Dairy-Based Probiotic-Fermented Functional Foods: An Update on Their Health-Promoting Properties
Abstract
1. Introduction
2. Dairy-Based Functional Food: Its Composition and Application in Human Health
2.1. Other Dairy-Based Fermented Products
2.2. Positive Influence of Fermented Whey-Based Products
2.3. Fermented Dairy Product as a Probiotic Vehicle
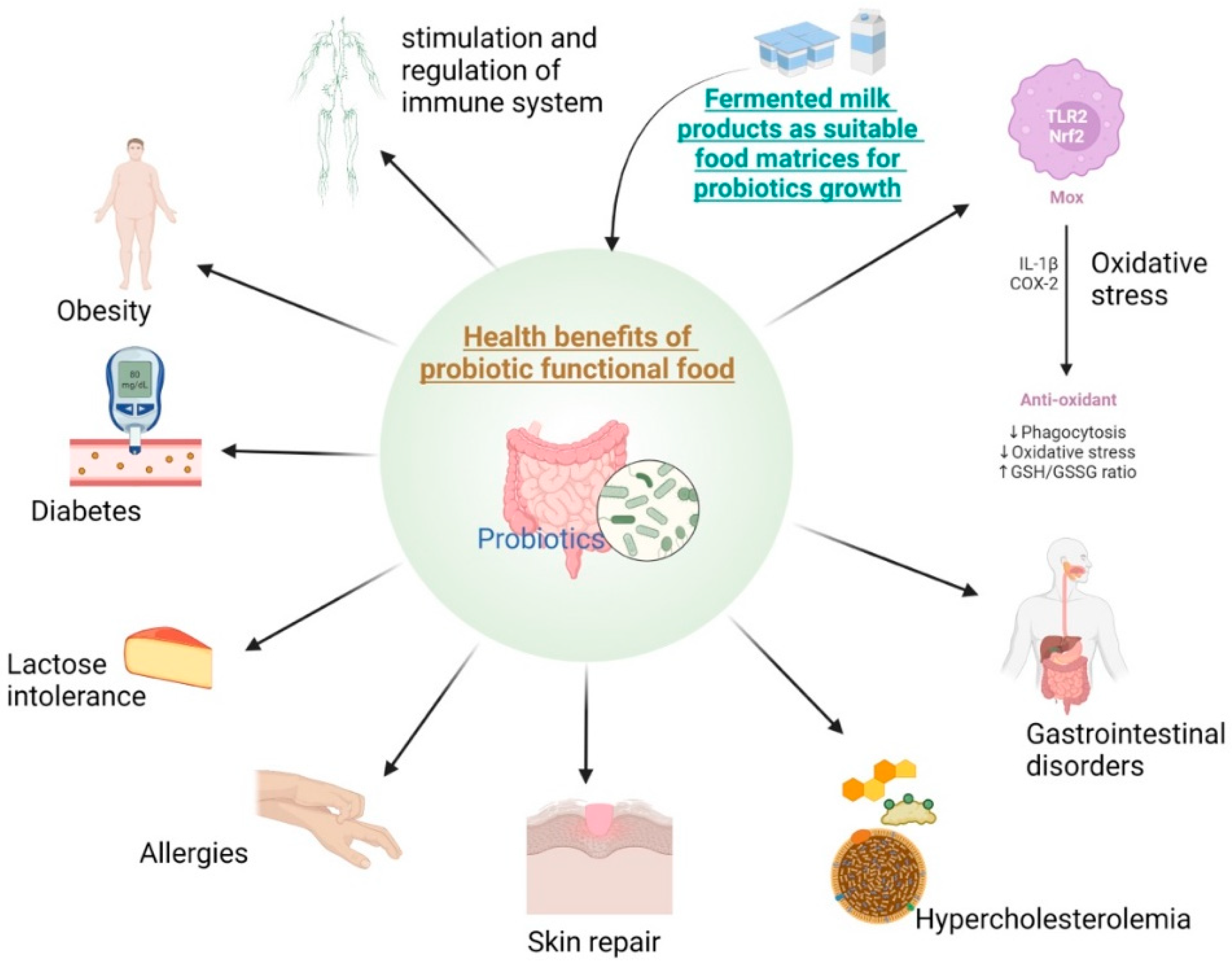
2.4. Health Benefits of Probiotic Functional Food
2.4.1. Obesity and Diabetes
2.4.2. Lactose Intolerance
2.4.3. Allergies
2.4.4. Oxidative Stress
2.4.5. Skin Repair
2.4.6. Hypercholesterolemia
2.4.7. Gastrointestinal Disorders
3. Probiotic-Fermented Products: Stimulation and Regulation of the Immune System
3.1. Probiotic-Fermented-Product-Mediated Modulation of DCs
3.2. Probiotic-Fermented-Product-Mediated Modulation of NK Cells
3.3. Probiotic-Fermented-Product-Mediated Modulation of Neutrophils
3.4. Probiotic-Fermented-Product-Mediated Modulation of Cytokines and Immunoglobulin
3.5. TLR-Mediated Immune Response and Probiotic-Fermented Products
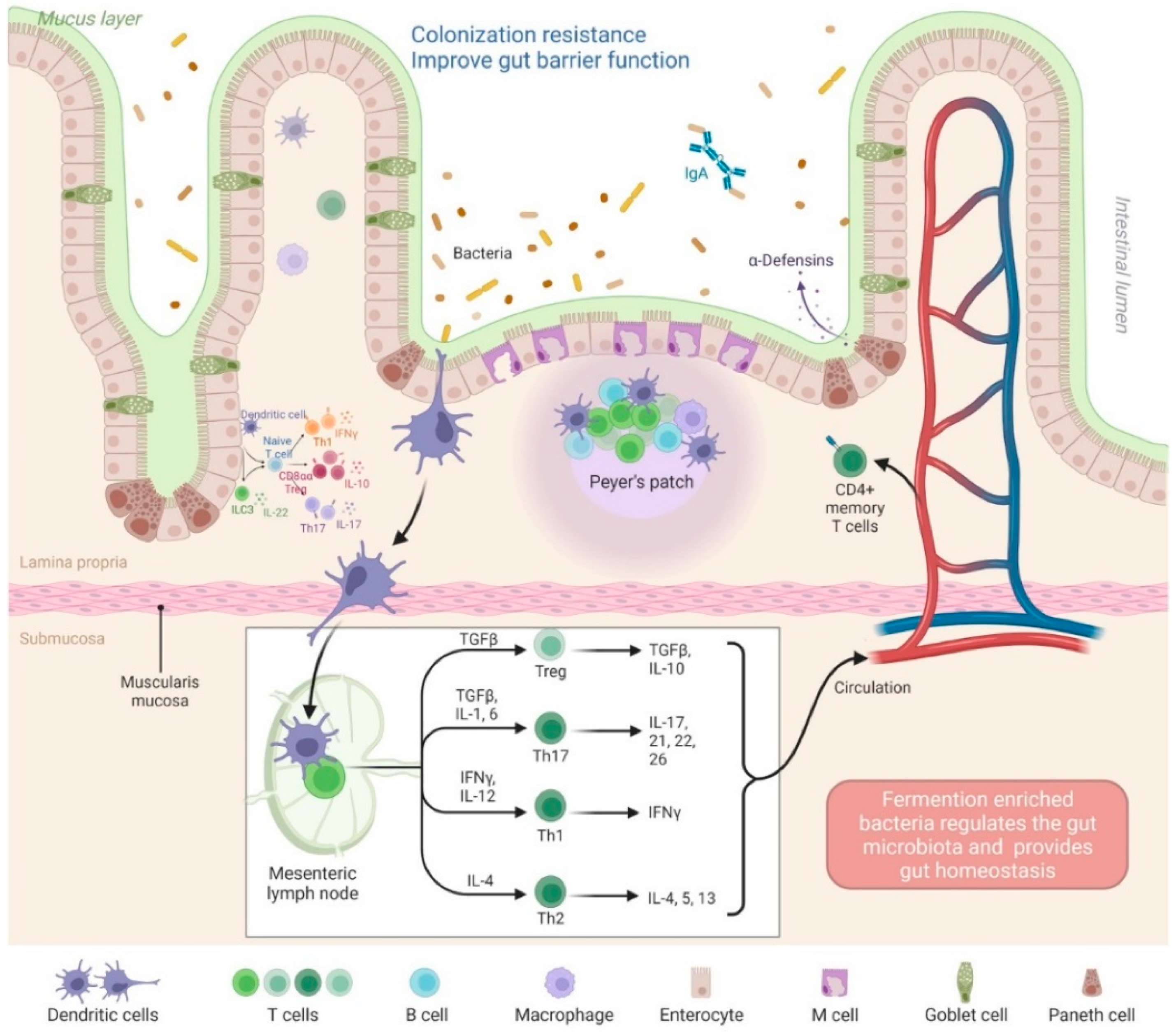
4. Probiotic-Fermented-Product-Mediated Reinforcement of Gut Barrier Function
4.1. Modulation of Tight Junctional Barrier Integrity
4.2. Modulation of Gut Permeability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Górska, W.H.; Rejman, K.L.W.; Czeczotko, M. Milk and dairy products and their nutritional contribution to the average polish diet. Nutrients 2019, 11, 1771. [Google Scholar] [CrossRef] [PubMed]
- Cena, H.; Calder, P.C. Defining a healthy diet: Evidence for the role of contemporary dietary patterns in health and disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef] [PubMed]
- Deledda, A.; Annunziata, G.; Tenore, G.C.; Palmas, V.; Manzin, A.; Velluzzi, F. Diet-derived antioxidants and their role in inflammation, obesity and gut microbiota modulation. Antioxidants 2021, 10, 708. [Google Scholar] [CrossRef] [PubMed]
- García, B.M.; Moreno, F.J.; Alférez, M.J.; Díaz-Castro, J.; López, A.I. New perspectives in fermented dairy products and their health relevance. J. Funct. Foods. 2020, 72, 104059. [Google Scholar] [CrossRef]
- Savaiano, D.A.; Hutkins, R.W. Yogurt, cultured fermented milk, and health: A systematic review. Nutr. Rev. 2021, 79, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Singh, P.; Nataraj, B.H.; Kokkiligadda, A.; Naithani, H.; Ali, S.A.; Nagpal, R. Fostering next-generation probiotics in human gut by targeted dietary modulation: An emerging perspective. Int. Food Res. J. 2021, 150, 110716. [Google Scholar] [CrossRef]
- Bintsis, T. Lactic acid bacteria as starter cultures: An update in their metabolism and genetics. AIMS Microbiol. 2018, 4, 665. [Google Scholar] [CrossRef]
- Rezac, S.; Kok, C.R.; Heermann, M.; Hutkins, R. Fermented foods as a dietary source of live organisms. Front. Microbiol. 2018, 9, 1785. [Google Scholar] [CrossRef]
- Nataraj, B.H.; Ali, S.A.; Behare, P.V.; Yadav, H. Postbiotics-parabiotics: The new horizons in microbial biotherapy and functional foods. Microb. Cell Factories 2020, 19, 168. [Google Scholar] [CrossRef] [PubMed]
- Auestad, N.; Layman, D.K. Dairy bioactive proteins and peptides: A narrative review. Nutr Rev. 2021, 79, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Ali, S.A.; Singh, S.K.; Bhushan, V.; Mathur, M.; Jamwal, S.; Kumar, S. Antimicrobial peptides in farm animals: An updated review on its diversity, function, modes of action and therapeutic prospects. Vet. Sci. 2020, 7, 206. [Google Scholar] [CrossRef] [PubMed]
- Solanki, S.S.; Singh, P.; Kashyap, P.; Sansi, M.S.; Ali, S.A. Promising role of defensins peptides as therapeutics to combat against viral infection. Microb. Pathog. 2021, 155, 104930. [Google Scholar] [CrossRef] [PubMed]
- Fortea, M.; Albert, B.M.; Abril, G.M.; Ganda Mall, J.P.; Serra-Ruiz, X.; Henao, P.A.; Expósito, E.; González-Castro, A.M.; Guagnozzi, D.; Lobo, B.; et al. Present and future therapeutic approaches to barrier dysfunction. Front. Nutr. 2021, 8, 718093. [Google Scholar] [CrossRef] [PubMed]
- Chopra, A.; Ali, S.A.; Bathla, S.; Rawat, P.; Vohra, V.; Kumar, S.; Mohanty, A.K. High-resolution mass spectrometer–based ultra-deep profile of milk whey proteome in Indian Zebu (Sahiwal) cattle. Front. Nutr. 2020, 7, 150. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.M.; Kang, E.A.; Park, J.M.; Oh, J.Y.; Lee, D.Y.; Choi, S.H.; Hahm, K.B. Dietary intake of fermented kimchi prevented colitis-associated cancer. J. Clin. Biochem. Nutr. 2020, 20–77. [Google Scholar] [CrossRef]
- Kaur, H.; Gupta, T.; Kapila, S.; Kapila, R. Protective effects of potential probiotic Lactobacillus rhamnosus (MTCC-5897) fermented whey on reinforcement of intestinal epithelial barrier function in a colitis-induced murine model. Food. Funct. 2021, 12, 6102–6116. [Google Scholar] [CrossRef]
- Matsuoka, K.; Uemura, Y.; Kanai, T.; Kunisaki, R.; Suzuki, Y.; Yokoyama, K.; Yoshimura, N.; Hibi, T. Efficacy of Bifidobacterium breve fermented milk in maintaining remission of ulcerative colitis. Dig. Dis. Sci. 2018, 63, 1910–1919. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, R.K.; Malhotra, S.; Pothuraju, R.; Shandilya, U.K. Lactobacillus rhamnosus NCDC17 ameliorates type-2 diabetes by improving gut function, oxidative stress and inflammation in high-fat-diet fed and streptozotocintreated rats. Benef. Microbes 2017, 8, 243–255. [Google Scholar] [CrossRef]
- Rong, J.; Zheng, H.; Liu, M.; Hu, X.; Wang, T.; Zhang, X.; Jin, F.; Wang, L. Probiotic and anti-inflammatory attributes of an isolate Lactobacillus helveticus NS8 from Mongolian fermented koumiss. BMC Microbiol. 2015, 15, 196. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; González-Córdova, A.F.; Hernández-Mendoza, A.; Torres-Inguanzo, E.H.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Randomized double-blind controlled clinical trial of the blood pressure–lowering effect of fermented milk with Lactococcus lactis: A pilot study. Int. J. Dairy Sci. 2018, 101, 2819–2825. [Google Scholar] [CrossRef]
- Revuelta, J.L.; Serrano-Amatriain, C.; Ledesma-Amaro, R.; Jiménez, A. Formation of folates by microorganisms: Towards the biotechnological production of this vitamin. Appl. Microbiol. Biotechnol. 2018, 102, 8613–8620. [Google Scholar] [CrossRef] [PubMed]
