Acute Cardiometabolic Responses to Multi-Modal Integrative Neuromuscular Training in Children
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Peak Aerobic Capacity Testing
2.3. Integrative Neuromuscular Training Protocol
2.4. Treadmill Protocol
2.5. Experimental Measurements: Oxygen Uptake and Heart Rate
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Aubert, S.; Barnes, J.; Abdeta, C.; Abi Nader, P.; Adeniyi, A.; Aguilar-Farias, N.; Andrade Tenesaca, D.; Bhawra, J.; Brazo-Sayavera, J.; Cardon, G.; et al. Global Matrix 3.0 Physical Activity Report Card Grades for Children and Youth: Results and Analysis From 49 Countries. J. Phys. Act. Health 2018, 15, S251–S273. [Google Scholar] [CrossRef] [PubMed]
- Faigenbaum, A.; Rial Rebullido, T.; MacDonald, J. The unsolved problem of paediatric physical inactivity: It is time for a new perspective. Acta Paediatr. 2018, 107, 1857–1859. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, B.; Henley, W.; Wilkin, T. Effectiveness of intervention on physical activity of children: Systematic review and meta-analysis of controlled trials with objectively measured outcomes (EarlyBird 54). BMJ 2012, 345, e5888. [Google Scholar] [CrossRef] [PubMed]
- Carlin, A.; Murphy, M.; Nevill, A.; Gallagher, A. Effects of a peer-led Walking In ScHools intervention (the WISH study) on physical activity levels of adolescent girls: a cluster randomised pilot study. Trials 2018, 19, 31. [Google Scholar] [CrossRef] [PubMed]
- Villa-González, E.; Ruiz, J.; Mendoza, J.; Chillón, P. Effects of a school-based intervention on active commuting to school and health-related fitness. BMC Public Health 2017, 17, 20. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Hermosa, A.; Martínez-Vizcaíno, V.; Alvarez-Bueno, C.; García-Prieto, J.; Pardo-Guijarro, M.; Sánchez-López, M. No association between active commuting to school, adiposity, fitness, and cognition in Spanish children: The MOVI-KIDS Study. J. Sch. Health 2018, 88, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Sandercock, G.; Cohen, D. Temporal trends in muscular fitness of English 10-year-olds 1998–2014: An allometric approach. J. Sci. Med. Sport 2019, 22, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Fraser, B.; Blizzard, L.; Tomkinson, G.; Lycett, K.; Wake, M.; Burgner, D.; Ranganathan, S.; Juonala, M.; Dwyer, T.; Venn, A.; et al. The great leap backward: changes in the jumping performance of Australian children aged 11-12-years between 1985 and 2015. J. Sports Sci. 2019, 37, 748–754. [Google Scholar] [CrossRef]
- Smith, J.; Eather, N.; Weaver, R.; Riley, N.; Beets, M.; Lubans, D. Behavioral correlates of muscular fitness in children and adolescents: A systematic review. Sports Med. 2019. epub ahead of print. [Google Scholar] [CrossRef]
- Myer, G.; Faigenbaum, A.; Ford, K.; Best, T.; Bergeron, M.; Hewett, T. When to initiate integrative neuromuscular training to reduce sports-related injuries and enhance health in youth? Curr. Sports Med. Rep. 2011, 10, 155–166. [Google Scholar] [CrossRef]
- Faigenbaum, A.; Farrell, A.; Fabiano, M.; Radler, T.; Naclerio, F.; Ratamess, N.; Kang, J.; Myer, G. Effects of integrated neuromuscular training on fitness performance in children. Pediatr. Exerc. Sci. 2011, 23, 573–584. [Google Scholar] [CrossRef]
- Duncan, M.; Eyre, E.; Oxford, S. The effects of 10 weeks Integrated Neuromuscular Training on fundamental movement skills and physical self-efficacy in 6–7 year old children. J. Strength Cond. Res. 2018, 32, 3348–3356. [Google Scholar] [CrossRef] [PubMed]
