The Formation of Aroma Compounds During Fermentation in Relation to Yeast Nutrient Source in Sauvignon Blanc Wine
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sauvignon Blanc Grapes
2.3. Fermentation and Yeast Nutrient Additions
2.4. Basic Analyses
2.5. Analysis of Glutathione, Varietal Thiols, Esters, Higher Alcohols, Other Volatile Compounds, and Yeast Assimilable Nitrogen (YAN)
2.5.1. Glutathione and Oxidized Glutathione
2.5.2. Varietal Thiols
2.5.3. Analysis of Esters, C6 Compounds, Aldehydes, and Lactones
2.5.4. Higher Alcohols
2.5.5. Yeast Assimilable Nitrogen Analysis
2.6. Sensory Analysis
2.7. Statistical Analysis
3. Results and Discussion
3.1. Fermentation and Basic Wine Parameters
3.2. Yeast Assimilable Nitrogen
3.3. Glutathione
3.4. Influence of Yeast Nutrients on the Aromatic Composition of Wine
3.4.1. Effect on Varietal Thiols
3.4.2. Effects on Esters, Higher Alcohols, and Other Volatile Compounds
Effect on Ethyl Esters
Effect on Acetate Esters
Effect on Higher Alcohols and Other Volatile Compounds
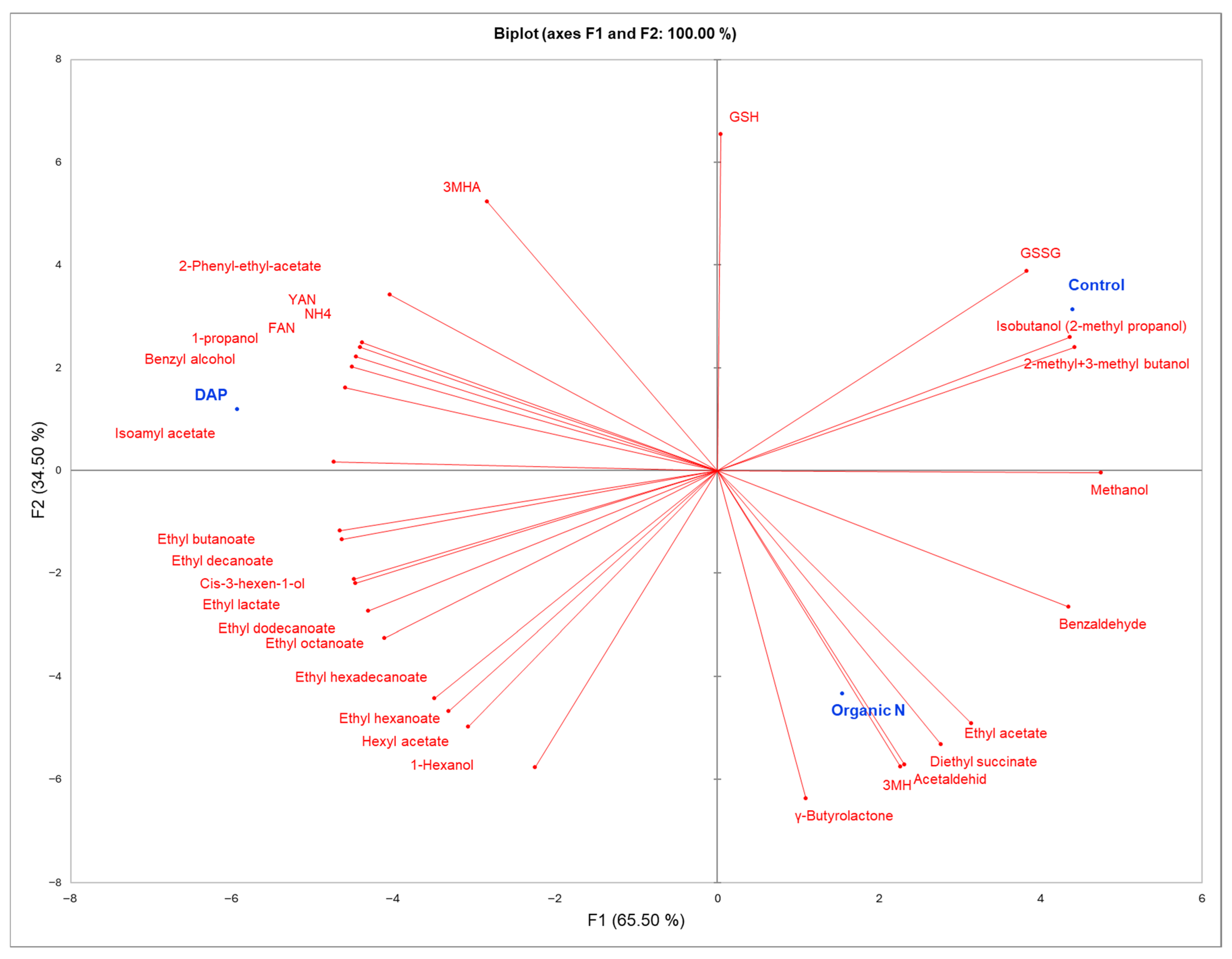
3.4.3. Principal Component Analysis (PCA)
3.5. Sensory Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gobert, A.; Tourdot-Maréchal, R.; Sparrow, C.; Morge, C.; Alexandre, H. Influence of nitrogen status in wine alcoholic fermentation. Food Microbiol. 2019, 83, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Sablayrolles, J.M.; Barre, P. Kinetics of alcoholic fermentation under anisothermal enological conditions. I. Influence of temperature evolution on the instantaneous rate of fermentation. Am. J. Enol. Vitic. 1993, 44, 127–133. [Google Scholar] [CrossRef]
- Bell, S.J.; Henschke, P.A. Implications of nitrogen nutrition for grape fermentation and wine. Aust. J. Grape Wine Res. 2005, 11, 242–295. [Google Scholar] [CrossRef]
- Bisson, L. Yeast and biochemistry of ethanol formation. In Principles and Practices of Winemaking; Boulton, R.B., Singleton, V.L., Bisson, L.F., Kunkee, R.E., Eds.; Chapman & Hall: New York, NY, USA, 1996; pp. 140–172. [Google Scholar]
- Bisson, L.F.; Butzke, C.E. Diagnosis and rectification of stuck and sluggish fermentations. Am. J. Enol. Vitic. 2000, 51, 168–177. [Google Scholar] [CrossRef]
- Wang, X.D.; Bohlscheid, J.C.; Edward, C.G. Fermentative activity and production of volatile compounds by Saccharomyces grown in synthetic grape juice media deficient in assimilable nitrogen and/or pantothenic acid. J. Appl. Microbiol. 2003, 94, 349–359. [Google Scholar] [CrossRef]
- Beltran, G.; Esteve, B.; Zarzoso, I.; Rozes, N.; Mas, A.; Guillamón, J.M. Influence of the timing of nitrogen additions during synthetic grape must fermentations on fermentation kinetics and nitrogen consumption. J. Agric. Food Chem. 2005, 53, 996–1002. [Google Scholar] [CrossRef]
- Barnett, J.A. A history of research on yeasts 13: Active transport and the uptake of various metabolites. Yeast 2008, 25, 689–731. [Google Scholar] [CrossRef]
- Scrimgeour, N.; Wilkes, E.; Bartowsky, E. Making sense of yeast assimilable nitrogen (YAN). Aust. N. Z. Grapegrow. Winemak. 2019, 47–50. [Google Scholar]
- Bouloumpasi, E.; Skendi, A.; Soufleros, E.H. Survey on yeast assimilable nitrogen status of musts from native and international grape varieties: Effect of variety and climate. Fermentation 2023, 9, 773. [Google Scholar] [CrossRef]
