Beyond the Sleep Lab: A Narrative Review of Wearable Sleep Monitoring
Abstract
1. Introduction
2. Historical Evolution of Sleep Monitoring
2.1. Early Foundations: Polysomnography
2.2. The Advent of Actigraphy
2.3. Transition to Electronic and Digital Monitoring
3. Emergence of Wearable Sleep Monitors
3.1. The Consumer-Grade Revolution
3.2. Integration of Multi-Sensor Technologies
3.3. Videosomnography for Sleep Monitoring in Infants and Children
3.4. Advances in Algorithm Development and Machine Learning
3.5. User-Centric Design and Data Accessibility
4. Validation and Accuracy of Wearable Sleep Monitors
4.1. Comparative Studies with Polysomnography
4.2. Methodological Considerations in Validation
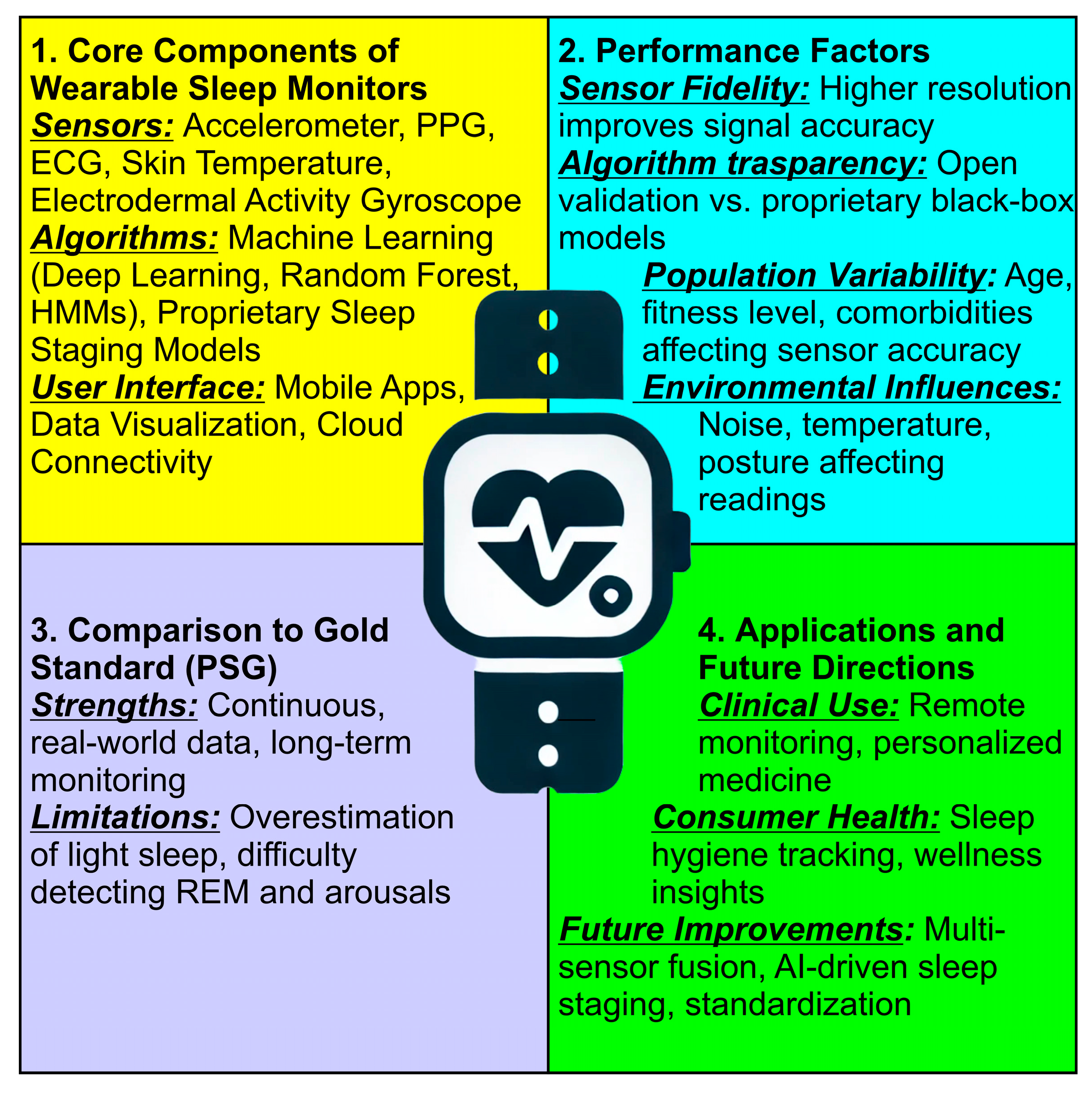
- Sensor Fidelity: The accuracy of sleep stage classification hinges on the quality of the raw physiological signals captured by wearable sensors. Higher-resolution accelerometers improve motion-based sleep–wake detection, while PPG sensors with greater sampling rates enhance HRV measurements, which are critical for distinguishing between sleep stages. The inclusion of additional biosensors, such as skin temperature and electrodermal activity sensors, may further refine sleep staging by providing complementary physiological markers [54].
