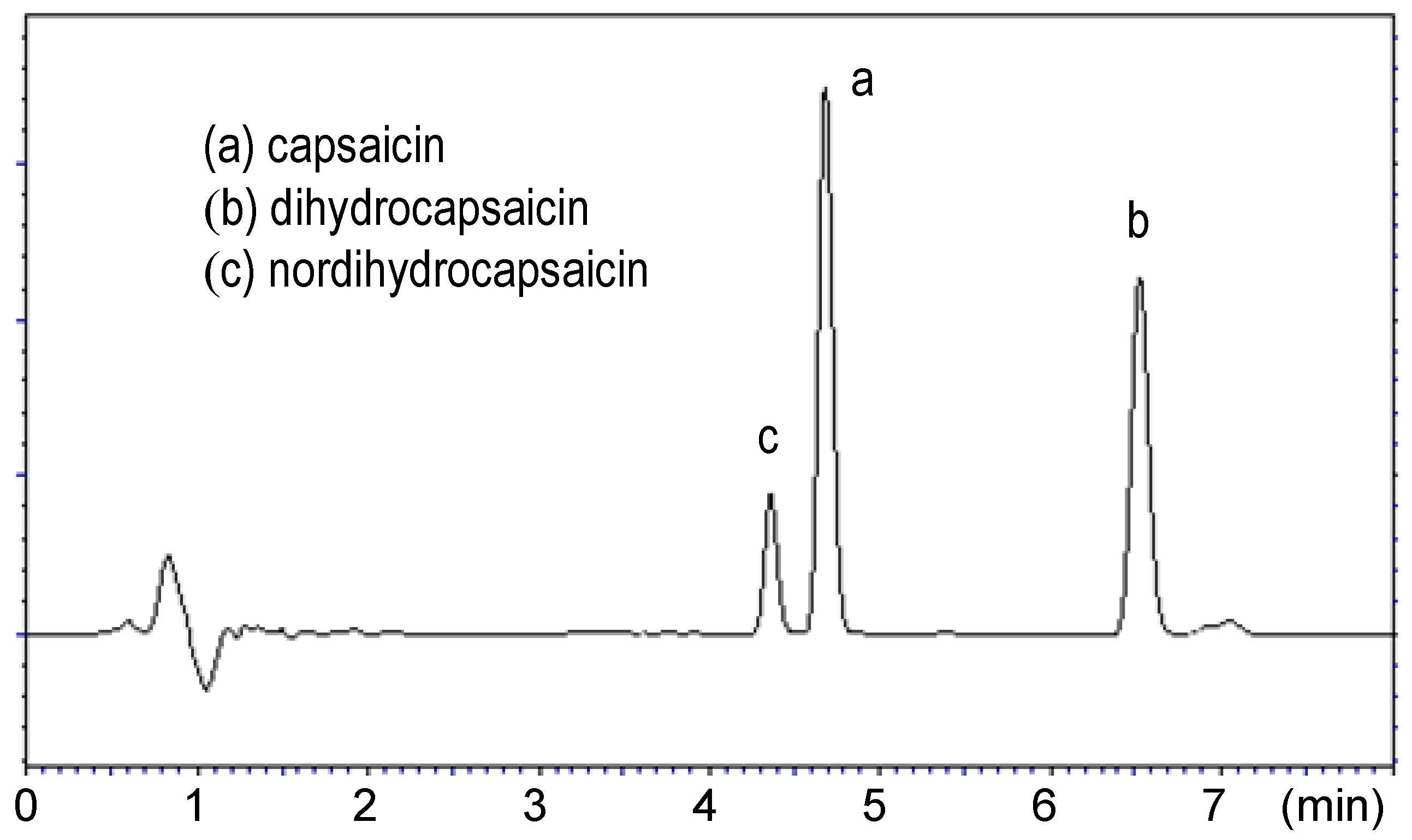
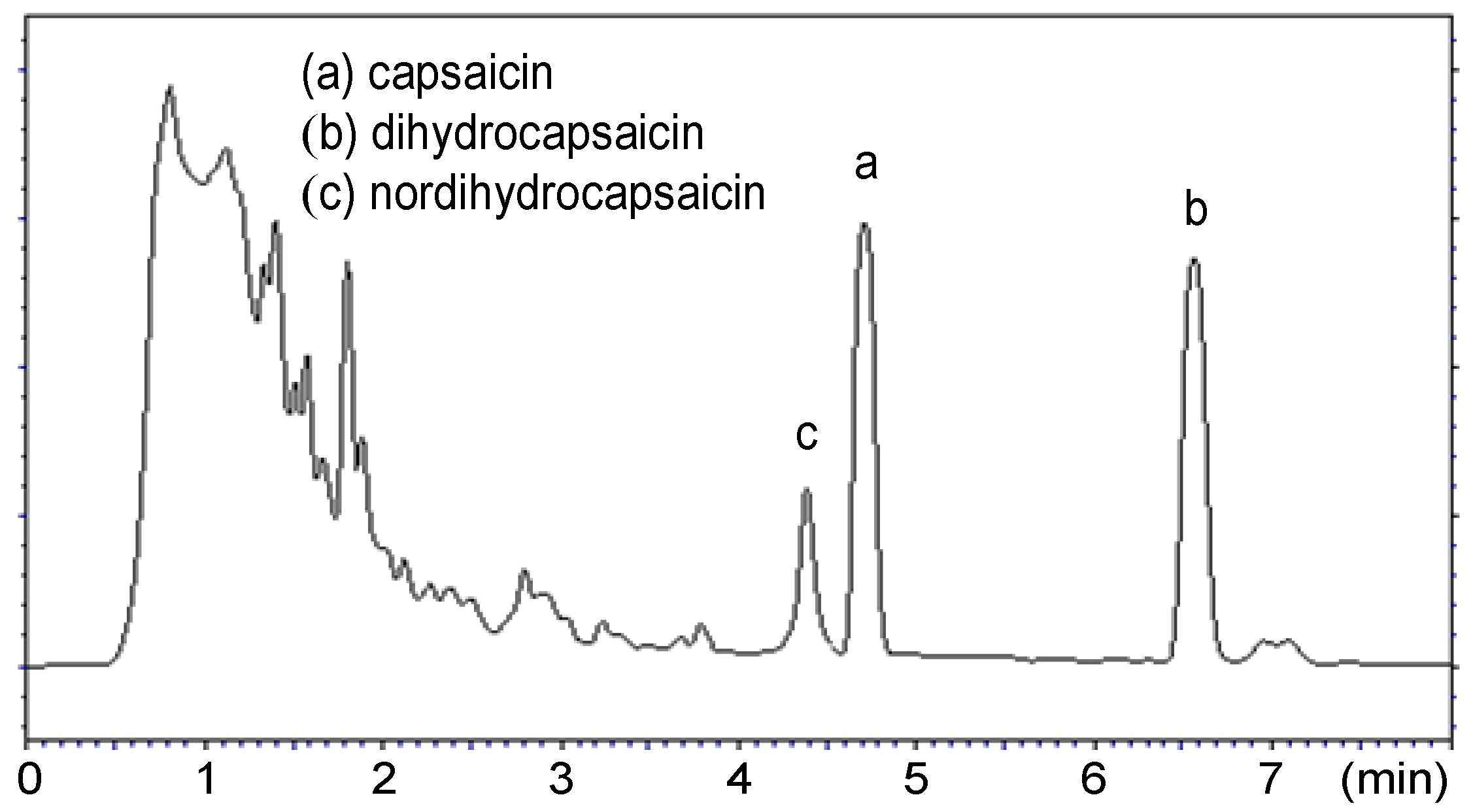
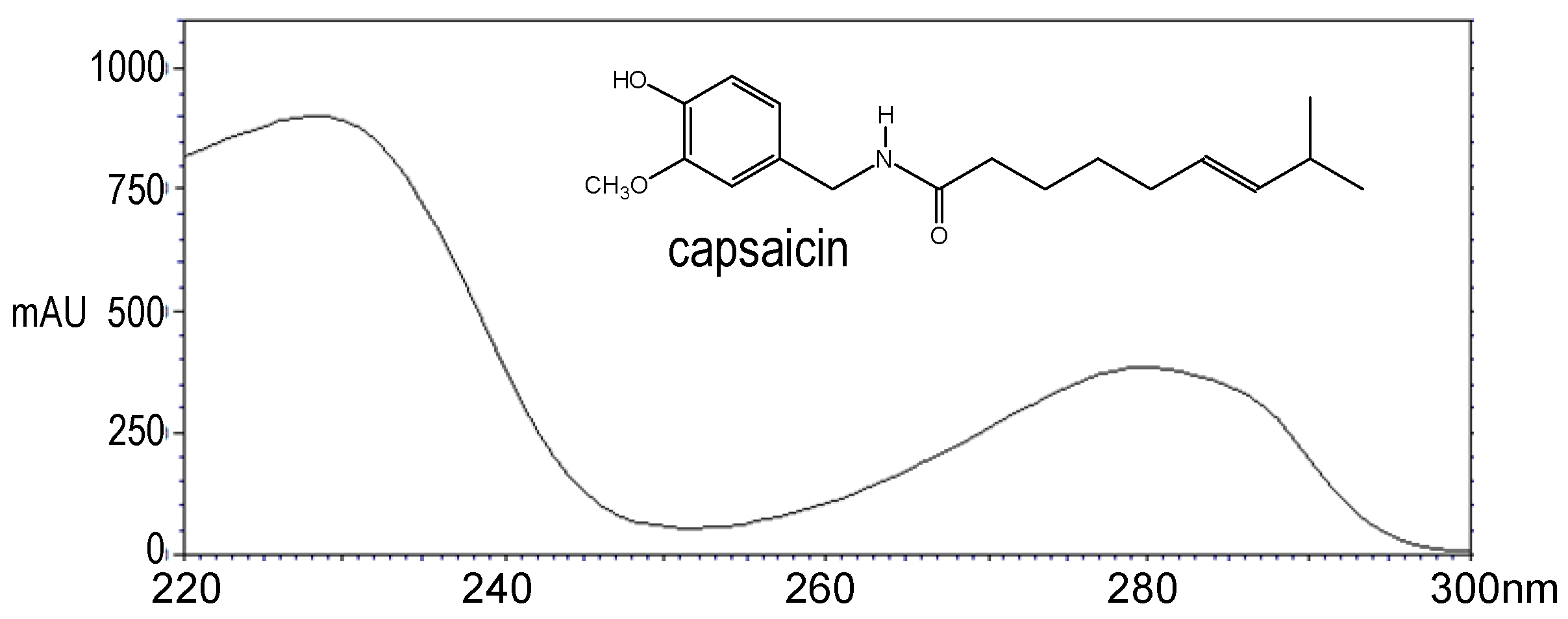
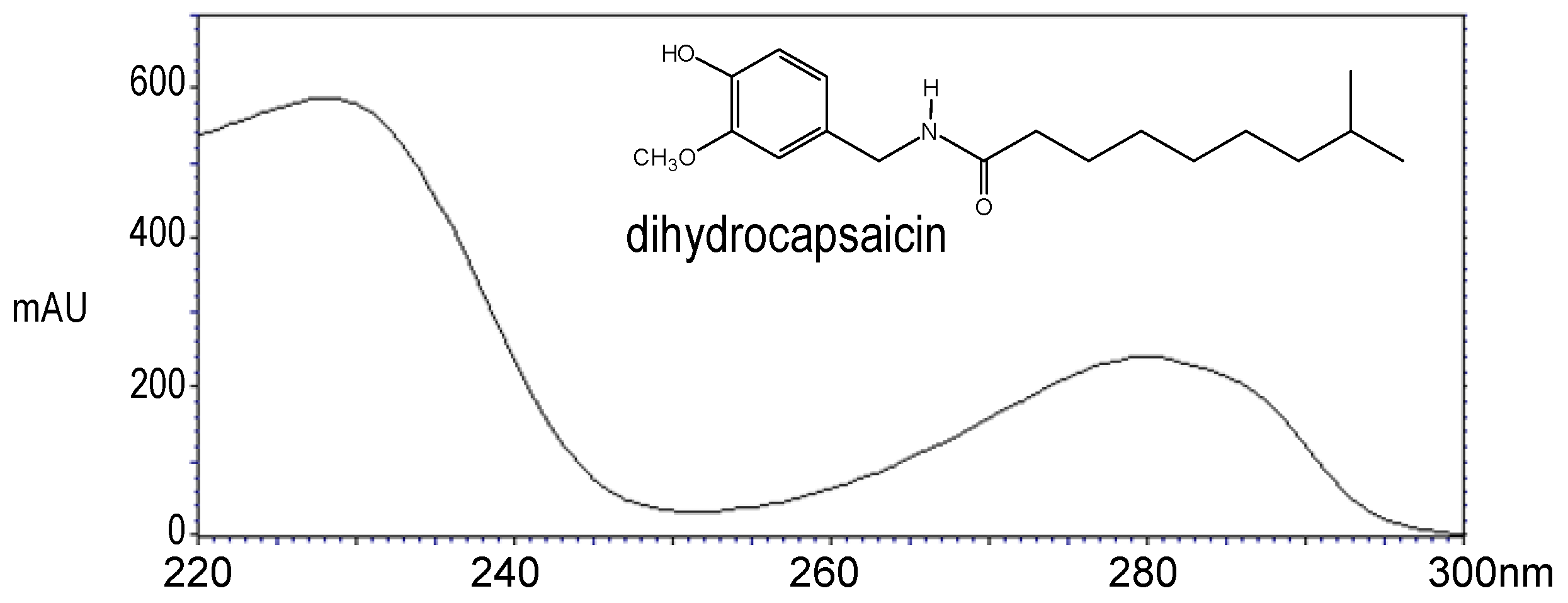
2. Results and Discussion
3. Experimental
3.1. Samples, Chemicals and Solvents
3.2. Extraction of Capsaicinoids
3.3. Liquid Chromatographic Analysis
- Betasil C18 column (particle size 3 μm, dimension 150 × 4.6 mm) from Thermo Electron (USA),
- column temperature: 60 °C, sample temperature: 20 °C, sample volume: 5 μL,
- UV detection wavelength at 222 nm,
- mobile phase: binary mixture water-acetonitrile (CH3CN) at a 50:50 ratio, flow rate: 1.5 mL/min.
3.4. Capsaicinoids Quantitation
3.5. Scoville Heat Unit Conversions
3.6. Estimation of Dietary Capsaicin Intake
4. Conclusions
Conflicts of Interest
Acknowledgments
References
- Perucka, I.; Materska, M. Phenylalanine ammonia-lyase and antioxidant activities of lipophilic fraction of fresh pepper fruits Capsicum annuum L. Innovat. Food Sci. Emerg. Tech. 2001, 2, 189–192. [Google Scholar] [CrossRef]
- Kosuge, S.; Furuta, M. Studies on the pungent principle of Capsicum. Part XIV: Chemical constitution of the pungent principle. Agric. Biol. Chem. 1970, 34, 248–256. [Google Scholar]
