The Aroma Volatile in ‘Nanguo’ Pear: A Review
Abstract
1. Introduction
2. The Methods of Extracting and Identifying Volatiles from the ‘Nanguo’ Pear
2.1. Solvent Extraction (SE)
2.2. Static Headspace Extraction (SHS)
2.3. Simultaneous Distillation Extraction (SDE)
2.4. Headspace Solid Phase Microextraction (HS-SPME)
3. Category and Composition of the Primary Aroma Components in ‘Nanguo’ Pears
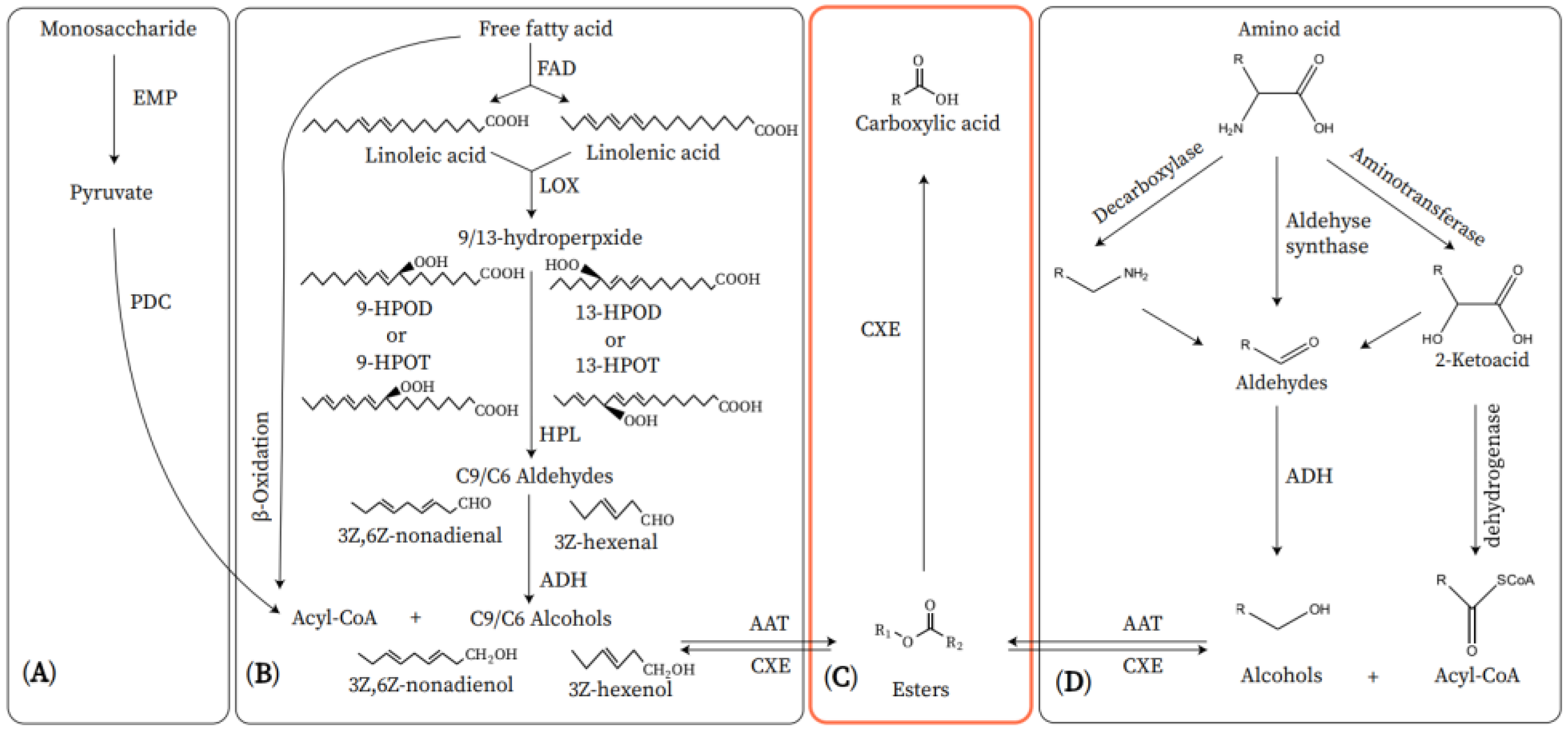
4. The Biosynthetic Pathway of VOCs in the ‘Nanguo’ Pear
4.1. Fatty Acid Pathway
4.2. Amino Acid Pathway of Ester Biosynthesis
4.3. Carbohydrate Pathway
Terpenoid Pathway
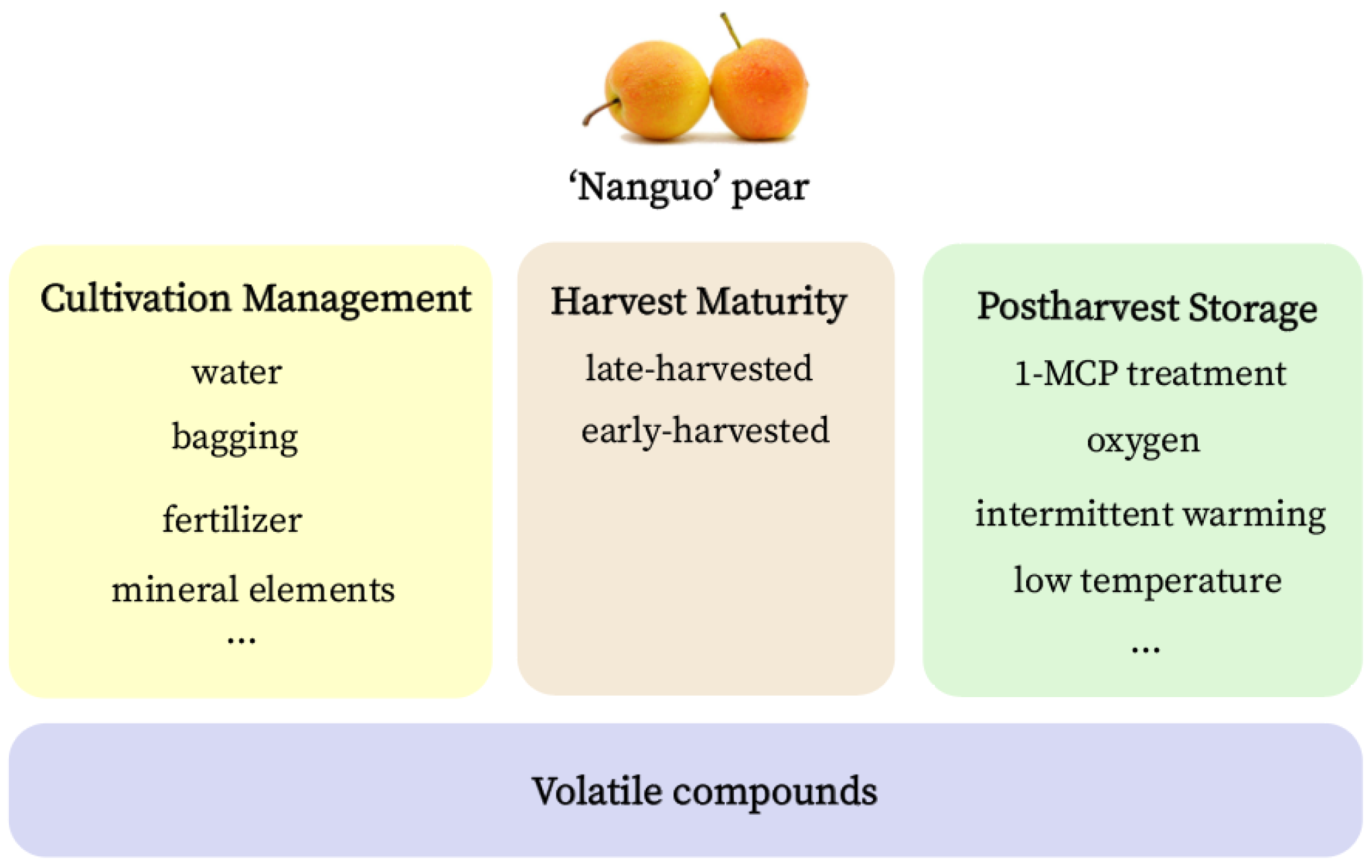
5. Factors That Influence the Production of Aroma Volatiles in the ‘Nanguo’ Pear
5.1. Conditions of Cultivation Management
5.2. Harvest Maturity
5.3. Postharvest Storage Methods and Conditions
6. Hormone Regulation of Aroma Volatiles in the ‘Nanguo’ Pear
6.1. Ethylene
6.2. Methyl Jasmonate
6.3. Salicylic Acid
6.4. Glycine Betaine
7. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of Plant-Derived Flavor Compounds. Plant J. 2008, 54, 712–732. [Google Scholar] [CrossRef] [PubMed]
- Goff, S.A.; Klee, H.J. Plant Volatile Compounds: Sensory Cues for Health and Nutritional Value? Science 2006, 311, 815–819. [Google Scholar] [CrossRef] [PubMed]
- El Hadi, M.; Zhang, F.-J.; Wu, F.-F.; Zhou, C.-H.; Tao, J. Advances in Fruit Aroma Volatile Research. Molecules 2013, 18, 8200–8229. [Google Scholar] [CrossRef] [PubMed]
- Klee, H.J.; Tieman, D.M. The Genetics of Fruit Flavour Preferences. Nat. Rev. Genet. 2018, 19, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Zhang, Y.; Li, H.; Gao, Q.; Cheng, Y.; Ogunyemi, S.O.; Guan, J. Fruit Bagging Reduces the Postharvest Decay and Alters the Diversity of Fruit Surface Fungal Community in ‘Yali’ Pear. BMC Microbiol. 2022, 22, 239. [Google Scholar] [CrossRef]
- Defilippi, B.G.; Manríquez, D.; Luengwilai, K.; González-Agüero, M. Chapter 1 Aroma Volatiles: Biosynthesis and Mechanisms of Modulation During Fruit Ripening. In Advances in Botanical Research; Academic Press: Cambridge, MA, USA, 2009; Volume 50, pp. 1–37. [Google Scholar]
- Spaho, N.; Gaši, F.; Leitner, E.; Blesić, M.; Akagić, A.; Žuljević, S.O.; Kurtović, M.; Ratković, D.Đ.; Murtić, M.S.; Akšić, M.F.; et al. Characterization of Volatile Compounds and Flavor in Spirits of Old Apple and Pear Cultivars from the Balkan Region. Foods 2021, 10, 1258. [Google Scholar] [CrossRef]
- Li, X.; Qi, L.; Zang, N.; Zhao, L.; Sun, Y.; Huang, X.; Wang, H.; Yin, Z.; Wang, A. Integrated Metabolome and Transcriptome Analysis of the Regulatory Network of Volatile Ester Formation during Fruit Ripening in Pear. Plant Physiol. Biochem. 2022, 185, 80–90. [Google Scholar] [CrossRef]
- Wang, R.; Shu, P.; Zhang, C.; Zhang, J.; Chen, Y.; Zhang, Y.; Du, K.; Xie, Y.; Li, M.; Ma, T.; et al. Integrative Analyses of Metabolome and Genome-wide Transcriptome Reveal the Regulatory Network Governing Flavor Formation in Kiwifruit (Actinidia chinensis). New Phytol. 2022, 233, 373–389. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, N.; Wang, J.; Zhang, H.; Li, D.; Shi, J.; Li, R.; Weeda, S.; Zhao, B.; Ren, S.; et al. Melatonin Promotes Ripening and Improves Quality of Tomato Fruit during Postharvest Life. J. Exp. Bot. 2015, 66, 657–668. [Google Scholar] [CrossRef]
- Cao, X.; Wei, C.; Duan, W.; Gao, Y.; Kuang, J.; Liu, M.; Chen, K.; Klee, H.; Zhang, B. Transcriptional and Epigenetic Analysis Reveals That NAC Transcription Factors Regulate Fruit Flavor Ester Biosynthesis. Plant J. 2021, 106, 785–800. [Google Scholar] [CrossRef]
- Luo, M.; Ge, W.; Sun, H.; Yang, Q.; Sun, Y.; Zhou, X.; Zhou, Q.; Ji, S. Salicylic Acid Treatment Alleviates Diminished Ester Production in Cold-Stored ‘Nanguo’ Pear by Promoting the Transcription of PuAAT. Postharvest Biol. Technol. 2022, 187, 111849. [Google Scholar] [CrossRef]
- Shi, F.; Zhou, X.; Zhou, Q.; Tan, Z.; Yao, M.; Wei, B.; Ji, S. Membrane Lipid Metabolism Changes and Aroma Ester Loss in Low-Temperature Stored Nanguo Pears. Food Chem. 2018, 245, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Wei, B.; Zhou, Q.; Tan, D.; Ji, S. 1-Methylcyclopropene Alleviates Chilling Injury by Regulating Energy Metabolism and Fatty Acid Content in ‘Nanguo’ Pears. Postharvest Biol. Technol. 2015, 109, 130–136. [Google Scholar] [CrossRef]