- Saubade, F.; Hemery, Y.M.; Rochette, I.; Guyot, J.P.; Humblot, C. Influence of fermentation and other processing steps on the folate content of a traditional African cereal-based fermented food. Int. J. Food Microbiol. 2018, 266, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Thumthanaruk, B.; Shetty, K. Changes in physico-chemical, astringency, volatile compounds and antioxidant activity of fresh and concentrated cashew apple juice fermented with Lactobacillus plantarum. J. Food Sci. Technol. 2018, 55, 3979–3990. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, M.; Fabritius, M.; Yang, B. Determination of vitamin K composition of fermented food. Food Chem. 2019, 275, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Merenstein, D.; Murphy, M.; Fokar, A.; Hernandez, R.K.; Park, H.; Nsouli, H.; Sanders, M.E.; Davis, B.A.; Niborski, V.; Tondu, F.; et al. Use of a fermented dairy probiotic drink containing Lactobacillus casei (DN-114 001) to decrease the rate of illness in kids: The DRINK study A patient-oriented, double-blind, cluster-randomized, placebo-controlled, clinical trial. Eur. J. Clin. Nutr. 2010, 64, 669–677. [Google Scholar] [CrossRef]
- Perna, A.; Intaglietta, I.; Simonetti, A.; Gambacorta, E. Donkey milk for manufacture of novel functional fermented beverages. J. Food Sci. 2015, 80, S1352–S1359. [Google Scholar] [CrossRef]
- Ho, S.W.; El-Nezami, H.; Shah, N.P. The protective effects of enriched citrulline fermented milk with Lactobacillus helveticus on the intestinal epithelium integrity against Escherichia coli infection. Sci. Rep. 2020, 10, 499. [Google Scholar] [CrossRef] [PubMed]
- Sivasankari, R.; Hemalatha, G.; Kanchana, S.; Umamaheswari, T. Standardization and evaluation of probiotic shrikhand. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 41–47. [Google Scholar] [CrossRef]
- Sevencan, N.O.; Isler, M.; Kapucuoglu, F.N.; Senol, A.; Kayhan, B.; Kiztanir, S.; Kockar, M.C. Dose-dependent effects of kefir on colitis induced by trinitrobenzene sulfonic acid in rats. Food Sci. Nutr. 2019, 7, 3110–3118. [Google Scholar] [CrossRef]
- Sánchez, E.; Nieto, J.C.; Vidal, S.; Santiago, A.; Martinez, X.; Sancho, F.J.; Sancho-Bru, P.; Mirelis, B.; Corominola, H.; Juárez, C.; et al. Fermented milk containing Lactobacillus paracasei subsp. paracasei CNCM I-1518 reduces bacterial translocation in rats treated with carbon tetrachloride. Sci. Rep. 2017, 7, 45712. [Google Scholar] [CrossRef]
- Akram, M.; Ali, S.A.; Behare, P.; Kaul, G. Dietary intake of probiotic fermented milk benefits the gut and reproductive health in mice fed with an obesogenic diet. Food Funct. 2022, 13, 737–752. [Google Scholar] [CrossRef]
- Yadav, R.; Vij, R.; Kapila, S.; Khan, S.H.; Kumar, N.; Meena, S.; Kapila, R. Milk fermented with probiotic strains Lactobacillus rhamnosus MTCC: 5957 and Lactobacillus rhamnosus MTCC: 5897 ameliorates the diet-induced hypercholesterolemia in rats. Ann. Microbiol. 2019, 69, 483–494. [Google Scholar] [CrossRef]
- Companys, J.; Pla-Pagà, L.; Calderón-Pérez, L.; Llauradó, E.; Solà, R.; Pedret, A.; Valls, R.M. Fermented dairy products, probiotic supplementation, and cardiometabolic diseases: A systematic review and meta-analysis. Adv. Nutr. 2020, 11, 834–863. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Chen, H.; Chen, H.; Ding, W. Effects of fermentation with Lactobacillus rhamnosus GG on product quality and fatty acids of goat milk yogurt. Int. J. Dairy Sci. 2016, 99, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, F.; Siroli, L.; Parolin, C.; Serrazanetti, D.I.; Vitali, B.; Lanciotti, R. Use of Lactobacillus crispatus to produce a probiotic cheese as potential gender food for preventing gynaecological infections. PLoS ONE 2019, 14, 0208906. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, K.; Kim, W. Cream cheese-derived Lactococcus chungangensis CAU 28 modulates the gut microbiota and alleviates atopic dermatitis in BALB/c mice. Sci. Rep. 2019, 9, 446. [Google Scholar] [CrossRef]
- Leeuwendaal, N.K.; Hayes, J.J.; Stanton, C.; O’Toole, P.W.; Beresford, T.P. Protection of candidate probiotic lactobacilli by Cheddar cheese matrix during simulated gastrointestinal digestion. J. Funct. Foods 2022, 92, 105042. [Google Scholar] [CrossRef]
- Yan, S.; Yang, B.; Ross, R.P.; Stanton, C.; Zhang, H.; Zhao, J.; Chen, W. Bifidobacterium longum subsp. longum YS108R fermented milk alleviates DSS induced colitis via anti-inflammation, mucosal barrier maintenance and gut microbiota modulation. J. Funct. Foods 2020, 73, 104153. [Google Scholar] [CrossRef]
- Lee, K.Y.; Tsai, Y.C.; Wang, S.Y.; Chen, Y.P.; Chen, M.J. Coculture Strategy for Developing Lactobacillus paracasei PS23 Fermented Milk with Anti-Colitis Effect. Foods 2021, 10, 2337. [Google Scholar] [CrossRef]
- Sumi, K.; Osada, K.; Sakuda, M.; Ashida, K.; Nakazato, K. Fermented milk retains beneficial effects on skeletal muscle protein anabolism after processing by centrifugation and supernatant removal. Int. J. Dairy Sci. 2021, 104, 1336–1350. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Han, X.; Cen, S.; Duan, H.; Feng, S.; Xue, Y.; Tian, F.; Zhao, J.; Zhang, H.; Zhai, Q.; et al. Beneficial effect of GABA-rich fermented milk on insomnia involving regulation of gut microbiota. Microbiol. Res. 2020, 233, 126409. [Google Scholar] [CrossRef]
- Tavakoli, M.; Najafi, M.B.H.; Mohebbi, M. Effect of the milk fat content and starter culture selection on proteolysis and antioxidant activity of probiotic yogurt. Heliyon 2019, 5, 01204. [Google Scholar] [CrossRef]
- Minj, S.; Anand, S. Whey proteins and its derivatives: Bioactivity, functionality, and current applications. Dairy 2020, 1, 233–258. [Google Scholar] [CrossRef]
- Wolber, F.M.; Broomfield, A.M.; Fray, L.; Cross, M.L.; Dey, D. Supplemental dietary whey protein concentrate reduces rotavirus-induced disease symptoms in suckling mice. J. Nutr. 2005, 135, 1470–1474. [Google Scholar] [CrossRef]
- Vasconcelos, Q.D.J.S.; Bachur, T.P.R.; Aragão, G.F. Whey protein supplementation and its potentially adverse effects on health: A systematic review. Appl. Physiol. Nutr. Metab. 2021, 46, 27–33. [Google Scholar] [CrossRef]
- Lorentz, A.; Müller, L. Probiotics in the Treatment of Inflammatory Bowel Disease in Adulthood: A Systematic Review. Gastrointestin. Liver Dis. 2022, 31, 74–84. [Google Scholar] [CrossRef]
- Karimi, R.; Mortazavian, A.M.; Amiri, R.A. Selective enumeration of probiotic microorganisms in cheese. Food Microbiol. 2012, 29, 1–9. [Google Scholar] [CrossRef]
- Januś, E.; Sablik, P.; Jakubowska, M.; Wróbel, K. Microbiological, sensory, and physicochemical quality of curd cheeses originating from direct sales. Vet. World 2021, 14, 3038. [Google Scholar] [CrossRef]