- Kawabata, F.; Inoue, N.; Yazawa, S.; Kawada, T.; Inoue, K.; Fushiki, T. Effects of CH-19 sweet, a non-pungent cultivar of red pepper, in decreasing the body weight and suppressing body fat accumulation by sympathetic nerve activation in humans. Biosci. Biotechnol. Biochem. 2006, 70, 2824–2835. [Google Scholar] [CrossRef] [PubMed]
- Hachiya, S.; Kawabata, F.; Ohnuki, K.; Inoue, N.; Yoneda, H.; Yazawa, S.; Fushiki, T. Effects of CH-19 Sweet, a non-pungent cultivar of red pepper, on sympathetic nervous activity, body temperature, heart rate, and blood pressure in humans. Biosci. Biotechnol. Biochem. 2007, 71, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Ohnuki, K.; Moritani, T.; Ishihara, K.; Fushiki, T. Capsaicin increases modulation of sympathetic nerve activity in rats: Measurement using power spectral analysis of heart rate fluctuations. Biosci. Biotechnol. Biochem. 2001, 65, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.L.; Liu, D.Y.; Ma, L.Q.; Luo, Z.D.; Cao, T.B.; Zhong, J.; Yan, Z.C.; Wang, L.J.; Zhao, Z.G.; Zhu, S.J.; Schrader, M.; Thilo, F.; Zhu, M.Z.; Tepel, M. Activation of transient receptor potential vanilloid type-1 channel prevents adipogenesis and obesity. Circ. Res. 2007, 100, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Deal, C.L.; Schnitzer, T.J.; Lipstein, E.; Seibold, J.R.; Stevens, R.M.; Levy, M.D.; Albert, D.; Renold, F. Treatment of arthritis with topical capsaicin: A double-blind trial. Clin. Ther. 1999, 13, 383–395. [Google Scholar]
- Moore, D.J.; Moore, D.M. Synergistic Capsicum-tea mixtures with anticancer activity. J. Pharm. Pharmacol. 2003, 55, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Szolcsanyi, J. Forty years in capsaicin research for sensory pharmacology and physiology. Neuropeptides 2004, 38, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Kempaiah, R.K.; Manjunatha, H.; Srinivasan, K. Protective effect of dietary capsaicin on induced oxidation of low-density lipoprotein in rats. Mol. Cell. Biochem. 2005, 275, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Moriyama, T.; Kobata, K. TRPV1 activation and induction of nociceptive response by a non-pungent capsaicin-like compound, capsiate. Neuropharmacology 2003, 44, 958–967. [Google Scholar] [CrossRef]
- Inoue, N.; Matsunaga, Y.; Satoh, H.; Takahashi, M. Enhanced energy expenditure and fat oxidation in humans with high BMI scores by the ingestion of novel and non-pungent capsaicin analogues (capsinoids). Biosci. Biotechnol. Biochem. 