- Sun, Y.; Luo, M.; Ge, W.; Zhou, X.; Zhou, Q.; Wei, B.; Cheng, S.; Ji, S. Phenylpropanoid Metabolism in Relation to Peel Browning Development of Cold-Stored ‘Nanguo’ Pears. Plant Sci. 2022, 322, 111363. [Google Scholar] [CrossRef]
- Zhou, X.; Dong, L.; Zhou, Q.; Wang, J.; Chang, N.; Liu, Z.; Ji, S. Effects of Intermittent Warming on Aroma-Related Esters of 1-Methylcyclopropene-Treated ‘Nanguo’ Pears during Ripening at Room Temperature. Sci. Hortic. 2015, 185, 82–89. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, X.; Wang, J.; Ji, S. Proteomic Analysis of the Potential Mechanism of Fading of Aroma-related Esters in “Nanguo” Pears after Long-term Refrigeration. J. Food Biochem. 2019, 43, e12771. [Google Scholar] [CrossRef] [PubMed]
- Li, G. Characterization of Aromatic Volatile Constituents in 11 Asian Pear Cultivars Belonging to Different Species. Afr. J. Agric. Res. 2012, 7, 4761–4770. [Google Scholar] [CrossRef]
- Qin, G.; Tao, S.; Cao, Y.; Wu, J.; Zhang, H.; Huang, W.; Zhang, S. Evaluation of the Volatile Profile of 33 Pyrus Ussuriensis Cultivars by HS-SPME with GC–MS. Food Chem. 2012, 134, 2367–2382. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zhang, P.; Fang, Z. Modern Technologies for Extraction of Aroma Compounds from Fruit Peels: A Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 1284–1307. [Google Scholar] [CrossRef]
- Solissolis, H.; Calderonsantoyo, M.; Schorrgalindo, S.; Lunasolano, G.; Ragazzosanchez, J. Characterization of Aroma Potential of Apricot Varieties Using Different Extraction Techniques. Food Chem. 2007, 105, 829–837. [Google Scholar] [CrossRef]
- Silvestre, C.I.C.; Santos, J.L.M.; Lima, J.L.F.C.; Zagatto, E.A.G. Liquid–Liquid Extraction in Flow Analysis: A Critical Review. Anal. Chim. Acta 2009, 652, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Li, X.; Huang, S.; Li, Z.; Zhang, J.; Hu, Z. Effect of Different Extraction Methods on the Datect of Aroma. Chem. Reag. 2018, 40, 784–788. [Google Scholar] [CrossRef]
- Ortega-Heras, M.; González-SanJosé, M.L.; Beltrán, S. Aroma Composition of Wine Studied by Different Extraction Methods. Anal. Chim. Acta 2002, 458, 85–93. [Google Scholar] [CrossRef]
- Zhu, M.; Li, E.; He, H. Determination of Volatile Chemical Constitutes in Tea by Simultaneous Distillation Extraction, Vacuum Hydrodistillation and Thermal Desorption. Chromatographia 2008, 68, 603–610. [Google Scholar] [CrossRef]
- Ibáñez, E.; López-Sebastián, S.; Ramos, E.; Tabera, J.; Reglero, G. Analysis of Volatile Fruit Components by Headspace Solid-Phase Microextraction. Food Chem. 1998, 63, 281–286. [Google Scholar] [CrossRef]
- Marín-San Román, S.; Rubio-Bretón, P.; Pérez-Álvarez, E.P.; Garde-Cerdán, T. Advancement in Analytical Techniques for the Extraction of Grape and Wine Volatile Compounds. Food Res. Int. 2020, 137, 109712. [Google Scholar] [CrossRef]
- Yi, X.-K.; Liu, G.-F.; Rana, M.M.; Zhu, L.-W.; Jiang, S.-L.; Huang, Y.-F.; Lu, W.-M.; Wei, S. Volatile Profiling of Two Pear Genotypes with Different Potential for White Pear Aroma Improvement. Sci. Hortic. 2016, 209, 221–228. [Google Scholar] [CrossRef]
- Luo, M.; Zhou, X.; Hao, Y.; Sun, H.; Zhou, Q.; Sun, Y.; Ji, S. Methyl Jasmonate Pretreatment Improves Aroma Quality of Cold-Stored ‘Nanguo’ Pears by Promoting Ester Biosynthesis. Food Chem. 2021, 338, 127846. [Google Scholar] [CrossRef]
- Yeh, C.-H.; Chou, C.-Y.; Wu, C.-S.; Chu, L.-P.; Huang, W.-J.; Chen, H.-C. Effects of Different Extraction Methods on Vanilla Aroma. Molecules 2022, 27, 4593. [Google Scholar] [CrossRef]
- Sostaric, T.; Boyce, M.C.; Spickett, E.E. Analysis of the Volatile Components in Vanilla Extracts and Flavorings by Solid-Phase Microextraction and Gas Chromatography. Available online: https://pubs.acs.org/doi/pdf/10.1021/jf000515%2B (accessed on 1 November 2022).
- Ji, S.-J.; Zhang, L.; Bu, Q.; Shen, Y.; Liu, X. Changes in Aromatic Components of Refrigerated Nanguo Pears during Shelf Period Analyzed by Electronic Nose Technique. Food Sci. 2012, 33, 123–126. [Google Scholar]
