1. Introduction
Flow through the human airway is characterized by regions of flow separation and the formation of secondary flow structures [
1,
2]. The oscillatory nature of airflow can facilitate gas exchange in higher generations of the human airway [
1,
3]. Normal respiratory rate (RR) in humans ranges between 10–15 breaths per minute (bpm), corresponding to a Womersley number (
) range of 2.41–2.95 based on the following relation:
where
is the angular frequency based on breathing time (BT, where BT = 60/RR seconds),
D is the tracheal diameter, and
is the kinematic viscosity of air. RR can be altered from the resting range of 10–15 bpm during exercise (such as walking RR = 20–60 bpm [
4] and yoga RR = 3–7 bpm [
5]), and also in mechanical ventilation strategies, such as high-frequency oscillatory ventilation (HFOV) [
6,
7,
8]. Further, inhalation time (IT) is about 45% of BT under normal breathing conditions [
6]. Characterizing the airflow inside the human airway during the entire breathing cycle is important for understanding particle deposition characteristics in lung diseases (e.g., chronic obstructive pulmonary disease or COPD), during the use of e-cigarettes, and in inhaled drug therapy [
9,
10]. Previous studies of inhaled drug delivery in human subjects with COPD and asthma have shown improved respiratory outcomes [
11] and increased aerosol deposition with increased inhalation time [
12]. However, computational fluid dynamics (CFD) studies examining particle deposition relevant to aerosols [
13,
14] and e-cigarettes [
13,
15,
16] typically consider either steady flow rates or only the inhalation phase for simulations. Studies examining unsteady breathing patterns over an entire breathing cycle are limited.
The respiratory system consists of airways with complex geometrical cross-sections, including the nasal cavity [
17,
18], mouth, pharynx, larynx, glottis [
19], and the trachea that further bifurcates up to 16–23 Generations (G) [
20] in addition to inter-subject geometry variability [
18,
21]. Due to the geometrical complexity, idealized geometries of the nasal cavity [
22], mouth-to-glottis section [
23], and airway generations [
24,
25] have been used in several studies. Weibel proposed an idealized airway geometry consisting of circular tubes at each generation [
24]. This geometry bifurcated symmetrically along the sagittal plane and was able to capture key aspects of airflow and its features [
26,
27]. A review on using idealized airway models can be found elsewhere [
21]. In addition to airway geometry, head rotation and posture can also influence airway fluid dynamics. Idealized airway model studies have included mouth-to-glottis geometry in the sagittal plane that incorporate head rotation [
23], and clinical studies of HFOV have reported prone positions to improve gas exchange, as compared to the supine position [
28]. Finally, voluntary exercises, such as freestyle swimming and some yoga breathing techniques, can be conducted with head rotation [
29].