2007, 71, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Mozsik, G.; Szolcsanyi, J.; Racz, I. Gastroprotection induced by capsaicin in healthy human subjects. World J. Gastroenterol. 2005, 11, 5180–5184. [Google Scholar] [PubMed]
- Backonja, M.M.; Malan, T.P.; Vanhove, G.F.; Tobias, J.K. NGX-4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia: A randomized, double-blind, controlled study with an open-label extension. Pain Med. 2010, 11, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Tesfaye, S. Advances in the management of diabetic peripheral neuropathy. Curr. Opin. Support. Palliat. Care 2009, 3, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Sawynok, J. Topical analgesics in neuropathic pain. Curr. Pharm. Des. 2005, 11, 2995–3004. [Google Scholar] [CrossRef] [PubMed]
- Derry, S.; Lloyd, R.; Moore, R.A.; McQuay, H.J. Topical capsaicin for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2009, 7, CD007393. [Google Scholar]
- Reyes-Escogido, M.L.; Gonzalez-Mondragon, E.G.; Vazquez-Tzompantzi, E. Chemical and Pharmacological Aspects of Capsaicin. Molecules 2011, 16, 1253–1270. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Carrillo, L.; Avila, H.M.; Dubrow, R. Chili pepper consumption and gastric cancer in Mexico: A case-control study. Am. J. Epidemiol. 