- Karakaya, D.; Ulucan, O.; Turkan, M. Electronic Nose and Its Applications: A Survey. Int. J. Autom. Comput. 2020, 17, 179–209. [Google Scholar] [CrossRef]
- Noguerol-Pato, R.; González-Álvarez, M.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gándara, J. Aroma Profile of Garnacha Tintorera-Based Sweet Wines by Chromatographic and Sensorial Analyses. Food Chem. 2012, 134, 2313–2325. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ai, L.; Mu, Z.; Liu, H.; Yan, X.; Ni, L.; Zhang, H.; Xia, Y. Flavor Compounds with High Odor Activity Values (OAV > 1) Dominate the Aroma of Aged Chinese Rice Wine (Huangjiu) by Molecular Association. Food Chem. 2022, 383, 132370. [Google Scholar] [CrossRef] [PubMed]
- Takeoka, G.R.; Buttery, R.G.; Flath, R.A. Volatile Constituents of Asian Pear (Pyrus serotina). J. Agric. Food Chem. 1992, 40, 1925–1929. [Google Scholar] [CrossRef]
- Lara, I.; Miró, R.M.; Fuentes, T.; Sayez, G.; Graell, J.; López, M.L. Biosynthesis of Volatile Aroma Compounds in Pear Fruit Stored under Long-Term Controlled-Atmosphere Conditions. Postharvest Biol. Technol. 2003, 29, 29–39. [Google Scholar] [CrossRef]
- Zhou, X.; Dong, L.; Li, R.; Zhou, Q.; Wang, J.; Ji, S. Low Temperature Conditioning Prevents Loss of Aroma-Related Esters from ‘Nanguo’ Pears during Ripening at Room Temperature. Postharvest Biol. Technol. 2015, 100, 23–32. [Google Scholar] [CrossRef]
- Bult, J.H.F. Sensory Evaluation of Character Impact Components in an Apple Model Mixture. Chem. Senses 2002, 27, 485–494. [Google Scholar] [CrossRef]
- Matsui, K. Green Leaf Volatiles: Hydroperoxide Lyase Pathway of Oxylipin Metabolism. Curr. Opin. Plant Biol. 2006, 9, 274–280. [Google Scholar] [CrossRef]
- Hjelmeland, A.K.; Ebeler, S.E. Glycosidically Bound Volatile Aroma Compounds in Grapes and Wine: A Review. Am. J. Enol. Vitic. 2015, 66, 1–11. [Google Scholar] [CrossRef]
- Liang, Z.; Fang, Z.; Pai, A.; Luo, J.; Gan, R.; Gao, Y.; Lu, J.; Zhang, P. Glycosidically Bound Aroma Precursors in Fruits: A Comprehensive Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 215–243. [Google Scholar] [CrossRef]
- Agrawal, R.; Verma, A.K.; Satlewal, A. Application of Nanoparticle-Immobilized Thermostable β-Glucosidase for Improving the Sugarcane Juice Properties. Innov. Food Sci. Emerg. Technol. 2016, 33, 472–482. [Google Scholar] [CrossRef]
- Wen, Y.-Q.; He, F.; Zhu, B.-Q.; Lan, Y.-B.; Pan, Q.-H.; Li, C.-Y.; Reeves, M.J.; Wang, J. Free and Glycosidically Bound Aroma Compounds in Cherry (Prunus avium L.). Food Chem. 2014, 152, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Li, J.M.; Huang, X.S.; Li, L.T.; Zheng, D.M.; Xue, C.; Zhang, S.L.; Wu, J. Proteome Analysis of Pear Reveals Key Genes Associated with Fruit Development and Quality. Planta 2015, 241, 1363–1379. [Google Scholar] [CrossRef] [PubMed]
- De Pooter, H.L.; Montens, J.P.; Willaert, G.A.; Dirinck, P.J.; Schamp, N.M. Treatment of Golden Delicious Apples with Aldehydes and Carboxylic Acids: Effect on the Headspace Composition. J. Agric. Food Chem. 1983, 31, 813–818. [Google Scholar] [CrossRef]
- Bartley, I.M.; Stoker, P.G.; Martin, A.D.E.; Hatfield, S.G.S.; Knee, M. Synthesis of Aroma Compounds by Apples Supplied with Alcohols and Methyl Esters of Fatty Acids. J. Sci. Food Agric. 1985, 36, 567–574. [Google Scholar] [CrossRef]
- Gonçalves, B.; Oliveira, I.; Bacelar, E.; Morais, M.C.; Aires, A.; Cosme, F.; Ventura-Cardoso, J.; Anjos, R.; Pinto, T. Aromas and Flavours of Fruits. In Generation of Aromas and Flavours; Vilela, A., Ed.; InTech: Cambridge, MA, USA, 2018; ISBN 978-1-78984-452-8. [Google Scholar]
- Graham, I.A.; Eastmond, P.J. Pathways of Straight and Branched Chain Fatty Acid Catabolism in Higher Plants. Prog. Lipid Res. 2002, 41, 156–181. [Google Scholar] [CrossRef] [PubMed]
- Noordermeer, M.A.; van Dijken, A.J.H.; Smeekens, S.C.M.; Veldink, G.A.; Vliegenthart, J.F.G. Characterization of Three Cloned and Expressed 13-Hydroperoxide Lyase Isoenzymes from Alfalfa with Unusual N-Terminal Sequences and Different Enzyme Kinetics. Eur. J. Biochem. 2000, 267, 2473–2482. [Google Scholar] [CrossRef]
- Song, M.S.; Kim, D.G.; Lee, S.H. Isolation and Characterization of a Jasmonic Acid Carboxyl Methyltransferase Gene from Hot Pepper(Capsicum annuum L.). J. Plant Biol. 2005, 48, 292–297. [Google Scholar] [CrossRef]