Previous studies examining the fluid dynamics of deep inspiration were conducted by prescribing steady inhalation flow through idealized [
30] and anatomically accurate physical models [
18,
31]. Borojeni et al. [
18] recently used computational fluid dynamics simulations to establish physiological ranges of nasal airflow variables, and documented inter-subject variability. Steady inhalation studies with varying airflow parameters (such as
,
) identified both axial (streamwise) and secondary (transverse) flow dispersion to be effective transport mechanisms [
30,
31]. Unsteady physiological flow through subject-specific airways has also been experimentally investigated [
32,
33,
34,
35]. Jalal et al. [
35] found that with respect to idealized airway models [
30], realistic airway geometry produced stronger secondary flows, as well as increased axial and secondary flow dispersion. Secondary flows in realistic airways propagated deeper in the bronchial tree, and were stronger during exhalation as compared to inhalation [
35]. Jalal et al. [
36] conducted magnetic resonance velocimetry (MRV) experiments on an idealized double bifurcation airway model across a range of
and
in the convective region of the flow regime diagram of Jan et al. [
3]. Strong secondary flows were observed for
≥ 6 during crossover from the inhalation to exhalation phase [
35]. Große et al. [
33] conducted particle image velocimetry (PIV) measurements of steady and oscillating flows at the first bifurcation of an anatomical silicone model of the human airway. At steady inhalation, they found that the asymmetrical bifurcation promoted the formation of flow structures that were responsible for continuous axial transport to the lung [
33]. Oscillating flow studies showed that the size of secondary flow structures strongly depended on the instantaneous values of
and
[
33]. Two-dimensional PIV measurements of oscillating flow through an asymmetric idealized model (based on Weibel et al. [
24] and Horsfield et al. [
25]) at normal and HFOV conditions showed mass exchange at higher frequencies [
32]. Experimental studies of HFOV flow through a subject-specific airway model [
37,
38] reported homogeneous ventilation at higher generations with increasing RR (and hence
). However, the above studies of oscillatory flow through the human airway only considered IT/BT = 50%. While IT/BT varies in normal conditions and is not strictly equal to 50% (e.g., IT/BT = 46% [
6]), IT/BT as low as 20% has been used in mechanical ventilation of patients with chronic obstructive pulmonary disease (COPD) and asthma [
4,
39,
40]. Further, breathing exercises that involve voluntary manipulation of the normal rhythm (e.g., hatha yoga) also result in changes to IT/BT. The effects of varying IT/BT on axial dispersion and secondary flows currently remains unknown.
In terms of the physical mechanisms driving flow and gas exchange in different portions of the human airway, Jan et al. [
3] used order-of-magnitude analysis to examine the relative importance of various terms in axial flow and secondary momentum equations. Based on the regional domination of viscosity, unsteadiness, and convective acceleration, they developed a flow regime map by relating
with the product of the ratio of stroke length (
L) to hydraulic diameter (
D) of the trachea when using a sinusoidal breathing profile with IT/BT = 50%. This map was developed to: (a) identify what approximation can be used in modeling flow in different regions of the airway; and (b) investigate the fluid dynamic phenomena that affect gas exchange in HFOV with equal inhalation and exhalation durations. To extend the applicability of their map to realistic breathing patterns in normal and HFOV conditions, this regime chart needs to be modified to account for variations in IT/BT.
HFOV was proposed by Lunkenheimer et al. [
41] to generate both active inspiration and expiration for eliminating entrained gas and gas decompression in airway models. The HFOV technique has since been used to overcome the injuries caused due to the use of conventional ventilation for patients with acute lung injury and acute respiratory distress syndrome (ARDS) [
1]. Fredburg et al. [
42] observed changes in local air distribution and pressure variations at resonant frequency with the use of HFOV, and speculated that lung ventilation can be controlled by varying frequencies. Clinical studies on ARDS patients have shown improved gas exchange with HFOV without compromise in oxygen delivery [
43]. Zhang et al. [
6] performed computations using realistic breathing patterns under normal conditions (
= 2.4) with IT/BT = 46% and HFOV (
= 23.3) with IT/BT = 50%, and characterized fluid flow in the fourth-generation airway. However, the extent to which active inspiration and expiration at larger
(characteristic to HFOV) affect the flow characteristics in upper airways remains fundamentally unclear.
In this study, we examine the effects of varying
and IT/BT on airway flow characteristics. Three-dimensional (3D) computational fluid dynamics (CFD) simulations were performed by prescribing an oscillatory inflow profile in an idealized double bifurcation airway model (two generations) with a mouth-to-glottis section. The aims of this study were to: elucidate the flow physics under varying inhalation to breathing time, and modify the flow regime diagram of Jan et al. [
3] to incorporate effects of varying IT/BT.