1994, 139, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Scoville, W.L. Note on Capsicum. J. Am. Pharm. Assoc. 1912, 1, 453–454. [Google Scholar] [CrossRef]
- Weiss, E.A. Spice Crops; CABI Publishing International: New York, NY, USA, 2002; p. 411. [Google Scholar]
- Nwokem, C.O.; Agbaji, E.B.; Kagbu, J.A.; Ekanem, E.J. Determination of capsaicin content and pungency level of five different peppers grown in Nigeria. NY Sci. J. 2010, 3, 17–21. [Google Scholar]
- Parrish, M. Liquid chromatographic method of determining capsaicinoids in capsicums and their extractives: Collaborative study. J. Assoc. Off. Anal. Chem. 1996, 79, 738–745. [Google Scholar]
- Thomas, B.V.; Schreiber, A.A.; Weisskopf, C.P. Simple method for quantitation of capsaicinoids in peppers using capillary gas chromatography. J. Agric. Food Chem. 1998, 46, 2655–2663. [Google Scholar] [CrossRef]
- Lopez-Hernandez, J.; Oruna-Concha, M.J.; Simal-Lozano, J.; Gonzales-Castro, M.J.; Vazquez-Blanco, M.E. Determination of capsaicin and dihydrocapsaicin in cayenne pepper and padron peppers by HPLC. Dtsch. Lebensmitt. Rundsch. 1996, 92, 393–395. [Google Scholar]