- Lambrechts, M.-G.; Pretorius, I.-S. Yeast and its importance to wine aroma: A review. S. Afr. J. Enol. Vitic. 2000, 21, 97–129. [Google Scholar] [CrossRef]
- Ugliano, M.; Fredrizzi, B.; Siebert, T.; Travis, B.; Magno, F.; Versini, G.; Henschke, P.A. Effect of nitrogen supplementation and Saccharomyces species on hydrogen sulfide and other volatile sulfur compounds in Shiraz fermentation and wine. J. Agric. Food Chem. 2009, 57, 4948–4955. [Google Scholar] [CrossRef] [PubMed]
- Cramer, A.C.; Vlassides, S.; Block, D.E. Kinetic model for nitrogen-limited wine fermentations. Biotechnol. Bioeng. 2002, 77, 49–60. [Google Scholar] [CrossRef]
- Taillandier, P.; Ramon Portugal, F.; Fuster, A.; Strehaiano, P. Effect of ammonium concentration on alcoholic fermentation kinetics by wine yeasts for high sugar content. Food Microbiol. 2007, 24, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Ochando, T.; Mouret, J.R.; Humbert-Goffard, A.; Sablayrolles, J.M.; Farines, V. Impact of initial lipid content and oxygen supply on alcoholic fermentation in champagne-like musts. Food Res. Int. 2017, 98, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Santamaría, P.; González-Arenzana, L.; Garijo, P.; Gutiérrez, A.R.; López, R. Nitrogen sources added to must: Effect on fermentations and on the Tempranillo red wine quality. Fermentation 2020, 6, 79. [Google Scholar] [CrossRef]
- Seguinot, P.; Rollero, S.; Sanchez, I.; Sablayrolles, J.-M.; Ortiz-Julien, A.; Camarasa, C.; Mouret, J.-R. Impact of the timing and the nature of nitrogen additions on the production kinetics of fermentative aromas by Saccharomyces cerevisiae during winemaking fermentation in synthetic media. Food Microbiol. 2018, 76, 29–39. [Google Scholar] [CrossRef]
- Barbosa, C.; Mendes-Faia, A.; Mendes-Ferreira, A. The nitrogen source impacts major volatile compounds released by Saccharomyces cerevisiae during alcoholic fermentation. Int. J. Food Microbiol. 2012, 160, 87–93. [Google Scholar] [CrossRef]
- Mendes-Ferreira, A.; Mendes-Faia, A.; Leao, C. Growth and fermentation patterns of Saccharomyces cerevisiae under different ammonium concentrations and its implication in wine-making industry. J. Appl. Microbiol. 2004, 97, 540–545. [Google Scholar] [CrossRef]
- Tominaga, T.; Furrer, A.; Henry, R.; Dubourdieu, D. Identification of new volatile thiols in the aroma of Vitis vinifera L. var. Sauvignon blanc wines. Flavour Fragr. J. 1998, 13, 159–162. [Google Scholar] [CrossRef]
- Murat, M.; Masneuf, I.; Darriet, P.; Lavigne, V.; Tominaga, T.; Dubourdieu, D. Effect of Saccharomyces cerevisiae yeast strains on the liberation of volatile thiols in Sauvignon blanc wine. Am. J. Enol. Vitic. 2001, 52, 136–139. [Google Scholar] [CrossRef]
- Lu, W.-J.; Li, H.-K.; Liu, C.-Y.; Lin, T.-Z.; Chang, C.-F.; Yang, T.-Y.; Hsu, P.-H.; Lin, V.H.T. Enzymatic production of aroma compound 3-mercapto-1-hexanol enantiomers via cysteine-S-conjugate β-lyase. LWT 2024, 205, 115–123. [Google Scholar] [CrossRef]
- Winter, G.; Van Der Westhuizen, T.; Higgins, V.; Curtin, C.; Ugliano, M. Contribution of cysteine and glutathione conjugates to the formation of the volatile thiols 3-mercaptohexan-1-ol (3MH) and 3-mercaptohexyl acetate (3MHA) during fermentation by Saccharomyces cerevisiae. Aust. J. Grape Wine Res. 2011, 17, 285–290. [Google Scholar] [CrossRef]