- Algorithm Transparency: Many consumer wearables rely on proprietary machine learning algorithms for sleep classification, often trained on limited datasets. While these models can demonstrate strong performance in controlled conditions, the lack of publicly available validation methodologies makes it difficult to assess their generalizability [55]. The use of black-box algorithms also limits cross-study comparisons, as different manufacturers employ distinct training datasets, feature selection processes, and scoring criteria [51].
- Population Variability: Individual differences significantly affect wearable sleep-tracking performance. Factors such as age, sex, cardiorespiratory fitness, and comorbidities (e.g., insomnia, sleep apnea) can alter sensor readings and algorithm predictions. Older adults, for instance, exhibit lower movement levels during sleep, potentially leading to misclassification of wake periods as sleep. Additionally, variations in HRV between individuals can affect the accuracy of sleep stage detection, particularly in devices that rely on PPG-derived HRV metrics.
- Environmental Influences: External conditions can interfere with sleep-tracking accuracy. Ambient temperature fluctuations may impact skin conductance and pulse wave characteristics, leading to inconsistencies in HRV-derived sleep staging. Background noise and disruptions, such as a partner’s movements, can confound accelerometer-based motion analysis. Even sleep posture plays a role, wrist-worn devices may experience signal attenuation if the user sleeps with their arm tucked under their body, potentially reducing sensor contact quality and affecting data reliability [54].
4.3. Recent Improvements and Emerging Standards
5. Clinical Applications
5.1. Remote Patient Monitoring and Telemedicine
5.2. Personalized Medicine and Predictive Analytics
5.3. Large-Scale Epidemiological Research
5.4. Integration with Other Health Monitoring Systems
6. Conclusions
- Core Components of Wearable Sleep Monitors (Yellow Section) lists the fundamental elements of wearable sleep tracking devices, including sensors, algorithms, and user interfaces; covers the role of accelerometers, PPG, ECG, skin temperature, and electrodermal activity in monitoring sleep, and mentions machine learning models and proprietary sleep staging approaches;
- Validation Factors (Blue Section) highlights the essential criteria for assessing wearable sleep monitor accuracy; includes sensor fidelity, algorithm transparency, population variability, and environmental influences, and emphasizes how external factors like temperature, noise, and posture affect data reliability;
- Comparison to Gold Standard PSG (Purple Section) discusses how wearables compare to polysomnography (PSG), the clinical gold standard; lists the strengths of wearables, such as continuous real-world monitoring, and identifies limitations, including the overestimation of light sleep and difficulty in detecting REM sleep and arousals;
- Applications and Future Directions (Green Section) explores potential applications in clinical settings and consumer health; mentions advancements such as multi-sensor fusion, AI-driven sleep staging, and standardization, and highlights the role of wearables in remote monitoring and personalized medicine.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rasch, B.; Born, J. About sleep’s role in memory. Physiol. Rev. 2013, 93, 681–766. [Google Scholar] [CrossRef] [PubMed]