- Mostafa, S.; Wang, Y.; Zeng, W.; Jin, B. Floral Scents and Fruit Aromas: Functions, Compositions, Biosynthesis, and Regulation. Front. Plant Sci. 2022, 13, 860157. [Google Scholar] [CrossRef]
- Rowan, D.D.; Lane, H.P.; Allen, J.M.; Fielder, S.; Hunt, M.B. Biosynthesis of 2-Methylbutyl, 2-Methyl-2-Butenyl, and 2-Methylbutanoate Esters in Red Delicious and Granny Smith Apples Using Deuterium-Labeled Substrates. J. Agric. Food Chem. 1996, 44, 3276–3285. [Google Scholar] [CrossRef]
- Pérez, A.G.; Olías, R.; Luaces, P.; Sanz, C. Biosynthesis of Strawberry Aroma Compounds through Amino Acid Metabolism. J. Agric. Food Chem. 2002, 50, 4037–4042. [Google Scholar] [CrossRef] [PubMed]
- Gonda, I.; Bar, E.; Portnoy, V.; Lev, S.; Burger, J.; Schaffer, A.A.; Tadmor, Y.; Gepstein, S.; Giovannoni, J.J.; Katzir, N.; et al. Branched-Chain and Aromatic Amino Acid Catabolism into Aroma Volatiles in Cucumis melo L. Fruit. J. Exp. Bot. 2010, 61, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Muhlemann, J.K.; Klempien, A.; Dudareva, N. Floral Volatiles: From Biosynthesis to Function: Floral Volatiles. Plant Cell Environ. 2014, 37, 1936–1949. [Google Scholar] [CrossRef]
- Ramya, M.; An, H.R.; Baek, Y.S.; Reddy, K.E.; Park, P.H. Orchid Floral Volatiles: Biosynthesis Genes and Transcriptional Regulations. Sci. Hortic. 2018, 235, 62–69. [Google Scholar] [CrossRef]
- Rinaldi, M.A.; Ferraz, C.A.; Scrutton, N.S. Alternative Metabolic Pathways and Strategies to High-Titre Terpenoid Production in Escherichia Coli. Nat. Prod. Rep. 2022, 39, 90–118. [Google Scholar] [CrossRef]
- Martínez-Rivas, F.J.; Blanco-Portales, R.; Moyano, E.; Alseekh, S.; Caballero, J.L.; Schwab, W.; Fernie, A.R.; Muñoz-Blanco, J.; Molina-Hidalgo, F.J. Strawberry Fruit FanCXE1 Carboxylesterase Is Involved in the Catabolism of Volatile Esters during the Ripening Process. Hortic. Res. 2022, 9, uhac095. [Google Scholar] [CrossRef]
- Goulet, C.; Mageroy, M.H.; Lam, N.B.; Floystad, A.; Tieman, D.M.; Klee, H.J. Role of an Esterase in Flavor Volatile Variation within the Tomato Clade. Proc. Natl. Acad. Sci. USA 2012, 109, 19009–19014. [Google Scholar] [CrossRef]
- Cao, X.; Xie, K.; Duan, W.; Zhu, Y.; Liu, M.; Chen, K.; Klee, H.; Zhang, B. Peach Carboxylesterase PpCXE1 Is Associated with Catabolism of Volatile Esters. J. Agric. Food Chem. 2019, 67, 5189–5196. [Google Scholar] [CrossRef]
- Alem, H.; Rigou, P.; Schneider, R.; Ojeda, H.; Torregrosa, L. Impact of Agronomic Practices on Grape Aroma Composition: A Review. J. Sci. Food Agric. 2019, 99, 975–985. [Google Scholar] [CrossRef]
- Wei, S.; Wang, S.; Zhang, Y.; Ran, K.; Wang, H. Effects of Different Soil Management Methods on the Soil Nutrients, enzyme Activity and Fruit Quality of Pear Orchards. Acta Prataculturae Sin. 2015, 24, 46–55. [Google Scholar] [CrossRef]
- Cvelbar Weber, N.; Koron, D.; Jakopič, J.; Veberič, R.; Hudina, M.; Baša Česnik, H. Influence of Nitrogen, Calcium and Nano-Fertilizer on Strawberry (Fragaria × ananassa Duch.) Fruit Inner and Outer Quality. Agronomy 2021, 11, 997. [Google Scholar] [CrossRef]
- Ojeda-Real, L.A.; Lobit, P.; Cárdenas-Navarro, R.; Grageda-Cabrera, O.; Farías-Rodríguez, R.; Valencia-Cantero, E.; Macías-Rodríguez, L. Effect of Nitrogen Fertilization on Quality Markers of Strawberry (Fragaria × ananassa Duch. Cv. Aromas). J. Sci. Food Agric. 2009, 89, 935–939. [Google Scholar] [CrossRef]
- Adomako, M.O.; Roiloa, S.; Yu, F.-H. Potential Roles of Soil Microorganisms in Regulating the Effect of Soil Nutrient Heterogeneity on Plant Performance. Microorganisms 2022, 10, 2399. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, X.; Fu, H.; Zhao, M.; Chen, W. Effects of Microbial Fertilizer on Apple Fruit Quality. IOP Conf. Ser. Earth Environ. Sci. 2020, 446, 032102. [Google Scholar] [CrossRef]
- Des Gachons, C.P.; Leeuwen, C.V.; Tominaga, T.; Soyer, J.-P.; Gaudillère, J.-P.; Dubourdieu, D. Influence of Water and Nitrogen Deficit on Fruit Ripening and Aroma Potential of Vitis vinifera L Cv Sauvignon Blanc in Field Conditions: Influence of Water and Nitrogen Deficit on Grape Aroma Potential. J. Sci. Food Agric. 2005, 85, 73–85. [Google Scholar] [CrossRef]