- Tang, H.; Ma, H.; Hou, Q.; Li, W.; Xu, H.; Liu, W.; Sun, Z.; Haobisi, H.; Menghe, B. Profiling of koumiss microbiota and organic acids and their effects on koumiss taste. BMC Microbiol. 2020, 20, 85. [Google Scholar] [CrossRef]
- Afzaal, M.; Saeed, F.; Anjum, F.; Waris, N.; Husaain, M.; Ikram, A.; Ateeq, H.; Muhammad Anjum, F.; Suleria, H. Nutritional and ethnomedicinal scenario of koumiss: A concurrent review. Food Sci. Nutr. 2021, 9, 6421–6428. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, C.; Xilin, T.; Ji, M.; Meng, X.; Zhao, Y.; Siqin, B.; Zhang, N.; Li, M. Effects of Koumiss on Intestinal Immune Modulation in Immunosuppressed Rats. Front. Nutr. 2022, 9, 765499. [Google Scholar] [CrossRef]
- Tiss, M.; Souiy, Z.; ben Abdeljelil, N.; Njima, M.; Achour, L.; Hamden, K. Fermented soy milk prepared using kefir grains prevents and ameliorates obesity, type 2 diabetes, hyperlipidemia and liver-kidney toxicities in HFFD-rats. J. Funct. Foods 2020, 67, 103869. [Google Scholar]
- Mohan, J.; Ali, S.A.; Suvartan, R.; Kapila, S.; Sharma, R.; Tomar, S.K.; Yadav, H. Bioavailability of biotransformed zinc enriched ahi in wistar rats. Int. J. Probiotics Prebiotics 2018, 13, 45. [Google Scholar]
- Gawande, K.; Kolhekar, M.; Kumari, M.; Kapila, S.; Sharma, P.; Ali, S.A.; Behare, P.V. Lactic acid bacteria based purified exopolysaccharide showed viscofying and hypercholesterolemic capabilites. Food Hydrocoll. 2021, 1, 100042. [Google Scholar] [CrossRef]
- Bengoa, A.A.; Dardis, C.; Garrote, G.L.; Abraham, A.G. Health-promoting properties of Lacticaseibacillus paracasei: A focus on kefir isolates and exopolysaccharide-producing strains. Foods 2021, 10, 2239. [Google Scholar] [CrossRef]
- Perazza, L.R.; Daniel, N.; Dubois, M.J.; Pilon, G.; Varin, T.V.; Blais, M.; Martinez Gonzales, J.L.; Bouchard, M.; Asselin, C.; Lessard, M.; et al. Distinct effects of milk-derived and fermented dairy protein on gut microbiota and cardiometabolic markers in diet-induced obese mice. J. Nutr. 2020, 150, 2673–2686. [Google Scholar] [CrossRef]
- Utz, V.E.M.; Perdigón, G.; de LeBlanc, A.D.M. Oral administration of milk fermented by Lactobacillus casei CRL431 was able to decrease metastasis from breast cancer in a murine model by modulating immune response locally in the lungs. J. Funct. Food. 2019, 54, 263–270. [Google Scholar]
- Gupta, C.; Prakash, D. Therapeutic potential of milk whey. Beverages 2017, 3, 31. [Google Scholar] [CrossRef]
- Mehra, R.; Kumar, H.; Kumar, N.; Ranvir, S.; Jana, A.; Buttar, H.S.; Telessy, I.G.; Awuchi, C.G.; Okpala, C.O.R.; Korzeniowska, M. Whey proteins processing and emergent derivatives: An insight perspective from constituents, bioactivities, functionalities to therapeutic applications. J. Funct. Foods 2021, 87, 104760. [Google Scholar]
- Hering, N.A.; Andres, S.; Fromm, A.; van Tol, E.A.; Amasheh, M.; Mankertz, J.; Fromm, M.; Schulzke, J.D. Transforming growth factor-β, a whey protein component, strengthens the intestinal barrier by upregulating claudin-4 in HT-29/B6 cells. J. Nutr. 2011, 141, 783–789. [Google Scholar] [CrossRef]
- Araujo, D.F.; Guerra, G.C.; Pintado, M.M.E.; Sousa, Y.R.; Algieri, F.; Rodriguez-Nogales, A.; Araujo Jr, R.F.; Galvez, J.; Queiroga, R.D.C.R.; Rodriguez-Cabezas, M.E. Intestinal anti-inflammatory effects of goat whey on DNBS-induced colitis in mice. PLoS ONE 2017, 12, e0185382. [Google Scholar] [CrossRef] [PubMed]
- Rusu, E.; Enache, G.; Cursaru, R.; Alexescu, A.; Radu, R.; Onila, O.; Cavallioti, T.; Rusu, F.; Posea, M.; Jinga, M.; et al. Prebiotics and probiotics in atopic dermatitis. Exp. Ther. Med. 2019, 18, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.N.; Sangild, P.T.; Li, Y.; Bering, S.B.; Chatterton, D.E. Processing of whey modulates proliferative and immune functions in intestinal epithelial cells. Int. J. Dairy Sci. 2016, 99, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.H.; Lee, H.A.; Lee, K.B.; Kim, S.H.; Park, K.Y.; Lee, K.W. Effects of glycated whey protein concentrate on pro-inflammatory cytokine expression and phagocytic activity in RAW264. 7 macrophages. Biol. Pharm. Bull. 2016, 39, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Stanifer, M.L.; Guo, C.; Doldan, P.; Boulant, S. Importance of type I and III interferons at respiratory and intestinal barrier surfaces. Front. Immunol. 2020, 11, 608645. [Google Scholar] [CrossRef]
- Rusu, D.; Drouin, R.; Pouliot, Y.; Gauthier, S.; Poubelle, P.E. A bovine whey protein extract stimulates human neutrophils to generate bioactive IL-1Ra through a NF-κB-and MAPK-dependent mechanism. J. Nutr. 2010, 140, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Dalziel, J.E.; Anderson, R.C.; Bassett, S.A.; Lloyd-West, C.M.; Haggarty, N.W.; Roy, N.C. Influence of bovine whey protein concentrate and hydrolysate preparation methods on motility in the isolated rat distal colon. Nutrients 2016, 8, 809. [Google Scholar] [CrossRef]
- Gupta, T.; Kaur, H.; Kapila, S.; Kapila, R. Potential probiotic Lacticaseibacillus rhamnosus MTCC-5897 attenuates Escherichia coli induced inflammatory response in intestinal cells. Arch. Microbiol. 2021, 203, 5703–5713. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, N.; Battista, N.; Prete, R.; Corsetti, A. Health-promoting role of lactiplantibacillus plantarum isolated from fermented foods. Microorganisms 2021, 9, 349. [Google Scholar]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional modulation of immune function: Analysis of evidence, mechanisms, and clinical relevance. Front. Immunol. 2019, 9, 3160. [Google Scholar] [CrossRef]
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef] [PubMed]
- Widyastuti, Y.; Febrisiantosa, A.; Tidona, F. Health-Promoting Properties of Lactobacilli in Fermented Dairy Products. Front. Microbiol. 2021, 12, 1189. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.M.; Ali, S.A.; Ceciliani, F.; Eckersall, P.D.; Hernandez-Castellano, L.E.; Han, R.; Zachut, M. Domestic animal proteomics in the 21st century: A global retrospective and viewpoint analysis. J. Proteom. 2021, 241, 104220. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Ali, S.A.; Kumar, S.; Mohanty, A.K.; Behare, P. Label-free quantitative proteomic analysis of Lactobacillus fermentum NCDC 400 during bile salt exposure. J. Proteom. 2017, 167, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Pragya, P.; Kaur, G.; Ali, S.A.; Bhatla, S.; Rawat, P.; Lule, V.; Behare, P. High-resolution mass spectrometry-based global proteomic analysis of probiotic strains Lactobacillus fermentum NCDC 400 and RS2. J. Proteom. 2017, 152, 121–130. [Google Scholar] [CrossRef]