- Eugene, A.R.; Masiak, J. The Neuroprotective Aspects of Sleep. MEDtube Sci. 2015, 3, 35–40. [Google Scholar]
- Besedovsky, L.; Lange, T.; Born, J. Sleep and immune function. Pflug. Arch. Eur. J. Physiol. 2012, 463, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Vandekerckhove, M.; Wang, Y.L. Emotion, emotion regulation and sleep: An intimate relationship. AIMS Neurosci. 2018, 5, 1–17. [Google Scholar] [CrossRef]
- Orr, W.C. Utilization of polysomnography in the assessment of sleep disorders. Med. Clin. N. Am. 1985, 69, 1153–1167. [Google Scholar] [CrossRef]
- Troester, M.M.; Quan, S.F.; Berry, R.R.; Plante, D.T.; Abreu, A.R.; Alzoubaidi, M.; Bandyopadhyay, A.; DelRosso, L.; Ebben, M.; Kwon, Y.; et al. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, Version 3; American Academy of Sleep Medicine: Darien, IL, USA, 2023. [Google Scholar]
- De Zambotti, M.; Cellini, N.; Goldstone, A.; Colrain, I.M.; Baker, F.C. Wearable Sleep Technology in Clinical and Research Settings. Med. Sci. Sports Exerc. 2019, 51, 1538–1557. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Choi, S.H. Technologies for sleep monitoring at home: Wearables and nearables. Biomed. Eng. Lett. 2023, 13, 313–327. [Google Scholar] [CrossRef]
- Shepard, J.W., Jr.; Buysse, D.J.; Chesson, A.L., Jr.; Dement, W.C.; Goldberg, R.; Guilleminault, C.; Harris, C.D.; Iber, C.; Mignot, E.; Mitler, M.M.; et al. History of the development of sleep medicine in the United States. J. Clin. Sleep Med. 2005, 1, 61–82. [Google Scholar] [CrossRef]
- Deak, M.; Epstein, L.J. The History of Polysomnography. Sleep Med. Clin. 2009, 4, 313–321. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; text revision; American Academy of Sleep Medicine: Darien, IL, USA, 2023. [Google Scholar]
- Natsky, A.N.; Chai-Coetzer, C.L.; Vakulin, A.; McEvoy, R.D.; Adams, R.J.; Kaambwa, B. Economic evaluation of diagnostic sleep studies for obstructive sleep apnoea in the adult population: A systematic review. Sleep Med. Rev. 2022, 62, 101608. [Google Scholar] [CrossRef]
- Kupfer, D.J.; Detre, T.P.; Foster, G.; Tucker, G.J.; Delgado, J. The application of Delgado’s telemetric mobility recorder for human studies. Behav. Biol. 1972, 7, 585–590. [Google Scholar] [CrossRef]
- Kripke, D.F.; Mullaney, D.J.; Messin, S.; Wyborney, V.G. Wrist actigraphic measures of sleep and rhythms. Electroencephalogr. Clin. Neurophysiol. 1978, 44, 674–676. [Google Scholar] [CrossRef] [PubMed]
- Ancoli-Israel, S.; Cole, R.; Alessi, C.; Chambers, M.; Moorcroft, W.; Pollak, C.P. The role of actigraphy in the study of sleep and circadian rhythms. Sleep 2003, 26, 342–392. [Google Scholar] [CrossRef] [PubMed]
- Lujan, M.R.; Perez-Pozuelo, I.; Grandner, M.A. Past, Present, and Future of Multisensory Wearable Technology to Monitor Sleep and Circadian Rhythms. Front. Digit. Health 2021, 3, 721919. [Google Scholar] [CrossRef]
- Smith, M.T.; McCrae, C.S.; Cheung, J.; Martin, J.L.; Harrod, C.G.; Heald, J.L.; Carden, K.A. Use of Actigraphy for the Evaluation of Sleep Disorders and Circadian Rhythm Sleep-Wake Disorders: An American Academy of Sleep Medicine Clinical Practice Guideline. J. Clin. Sleep Med. 2018, 14, 1231–1237. [Google Scholar] [CrossRef]