- Harsch, M.J.; Richard, C.; Gardner, R. Yeast genes involved in sulfur and nitrogen metabolism affect the production of volatile thiols from Sauvignon Blanc musts. Appl. Microbiol. Biotechnol. 2013, 97, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Subileau, M.; Schneider, R.; Salmon, J.M.; Degryse, E. Nitrogen catabolite repression modulates the production of aromatic thiols characteristic of Sauvignon Blanc at the level of precursor transport. FEMS Yeast Res. 2008, 8, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Thibon, C.; Marullo, P.; Claisse, O.; Cullin, C.; Dubourdieu, D.; Tominaga, T. Nitrogen catabolic repression controls the release of volatile thiols by Saccharomyces cerevisiae during wine fermentation. FEMS Yeast Res. 2008, 8, 1076–1086. [Google Scholar] [CrossRef]
- Pinu, F.R.; Edwards, P.J.B.; Jouanneau, S.; Kilmartin, P.A.; Gardner, R.C.; Villas-Boas, S.G. Sauvignon blanc metabolomics: Grape juice metabolites affecting the development of varietal thiols and other aroma compounds in wine. Metabolomics 2013, 10, 556–573. [Google Scholar] [CrossRef]
- Deroite, A.; Legras, J.-L.; Rigou, P.; Ortiz-Julien, A.; Dequin, S. Lipids modulate acetic acid and thiol final concentrations in wine during fermentation by Saccharomyces cerevisiae × Saccharomyces kudriavzevii hybrids. AMB Express 2018, 8, 130. [Google Scholar] [CrossRef]
- Binati, L.R.; Larini, I.; Salvetti, E.; Torriani, S. Glutathione production by non-Saccharomyces yeasts and its impact on winemaking: A review. Food Res. Int. 2022, 156, 111–123. [Google Scholar] [CrossRef]
- Herbst, M.; Kilmartin, P.A.; Nicolau, L. Copper(II)-catalysed oxidation of phenolic compounds in model wine. In Proceedings of the Sixth International Cool Climate Symposium for Viticulture and Oenology; Creasy, G.L., Steans, G.F., Eds.; New Zealand Society for Viticulture and Oenology: Christchurch, New Zealand, 2006. [Google Scholar]
- Singleton, V.L.; Salgues, M.; Zaya, J.; Trousdale, E. Caftaric acid disappearance and conversion to products of enzymic oxidation in grape must and wine. Am. J. Enol. Vitic. 1985, 36, 50–56. [Google Scholar] [CrossRef]
- Kritzinger, E.C.; Stander, M.A.; Du Toit, W.J. Assessment of glutathione levels in model solution and grape ferments supplemented with glutathione-enriched inactive dry yeast preparations using a novel UPLC-MS/MS method. Food Addit. Contam. A 2013, 30, 80–92. [Google Scholar]
- Dragojlović, D.; Philipp, C.; Korntheuer, K.; Bader, C.; Patzl-Fischerleitner, E.; Eder, R. Mitteilungen Klosterneuburg. Acta Vitic. Vinic. 2018, 68, 250–257. [Google Scholar]
- OIV. Compendium of International Methods of Wine and Must Analysis; OIV-MA-AS313-27:2019 (Acetic Acid) and OIV-MA-AS311-10:R2018 (Glucose + Fructose); OIV: Dijon, France, 2021. [Google Scholar]
- Commission Regulation (EEC). Commission Regulation (EEC) No 2676/90 of 17 September 1990 determining Community methods for the analysis of wines. Off. J. Eur. Union L 1990, 272, 1–192. Available online: http://data.europa.eu/eli/reg/1990/2676/oj/eng (accessed on 1 January 2020).