- Foss, K.; Thomas, S.; Khoury, J.; Myer, G.; Hewett, T. A school-based neuromuscular training program and sport-related injury incidence: A prospective randomized controlled clinical trial. J. Athl. Train. 2018, 53, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Faigenbaum, A.; Kang, J.; Ratamess, N.; Farrell, A.; Ellis, N.; Vought, I.; Bush, J. Acute cardiometabolic responses to medicine ball interval training in children. Int. J. Exerc. Sci. 2018, 11, 886–899. [Google Scholar] [PubMed]
- Faigenbaum, A.; Kang, J.; Ratamess, N.; Farrell, A.; Golda, S.; Stranieri, A.; Coe, J.; Bush, J. Acute cardiometabolic responses to battling rope exercise in children. J. Strength Cond. Res. 2018, 32, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Panagoulis, C.; Chatzinikolaou, A.; Avloniti, A.; Leontsini, D.; Deli, C.; Draganidis, D.; Stampoulis, T.; Oikonomou, T.; Papanikolaou, K.; Rafailakis, L.; et al. In-season integrative neuromuscular strength training improves performance of early adolescent soccer athletes. J. Strength Cond. Res. 2018. epub before print. [Google Scholar] [CrossRef]
- Tarp, J.; Child, A.; White, T.; Westgate, K.; Bugge, A.; Grøntved, A.; Wedderkopp, N.; Andersen, L.; Cardon, G.; Davey, R.; et al. Physical activity intensity, bout-duration, and cardiometabolic risk markers in children and adolescents. Int. J. Obes. (Lond.) 2018, 42, 1639–1650. [Google Scholar] [CrossRef]
- Aadland, E.; Andersen, L.; Anderssen, S.; Resaland, G.; Kvalheim, O. Associations of volumes and patterns of physical activity with metabolic health in children: A multivariate pattern analysis approach. Prev. Med. 2018, 115, 12–18. [Google Scholar] [CrossRef]
- Hamer, M.; Stamatakis, E. Relative proportion of vigorous physical activity, total volume of moderate to vigorous activity, and body mass index in youth: the Millennium Cohort Study. Int. J. Obes. (Lond.) 2018, 42, 1239–1242. [Google Scholar] [CrossRef]
- Harris, N.; Dulson, D.; Logan, G.; Warbrick, I.; Merien, F.; Lubans, D. Acute responses to resistance and high-intensity interval training in early adolescents. J. Strength Cond. Res. 2016, 31, 1177–1186. [Google Scholar] [CrossRef]
- Bond, B.; Weston, K.; Williams, C.; Barker, A. Perspectives on high-intensity interval exercise for health promotion in children and adolescents. Open Access J. Sports Med. 2017, 8, 243–265. [Google Scholar] [CrossRef] [PubMed]
- Thivel, D.; Masurier, J.; Baquet, G.; Timmons, B.; Pereira, B.; Berthoin, S.; Duclos, M.; Aucouturier, J. High-intensity interval training in overweight and obese children and adolescents: systematic review and meta-analysis. J. Sports Med. Phys. Fitness 2019, 59, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Educating the Student Body: Taking Physical Activity and Physical Education to School; The National Academies Press: Washington, DC, USA, 2013. [Google Scholar]
- Kotte, E.; DE Groot, J.; Bongers, B.; Winkler, A.; Takken, T. Validity and reproducibility of a new treadmill protocol: The fitkids treadmill test. Med. Sci. Sports Exerc. 2015, 47, 2241–2247. [Google Scholar] [CrossRef]
- Armstrong, N.; McManus, A. Aerobic Fitness. In Oxford Textbook of Children’s Sport and Exercise Medicine, 3rd ed.; Armstrong, N., van Mechelen, W., Eds.; Oxford University Press: Oxford, UK, 2017; pp. 161–180. [Google Scholar]
- Faigenbaum, A.D.; Milliken, L.A.; Cloutier, G.; Westcott, W.L. Perceived exertion during resistance exercise by children. Percept. Mot. Skills 2004, 98, 627–637. [Google Scholar] [CrossRef]
- Faigenbaum, A.; Bush, J.; McLoone, R.; Kreckel, M.; Farrell, A.; Ratamess, N.; Kang, J. Benefits of strength and skill-based training during primary school physical education. J. Strength Cond. Res. 2015, 29, 1255–1262. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. Physical Activity Guidelines for Americans; U.S. Department of Health and Human Services: Washington, DC, USA, 2018; p. 49.