- Ali, S.A.; Kumar, S.; Mohanty, A.K.; Behare, P. Draft genome sequence of Lactobacillus fermentum NCDC 400, isolated from a traditional Indian dairy product. Genome Announc. 2018, 6, e01492-17. [Google Scholar]
- Ali, S.A.; Singh, P.; Tomar, S.K.; Mohanty, A.K.; Behare, P. Proteomics fingerprints of systemic mechanisms of adaptation to bile in Lactobacillus fermentum. J. Proteom. 2020, 213, 103600. [Google Scholar] [CrossRef]
- Behare, P.V.; Ali, S.A.; McAuliffe, O. Draft genome sequences of Fructobacillus fructosus DPC 7238 and Leuconostoc mesenteroides DPC 7261, mannitol-producing organisms isolated from fructose-rich honeybee-resident flowers on an Irish farm. Microbiol. Resour. Announc. 2020, 9, e01297-20. [Google Scholar] [CrossRef]
- Ali, S.A.; Singh, A.K.; Tomar, S.K.; Behare, P. Genome Sequence of Lacticaseibacillus rhamnosus Strain NCDC610, Isolated from a Traditional Cereal-Based Fermented Milk Product (Raabadi). Microbiol. Resour. Announc. 2021, 10, e00672-21. [Google Scholar]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef]
- Marco, M.L.; Sanders, M.E.; Gänzle, M.; Arrieta, M.C.; Cotter, P.D.; De Vuyst, L.; Hill, C.; Holzapfel, W.; Lebeer, S.; Merenstein, D.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on fermented foods. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 196–208. [Google Scholar] [CrossRef]
- Hickey, C.D.; Sheehan, J.J.; Wilkinson, M.G.; Auty, M.A. Growth and location of bacterial colonies within dairy foods using microscopy techniques: A review. Front. Microbiol. 2015, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Nyanzi, R.; Jooste, P.J.; Buys, E.M. Invited review: Probiotic yogurt quality criteria, regulatory framework, clinical evidence, and analytical aspects. Int. J. Dairy Sci. 2021, 104, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in food systems: Significance and emerging strategies towards improved viability and delivery of enhanced beneficial value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Sun-Waterhouse, D.; Waterhouse, G.I.; Cui, C.; Ruan, Z. Fermentation-enabled wellness foods: A fresh perspective. Food Sci. Hum. Wellness 2019, 8, 203–243. [Google Scholar] [CrossRef]
- Mahmoudi, R.; Tajik, H.; Ehsani, A.; Zare, P. Physicochemical and hygienic effects of Lactobacillus acidophilus in Iranian white cheese. Vet. Res. Forum 2012, 3, 193. [Google Scholar]
- Wendel, U. Assessing Viability and Stress Tolerance of Probiotics-A Review. Front. Microbiol 2021, 12, 818468. [Google Scholar] [CrossRef]
- Damián, M.R.; Cortes-Perez, N.G.; Quintana, E.T.; Ortiz-Moreno, A.; Garfias Noguez, C.; Cruceño-Casarrubias, C.E.; Sánchez Pardo, M.E.; Bermúdez-Humarán, L.G. Functional Foods, Nutraceuticals and Probiotics: A Focus on Human Health. Microorganisms 2022, 10, 1065. [Google Scholar] [CrossRef]
- Panicker, A.S.; Ali, S.A.; Anand, S.; Panjagari, N.R.; Kumar, S.; Mohanty, A.K.; Behare, P.V. Evaluation of some in vitro probiotic properties of Lactobacillus fermentum Strains. J. Food Sci. Technol. 2018, 55, 2801–2807. [Google Scholar] [CrossRef]
- Koirala, S.; Anal, A.K. Probiotics-based foods and beverages as future foods and their overall safety and regulatory claims. J. Food Agric. Environ. 2021, 3, 100013. [Google Scholar] [CrossRef]
- Hendler, R.; Zhang, Y. Probiotics in the treatment of colorectal cancer. Medicines 2018, 5, 101. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tran, D.Q.; Rhoads, J.M. Probiotics in disease prevention and treatment. J. Clin. Pharmacol. 2018, 58, S164–S179. [Google Scholar] [CrossRef]
- Begum, P.S.; Madhavi, G.; Rajagopal, S.; Viswanath, B.; Razak, M.A.; Venkataratnamma, V. Probiotics as functional foods: Potential effects on human health and its impact on neurological diseases. Int. J. Nutr. Pharmacol. Neurol. Dis. 2017, 7, 23. [Google Scholar]
- Gomes, A.C.; Bueno, A.A.; de Souza, R.G.M.; Mota, J.F. Gut microbiota, probiotics and diabetes. Nutr. J. 2014, 13, 60. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.J.; Sui, H.; Qi, C.; Li, Q.; Zhang, J.; Wu, S.F.; Mei, M.Z.; Lu, Y.Y.; Wan, Y.T.; Chang, H.; et al. Ursolic acid inhibits proliferation and reverses drug resistance of ovarian cancer stem cells by downregulating ABCG2 through suppressing the expression of hypoxia-inducible factor-1α in vitro. Oncol. Rep. 2016, 36, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Alihosseini, N.; Moahboob, S.A.; Farrin, N.; Mobasseri, M.; Taghizadeh, A.; Ostadrahimi, A.R. Effect of probiotic fermented milk (kefir) on serum level of insulin and homocysteine in type 2 diabetes patients. Acta. Endocrinol. 2017, 13, 431. [Google Scholar] [CrossRef] [PubMed]
- Widodo, W.; Harsita, P.A.; Sukarno, A.S.; Nurrochmad, A. Antidiabetic effect of milk fermented using intestinal probiotics. Food Sci. Nutr. 2019, 49, 1063–1074. [Google Scholar] [CrossRef]
- Yadav, R.; Dey, D.K.; Vij, R.; Meena, S.; Kapila, R.; Kapila, S. Evaluation of anti-diabetic attributes of Lactobacillus rhamnosus MTCC: 5957, Lactobacillus rhamnosus MTCC: 5897 and Lactobacillus fermentum MTCC: 5898 in streptozotocin induced diabetic rats. Microb. Pathog. 2018, 125, 454–462. [Google Scholar] [CrossRef]
- Misselwitz, B.; Butter, M.; Verbeke, K.; Fox, M.R. Update on lactose malabsorption and intolerance: Pathogenesis, diagnosis and clinical management. Gut 2019, 68, 2080–2091. [Google Scholar] [CrossRef]
- Szilagyi, A.; Ishayek, N. Lactose intolerance, dairy avoidance, and treatment options. Nutrients 2018, 10, 1994. [Google Scholar] [CrossRef]
- Facioni, M.S.; Raspini, B.; Pivari, F.; Dogliotti, E.; Cena, H. Nutritional management of lactose intolerance: The importance of diet and food labelling. J. Transl. Med. 2020, 18, 260. [Google Scholar] [CrossRef] [PubMed]
- Forsgård, R.A. Lactose digestion in humans: Intestinal lactase appears to be constitutive whereas the colonic microbiome is adaptable. Am. J. Clin. Nutr. 2019, 110, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, U.; Malida, R.; Panda, R.; Halder, T.; Roymahapatra, G. Variety of yogurt and its health aspects—A brief review. Int. J. Innov. Pract. Appl. Res. 2017, 7, 56–66. [Google Scholar]
