Synthesis and Bioactivity of New Phosphorylated R,R’-substituted Sulfoximines
Abstract
:Introduction
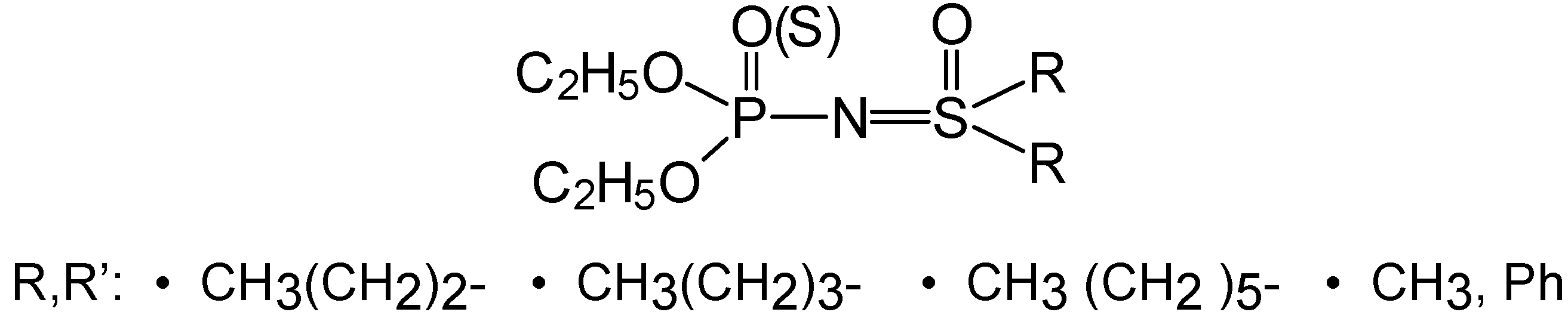
Results and Discussion
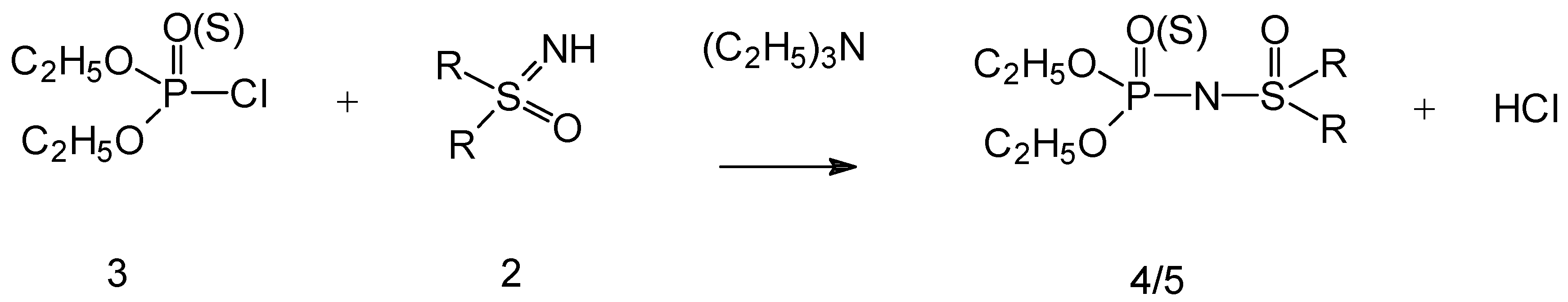
Chemistry
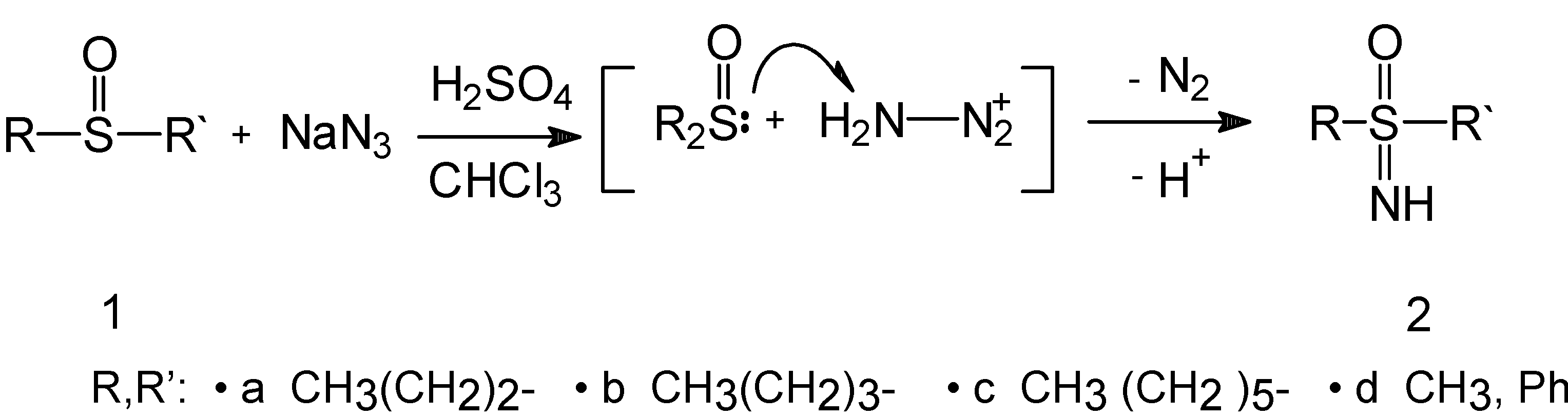
Toxicity and anticholinesterasic assays
| COMPOUND | LD50 µg/ insect | Confidence interval 95% |
|---|---|---|
| Dipropyl | 1.4 | 1.3-1.5 |
| Dibutyl | 1.4 | 1.2-1.5 |
| Dihexyl | 4.9 | 4.5-5.4 |
| Methyl phenyl | 0.5 | 0.3-0.7 |
| COMPOUND | Ki mol-1min-1 |
|---|---|
| Dipropyl | 2.9 x 106 |
| Dibutyl | 1.4 x 106 |
| Dihexyl | 1.3 x 104 |
| Methyl phenyl | 7.4 x 106 |
| Paraoxon | 1.17 x 106 |
Conclusions
Experimental
General
Synthesis of R,R’-substituted sulfoximines 2a-d:
Synthesis of O,O-diethyl-N-(R,R’-disubstituted sulfoximine) phosphoroamidates 4a-d or amido-thionates 5a-d:
Insecticidal activity (LD50)
Anticholinesterase activity
Acknowledgements
References
- Eto, M. Organophosphorus Pesticides: Organic and Biological Chemistry; CRC Press: Cleveland, Ohio, USA , 1974. [Google Scholar]
- Quistad, G.B.; Fukuto, T.R.; Metcalf, R.L. J. Agr. Food Chem. 1970, 18, 189–193.
