Abstract
Background: This review examines the expanding influence of artificial intelligence (AI) in the detection and management of uveal melanoma (UM). Methods: This work delves into the application of AI technologies such as machine learning (ML), deep learning (DL), and convolutional neural networks (CNNs) in various diagnostic procedures, molecular profiling, and predictive analysis. Results: The discussion underscores AI’s potential to enhance diagnostic precision and efficiency. Particular focus is placed on its role in histopathological assessments of UM, where algorithms facilitate the analysis of whole-slide images (WSIs). AI contributes to more accurate tumor classification, assists in planning treatments, and improves the prediction of the prognostic indicators and molecular characteristics of the tumor. Conclusions: Despite these promising developments, this review acknowledges existing hurdles to AI implementation, including issues with data standardization and the interpretability of AI models. It emphasizes the need for further research to fully integrate AI into clinical workflows, ultimately aiming to improve patient care and outcomes.
1. Introduction
Uveal melanoma (UM) is the most frequent primary intraocular malignancy in adults. The most common site is the choroid (85–90%), whereas the iris and ciliary body represent rare locations (9–15%). Ciliary body melanomas are those with the poorest prognosis, whereas UMs arising in the iris generally have a better prognosis because they are discovered at an early stage. Around half of patients with UM develop metastasis, with the liver representing the most common metastatic site, resulting in death within a year. Patients with metastatic disease have a median survival of 4–15 months [1].
Ocular management for UM aims to retain the eye-ball and visual function. Enucleation used to be the preferred treatment but has now been replaced with a combination of phototherapy, radiotherapy, and local tumor resection. Different features, such as tumor diameter and location, the presence of vitreous hemorrhage, retinal detachment, or retinal involvement, have an impact on treatment decisions. Unfortunately, there is no definitive therapeutic strategy for metastatic UM [1].
In recent years, artificial intelligence (AI) has proven to be a valuable asset in oncology, including in its application in uveal melanoma (UM) [2]. This review aims to provide an overview of the most recent advancements in AI utilization for UM, focusing on studies published up to 2024. AI technologies such as machine learning (ML), deep learning (DL), and convolutional neural networks (CNNs) are pivotal in medical imaging and diagnostics. These tools are engineered to handle large datasets, recognize complex patterns, and generate insights that support clinical decision-making. In the context of UM, AI has been employed across various diagnostic domains, including image interpretation, molecular analysis, and predictive modeling.
2. Materials and Methods
2.1. Search Strategy
The authors conducted a comprehensive electronic search of the literature up to November 2024, utilizing the following databases to identify pertinent studies: PubMed/MEDLINE, Cochrane Library (including the Cochrane Database of Systematic Reviews and Cochrane Central Register of Controlled Trials—CENTRAL), Web of Science (Science and Social Science Citation Index), and Scopus. The search strategy combined both MeSH and non-MeSH terms, as detailed below.
(i) PubMed/MEDLINE: ((artificial intelligence [Title/Abstract]) OR (machine learning [Title/Abstract]) OR (Deep Learning [Title/Abstract]) OR (Convolutional Neural Networks [Title/Abstract]) OR (Histopathology [Title/Abstract]) OR (Molecular Diagnosis [Title/Abstract]) OR (Genomics [Title/Abstract])) AND (Uveal Melanoma [Title/Abstract]). Filters applied: English language. (ii) Cochrane Library: “artificial intelligence” in Title Abstract Keyword AND “uveal melanoma” in Title Abstract Keyword (including word variations). Language: English. (iii) Web of Science: “artificial intelligence” (Topic) AND “uveal melanoma” (Topic) OR “machine learning” (Topic) AND “uveal melanoma” (Topic), restricted to English language. (iv) Scopus: (TITLE-ABS-KEY (artificial intelligence) AND TITLE-ABS-KEY (uveal melanoma)) OR (TITLE-ABS-KEY (deep learning) AND TITLE-ABS-KEY (uveal melanoma)) AND (LIMIT-TO (LANGUAGE, “English”)).
Additionally, the reference lists of relevant publications were manually screened to ensure the inclusion of any studies potentially missed during the electronic search.
2.2. Inclusion and Exclusion Criteria
The studies included in the review were published in the period from September 2008 to November 2024. Articles that did not focus on uveal melanoma or approached it without emphasizing the use of AI in histopathological or molecular diagnosis, or in patient outcomes, were excluded. The review also omitted comments, opinions, perspectives, guidelines, editorials, case reports, previously published systematic reviews or meta-analyses, and papers in languages other than English. Publications limited to abstracts or those providing minimal or non-informative content were also excluded.
The inclusion criteria prioritized articles written in English with full-text availability, particularly those offering comprehensive analyses or detailed discussions of AI applications in UM. These applications included diagnosis, prognostic prediction, clinical management, and insights into molecular characteristics. Specifically, the inclusion criteria were as follows:
- (i)
- Involve patients diagnosed with UM;
- (ii)
- Use AI for histopathological or molecular diagnosis of UM;
- (iii)
- Use AI in predicting patient outcomes associated with UM.