- Butte, N.; Watson, K.; Ridle, Y.K.; Zakeri, I.; McMurray, R.; Pfeiffer, K.; Crouter, S.; Herrmann, S.; Bassett, D.; Long, A.; et al. A Youth Compendium of Physical Activities: Activity Codes and Metabolic Intensities. Med. Sci. Sports Exerc. 2018, 50, 246–256. [Google Scholar] [CrossRef]
- Chuensiri, N.; Tanaka, H.; Suksom, D. The acute effects of supramaximal high-intensity intermittent exercise on vascular function in lean vs. obese prepubescent boys. Pediatr. Exerc. Sci. 2015, 27, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Baquet, G.; Gamelin, F.; Aucouturier, J.; Berthoin, S. Cardiorespiratory responses to continuous and intermittent exercise in children. Int. J. Sports Med. 2017, 38, 755–762. [Google Scholar] [CrossRef]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Lippincott, Williams and Wilkins: Baltimore, MD, USA, 2018. [Google Scholar]
- Eddolls, W.; McNarry, M.; Stratton, G.; Winn, C.; Mackintosh, K. High-intensity interval training interventions in children and adolescents: A systematic review. Sports Med. 2017, 47, 2326–2374. [Google Scholar] [CrossRef]
- Birat, A.; Bourdier, P.; Piponnier, E.; Blazevich, A.; Maciejewski, H.; Duché, P.; Ratel, S. Metabolic and fatigue profiles are comparable between prepubertal children and well-trained adult endurance athletes. Front. Pediatr. 2018, 9, 387. [Google Scholar] [CrossRef]
- Ratamess, N.; Rosenberg, J.; Klei, S.; Dougherty, B.; Kang, J.; Smith, C.; Ross, R.; Faigenbaum, A. Comparison of the acute metabolic responses to traditional resistance, body-weight, and battling rope exercise. J. Strength Cond. Res. 2015, 29, 47–57. [Google Scholar] [CrossRef]