- Vanzo, A.; Janeš, L.; Požgan, F.; Velikonja Bolta, Š.; Sivilotti, P.; Lisjak, K. UHPLC-MS/MS determination of varietal thiol precursors in Sauvignon blanc grapes. Sci. Rep. 2017, 7, 13122. [Google Scholar] [CrossRef]
- Šuklje, K.; Baša Česnik, H.; Janeš, L.; Kmecl, V.; Vanzo, A.; Deloire, A.; Sivilotti, P.; Lisjak, K. The effect of leaf area to yield ratio on secondary metabolites in grapes and wines of Vitis vinifera L. cv. Sauvignon Blanc. OENO One 2013, 47, 83–97. [Google Scholar] [CrossRef]
- Tominaga, T.; Dubourdieu, D. A novel method for quantification of 2-methyl-3-furanthiol and 2-furanmethanethiol in wines made from Vitis vinifera grape varieties. J. Agric. Food Chem. 2006, 54, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Bavčar, D.; Baša Česnik, H. Validation of the method for the determination of some wine volatile compounds. Acta Agric. Slov. 2011, 97, 285–293. [Google Scholar] [CrossRef]
- Bavčar, D.; Baša Česnik, H.; Čuš, F.; Košmerl, T. The influence of skin contact during alcoholic fermentation on the aroma composition of Ribolla Gialla and Malvasia Istriana Vitis vinifera (L.) grape wines. Int. J. Food Sci. Technol. 2011, 46, 1801–1808. [Google Scholar] [CrossRef]
- OIV. Compendium of International Methods of Wine and Must Analysis; OIV-MA-AS312-01 (Method for Wine Distillate); OIV: Dijon, France, 2021. [Google Scholar]
- Dukes, B.C.; Butzke, C.E. Rapid determination of primary amino acids in grape juice using an o-phthaldialdehyde/N-acetyl-L-cysteine spectrophotometric assay. Am. J. Enol. Vitic. 1998, 49, 125–133. [Google Scholar] [CrossRef]
- Zoecklein, B.W.; Fugelsang, K.C.; Gump, B.H.; Nury, F.S. Wine Analysis and Production, 1st ed.; Chapman & Hall: Gaithersburg, MD, USA, 1999. [Google Scholar]
- Zoecklein, B.W.; Gump, B.H.; Fugelsang, K.C. Nutritional status of grape juice. In Methods in Biotechnology; Humana Press: Totowa, NJ, USA, 2002; pp. 413–423. [Google Scholar]
- Hernández-Orte, P.; Bely, M.; Cacho, J.; Ferreira, V. Impact of ammonium additions on volatile acidity, ethanol, and aromatic compound production by different Saccharomyces cerevisiae strains during fermentation in controlled synthetic media. Aust. J. Grape Wine Res. 2006, 12, 150–160. [Google Scholar] [CrossRef]
- Tronchoni, J.; Gamero, A.; Arroyo-López, F.N.; Barrio, E.; Querol, A. Differences in the glucose and fructose consumption profiles in diverse Saccharomyces wine species and their hybrids during grape juice fermentation. Int. J. Food Microbiol. 2009, 134, 237–243. [Google Scholar] [CrossRef]
- Blateyron, L.; Sablayrolles, J.M. Stuck and slow fermentations in enology: Statistical study of causes and effectiveness of combined additions of oxygen and diammonium phosphate. J. Biosci. Bioeng. 2001, 91, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Christofi, S.; Papanikolaou, S.; Dimopoulou, M.; Terpou, A.; Cioroiu, I.B.; Cotea, V.; Kallithraka, S. Effect of yeast assimilable nitrogen content on fermentation kinetics, wine chemical composition and sensory character in the production of Assyrtiko wines. Appl. Sci. 2022, 12, 1405. [Google Scholar] [CrossRef]
- Binati, L.R.; Lemos, W.J.F.; Torriani, S. Contribution of non-Saccharomyces yeasts to increase glutathione concentration in wine. Aust. J. Grape Wine Res. 2021, 27, 290–294. [Google Scholar] [CrossRef]
- Bekker, M.-Z.; Cuijvers, K.-M.; Kulcsar, A.-C.; Sanders, R.-D.; Capone, D.-L.; Jeffery, D.-W.; Schmidt, S.-A. Effects of yeast strain and juice nitrogen status on glutathione utilisation during fermentation of model media. Aust. J. Grape Wine Res. 2023, 2023, 8041096. [Google Scholar] [CrossRef]