- Bentley, H.R.; Mc Dermott, E.E.; Moran, T.; Pace, J.; Whitehead, J.K. Proc. Roy. Soc. 1950, B137, 402–417.
- Johnson, C.R. “Sulphoximides”. In Comprehensive Organic Chemistry: Synthesis and Reactions of Organic Compounds; Barton, D., Ollis, W.D., Eds.; Pergamon Press: Oxford-New York, 1979; Vol 3, chapter 11.11; pp. 223–232, (Edited by Jones N., Univ of Sheffield). [Google Scholar]
- O`Brien, R.D. Drug Design; Ariens, E.J., Ed.; Academic Press: New York, 1971; Vol II, chap. 3. [Google Scholar]
- Hollingworth, R.M.; Fukuto, T.R.; Metcalf, R.L. J. Agric. Food Chem. 1967, 15, 235–241. [CrossRef]
- Bentley, H.R.; Mc Dermott, E.E.; Whitehead, J.K. Proc. Roy. Soc. 1951, B 138, 265–272.
- Bellozas Reinhard, M.; Mastrantonio, G.; Jios, J.; Della Vedova, C.O. – to be published –. 2005.
- Oae, S.; Harada, K.; Tsujihara, K.; Furukawa, N. J. Sulfur Chem. 1972 A, 49–61.
- Wieczorkowski, J.; Jakobsen, P.; Treppendahl, S. Acta Chem. Scand. 1983, B 37, 27–30.
- Modro, T.A.; Wieczorkowski, J. Org. Mass Spectrom. 1989, 24, 839–840.
- Mastantonio Garrido, G. Estudio Conformacional de Compuestos Organofosforados y sus Mecanismos de Acción Tóxica. Ph.D. Thesis, La Plata Nacional University, Argentina, 2003. www.sedici.edu.ar/search/request.php?id_document=ARG-UNLP-TPG-0000000038. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.H. Biochem. Pharmacol. 1961, 7, 88–95.
- O`Brien, R. D. Insecticides,Action and Metabolism; Academic Press: New York, 1967. [Google Scholar]
- Leonard, N.J.; Johnnson, C.R. J.Org.Chem. 1961, 27, 282–284.
- Whitehead, J.K.; Bentley, H.R. J. Chem. Soc. 1952, 1572–74.
- Picollo, M.I.; Word, E.J.; Zerba, E.N.; Licastro, S.A.; Ruveda, M.A. Acta Bioquim. Clin. Latinoam. 1976, 10, 67–70.
- Lichfield, J.T.; Wilcoxon, F.J. J. Exp. Therap. 1949, 96, 99–100.
- Fukuto, T.R.; Mercalf, R. L. J. Agric. Food. Chem. 1956, 4, 930–935. [CrossRef]
- Sample Availability: Contact the authors.
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Reinhard, M.B.; De Licastro, S.A. Synthesis and Bioactivity of New Phosphorylated R,R’-substituted Sulfoximines. Molecules 2005, 10, 1369-1376. https://doi.org/10.3390/10111369
Reinhard MB, De Licastro SA. Synthesis and Bioactivity of New Phosphorylated R,R’-substituted Sulfoximines. Molecules. 2005; 10(11):1369-1376. https://doi.org/10.3390/10111369
Chicago/Turabian StyleReinhard, Monica Bellozas, and Susana Arnstein De Licastro. 2005. "Synthesis and Bioactivity of New Phosphorylated R,R’-substituted Sulfoximines" Molecules 10, no. 11: 1369-1376. https://doi.org/10.3390/10111369
APA StyleReinhard, M. B., & De Licastro, S. A. (2005). Synthesis and Bioactivity of New Phosphorylated R,R’-substituted Sulfoximines. Molecules, 10(11), 1369-1376. https://doi.org/10.3390/10111369