2. Methods
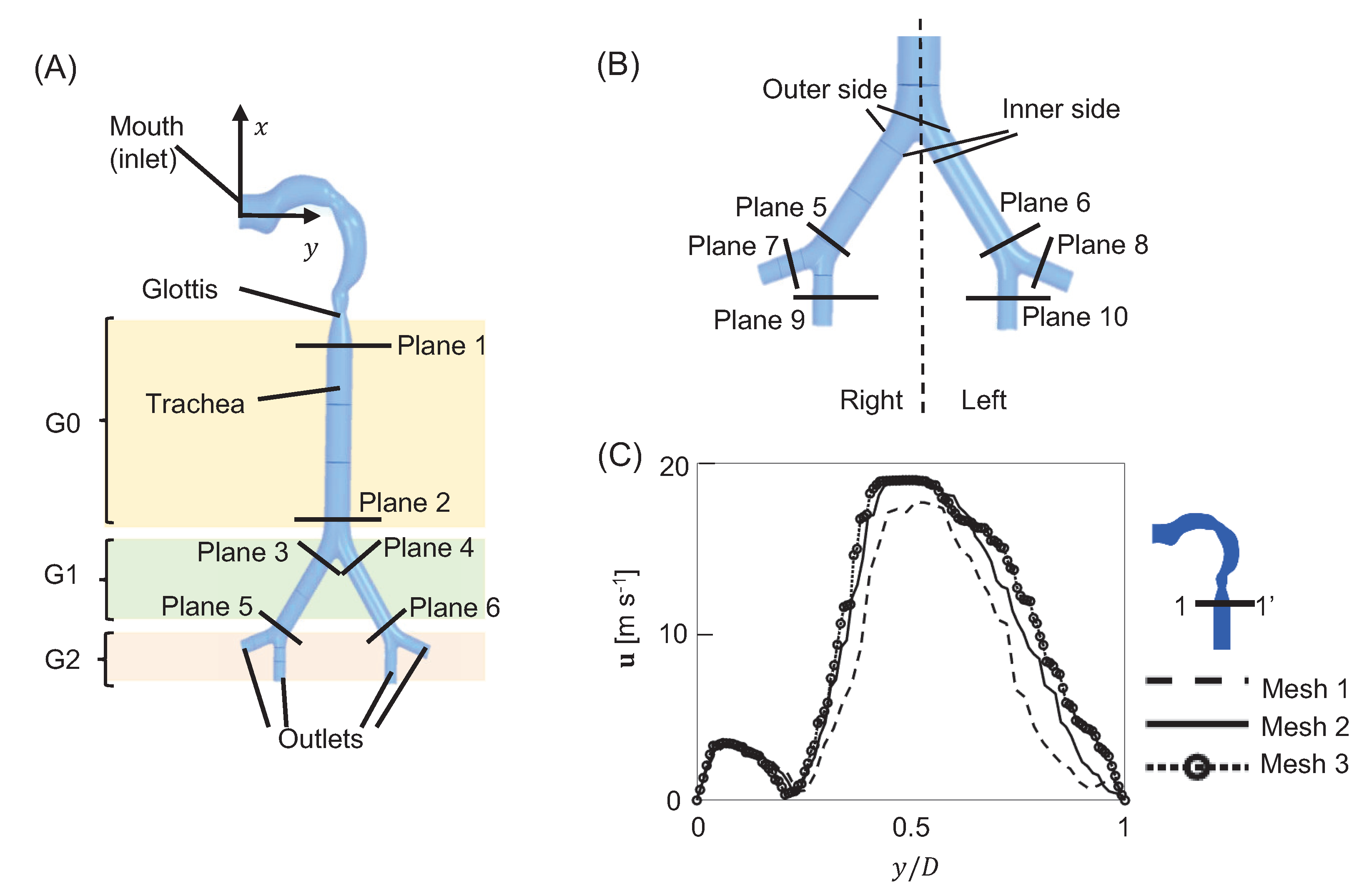
An idealized airway model [
24] with the addition of an idealized mouth-to-glottis section (previously used in a particle deposition study [
23]) was designed in Solidworks (Dassault Systèmes SolidWorks Corporation, Waltham, MA) (
Figure 1A). The airway includes the following: (i) a circular mouth-inlet of 20 mm diameter; (ii) a circular glottis of 8 mm diameter; (iii) a smooth trachea with a uniform, circular cross-section of 120 mm length and diameter (
D) = 18 mm; and (iv) two generations (G) G1 and G2 of diameters 12.2 and 8.3 mm, respectively. The lengths of G1 and G2 were equal to 47 mm and 19 mm, respectively. All the bifurcation angles of the airway geometry were identically equal to 70°. The airway geometry was symmetric with respect to the coronal plane (defined along
x-
y Plane at
z = 0 m,
Figure 1A). Meshing was performed in ANSYS 2019 R3 (ANSYS, Inc., Canonsburg, PA, USA) by importing an STL geometry file into ICEM-CFD. The surface was modified to create parts such as an inlet, walls, and outlets using the angle method, and a material region was created. Octree mesh was created using minimum and maximum element sizes, along with curvature refinement. Walls were selected to generate prism layers with growth rate = 1.1, and volume mesh was computed. A total of three different meshes, each consisting of tetrahedral cells with five prism layers on walls, were generated for mesh independence tests.
for all the meshes were <5, corresponding to peak velocity condition in the trachea. Previous studies have reported that
< 5 in the glottis region and
< 1 in the trachea are sufficient for resolving flow in the near-wall region when using the
k-
SST turbulence model [
44,
45].
Three-dimensional CFD simulations of transient, incompressible flow through the above airway geometry were performed in ANSYS Fluent.
k-
SST turbulence model, used in previous studies of flow through the human airway [
46,
47], was chosen for all simulations in this study. Sinusoidal profiles were used as a simplified representation of realistic breathing waveforms [
7]. The Reynolds number at the trachea during peak inhalation (
) was defined as