- Smith, M.T.; McCrae, C.S.; Cheung, J.; Martin, J.L.; Harrod, C.G.; Heald, J.L.; Carden, K.A. Use of Actigraphy for the Evaluation of Sleep Disorders and Circadian Rhythm Sleep-Wake Disorders: An American Academy of Sleep Medicine Systematic Review, Meta-Analysis, and GRADE Assessment. J. Clin. Sleep Med. 2018, 14, 1209–1230. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A. The role and validity of actigraphy in sleep medicine: An update. Sleep Med. Rev. 2011, 15, 259–267. [Google Scholar] [CrossRef]
- Martin, J.L.; Hakim, A.D. Wrist Actigraphy. Chest 2011, 139, 1514–1527. [Google Scholar] [CrossRef]
- Gao, C.; Li, P.; Morris, C.J.; Zheng, X.; Ulsa, M.C.; Gao, L.; Scheer, F.; Hu, K. Actigraphy-Based Sleep Detection: Validation with Polysomnography and Comparison of Performance for Nighttime and Daytime Sleep During Simulated Shift Work. Nat. Sci. Sleep 2022, 14, 1801–1816. [Google Scholar] [CrossRef]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.L.; Quan, S.F. The Aasm Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specifications, 1st ed.; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Kemp, B.; Varri, A.; Rosa, A.C.; Nielsen, K.D.; Gade, J. A simple format for exchange of digitized polygraphic recordings. Electroencephalogr. Clin. Neurophysiol. 1992, 82, 391–393. [Google Scholar] [CrossRef]
- Choo, B.P.; Mok, Y.; Oh, H.C.; Patanaik, A.; Kishan, K.; Awasthi, A.; Biju, S.; Bhattacharjee, S.; Poh, Y.; Wong, H.S. Benchmarking performance of an automatic polysomnography scoring system in a population with suspected sleep disorders. Front. Neurol. 2023, 14, 1123935. [Google Scholar] [CrossRef] [PubMed]
- Alsolai, H.; Qureshi, S.; Iqbal, S.M.Z.; Vanichayobon, S.; Henesey, L.E.; Lindley, C.; Karrila, S. A systematic review of literature on automated sleep scoring. IEEE Access 2022, 10, 79419–79443. [Google Scholar] [CrossRef]
- Bailly, S.; Mendelson, M.; Baillieul, S.; Tamisier, R.; Pepin, J.L. The Future of Telemedicine for Obstructive Sleep Apnea Treatment: A Narrative Review. J. Clin. Med. 2024, 13, 2700. [Google Scholar] [CrossRef]
- Wright, S.P.; Hall Brown, T.S.; Collier, S.R.; Sandberg, K. How consumer physical activity monitors could transform human physiology research. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R358–R367. [Google Scholar] [CrossRef]
- Mantua, J.; Gravel, N.; Spencer, R.M. Reliability of Sleep Measures from Four Personal Health Monitoring Devices Compared to Research-Based Actigraphy and Polysomnography. Sensors 2016, 16, 646. [Google Scholar] [CrossRef]
- Charlton, P.H.; Kyriaco, P.A.; Mant, J.; Marozas, V.; Chowienczyk, P.; Alastruey, J. Wearable Photoplethysmography for Cardiovascular Monitoring. Proc. IEEE. Inst. Electr. Electron. Eng. 2022, 110, 355–381. [Google Scholar] [CrossRef]
- Wu, X.; Yang, J.; Pan, Y.; Zhang, X.; Luo, Y. Automatic sleep-stage scoring based on photoplethysmographic signals. Physiol. Meas. 2020, 41, 065008. [Google Scholar] [CrossRef]