- Peng, T.; Liu, G.; Bu, Q.; Wang, X.; Zhan, D. Effect of Bagging on Quality and Aroma of Cold-Stored Nanguo Pears During Shelf Life. Hortic. Seed 2022, 7, 13–16. [Google Scholar] [CrossRef]
- Kader, A.A. A Perspective on Postharvest Horticulture (1978–2003). HortScience 2003, 38, 1004–1008. [Google Scholar] [CrossRef]
- Lester, G. Consumer Preference Quality Attributes of Melon Fruits. Acta Hortic. 2006, 712, 175–182. [Google Scholar] [CrossRef]
- Yao, M.; Zhou, X.; Zhou, Q.; Shi, F.; Wei, B.; Cheng, S.; Tan, Z.; Ji, S. Low Temperature Conditioning Alleviates Loss of Aroma-Related Esters of ‘Nanguo’ Pears by Regulation of Ethylene Signal Transduction. Food Chem. 2018, 264, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Zhou, X.; Ji, Y.; Luo, M.; Sun, Y.; Zhou, Q.; Ji, S. Potential of Ethylene in Alleviating Cold-Induced Volatile Esters Loss of ‘Nanguo’ Pears by Regulating the Lipoxygenase Pathway. Environ. Exp. Bot. 2022, 194, 104723. [Google Scholar] [CrossRef]
- Zlatić, E.; Zadnik, V.; Fellman, J.; Demšar, L.; Hribar, J.; Čejić, Ž.; Vidrih, R. Comparative Analysis of Aroma Compounds in ‘Bartlett’ Pear in Relation to Harvest Date, Storage Conditions, and Shelf-Life. Postharvest Biol. Technol. 2016, 117, 71–80. [Google Scholar] [CrossRef]
- Watkins, C.B. The Use of 1-Methylcyclopropene (1-MCP) on Fruits and Vegetables. Biotechnol. Adv. 2006, 24, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Song, J.; Du, L.; Forney, C.; Campbell-Palmer, L.; Fillmore, S.; Wismer, P.; Zhang, Z. Ethylene and 1-MCP Regulate Major Volatile Biosynthetic Pathways in Apple Fruit. Food Chem. 2016, 194, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Zhang, L.; Lv, J.; Zhang, Y.; Sun, M.; Chen, J.; Ge, Y. Effects of 1-Methylcyclopropene (1-MCP) Treatment on Ethanol Fermentation of Nanguo Pear Fruit during Ripening. J. Food Biochem. 2022, 46, e14035. [Google Scholar] [CrossRef]
- Wolters, H.; Jürgens, G. Survival of the Flexible: Hormonal Growth Control and Adaptation in Plant Development. Nat. Rev. Genet. 2009, 10, 305–317. [Google Scholar] [CrossRef]
- Oetiker, J.H.; Yang, S.F. The Role of Ethylene in Fruit Ripening. Acta Hortic. 1995, 398, 167–178. [Google Scholar] [CrossRef]
- Paul, V.; Pandey, R.; Srivastava, G.C. The Fading Distinctions between Classical Patterns of Ripening in Climacteric and Non-Climacteric Fruit and the Ubiquity of Ethylene—An Overview. J. Food Sci. Technol. 2012, 49, 1118–1126. [Google Scholar] [CrossRef]
- Li, X.; Gao, S.; Yang, L.; Yin, M.; Li, J.; Zhang, H.; Ji, S. Ethylene Promotes Ester Biosynthesis through PuERF13/PuDof2.5 Synergically Activated PuAAT1 during Ripening of Cold-Stored ‘Nanguo’ Pear. Postharvest Biol. Technol. 2023, 195, 112108. [Google Scholar] [CrossRef]
- Günther, C.S.; Marsh, K.B.; Winz, R.A.; Harker, R.F.; Wohlers, M.W.; White, A.; Goddard, M.R. The Impact of Cold Storage and Ethylene on Volatile Ester Production and Aroma Perception in ‘Hort16A’ Kiwifruit. Food Chem. 2015, 169, 5–12. [Google Scholar] [CrossRef]
- Cai, H.; An, X.; Han, S.; Jiang, L.; Yu, M.; Ma, R.; Yu, Z. Effect of 1-MCP on the Production of Volatiles and Biosynthesis-Related Gene Expression in Peach Fruit during Cold Storage. Postharvest Biol. Technol. 2018, 141, 50–57. [Google Scholar] [CrossRef]
- Larsen, P.B. Mechanisms of Ethylene Biosynthesis and Response in Plants. Essays Biochem. 2015, 58, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Kieber, J.J.; Rothenberg, M.; Roman, G.; Feldmann, K.A.; Ecker, J.R. CTR1, a Negative Regulator of the Ethylene Response Pathway in Arabidopsis, Encodes a Member of the Raf Family of Protein Kinases. Cell 1993, 72, 427–441. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-F.; Etheridge, N.; Schaller, G.E. Ethylene Signal Transduction. Ann. Bot. 2005, 95, 901–915. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, S.; Schmidt, R.J. Diversity and Similarity among Recognition Sequences of Dof Transcription Factors. Plant J. 1999, 17, 209–214. [Google Scholar] [CrossRef]