- Monsereenusorn, Y. Subchronic toxicity studies of capsaicin and capsicum in rats. Res. Commun. Chem. Pathol. Pharmacol. 1983, 41, 95–110. [Google Scholar] [PubMed]
- Council of Europe. Committee of experts on flavouring substances. Datasheet on Capsaicin, 2001. [Google Scholar]
- Govindarajan, V.S.; Sathyanarayana, M.N. Capsicum: Production, technology, chemistry, and quality. Part V. Impact on physiology, pharmacology, nutrition, and metabolism; structure, pungency, pain, and desensitization sequences. Crit. Rev. Food Sci. Nutr. 1991, 29, 435–473. [Google Scholar] [CrossRef] [PubMed]
- CREDOC/OCA (Observatoire des Consommations Alimentaires). Estimation des niveaux d’ingestion de substances aromatisantes safrole, estragole, coumarine et capsaicine. Note Technique No. 98, 1998; 25. [Google Scholar]
- Collins, M.D.; Mayer-Wasmund, L.; Bosland, P.W. Improved method for quantifying capsaicinoids in Capsicum using high performance liquid chromatography. HortScience 1995, 30, 137–139. [Google Scholar]
- Todd, P.; Bensinger, M.; Biftu, T. Determination of pungency due to Capsicum by gas-liquid chromatography. J. Food Sci. 1977, 42, 660–665. [Google Scholar] [CrossRef]