where
is the mean flow speed in the trachea,
is the kinematic viscosity of air (1.46 × 10
−5 m
2 s
−1), and
D is the tracheal diameter = 18 mm.
was maintained constant in all breathing waveforms at
= 4200, which is in the physiological range [
14,
31]. The peak inhalation flow rate (
) was determined using the relation:
where
is the kinematic viscosity of air (1.46 × 10
−5 m
2 s
−1) and
is the cross-sectional area along the transverse cross-sectional Plane 2 with diameter
D = 18 mm. Inhalation and exhalation volumes were kept equal for a given RR and IT. The peak exhalation flow rate (
) was defined based on IT, BT, and
using the equation below:
such that
=
when IT = 0.5 BT, and
when IT < 0.5 BT. Instantaneous profiles of inhalation velocity (
) and exhalation velocity (
) were defined using the equations:
where
is the area of the inlet. The above relations were used to develop nine oscillatory profiles of time-varying inflow velocity (
), prescribed perpendicular to the inlet, and prescribed as initial conditions. The tests conducted here include three RRs (10 bpm, 100 bpm, and 1000 bpm (later being clinically impractical in adults)), each for IT/BT = 25%, 33%, and 50% [
4,
39,
40,
48] to examine fluid dynamics across several orders of magnitude variation in
. We expect that our findings can inform future studies of low Reynolds number (
) airway flows (such as in children), where HFOV is clinically used at higher frequencies than in adults (e.g., RR = 960 bpm in neonates [
49,
50]). All the oscillatory velocity profiles consisted of positive velocities for inhalation phases (
from 0° to 180°) and negative velocities for the exhalation phase (
from 180° to 360°).
Boundary conditions included zero pressures at the outlets and no-slip walls. To characterize the oscillatory flow relative to viscous effects,
was calculated for each condition using Equation (
1), where
= 1.46 × 10
−5 m
2 s
−1 was used as the kinematic viscosity of air.
denotes mean flow speed in the trachea, and was calculated to be 3.4 m s
−1 by equating flow rates at the inlet and the trachea via mass conservation for an incompressible flow:
where
is the peak inhalation velocity calculated from Equation (
5).