3. Results
Our search of the literature identified 24 significant studies focusing on the application of AI in diagnosis, molecular analysis, and outcome prediction for patients with uveal melanoma (UM) [2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27]. In the realm of histopathology, AI algorithms, particularly deep learning (DL), have been utilized to identify UM and differentiate it from benign tumors such as choroidal nevi. Despite these advancements, challenges such as limited dataset availability and uncertain clinical applicability persist [28,29]. From a molecular perspective, AI has been employed to examine gene expression profiles (GEPs) and methylation patterns in UM. However, the field faces obstacles, including the need for larger, more diverse datasets and additional validation of the findings. AI models have demonstrated potential in improving treatment outcome predictions and stratifying prognostic risks, particularly concerning metastatic potential. Nevertheless, issues such as data biases, insufficient sample sizes, and the necessity for more extensive studies continue to hinder broader application. Relevant information was systematically extracted and is presented narratively. Figure 1 summarizes the main applications of AI in UM diagnosis and prognosis.
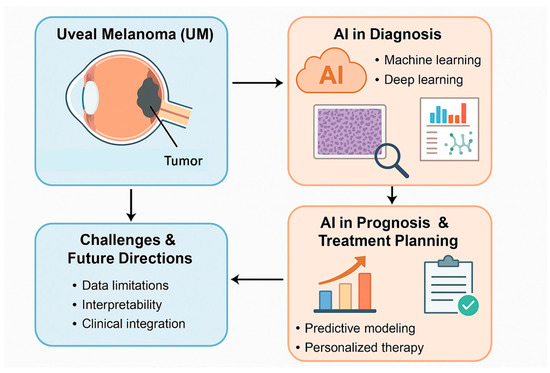
Figure 1.
The main applications of AI in UM are summarized.
4. Discussion
4.1. AI in Histopathological Diagnosis
The methods commonly used to diagnose UM assess the tumor’s diameter and thickness to classify it into four T categories (from T1 to T4). Further staging considers ciliary body involvement and extraocular extension, which help define progressively higher-risk categories for metastatic death (ranging from T1a to T4e) [30].
Following this, lymph node involvement and distant metastases are evaluated, allowing the T stages to be grouped into AJCC stages, with progressively worse prognoses (ranging from stage I to IV). Another tumor-related feature associated with high mortality is the presence of epithelioid cells compared to spindle cells [30]. Additionally, a high mitotic figure count—indicating the increased proliferative activity of tumor cells—has also been linked to a poorer prognosis [31]. Histological analysis can also be used to examine blood vessels within the tumor. Microvascular density, assessed by staining for vascular markers such as CD31, CD34, and von Willebrand factor, is predictive of a worse prognosis [32]. Moreover, UM may exhibit extravascular loops and networks, best visualized with periodic acid–Schiff staining and a dark green filter. These structures are associated with poor prognosis [33].
In routine diagnostic pathology, the immunohistochemical expression of S100 and SOX10 has traditionally been considered the most sensitive marker of melanocytic lineage/differentiation when dealing with metastases from an unknown primary tumor. However, most studies on S100 immunoreactivity in melanocytic neoplasms have primarily focused on CM. Some studies, however, suggest a significant lack of S100 immunoreactivity in metastatic UM, in stark contrast to the widespread positivity seen in both primary UM and metastatic CM [34].
One of the primary applications of ML in this field is prognostication in uveal melanoma (UM). For example, DL-based analysis of digital pathology images shows great potential, particularly in predicting the gene expression profile (GEP) of UM tumors. Looking ahead, we anticipate that more advanced DL techniques—such as generative adversarial networks (GANs), few-shot learning, and zero-shot learning—will be increasingly used to address the challenges posed by the rarity of ocular oncology cases [35].
A study by Tahmasebzadeh et al. concluded that artificial intelligence is an emerging tool in image analysis, enhancing diagnostic workflows from data and image processing to clinical decision-making. It contributes to more personalized treatment approaches, improves response prediction, and refines prognosis assessment [36].
AI application in the histopathological analysis of UM represents a promising frontier. UM is a rare yet aggressive cancer of the eye, and its accurate diagnosis and prognosis assessment are crucial for determining appropriate patient treatment and management [37]. The histopathological examination of tissue samples remains the gold standard for diagnosing and classifying UM. However, the complexity of histological data and the subjective nature of human interpretation pose significant challenges in ensuring accurate diagnoses. AI, in particular DL algorithms such as CNNs, has shown potential in overcoming these limitations by providing a more reliable, reproducible, and efficient method of analyzing histopathological images.
One of the key advantages of AI in this domain is its ability to achieve high diagnostic accuracy in distinguishing between benign choroidal nevi and malignant UMs. Studies have demonstrated that AI algorithms outperform traditional diagnostic methods in certain contexts. For instance, a study by Dadzie et al. [2] highlighted that AI models could identify small melanomas with superior sensitivity compared to both resident and consultant ophthalmologists. The algorithm achieved comparable sensitivity and specificity to ocular oncologists, proving that AI can be an effective tool in complex diagnostic scenarios, especially when differential diagnosis is challenging. Moreover, AI shows consistency in assessing the same image every time it is analyzed, without human interobserver variability, and this may represent a crucial advantage in ensuring reliable results across different healthcare settings.