- Logacjov, A.; Skarpsno, E.S.; Kongsvold, A.; Bach, K.; Mork, P.J. A Machine Learning Model for Predicting Sleep and Wakefulness Based on Accelerometry, Skin Temperature and Contextual Information. Nat. Sci. Sleep 2024, 16, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Frasier, K.; Li, V.; Sobotka, M.; Vinagolu-Baur, J.; Herrick, G. The role of wearable technology in real-time skin health monitoring. JEADV Clin. Pract. 2024, 4, 21–29. [Google Scholar] [CrossRef]
- Hwang, S.H.; Seo, S.; Yoon, H.N.; Jung, D.W.; Baek, H.J.; Cho, J.; Choi, J.W.; Lee, Y.J.; Jeong, D.U.; Park, K.S. Sleep Period Time Estimation Based on Electrodermal Activity. IEEE J. Biomed. Health Inform. 2017, 21, 115–122. [Google Scholar] [CrossRef]
- Nechyporenko, A.; Frohme, M.; Strelchuk, Y.; Omelchenko, V.; Gargin, V.; Ishchenko, L.; Alekseeva, V. Galvanic Skin Response and Photoplethysmography for Stress Recognition Using Machine Learning and Wearable Sensors. Appl. Sci. 2024, 14, 11997. [Google Scholar] [CrossRef]
- de Zambotti, M.; Rosas, L.; Colrain, I.M.; Baker, F.C. The Sleep of the Ring: Comparison of the OURA Sleep Tracker Against Polysomnography. Behav. Sleep Med. 2019, 17, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Perez-Pozuelo, I.; Posa, M.; Spathis, D.; Westgate, K.; Wareham, N.; Mascolo, C.; Brage, S.; Palotti, J. Detecting sleep outside the clinic using wearable heart rate devices. Sci. Rep. 2022, 12, 7956. [Google Scholar] [CrossRef]
- Breda, M.; Lucchini, M.; Barnett, N.; Bruni, O. A cross-sectional study on the relationship between infant sleep, temperament, and bedtime practices. J. Clin. Sleep Med. 2024, 20, 1965–1974. [Google Scholar] [CrossRef]
- Breda, M.; Lucchini, M.; Barnett, N.; Bruni, O. Early identification of sleep phenotypes in infants by videosomnography: A cross-sectional study. J. Clin. Sleep Med. 2025, 21, 867–874. [Google Scholar] [CrossRef]
- Tikotzky, L.; Ran-Peled, D.; Ben-Zion, H. A preliminary study on the performance of the Nanit auto-videosomnography scoring system against observed video scoring and actigraphy to estimate sleep-wake states in infants. Sleep Health 2023, 9, 611–617. [Google Scholar] [CrossRef]
- Liang, M.; Cai, Z.; Zhang, H.; Huang, C.; Meng, Y.; Zhao, L.; Li, D.; Ma, X.; Zhao, X. Machine Learning-based Analysis of Rectal Cancer MRI Radiomics for Prediction of Metachronous Liver Metastasis. Acad. Radiol. 2019, 26, 1495–1504. [Google Scholar] [CrossRef]
- Li, C.; Qi, Y.; Ding, X.; Zhao, J.; Sang, T.; Lee, M. A Deep Learning Method Approach for Sleep Stage Classification with EEG Spectrogram. Int. J. Environ. Res. Public Health 2022, 19, 6322. [Google Scholar] [CrossRef]
- Chen, W.; Cai, Y.; Li, A.; Su, Y.; Jiang, K. Single-Channel Sleep EEG Classification Method Based on LSTM and Hidden Markov Model. Brain Sci. 2024, 14, 1087. [Google Scholar] [CrossRef]
- Wang, Y.; Ye, S.; Xu, Z.; Chu, Y.; Zhang, J.; Yu, W. Research on Sleep Staging Based on Support Vector Machine and Extreme Gradient Boosting Algorithm. Nat. Sci. Sleep 2024, 16, 1827–1847. [Google Scholar] [CrossRef] [PubMed]
- Sri, T.R.; A, R.R.; Madala, J.; Duddukuru, S.L.; Reddipalli, R.; Polasi, P.K. A Systematic Review on Deep Learning Models for Sleep Stage Classification. In Proceedings of the 2022 6th International Conference on Trends in Electronics and Informatics (ICOEI), Tirunelveli, India, 28–30 April 2022; pp. 1505–1511. [Google Scholar]