After prescribing the velocity profile, each simulation was standard-initialized with initial x-velocity, y-velocity, and z-velocity to be equal to 0 m s−1. Simulations were conducted at the High-Performance Computing Center at Oklahoma State University using 16 processor units. The solution method included a coupled scheme, and second-order spatial discretization. A uniform time-step of 10−3 s was used for running simulations for one breathing cycle. All the processing results were auto-saved at 12.5% inhalation time to maintain uniformity across all breathing frequencies and IT/BT ratios.
Mesh independence tests were performed on the airway model for different mesh sizes (
Table 1) at a time-step of 10
s for HFOV velocity profiles (
= 4200,
= 24.1, IT/BT = 50%) for one breathing cycle. This particular case was used in mesh independence tests to decrease the solution time. Three-dimensional velocity (
) was extracted from each converged simulation along a line 1-1’ at the upper trachea in the coronal plane (
z = 0 m; see inset in
Figure 1C).
Figure 1C shows the extracted velocity magnitude along the line 1-1’ for Mesh 1 (dashed line), Mesh 2 (solid line), and Mesh 3 (dotted line with circle markers) as a function of non-dimensional diameter
y/
D, where
D = 18 mm is the trachea diameter. The velocity of Mesh 2 was different compared to Mesh 1, as the smaller element size increased the spatial resolution. However, the spatial resolution of Mesh 2 was nearly the same as that of Mesh 3, but the simulation time of Mesh 3 was four times longer than Mesh 2, due to the larger number of cells. Since the velocity profiles at 1-1’ for Meshes 2 and 3 were nearly identical (
Figure 1C), Mesh 2 was selected for use in this study. In addition, the solution from each mesh study was analyzed further by measuring bulk velocity at a lower trachea (Plane 2). Average velocities of 3.4256 m s
−1, 3.4281 m s
−1 and 3.4298 m s
−1 was found, respectively, in mesh independence test simulations. Since the error in average velocity from Mesh 3 to Mesh 2 (0.05%) was less than the error between Mesh 3 and Mesh 1 (0.1%), Mesh 2 was selected for all simulations and time-step independence studies.
Time-step validation for higher
was conducted on the finalized Mesh 2. For time-step size validation, three different time-step sizes (0.0005 s, 0.001 s, and 0.00375 s) were used with inlet velocity profile corresponding to
= 4200 and
= 24.1 at IT/BT = 50% with the same computational setup as described earlier. Velocity profiles were extracted from the completed simulations along the coronal plane in the upper trachea (Plane 1 in
Table 2). The three velocity profiles showed no major variation at the center of the airway, with minor differences in near-wall velocity (see
Figure S1 in Supplementary Materials). As the error in velocity prediction was better in
= 0.001 s when comparing
= 0.00375 s and simulation time for
= 0.001 s was lower compared to
= 0.0005 s, the time-step of
= 0.001 s was used for all the simulations reported in this study.
Steady inhalation with similar boundary conditions, as described for transient simulations, were simulated at inlet flow rates of 10, 15, 30, 45, and 60 L min−1. Pressure drop () was estimated for both validation (steady flow simulations) and varying and IT/BT (transient flow simulations) in ANSYS Fluent.
Solution files from ANSYS Fluent were imported to ANSYS CFD-POST for further analysis and generating results. All
-tested, velocity magnitude contours were extracted along the planes in
Table 2 and visualized in Tecplot 360 (Tecplot Inc., Bellevue, WA, USA). The
Q-criterion previously used in a study of steady airflow through a similar geometry [
46] was used to identify flow vortex structures at various breathing conditions.
Q = 100,000 s
−2 was used for isosurfaces at peak inhalation, and
Q = 15,000 s
−2 was used for isosurfaces at peak exhalation.