- Riddoch, C.; Mattocks, C.; Deere, K.; Saunders, J.; Kirkby, J.; Tilling, K.; Leary, S.; Blair, S.; Ness, A. Objective measurement of levels and patterns of physical activity. Arch. Dis. Childhood 2007, 92, 963–969. [Google Scholar] [CrossRef]
- Robertson, R.; Goss, F.; Aaron, D.; Nagle, E.; Gallagher, M.; Kane, I.; Tessmer, K.; Schafer, M.; Hunt, S. Concurrent muscle hurt and perceived exertion of children during resistance exercise. Med. Sci. Sports Exerc. 2009, 41, 1146–1154. [Google Scholar] [CrossRef]
- Malik, A.; Williams, C.; Bond, B.; Weston, K.; Barker, A. Acute cardiorespiratory, perceptual and enjoyment responses to high-intensity interval exercise in adolescents. Eur. J. Sport Sci. 2017, 17, 1335–1342. [Google Scholar] [CrossRef]
- Sebire, S.; Jago, R.; Fox, K.; Edwards, M.; Thompson, J. Testing a self-determination theory model of children’s physical activity motivation: a cross-sectional study. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 111. [Google Scholar] [CrossRef]
- Ratamess, N. ACSM’s Foundations of Strength Training and Conditioning; Lippincott, Williams and Wilkins: Philadelphia, PA, USA, 2012. [Google Scholar]
- Nikolaidis, P.; Kintziou, E.; Georgoudis, G.; Afonso, J.; Vancini, R.; Knechtle, B. The effect of body mass index on acute cardiometabolic responses to graded exercise testing in children: A narrative review. Sports 2018, 6, 103. [Google Scholar] [CrossRef]
- Murphy, J.; Button, D.; Chaouachi, A.; Behm, D. Prepubescent males are less susceptible to neuromuscular fatigue following resistance exercise. Eur. J. Appl. Physiol. 2014, 114, 825–835. [Google Scholar] [CrossRef]