In addition to diagnostic accuracy, AI’s ability to integrate multiple data sources, such as histopathological images, clinical features, and genetic information, has further enhanced its value.
Another notable advantage of AI in histopathology is its ability to reduce human error and subjectivity in interpretation. Histopathological evaluation is often prone to interobserver and intraobserver variability, especially when assessing small or ambiguous lesions. AI algorithms trained on large datasets can provide consistent results by focusing on objective image features, significantly decreasing the potential for misclassification or oversight. As Chandrabhatla et al. pointed out, the use of AI models can be particularly beneficial in routine screenings and early detection, as the system can analyze large volumes of data efficiently, helping clinicians save time and focus on more complex cases [3].
Furthermore, the ability of AI to automatically segment and highlight regions of interest within histopathological images is another strength. Sabazade et al. noted that AI’s segmentation capabilities help isolate key features such as tumor boundaries, areas of infiltration, and abnormal tissue structures, thereby improving diagnostic clarity [4]. This segmentation not only increases classification accuracy but also makes the model’s decision-making process more interpretable, providing clinicians with insights into the specific features that contributed to the diagnosis. This transparency is critical for fostering trust between healthcare providers and the AI system, as it enables clinicians to better understand and validate AI-driven predictions.
However, despite these significant advantages, there are notable limitations that must be addressed for AI to reach its full potential in the histopathological analysis of UM. One of the most pressing challenges is the quality and diversity of the datasets used to train AI models. As highlighted by Wan et al., the accuracy of AI models is heavily dependent on the quality of the data they are trained on [5]. Small or non-representative datasets may lead to overfitting, where the model becomes highly specialized to the training data but fails to generalize well to new or unseen cases [5]. The rarity of UM further exacerbates this issue, as limited samples from diverse populations may hinder the model’s ability to detect atypical lesions or those with unusual presentations.
Moreover, while CNNs have demonstrated excellent performance in analyzing histopathological images, they are often considered “black-box” models. This means that the decision-making process of the AI system is not always transparent, which can be a barrier to clinical adoption. The lack of interpretability can make it difficult for clinicians to understand why the AI made a particular prediction or diagnosis, limiting their trust in the system. Techniques like saliency maps, as proposed by Zhang et al., could help address this issue by highlighting the key image features that influenced the model’s decision, thereby improving interpretability and fostering clinician confidence [6].
Additionally, AI’s ability to process only the data it has been trained on raises concerns regarding the generalizability of these models across different clinical settings. As noted by Sabazade et al., the algorithm they studied was trained on a limited set of images, which could potentially reduce its effectiveness when applied to different imaging systems or institutions with varying practices and equipment [4]. For AI to be successfully integrated into clinical practice, it must be trained on larger, more diverse datasets that include a wide range of image qualities and lesion types. This would ensure that the models are robust enough to perform well across different real-world scenarios.
Lastly, the implementation of AI in histopathology also requires validation through large-scale, prospective clinical studies. While retrospective studies have shown promising results, the true clinical value of AI can only be fully realized when it is tested in prospective trials with broader patient populations. Iddir et al. highlighted the importance of continued validation in diverse clinical settings to confirm the efficacy and reliability of AI algorithms in real-world applications [7].
In conclusion, while AI holds significant promise for improving the diagnosis, prognosis, and treatment of UM through histopathological analysis, challenges related to data quality, interpretability, and generalizability remain. Overcoming these limitations will require further research, improved dataset diversity, and the development of more transparent AI models to ensure their successful integration into routine clinical practice. The main advantages and limitations of AI applications in the histopathological diagnosis of UM are summarized in Table 1. AI, particularly DL and CNNs, enhances diagnostic accuracy and consistency in identifying UM, especially in distinguishing malignant tumors from benign nevi. Strengths include a reduction in human error, improved reproducibility, and the effective integration of multi-source data. However, limitations persist due to small, non-diverse datasets, interpretability challenges, and the lack of large-scale clinical validation.

Table 1.
AI applications in UM histopathological diagnosis.
4.2. AI in Molecular and Genomic Analysis
The BAP1 gene, situated on chromosome 3p21.1, encodes a nuclear ubiquitinase that plays a crucial role in the epigenetic regulation of chromatin. It is a key tumor suppressor, and its mutational inactivation is a pivotal step in the development of metastatic potential in uveal melanoma (UM) [10].
In a 2019 study, Sun et al. utilized a densely connected neural network to predict BAP1 expression from ophthalmic histopathology images, achieving a remarkable accuracy of 97% [11]. Similarly, Zhang et al. developed deep learning models to estimate nuclear BAP1 expression using hematoxylin and eosin (H&E)-stained histopathology slides, achieving an accuracy of 93% [12]. Gene expression profiling (GEP) is considered the best indicator to predict the survival of patients with UM, with two identified classes showing a significant difference in long-term survival (92-month reported survival is 95% and 31% for Class 1 and Class 2, respectively). Therefore, considering the importance of correctly assessing the GEP of UM, Liu et al. developed a DL model with a dual-attention feature extraction mechanism to directly predict the GEP from digital H&E-stained cytopathology images, achieving 91.7% sensitivity, specificity, and accuracy [13].