Integral parameters (
,
) have been used in previous studies of flow through the human airway to examine the relative importance of axial flow streaming and lateral dispersion mechanisms [
30,
34]. Axial (i.e., streamwise direction) flow uniformity at a cross-sectional plane was quantified using the integral parameter
[
30,
34]:
where
is the 3D velocity field for a specific phase-point in the cycle,
is the unit normal vector at a given cross-section of the airway, and
A is the cross-sectional area. Lateral dispersion arising on account of secondary (i.e., transverse direction) flow at a cross-sectional plane was quantified using integral parameter
[
30,
34]:
and
were calculated to examine the effects of varying
and IT/BT at G0, G1 and G2 (
Figure 1A,
Table 2).
4. Discussion
Particle deposition and gas exchange within the human respiratory system are intricately dependent on flow through the airway. While previous studies have identified that axial flow streaming and lateral dispersion are effective transport mechanisms in the human airway [
30,
31,
34,
35,
36], the effects of varying inhalation duration (IT/BT) in conjunction with the respiratory rate (RR) on airway flow characteristics remain unknown. The majority of airway fluid dynamics studies have either focused only on inhalation, or examined both inhalation and exhalation with IT/BT = 50%. Inter-subject variability of IT/BT < 50% can occur in voluntary and involuntary breathing exercises. Further, clinical therapies, such as HFOV, allow for patient-specific tuning of IT/BT and RR. Studies of varying IT/BT alongside RR can help identify fundamental differences in axial and lateral flow patterns, potentially valuable for improving airway flow distribution in HFOV therapy. We examined the role of varying IT/BT from 25% to 50% and RR from 10 bpm to 1000 bpm using 3D CFD simulations of airflow through an idealized double bifurcation airway model.
Axial and secondary flows were observed across all
and IT/BT conditions examined in this study (refer Movie 1–3). Several differences can be observed when comparing flow fields at
= 24.1 to those at lower
. At the upper tracheal section (Plane 1) for IT/BT = 25%, asymmetric axial velocity distribution was observed for
= 2.41, as opposed to a centrally concentrated distribution of axial velocity for
= 24.1 during inhalation. The lower tracheal section (Plane 2) for IT/BT = 25% uniform axial velocity distribution for all
. Asymmetric distribution of axial velocity was observed following the first and second bifurcations at lower
. Across all
and IT/BT conditions,
= 2.41 and 7.61 showed the strongest axial flow streaming (
) and lateral flow dispersion at Plane 9 and Plane 10 during inhalation, while during exhalation, all
and IT/BT = 50% showed both strongest axial flow streaming (
) and secondary flow strength (
) at Planes 7–10 (G2). The latter observation regarding
was in partial agreement with findings of a previous MRV study of flow through an idealized double bifurcation model [
36], where secondary flows during inhalation and exhalation were reported to be strengthened with increasing
. Our results showed increased secondary flow strength at the end of inhalation, with an increase in
along with equal secondary flow strengths for planes in a given generation (
=
,
=
=
=
). In terms of secondary flow fields, we observed the generation of vortices at all
during inhalation and exhalation. These secondary flow vortices can be expected to impact particle deposition in applications such as targeted drug delivery.
Similar to the observations of Banko et al. [
31,
34] where
= 7 and IT/BT = 50%, a single-sided axial vortex (see Plane 1 at Phase B in
Figure 3 and
Figure 7 ) was observed during peak inhalation in the trachea for
= 2.41 across all IT/BT ratios. Banko et al. [
34] noted that the vortex at peak inhalation was not previously observed in studies using idealized symmetric geometries. Compared to idealized airway models, our airway geometry includes the mouth-to-glottis section in the coronal plane instead of the sagittal plane. Despite the left–right symmetry in our model starting from the glottis, the presence of a mouth-to-glottis section on the anatomical right side promoted single-sided axial swirls at lower
(vortex structure from
Figure 11). The effect of the glottis can also be seen during inhalation, through axial flow velocity contours in all
and IT/BT in the upper trachea.