- Singh, T.; Alexander, M.; Gauvreau, K.; Curran, T.; Rhodes, Y.; Rhodes, J. Recovery of oxygen consumption after maximal exercise in children. Med. Sci. Sports Exerc. 2011, 43, 555–559. [Google Scholar] [CrossRef]
- Hollis, J.; Williams, A.; Sutherland, R.; Campbell, E.; Nathan, N.; Wolfenden, L.; Morgan, P.; Lubans, D.; Wiggers, J. A systematic review and meta-analysis of moderate-to-vigorous physical activity levels in elementary school physical education lessons. Prev. Med. 2016, 86, 34–54. [Google Scholar] [CrossRef]
- Schlechter, C.; Rosenkranz, R.; Milliken, G.; Dzewaltowski, D. Physical activity levels during youth sport practice: Does coach training or experience have an influence? J. Sports Sci. 2017, 35, 22–28. [Google Scholar] [CrossRef]
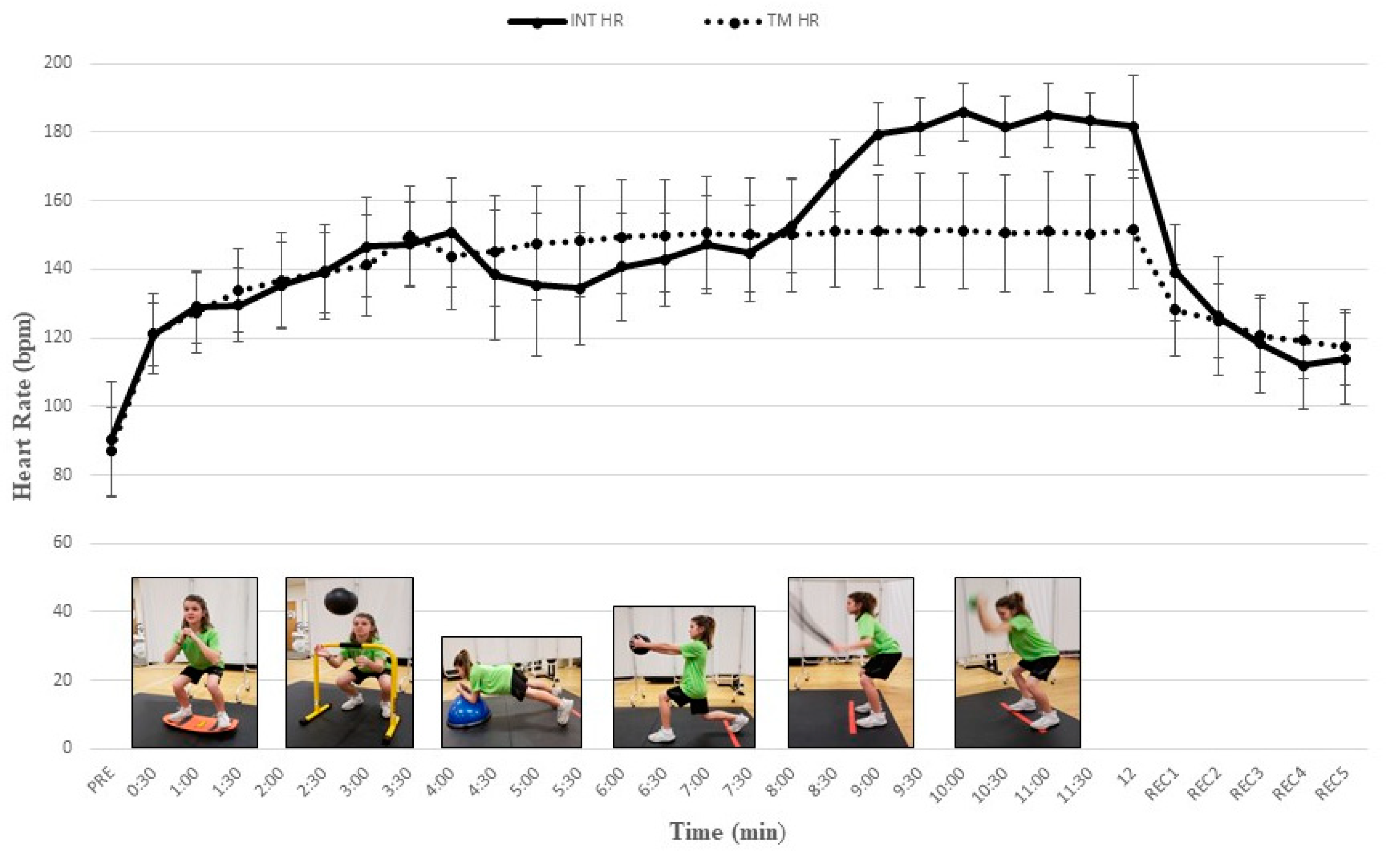
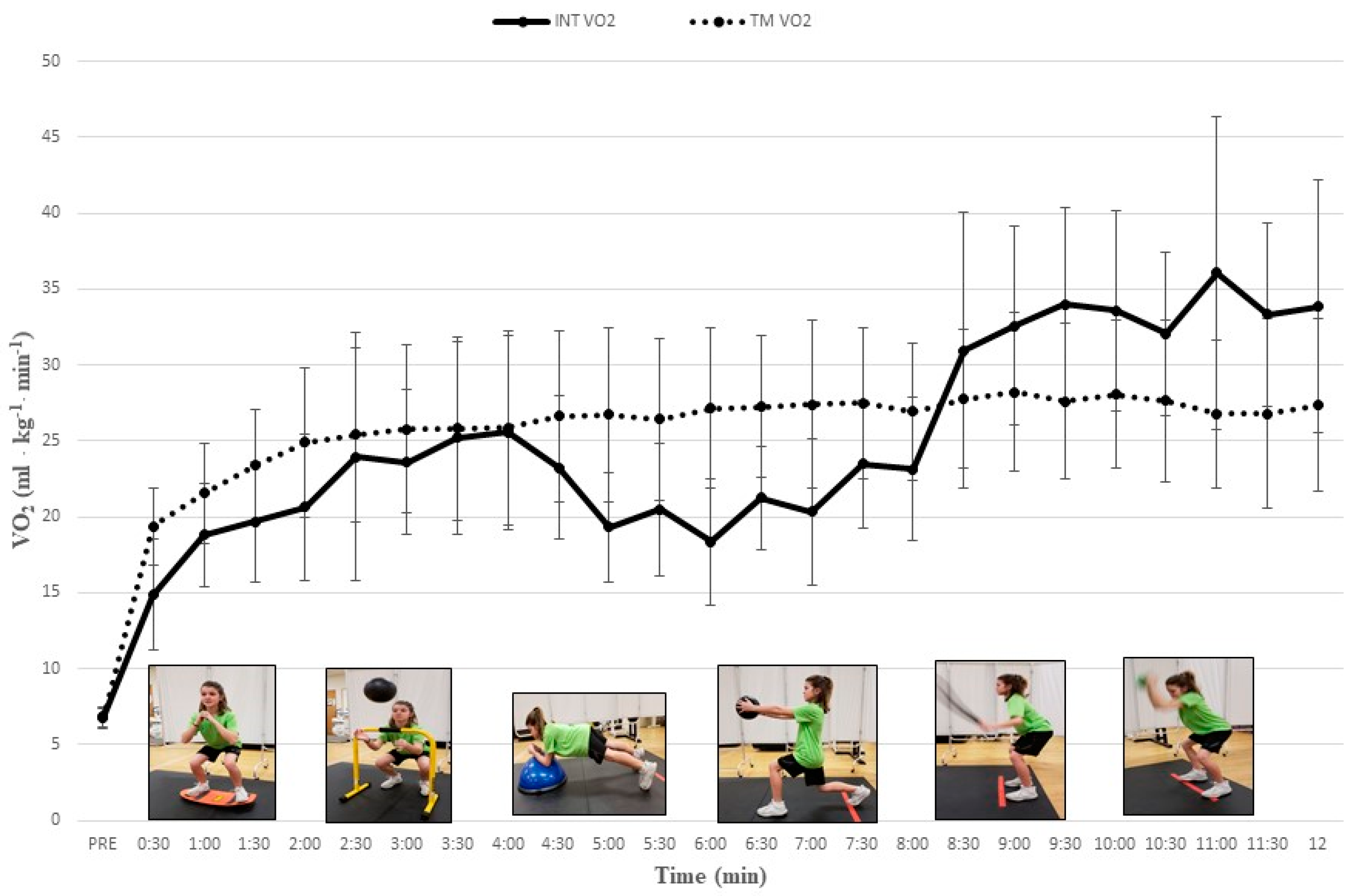
| Interval (min) | INT EX/set | VO2 | VE | HR | RER | ||||
|---|---|---|---|---|---|---|---|---|---|
| INT | TM | INT | TM | INT | TM | INT | TM | ||
| 1 (0–0.5) | 1/1 | 14.9 ± 3.6 b−l | 19.2 ± 2.5 | 14.9 ± 4.5 b−l | 16.8 ± 2.5 | 121.1 ± 9.0 b−d,g−l | 121.1 ± 11.7 | 0.88 ± 0.07 j−l | 0.82 ± 0.05 |
| 2 (1.0–1.5) | 1/2 | 19.7 ± 3.9 a,d,h−l | 23.4 ± 3.7 | 19.7 ± 6.6 a,d,e,h−l | 20.8 ± 4.5 | 129.6 ± 10.9 a,c−d,g−l | 133.8 ± 12.2 | 0.88 ± 0.08 j−l | 0.85 ± 0.06 |
| 3 (2.0–2.5) | 2/1 | 23.9 ± 8.2 a,j−l | 25.4 ± 5.7 | 24.7 ± 7.7 a,i−l | 23.8 ± 6.0 | 139.1 ± 11.6 a,b,d,i−l | 139.3 ± 13.7 | 0.90 ± 0.07 j−l | 0.89 ± 0.04 |
| 4 (3.0–3.5) | 2/2 | 25.2 ± 6.3 a,b,j−l | 25.8 ± 6.6 | 27.9 ± 9.2 a,b,j−l | 24.2 ± 6.6 | 147.3 ± 12.4 a−c,i−l | 146.0 ± 14.5 | 0.93 ± 0/07 j−l | 0.90 ± 0.05 |
| 5 (4.0–4.5) | 3/1 | 23.2 ± 4.7 a,j−l | 26.6 ± 5.6 | 26.7 ± 9.5 a,b,i−l | 25.4 ± 7.0 | 138.4 ± 19.0 i−l | 145.1 ± 16.1 | 0.96 ± 0.07 j−l | 0.90 ± 0.06 |