- Purnell, L.; Sierra, M.; Lisker, S.; Lim, M.S.; Bailey, E.; Sarkar, U.; Lyles, C.R.; Nguyen, K.H. Acceptability and Usability of a Wearable Device for Sleep Health Among English- and Spanish-Speaking Patients in a Safety Net Clinic: Qualitative Analysis. JMIR Form. Res. 2023, 7, e43067. [Google Scholar] [CrossRef] [PubMed]
- Van de Water, A.T.; Holmes, A.; Hurley, D.A. Objective measurements of sleep for non-laboratory settings as alternatives to polysomnography—A systematic review. J. Sleep Res. 2011, 20, 183–200. [Google Scholar] [CrossRef] [PubMed]
- de Zambotti, M.; Baker, F.C.; Colrain, I.M. Validation of Sleep-Tracking Technology Compared with Polysomnography in Adolescents. Sleep 2015, 38, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Chinoy, E.D.; Cuellar, J.A.; Huwa, K.E.; Jameson, J.T.; Watson, C.H.; Bessman, S.C.; Hirsch, D.A.; Cooper, A.D.; Drummond, S.P.A.; Markwald, R.R. Performance of seven consumer sleep-tracking devices compared with polysomnography. Sleep 2021, 44, zsaa291. [Google Scholar] [CrossRef]
- Miller, D.J.; Sargent, C.; Roach, G.D. A Validation of Six Wearable Devices for Estimating Sleep, Heart Rate and Heart Rate Variability in Healthy Adults. Sensors 2022, 22, 6317. [Google Scholar] [CrossRef]
- Grandner, M.A.; Rosenberger, M.E. Chapter 12—Actigraphic sleep tracking and wearables: Historical context, scientific applications and guidelines, limitations, and considerations for commercial sleep devices. In Sleep and Health; Grandner, M.A., Ed.; Academic Press: Cambridge, UK, 2019; pp. 147–157. [Google Scholar] [CrossRef]
- Menghini, L.; Cellini, N.; Goldstone, A.; Baker, F.C.; de Zambotti, M. A standardized framework for testing the performance of sleep-tracking technology: Step-by-step guidelines and open-source code. Sleep 2021, 44, zsaa170. [Google Scholar] [CrossRef]
- Hamill, K.; Jumabhoy, R.; Kahawage, P.; de Zambotti, M.; Walters, E.M.; Drummond, S.P.A. Validity, potential clinical utility and comparison of a consumer activity tracker and a research-grade activity tracker in insomnia disorder II: Outside the laboratory. J. Sleep Res. 2020, 29, e12944. [Google Scholar] [CrossRef]
- Imtiaz, S.A. A Systematic Review of Sensing Technologies for Wearable Sleep Staging. Sensors 2021, 21, 1562. [Google Scholar] [CrossRef]
- de Zambotti, M.; Goldstein, C.; Cook, J.; Menghini, L.; Altini, M.; Cheng, P.; Robillard, R. State of the science and recommendations for using wearable technology in sleep and circadian research. Sleep 2024, 47, zsad325. [Google Scholar] [CrossRef]
- Benedetti, D.; Menghini, L.; Vallat, R.; Mallett, R.; Kiss, O.; Faraguna, U.; Baker, F.C.; de Zambotti, M. Call to action: An open-source pipeline for standardized performance evaluation of sleep-tracking technology. Sleep 2023, 46, zsac304. [Google Scholar] [CrossRef]
- Coelho, J.; Pecune, F.; Levavasseur, Y.; De Sevin, E.; D’Incau, E.; Sagaspe, P.; Sanchez-Ortuno, M.M.; Micoulaud-Franchi, J.A.; Philip, P. From improved sleep regularity to reduced sleep complaints and mental health conditions: A population-based interventional study using a smartphone-based virtual agent. Sleep 2023, 46, zsad165. [Google Scholar] [CrossRef] [PubMed]
- Vohra, K.P.; Johnson, K.G.; Dalal, A.; Ibrahim, S.; Krishnan, V.; Abbasi-Feinberg, F.; Abreu, A.R.; Bandyopadhyay, A.; Gurubhagavatula, I.; Kuhlmann, D.; et al. Recommendations for permanent sleep telehealth: An American Academy of Sleep Medicine position statement. J. Clin. Sleep Med. 2025, 21, 401–404. [Google Scholar] [CrossRef]