Choi et al. [
20] used large eddy simulations to examine the effect of upper airway truncation on the flow in the trachea, and found that the airways above the glottis were crucial for generating turbulence in the trachea. Our results showing the formation of vortex structures point to the importance of including the upper airway structure and its orientation relative to the sagittal plane, which was also noted previously by Lin et al. [
17]. Obstructions such as tongue and upper mouth geometry that were not included in our model can also enhance turbulence in the airway flow, and has been noted by Lin et al. [
17]. Further studies on subject-specific airways are needed to isolate how each of these anatomical structures impact fluid dynamics and examine the importance of
and IT/BT in generating asymmetric axial flow in the airway.
Finally, for a given peak inhalation
, a change in IT/BT ratio for a given
affected the tidal volume. With an increase in IT/BT ratio, tidal inhalation volume increased. The exhalation tidal volume was matched with tidal inhalation volume to maintain equal volumes in both inhalation and exhalation for involuntary breathing frequencies, such that lower IT/BT ratio breathing has smaller
-based exhalation velocity profiles. The lower exhalation velocity profile affected the axial flow streaming in all generations, showing lower strength of primary and secondary flows. Using varying IT/BT ratio might be beneficial with peak inhalation and exhalation of equal
, such that exhalation tidal volume would be more than an inhalation tidal volume, resulting in a greater amount of gases that would have been expelled out, which is indeed necessary for some situations, such as in ARDS [
41].
Modified Flow Regime Diagram
Jan et al. [
3] developed a diagram to classify the flow regime within different sections of the human airway, using an order of magnitude analysis starting from the Navier–Stokes’ equations. This diagram was used to characterize the influence of fluid viscosity, unsteadiness, and convective acceleration in an idealized airway bifurcation for
ranging from 2.3 to 21.3. Their flow map examined
versus non-dimensional stroke length
, where
L is the stroke length (i.e., axial length of travel of a fluid particle) that can be calculated as the ratio of stroke volume (SV) swept in a cycle to the tracheal cross-sectional area) and
D is the tracheal diameter. For a sinusoidal waveform (as considered in this study), Jan et al. [
3] showed that
is related to dimensionless stroke length (
) and
as
. However, their classification was restricted to oscillatory (sinusoidal) breathing cycle with IT/BT = 50%.
Table 3 shows the SV,
L,
evaluated for this study at the upper tracheal cross-section (Plane 1) across the various conditions of
and IT/BT, where
= 4200, based on
and
D. It can be seen that
does not equal
when IT/BT ≠ 50%.
To include the effect of IT/BT ≠ 50%, we examined the use of a modified dimensionless stroke length, where the parameter (BT/2 IT) is included as a multiple of
(i.e., (
)(BT/2 IT)). The modified dimensionless stroke length matched
for all
and IT/BT conditions considered in this study (
Table 3), showing the importance of including IT/BT to classify the operating flow regime accurately. To examine the operating flow regimes at the cross-sectional locations where we analyzed the 3D CFD data (Planes 1–10), we calculated local values of the Reynolds number (
) and Womersley number (
) as follows:
where
and
denote the axial velocity and in-plane airway diameter, respectively, at a given cross-sectional plane (
Table S1 in the Supplementary Materials).
and
are interrelated via the modified dimensionless stroke length as:
.
The values of
and
for a given test condition (i.e.,
, IT/BT) were averaged over the different planes included within a particular generation (see
Table 2 for plane locations). The regime diagram for classifying the flow regime at different generations is shown in
Figure 16, where
is plotted along the
x-axis and the modified dimensionless stroke length ((2
/
)(BT/2 IT)) is plotted along the
y-axis. For a given
and generation number, varying IT/BT did not noticeably alter flow regime location when using the modified dimensionless stroke length. For
= 2.41, the trachea (G0) was in the turbulent zone of the regime diagram.