| 6 (5.0–5.5) | 3/2 | 20.5 ± 4.3 a,h−l | 26.4 ± 5.3 * | 23.4 ± 9.4 a,h−l | 25.0 ± 6.9 | 134.3 ± 16.5 i−l | 148.1 ± 16.2 | 0.94 ± 0.06 j−l | 0.90 ± 0.06 |
| 7 (6.0–6.5) | 4/1 | 21.2 ± 3.4 a,h−l | 27.2 ± 4.7 * | 24.3 ± 9.1 a,b,i−l | 25.8 ± 5.8 | 142.8 ± 13.6 a,b,i−l | 150.0 ± 16.4 | 0.95 ± 0.07 j−l | 0.90 ± 0.04 |
| 8 (7.0–7.5) | 4/2 | 23.4 ± 4.2 a,b,f−l | 27.5 ± 4.9 * | 26.7 ± 9.1 a,b,f,i−l | 26.6 ± 7.0 | 144.8 ± 14.0 a,b,i−l | 150.1 ± 16.7 | 0.90 ± 0.05 j−l | 0.90 ± 0.05 |
| 9 (8.0–8.5) | 5/1 | 30.9 ± 9.1 a,b,f,g | 27.8 ± 4.6 | 38.3 ± 14.2 a−c,e−j | 26.7 ± 6.0 | 167.4 ± 10.5 a−h,j−l | 151.1 ± 16.1 | 0.95 ± 0.08 j−l | 0.91 ± 0.05 |
| 10 (9.0–9.5) | 5/2 | 34.0 ± 6.4 a−h | 27.6 ± 5.1 * | 52.2 ± 12.4 a−i | 26.4 ± 5.6 * | 181.6 ± 8.3 a−i, k,l | 151.3 ± 16.7 * | 1.15 ± 0.07 a−i | 0.90 ± 0.05 * |
| 11 (10–10.5) | 6/1 | 32.0 ± 5.4 a−h | 27.6 ± 5.3 * | 46.2 ± 10.2 a−h | 26.7 ± 5.7 * | 181.5 ± 9.0 a−i | 150.5 ± 17.2 * | 1.12 ± 0.07 a−i | 0.90 ± 0.05 * |
| 12 (11–11.5) | 6/2 | 33.3 ± 6.0 a−h | 26.8 ± 6.2 * | 48.6 ± 11.0 a−h | 25.1 ± 6.5 * | 183.5 ± 7.9 a−i | 150.4 ± 17.3 * | 1.07 ± 0.06 a−i | 0.89 ± 0.05 * |
| Interval (min) | INT EX/Set | % VO2 Peak | %HR Peak | ||
|---|---|---|---|---|---|
| INT | TM | INT | TM | ||
| 1 (0–0.5) | 1/1 | 28.2 | 36.3 | 61.0 | 61.0 |
| 2 (1.0–1.5) | 1/2 | 37.2 | 44.2 | 65.3 | 67.4 |
| 3 (2.0–2.5) | 2/1 | 45.1 | 48.0 | 70.1 | 70.1 |
| 4 (3.0–3.5) | 2/2 | 47.6 | 48.8 | 74.2 | 73.5 |
| 5 (4.0–4.5) | 3/1 | 43.8 | 50.2 | 69.7 | 73.1 |
| 6 (5.0–5.5) | 3/2 | 38.7 | 49.9 | 67.6 | 74.6 |
| 7 (6.0–6.5) | 4/1 | 40.0 | 51.4 | 71.9 | 75.6 |
| 8 (7.0–7.5) | 4/2 | 44.2 | 52.0 | 72.9 | 75.6 |