- Defilippi, B.G.; Kader, A.A.; Dandekar, A.M. Apple Aroma: Alcohol Acyltransferase, a Rate Limiting Step for Ester Biosynthesis, Is Regulated by Ethylene. Plant Sci. 2005, 168, 1199–1210. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, C.; Miao, Y.; Deng, L.; Zhang, B.; Meng, J.; Wang, Y.; Pan, L.; Niu, L.; Liu, H.; et al. Interaction between PpERF5 and PpERF7 Enhances Peach Fruit Aroma by Upregulating PpLOX4 Expression. Plant Physiol. Biochem. 2022, 185, 378–389. [Google Scholar] [CrossRef]
- Fonseca, S.; Hackler, L., Jr.; Zvara, Á.; Ferreira, S.; Baldé, A.; Dudits, D.; Pais, M.S.; Puskás, L.G. Monitoring Gene Expression along Pear Fruit Development, Ripening and Senescence Using CDNA Microarrays. Plant Sci. 2004, 167, 457–469. [Google Scholar] [CrossRef]
- Ho, T.-T.; Murthy, H.N.; Park, S.-Y. Methyl Jasmonate Induced Oxidative Stress and Accumulation of Secondary Metabolites in Plant Cell and Organ Cultures. Int. J. Mol. Sci. 2020, 21, 716. [Google Scholar] [CrossRef]
- Murthy, H.N.; Lee, E.-J.; Paek, K.-Y. Production of Secondary Metabolites from Cell and Organ Cultures: Strategies and Approaches for Biomass Improvement and Metabolite Accumulation. Plant Cell Tissue Organ Cult. PCTOC 2014, 118, 1–16. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Pérez-Álvarez, E.P.; Rubio-Bretón, P.; Garde-Cerdán, T. Changes on Grape Volatile Composition through Elicitation with Methyl Jasmonate, Chitosan, and a Yeast Extract in Tempranillo (Vitis vinifera L.) Grapevines. Sci. Hortic. 2019, 244, 257–262. [Google Scholar] [CrossRef]
- Mwenda, C.M.; Matsui, K. The Importance of Lipoxygenase Control in the Production of Green Leaf Volatiles by Lipase-Dependent and Independent Pathways. Plant Biotechnol. 2014, 31, 445–452. [Google Scholar] [CrossRef]
- Farmaki, T.; Sanmartin, M.; Jimenez, P.; Paneque, M.; Sanz, C.; Vancanneyt, G.; Leon, J.; Sanchez-Serrano, J.J. Differential Distribution of the Lipoxygenase Pathway Enzymes within Potato Chloroplasts. J. Exp. Bot. 2006, 58, 555–568. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Wang, A. Recent Advances in Phytohormone Regulation of Apple-Fruit Ripening. Plants 2021, 10, 2061. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Ji, S.; Cheng, S.; Zhou, Q.; Zhou, X.; Luo, M.; Ma, M.; Hu, M.-S.; Wei, B. Methyl Jasmonate Alleviates the Reduced Release of Aroma-Related Esters in ‘Nanguo’ Pears by Regulating Ethylene Biosynthesis and Signal Transduction. Int. J. Food Sci. Technol. 2021, 56, 814–824. [Google Scholar] [CrossRef]
- Cai, H.; Han, S.; Yu, M.; Ma, R.; Yu, Z. The Alleviation of Methyl Jasmonate on Loss of Aroma Lactones Correlated with Ethylene Biosynthesis in Peaches. J. Food Sci. 2020, 85, 2389–2397. [Google Scholar] [CrossRef]
- Kim, D.S.; Hwang, B.K. An Important Role of the Pepper Phenylalanine Ammonia-Lyase Gene (PAL1) in Salicylic Acid-Dependent Signalling of the Defence Response to Microbial Pathogens. J. Exp. Bot. 2014, 65, 2295–2306. [Google Scholar] [CrossRef]
- White, R.F. Acetylsalicylic Acid (Aspirin) Induces Resistance to Tobacco Mosaic Virus in Tobacco. Virology 1979, 99, 410–412. [Google Scholar] [CrossRef]
- Baswal, A.K.; Dhaliwal, H.S.; Singh, Z.; Mahajan, B.V.C.; Gill, K.S. Postharvest Application of Methyl Jasmonate, 1-Methylcyclopropene and Salicylic Acid Extends the Cold Storage Life and Maintain the Quality of ‘Kinnow’ Mandarin (Citrus nobilis Lour × C. deliciosa Tenora) Fruit. Postharvest Biol. Technol. 2020, 161, 111064. [Google Scholar] [CrossRef]
- Wang, L.; Baldwin, E.A.; Plotto, A.; Luo, W.; Raithore, S.; Yu, Z.; Bai, J. Effect of Methyl Salicylate and Methyl Jasmonate Pre-Treatment on the Volatile Profile in Tomato Fruit Subjected to Chilling Temperature. Postharvest Biol. Technol. 2015, 108, 28–38. [Google Scholar] [CrossRef]
- Mansour, M.M.F. Protection of Plasma Membrane of Onion Epidermal Cells by Glycinebetaine and Proline against NaCl Stress. Plant Physiol. Biochem. 1998, 36, 767–772. [Google Scholar] [CrossRef]
- Gonçalves, B.; Morais, M.C.; Sequeira, A.; Ribeiro, C.; Guedes, F.; Silva, A.P.; Aires, A. Quality Preservation of Sweet Cherry Cv. “staccato” by Using Glycine-Betaine or Ascophyllum Nodosum. Food Chem. 2020, 322, 126713. [Google Scholar] [CrossRef]