- Schneider, R.; Charrier, F.; Razungles, A.; Baumes, R. Evidence for an alternative biogenetic pathway leading to 3-mercaptohexanol and 4-mercapto-4-methylpentan-2-one in wines. Anal. Chim. Acta 2006, 563, 58–64. [Google Scholar] [CrossRef]
- Swiegers, J.H.; Willmott, R.; Hill-Ling, A.; Capone, D.L.; Pardon, K.H.; Elsey, G.M.; Howell, K.S.; de Barros Lopes, M.A.; Sefton, M.A.; Lilly, M.; et al. Modulation of volatile thiol and ester aromas by modified wine yeast. In Flavour Science: Recent Advances and Trends; Elsevier: Amsterdam, The Netherlands, 2006; Volume 43, pp. 123–142. [Google Scholar]
- Verstrepen, K.-J.; Van Laere, S.-D.; Vanderhaegen, B.-M.; Derdelinckx, G.; Dufour, J.-P.; Pretorius, I.S.; Winderickx, J.; Thevelein, J.M.; Delvaux, F.R. Expression levels of the yeast alcohol acetyltransferase genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of volatile esters. Appl. Environ. Microbiol. 2003, 69, 5228–5237. [Google Scholar] [CrossRef]
- Yoshimoto, H.; Fukushige, T.; Yonezawa, T.; Sone, H. Genetic and physiological analysis of branched-chain alcohols and isoamyl acetate production in Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2002, 59, 501–508. [Google Scholar] [CrossRef]
- Duc, C.; Maçna, F.; Sanchez, I.; Galeote, V.; Delpech, S.; Silvano, A.; Mouret, J.-R. Large-scale screening of thiol and fermentative aroma production during wine alcoholic fermentation: Exploring the effects of assimilable nitrogen and peptides. Fermentation 2020, 6, 98. [Google Scholar] [CrossRef]
- Waterhouse, A.; Sacks, G.L.; Jeffery, D.W. Understanding Wine Chemistry; Wiley: Hoboken, NJ, USA, 2016. [Google Scholar]
- Swiegers, J.-H.; Pretorius, I.-S. Yeast modulation of wine flavor. Adv. Appl. Microbiol. 2005, 57, 131–175. [Google Scholar]
- Saerens, S.M.G.; Delvaux, F.R.; Verstrepen, K.J.; Van Dijck, P.; Thevelein, J.M.; Delvaux, F.R. Parameters affecting ethyl ester production by Saccharomyces cerevisiae during fermentation. Appl. Environ. Microbiol. 2008, 74, 454–461. [Google Scholar] [CrossRef]
- Mouret, J.-R.; Perez, M.; Angenieux, M.; Nicolle, P.; Farines, V.; Sablayrolles, J.-M. Online-based kinetic analysis of higher alcohol and ester synthesis during winemaking fermentations. Food Bioproc. Technol. 2014, 7, 1235–1245. [Google Scholar] [CrossRef]
- Torrea, D.; Varela, C.; Ugliano, M.; Ancin-Azpilicueta, C.; Francis, L.I.; Henschke, P.-A. Comparison of inorganic and organic nitrogen supplementation of grape juice: Effect on volatile composition and aroma profile of a Chardonnay wine fermented with Saccharomyces cerevisiae yeast. Food Chem. 2011, 127, 1072–1083. [Google Scholar] [CrossRef] [PubMed]
- Vilanova, M.; Siebert, T.E.; Varela, C.; Pretorius, I.S.; Henschke, P.A. Effect of ammonium nitrogen supplementation of grape juice on wine volatiles and non-volatiles composition of the aromatic grape variety Albariño. Food Chem. 2012, 133, 124–131. [Google Scholar] [CrossRef]
- Godillot, J.; Sanchez, I.; Perez, M.; Picou, C.; Galeote, V.; Sablayrolles, J.-M.; Farines, V.; Mouret, J.-R. The timing of nitrogen addition impacts yeast gene expression and the production of aroma compounds during wine fermentation. Front. Microbiol. 2022, 13, 829786. [Google Scholar] [CrossRef]