With increasing
beyond 2.41, flow through the trachea was in the unsteady-convective zone, meaning that both unsteady and convective acceleration terms are important in the Navier–Stokes’ equations. With increased generation number (i.e., moving further down the airway) for either
= 2.41 or
= 7.61, the flow regime tends towards the viscous-convective zone so that unsteady effects are not dominant (quasisteady). At
= 24.1, all three generations are in the weak turbulence (
≈ 100–200 [
3]), such that unsteady effects on the flow field are the most dominant. Our flow regime results agree with MRV studies on a subject-specific anatomical model at
= 7 [
34] and on a planar double bifurcation model at
= 6 and
= 12 [
36]. At
relevant to HFOV, gas exchange and particle deposition at higher airway generations are expected to be most impacted by the unsteady acceleration of the flow. By contrast, viscous forces and convective acceleration are expected to be more influential at lower
.
Investigating the 3D fluid dynamics in anatomically accurate airway models and physiologically relevant breathing patterns can be computationally demanding. Inter-subject variability in geometry can present challenges [
18] in identifying salient features of the fluid flow and in developing modified scaling relations as in this study. In this regard, studies of idealized airway models with sinusoidal breathing patterns can help in providing a basic understanding of flow physics at relatively low computational cost. The current study investigated the flow features inside the airways for an idealized airway geometry, varying clinically significant parameters (IT/BT and RR). The modified dimensionless stroke length identified in this work can be useful for fluid dynamic studies of clinically relevant situations where IT/BT variation (e.g., mechanical ventilation in COPD and asthma patients [
4,
39,
40,
48] and RR variation (e.g., HFOV in neonates) are observed. Future studies using subject-specific geometries are needed to examine the clinical significance of our findings.
5. Conclusions
Using 3D CFD simulations, we examined the roles of varying respiratory rate and inhalation duration (IT/BT) on flow through an idealized human airway model consisting of a mouth-to-glottis section and two generations. Axial and secondary flows were observed throughout the model at all conditions of and IT/BT. For = 2.41, the strong non-axisymmetric axial flow was observed in the trachea during inhalation. For = 24.1, centrally concentrated axial flow was observed during inhalation in the upper trachea, followed by uniformly distributed weak axial flow in the lower trachea. On account of secondary flow, lateral dispersion was found to be the dominant transport mechanism across all test conditions. For all , increase in IT/BT ratio increased the axial flow at early inhalation. In addition, secondary flow after peak inhalation increased during small , and decreased during large . For all increased IT/BT showed increased axial and secondary flows strength during each phase of exhalation, respectively. As changing IT/BT changes the stroke length (L) of an oscillatory flow, we observed a breakdown in the theoretically expected / = 2 L/D relation for IT/BT ≠ 50%. We developed a modified dimensionless stroke length including IT/BT to correct this discrepancy. While a lower regime was dominated by viscous forces and convective acceleration, unsteady acceleration was dominant for higher .
A central limitation of our study is the simplification of our model geometry. A range of morphological complexities observed from G0–G2, including (but not limited to) asymmetric branching and non-circular lumen of the airway branches, are expected to alter the respiratory flows. The general trends reported here relative to
and IT/BT, specifically, the importance of secondary flows with increasing
as well as the modified dimensionless stroke length accounting for IT/BT, are expected to be applicable in anatomically realistic airways. Additional model simplifications, such as the use of rigid walled vessels and a sinusoidal flow profile, should also be noted as limitations of this study, while changes to velocity profiles have shown to modestly impact the flow physics [
7], including airway wall motion has been reported to influence axial and secondary flows compared to rigid-walled airways [
54]. Finally, RANS models (such as in this study) have well-known limitations in modeling unsteady turbulent flows and comparing our study findings with higher-fidelity turbulence models (e.g., large eddy simulations) are needed to examine how the choice of turbulence model impacts time-varying flow physics.