- Palmese, F.; Druda, Y.; Benintende, V.; Fuda, D.; Sicbaldi, M.; Di Florio, P.; Butt, A.H.; Bedogni, G.; Chiari, L.; Silvani, A.; et al. Wearable sensors for monitoring caregivers of people with dementia: A scoping review. Eur. Geriatr. Med. 2024, 16, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Garbarino, S.; Bragazzi, N.L. Revolutionizing Sleep Health: The Emergence and Impact of Personalized Sleep Medicine. J. Pers. Med. 2024, 14, 598. [Google Scholar] [CrossRef]
- Charlton, P.H.; Allen, J.; Bailon, R.; Baker, S.; Behar, J.A.; Chen, F.; Clifford, G.D.; Clifton, D.A.; Davies, H.J.; Ding, C.; et al. The 2023 wearable photoplethysmography roadmap. Physiol. Meas. 2023, 44, 111001. [Google Scholar] [CrossRef]
- Vural, B.; Uludag, I.; Ince, B.; Ozyurt, C.; Ozturk, F.; Sezginturk, M.K. Fluid-based wearable sensors: A turning point in personalized healthcare. Turk. J. Chem. 2023, 47, 944–967. [Google Scholar] [CrossRef]
- AbbasiMoradi, F.; Mogavero, M.P.; Palomino, M.; Ferri, R.; DelRosso, L.M. Age related disparities in sleep apnea diagnosis using a wearable device: Implications of 4% vs. 3% hypopnea scoring criteria. Sleep Med. 2024, 118, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.J.; Pawelek, J.B.; Warshawsky, S.; Quer, G.; Trieu, M.; Pandit, J.A.; Owens, R.L. Using New Technologies and Wearables for Characterizing Sleep in Population-based Studies. Curr. Sleep Med. Rep. 2024, 10, 82–92. [Google Scholar] [CrossRef]
- Willetts, M.; Hollowell, S.; Aslett, L.; Holmes, C.; Doherty, A. Statistical machine learning of sleep and physical activity phenotypes from sensor data in 96,220 UK Biobank participants. Sci. Rep. 2018, 8, 7961. [Google Scholar] [CrossRef] [PubMed]
- Denny, J.C.; Rutter, J.L.; Goldstein, D.B.; Philippakis, A.; Smoller, J.W.; Jenkins, G.; Dishman, E. The “All of Us” Research Program. N. Engl. J. Med. 2019, 381, 668–676. [Google Scholar] [CrossRef]
- Del Pozo Cruz, B.; McGregor, D.E.; Del Pozo Cruz, J.; Buman, M.P.; Palarea-Albaladejo, J.; Alfonso-Rosa, R.M.; Chastin, S.F.M. Integrating Sleep, Physical Activity, and Diet Quality to Estimate All-Cause Mortality Risk: A Combined Compositional Clustering and Survival Analysis of the National Health and Nutrition Examination Survey 2005–2006 Cycle. Am. J. Epidemiol. 2020, 189, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Mennella, C.; Maniscalco, U.; De Pietro, G.; Esposito, M. Ethical and regulatory challenges of AI technologies in healthcare: A narrative review. Heliyon 2024, 10, e26297. [Google Scholar] [CrossRef] [PubMed]


| Device/Platform (Representative Model) | Approx. Era | Primary Sensing Technology | Measured Parameters | Main Advantages | Main Limitations |
|---|---|---|---|---|---|
| Early wrist actigraphs | 1970s | Piezoelectric accelerometer | Total sleep time (TST), sleep efficiency (SE), sleep onset latency (SOL) | Non-invasive, inexpensive, allows long-term home monitoring | Cannot differentiate sleep stages; misclassifies quiet wake as sleep |