As the GEP represents a pivotal prognostic factor in UM, Akram et al. tried to predict molecular subclasses of UM based on H&E-stained sections through a CNN [14]. Their models correctly identified 14/20 BAP1mut, 8/10 EIF1AXmut, and 11/17 SF3B1mut UM, suggesting that these models were able to predict the molecular signature of the tumor based on some morphological features.
Liu et al. conducted a study to develop a DL system that can determine patient survival using smeared cytologic aspirates from fine-needle aspiration biopsy (FNAB) samples, with the GEP as the reference standard [15]. This DL system predicted the GEP of the tumor in 15/20 cases (75%).
Genomic studies have revealed that uveal melanoma (UM) is driven by a small number of key driver events, resulting in four distinct molecular subtypes associated with varying metastatic risks. These subtypes are as follows: Class A (Disomy of chromosome 3 without hotspot mutations in SF3B1); Class B (Disomy of chromosome 3 with hotspot SF3B1 mutations); Class C (Monosomy of chromosome 3 with BAP1 mutations but no amplification of chromosome 8q); and Class D (Monosomy of chromosome 3 with BAP1 mutations, amplification of chromosome 8q, and an inflammatory infiltrate).
Metastatic risk is stratified by subtype, being low in Class A, intermediate in Class B, and high in Classes C and D [16].
In a 2024 study, Reggiani et al. utilized machine learning (ML) approaches to identify genes linked to copy number alterations (CNAs) and developed a model capable of distinguishing monosomic from disomic chromosome 3 samples, leveraging data from The Cancer Genome Atlas (TCGA) UM cohort [17]. Similarly, Xie et al. [18] conducted bioinformatics analyses of gene expression profiles in UM, including cases with metastasis, identifying several hub genes (e.g., FN1, APOB, F2, SERPINC1, SERPINA1, APOA1, FGG, PROC, ITIH2, VCAN, TFPI, CXCL8, CDH2, and HP) that may play crucial roles in the metastatic progression of UM.
Another interesting aspect to consider for predicting the clinical behavior of UM is the methylation profile of the tumor. In this regard, Hou et al. used ML models to detect DNA methylation signatures in UM, which may be of prognostic relevance [19]. They identified two risk groups, with significantly different prognosis in terms of survival.
The main advantages and limitations of AI applications to assess the GEP and molecular features of UM are summarized in Table 2. AI applications in this field have shown strong performance using routine histological images. These tools offer non-invasive, rapid molecular profiling potential. Limitations include reliance on small datasets, a lack of population diversity, and the need for broader validation across centers and staining techniques.

Table 2.
AI applications to assess UM molecular features.
4.3. AI in Prognostic Prediction
The possibility of applying AI models to identify prognostic factors in patients affected by UM has been investigated in different studies during recent years. In 2022, Bassi et al. analyzed the possibility of predicting treatment outcomes in patients with UM. In particular, they highlighted the role of AI models in weighing the significance of biomarkers, such as lactate dehydrogenase (LDH), against other prognostic variables with respect to treatment outcomes [20].
Lever et al. analyzed hepatic metastases from UM by using ML tools, showing that substantial binary risk stratification can be performed, based on (i) clinical and laboratory parameters, (ii) measures of quantitative overall hepatic tumor burden, and (iii) radiomic parameters [21]. However, the combination of two or all three domains failed to improve prognostic stratification. They also identified highly relevant parameters, including LDH, platelet count, aspartate aminotransferase (AST), and the metastasis-free interval at the first diagnosis of metastatic disease, as predictors for time-to-treatment failure and overall survival (OS). The risk stratification models based on this ML algorithm identified a comparable and independent prognostic value of clinical, radiological, and radiomic parameters in UM patients with hepatic metastases.
In recent years, there has been growing interest in utilizing AI to identify factors predictive of metastatic disease. In a study aimed at predicting survival in patients with uveal melanoma (UM), Chen et al. employed random forest, a classical machine learning (ML) method, to develop two predictive models for estimating the risk of death and metastasis within two years [22]. These models were trained using demographic characteristics, ophthalmic examination variables (notably, intraocular pressure), and tumor-specific features, including largest basal diameter, thickness, pigmentation, location, macroscopic appearance, optic nerve and ciliary body involvement, subretinal fluid, intraocular hemorrhage, extraocular extension, TNM stage, and initial treatment. The study identified largest basal diameter, thickness, tumor size, intraocular pressure, and initial treatment as the most significant parameters. For death prediction, the model achieved an AUC of 0.883 and an accuracy of 76.9%, while for metastasis prediction, it achieved an AUC of 0.846 and an accuracy of 74.9%.