- Markowiak, P.; Śliżewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Dennis-Wall, J.C.; Culpepper, T.; Nieves Jr, C.; Rowe, C.C.; Burns, A.M.; Rusch, C.T.; Federico, A.; Ukhanova, M.; Waugh, S.; Mai, V.; et al. Probiotics (Lactobacillus gasseri KS-13, Bifidobacterium bifidum G9-1, and Bifidobacterium longum MM-2) improve rhinoconjunctivitis-specific quality of life in individuals with seasonal allergies: A double-blind, placebo-controlled, randomized trial. Am. J. Clin. Nutr. 2017, 105, 758–767. [Google Scholar] [CrossRef]
- Liu, M.Y.; Yang, Z.Y.; Dai, W.K.; Huang, J.Q.; Li, Y.H.; Zhang, J.; Qiu, C.Z.; Wei, C.; Zhou, Q.; Sun, X.; et al. Protective effect of Bifidobacterium infantis CGMCC313-2 on ovalbumin-induced airway asthma and β-lactoglobulin-induced intestinal food allergy mouse models. World, J. Gastroenterol. 2017, 23, 2149. [Google Scholar] [CrossRef]
- Saliganti, V.; Kapila, R.; Sharma, R.; Kapila, S. Feeding probiotic Lactobacillus rhamnosus (MTCC 5897) fermented milk to suckling mothers alleviates ovalbumin-induced allergic sensitisation in mice offspring. Br. J. Nutr. 2015, 114, 1168–1179. [Google Scholar] [CrossRef]
- Li Wang, H.; Ding, F.; Xiao, L.; Shi, R.; Wang, H.; Han, W.; Huang, Z. Food-derived antioxidant polysaccharides and their pharmacological potential in neurodegenerative diseases. Nutrients 2017, 9, 778. [Google Scholar]
- Wang, Y.; Wu, Y.; Wang, Y.; Fu, A.; Gong, L.; Li, W.; Li, Y. Bacillus amyloliquefaciens SC06 alleviates the oxidative stress of IPEC-1 via modulating Nrf2/Keap1 signaling pathway and decreasing ROS production. Appl. Microbiol. Biotechnol. 2017, 101, 3015–3026. [Google Scholar] [CrossRef]
- Nayik, G.A.; Jagdale, Y.D.; Gaikwad, S.A.; Devkatte, A.N.; Dar, A.H.; Dezmirean, D.S.; Bobis, O.; Ranjha, M.M.A.; Ansari, M.J.; Hemeg, H.A.; et al. Recent Insights into Processing Approaches and Potential Health Benefits of Goat Milk and Its Products: A Review. Front. Nutr. 2021, 8, 789117. [Google Scholar] [CrossRef]
- Salem, I.; Ramser, A.; Isham, N.; Ghannoum, M.A. The gut microbiome as a major regulator of the gut-skin axis. Front. Microbiol. 2018, 9, 1459. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, A.D.; Leoni, J.; Paz, M.L.; González Maglio, D.H. Lipoteichoic Acid from Lacticaseibacillus rhamnosus GG Modulates Dendritic Cells and T Cells in the Gut. Nutrients 2022, 14, 723. [Google Scholar] [CrossRef] [PubMed]
- Khmaladze, I.; Butler, É.; Fabre, S.; Gillbro, J.M. Lactobacillus reuteri DSM 17938—A comparative study on the effect of probiotics and lysates on human skin. Exp. Dermatol. 2019, 28, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Kober, M.M.; Bowe, W.P. The effect of probiotics on immune regulation, acne, and photoaging. Int. J. Women’s Dermatol. 2015, 1, 85–89. [Google Scholar] [CrossRef]
- Bianconi, V.; Banach, M.; Pirro, M.; Panel, I.L.E. Why patients with familial hypercholesterolemia are at high cardiovascular risk? Beyond LDL-C levels. Trends Cardiovasc Med. 2021, 31, 205–215. [Google Scholar] [CrossRef]
- Jesch, E.D.; Carr, T.P. Food ingredients that inhibit cholesterol absorption. Prev. Nutr. Food Sci. 2017, 22, 67. [Google Scholar]
- Machlik, M.L.; Hopstock, L.A.; Wilsgaard, T.; Hansson, P. Associations Between Intake of Fermented Dairy Products and Blood Lipid Concentrations Are Affected by Fat Content and Dairy Matrix–The Tromsø Study: Tromsø7. Front. Nutr. 2021, 8, 773468. [Google Scholar] [CrossRef]
- Jakubczyk, D.; Leszczyńska, K.; Górska, S. The effectiveness of probiotics in the treatment of inflammatory bowel disease (IBD)—A critical review. Nutrients 2020, 12, 1973. [Google Scholar] [CrossRef]
- Sartor, R.B. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: Antibiotics, probiotics, and prebiotics. Gastroenterology 2004, 126, 1620–1633. [Google Scholar] [CrossRef]
- Dore, M.P.; Bibbò, S.; Fresi, G.; Bassotti, G.; Pes, G.M. Side effects associated with probiotic use in adult patients with inflammatory bowel disease: A systematic review and meta-analysis of randomized controlled trials. Nutrients 2019, 11, 2913. [Google Scholar] [CrossRef]
- Gomi, A.; Yamaji, K.; Watanabe, O.; Yoshioka, M.; Miyazaki, K.; Iwama, Y.; Urita, Y. Bifidobacterium bifidum YIT 10347 fermented milk exerts beneficial effects on gastrointestinal discomfort and symptoms in healthy adults: A double-blind, randomized, placebo-controlled study. Int. J. Dairy Sci. 2018, 101, 4830–4841. [Google Scholar] [CrossRef] [PubMed]
- Eales, J.; Gibson, P.; Whorwell, P.; Kellow, J.; Yellowlees, A.; Perry, R.H.; Edwards, M.; King, S.; Wood, H.; Glanville, J. Systematic review and meta-analysis: The effects of fermented milk with Bifidobacterium lactis CNCM I-2494 and lactic acid bacteria on gastrointestinal discomfort in the general adult population. Therap. Adv. Gastroenter. 2017, 10, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Kaur, H.; Gupta, T.; Kapila, S.; Kapila, R. Role of fermented dairy foods in human health. Ind. J. Dairy Sci. 2020, 73, 97–110. [Google Scholar] [CrossRef]
- Gómez-Llorente, C.; Munoz, S.; Gil, A. Role of Toll-like receptors in the development of immunotolerance mediated by probiotics. Proc. Nutr. Soc. 2010, 69, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Carstensen, L.S.; Lie-Andersen, O.; Obers, A.; Crowther, M.D.; Svane, I.M.; Hansen, M. Long-term exposure to inflammation induces differential cytokine patterns and apoptosis in dendritic cells. Front. Immunol. 2019, 10, 2702. [Google Scholar] [CrossRef]
- Jeffrey, M.P.; Taggart, H.J.; Strap, J.L.; Edun, G.; Green-Johnson, J.M. Milk fermented with Lactobacillus rhamnosus R0011 induces a regulatory cytokine profile in LPS-challenged U937 and THP-1 macrophages. Curr. Res. Nutr. Food Sci. 2020, 3, 51–58. [Google Scholar] [CrossRef]
- Fernandez, E.M.; Valenti, V.; Rockel, C.; Hermann, C.; Pot, B.; Boneca, I.G.; Grangette, C. Anti-inflammatory capacity of selected lactobacilli in experimental colitis is driven by NOD2-mediated recognition of a specific peptidoglycan-derived muropeptide. Gut 2011, 60, 1050–1059. [Google Scholar] [CrossRef]
- Kwon, H.K.; Lee, C.G.; So, J.S.; Chae, C.S.; Hwang, J.S.; Sahoo, A.; Nam, J.H.; Rhee, J.H.; Hwang, K.C.; Im, S.H. Generation of regulatory dendritic cells and CD4+ Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc. Natl. Acad. Sci. USA 2010, 107, 2159–2164. [Google Scholar] [CrossRef]
- Miller, L.E.; Lehtoranta, L.; Lehtinen, M.J. Short-term probiotic supplementation enhances cellular immune function in healthy elderly: Systematic review and meta-analysis of controlled studies. Nutr. Res. 2019, 64, 1–8. [Google Scholar] [CrossRef]