- Luo, M.; Sun, H.; Ge, W.; Sun, Y.; Zhou, X.; Zhou, Q.; Ji, S. Effect of Glycine Betaine Treatment on Aroma Production of ‘Nanguo’ Pears After Long-Term Cold Storage–Possible Involvement of Ethylene Synthesis and Signal Transduction Pathways. Food Bioprocess Technol. 2022, 15, 1327–1342. [Google Scholar] [CrossRef]
- Luo, M.; Zhou, X.; Sun, H.; Zhou, Q.; Wei, B.; Cheng, S.; Ji, S. Glycine Betaine Treatment Alleviates Loss of Aroma-Related Esters in Cold-Stored ‘Nanguo’ Pears by Regulating the Lipoxygenase Pathway. Food Chem. 2020, 316, 126335. [Google Scholar] [CrossRef] [PubMed]
- Aghdam, M.S.; Jannatizadeh, A.; Nojadeh, M.S.; Ebrahimzadeh, A. Exogenous Melatonin Ameliorates Chilling Injury in Cut Anthurium Flowers during Low Temperature Storage. Postharvest Biol. Technol. 2019, 148, 184–191. [Google Scholar] [CrossRef]
- Aghdam, M.S.; Fard, J.R. Melatonin Treatment Attenuates Postharvest Decay and Maintains Nutritional Quality of Strawberry Fruits (Fragaria × anannasa Cv. Selva) by Enhancing GABA Shunt Activity. Food Chem. 2017, 221, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Luo, M.; Zhou, X.; Zhou, Q.; Ji, S. Influence of Melatonin Treatment on Peel Browning of Cold-Stored “Nanguo” Pears. Food Bioprocess Technol. 2020, 13, 1478–1490. [Google Scholar] [CrossRef]
- Wei, S.; Jiao, H.; Wang, H.; Ran, K.; Dong, R.; Dong, X.; Yan, W.; Wang, S. The Mechanism Analysis of Exogenous Melatonin in Limiting Pear Fruit Aroma Decrease under Low Temperature Storage. PeerJ 2022, 10, e14166. [Google Scholar] [CrossRef]
- Cliff, M.; Stanich, K.; Trujillo, J.M.; Toivonen, P. Determination and Prediction of Odor Thresholds for Odor Active Volatiles in A Neutral Apple Juice Matrix. J. Fruit Qual. 2011, 34, 151–228. [Google Scholar] [CrossRef]
- Yan, J.; Ban, Z.; Lu, H.; Li, D.; Poverenov, E.; Luo, Z.; Li, L. The Aroma Volatile Repertoire in Strawberry Fruit: A Review: The Aroma Volatile Repertoire in Strawberry Fruit. J. Sci. Food Agric. 2018, 98, 4395–4402. [Google Scholar] [CrossRef]


| No. | Compounds | 0 DAH (µg kg−1) | 5 DAH (µg kg−1) | 10 DAH (µg kg−1) | 15 DAH (µg kg−1) |
|---|---|---|---|---|---|
| Esters | |||||
| 1 | Ethyl Acetate | — | — | — | 180.82 ± 2.40 |
| 2 | Ethyl Butanoate | — | — | 352.58 ± 3.40 | 550.58 ± 5.40 |
| 3 | Butyl Acetate | — | — | 38.58 ± 0.96 | — |
| 4 | Ethyl Valerate | — | — | 51.10 ± 2.26 | 71.12 ± 3.92 |
| 5 | Methyl Hexoate | — | — | 81.62 ± 8.10 | 223.08 ± 16.28 |
| 6 | Ethyl Hexanoate | — | — | 1230.34 ± 49.12 | 3521.68 ± 472.54 |
| 7 | Hexyl Acetate | — | — | 845.88 ± 10.42 | 1082.00 ± 31.72 |
| 8 | Ethyl Hex-3-Enoate | — | — | 13.16 ± 0.52 | — |
| 9 | Heptyl Hexanoate | — | — | 24.72 ± 1.28 | — |
| 10 | Hex-2-Enoic Acid Ethyl Ester | — | — | — | 47.00 ± 0.74 |
| 11 | Heptylacetat | — | — | 14.98 ± 1.10 | 39.6 ± 1.22 |
| 12 | Methyl Octylate | — | — | — | 31.26 ± 1.18 |
| 13 | Hexyl Butyrate | — | 13.56 ± 0.26 | 20.44 ± 0.60 | — |
| 14 | Ethyl Caprylate | — | — | 51.54 ± 0.78 | 285.88 ± 8.42 |
| 15 | Ethyl (Z)-Oct-4-Enoate | — | — | 12.08 ± 0.68 | — |
| 16 | Octyl Acetate | — | — | — | 34.72 ± 0.18 |
| 17 | Ethyl Trans-2-Octenoate | — | — | — | 111.5 ± 2.30 |
| 18 | Chloroformic Acid N-Octyl Ester | — | — | 24.32 ± 0.22 | 76.52 ± 0.90 |
| 19 | Ethyl 3-Methylthiopropionate | — | — | 22.42 ± 1.14 | 55.92 ± 0.34 |
| 20 | Hexyl Hexanoate | — | 25.44 ± 2.68 | 131.26 ± 4.18 | — |
| 21 | Ethyl Caprate | — | — | — | 35.96 ± 3.78 |
| 22 | Ethyl 3-Hydroxyhexanoate | — | — | — | 50.94 ± 1.44 |
| 23 | Ethyl Phenylacetate | — | — | — | 59.28 ± 0.60 |
| 24 | Methyl (2Z,4E)-2,4-Decadienoate | — | — | — | 132.58 ± 0.96 |
| 25 | Phenethyl Acetate | — | — | 35 ± 1.40 | 74.4 ± 0.84 |
| 26 | Fema 3148 | — | — | 18.56 ± 0.40 | 457.04 ± 14.06 |
| 27 | Pentanoic Acid, 2,2,4-Trimethyl-3-Carboxyisopropyl, Isobutyl Ester | 10.72 ± 1.04 | — | — | 27.34 ± 2.68 |
| 28 | Phosphorochloridic Acid, Propyl Undecyl Ester | — | — | — | 70.46 ± 1.10 |
| 29 | Ethyl 9-Tetradecenoate | — | — | — | 74.4 ± 5.10 |
| 30 | Isophthalic Acid, Ethyl 2-Propylphenyl Ester | 9.68 ± 0.34 | — | — | — |
| 31 | Ethyl Palmitate | — | — | — | 90.64 ± 4.88 |
| Ketones | |||||
| 32 | 6-Methylhept-5-En-2-One | — | — | 43.18 ± 0.56 | 134.58 ± 0.90 |
| 33 | (E)-4-Oxohex-2-Enal | 80.00 ± 2.00 | 66.7 ± 2.70 | 15.38 ± 0.54 | — |