Similarly, Donizy et al. utilized classical ML models to identify predictors of metastasis and survival in a cohort of 164 patients with UM who underwent ocular enucleation without prior treatment [23]. Three models were constructed to identify predictors of overall survival (OS) and progression-free survival (PFS). Across all three models, the following features emerged as significant predictors of OS: age, ciliary body infiltration, mitotic rate per 1 mm2, BAP1 status, and nucleoli size. For PFS, the most significant predictors included tumor-infiltrating lymphocyte and macrophage densities, largest basal diameter, nucleoli size, and BAP1 status. However, a potential limitation of this study was the inclusion of only patients who underwent ocular enucleation. This may introduce selection and outcome biases, as current UM management more commonly involves local therapies such as plaque brachytherapy or proton beam radiation rather than enucleation.
In contrast, Luo et al. developed a machine learning model to predict the 4-year risk of death and metastasis in patients with UM treated with iodine-125 plaque brachytherapy [24]. This model incorporated demographic data (age and sex), ocular features (laterality, corrected visual acuity, and intraocular pressure), and tumor characteristics (presence of subretinal fluid, optic nerve and ciliary body involvement, vitreous hemorrhage, tumor thickness, tumor shape, and basal diameter) derived from multimodal imaging. Notably, the model’s performance improved significantly when data from three follow-up visits were included, rather than data from a single visit. With this additional postoperative information, the model achieved an accuracy of 83% for predicting death and 79.5% for predicting metastasis. These findings underscore the importance of incorporating longitudinal follow-up data to enhance the accuracy of predictive models for UM-related outcomes.
In a recent retrospective study, Wu et al. investigated potential risk factors for metastatic disease in patients with UM using ML models [25]. The study identified higher tumor staging, specific histologic features (mixed epithelioid and spindle cells, as well as epithelioid cell melanoma), primary tumor location in the ciliary body, surgical treatment, and systemic therapy as significant risk factors for distant metastasis (p-value < 0.05). The models achieved an accuracy of 78.8%. One of the study’s key findings was the identification of the ciliary body as the tumor location associated with the highest metastatic risk, potentially due to the distinct vascular supply of the anterior chamber and iris compared to the choroid. Another critical insight from this study was the importance of early diagnosis and treatment in preventing distant metastasis. Radiation therapy, in particular, was highlighted as playing a pivotal role in reducing metastatic risk by controlling local tumor growth, inhibiting angiogenesis, inducing apoptosis, and stimulating immune system activation.
Damato et al. utilized a neural network to estimate survival probabilities following surgical treatment for UM and stratified risks based on various clinico-pathological features. The neural network generated survival curves comparable to Kaplan–Meier estimates, indicating that predicted survival closely aligned with observed outcomes [26].
Another significant challenge in UM research involves the potential malignant transformation of a choroidal nevus into melanoma. The accurate prediction of this transformation carries substantial implications for treatment strategies, ocular morbidity, prognosis, and mortality. Tailor et al. addressed this issue by developing an ML model designed to predict the malignant transformation of choroidal nevi. The model, based on tabular multimodal imaging data from a multicenter study with internal and external validation, achieved a sensitivity of 86.9% and a specificity of 98.4% [27]. Key predictive factors for malignant transformation included lesion thickness, lesion diameter, and tumor shape (particularly, dome-shaped lesions). This area remains a critical focus in ophthalmic pathology, requiring further investigation due to the current lack of definitive criteria for distinguishing between a choroidal nevus and a small UM.
The primary AI applications, along with their respective advantages and limitations, in predicting UM prognosis and metastatic risk are summarized in Table 3. AI shows promise in predicting metastatic risk and overall survival by leveraging clinical, imaging, and histological data. Models have demonstrated good accuracy and can support early intervention strategies. However, many studies are retrospective with limited diversity, potential selection bias, and insufficient external validation, reducing generalizability.

Table 3.
AI applications to predict UM prognosis and metastatic risk.
5. Conclusions
Various types of AI algorithms were evaluated in the included studies. Compared to ML, DL algorithms require larger datasets for training to achieve optimal performance. Consequently, when training datasets are limited or of suboptimal quality (e.g., lack of heterogeneity), the performance of these algorithms may be adversely affected.
In recent years, AI has increasingly been recognized as a valuable tool for the diagnosis and management of various tumors, including central nervous system tumors and head and neck cancers [28,29].
The application of AI in the histopathological and molecular diagnosis of UM, similar to its use in other healthcare domains, presents several technical, regulatory, and clinical challenges. From a technical perspective, AI models, particularly those based on DL, require extensive, well-annotated datasets, which are difficult to standardize across institutions, especially for rare diseases like UM. Additionally, DL models are often perceived as “black boxes”, making their decision-making processes difficult to interpret, which can be a concern for clinicians who require transparency to trust AI-driven diagnoses. Furthermore, the high computational requirements of complex AI models may limit their feasibility in smaller healthcare settings.
Regulatory challenges include ensuring data privacy, maintaining security, and adhering to established standards. Certifying AI models for clinical use is particularly challenging for evolving systems, and liability concerns related to potential AI misdiagnoses remain unresolved. Clinically, one of the main obstacles is gaining the trust of healthcare professionals, who may hesitate to rely on AI tools if the underlying algorithms are not easily interpretable. Moreover, AI models need to integrate seamlessly into existing clinical workflows to avoid disruptions. Ensuring that these systems generalize well and perform effectively across diverse patient populations is essential for their widespread adoption in clinical practice.