- Gui, Q.; Wang, A.; Zhao, X.; Huang, S.; Tan, Z.; Xiao, C.; Yang, Y. Effects of probiotic supplementation on natural killer cell function in healthy elderly individuals: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2020, 74, 1630–1637. [Google Scholar] [CrossRef] [PubMed]
- Shida, K.; Sato, T.; Iizuka, R.; Hoshi, R.; Watanabe, O.; Igarashi, T.; Miyazaki, K.; Nanno, M.; Ishikawa, F. Daily intake of fermented milk with Lactobacillus casei strain Shirota reduces the incidence and duration of upper respiratory tract infections in healthy middle-aged office workers. Eur. J. Nutr. 2017, 56, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Rowland, I.; Tuohy, K.M.; Thomas, L.V.; Yaqoob, P. Selective effects of Lactobacillus casei Shirota on T cell activation, natural killer cell activity and cytokine production. Clin. Exp. Immunol. 2010, 161, 378–388. [Google Scholar] [CrossRef]
- Ko, M.W.; Kaur, K.; Safaei, T.; Chen, W.; Sutanto, C.; Wong, P.; Jewett, A. Defective Patient NK Function Is Reversed by AJ2 Probiotic Bacteria or Addition of Allogeneic Healthy Monocytes. Cells 2022, 11, 697. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Kozlowska, A.K.; Topchyan, P.; Ko, M.W.; Ohanian, N.; Chiang, J.; Cook, J.; Maung, P.O.; Park, S.H.; Cacalano, N.; et al. Probiotic-treated super-charged NK cells efficiently clear poorly differentiated pancreatic tumors in Hu-BLT mice. Cancers 2019, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Suzuki, T.; Shimada, S.I.; Shida, K.; Nanno, M.; Okumura, K. Interleukin-12 is involved in the enhancement of human natural killer cell activity by Lactobacillus casei Shirota. Clin. Exp. Immunol. 2006, 146, 109–115. [Google Scholar] [CrossRef]
- Xu, X.; Hicks, C.; Li, Y.; Su, J.; Shiloach, J.; Kaufman, J.B.; Fitz, Y.; Eichacker, P.Q.; Cui, X. Purified cell wall from the probiotic bacterium Lactobacillus gasseri activates systemic inflammation and, at higher doses, produces lethality in a rat model. Crit. Care. 2014, 18, R140. [Google Scholar] [CrossRef]
- Vong, L.; Lorentz, R.J.; Assa, A.; Glogauer, M.; Sherman, P.M. Probiotic Lactobacillus rhamnosus inhibits the formation of neutrophil extracellular traps. J. Immunol. 2014, 192, 1870–1877. [Google Scholar] [CrossRef]
- Frank, K.; Paust, S. Dynamic natural killer cell and T cell responses to influenza infection. Front. Cell. Infect. Microbiol. 2020, 10, 425. [Google Scholar] [CrossRef]
- Li, G.; Lin, J.; Zhang, C.; Gao, H.; Lu, H.; Gao, X.; Zhu, R.; Li, Z.; Li, M.; Liu, Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes 2021, 13, 1968257. [Google Scholar] [CrossRef]
- Roselli, M.; Finamore, A.; Britti, M.S.; Mengheri, E. Probiotic bacteria Bifidobacterium animalis MB5 and Lactobacillus rhamnosus GG protect intestinal Caco-2 cells from the inflammation-associated response induced by enterotoxigenic Escherichia coli K88. Br. J. Nutr. 2006, 95, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Raheem, A.; Liang, L.; Zhang, G.; Cui, S. Modulatory effects of probiotics during pathogenic infections with emphasis on immune regulation. Front. Immunol. 2021, 12, 616713. [Google Scholar] [CrossRef] [PubMed]
- De Moreno De Leblanc, A.; Perdigón, G. The application of probiotic fermented milks in cancer and intestinal inflammation. Proc. Nutr. Soc. 2010, 69, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, M.; Moridnia, A.; Mortazavi, D.; Salehi, M.; Bagheri, M.; Sheikhi, A. Kefir: A powerful probiotics with anticancer properties. Med. Oncol. 2017, 34, 183. [Google Scholar] [CrossRef]
- Mori, N.; Kano, M.; Masuoka, N.; Konno, T.; Suzuki, Y.; Miyazaki, K.; Ueki, Y. Effect of probiotic and prebiotic fermented milk on skin and intestinal conditions in healthy young female students. Biosci. Microbiota Food Health 2016, 35, 105–112. [Google Scholar] [CrossRef]
- Strzepa, A.; Gurski, C.J.; Dittel, L.J.; Szczepanik, M.; Pritchard Jr, K.A.; Dittel, B.N. Neutrophil-derived myeloperoxidase facilitates both the induction and elicitation phases of contact hypersensitivity. Front. Immunol. 2021, 11, 3552. [Google Scholar] [CrossRef]
- Cristofori, F.; Dargenio, V.N.; Dargenio, C.; Miniello, V.L.; Barone, M.; Francavilla, R. Anti-inflammatory and immunomodulatory effects of probiotics in gut inflammation: A door to the body. Front. Immunol. 2021, 12, 178. [Google Scholar] [CrossRef]
- Galdeano, C.M.; Cazorla, S.I.; Dumit, J.M.L.; Vélez, E.; Perdigón, G. Beneficial effects of probiotic consumption on the immune system. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar]
- Cordeiro, M.A.; Souza, E.L.S.; Arantes, R.M.E.; Balthazar, C.F.; Guimarães, J.T.; Scudino, H.; Silva, H.L.A.; Rocha, R.S.; Freitas, M.Q.; Esmerino, E.A.; et al. Fermented whey dairy beverage offers protection against Salmonella enterica ssp. enterica serovar Typhimurium infection in mice. Int. J. Dairy Sci. 2019, 102, 6756–6765. [Google Scholar] [CrossRef]
- Beh, B.K.; Mohamad, N.E.; Yeap, S.K.; Ky, H.; Boo, S.Y.; Chua, J.Y.H.; Tan, S.W.; Ho, W.Y.; Sharifuddin, S.A.; Long, K.; et al. Anti-obesity and anti-inflammatory effects of synthetic acetic acid vinegar and Nipa vinegar on high-fat-diet-induced obese mice. Sci. Rep. 2017, 7, 6664. [Google Scholar] [CrossRef]
- Hunsche, C.; Cruces, J.; De la Fuente, M. Improvement of redox state and functions of immune cells as well as of behavioral response in aged mice after two-week supplementation of fermented milk with probiotics. Curr. Microbiol. 2019, 76, 1278–1289. [Google Scholar] [CrossRef] [PubMed]
- Karaffová, V.; Mudroňová, D.; Mad’ar, M.; Hrčková, G.; Faixová, D.; Gancarčíková, S.; Ševčíková, Z.; Nemcová, R. Differences in Immune Response and Biochemical Parameters of Mice Fed by Kefir Milk and Lacticaseibacillus paracasei Isolated from the Kefir Grains. Microorganisms 2021, 9, 831. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Zhang, Q.; de Haan, B.J.; Zhang, H.; Faas, M.M.; de Vos, P. Identification of TLR2/TLR6 signalling lactic acid bacteria for supporting immune regulation. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hee, K.C.; Geun, K.H.; Yun, K.J.; Ra, K.N.; Jun, J.B.; Hye, J.J. Probiotic genomic DNA reduces the production of pro-inflammatory cytokine tumor necrosis factor-alpha. FEMS Microbiol Lett. 2012, 328, 13–19. [Google Scholar]
- Jia, G.; Liu, X.; Che, N.; Xia, Y.; Wang, G.; Xiong, Z.; Zhang, H.; Ai, L. Human-origin Lactobacillus salivarius AR809 protects against immunosuppression in S. aureus-induced pharyngitis via Akt-mediated NF-κB and autophagy signaling pathways. Food Funct. 2020, 11, 270–284. [Google Scholar] [CrossRef]