- Sanatombi, K.; Sharma, G.J. Capsaicin content and pungency of different Capsicum spp. cultivars. Not. Bot. Hort. Agrobot. Cluj. 2008, 36, 89–90. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |

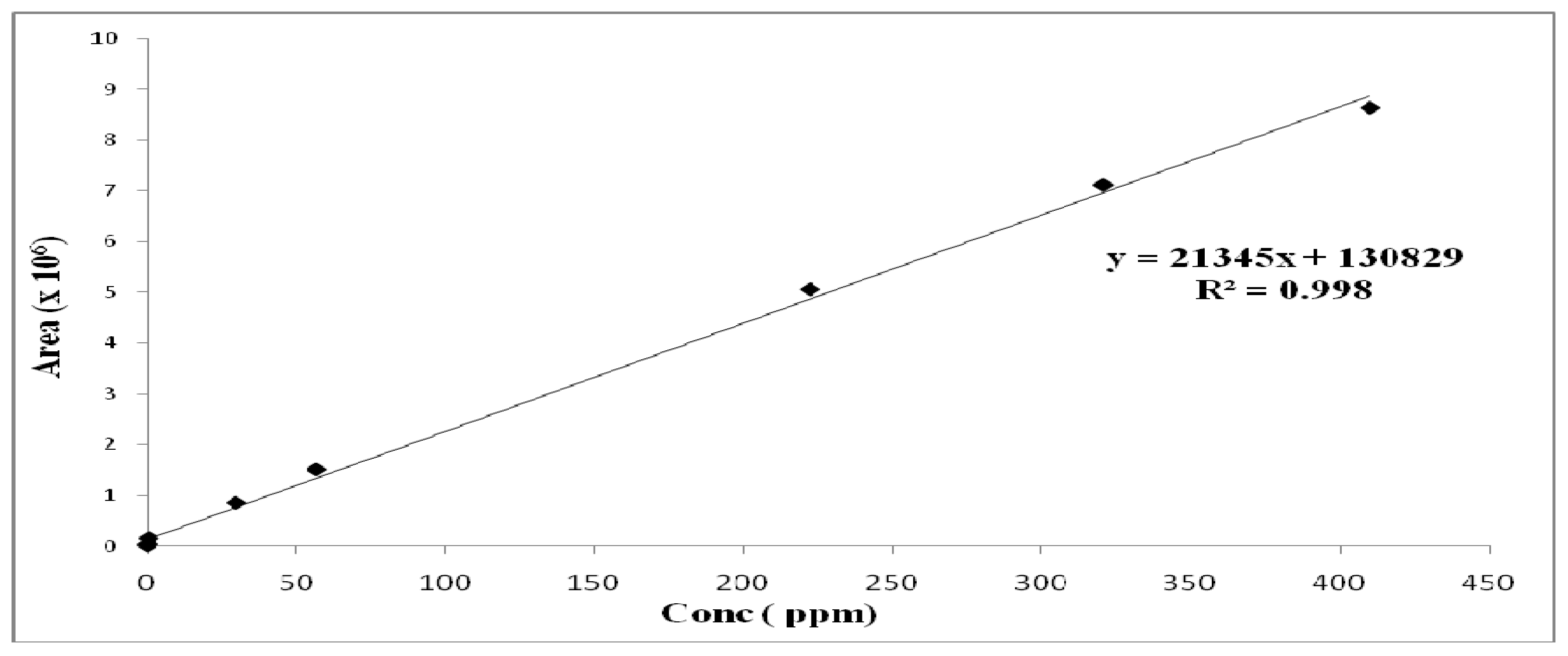
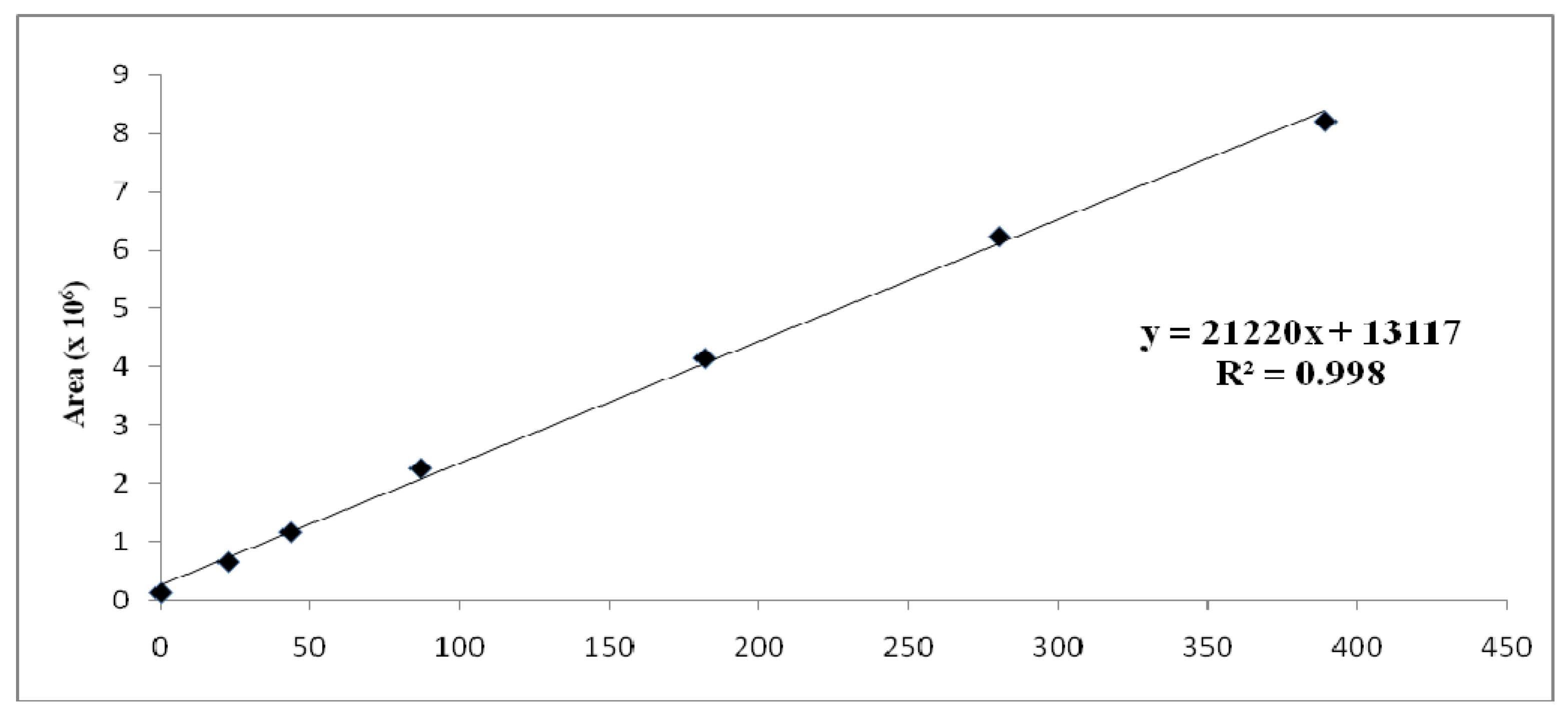
| Parameter | Capsaicin | Dihydrocapsaicin |
|---|---|---|
| Average peak area | 2221184.0 | 1617453.4 |
| SD | 22485.5 | 9286.3 |
| %RSD | 1.01 | 0.57 |
| LOD | 0.09 | 0.1 |
| LOQ | 0.3 | 0.36 |
| Pepper type | Capsaicin (µg/g) | Dihydrocapsaicin (µg/g) | Scoville heat units (SHU) | Levels of pungency |
|---|---|---|---|---|
| Hot chili | 4249.0 ± 190.3 | 4482.2 ± 35.6 | 67984.60 | highly pungent |
| Red chili | 309.3 ± 4.2 | 238.2 ± 2.6 | 4949.08 | moderately pungent |
| Green chili | 138.5 ± 5.2 | 146.4 ± 4.2 | 2216.58 | mildly pungent |
| Green pepper | 1.0 ± 0.9 | ND | 15.83 | non-pungent |
| Red pepper | ND | ND | 0 | non-pungent |
| Yellow pepper | ND | ND | 0 | non-pungent |
| Pepper type | Capsaicin (µg/g) | Daily consumption (g) | Capsaicin daily intake (mg/person/day) |
|---|---|---|---|
| Hot chili | 4249.0 | 1.5 | 6.374 |
| Red chili | 309.3 | 3 | 0.928 |
| Green chili | 138.5 | 2 | 0.277 |
| Green pepper | 0.99 | 5 | 0.005 |
| Red pepper | 0 | 2 | 0 |
| Yellow pepper | 0 | 2 | 0 |
| Average daily intake (mg/person/day) | 7.584 | ||
| Average daily intake (mg/kg body wt/day) | 0.108 | ||
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Othman, Z.A.A.; Ahmed, Y.B.H.; Habila, M.A.; Ghafar, A.A. Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruit Samples using High Performance Liquid Chromatography. Molecules 2011, 16, 8919-8929. https://doi.org/10.3390/molecules16108919
Othman ZAA, Ahmed YBH, Habila MA, Ghafar AA. Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruit Samples using High Performance Liquid Chromatography. Molecules. 2011; 16(10):8919-8929. https://doi.org/10.3390/molecules16108919
Chicago/Turabian StyleOthman, Zeid Abdullah Al, Yacine Badjah Hadj Ahmed, Mohamed Abdelaty Habila, and Ayman Abdel Ghafar. 2011. "Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruit Samples using High Performance Liquid Chromatography" Molecules 16, no. 10: 8919-8929. https://doi.org/10.3390/molecules16108919
APA StyleOthman, Z. A. A., Ahmed, Y. B. H., Habila, M. A., & Ghafar, A. A. (2011). Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruit Samples using High Performance Liquid Chromatography. Molecules, 16(10), 8919-8929. https://doi.org/10.3390/molecules16108919