Further research is necessary to explore AI applications in UM. Longitudinal studies assessing the long-term impact of AI on patient outcomes are particularly important. It is also crucial to evaluate how AI-assisted diagnoses influence treatment decisions, disease progression, and patient outcomes. Including diverse patient populations in studies is another critical factor to ensure AI models are trained on representative datasets, thereby reducing biases and improving their generalizability. Notably, AI can facilitate collaboration among physicians from various specialties (e.g., oncologists, radiologists, pathologists, and surgeons), fostering the development of integrated and comprehensive care strategies.
Finally, the ethical and social implications of AI adoption in healthcare cannot be overlooked. Research must address concerns related to patient privacy and the potential for algorithmic bias, as well as investigate how AI integration might influence equity and access to care across different populations. It is important to note that AI algorithms, particularly those based on deep learning (DL), have been increasingly applied in recent years for the detection of uveal melanoma (UM) and the differentiation of benign tumors, such as choroidal nevi, from UM. The applications of AI in UM extend beyond histopathological diagnosis to include molecular analysis, such as gene expression profiling (GEP) and methylation patterns, which are crucial for predicting patient prognosis.
In summary, while AI holds great potential to revolutionize the diagnosis and prognosis of UM, coordinated efforts are essential to overcome current challenges related to data standardization, model interpretability, and regulatory compliance. By addressing these critical issues and emphasizing future research that incorporates population diversity and ethical considerations, we can facilitate the seamless integration of AI into clinical workflows. Ultimately, this will enhance early detection, optimize treatment strategies, and improve patient outcomes in UM management.
Author Contributions
Conceptualization, S.S., G.B. and M.M.; methodology, S.S. and G.B.; validation, S.S., G.B. and M.M.; formal analysis, S.S., G.B. and R.C.; investigation, S.S. and G.B.; resources, A.R., T.A. and A.L.; data curation, G.B., R.C. and M.M.; writing—original draft preparation, S.S. and M.M.; writing—review and editing, S.S., G.B. and M.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data presented in this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
UM: uveal melanoma; AI: artificial intelligence; ML: machine learning; DL: deep learning; CNN: convolutional neural network; WSI: whole-slide imaging; GEP: gene expression profile; AUC: area under the curve; UWF: ultra-widefield; US: ultrasound; H&E: hematoxylin and eosin; FNAB: fine-needle aspiration biopsy; CNA: copy number alteration; TCGA: The Cancer Genome Atlas; GEO: Gene Expression Omnibus; LDH: lactate dehydrogenase; AST: aspartate aminotransferase; OS: overall survival; PFS: progression-free survival.
References
- Bai, H.; Bosch, J.J.; Heindl, L.M. Current management of uveal melanoma: A review. Clin. Exp. Ophthalmol. 2023, 51, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Dadzie, A.K.; Iddir, S.P.; Abtahi, M.; Ebrahimi, B.; Le, D.; Ganesh, S.; Son, T.; Heiferman, M.J.; Yao, X. Colour fusion effect on deep learning classification of uveal melanoma. Eye 2024, 38, 2781–2787. [Google Scholar] [CrossRef]
- Chandrabhatla, A.S.; Horgan, T.M.; Cotton, C.C.; Ambati, N.K.; Shildkrot, Y.E. Clinical Applications of Machine Learning in the Management of Intraocular Cancers: A Narrative Review. Invest. Ophthalmol. Vis. Sci. 2023, 64, 29. [Google Scholar] [CrossRef]
- Sabazade, S.; Lumia Michalski, M.A.; Bartoszek, J.; Fili, M.; Holmström, M.; Stålhammar, G. Development and Validation of a Deep Learning Algorithm for Differentiation of Choroidal Nevi from Small Melanoma in Fundus Photographs. Ophthalmol. Sci. 2024, 5, 100613. [Google Scholar] [CrossRef]