- Hoarau, C.; Lagaraine, C.; Martin, L.; Velge-Roussel, F.; Lebranchu, Y. Supernatant of Bifidobacterium breve induces dendritic cell maturation, activation, and survival through a Toll-like receptor 2 pathway. J. Allergy Clin. Immunol. 2006, 117, 696–702. [Google Scholar] [CrossRef]
- Gupta, T.; Kaur, H.; Kapila, S.; Kapila, R. Lactobacillus fermentum (MTCC-5898) alleviates Escherichia coli-induced inflammatory responses in intestinal epithelial cells by modulating immune genes and NF-κB signalling. J. Appl Microbiol. 2021, 131, 3008–3017. [Google Scholar] [CrossRef]
- Javanshir, N.; Hosseini, G.N.G.; Sadeghi, M.; Esmaeili, R.; Satarikia, F.; Ahmadian, G.; Allahyari, N. Evaluation of the Function of Probiotics, Emphasizing the Role of their Binding to the Intestinal Epithelium in the Stability and their Effects on the Immune System. Biol. Proced. Online. 2021, 23, 23. [Google Scholar]
- McGuckin, M.A.; Lindén, S.K.; Sutton, P.; Florin, T.H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011, 9, 265–278. [Google Scholar] [CrossRef]
- Kaur, H.; Ali, S.A. Probiotics and Gut Microbiota: Mechanistic Insights into Gut Immune Homeostasis through TLR Pathway Regulation. Food Funct. 2022, 13, 7423–7447. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Hemarajata, P.; Versalovic, J. Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Thrapeutic Adv. Gastroenterol. 2013, 6, 39–51. [Google Scholar] [CrossRef]
- Krishna Rao, R.; Samak, G. Protection and restitution of gut barrier by probiotics: Nutritional and clinical implications. Curr. Nutr. Food Sci. 2013, 9, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- Bonvicini, F.; Pagnotta, E.; Punzo, A.; Calabria, D.; Simoni, P.; Mirasoli, M.; Passerini, N.; Bertoni, S.; Ugolini, L.; Lazzeri, L.; et al. Effect of Lactobacillus acidophilus fermented broths enriched with Eruca sativa seed extracts on intestinal barrier and inflammation in a co-culture system of an enterohemorrhagic Escherichia coli and human intestinal cells. Nutrients 2020, 12, 3064. [Google Scholar] [CrossRef]
- Zhai, Z.; Torres-Fuentes, C.; Heeney, D.D.; Marco, M.L. Synergy between probiotic Lactobacillus casei and milk to maintain barrier integrity of intestinal epithelial cells. J. Agric. Food Chem. 2019, 67, 1955–1962. [Google Scholar] [CrossRef]
- Rokana, N.; Singh, R.; Mallappa, R.H.; Batish, V.K.; Grover, S. Modulation of intestinal barrier function to ameliorate Salmonella infection in mice by oral administration of fermented milks produced with Lactobacillus plantarum MTCC 5690–a probiotic strain of Indian gut origin. J. Med. Microbiol. 2016, 65, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.P.; Hsu, C.A.; Hung, W.T.; Chen, M.J. Effects of Lactobacillus paracasei 01 fermented milk beverage on protection of intestinal epithelial cell in vitro. J. Sci. Food Agric. 2016, 96, 2154–2160. [Google Scholar] [CrossRef]
- Vinderola, G.; Matar, C.; Perdigón, G. Milk fermented by Lactobacillus helveticus R389 and its non-bacterial fraction confer enhanced protection against Salmonella enteritidis serovar Typhimurium infection in mice. Immunobiology 2007, 212, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Usuda, H.; Okamoto, T.; Wada, K. Leaky gut: Effect of dietary fiber and fats on microbiome and intestinal barrier. Int. J. Mol. Sci. 2007, 22, 7613. [Google Scholar] [CrossRef]
- Bruno, C.; Paparo, L.; Pisapia, L.; Romano, A.; Cortese, M.; Punzo, E.; Canani, R. Protective effects of the postbiotic deriving from cow’s milk fermentation with L. paracasei CBA L74 against Rotavirus infection in human enterocytes. Sci. Rep. 2022, 12, 16268. [Google Scholar]
- Zhu, Y.; Xu, Y.; Wang, X.; Rao, L.; Yan, X.; Gao, R.; Shen, T.; Zhou, Y.; Kong, C.; Zhou, L. Probiotic Cocktail Alleviates Intestinal Inflammation Through Improving Gut Microbiota and Metabolites in Colitis Mice. Front. Cell. Infect. Microbiol. 2022, 760. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.S.; Corredig, M.; Morales-Rayas, R.; Hassan, A.; Griffiths, M.W.; LaPointe, G. Effect of fermented milk from Lactococcus lactis ssp. cremoris strain JFR1 on Salmonella invasion of intestinal epithelial cells. Int. J. Dairy Sci. 2019, 102, 6802–6819. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Han, D.; Wang, Y.; Wang, Q.; Yan, X.; Wang, S.; Liu, X.; Song, W.; Ma, Y. Lactobacillus casei protects intestinal mucosa from damage in chicks caused by Salmonella pullorum via regulating immunity and the Wnt signaling pathway and maintaining the abundance of gut microbiota. Poult. Sci. 2021, 100, 101283. [Google Scholar] [CrossRef]
- Liu, Q.; Yu, Z.; Tian, F.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Surface components and metabolites of probiotics for regulation of intestinal epithelial barrier. Microb. Cell Factories 2020, 19, 23. [Google Scholar] [CrossRef]
- Leeuwendaal, N.K.; Stanton, C.; O’Toole, P.W.; Beresford, T.P. Fermented Foods, Health and the Gut Microbiome. Nutrients 2022, 14, 1527. [Google Scholar] [CrossRef]
- Ghosh, S.; Whitley, C.S.; Haribabu, B.; Jala, V.R. Regulation of intestinal barrier function by microbial metabolites. Cell Mol. Gastroenterol. Hepatol. 2021, 11, 1463–1482. [Google Scholar] [CrossRef]


| Food Matrices | Microorganism | Health-Promoting Compounds | Disease | Reference |
|---|---|---|---|---|
| Kimchi | Lactobacillus plantarum (L. plantarum) | Several prebiotics and the presence of vitamins, minerals, and dietary fibers and O-linked β-N-acetylglucosamine (O-GlcNAc) | Colon cancer | [15] |
| Whey | L.rhamnosus MTCC 5897 | Probiotic released metabolites | Ulcerative colitis | [16] |
| Milk | Bifdobacterium breve | Bacteria and released components | Ulcerative colitis | [17] |
| Milk | L. rhamnosus NCDC 17 and L. rhamnosus GG | Metabolites | STZ-induced diabetic Wistar rats in high-fat diet (T2DM) | [18] |
| Koumiss | L. helveticus NS8 | Bacterial surface components | Gastrointestinal health | [19] |
| Skimmed milk | Lactococcus lactis strain NRRL B-50571 | ACE 3 inhibitory peptides and GABA | Anti-hypertensive activity | [20] |
| Milk, yogurt | LAB and Bifidobacteria species | Folate (vitamin B9), vitamin K, riboflavin (vitamin B2) | Vitamin deficiency | [21,22] |
| Cashew apple juice, kimchi | 5 probiotic strains (Lb. acidophilus, Lb. casei, Lb. plantarum, Lb. mesenteroides and B. longum) | Vitamin K (Phylloquinone) | Vitamin deficiency | [23,24] |
| Milk | L. casei DN-114 00 | - | Common infectious diseases (CIDs) | [25] |