| 9 (8.0–8.5) | 5/1 | 58.4 | 52.5 | 84.3 | 76.1 |
| 10 (9.0–9.5) | 5/2 | 64.3 | 52.2 | 91.5 | 76.2 |
| 11 (10–10.5) | 6/1 | 60.5 | 52.2 | 91.4 | 75.8 |
| 12 (11–11.5) | 6/2 | 62.9 | 50.7 | 92.4 | 75.8 |
| INT | TM | |
|---|---|---|
| VO2 (mL∙kg−1∙min−1) | 25.4 ± 4.5 | 26.2 ± 4.5 |
| VE (l∙min−1) | 29.79 ± 8.0 | 24.7 ± 5.5 * |
| HR (beats∙min−1) | 153.4 ± 10.6 | 145.2 ± 15.0 * |
| RER | 0.96 ± 0.04 | 0.89 ± 0.05 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faigenbaum, A.D.; Kang, J.; Ratamess, N.A.; Farrell, A.C.; Belfert, M.; Duffy, S.; Jenson, C.; Bush, J. Acute Cardiometabolic Responses to Multi-Modal Integrative Neuromuscular Training in Children. J. Funct. Morphol. Kinesiol. 2019, 4, 39. https://doi.org/10.3390/jfmk4020039
Faigenbaum AD, Kang J, Ratamess NA, Farrell AC, Belfert M, Duffy S, Jenson C, Bush J. Acute Cardiometabolic Responses to Multi-Modal Integrative Neuromuscular Training in Children. Journal of Functional Morphology and Kinesiology. 2019; 4(2):39. https://doi.org/10.3390/jfmk4020039
Chicago/Turabian StyleFaigenbaum, Avery D., Jie Kang, Nicholas A. Ratamess, Anne C. Farrell, Mina Belfert, Sean Duffy, Cara Jenson, and Jill Bush. 2019. "Acute Cardiometabolic Responses to Multi-Modal Integrative Neuromuscular Training in Children" Journal of Functional Morphology and Kinesiology 4, no. 2: 39. https://doi.org/10.3390/jfmk4020039
APA StyleFaigenbaum, A. D., Kang, J., Ratamess, N. A., Farrell, A. C., Belfert, M., Duffy, S., Jenson, C., & Bush, J. (2019). Acute Cardiometabolic Responses to Multi-Modal Integrative Neuromuscular Training in Children. Journal of Functional Morphology and Kinesiology, 4(2), 39. https://doi.org/10.3390/jfmk4020039