| 34 | 1,5,6,7-Tetrahydro-4H-Indol-4-One | — | 27.74 ± 2.74 | — | — |
| 35 | N-Cyclobutylidenehydroxylamine | 62.88 ± 3.32 | 42.80 ± 0.42 | — | — |
| Terpenoids | |||||
| 36 | Cis-Β-Farnesene | — | — | 12.42 ± 0.48 | 23.7 ± 1.12 |
| 37 | (E)-3,7-Dimethylocta-1,3,6-Triene | — | — | 30.56 ± 1.88 | 39.06 ± 1.28 |
| 38 | Zingiberene | — | — | 26.76 ± 1.58 | — |
| 39 | 2,6-Dimethyl-6-(4-Methyl-3-Pentenyl)Bicyclo[3.1.1]Hept-2-Ene | — | — | — | 35.92 ± 3.02 |
| 40 | Farnesene | — | 534.42 ± 53.72 | 3606.7 ± 83.56 | 7756.26 ± 88.28 |
| 41 | (-)-Β-Chamigrene | — | — | 72.8 ± 0.62 | — |
| 42 | 1,3-Cyclopentadiene, 1,3-Bis(1-Methylethyl)- | — | — | 33.82 ± 1.06 | — |
| 43 | (3Z,6E)-3,7,11-Trimethyldodeca-1,3,6,10-Tetraene | — | — | 29.12 ± 1.36 | 44.00 ± 2.1- |
| 44 | 2,5-Cyclohexadiene, 1,4-Diethyl-1,4-Dimethyl- | — | — | — | 173.44 ± 0.6- |
| 45 | 1H-Indene, 2,3,3A,4-Tetrahydro-3,3A,6-Trimethyl-1-(1-Methylethyl)- | — | — | 17.8 ± 0.16 | — |
| 46 | Cyperene | — | — | — | 33.6 ± 1.1- |
| 47 | Guaiazulene | — | — | 56.2 ± 1.5- | 70.96 ± 0.18 |
| 48 | Isolongifolene, 9,10-Dehydro- | — | — | — | 28.04 ± 1.66 |
| 49 | .Beta.-Vatirenene | — | — | — | 28.46 ± 1.64 |
| 50 | Aromadendrene, Dehydro- | — | — | 18.7 ± 0.34 | 38.10 ± 1.68 |
| Aldehydes | — | — | — | — | |
| 51 | Hexanal | 619.12 ± 4.60 | 1563.84 ± 19.48 | 818.24 ± 14.06 | 742.60 ± 23.88 |
| 52 | 3-Hexenal | 86.72 ± 4.90 | — | — | — |
| 53 | Trans-2-Hexenal | 273.02 ± 14.34 | 1621.42 ± 39.26 | 1013.08 ± 11.68 | 704.04 ± 7.98 |
| 54 | (E,E)-2,4-Heptadienal | — | 9.08 ± 0.10 | — | — |
| Alcohols | |||||
| 55 | 1-Hexanol | — | — | — | 32.20 ± 0.80 |
| Benzenes | |||||
| 56 | Benzaldehyde | 20.02 ± 0.60 | 23.88 ± 0.54 | — | — |
| 57 | Benzeneethanamine, N-Butyl-.Beta.,4-Bis[(Trimethylsilyl)Oxy]- | — | 8.68 ± 0.02 | — | — |
| 58 | Silane, [[4-[1,2-Bis[(Trimethylsilyl)Oxy]Ethyl]-1,2-Phenylene]Bis(Oxy)]Bis[Trimethyl- | 8.68 ± 0.40 | 12.52 ± 0.74 | 16.74 ± 0.18 | — |
| 59 | Phenylacetaldehyde | 21.08 ± 0.60 | 14.98 ± 2.08 | 18.24 ± 1.64 | — |
| 60 | 1H-Benzocycloheptene, 2,4A,5,6,7,8,9,9A-Octahydro-3,5,5-Trimethyl-9-Methylene- | — | 11.96 ± 1.18 | — | 111.62 ± 1.48 |
| 61 | Phenylacetaldehyde | 47.12 ± 1.8 | 26.76 ± 2.48 | 53.34 ± 1.00 | 56.14 ± 2.68 |
| 62 | Hexestrol, O-Trifluoroacetyl- | — | 23.52 ± 1.84 | 91.3 ± 2.66 | — |
| 63 | 4-Tert-Butylphenol | — | — | 155.8 ± 2.12 | — |
| 64 | 4-Butyl-Benzonitrile | — | — | 17.34 ± 0.62 | — |
| 65 | 1,2,3,4-Tetrahydrophenanthren-9-Ol | — | 20.3 ± 1.60 | — | — |
| 66 | 2,4-Di-T-Butylphenol | 206.46 ± 28.52 | 106.88 ± 5.76 | 198.00 ± 9.36 | 151.7 ± 12.82 |
| Others | |||||
| 67 | Hexamethylcyclotrisiloxane | 9.30 ± 0.58 | 10.92 ± 0.20 | — | — |
| 68 | Octamethylcyclotetrasiloxane | — | 12.16 ± 1.06 | — | — |
| 69 | 4-[2-(5-Nitro-Furan-2-Yl)-Vinyl]-Quinolin-2-Ylamine | 12.36 ± 0.38 | — | — | — |
| 70 | Cyclohexene Oxide | 16.34 ± 1.76 | — | — | — |
| 71 | Methyl Cyclohexane | — | 16.18 ± 0.98 | — | — |
| 72 | Dodecamethylcyclohexasiloxane | 8.02 ± 0.50 | 9.68 ± 1.12 | — | — |
| 73 | Methylcyclopentane | — | — | 40.94 ± 0.44 | — |
| 74 | Tetradecamethyl Cycloheptasiloxane | 15.58 ± 0.18 | 25.68 ± 0.44 | 24.08 ± 1.20 | 49.28 ± 2.40 |
| 75 | Piperazine | 8.52 ± 0.18 | — | — | — |
| 76 | N-Heptadecane | — | 12.00 ± 0.32 | — | — |
| 77 | Ethyl 3-Methylsulfanylprop-2-Enoate | — | — | 36.24 ± 0.18 | 71.4 ± 1.56 |
| 78 | Tricyclo[3.1.0.0(2,4)]Hexane, 3,6-Diethyl-3,6-Dimethyl-, Trans- | — | — | 27.48 ± 0.64 | 48.68 ± 0.20 |
| 79 | (1S,5S)-9-Borabicyclo[3.3.1]Nonan-9-Ol | — | — | — | 48.26 ± 2.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Yin, Z. The Aroma Volatile in ‘Nanguo’ Pear: A Review. Horticulturae 2023, 9, 339. https://doi.org/10.3390/horticulturae9030339
Zhang Z, Yin Z. The Aroma Volatile in ‘Nanguo’ Pear: A Review. Horticulturae. 2023; 9(3):339. https://doi.org/10.3390/horticulturae9030339
Chicago/Turabian StyleZhang, Zhuoran, and Zepeng Yin. 2023. "The Aroma Volatile in ‘Nanguo’ Pear: A Review" Horticulturae 9, no. 3: 339. https://doi.org/10.3390/horticulturae9030339
APA StyleZhang, Z., & Yin, Z. (2023). The Aroma Volatile in ‘Nanguo’ Pear: A Review. Horticulturae, 9(3), 339. https://doi.org/10.3390/horticulturae9030339