- Wan, Q.; Ren, X.; Wei, R.; Yue, S.; Wang, L.; Yin, H.; Tang, J.; Zhang, M.; Ma, K.; Deng, Y.P. Deep learning classification of uveal melanoma based on histopathological images and identification of a novel indicator for prognosis of patients. Biol. Proced. Online 2023, 25, 15. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.; Zhang, K.; Hui, S.; Feng, Y.; Luo, J.; Li, Y.; Wei, W. Validation of the Relationship Between Iris Color and Uveal Melanoma Using Artificial Intelligence With Multiple Paths in a Large Chinese Population. Front. Cell Dev. Biol. 2021, 9, 713209. [Google Scholar] [CrossRef] [PubMed]
- Iddir, S.P.; Love, J.; Ma, J.S.; Bryan, J.M.; Ganesh, S.; Heiferman, M.J.; Yi, D. Predicting Malignant Transformation of Choroidal Nevi Using Machine Learning. Res. Sq. 2023; rs.3. [Google Scholar] [CrossRef]
- Kulbay, M.; Marcotte, E.; Remtulla, R.; Lau, T.H.A.; Paez-Escamilla, M.; Wu, K.Y.; Burnier, M.N. Uveal Melanoma: Comprehensive Review of Its Pathophysiology, Diagnosis, Treatment, and Future Perspectives. Biomedicines 2024, 12, 1758. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, L.; Runkel, C.B.; Künzel, S.; Kabiri, P.; Rübsam, A.; Bonaventura, T.; Marquardt, P.; Haas, V.; Biniaminov, N.; Biniaminov, S.; et al. Using Deep Learning to Distinguish Highly Malignant Uveal Melanoma from Benign Choroidal Nevi. J. Clin. Med. 2024, 13, 4141. [Google Scholar] [CrossRef]
- Murali, R.; Wiesner, T.; Scolyer, R.A. Tumours associated with BAP1 mutations. Pathology 2013, 45, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Zhou, W.; Qi, X.; Zhang, G.; Girnita, L.; Seregard, S.; Grossniklaus, H.E.; Yao, Z.; Zhou, X.; Stålhammar, G. Prediction of BAP1 Expression in Uveal Melanoma Using Densely-Connected Deep Classification Networks. Cancers 2019, 11, 1579. [Google Scholar] [CrossRef]
- Zhang, H.; Kalirai, H.; Acha-Sagredo, A.; Yang, X.; Zheng, Y.; Coupland, S.E. Piloting a Deep Learning Model for Predicting Nuclear BAP1 Immunohistochemical Expression of Uveal Melanoma from Hematoxylin-and-Eosin Sections. Transl. Vis. Sci. Technol. 2020, 9, 50. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.Y.A.; Chen, H.; Gomez, C.; Correa, Z.M.; Unberath, M. Direct Gene Expression Profile Prediction for Uveal Melanoma from Digital Cytopathology Images via Deep Learning and Salient Image Region Identification. Ophthalmol. Sci. 2022, 3, 100240. [Google Scholar] [CrossRef] [PubMed]
- Akram, F.; de Bruyn, D.P.; van den Bosch, Q.C.C.; Trandafir, T.E.; van den Bosch, T.P.P.; Verdijk, R.M.; de Klein, A.; Kiliç, E.; Stubbs, A.P.; Brosens, E.; et al. Prediction of molecular subclasses of uveal melanoma by deep learning using routine haematoxylin-eosin-stained tissue slides. Histopathology 2024, 85, 909–919. [Google Scholar] [CrossRef]
- Liu, T.Y.A.; Zhu, H.; Chen, H.; Arevalo, J.F.; Hui, F.K.; Yi, P.H.; Wei, J.; Unberath, M.; Correa, Z.M. Gene Expression Profile Prediction in Uveal Melanoma Using Deep Learning: A Pilot Study for the Development of an Alternative Survival Prediction Tool. Ophthalmol. Retina. 2020, 4, 1213–1215. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.G.; Shih, J.; Yau, C.; Gibb, E.A.; Oba, J.; Mungall, K.L.; Hess, J.M.; Uzunangelov, V.; Walter, V.; Danilova, L.; et al. Integrative Analysis Identifies Four Molecular and Clinical Subsets in Uveal Melanoma. Cancer Cell 2017, 32, 204–220.e15. [Google Scholar] [CrossRef]
- Reggiani, F.; El Rashed, Z.; Petito, M.; Pfeffer, M.; Morabito, A.; Tanda, E.T.; Spagnolo, F.; Croce, M.; Pfeffer, U.; Amaro, A. Machine Learning Methods for Gene Selection in Uveal Melanoma. Int. J. Mol. Sci. 2024, 25, 1796. [Google Scholar] [CrossRef]
- Xie, J.; Wu, Z.; Xu, X.; Liang, G.; Xu, J. Screening and identification of key genes and pathways in metastatic uveal melanoma based on gene expression using bioinformatic analysis. Medicine 2020, 99, e22974. [Google Scholar] [CrossRef]
- Hou, P.; Bao, S.; Fan, D.; Yan, C.; Su, J.; Qu, J.; Zhou, M. Machine learning-based integrative analysis of methylome and transcriptome identifies novel prognostic DNA methylation signature in uveal melanoma. Brief. Bioinform. 2021, 22, bbaa371. [Google Scholar] [CrossRef]
- Bassi, A.; Krance, S.H.; Pucchio, A.; Pur, D.R.; Miranda, R.N.; Felfeli, T. The Application of Artificial Intelligence in the Analysis of Biomarkers for Diagnosis and Management of Uveitis and Uveal Melanoma: A Systematic Review. Clin. Ophthalmol. 2022, 16, 2895–2908. [Google Scholar] [CrossRef]