| Mini-Motionlogger (Ambulatory Monitoring, Inc. AMI; Ardsley, NY, USA), Actiwatch (Philips Respironics, Inc.; Murrysville, PA, USA) | 1990s−2000s | Triaxial accelerometer ± ambient-light sensor | Activity counts, sleep–wake cycles, circadian phase | Widely validated; incorporates light exposure data | Limited staging resolution; motion artifacts in low-movement conditions |
| Fitbit Flex (Fitbit; San Francisco, CA, USA), Jawbone UP (Jawbone, AliphCom, Inc.; San Francisco, CA, USA) | Early 2010s | 3-axis accelerometer | Sleep duration, SE, bedtime/wake time | Broad consumer accessibility; continuous use | Proprietary algorithms; low staging accuracy |
| Oura Ring (Oura Health Oy; Oulu, Finland) | 2015–present | Photoplethysmography (PPG), temperature, 3-axis accelerometer | TST, SE, HR/HRV, light/deep/REM staging | Multi-sensor integration; good HRV precision | Underestimates wake; algorithm not transparent |
| Whoop Strap (WHOOP, Inc.; Boston, MA, USA), Garmin Vivosmart (Garmin International, Inc.; Olathe, KS, USA) | 2018–present | PPG, galvanic-skin-response (GSR), temperature, accelerometer | Sleep stages, HRV, recovery index | High-frequency sampling; cloud analytics | Sleep staging less accurate than PSG; proprietary scoring |
| Dreem Headband (Dreem/Rythm SAS; Paris, France), Muse S Headband (InteraXon Inc.; Toronto, ON, Canada) | 2018–present | Dry-EEG, PPG, accelerometer | EEG-based sleep stages, HR, respiration | Direct EEG signal; near-PSG accuracy | Less comfortable for long-term home use |
| Apple Watch Series 6+ (Apple Inc.; Cupertino, CA, USA) | 2020–present | PPG, accelerometer, SpO2 | Sleep duration, HR/HRV, oxygen saturation | Large user base; seamless integration with health platforms | Simplified staging; restricted raw-data access |
| Nanit System (video-based) (Nanit, Udisense Inc.; New York, NY, USA) | 2020s | Computer-vision motion & respiration analysis | Sleep–wake patterns, respiratory motion | Non-contact, infant-friendly | Low wake specificity; dependent on lighting/environment |
| Experimental biochemical patches | 2020s | Sweat-based biochemical sensors (electrolytes, cortisol) + temperature | Stress biomarkers, circadian trends | Adds metabolic insight; expanding research field | Not yet validated for clinical sleep staging |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mogavero, M.P.; Lanza, G.; Bruni, O.; Ferini-Strambi, L.; Silvani, A.; Faraguna, U.; Ferri, R. Beyond the Sleep Lab: A Narrative Review of Wearable Sleep Monitoring. Bioengineering 2025, 12, 1191. https://doi.org/10.3390/bioengineering12111191
Mogavero MP, Lanza G, Bruni O, Ferini-Strambi L, Silvani A, Faraguna U, Ferri R. Beyond the Sleep Lab: A Narrative Review of Wearable Sleep Monitoring. Bioengineering. 2025; 12(11):1191. https://doi.org/10.3390/bioengineering12111191
Chicago/Turabian StyleMogavero, Maria P., Giuseppe Lanza, Oliviero Bruni, Luigi Ferini-Strambi, Alessandro Silvani, Ugo Faraguna, and Raffaele Ferri. 2025. "Beyond the Sleep Lab: A Narrative Review of Wearable Sleep Monitoring" Bioengineering 12, no. 11: 1191. https://doi.org/10.3390/bioengineering12111191
APA StyleMogavero, M. P., Lanza, G., Bruni, O., Ferini-Strambi, L., Silvani, A., Faraguna, U., & Ferri, R. (2025). Beyond the Sleep Lab: A Narrative Review of Wearable Sleep Monitoring. Bioengineering, 12(11), 1191. https://doi.org/10.3390/bioengineering12111191