- Rollero, S.; Bloem, A.; Camarasa, C.; Sanchez, I.; Ortiz-Julien, A.; Sablayrolles, J.-M.; Dequin, S.; Mouret, J.-R. Combined effects of nutrients and temperature on the production of fermentative aromas by Saccharomyces cerevisiae during wine fermentation. Appl. Microbiol. Biotechnol. 2014, 99, 2291–2304. [Google Scholar] [CrossRef]
- Saerens, S.M.; Delvaux, F.R.; Verstrepen, K.J.; Thevelein, J.M. Production and biological function of volatile esters in Saccharomyces cerevisiae. Microb. Biotechnol. 2010, 3, 165–177. [Google Scholar] [CrossRef]
- Garde-Cerdán, T.; Ancín-Azpilicueta, C. Effect of the addition of different quantities of amino acids to nitrogen-deficient must on the formation of esters, alcohols, and acids during wine alcoholic fermentation. LWT 2008, 41, 505–510. [Google Scholar] [CrossRef]
- Amerine, M.-A.; Roessler, E.B. Wines: Their Sensory Evaluation; Freeman: New York, NY, USA, 1983. [Google Scholar]
- Rapp, A.; Mandery, H. Wine aroma. Experientia 1986, 42, 873–884. [Google Scholar] [CrossRef]
- Boulton, R.B.; Singleton, V.L.; Bisson, L.F.; Kunkee, R.E. Yeast and biochemistry of ethanol fermentation. In Principles and Practices of Winemaking; Boulton, R.B., Ed.; Aspen Publishers: Gaithersburg, MD, USA, 1996; pp. 139–172. [Google Scholar]
- Benkwitz, F.; Nicolau, L.; Lund, C.; Beresford, M.; Wohlers, M.; Kilmartin, P.A. Evaluation of key odorants in Sauvignon blanc wines using three different methodologies. J. Agric. Food Chem. 2012, 60, 6293–6302. [Google Scholar] [CrossRef]
- Rapp, A.; Versini, G. Influence of nitrogen compounds in grapes on aroma compounds of wines. Dev. Food Sci. 1991, 37, 1659–1694. [Google Scholar]
- Hernández-Orte, P.; Ibarz, M.; Cacho, J.; Ferreira, V. Effect of the addition of ammonium and amino acids to musts of Airén variety on aromatic composition and sensory properties of the obtained wine. Food Chem. 2005, 89, 163–174. [Google Scholar] [CrossRef]
- Gobbi, M.; Comitini, F.; D’Ignazi, G.; Ciani, M. Effects of nutrient supplementation on fermentation kinetics, H2S evolution, and aroma profile in Verdicchio DOC wine production. Eur. Food Res. Technol. 2013, 236, 145–154. [Google Scholar] [CrossRef]

| Days of Fermentation | Day 3 | Day 5 | Day 7 | Day 10 | Day 13 | Day 18 |
|---|---|---|---|---|---|---|
| Nutrient additions (mg/L) | ||||||
| Control | 0 | 0 | 0 | – | 0 | – |
| DAP | +200 | +50 | +50 | – | +30 | – |
| Organic N | +200 | +200 | +200 | – | +125 | – |
| Sampling for secondary metabolites | ✔ | – | ✔ | ✔ | – | ✔ |
| Stage of Fermentation | NH4+ (mg N/L) | FAN (mg N/L) | ||||
|---|---|---|---|---|---|---|
| Control | DAP | Organic N | Control | DAP | Organic N | |
| Early stage | 25.7 ± 7.1 a | 60.3 ± 12.7 b | 26.0 ± 5.6 a | 68.3 ± 13.9 a | 66.3 ± 4.9 a | 60.3 ± 12.7 a |
| Mid stage | 0.7 ± 0.6 a | 0.7 ± 0.6 a | 1.3 ± 0.6 a | 49.3 ± 36.9 a | 27.7 ± 5.7 a | 20.7 ± 1.2 a |
| Late stage | 1.0 ± 0.0 a | 12.3 ± 0.6 b | 1.0 ± 0.0 a | 16.7 ± 1.2 a | 25.3 ± 2.5 b | 20.3 ± 2.5 ab |
| After fermentation | 2.0 ± 0.0 b | 8.7 ± 0.6 c | 1.0 ± 0.0 a | 31.7 ± 0.6 a | 35.3 ± 0.6 b | 31.3 ± 1.2 c |
| Compound | Treatment | Early Stage | Mid Stage | Late Stage | Young Wine |
|---|---|---|---|---|---|
| GSH (mg/L) | Control | 5.3 ± 1.1 a | 5.9 ± 0.2 a | 3.4 ± 0.1 b | 1.8 ± 0.1 a |
| DAP | 5.8 ± 0.5 a | 6.8 ± 0.6 b | 3.3 ± 0.1 b | 1.2 ± 0.1 a | |
| Organic N | 4.7 ± 0.7 a | 5.4 ± 0.2 a | 2.7 ± 0.3 a | 1.4 ± 0.1 a | |
| GSSG (mg/L) | Control | 0.9 ± 0.0 a | 0.9 ± 0.0 a | 0.9 ± 0.0 a | 0.7 ± 0.0 a |
| DAP | 0.9 ± 0.0 a | 0.9 ± 0.0 a | 1.0 ± 0.1 a | 0.7 ± 0.0 a | |
| Organic N | 0.9 ± 0.0 a | 0.9 ± 0.0 a | 0.9 ± 0.0 a | 0.7 ± 0.0 a |