| Milk | L. acidophilus and L. casei | - | Alleviation of lactose intolerance | [26] |
| Milk | L. helveticus LH511 | Bioactive components in supernatant | Barrier dysfunction | [27] |
| Shrikhand | L. acidophilus NCDC14 | - | Gastrointestinal health | [28] |
| Kefir | Lactobacillus lactis subs., Leuconostoc subs., Streptococcus thermophilus, Lactobacillus subs., and yeast of kefir | - | Inflammatory bowel disease (IBD) | [29] |
| Milk | L. paracasei CNCM I 1518 | - | Cirrhosis | [30] |
| Milk | L. fermentum NCDC 400 and L. rhamnosus NCDC 610 | - | Obesity and other related metabolic syndromes | [31] |
| Milk | L. rhamnosus MTCC 5897 | - | Hypercholestrolemia | [32] |
| Milk | L. acidophilus, L. amylovorus, L. bravis, L. bulgaricus, L. casei, L. curvatus, L. fermentum, L. gasseri, L. helveticus, L. lactis, L. paracasei, L. plantarum, L. rhamnosus, L. reuteri, L. salivarius, B. lactis, B. breve, B. bifidum, B. longum, B. infantis, Pediococcus pentosaceus, and Streptococcus thermophilus. | - | Cardiometabolic diseases (CMDs) | [33] |
| Yogurt, capsules | L. rhamnosus GG (ATCC 53013) | Long chain fatty acids | Acute pediatric diarrhea, antibiotic-associated diarrhea | [34] |
| Squacquerone cheese | L. crispatus BC4 | Calcium, vitamins with antimicrobial properties | Vaginal infections | [35] |
| Cream cheese | L. chungangensis CAU28 | Short chain fatty acid | Atopic dermatitis | [36] |
| Cheddar cheese | Lacticaseibacillus rhamnosus and Lc. paracasei | γ aminobutyric acid (GABA) | Gastrointestinal digestion | [37] |
| Milk | B. longum subsp. longum YS108R; L. paracasei PS23 | Exoplysaacharide (EPS); short chain fatty acids (SCFA) | Ulcerative colitis | [38,39] |
| Milk | Lactobacillus spp. | Free amino acids and lactate | Bone health | [40] |
| Milk | L. brevis DL1-11 | γ aminobutyric acid (GABA), short-chain fatty acids (SCFAs), such as butyric acid | Insomnia | [41] |
| Milk | L. acidophilus | Phenolic compounds, GABA 1, peptides, CLA 2, folates (vitamin B9) | Antioxidant activity related disorders | [42] |
| Formulation | Probiotic Strain | Associated Mechanism | Disorder | Model Organism | Reference |
|---|---|---|---|---|---|
| Kefir drink | Commercial kefir starter culture (Danisco®) | Fermented kefir ameliorates the colitis-induced symptoms in dose-dependent manner, relieves from disease-induced diarrhea and macroscopic damages caused in mucosal wall | Inflammatory bowel disease (IBD) | Rat | [29] |
| Whey drink | L. rhamnosus MTCC 5897 | Increases the anti-inflammatory cytokines to ameliorate the colitis-induced inflammation, enhances the tight junctional epithelial barrier integrity in mice | Ulcerative colitis | Albino mice | [16] |
| Fermented milk | B. longum subsp. longum YS108R | Reduces inflammatory cytokine concentrations in serum as well as their mRNA expression levels in the colon by increasing tight junction protein and MUC2 expression | DSS-induced colitis | C57BL/6J mice | [38] |
| Fermented milk | L. rhamnosus MTCC: 5957 and L. rhamnosus MTCC: 5897 | Reduces the high-cholesterol diet (HCD)-induced body weight, hyperlipidemia, and hepatic lipids (total cholesterol and triacylglycerol), decreases the TNF-alpha, IL-6 in the liver | Diet-induced hypercholesterolemia | Wistar rats | [32] |
| Fermented milk | L.paracasei PS23 | Strengthened tight junction through the modification of specific cecal bacteria and upregulation of short-chain fatty acids | DSS-induced colitis | Mice | [39] |
| Fermented milk | L. fermentum | Bioavailability of zinc | Zinc depletion | Wistar rats | [53] |
| Milk; fermented milk | L. fermentum | Improvement in the hypercholesterolemic effect | Hypercholesterolemic | Wistar rats | [54] |
| Cream cheese | L. chungangensis CAU28 | Maintains Th1 and Th2 balance as well as reduces IgE immunoglobulin associated with immune homeostasis | Atopic dermatitis | Balb/c mice | [36] |
| Fermented milk | L. paracasei CNCM I 1518 | Decreases bacterial translocation, gut dysbiosis, and ileal oxidative damage and increases ileal β-defensin-1 expression in rats treated with CCl4 | Cirrhosis | Sprague-Dawley rats | [55] |
| Fermented milk | Lactobacillus spp. | Improves amino acid absorption that in turn enhances postprandial MPS via Akt-independent mechanisms | Skeletal muscle protein anabolism | Sprague-Dawley rats | [40] |
| Yogurt and fermented milk | FMP: Lactobacillus helveticus; -Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus | Greek-style yogurt protein (YP) increased the expression of genes involved in jejunal and ileal immunity and integrity in WT mice; as well as YP also improved insulin sensitivity by 65% in LRKO mice, Lactobacillus helveticus fermented milk protein (FMP) and YP both modulate the intestinal microbiota | Cardiometabolic markers and intestinal microbiota | C57BL/6J wild-type (WT) and atherosclerotic (LRKO) male mice | [56] |
| Fermented milk | Lacticaseibacillus casei CRL431 | Probiotic-fermented milk (PFM) had the highest percentages for CD4+ and CD4+ CD8+ cells. While IL-10, TNF-α, IFN-γ, and IL-6 concentrations decreased significantly in the PFM group that increased in mice from the milk group | Breast cancer | Female BALB/c mice | [57] |
| Fermented milk | Lactobacillus brevis DL1-11 | Modulates the composition of the gut microbiota, such as significant increases in the relative abundances of Ruminococcus, Adlercreutzia, and Allobaculum. Moreover, these changes may be associated with significant increases in the production of SCFAs in the intestine | Insomnia | Rat | [41] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaur, H.; Kaur, G.; Ali, S.A. Dairy-Based Probiotic-Fermented Functional Foods: An Update on Their Health-Promoting Properties. Fermentation 2022, 8, 425. https://doi.org/10.3390/fermentation8090425
Kaur H, Kaur G, Ali SA. Dairy-Based Probiotic-Fermented Functional Foods: An Update on Their Health-Promoting Properties. Fermentation. 2022; 8(9):425. https://doi.org/10.3390/fermentation8090425
Chicago/Turabian StyleKaur, Harpreet, Gurjeet Kaur, and Syed Azmal Ali. 2022. "Dairy-Based Probiotic-Fermented Functional Foods: An Update on Their Health-Promoting Properties" Fermentation 8, no. 9: 425. https://doi.org/10.3390/fermentation8090425
APA StyleKaur, H., Kaur, G., & Ali, S. A. (2022). Dairy-Based Probiotic-Fermented Functional Foods: An Update on Their Health-Promoting Properties. Fermentation, 8(9), 425. https://doi.org/10.3390/fermentation8090425