- Lever, M.; Bogner, S.; Giousmas, M.; Mairinger, F.D.; Baba, H.A.; Richly, H.; Gromke, T.; Schuler, M.; Bechrakis, N.E.; Kalkavan, H. Prognostic value of clinical and radiomic parameters in patients with liver metastases from uveal melanoma. Pigment. Cell Melanoma Res. 2024, 37, 831–838. [Google Scholar] [CrossRef]
- Chen, Y.N.; Wang, Y.N.; Chen, M.X.; Zhang, K.; Chen, R.T.; Fang, R.; Wang, H.; Zhang, H.H.; Huang, Y.N.; Feng, Y.; et al. Machine learning models for outcome prediction of Chinese uveal melanoma patients: A 15-year follow-up study. Cancer Commun. 2022, 42, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Donizy, P.; Krzyzinski, M.; Markiewicz, A.; Karpinski, P.; Kotowski, K.; Kowalik, A.; Orlowska-Heitzman, J.; Romanowska-Dixon, B.; Biecek, P.; Hoang, M.P. Machine learning models demonstrate that clinicopathologic variables are comparable to gene expression prognostic signature in predicting survival in uveal melanoma. Eur. J. Cancer. 2022, 174, 251–260. [Google Scholar] [CrossRef]
- Luo, J.; Chen, Y.; Yang, Y.; Zhang, K.; Liu, Y.; Zhao, H.; Dong, L.; Xu, J.; Li, Y.; Wei, W. Prognosis Prediction of Uveal Melanoma After Plaque Brachytherapy Based on Ultrasound With Machine Learning. Front. Med. 2022, 8, 777142. [Google Scholar] [CrossRef]
- Wu, S.N.; Qin, D.Y.; Zhu, L.; Guo, S.J.; Li, X.; Huang, C.H.; Hu, J.; Liu, Z. Uveal melanoma distant metastasis prediction system: A retrospective observational study based on machine learning. Cancer Sci. 2024, 115, 3107–3126. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.; Eleuteri, A.; Fisher, A.C.; Coupland, S.E.; Taktak, A.F. Artificial neural networks estimating survival probability after treatment of choroidal melanoma. Ophthalmology 2008, 115, 1598–1607. [Google Scholar] [CrossRef] [PubMed]
- Tailor, P.D.; Kopinski, P.K.; D’Souza, H.S.; Leske, D.A.; Olsen, T.W.; Shields, C.L.; Shields, J.A.; Dalvin, L.A. Predicting Choroidal Nevus Transformation to Melanoma Using Machine Learning. Ophthalmol. Sci. 2024, 5, 100584. [Google Scholar] [CrossRef]
- Broggi, G.; Mazzucchelli, M.; Salzano, S.; Barbagallo, G.M.V.; Certo, F.; Zanelli, M.; Palicelli, A.; Zizzo, M.; Koufopoulos, N.; Magro, G.; et al. The emerging role of artificial intelligence in neuropathology: Where are we and where do we want to go? Pathol. Res. Pract. 2024, 263, 155671. [Google Scholar] [CrossRef] [PubMed]
- Broggi, G.; Maniaci, A.; Lentini, M.; Palicelli, A.; Zanelli, M.; Zizzo, M.; Koufopoulos, N.; Salzano, S.; Mazzucchelli, M.; Caltabiano, R. Artificial Intelligence in Head and Neck Cancer Diagnosis: A Comprehensive Review with Emphasis on Radiomics, Histopathological, and Molecular Applications. Cancers 2024, 16, 3623. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef]
- McLean, M.J.; Foster, W.D.; Zimmerman, L.E. Prognostic factors in small malignant melanomas of choroid and ciliary body. Arch. Ophthalmol. 1977, 95, 48–58. [Google Scholar] [CrossRef]
- Folberg, R.; Hendrix, M.J.; Maniotis, A.J. Vasculogenic mimicry and tumor angiogenesis. Am. J. Pathol. 2000, 156, 361–381. [Google Scholar] [CrossRef] [PubMed]
- Folberg, R.; Rummelt, V.; Parys-Van Ginderdeuren, R.; Hwang, T.; Woolson, R.F.; Pe’er, J.; Gruman, L.M. The prognostic value of tumor blood vessel morphology in primary uveal melanoma. Ophthalmology 1993, 100, 1389–1398. [Google Scholar] [CrossRef] [PubMed]
- Salzano, S.; Vecchio, G.M.; Failla, M.; Russo, A.; Avitabile, T.; Longo, A.; Caltabiano, R.; Broggi, G. Metastases from uveal melanoma may lack S100 expression: A clinico-pathologic and immunohistochemical study with emphasis on potential causes and diagnostic implications. Ann. Diagn. Pathol. 2025, 76, 152464. [Google Scholar] [CrossRef] [PubMed]
- Koseoglu, N.D.; Corrêa, Z.M.; Liu, T.Y.A. Artificial intelligence for ocular oncology. Curr. Opin. Ophthalmol. 2023, 34, 437–440. [Google Scholar] [CrossRef]
- Tahmasebzadeh, A.; Sadeghi, M.; Naseripour, M.; Mirshahi, R.; Ghaderi, R. Artificial intelligence and different image modalities in uveal melanoma diagnosis and prognosis: A narrative review. Photodiagnosis Photodyn. Ther. 2025, 52, 104528. [Google Scholar] [CrossRef]
- Longhitano, L.; Giallongo, S.; Orlando, L.; Broggi, G.; Longo, A.; Russo, A.; Caltabiano, R.; Giallongo, C.; Barbagallo, I.; Di Rosa, M.; et al. Lactate Rewrites the Metabolic Reprogramming of Uveal Melanoma Cells and Induces Quiescence Phenotype. Int. J. Mol. Sci. 2022, 24, 24. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).