| Compound | Treatment | Early Stage | Mid Stage | Late Stage | Young Wine |
|---|---|---|---|---|---|
| 4MSP (ng/L) | Control | 14.3 ± 0.5 a | 14.2 ± 2.5 a | 13.6 ± 0.7 b | 12.9 ± 0.2 a |
| DAP | 16.5 ± 2.6 ab | 13.1 ± 2.0 a | 11.2 ± 1.4 a | 14.7 ± 0.2 a | |
| Organic N | 21.4 ± 4.9 b | 16.0 ± 1.9 a | 11.9 ± 1.2 ab | 20.1 ± 1.5 b | |
| 3SH (ng/L) | Control | 429 ± 16.5 a | 306 ± 54.5 a | 445 ± 59.6 b | 530 ± 34.6 a |
| DAP | 370 ± 59 a | 297 ± 13.1 a | 265 ± 43.9 a | 491 ± 80.8 a | |
| Organic N | 517 ± 126.3 a | 353 ± 9.3 a | 587 ± 6.7 c | 647 ± 1.4 b | |
| 3SHA (ng/L) | Control | 32 ± 3.7 a | 33 ± 3.0 a | 43 ± 2.5 b | 47 ± 5.5 a |
| DAP | 31 ± 0.4 a | 33 ± 2.5 a | 38 ± 1.5 ab | 49 ± 3.7 a | |
| Organic N | 32 ± 2.6 a | 31 ± 0.8 a | 35 ± 2.9 a | 43 ± 7.0 a |
| Compound | Treatment | Early Stage | Mid Stage | Late Stage | Young Wine |
|---|---|---|---|---|---|
| Ethyl esters (µg/L) | |||||
| Ethyl butanoate | Control | 2 ± 0.8 a | 95 ± 9 a | 134 ± 16 a | 111 ± 13 a |
| DAP | 2 ± 0.3 ab | 181 ± 36 b | 238 ± 33 b | 170 ± 6 c | |
| Organic N | 3 ± 0.2 b | 110 ± 28 a | 141 ± 8 a | 137 ± 4 b | |
| Ethyl hexanoate | Control | 25 ± 6 a | 455 ± 4 a | 619 ± 77 a | 630 ± 25 a |
| DAP | 20 ± 2 a | 748 ± 102 b | 779 ± 77 b | 733 ± 25 b | |
| Organic N | 20 ± 2 a | 541 ± 117 a | 575 ± 43 a | 736 ± 17 b | |
| Ethyl octanoate | Control | 113 ± 38 a | 463 ± 50 a | 587 ± 25 a | 1745 ± 6 a |
| DAP | 100 ± 26 a | 653 ± 72 ab | 762 ± 41 b | 2084 ± 8 b | |
| Organic N | 78 ± 2 a | 708 ± 203 b | 626 ± 37 a | 1995 ± 4 b | |
| Ethyl decanoate | Control | 41 ± 0.3 a | 97 ± 16 a | 103 ± 11 a | 361 ± 8 a |
| DAP | 40 ± 1 a | 141 ± 25 a | 158 ± 9 b | 747 ± 64 c | |
| Organic N | 39 ± 2 a | 166 ± 75 a | 141 ± 14 b | 538 ± 2 b | |
| Ethyl dodecanoate | Control | 23 ± 16 a | 43 ± 10 a | 38 ± 14 a | 31 ± 0.4 a |
| DAP | 21 ± 15 a | 33 ± 4 a | 34 ± 3 a | 186 ± 36 c | |
| Organic N | 14 ± 1 a | 81 ± 55 a | 40 ± 4 a | 132 ± 3 b | |
| Ethyl hexadecanoate | Control | 17 ± 1 a | 35 ± 8 a | 10 ± 4 ab | 21 ± 0.2 a |
| DAP | 14 ± 1 a | 16 ± 4 a | 6 ± 1 a | 145 ± 21 b | |
| Organic N | 15 ± 2 a | 77 ± 61 a | 13 ± 1 b | 140 ± 20 b | |
| Total ethyl esters | Control | 221 | 1188 | 1491 | 2899 |
| DAP | 197 | 1772 | 1977 | 4065 | |
| Organic N | 169 | 1683 | 1539 | 3678 | |
| Acetate esters | |||||
| Ethyl acetate (mg/L) | Control | n.d. | 15 ± 6 a | 44 ± 8 a | 43 ± 1 b |
| DAP | n.d. | 31 ± 3 b | 57 ± 2 ab | 36 ± 1 a | |
| Organic N | n.d. | 21 ± 4 ab | 69 ± 4 b | 50 ± 0.3 c | |
| Isoamyl acetate (µg/L) | Control | 15 ± 3 a | 659 ± 34 a | 782 ± 37 a | 515 ± 38 a |
| DAP | 14 ± 1 a | 1110 ± 241 b | 1218 ± 167 b | 767 ± 11 c | |
| Organic N | 17 ± 2 a | 632 ± 113 a | 736 ± 75 a | 578 ± 14 b | |
| Ethyl lactate (mg/L) | Control | 129 ± 44 b | 2832 ± 352 | 5257 ± 126 a | 4148 ± 44 a |
| DAP | 68 ± 12 a | – | 9308 ± 156 b | 4148 ± 44 a | |
| Organic N | 105 ± 19 ab | 4755 ± 1153 | 5165 ± 603 a | 5513 ± 72 b | |
| Hexyl acetate (µg/L) | Control | 46 ± 4 b | 196 ± 9 a | 137 ± 20 a | 119 ± 8 a |
| DAP | 36 ± 1 a | 236 ± 37 b | 184 ± 24 b | 140 ± 3 b | |
| Organic N | 39 ± 6 ab | 190 ± 21 a | 133 ± 13 a | 142 ± 3 b | |
| 2-Phenylethyl acetate (µg/L) | Control | n.d. | 49 ± 2 a | 57 ± 2 ab | 47 ± 1 ab |
| DAP | n.d. | 61 ± 10 a | 62 ± 4 b | 49 ± 2 b | |
| Organic N | n.d. | 52 ± 9 a | 53 ± 7 a | 46 ± 0.5 a | |
| Diethyl succinate (µg/L) | Control | n.d. | 85 ± 18 a | 288 ± 73 a | 815 ± 18 b |
| DAP | n.d. | 185 ± 60 a | 388 ± 48 a | 763 ± 28 a | |
| Organic N | n.d. | 140 ± 59 a | 296 ± 83 a | 906 ± 9 c | |
| Total acetate esters | Control | 79 | 989 | 1264 | 1496 |
| DAP | 50 | 1592 | 1852 | 1719 | |
| Organic N | 106 | 1014 | 1218 | 1672 | |
| Higher alcohols (mg/L) | |||||
| 1-Propanol | Control | n.d. | n.d. | 9 ± 0.2 a | 10 ± 0.3 a |
| DAP | n.d. | n.d. | 18 ± 1 b | 16 ± 0.4 b | |
| Organic N | n.d. | n.d. | 10 ± 0.3 a | 9 ± 1.1 a | |
| Isobutanol (2-methylpropanol) | Control | n.d. | 12 ± 1 b | 19 ± 1 a | 19 ± 1 b |
| DAP | n.d. | 14 ± 2 b | 20 ± 1 a | 17 ± 0.4 a | |
| Organic N | n.d. | 8 ± 0.3 a | 20 ± 0.5 a | 18 ± 1 a | |
| 2-Methyl + 3-methylbutanol | Control | 12 ± 3 a | 97 ± 28 a | 170 ± 8 a | 173 ± 2 c |
| DAP | 8 ± 8 a | 120 ± 10 a | 153 ± 13 a | 140 ± 1 a | |
| Organic N | 10 ± 3 a | 90 ± 11 a | 161 ± 2 a | 152 ± 2 b | |
| Total higher alcohols | Control | 12 | 109 | 198 | 202 |
| DAP | 8 | 134 | 191 | 173 | |
| Organic N | 10 | 98 | 191 | 179 | |
| Other compounds (µg/L) | |||||
| 1-Hexanol | Control | 1181 ± 156 a | 1391 ± 87 a | 1551 ± 59 b | 1469 ± 79 a |
| DAP | 1314 ± 90 a | 1405 ± 47 a | 1581 ± 15 b | 1559 ± 28 ab | |
| Organic N | 1325 ± 90 a | 1339 ± 153 a | 1428 ± 32 a | 1598 ± 23 b | |
| cis-3-Hexen-1-ol | Control | 128 ± 17 a | 150 ± 11 a | 160 ± 7 b | 153 ± 7 a |
| DAP | 144 ± 11 ab | 155 ± 6 a | 163 ± 3 b | 159 ± 3 a | |
| Organic N | 151 ± 10 b | 149 ± 17 a | 149 ± 1 a | 156 ± 3 a | |
| Methanol (mg/L) | Control | 35 ± 4 | 23 ± 13 a | 38 ± 2 a | 41 ± 0.3 b |
| DAP | 25.5 | 28 ± 7 a | 42 ± 1 a | 36 ± 1 a | |
| Organic N | 27.2 | 25 ± 1 a | 41 ± 1 a | 38 ± 3 b | |
| γ-Butyrolactone | Control | 601 ± 84 a | 1441 ± 107 a | 2324 ± 286 a | 3252 ± 123 a |
| DAP | 587 ± 56 a | 1808 ± 194 a | 2722 ± 108 b | 3272 ± 92 a | |
| Organic N | 611 ± 35 a | 1827 ± 385 a | 2836 ± 179 b | 4121 ± 75 b | |
| Benzyl alcohol | Control | 29 ± 4 a | 26 ± 2 a | 37 ± 2 ab | 78 ± 2 a |
| DAP | 28 ± 1 a | 27 ± 1 a | 39 ± 0.2 b | 84 ± 3 b | |
| Organic N | 25 ± 1 a | 27 ± 2 a | 36 ± 1 a | 78 ± 1 a | |
| Acetaldehyde | Control | 29 ± 1 a | 36 ± 11 a | 24 ± 2 ab | 24 ± 5 a |
| DAP | 21 ± 4 a | 41 ± 2 a | 19 ± 1 a | 20 ± 4 a | |
| Organic N | 25 ± 5 a | 39 ± 6 a | 31 ± 6 b | 29 ± 8 b |
| Control | DAP | Organic N | |
|---|---|---|---|
| Tropical fruit | 3.8 ± 2.1 B | 5.7 ± 1.6 A | 1.4 ± 0.9 C |
| Varietal aroma | 2.6 ± 1.1 B | 5.6 ± 1.8 A | 2.6 ± 2.1 B |
| Wine body | 3.0 ± 2.0 A | 4.3 ± 2.8 A | 3.3 ± 2.9 A |
| Overall wine quality | 1.9 ± 1.5 B | 5.9 ± 3.0 A | 2.8 ± 1.9 B |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Temelkova, Z.L.; Česnik, H.B.; Vanzo, A.; Lisjak, K. The Formation of Aroma Compounds During Fermentation in Relation to Yeast Nutrient Source in Sauvignon Blanc Wine. Fermentation 2026, 12, 183. https://doi.org/10.3390/fermentation12040183
Temelkova ZL, Česnik HB, Vanzo A, Lisjak K. The Formation of Aroma Compounds During Fermentation in Relation to Yeast Nutrient Source in Sauvignon Blanc Wine. Fermentation. 2026; 12(4):183. https://doi.org/10.3390/fermentation12040183
Chicago/Turabian StyleTemelkova, Zorica Lelova, Helena Baša Česnik, Andreja Vanzo, and Klemen Lisjak. 2026. "The Formation of Aroma Compounds During Fermentation in Relation to Yeast Nutrient Source in Sauvignon Blanc Wine" Fermentation 12, no. 4: 183. https://doi.org/10.3390/fermentation12040183
APA StyleTemelkova, Z. L., Česnik, H. B., Vanzo, A., & Lisjak, K. (2026). The Formation of Aroma Compounds During Fermentation in Relation to Yeast Nutrient Source in Sauvignon Blanc Wine. Fermentation, 12(4), 183. https://doi.org/10.3390/fermentation12040183





